Vaccine Design against Chagas Disease Focused on the Use of Nucleic Acids
Abstract
1. Introduction
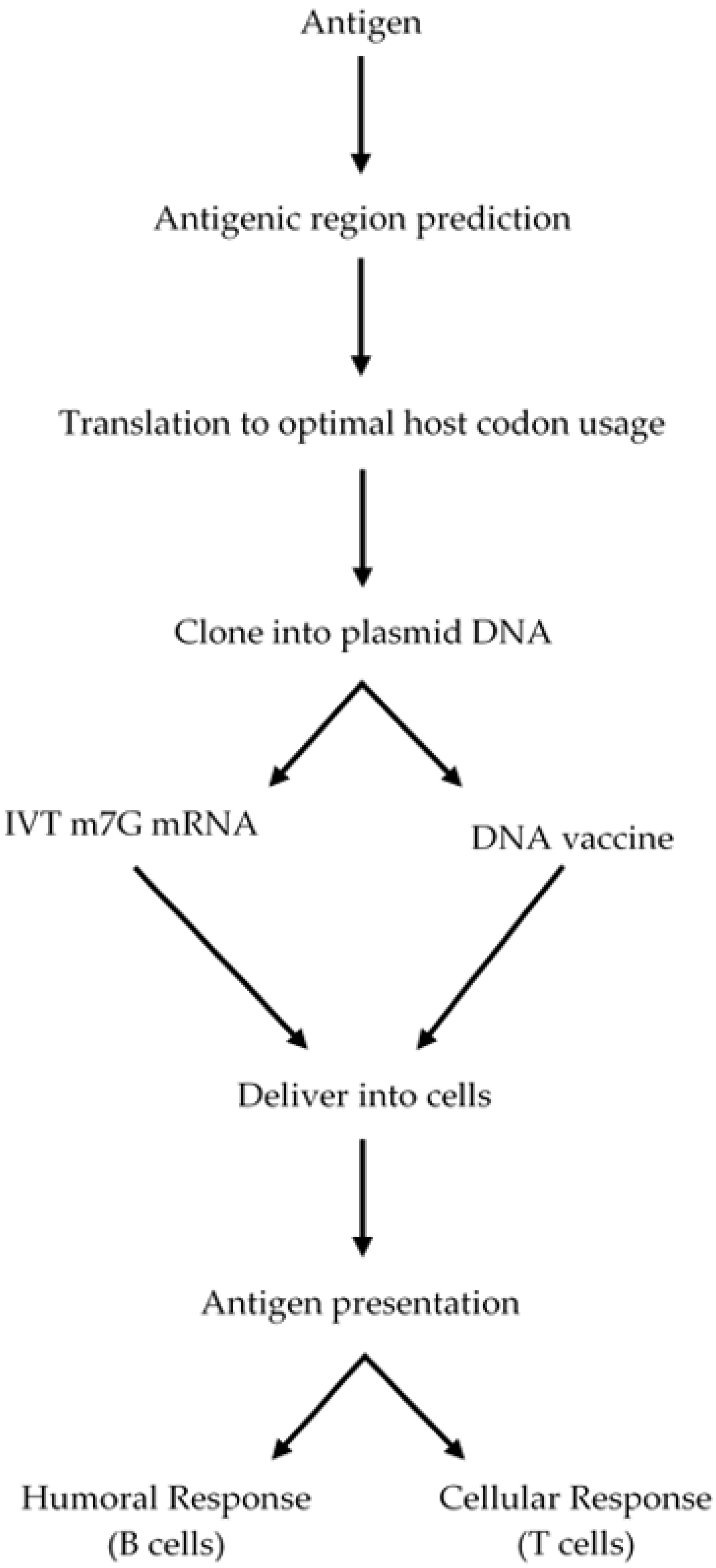
2. Vaccine Rationale
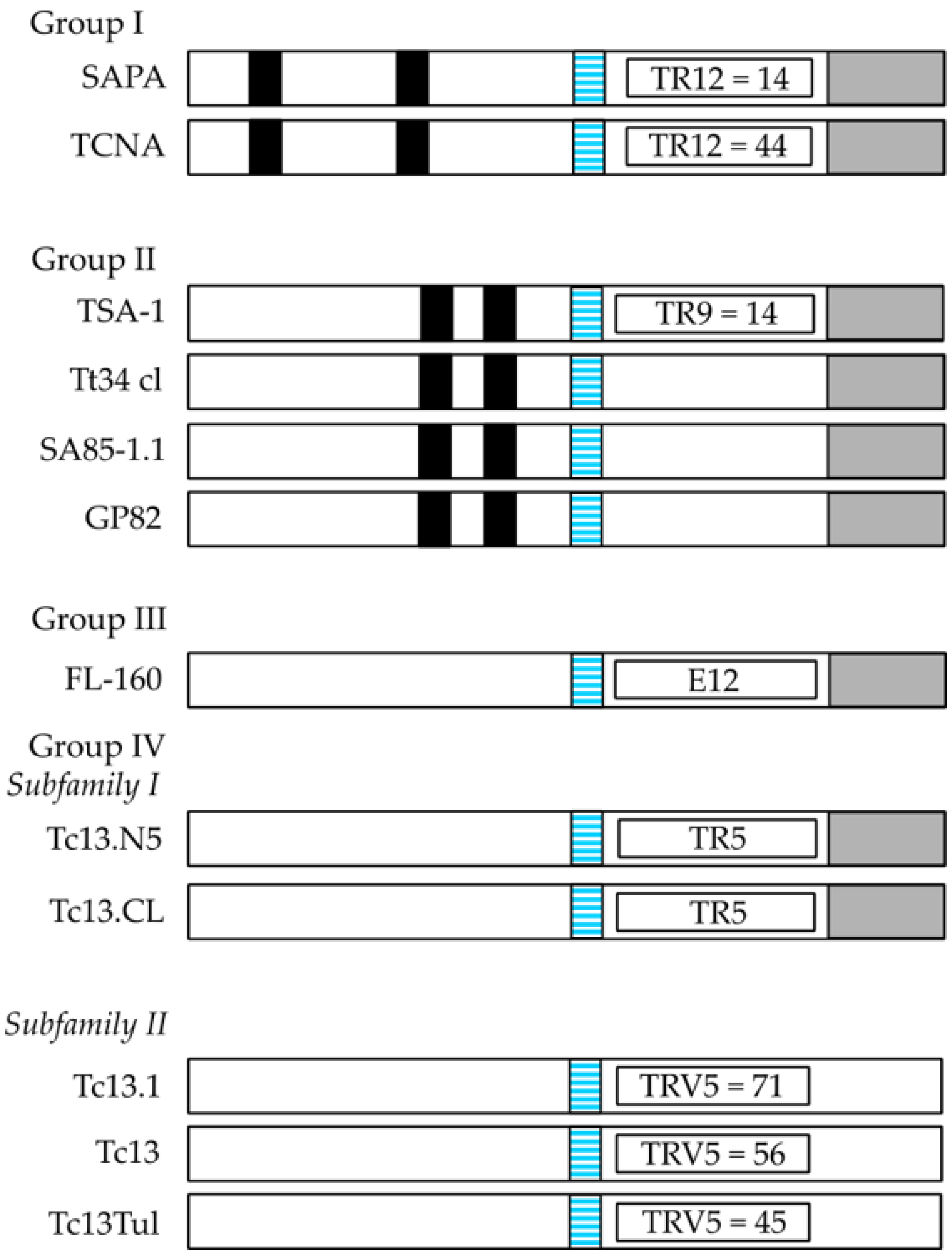
2.1. Trypanosoma Cruzi Surface Antigens
2.2. Most Likely Candidate Antigens for Vaccine Development
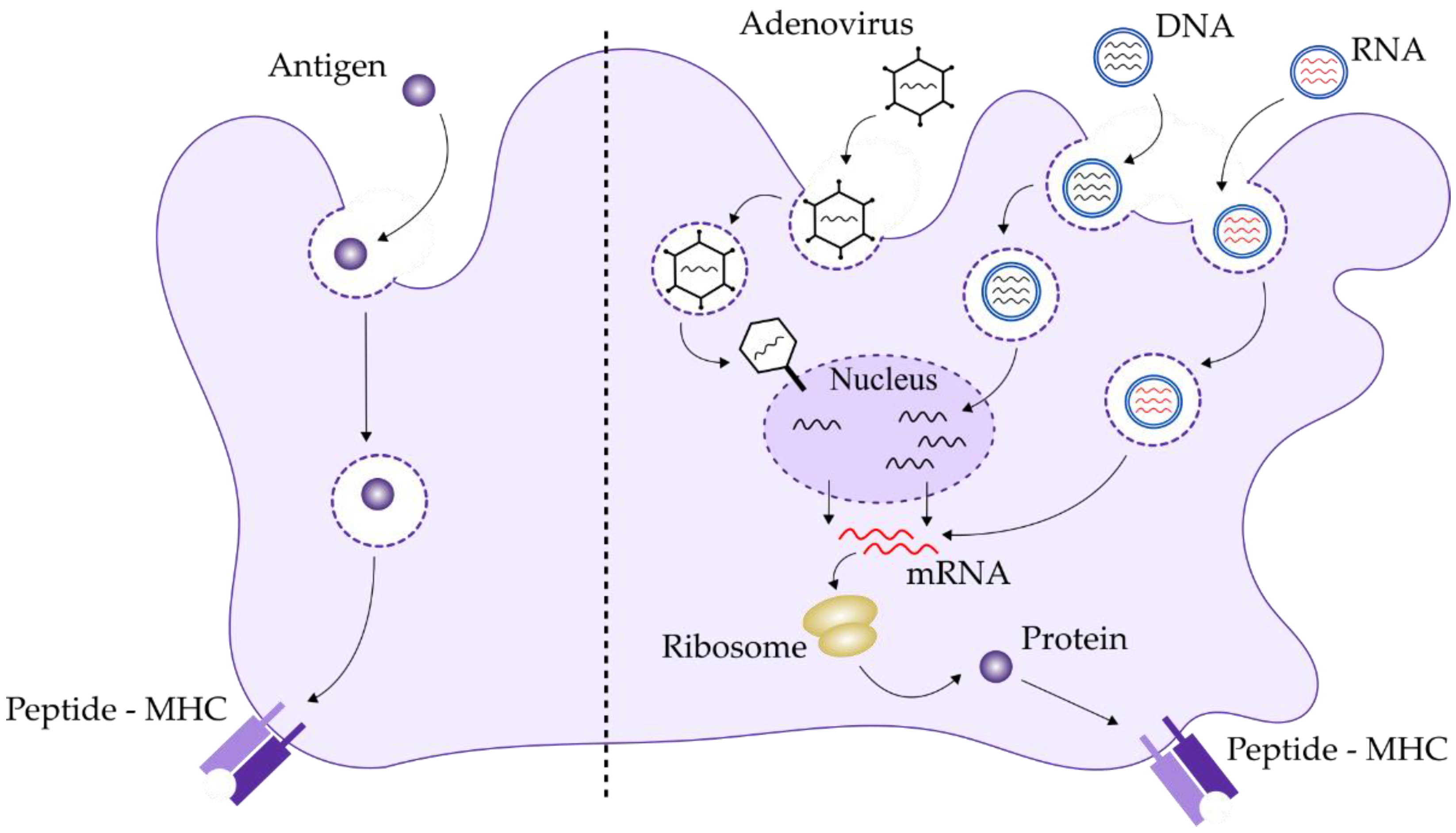
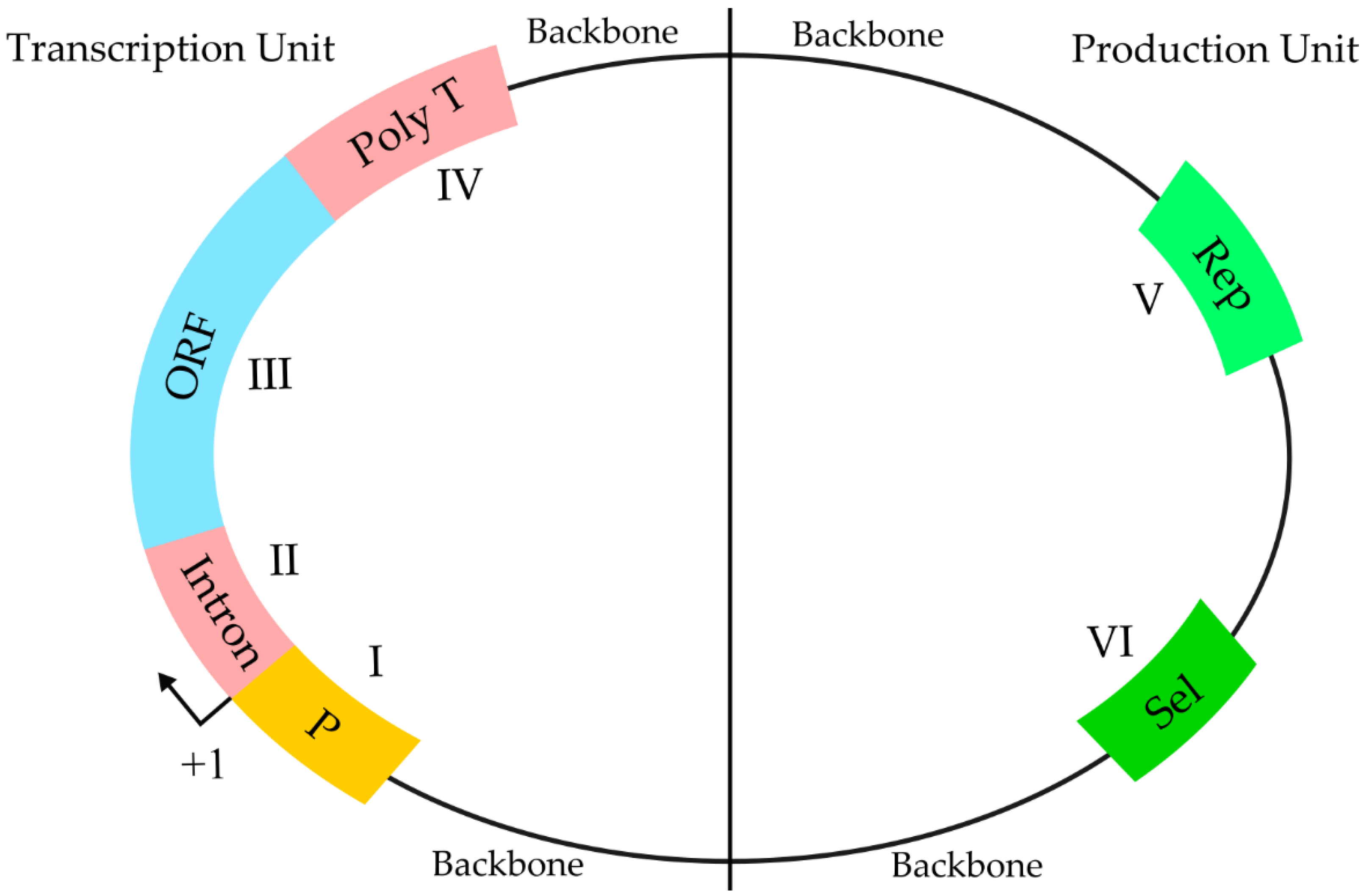
2.3. DNA-Based Vaccines
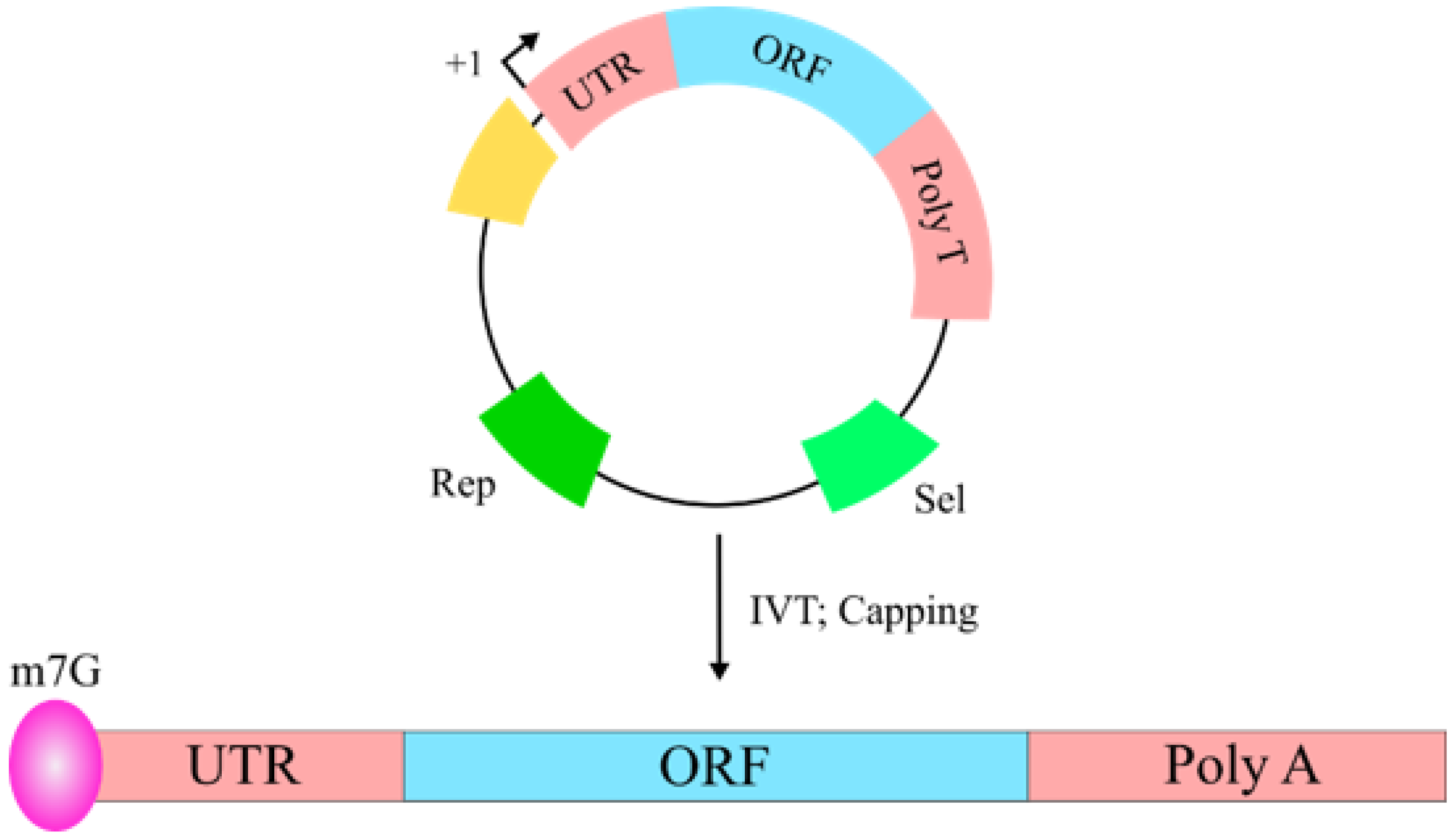
2.4. RNA-Based Vaccines
2.5. Live Attenuated Vaccines
2.6. T4 Bacteriophage Nanoparticles for Vaccine Delivery
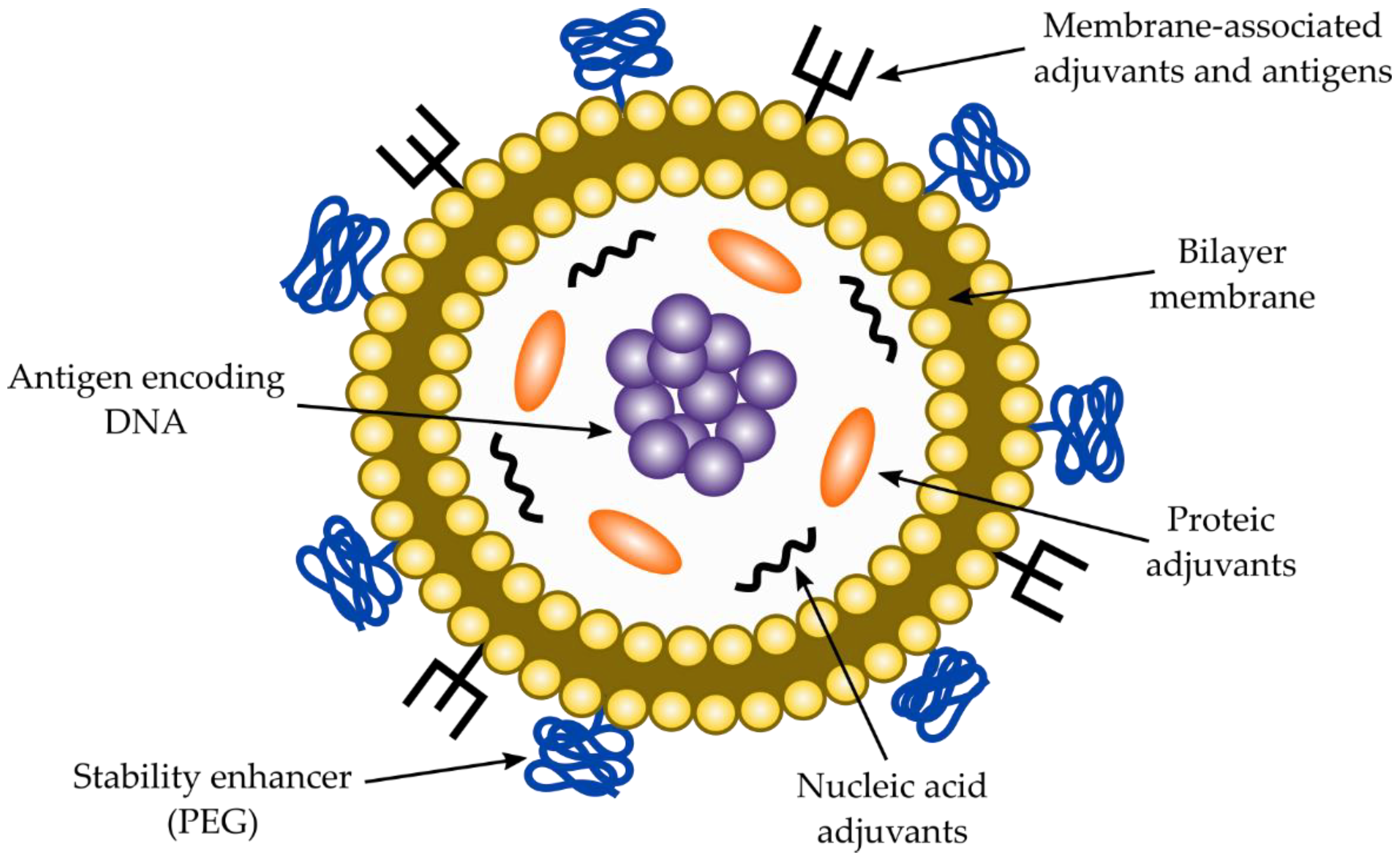
2.7. Co-Adjuvants
2.7.1. Co-Adjuvants for Protein Antigen-Based Vaccines
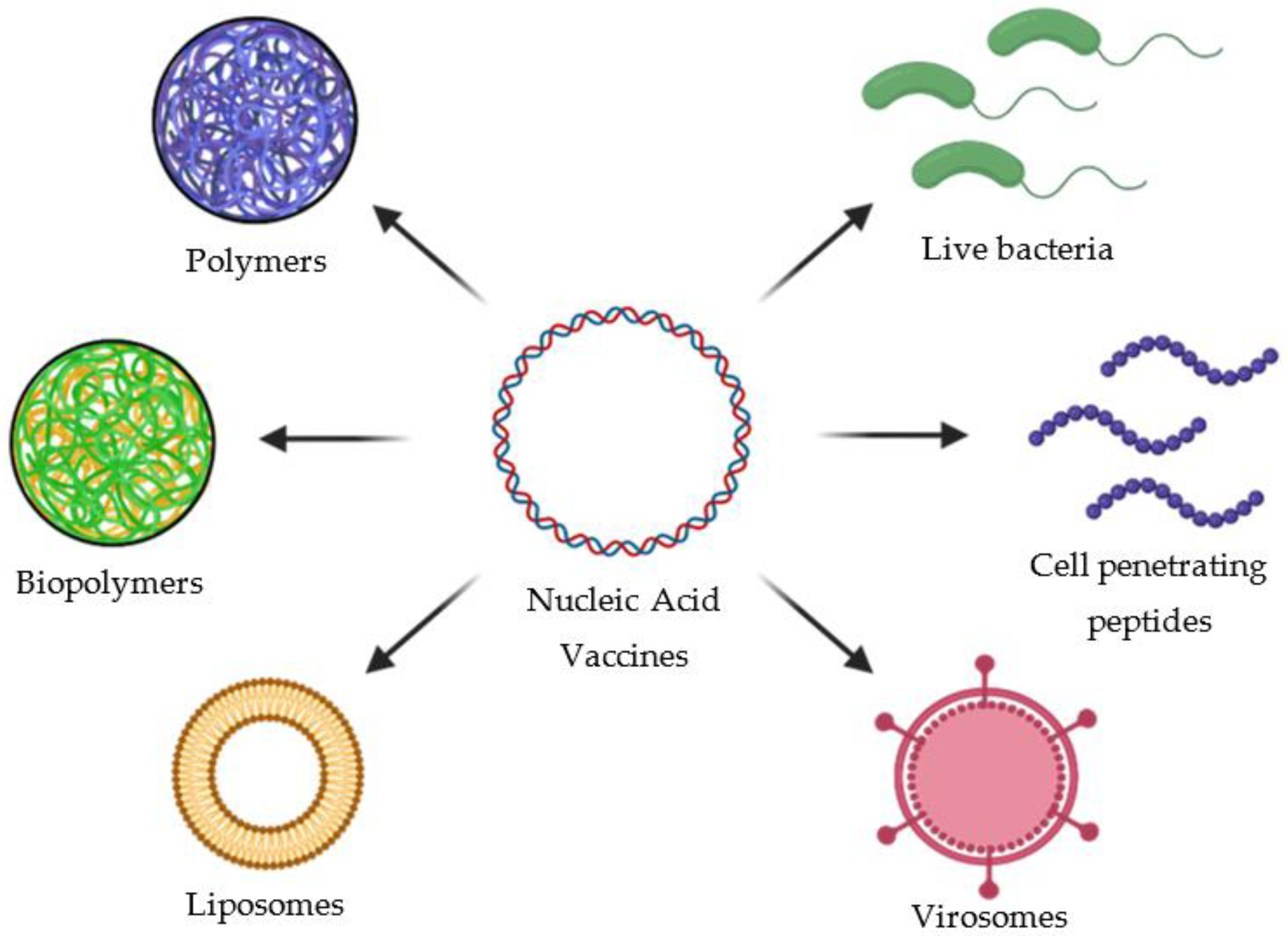
2.7.2. Co-Adjuvants for Nucleic Acid-Based Vaccines
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yeung, C.; Mendoza, I.; Echeverria, L.E.; Baranchuk, A. Chagas’ cardiomyopathy and Lyme carditis: Lessons learned from two infectious diseases affecting the heart. Trends Cardiovasc. Med. 2021, 31, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Antinori, S.; Galimberti, L.; Bianco, R.; Grande, R.; Galli, M.; Corbellino, M. Chagas disease in Europe: A review for the internist in the globalized world. Eur. J. Intern. Med. 2017, 43, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Molina, J.A.; Molina, I. Chagas disease. Lancet 2018, 391, 82–94. [Google Scholar] [CrossRef]
- Kourbeli, V.; Chontzopoulou, E.; Moschovou, K.; Pavlos, D.; Mavromoustakos, T.; Papanastasiou, I.P. An overview on target-based drug design against kinetoplastid protozoan infections: Human African trypanosomiasis, Chagas disease and leishmaniases. Molecules 2021, 26, 4629. [Google Scholar] [CrossRef]
- Vermelho, A.B.; Rodrigues, G.C.; Supuran, C.T. Why hasn’t there been more progress in new Chagas disease drug discovery? Expert Opin. Drug Discov. 2020, 15, 145–158. [Google Scholar] [CrossRef]
- Vekemans, J.; Hasso-Agopsowicz, M.; Kang, G.; Hausdorff, W.P.; Fiore, A.; Tayler, E.; Klemm, E.J.; Laxminarayan, R.; Srikantiah, P.; Friede, M.; et al. Leveraging vaccines to reduce antibiotic use and prevent antimicrobial resistance: A World Health Organization action framework. Clin. Infect. Dis. 2021, 73, e1011–e1017. [Google Scholar] [CrossRef]
- Bartsch, S.M.; Avelis, C.M.; Asti, L.; Hertenstein, D.L.; Ndeffo-Mbah, M.; Galvani, A.; Lee, B.Y. The economic value of identifying and treating Chagas disease patients earlier and the impact on Trypanosoma cruzi transmission. PLoS Negl. Trop. Dis. 2018, 12, e0006809. [Google Scholar] [CrossRef]
- Lee, B.Y.; Bartsch, S.M.; Skrip, L.; Hertenstein, D.L.; Avelis, C.M.; Ndeffo-Mbah, M.; Tilchin, C.; Dumonteil, E.O.; Galvani, A. Are the London Declaration’s 2020 goals sufficient to control Chagas disease?: Modeling scenarios for the Yucatan Peninsula. PLoS Negl. Trop. Dis. 2018, 12, e0006337. [Google Scholar] [CrossRef]
- Dumonteil, E.; Bottazzi, M.E.; Zhan, B.; Heffernan, M.J.; Jones, K.; Valenzuela, J.G.; Kamhawi, S.; Ortega, J.; Ponce de Leon Rosales, S.; Lee, B.Y.; et al. Accelerating the development of a therapeutic vaccine for human Chagas disease: Rationale and prospects. Expert Rev. Vaccines 2012, 11, 1043–1055. [Google Scholar] [CrossRef]
- Bottazzi, M.E. Vaccines against neglected tropical diseases: Promising interventions to rescue the poorest populations in the Americas. Immunotherapy 2014, 6, 117–119. [Google Scholar] [CrossRef]
- Camargo, E.P.; Gazzinelli, R.T.; Morel, C.M.; Precioso, A.R. Why do we still have not a vaccine against Chagas disease? Mem Inst Oswaldo Cruz Rio De Jan. 2021, 116. [Google Scholar] [CrossRef]
- Engman, D.M.; Dragon, E.A.; Donelson, J.E. Human humoral immunity to hsp70 during Trypanosoma cruzi infection. J. Immunol. 1990, 144, 3987–3991. [Google Scholar] [PubMed]
- Campetella, O.; Sánchez, D.; Cazzulo, J.J.; Frasch, A.C. A superfamily of Trypanosoma cruzi surface antigens. Parasitol. Today 1992, 8, 378–381. [Google Scholar] [CrossRef]
- De Marchi, C.R.; Di Noia, J.M.; Frasch, A.C.C.; Neto, V.A.; Almeida, I.C.; Buscaglia, C.A. Evaluation of a recombinant Trypanosoma cruzi mucin-like antigen for serodiagnosis of Chagas’ disease. Clin. Vaccine Immunol. 2011, 18, 1850–1855. [Google Scholar] [CrossRef] [PubMed]
- Buscaglia, C.A.; Campo, V.A.; Frasch, A.C.C.; Di Noia, J.M. Trypanosoma cruzi surface mucins: Host-dependent coat diversity. Nat. Rev. Microbiol. 2006, 4, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Cánepa, G.E.; Mesías, A.C.; Yu, H.; Chen, X.; Buscaglia, C.A. Structural features affecting trafficking, processing, and secretion of Trypanosoma cruzi mucins. J. Biol. Chem. 2012, 287, 26365–26376. [Google Scholar] [CrossRef]
