COVID-19 Vaccine Hesitancy among Parents of Children under Five Years in the United States
Abstract
:1. Introduction
2. Materials and Methods
2.1. Demographic Items
2.2. Survey Items Assessing COVID-19 Pediatric Vaccination Facilitators and Barriers
2.3. Intent to Vaccinate
2.4. Open Ended Response to Reasons for Vaccine Intent
3. Results
3.1. Intent to Vaccinate and Demographic Characteristics
3.2. Parental Attitudes toward COVID-19 Pediatric Vaccination Survey Items
3.3. Relationships among Intent to Vaccinate, Parental Attitudes, and Demographics
3.4. In Their Own Words: Qualitative Data on Parents’ Reasons for Their Intention to Vaccinate or Not Vaccinate Their Child
3.4.1. Theme 1—Vaccine Safety Concerns
3.4.2. Theme 2—Distrust in Government, Science, or the Pharmaceutical Industry
3.4.3. Theme 3—The Vaccine Is Unnecessary
3.4.4. Theme 4—My Child Is Too Young
3.4.5. Theme 5—The Vaccine Protects My Child and Others
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FDA Coronavirus (COVID-19) Update: FDA Authorizes Moderna and Pfizer-BioNTech COVID-19 Vaccines for Children Down to 6 Months of Age. Available online: https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-moderna-and-pfizer-biontech-covid-19-vaccines-children (accessed on 20 July 2022).
- Feming-Dutra, K.C. COVID-19 Epidemiology in Children Ages 6 Months–4 Years. Available online: https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2022-06-17-18/02-COVID-Fleming-Dutra-508.pdf (accessed on 11 August 2022).
- CDC. COVID Data Tracker. Available online: https://covid.cdc.gov/covid-data-tracker (accessed on 20 July 2022).
- Fisher, C.B.; Gray, A.; Sheck, I. COVID-19 Pediatric Vaccine Hesitancy among Racially Diverse Parents in the United States. Vaccines 2022, 10, 31. [Google Scholar] [CrossRef]
- Szilagyi, P.G.; Shah, M.D.; Delgado, J.R.; Thomas, K.; Vizueta, N.; Cui, Y.; Vangala, S.; Shetgiri, R.; Kapteyn, A. Parents’ Intentions and Perceptions About COVID-19 Vaccination for Their Children: Results from a National Survey. Pediatrics 2021, 148, e2021052335. [Google Scholar] [CrossRef] [PubMed]
- Kaiser Family Foundation. The Last Major Phase of the COVID-19 Vaccination Roll-Out: Children under 5. Available online: https://www.kff.org/coronavirus-covid-19/issue-brief/the-last-major-phase-of-the-covid-19-vaccination-roll-out-children-under-5/ (accessed on 11 August 2022).
- Shmueli, L. Predicting Intention to Receive COVID-19 Vaccine among the General Population Using the Health Belief Model and the Theory of Planned Behavior Model. BMC Public Health 2021, 21, 804. [Google Scholar] [CrossRef] [PubMed]
- Limbu, Y.B.; Gautam, R.K.; Pham, L. The Health Belief Model Applied to COVID-19 Vaccine Hesitancy: A Systematic Review. Vaccines 2022, 10, 973. [Google Scholar] [CrossRef]
- Alfieri, N.L.; Kusma, J.D.; Heard-Garris, N.; Davis, M.M.; Golbeck, E.; Barrera, L.; Macy, M.L. Parental COVID-19 Vaccine Hesitancy for Children: Vulnerability in an Urban Hotspot. BMC Public Health 2021, 21, 1662. [Google Scholar] [CrossRef] [PubMed]
- Pippert, C.H.; Trujillo, K.L.; Lazer, D.; Baum, M.; Simonson, M.D.; Ognyanova, K.; Perlis, R.; Druckman, J.; Santillana, M.; Quintana, A.; et al. The COVID States Project #68: Heightened Parental Concerns about COVID-19 Vaccinations for Children; OSFPREPRINT. 2021. Available online: https://osf.io/4u6hb/ (accessed on 11 August 2022).
- Ruggiero, K.M.; Wong, J.; Sweeney, C.F.; Avola, A.; Auger, A.; Macaluso, M.; Reidy, P. Parents’ Intentions to Vaccinate Their Children Against COVID-19. J. Pediatr. Health Care 2021, 35, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Shacham, E.; Little, G.; Scroggins, S.; Fredman, A.; Wray, R.; Charney, R. Intent to Vaccinate Children against COVID-19: Low Levels of Trust in Public Health Agencies Trust across Rural Communities; SocArXiv Papers. 2021. Available online: https://osf.io/preprints/socarxiv/9f3r6/ (accessed on 11 August 2022).
