Leveraging Beneficial Off-Target Effects of Live-Attenuated Rotavirus Vaccines
Abstract
1. Introduction
2. Off-Target Effects of Vaccines
3. Mechanisms of Off-Target Effects of Vaccination: Major Lessons from Bacille Calmette Guérin
4. Off-Target Effects from Live and Non-Live Vaccines
5. Sex Differential Off-Target Effects
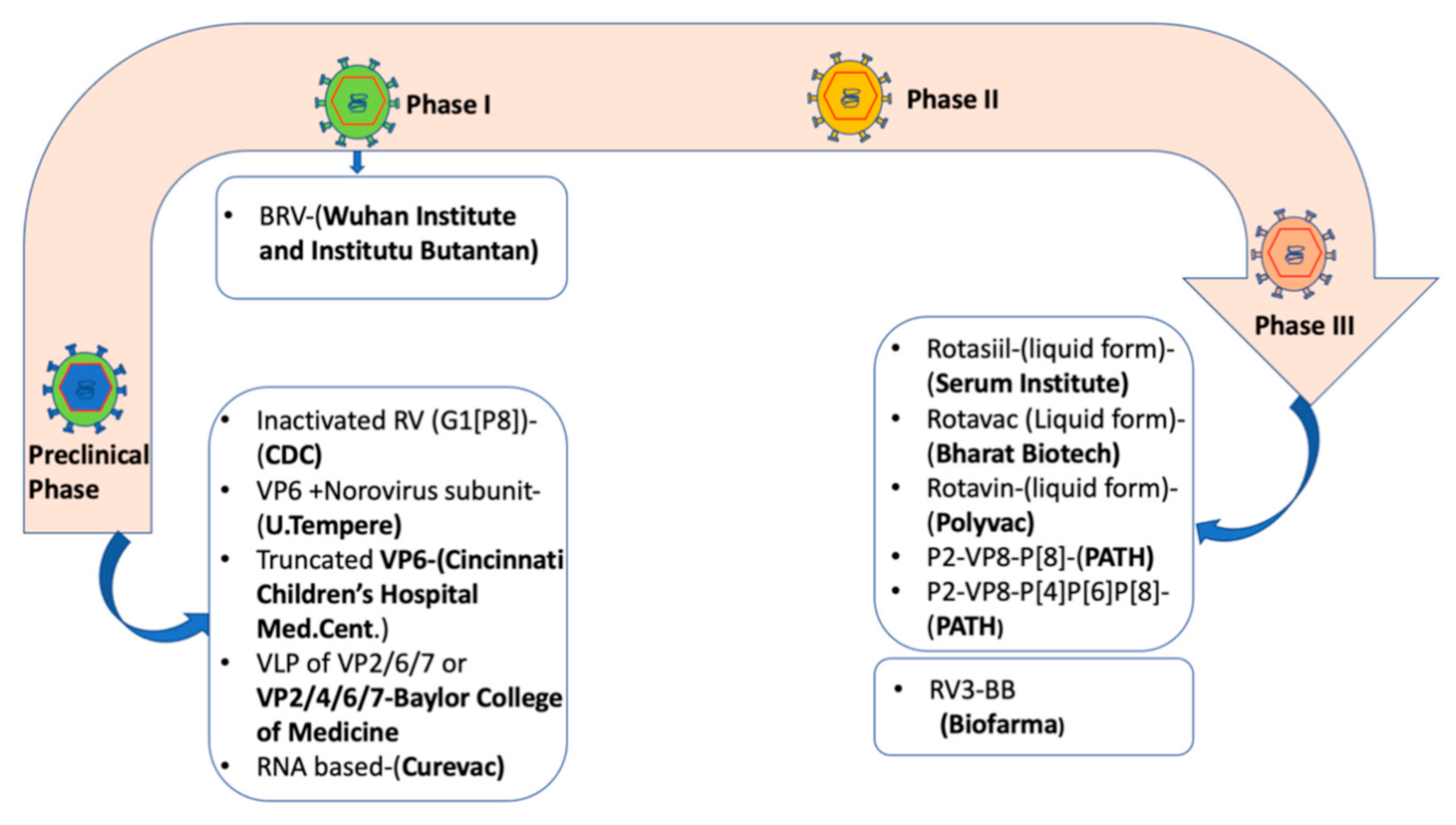
6. Current Rotavirus Vaccines
7. The Need to Improve the Performance of Rotavirus Vaccines
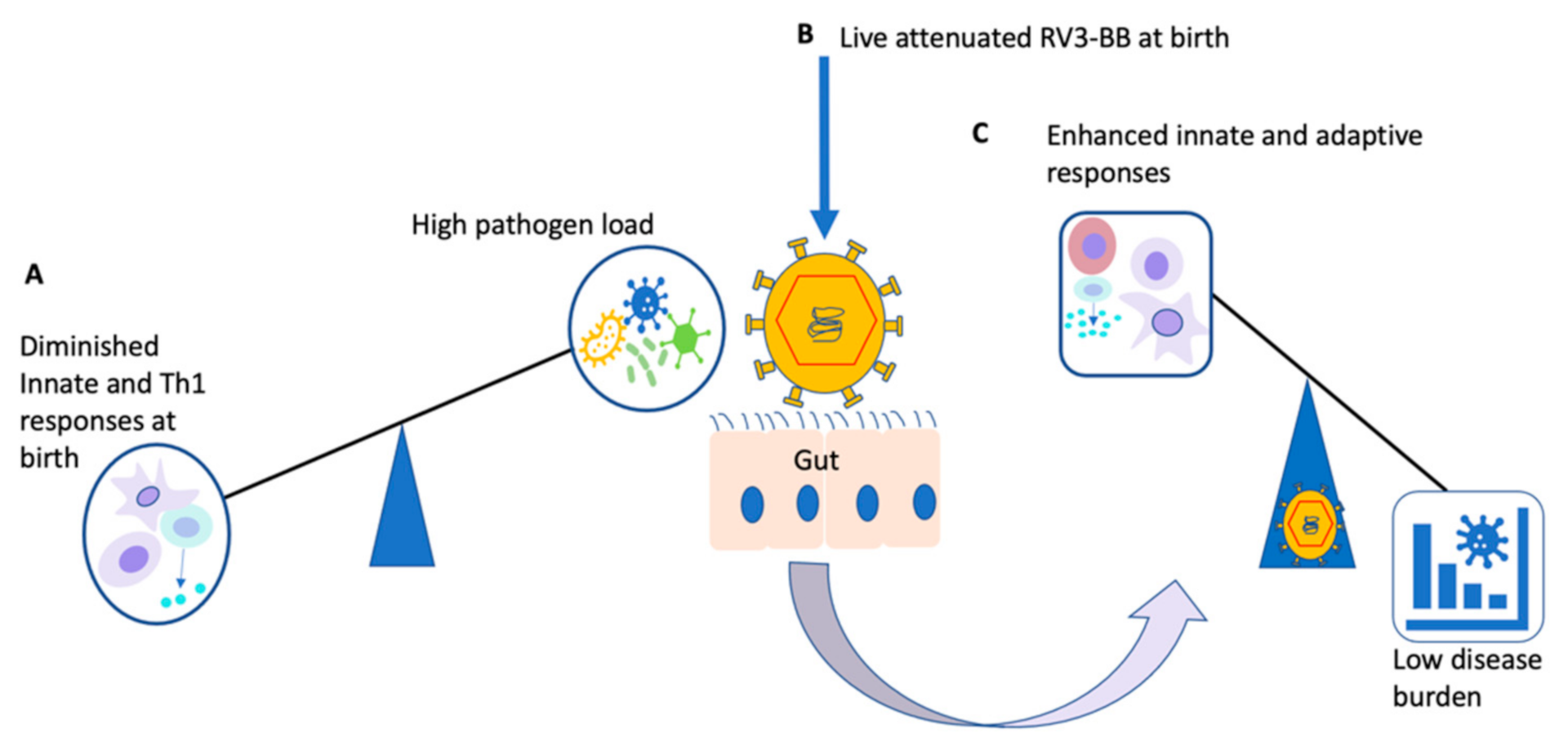
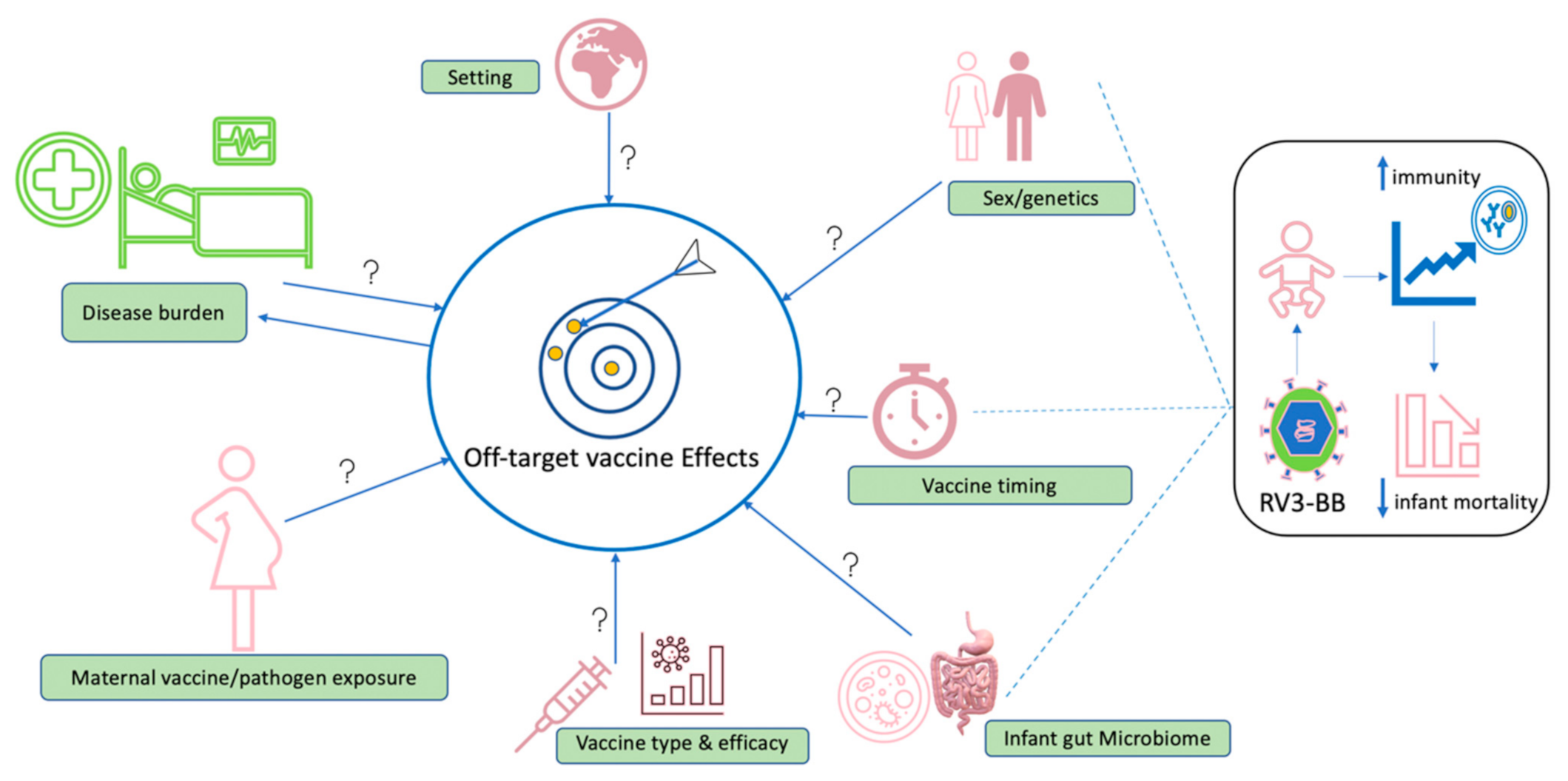
8. Future Considerations
- How long do vaccine-induced off-target effects last?
- To what extent do external factors such as maternal vaccine exposure, birth method and vaccine type influence occurrence of off-target effects?
- What mechanistic approaches can separate vaccine-induced off-target effects versus environmentally induced off-target effects (such as early pathogen exposure, infant gut microbiome, maternal vaccine/pathogen exposure)?
- At what age does sex influence the occurrence of off-target effects?
- Where adequate evidence on sex interaction with vaccine-induced effects is gathered, what is the practicability of developing vaccines tailored to specific sex or about interindividual variations that occur naturally?