- Pech-Canul, A.d.l.C.; Monteón, V.; Solís-Oviedo, R.L. A brief view of the surface membrane proteins from Trypanosoma cruzi. J. Parasitol. Res. 2017. [Google Scholar] [CrossRef]
- Barreto-Bergter, E.; Vermelho, A.B. Structures of glycolipids found in trypanosomatids: Contribution to parasite functions. Open Parasitol. J. 2010, 4, 84–97. [Google Scholar] [CrossRef]
- Cánepa, G.E.; Degese, M.S.; Budu, A.; Garcia, C.R.S.; Buscaglia, C.A. Involvement of TSSA (trypomastigote small surface antigen) in Trypanosoma cruzi invasion of mammalian cells. Biochem. J. 2012, 444, 211–218. [Google Scholar] [CrossRef]
- Nakayasu, E.S.; Yashunsky, D.V.; Nohara, L.L.; Torrecilhas, A.C.T.; Nikolaev, A.V.; Almeida, I.C. GPIomics: Global analysis of glycosylphosphatidylinositol-anchored molecules of Trypanosoma cruzi. Mol. Syst. Biol. 2009, 5, 261. [Google Scholar] [CrossRef]
- Urban, I.; Santurio, L.B.; Chidichimo, A.; Yu, H.; Chen, X.; Mucci, J.; Agüero, F.; Buscaglia, C.A. Molecular diversity of the Trypanosoma cruzi TcSMUG family of mucin genes and proteins. Biochem. J. 2011, 438, 303–313. [Google Scholar] [CrossRef] [PubMed]
- De Pablos, L.M.; Osuna, A. Conserved regions as markers of different patterns of expression and distribution of the mucin-associated surface proteins of Trypanosoma cruzi. Infect. Immun. 2012, 80, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Schenkman, S.; Jiang, M.S.; Hart, G.W.; Nussenzweig, V. A novel cell surface trans-sialidase of Trypanosoma cruzi generates a stage-specific epitope required for invasion of mammalian cells. Cell 1991, 65, 1117–1125. [Google Scholar] [CrossRef]
- El-Sayed, N.M.; Myler, P.J.; Bartholomeu, D.C.; Nilsson, D.; Aggarwal, G.; Tran, A.-N.; Ghedin, E.; Worthey, E.A.; Delcher, A.L.; Blandin, G.; et al. The genome sequence of Trypanosoma cruzi, etiologic agent of Chagas disease. Science 2005, 309, 409–415. [Google Scholar] [CrossRef]
- Frasch, A.C.C. Functional diversity in the trans-sialidase and mucin families in Trypanosoma cruzi. Parasitol. Today 2000, 16, 282–286. [Google Scholar] [CrossRef]
- Freitas, L.M.; dos Santos, S.L.; Rodrigues-Luiz, G.F.; Mendes, T.A.O.; Rodrigues, T.S.; Gazzinelli, R.T.; Teixeira, S.M.R.; Fujiwara, R.T.; Bartholomeu, D.C. Genomic analyses, gene expression and antigenic profile of the trans-sialidase superfamily of Trypanosoma cruzi reveal an undetected level of complexity. PLoS ONE 2011, 6, e25914. [Google Scholar] [CrossRef]
- Chiurillo, M.A.; Cortez, D.R.; Lima, F.M.; Cortez, C.; Ramírez, J.L.; Martins, A.G.; Serrano, M.G.; Teixeira, M.M.G.; da Silveira, J.F. The diversity and expansion of the trans-sialidase gene family is a common feature in Trypanosoma cruzi clade members. Infect. Genet. Evol. 2016, 37, 266–274. [Google Scholar] [CrossRef]
- Dc-Rubin, S.S.C.; Schenkman, S. Trypanosoma cruzi trans-sialidase as a multifunctional enzyme in Chagas’ disease. Cell. Microbiol. 2012, 14, 1522–1530. [Google Scholar] [CrossRef]
- Freire-de-Lima, L.; Fonseca, L.M.; Oeltmann, T.; Mendoca-Previato, L.; Previato, J.O. The trans-sialidase, the major Trypanosoma cruzi virulence factor: Three decades of studies. Glycobiology 2015, 25, 1142–1149. [Google Scholar] [CrossRef]
- Lantos, A.B.; Carlevaro, G.; Araoz, B.; Diaz, P.R.; de los Milagros Camara, M.; Buscaglia, C.A.; Bossi, M.; Yu, H.; Chen, X.; Bertozzi, C.R.; et al. Sialic acid glycobiology unveils Trypanosoma cruzi trypomastigote membrane physiology. PLoS Pathog. 2016, 12, e1005559. [Google Scholar] [CrossRef]
- Bivona, A.E.; Alberti, A.S.; Cerny, N.; Trinitario, S.N.; Malchiodi, E.L. Chagas disease vaccine design: The search for an efficient Trypanosoma cruzi immune-mediated control. Biochim. Et Biophys. Acta (BBA)-Mol. Basis Dis. 2020, 1866, 165658. [Google Scholar] [CrossRef] [PubMed]
- Schenkman, S.; Eichinger, D.; Pereira, M.E.; Nussenzweig, V. Structural and functional properties of Trypanosoma trans-sialidase. Annu. Rev. Microbiol. 1994, 48, 499–523. [Google Scholar] [CrossRef] [PubMed]
- Bayer-Santos, E.; Aguilar-Bonavides, C.; Rodrigues, S.P.; Cordero, E.M.; Marques, A.F.; Varela-Ramirez, A.; Choi, H.; Yoshida, N.; Silveira, J.F.; Almeida, I.C. Proteomic analysis of Trypanosoma cruzi secretome: Characterization of two populations of extracellular vesicles and soluble proteins. J. Proteome Res. 2013, 12, 883–897. [Google Scholar] [CrossRef] [PubMed]
- Reyes, M.B.; Lorca, M.; Muñoz, P.; Frasch, A.C. Fetal IgG specificities against Trypanosoma cruzi antigens in infected newborns. Proc. Natl. Acad. Sci. USA 1990, 87, 2846–2850. [Google Scholar] [CrossRef] [PubMed]
