COVID-19 Vaccine Hesitancy: The Perils of Peddling Science by Social Media and the Lay Press
Abstract
:1. Introduction
2. Methodology
3. Results
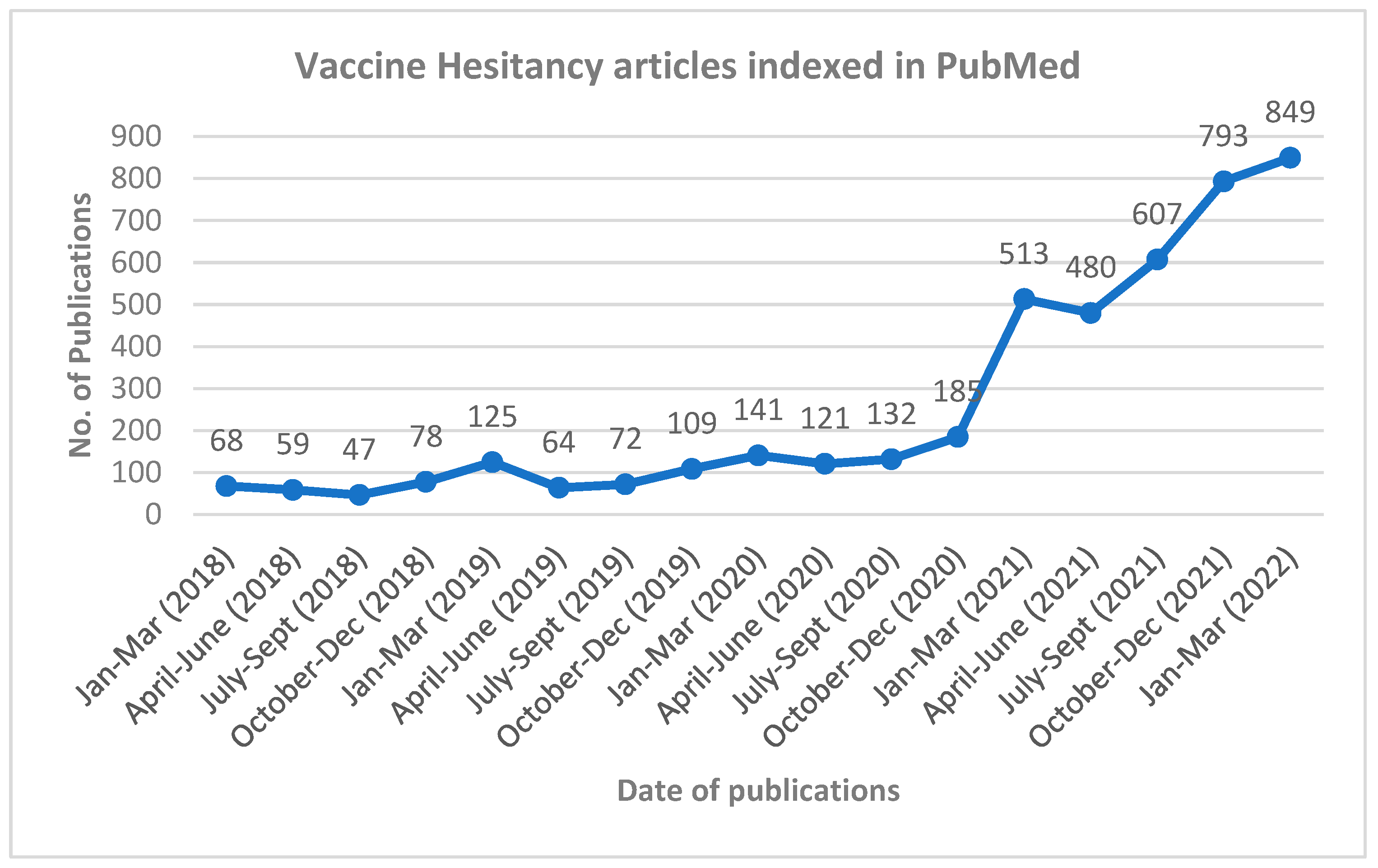
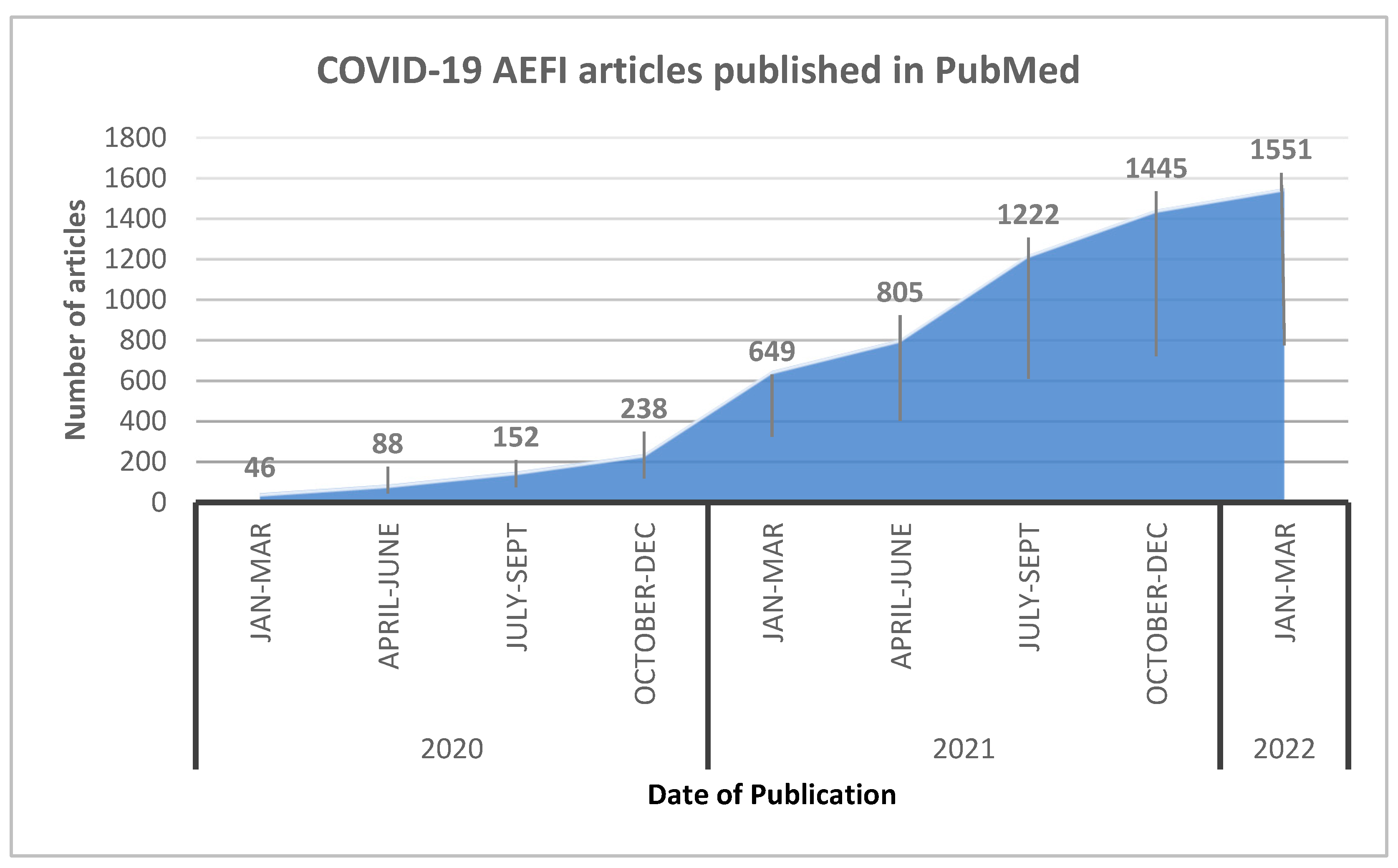
