Multifunctional Plant Virus Nanoparticles for Targeting Breast Cancer Tumors
Abstract
1. Introduction
- Their inherent safety, their non-infectious nature in mammals, biocompatibility, and biodegradability;
- Their well-defined structural features, such as unique shapes and sizes, can be monodispersed for loading targeted molecular antitumor therapies onto their internal cavity and their interior and exterior surfaces by the assembly, infusion, or internal surface modification;
- Their ability to self-assemble;
- Their ability to be localized to the target site by chemical and genetic programmability;
- Their simple and inexpensive production;
- Their inherent immunogenicity enables them to act as nano-adjuvants and nanovaccines in cancer immunotherapy
2. Multifunctional PVNPs in Cancer Therapy
3. PVNP-Based BC Tumor Therapies
- Nanoparticulate features such as composition, size, and surface properties;
- Loading and targetability via nano-engineering;
- Inherent immune stimulatory ability.
- The nature of tumor-associated vasculature in comparison to normal vasculature;
- Overexpression of tumor-cell-based biomarkers in comparison to normal cells;
3.1. PVNP-Based Non-Targeted Delivery for BC Tumor
3.1.1. Small Molecule Drug Delivery
3.1.2. Nucleic Acid Delivery
3.1.3. Peptide/Protein Delivery
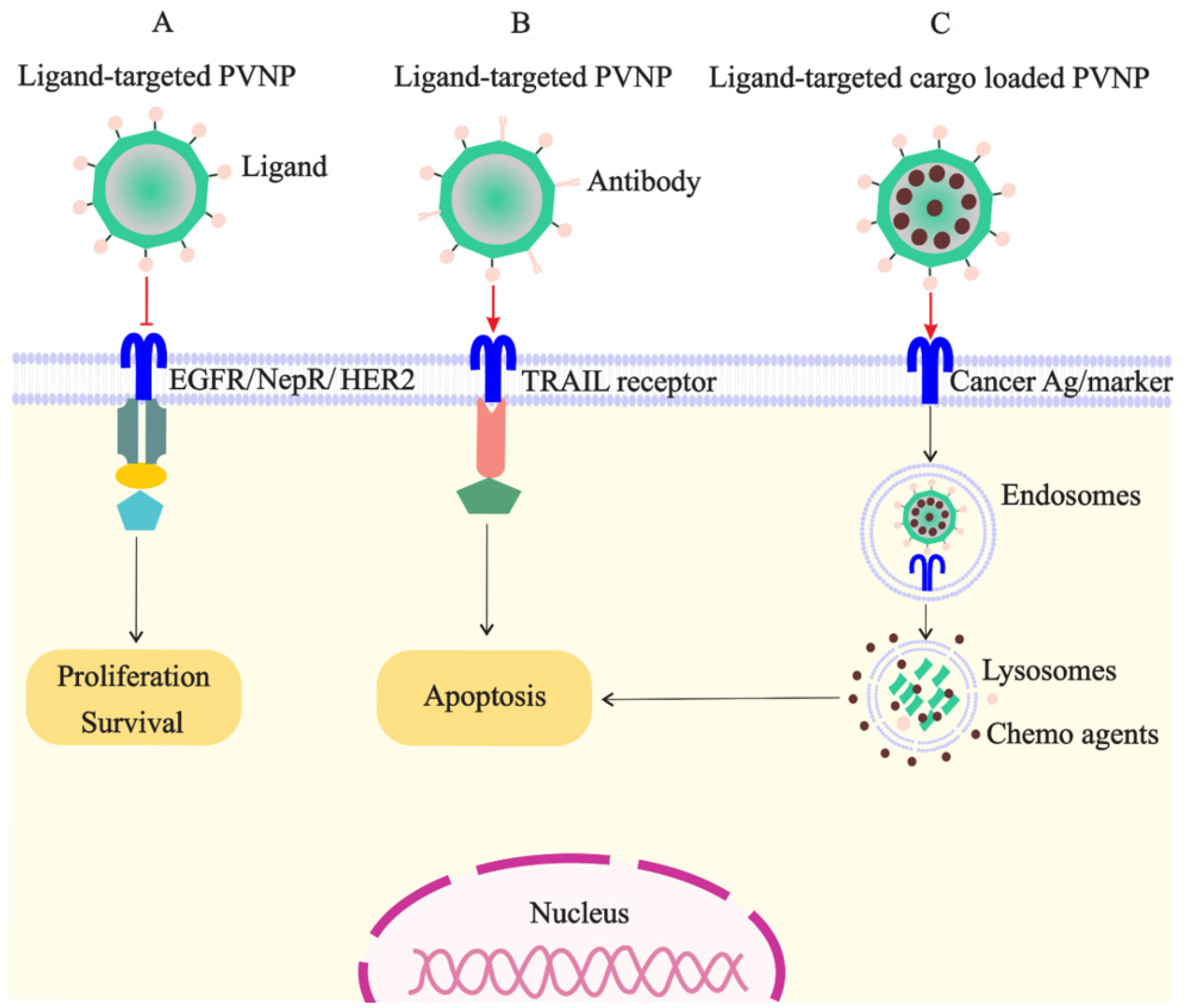
3.2. PVNP-Based Targeted Delivery for BC Tumor
- FR-based drug agonists (e.g., MTX) conjugated to PVNPs;
- Dual targeting by presenting FA and conjugation of drug agonist (e.g., MTX) on surface PVNPs [68].