- Prioli, R.P.; Ortega-Barria, E.; Mejia, J.S.; Pereira, M.E. Mapping of a B-cell epitope present in the neuraminidase of Trypanosoma cruzi. Mol. Biochem. Parasitol. 1992, 52, 85–96. [Google Scholar] [CrossRef]
- Leguizamón, M.S.; Campetella, O.E.; Gonzalez-Cappa, S.M.; Frasch, A.C. Mice infected with Trypanosoma cruzi produce antibodies against the enzymatic domain of trans-sialidase that inhibit its activity. Infect. Immun. 1994, 62, 3441–3446. [Google Scholar] [CrossRef]
- Prioli, R.P.; Rosenberg, I.; Pereira, M.E. High-and low-density lipoproteins enhance infection of Trypanosoma cruzi in vitro. Mol. Biochem. Parasitol. 1990, 38, 191–198. [Google Scholar] [CrossRef]
- Prioli, R.P.; Rosenberg, I.; Pereira, M.E. Specific inhibition of Trypanosoma cruzi neuraminidase by the human plasma glycoprotein “cruzin”. Proc. Natl. Acad. Sci. USA 1987, 84, 3097–3101. [Google Scholar] [CrossRef]
- Prioli, R.P.; Ordovas, J.M.; Rosenberg, I.; Schaefer, E.J.; Pereira, M.E. Similarity of cruzin, an inhibitor of Trypanosoma cruzi neuraminidase, to high-density lipoprotein. Science 1987, 238, 1417–1419. [Google Scholar] [CrossRef]
- Briones, M.R.; Egima, C.M.; Schenkman, S. Trypanosoma cruzi trans-sialidase gene lacking C-terminal repeats and expressed in epimastigote forms. Mol. Biochem. Parasitol. 1995, 70, 9–17. [Google Scholar] [CrossRef]
- Rubin-De-Celis, S.S.C.; Uemura, H.; Yoshida, N.; Schenkman, S. Expression of trypomastigote trans-sialidase in metacyclic forms of Trypanosoma cruzi increases parasite escape from its parasitophorous vacuole. Cell. Microbiol. 2006, 8, 1888–1898. [Google Scholar] [CrossRef] [PubMed]
- Magdesian, M.H.; Giordano, R.; Ulrich, H.; Juliano, M.A.; Juliano, L.; Schumacher, R.I.; Colli, W.; Alves, M.J. Infection by Trypanosoma cruzi: Identification of a parasite ligand and its host cell receptor. J. Biol. Chem. 2001, 276, 19382–19389. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, R.R.; Giordano, R.J.; Barbu, E.M.; Torrecilhas, A.C.; Kobayashi, G.S.; Langley, R.R.; Arap, W.; Pasqualina, R.; Colli, W.; Alves, M.J.M. Role of the gp85/trans-sialidases in Trypanosoma cruzi tissue tropism: Preferential binding of a conserved peptide motif to the vasculature in vivo. PLoS Negl. Trop. Dis. 2010, 4, e864. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, A.A.R.; de Vasconcelos, V.d.C.S.; Colli, W.; Alves, M.J.M.; Giordano, R.J. Trypanosoma cruzi binds to cytokeratin through conserved peptide motifs found in the laminin-G-like domain of the gp85/trans-sialidase proteins. PLoS Negl. Trop. Dis. 2015, 9, e0004099. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.; Wleklinski, M.; Aruffo, A.; Farr, A.; Coder, D.; Kahn, M. Trypanosoma cruzi amastigote adhesion to macrophages is facilitated by the mannose receptor. J. Exp. Med. 1995, 182, 1243–1258. [Google Scholar] [CrossRef]
- Staquicini, D.I.; Martins, R.M.; Macedo, S.; Sasso, G.R.S.; Atayde, V.D.; Juliano, M.A.; Yoshida, N. Role of GP82 in the selective binding to gastric mucin during oral infection with Trypanosoma cruzi. PLoS Negl. Trop. Dis. 2010, 4, e613. [Google Scholar] [CrossRef]
- Kipnis, T.L.; David, J.R.; Alper, C.A.; Sher, A.; da Silva, W.D. Enzymatic treatment transforms trypomastigotes of Trypanosoma cruzi into activators of alternative complement pathway and potentiates their uptake by macrophages. Proc. Natl. Acad. Sci. USA 1981, 78, 602–605. [Google Scholar] [CrossRef]
- Beucher, M.; Norris, K.A. Sequence diversity of the Trypanosoma cruzi complement regulatory protein family. Infect. Immun. 2008, 76, 750–758. [Google Scholar] [CrossRef]
- Matsumoto, T.K.; Cotrim, P.C.; da Silveira, J.F.; Stolf, A.M.S.; Umezawa, E.S. Trypanosoma cruzi: Isolation of an immunodominant peptide of TESA (trypomastigote excreted-secreted antigens) by gene cloning. Diagn. Microbiol. Infect. Dis. 2002, 42, 187–192. [Google Scholar] [CrossRef]
- Berrizbeitia, M.; Ndao, M.; Bubis, J.; Gottschalk, M.; Aché, A.; Lacouture, S.; Medina, M.; Ward, B.J. Purified excreted-secreted antigens from Trypanosoma cruzi trypomastigotes as tools for diagnosis of Chagas’ disease. J. Clin. Microbiol. 2006, 44, 291–296. [Google Scholar] [CrossRef]
- García, E.A.; Ziliani, M.; Agüero, F.; Bernabó, G.; Sánchez, D.O.; Tekiel, V. TcTASV: A novel protein family in Trypanosoma cruzi identified from a subtractive trypomastigote cDNA library. PLoS Negl. Trop. Dis. 2010, 4, e841. [Google Scholar] [CrossRef] [PubMed]
- Floridia-Yapur, N.; Monje-Rumi, M.; Ragone, P.; Lauthier, J.J.; Tomasini, N.; D’Amato, A.A.; Diosque, P.; Cimino, R.; Gil, J.F.; Sanchez, D.O.; et al. TcTASV antigens of Trypanosoma cruzi: Utility for diagnosis and high accuracy as biomarkers of treatment efficacy in pediatric patients. Am. J. Trop. Med. Hyg. 2019, 101, 1135. [Google Scholar] [CrossRef] [PubMed]