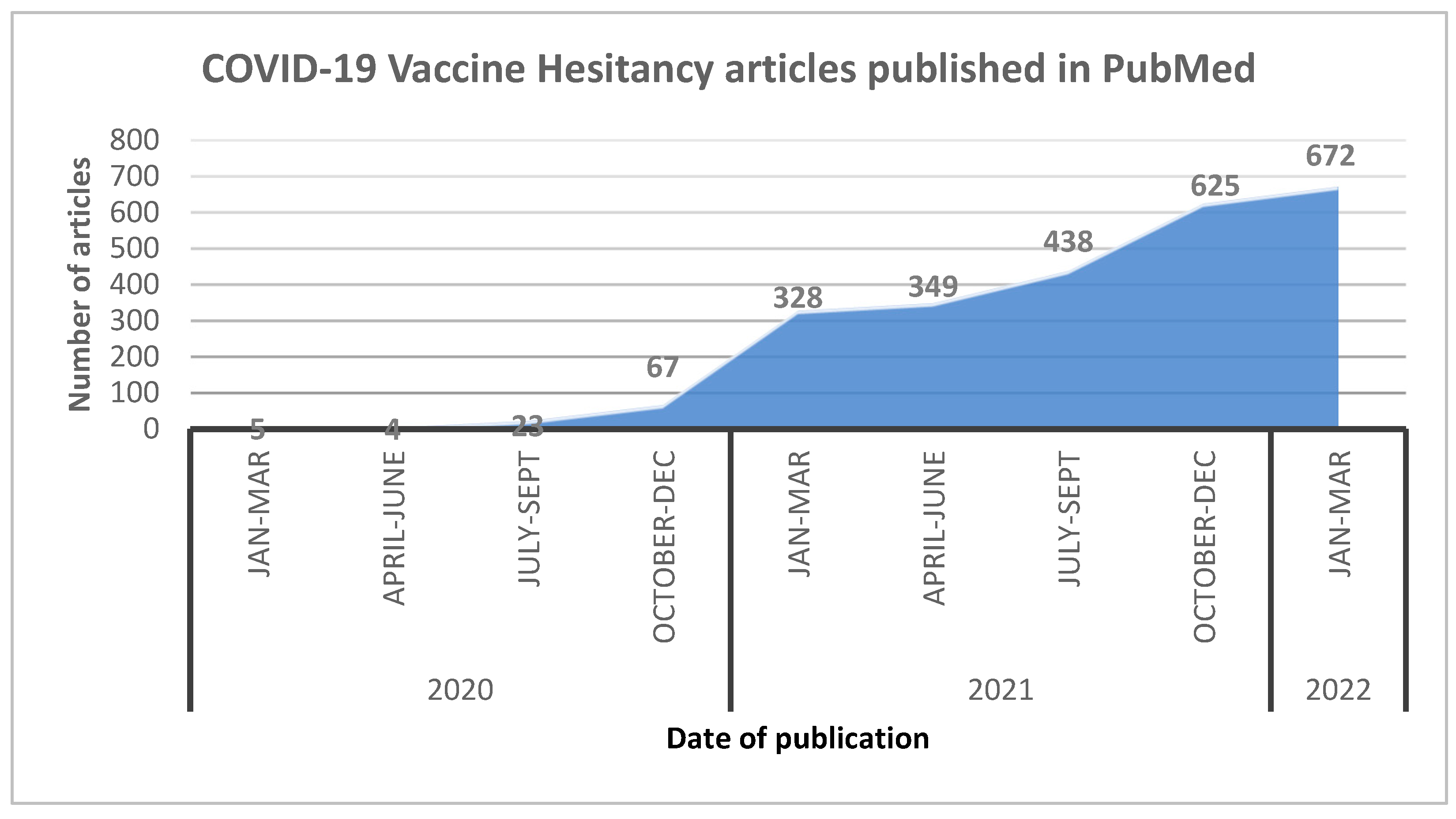
Vaccine Hesitancy and AEFI Literatures
4. Discussion
4.1. COVID-19 Vaccine Hesitancy
4.2. Adverse Events following COVID-19 Vaccination
4.3. Recommendations to Overcome VH
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boston, 677 Huntington Avenue; Ma 02115 +1495-1000 Barry, R. Bloom’s Faculty Website. Available online: https://www.hsph.harvard.edu/barry-bloom/ (accessed on 6 May 2022).
