Equids’ Core Vaccines Guidelines in North America: Considerations and Prospective
Abstract
1. Introduction
2. Establishment of Guidelines in Dogs and Cats
3. Guidelines of Equine Core Vaccines
3.1. Tetanus
3.2. Rabies
3.3. Equine Arboviroses
3.3.1. Eastern (EEE) and Western Encephalitis (WEE), Venezuelan Encephalitis (VEE)
3.3.2. West Nile Virus (WNV)
4. Other Considerations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAEP | American Association of Equine Practitioners |
| AAFP | American Association of Feline Practitioners |
| AAHA | American Animal Hospital Association |
| EEE | Eastern Equine Encephalitis |
| EHV | Equine Herpesvirus |
| EVA | Equine Viral Arteritis |
| EWT | Encephalomyelitis-West Nile |
| PPID | Pituitary Pars Intermedia Dysfunction |
| WEE | Western Equine Encephalitis |
| VEE | Venezuelan Equine Encephalitis |
| WNV | West Nile virus |
| WSAVA | World Small Animal Veterinary Association |
References
- Aida, V.; Pliasas, V.C.; Neasham, P.J.; North, J.F.; McWhorter, K.L.; Glover, S.R.; Kyriakis, C.S. Novel Vaccine Technologies in Veterinary Medicine: A Herald to Human Medicine Vaccines. Front. Vet. Sci. 2021, 8, 340. [Google Scholar] [CrossRef] [PubMed]
- Cohen, N.D.; Bordin, A.I. Principles of vaccination. In Equine Clinical Immunology; John Wiley & Sons, Inc.: Chichester, UK, 2015; pp. 263–278. [Google Scholar]
- Wilson, J. Vaccine efficacy and controversies. In Proceedings of the 51st Annual Convention of the American Association of Equine Practitioners, Seattle, WA, USA, 3–7 December 2005; pp. 409–420. [Google Scholar]
- Barros, T.; Moran, G.; Uberti, B. Reactive Seizures After Vaccination in a Thoroughbred Broodmare. J. Equine Vet. Sci. 2019, 73, 106–109. [Google Scholar] [CrossRef]
- American Association of Equine Practitioners (AAEP). Vaccination Guidelines. Available online: https://aaep.org/guidelines/vaccination-guidelines (accessed on 26 December 2021).
- Wilson, W. Strategies for vaccinating mares, foals, and weanlings. In Proceedings of the 51st Annual Convention of the American Association of Equine Practitioners, Seattle, WA, USA, 3–7 December 2005; pp. 421–438. [Google Scholar]
- Stone, A.E.; Brummet, G.O.; Carozza, E.M.; Kass, P.H.; Petersen, E.P.; Sykes, J.; Westman, M.E. 2020 AAHA/AAFP feline vaccination guidelines. J. Am. Anim. Hosp. Assoc. 2020, 56, 249–265. [Google Scholar] [CrossRef]
- Ford, R.B.; Larson, L.J.; McClure, K.D.; Schultz, R.D.; Welborn, L.V. 2017 AAHA canine vaccination guidelines. J. Am. Anim. Hosp. Assoc. 2017, 53, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Bittle, J.; Grant, W.; Scott, F. Canine and feline immunization guidelines—1982. J. Am. Vet. Med. Assoc. 1982, 181, 332–335. [Google Scholar] [PubMed]
- Gumley, N. Revaccination guidelines revisited. Can. Vet. J. 2000, 41, 26. [Google Scholar] [PubMed]
- Klingborg, D.J.; Hustead, D.R.; Curry-Galvin, E.A.; Gumley, N.R.; Henry, S.C.; Bain, F.T.; Paul, M.A.; Boothe, D.M.; Blood, K.S.; Huxsoll, D.L. AVMA Council on Biologic and Therapeutic Agents’ report on cat and dog vaccines. J. Am. Vet. Med. Assoc. 2002, 221, 1401–1407. [Google Scholar] [CrossRef] [PubMed]
- American Veterinary Medical Association. Vaccination Principles. Available online: https://www.avma.org/resources-tools/avma-policies/vaccination-principles (accessed on 26 December 2021).
- Paul, M.A.; Appel, M.; Barrett, R.; Carmichael, L.E. Report of the American Animal Hospital Association (AAHA) Canine Vaccine Task Force: Executive summary and 2003 canine vaccine guidelines and recommendations. J. Am. Anim. Hosp. Assoc. 2003, 39, 119. [Google Scholar] [PubMed]
- Day, M.J.; Horzinek, M.; Schultz, R.; Squires, R. WSAVA guidelines for the vaccination of dogs and cats. J. Small Anim. Pract. 2016, 57, E1. [Google Scholar] [CrossRef]
- American Animal Hospital Association (AAHA). Lifestyle-Based Vaccine Calculator. Available online: https://www.aaha.org/aaha-guidelines/2020-aahaaafp-feline-vaccination-guidelines/feline-vaccine-calculator/ (accessed on 26 December 2021).
- Wilson, W.D.; Pusterla, N.; Long, D.C. Immunoprophylaxis. In Equine Infectious Diseases, 2nd ed.; Sellon, D.C., Long, M.T., Eds.; Elsevier Health Sciences: Saint Louis, MO, USA, 2013. [Google Scholar]
- Broaddus, C.C.; Balasuriya, U.B.; White, J.L.; Timoney, P.J.; Funk, R.A.; Holyoak, G.R. Evaluation of the safety of vaccinating mares against equine viral arteritis during mid or late gestation or during the immediate postpartum period. J. Am. Vet. Med. Assoc. 2011, 238, 741–750. [Google Scholar] [CrossRef]