- Caeiro, L.D.; Alba-Soto, C.D.; Rizzi, M.; Solana, M.E.; Rodriguez, G.; Chidichimo, A.M.; Rodriguez, M.E.; Sánchez, D.O.; Levy, G.V.; Tekiel, V. The protein family TcTASV-C is a novel Trypanosoma cruzi virulence factor secreted in extracellular vesicles by trypomastigotes and highly expressed in bloodstream forms. PLoS Negl. Trop. Dis. 2018, 12, e0006475. [Google Scholar] [CrossRef] [PubMed]
- Atwood, J.A., 3rd; Weatherly, D.B.; Minning, T.A.; Bundy, B.; Cavola, C.; Opperdoes, F.R.; Orlando, R.; Tarleton, R.L. The Trypanosoma cruzi proteome. Science 2005, 309, 473–476. [Google Scholar] [CrossRef]
- Ouaissi, M.A.; Dubremetz, J.F.; Schöneck, R.; Fernandez-Gomez, R.; Gomez-Corvera, R.; Billaut-Mulot, O.; Taibi, A.; Loyens, M.; Tartar, A.; Sergheraert, C. Trypanosoma cruzi: A 52-kDa protein sharing sequence homology with glutathione S-transferase is localized in parasite organelles morphologically resembling reservosomes. Exp. Parasitol. 1995, 81, 453–461. [Google Scholar] [CrossRef]
- Allaoui, A.; François, C.; Zemzoumi, K.; Guilvard, E.; Ouaissi, A. Intracellular growth and metacyclogenesis defects in Trypanosoma cruzi carrying a targeted deletion of a Tc52 protein-encoding allele. Mol. Microbiol. 1999, 32, 1273–1286. [Google Scholar] [CrossRef]
- Schöneck, R.; Plumas-Marty, B.; Taibi, A.; Billaut-Mulot, O.; Loyens, M.; Gras-Masse, H.; Capron, A.; Ouaissi, A. Trypanosoma cruzi cDNA encodes a tandemly repeated domain structure characteristic of small stress proteins and glutathione S-transferases. Biol. Cell 1994, 80, 1–10. [Google Scholar] [CrossRef]
- Matos, M.N.; Cazorla, S.I.; Bivona, A.E.; Morales, C.; Guzmán, C.A.; Malchiodi, E.L. Tc52 amino-terminal-domain DNA carried by attenuated Salmonella enterica serovar Typhimurium induces protection against a Trypanosoma cruzi lethal challenge. Infect. Immun. 2014, 82, 4265–4275. [Google Scholar] [CrossRef]
- Matos, M.N.; Alberti, A.S.; Morales, C.; Cazorla, S.I.; Malchiodi, E.L. A prime-boost immunization with Tc52 N-terminal domain DNA and the recombinant protein expressed in Pichia pastoris protects against Trypanosoma cruzi infection. Vaccine 2016, 34, 3243–3251. [Google Scholar] [CrossRef]
- Matos, M.N.; Cazorla, S.I.; Schulze, K.; Ebensen, T.; Guzmán, C.A.; Malchiodi, E.L. Immunization with Tc52 or its amino terminal domain adjuvanted with c-di-AMP induces Th17+ Th1 specific immune responses and confers protection against Trypanosoma cruzi. PLoS Negl. Trop. Dis. 2017, 11, e0005300. [Google Scholar] [CrossRef]
- Santana, J.M.; Grellier, P.; Schrével, J.; Teixeira, A.R. A Trypanosoma cruzi-secreted 80 kDa proteinase with specificity for human collagen types I and IV. Biochem. J. 1997, 325, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Bastos, I.M.D.; Grellier, P.; Martins, N.F.; Cadavid-Restrepo, G.; de Souza-Ault, M.R.; Augustyns, K.; Teixeira, A.R.L.; Schrével, J.; Maigret, B.; da Silveira, J.F.; et al. Molecular, functional and structural properties of the prolyl oligopeptidase of Trypanosoma cruzi (POP Tc80), which is required for parasite entry into mammalian cells. Biochem. J. 2005, 388, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Bivona, A.E.; Alberti, A.S.; Matos, M.N.; Cerny, N.; Cardoso, A.C.; Morales, C.; González, G.; Cazorla, S.I.; Malchiodi, E.L. Trypanosoma cruzi 80 kDa prolyl oligopeptidase (Tc80) as a novel immunogen for Chagas disease vaccine. PLoS Negl. Trop. Dis. 2018, 12, e0006384. [Google Scholar] [CrossRef] [PubMed]
- Campetella, O.; Henriksson, J.; Aslund, L.; Frasch, A.C.; Pettersson, U.; Cazzulo, J.J. The major cysteine proteinase (cruzipain) from Trypanosoma cruzi is encoded by multiple polymorphic tandemly organized genes located on different chromosomes. Mol. Biochem. Parasitol. 1992, 50, 225–234. [Google Scholar] [CrossRef]
- Beaulieu, C.; Isabel, E.; Fortier, A.; Massé, F.; Mellon, C.; Méthot, N.; Ndao, M.; Nicoll-Griffith, D.; Lee, D.; Park, H.; et al. Identification of potent and reversible cruzipain inhibitors for the treatment of Chagas disease. Bioorganic Med. Chem. Lett. 2010, 20, 7444–7449. [Google Scholar] [CrossRef]
- Duschak, V.G. Major kinds of drug targets in Chagas disease or American Trypanosomiasis. Curr. Drug Targets 2019, 20, 1203–1216. [Google Scholar] [CrossRef]
- Souto-Padron, T.; Campetella, O.E.; Cazzulo, J.J.; de Souza, W. Cysteine proteinase in Trypanosoma cruzi: Immunocytochemical localization and involvement in parasite-host cell interaction. J. Cell Sci. 1990, 96, 485–490. [Google Scholar] [CrossRef]
