Waning Humoral Response after COVID-19 mRNA Vaccination in Maintenance Dialysis Patients and Recovery after a Complementary Third Dose
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.1.1. First Protocol
2.1.2. Second Protocol
2.2. Study Population
2.3. Procedures and Analytical Methods
2.4. Statistical Analyses
3. Results
3.1. Patient Characteristics
3.2. Waning Anti-S IgG Antibodies after 3 and 6 Months (First Protocol)
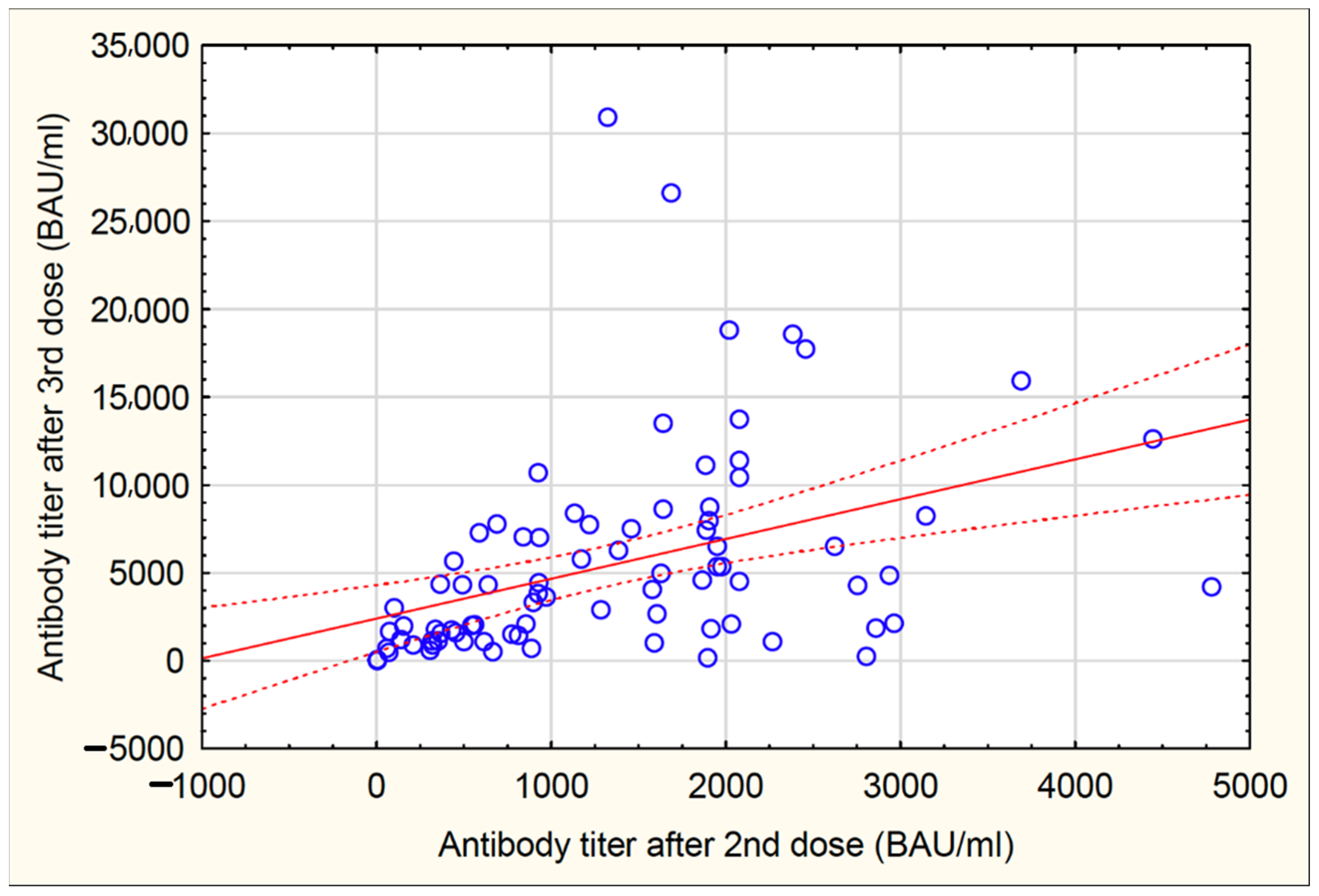
3.3. Anti-S IgG Antibody Titer after the Third Vaccine Dose (Second Protocol)
3.4. Reactogenicity to the Third Vaccine Dose
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dickerman, B.A.; Gerlovin, H.; Madenci, A.L.; Kurgansky, K.E.; Ferolito, B.R.; Figueroa Muñiz, M.J.; Gagnon, D.R.; Gaziano, J.M.; Cho, K.; Casas, J.P.; et al. Comparative effectiveness of BNT162b2 and mRNA-1273 Vaccines in U.S. Veterans. N. Engl. J. Med. 2021, 386, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Levin, E.G.; Lustig, Y.; Cohen, C.; Fluss, R.; Indenbaum, V.; Amit, S.; Doolman, R.; Asraf, K.; Mendelson, E.; Ziv, A.; et al. Waning immune humoral response to BNT162b2 covid-19 vaccine over 6 months. N. Engl. J. Med. 2021, 385, e84. [Google Scholar] [CrossRef] [PubMed]
- Sette, A.; Crotty, S. Adaptive immunity to SARS-CoV-2 and COVID-19. Cell 2021, 184, 861–880. [Google Scholar] [CrossRef] [PubMed]
- Barda, N.; Dagan, N.; Cohen, C.; Hernan, M.A.; Lipsitch, M.; Kohane, I.S.; Reis, B.Y.; Balicer, R.D. Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: An observational study. Lancet 2021, 398, P2093–P2100. [Google Scholar] [CrossRef]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef]
- Hilbrands, L.B.; Duivenvoorden, R.; Vart, P.; Franssen, C.F.M.; Hemmelder, M.H.; Jager, K.J.; Kieneker, L.M.; Noordzij, M.; Pena, M.J.; Vries, H.; et al. COVID-19-related mortality in kidney transplant and dialysis patients: Results of the ERACODA collaboration. Nephrol. Dial. Transplant. 2020, 35, 1973–1983. [Google Scholar] [CrossRef]
