Poloxamer-188 Adjuvant Efficiently Maintains Adaptive Immunity of SARS-CoV-2 RBD Subunit Vaccination through Repressing p38MAPK Signaling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Vaccine Formulations and Immunization of Mice
2.2. Neutralization Assay Using SARS-CoV-2 Pseudoviruses
2.3. Binding Assay of S-Protein and Human ACE2
2.4. Cytokine and Apoptosis Assays
2.5. RT-qPCR
2.6. Western Blotting
2.7. Statistical Analysis
3. Results
3.1. Subsection
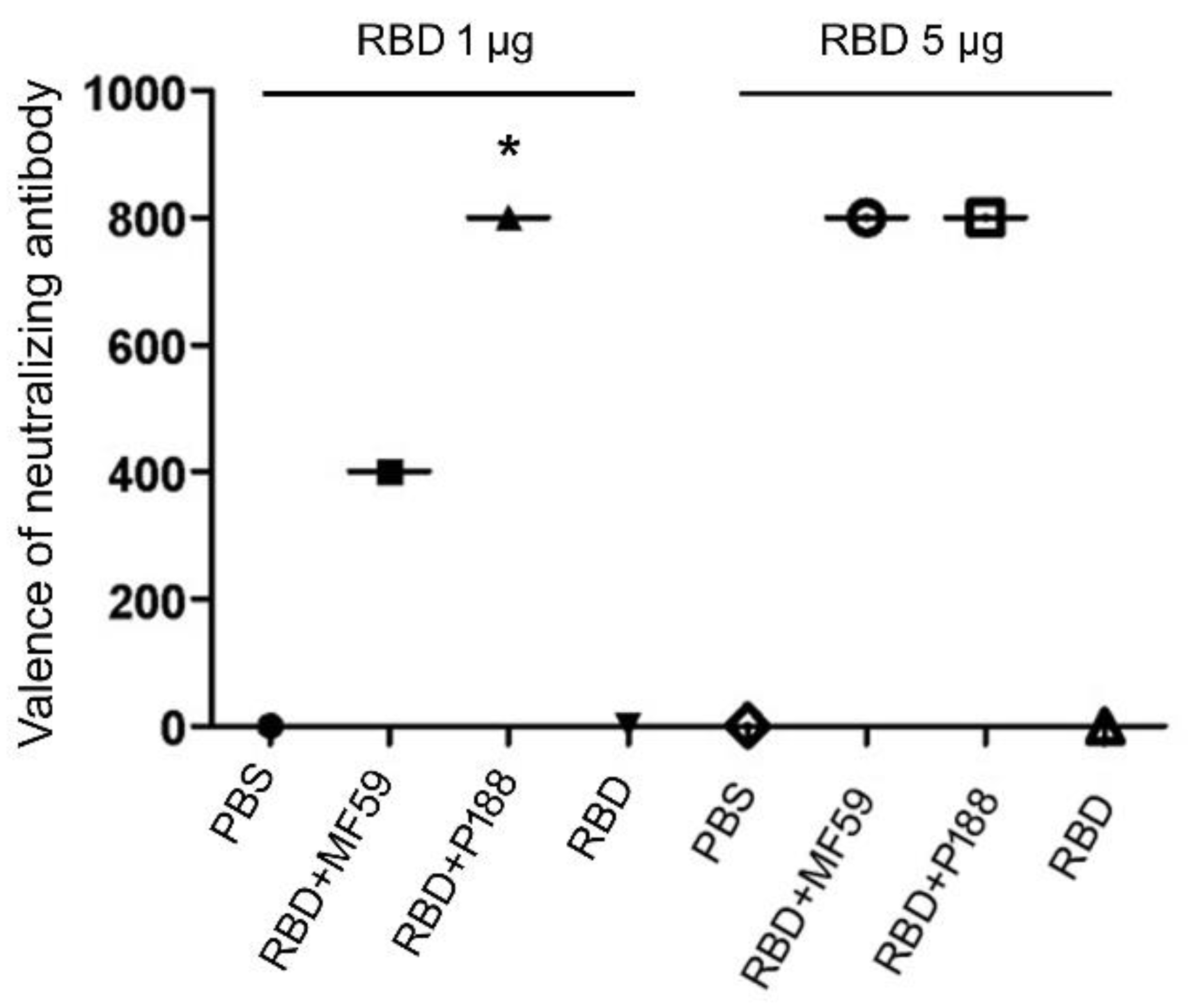
3.1.1. P188 Enhanced the Production of Neutralizing Antibody
3.1.2. P188-Adjuvanted RBD Vaccine Induced Antibodies That Block S Protein-hACE Interaction
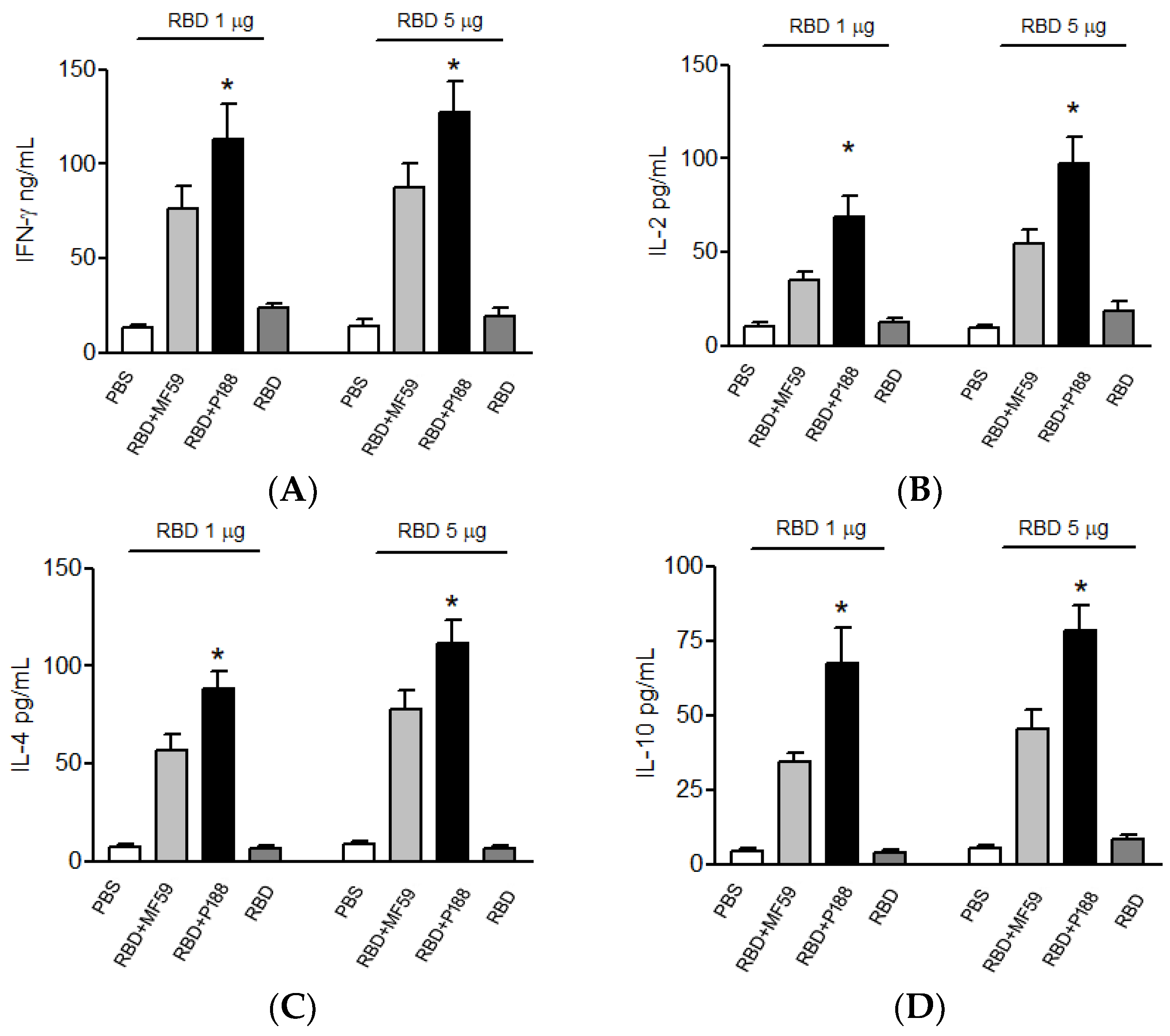
3.1.3. Splenocytes of the P188-Adjuvanted RBD Vaccine Group Produces Long-Lasting IFN-γ, IL-2, IL-10, and IL-4
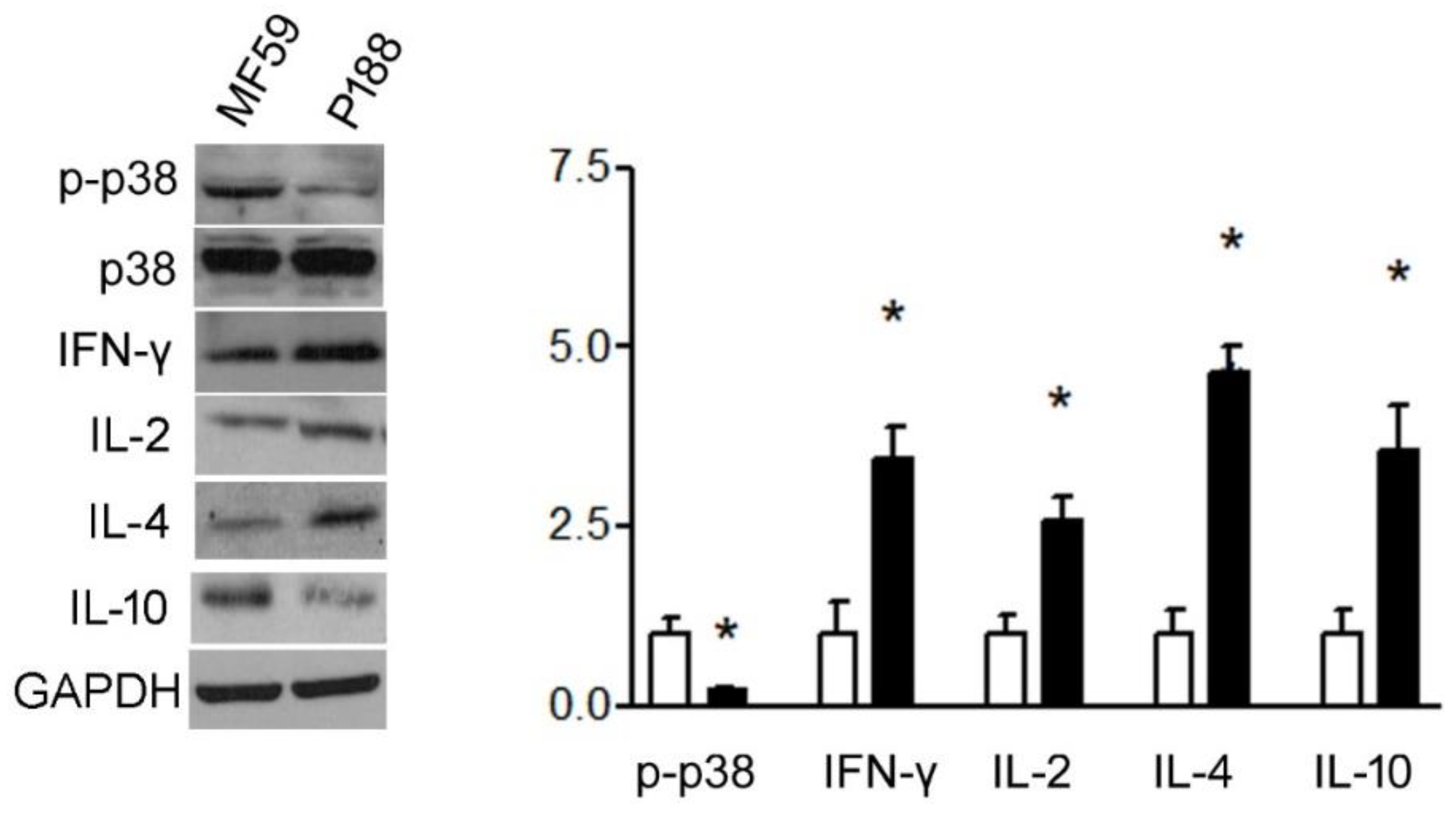
3.1.4. P188 Enhanced IFN-γ, IL-2, IL-10, and IL-4 Expression in Splenocytes by Inhibiting p38MAPK
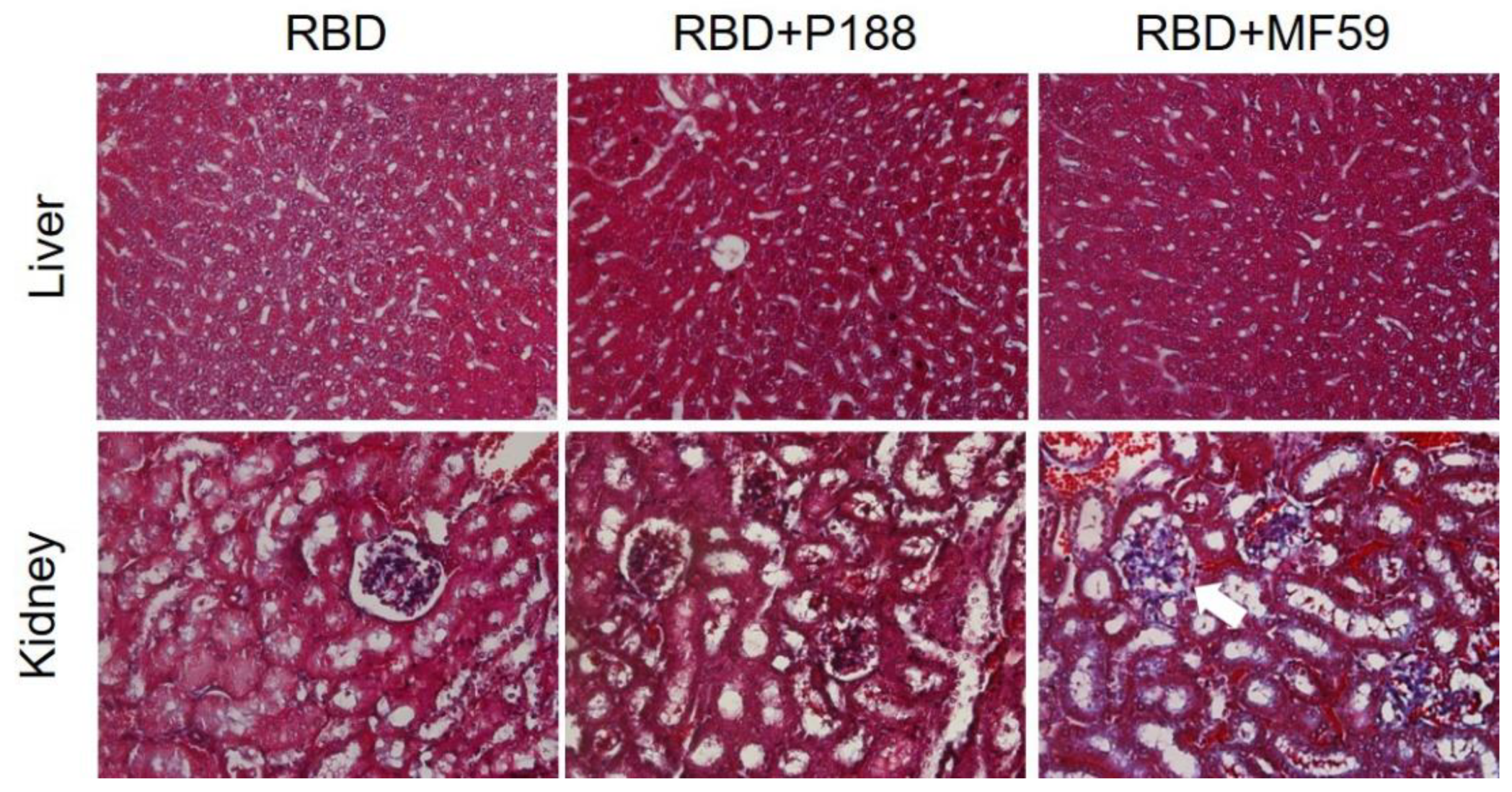
3.1.5. P188 Did Not Induce Systemic Adverse Effect after Vaccination
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Böger, B.; Fachi, M.M.; Vilhena, R.O.; Cobre, A.F.; Tonin, F.S.; Pontarolo, R. Systematic review with meta-analysis of the accuracy of diagnostic tests for COVID-19. Am. J. Infect. Control 2021, 49, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef]