- Teasdale, C.A.; Borrell, L.N.; Shen, Y.; Kimball, S.; Rinke, M.L.; Fleary, S.A.; Nash, D. Parental Plans to Vaccinate Children for COVID-19 in New York City. Vaccine 2021, 39, 5082–5086. [Google Scholar] [CrossRef] [PubMed]
- Teherani, M.; Banskota, S.; Camacho-Gonzalez, A.; Smith, A.G.C.; Anderson, E.J.; Kao, C.M.; Crepy D’Orleans, C.; Shane, A.L.; Lu, A.; Jaggi, P. Intent to Vaccinate SARS-CoV-2 Infected Children in US Households: A Survey. Vaccines 2021, 9, 1049. [Google Scholar] [CrossRef] [PubMed]
- Temsah, M.-H.; Alhuzaimi, A.N.; Aljamaan, F.; Bahkali, F.; Al-Eyadhy, A.; Alrabiaah, A.; Alhaboob, A.; Bashiri, F.A.; Alshaer, A.; Temsah, O.; et al. Parental Attitudes and Hesitancy About COVID-19 vs. Routine Childhood Vaccinations: A National Survey. Front. Public Health 2021, 9, 752323. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Guo, Y.; Zhou, Q.; Tan, Z.; Cao, J. The Mediating Roles of Medical Mistrust, Knowledge, Confidence and Complacency of Vaccines in the Pathways from Conspiracy Beliefs to Vaccine Hesitancy. Vaccines 2021, 9, 1342. [Google Scholar] [CrossRef] [PubMed]
- Teasdale, C.A.; Borrell, L.N.; Kimball, S.; Rinke, M.L.; Rane, M.; Fleary, S.A.; Nash, D. Plans to Vaccinate Children for Coronavirus Disease 2019: A Survey of United States Parents. J. Pediatr. 2021, 237, 292–297. [Google Scholar] [CrossRef]
- Gray, A.; Fisher, C.B. Determinants of COVID-19 Vaccine Uptake in Adolescents 12–17 Years Old: Examining Pediatric Vaccine Hesitancy Among Racially Diverse Parents in the United States. Front. Public Health 2022, 10, 844310. [Google Scholar] [CrossRef]
- Kaiser Family Foundation. Latest Data on COVID-19 Vaccinations by Race/Ethnicity; USA. 2022. Available online: https://www.kff.org/coronavirus-covid-19/issue-brief/latest-data-on-covid-19-vaccinations-by-race-ethnicity/ (accessed on 11 August 2022).
- CDC. Omicron Variant: What You Need to Know. Available online: https://www.cdc.gov/coronavirus/2019-ncov/variants/omicron-variant.html (accessed on 25 July 2022).
- Sigal, A.; Milo, R.; Jassat, W. Estimating Disease Severity of Omicron and Delta SARS-CoV-2 Infections. Nat. Rev. Immunol. 2022, 22, 267–269. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.E.; Jones, J.M.; Deng, Y.; Nycz, E.; Lee, A.; Iachan, R.; Gundlapalli, A.V.; Hall, A.J.; MacNeil, A. Seroprevalence of Infection-Induced SARS-CoV-2 Antibodies—United States, September 2021–February 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 606–608. [Google Scholar] [CrossRef] [PubMed]
- Kaiser Family Foundation. COVID-19 Vaccine Monitor: Parents and the Pandemic. Available online: https://www.kff.org/coronavirus-covid-19/poll-finding/kff-covid-19-vaccine-monitor-parents-and-the-pandemic/ (accessed on 25 July 2022).