- Orenstein, W.A.; Ahmed, R. Simply Put: Vaccination Saves Lives. Proc. Natl. Acad. Sci. USA 2017, 114, 4031–4033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeyanathan, M.; Afkhami, S.; Smaill, F.; Miller, M.S.; Lichty, B.D.; Xing, Z. Immunological Considerations for COVID-19 Vaccine Strategies. Nat. Rev. Immunol. 2020, 20, 615–632. [Google Scholar] [CrossRef] [PubMed]
- Chapter-Vaccines.Pdf. Available online: https://www.who.int/publications/10-year-review/chapter-vaccines.pdf (accessed on 10 March 2022).
- Harris, K.M.; Maurer, J.; Kellermann, A.L. Influenza Vaccine--Safe, Effective, and Mistrusted. N. Engl. J. Med. 2010, 363, 2183–2185. [Google Scholar] [CrossRef] [PubMed]
- Horne, Z.; Powell, D.; Hummel, J.E.; Holyoak, K.J. Countering Antivaccination Attitudes. Proc. Natl. Acad. Sci. USA 2015, 112, 10321–10324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hornsey, M.J.; Harris, E.A.; Fielding, K.S. The Psychological Roots of Anti-Vaccination Attitudes: A 24-Nation Investigation. Health Psychol. 2018, 37, 307–315. [Google Scholar] [CrossRef]
- Aschwanden, C. Five Reasons Why COVID Herd Immunity Is Probably Impossible. Nature 2021, 591, 520–522. [Google Scholar] [CrossRef]
- Frontiers|The Relationship Between the COVID-19 Pandemic and Vaccine Hesitancy: A Scoping Review of Literature Until August 2021|Public Health. Available online: https://www.frontiersin.org/articles/10.3389/fpubh.2021.747787/full (accessed on 27 April 2022).
- Machingaidze, S.; Wiysonge, C.S. Understanding COVID-19 Vaccine Hesitancy. Nat. Med. 2021, 27, 1338–1339. [Google Scholar] [CrossRef]
- Ten Health Issues WHO Will Tackle This Year. Available online: https://www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019 (accessed on 10 March 2022).
- Puri, N.; Coomes, E.A.; Haghbayan, H.; Gunaratne, K. Social Media and Vaccine Hesitancy: New Updates for the Era of COVID-19 and Globalized Infectious Diseases. Hum. Vaccin. Immunother. 2020, 16, 2586–2593. [Google Scholar] [CrossRef]
- Abdool Karim, S.S.; de Oliveira, T. New SARS-CoV-2 Variants–Clinical, Public Health, and Vaccine Implications. N. Engl. J. Med. 2021, 384, 1866–1868. [Google Scholar] [CrossRef]
- Karafillakis, E.; Damme, P.V.; Hendrickx, G.; Larson, H.J. COVID-19 in Europe: New Challenges for Addressing Vaccine Hesitancy. Lancet 2022, 399, 699–701. [Google Scholar] [CrossRef]
- Dubé, E.; Vivion, M.; MacDonald, N.E. Vaccine Hesitancy, Vaccine Refusal and the Anti-Vaccine Movement: Influence, Impact and Implications. Expert Rev. Vaccines 2015, 14, 99–117. [Google Scholar] [CrossRef] [PubMed]
- Paules, C.I.; Marston, H.D.; Fauci, A.S. Measles in 2019–Going Backward. N. Engl. J. Med. 2019, 380, 2185–2187. [Google Scholar] [CrossRef] [PubMed]
- Putman, M.; Kennedy, K.; Sirotich, E.; Liew, J.W.; Sattui, S.E.; Moni, T.T.; Akpabio, A.A.; Alpizar-Rodriguez, D.; Angevare, S.; Beesley, R.P.; et al. COVID-19 Vaccine Perceptions and Uptake: Results from the COVID-19 Global Rheumatology Alliance Vaccine Survey. Lancet Rheumatol. 2022, 4, e237–e240. [Google Scholar] [CrossRef]
