The Protective Effects of Influenza Vaccination in Elderly Patients with Breast Cancer in Taiwan: A Real-World Evidence-Based Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Background Information
2.2. Study Design
2.3. Two Population-Based Study Cohorts
2.4. Independent Variable
2.5. Outcome Measures
2.6. Covariates
2.7. Statistical Analysis
3. Results
3.1. Descriptive Results
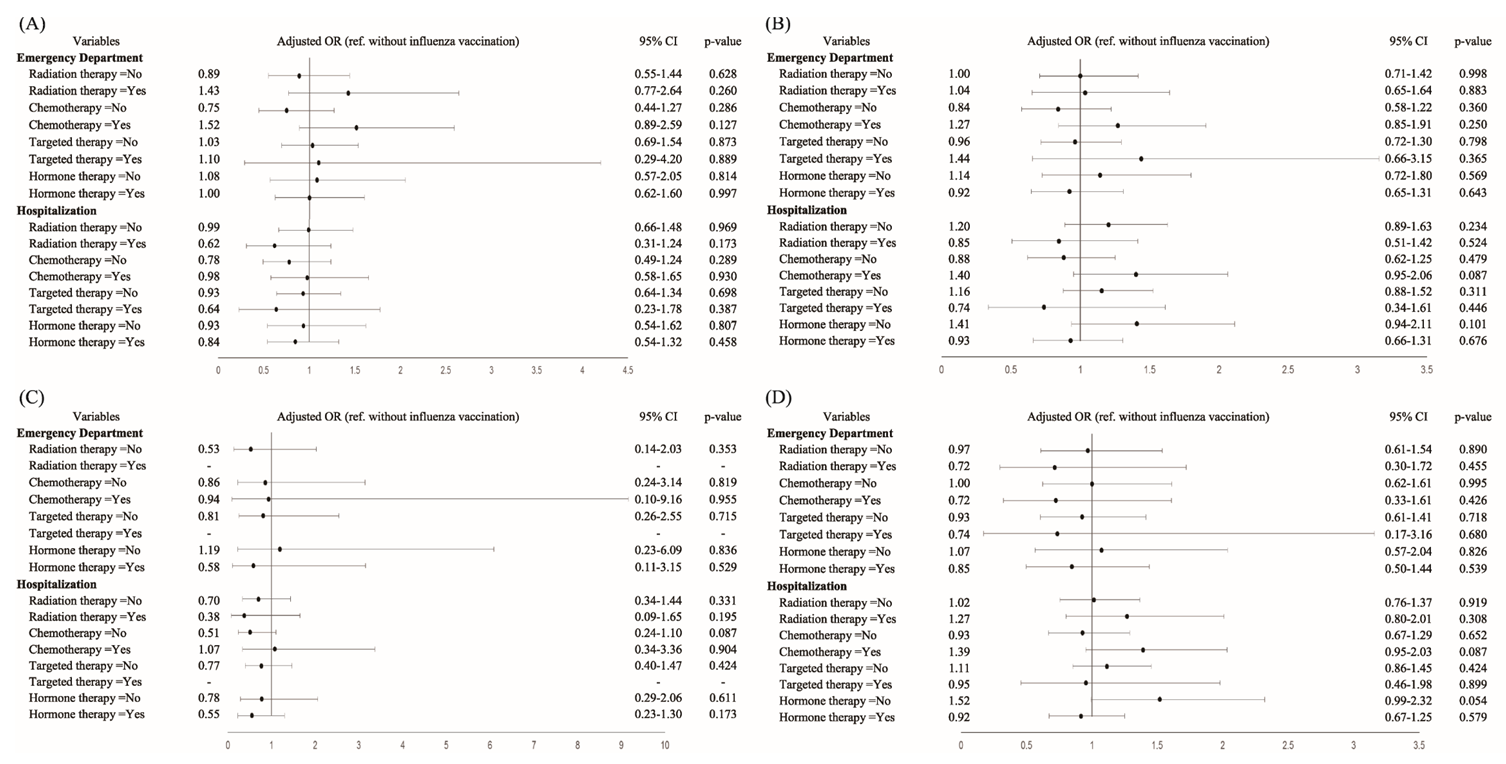
3.2. The Results of Elderly Patients with Newly Diagnosed Breast Cancer
3.3. Results of General Elderly Women Cohort
| Variables | Breast Cancer Cohort | General Women Cohort | ||||||
|---|---|---|---|---|---|---|---|---|
| Without Influenza Vaccine (N = 3982) | With Influenza Vaccine (N = 1991) | With Influenza Vaccine vs. without Influenza Vaccine (Ref.) | Without Influenza Vaccine (N = 585,327) | With Influenza Vaccine (N = 585,327) | With Influenza Vaccine vs. without Influenza Vaccine (Ref.) | |||
| Incident (‰) | Incident (‰) | aOR 1 | 95% CI | Incident (‰) | Incident (‰) | aOR 2 | 95% CI | |
| All-cause mortality | 17.58 | 21.09 | 1.39 | 0.90–2.16 | 28.65 | 16.80 | 0.58 *** | 0.56–0.59 |
| Emergency admission | ||||||||
| Influenza and pneumonia | 16.57 | 16.07 | 1.04 | 0.65–1.68 | 22.89 | 19.93 | 0.87 *** | 0.85–0.90 |
| Respiratory diseases | 36.92 | 30.14 | 1.02 | 0.73–1.43 | 38.88 | 34.23 | 0.88 *** | 0.86–0.90 |
| Respiratory failure | 1.26 | 2.01 | 1.61 | 0.35–7.36 | 3.81 | 2.57 | 0.68 *** | 0.64–0.73 |
| Heart disease | 16.32 | 14.57 | 1.11 | 0.68–1.82 | 23.93 | 19.05 | 0.80 *** | 0.78–0.82 |
| Hospitalization | ||||||||
| Influenza and pneumonia | 20.59 | 22.60 | 1.25 | 0.83–1.89 | 31.56 | 28.34 | 0.90 *** | 0.88–0.92 |
| Respiratory diseases | 34.66 | 41.19 | 1.38 * | 1.01–1.89 | 46.13 | 40.54 | 0.88 *** | 0.86–0.90 |
| Respiratory failure | 7.53 | 6.53 | 0.92 | 0.44–1.91 | 12.81 | 9.55 | 0.75 *** | 0.73–0.78 |
| Heart disease | 37.67 | 44.70 | 1.27 | 0.94–1.72 | 39.48 | 34.31 | 0.87 *** | 0.85–0.89 |
| Variables | Breast Cancer Cohort | General Women Cohort | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | Mean | SD | Exp (β) 1 | 95% CI | N | Mean | SD | Exp (β) 2 | 95% CI | |
| Influenza and pneumonia | ||||||||||
| Without influenza vaccine (ref.) | 82 | 86,777 | 104,359 | 18,470 | 119,784 | 184,895 | ||||