- Hooker, B.; Kern, J.; Geier, D.; Haley, B.; Sykes, L.; King, P.; Geier, M. Methodological issues and evidence of malfeasance in research purporting to show Thimerosal in vaccines is safe. BioMed Res. Int. 2014, 2014, 247218. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.G.; Bello, N.; Bryan, A.; Hankins, K.; Wilkerson, M. Characterisation of immune responses in healthy foals when a multivalent vaccine protocol was initiated at age 90 or 180 days. Equine Vet. J. 2015, 47, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Pusterla, N.; Collier, J.; Mapes, S.; Wattanaphasak, S.; Gebhart, C. Effects of administration of an avirulent live vaccine of Lawsonia intracellularis on mares and foals. Vet. Rec. 2009, 164, 783. [Google Scholar] [CrossRef] [PubMed]
- Cauchard, J.; Sevin, C.; Ballet, J.-J.; Taouji, S. Foal IgG and opsonizing anti-Rhodococcus equi antibodies after immunization of pregnant mares with a protective VapA candidate vaccine. Vet. Microbiol. 2004, 104, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Martens, R.; Martens, J.G.; Fiske, R. Failure of passive immunisation by colostrum from immunised mares to protect foals against Rhodococcus equi pneumonia. Equine Vet. J. 1991, 23, 19–22. [Google Scholar] [CrossRef]
- Sturgill, T.L.; Horohov, D.W. Vaccination response of young foals to keyhole limpet hemocyanin: Evidence of effective priming in the presence of maternal antibodies. J. Equine Vet. Sci. 2010, 30, 359–364. [Google Scholar] [CrossRef]
- Ryan, C.; Giguère, S. Equine neonates have attenuated humoral and cell-mediated immune responses to a killed adjuvanted vaccine compared to adult horses. Clin. Vaccine Immunol. 2010, 17, 1896–1902. [Google Scholar] [CrossRef] [PubMed]
- Wilson, W.D.; Mihalyi, J.; Hussey, S.; Lunn, D. Passive transfer of maternal immunoglobulin isotype antibodies against tetanus and influenza and their effect on the response of foals to vaccination. Equine Vet. J. 2001, 33, 644–650. [Google Scholar] [CrossRef]
- Cullinane, A.; Weld, J.; Osborne, M.; Nelly, M.; McBride, C.; Walsh, C. Field studies on equine influenza vaccination regimes in thoroughbred foals and yearlings. Vet. J. 2001, 161, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Wilson, W.D. Vaccination programs for goals and weanlings. In Proceedings of the 45th Annual Convention, Albuquerque, NM, USA, 5–8 December 1999; pp. 254–263. [Google Scholar]
- Muirhead, T.; McClure, J.; Wichtel, J.; Stryhn, H.; Markham, R.; McFarlane, D.; Lunn, D. The effect of age on the immune response of horses to vaccination. J. Comp. Pathol. 2010, 142, S85–S90. [Google Scholar] [CrossRef] [PubMed]
- Muirhead, T.; McClure, J.; Wichtel, J.; Stryhn, H.; Frederick Markham, R.; McFarlane, D.; Lunn, D. The effect of age on serum antibody titers after rabies and influenza vaccination in healthy horses. J. Vet. Intern. Med. 2008, 22, 654–661. [Google Scholar] [CrossRef] [PubMed]
- McFarlane, D. Equine pituitary pars intermedia dysfunction. Vet. Clin. Equine Pract. 2011, 27, 93–113. [Google Scholar] [CrossRef] [PubMed]
- Recknagel, S.; Snyder, A.; Brueser, B.; Schusser, G.F. Immunization strategies and seroprotection against tetanus in horses in central Germany. Pferdeheilkunde 2015, 31, 469–476. [Google Scholar] [CrossRef]
- Kay, G.; Souhail, M.; Mazan, M. Recurrent tetanus in a donkey. Equine Vet. Educ. 2021. [Google Scholar] [CrossRef]
- Wilson, W.D. Assessment of Vaccination Status and Susceptibility to Infection. In Interpretation of Equine Laboratory Diagnostics; John Wiley & Sons: Hoboken, NJ, USA, 2017; p. 227. [Google Scholar]
- Kendall, A.; Anagrius, K.; Gånheim, A.; Rosanowski, S.; Bergström, K. Duration of tetanus immunoglobulin G titres following basic immunisation of horses. Equine Vet. J. 2016, 48, 710–713. [Google Scholar] [CrossRef] [PubMed]
- Löhrer, J.; Radvila, P. Active tetanus prophylaxis in the horse and the duration of immunity. Schweiz. Arch. Fur Tierheilkd. 1970, 112, 307–314. [Google Scholar]
- Recknagel, S.; Snyder, A.; Blanke, A.; Uhlig, A.; Brüser, B.; Schusser, G.F. Evaluation of an immunochromatographic dipstick test for the assessment of tetanus immunity in horses. Berl. Und Munch. Tierarztl. Wochenschr. 2015, 128, 376–383. [Google Scholar]
- Green, S.L.; Little, C.B.; Baird, J.D.; Tremblay, R.R.; Smith-Maxie, L.L. Tetanus in the horse: A review of 20 cases (1970 to 1990). J. Vet. Intern. Med. 1994, 8, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Van Galen, G.; Saegerman, C.; Rijckaert, J.; Amory, H.; Armengou, L.; Bezdekova, B.; Durie, I.; Findshøj Delany, R.; Fouché, N.; Haley, L. Retrospective evaluation of 155 adult equids and 21 foals with tetanus in Western, Northern, and Central Europe (2000–2014). Part 1: Description of history and clinical evolution. J. Vet. Emerg. Crit. Care 2017, 27, 684–696. [Google Scholar] [CrossRef]
- Jansen, B.; Knoetze, P. The Immune Response of Horses to Tetanus Toxoid. Onderstepoort J. Vet. Res. 1979, 46, 211–216. [Google Scholar]
- Heldens, J.; Pouwels, H.; Derks, C.; Van de Zande, S.; Hoeijmakers, M. Duration of immunity induced by an equine influenza and tetanus combination vaccine formulation adjuvanted with ISCOM-Matrix. Vaccine 2010, 28, 6989–6996. [Google Scholar] [CrossRef]