- Nordström, P.; Ballin, M.; Nordström, A. Risk of Infection, Hospitalisation, and Death up to 9 Months after a Second Dose of COVID-19 Vaccine: A Retrospective, Total Population Cohort Study in Sweden. Lancet 2022, 399, 814–823. [Google Scholar] [CrossRef]
- Paul, E.; Fancourt, D. Predictors of Uncertainty and Unwillingness to Receive the COVID-19 Booster Vaccine: An Observational Study of 22,139 Fully Vaccinated Adults in the UK. Lancet Reg. Health Eur. 2022, 14, 100317. [Google Scholar] [CrossRef]
- Solís Arce, J.S.; Warren, S.S.; Meriggi, N.F.; Scacco, A.; McMurry, N.; Voors, M.; Syunyaev, G.; Malik, A.A.; Aboutajdine, S.; Adeojo, O.; et al. COVID-19 Vaccine Acceptance and Hesitancy in Low- and Middle-Income Countries. Nat. Med. 2021, 27, 1385–1394. [Google Scholar] [CrossRef]
- Mohamed, N.A.; Solehan, H.M.; Rani, M.D.M.; Ithnin, M.; Isahak, C.I.C. Knowledge, Acceptance and Perception on COVID-19 Vaccine among Malaysians: A Web-Based Survey. PLoS ONE 2021, 16, e0256110. [Google Scholar] [CrossRef]
- Lucia, V.C.; Kelekar, A.; Afonso, N.M. COVID-19 Vaccine Hesitancy among Medical Students. J. Public Health (Oxf.) 2021, 43, 445–449. [Google Scholar] [CrossRef]
- Kelekar, A.K.; Lucia, V.C.; Afonso, N.M.; Mascarenhas, A.K. COVID-19 Vaccine Acceptance and Hesitancy among Dental and Medical Students. J. Am. Dent. Assoc. 2021, 152, 596–603. [Google Scholar] [CrossRef]
- Jain, J.; Saurabh, S.; Kumar, P.; Verma, M.K.; Goel, A.D.; Gupta, M.K.; Bhardwaj, P.; Raghav, P.R. COVID-19 Vaccine Hesitancy among Medical Students in India. Epidemiol. Infect. 2021, 149, e132. [Google Scholar] [CrossRef]
- Krishnamurthy, K.; Sobers, N.; Kumar, A.; Ojeh, N.; Scott, A.; Cave, C.; Gupta, S.; Bradford-King, J.; Sa, B.; Adams, O.P.; et al. COVID-19 Vaccine Intent Among Health Care Professionals of Queen Elizabeth Hospital, Barbados. J. Multidiscip. Healthc. 2021, 14, 3309–3319. [Google Scholar] [CrossRef] [PubMed]
- Paris, C.; Bénézit, F.; Geslin, M.; Polard, E.; Baldeyrou, M.; Turmel, V.; Tadié, É.; Garlantezec, R.; Tattevin, P. COVID-19 Vaccine Hesitancy among Healthcare Workers. Infect. Dis. Now 2021, 51, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Xu, X.-W.; Yang, J.; Zheng, J.; Dai, S.-M.; Zhou, J.; Zhang, Q.-M.; Ruan, Y.; Ling, C.-Q. Knowledge about, Attitude and Acceptance towards, and Predictors of Intention to Receive the COVID-19 Vaccine among Cancer Patients in Eastern China: A Cross-Sectional Survey. J. Integr. Med. 2022, 20, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Samanta, S.; Banerjee, J.; Kar, S.S.; Ali, K.M.; Giri, B.; Pal, A.; Dash, S.K. Awareness, knowledge and acceptance of COVID-19 vaccine among the people of West Bengal, India: A web-based survey. Vacunas 2022, 23, S46–S55. [Google Scholar] [CrossRef]
- Mahmud, S.; Mohsin, M.; Khan, I.A.; Mian, A.U.; Zaman, M.A. Knowledge, Beliefs, Attitudes and Perceived Risk about COVID-19 Vaccine and Determinants of COVID-19 Vaccine Acceptance in Bangladesh. PLoS ONE 2021, 16, e0257096. [Google Scholar] [CrossRef]