| With influenza vaccine | 45 | 79,516 | 120,849 | 0.99 | 0.65–1.52 | 16,589 | 105,472 | 166,555 | 0.87 *** | 0.85–0.89 |
| Respiratory diseases | ||||||||||
| Without influenza vaccine (ref.) | 138 | 136,069 | 207,251 | 26,999 | 141,979 | 238,490 | ||||
| With influenza vaccine | 82 | 127,108 | 317,341 | 0.89 | 0.62–1.27 | 23,730 | 123,530 | 218,378 | 0.87 *** | 0.85–0.88 |
| Respiratory failure | ||||||||||
| Without influenza vaccine (ref.) | 30 | 247,260 | 212,202 | 7500 | 258,238 | 341,586 | ||||
| With influenza vaccine | 13 | 223,307 | 197,064 | 0.81 | 0.48–1.34 | 5591 | 240,661 | 335,844 | 0.93 *** | 0.90–0.97 |
| Heart disease | ||||||||||
| Without influenza vaccine (ref.) | 150 | 121,610 | 181,776 | 23,111 | 117,837 | 180,032 | ||||
| With influenza vaccine | 89 | 142,199 | 230,031 | 1.10 | 0.81–1.50 | 20,083 | 107,633 | 168,938 | 0.92 *** | 0.90–0.93 |
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hibberd, P.L.; Rubin, R.H. Approach to immunization in the immunosuppressed host. Infect. Dis. Clin. N. Am. 1990, 4, 123–142. [Google Scholar] [CrossRef]
- Grohskopf, L.A.; Alyanak, E.; Ferdinands, J.M.; Broder, K.R.; Blanton, L.H.; Talbot, H.K.; Fry, A.M. Prevention and control of seasonal influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices, United States, 2021–2022 Influenza Season. MMWR Recomm. Rep. 2021, 70, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Ortbals, D.W.; Liebhaber, H.; Presant, C.A.; Van Amburg, A.L., 3rd; Lee, J.Y. Influenza immunization of adult patients with malignant diseases. Ann. Intern. Med. 1977, 87, 552–557. [Google Scholar] [CrossRef]
- Rubin, L.G.; Levin, M.J.; Ljungman, P.; Davies, E.G.; Avery, R.; Tomblyn, M.; Bousvaros, A.; Dhanireddy, S.; Sung, L.; Keyserling, H.; et al. 2013 IDSA clinical practice guideline for vaccination of the immunocompromised host. Clin. Infect. Dis. 2014, 58, e44–e100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taplitz, R.A.; Kennedy, E.B.; Bow, E.J.; Crews, J.; Gleason, C.; Hawley, D.K.; Langston, A.A.; Nastoupil, L.J.; Rajotte, M.; Rolston, K.V.; et al. Antimicrobial prophylaxis for adult patients with cancer-related immunosuppression: ASCO and IDSA clinical practice guideline update. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2018, 36, 3043–3054. [Google Scholar] [CrossRef]
