Benefits of Inactivated Vaccine and Viral Vector Vaccine Immunization on COVID-19 Infection in Kidney Transplant Recipients
Abstract
:1. Introduction
2. Methods
2.1. Study Design and Participants
2.2. Outcomes
2.3. Management during COVID-19 Infection
2.4. Statistical Analysis
3. Results
3.1. Patients
3.2. Hospital Course
3.3. Clinical Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AKIN | Acute Kidney Injury Network |
| CI | confidence interval |
| CNIs | calcineurin inhibitors |
| COVID-19 | coronavirus virus disease 2019 |
| eGFR | estimated glomerular filtration rate |
| IQR | interquartile range |
| KDIGO | Kidney Disease: Improving Global Outcomes |
| mRNA | messenger ribonucleic acid |
| mTOR | mammalian target of rapamycin |
| RR | relative risk ratio |
| RT-PCR | real-time polymerase chain reaction |
| SARS-CoV2 | severe acute respiratory syndrome coronavirus 2 |
| SD | standard deviation |
References
- El Sahly, H.M.; Baden, L.R.; Essink, B.; Doblecki-Lewis, S.; Martin, J.M.; Anderson, E.J.; Campbell, T.B.; Clark, J.; Jackson, L.A.; Fichtenbaum, C.J.; et al. Efficacy of the mRNA-1273 SARS-CoV-2 Vaccine at Completion of Blinded Phase. N. Engl. J. Med. 2021, 385, 1774–1785. [Google Scholar] [CrossRef] [PubMed]
- Heath, P.T.; Galiza, E.P.; Baxter, D.N.; Boffito, M.; Browne, D.; Burns, F.; Chadwick, D.R.; Clark, R.; Cosgrove, C.; Galloway, J.; et al. Safety and Efficacy of NVX-CoV2373 Covid-19 Vaccine. N. Engl. J. Med. 2021, 385, 1172–1183. [Google Scholar] [CrossRef] [PubMed]
- Jara, A.; Undurraga, E.A.; González, C.; Paredes, F.; Fontecilla, T.; Jara, G.; Pizarro, A.; Acevedo, J.; Leo, K.; Leon, F.; et al. Effectiveness of an Inactivated SARS-CoV-2 Vaccine in Chile. N. Engl. J. Med. 2021, 385, 875–884. [Google Scholar] [CrossRef]
- Rotondo, J.C.; Martini, F.; Maritati, M.; Mazziotta, C.; Di Mauro, G.; Lanzillotti, C.; Barp, N.; Gallerani, A.; Tognon, M.; Contini, C. SARS-CoV-2 Infection: New Molecular, Phylogenetic, and Pathogenetic Insights. Efficacy of Current Vaccines and the Potential Risk of Variants. Viruses 2021, 13, 1687. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.J.; Moreira, E.D., Jr.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Polack, F.P.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine through 6 Months. N. Engl. J. Med. 2021, 385, 1761–1773. [Google Scholar] [CrossRef] [PubMed]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Perkins, G.B.; Tunbridge, M.; Salehi, T.; Chai, C.S.; Kireta, S.; Johnston, J.; Penko, D.; Nitschke, J.; Yeow, A.; Al-Delfi, Z.; et al. Concurrent vaccination of kidney transplant recipients and close household cohabitants against COVID-19. Kidney Int. 2022. [Google Scholar] [CrossRef] [PubMed]