3.3. PVNP-Based Targeted Immunotherapies
3.4. PVNP -Based Combinational Therapies for BC
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cai, H.; Wang, C.; Shukla, S.; Steinmetz, N.F. Cowpea mosaic virus immunotherapy combined with cyclophosphamide reduces breast cancer tumor burden and inhibits lung metastasis. Adv. Sci. 2019, 6, 1802281. [Google Scholar] [CrossRef] [PubMed]
- Barzaman, K.; Karami, J.; Zarei, Z.; Hosseinzadeh, A.; Kazemi, M.H.; Moradi-Kalbolandi, S.; Safari, E.; Farahmand, L. Breast cancer: Biology, biomarkers, and treatments. Int. Immunopharmacol. 2020, 84, 106535. [Google Scholar] [CrossRef]
- Lin, H.-J.; Liu, Y.; Lofland, D.; Lin, J. Breast cancer tumor microenvironment and molecular aberrations hijack tumoricidal immunity. Cancers 2022, 14, 285. [Google Scholar] [CrossRef] [PubMed]
- Ediriwickrema, A.; Saltzman, W.M. Nanotherapy for cancer: Targeting and multifunctionality in the future of cancer therapies. ACS Biomater. Sci. Eng. 2015, 1, 64–78. [Google Scholar] [CrossRef]
- Malliappan, S.P.; Kandasamy, P.; Chidambaram, S.; Venkatasubbu, D.; Perumal, S.K.; Sugumaran, A. Breast cancer targeted treatment strategies: Promising nanocarrier approaches. Anti-Cancer Agents Med. Chem. (Former. Curr. Med. Chem.-Anti-Cancer Agents) 2020, 20, 1300–1310. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, K.; Wang, Y.; Gao, F.; Liu, Q.; Zhang, C.; Li, P.; Zhang, J.; Chen, J. Targeting engineered nanoparticles for breast cancer therapy. Pharmaceutics 2021, 13, 1829. [Google Scholar] [CrossRef]
- Jahan, S.; Karim, M.; Chowdhury, E.H. Nanoparticles targeting receptors on breast cancer for efficient delivery of chemotherapeutics. Biomedicines 2021, 9, 114. [Google Scholar] [CrossRef]
- Chung, Y.H.; Cai, H.; Steinmetz, N.F. Viral nanoparticles for drug delivery, imaging, immunotherapy, and theranostic applications. Adv. Drug Deliv. Rev. 2020, 156, 214–235. [Google Scholar]
- Wen, A.M.; Steinmetz, N.F. Design of virus-based nanomaterials for medicine, biotechnology, and energy. Chem. Soc. Rev. 2016, 45, 4074–4126. [Google Scholar] [CrossRef]
- Buzón, P.; Maity, S.; Roos, W.H. Physical virology: From virus self-assembly to particle mechanics. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1613. [Google Scholar] [CrossRef]
- Masarapu, H.; Patel, B.K.; Chariou, P.L.; Hu, H.; Gulati, N.M.; Carpenter, B.L.; Ghiladi, R.A.; Shukla, S.; Steinmetz, N.F. Physalis mottle virus-like particles as nanocarriers for imaging reagents and drugs. Biomacromolecules 2017, 18, 4141–4153. [Google Scholar] [PubMed]
- Shahgolzari, M.; Dianat-Moghadam, H.; Fiering, S. Multifunctional plant virus nanoparticles in the next generation of cancer immunotherapies. Semin. Cancer Biol. 2021, in press. [Google Scholar]
- Shahgolzari, M.; Pazhouhandeh, M.; Milani, M.; Yari Khosroushahi, A.; Fiering, S. Plant viral nanoparticles for packaging and in vivo delivery of bioactive cargos. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1629. [Google Scholar] [PubMed]
- Wen, A.M.; Rambhia, P.H.; French, R.H.; Steinmetz, N.F. Design rules for nanomedical engineering: From physical virology to the applications of virus-based materials in medicine. J. Biol. Phys. 2013, 39, 301–325. [Google Scholar]
- Lebel, M.-È.; Chartrand, K.; Leclerc, D.; Lamarre, A. Plant viruses as nanoparticle-based vaccines and adjuvants. Vaccines 2015, 3, 620–637. [Google Scholar]
- Shahgolzari, M.; Fiering, S. Emerging Potential of Plant Virus Nanoparticles (PVNPs) in Anticancer Immunotherapies. J. Cancer Immunol. 2022, 4, 22. [Google Scholar]
- Mittal, S.; Brown, N.J.; Holen, I. The breast tumor microenvironment: Role in cancer development, progression and response to therapy. Expert Rev. Mol. Diagn. 2018, 18, 227–243. [Google Scholar] [PubMed]