- Liefman, C. Combined active-passive immunisation of horses against tetanus. Aust. Vet. J. 1980, 56, 119–122. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.M.; Watson, J.L.; Brault, S.A.; Edman, J.M.; Moore, S.M.; Kass, P.H.; Wilson, W.D. Duration of serum antibody response to rabies vaccination in horses. J. Am. Vet. Med. Assoc. 2016, 249, 411–418. [Google Scholar] [CrossRef] [PubMed]
- National Association of State Public Health Veterinarians; Compendium of Animal Rabies Prevention and Control Committee; Brown, C.M.; Slavinski, S.; Ettestad, P.; Sidwa, T.J.; Sorhage, F.E. Compendium of animal rabies prevention and control, 2016. J. Am. Vet. Med. Assoc. 2016, 248, 505–517. [Google Scholar] [CrossRef] [PubMed]
- Hooper, D.C.; Morimoto, K.; Bette, M.; Weihe, E.; Koprowski, H.; Dietzschold, B. Collaboration of antibody and inflammation in clearance of rabies virus from the central nervous system. J. Virol. 1998, 72, 3711–3719. [Google Scholar] [CrossRef] [PubMed]
- Wiktor, T.; Doherty, P.; Koprowski, H. In vitro evidence of cell-mediated immunity after exposure of mice to both live and inactivated rabies virus. Proc. Natl. Acad. Sci. USA 1977, 74, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Wiktor, T.J.; Doherty, P.C.; Koprowski, H. Suppression of cell-mediated immunity by street rabies virus. J. Exp. Med. 1977, 145, 1617–1622. [Google Scholar] [CrossRef]
- Fischer, L.; Minke, J.; Dufay, N.; Baudu, P.; Audonnet, J.-C. Rabies DNA vaccine in the horse: Strategies to improve serological responses. Vaccine 2003, 21, 4593–4596. [Google Scholar] [CrossRef]
- Green, S.; Smith, L.; Vernau, W.; Beacock, S. Rabies in horses: 21 cases (1970–1990). J. Am. Vet. Med. Assoc. 1992, 200, 1133–1137. [Google Scholar]
- Chappuis, G. Neonatal immunity and immunisation in early age: Lessons from veterinary medicine. Vaccine 1998, 16, 1468–1472. [Google Scholar] [CrossRef]
- Chapman, G.E.; Baylis, M.; Archer, D.; Daly, J.M. The challenges posed by equine arboviruses. Equine Vet. J. 2018, 50, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Chaintoutis, S.C.; Diakakis, N.; Papanastassopoulou, M.; Banos, G.; Dovas, C.I. Evaluation of cross-protection of a lineage 1 West Nile virus inactivated vaccine against natural infections from a virulent lineage 2 strain in horses, under field conditions. Clin. Vaccine Immunol. 2015, 22, 1040–1049. [Google Scholar] [CrossRef][Green Version]
- Fehér, O.; Bakonyi, T.; Barna, M.; Nagy, A.; Takács, M.; Szenci, O.; Joó, K.; Sárdi, S.; Korbacska-Kutasi, O. Serum neutralising antibody titres against a lineage 2 neuroinvasive West Nile Virus strain in response to vaccination with an inactivated lineage 1 vaccine in a European endemic area. Vet. Immunol. Immunopathol. 2020, 227, 110087. [Google Scholar] [CrossRef] [PubMed]
- Minke, J.; Siger, L.; Cupillard, L.; Powers, B.; Bakonyi, T.; Boyum, S.; Nowotny, N.; Bowen, R. Protection provided by a recombinant ALVAC®-WNV vaccine expressing the prM/E genes of a lineage 1 strain of WNV against a virulent challenge with a lineage 2 strain. Vaccine 2011, 29, 4608–4612. [Google Scholar] [CrossRef]
- Waldridge, B.M.; Wenzel, J.; Ellis, A.; Rowe-Morton, S.E.; Bridges, E.R.; D’Andrea, G.; Wint, R. Serologic responses to eastern and western equine encephalomyelitis vaccination in previously vaccinated horses. Vet. Ther. Res. Appl. Vet. Med. 2003, 4, 242–248. [Google Scholar]
- Gibbs, E.P.J.; Wilson, J.H.; All, B. Studies on passive immunity and the vaccination of foals against eastern equine encephalitis in Florida. In Proceedings of Equine Infectious Diseases V: Proceedings of the 5th International Conference; The University Press of Kentucky: Lexington, KY, USA, 1988; pp. 201–205. [Google Scholar]
- Calisher, C.H.; Sasso, D.R.; Sather, G.E. Possible evidence for interference with Venezuelan equine encephalitis virus vaccination of equines by pre-existing antibody to eastern or western equine encephalitis virus, or both. Appl. Microbiol. 1973, 26, 485–488. [Google Scholar] [CrossRef] [PubMed]
- Zacks, M.A.; Paessler, S. Encephalitic alphaviruses. Vet. Microbiol. 2010, 140, 281–286. [Google Scholar] [CrossRef]
- Walton, T.; Jochim, M.; Barber, T.; Thompson, L. Cross-protective immunity between equine encephalomyelitis viruses in equids. Am. J. Vet. Res. 1989, 50, 1442–1446. [Google Scholar]
- Barber, T.; Walton, T.; Lewis, K. Efficacy of trivalent inactivated encephalomyelitis virus vaccine in horses. Am. J. Vet. Res. 1978, 39, 621–625. [Google Scholar]
- Ferguson, J.; Reeves, W.C.; Hardy, J.L. Studies on immunity to alphaviruses in foals. Am. J. Vet. Res. 1979, 40, 5–10. [Google Scholar]
