Pseudomonas aeruginosa: Recent Advances in Vaccine Development
Abstract
1. An Introduction to Pseudomonas aeruginosa
2. Virulence Factors
2.1. Biofilm Formation
2.2. Antimicrobial Resistance
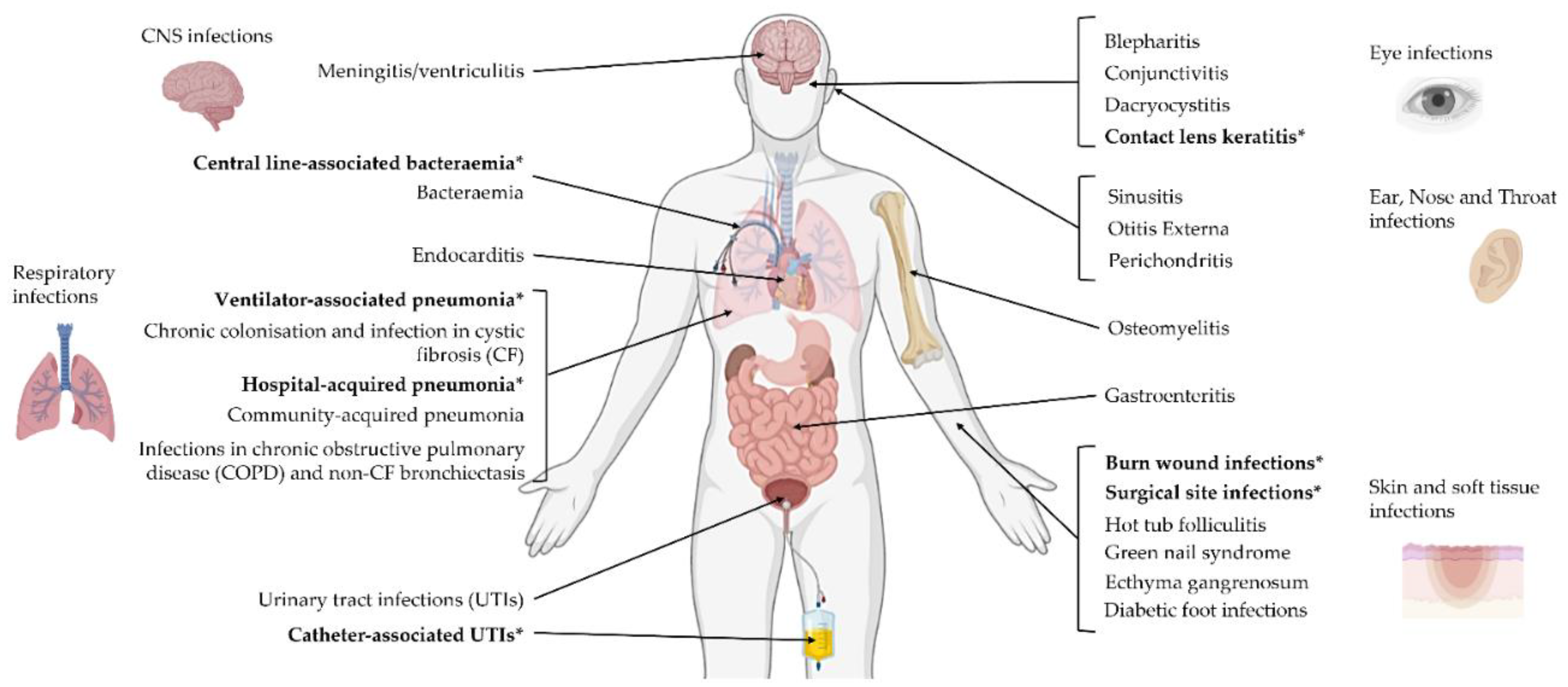
3. P. aeruginosa Infections
4. A History of Clinical Vaccine Development against P. aeruginosa
4.1. Lipopolysaccharide (LPS)
4.2. Alginate
4.3. Flagellar Antigens
4.4. Whole-Cell Killed
4.5. Outer Membrane Proteins
5. New Developments in P. aeruginosa Vaccinology
5.1. Immune Response against P. aeruginosa and Its Importance in Vaccine Design
Advantages of Mucosal Immunity
- Sites of mucosal administration are typically easily accessible and highly vascularised, allowing for rapid antigen uptake;
- Stimulation of all arms of the immune system—antigen-specific IgA at the mucosa, systemic IgG, and cell-mediated responses;
- Existence of the “common mucosal immune system” confers an immune response at mucosal sites distant from the site of vaccine delivery;
- No requirement for needles during administration, thus potentially increasing uptake and eliminating the risk of transmitting blood-borne diseases such as hepatitis B or HIV.