- Puchalska-Reglińska, E.; Debska-Slizien, A.; Biedunkiewicz, B.; Tylicki, P.; Polewska, K.; Rutkowski, B.; Gellert, R.; Tylicki, L. Extremely high mortality in COVID-19 hemodialyzed patients before the anti-SARS-CoV-2 vaccination era. Large database from the North of Poland. Pol. Arch. Intern. Med. 2021, 131, 643–648. [Google Scholar] [CrossRef]
- Tylicki, L.; Puchalska-Reglińska, E.; Tylicki, P.; Och, A.; Polewska, K.; Biedunkiewicz, B.; Parczewska, A.; Szabat, K.; Wolf, J.; Dębska-Ślizień, A. Predictors of mortality in hemodialyzed patients after SARS-CoV-2 infection. J. Clin. Med. 2022, 11, 285. [Google Scholar] [CrossRef]
- Tylicki, L.; Biedunkiewicz, B.; Dabrowska, M.; Slizien, W.; Tylicki, P.; Polewska, K.; Rosenberg, I.; Rodak, S.; Debska-Slizien, A. Humoral response to SARS-CoV-2 vaccination promises to improve the catastrophic prognosis of hemodialysis patients as a result of COVID-19: The COViNEPH project. Pol. Arch. Intern. Med. 2021, 131, 797–801. [Google Scholar] [CrossRef]
- Tylicki, L.; Piotrowska, M.; Biedunkiewicz, B.; Zielinski, M.; Dabrowska, M.; Tylicki, P.; Polewska, K.; Trzonkowski, P.; Lichodziejewska-Niemierko, M.; Debska-Slizien, A. Humoral response to COVID19 vaccination in patients treated with peritoneal dialysis: The COViNEPH project. Pol. Arch. Intern. Med. 2021, 131, 16091. [Google Scholar] [CrossRef]
- Grupper, A.; Sharon, N.; Finn, T.; Cohen, R.; Israel, M.; Agbaria, A.; Rechavi, Y.; Schwartz, I.F.; Schwartz, D.; Lellouch, Y.; et al. Humoral response to the Pfizer BNT162b2 vaccine in patients undergoing maintenance hemodialysis. Clin. J. Am. Soc. Nephrol. 2021, 16, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Davidovic, T.; Schimpf, J.; Abbassi-Nik, A.; Stockinger, R.; Sprenger-Mahr, H.; Lhotta, K.; Zitt, E. Waning humoral response 6 months after SARS-CoV-2 vaccination with the mRNA-BNT162b2 vaccine in hemodialysis patients: Time for a boost. Kidney Int. 2021, 100, 1334–1335. [Google Scholar] [CrossRef] [PubMed]