- Meerveld-Eggink, A.; de Weerdt, O.; van der Velden, A.M.T.; Los, M.; van der Velden, A.W.G.; Stouthard, J.M.L.; Nijziel, M.R.; Westerman, M.; Beeker, A.; van Beek, R.; et al. Response to influenza virus vaccination during chemotherapy in patients with breast cancer. Ann. Oncol. 2011, 22, 2031–2035. [Google Scholar] [CrossRef]
- Wumkes, M.L.; van der Velden, A.M.; Los, M.; Leys, M.B.; Beeker, A.; Nijziel, M.R.; van der Velden, A.W.; Westerman, M.; Meerveld-Eggink, A.; Rimmelzwaan, G.F.; et al. Serum antibody response to influenza virus vaccination during chemotherapy treatment in adult patients with solid tumours. Vaccine 2013, 31, 6177–6184. [Google Scholar] [CrossRef]
- Health Promotion Administration. Taiwan Cancer Registry Annual Report, 2019; Ministry of Health and Welfare: Taiwan, 2021. [Google Scholar]
- Giuliano, A.E.; Hunt, K.K.; Ballman, K.V.; Beitsch, P.D.; Whitworth, P.W.; Blumencranz, P.W.; Leitch, A.M.; Saha, S.; McCall, L.M.; Morrow, M. Axillary dissection vs. no axillary dissection in women with invasive breast cancer and sentinel node metastasis: A randomized clinical trial. JAMA 2011, 305, 569–575. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Lu, C.Y.; Chen, C.H.; Chen, H.M.; Wu, S.Y. Effect of pathologic stages on postmastectomy radiation therapy in breast cancer receiving neoadjuvant chemotherapy and total mastectomy: A cancer database analysis. Breast 2020, 54, 70–78. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, C.Y.; Qin, L.; Chen, H.M.; Wu, S.Y. Breast-conserving surgery with or without irradiation in women with invasive ductal carcinoma of the breast receiving preoperative systemic therapy: A cohort study. Breast 2020, 54, 139–147. [Google Scholar] [CrossRef]
- Zhang, J.; Lu, C.Y.; Chen, H.M.; Wu, S.Y. Neoadjuvant chemotherapy or endocrine therapy for invasive ductal carcinoma of the breast With high hormone receptor positivity and human epidermal growth factor receptor 2 negativity. JAMA Netw. Open 2021, 4, e211785. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network (NCCN). Clinical Practice Guidelines in Oncology: Breast Cancer. Available online: https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf (accessed on 11 November 2021).