5.2. Lessons from the Past: Enhancing and Optimising Unsuccessful Clinical Vaccines
5.3. Novel Vaccine Formulations
5.3.1. Antigen Discovery
5.3.2. Adjuvant Selection
5.3.3. Vaccine Delivery Systems
5.3.4. Nanoparticles
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Mulani, M.S.; Kamble, E.; Kumkar, S.N.; Tawre, M.S.; Pardesi, K.R. Emerging Strategies to Combat ESKAPE Pathogens in the Era of Antimicrobial Resistance: A Review. Front. Microbiol. 2019, 10, 539. [Google Scholar] [CrossRef]
- Founou, R.C.; Founou, L.L.; Essack, S.Y. Clinical and economic impact of antibiotic resistance in developing countries: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0189621. [Google Scholar] [CrossRef]
- Lansbury, L.; Lim, B.; Baskaran, V.; Lim, W.S. Co-infections in people with COVID-19: A systematic review and meta-analysis. J. Infect. 2020, 81, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Moradali, M.F.; Ghods, S.; Rehm, B.H.A. Pseudomonas aeruginosa Lifestyle: A Paradigm for Adaptation, Survival, and Persistence. Front. Cell. Infect. Microbiol. 2017, 7, 39. [Google Scholar] [CrossRef] [PubMed]
- Jurado-Martín, I.; Sainz-Mejías, M.; McClean, S. Pseudomonas aeruginosa: An Audacious Pathogen with an Adaptable Arsenal of Virulence Factors. Int. J. Mol. Sci. 2021, 22, 3128. [Google Scholar] [CrossRef]
- Barken, K.B.; Pamp, S.J.; Yang, L.; Gjermansen, M.; Bertrand, J.J.; Klausen, M.; Givskov, M.; Whitchurch, C.B.; Engel, J.N.; Tolker-Nielsen, T. Roles of type IV pili, flagellum-mediated motility and extracellular DNA in the formation of mature multicellular structures in Pseudomonas aeruginosa biofilms. Environ. Microbiol. 2008, 10, 2331–2343. [Google Scholar] [CrossRef] [PubMed]
- Deveaux, W.; Selvarajoo, K. Searching for simple rules in Pseudomonas aeruginosa biofilm formation. BMC Res. Notes 2019, 12, 763. [Google Scholar] [CrossRef]
- Thi, M.T.; Wibowo, D.; Rehm, B.H.A. Pseudomonas aeruginosa Biofilms. Int. J. Mol. Sci. 2020, 21, 8671. [Google Scholar] [CrossRef] [PubMed]
- Moser, C.; Jensen, P.; Thomsen, K.; Kolpen, M.; Rybtke, M.; Lauland, A.S.; Trøstrup, H.; Tolker-Nielsen, T. Immune Responses to Pseudomonas aeruginosa Biofilm Infections. Front. Immunol. 2021, 12, 625597. [Google Scholar] [CrossRef]
- López, C.A.; Zgurskaya, H.; Gnanakaran, S. Molecular characterization of the outer membrane of Pseudomonas aeruginosa. Biochim. Et Biophys. Acta (BBA) Biomembr. 2020, 1862, 183151. [Google Scholar] [CrossRef] [PubMed]
- Pang, Z.; Raudonis, R.; Glick, B.R.; Lin, T.-J.; Cheng, Z. Antibiotic resistance in Pseudomonas aeruginosa: Mechanisms and alternative therapeutic strategies. Biotechnol. Adv. 2019, 37, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Munita Jose, M.; Arias Cesar, A.; Kudva Indira, T.; Zhang, Q. Mechanisms of Antibiotic Resistance. Microbiol. Spectr. 2016, 4, 481–511. [Google Scholar]
- Breidenstein, E.B.; de la Fuente-Núñez, C.; Hancock, R.E. Pseudomonas aeruginosa: All roads lead to resistance. Trends Microbiol. 2011, 19, 419–426. [Google Scholar] [CrossRef]
- Bisht, K.; Baishya, J.; Wakeman, C.A. Pseudomonas aeruginosa polymicrobial interactions during lung infection. Curr. Opin. Microbiol. 2020, 53, 1–8. [Google Scholar] [CrossRef]
- Raman, G.; Avendano, E.E.; Chan, J.; Merchant, S.; Puzniak, L. Risk factors for hospitalized patients with resistant or multidrug-resistant Pseudomonas aeruginosa infections: A systematic review and meta-analysis. Antimicrob. Resist. Infect. Control 2018, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- Coates, M.; Blanchard, S.; MacLeod, A.S. Innate antimicrobial immunity in the skin: A protective barrier against bacteria, viruses, and fungi. PLOS Pathog. 2018, 14, e1007353. [Google Scholar] [CrossRef]
- Elmassry, M.M.; Mudaliar, N.S.; Colmer-Hamood, J.A.; Francisco, M.J.S.; Griswold, J.A.; Dissanaike, S.; Hamood, A.N. New markers for sepsis caused by Pseudomonas aeruginosa during burn infection. Metabolomics 2020, 16, 40. [Google Scholar] [CrossRef] [PubMed]
- Mahar, P.; Padiglione, A.A.; Cleland, H.; Paul, E.; Hinrichs, M.; Wasiak, J. Pseudomonas aeruginosa bacteraemia in burns patients: Risk factors and outcomes. Burns 2010, 36, 1228–1233. [Google Scholar] [CrossRef]
- England, P.H. Laboratory Surveillance of Pseudomonas and Stenotrophomonas spp. Bacteraemia in England; Wales and Northern Ireland: London, UK, 2018. [Google Scholar]
- Weintrob, A.C.; Murray, C.K.; Xu, J.; Krauss, M.; Bradley, W.; Warkentien, T.E.; Lloyd, B.A.; Tribble, D.R. Early Infections Complicating the Care of Combat Casualties from Iraq and Afghanistan. Surg. Infect. 2018, 19, 286–297. [Google Scholar] [CrossRef]
- Kollef, M.H.; Chastre, J.; Fagon, J.-Y.; François, B.; Niederman, M.S.; Rello, J.; Torres, A.; Vincent, J.-L.; Wunderink, R.; Go, K.W.; et al. Global Prospective Epidemiologic and Surveillance Study of Ventilator-Associated Pneumonia due to Pseudomonas aeruginosa. Crit. Care Med. 2014, 42, 2178–2187. [Google Scholar] [CrossRef] [PubMed]
- Pitiriga, V.; Kanellopoulos, P.; Bakalis, I.; Kampos, E.; Sagris, I.; Saroglou, G.; Tsakris, A. Central venous catheter-related bloodstream infection and colonization: The impact of insertion site and distribution of multidrug-resistant pathogens. Antimicrob. Resist. Infect. Control 2020, 9, 189. [Google Scholar] [CrossRef] [PubMed]