- Berasain, P.; Carmona, C.; Frangione, B.; Cazzulo, J.J.; Goñi, F. Specific cleavage sites on human IgG subclasses by cruzipain, the major cysteine proteinase from Trypanosoma cruzi. Mol. Biochem. Parasitol. 2003, 130, 23–29. [Google Scholar] [CrossRef]
- Duschak, V.G.; Riarte, A.; Segura, E.L.; Laucella, S.A. Humoral immune response to cruzipain and cardiac dysfunction in chronic Chagas disease. Immunol. Lett. 2001, 78, 135–142. [Google Scholar] [CrossRef]
- Giordanengo, L.; Guiñazú, N.; Stempin, C.; Fretes, R.; Cerban, F.; Gea, S. Cruzipain, a major Trypanosoma cruzi antigen, conditions the host immune response in favor of parasite. Eur. J. Immunol. 2002, 32, 1003–1011. [Google Scholar] [CrossRef]
- Santos, V.C.; Rocha-Oliveira, A.E.; Broilo-Campos, A.C.; Reis-Cunha, J.L.; Bartholomeu, D.C.; Teixeira, S.M.R.; Lima, A.P.C.A.; Ferreira, R.S. The gene repertoire of the main cysteine protease of Trypanosoma cruzi, cruzipain, reveals four sub-types with distinct active sites. Sci. Rep. 2021, 11, 18231. [Google Scholar] [CrossRef] [PubMed]
- Cazorla, S.I.; Frank, F.M.; Becker, P.D.; Arnaiz, M.; Mirkin, G.A.; Corral, R.S.; Guzmán, C.A.; Malchiodi, E.L. Redirection of the immune response to the functional catalytic domain of the cystein proteinase cruzipain improves protective immunity against Trypanosoma cruzi infection. J. Infect. Dis. 2010, 202, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Duschak, V.G.; Couto, A.S. Cruzipain, the major cysteine protease of Trypanosoma cruzi: A sulfated glycoprotein antigen as relevant candidate for vaccine development and drug target. A review. Curr. Med. Chem. 2009, 16, 3174–3202. [Google Scholar] [CrossRef]
- Cuevas, I.C.; Cazzulo, J.J.; Sánchez, D.O. gp63 homologues in Trypanosoma cruzi: Surface antigens with metalloprotease activity and a possible role in host cell infection. Infect. Immun. 2003, 71, 5739–5749. [Google Scholar] [CrossRef]
- Teixeira, S.M.; Russell, D.G.; Kirchhoff, L.V.; Donelson, J.E. A differentially expressed gene family encoding “amastin,” a surface protein of Trypanosoma cruzi amastigotes. J. Biol. Chem. 1994, 269, 20509. [Google Scholar] [CrossRef]
- Cruz, M.C.; Souza-Melo, N.; da Silva, C.V.; DaRocha, W.D.; Bahia, D.; Araújo, P.R.; Teixeira, S.R.; Mortara, R.A. Trypanosoma cruzi: Role of δ-amastin on extracellular amastigote cell invasion and differentiation. PLoS ONE 2012, 7, e51804. [Google Scholar] [CrossRef]
- Gunter, S.M.; Jones, K.M.; Zhan, B.; Essigmann, H.T.; Murray, K.O.; Garcia, M.N.; Gorchakov, R.; Bottazzi, M.E.; Hotez, P.J.; Brown, E.L. Identification and characterization of the Trypanosoma cruzi B-cell superantigen Tc24. Am. J. Trop. Med. Hyg. 2016, 94, 114. [Google Scholar] [CrossRef]
- Arnal, A.; Villanueva-Lizama, L.; Teh-Poot, C.; Herrera, C.; Dumonteil, E. Extent of polymorphism and selection pressure on the Trypanosoma cruzi vaccine candidate antigen Tc24. Evol. Appl. 2020, 13, 2663–2672. [Google Scholar] [CrossRef]
- Versteeg, L.; Adhikari, R.; Poveda, C.; Villar-Mondragon, M.J.; Jones, K.M.; Hotez, P.J.; Bottazzi, M.E.; Tijhaar, E.; Pollet, J. Location and expression kinetics of Tc24 in different life stages of Trypanosoma cruzi. PLoS Negl. Trop. Dis. 2021, 15, e0009689. [Google Scholar] [CrossRef]
- Gunter, S.M.; Versteeg, L.; Jones, K.M.; Keegan, B.P.; Strych, U.; Bottazzi, M.E.; Hotez, P.J.; Brown, E.L. Covalent vaccination with Trypanosoma cruzi Tc24 induces catalytic antibody production. Parasite Immunol. 2018, 40, e12585. [Google Scholar] [CrossRef]
- Rappuoli, R. Reverse vaccinology. Curr. Opin. Microbiol. 2000, 3, 445–450. [Google Scholar] [CrossRef]
- Moxon, R.; Reche, P.A.; Rappuoli, R. Reverse vaccinology. Front. Immunol. 2019, 10, 2776. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, V.; Sinha, M.; Luxon, B.; Garg, N. Utility of the Trypanosoma cruzi sequence database for identification of potential vaccine candidates by in silico and in vitro screening. Infect. Immun. 2004, 72, 6245–6254. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bhatia, V.; Garg, N.J. Previously unrecognized vaccine candidates control Trypanosoma cruzi infection and immunopathology in mice. Clin. Vaccine Immunol. 2008, 15, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Garg, N.J. A two-component DNA-prime/protein-boost vaccination strategy for eliciting long-term, protective T cell immunity against Trypanosoma cruzi. PLoS Pathog. 2015, 11, e1004828. [Google Scholar] [CrossRef] [PubMed]
- das Neves, R.F.C.; Adade, C.M.; Fernandes, A.C.S.; Lopes, A.H.; Souto-Padrón, T. Trypanosoma cruzi: Cell surface dynamics in trypomastigotes of different strains. Parasitology 2020, 147, 310–321. [Google Scholar] [CrossRef]