- Izda, V.; Jeffries, M.A.; Sawalha, A.H. COVID-19: A review of therapeutic strategies and vaccine candidates. Clin. Immunol. 2021, 222, 108634. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Shin, B.; Kang, K.; Park, S.; Beck, B.R. Share Target-Centered Drug Repurposing Predictions of Human Angiotensin-Converting Enzyme 2 (ACE2) and Transmembrane Protease Serine Subtype 2 (TMPRSS2) Interacting Approved Drugs for Coronavirus Disease 2019 (COVID-19) Treatment through a Drug-Target Interaction Deep Learning Model. Viruses 2020, 12, 1325. [Google Scholar]
- Singh, H.; Choudhari, R.; Nema, V.; Khan, A.A. ACE2 and TMPRSS2 polymorphisms in various diseases with special reference to its impact on COVID-19 disease. Microb. Pathog. 2021, 150, 104621. [Google Scholar] [CrossRef]
- Corbett, K.S.; Edwards, D.K.; Leist, S.R.; Abiona, O.M.; Boyoglu-Barnum, S.; Gillespie, R.A.; Himansu, S.; Schäfer, A.; Ziwawo, C.T.; DiPiazza, A.T.; et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 2020, 586, 567–571. [Google Scholar] [CrossRef]
- Cho, H.; Jang, Y.; Park, K.H.; Choi, H.; Nowakowska, A.; Lee, H.J.; Kim, M.; Kang, M.H.; Ki, J.H.; Shin, H.Y.; et al. Human endogenous retrovirus-enveloped baculoviral DNA vaccines against MERS-CoV and SARS-CoV2. NPJ Vaccines 2021, 6, 37. [Google Scholar] [CrossRef]
- Park, K.S.; Sun, X.Q.; Aikins, M.E.; Moon, J.J. Non-viral COVID-19 vaccine delivery systemsAdv. Drug Deliv. Rev. 2021, 169, 137. [Google Scholar] [CrossRef]
- Lai, K.Y.; Au, S.Y.; Fong, K.M.; Choi, P.Y.I.; Eichinger, S.; Warkentin, T.E.; Greinacher, A. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N. Engl. J. Med. 2021, 384, 2092. [Google Scholar]