- Olejnickova, K.; Hola, V.; Ruzicka, F. Catheter-related infections caused by Pseudomonas aeruginosa: Virulence factors involved and their relationships. Pathog. Dis. 2014, 72, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Safdar, N.; Dezfulian, C.; Collard, H.R.; Saint, S. Clinical and economic consequences of ventilator-associated pneumonia: A systematic review. Crit. Care Med. 2005, 33, 2184–2193. [Google Scholar] [CrossRef]
- Mehta, A.C.; Muscarella, L.F. Bronchoscope-Related “Superbug” Infections. Chest 2020, 157, 454–469. [Google Scholar] [CrossRef]
- Baidya, S.; Sharma, S.; Mishra, S.K.; Kattel, H.P.; Parajuli, K.; Sherchand, J.B. Biofilm Formation by Pathogens Causing Ventilator-Associated Pneumonia at Intensive Care Units in a Tertiary Care Hospital: An Armor for Refuge. BioMed Res. Int. 2021, 2021, 8817700. [Google Scholar] [CrossRef]
- Bédard, E.; Prévost, M.; Déziel, E. Pseudomonas aeruginosa in premise plumbing of large buildings. MicrobiologyOpen 2016, 5, 937–956. [Google Scholar] [CrossRef] [PubMed]
- Arancibia, F.; Bauer, T.T.; Ewig, S.; Mensa, J.; Gonzalez, J.; Niederman, M.S.; Torres, A. Community-Acquired Pneumonia Due to Gram-Negative Bacteria and Pseudomonas aeruginosa: Incidence, Risk, and Prognosis. Arch. Intern. Med. 2002, 162, 1849–1858. [Google Scholar] [CrossRef] [PubMed]
- Kaier, K.; Heister, T.; Götting, T.; Wolkewitz, M.; Mutters, N. Measuring the in-hospital costs of Pseudomonas aeruginosa pneumonia: Methodology and results from a German teaching hospital. BMC Infect. Dis. 2019, 19, 1028. [Google Scholar] [CrossRef]
- Restrepo, M.I.; Babu, B.L.; Reyes, L.F.; Chalmers, J.D.; Soni, N.J.; Sibila, O.; Faverio, P.; Cilloniz, C.; Rodriguez-Cintron, W.; Aliberti, S. Burden and risk factors for Pseudomonas aeruginosa community-acquired pneumonia: A multinational point prevalence study of hospitalised patients. Eur. Respir. J. 2018, 52, 1701190. [Google Scholar] [CrossRef]
- Elborn, J.S. Cystic fibrosis. Lancet 2016, 388, 2519–2531. [Google Scholar] [CrossRef]
- Pressler, T.; Bohmova, C.; Conway, S.; Dumcius, S.; Hjelte, L.; Høiby, N.; Kollberg, H.; Tümmler, B.; Vavrova, V. Chronic Pseudomonas aeruginosa infection definition: EuroCareCF Working Group report. J. Cyst. Fibros. 2011, 10, S75–S78. [Google Scholar] [CrossRef]
- Rossi, E.; La Rosa, R.; Bartell, J.A.; Marvig, R.L.; Haagensen, J.A.J.; Sommer, L.M.; Molin, S.; Johansen, H.K. Pseudomonas aeruginosa adaptation and evolution in patients with cystic fibrosis. Nat. Rev. Genet. 2021, 19, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Rau, M.H.; Hansen, S.K.; Johansen, H.K.; Thomsen, L.E.; Workman, C.T.; Nielsen, K.F.; Jelsbak, L.; Høiby, N.; Yang, L.; Molin, S. Early adaptive developments of Pseudomonas aeruginosa after the transition from life in the environment to persistent colonization in the airways of human cystic fibrosis hosts. Environ. Microbiol. 2010, 12, 1643–1658. [Google Scholar]
- Waters Christopher, M.; Goldberg Joanna, B. Pseudomonas aeruginosa in cystic fibrosis: A chronic cheater. Proc. Natl. Acad. Sci. USA 2019, 116, 6525–6527. [Google Scholar] [CrossRef] [PubMed]
- Ehsan, Z.; Clancy, J.P. Management of Pseudomonas aeruginosa infection in cystic fibrosis patients using inhaled antibiotics with a focus on nebulized liposomal amikacin. Futur. Microbiol. 2015, 10, 1901–1912. [Google Scholar] [CrossRef] [PubMed]
- Hartl, D.; Gaggar, A.; Bruscia, E.; Hector, A.; Marcos, V.; Jung, A.; Greene, C.; McElvaney, G.; Mall, M.; Döring, G. Innate immunity in cystic fibrosis lung disease. J. Cyst. Fibros. 2012, 11, 363–382. [Google Scholar] [CrossRef]
- Avendaño-Ortiz, J.; Llanos-González, E.; Toledano, V.; del Campo, R.; Cubillos-Zapata, C.; Lozano-Rodríguez, R.; Ismail, A.; Prados, C.; Gómez-Campelo, P.; Aguirre, L.A.; et al. Pseudomonas aeruginosa colonization causes PD-L1 overexpression on monocytes, impairing the adaptive immune response in patients with cystic fibrosis. J. Cyst. Fibros. 2019, 18, 630–635. [Google Scholar] [CrossRef]
- Costas, L.M.; Martí, S.; Ardanuy, C.; Liñares, J.; Santos, S.; Dorca, J.; Garcia, M.; Monsó, E. Specific immune response against Pseudomonas aeruginosa and haemophilus influenzae in severe COPD. Eur. Respir. J. 2016, 48, OA1790. [Google Scholar]
- Kwok, W.C.; Ho, J.C.M.; Tam, T.C.C.; Ip, M.S.M.; Lam, D.C.L. Risk factors for Pseudomonas aeruginosa colonization in non-cystic fibrosis bronchiectasis and clinical implications. Respir. Res. 2021, 22, 132. [Google Scholar] [CrossRef]
- Wesley Alexander, J.; Fisher, M.W. Vaccination for Pseudomonas aeruginosa. Am. J. Surg. 1970, 120, 512. [Google Scholar] [CrossRef]
- Young, L.S.; Meyer, R.D.; Armstrong, D. Pseudomonas aeruginosa Vaccine in Cancer Patients. Ann. Intern. Med. 1973, 79, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Pennington, J.E. Preliminary Investigations of Pseudomonas aeruginosa Vaccine in Patients with Leukemia and Cystic Fibrosis. J. Infect. Dis. 1974, 130, S159–S162. [Google Scholar] [CrossRef] [PubMed]