- In the context of a neonatal live-attenuated RV3-BB vaccine, can co-administration of RV3-BB with known off-target inducers such as BCG affect induction of anticipated off-target effects?
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Children: Improving Survival and Well-Being. Available online: https://www.who.int/news-room/fact-sheets/detail/children-reducing-mortality (accessed on 11 October 2021).
- Clark, A.; Black, R.; Tate, J.; Roose, A.; Kotloff, K.; Lam, D.; Blackwelder, W.; Parashar, U.; Lanata, C.; Kang, G.; et al. Estimating global, regional and national rotavirus deaths in children aged <5 years. PLoS ONE 2017, 12, e0183392. [Google Scholar] [PubMed]
- WHO. Diarrhoeal Disease. Available online: https://www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease (accessed on 11 October 2021).
- WHO. Vaccines and Immunization 2021. Available online: https://www.who.int/health-topics/vaccines-and-immunization#tab=tab_1 (accessed on 8 December 2021).
- Cárcamo-Calvo, R.; Muñoz, C.; Buesa, J.; Rodríguez-Díaz, J.; Gozalbo-Rovira, R. The rotavirus vaccine landscape, an update. Pathogens 2021, 10, 520. [Google Scholar] [CrossRef] [PubMed]
- Burke, R.M.; Tate, J.E.; Kirkwood, C.D.; Steele, A.D.; Parashar, U.D. Current and new rotavirus vaccines. Curr. Opin. Infect. Dis. 2019, 32, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Troeger, C.; Khalil, I.A.; Rao, P.C.; Cao, S.; Blacker, B.F.; Ahmed, T.; Armah, G.; Bines, J.E.; Brewer, T.G.; Colombara, D.V.; et al. Rotavirus vaccination and the global burden of rotavirus diarrhea among children younger than 5 year. JAMA Pediatr. 2018, 172, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.I. Unexpected benefits of rotavirus vaccination in the United States. J. Infect. Dis. 2011, 204, 975–977. [Google Scholar] [CrossRef] [PubMed]
- Bar-Zeev, N.; King, C.; Phiri, T.; Beard, J.; Mvula, H.; Crampin, A.C.; Heinsbroek, E.; Lewycka, S.; Tate, J.E.; Parashar, U.D.; et al. Impact of monovalent rotavirus vaccine on diarrhoea-associated post-neonatal infant mortality in rural communities in Malawi: A population-based birth cohort study. Lancet Glob. Health 2018, 6, e1036–e1044. [Google Scholar] [CrossRef]
- Lopman, B.A.; Curns, A.T.; Yen, C.; Parashar, U.D. Infant rotavirus vaccination may provide indirect protection to older children and adults in the United States. J. Infect. Dis. 2011, 204, 980–986. [Google Scholar] [CrossRef]
- Uthayakumar, D.; Paris, S.; Chapat, L.; Freyburger, L.; Poulet, H.; De Luca, K. Non-specific Effects of Vaccines Illustrated Through the BCG Example: From Observations to Demonstrations. Front. Immunol. 2018, 9, 2869. [Google Scholar] [CrossRef]
- Shann, F. The nonspecific effects of vaccines and the expanded program on immunization. J. Infect. Dis. 2011, 204, 182–184. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Soares-weiser, K.; López-lópez, J.A.; Kakourou, A.; Chaplin, K.; Christensen, H.; Martin, N.K.; Sterne, J.A.C.; Reingold, A.L. Association of BCG, DTP, and measles containing vaccines with childhood mortality: Systematic review. BMJ 2016, 355, i5170. [Google Scholar] [CrossRef]
- Kleinnijenhuis, J.; Quintin, J.; Preijers, F.; Joosten, L.A.B.; Ifrim, D.C.; Saeed, S.; Jacobs, C.; Van Loenhout, J.; De Jong, D.; Hendrik, S.; et al. Bacille Calmette-Guérin induces NOD2-dependent nonspecific protection from reinfection via epigenetic reprogramming of monocytes. Proc. Natl. Acad. Sci. USA 2012, 109, 17537–17542. [Google Scholar] [CrossRef] [PubMed]
- de Bree, L.C.J.; Koeken, V.A.C.M.; Joosten, L.A.B.; Aaby, P.; Benn, C.S.; van Crevel, R.; Netea, M.G. Non-specific effects of vaccines: Current evidence and potential implications. Semin. Immunol. 2018, 39, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Shann, F. Nonspecific Effects of Vaccines and the Reduction of Mortality in Children. Clin. Ther. 2013, 35, 109–114. [Google Scholar] [CrossRef] [PubMed]
- WHO. Meeting of Global Advisory Committee on Vaccine Safety. Wkly. Epidemiol. Rec. 2008, 83, 287–292. [Google Scholar]
- Pennington, S.H.; Ferreira, D.M.; Caamaño-Gutiérrez, E.; Reiné, J.; Hewitt, C.; Hyder-Wright, A.D.; Gordon, S.B.; Gordon, M.A. Nonspecific effects of oral vaccination with live-attenuated Salmonella Typhi strain Ty21a. Sci. Adv. 2020, 5, eaau6849. [Google Scholar] [CrossRef] [PubMed]
- Aaby, P.; Benn, C.S.; Flanagan, K.L.; Klein, S.L.; Kollmann, T.R.; Lynn, D.J.; Shann, F. The non-specific and sex-differential effects of vaccines. Nat. Rev. Immunol. 2020, 20, 464–470. [Google Scholar] [CrossRef]
- Sankoh, O.; Welaga, P.; Debpuur, C.; Zandoh, C.; Gyaase, S.; Poma, M.A.; Mutua, M.K.; Hanifi, S.M.M.A.; Martins, C.; Nebie, E.; et al. The non-specific effects of vaccines and other childhood interventions: The contribution of INDEPTH Health and Demographic Surveillance Systems. Int. J. Epidemiol. 2014, 43, 645–653. [Google Scholar] [CrossRef]