- Khatibzadeh, S.M.; Gold, C.B.; Keggan, A.E.; Perkins, G.A.; Glaser, A.L.; Dubovi, E.J.; Wagner, B. West Nile virus–specific immunoglobulin isotype responses in vaccinated and infected horses. Am. J. Vet. Res. 2015, 76, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Siger, L.; Bowen, R.; Karaca, K.; Murray, M.; Jagannatha, S.; Echols, B.; Nordgren, R.; Minke, J.M. Evaluation of the efficacy provided by a recombinant canarypox-vectored equine West Nile virus vaccine against an experimental West Nile virus intrathecal challenge in horses. Vet. Ther. 2006, 7, 249–256. [Google Scholar] [PubMed]
- Long, M.; Gibbs, E.; Mellencamp, M.; Bowen, R.; Seino, K.; Zhang, S.; Beachboard, S.; Humphrey, P. Efficacy, duration, and onset of immunogenicity of a West Nile virus vaccine, live Flavivirus chimera, in horses with a clinical disease challenge model. Equine Vet. J. 2007, 39, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Siger, L.; Bowen, R.A.; Karaca, K.; Murray, M.J.; Gordy, P.W.; Loosmore, S.M.; Audonnet, J.-C.F.; Nordgren, R.M.; Minke, J.M. Assessment of the efficacy of a single dose of a recombinant vaccine against West Nile virus in response to natural challenge with West Nile virus-infected mosquitoes in horses. Am. J. Vet. Res. 2004, 65, 1459–1462. [Google Scholar] [CrossRef]
- Davidson, A.H.; Traub-Dargatz, J.L.; Rodeheaver, R.M.; Ostlund, E.N.; Pedersen, D.D.; Moorhead, R.G.; Stricklin, J.B.; Dewell, R.D.; Roach, S.D.; Long, R.E. Immunologic responses to West Nile virus in vaccinated and clinically affected horses. J. Am. Vet. Med. Assoc. 2005, 226, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.; Hathaway, D.; Jennings, N.; Champ, D.; Chiang, Y.; Chu, H. Equine vaccine for West Nile virus. Dev. Biol. 2003, 114, 221–227. [Google Scholar]
- Long, M.T.; Jeter, W.; Hernandez, J.; Sellon, D.C.; Gosche, D.; Gillis, K.; Bille, E.; Gibbs, E.P. Diagnostic performance of the equine IgM capture ELISA for serodiagnosis of West Nile virus infection. J. Vet. Intern. Med. 2006, 20, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Long, M.T. West Nile virus and equine encephalitis viruses: New perspectives. Vet. Clin. Equine Pract. 2014, 30, 523–542. [Google Scholar] [CrossRef] [PubMed]
- Long, M.; Porter, M.; Hernandez, J.; Giguere, S.; Fontaine, G.; Jodoin, E.; Gillis, K. Preliminary data regarding the subclinical exposure rate of horses to West Nile virus during the 2001 Florida enzootic. Proc. Am. Assoc. Equine Pract 2003, 49, 397–398. [Google Scholar]
- Durand, B.; Dauphin, G.; Zeller, H.; Labie, J.; Schuffenecker, I.; Murri, S.; Moutou, F.; Zientara, S. Serosurvey for West Nile virus in horses in southern France. Vet. Rec. 2005, 157, 711. [Google Scholar] [CrossRef] [PubMed]
- Roehrig, J.T.; Nash, D.; Maldin, B.; Labowitz, A.; Martin, D.A.; Lanciotti, R.S.; Campbell, G.L. Persistence of virus-reactive serum immunoglobulin M antibody in confirmed West Nile virus encephalitis cases. Emerg. Infect. Dis. 2003, 9, 376. [Google Scholar] [CrossRef]
- Kitai, Y.; Shirafuji, H.; Kanehira, K.; Kamio, T.; Kondo, T.; Konishi, E. Specific antibody responses to West Nile virus infections in horses preimmunized with inactivated Japanese encephalitis vaccine: Evaluation of blocking enzyme-linked immunosorbent assay and complement-dependent cytotoxicity assay. Vector-Borne Zoonotic Dis. 2011, 11, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Vest, D.J.; Cohen, N.D.; Berezowski, C.J.; Morehead, J.P.; Blodgett, G.P.; Blanchard, T.L. Evaluation of administration of West Nile virus vaccine to pregnant broodmares. J. Am. Vet. Med. Assoc. 2004, 225, 1894–1897. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, P.A.; Glaser, A.L.; McDonnell, S.M. Passive transfer of naturally acquired specific immunity against West Nile Virus to foals in a semi-feral pony herd. J. Vet. Intern. Med. 2006, 20, 1045–1047. [Google Scholar] [CrossRef]
- Gardner, I.A.; Wong, S.J.; Ferraro, G.L.; Balasuriya, U.B.; Hullinger, P.J.; Wilson, W.D.; Shi, P.-Y.; MacLachlan, N.J. Incidence and effects of West Nile virus infection in vaccinated and unvaccinated horses in California. Vet. Res. 2007, 38, 109–116. [Google Scholar] [CrossRef]
- Boyle, A.; Timoney, J.F.; Newton, J.; Hines, M.; Waller, A.; Buchanan, B. Streptococcus equi infections in horses: Guidelines for treatment, control, and prevention of strangles—revised consensus statement. J. Vet. Intern. Med. 2018, 32, 633–647. [Google Scholar] [CrossRef] [PubMed]
- Andersen, S.A.; Petersen, H.H.; Ersbøll, A.K.; Falk-Rønne, J.; Jacobsen, S. Vaccination elicits a prominent acute phase response in horses. Vet. J. 2012, 191, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Arfuso, F.; Giudice, E.; Di Pietro, S.; Piccione, G.; Giannetto, C. Modulation of Serum Protein Electrophoretic Pattern and Leukocyte Population in Horses Vaccinated against West Nile Virus. Animals 2021, 11, 477. [Google Scholar] [CrossRef]
- Holmes, M.; Townsend, H.; Hussey, S.; Breathnach, C.; Barnett, C.; Holland, R.; Lunn, D. Immune responses to commercial equine vaccines. In Proceedings of the 49th Annual Convention of the American Association of Equine Practitioners, New Orleans, LA, USA, 21–25 November 2003; pp. 224–226. [Google Scholar]