- Chung, J.Y.; Thone, M.N.; Kwon, Y.J. COVID-19 vaccines: The status and perspectives in delivery points of view. Adv. Drug Deliv. Rev. 2021, 170, 1–25. [Google Scholar] [CrossRef]
- Nelde, A.; Rammensee, H.G.; Walz, J.S. The Peptide Vaccine of the Future. Mol. Cell. Proteom. 2021, 20, 100022. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Lauritano, D.; Ronconi, G.; Calvisi, V.; Conti, P.J. Antibodies for COVID-19—Which, when and how long? J. Biol. Regul. Homeost. Agents 2021, 35, 417–422. [Google Scholar] [PubMed]
- Ko, E.J.; Kang, S.M. Immunology and efficacy of MF59-adjuvanted vaccines. Hum. Vaccines Immunother. 2018, 14, 3041–3045. [Google Scholar] [CrossRef]
- Chappell, K.J.; Mordant, F.L.; Li, Z.; Wijesundara, D.K.; Ellenberg, P.; Lackenby, J.A.; Cheung, S.T.M.; Modhiran, N.; Avumegah, M.S.; Henderson, C.L.; et al. Safety and immunogenicity of an MF59-adjuvanted spike glycoprotein-clamp vaccine for SARS-CoV-2: A randomised, double-blind, placebo-controlled, phase 1 trial. Lancet Infect. Dis. 2021, 21, 1383–1394. [Google Scholar] [CrossRef]
- Cerpa-Cruz, S.; Paredes-Casillas, P.; Landeros Navarro, E.; Bernard-Medina, A.G.; Martínez-Bonilla, G.; Gutiérrez-Ureña, S. Adverse events following immunization with vaccines containing adjuvants. Immunol. Res. 2013, 56, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Duggal, T.; Segal, P.; Shah, M. Antineutrophil cytoplasmic antibody vasculitis associated with influenza vaccination. Am. J. Nephrol. 2013, 38, 174–178. [Google Scholar] [CrossRef]