- Bruminhent, J.; Setthaudom, C.; Chaumdee, P.; Boongird, S.; Kiertiburanakul, S.; Malathum, K.; Nongnuch, A.; Phuphuakrat, A.; Jirasiritham, S.; Janphram, C.; et al. SARS-CoV-2-specific humoral and cell-mediated immune responses after immunization with inactivated COVID-19 vaccine in kidney transplant recipients (CVIM 1 study). Am. J. Transplant. 2021, 22, 813–822. [Google Scholar] [CrossRef]
- Benotmane, I.; Gautier-Vargas, G.; Cognard, N.; Olagne, J.; Heibel, F.; Braun-Parvez, L.; Martzloff, J.; Perrin, P.; Moulin, B.; Fafi-Kremer, S.; et al. Low immunization rates among kidney transplant recipients who received 2 doses of the mRNA-1273 SARS-CoV-2 vaccine. Kidney Int. 2021, 99, 1498–1500. [Google Scholar] [CrossRef]
- Benotmane, I.; Gautier-Vargas, G.; Cognard, N.; Olagne, J.; Heibel, F.; Braun-Parvez, L.; Martzloff, J.; Perrin, P.; Moulin, B.; Fafi-Kremer, S.; et al. Weak anti-SARS-CoV-2 antibody response after the first injection of an mRNA COVID-19 vaccine in kidney transplant recipients. Kidney Int. 2021, 99, 1487–1489. [Google Scholar] [CrossRef]
- Korth, J.; Jahn, M.; Dorsch, O.; Anastasiou, O.; Sorge-Hädicke, B.; Eisenberger, U.; Gäckler, A.; Dittmer, U.; Witzke, O.; Wilde, B.; et al. Impaired Humoral Response in Renal Transplant Recipients to SARS-CoV-2 Vaccination with BNT162b2 (Pfizer-BioNTech). Viruses 2021, 13, 756. [Google Scholar] [CrossRef] [PubMed]
- Malinis, M.; Cohen, E.; Azar, M.M. Effectiveness of SARS-CoV-2 vaccination in fully vaccinated solid organ transplant recipients. Am. J. Transplant. 2021, 21, 2916–2918. [Google Scholar] [CrossRef] [PubMed]
- Miele, M.; Busà, R.; Russelli, G.; Sorrentino, M.C.; Di Bella, M.; Timoneri, F.; Mularoni, A.; Panarello, G.; Vitulo, P.; Conaldi, P.G.; et al. Impaired anti-SARS-CoV-2 humoral and cellular immune response induced by Pfizer-BioNTech BNT162b2 mRNA vaccine in solid organ transplanted patients. Am. J. Transplant. 2021, 21, 2919–2921. [Google Scholar] [CrossRef] [PubMed]
- Akalin, E.; Azzi, Y.; Bartash, R.; Seethamraju, H.; Parides, M.; Hemmige, V.; Ross, M.; Forest, S.; Goldstein, Y.D.; Ajaimy, M.; et al. Covid-19 and Kidney Transplantation. N. Engl. J. Med. 2020, 382, 2475–2477. [Google Scholar] [CrossRef] [PubMed]