- Montalti, M.; Rallo, F.; Guaraldi, F.; Bartoli, L.; Po, G.; Stillo, M.; Perrone, P.; Squillace, L.; Dallolio, L.; Pandolfi, P.; et al. Would Parents Get Their Children Vaccinated Against SARS-CoV-2? Rate and Predictors of Vaccine Hesitancy According to a Survey over 5000 Families from Bologna, Italy. Vaccines 2021, 9, 366. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, S.A.; Savas, L.S.; Baumler, E.; Nyitray, A.G.; Mullen, P.D.; Vernon, S.W.; Fernandez, M.E. Parental Predictors of HPV Vaccine Initiation among Low-Income Hispanic Females Aged 11–17 years. Vaccine 2018, 36, 5084–5090. [Google Scholar] [CrossRef] [PubMed]
- Braun, V.; Clarke, V. Using Thematic Analysis in Psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Cohen, J. Weighted Kappa: Nominal Scale Agreement Provision for Scaled Disagreement or Partial Credit. Psychol. Bull. 1968, 70, 213–220. [Google Scholar] [CrossRef]
- Bujang, M.A.; Sa’at, N.; Sidik, T.M.I.T.A.B.; Joo, L.C. Sample Size Guidelines for Logistic Regression from Observational Studies with Large Population: Emphasis on the Accuracy Between Statistics and Parameters Based on Real Life Clinical Data. Malays. J. Med. Sci. 2018, 25, 122–130. [Google Scholar] [CrossRef] [PubMed]
- FDA. FDA Briefing Document EUA Amendment Request for Pfizer-BioNTech COVID-19 Vaccine for Use in Children 6 Months through 4 Years of Age. 2022. Available online: https://www.fda.gov/media/159195/download (accessed on 27 July 2022).
- FDA. FDA Briefing Document EUA Amendment Request for Use of the Moderna COVID-19 Vaccine for Use in Children 6 Months through 17 Years of Age. 2022. Available online: https://www.fda.gov/media/159189/download (accessed on 27 July 2022).
- CDC, Polio Vaccine Effectiveness and Duration of Protection. Available online: https://www.cdc.gov/vaccines/vpd/polio/hcp/effectiveness-duration-protection.html (accessed on 25 July 2022).
- CDC, About Measles Vaccination. Available online: https://www.cdc.gov/vaccines/vpd/measles/index.html (accessed on 25 July 2022).
| Total Sample n = 411 | Resistant n = 190 (46.2%) | Unsure n = 93 (22.6%) | Accepting n = 128 (31.1%) | X2 | |
|---|---|---|---|---|---|
| n (%) | n (%) | n (%) | n (%) | ||
| Parent age, M (SD), Range = 21–50 years | 32.14 (6.18) | 31.59 (6.19) | 31.26 (6.16) | 33.59 (5.97) | F = 5.36 ** |
| Child’s age (youngest between 1–4-years) | 6.96 n.s. | ||||
| 1 year | 127 (30.9) | 58 (30.5%) | 33 (35.5%) | 36 (28.1%) | |
| 2 years | 119 (29.0) | 46 (24.2%) | 30 (32.3%) | 43 (33.6%) | |
| 3 years | 94 (22.9) | 50 (26.3%) | 18 (19.4%) | 26 (20.3%) | |
| 4 years | 71 (17.3) | 36 (18.9%) | 12 (12.9%) | 23 (18.0%) | |
| Race/Ethnicity | 9.55 n.s. | ||||