- Noh, M.-G.; Kim, S.S.; Kim, Y.J.; Jung, T.-Y.; Jung, S.; Rhee, J.-H.; Lee, J.-H.; Lee, J.-S.; Cho, J.-H.; Moon, K.-S. Evolution of the tumor microenvironment toward immune-suppressive seclusion during brain metastasis of breast cancer: Implications for targeted therapy. Cancers 2021, 13, 4895. [Google Scholar]
- Li, J.; Burgess, D.J. Nanomedicine-based drug delivery towards tumor biological and immunological microenvironment. Acta Pharm. Sin. B 2020, 10, 2110–2124. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar]
- Franzen, S.; Lommel, S.A. Targeting cancer with ‘smart bombs’: Equipping plant virus nanoparticles for a ‘seek and destroy’mission. Nanomedicine 2009, 4, 575–588. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Shukla, S.; Steinmetz, N.F. The antitumor efficacy of CpG oligonucleotides is improved by encapsulation in plant virus-like particles. Adv. Funct. Mater. 2020, 30, 1908743. [Google Scholar] [CrossRef] [PubMed]
- Lam, P.; Steinmetz, N.F. Plant viral and bacteriophage delivery of nucleic acid therapeutics. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2018, 10, e1487. [Google Scholar] [CrossRef] [PubMed]
- Beatty, P.H.; Lewis, J.D. Cowpea mosaic virus nanoparticles for cancer imaging and therapy. Adv. Drug Deliv. Rev. 2019, 145, 130–144. [Google Scholar] [CrossRef]
- Pitek, A.S.; Wen, A.M.; Shukla, S.; Steinmetz, N.F. The protein corona of plant virus nanoparticles influences their dispersion properties, cellular interactions, and in vivo fates. Small 2016, 12, 1758–1769. [Google Scholar] [CrossRef]
- Bruckman, M.A.; Randolph, L.N.; VanMeter, A.; Hern, S.; Shoffstall, A.J.; Taurog, R.E.; Steinmetz, N.F. Biodistribution, pharmacokinetics, and blood compatibility of native and PEGylated tobacco mosaic virus nano-rods and-spheres in mice. Virology 2014, 449, 163–173. [Google Scholar] [CrossRef]
- Nkanga, C.I.; Steinmetz, N.F. The pharmacology of plant virus nanoparticles. Virology 2021, 556, 39–61. [Google Scholar] [CrossRef]
- Shukla, S. Serum albumin ‘camouflage’of plant virus based nanoparticles prevents their antibody recognition and enhances pharmacokinetics. Biomaterials 2016, 89, 89–97. [Google Scholar]
- Steinmetz, N.F.; Manchester, M. PEGylated viral nanoparticles for biomedicine: The impact of PEG chain length on VNP cell interactions in vitro and ex vivo. Biomacromolecules 2009, 10, 784–792. [Google Scholar] [CrossRef]
- Shukla, S.; Ablack, A.L.; Wen, A.M.; Lee, K.L.; Lewis, J.D.; Steinmetz, N.F. Increased tumor homing and tissue penetration of the filamentous plant viral nanoparticle Potato virus X. Mol. Pharm. 2013, 10, 33–42. [Google Scholar] [CrossRef]
- Chauhan, V.P.; Popović, Z.; Chen, O.; Cui, J.; Fukumura, D.; Bawendi, M.G.; Jain, R.K. Fluorescent nanorods and nanospheres for real-time in vivo probing of nanoparticle shape-dependent tumor penetration. Angew. Chem. 2011, 123, 11619–11622. [Google Scholar]
- Geng, Y.; Dalhaimer, P.; Cai, S.; Tsai, R.; Tewari, M.; Minko, T.; Discher, D.E. Shape effects of filaments versus spherical particles in flow and drug delivery. Nat. Nanotechnol. 2007, 2, 249–255. [Google Scholar] [PubMed]
- Shukla, S.; DiFranco, N.A.; Wen, A.M.; Commandeur, U.; Steinmetz, N.F. To target or not to target: Active vs. passive tumor homing of filamentous nanoparticles based on potato virus X. Cell. Mol. Bioeng. 2015, 8, 433–444. [Google Scholar] [PubMed]
- Zhong, L.; Li, Y.; Xiong, L.; Wang, W.; Wu, M.; Yuan, T.; Yang, W.; Tian, C.; Miao, Z.; Wang, T. Small molecules in targeted cancer therapy: Advances, challenges, and future perspectives. Signal Transduct. Target. Ther. 2021, 6, 1–48. [Google Scholar]