- Holmes, M.A.; Townsend, H.G.; Kohler, A.K.; Hussey, S.; Breathnach, C.; Barnett, C.; Holland, R.; Lunn, D. Immune responses to commercial equine vaccines against equine herpesvirus-1, equine influenza virus, eastern equine encephalomyelitis, and tetanus. Vet. Immunol. Immunopathol. 2006, 111, 67–80. [Google Scholar] [CrossRef]
- Cortese, V.; Hankins, K.; Holland, R.; Syvrud, K. Serologic responses of West Nile virus seronegative mature horses to West Nile virus vaccines. J. Equine Vet. Sci. 2013, 33, 1101–1105. [Google Scholar] [CrossRef]
- Chapuis, R.J.; Smith, J.S.; French, H.M.; Toka, F.N.; Peterson, E.W.; Little, E.L. Nonlinear Mixed-Effect Pharmacokinetic Modeling and Distribution of Doxycycline in Healthy Female Donkeys after Multiple Intragastric Dosing–Preliminary Investigation. Animals 2021, 11, 2047. [Google Scholar] [CrossRef] [PubMed]
- Barrandeguy, M.E.; Carossino, M. Infectious diseases in donkeys and mules: An overview and update. J. Equine Vet. Sci. 2018, 65, 98–105. [Google Scholar] [CrossRef]
- Day, M. The CALLISTO project: A summary. J. Comp. Pathol. 2016, 155, S1–S7. [Google Scholar] [CrossRef] [PubMed]
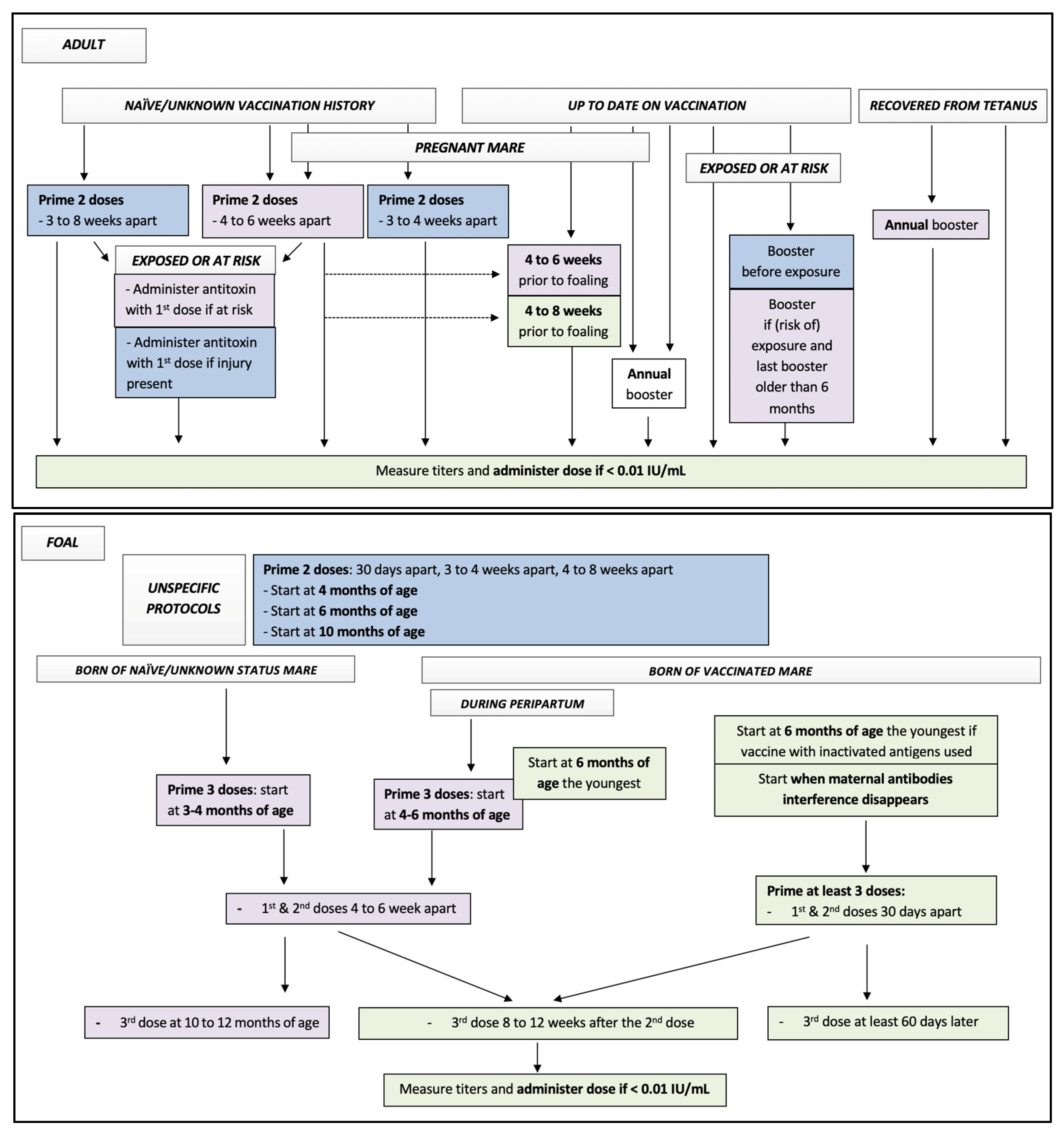

| Vaccine: AAEP Protocol | Labelled Protocols of Equine Core Vaccine Approved in North America | Suggested Additional Recommendations |
|---|---|---|
Adult
Foal, primary vaccination
| Monovalent vaccines Tetguard™ (Boehringer Ingelheim)
Tetanus Toxoid/Super-Tet/Prestige® Tetanus (Intervet/Merck)
Tetanus toxoid (Zoetis)
Multivalent vaccines Core EQ Innovator®/West Nile-Innovator® + EWT (Zoetis)
Equi-Jec® 5/Equi-Jec® 6/Equi-Jec® 7/Equi-Jec® WNV + EWT (Boehringer Ingelheim)
Vetera® EWT/Vetera® VEWT/Vetera® 4XP +WNV/Vetera® 5XP/Vetera® 6XP/Vetera® EWT + WNV/Vetera® VEWT + WNV (Boehringer Ingelheim)
| Pregnant mare:
Foal, primary vaccination
Test serum titer (peer review case reports [31,32])
|
| Infectious Agent and Description | Vaccines Labelled in the USA | Vaccine Description | Reported Efficacy and Safety |
|---|---|---|---|
Clostridium tetani (TT)
| GoatVac T (Durvet)—not in market since 2007 (personal communication with Durvet) | Tetanus toxoids | Efficacy and Safety—Study data were evaluated by USDA-APHIS prior to product licensure and met regulatory standards for acceptance at the time of submission. (https://www.aphis.usda.gov/wcm/connect/3bba1d24-a8c7-4767-a11f-ead24b1b3b64/188-860100.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-3bba1d24-a8c7-4767-a11f-ead24b1b3b64-mvhp..F) (accessed on 3 February 2022) |
| TetguardTM (Boehringer Ingelheim) | Purified toxoid Alum precipitated Preservative-Thimerosal | Study data were evaluated by USDA-APHIS prior to product licensure and met regulatory standards for acceptance at the time of submission. No data are published as this study was submitted to USDA-APHIS prior to 1 January 2007, and APHIS only requires publication of data submitted after that date. (https://www.aphis.usda.gov/wcm/connect/7bbeb060-a2c3-45f6-befc-c6dbb31c7d31/165A-860101.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-7bbeb060-a2c3-45f6-befc-c6dbb31c7d31-m9P8gXL) (accessed on 3 February 2022) | |
| Tetanus Toxoid/Prestige® Tetanus (previously Super-Tet) (Intervet/Merck) | Antigen purification system Tetanus toxoid Havlogen® adjuvant Preservative-Thimerosal | Efficacy—demonstrated in laboratory animals (Guinea pigs) according to 9CFR 113.114(c). Satisfactory result is an antitoxin titer of at least 2.0 A.U. per mL for the serum pool. (https://www.aphis.usda.gov/wcm/connect/7bbeb060-a2c3-45f6-befc-c6dbb31c7d31/165A-860101.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-7bbeb060-a2c3-45f6-befc-c6dbb31c7d31-m9P8gXL) (accessed on 3 February 2022) Safety—of 552 horses, 298 received 2 doses IM 3 to 4 weeks apart for primary immunization and 254 horses received 1 dose IM. No horses showed muscular swelling, pain, or stiffness. (https://www.aphis.usda.gov/wcm/connect/7bbeb060-a2c3-45f6-befc-c6dbb31c7d31/165A-860101.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-7bbeb060-a2c3-45f6-befc-c6dbb31c7d31-m9P8gXL) (accessed on 3 February 2022) | |
| Tetanus toxoid (Zoetis) | Tetanus toxoid | Efficacy—demonstrated in laboratory animal (guinea pigs) requirements were evaluated by USDA-APHIS prior to product licensure and met regulatory standards for acceptance per 9 CFR 113.114 (date not specified). (https://www.aphis.usda.gov/wcm/connect/0f81ce58-550f-4b18-b406-f8df3e6f2bcb/190-48R521.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-0f81ce58-550f-4b18-b406-f8df3e6f2bcb-mCiisji) (accessed on 3 February 2022) |
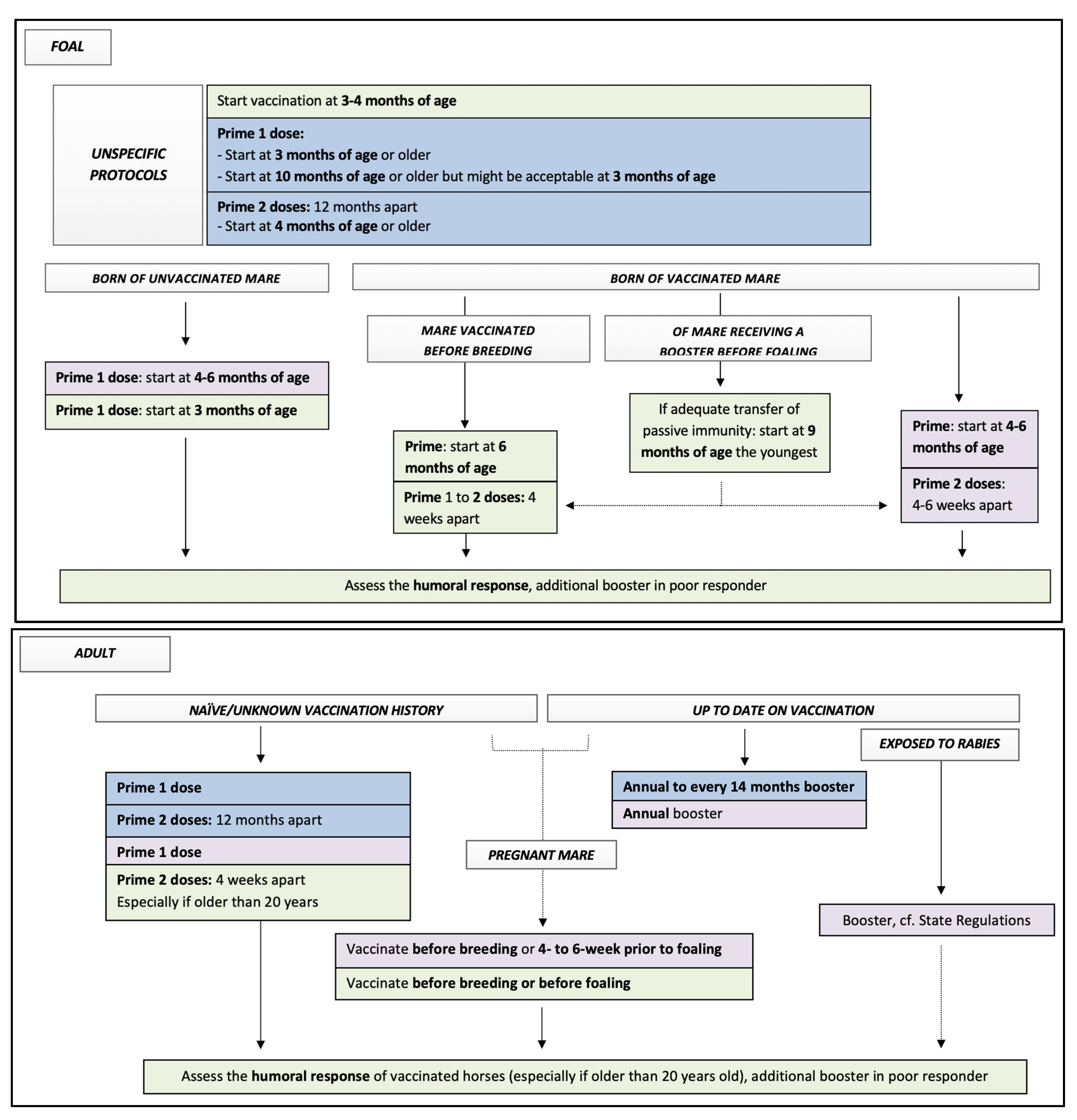
| Vaccine: AAEP Protocol | Labelled Protocols of Equine Core Vaccine Approved in North America | Suggested Additional Recommendations |