- Kuo, C.N.; Liao, Y.M.; Kuo, L.N.; Tsai, H.J.; Chang, W.C.; Yen, Y. Cancers in Taiwan: Practical insight from epidemiology, treatments, biomarkers, and cost. J. Formos. Med. Assoc. 2020, 119, 1731–1741. [Google Scholar] [CrossRef] [PubMed]
- Keam, B.; Kim, M.K.; Choi, Y.; Choi, S.J.; Choe, P.G.; Lee, K.H.; Kim, T.M.; Kim, T.Y.; Oh, D.Y.; Kim, D.W.; et al. Optimal timing of influenza vaccination during 3-week cytotoxic chemotherapy cycles. Cancer 2017, 123, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Gribabis, D.A.; Panayiotidis, P.; Boussiotis, V.A.; Hannoun, C.; Pangalis, G.A. Influenza virus vaccine in B-cell chronic lymphocytic leukaemia patients. Acta Haematol. 1994, 91, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Anderson, H.; Petrie, K.; Berrisford, C.; Charlett, A.; Thatcher, N.; Zambon, M. Seroconversion after influenza vaccination in patients with lung cancer. Br. J. Cancer 1999, 80, 219–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brydak, L.B.; Calbecka, M. Immunogenicity of influenza vaccine in patients with hemato-oncological disorders. Leuk. Lymphoma 1999, 32, 369–374. [Google Scholar] [CrossRef]
- Lo, W.; Whimbey, E.; Elting, L.; Couch, R.; Cabanillas, F.; Bodey, G. Antibody response to a two-dose influenza vaccine regimen in adult lymphoma patients on chemotherapy. Eur. J. Clin. Microbiol. Infect. Dis. 1993, 12, 778–782. [Google Scholar] [CrossRef]
- Ljungman, P.; Nahi, H.; Linde, A. Vaccination of patients with haematological malignancies with one or two doses of influenza vaccine: A randomised study. Br. J. Haematol. 2005, 130, 96–98. [Google Scholar] [CrossRef]
- Beck, C.R.; McKenzie, B.C.; Hashim, A.B.; Harris, R.C.; Nguyen-Van-Tam, J.S. Influenza vaccination for immunocompromised patients: Systematic review and meta-analysis by etiology. J. Infect. Dis. 2012, 206, 1250–1259. [Google Scholar] [CrossRef] [Green Version]
- Yri, O.E.; Torfoss, D.; Hungnes, O.; Tierens, A.; Waalen, K.; Nordoy, T.; Dudman, S.; Kilander, A.; Wader, K.F.; Ostenstad, B.; et al. Rituximab blocks protective serologic response to influenza A (H1N1) 2009 vaccination in lymphoma patients during or within 6 months after treatment. Blood 2011, 118, 6769–6771. [Google Scholar] [CrossRef]
- Austin, P.C. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivar. Behav. Res. 2011, 46, 399–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, J.C.; Wang, T.J.; Sung, L.C.; Kao, P.F.; Yang, T.Y.; Hao, W.R.; Chen, C.C.; Hsu, Y.P.; Wu, S.Y. Influenza vaccination reduces hemorrhagic stroke risk in patients with atrial fibrillation: A population-based cohort study. Int. J. Cardiol. 