- Lin, R.D.; Steinmetz, N.F. Tobacco mosaic virus delivery of mitoxantrone for cancer therapy. Nanoscale 2018, 10, 16307–16313. [Google Scholar]
- Parhizkar, E.; Rafieipour, P.; Sepasian, A.; Alemzadeh, E.; Dehshahri, A.; Ahmadi, F. Synthesis and cytotoxicity evaluation of gemcitabine-tobacco mosaic virus conjugates. J. Drug Deliv. Sci. Technol. 2021, 62, 102388. [Google Scholar]
- Czapar, A.E.; Zheng, Y.-R.; Riddell, I.A.; Shukla, S.; Awuah, S.G.; Lippard, S.J.; Steinmetz, N.F. Tobacco mosaic virus delivery of phenanthriplatin for cancer therapy. ACS Nano 2016, 10, 4119–4126. [Google Scholar]
- Bruckman, M.A.; Czapar, A.E.; VanMeter, A.; Randolph, L.N.; Steinmetz, N.F. Tobacco mosaic virus-based protein nanoparticles and nanorods for chemotherapy delivery targeting breast cancer. J. Control. Release 2016, 231, 103–113. [Google Scholar]
- Alemzadeh, E.; Dehshahri, A.; Dehghanian, A.R.; Afsharifar, A.; Behjatnia, A.A.; Izadpanah, K.; Ahmadi, F. Enhanced anti-tumor efficacy and reduced cardiotoxicity of doxorubicin delivered in a novel plant virus nanoparticle. Colloids Surf. B Biointerfaces 2019, 174, 80–86. [Google Scholar]
- Hu, H.; Steinmetz, N.F. Doxorubicin-Loaded Physalis Mottle Virus Particles Function as a pH-Responsive Prodrug Enabling Cancer Therapy. Biotechnol. J. 2020, 15, 2000077. [Google Scholar]
- Mirza, Z.; Karim, S. Nanoparticles-based drug delivery and gene therapy for breast cancer: Recent advancements and future challenges. In Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 226–237. [Google Scholar]
- Lam, P.; Steinmetz, N.F. Delivery of siRNA therapeutics using cowpea chlorotic mottle virus-like particles. Biomater. Sci. 2019, 7, 3138–3142. [Google Scholar] [PubMed]
- Chadar, R.; Kesharwani, P. Nanotechnology-based siRNA delivery strategies for treatment of triple negative breast cancer. Int. J. Pharm. 2021, 605, 120835. [Google Scholar] [PubMed]
- Rohovie, M.J.; Nagasawa, M.; Swartz, J.R. Virus-like particles: Next-generation nanoparticles for targeted therapeutic delivery. Bioeng. Transl. Med. 2017, 2, 43–57. [Google Scholar] [PubMed]
- Carignan, D.; Herblot, S.; Laliberté-Gagné, M.-È.; Bolduc, M.; Duval, M.; Savard, P.; Leclerc, D. Activation of innate immunity in primary human cells using a plant virus derived nanoparticle TLR7/8 agonist. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2317–2327. [Google Scholar]
- Villagrana-Escareño, M.V.; Reynaga-Hernández, E.; Galicia-Cruz, O.G.; Durán-Meza, A.L.; la Cruz-González, D.; Hernández-Carballo, C.Y.; Ruíz-García, J. VLPs derived from the CCMV plant virus can directly transfect and deliver heterologous genes for translation into mammalian cells. BioMed Res. Int. 2019, 2019, 4630891. [Google Scholar]
- Biddlecome, A.; Habte, H.H.; McGrath, K.M.; Sambanthamoorthy, S.; Wurm, M.; Sykora, M.M.; Knobler, C.M.; Lorenz, I.C.; Lasaro, M.; Elbers, K. Delivery of self-amplifying RNA vaccines in in vitro reconstituted virus-like particles. PLoS ONE 2019, 14, e0215031. [Google Scholar]
- Pretto, C.; van Hest, J.C. Versatile reversible cross-linking strategy to stabilize CCMV virus like particles for efficient siRNA delivery. Bioconjugate Chem. 2019, 30, 3069–3077. [Google Scholar]
- Nuñez-Rivera, A.; Fournier, P.G.; Arellano, D.L.; Rodriguez-Hernandez, A.G.; Vazquez-Duhalt, R.; Cadena-Nava, R.D. Brome mosaic virus-like particles as siRNA nanocarriers for biomedical purposes. Beilstein J. Nanotechnol. 2020, 11, 372–382. [Google Scholar]
- Xue, F.; Cornelissen, J.J.; Yuan, Q.; Cao, S. Delivery of MicroRNAs by plant virus-based nanoparticles to functionally alter the osteogenic differentiation of human mesenchymal stem cells. Chin. Chem. Lett. 2022, in press. [Google Scholar] [CrossRef]