|---|---|---|
Adult
Foal and Weanling, primary vaccination
| Monovalent vaccines Prestige® EquiRab® (Merk)
Rabisin/Imrab Large Animal (Merial/Boehringer Ingelheim)
Multivalent vaccine Core EQ Innovator® (Zoetis)
| Adult
Foal:
Assess the humoral response of vaccinated horses. Additional booster in poor responders might be beneficial (based on peer review cohort studies [28,29,42]) |
| Infectious Agent and Description | Vaccines Labelled in the USA | Vaccine Description | Reported Efficacy and Safety |
|---|---|---|---|
Rabies Virus
| EquiRab and Prestige EquiRab (Merck) | Inactivated virus + Havlogen® adjuvant Antibiotic-neomycin + polymyxin B Preservative-Thimerosal | Efficacy—Of 37 animals, 26 4-month-old horses vaccinated once IM, and 11 were challenged with Rabies virus 14-months after with no deaths due to Rabies. (https://www.aphis.usda.gov/wcm/connect/577930f8-81c2-426a-8488-a1570b630955/165A-190523.pdf?MOD=AJPERES&CACHEID=ROOTWORKSPACE-577930f8-81c2-426a-8488-a1570b630955-mdG7YyP) (accessed on 3 February 2022) Safety—992 horses with 413 4-month-old animals received 1 dose IM. No animals showed pain or swelling in the injection site |
| Rabisin/Imrab Large Animal (Boehringer Ingelheim) | Tetanus toxoid with betapropiolactone + adjuvant aluminum hydroxide Antibiotic-Gentamicin Preservative-Thimerosal | Study data were evaluated by USDA-APHIS prior to product licensure and met regulatory standards for acceptance at the time of submission. Study data, however, are no longer available. https://www.aphis.usda.gov/wcm/connect/577930f8-81c2-426a-8488-a1570b630955/165A-190523.pdf?MOD=AJPERES&CACHEID=ROOTWORKSPACE-577930f8-81c2-426a-8488-a1570b630955-mdG7YyP) (accessed on 3 February 2022) | |
| Study results applicable to Intramuscular route of administration. Study data were evaluated by USDA-APHIS prior to product licensure and met regulatory standards for acceptance at the time of submission. No data are published as this study was submitted to USDA-APHIS prior to 1 January 2007, and APHIS only requires publication of data submitted after that date.(https://www.aphis.usda.gov/wcm/connect/577930f8-81c2-426a-8488-a1570b630955/165A-190523.pdf?MOD=AJPERES&CACHEID=ROOTWORKSPACE-577930f8-81c2-426a-8488-a1570b630955-mdG7YyP) (accessed on 3 February 2022) |
| Vaccine: AAEP Protocol | Labelled Protocols of Equine Core Vaccine Approved in North America | Suggested Additional Recommendations |
|---|---|---|
Adults
Foal regardless vaccination status of the mares
| Prestige® 3/Prestige® 4/Prestige® 5/Prestige® 3 + WNV/Prestige® 5 + WNV (Intervet/Merck)
Equi-Jec® 5/Equi-Jec® 6/Equi-Jec® 7/Equi-Jec® WNV + EWT (Boehringer Ingelheim)
Vetera® EWT/Vetera® VEWT/Vetera® 4XP +WNV/Vetera® 5XP/Vetera® 6XP/Vetera® EWT + WNV/Vetera® VEWT + WNV/Vetera GoldXP (Boehringer Ingelheim)
Core EQ Innovator®/West Nile-Innovator® + EWT (Zoetis)
|
|
| Infectious Agents and Description | Vaccines Labelled in the USA | Vaccine Description | Reported Efficacy and Safety |
|---|---|---|---|
Eastern Equine Encephalitis Virus (EEE) and Western Equine Encephalitis Virus (WEE)
| Encevac TC-4 (Intervet/Merck; EIV, TT, EEEV, WEEV, WNV) | Killed virus Tetanus toxoid | Efficacy TT—was demonstrated in laboratory animals (guinea pigs) according to 9CFR 113.114(c). Satisfactory result is an antitoxin titer of at least 2.0 A.U. per mL for the serum pool. Efficacy EEEV—was demonstrated in laboratory animals (guinea pigs) according to 9CFR 113.207(b). Satisfactory test result is a Virus Neutralization Titer of ≥1:40 in at least 9 out of 10 vaccinates (2nd stage—at least 17 out of 20 vaccinates). Efficacy WEEV—was demonstrated in laboratory animals (guinea pigs) according to 9CFR 113.207(b). Satisfactory test result is a Virus Neutralization Titer of ≥1:40 in at least 9 of the vaccinates. Efficacy EHV-1—16/32, 11 months of age horses received 2 vaccination IM doses 3 weeks apart were challenged with EHV-1, DA35 strain. 6/16 horses were positive to virus isolation. 1. Efficacy EHV-4—21/36, 6 months of age horses received 2 vaccination IM doses 3 weeks apart were challenged with EHV-4. Only 2 animals had clinical signs. 2.Efficacy EHV-4—16/21, 6 months of age horses received 2 vaccination IM doses 3 weeks apart; were challenged with EHV-4. Horses shed the virus for a minimum of 3 days and a maximum of 14 days, also, 75% presented nasal discharge. Efficacy EIV—18 horses were challenged with A/equine/Kentucky/99 six months post 2nd IM vaccination 3 weeks apart. 14/18 horses showed clinical signs and 12/18 horses 12/18 horses were positive to virus isolation 14 days after challenge. Safety—298 horses received 2 vaccination IM doses 3–4-weeks apart. 96.47% of the horses had no evidence of reactions. (https://www.aphis.usda.gov/wcm/connect/4f1c0212-8505-43ff-add1-e096881ebfac/165a-4855r2.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-4f1c0212-8505-43ff-add1-e096881ebfac-mVCsR7z) (accessed on 3 February 2022) |