2017, 232, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.C.; Tung, H.J.; Huang, Y.T.; Lu, C.T.; Ernawaty, E.; Wu, S.Y. Effect of influenza vaccination on mortality and risk of hospitalization in elderly individuals with and without disabilities: A nationwide, population-based cohort study. Vaccines 2020, 8, 112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, Y.C.; Yu-Tung, H.; Chen, L.S.; Tung, H.J.; Huang, K.H.; Ernawaty, E.; Wu, S.Y. Protective effect of seasonal influenza vaccination in elderly individuals with disability in Taiwan: A propensity score-matched, nationwide, population-based cohort study. Vaccines 2020, 8, 140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyer, D.; Shearer, M.P.; Chih, Y.C.; Hsu, Y.C.; Lin, Y.C.; Nuzzo, J.B. Taiwan’s annual seasonal influenza mass vaccination program-lessons for pandemic planning. Am. J. Public Health 2018, 108, S188–S193. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.Y.; Su, C.C.; Shao, S.C.; Sung, S.F.; Lin, S.J.; Kao Yang, Y.H.; Lai, E.C. Taiwan’s National Health Insurance Research Database: Past and future. Clin. Epidemiol. 2019, 11, 349–358. [Google Scholar] [CrossRef] [Green Version]
- Lin, K.C.; Chen, T.M.; Yuan, K.S.; Wu, A.T.H.; Wu, S.Y. Assessment of predictive scoring system for 90-Day mortality among patients with locally advanced head and neck squamous cell carcinoma who have completed concurrent chemoradiotherapy. JAMA Netw. Open 2020, 3, e1920671. [Google Scholar] [CrossRef] [Green Version]
- Wu, S.Y.; Chang, C.L.; Chen, C.I.; Huang, C.C. Comparison of acute and chronic surgical complications following robot-assisted, laparoscopic, and traditional open radical prostatectomy among men in Taiwan. JAMA Netw. Open 2021, 4, e2120156. [Google Scholar] [CrossRef]
- Chang, Y.C.; Chou, Y.J.; Liu, J.Y.; Yeh, T.F.; Huang, N. Additive benefits of pneumococcal and influenza vaccines among elderly persons aged 75 years or older in Taiwan--a representative population-based comparative study. J. Infect. 2012, 65, 231–238. [Google Scholar] [CrossRef]
- Fishman, J.A. Opportunistic infections—Coming to the limits of immunosuppression? Cold Spring Harb. Perspect. Med. 2013, 3, a015669. [Google Scholar] [CrossRef]
- McCormick, R.D. Infections in patients with solid tumors. Nurs. Clin. N. Am. 1985, 20, 199–205. [Google Scholar]
- Morey, J.N.; Boggero, I.A.; Scott, A.B.; Segerstrom, S.C. Current directions in stress and human immune function. Curr. Opin. Psychol. 2015, 5, 13–17. [Google Scholar] [CrossRef] [Green Version]
- Cohen, S.; Janicki-Deverts, D.; Doyle, W.J.; Miller, G.E.; Frank, E.; Rabin, B.S.; Turner, R.B. Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proc. Natl. Acad. Sci. USA 2012, 109, 5995–5999. [Google Scholar] [CrossRef] [Green Version]