| Non-Hispanic Asian | 101 (24.6) | 36 (18.9) | 26 (28.0) | 39 (30.5) | |
| Non-Hispanic Black | 103 (25.1) | 54 (28.4) | 25 (26.9) | 24 (18.8) | |
| Hispanic/Latina | 103 (25.1) | 47 (24.7) | 24 (25.8) | 32 (25.0) | |
| Non-Hispanic White | 104 (25.3) | 53 (27.9) | 18 (19.4) | 33 (25.8) | |
| Education | 13.02 ** | ||||
| Did not attend college | 134 (32.6) | 79 (41.6) | 24 (25.8) | 31 (24.2) | |
| Some college or higher | 277 (67.4) | 111 (58.4) | 69 (74.2) | 97 (75.8) | |
| Annual household income (n = 402) | 15.72 ** | ||||
| <$20,000 | 81 (20.1) | 41 (21.9) | 18 (19.4) | 22 (18.0) | |
| Between $20,000 and 50,999 | 140 (34.8) | 72 (38.5) | 40 (43.0) | 28 (23.0) | |
| $51,000 and above | 181 (45.0) | 74 (39.6) | 35 (37.6) | 73 (59.0) | |
| Financial security | 7.54 * | ||||
| Cannot make ends meet | 97 (23.6) | 56 (29.5) | 20 (21.5) | 21 (16.4) | |
| Have just enough or comfortable | 314 (76.4) | 134 (70.5) | 73 (78.5) | 107 (83.6) | |
| Region of residence (n = 404) | 10.98 n.s. | ||||
| Northeast | 59 (14.6) | 25 (13.5) | 17 (18.5) | 17 (13.4) | |
| Midwest | 85 (21.0) | 47 (25.4) | 16 (17.4) | 22 (17.3) | |
| South | 162 (40.1) | 79 (42.7) | 36 (39.1) | 47 (37.0) | |
| West | 98 (24.3) | 34 (18.4) | 23 (25.0) | 41 (32.3) | |
| Mother’s vaccine status | 100.69 *** | ||||
| No | 130 (31.6) | 105 (55.3) | 21 (22.6) | 4 (3.1) | |
| Yes | 281 (68.4) | 85 (44.7) | 72 (77.4) | 124 (96.9) | |
| Mother had COVID-19 (n = 409) | 8.30 * | ||||
| No | 256 (62.6) | 106 (55.8) | 59 (64.1) | 91 (71.7) | |
| Yes | 153 (37.4) | 84 (44.2) | 33 (35.9) | 36 (28.3) | |
| Child’s (1–4 years) Routine Vaccinations: MMR, Polio, DtaP, Chickenpox | |||||
| Received all four | 277 (67.4) | 121 (63.7) | 64 (68.8) | 92 (71.9) | |
| Received at least one | 368 (89.5) | 164 (86.3) | 87 (93.5) | 117 (91.4) | 4.17 n.s. |
| Received none | 47 (11.1) | 26 (13.7) | 6 (6.5) | 11 (8.6) | |
| Child (1–4 years) received annual flu vaccine | 37.84 *** | ||||
| Yes | 254 (61.8) | 89 (46.8) | 62 (66.7) | 103 (80.5) | |
| No | 157 (38.2) | 101 (53.2) | 31 (33.3) | 25 (19.5) | |
| Other children aged 5–18 have received COVID-19 vaccine (n = 199) | 76.58 *** | ||||
| Yes | 73 (36.7) | 11 (10.6) | 16 (43.2) | 46 (79.3) | |
| No | 126 (63.3) | 93 (89.4) | 21 (56.8) | 12 (20.7) |
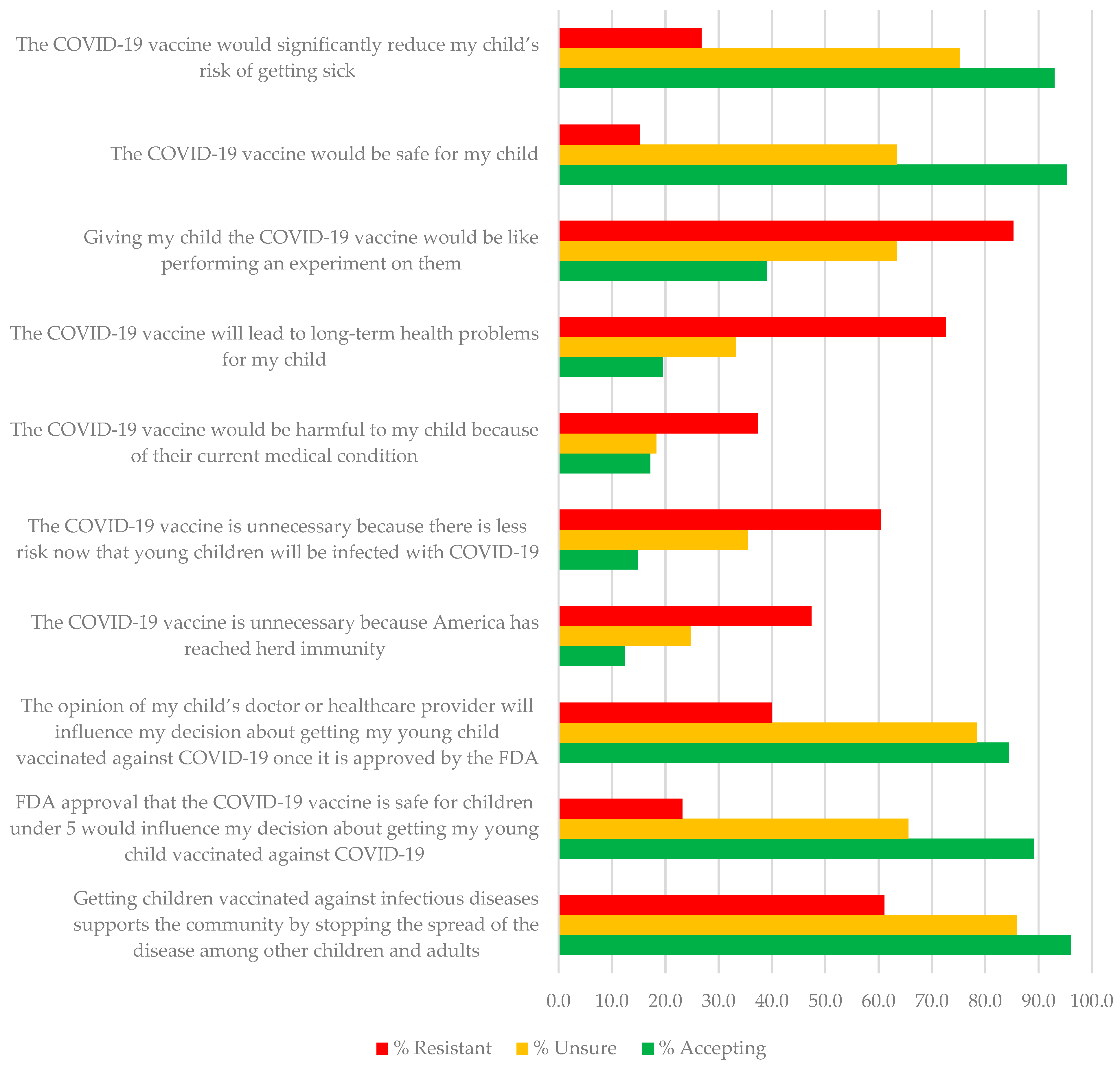
| Survey Item or Scale | Total n = 411 | Resistant n = 190 | Unsure n = 93 | Accepting n = 128 | X2 (2) | ||||
|---|---|---|---|---|---|---|---|---|---|
| M (SD) | n (%) | M (SD) | n (%) | M (SD) | n (%) | M (SD) | N (%) | ||
| The COVID-19 vaccine would significantly reduce my child’s risk of getting sick (Vaccine Efficacy) | 3.57 (1.59) | 240 (58.4) | 2.46 (1.32) | 51 (26.8) | 3.96 (1.00) | 70 (75.3) | 4.94 (0.99) | 119 (93.0) | 151.73 *** |