- Bolles, M.; Deming, D.; Long, K.; Agnihothram, S.; Whitmore, A.; Ferris, M.; Funkhouser, W.; Gralinski, L.; Totura, A.; Heise, M.; et al. A double-inactivated severe acute respiratory syndrome coronavirus vaccine provides incomplete protection in mice and induces increased eosinophilic proinflammatory pulmonary response upon challenge. J. Virol. 2011, 85, 12201–12215. [Google Scholar] [CrossRef] [Green Version]
- Soliman, K.A.; Ullah, K.; Shah, A.; Jones, D.S.; Singh, T.R.R. Poloxamer-based in situ gelling thermoresponsive systems for ocular drug delivery applications. Drug Discov. Today 2019, 24, 1575–1586. [Google Scholar] [CrossRef]
- Francke, N.M.; Bunjes, H. Drug localization and its effect on the physical stability of poloxamer 188-stabilized colloidal lipid emulsions. Int. J. Pharm. 2021, 599, 120394. [Google Scholar]
- Kilinc, D.; Gallo, G.; Barbee, K.A. Mechanically-induced membrane portion causes axonal beading and localized cytoskeletal damage. Exp. Neurol. 2008, 212, 422–430. [Google Scholar] [CrossRef]
- Luo, C.L.; Chen, X.P.; Li, L.L.; Li, Q.Q.; Li, B.X.; Xue, A.M.; Xu, H.F.; Dai, D.K.; Shen, Y.W.; Tao, L.Y.; et al. Poloxamer 188 attenuates in vitro traumatic brain injury-induced mitochondrial and lysosomal membrane permeabilization damage in cultured primary neurons. J. Neurotrauma 2013, 30, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Markham, A.; Thomas, S.J.; Wang, N.; Huang, L.; Clemens, M.; Rajagopalan, N. Solution Stability of Poloxamer 188 Under Stress Conditions. J. Pharm. Sci. 2019, 108, 1264–1271. [Google Scholar] [CrossRef]