- Aaby, P.; Martins, C.L.; Garly, M.L.; Balé, C.; Andersen, A.; Rodrigues, A.; Ravn, H.; Lisse, I.M.; Benn, C.S.; Whittle, H.C. Non-specific effects of standard measles vaccine at 4.5 and 9 months of age on childhood mortality: Randomised controlled trial. BMJ 2010, 341, 1262. [Google Scholar] [CrossRef]
- Roth, A.E.; Benn, C.S.; Ravn, H.; Rodrigues, A.; Lisse, I.M.; Yazdanbakhsh, M.; Whittle, H.; Aaby, P. Effect of revaccination with BCG in early childhood on mortality: Randomised trial in Guinea-Bissau. BMJ 2010, 340, 749. [Google Scholar] [CrossRef]
- Moorlag, S.J.C.F.M.; Arts, R.J.W.; van Crevel, R.; Netea, M.G. Non-specific effects of BCG vaccine on viral infections. Clin. Microbiol. Infect. 2019, 25, 1473–1478. [Google Scholar] [CrossRef]
- De Groot, A.S.; Ardito, M.; McClaine, E.M.; Moise, L.; Martin, W.D. Immunoinformatic comparison of T-cell epitopes contained in novel swine-origin influenza A (H1N1) virus with epitopes in 2008-2009 conventional influenza vaccine. Vaccine 2009, 27, 5740–5747. [Google Scholar] [CrossRef] [PubMed]
- Saadatian-Elahi, M.; Aaby, P.; Shann, F.; Netea, M.G.; Levy, O.; Louis, J.; Picot, V.; Greenberg, M.; Warren, W. Heterologous vaccine effects. Vaccine 2016, 34, 3923–3930. [Google Scholar] [CrossRef]
- Goodridge, H.S.; Ahmed, S.S.; Curtis, N.; Kollmann, T.R.; Levy, O.; Netea, M.G.; Pollard, A.J.; Van Crevel, R.; Wilson, C.B. Harnessing the beneficial heterologous effects of vaccination. Nat. Rev. Immunol. 2016, 16, 392–400. [Google Scholar] [CrossRef]
- Kleinnijenhuis, J.; Quintin, J.; Preijers, F.; Benn, C.S.; Joosten, L.A.B.; Jacobs, C.; Van Loenhout, J.; Xavier, R.J.; Aaby, P.; Van Der Meer, J.W.M.; et al. Long-lasting effects of bcg vaccination on both heterologous th1/th17 responses and innate trained immunity. J. Innate Immun. 2014, 6, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.G.; Kleinnijenhuis, J.; Netea, M.G.; Dockrell, H.M. Whole Blood Profiling of Bacillus Calmette–Guérin-Induced Trained Innate Immunity in Infants Identifies Epidermal Growth Factor, IL-6, Platelet-Derived Growth Factor-AB/BB, and Natural Killer Cell Activation. Front. Immunol. 2017, 8, 644. [Google Scholar] [CrossRef] [PubMed]
- Ritz, N.; Mui, M.; Balloch, A.; Curtis, N. Non-specific effect of Bacille Calmette-Guérin vaccine on the immune response to routine immunisations. Vaccine 2013, 31, 3098–3103. [Google Scholar] [CrossRef]
- Kollmann, T.R.; Kampmann, B.; Mazmanian, S.K.; Marchant, A.; Levy, O. Protecting the Newborn and Young Infant from Infectious Diseases: Lessons from Immune Ontogeny. Immunity 2017, 46, 350–363. [Google Scholar] [CrossRef]
- Brook, B.; Harbeson, D.J.; Shannon, C.P.; Cai, B.; He, D.; Ben-Othman, R.; Francis, F.; Huang, J.; Varankovich, N.; Liu, A.; et al. BCG vaccination-induced emergency granulopoiesis provides rapid protection from neonatal sepsis. Sci. Transl. Med. 2020, 12, eaax4517. [Google Scholar] [CrossRef]
- Cuenca, A.G.; Cuenca, A.L.; Gentile, L.F.; Efron, P.A.; Islam, S.; Moldawer, L.L.; Kays, D.W.; Larson, S.D. Delayed emergency myelopoiesis following polymicrobial sepsis in neonates. Innate Immun. 2015, 21, 386–391. [Google Scholar] [CrossRef]
- Netea, M.G.; Joosten, L.A.B.; Latz, E.; Mills, K.H.G.; Stunnenberg, H.G.; Neill, L.A.J.O.; Xavier, R.J. Trained immunity: A programme of innate immune memory in health and disease. Science 2017, 352, aaf1098. [Google Scholar] [CrossRef]
- Schnack, L.; Sohrabi, Y.; Lagache, S.M.M.; Kahles, F.; Bruemmer, D.; Waltenberger, J.; Findeisen, H.M. Mechanisms of trained innate immunity in oxLDL primed human coronary smooth muscle cells. Front. Immunol. 2019, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Naik, S.; Larsen, S.B.; Gomez, N.C.; Alaverdyan, K.; Yuan, S.; Polak, L.; Kulukian, A.; Chai, S.; Fuchs, E. Inflammatory memory sensitizes skin epithelial stem cells to tissue damage. Nature 2018, 550, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Sun, Y.; Drummer, C.; Nanayakkara, G.K.; Shao, Y.; Saaoud, F.; Johnson, C.; Zhang, R.; Yu, D.; Li, X.; et al. Increased acetylation of H3K14 in the genomic regions that encode trained immunity enzymes in lysophosphatidylcholine-activated human aortic endothelial cells—Novel qualification markers for chronic disease risk factors and conditional DAMPs. Redox Biol. 2019, 24, 101221. [Google Scholar] [CrossRef]
- Hamada, A.; Torre, C.; Drancourt, M.; Ghigo, E. Trained immunity carried by non-immune cells. Front. Microbiol. 2019, 10, 3225. [Google Scholar] [CrossRef]
- dos Santos, J.C.; Barroso de Figueiredo, A.M.; Teodoro Silva, M.V.; Cirovic, B.; de Bree, L.C.J.; Damen, M.S.M.A.; Moorlag, S.J.C.F.M.; Gomes, R.S.; Helsen, M.M.; Oosting, M.; et al. β-Glucan-Induced Trained Immunity Protects against Leishmania braziliensis Infection: A Crucial Role for IL-32. Cell Rep. 2019, 28, 2659–2672. [Google Scholar] [CrossRef] [PubMed]
- Milutinović, B.; Kurtz, J. Immune memory in invertebrates. Semin. Immunol. 2016, 28, 328–342. [Google Scholar] [CrossRef]
- Melillo, D.; Marino, R.; Italiani, P.; Boraschi, D. Innate Immune Memory in Invertebrate Metazoans: A Critical Appraisal. Front. Immunol. 2018, 9, 1915. [Google Scholar] [CrossRef]