- Nie, X.; Kitaoka, S.; Tanaka, K.; Segi-Nishida, E.; Imoto, Y.; Ogawa, A.; Nakano, F.; Tomohiro, A.; Nakayama, K.; Taniguchi, M.; et al. The innate immune receptors TLR2/4 mediate repeated social defeat stress-induced social avoidance through prefrontal microglial activation. Neuron 2018, 99, 464–479 e467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schuemie, M.J.; Cepeda, M.S.; Suchard, M.A.; Yang, J.; Tian, Y.; Schuler, A.; Ryan, P.B.; Madigan, D.; Hripcsak, G. How confident are we about observational findings in healthcare: A benchmark study. Harv. Data Sci. Rev. 2020, 2, 10-1162. [Google Scholar] [CrossRef] [Green Version]
- Kao, P.F.; Liu, J.C.; Hsu, Y.P.; Sung, L.C.; Yang, T.Y.; Hao, W.R.; Lin, Y.C.; Wu, S.Y. Influenza vaccination might reduce the risk of ischemic stroke in patients with atrial fibrillation: A population-based cohort study. Oncotarget 2017, 8, 112697–112711. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.C.; Hsu, Y.P.; Kao, P.F.; Hao, W.R.; Liu, S.H.; Lin, C.F.; Sung, L.C.; Wu, S.Y. Influenza vaccination reduces dementia Risk in chronic kidney disease patients: A population-based cohort study. Medicine 2016, 95, e2868. [Google Scholar] [CrossRef]
- Srivastav, A.; O’Halloran, A.; Lu, P.J.; Williams, W.W. Influenza vaccination coverage among English-Speaking Asian Americans. Am. J. Prev. Med. 2018, 55, e123–e137. [Google Scholar] [CrossRef]
- Cheng, C.L.; Lee, C.H.; Chen, P.S.; Li, Y.H.; Lin, S.J.; Yang, Y.H. Validation of acute myocardial infarction cases in the National Health Insurance Research Database in Taiwan. J. Epidemiol. 2014, 24, 500–507. [Google Scholar] [CrossRef] [Green Version]
- Hsing, A.W.; Ioannidis, J.P. Nationwide population science: Lessons from the Taiwan National Health Insurance Research Database. JAMA Intern. Med. 2015, 175, 1527–1529. [Google Scholar] [CrossRef]
| Variables | Breast Cancer Cohort | General Women Cohort | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1:2 Matching | 1:1 Matching | |||||||||
| Without | With | p-Value | Without | With | p-Value | |||||
| Influenza Vaccine | Influenza Vaccine | Influenza Vaccine | Influenza Vaccine | |||||||
| N | % | N | % | N | % | N | % | |||
| Total | 3982 | 66.67 | 1991 | 33.33 | 585,327 | 50 | 585,327 | 50 | ||
| Age (year) 1 (mean ± SD) | 72.29 ± 5.96 | 72.42 ± 5.65 | 0.996 | 74.89 ± 7.74 | 74.61 ± 7.25 | 0.654 | ||||
| 65–69 | 1571 | 39.45 | 783 | 39.33 | 185,230 | 31.65 | 185,381 | 31.67 | ||
| 70–74 | 1119 | 28.1 | 561 | 28.18 | 127,390 | 21.76 | 126,979 | 21.69 | ||
| ≥75 | 1292 | 32.45 | 647 | 32.5 | 272,707 | 46.59 | 272,967 | 46.63 | ||
| Salary (NTD) 1 | 0.738 | 0.37 | ||||||||
| ≤20,008 | 1578 | 39.63 | 777 | 39.03 | 183,417 | 31.34 | 183,635 | 31.37 | ||
| 20,009–22,800 | 1302 | 32.7 | 641 | 32.19 | 239,694 | 40.95 | 240,282 | 41.05 | ||
| 22,801–38,200 | 397 | 9.97 | 216 | 10.85 | 66,595 | 11.38 | 66,413 | 11.35 | ||
| ≥38,201 | 705 | 17.7 | 357 | 17.93 | 95,621 | 16.34 | 94,997 | 16.23 | ||