- Rodrigues da Cunha, G.M.; Azevedo, M.A.; Nogueira, D.S.; de Carvalho Clímaco, M.; Ayala, E.V.; Chunga, J.A.J.; La Valle, R.J.Y.; da Cunha Galvão, L.M.; Chiari, E.; Brito, C.R.N.; et al. α-Gal immunization positively impacts Trypanosoma cruzi colonization of heart tissue in a mouse model. PLoS Negl. Trop. Dis. 2021, 15, e0009613. [Google Scholar] [CrossRef]
- Alberti, A.S.; Bivona, A.E.; Cerny, N.; Schulze, K.; Weißmann, S.; Ebensen, T.; Morales, C.; Padilla, A.M.; Cazorla, S.I.; Tarleton, R.L.; et al. Engineered trivalent immunogen adjuvanted with a STING agonist confers protection against Trypanosoma cruzi infection. NPJ Vaccines 2017, 2, 9. [Google Scholar] [CrossRef]
- Pereira, I.R.; Vilar-Pereira, G.; Marques, V.; da Silva, A.A.; Caetano, B.; Moreira, O.C.; Machado, A.V.; Bruna-Romero, O.; Rodrigues, M.M.; Gazzinelli, R.T. A human type 5 adenovirus-based Trypanosoma cruzi therapeutic vaccine re-programs immune response and reverses chronic cardiomyopathy. PLoS Pathog. 2015, 11, e1004594. [Google Scholar] [CrossRef]
- Lokugamage, N.; Choudhuri, S.; Davies, C.; Chowdhury, I.H.; Garg, N.J. Antigen-based nano-immunotherapy controls parasite persistence, inflammatory and oxidative stress, and cardiac fibrosis, the hallmarks of chronic Chagas cardiomyopathy, in a mouse model of Trypanosoma cruzi infection. Vaccines 2020, 8, 96. [Google Scholar] [CrossRef]
- Bontempi, I.; Leal, K.; Prochetto, E.; Díaz, G.; Cabrera, G.; Bortolotti, H.R.; Borsuk, S.; Dellagostin, O.A.; Marcipar, I. Recombinant Mycobacterium bovis BCG is a promising platform to develop vaccines against Trypansoma cruzi infection. Clin. Exp. Immunol. 2020, 201, 306–316. [Google Scholar] [CrossRef] [PubMed]
- Tenney, T.D.; Curtis-Robles, R.; Snowden, K.F.; Hamer, S.A. Shelter dogs as sentinels for Trypanosoma cruzi transmission across Texas. Emerg. Infect. Dis. 2014, 20, 1323. [Google Scholar] [CrossRef] [PubMed]
- Cazorla, S.I.; Matos, M.N.; Cerny, N.; Ramirez, C.; Alberti, A.S.; Bivona, A.E.; Morales, C.; Guzmán, C.A.; Malchiodi, E.L. Oral multicomponent DNA vaccine delivered by attenuated Salmonella elicited immunoprotection against American trypanosomiasis. J. Infect. Dis. 2015, 211, 698–707. [Google Scholar] [CrossRef] [PubMed]
- Barry, M.A.; Versteeg, L.; Wang, Q.; Pollet, J.; Zhan, B.; Gusovsky, F.; Bottazzi, M.E.; Hotez, P.J.; Jones, K.M. A therapeutic vaccine prototype induces protective immunity and reduces cardiac fibrosis in a mouse model of chronic Trypanosoma cruzi infection. PLoS Negl. Trop. Dis. 2019, 13, e0007413. [Google Scholar] [CrossRef]
- Villanueva-Lizama, L.E.; Cruz-Chan, J.V.; Aguilar-Cetina, A.D.C.; Herrera-Sanchez, L.F.; Rodriguez-Perez, J.M.; Rosado-Vallado, M.E.; Ramirez-Sierra, M.J.; Ortega-Lopez, J.; Jones, K.; Hotez, P.; et al. Trypanosoma cruzi vaccine candidate antigens Tc24 and TSA-1 recall memory immune response associated with HLA-A and-B supertypes in Chagasic chronic patients from Mexico. PLoS Negl. Trop. Dis. 2018, 12, e0006240. [Google Scholar] [CrossRef]
- Liu, M.A. DNA vaccines: A review. J. Intern. Med. 2003, 253, 402–410. [Google Scholar] [CrossRef]
- Franck, C.O.; Fanslau, L.; Popov, A.B.; Tyagi, P.; Fruk, L. Biopolymer-based Carriers for DNA Vaccine Design. Angew. Chem. 2021, 133, 13333–13351. [Google Scholar] [CrossRef]
- Shafaati, M.; Saidijam, M.; Soleimani, M.; Hazrati, F.; Mirzaei, R.; Amirheidari, B.; Tanzadehpanah, H.; Karampoor, S.; Kazemi, S.; Yavari, B.; et al. A brief review on DNA vaccines in the era of COVID-19. Future Virol. 2022, 17, 49–66. [Google Scholar] [CrossRef]
- Krinner, S.; Heitzer, A.; Asbach, B.; Wagner, R. Interplay of promoter usage and intragenic CpG content: Impact on GFP reporter gene expression. Hum. Gene Ther. 2015, 26, 826–840. [Google Scholar] [CrossRef]
- Juven-Gershon, T.; Cheng, S.; Kadonaga, J.T. Rational design of a super core promoter that enhances gene expression. Nat. Methods 2006, 3, 917–922. [Google Scholar] [CrossRef]
- Pardi, N.; Hogan, M.J.; Porter, F.W.; Weissman, D. mRNA vaccines—a new era in vaccinology. Nat. Rev. Drug Discov. 2018, 17, 261–279. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, N.; Weissman, D.; Whitehead, K.A. mRNA vaccines for infectious diseases: Principles, delivery and clinical translation. Nat. Rev. Drug Discov. 2021, 20, 817–838. [Google Scholar] [CrossRef] [PubMed]
- Balmayor, E.R. Synthetic mRNA–emerging new class of drug for tissue regeneration. Curr. Opin. Biotechnol. 2022, 74, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Krammer, F. SARS-CoV-2 vaccines in development. Nature 2020, 586, 516–527. [Google Scholar] [CrossRef]
