Designing a Novel Peptide-Based Multi-Epitope Vaccine to Evoke a Robust Immune Response against Pathogenic Multidrug-Resistant Providencia heimbachae
Abstract
:1. Introduction
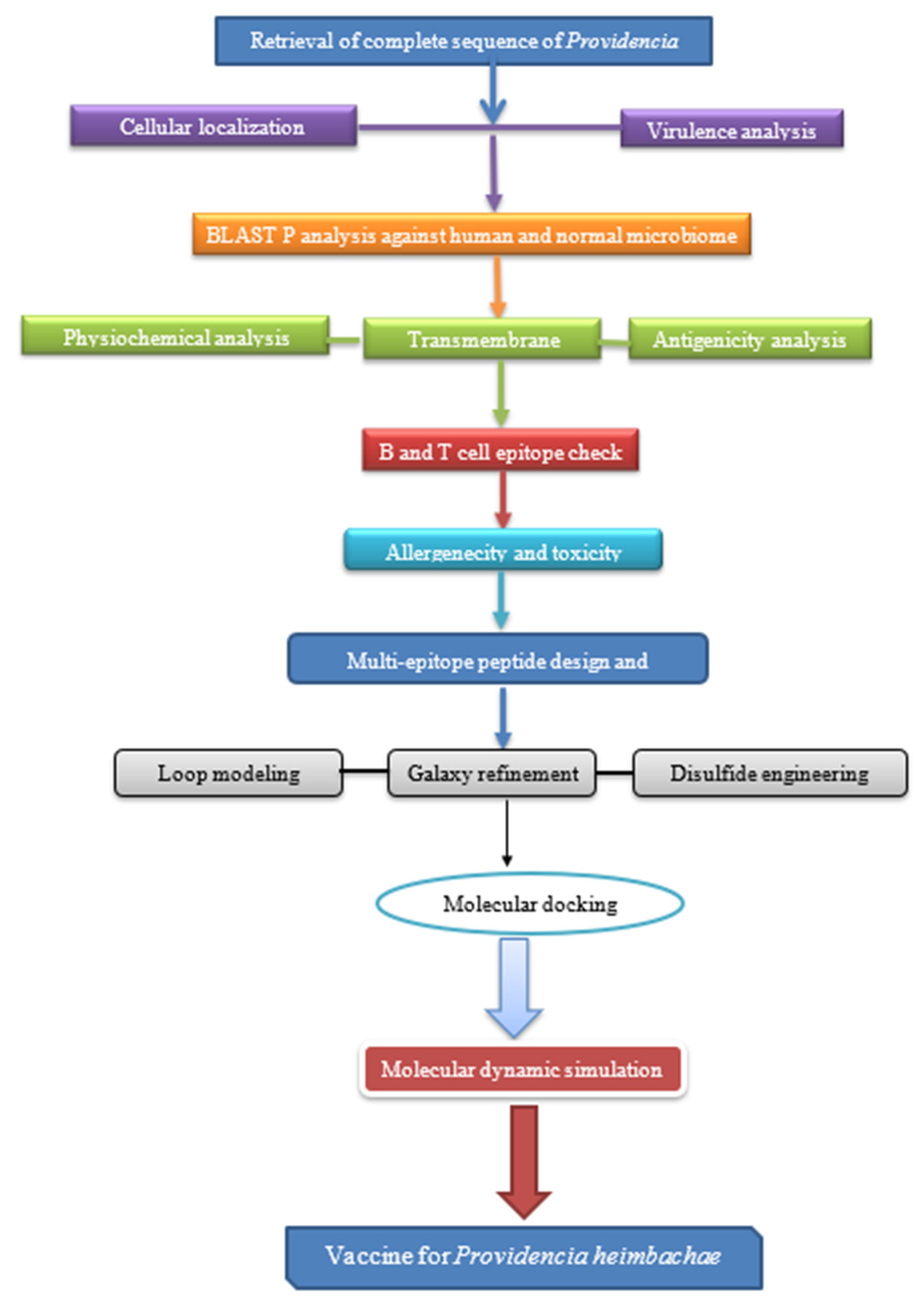
2. Materials and Methods
2.1. Retrieval of Complete Proteome of P. heimbachae
2.2. CD-Hit Analysis
2.3. Subcellular Localization
2.4. Virulent Protein Analysis
2.5. BLAST p Analysis
2.6. Physiochemical Analysis
2.7. Transmembrane Helices
2.8. Antigenicity Prediction
2.9. Epitopes Prediction Phase
2.10. Allergenicity and Toxicity Prediction
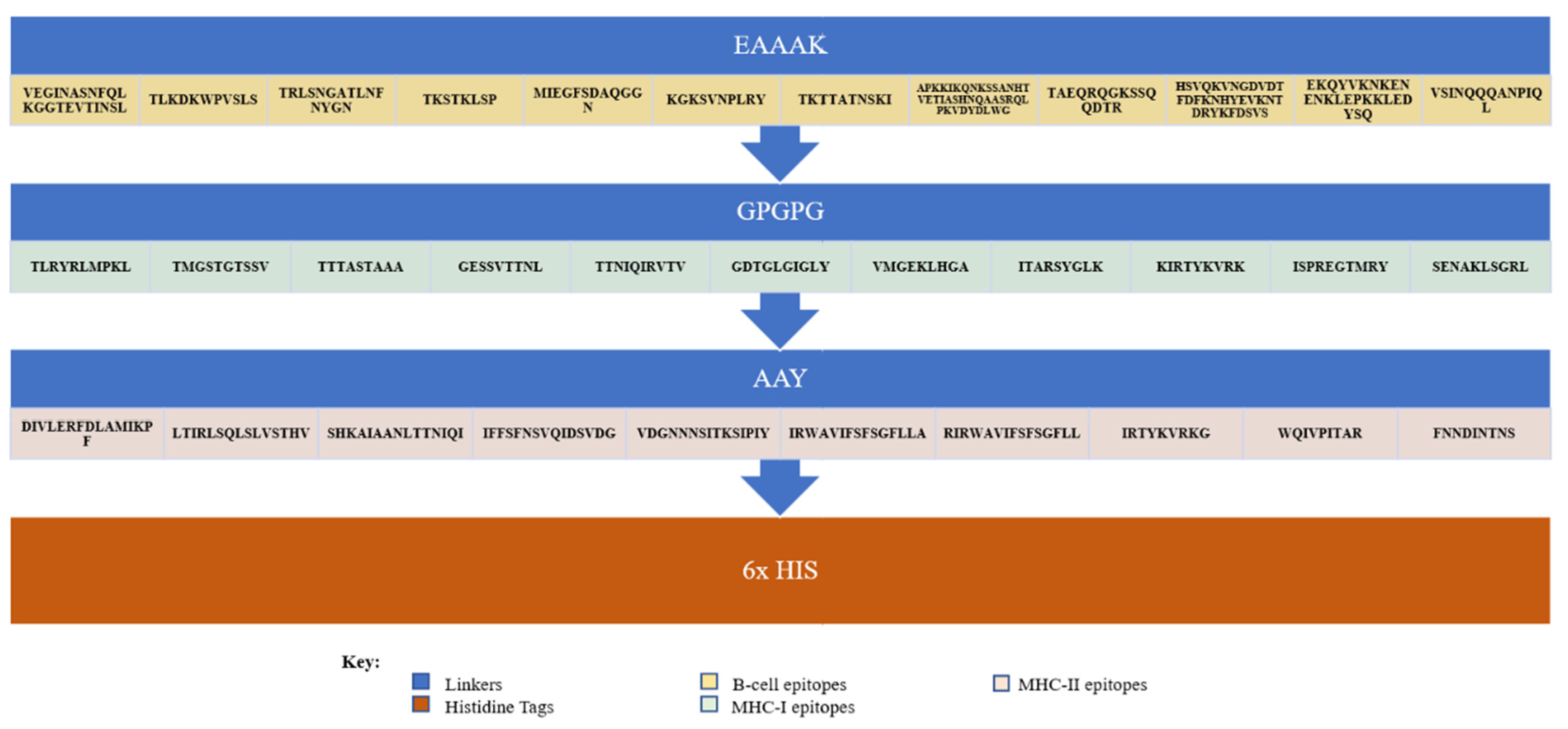
2.11. Multi-Epitope Vaccine Designing and Processing
2.12. Loop Modeling
2.13. Galaxy Refinement
2.14. Disulfide Engineering
2.15. Codon Optimization
2.16. Molecular Docking
2.17. Molecular Dynamic Simulation (MDS)
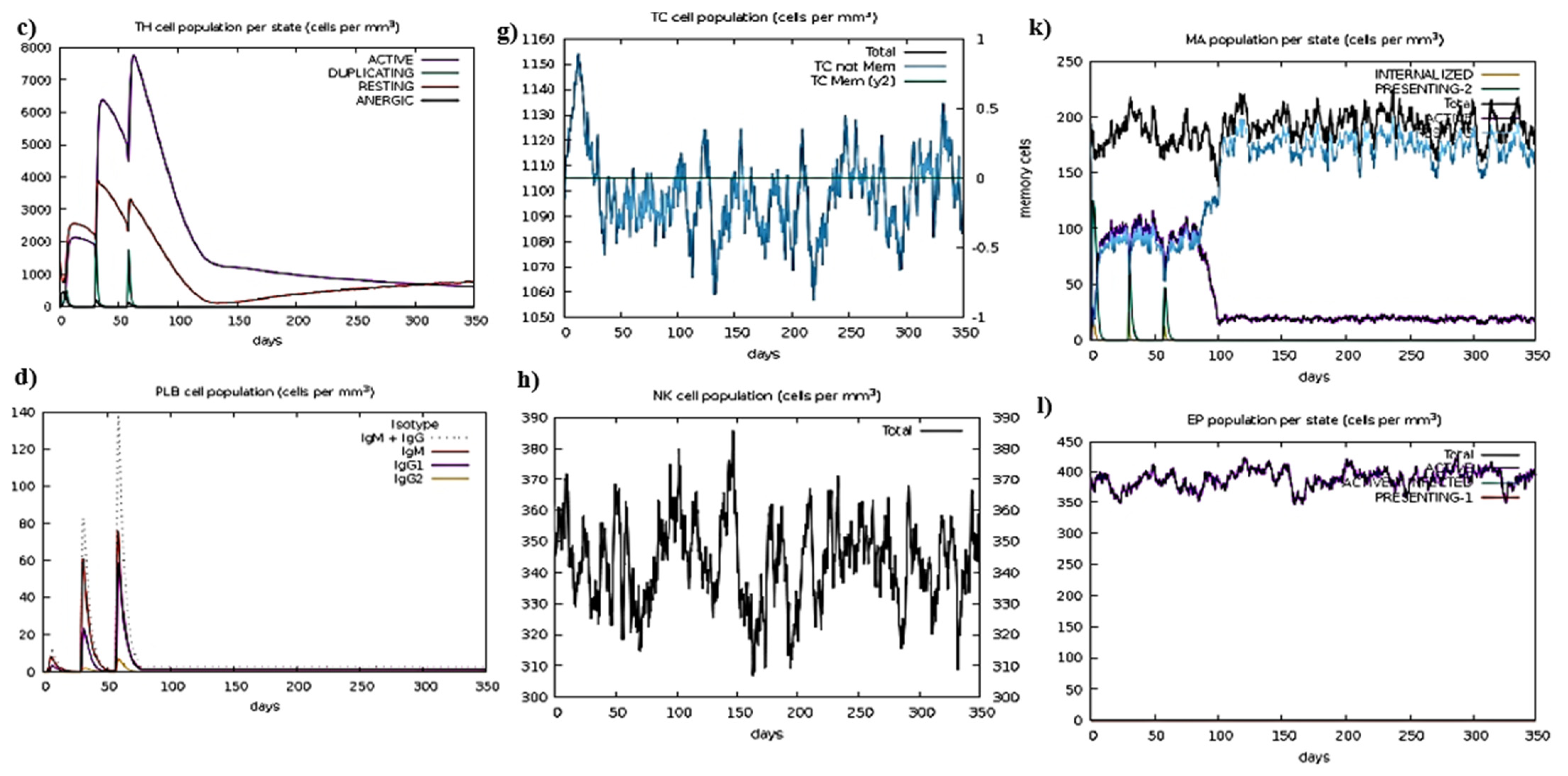
2.18. Immune Simulation
3. Results
3.1. Retrieval of Proteomic Sequence, Subcellular Localization, and Transmembrane Alpha-Helices Identification
3.2. Epitope Prediction and Population Coverage
3.3. Peptide-Based Vaccine Construction
3.4. Structure Prediction and Validation
3.5. Docking of Multiple Epitope Vaccine Construct
3.6. Insilco Cloning and Immune Simulation of Construct
3.7. Molecular Dynamic Simulation