- Anand, S.; Montez-Rath, M.E.; Han, J.; Garcia, P.; Cadden, L.; Hunsader, P.; Morgan, C.; Kerschmann, R.; Beyer, P.; Dittrich, M.; et al. SARS-CoV-2 Vaccine Antibody Response and Breakthrough Infection in Patients Receiving Dialysis. Ann. Intern. Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Bensouna, I.; Caudwell, V.; Kubab, S.; Acquaviva, S.; Pardon, A.; Vittoz, N.; Bozman, D.F.; Hanafi, L.; Faucon, A.L.; Housset, P. SARS-CoV-2 Antibody Response After a Third Dose of the BNT162b2 Vaccine in Patients Receiving Maintenance Hemodialysis or Peritoneal Dialysis. Am. J. Kidney Dis. 2021, 79, 185–192.e1. [Google Scholar] [CrossRef] [PubMed]
- Tillmann, F.P.; Figiel, L.; Ricken, J.; Still, H.; Korte, C.; Plassmann, G.; von Landenberg, P. Evolution of SARS-CoV-2-neutralizing antibodies after two standard dose vaccinations, risk factors for non-response and effect of a third dose booster vaccination in non-responders on hemodialysis: A prospective multi-centre cohort study. J. Clin. Med. 2021, 10, 5113. [Google Scholar] [CrossRef]
- Dimeglio, C.; Herin, F.; Martin-Blondel, G.; Miedouge, M.; Izopet, J. Antibody titers and protection against a SARS-CoV-2 infection. J. Infect. 2022, 84, 248–288. [Google Scholar] [CrossRef]
- Bonelli, F.; Blocki, F.A.; Bunnell, T.; Chu, E.; De La, O.A.; Grenache, D.G.; Marzucchi, G.; Montomoli, E.; Okoye, L.; Pallavicini, L.; et al. Evaluation of the automated LIAISON((R)) SARS-CoV-2 TrimericS IgG assay for the detection of circulating antibodies. Clin. Chem. Lab. Med. 2021, 59, 1463–1467. [Google Scholar] [CrossRef]