- Sohrabi, Y.; Dos Santos, J.C.; Dorenkamp, M.; Findeisen, H.; Godfrey, R.; Netea, M.G.; Joosten, L.A.B. Trained immunity as a novel approach against COVID-19 with a focus on Bacillus Calmette–Guérin vaccine: Mechanisms, challenges and perspectives. Clin. Transl. Immunol. 2020, 9, e1228. [Google Scholar] [CrossRef]
- Bryant, C.; Kerr, I.D.; Debnath, M.; Kenny, K.H.A.; Ratnam, J.; Ferreira, R.S.; Jaishankar, P.; Zhao, D.; Arkin, M.R.; McKerrow, J.H.; et al. Candida albicans Infection Affords Protection against Reinfection via Functional Reprogramming of Monocytes. Cell Host Microbe 2020, 19, 6218–6221. [Google Scholar] [CrossRef]
- Arts, R.J.W.; Moorlag, S.J.C.F.M.; Novakovic, B.; Li, Y.; Wang, S.Y.; Oosting, M.; Kumar, V.; Xavier, R.J.; Wijmenga, C.; Joosten, L.A.B.; et al. BCG Vaccination Protects against Experimental Viral Infection in Humans through the Induction of Cytokines Associated with Trained Immunity. Cell Host Microbe 2018, 23, 89–100. [Google Scholar] [CrossRef]
- Bai, W.; Liu, H.; Ji, Q.; Zhou, Y.; Liang, L.; Zheng, R.; Chen, J.; Liu, Z.; Yang, H.; Zhang, P.; et al. TLR3 regulates mycobacterial RNA-induced IL-10 production through the PI3K/AKT signaling pathway. Cell. Signal. 2014, 26, 942–950. [Google Scholar] [CrossRef] [PubMed]
- Speth, M.T.; Repnik, U.; Müller, E.; Spanier, J.; Kalinke, U.; Corthay, A.; Griffiths, G. Poly(I:C)-Encapsulating Nanoparticles Enhance Innate Immune Responses to the Tuberculosis Vaccine Bacille Calmette-Guérin (BCG) via Synergistic Activation of Innate Immune Receptors. Mol. Pharm. 2017, 14, 4098–4112. [Google Scholar] [CrossRef] [PubMed]
- Aaby, P.; Roth, A.; Ravn, H.; Napirna, B.M.; Rodrigues, A.; Lisse, I.M.; Stensballe, L.; Diness, B.R.; Lausch, K.R.; Lund, N.; et al. Randomized trial of BCG vaccination at birth to low-birth-weight children: Beneficial nonspecific effects in the neonatal period? J. Infect. Dis. 2011, 204, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Biering-Sørensen, S.; Aaby, P.; Napirna, B.M.; Roth, A.; Ravn, H.; Rodrigues, A.; Whittle, H.; Benn, C.S. Small randomized trial among low-birth-weight children receiving bacillus Calmette-Guéerin vaccination at first health center contact. Pediatr. Infect. Dis. J. 2012, 31, 306–308. [Google Scholar] [CrossRef]
- Aaby, P.; Ravn, H.; Roth, A.; Rodrigues, A.; Lisse, I.M.; Diness, B.R.; Lausch, K.R.; Lund, N.; Rasmussen, J.; Biering-Sørensen, S.; et al. Early diphtheria-tetanus-pertussis vaccination associated with higher female mortality and no difference in male mortality in a cohort of low birthweight children: An observational study within a randomised trial. Arch. Dis. Child. 2012, 97, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Aaby, P.; Jensen, H.; Gomes, J.; Fernandes, M.; Lisse, I.M. The introduction of diphtheria-tetanus-pertussis vaccine and child mortality in rural Guinea-Bissau: An observational study. Int. J. Epidemiol. 2004, 33, 374–380. [Google Scholar] [CrossRef]
- Shann, F. Commentary: BCG vaccination halves neonatal mortality. Pediatr. Infect. Dis. J. 2012, 31, 308–309. [Google Scholar] [CrossRef]
- Bardenheier, B.H.; McNeil, M.M.; Wodi, A.P.; McNicholl, J.M.; DeStefano, F. Risk of non-targeted infectious disease hospitalizations among U.S. children following inactivated and live vaccines, 2005–2014. Clin. Infect. Dis. 2017, 65, 729–737. [Google Scholar] [CrossRef]
- Bucardo, F.; Reyes, Y.; Svensson, L.; Nordgren, J. Predominance of norovirus and sapovirus in nicaragua after implementation of universal rotavirus vaccination. PLoS ONE 2014, 9, e98201. [Google Scholar] [CrossRef]
- Becker-Dreps, S.; Cuthbertson, C.C.; Bucardo, F.; Vinje, J.; Paniagua, M.; Giebultowicz, S.; Espinoza, F.; Emch, M. Environmental factors associated with childhood norovirus diarrhoea in León, Nicaragua. Epidemiol. Infect. 2017, 145, 1597–1605. [Google Scholar] [CrossRef][Green Version]
- Iturriza-Gómara, M.; Jere, K.C.; Hungerford, D.; Bar-Zeev, N.; Shioda, K.; Kanjerwa, O.; Houpt, E.R.; Operario, D.J.; Wachepa, R.; Pollock, L.; et al. Etiology of Diarrhea among Hospitalized Children in Blantyre, Malawi, following Rotavirus Vaccine Introduction: A Case-Control Study. J. Infect. Dis. 2019, 220, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Burnett, E.; Parashar, U.; Tate, J. Rotavirus Vaccines: Effectiveness, Safety, and Future Directions. Pediatr. Drugs 2018, 20, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Organizations, W.H.; Record, W.E.; States, M.; Group, S.A.; Africa, S. Rotavirus vaccines WHO position paper: January 2013—Recommendations. Vaccine 2013, 31, 6170–6171. [Google Scholar] [CrossRef]
- Aaby, P.; Benn, C.; Nielsen, J.; Lisse, I.M.; Rodrigues, A.; Ravn, H. Testing the hypothesis that diphtheria–tetanus–pertussis vaccine has negative non-specific and sex-differential effects on child survival in high-mortality countries. BMJ Open 2012, 2, e000707. [Google Scholar] [CrossRef]