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohr O’Hara, C.; Steigerwalt, A.G.; Green, D.; McDowell, M.; Hill, B.C.; Brenner, D.J.; Miller, J.M. Isolation of Providencia heimbachae from human feces. J. Clin. Microbiol. 1999, 37, 3048–3050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Müller, H.; O’hara, C.; Fanning, G.; Hickman-Brenner, F.; Swenson, J.; Brenner, D.J. Providencia heimbachae, a new species of Enterobacteriaceae isolated from animals. Int. J. Syst. Evol. Microbiol. 1986, 36, 252–256. [Google Scholar] [CrossRef] [Green Version]
- Gul, S.; Ahmad, S.; Ullah, A.; Ismail, S.; Khurram, M.; Qamar, M.T.U.; Hakami, A.R.; Alkhathami, A.G.; Alrumaihi, F.; Allemailem, K.S. Designing a Recombinant Vaccine against Providencia rettgeri Using Immunoinformatics Approach. Vaccines 2022, 10, 189. [Google Scholar] [CrossRef] [PubMed]
- Senior, B. Media and tests to simplify the recognition and identification of members of the Proteeae. J. Med. Microbiol. 1997, 46, 39–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parks, D.H.; Imelfort, M.; Skennerton, C.T.; Hugenholtz, P.; Tyson, G.W. CheckM: Assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 2015, 25, 1043–1055. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, S.; Jeong, S.H.; Lee, H.; Hong, J.S.; Park, M.