- Pennington, J.E.; Reynolds, H.Y.; Wood, R.E.; Robinson, R.A.; Levine, A.S. Use of a Pseudomonas aeruginosa vaccine in patients with acute leukemia and cystic fibrosis. Am. J. Med. 1975, 58, 629–636. [Google Scholar] [CrossRef]
- Jones, R.J.; Roe, E.A.; Lowbury, E.J.L.; Miler, J.J.; Spilsbury, J.F. A new Pseudomonas vaccine: Preliminary trial on human volunteers. J. Hyg. 1976, 76, 429–439. [Google Scholar] [CrossRef]
- Elizabeth, A.R.; Roderick, J.J. Immunization of Burned Patients against Pseudomonas aeruginosa Infection at Safdarjang Hospital, New Delhi. Rev. Infect. Dis. 1983, 5, S922–S930. [Google Scholar]
- Langford, D.T.; Hiller, J. Prospective, controlled study of a polyvalent pseudomonas vaccine in cystic fibrosis--three year results. Arch. Dis. Child. 1984, 59, 1131–1134. [Google Scholar] [CrossRef] [PubMed]
- Cryz, S.J.; Fürer, E.; Cross, A.S.; Wegmann, A.; Germanier, R.; Sadoff, J.C. Safety and immunogenicity of a Pseudomonas aeruginosa O-polysaccharide toxin A conjugate vaccine in humans. J. Clin. Investig. 1987, 80, 51–56. [Google Scholar] [CrossRef]
- Cryz, S.J.; Sadoff, J.C.; Fürer, E. Immunization with a Pseudomonas aeruginosa immunotype 5 O polysaccharide-toxin A conjugate vaccine: Effect of a booster dose on antibody levels in humans. Infect. Immun. 1988, 56, 1829–1830. [Google Scholar] [CrossRef] [PubMed]
- Schaad, U.; Wedgwood, J.; Ruedeberg, A.; Lang, A.; Que, J.; Furer, E.; Cryz, S. Safety and immunogenicity of Pseudomonas aeruginosa conjugate A vaccine in cystic fibrosis. Lancet 1991, 338, 1236–1237. [Google Scholar] [CrossRef]
- Döring, G. Prevention of Pseudomonas aeruginosa infection in cystic fibrosis patients. Int. J. Med Microbiol. 2010, 300, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Crowe, B.A.; Enzersberger, O.; Schober-Bendixen, S.; Mitterer, A.; Mundt, W.; Livey, I.; Pabst, H.; Kaeser, R.; Eibl, M.; Eibl, J.; et al. The First Clinical Trial of Immuno’s Experimental Pseudomonas aeruginosa Flagellar Vaccines. Pseudomonas Aeruginosa Hum. Dis. 1991, 44, 143–156. [Google Scholar] [CrossRef]
- Döring, G.; Pfeiffer, C.; Weber, U.; Mohr-Pennert, A.; Dorner, F. Parenteral application of a Pseudomonas aeruginosa flagella vaccine elicits specific anti-flagella antibodies in the airways of healthy individuals. Am. J. Respir. Crit. Care Med. 1995, 151, 983–985. [Google Scholar] [CrossRef] [PubMed]
- Döring, G.; Meisner, C.; Stern, M. For the Flagella Vaccine Trial Study Group A double-blind randomized placebo-controlled phase III study of a Pseudomonas aeruginosa flagella vaccine in cystic fibrosis patients. Proc. Natl. Acad. Sci. USA 2007, 104, 11020–11025. [Google Scholar] [CrossRef] [PubMed]
- Pier, G.B.; DesJardin, D.; Grout, M.; Garner, C.; Bennett, S.E.; Pekoe, G.; Fuller, S.A.; Thornton, M.O.; Harkonen, W.S.; Miller, H.C. Human immune response to Pseudomonas aeruginosa mucoid exopolysaccharide (alginate) vaccine. Infect. Immun. 1994, 62, 3972–3979. [Google Scholar] [CrossRef] [PubMed]
- Cripps, A.W.; Dunkley, M.L.; Clancy, R.L.; Kyd, J. Vaccine strategies against Pseudomonas aeruginosa infection in the lung. Behring Inst. Mitt. 1997, 98, 262–268. [Google Scholar]
- Cripps, A.W.; Peek, K.; Dunkley, M.; Vento, K.; Marjason, J.K.; McIntyre, M.E.; Sizer, P.; Croft, D.; Sedlak-Weinstein, L. Safety and Immunogenicity of an Oral Inactivated Whole-Cell Pseudomonas aeruginosa Vaccine Administered to Healthy Human Subjects. Infect. Immun. 2006, 74, 968–974. [Google Scholar] [CrossRef]
- Mansouri, E.; Gabelsberger, J.; Knapp, B.; Hundt, E.; Lenz, U.; Hungerer, K.D.; Gilleland Jr, H.E.; Staczek, J.; Domdey, H.; von Specht, B.U. Safety and Immunogenicity of a Pseudomonas aeruginosa Hybrid Outer Membrane Protein F-I Vaccine in Human Volunteers. Infect. Immun. 1999, 67, 1461–1470. [Google Scholar] [CrossRef]
- Mansouri, E.; Blome-Eberwein, S.; Gabelsberger, J.; Germann, G.N.; Specht, B.-U. Clinical study to assess the immunogenicity and safety of a recombinant Pseudomonas aeruginosa OprF-OprI vaccine in burn patients. FEM Immunol. Med. Microbiol. 2003, 37, 161–166. [Google Scholar] [CrossRef]
- Westritschnig, K.; Hochreiter, R.; Wallner, G.; Firbas, C.; Schwameis, M.; Jilma, B. A randomized, placebo-controlled phase I study assessing the safety and immunogenicity of a Pseudomonas aeruginosa hybrid outer membrane protein OprF/I vaccine (IC43) in healthy volunteers. Hum. Vaccines Immunother. 2014, 10, 170–183. [Google Scholar] [CrossRef]
- Rello, J.; Krenn, C.-G.; Locker, G.; Pilger, E.; Madl, C.; Balica, L.; Dugernier, T.; Laterre, P.-F.; Spapen, H.; Depuydt, P.; et al. A randomized placebo-controlled phase II study of a Pseudomonas vaccine in ventilated ICU patients. Crit. Care 2017, 21, 22. [Google Scholar] [CrossRef] [PubMed]
- Adlbrecht, C.; Wurm, R.; Depuydt, P.; Spapen, H.; Lorente, J.A.; Staudinger, T.; Creteur, J.; Zauner, C.; Meier-Hellmann, A.; Eller, P.; et al. Efficacy, immunogenicity, and safety of IC43 recombinant Pseudomonas aeruginosa vaccine in mechanically ventilated intensive care patients—a randomized clinical trial. Crit. Care 2020, 24, 74. [Google Scholar] [CrossRef] [PubMed]