- Czeiszperger, T.L.; Wang, M.P.; Chung, C.S. Membrane stabilizer Poloxamer 188 improves yield of primary isolated rat cardiomyocytes without impairing function. Physiol. Rep. 2020, 8, e14382. [Google Scholar] [CrossRef] [PubMed]
- Mehdipour, M.; Daghigh-Kia, H.; Martínez-Pastor, F. Poloxamer 188 exerts a cryoprotective effect on rooster sperm and allows decreasing glycerol concentration in the freezing extender. Poult. Sci. 2020, 99, 6212–6220. [Google Scholar] [CrossRef] [PubMed]
- Grapentin, C.; Müller, C.; Kishore, R.S.K.; Adler, M.; ElBialy, I.; Friess, W.; Huwyler, J.; Khan, T.A. Protein-Polydimethylsiloxane Particles in Liquid Vial Monoclonal Antibody Formulations Containing Poloxamer 188. J. Pharm. Sci. 2020, 109, 2393–2404. [Google Scholar] [CrossRef] [PubMed]
- Serbest, G.; Horwitz, J.; Jost, M.; Barbee, K. Mechanisms of cell death and neuroprotection by poloxamer 188 after mechanical trauma. FASEB J. 2006, 20, 308–310. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, X.; Darrah, P.A.; Mosser, D.M. The regulation of Th1 responses by the p38 MAPK. J. Immunol. 2010, 185, 6205–6213. [Google Scholar] [CrossRef] [PubMed]
- Jewell, R.C.; Khor, S.P.; Kisor, D.F.; LaCroix, K.A.; Wargin, W.A. Pharmacokinetics of RheothRx injection in healthy male volunteers. J. Pharm. Sci. 1997, 86, 808–812. [Google Scholar] [CrossRef]
- Adams-Graves, P.; Kedar, A.; Koshy, M.; Steinberg, M.; Veith, R.; Ward, D.; Crawford, R.; Edwards, S.; Bustrack, J.; Emanuele, M. RheothRx (poloxamer 188) injection for the acute painful episode of sickle cell disease: A pilot study. Blood 1997, 90, 2041–2046. [Google Scholar] [CrossRef]