- Cristelli, M.P.; Viana, L.A.; Dantas, M.T.; Martins, S.B.; Fernandes, R.; Nakamura, M.R.; Santos, D.W.; Taddeo, J.B.; Azevedo, V.F.; Foresto, R.D.; et al. The Full Spectrum of COVID-19 Development and Recovery among Kidney Transplant Recipients. Transplantation 2021, 105, 1433–1444. [Google Scholar] [CrossRef]
- Danziger-Isakov, L.; Blumberg, E.A.; Manuel, O.; Sester, M. Impact of COVID-19 in solid organ transplant recipients. Am. J. Transplant. 2021, 21, 925–937. [Google Scholar] [CrossRef]
- Requião-Moura, L.R.; de Sandes-Freitas, T.V.; Viana, L.A.; Cristelli, M.P.; de Andrade, L.G.M.; Garcia, V.D.; de Oliveira, C.M.C.; Esmeraldo, R.D.M.; Filho, M.A.; Pacheco-Silva, A.; et al. High mortality among kidney transplant recipients diagnosed with coronavirus disease 2019: Results from the Brazilian multicenter cohort study. PLoS ONE 2021, 16, e0254822. [Google Scholar] [CrossRef]
- Tartof, S.Y.; Slezak, J.M.; Fischer, H.; Hong, V.; Ackerson, B.K.; Ranasinghe, O.N.; Frankland, T.B.; Ogun, O.A.; Zamparo, J.M.; Gray, S.; et al. Effectiveness of mRNA BNT162b2 COVID-19 vaccine up to 6 months in a large integrated health system in the USA: A retrospective cohort study. Lancet 2021, 398, 1407–1416. [Google Scholar] [CrossRef]
- Yoda, T.; Suksatit, B.; Tokuda, M.; Katsuyama, H. Analysis of People’s Attitude toward COVID-19 Vaccine and Its Information Sources in Thailand. Cureus 2022, 14, 22215. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Interim Recommendations for Use of the Inactivated COVID-19 Vaccine, CoronaVac, Developed by Sinovac: Interim Guidance, First Issued 24 May 2021, Updated 21 October 2021, Updated 15 March 2022; World Health Organization: Geneva, Switzerland, 2022. [Google Scholar]
- World Health Organization (WHO). Interim Recommendations for Use of the ChAdOx1-S [Recombinant] Vaccine against COVID-19 (AstraZeneca COVID-19 Vaccine AZD1222, SII Covishield, SK Bioscience); World Health Organization (WHO): Geneva, Switzerland, 2021. [Google Scholar]
- Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Section 2: AKI Definition. Kidney Int. Suppl. 2012, 2, 19–36. [Google Scholar] [CrossRef] [Green Version]