- McAndrews, K.M.; Dowlatshahi, D.P.; Dai, J.; Becker, L.M.; Hensel, J.; Snowden, L.M.; Leveille, J.M.; Brunner, M.R.; Holden, K.W.; Hopkins, N.S.; et al. Heterogeneous antibodies against SARS-CoV-2 spike receptor binding domain and nucleocapsid with implications for COVID-19 immunity. JCI Insight 2020, 5, e142386. [Google Scholar] [CrossRef]
- Polewska, K.; Tylicki, P.; Biedunkiewicz, B.; Rucinska, A.; Szydlowska, A.; Kubanek, A.; Rosenberg, I.; Rodak, S.; Slizien, W.; Renke, M.; et al. Safety and tolerability of the BNT162b2 mRNA COVID-19 vaccine in dialyzed patients. COViNEPH Project. Medicina 2021, 57, 732. [Google Scholar] [CrossRef]
- Tillmann, F.P.; Still, H.; von Landenberg, P. Long-term trajectories of SARS-CoV-2 neutralizing antibodies and predictive value of first dose vaccination-induced IgG-antibodies in hemodialysis patients. Int. Urol. Nephrol. 2021, 1–7. [Google Scholar] [CrossRef]
- Bayart, J.L.; Douxfils, J.; Gillot, C.; David, C.; Mullier, F.; Elsen, M.; Eucher, C.; Van Eeckhoudt, S.; Roy, T.; Gerin, V.; et al. Waning of IgG, total and neutralizing antibodies 6 months post-vaccination with BNT162b2 in healthcare workers. Vaccines 2021, 9, 1092. [Google Scholar] [CrossRef] [PubMed]
- Speer, C.; Schaier, M.; Nusshag, C.; Tollner, M.; Buylaert, M.; Kalble, F.; Reichel, P.; Grenz, J.; Susal, C.; Zeier, M.; et al. Longitudinal humoral responses after COVID-19 vaccination in peritoneal and hemodialysis patients over twelve weeks. Vaccines 2021, 9, 1130. [Google Scholar] [CrossRef] [PubMed]
- Dekervel, M.; Henry, N.; Torreggiani, M.; Pouteau, L.M.; Imiela, J.P.; Mellaza, C.; Garnier, A.S.; Dujardin, A.; Asfar, M.; Ducancelle, A.; et al. Humoral response to a third injection of BNT162b2 vaccine in patients on maintenance haemodialysis. Clin. Kidney J. 2021, 14, 2349–2355. [Google Scholar] [CrossRef] [PubMed]