-J.; Song, W. Emergence of multidrug-resistant Providencia rettgeri isolates co-producing NDM-1 carbapenemase and PER-1 extended-spectrum β-lactamase causing a first outbreak in Korea. Ann. Clin. Microbiol. 2018, 17, 20. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, L.; Song, M.; Luo, J.; Liu, H.; Xue, K.; Huang, C.; Chen, H.; Ge, J. Providencia heimbachae Associated with Post-weaning Diarrhea in Piglets: Identification, Phenotype, and Pathogenesis. Curr. Microbiol. 2021, 79, 1. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.; Wei, Y.; Zhang, S.; Cheng, J.; Cheng, X.; Qian, C.; Wang, Y.; Zhang, Y.; Yin, Z.; Chen, H. Comparative Genomic Analysis Reveals Genetic Mechanisms of the Variety of Pathogenicity, Antibiotic Resistance, and Environmental Adaptation of Providencia Genus. Front. Microbiol. 2020, 11, 572642. [Google Scholar] [CrossRef]
- Russell, A.D.; Path, F.R.C. Chlorhexidine: Antibacterial action and bacterial resistance. Infection 1986, 14, 212–215. [Google Scholar] [CrossRef]
- Fathollahi, M.; Fathollahi, A.; Motamedi, H.; Moradi, J.; Alvandi, A.; Abiri, R. In silico vaccine design and epitope mapping of New Delhi metallo-beta-lactamase (NDM): An immunoinformatics approach. BMC Bioinform. 