- Jairoun, A.A.; Al-Hemyari, S.S.; El-Dahiyat, F.; Jairoun, M.; Shahwan, M.; Al Ani, M.; Habeb, M.; Babar, Z.-U.-D. Assessing Public Knowledge, Attitudes and Determinants of Third COVID-19 Vaccine Booster Dose Acceptance: Current Scenario and Future Perspectives. J. Pharm. Policy Pract. 2022, 15, 26. [Google Scholar] [CrossRef]
- Cascini, F.; Pantovic, A.; Al-Ajlouni, Y.; Failla, G.; Ricciardi, W. Attitudes, Acceptance and Hesitancy among the General Population Worldwide to Receive the COVID-19 Vaccines and Their Contributing Factors: A Systematic Review. EClinicalMedicine 2021, 40, 101113. [Google Scholar] [CrossRef]
- Gallè, F.; Sabella, E.A.; Roma, P.; Ferracuti, S.; Da Molin, G.; Diella, G.; Montagna, M.T.; Orsi, G.B.; Liguori, G.; Napoli, C. Knowledge and Lifestyle Behaviors Related to COVID-19 Pandemic in People over 65 Years Old from Southern Italy. Int. J. Environ. Res. Public Health 2021, 18, 10872. [Google Scholar] [CrossRef]
- Tsutsumi, S.; Maeda, N.; Tashiro, T.; Arima, S.; Mizuta, R.; Fukui, K.; Naito, K.; Komiya, M.; Urabe, Y. Willingness to Receive the COVID-19 Vaccination and the Psychological State of Japanese University Students: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2022, 19, 1654. [Google Scholar] [CrossRef]
- Abbasi, J. Widespread Misinformation About Infertility Continues to Create COVID-19 Vaccine Hesitancy. JAMA 2022, 327, 1013–1015. [Google Scholar] [CrossRef]
- Islam, M.S.; Kamal, A.-H.M.; Kabir, A.; Southern, D.L.; Khan, S.H.; Hasan, S.M.M.; Sarkar, T.; Sharmin, S.; Das, S.; Roy, T.; et al. COVID-19 Vaccine Rumors and Conspiracy Theories: The Need for Cognitive Inoculation against Misinformation to Improve Vaccine Adherence. PLoS ONE 2021, 16, e0251605. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Causality Assessment of an Adverse Event Following Immunization (AEFI): User Manual for the Revised WHO Classification; World Health Organization: Geneva, Switzerland, 2019; ISBN 978-92-4-151699-0.
- Adverse Events Following Immunization (AEFI). Available online: https://www.who.int/teams/regulation-prequalification/regulation-and-safety/pharmacovigilance/health-professionals-info/aefi (accessed on 10 March 2022).
- Hoeve, C.E.; Gadroen, K.; Kwa, M.S.G.; van Haren, A.; Sturkenboom, M.C.J.M.; Straus, S.M.J.M. Fatal Outcomes Following Immunization Errors as Reported to the EudraVigilance: A Case Series. Vaccine 2020, 38, 3086–3095. [Google Scholar] [CrossRef]
- Kaur, U.; Ojha, B.; Pathak, B.K.; Singh, A.; Giri, K.R.; Singh, A.; Das, A.; Misra, A.; Yadav, A.K.; Kansal, S.; et al. A Prospective Observational Safety Study on ChAdOx1 NCoV-19 Corona Virus Vaccine (Recombinant) Use in Healthcare Workers- First Results from India. EClinicalMedicine 2021, 38, 101038. [Google Scholar] [CrossRef] [PubMed]
- CDC What to Expect after Getting a COVID-19 Vaccine. Available online: https://www.cdc.gov/coronavirus/2019-ncov/vaccines/expect/after.html (accessed on 10 March 2022).