- Mishra, P.; Pandey, C.M.; Singh, U.; Keshri, A.; Sabaretnam, M. Selection of appropriate statistical methods for data analysis. Ann. Card. Anaesth. 2019, 22, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Kishore, J.; Goel, M.; Khanna, P. Understanding survival analysis: Kaplan-Meier estimate. Int. J. Ayurveda Res. 2010, 1, 274–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonanad, C.; García-Blas, S.; Tarazona-Santabalbina, F.; Sanchis, J.; Bertomeu-González, V.; Fácila, L.; Ariza, A.; Núñez, J.; Cordero, A. The Effect of Age on Mortality in Patients With COVID-19: A Meta-Analysis With 611,583 Subjects. J. Am. Med. Dir. Assoc. 2020, 21, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Trecarichi, E.M.; Mazzitelli, M.; Serapide, F.; Pelle, M.C.; Tassone, B.; Arrighi, E.; Perri, G.; Fusco, P.; Scaglione, V.; Davoli, C.; et al. Clinical characteristics and predictors of mortality associated with COVID-19 in elderly patients from a long-term care facility. Sci. Rep. 2020, 10, 20834. [Google Scholar] [CrossRef]
- Voysey, M.; Costa Clemens, S.A.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: A pooled analysis of four randomised trials. Lancet 2021, 397, 881–891. [Google Scholar] [CrossRef]
- Maggiore, U.; Abramowicz, D.; Crespo, M.; Mariat, C.; Mjoen, G.; Peruzzi, L.; Sever, M.S.; Oniscu, G.C.; Hilbrands, L.; Watschinger, B. How should I manage immunosuppression in a kidney transplant patient with COVID-19? An ERA-EDTA DESCARTES expert opinion. Nephrol. Dial. Transplant. 2020, 35, 899–904. [Google Scholar] [CrossRef]
- Angelico, R.; Blasi, F.; Manzia, T.; Toti, L.; Tisone, G.; Cacciola, R. The Management of Immunosuppression in Kidney Transplant Recipients with COVID-19 Disease: An Update and Systematic Review of the Literature. Medicina 2021, 57, 435. [Google Scholar] [CrossRef]
- López, V.; Vázquez, T.; Alonso-Titos, J.; Cabello, M.; Alonso, A.; Beneyto, I.; Crespo, M.; Díaz-Corte, C.; Franco, A.; González-Roncero, F.; et al. Recommendations on management of the SARS-CoV-2 coronavirus pandemic (COVID-19) in kidney transplant patients. Nefrología 2020, 40, 265–271. [Google Scholar] [CrossRef]
- Pongpirul, W.A.; Wiboonchutikul, S.; Charoenpong, L.; Panitantum, N.; Vachiraphan, A.; Uttayamakul, S.; Pongpirul, K.; Manosuthi, W.; Prasithsirikul, W. Clinical course and potential predictive factors for pneumonia of adult patients with Coronavirus Disease 2019 (COVID-19): A retrospective observational analysis of 193 confirmed cases in Thailand. PLoS Negl. Trop. Dis. 2020, 14, e0008806. [Google Scholar] [CrossRef]