- Stervbo, U.; Blazquez-Navarro, A.; Blanco, E.V.; Safi, L.; Meister, T.L.; Paniskaki, K.; Stockhausen, M.; Marheinecke, C.; Zimmer, G.; Wellenkotter, J.; et al. Improved cellular and humoral immunity upon a second BNT162b2 and mRNA-1273 boost in prime-boost vaccination no/low responders with end-stage renal disease. Kidney Int. 2021, 100, 1335–1337. [Google Scholar] [CrossRef] [PubMed]
- Ducloux, D.; Colladant, M.; Chabannes, M.; Yannaraki, M.; Courivaud, C. Humoral response after 3 doses of the BNT162b2 mRNA COVID-19 vaccine in patients on hemodialysis. Kidney Int. 2021, 100, 702–704. [Google Scholar] [CrossRef]
- Amodio, E.; Capra, G.; Casuccio, A.; Grazia, S.; Genovese, D.; Pizzo, S.; Calamusa, G.; Ferraro, D.; Giammanco, G.M.; Vitale, F.; et al. Antibodies responses to SARS-CoV-2 in a large cohort of vaccinated subjects and seropositive patients. Vaccines 2021, 9, 714. [Google Scholar] [CrossRef]
- Debska-Slizien, A.; Slizien, Z.; Muchlado, M.; Kubanek, A.; Piotrowska, M.; Dabrowska, M.; Tarasewicz, A.; Chamienia, A.; Biedunkiewicz, B.; Renke, M.; et al. Predictors of humoral response to mRNA COVID19 vaccines in kidney transplant recipients: A longitudinal study-the COViNEPH project. Vaccines 2021, 9, 1165. [Google Scholar] [CrossRef]
- Boulanger, H.; Saksi, S.A.; Achiche, J.; Batusanski, F.; Stawiarski, N.; Diddaoui, A.; Fromentin, L.; Chawki, M. SARS-CoV-2 antibodies in hemodialysis patients six months after infection compared to healthcare workers. Int. J. Nephrol. 2021, 2021, 4747221. [Google Scholar] [CrossRef]
- Zhong, D.; Xiao, S.; Debes, A.K.; Egbert, E.R.; Caturegli, P.; Colantuoni, E.; Milstone, A.M. Durability of antibody levels after vaccination with mRNA SARS-CoV-2 vaccine in individuals with or without prior infection. JAMA 2021, 326, 2524–2526. [Google Scholar] [CrossRef]
- Debska-Slizien, A.; Muchlado, M.; Slizien, Z.; Kubanek, A.; Piotrowska, M.; Dabrowska, M.; Bzoma, B.; Konopa, J.; Renke, M.; Biedunkiewicz, B.; et al. Significant humoral response to mRNA COVID-19 vaccine in kidney transplant recipients with prior exposure to SARS-CoV-2. The COViNEPH Project. Pol. Arch. Intern. Med. 2021, 132. [Google Scholar] [CrossRef]