| The COVID-19 vaccine would be safe for my child (Vaccine Safety) | 3.42 (1.57) | 210 (51.1) | 2.27 (1.25) | 29 (15.3) | 3.75 (0.99) | 59 (63.4) | 4.89 (0.86) | 122 (95.3) | 203.45 *** |
| Giving my child the COVID-19 vaccine would be like performing an experiment on them (Vaccine Distrust) | 3.98 (1.52) | 271 (65.9) | 4.74 (1.35) | 162 (85.3) | 3.72 (1.04) | 59 (63.4) | 3.02 (1.44) | 50 (39.1) | 73.01 *** |
| The COVID-19 vaccine will lead to long-term health problems for my child (Vaccine Long-Term Health Problems) | 3.43 (1.40) | 194 (47.2) | 4.16 (1.29) | 138 (72.6) | 3.20 (0.95) | 31 (33.3) | 2.52 (1.23) | 25 (19.5) | 95.80 *** |
| The COVID-19 vaccine would be harmful to my child because of their current medical condition (Child’s Medical Condition) | 2.73 (1.40) | 110 (26.8) | 3.14 (1.48) | 71 (37.4) | 2.56 (1.07) | 17 (18.3) | 2.25 (1.30) | 22 (17.2) | 20.31 *** |
| The COVID-19 vaccine is unnecessary because there is less risk now that young children will be infected with COVID-19 (Less Risk of Infection) | 3.16 (1.47) | 167 (40.6) | 3.82 (1.46) | 115 (60.5) | 3.03 (1.14) | 33 (35.5) | 2.28 (1.20) | 19 (14.8) | 67.48 *** |
| The COVID-19 vaccine is unnecessary because America has reached herd immunity (Herd Immunity Has Been Reached) | 2.82 (1.49) | 129 (31.4) | 3.45 (1.40) | 90 (47.4) | 2.71 (1.21) | 23 (24.7) | 1.95 (1.19) | 16 (12.5) | 45.65 *** |
| The opinion of my child’s doctor or healthcare provider will influence my decision about getting my young child vaccinated against COVID-19 once it is approved by the FDA (Confidence in Provider) | 3.75 (1.53) | 257 (62.5) | 2.95 (1.49) | 76 (40.0) | 4.25 (1.10) | 73 (78.5) | 4.58 (1.25) | 108 (84.4) | 77.35 *** |
| FDA approval that the COVID-19 vaccine is safe for children under 5 would influence my decision about getting my young child vaccinated against COVID-19 (Confidence in FDA) | 3.48 (1.64) | 219 (53.3) | 2.44 (1.42) | 44 (23.2) | 3.72 (1.17) | 61 (65.6) | 4.84 (1.05) | 114 (89.1) | 140.76 *** |
| Getting children vaccinated against infectious diseases supports the community by stopping the spread of the disease among other children and adults (Stopping Community Spread) | 4.22 (1.39) | 319 (77.6) | 3.50 (1.42) | 116 (61.1) | 4.37 (1.00) | 80 (86.0) | 5.18 (0.87) | 123 (96.1) | 58.94 *** |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Intent to Vaccinate | - | ||||||||||||||||
| 2. Belief in Vaccine Efficacy | 0.71 *** | - | |||||||||||||||
| 3. Belief in Vaccine Safety | 0.75 *** | 0.82 *** | - | ||||||||||||||
| 4. Vaccine Distrust | −0.52 *** | −0.57 *** | −0.60 *** | - | |||||||||||||