- Karattuthodi, M.S.; Chandrasekher, D.; Panakkal, L.M.; Shinu, C.; Salman, M.; Megha; Muhammad, S.E.; Fasil, M.; Mohammad, A.M.; Reji, M. Pharmacist-Directed Sputnik V (GAM-COVID-VAC) Surveillance Program: A Prospective Observational Study in Southern India. J. Basic Clin. Physiol. Pharm. 2022. [Google Scholar] [CrossRef]
- Safety Information for Hepatitis B Vaccines|Vaccine Safety|CDC. Available online: https://www.cdc.gov/vaccinesafety/vaccines/hepatitis-b-vaccine.html (accessed on 10 March 2022).
- Castells, M.C.; Phillips, E.J. Maintaining Safety with SARS-CoV-2 Vaccines. N. Engl. J. Med. 2021, 384, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Dudley, M.Z.; Chen, X.; Bai, X.; Dong, K.; Zhuang, T.; Salmon, D.; Yu, H. Evaluation of the Safety Profile of COVID-19 Vaccines: A Rapid Review. BMC Med. 2021, 19, 173. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.; Xu, L.; Bai, Y.; Liu, Z.; Liu, Z.; Farrington, P.; Zhan, S. Self-Controlled Case Series Design in Vaccine Safety: A Systematic Review. Expert Rev. Vaccines 2022, 21, 313–324. [Google Scholar] [CrossRef]
- Wan, E.Y.F.; Chui, C.S.L.; Lai, F.T.T.; Chan, E.W.Y.; Li, X.; Yan, V.K.C.; Gao, L.; Yu, Q.; Lam, I.C.H.; Chun, R.K.C.; et al. Bell’s Palsy Following Vaccination with MRNA (BNT162b2) and Inactivated (CoronaVac) SARS-CoV-2 Vaccines: A Case Series and Nested Case-Control Study. Lancet Infect. Dis. 2022, 22, 64–72. [Google Scholar] [CrossRef]
- Fatal Cerebral Haemorrhage after COVID-19 Vaccine–PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/33928772/ (accessed on 10 March 2022).
- Thrombotic Thrombocytopenia after ChAdOx1 NCov-19 Vaccination|NEJM. Available online: https://www.nejm.org/doi/full/10.1056/NEJMoa2104840 (accessed on 10 March 2022).
- Althaus, K.; Möller, P.; Uzun, G.; Singh, A.; Beck, A.; Bettag, M.; Bösmüller, H.; Guthoff, M.; Dorn, F.; Petzold, G.C.; et al. Antibody-Mediated Procoagulant Platelets in SARS-CoV-2-Vaccination Associated Immune Thrombotic Thrombocytopenia. Haematologica 2021, 106, 2170–2179. [Google Scholar] [CrossRef]
- Ong, H.T. Comparison of Vaccine Efficacy Must Be Based on Good Clinical Data. Lancet Reg. Health West. Pac. 2022, 20, 100410. [Google Scholar] [CrossRef]
- Song, H.; Pei, X.; Liu, Z.; Shen, C.; Sun, J.; Liu, Y.; Zhou, L.; Sun, F.; Xiao, X. Pharmacovigilance in China: Evolution and Future Challenges. Br. J. Clin. Pharmacol. 2022. [Google Scholar] [CrossRef]
- Malhotra, H.S.; Gupta, P.; Prabhu, V.; Kumar Garg, R.; Dandu, H.; Agarwal, V. COVID-19 Vaccination-Associated Myelitis. QJM 2021, 114, 591–593. [Google Scholar] [CrossRef] [PubMed]
- Iguchi, T.; Umeda, H.; Kojima, M.; Kanno, Y.; Tanaka, Y.; Kinoshita, N.; Sato, D. Cumulative Adverse Event Reporting of Anaphylaxis After MRNA COVID-19 Vaccine (Pfizer-BioNTech) Injections in Japan: The First-Month Report. Drug Saf. 2021, 44, 1209–1214. [Google Scholar] [CrossRef] [PubMed]
- Hlatshwako, T.G.; Shah, S.J.; Kosana, P.; Adebayo, E.; Hendriks, J.; Larsson, E.C.; Hensel, D.J.; Erausquin, J.T.; Marks, M.; Michielsen, K.; et al. Online Health Survey Research during COVID-19. Lancet Digit. Health 2021, 3, e76–e77. [Google Scholar] [CrossRef]
- Dirty Data: The Effects of Screening Respondents Who Provide Low-Quality Data in Survey Research. Available online: https://www.springerprofessional.de/en/dirty-data-the-effects-of-screening-respondents-who-provide-low-/14981420 (accessed on 10 March 2022).