2021, 22, 458. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, K.; Marchaim, D.; Divine, G.W.; Pogue, J.M.; Kumar, S.; Lephart, P.; Risko, K.; Sobel, J.D.; Kaye, K.S. Growing prevalence of Providencia stuartii associated with the increased usage of colistin at a tertiary health care center. Int. J. Infect. Dis. 2012, 16, e646–e648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anand, U.; Jakhmola, S.; Indari, O.; Jha, H.C.; Chen, Z.-S.; Tripathi, V.; Pérez de la Lastra, J.M. Potential Therapeutic Targets and Vaccine Development for SARS-CoV-2/COVID-19 Pandemic Management: A Review on the Recent Update. Front. Immunol. 2021, 12, 658519. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, S.H.; Asghari, A.; Farhadi, M.; Shamshiri, A.R.; Kabir, A.; Kamrava, S.K.; Jalessi, M.; Mohebbi, A.; Alizadeh, R.; Honarmand, A.A.; et al. Coincidence of COVID-19 epidemic and olfactory dysfunction outbreak in Iran. Med. J. Islam. Repub. Iran 2020, 34, 62. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, C.R.; Scarbrough, D. Implementing Bioinformatic Tools to Predict Vaccine Potential from Prioritized Staphylococcus aureus antigens. 2020. Available online: https://scholarworks.boisestate.edu/icur/2020/Poster_Session/119/ (accessed on 20 June 2022).