- Jelley-Gibbs, D.M.; Dibble, J.P.; Filipson, S.; Haynes, L.; Kemp, R.A.; Swain, S.L. Repeated stimulation of CD4 effector T cells can limit their protective function. J. Exp. Med. 2005, 201, 1101–1112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blank, C.U.; Haining, W.N.; Held, W.; Hogan, P.G.; Kallies, A.; Lugli, E.; Lynn, R.C.; Philip, M.; Rao, A.; Restifo, N.P.; et al. Defining ‘T cell exhaustion’. Nat. Rev. Immunol. 2019, 19, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Bonura, F.; De Grazia, S.; Bonura, C.; Sanfilippo, G.L.; Giammanco, G.M.; Amodio, E.; Ferraro, D. Differing kinetics of anti-spike protein IgGs and neutralizing antibodies against SARS-CoV-2 after Comirnaty (BNT162b2) immunization. J. Appl. Microbiol. 2022. [Google Scholar] [CrossRef] [PubMed]
| IN-Ds N = 109 | PI-Ds N = 32 | Controls N = 20 | p-Value * | |
|---|---|---|---|---|
| Age years | 69 (57–75) | 65 (58–74) | 53 (47–69.5) | 0.09 |
| Male sex Female sex | 69 (63.30) 40 (36.70) | 22 (68.75) 10 (31.25) | 13 (65) 7 (35) | 0.57 0.57 |
| CCI | 6 (4–8) | 6.5 (4–8) | 1 (1–3) | <0.001 |
| Diabetes mellitus | 40 (36.69) | 6 (18.75) | 0 (0) | 0.059 |
| BMI kg/m2 | 24.9 (22.4–29.3) | 26.1 (23.2–29.1) | 27 (24.5–30.5) | 0.5 |
| Dialysis vintage months | 36 (15–74) | 39 (19.5–84) | na | 0.71 |
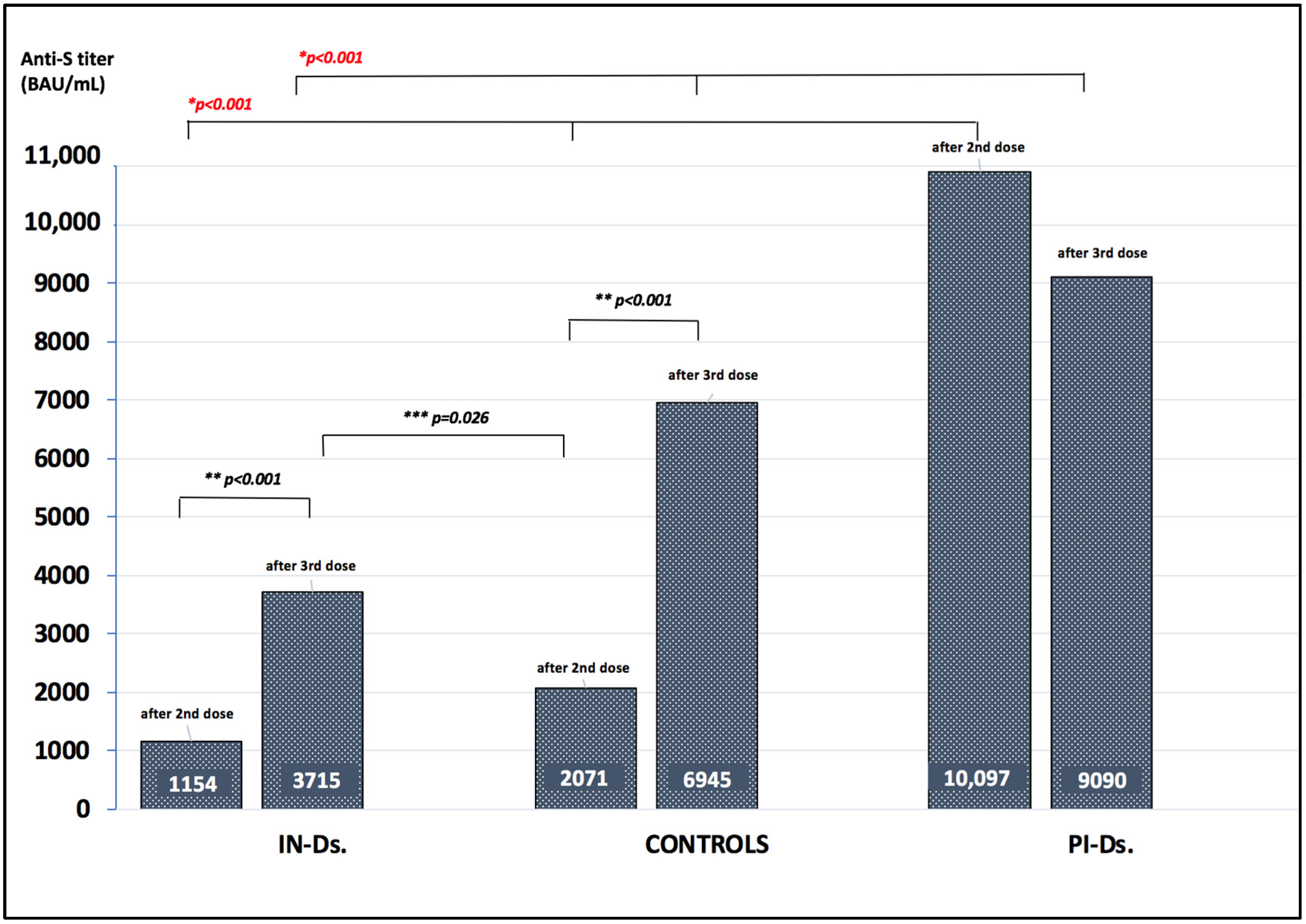
| Anti-S IgG after 2nd dose (BAU/mL) | 933 (528–1906) | 10,907 (2502–18,031) | 2070 (1703–3068) | <0.001 |
| Anti-S IgG 3 months after 2nd dose (BAU/mL) | 159 (42.3–357) | 2945 (1600–11,500) | 508 (422–1127) | <0.001 |
| Anti-S IgG 6 months after 2nd dose (BAU/mL) | 66 (24–127) | 723 (474–1110) | 231 (103–336) | <0.001 |
| IN-Ds N = 104 | PI-Ds N = 35 | Controls N = 20 | p-Value * | |
|---|---|---|---|---|