- Meshram, H.S.; Kute, V.B.; Shah, N.; Chauhan, S.; Navadiya, V.V.; Patel, A.H.; Patel, H.V.; Engineer, D.; Banerjee, S.; Rizvi, J.; et al. COVID-19 in Kidney Transplant Recipients Vaccinated With Oxford-AstraZeneca COVID-19 Vaccine (Covishield): A Single-center Experience From India. Transplantation 2021, 105, e100–e103. [Google Scholar] [CrossRef]
- Prasad, N.; Yadav, B.; Singh, M.; Gautam, S.; Bhadauria, D.; Patel, M.; Kushwaha, R.; Yadav, D.; Singh, A.; Yachha, M.; et al. Humoral Immune Response of SARS-CoV-2 Infection and Anti-SARS-CoV-2 Vaccination in Renal Transplant Recipients. Vaccines 2022, 10, 385. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.; Klemis, V.; Schub, D.; Schneitler, S.; Reichert, M.C.; Wilkens, H.; Sester, U.; Sester, M.; Mihm, J. Cellular immunity predominates over humoral immunity after homologous and heterologous mRNA and vector-based COVID-19 vaccine regimens in solid organ transplant recipients. Am. J. Transplant. 2021, 21, 3990–4002. [Google Scholar] [CrossRef] [PubMed]
- Masset, C.; Kerleau, C.; Garandeau, C.; Ville, S.; Cantarovich, D.; Hourmant, M.; Kervella, D.; Houzet, A.; Guillot-Gueguen, C.; Guihard, I.; et al. A third injection of the BNT162b2 mRNA COVID-19 vaccine in kidney transplant recipients improves the humoral immune response. Kidney Int. 2021, 100, 1132–1135. [Google Scholar] [CrossRef] [PubMed]
| Patient Characteristics | Total (N = 45) | Vaccinated (N = 23) | Unvaccinated (N = 22) | p-Value |
|---|---|---|---|---|
| Age (y), mean(SD) | 52 (13) | 55 (11) | 48 (15) | 0.04 |
| Gender, n (%) | 0.12 | |||
| Male | 30 (67) | 18 (78) | 12 (55) | |
| Female | 15 (33) | 5 (22) | 10 (45) | |
| BMI (kg/m2), mean (SD) | 25.11 (5.68) | 25.19 (5.49) | 25.03 (6.00) | 0.92 |
| Kidney transplant status | 0.67 | |||
| Deceased donor transplantation, n (%) | 30 (67) | 16 (70) | 14 (64) | |
| Living related transplantation, n (%) | 15 (33) | 7 (30) | 8 (36) | |
| Post-transplant time (y), median (IQR) | 5.4 (2.63–8.65) | 4.6 (2.74–7.69) | 6.72 (2.28–9.66) | 0.86 |
| Baseline creatinine (mg/dL), mean (SD) | 1.45 (0.67) | 1.46 (0.52) | 1.43 (0.81) | 0.85 |
| eGFR (mL/min/1.73 m2), mean (SD) | 62.1 (26.5) | 60.6 (26.3) | 63.7 (27.2) | 0.69 |
| Immunosuppressive regimens, n(%) | ||||
| Calcineurin inhibitors | 41 (91) | 22 (96) | 19 (86) | 0.35 |
| Mycophenolate | 42 (93) | 23 (100) | 19 (86) | 0.11 |
| mTOR inhibitors | 3 (7) | 1 (4) | 2 (9) | 0.61 |