- Aaby, P.; Benn, C.S. Developing the concept of beneficial non-specific effect of live vaccines with epidemiological studies. Clin. Microbiol. Infect. 2019, 25, 1459–1467. [Google Scholar] [CrossRef]
- Aaby, P.; Kollmann, T.R.; Benn, C.S. Nonspecific effects of neonatal and infant vaccination: Public-health, immunological and conceptual challenges. Nat. Immunol. 2014, 15, 895–900. [Google Scholar] [CrossRef]
- Aaby, P.; Jensen, H.; Samb, B.; Cisse, B.; Sodemann, M.; Jakobsen, M.; Poulsen, A.; Rodrigues, A.; Lisse, I.M.; Simondon, F.; et al. Differences in female-male mortality after high-titre measles vaccine and association with subsequent vaccination with diphtheria-tetanus-pertussis and inactivated poliovirus: Reanalysis of West African studies. Lancet 2003, 361, 2183–2189. [Google Scholar] [CrossRef]
- Aaby, P.; Garly, M.L.; Nielsen, J.; Ravn, H.; Martins, C.; Balé, C.; Rodrigues, A.; Benn, C.S.; Lisse, I.M. Increased female-male mortality ratio associated with inactivated polio and diphtheria-tetanus-pertussis vaccines: Observations from vaccination trials in Guinea-Bissau. Pediatr. Infect. Dis. J. 2007, 26, 247–252. [Google Scholar] [CrossRef]
- Benn, C.S.; Fisker, A.B.; Rieckmann, A.; Sørup, S.; Aaby, P. Personal View Vaccinology: Time to change the paradigm? Lancet Infect. Dis. 2020, 20, e274–e283. [Google Scholar] [CrossRef]
- Klein, S.L.; Shann, F.; Moss, W.J.; Benn, C.S.; Aabye, P. RTS, S malaria vaccine and increased mortality in girls. mBio 2016, 7, e00514-16. [Google Scholar] [CrossRef]
- WHO. Meeting of the strategic Advisory Group of Experts on Immunization. Wkly. Epidemiol. Rec. 2014, 21, 221–236. [Google Scholar]
- Noho-Konteh, F.; Adetifa, J.U.; Cox, M.; Hossin, S.; Reynolds, J.; Le, M.T.; Sanyang, L.C.; Drammeh, A.; Plebanski, M.; Forster, T.; et al. Sex-Differential Non-Vaccine-Specific Immunological Effects of Diphtheria-Tetanus-Pertussis and Measles Vaccination. Clin. Infect. Dis. 2016, 63, 1213–1226. [Google Scholar] [CrossRef]
- Saso, A.; Kampmann, B. Vaccine responses in newborns. Semin. Immunopathol. 2017, 39, 627–642. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, K.L.; Klein, S.L.; Skakkebaek, N.E.; Marriott, I.; Marchant, A.; Selin, L.; Fish, E.N.; Prentice, A.M.; Whittle, H.; Benn, C.S.; et al. Sex differences in the vaccine-specific and non-targeted effects of vaccines. Vaccine 2011, 29, 2349–2354. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, K.L.; Fink, A.L.; Plebanski, M.; Klein, S.L. Sex and gender differences in the outcomes of vaccination over the life course. Annu. Rev. Cell Dev. Biol. 2017, 33, 577–599. [Google Scholar] [CrossRef]
- Fink, A.L.; Engle, K.; Ursin, R.L.; Tang, W.Y.; Klein, S.L. Biological sex affects vaccine efficacy and protection against influenza in mice. Proc. Natl. Acad. Sci. USA 2018, 115, 12477–12482. [Google Scholar] [CrossRef]
- Potluri, T.; Fink, A.L.; Sylvia, K.E.; Dhakal, S.; Vermillion, M.S.; vom Steeg, L.; Deshpande, S.; Narasimhan, H.; Klein, S.L. Age-associated changes in the impact of sex steroids on influenza vaccine responses in males and females. NPJ Vaccines 2019, 4, 29. [Google Scholar] [CrossRef]
- Bishop, R.F.; Barnes, G.L.; Cipriani, E.; Lund, J.S. Clinical Immunity after Neonatal Rotavirus Infection—A Prospective Longitudinal Study in Young Children. N. Engl. J. Med. 1983, 309, 72–76. [Google Scholar] [CrossRef]
- Simonsen, L.; Viboud, C.; Elixhauser, A.; Taylor, R.J.; Kapikian, A.Z. More on RotaShield and intussusception: The role of age at the time of vaccination. J. Infect. Dis. 2005, 192 (Suppl. 1), S36–S43. [Google Scholar] [CrossRef]
- Rennels, M.; Parashar, U.; Holman, R.; Le, C.; Chang, H.; Glass, R. Lack Of an Apparent Association Between Intussusception and wild or vaccine Rotavirus infection. Pediatr. Infect. Dis. J. 1998, 17, 924–925. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.M. Rotavirus Vaccines: Why Continued Investment in Research Is Necessary. Curr. Clin. Microbiol. Rep. 2018, 5, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Grant, L.; Watt, J.; Moulton, L.; Weatherholtz, R.; Reid, R.; Santosham, M.; O’Brien, K. Lack of nonspecific protection against all-cause nonrotavirus gastroenteritis by vaccination with orally administered rotavirus vaccine. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 635–640. [Google Scholar] [CrossRef] [PubMed]
- King, C.; Bar-Zeev, N.; Phiri, T.; Beard, J.; Mvula, H.; Crampin, A.; Heinsbroek, E.; Hungerford, D.; Lewckya, S.; Verani, J.; et al. Population impact and effectiveness of sequential 13-valent pneumococcal conjugate and monovalent rotavirus vaccine introduction on infant mortality: Prospective birth cohort studies from Malawi. BMJ Glob. Health 2020, 5, e002669. [Google Scholar] [CrossRef]
- Bergman, H.; Henschke, N.; Hungerford, D.; Pitan, F.; Ndwandwe, D.; Cunliffe, N.; Soares-Weiser, K. Vaccines for preventing rotavirus diarrhoea: Vaccines in use. Cochrane Database Syst. Rev. 2021, 2021, CD008521. [Google Scholar] [CrossRef]