- Larbig, M.; Mansouri, E.; Freihorst, J.; Tümmler, B.; Köhler, G.; Domdey, H.; Knapp, B.; Hungerer, K.; Hundt, E.; Gabelsberger, J.; et al. Safety and immunogenicity of an intranasal Pseudomonas aeruginosa hybrid outer membrane protein F-I vaccine in human volunteers. Vaccine 2001, 19, 2291–2297. [Google Scholar] [CrossRef]
- Göcke, K.; Baumann, U.; Hagemann, H.; Gabelsberger, J.; Hahn, H.; Freihorst, J.; von Specht, B.U. Mucosal vaccination with a recombinant OprF-I vaccine of Pseudomonas aeruginosa in healthy volunteers: Comparison of a systemic vs. a mucosal booster schedule. FEMS Immunol. Med. Microbiol. 2003, 37, 167–171. [Google Scholar] [CrossRef]
- Baumann, U.; Göcke, K.; Gewecke, B.; Freihorst, J.; Von Specht, B.U. Assessment of pulmonary antibodies with induced sputum and bronchoalveolar lavage induced by nasal vaccination against Pseudomonas aeruginosa: A clinical phase I/II study. Respir. Res. 2007, 8, 57. [Google Scholar] [CrossRef]
- Lang, A.B.; Rüdeberg, A.; Schöni, M.H.; Que, J.U.; Fürer, E.; Schaad, U.B. Vaccination of Cystic Fibrosis Patients Against Pseudomonas aeruginosa Reduces the Proportion of Patients Infected and Delays Time to Infection. Pediatr. Infect. Dis. J. 2004, 23, 504–510. [Google Scholar] [CrossRef]
- Zuercher, A.W.; Imboden, M.A.; Jampen, S.; Bosse, D.; Ulrich, M.; Chtioui, H.; Lauterburg, B.H.; Lang, A.B. Cellular immunity in healthy volunteers treated with an octavalent conjugate Pseudomonas aeruginosa vaccine. Clin. Exp. Immunol. 2006, 142, 381–387. [Google Scholar] [CrossRef]
- Pier, G.B.; Saunders, J.M.; Ames, P.; Edwards, M.S.; Auerbach, H.; Goldfarb, J.; Speert, D.P.; Hurwitch, S. Opsonophagocytic Killing Antibody to Pseudomonas aeruginosa Mucoid Exopolysaccharide in Older Noncolonized Patients with Cystic Fibrosis. N. Engl. J. Med. 1987, 317, 793–798. [Google Scholar] [CrossRef]
- Meluleni, G.J.; Grout, M.; Evans, D.; Pier, G. Mucoid Pseudomonas aeruginosa growing in a biofilm in vitro are killed by opsonic antibodies to the mucoid exopolysaccharide capsule but not by antibodies produced during chronic lung infection in cystic fibrosis patients. J. Immunol. 1995, 155, 2029–2038. [Google Scholar]
- Mutharia, L.M.; Hancock, R.E. Surface localization of Pseudomonas aeruginosa outer membrane porin protein F by using monoclonal antibodies. Infect. Immun. 1983, 42, 1027–1033. [Google Scholar] [CrossRef]
- Von Specht, B.U.; Knapp, B.; Muth, G.; Bröker, M.; Hungerer, K.D.; Diehl, K.D.; Massarrat, K.; Seemann, A.; Domdey, H. Protection of immunocompromised mice against lethal infection with Pseudomonas aeruginosa by active or passive immunization with recombinant P. aeruginosa outer membrane protein F and outer membrane protein I fusion proteins. Infect. Immun. 1995, 63, 1855–1862. [Google Scholar] [CrossRef] [PubMed]
- Von Specht, B.U.; Gabelsberger, J.; Knapp, B.; Hundt, E.; Schmidt-Pilger, H.; Bauernsachs, S.; Lenz, U.; Domdey, H. Immunogenic efficacy of differently produced recombinant vaccines candidates against Pseudomonas aeruginosa infections. J. Biotechnol. 2000, 83, 3–12. [Google Scholar] [CrossRef]
- Kaye, K.S.; Pogue, J.M. Infections Caused by Resistant Gram-Negative Bacteria: Epidemiology and Management. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2015, 35, 949–962. [Google Scholar] [CrossRef] [PubMed]
- Sorichter, S.; Baumann, U.; Baumgart, A.; Walterspacher, S.; von Specht, B.-U. Immune responses in the airways by nasal vaccination with systemic boosting against Pseudomonas aeruginosa in chronic lung disease. Vaccine 2009, 27, 2755–2759. [Google Scholar] [CrossRef]
- Sainz-Mejías, M.; Jurado-Martín, I.; McClean, S. Understanding Pseudomonas aeruginosa–Host Interactions: The Ongoing Quest for an Efficacious Vaccine. Cells 2020, 9, 2617. [Google Scholar] [CrossRef]
- Baker, S.M.; McLachlan, J.B.; Morici, L.A. Immunological considerations in the development of Pseudomonas aeruginosa vaccines. Hum. Vaccines Immunother. 2020, 16, 412–418. [Google Scholar] [CrossRef]
- Lavoie, E.G.; Wangdi, T.; Kazmierczak, B.I. Innate immune responses to Pseudomonas aeruginosa infection. Microbes Infect. 2011, 13, 1133–1145. [Google Scholar] [CrossRef]
- Mauch, R.; Jensen, P.; Moser, C.; Levy, C.; Høiby, N. Mechanisms of humoral immune response against Pseudomonas aeruginosa biofilm infection in cystic fibrosis. J. Cyst. Fibros. 2018, 17, 143–152. [Google Scholar] [CrossRef]
- McIsaac, S.M.; Stadnyk, A.W.; Lin, T.-J. Toll-like receptors in the host defense against Pseudomonas aeruginosa respiratory infection and cystic fibrosis. J. Leukoc. Biol. 2012, 92, 977–985. [Google Scholar] [CrossRef]
- Lin, C.K.; Kazmierczak, B.I. Inflammation: A Double-Edged Sword in the Response to Pseudomonas aeruginosa Infection. J. Innate Immun. 2017, 9, 250–261. [Google Scholar] [CrossRef]
- Jensen, E.T.; Kharazmi, A.; Garred, P.; Kronborg, G.; Fomsgaard, A.; Mollnes, T.E.; Høiby, N. Complement activation by Pseudomonas aeruginosa biofilms. Microb. Pathog. 1993, 15, 377–388. [Google Scholar] [CrossRef]