| Age years | 70 (58.5–76) | 62 (46–70) | 53 (47–69.5) | 0.002 |
| Male sex Female sex | 65 (62.5) 39 (37.5) | 24 (68.57) 11 (31.43) | 13 (65) 7 (35) | 0.52 0.52 |
| CCI | 6.5 (4–8) | 6 (3–7) | 1 (1–3) | <0.001 |
| Diabetes mellitus | 36 (34.61) | 8 (22.86) | 0 | 0.019 |
| BMI kg/m2 | 25.6 (22.7–29.2) | 26.3 (22.86–28.1) | 27 (24.5–30.5) | 0.40 |
| Dialysis vintage months | 33.5 (11–68.5) | 36 (10–60) | 0.77 | |
| BNT162b2 vaccination mRNA-1273 vaccination Anti-S IgG after 2nd dose (BAU/mL) | 96 (92.3) 8 (7.7) 1154 (474–1952) | 30 (85.7) 5 (14.3) 10,907 (1342–13,754) | 20 (100) 0 (0) 2070 (1703–3068) | 0.40 0.40 <0.001 |
| Anti-S IgG before 3rd dose (BAU/mL) | 72 (25–160) | 1080 (474–1660) | 231 (102–336) | <0.001 |
| Anti-S IgG after 3rd dose (BAU/mL) | 3715 (1470–7325) | 9090 (3300–15,000) | 6945 (2130–11,800) | <0.001 |
| Follow-Up | IN-Ds | CONTROLS | PI-Ds | p-Value | |
|---|---|---|---|---|---|
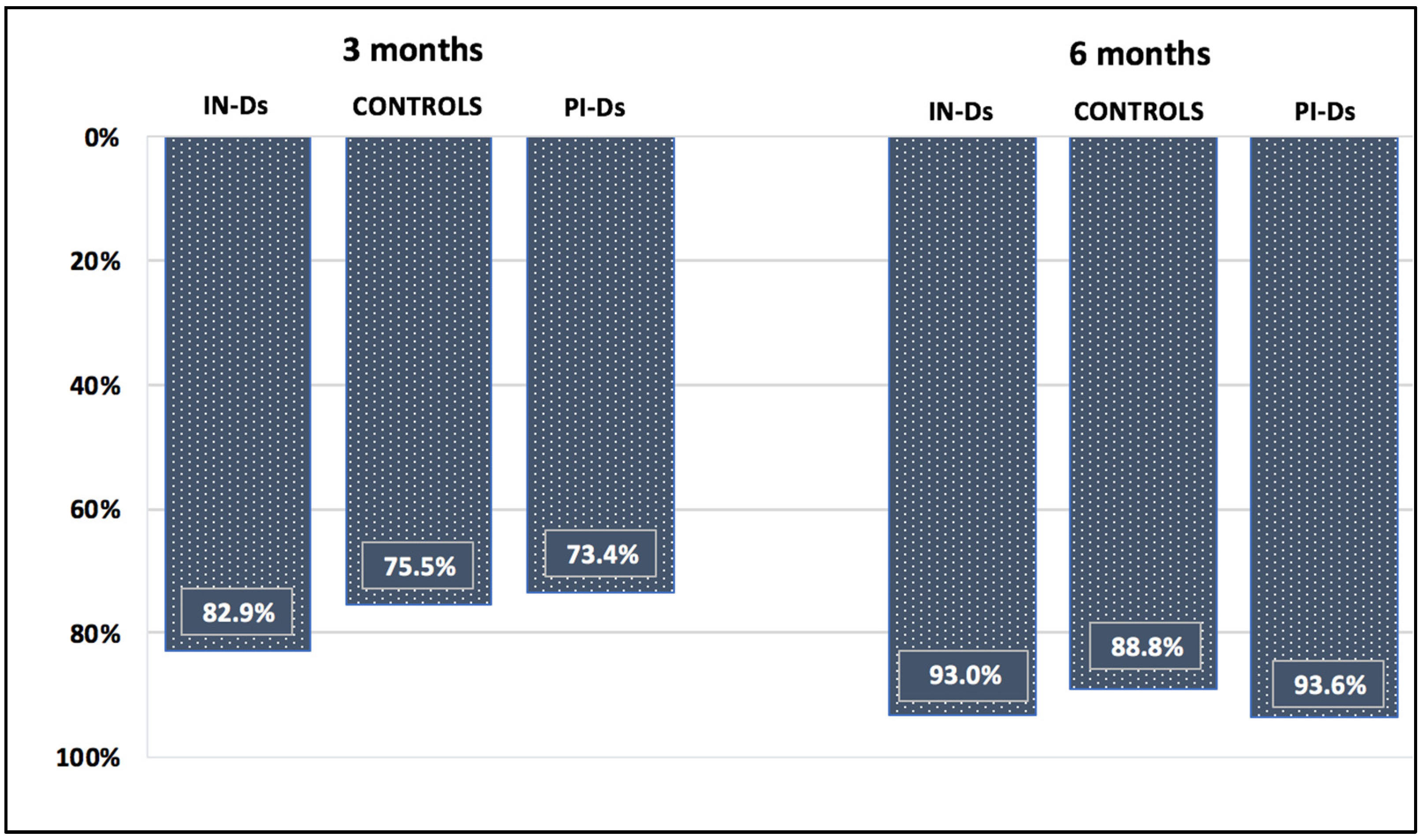
| Seroconversion titer > 33.8 BAU/mL | 3 months | 82.6% | 100% | 100% | 0.05 |
| 6 months | 67.9% | 95% | 96.9% | <0.001 | |
| Protective titer > 141 BAU/mL | 3 months | 47.7% | 95% | 96.9% | <0.001 |
| 6 months | 23.8% | 70% | 93.7% | <0.001 |
| N (%) | Anti-S IgG 2nd Dose | p-Value | Anti-S IgG 3rd Dose | p-Value | |
|---|---|---|---|---|---|
| Age < 70 years Age ≥ 70 | 56 (53.85) 48 (46.15) | 1578 (637–1919) 926 (320–1919) | 0.06 | 4005 (1640–6865) 3460 (1160–7565) | 0.82 |
| Male sex Female sex | 65 (62.5) 39 (37.5) | 1134 (455–1905) 1219 (494–2080) | 0.56 | 3810 (1130–7250) 3490 (1640–7510) | 0.92 |
| CCI ≥ 6.5 CCI < 6.5 | 53 (50.96) 51 (49.04) | 926 (330–1936) 1423 (629–2000) | 0.19 | 3810 (1190–7400) 3490 (1520–7250) | 0.94 |
| BMI ≥ 25.6 kg/m2 BMI < 25.6 | 52 (50) 52 (50) | 1175 (494–1908) 1133 (445–1953) | 0.95 | 3650 (1600–7250) 3795 (1410–7325) | 0.95 |
| Diabetes No diabetes | 36 (34.6) 68 (65.4) | 932 (445–1498) 1336 (494–2033) | 0.11 | 4630 (1160–7990) 3140 (1530–6755) | 0.46 |