- Clarke, E.; Desselberger, U. Correlates of protection against human rotavirus disease and the factors influencing protection in low-income settings. Mucosal Immunol. 2015, 8, 1–17. [Google Scholar] [CrossRef]
- Bines, J.E.; Kotloff, K.L. Next-generation rotavirus vaccines: Important progress but work still to be done. Lancet Infect. Dis. 2020, 20, 762–764. [Google Scholar] [CrossRef]
- Fix, A.; Kirkwood, C.D.; Steele, D.; Flores, J. Next-generation rotavirus vaccine developers meeting: Summary of a meeting sponsored by PATH and the bill & melinda gates foundation (19–20 June 2019, Geneva). Vaccine 2020, 38, 8247–8254. [Google Scholar] [CrossRef]
- Bishop, R.F.; Tzipori, S.R.; Coulson, B.S.; Unicomb, L.E.; Albert, M.J.; Barnes, G.L. Heterologous protection against rotavirus-induced disease in gnotobiotic piglets. J. Clin. Microbiol. 1986, 24, 1023–1028. [Google Scholar] [CrossRef] [PubMed]
- Bines, J.E.; Danchin, M.; Jackson, P.; Handley, A.; Watts, E.; Lee, K.J.; West, A.; Cowley, D.; Chen, M.Y.; Barnes, G.L.; et al. Safety and immunogenicity of RV3-BB human neonatal rotavirus vaccine administered at birth or in infancy: A randomised, double-blind, placebo-controlled trial. Lancet Infect. Dis. 2015, 15, 1389–1397. [Google Scholar] [CrossRef]
- Segovia, M.J.G. Human neonatal rotavirus vaccine (RV3-BB) to target rotavirus from birth. Acta Pediatr. Esp. 2018, 76, 62–63. [Google Scholar] [CrossRef]
- Boniface, K.; Byars, S.G.; Cowley, D.; Kirkwood, C.D.; Bines, J.E. Human neonatal rotavirus vaccine (RV3-BB) produces vaccine take irrespective of histo-blood group antigen status. J. Infect. Dis. 2020, 221, 1070–1078. [Google Scholar] [CrossRef] [PubMed]
- Pollock, L.; Bennett, A.; Jere, K.C.; Dube, Q.; Mandolo, J.; Bar-Zeev, N.; Heyderman, R.S.; Cunliffe, N.A.; Iturriza-Gomara, M. Nonsecretor Histo-blood Group Antigen Phenotype Is Associated with Reduced Risk of Clinical Rotavirus Vaccine Failure in Malawian Infants. Clin. Infect. Dis. 2019, 69, 1313–1319. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Hagbom, M.; Svensson, L.; Nordgren, J. The impact of human genetic polymorphisms on rotavirus susceptibility, epidemiology, and vaccine take. Viruses 2020, 12, 324. [Google Scholar] [CrossRef]
- Witte, D.A. Neonatal Rotavirus Vaccine (RV3-BB) immunogenicity and safety in a neonatal and infant administration schedule in Malawi: A randomized, double-blind, four-arm parallel group dose-ranging study. Lancet Infect. Dis. 2022. [Google Scholar] [CrossRef]
- Witte, D.; Handley, A.; Jere, K.C.; Bogandovic-Sakran, N.; Mpakiza, A.; Turner, A.; Pavlic, D.; Boniface, K.; Mandolo, J.; Suryawijaya Ong, D.; et al. Neonatal Rotavirus Vaccine (RV3-BB) Immunogenicity and Safety in a Neonatal and Infant Administration Schedule in Malawi: A Randomized, Double-Blind, Four-Arm Parallel Group Dose-Ranging Study. SSRN Electron. J. 2021, 3099, 1–11. [Google Scholar] [CrossRef]
- Bar-Zeev, N.; Kapanda, L.; Tate, J.E.; Jere, K.C.; Iturriza-Gomara, M.; Nakagomi, O.; Mwansambo, C.; Costello, A.; Parashar, U.D.; Heyderman, R.S.; et al. Effectiveness of a monovalent rotavirus vaccine in infants in Malawi after programmatic roll-out: An observational and case-control study. Lancet Infect. Dis. 2015, 15, 422–428. [Google Scholar] [CrossRef]
- Mandolo, J.J.; Henrion, M.Y.R.; Mhango, C.; Chinyama, E.; Wachepa, R.; Kanjerwa, O.; Malamba-Banda, C.; Shawa, I.T.; Hungerford, D.; Kamng’ona, A.W.; et al. Reduction in severity of all-cause gastroenteritis requiring hospitalisation in children vaccinated against rotavirus in Malawi. Viruses 2021, 13, 2491. [Google Scholar] [CrossRef]
- Hungerford, D.J.; French, N.; Iturriza-Gómara, M.; Read, J.M.; Cunliffe, N.A.; Vivancos, R. Reduction in hospitalisations for acute gastroenteritis-associated childhood seizures since introduction of rotavirus vaccination: A time-series and change-point analysis of hospital admissions in England. J. Epidemiol. Community Health 2019, 73, 1020–1025. [Google Scholar] [CrossRef]
- Lee, Y.J.; Lee, J.Y.; Jang, Y.H.; Seo, S.U.; Chang, J.; Seong, B.L. Non-specific effect of vaccines: Immediate protection against respiratory syncytial virus infection by a live attenuated influenza vaccine. Front. Microbiol. 2018, 9, 83. [Google Scholar] [CrossRef]
- Seo, S.U.; Lee, K.H.; Byun, Y.H.; Kweon, M.N.; Seong, B.L. Immediate and broad-spectrum protection against heterologous and heterotypic lethal challenge in mice by live influenza vaccine. Vaccine 2007, 25, 8067–8076. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, S.; Joshi, S.R.; Ueda, I.; Wilson, J.; Blevins, T.P.; Siconolfi, B.; Meng, H.; Devine, L.; Raddassi, K.; Tsang, S.; et al. Prolonged proinflammatory cytokine production in monocytes modulated by interleukin 10 after influenza vaccination in older adults. J. Infect. Dis. 2015, 211, 1174–1184. [Google Scholar] [CrossRef]