- Marqus, S.; Pirogova, E.; Piva, T.J. Evaluation of the use of therapeutic peptides for cancer treatment. J. Biomed. Sci. 2017, 24, 1–15. [Google Scholar]
- Esfandiari, N.; Arzanani, M.K.; Soleimani, M.; Kohi-Habibi, M.; Svendsen, W.E. A new application of plant virus nanoparticles as drug delivery in breast cancer. Tumor Biol. 2016, 37, 1229–1236. [Google Scholar]
- Shukla, S.; Wen, A.M.; Commandeur, U.; Steinmetz, N.F. Presentation of HER2 epitopes using a filamentous plant virus-based vaccination platform. J. Mater. Chem. B 2014, 2, 6249–6258. [Google Scholar] [PubMed]
- Shukla, S.; Myers, J.T.; Woods, S.E.; Gong, X.; Czapar, A.E.; Commandeur, U.; Huang, A.Y.; Levine, A.D.; Steinmetz, N.F. Plant viral nanoparticles-based HER2 vaccine: Immune response influenced by differential transport, localization and cellular interactions of particulate carriers. Biomaterials 2017, 121, 15–27. [Google Scholar]
- Esfandiari, N.; Arzanani, M.K.; Koohi-Habibi, M. The study of toxicity and pathogenicity risk of Potato Virus X/Herceptin nanoparticles as agents for cancer therapy. Cancer Nanotechnol. 2018, 9, 1–13. [Google Scholar]
- Park, J.; Chariou, P.L.; Steinmetz, N.F. Site-specific antibody conjugation strategy to functionalize virus-based nanoparticles. Bioconjugate Chem. 2020, 31, 1408–1416. [Google Scholar]
- Brunel, F.M.; Lewis, J.D.; Destito, G.; Steinmetz, N.F.; Manchester, M.; Stuhlmann, H.; Dawson, P.E. Hydrazone ligation strategy to assemble multifunctional viral nanoparticles for cell imaging and tumor targeting. Nano Lett. 2010, 10, 1093–1097. [Google Scholar]
- Le, D.H.; Commandeur, U.; Steinmetz, N.F. Presentation and delivery of tumor necrosis factor-related apoptosis-inducing ligand via elongated plant viral nanoparticle enhances antitumor efficacy. ACS Nano 2019, 13, 2501–2510. [Google Scholar]
- Zhong, H.-H.; Wang, H.-Y.; Li, J.; Huang, Y.-Z. TRAIL-based gene delivery and therapeutic strategies. Acta Pharmacol. Sin. 2019, 40, 1373–1385. [Google Scholar]
- Dianat-Moghadam, H.; Heidarifard, M.; Mahari, A.; Shahgolzari, M.; Keshavarz, M.; Nouri, M.; Amoozgar, Z. TRAIL in oncology: From recombinant TRAIL to nano-and self-targeted TRAIL-based therapies. Pharmacol. Res. 2020, 155, 104716. [Google Scholar]
- Chariou, P.L.; Lee, K.L.; Wen, A.M.; Gulati, N.M.; Stewart, P.L.; Steinmetz, N.F. Detection and imaging of aggressive cancer cells using an epidermal growth factor receptor (EGFR)-targeted filamentous plant virus-based nanoparticle. Bioconjugate Chem. 2015, 26, 262–269. [Google Scholar]
- Seshacharyulu, P.; Ponnusamy, M.P.; Haridas, D.; Jain, M.; Ganti, A.K.; Batra, S.K. Targeting the EGFR signaling pathway in cancer therapy. Expert Opin. Ther. Targets 2012, 16, 15–31. [Google Scholar] [PubMed]
- Gupta, S.; Wilder, H.; Rao, A.; Vullev, V.; Anvari, B. Plant virus-resembling optical nano-materials conjugated with anti-EGFR for targeted cancer imaging. In Reporters, Markers, Dyes, Nanoparticles, and Molecular Probes for Biomedical Applications IV; SPIE: Bellingham, WA, USA, 2012; pp. 120–126. [Google Scholar]
- Scaranti, M.; Cojocaru, E.; Banerjee, S.; Banerji, U. Exploiting the folate receptor α in oncology. Nat. Rev. Clin. Oncol. 2020, 17, 349–359. [Google Scholar] [PubMed]
- Ren, Y.; Wong, S.M.; Lim, L.-Y. Folic acid-conjugated protein cages of a plant virus: A novel delivery platform for doxorubicin. Bioconjugate Chem. 2007, 18, 836–843. [Google Scholar]
- Destito, G.; Yeh, R.; Rae, C.S.; Finn, M.; Manchester, M. Folic acid-mediated targeting of cowpea mosaic virus particles to tumor cells. Chem. Biol. 2007, 14, 1152–1162. [Google Scholar] [PubMed]
- Barwal, I.; Kumar, R.; Kateriya, S.; Dinda, A.K.; Yadav, S.C. Targeted delivery system for cancer cells consist of multiple ligands conjugated genetically modified CCMV capsid on doxorubicin GNPs complex. Sci. Rep. 2016, 6, 1–15. [Google Scholar]