| Urbanization 1 | 0.891 | 0.715 | ||||||||
| Level 1 | 1190 | 29.88 | 588 | 29.53 | 138,491 | 23.66 | 137,900 | 23.56 | ||
| Level 2 | 1389 | 34.88 | 678 | 34.05 | 170,650 | 29.15 | 170,668 | 29.16 | ||
| Level 3 | 610 | 15.32 | 319 | 16.02 | 96,335 | 16.46 | 96,361 | 16.46 | ||
| Level 4 | 513 | 12.88 | 249 | 12.51 | 97,560 | 16.67 | 97,769 | 16.7 | ||
| Level 5 | 58 | 1.46 | 34 | 1.71 | 18,750 | 3.2 | 18,684 | 3.19 | ||
| Level 6 | 123 | 3.09 | 67 | 3.37 | 32,906 | 5.62 | 33,278 | 5.69 | ||
| Level 7 | 99 | 2.49 | 56 | 2.81 | 30,635 | 5.23 | 30,667 | 5.24 | ||
| CCI score 1,2 | 0.85 | 0.567 | ||||||||
| 0 | 1065 | 26.75 | 537 | 26.97 | 155,433 | 26.55 | 155,844 | 26.63 | ||
| 1 | 1007 | 25.29 | 517 | 25.97 | 157,954 | 26.99 | 158,133 | 27.02 | ||
| 2 | 481 | 12.08 | 245 | 12.31 | 106,383 | 18.17 | 105,843 | 18.08 | ||
| 3 | 1429 | 35.89 | 692 | 34.76 | 165,557 | 28.28 | 165,507 | 28.28 | ||
| Cancer stage 1 | 0.48 | |||||||||
| Stage I | 1534 | 38.52 | 766 | 38.47 | ||||||
| Stage II | 1869 | 46.94 | 913 | 45.86 | ||||||
| Stage III | 579 | 14.54 | 312 | 15.67 | ||||||
| Radiotherapy | 0.279 | |||||||||
| No | 2614 | 65.65 | 1335 | 67.05 | ||||||
| Yes | 1368 | 34.35 | 656 | 32.95 | ||||||
| Chemotherapy | <0.001 | |||||||||
| No | 2305 | 57.89 | 1354 | 68.01 | ||||||
| Yes | 1677 | 42.11 | 637 | 31.99 | ||||||
| Target therapy. | 0.003 | |||||||||
| No | 3553 | 89.23 | 1825 | 91.66 | ||||||
| Yes | 429 | 10.77 | 166 | 8.34 | ||||||
| Hormone treatment | <0.001 | |||||||||
| No | 1161 | 29.16 | 479 | 24.06 | ||||||
| Yes | 2821 | 70.84 | 1512 | 75.94 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, S.-Y.; Tung, H.-J.; Huang, K.-H.; Lee, C.B.; Tsai, T.-H.; Chang, Y.-C. The Protective Effects of Influenza Vaccination in Elderly Patients with Breast Cancer in Taiwan: A Real-World Evidence-Based Study. Vaccines 2022, 10, 1144. https://doi.org/10.3390/vaccines10071144
Wu S-Y, Tung H-J, Huang K-H, Lee CB, Tsai T-H, Chang Y-C. The Protective Effects of Influenza Vaccination in Elderly Patients with Breast Cancer in Taiwan: A Real-World Evidence-Based Study. Vaccines. 2022; 10(7):1144. https://doi.org/10.3390/vaccines10071144
Chicago/Turabian StyleWu, Szu-Yuan, Ho-Jui Tung, Kuang-Hua Huang, Chiachi Bonnie Lee, Tung-Han Tsai, and Yu-Chia Chang. 2022. "The Protective Effects of Influenza Vaccination in Elderly Patients with Breast Cancer in Taiwan: A Real-World Evidence-Based Study" Vaccines 10, no. 7: 1144. https://doi.org/10.3390/vaccines10071144
APA StyleWu, S.-Y., Tung, H.-J., Huang, K.-H., Lee, C. B., Tsai, T.-H., & Chang, Y.-C. (2022). The Protective Effects of Influenza Vaccination in Elderly Patients with Breast Cancer in Taiwan: A Real-World Evidence-Based Study. Vaccines, 10(7), 1144. https://doi.org/10.3390/vaccines10071144