- Casella, J.F.; Barton, B.A.; Kanter, J.; Black, L.V.; Majumdar, S.; Inati, A.; Wali, Y.; Drachtman, R.A.; Abboud, M.R.; Kilinc, Y.; et al. Effect of Poloxamer 188 vs Placebo on Painful Vaso-Occlusive Episodes in Children and Adults With Sickle Cell Disease: A Randomized Clinical Trial. JAMA 2021, 325, 1513–1523. [Google Scholar] [CrossRef]
- Paprikar, A.; Soni, A.; Kaushal, N.; Lin, S. Sublingual insulin administration: Application of hydroxypropyl beta-cyclodextrin and poloxamer188 as permeation enhancers. Pharm. Dev. Technol. 2021, 26, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Jin, X.; Zhang, Z.; Lv, H. Preparation and in vitro antitumor effects on MDA-MB-231 cells of niclosamide nanocrystals stabilized by poloxamer188 and PBS. Int. J. Pharm. 2020, 584, 119432. [Google Scholar] [CrossRef] [PubMed]
- Guler, N.; Abro, S.; Emanuele, M.; Iqbal, O.; Hoppensteadt, D.; Fareed, J. The Protective Effect of Poloxamer-188 on Platelet Functions. Clin. Appl. Thromb. Hemost. 2017, 23, 987–991. [Google Scholar] [CrossRef] [PubMed]
- Lakshmikuttyamma, A.; Sun, Y.; Lu, B.; Undieh, A.S.; Shoyele, S. A Stable and efficient transfection of siRNA for mutated KRAS silencing using novel hybrid nanoparticles. Mol. Pharm. 2014, 11, 4415–4424. [Google Scholar] [CrossRef]




| 1 μg RBD | 5 μg RBD | |||||||
|---|---|---|---|---|---|---|---|---|
| Group | PBS | RBD + MF59 | RBD + P188 | RBD | PBS | RBD + MF59 | RBD + P188 | RBD |