- Wong, P.T.; Choi, S.K. Mechanisms and implications of dual-acting methotrexate in folate-targeted nanotherapeutic delivery. Int. J. Mol. Sci. 2015, 16, 1772–1790. [Google Scholar]
- Peng, J.; Yin, Y.; Liang, H.; Lu, Y.; Zheng, H.; Wu, G.; Rao, S.; Chen, J.; Yan, F.; Hu, J. Tumor Microenvironment Responsive Pepper Mild Mottle Virus-Based Nanotubes for Targeted Delivery and Controlled Release of Paclitaxel. Front. Bioeng. Biotechnol. 2021, 9, 763661. [Google Scholar]
- Cai, H.; Shukla, S.; Wang, C.; Masarapu, H.; Steinmetz, N.F. Heterologous prime-boost enhances the antitumor immune response elicited by plant-virus-based cancer vaccine. J. Am. Chem. Soc. 2019, 141, 6509–6518. [Google Scholar]
- Bianchini, G.; Gianni, L. The immune system and response to HER2-targeted treatment in breast cancer. Lancet Oncol. 2014, 15, e58–e68. [Google Scholar]
- Shukla, S.; Jandzinski, M.; Wang, C.; Gong, X.; Bonk, K.W.; Keri, R.A.; Steinmetz, N.F. A viral nanoparticle cancer vaccine delays tumor progression and prolongs survival in a HER2+ tumor mouse model. Adv. Ther. 2019, 2, 1800139. [Google Scholar]
- Abraham, A.; Natraj, U.; Karande, A.A.; Gulati, A.; Murthy, M.; Murugesan, S.; Mukunda, P.; Savithri, H.S. Intracellular delivery of antibodies by chimeric Sesbania mosaic virus (SeMV) virus like particles. Sci. Rep. 2016, 6, 1–12. [Google Scholar]
- Rachner, T.D.; Kasimir-Bauer, S.; Goebel, A.; Erdmann, K.; Hoffmann, O.; Rauner, M.; Hofbauer, L.C.; Kimmig, R.; Bittner, A.-K. Soluble Neuropilin-1 is an independent marker of poor prognosis in early breast cancer. J. Cancer Res. Clin. Oncol. 2021, 147, 2233–2238. [Google Scholar] [CrossRef] [PubMed]
- Bachelder, R.E.; Wendt, M.A.; Mercurio, A.M. Vascular endothelial growth factor promotes breast carcinoma invasion in an autocrine manner by regulating the chemokine receptor CXCR4. Cancer Res. 2002, 62, 7203–7206. [Google Scholar] [PubMed]
- Seifi-Alan, M.; Shams, R.; Bandehpour, M.; Mirfakhraie, R.; Ghafouri-Fard, S. Neuropilin-1 expression is associated with lymph node metastasis in breast cancer tissues. Cancer Manag. Res. 2018, 10, 1969. [Google Scholar] [CrossRef] [PubMed]
- Gamper, C.; Spenlé, C.; Boscá, S.; van der Heyden, M.; Erhardt, M.; Orend, G.; Bagnard, D.; Heinlein, M. Functionalized tobacco mosaic virus coat protein monomers and oligomers as nanocarriers for anti-cancer peptides. Cancers 2019, 11, 1609. [Google Scholar] [CrossRef]
- Kondo, E.; Iioka, H.; Saito, K. Tumor-homing peptide and its utility for advanced cancer medicine. Cancer Sci. 2021, 112, 2118–2125. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Li, J.; Yang, H.; Seoung, J.; Lim, H.-D.; Kim, G.-J.; Shin, H.-J. Targeted cowpea chlorotic mottle virus-based nanoparticles with tumor-homing peptide F3 for photothermal therapy. Biotechnol. Bioprocess Eng. 2017, 22, 700–708. [Google Scholar] [CrossRef]
- Hynes, R.O. Integrins: Bidirectional, allosteric signaling machines. Cell 2002, 110, 673–687. [Google Scholar] [CrossRef]
- Castel, S.; Pagan, R.; Mitjans, F.; Piulats, J.; Goodman, S.; Jonczyk, A.; Huber, F.; Vilaró, S.; Reina, M. RGD peptides and monoclonal antibodies, antagonists of αv-integrin, enter the cells by independent endocytic pathways. Lab. Investig. 2001, 81, 1615–1626. [Google Scholar] [CrossRef]
- Hovlid, M.L.; Steinmetz, N.F.; Laufer, B.; Lau, J.L.; Kuzelka, J.; Wang, Q.; Hyypiä, T.; Nemerow, G.R.; Kessler, H.; Manchester, M. Guiding plant virus particles to integrin-displaying cells. Nanoscale 2012, 4, 3698–3705. [Google Scholar]