- Ross, J. Control of messenger RNA stability in higher eukaryotes. Trends Genet. 1996, 12, 171–175. [Google Scholar] [CrossRef]
- Grudzien, E.W.A.; Stepinski, J.; Jankowska-Anyszka, M.; Stolarski, R.; Darzynkiewicz, E.; Rhoads, R.E. Novel cap analogs for in vitro synthesis of mRNAs with high translational efficiency. RNA 2004, 10, 1479–1487. [Google Scholar] [CrossRef]
- Maruggi, G.; Zhang, C.; Li, J.; Ulmer, J.B.; Yu, D. mRNA as a transformative technology for vaccine development to control infectious diseases. Mol. Ther. 2019, 27, 757–772. [Google Scholar] [CrossRef]
- Urbina, F.; Morales-Pison, S.; Maldonado, E. Enzymatic protein biopolymers as a tool to synthetize eukaryotic messenger ribonucleic acid (mRNA) with uses in vaccination, immunotherapy and nanotechnology. Polymers 2020, 12, 1633. [Google Scholar] [CrossRef]
- Sánchez-Valdéz, F.J.; Brandán, C.P.; Ferreira, A.; Basombrío, M.A. Gene-deleted live-attenuated Trypanosoma cruzi parasites as vaccines to protect against Chagas disease. Expert Rev. Vaccines 2015, 14, 681–697. [Google Scholar] [CrossRef]
- Solana, J.C.; Moreno, J.; Iborra, S.; Soto, M.; Requena, J.M. Live attenuated vaccines, a favorable strategy to provide long-term immunity against protozoan diseases. Trends Parasitol. 2021, 38, 316–334. [Google Scholar] [CrossRef]
- Rodríguez, E.V.A.; Furlan, C.L.A.; Vernengo, F.F.; Montes, C.L.; Gruppi, A. Understanding CD8+ T cell immunity to Trypanosoma cruzi and how to improve it. Trends Parasitol. 2019, 35, 899–917. [Google Scholar] [CrossRef] [PubMed]
- Basombrío, M.A.; Besuschio, S. Trypanosoma cruzi culture used as vaccine to prevent chronic Chagas’ disease in mice. Infect. Immun. 1982, 36, 351–356. [Google Scholar] [CrossRef]
- Basombrío, M.A.; Segura, M.A.; Mora, M.C.; Gomez, L. Field trial of vaccination against American trypanosomiasis (Chagas’ disease) in dogs. Am. J. Trop. Med. Hyg. 1993, 49, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Barrio, A.B.; Van Voorhis, W.C.; Basombrío, M.A. Trypanosoma cruzi: Attenuation of virulence and protective immunogenicity after monoallelic disruption of the cub gene. Exp. Parasitol. 2007, 117, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Zago, M.P.; Barrio, A.B.; Cardozo, R.M.; Duffy, T.; Schijman, A.G.; Basombrío, M.A. Impairment of infectivity and immunoprotective effect of a LYT1 null mutant of Trypanosoma cruzi. Infect. Immun. 2008, 76, 443–451. [Google Scholar] [CrossRef]
- Tao, P.; Mahalingam, M.; Marasa, B.S.; Zhang, Z.; Chopra, A.K.; Rao, V.B. In vitro and in vivo delivery of genes and proteins using the bacteriophage T4 DNA packaging machine. Proc. Natl. Acad. Sci. USA 2013, 110, 5846–5851. [Google Scholar] [CrossRef]
- Tao, P.; Zhu, J.; Mahalingam, M.; Batra, H.; Rao, V.B. Bacteriophage T4 nanoparticles for vaccine delivery against infectious diseases. Adv. Drug Deliv. Rev. 2019, 145, 57–72. [Google Scholar] [CrossRef]
- Yap, M.L.; Rossmann, M.G. Structure and function of bacteriophage T4. Future Microbiol. 2014, 9, 1319–1327. [Google Scholar] [CrossRef]
- Bode, C.; Zhao, G.; Steinhagen, F.; Kinjo, T.; Klinman, D.M. CpG DNA as a vaccine adjuvant. Expert Rev. Vaccines 2011, 10, 499–511. [Google Scholar] [CrossRef]
- Chu, R.S.; Targoni, O.S.; Krieg, A.M.; Lehmann, P.V.; Harding, C.V. CpG oligodeoxynucleotides act as adjuvants that switch on T helper 1 (Th1) immunity. J. Exp. Med. 1997, 186, 1623–1631. [Google Scholar] [CrossRef]
- Roman, M.; Martin-Orozco, E.; Goodman, J.S.; Nguyen, M.-D.; Sato, Y.; Ronaghy, A.; Kornbluth, R.S.; Richman, D.D.; Carson, D.A.; Raz, E. Immunostimulatory DNA sequences function as T helper-1-promoting adjuvants. Nat. Med. 1997, 3, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Krieg, A.M.; Yi, A.K.; Matson, S.; Waldschmidt, T.J.; Bishop, G.A.; Teasdale, R.; Koretzky, G.A.; Klinman, D.M. CpG motifs in bacterial DNA trigger direct B-cell activation. Nature 1995, 374, 546–549. [Google Scholar] [CrossRef] [PubMed]
- Frank, F.M.; Petray, P.B.; Cazorla, S.I.; Muñoz, M.C.; Corral, R.S.; Malchiodi, E.L. Use of a purified Trypanosoma cruzi antigen and CpG oligodeoxynucleotides for immunoprotection against a lethal challenge with trypomastigotes. Vaccine 2003, 22, 77–86. [Google Scholar] [CrossRef]
- Schnapp, A.R.; Eickhoff, C.S.; Sizemore, D.; Curtiss, R., 3rd; Hoft, D.F. Cruzipain induces both mucosal and systemic protection against Trypanosoma cruzi in mice. Infect. Immun. 2002, 70, 5065–5074. [Google Scholar] [CrossRef] [PubMed]