| GPT (U/I) | 51.14 ± 13.12 | 76.11 ± 21.13 | 61.15 ± 19.17 | 59.71 ± 22.63 | 57.17 ± 19.11 | 78.37 ± 18.93 | 69.17 ± 14.73 | 75.43 ± 19.77 |
| Blood urea nitrogen (BUN) (mg/dL) | 18.24 ± 3.51 | 16.82 ± 6.73 | 18.33 ± 5.61 | 21.11 ± 7.37 | 19.43 ± 8.11 | 22.16 ± 7.23 | 19.57 ± 8.36 | 15.78 ± 9.05 |
| Creatinine (Cr) (μmol/L) | 17.83 ± 4.27 | 18.79 ± 6.53 | 16.93 ± 6.53 | 20.17 ± 4.81 | 23.11 ± 7.19 | 15.85 ± 5.67 | 17.74 ± 4.71 | 19.45 ± 4.83 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-H.; Lin, Y.-J.; Cheng, L.-T.; Lin, C.-H.; Ke, G.-M. Poloxamer-188 Adjuvant Efficiently Maintains Adaptive Immunity of SARS-CoV-2 RBD Subunit Vaccination through Repressing p38MAPK Signaling. Vaccines 2022, 10, 715. https://doi.org/10.3390/vaccines10050715
Chen C-H, Lin Y-J, Cheng L-T, Lin C-H, Ke G-M. Poloxamer-188 Adjuvant Efficiently Maintains Adaptive Immunity of SARS-CoV-2 RBD Subunit Vaccination through Repressing p38MAPK Signaling. Vaccines. 2022; 10(5):715. https://doi.org/10.3390/vaccines10050715
Chicago/Turabian StyleChen, Chao-Hung, Yu-Jen Lin, Li-Ting Cheng, Chien-Hung Lin, and Guan-Ming Ke. 2022. "Poloxamer-188 Adjuvant Efficiently Maintains Adaptive Immunity of SARS-CoV-2 RBD Subunit Vaccination through Repressing p38MAPK Signaling" Vaccines 10, no. 5: 715. https://doi.org/10.3390/vaccines10050715
APA StyleChen, C.-H., Lin, Y.-J., Cheng, L.-T., Lin, C.-H., & Ke, G.-M. (2022). Poloxamer-188 Adjuvant Efficiently Maintains Adaptive Immunity of SARS-CoV-2 RBD Subunit Vaccination through Repressing p38MAPK Signaling. Vaccines, 10(5), 715. https://doi.org/10.3390/vaccines10050715