| Prestige®3 (Merck; TT, EEEV, WEEV) | |||
| Prestige®3 + WNV (Merck; TT, EEEV, WEEV, WNV) | |||
| Prestige®4 (Merck; EIV, TT, EEEV, WEEV) | Killed virus Tetanus toxoid Antibiotic-Gentamicin Preservatives-Thimerosal | ||
| Prestige®5 (Merck; EIV, EHV-1/4, TT, EEEV, WEEV) | |||
| Prestige®5 + WNV (Merck; TT, EEEV, WEEV, EIV, EHV-1/4, WNV) | |||
| Vetera EWT (Boehringer Ingelheim; TT, EEEV, WEEV | Inactivated virus Tetanus toxoid Established Carbimmune® adjuvant | Efficacy TT—10 guinea pigs, 6 weeks after the injection, vaccinate serum samples were collected and pooled, then tested for antitoxin content by indirect ELISA. A satisfactory value, which met the requirements per 9 CFR 113.114(c), was achieved. Efficacy EEEV+WEEV+VEEV—10/12 guinea pigs, Serum samples were tested by a plaque reduction, serum neutralization test, 14 to 21 days after the second injection. Vaccinates and controls were evaluated in terms of Eastern and Western equine encephalomyelitis per the criteria in 9 CFR 113.207(b) and the requirements were met. Efficacy EHV-1—20/40, 4–5 months old horses received 2 vaccination IM doses 3 weeks apart and were challenged with EHV-1 15-days the last dose. Only 6 horses had no nasal discharge. Efficacy EHV-4—20/40, 4-months old horses received 2 vaccination IM doses 3 weeks apart and were challenged 4-days after the last dose. 12/20 had mild and 1/20 had moderate clinical signs 1. Efficacy EIV—20/30 hoses, 5–6-months-old received 2 IM vaccination doses 21 days apart and were challenged with Influenza A/eq/Ohio/2003 administered 184 days post-final vaccination. 9/20 horses had clinical signs. 2. Efficacy EIV—18/37 horses, 9–10-months-oldreceived 2 IM vaccination doses 21 days apart and were challenged with Influenza A/eq/Ohio/2003 administered 3 weeks post-final vaccination. 0/18 horses shed virus. Efficacy WNV—20/30 horses, 4–5-months-old received 2 IM vaccination doses 25 days apart and 10/20 horses were challenged 380 days after the 2nd vaccination dose and 10/20 horses were challenged 408 days after the 2nd vaccination dose. 2/20 horses had viremia and 1/20 horses died. Safety all—622 horses vaccinated with 2 IM doses 21 days apart. Only 8/622 horses showed transient injection site swelling. (https://www.aphis.usda.gov/wcm/connect/283444e4-a323-41e3-b741-f2c022f332c9/124_484721.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-283444e4-a323-41e3-b741-f2c022f332c9-mCXh75k) (accessed on 3 February 2022) | |
| Vetera EWT + WNV (Boehringer Ingelheim; TT, EEEV, WEEV, WNV) | |||
| Vetera VEWT (Boehringer Ingelheim; TT, EEEV, WEEV, VEEV) | |||
| Vetera VEWT + WNV (Boehringer Ingelheim; TT, EEEV, WEEV, VEEV, WNV) | |||
| Vetera 4XP + WNV (Boehringer Ingelheim; EIV, TT, EEEV, WEEV, WNV) | |||
| Vetera 5XP (Boehringer Ingelheim; EIV, EHV-1/4, TT, EEEV, WEEV) | |||
| Vetera 6XP (Boehringer Ingelheim; EIV, EHV-1/4, TT, EEEV, WEEV, VEEV) | |||
| Vetera GoldXP (Boehringer Ingelheim; EIV, EHV-1/4, TT, EEEV, WEEV, VEEV) | |||
| Equi-Jec® WNV+EWT (Boehringer Ingelheim; TT, EEEV, WEEV, WNV) | Inactivated virus Tetanus toxoid Preservative-Formaldehyde Antibiotic-Gentamicin | ||
| Equi-Jec 7 (Boehringer Ingelheim; TT, EEEV, WEEV, VEEV, EIV, EHV-1/4, WNV) | |||
| Equi-Jec 6 (Boehringer Ingelheim; TT, EEEV, WEEV, EIV, EHV-1/4, WNV) | |||
| Core EQ Innovator®/West Nile-Innovator® + EWT (Zoetis; RV, TT, WNV, EEE, WEE) | Inactivated virus Tetanus Toxoid Adjuvant-MetaStim® | Efficacy (TT, EEE, WEE)—evaluated by USDA-APHIS in guinea pigs prior to product licensure and met regulatory standards for acceptance per 9 CFR 113.207(b)(2). Efficacy WNV—32, 9–11-months-old horses received 2 vaccination doses 3 weeks apart and challenged 12 months after. 1/19 horses were viremic. Efficacy RV—39, 3-month-old horses received 2 IM vaccination doses 3–4 weeks apart. 2/25 animals were positive—The requirements of 9 CFR 113.209 were met. (https://www.aphis.usda.gov/wcm/connect/0f81ce58-550f-4b18-b406-f8df3e6f2bcb/190-48R521.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-0f81ce58-550f-4b18-b406-f8df3e6f2bcb-mCiisji) (accessed on 3 February 2022) Safety—682 horses (https://www.aphis.usda.gov/wcm/connect/0f81ce58-550f-4b18-b406-f8df3e6f2bcb/190-48R521.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-0f81ce58-550f-4b18-b406-f8df3e6f2bcb-mCiisji) (accessed on 3 February 2022) | |
| Core EQ Innovator® + V (Zoetis; RV, TT, WNV, EEE, WEE, VEE) |
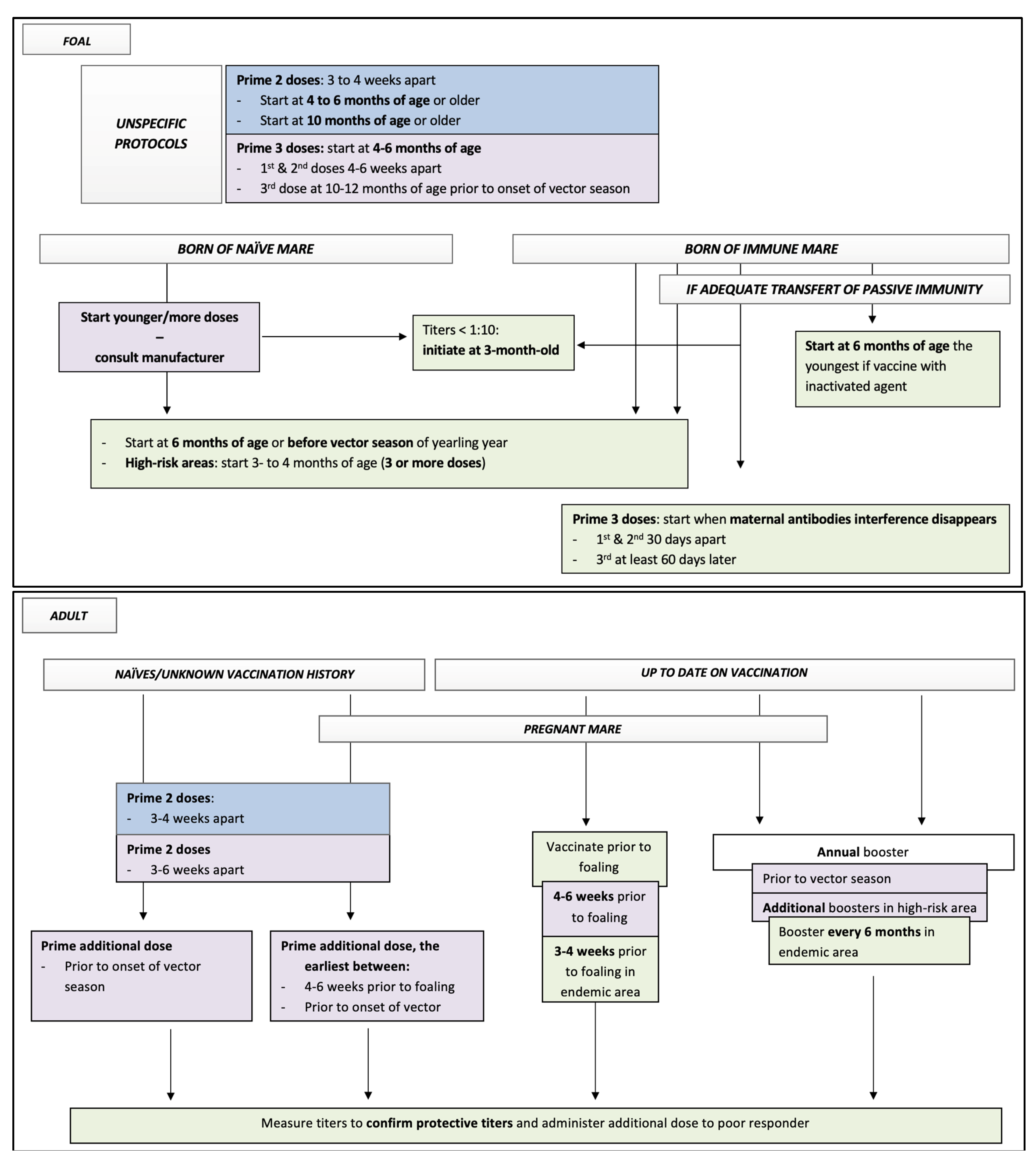
| Vaccine: AAEP Protocol | Labelled Protocols of Equine core Vaccine Approved in North America | Suggested Additional Recommendations |
|---|---|---|
Adult
Foals
| Monovalent vaccines West Nile-Innovator® (Zoetis)
Equi-Jec® WNV (Boehringer Ingelheim)
Vetera® WNV (Boehringer Ingelheim)