| 5. Vaccine long-term health problems | −0.54 *** | −0.53 *** | −0.59 *** | 0.69 *** | - | ||||||||||||
| 6. Child’s medical condition | −0.29 *** | −0.31 *** | −0.30 *** | 0.43 *** | 0.57 *** | - | |||||||||||
| 7. Less Risk of Infection | −0.47 *** | −0.46 *** | −0.45 *** | 0.51 *** | 0.54 *** | 0.47 *** | - | ||||||||||
| 8. Herd Immunity Has Been Reached | −0.44 *** | −0.42 *** | −0.44 *** | 0.43 *** | 0.53 *** | 0.46 *** | 0.69 *** | - | |||||||||
| 9. Confidence in Provider | 0.50 *** | 0.59 *** | 0.57 *** | −0.38 *** | −0.39 *** | −0.16 *** | −0.28 *** | −0.25 *** | - | ||||||||
| 10. Confidence in FDA | 0.66 *** | 0.69 *** | 0.70 *** | −0.48 *** | −0.44 *** | −0.17 ** | −0.37 *** | −0.32 *** | 0.67 *** | - | |||||||
| 11. Stopping Spread | 0.53 *** | 0.60 *** | 0.60 *** | −0.34 *** | −0.38 *** | −0.28 *** | −0.32 *** | −0.35 *** | 0.54 *** | 0.58 *** | - | ||||||
| 12. Community Support | 0.48 *** | 0.44 *** | 0.44 *** | −0.29 *** | −0.32 *** | −0.16 ** | −0.31 *** | −0.33 *** | 0.32 *** | 0.37 *** | 0.34 *** | - | |||||
| 13. Parent Age | 0.12 * | 0.08 | 0.12 * | 0.00 | 0.01 | 0.04 | −0.01 | −0.09 | 0.13 ** | 0.11 * | 0.11 * | 0.11 * | - | ||||
| 14. Education | 0.28 *** | 0.19 ** | 0.24 *** | −0.09 | −0.12 * | −0.08 | −0.11 * | −0.11 * | 0.10 * | 0.19 *** | 0.14 ** | 0.15 ** | 0.23 *** | - | |||
| 15. Household Income | 0.16 ** | 0.11 * | 0.13 * | −0.09 | −0.17 ** | −0.17 *** | −0.05 | −0.06 | 0.10 * | 0.12 * | 0.13 * | 0.06 | 0.27 *** | 0.51 *** | - | ||
| 16. Mother Vaccinated | 0.53 *** | 0.51 ** | 0.53 *** | −0.35 *** | −0.36 *** | −0.16 *** | −0.30 *** | −0.27 *** | 0.43 *** | 0.51 *** | 0.35 *** | 0.33 *** | 0.16 ** | 0.29 *** | 0.29 *** | ||
| 17. Child Age | −0.02 | −0.05 | −0.05 | 0.04 | −0.02 | 0.01 | 0.01 | −0.004 | −0.04 | −0.10 * | −0.09 | −0.05 | 0.19 *** | 0.03 | 0.10 * | −0.04 | - |
| 18. Children 5–18 Vaccinated (n = 199) | 0.62 *** | 0.55 *** | 0.60 *** | −0.43 *** | −0.40 *** | −0.19 ** | −0.37 *** | −0.30 *** | 0.37 *** | 0.47 *** | 0.44 *** | 0.29 *** | 0.21 ** | 0.21 ** | 0.12 | 0.47 *** | 0.04 |
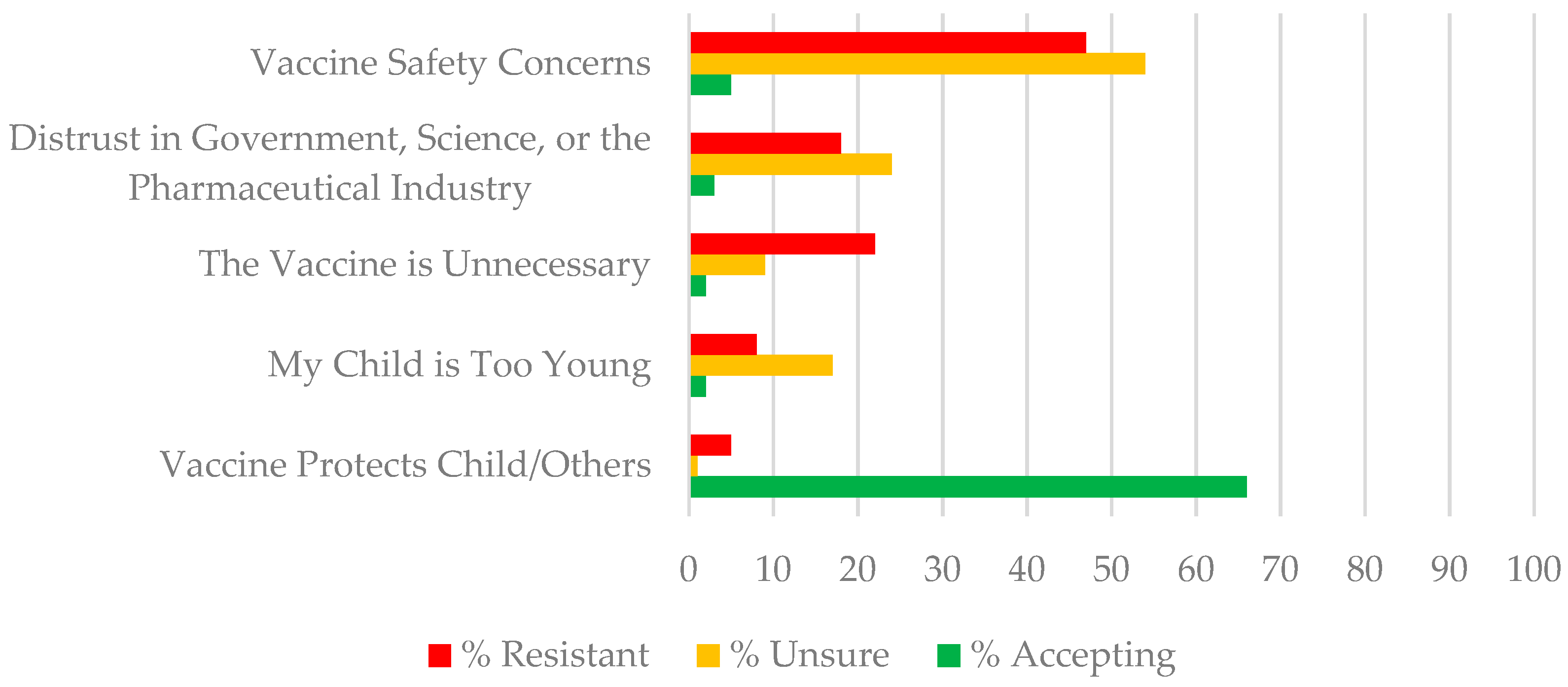
| Theme | Total Sample | Illustrative Statements | |||
|---|---|---|---|---|---|
| Resistant | Unsure | Accepting | |||
| N | % | ||||
| Theme 1: Vaccine Safety Concerns This theme reflects concern that vaccines in general or the COVID-19 vaccine specifically is unsafe including: concerns about immediate and long-term side effects, and the rushed nature of vaccine approval. | 146 | 33% | “I had a pretty bad reaction to my COVID vaccines, as well as many of my family members, I don’t want to risk my child having an even worse reaction to a vaccine.” “There is absolutely no