- Johansen, H.K.; Nørregaard, L.L.; Gøtzsche, P.C.; Pressler, T.; Koch, C.B.; Høiby, N. Antibody response to Pseudomonas aeruginosa in cystic fibrosis patients: A marker of therapeutic success?—A 30-year Cohort study of survival in Danish CF patients after onset of chronic P. aeruginosa lung infection. Pediatric Pulmonol. 2004, 37, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.; Martin, L.; Bright-Thomas, R.; Dodd, M.; McDowell, A.; Moffitt, K.; Elborn, J.; Webb, A. Inflammatory markers in cystic fibrosis patients with transmissible Pseudomonas aeruginosa. Eur. Respir. J. 2003, 22, 503–506. [Google Scholar] [CrossRef] [PubMed]
- Hellfritzsch, M.; Scherließ, R. Mucosal Vaccination via the Respiratory Tract. Pharmaceutics 2019, 11, 375. [Google Scholar] [CrossRef] [PubMed]
- Lavelle, E.C.; Ward, R.W. Mucosal vaccines—Fortifying the frontiers. Nat. Rev. Immunol. 2022, 22, 236–250. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Mu, Y.; Lu, L.; Tian, Y.; Yuan, F.; Zhou, B.; Yu, C.; Wang, Z.; Li, X.; Lei, S.; et al. Hydrogen peroxide-inactivated bacteria induces potent humoral and cellular immune responses and releases nucleic acids. Int. Immunopharmacol. 2019, 69, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Ma, X.; Jiang, B.; Pan, H.; Liao, X.; Zhang, L.; Li, W.; Luo, Y.; Shen, Z.; Cheng, X.; et al. A novel inactivated whole-cell Pseudomonas aeruginosa vaccine that acts through the cGAS-STING pathway. Signal Transduct. Target. Ther. 2021, 6, 353. [Google Scholar] [CrossRef]
- Meynet, E.; Laurin, D.; Lenormand, J.L.; Camara, B.; Toussaint, B.; Le Gouëllec, A. Killed but metabolically active Pseudomonas aeruginosa -based vaccine induces protective humoral- and cell-mediated immunity against Pseudomonas aeruginosa pulmonary infections. Vaccine 2018, 36, 1893–1900. [Google Scholar] [CrossRef]
- Jing, H.; Zhang, X.; Zou, J.; Yuan, Y.; Chen, Z.; Liu, D.; Wu, W.; Yang, F.; Lu, D.; Zou, Q.; et al. Oligomerization of IC43 resulted in improved immunogenicity and protective efficacy against Pseudomonas aeruginosa lung infection. Int. J. Biol. Macromol. 2020, 159, 174–182. [Google Scholar] [CrossRef]
- Ding, B.; von Specht, B.-U.; Li, Y. OprF/I-vaccinated sera inhibit binding of human interferon-gamma to Pseudomonas aeruginosa. Vaccine 2010, 28, 4119–4122. [Google Scholar] [CrossRef]
- Rhee, J.H. Chapter 19—Current and New Approaches for Mucosal Vaccine Delivery. In Mucosal Vaccines, 2nd ed.; Kiyono, H., Pascual, D.W., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 325–356. [Google Scholar]
- Moyle, P.M.; Toth, I. Modern Subunit Vaccines: Development, Components, and Research Opportunities. ChemMedChem 2013, 8, 360–376. [Google Scholar] [CrossRef] [PubMed]
- Moxon, R.; Reche, P.A.; Rappuoli, R. Editorial: Reverse Vaccinology. Front. Immunol. 2019, 10, 2776. [Google Scholar] [CrossRef] [PubMed]
- Rashid, M.I.; Naz, A.; Ali, A.; Andleeb, S. Prediction of vaccine candidates against Pseudomonas aeruginosa: An integrated genomics and proteomics approach. Genomics 2017, 109, 274–283. [Google Scholar] [CrossRef]
- Bianconi, I.; Franco, B.A.; Scarselli, M.; Dalsass, M.; Buccato, S.; Colaprico, A.; Marchi, S.; Masignani, V.; Bragonzi, A. Genome-Based Approach Delivers Vaccine Candidates Against Pseudomonas aeruginosa. Front. Immunol. 2019, 9, 3021. [Google Scholar] [CrossRef] [PubMed]
- Day, C.J.; Hartley-Tassell, L.E.; Seib, K.L.; Tiralongo, J.; Bovin, N.; Savino, S.; Masignani, V.; Jennings, M.P. Lectin activity of Pseudomonas aeruginosa vaccine candidates PSE17-1, PSE41-5 and PSE54. Biochem. Biophys. Res. Commun. 2019, 513, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Solanki, V.; Tiwari, M.; Tiwari, V. Prioritization of potential vaccine targets using comparative proteomics and designing of the chimeric multi-epitope vaccine against Pseudomonas aeruginosa. Sci. Rep. 2019, 9, 5240. [Google Scholar] [CrossRef]
- Wu, W.; Huang, J.; Duan, B.; Traficante, D.C.; Hong, H.; Risech, M.; Lory, S.; Priebe, G.P. Th17-stimulating Protein Vaccines Confer Protection against Pseudomonas aeruginosa Pneumonia. Am. J. Respir. Crit. Care Med. 2012, 186, 420–427. [Google Scholar] [CrossRef]
- Mall, M.A. Role of Cilia, Mucus, and Airway Surface Liquid in Mucociliary Dysfunction: Lessons from Mouse Models. J. Aerosol Med. Pulm. Drug Deliv. 2008, 21, 13–24. [Google Scholar] [CrossRef]
- Baker, S.M.; Pociask, D.; Clements, J.D.; McLachlan, J.B.; Morici, L.A. Intradermal vaccination with a Pseudomonas aeruginosa vaccine adjuvanted with a mutant bacterial ADP-ribosylating enterotoxin protects against acute pneumonia. Vaccine 2019, 37, 808–816. [Google Scholar] [CrossRef]
- Das, S.; Howlader, D.R.; Zheng, Q.; Ratnakaram, S.S.K.; Whittier, S.K.; Lu, T.; Keith, J.D.; Picking, W.D.; Birket, S.E.; Picking, W.L. Development of a Broadly Protective, Self-Adjuvanting Subunit Vaccine to Prevent Infections by Pseudomonas aeruginosa. Front. Immunol. 2020, 11, 583008. [Google Scholar] [CrossRef]
- Gbian, D.; Omri, A. The Impact of an Efflux Pump Inhibitor on the Activity of Free and Liposomal Antibiotics against Pseudomonas aeruginosa. Pharmaceutics 2021, 13, 577. [Google Scholar] [CrossRef] [PubMed]
- Solleti, V.S.; Alhariri, M.; Halwani, M.; Omri, A. Antimicrobial properties of liposomal azithromycin for Pseudomonas infections in cystic fibrosis patients. J. Antimicrob. Chemother. 2015, 70, 784–796. [Google Scholar] [CrossRef] [PubMed]