- Abbas, G.; Zafar, I.; Ahmad, S.; Azam, S.S. Immunoinformatics design of a novel multi-epitope peptide vaccine to combat multi-drug resistant infections caused by Vibrio vulnificus. Eur. J. Pharm. Sci. 2020, 142, 105160. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Ranaghan, K.E.; Azam, S.S. Combating tigecycline resistant Acinetobacter baumannii: A leap forward towards multi-epitope based vaccine discovery. Eur. J. Pharm. Sci. 2019, 132, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Sahoo, P.; Sahoo, G.; Behera, P. Comparative Insilco physiochemical and phylogenetic analysis of insulin like growth factor 1 receptor (IGF-1R) in domestic animals. Indian J. Anim. Res. 2019, 53, 1033–1035. [Google Scholar] [CrossRef]
- Lüthje, S.; Ramanathan, K. In Silico Analysis of Class III Peroxidases: Hypothetical Structure, Ligand Binding Sites, Posttranslational Modifications, and Interaction with Substrates. In Plant Proteomics; Springer: Berlin/Heidelberg, Germany, 2020; pp. 325–339. [Google Scholar]
- Chen, H.; Gao, Z.; Bai, S.; Liu, X.; Han, S.; Xiao, Y.; Liu, F.; Yu, Y.; Sun, H.; Yang, X. Immunogenicity and safety of sabin-strain based inactivated poliovirus vaccine replacing salk-strain based inactivated poliovirus vaccine: An innovative application of different strain-IPVs replacement. Vaccine 2021, 39, 2467–2474. [Google Scholar] [CrossRef] [PubMed]
- Dhanda, S.K.; Mahajan, S.; Paul, S.; Yan, Z.; Kim, H.; Jespersen, M.C.; Jurtz, V.; Andreatta, M.; Greenbaum, J.A.; Marcatili, P. IEDB-AR: Immune epitope database—analysis resource in 2019. Nucleic Acids Res. 2019, 47, W502–W506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, S.; Azam, S.S. A novel approach of virulome based reverse vaccinology for exploring and validating peptide-based vaccine candidates against the most troublesome nosocomial pathogen: Acinetobacter baumannii. J. Mol. Graph. Model. J. 2018, 83, 1–11. [Google Scholar] [CrossRef]
- Saadi, M.; Karkhah, A.; Nouri, H.R. Development of a multi-epitope peptide vaccine inducing robust T cell responses against brucellosis using immunoinformatics based approaches. Infect. Genet. Evol. 2017, 51, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, M.; Saha, S. Structural insight into the role of novel SARS-CoV-2 E protein: A potential target for vaccine development and other therapeutic strategies. PLoS ONE 2020, 15, e0237300. [Google Scholar] [CrossRef]
- Qamar, M.T.U.; Ismail, S.A.S.; Mirza, M.U.; Abbasi, S.W.; Ashfaq, U.A.; Chen, L.-L. Development of a novel multi-epitope vaccine against crimean-congo hemorrhagic fever virus: An integrated reverse vaccinology, vaccine informatics and biophysics approach. Front. Immunol. 2021, 12, 2313. [Google Scholar]
- Devi, A.; Chaitanya, N.S. In silico designing of multi-epitope vaccine construct against human coronavirus infections. J. Biomol. Struct. Dyn. 2021, 39, 6903–6917. [Google Scholar] [CrossRef] [PubMed]
- Samad, A.; Ahammad, F.; Nain, Z.; Alam, R.; Imon, R.R.; Hasan, M.; Rahman, M.S. Designing a multi-epitope vaccine against SARS-CoV-2: An immunoinformatics approach. J. Biomol. Struct. Dyn. 2022, 40, 14–30. [Google Scholar] [CrossRef]
- Krishnan, S.; Joshi, A.; Akhtar, N.; Kaushik, V. Immunoinformatics designed T cell multi epitope dengue peptide vaccine derived from non-structural proteome. Microb. Pathog. 2021, 150, 104728. [Google Scholar] [CrossRef] [PubMed]