long-term data available for the vaccine. It was rushed and there is no knowledge of long-term side effects.” “This vaccine has been approved for emergency purposes only. It’s the only vaccine in history who’s been used without long term testing. I wouldn’t subject my children to this kind of thing” | “I will wait a couple of months. Because of the continuous trials about the doses and the vaccine not working in this age group. I have followed these updates and I want to be sure.” “I don’t feel like there’s been thorough research about it. I definitely don’t go off word of mouth from the CDC or FDA especially when it comes to the health of my kids. If the pros outweigh the cons then surely they will get vaccines, but we’ve went 2 years without catching COVID so…” | “I would allow time for other parents to see how their children react but then I would because I know it would keep my child healthy and to be a member of society it is required a lot” “I’m afraid because the words “emergency approval” kind of scares me especially when it comes to my children. I have a 3 year-old and an 8 month-old. Emergency makes it seemed like it wasn’t tested as long as it needs to show proper results. However, most likely I will vaccinate my children.” |
| Theme 2: Distrust in Government, Science, or the Pharmaceutical Industry This theme reflects a strong distrust in the vaccine research and promotion in general or COVID-19 vaccine specifically for reasons including: not wanting their child to be a guinea pig; distrust of government or pharmaceutical company intentions; and distrust created by mixed media messaging | 88 | 20% | “The pharmaceuticals are refusing to release the study records to see how safe and effective this vaccine actually is” “COVID19 was launched as a pandemic to distract the public from being attentive to the Black Lives Matter movement.” | “I’m unsure because we hear new stuff all the time about all of the COVID vaccines and you don’t know what to believe any more” “I definitely don’t go off word of mouth from the CDC or FDA especially when it comes to the health of my kids.” | “I’m like 99 percent sure I would do it, but depending on how rushed it was I might feel some reservations about it.” |
| Theme 3: The Vaccine is Unnecessary This theme includes statements rejecting the vaccine as unnecessary because: Most people do not get COVID-19 anymore, children are not susceptible to COVID-19, COVID-19 symptoms are mild, natural immunity is preferable, or that it does not work on evidence that vaccinated people still can become infected, | 53 | 12% | “The children had it including my three-year-old and the symptoms for him were very minor