- Evtushenko, E.A.; Ryabchevskaya, E.M.; Nikitin, N.A.; Atabekov, J.G.; Karpova, O.V. Plant virus particles with various shapes as potential adjuvants. Sci. Rep. 2020, 10, 1–10. [Google Scholar]
- Balke, I.; Zeltins, A. Use of plant viruses and virus-like particles for the creation of novel vaccines. Adv. Drug Deliv. Rev. 2019, 145, 119–129. [Google Scholar] [PubMed]
- Chariou, P.L.; Beiss, V.; Ma, Y.; Steinmetz, N.F. In situ vaccine application of inactivated CPMV nanoparticles for cancer immunotherapy. Mater. Adv. 2021, 2, 1644–1656. [Google Scholar]
- Chung, Y.H.; Church, D.; Koellhoffer, E.C.; Osota, E.; Shukla, S.; Rybicki, E.P.; Pokorski, J.K.; Steinmetz, N.F. Integrating plant molecular farming and materials research for next-generation vaccines. Nat. Rev. Mater. 2022, 7, 372–388. [Google Scholar]
- Lizotte, P.; Wen, A.; Sheen, M.; Fields, J.; Rojanasopondist, P.; Steinmetz, N.; Fiering, S. In situ vaccination with cowpea mosaic virus nanoparticles suppresses metastatic cancer. Nat. Nanotechnol. 2016, 11, 295–303. [Google Scholar]
- Beiss, V.; Mao, C.; Fiering, S.N.; Steinmetz, N.F. Cowpea Mosaic Virus Outperforms Other Members of the Secoviridae as In Situ Vaccine for Cancer Immunotherapy. Mol. Pharm. 2022, 19, 1573–1585. [Google Scholar] [PubMed]
- Murray, A.A.; Wang, C.; Fiering, S.; Steinmetz, N.F. In situ vaccination with cowpea vs tobacco mosaic virus against melanoma. Mol. Pharm. 2018, 15, 3700–3716. [Google Scholar]
- Lebel, M.-È.; Chartrand, K.; Tarrab, E.; Savard, P.; Leclerc, D.; Lamarre, A. Potentiating cancer immunotherapy using papaya mosaic virus-derived nanoparticles. Nano Lett. 2016, 16, 1826–1832. [Google Scholar]
- Wang, C.; Beiss, V.; Steinmetz, N.F. Cowpea mosaic virus nanoparticles and empty virus-like particles show distinct but overlapping immunostimulatory properties. J. Virol. 2019, 93, e00129-19. [Google Scholar]
- Comas-Garcia, M.; Garmann, R.F.; Singaram, S.W.; Ben-Shaul, A.; Knobler, C.M.; Gelbart, W.M. Characterization of viral capsid protein self-assembly around short single-stranded RNA. J. Phys. Chem. B 2014, 118, 7510–7519. [Google Scholar]
- Wang, C.; Steinmetz, N.F. CD47 Blockade and Cowpea Mosaic Virus Nanoparticle In Situ Vaccination Triggers Phagocytosis and Tumor Killing. Adv. Healthc. Mater. 2019, 8, 1801288. [Google Scholar] [CrossRef] [PubMed]
- Shahgolzari, M.; Pazhouhandeh, M.; Milani, M.; Fiering, S.; Khosroushahi, A.Y. Alfalfa mosaic virus nanoparticles-based in situ vaccination induces antitumor immune responses in breast cancer model. Nanomedicine 2020, 16, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Nuzzaci, M.; Piazzolla, G.; Vitti, A.; Lapelosa, M.; Tortorella, C.; Stella, I.; Natilla, A.; Antonaci, S.; Piazzolla, P. Cucumber mosaic virus as a presentation system for a double hepatitis C virus-derived epitope. Arch. Virol. 2007, 152, 915–928. [Google Scholar] [PubMed]
- Zeltins, A.; West, J.; Zabel, F.; El Turabi, A.; Balke, I.; Haas, S.; Maudrich, M.; Storni, F.; Engeroff, P.; Jennings, G.T. Incorporation of tetanus-epitope into virus-like particles achieves vaccine responses even in older recipients in models of psoriasis, Alzheimer’s and cat allergy. npj Vaccines 2017, 2, 1–13. [Google Scholar] [CrossRef]
- Mohsen, M.O.; Heath, M.D.; Cabral-Miranda, G.; Lipp, C.; Zeltins, A.; Sande, M.; Stein, J.V.; Riether, C.; Roesti, E.; Zha, L. Vaccination with nanoparticles combined with micro-adjuvants protects against cancer. J. Immunother. Cancer 2019, 7, 1–12. [Google Scholar]
- Shukla, S.; Wang, C.; Beiss, V.; Cai, H.; Washington, T.; Murray, A.A.; Gong, X.; Zhao, Z.; Masarapu, H.; Zlotnick, A. The unique potency of Cowpea mosaic virus (CPMV) in situ cancer vaccine. Biomater. Sci. 2020, 8, 5489–5503. [Google Scholar] [CrossRef]