- Heurtault, B.; Gentine, P.; Thomann, J.-S.; Baehr, C.; Frisch, B.; Pons, F. Design of a Liposomal Candidate Vaccine Against Pseudomonas aeruginosa and its Evaluation in Triggering Systemic and Lung Mucosal Immunity. Pharm. Res. 2009, 26, 276–285. [Google Scholar] [CrossRef]
- Mayeux, G.; Gayet, L.; Liguori, L.; Odier, M.; Martin, D.K.; Cortès, S.; Schaack, B.; Lenormand, J.-L. Cell-free expression of the outer membrane protein OprF of Pseudomonas aeruginosa for vaccine purposes. Life Sci. Alliance 2021, 4, e202000958. [Google Scholar] [CrossRef]
- Ellis Terri, N.; Leiman Sara, A.; Kuehn Meta, J. Naturally Produced Outer Membrane Vesicles from Pseudomonas aeruginosa Elicit a Potent Innate Immune Response via Combined Sensing of Both Lipopolysaccharide and Protein Components. Infect. Immun. 2010, 78, 3822–3831. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wang, X.; Sun, X.; Guan, Z.; Sun, W.; Burns Drusilla, L. Outer Membrane Vesicles Displaying a Heterologous PcrV-HitA Fusion Antigen Promote Protection against Pulmonary Pseudomonas aeruginosa Infection. mSphere 2021, 6, e00699-21. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wang, X.; Sun, X.; Cimino, J.; Guan, Z.; Sun, W. Recombinant Pseudomonas Bionanoparticles Induce Protection against Pneumonic Pseudomonas aeruginosa Infection. Infect. Immun. 2021, 89, IAI0039621. [Google Scholar] [CrossRef]
- Micoli, F.; MacLennan, C.A. Outer membrane vesicle vaccines. Semin. Immunol. 2020, 50, 101433. [Google Scholar] [CrossRef]
- Bumann, D.; Behre, C.; Behre, K.; Herz, S.; Gewecke, B.; Gessner, J.E.; von Specht, B.U.; Baumann, U. Systemic, nasal and oral live vaccines against Pseudomonas aeruginosa: A clinical trial of immunogenicity in lower airways of human volunteers. Vaccine 2010, 28, 707–713. [Google Scholar] [CrossRef]
- Zhang, M.; Sun, C.; Gu, J.; Yan, X.; Wang, B.; Cui, Z.; Sun, X.; Tong, C.; Feng, X.; Lei, L.; et al. Salmonella Typhimurium strain expressing OprF-OprI protects mice against fatal infection by Pseudomonas aeruginosa. Microbiol. Immunol. 2015, 59, 533–544. [Google Scholar] [CrossRef]
- Aguilera-Herce, J.; García-Quintanilla, M.; Romero-Flores, R.; McConnell, M.J.; Ramos-Morales, F. A Live Salmonella Vaccine Delivering PcrV through the Type III Secretion System Protects against Pseudomonas aeruginosa. mSphere 2019, 4, e00116-19. [Google Scholar] [CrossRef]
- Chen, H.; Ji, H.; Kong, X.; Lei, P.; Yang, Q.; Wu, W.; Jin, L.; Sun, D. Bacterial Ghosts-Based Vaccine and Drug Delivery Systems. Pharmaceutics 2021, 13, 1892. [Google Scholar] [CrossRef]
- Sheweita, S.A.; Amara, A.A.; Gamal, H.; Ghazy, A.A.; Hussein, A.; Bahey-El-Din, M. Bacterial Ghosts of Pseudomonas aeruginosa as a Promising Candidate Vaccine and Its Application in Diabetic Rats. Vaccines 2022, 10, 910. [Google Scholar] [CrossRef]
- Rodgers, A.M.; McCrudden, M.T.C.; Vincente-Perez, E.M.; Dubois, A.V.; Ingram, R.J.; Larrañeta, E.; Kissenpfennig, A.; Donnelly, R.F. Design and characterisation of a dissolving microneedle patch for intradermal vaccination with heat-inactivated bacteria: A proof of concept study. Int. J. Pharm. 2018, 549, 87–95. [Google Scholar] [CrossRef]
- Cui, Z.; Han, D.; Sun, X.; Zhang, M.; Feng, X.; Sun, C.; Gu, J.; Tong, C.; Lei, L.; Han, W. Mannose-modified chitosan microspheres enhance OprF-OprI-mediated protection of mice against Pseudomonas aeruginosa infection via induction of mucosal immunity. Appl. Microbiol. Biotechnol. 2015, 99, 667–680. [Google Scholar] [CrossRef]
- Najafzadeh, F.; Tanomand, A.; Hadadi, A.; Majidi, J. Immunological Properties of Exotoxin A Toxoid—Detoxified Lipopolysaccharide—Gold Nanoparticles Conjugate Against Pseudomonas aeruginosa Infection. Iran. J. Immunol. 2021, 18, 292–303. [Google Scholar] [PubMed]
- Chen, Y.; Yang, F.; Yang, J.; Hou, Y.; He, L.; Hu, H.; Lv, F. Aluminum (oxy) Hydroxide Nanorods Activate an Early Immune Response in Pseudomonas aeruginosa Vaccine. ACS Appl. Mater. Interfaces 2018, 10, 43533–43542. [Google Scholar] [CrossRef] [PubMed]
- Howlader, D.R.; Das, S.; Lu, T.; Hu, G.; Varisco, D.J.; Dietz, Z.K.; Walton, S.P.; Ratnakaram, S.S.K.; Gardner, F.M.; Ernst, R.K.; et al. Effect of Two Unique Nanoparticle Formulations on the Efficacy of a Broadly Protective Vaccine Against Pseudomonas aeruginosa. Front. Pharmacol. 2021, 12, 2137. [Google Scholar] [CrossRef]
- Safari Zanjani, L.; Shapouri, R.; Dezfulian, M.; Mahdavi, M.; Shafiee Ardestani, M. Exotoxin A-PLGA nanoconjugate vaccine against Pseudomonas aeruginosa infection: Protectivity in murine model. World J. Microbiol. Biotechnol. 2019, 35, 94. [Google Scholar] [CrossRef]
- Azimi, S.; Zanjani, L.S. Immunization against Pseudomonas aeruginosa using Alg-PLGA nano-vaccine. Iran. J. Basic Med Sci. 2021, 24, 476–482. [Google Scholar] [CrossRef]
- Gonzaga, Z.J.C.; Zhang, J.; Rehm, B.H.A. Intranasal Delivery of Antigen-Coated Polymer Particles Protects against Pseudomonas aeruginosa Infection. ACS Infect. Dis. 2022, 8, 744–756. [Google Scholar] [CrossRef]


| Intrinsic | Acquired | Adaptive |
|---|---|---|
| Restricted outer membrane permeability | Mutational changes | Biofilm formation |
| Antibiotic-inactivating enzymes | Over-expression of resistance genes | Persister cells |