to none. We actually had it twice and both times his symptoms were pretty minor. I also feel like they should have some immunity against the virus now and getting shots every few months, without enough years gone by to see the side effects of the shot, just isn’t an option for us.” | “I’m unsure because the effectiveness in the vaccine for children 5–11 has decreased” | “I’m a full time homemaker with 2 year twins who aren’t in daycare and we rarely go outside the community. Community spread has been consistently low and we have spent two years with COVID so far. These variables make me think twice about getting them vaccinated but I will probably end up choosing to vaccinate.” |
| Theme 4: Child Too Young This theme reflected primary concern about increased vaccination risk for young children. | 35 | 8% | “My child is only three I don’t feel comfortable getting her the COVID vaccination” | “I don’t if at my child’s age she could get the vaccine. I know older kids can get it but mine is only 2 1/2 years old” | “I might plan to when he is 6 because I believe in vaccinating my child.” |
| Theme 5: The Vaccine Protects My Child and Others This theme reflected confidence that the vaccine would be effective in protecting their child and others from COVID-19 infection. | 87 | 21.2% | “They will get it to help out” | “I want my child to be protected but I am also scared because instead of helping her become immune it may cause severe problems to her health” | “I want to insure that I have done what I can to protect my children. The pros outweigh the cons. The risk of COVID is horrible, the symptoms that COVID long haulers are dealing with are horrible. I’m not going to stop something that can prevent that for my child.” |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fisher, C.B.; Bragard, E.; Jaber, R.; Gray, A. COVID-19 Vaccine Hesitancy among Parents of Children under Five Years in the United States. Vaccines 2022, 10, 1313. https://doi.org/10.3390/vaccines10081313
Fisher CB, Bragard E, Jaber R, Gray A. COVID-19 Vaccine Hesitancy among Parents of Children under Five Years in the United States. Vaccines. 2022; 10(8):1313. https://doi.org/10.3390/vaccines10081313
Chicago/Turabian StyleFisher, Celia B., Elise Bragard, Rimah Jaber, and Aaliyah Gray. 2022. "COVID-19 Vaccine Hesitancy among Parents of Children under Five Years in the United States" Vaccines 10, no. 8: 1313. https://doi.org/10.3390/vaccines10081313
APA StyleFisher, C. B., Bragard, E., Jaber, R., & Gray, A. (2022). COVID-19 Vaccine Hesitancy among Parents of Children under Five Years in the United States. Vaccines, 10(8), 1313. https://doi.org/10.3390/vaccines10081313