- Lebel, M.-È.; Daudelin, J.-F.; Chartrand, K.; Tarrab, E.; Kalinke, U.; Savard, P.; Labrecque, N.; Leclerc, D.; Lamarre, A. Nanoparticle adjuvant sensing by TLR7 enhances CD8+ T cell–mediated protection from Listeria monocytogenes infection. J. Immunol. 2014, 192, 1071–1078. [Google Scholar] [CrossRef]
- Boone, C.E.; Wang, L.; Gautam, A.; Newton, I.G.; Steinmetz, N.F. Combining nanomedicine and immune checkpoint therapy for cancer immunotherapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2022, 14, e1739. [Google Scholar] [CrossRef]
- Lee, K.L.; Murray, A.A.; Le, D.H.; Sheen, M.R.; Shukla, S.; Commandeur, U.; Fiering, S.; Steinmetz, N.F. Combination of plant virus nanoparticle-based in situ vaccination with chemotherapy potentiates antitumor response. Nano Lett. 2017, 17, 4019–4028. [Google Scholar]
- Patel, R.; Czapar, A.E.; Fiering, S.; Oleinick, N.L.; Steinmetz, N.F. Radiation therapy combined with cowpea mosaic virus nanoparticle in situ vaccination initiates immune-mediated tumor regression. ACS Omega 2018, 3, 3702–3707. [Google Scholar] [CrossRef]
- Wang, C.; Steinmetz, N.F. A combination of cowpea mosaic virus and immune checkpoint therapy synergistically improves therapeutic efficacy in three tumor models. Adv. Funct. Mater. 2020, 30, 2002299. [Google Scholar] [CrossRef] [PubMed]
- Gautam, A.; Beiss, V.; Wang, C.; Wang, L.; Steinmetz, N.F. Plant viral nanoparticle conjugated with anti-PD-1 peptide for ovarian cancer immunotherapy. Int. J. Mol. Sci. 2021, 22, 9733. [Google Scholar] [PubMed]
- Koellhoffer, E.C.; Mao, C.; Beiss, V.; Wang, L.; Fiering, S.N.; Boone, C.E.; Steinmetz, N.F. Inactivated Cowpea Mosaic Virus in Combination with OX40 Agonist Primes Potent Antitumor Immunity in a Bilateral Melanoma Mouse Model. Mol. Pharm. 2022, 19, 592–601. [Google Scholar] [PubMed]
- Compte, M.; Harwood, S.L.; Muñoz, I.G.; Navarro, R.; Zonca, M.; Perez-Chacon, G.; Erce-Llamazares, A.; Merino, N.; Tapia-Galisteo, A.; Cuesta, A.M. A tumor-targeted trimeric 4-1BB-agonistic antibody induces potent anti-tumor immunity without systemic toxicity. Nat. Commun. 2018, 9, 1–13. [Google Scholar]
- Koellhoffer, E.C.; Steinmetz, N.F. Cowpea Mosaic Virus and Natural Killer Cell Agonism for In Situ Cancer Vaccination. Nano Lett. 2022, 22, 5348–5356. [Google Scholar] [CrossRef]
- Yuan, J.; Shi, X.; Chen, C.; He, H.; Liu, L.; Wu, J.; Yan, H. High expression of CD47 in triple negative breast cancer is associated with epithelial-mesenchymal transition and poor prognosis. Oncol. Lett. 2019, 18, 3249–3255. [Google Scholar]
- Nkanga, C.I.; Ortega-Rivera, O.A.; Steinmetz, N.F. Photothermal immunotherapy of melanoma using TLR-7 agonist laden tobacco mosaic virus with polydopamine coat. Nanomed. Nanotechnol. Biol. Med. 2022, 44, 102573. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shahgolzari, M.; Dianat-Moghadam, H.; Yavari, A.; Fiering, S.N.; Hefferon, K. Multifunctional Plant Virus Nanoparticles for Targeting Breast Cancer Tumors. Vaccines 2022, 10, 1431. https://doi.org/10.3390/vaccines10091431
Shahgolzari M, Dianat-Moghadam H, Yavari A, Fiering SN, Hefferon K. Multifunctional Plant Virus Nanoparticles for Targeting Breast Cancer Tumors. Vaccines. 2022; 10(9):1431. https://doi.org/10.3390/vaccines10091431
Chicago/Turabian StyleShahgolzari, Mehdi, Hassan Dianat-Moghadam, Afagh Yavari, Steven N. Fiering, and Kathleen Hefferon. 2022. "Multifunctional Plant Virus Nanoparticles for Targeting Breast Cancer Tumors" Vaccines 10, no. 9: 1431. https://doi.org/10.3390/vaccines10091431
APA StyleShahgolzari, M., Dianat-Moghadam, H., Yavari, A., Fiering, S. N., & Hefferon, K. (2022). Multifunctional Plant Virus Nanoparticles for Targeting Breast Cancer Tumors. Vaccines, 10(9), 1431. https://doi.org/10.3390/vaccines10091431