| HD patients PD patients | 94 (90.4) 10 (9.6) | 933 (445–1918) 1641 (1578–2080) | 0.08 | 3890 (1400–7250) 2975 (2080–8600) | 0.65 |
| BNT162b2 mRNA-1273 | 96 (92.3) 8 (7.7) | 1154 (475–1952) no data | na | 3890 (1410–7140) 2865 (1680–9595) | 0.68 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biedunkiewicz, B.; Tylicki, L.; Ślizień, W.; Lichodziejewska-Niemierko, M.; Dąbrowska, M.; Kubanek, A.; Rodak, S.; Polewska, K.; Tylicki, P.; Renke, M.; et al. Waning Humoral Response after COVID-19 mRNA Vaccination in Maintenance Dialysis Patients and Recovery after a Complementary Third Dose. Vaccines 2022, 10, 433. https://doi.org/10.3390/vaccines10030433
Biedunkiewicz B, Tylicki L, Ślizień W, Lichodziejewska-Niemierko M, Dąbrowska M, Kubanek A, Rodak S, Polewska K, Tylicki P, Renke M, et al. Waning Humoral Response after COVID-19 mRNA Vaccination in Maintenance Dialysis Patients and Recovery after a Complementary Third Dose. Vaccines. 2022; 10(3):433. https://doi.org/10.3390/vaccines10030433
Chicago/Turabian StyleBiedunkiewicz, Bogdan, Leszek Tylicki, Waldemar Ślizień, Monika Lichodziejewska-Niemierko, Małgorzata Dąbrowska, Alicja Kubanek, Sylwia Rodak, Karolina Polewska, Piotr Tylicki, Marcin Renke, and et al. 2022. "Waning Humoral Response after COVID-19 mRNA Vaccination in Maintenance Dialysis Patients and Recovery after a Complementary Third Dose" Vaccines 10, no. 3: 433. https://doi.org/10.3390/vaccines10030433
APA StyleBiedunkiewicz, B., Tylicki, L., Ślizień, W., Lichodziejewska-Niemierko, M., Dąbrowska, M., Kubanek, A., Rodak, S., Polewska, K., Tylicki, P., Renke, M., & Dębska-Ślizień, A. (2022). Waning Humoral Response after COVID-19 mRNA Vaccination in Maintenance Dialysis Patients and Recovery after a Complementary Third Dose. Vaccines, 10(3), 433. https://doi.org/10.3390/vaccines10030433