- Araújo, A.F.S.; de Alencar, B.C.G.; Vasconcelos, J.R.C.; Hiyane, M.I.; Marinho, C.R.F.; Penido, M.L.O.; Boscardin, S.B.; Hoft, D.F.; Gazzinelli, R.T.; Rodrigues, M.M. CD8+-T-cell-dependent control of Trypanosoma cruzi infection in a highly susceptible mouse strain after immunization with recombinant proteins based on amastigote surface protein 2. Infect. Immun. 2005, 73, 6017–6025. [Google Scholar] [CrossRef] [PubMed]
- Hoft, D.F.; Eickhoff, C.S.; Giddings, O.K.; Vasconcelos, J.R.C.; Rodrigues, M.M. Trans-sialidase recombinant protein mixed with CpG motif-containing oligodeoxynucleotide induces protective mucosal and systemic Trypanosoma cruzi immunity involving CD8+ CTL and B cell-mediated cross-priming. J. Immunol. 2007, 179, 6889–6900. [Google Scholar] [CrossRef]
- Giddings, O.K.; Eickhoff, C.S.; Sullivan, N.L.; Hoft, D.F. Intranasal vaccinations with the trans-sialidase antigen plus CpG Adjuvant induce mucosal immunity protective against conjunctival Trypanosoma cruzi challenges. Infect. Immun. 2010, 78, 1333–1338. [Google Scholar] [CrossRef]
- Ouaissi, A.; Guilvard, E.; Delneste, Y.; Caron, G.; Magistrelli, G.; Herbault, N.; Thieblemont, N.; Jeannin, P. The Trypanosoma cruzi Tc52-released protein induces human dendritic cell maturation, signals via Toll-like receptor 2, and confers protection against lethal infection. J. Immunol. 2002, 168, 6366–6374. [Google Scholar] [CrossRef]
- Cazorla, S.I.; Frank, F.M.; Becker, P.D.; Corral, R.S.; Guzmán, C.A.; Malchiodi, E.L. Prime-boost immunization with cruzipain co-administered with MALP-2 triggers a protective immune response able to decrease parasite burden and tissue injury in an experimental Trypanosoma cruzi infection model. Vaccine 2008, 26, 1999–2009. [Google Scholar] [CrossRef][Green Version]
- Zygmunt, B.M.; Weissmann, S.F.; Guzman, C.A. NKT cell stimulation with α-galactosylceramide results in a block of Th17 differentiation after intranasal immunization in mice. PLoS ONE 2012, 7, e30382. [Google Scholar] [CrossRef]
- Abbasi, S.; Uchida, S. Multifunctional Immunoadjuvants for Use in Minimalist Nucleic Acid Vaccines. Pharmaceutics 2021, 13, 644. [Google Scholar] [CrossRef] [PubMed]
- Assis, B.R.D.; da Silva, C.D.; Santiago, M.G.; Ferreira, L.A.M.; Goulart, G.A.C. Nanotechnology in adjuvants and vaccine development: What should we know? Nanomedicine 2021, 16, 2565–2568. [Google Scholar] [CrossRef] [PubMed]
- Alfagih, I.M.; Aldosari, B.; AlQuadeib, B.; Almurshedi, A.; Alfagih, M.M. Nanoparticles as adjuvants and nanodelivery systems for mRNA-based vaccines. Pharmaceutics 2021, 13, 45. [Google Scholar] [CrossRef] [PubMed]
- Cappellano, G.; Abreu, H.; Casale, C.; Dianzani, U.; Chiocchetti, A. Nano-microparticle platforms in developing next-generation vaccines. Vaccines 2021, 9, 606. [Google Scholar] [CrossRef] [PubMed]
- Tenchov, R.; Bird, R.; Curtze, A.E.; Zhou, Q. Lipid Nanoparticles—From Liposomes to mRNA Vaccine Delivery, a Landscape of Research Diversity and Advancement. ACS Nano 2021, 15, 16982–17015. [Google Scholar] [CrossRef]
- Li, Y.; Tenchov, R.; Smoot, J.; Liu, C.; Watkins, S.; Zhou, Q. A comprehensive review of the global efforts on COVID-19 vaccine development. ACS Cent. Sci. 2021, 7, 512–533. [Google Scholar] [CrossRef]
- Arce-Fonseca, M.; Rios-Castro, M.; Carrillo-Sánchez, S.d.C.; Martínez-Cruz, M.; Rodríguez-Morales, O. Prophylactic and therapeutic DNA vaccines against Chagas disease. Parasites Vectors 2015, 8, 121. [Google Scholar] [CrossRef]
- Lokugamage, N.; Chowdhury, I.H.; Biediger, R.J.; Market, R.V.; Khounlo, S.; Warier, N.D.; Hwang, S.-A.; Actor, J.K.; Woodside, D.G.; Marathi, U.; et al. Use of a small molecule integrin activator as a systemically administered vaccine adjuvant in controlling Chagas disease. NPJ Vaccines 2021, 6, 114. [Google Scholar] [CrossRef]
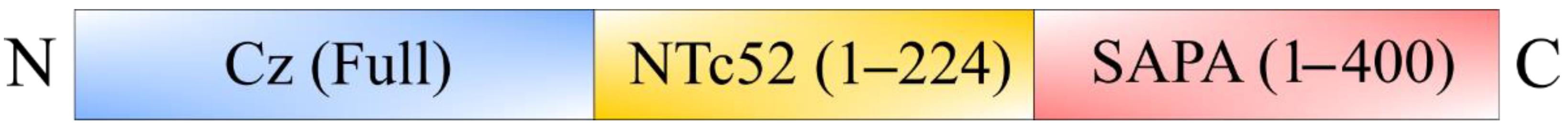
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maldonado, E.; Morales-Pison, S.; Urbina, F.; Solari, A. Vaccine Design against Chagas Disease Focused on the Use of Nucleic Acids. Vaccines 2022, 10, 587. https://doi.org/10.3390/vaccines10040587
Maldonado E, Morales-Pison S, Urbina F, Solari A. Vaccine Design against Chagas Disease Focused on the Use of Nucleic Acids. Vaccines. 2022; 10(4):587. https://doi.org/10.3390/vaccines10040587
Chicago/Turabian StyleMaldonado, Edio, Sebastian Morales-Pison, Fabiola Urbina, and Aldo Solari. 2022. "Vaccine Design against Chagas Disease Focused on the Use of Nucleic Acids" Vaccines 10, no. 4: 587. https://doi.org/10.3390/vaccines10040587
APA StyleMaldonado, E., Morales-Pison, S., Urbina, F., & Solari, A. (2022). Vaccine Design against Chagas Disease Focused on the Use of Nucleic Acids. Vaccines, 10(4), 587. https://doi.org/10.3390/vaccines10040587