- Lanthier, P.A.; Huston, G.E.; Moquin, A.; Eaton, S.M.; Szaba, F.M.; Kummer, L.W.; Tighe, M.P.; Kohlmeier, J.E.; Blair, P.J.; Broderick, M.; et al. Live attenuated influenza vaccine (LAIV) impacts innate and adaptive immune responses. Vaccine 2011, 29, 7849–7856. [Google Scholar] [CrossRef]
- Koyama, S.; Ishii, K.J.; Kumar, H.; Tanimoto, T.; Coban, C.; Uematsu, S.; Kawai, T.; Akira, S. Differential Role of TLR- and RLR-Signaling in the Immune Responses to Influenza A Virus Infection and Vaccination. J. Immunol. 2007, 179, 4711–4720. [Google Scholar] [CrossRef] [PubMed]
- Lund, J.M.; Alexopoulou, L.; Sato, A.; Karow, M.; Adams, N.C.; Gale, N.W.; Iwasaki, A.; Flavell, R.A. Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc. Natl. Acad. Sci. USA 2004, 101, 5598–5603. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Tang, Y.; Huang, G. Innate immune responses in RNA viral infection. Front. Med. 2021, 15, 333–346. [Google Scholar] [CrossRef] [PubMed]
- Kelvin, A.A.; Zambon, M. Influenza imprinting in childhood and the influence on vaccine response later in life. Eurosurveillance 2019, 24, 1900720. [Google Scholar] [CrossRef]
- Freyne, B.; Messina, N.L.; Donath, S.; Germano, S.; Bonnici, R.; Gardiner, K.; Casalaz, D.; Robins-Browne, R.M.; Netea, M.G.; Flanagan, K.L.; et al. Neonatal BCG Vaccination Reduces Interferon-γResponsiveness to Heterologous Pathogens in Infants from a Randomized Controlled Trial. J. Infect. Dis. 2020, 221, 1999–2009. [Google Scholar] [CrossRef]
- Dowling, D.J.; Levy, O. Ontogeny of early life immunity. Trends Immunol. 2014, 35, 299–310. [Google Scholar] [CrossRef]
- Morris, M.C.; Surendran, N. Neonatal vaccination: Challenges and intervention strategies. Neonatology 2016, 109, 161–169. [Google Scholar] [CrossRef]
- Burt, T.D. Fetal Regulatory T Cells and Peripheral Immune Tolerance In Utero: Implications for Development and Disease. Am. J. Reprod. Immunol. 2013, 69, 346–358. [Google Scholar] [CrossRef] [PubMed]
- Boer, M.C.; Joosten, S.A.; Ottenhoff, T.H.M. Regulatory T-cells at the interface between human host and pathogens in infectious diseases and vaccination. Front. Immunol. 2015, 6, 217. [Google Scholar] [CrossRef]
- Pulendran, B.; Arunachalam, P.S.; O’Hagan, D.T. Emerging concepts in the science of vaccine adjuvants. Nat. Rev. Drug Discov. 2021, 20, 454–475. [Google Scholar] [CrossRef] [PubMed]
- Twitchell, E.L.; Tin, C.; Wen, K.; Zhang, H.; Becker-Dreps, S.; Azcarate-Peril, M.A.; Vilchez, S.; Li, G.; Ramesh, A.; Weiss, M.; et al. Modeling human enteric dysbiosis and rotavirus immunity in gnotobiotic pigs. Gut Pathog. 2016, 8, 51. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Donovan, S.M. Human microbiota-associated swine: Current progress and future opportunities. ILAR J. 2015, 56, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Tokuhara, D.; Hikita, N. Cord blood-based approach to assess candidate vaccine adjuvants designed for neonates and infants. Vaccines 2021, 9, 95. [Google Scholar] [CrossRef] [PubMed]
| Product Name | Manufacturer | Composition | Doses | Route of Administration | Regulatory Status |
|---|---|---|---|---|---|
| Rotarix® | GSK | G1P[8]-Live attenuated | 2 | Oral | WHO prequalified |
| Rotavac® | Bharat Biologicals | G9[P11]-Live attenuated | 3 | Oral | WHO prequalified |
| RotaTeq® | Merck | G1, G2, G3, G4, P[8]-Live attenuated | 3 | Oral | WHO prequalified |
| Rotasiil® | Serum Institute of India | G1, G2, G3, G4, G9-Live attenuated | 3 | Oral | WHO prequalified |
| Rotavin-M1® | POLYVAC | G1P[8]-Live attenuated | 3 | Oral | Nationally licensed |
| Lanzhou Lamb® | Lanzhou Institute of Biological Products | G10P[12]-Live attenuated | 1 | Oral | Nationally licensed |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benedicto-Matambo, P.; Bines, J.E.; Malamba-Banda, C.; Shawa, I.T.; Barnes, K.; Kamng’ona, A.W.; Hungerford, D.; Jambo, K.C.; Iturriza-Gomara, M.; Cunliffe, N.A.; et al. Leveraging Beneficial Off-Target Effects of Live-Attenuated Rotavirus Vaccines. Vaccines 2022, 10, 418. https://doi.org/10.3390/vaccines10030418
Benedicto-Matambo P, Bines JE, Malamba-Banda C, Shawa IT, Barnes K, Kamng’ona AW, Hungerford D, Jambo KC, Iturriza-Gomara M, Cunliffe NA, et al. Leveraging Beneficial Off-Target Effects of Live-Attenuated Rotavirus Vaccines. Vaccines. 2022; 10(3):418. https://doi.org/10.3390/vaccines10030418
Chicago/Turabian StyleBenedicto-Matambo, Prisca, Julie E. Bines, Chikondi Malamba-Banda, Isaac T. Shawa, Kayla Barnes, Arox W. Kamng’ona, Daniel Hungerford, Kondwani C. Jambo, Miren Iturriza-Gomara, Nigel A. Cunliffe, and et al. 2022. "Leveraging Beneficial Off-Target Effects of Live-Attenuated Rotavirus Vaccines" Vaccines 10, no. 3: 418. https://doi.org/10.3390/vaccines10030418
APA StyleBenedicto-Matambo, P., Bines, J. E., Malamba-Banda, C., Shawa, I. T., Barnes, K., Kamng’ona, A. W., Hungerford, D., Jambo, K. C., Iturriza-Gomara, M., Cunliffe, N. A., Flanagan, K. L., & Jere, K. C. (2022). Leveraging Beneficial Off-Target Effects of Live-Attenuated Rotavirus Vaccines. Vaccines, 10(3), 418. https://doi.org/10.3390/vaccines10030418