- Setiawan, T.; Rizarullah, R. Predicting Multi-Epitope Peptide Cancer Vaccine from Novel TAA Topo48. J. Sci. Appl. Technol. 2021, 5, 171–178. [Google Scholar] [CrossRef]
- Debnath, S.; Sen, D. Mushrooms are potential foods against cancer: Identified by molecular docking and molecular dynamics simulation. Nat. Prod. Res. 2021, 36, 2604–2609. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, A. In silico Multi Subunit Vaccine Design Referring Spike Glycoprotein of SARS-CoV-2 (COVID-19): The World Pandemic. Indian J. Pharm. Sci. 2021, 83, 21–31. [Google Scholar] [CrossRef]
- Omoniyi, A.A.; Adebisi, S.S.; Musa, S.A.; Nzalak, J.O.; Danborno, B.; Bauchi, Z.M.; Badmus, I.T.; Olatomide, O.D.; Oladimeji, O.J.; Nyengaard, J.R. Immunoinformatics Analysis and In-Silico Design of Multi-Epitopes Vaccine against Lassa Virus. Sci. Rep. 2022, 12, 8736. [Google Scholar] [CrossRef]
- Ehsan, N.; Ahmad, S.; Navid, A.; Azam, S.S. Identification of potential antibiotic targets in the proteome of multi-drug resistant Proteus mirabilis. Meta Gene 2018, 18, 167–173. [Google Scholar] [CrossRef]
- Di, H.; Liang, S.; Li, Q.; Shi, L.; Shima, A.; Meng, H.; Yan, H.; Yamasaki, S. Providencia in retail meats from Guangzhou, China and Osaka, Japan: Prevalence, antimicrobial resistance and characterization of classes 1, 2 and 3 integrons. J. Vet. Med. Sci. 2018, 80, 829–835. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jneid, J.; Benamar, S.; Pagnier, I.; Levy, P.-Y.; Lavigne, J.-P.; la Scola, B. Draft genome sequence of Providencia heimbachae, isolated from a diabetic foot ulcer. Genome Announc. 2016, 4, e00276-16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Da Fiúza, T.S.; de Souza, G.A. Identification of core immunogenic peptides of Shigella sonnei for a Peptide-Based Vaccine. Bio-Manguinhos 2021, 5, 102. [Google Scholar]
- Dar, H.A.; Ismail, S.; Waheed, Y.; Ahmad, S.; Jamil, Z.; Aziz, H.; Hetta, H.F.; Muhammad, K. Designing a multi-epitope vaccine against Mycobacteroides abscessus by pangenome-reverse vaccinology. Sci. Rep. 2021, 11, 11197. [Google Scholar] [CrossRef] [PubMed]
- Bidmos, F.A.; Siris, S.; Gladstone, C.A.; Langford, P.R. Bacterial Vaccine Antigen Discovery in the Reverse Vaccinology 2.0 Era: Progress and Challenges. Front. Immunol. 2018, 9, 2315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ismail, S.; Ahmad, S.; Azam, S.S. Vaccinomics to design a novel single chimeric subunit vaccine for broad-spectrum immunological applications targeting nosocomial Enterobacteriaceae pathogens. Eur. J. Pharm. Sci. 2020, 146, 105258. [Google Scholar] [CrossRef]
- Jafari, E.; Mahmoodi, S. Design, expression, and purification of a multi-epitope vaccine against Helicobacter Pylori based on Melittin as an adjuvant. Microb. Pathog. 2021, 157, 104970. [Google Scholar] [CrossRef]
- Somvanshi, V.S.; Lang, E.; Sträubler, B.; Spröer, C.; Schumann, P.; Ganguly, S.; Saxena, A.K.; Stackebrandt, E. Providencia vermicola sp. nov.; isolated from infective juveniles of the entomopathogenic nematode Steinernema thermophilum. Int. J. Syst. Evol. Microbiol. 2006, 56, 629–633. [Google Scholar] [CrossRef] [Green Version]
- Rizwan, M.; Naz, A.; Ahmad, J.; Naz, K.; Obaid, A.; Parveen, T.; Ahsan, M.; Ali, A. VacSol: A high throughput in silico pipeline to predict potential therapeutic targets in prokaryotic pathogens using subtractive reverse vaccinology. BMC Bioinform. 2017, 18, 106. [Google Scholar] [CrossRef] [Green Version]