Multivalent vaccines Prestige® 3 + WNV (previously Encevac T + WNV)/Prestige® 5 + WNV (Intervet/Merck)
Equi-Jec® 6/Equi-Jec® 7/Equi-Jec® WNV + EWT (Boehringer Ingelheim)
Vetera® 4XP + WNV/Vetera® 5XP/Vetera® 6XP/Vetera® EWT + WNV/Vetera® VEWT + WNV (Boehringer Ingelheim)
Core EQ Innovator®/West Nile-Innovator® + EWT (Zoetis)
| Adult
Foal
Assess the humoral response of vaccinated horses. Additional booster in poor responders might be beneficial (based on peer review report [61]) |
| Infectious Agent and Description | Vaccines Labelled in the USA | Vaccine Description | Reported Efficacy and Safety |
|---|---|---|---|
West Nile Virus (WNV)
| West Nile-Innovator® (Zoetis) | Killed virus | Efficacy TT, EEE, and WEE—evaluated by USDA-APHIS in guinea pigs prior to product licensure and met regulatory standards for acceptance per 9 CFR 113.207(b)(2). Efficacy WNV—32, 9–11-months-old horses received 2 vaccination doses 3 weeks apart and challenged 12 months after. 1/19 horses were viremic. Efficacy RV—39, 3-month-old horses received 2 IM vaccination doses 3–4 weeks apart. 2/25 animals were positive—The requirements of 9 CFR 113.209 were met. (https://www.aphis.usda.gov/wcm/connect/0f81ce58-550f-4b18-b406-f8df3e6f2bcb/190-48R521.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-0f81ce58-550f-4b18-b406-f8df3e6f2bcb-mCiisji) (accessed on 3 February 2022) Safety—682 horses (https://www.aphis.usda.gov/wcm/connect/0f81ce58-550f-4b18-b406-f8df3e6f2bcb/190-48R521.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-0f81ce58-550f-4b18-b406-f8df3e6f2bcb-mCiisji) (accessed on 3 February 2022) |
| EquiNile (Intervet/Merck) | Inactivated chimera flavivirus | Efficacy—Of 40 horses, 20 vaccinates were challenged; 10 at 60 days post-2nd vaccination and 10 at 91 days post-2nd vaccination. (https://www.aphis.usda.gov/wcm/connect/5659ee49-dd51-48bb-989f-6bfc5db10e31/165A-1995R2.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-5659ee49-dd51-48bb-989f-6bfc5db10e31-m9P7PYj) (accessed on 3 February 2022) Safety—1255 horses received 2-doses 3–4 weeks apart. 1149/1255 horses had no evidence of reactions. (https://www.aphis.usda.gov/wcm/connect/5659ee49-dd51-48bb-989f-6bfc5db10e31/165A-1995R2.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-5659ee49-dd51-48bb-989f-6bfc5db10e31-m9P7PYj) (accessed on 3 February 2022) | |
| PreveNile (Merck) | Live attenuated chimeric vaccine—No longer available | No longer available | |
| Vetera WNV (Boehringer Ingelheim) | Inactivated virus + Established Carbimmune® adjuvant | 1. Efficacy—19/28 horses, 4–5-months-old were vaccinated with 2 IM doses 21 days apart. 10/19 horses were challenged intrathecally 14 days after 2nd vaccination dose and 9/19 horses were challenged intrathecally 28 days after 2nd vaccination dose. 1/19 horses was viremic. 2. Efficacy—20/30 horses were vaccinated with 2 doses 25 days apart. 10/20 were challenged 380 days after 2nd vaccination and 10/20 were challenged 408 days after 2nd vaccination dose with WNV. 1/20 vaccinates died and 2/20 vaccinates had viremia. 3. Efficacy—20/30 horses, 4–5-months-old were vaccinated with 2 IM doses 25 days apart. 10/20 horses were challenged 380 days after 2nd vaccination dose and 10/20 horses were challenged 408 days after 2nd vaccination dose. 2/20 horses had viremia and 1/20 died. Safety—325 pregnant mares were vaccinated with 2 IM doses 16–28 days apart, during 1st, 2nd, and 3rd trimester. No evidence of disease, abortion or death due to vaccination. Safety all—556 horses received 2 vaccination IM doses 3–4 weeks apart. 99.3% of horses after 1st dose showed no reactions and 98% of horses after the 2nd dose showed no reactions. (https://www.aphis.usda.gov/wcm/connect/aafa450c-43ea-476c-bfdc-ab3b2638486b/124_199520.pdf?MOD=AJPERES&CONVERT_TO=url&CACHEID=ROOTWORKSPACE-aafa450c-43ea-476c-bfdc-ab3b2638486b-nKD-5yM) (accessed on 3 February 2022) | |
| Equi-Jec® WNV (Boehringer Ingelheim) | Preservative-Formaldehyde Antibiotic-Gentamicin |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Desanti-Consoli, H.; Bouillon, J.; Chapuis, R.J.J. Equids’ Core Vaccines Guidelines in North America: Considerations and Prospective. Vaccines 2022, 10, 398. https://doi.org/10.3390/vaccines10030398
Desanti-Consoli H, Bouillon J, Chapuis RJJ. Equids’ Core Vaccines Guidelines in North America: Considerations and Prospective. Vaccines. 2022; 10(3):398. https://doi.org/10.3390/vaccines10030398
Chicago/Turabian StyleDesanti-Consoli, Hélène, Juliette Bouillon, and Ronan J. J. Chapuis. 2022. "Equids’ Core Vaccines Guidelines in North America: Considerations and Prospective" Vaccines 10, no. 3: 398. https://doi.org/10.3390/vaccines10030398
APA StyleDesanti-Consoli, H., Bouillon, J., & Chapuis, R. J. J. (2022). Equids’ Core Vaccines Guidelines in North America: Considerations and Prospective. Vaccines, 10(3), 398. https://doi.org/10.3390/vaccines10030398