| Efflux pumps | Horizontal transfer of resistance genes |
| Antigen | Formulation | Phase | Dosage, Administration, Adjuvant | Population | Outcomes | Study and Reference |
|---|---|---|---|---|---|---|
| Lipopolysaccharide | LPS extracts from 7 PA serotypes (Pseudogen®) | II | NS | 72 burns | Prevented development of sepsis and subsequent death | Alexander and Fisher (1970) [42] |
| II | NS, IM, 5 doses, none | 361 cancer | Reduced mortality, slight reduction in fatal PA infection but associated with adverse events | Young et al. (1973) [43] | ||
| II | 6–12 µg/kg, IM, 6 doses, none | 12 CF, 22 acute leukaemia | No clinical benefit, CF patients showed antibody response adverse events in 95% leukaemia patients | Pennington et al. (1975) [44,45] | ||
| LPS extracts from 16 PA serotypes (PEV-01) | I | 0.5 mL, SC, 3 doses, none | 15 healthy | Variable antibody response, no toxic adverse events | Jones et al. (1976) [46] | |
| II | 0.5 mL, SC, 3 doses, none | 746 burns | Reduced mortality in adults and children, variable antibody response, overall increase in bactericidal capacity of blood | Roe and Jones (1983) [47] | ||
| II | 0.25, 0.5 mL, SC, 3 doses, none | 34 CF | No reduction in colonization, vaccinated and colonized individuals suffered most rapid deterioration | Langford and Hiller (1984) [48] | ||
| LPS extracts from 8 PA serotypes conjugated to Exotoxin A (Aerugen®) | I | 0.5 mL, SC, 2 doses, none | 20 healthy | Safe, anti-exotoxin and anti-LPS IgG produced, boosted at 15 months | Cryz et al. (1987, 1988) [49,50] | |
| II | 6–12 µg/kg, IM, 3 doses, none | 30 CF, non-colonised | High affinity IgG response to exotoxin and LPS, no change in clinical status | Schaad et al. (1991) [51] | ||
| III | NS | 476 CF | No clinical difference between groups at interim analysis—study stopped | Döring (2010) [52] | ||
| Flagellum | Monovalent | I | 40 µg, IM, NS, Al(OH)3 | 220 healthy | High serum and respiratory mucosal anti-flagella antibody titres | Crowe et al. (1991) [53] |
| II | 40 µg, IM, NS, none | 10 healthy | Döring et al. (1995) [54] | |||
| Bivalent | III | 40 µg, IM, 4 doses, Al(OH)3 and thiomersal | 483 CF | Long-lasting serum anti-flagella serotype-specific antibodies, 34% protected against acute infection, 51% protected against chronic infection | Döring et al. (2007) [55] | |
| Alginate | 2 preparations of MEP extracts from mucoid PA | I | 10, 50, 100, 150 µg, IM, 2 doses, none | 28 healthy | Poorly functioning opsonizing antibodies, not augmented by booster dose | Pier et al. (1994) [56] |
| Whole-cell killed | Pseudostat® | I | NS, PO, NS, NS | 9 bronchiectasis | Induction of specific lymphocyte response, decrease in bacterial sputum counts | Cripps et al. (1997) [57] |
| I | 150 mg, PO, 2 doses, none | 30 healthy | IgG and IgA opsonizing antibodies, 20 adverse events | Cripps et al. (2006) [58] | ||
| Outer Membrane Proteins | OprF-OprI systemic formulation (IC43) | I | 20, 50, 100, 500 µg, IM, 4 doses, Al(OH)3 | 32 healthy | Complement binding and opsonizing antibodies present at 6-months post 3rd vaccine | McGhee et al. (1999) [59] |
| I | 100 µg, IM, 3 doses, Al(OH)3 | 8 burns | Well-tolerated, seroconversion in 7 subjects | Mansouri et al. (2003) [60] | ||
| I | 50, 100, 200 µg, IM, 2 doses, Al(OH)3 | 163 healthy | Safe and well-tolerated, induced specific IgG response vs placebo, higher doses were not more effective | Westritschnig et al. (2014) [61] | ||
| II | 100, 200 µg, IM, 2 doses, Al(OH)3 | 401 mechanically ventilated ICU | Increased IgG persisting until day 70 post-vaccination, not powered to assess infection rates/mortality | Rello et al. (2017) [62] | ||
| II/III | 100 µg, IM, 2 doses, none | 799 mechanically ventilated ICU | Well-tolerated and immunogenic, no difference in survival or mortality vs. placebo | Adlbrecht et al. (2020) [63] | ||
| OprF-OprI mucosal formulation (IC43) | I | 500 µg, IN, 3 doses | 8 healthy | Safe and well-tolerated, 6 subjects showed increased serum IgG and IgA | Larbig et al. (2001) [64] | |
| OprF-OprI systemic and mucosal formulations (Comparative study) | I/II | Mucosal 1 mg, IN, 2 doses (+1 booster), noneSystemic 100 µg, IM, (1 booster), Al(OH)3 | 12 healthy (6 mucosal only, 6 mucosal with systemic booster) | Safe and well-tolerated, immunogenic in all, serum IgG higher with systemic booster | Göcke et al. (2003) [65] Baumann et al. (2007) [66] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Killough, M.; Rodgers, A.M.; Ingram, R.J. Pseudomonas aeruginosa: Recent Advances in Vaccine Development. Vaccines 2022, 10, 1100. https://doi.org/10.3390/vaccines10071100
Killough M, Rodgers AM, Ingram RJ. Pseudomonas aeruginosa: Recent Advances in Vaccine Development. Vaccines. 2022; 10(7):1100. https://doi.org/10.3390/vaccines10071100
Chicago/Turabian StyleKillough, Matthew, Aoife Maria Rodgers, and Rebecca Jo Ingram. 2022. "Pseudomonas aeruginosa: Recent Advances in Vaccine Development" Vaccines 10, no. 7: 1100. https://doi.org/10.3390/vaccines10071100
APA StyleKillough, M., Rodgers, A. M., & Ingram, R. J. (2022). Pseudomonas aeruginosa: Recent Advances in Vaccine Development. Vaccines, 10(7), 1100. https://doi.org/10.3390/vaccines10071100