- Nielsen, M.; Lundegaard, C.; Lund, O. Prediction of MHC class II binding affinity using SMM-align, a novel stabilization matrix alignment method. BMC Bioinform. 2007, 8, 238. [Google Scholar] [CrossRef] [Green Version]
- Naveed, M.; Tehreem, S.; Arshad, S.; Bukhari, S.A.; Shabbir, M.A.; Essa, R.; Ali, N.; Zaib, S.; Khan, A.; Al-Harrasi, A. Design of a novel multiple epitope-based vaccine: An immunoinformatics approach to combat SARS-CoV-2 strains. J. Infect. Public Health 2021, 14, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Naveed, M.; Ali, U.; Karobari, M.I.; Ahmed, N.; Mohamed, R.N.; Abullais, S.S.; Kader, M.A.; Marya, A.; Messina, P.; Scardina, G.A. A Vaccine Construction against COVID-19-Associated Mucormycosis Contrived with Immunoinformatics-Based Scavenging of Potential Mucoralean Epitopes. Vaccines 2022, 10, 664. [Google Scholar] [CrossRef] [PubMed]
- MacLean, R.C.; Millan, A.S. The evolution of antibiotic resistance. Science 2019, 365, 1082–1083. [Google Scholar] [CrossRef] [PubMed]



| Property | Measurement | Indication |
|---|---|---|
| Total Number of Amino Acid | 578 | Appropriate |
| Molecular Weight | 62067.34 | Appropriate |
| Formula | C2761H4383N791O826S6 | - |
| Theoretical pI | 10.07 | Basic |
| Total number of positively charged residues (Arg + Lys) | 81 | - |
| Total number of negatively charged residues (Asp + Glu) | 35 | - |
| Total Number of Atoms | 8767 | - |
| Instability index (II) | 18.21 | Stable |
| Aliphatic Index | 68.58 | Thermostable |
| Grand Average of Hydropathicity (GRAVY) | −0.558 | Hydrophilic |
| Antigenicity VaxiJen | 1.05 | Antigenic |
| Allergenicity | Non-Allergen | Non-allergenic |
| Toxicity | Non-toxic | Non-toxic |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naveed, M.; Sheraz, M.; Amin, A.; Waseem, M.; Aziz, T.; Khan, A.A.; Ghani, M.; Shahzad, M.; Alruways, M.W.; Dablool, A.S.; et al. Designing a Novel Peptide-Based Multi-Epitope Vaccine to Evoke a Robust Immune Response against Pathogenic Multidrug-Resistant Providencia heimbachae. Vaccines 2022, 10, 1300. https://doi.org/10.3390/vaccines10081300
Naveed M, Sheraz M, Amin A, Waseem M, Aziz T, Khan AA, Ghani M, Shahzad M, Alruways MW, Dablool AS, et al. Designing a Novel Peptide-Based Multi-Epitope Vaccine to Evoke a Robust Immune Response against Pathogenic Multidrug-Resistant Providencia heimbachae. Vaccines. 2022; 10(8):1300. https://doi.org/10.3390/vaccines10081300
Chicago/Turabian StyleNaveed, Muhammad, Mohsin Sheraz, Aatif Amin, Muhammad Waseem, Tariq Aziz, Ayaz Ali Khan, Mustajab Ghani, Muhammad Shahzad, Mashael W. Alruways, Anas S. Dablool, and et al. 2022. "Designing a Novel Peptide-Based Multi-Epitope Vaccine to Evoke a Robust Immune Response against Pathogenic Multidrug-Resistant Providencia heimbachae" Vaccines 10, no. 8: 1300. https://doi.org/10.3390/vaccines10081300
APA StyleNaveed, M., Sheraz, M., Amin, A., Waseem, M., Aziz, T., Khan, A. A., Ghani, M., Shahzad, M., Alruways, M. W., Dablool, A. S., Elazzazy, A. M., Almalki, A. A., Alamri, A. S., & Alhomrani, M. (2022). Designing a Novel Peptide-Based Multi-Epitope Vaccine to Evoke a Robust Immune Response against Pathogenic Multidrug-Resistant Providencia heimbachae. Vaccines, 10(8), 1300. https://doi.org/10.3390/vaccines10081300