| Steroids | 43 (96) | 21 (91) | 22 (100) | 0.49 |
| Comorbidity, n (%) | ||||
| Diabetes | 20 (44) | 10 (43) | 10 (45) | 0.89 |
| Hypertension | 32 (71) | 18 (78) | 14 (64) | 0.34 |
| Ischemic heart disease | 7 (16) | 5 (22) | 2 (9) | 0.41 |
| Obesity | 18 (60) | 9 (39) | 9 (41) | 0.90 |
| Vaccination status, n (%) | ||||
| ChAdOxl-1 dose | 16 (69) | |||
| ChAdOx1-2 dose | 2 (9) | |||
| CoronaVac-2 dose | 5 (22) | |||
| Interval after last vaccine (d), median (IQR) | 49 (21–62) |
| Clinical Course, n (%) | Total (N = 45) | Vaccinated (N = 23) | Unvaccinated (N = 22) | p-Value |
|---|---|---|---|---|
| Clinical manifestation | ||||
| Asymptomatic | 2 (5) | 1 (4) | 1 (5) | 0.95 |
| Fever | 41 (93) | 22 (96) | 19 (90) | 0.50 |
| Upper respiratory tract symptoms | 15 (34) | 9 (39) | 6 (29) | 0.46 |
| Pneumonia | 31 (70) | 16 (70) | 15 (71) | 0.89 |
| Diarrhea | 2 (5) | 1 (4) | 1 (5) | 0.95 |
| Treatment | ||||
| Favipiravir | 41 (91) | 21 (91) | 20 (91) | 0.96 |
| Remdesivir | 2 (4) | 0 (0) | 2 (9) | 0.14 |
| Dexamethasone | 17 (38) | 11 (48) | 6 (27) | 0.16 |
| Methylprednisolone | 3 (7) | 1 (4) | 2 (9) | 0.52 |
| Tocilizumab | 1 (2) | 1 (4) | 0 (0) | 0.32 |
| Baricitinib | 2 (4) | 0 (0) | 2 (9) | 0.32 |
| Hemoperfusion | 1 (2) | 1 (4) | 0 (0) | 0.32 |
| Oxygen therapy | 0.55 | |||
| No requirement | 17 (39) | 11 (48) | 6 (29) | |
| Nasal cannula | 15 (34) | 7 (30) | 8 (38) | |
| High flow nasal cannula | 6 (14) | 3 (13) | 3 (14) | |
| Invasive mechanical ventilation | 6 (14) | 2 (9) | 4 (19) | |
| Immunosuppressive management | ||||
| Calcineurin inhibitors (N = 41) | 0.45 | |||
| Continue | 34 (83) | 18 (82) | 16 (84) | |
| Reduce dose | 4 (10) | 3 (14) | 1 (5) | |
| Discontinue | 3 (7) | 1 (4) | 2 (11) | |
| Mycophenolate (N = 42) | 0.47 | |||
| Continue | 12 (29) | 7 (30) | 5 (26) | |
| Discontinue | 30 (71) | 16 (70) | 14 (74) | |
| mTOR inhibitors (N = 3) | 0.61 | |||
| Continue | 3 (100) | 1 (100) | 2 (100) |
| Outcome, n (%) | Total | Vaccinated | Unvaccinated | RR † (95% CI) | p-Value | ||
|---|---|---|---|---|---|---|---|
| (N = 45) | Any Vaccine (N = 23) | Full Dose ‡ (N = 7) | AZ 1 Dose (N = 16) | (N = 22) | |||
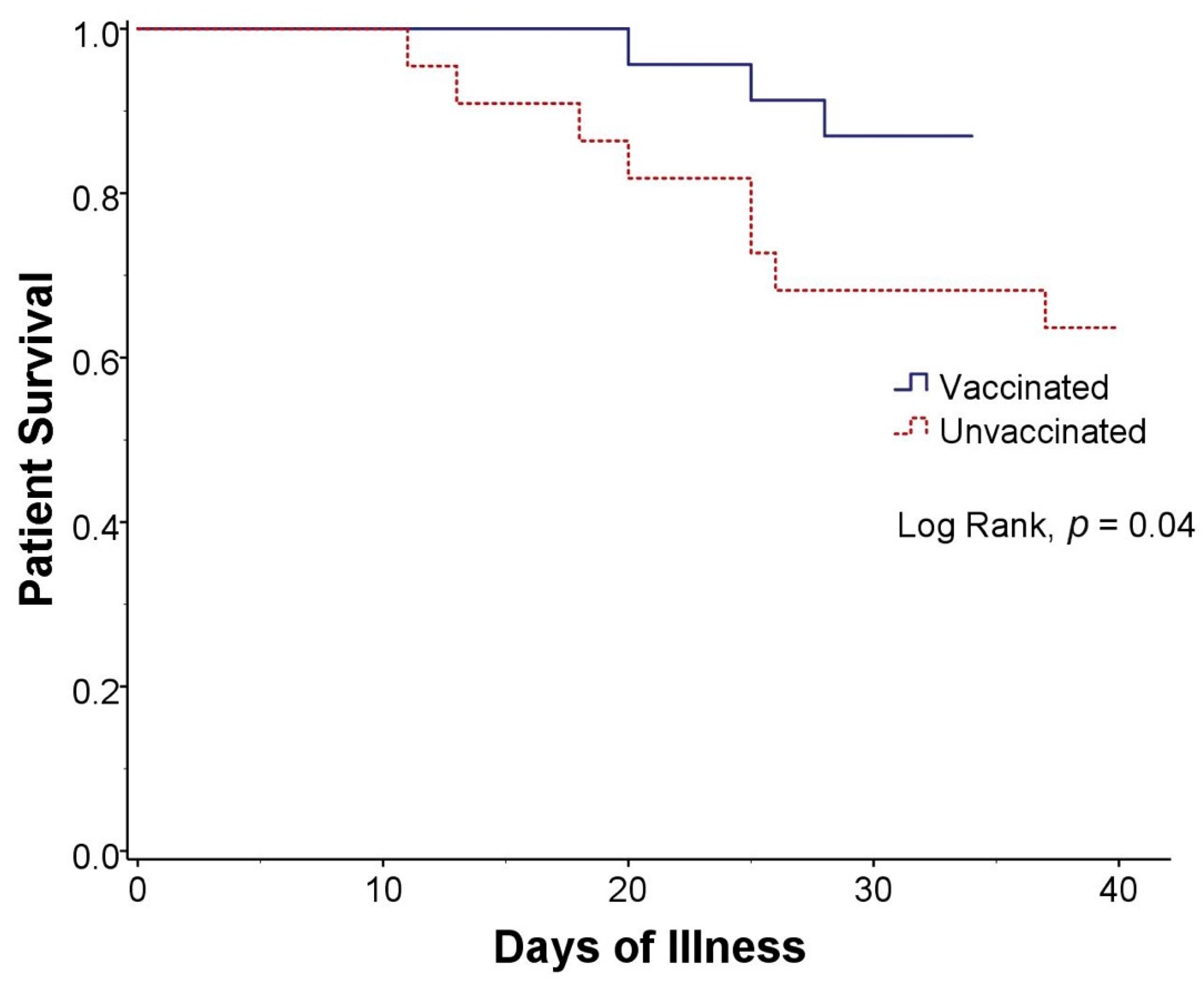
| Death | 11 (24) | 3 (13) | 0 (0) | 3 (19) | 8 (36) | 0.56 (0.29–0.83) | 0.03 |
| Oxygen requirement | 28 (62) | 11 (50) | 3 (43) | 9 (56) | 17 (74) | 0.35 (0.1–1.23) | 0.10 |