- Joshi, R.K.; Muralidharan, C.G.; Gulati, D.S.; Mopagar, V.; Dev, J.K.P.; Kuthe, S.; Rather, A.A.; Sahoo, A.K. Higher Incidence of Reported Adverse Events Following Immunisation (AEFI) after First Dose of COVID-19 Vaccine among Previously Infected Health Care Workers. Med. J. Armed. Forces India 2021, 77, S505–S507. [Google Scholar] [CrossRef] [PubMed]
- Myers, M.G.; Cairns, J.A.; Singer, J. The Consent Form as a Possible Cause of Side Effects. Clin. Pharmacol. Ther. 1987, 42, 250–253. [Google Scholar] [CrossRef]
- Webster, R.K.; Rubin, G.J. Influencing Side-Effects to Medicinal Treatments: A Systematic Review of Brief Psychological Interventions. Front. Psychiatry 2019, 9, 775. [Google Scholar] [CrossRef]
- Leibowitz, K.A.; Howe, L.C.; Crum, A.J. Changing Mindsets about Side Effects. BMJ Open 2021, 11, e040134. [Google Scholar] [CrossRef]
- The Spread of True and False News Online. Available online: https://www.science.org/doi/10.1126/science.aap9559 (accessed on 27 April 2022).
- Lay Epidemiology and Vaccine Acceptance|Vaccination|JAMA|JAMA Network. Available online: https://jamanetwork.com/journals/jama/fullarticle/2781943 (accessed on 10 March 2022).
- Garett, R.; Young, S.D. Online Misinformation and Vaccine Hesitancy. Transl. Behav. Med. 2021, 11, 2194–2199. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thorakkattil, S.A.; Abdulsalim, S.; Karattuthodi, M.S.; Unnikrishnan, M.K.; Rashid, M.; Thunga, G. COVID-19 Vaccine Hesitancy: The Perils of Peddling Science by Social Media and the Lay Press. Vaccines 2022, 10, 1059. https://doi.org/10.3390/vaccines10071059
Thorakkattil SA, Abdulsalim S, Karattuthodi MS, Unnikrishnan MK, Rashid M, Thunga G. COVID-19 Vaccine Hesitancy: The Perils of Peddling Science by Social Media and the Lay Press. Vaccines. 2022; 10(7):1059. https://doi.org/10.3390/vaccines10071059
Chicago/Turabian StyleThorakkattil, Shabeer Ali, Suhaj Abdulsalim, Mohammed Salim Karattuthodi, Mazhuvanchery Kesavan Unnikrishnan, Muhammed Rashid, and Girish Thunga. 2022. "COVID-19 Vaccine Hesitancy: The Perils of Peddling Science by Social Media and the Lay Press" Vaccines 10, no. 7: 1059. https://doi.org/10.3390/vaccines10071059
APA StyleThorakkattil, S. A., Abdulsalim, S., Karattuthodi, M. S., Unnikrishnan, M. K., Rashid, M., & Thunga, G. (2022). COVID-19 Vaccine Hesitancy: The Perils of Peddling Science by Social Media and the Lay Press. Vaccines, 10(7), 1059. https://doi.org/10.3390/vaccines10071059