| Mechanical ventilation | 6 (14) | 2 (9) | 0 (0) | 2 (13) | 4 (19) | 0.57 (0.12–2.73) | 0.48 |
| Bacterial pneumonia | 14 (31) | 6 (26) | 1 (14) | 5 (31) | 8 (36) | 0.7 (0.2–2.51) | 0.59 |
| IPA | 5 (11) | 1 (4) | 0 (0) | 1 (6) | 4 (18) | 0.23 (0.02–2.21) | 0.20 |
| Pulmonary embolism | 4 (9) | 3 (13) | 0 (0) | 3 (19) | 1 (5) | 1.5 (0.41–6.93) | 0.69 |
| Septicemia | 4 (9) | 2 (9) | 1 (14) | 1 (6) | 2 (9) | 1.05 (0.14–8.18) | 0.96 |
| Acute kidney injury | 21 (47) | 11 (50) | 1 (14) | 10 (63) | 10 (46) | 0.76 (0.23–2.46) | 0.64 |
| AKIN stage 1 | 12 (27) | 7 (30) | 1 (14) | 6 (38) | 5 (23) | 1.06 (0.28–3.98) | 0.93 |
| AKIN stage 2 | 5 (11) | 1 (4) | 0 (0) | 1 (6) | 4 (18) | 0.23 (0.03–2.21) | 0.20 |
| AKIN stage 3 | 3 (7) | 2 (9) | 0 (0) | 2 (13) | 1 (5) | 2.2 (0.19–16.12) | 0.53 |
| Required KRT | 2 (4) | 1 (4) | 0 (0) | 1 (6) | 1 (5) | 0.98 (0.23–4.76) | 0.58 |
| Factors | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | |
| Vaccine | 0.56 (0.29–0.83) | 0.03 | 0.54 (0.24–0.83) | 0.03 |
| Recipient age | 1.01 (0.96–1.07) | 0.56 | ||
| Deceased donor transplantation | 1.46 (0.23–6.53) | 0.63 | ||
| Obesity | 2.20 (0.55–8.74) | 0.26 | ||
| Diabetes | 4.89 (1.09–21.95) | 0.04 | 5.37 (1.07–26.95) | 0.04 |
| Sepsis | 3.56 (0.44–28.89) | 0.24 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sutharattanapong, N.; Thotsiri, S.; Kantachuvesiri, S.; Wiwattanathum, P. Benefits of Inactivated Vaccine and Viral Vector Vaccine Immunization on COVID-19 Infection in Kidney Transplant Recipients. Vaccines 2022, 10, 572. https://doi.org/10.3390/vaccines10040572
Sutharattanapong N, Thotsiri S, Kantachuvesiri S, Wiwattanathum P. Benefits of Inactivated Vaccine and Viral Vector Vaccine Immunization on COVID-19 Infection in Kidney Transplant Recipients. Vaccines. 2022; 10(4):572. https://doi.org/10.3390/vaccines10040572
Chicago/Turabian StyleSutharattanapong, Napun, Sansanee Thotsiri, Surasak Kantachuvesiri, and Punlop Wiwattanathum. 2022. "Benefits of Inactivated Vaccine and Viral Vector Vaccine Immunization on COVID-19 Infection in Kidney Transplant Recipients" Vaccines 10, no. 4: 572. https://doi.org/10.3390/vaccines10040572
APA StyleSutharattanapong, N., Thotsiri, S., Kantachuvesiri, S., & Wiwattanathum, P. (2022). Benefits of Inactivated Vaccine and Viral Vector Vaccine Immunization on COVID-19 Infection in Kidney Transplant Recipients. Vaccines, 10(4), 572. https://doi.org/10.3390/vaccines10040572






