Progress towards the Elusive Mastitis Vaccines
Abstract
1. Introduction: Are Efficacious Mastitis Vaccines Feasible?
2. Possible Reasons for the Current Mastitis Vaccine Shortcomings
2.1. Obstacles to Effective Vaccination That Are Peculiar to MG Immunobiology and the Diversity of Pathogens
2.2. The Issue of Protection Induced by MG Infections
2.3. Is Mastitis an Infection without Virulence Factors?
2.4. Co-Evolution of the MG and Mastitis-Causing Pathogens
2.5. Hypothesis: The Immune Response to Mastitis-Causing Bacteria Is Biased towards Tolerance or Acceptance
3. What Can Be Proposed to Get out of the Mastitis Vaccine Predicament?
3.1. Progress in Immunology
3.1.1. Filling the Main Knowledge Gaps in Cell-Mediated Immunity in the MG
3.1.2. Promote T Cell Immunity in the Framework of MG Immunobiology
3.1.3. Define Valid Correlates of MG Protection
3.2. Progress in Vaccinology
3.2.1. Capitalize on Systems Vaccinology
3.2.2. Improve Knowledge of Adjuvants for Ruminants to Guide the Immune Response
3.2.3. Make the Most of Vaccine Delivery
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ruegg, P.L. A 100-Year Review: Mastitis detection, management, and prevention. J. Dairy Sci. 2017, 100, 10381–10397. [Google Scholar] [CrossRef] [PubMed]
- Rainard, P.; Gilbert, F.B.; Germon, P.; Foucras, G. Invited review: A critical appraisal of mastitis vaccines for dairy cows. J. Dairy Sci. 2021, 104, 10427–10448. [Google Scholar] [CrossRef] [PubMed]
- Van Regenmortel, M.H. The rational design of biological complexity: A deceptive metaphor. Proteomics 2007, 7, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.C. The problem of immunization against staphylococcal mastitis. Br. Vet. J. 1978, 134, 412–420. [Google Scholar] [CrossRef]
- Casadevall, A.; Pirofski, L.A. Exploiting the redundancy in the immune system: Vaccines can mediate protection by eliciting “unnatural” immunity. J. Exp. Med. 2003, 197, 1401–1404. [Google Scholar] [CrossRef]
- Pulendran, B.; Li, S.; Nakaya, H.I. Systems vaccinology. Immunity 2010, 33, 516–529. [Google Scholar] [CrossRef]
- Rappuoli, R.; Bottomley, M.J.; D’Oro, U.; Finco, O.; De Gregorio, E. Reverse vaccinology 2.0: Human immunology instructs vaccine antigen design. J. Exp. Med. 2016, 213, 469–481. [Google Scholar] [CrossRef]
- Colditz, I.G.; Watson, D.L. The immunophysiological basis for vaccinating ruminants against mastitis. Aust. Vet. J. 1985, 62, 145–153. [Google Scholar] [CrossRef]
- Craven, N.; Williams, M.R. Defences of the bovine mammary gland against infection and prospects for their enhancement. Vet. Immunol. Immunopathol. 1985, 10, 71–127. [Google Scholar] [CrossRef]
- Anderson, J.C. Progressive pathology of staphylococcal mastitis with a note on control, immunisation and therapy. Vet. Rec. 1982, 110, 372–376. [Google Scholar] [CrossRef]
- Paape, M.J.; Guidry, A.J. Effect of fat and casein on intracellular killing of Staphylococcus aureus by milk leukocytes. Proc. Soc. Exp. Biol. Med. 1977, 155, 588–593. [Google Scholar] [CrossRef]
- Goldberg, J.J.; Pankey, J.W.; Politis, I.; Zavizion, B.; Bramley, A.J. Effect of oxygen tension on killing of Escherichia coli by bovine polymorphonuclear neutrophil leucocytes in vitro. J. Dairy Res. 1995, 62, 331–338. [Google Scholar] [CrossRef]
- Rainard, P. The complement in milk and defense of the bovine mammary gland against infections. Vet. Res. 2003, 34, 647–670. [Google Scholar] [CrossRef]
- Richardson, E.J.; Bacigalupe, R.; Harrison, E.M.; Weinert, L.A.; Lycett, S.; Vrieling, M.; Robb, K.; Hoskisson, P.A.; Holden, M.T.G.; Feil, E.J.; et al. Gene exchange drives the ecological success of a multi-host bacterial pathogen. Nat. Ecol. Evol. 2018, 2, 1468–1478. [Google Scholar] [CrossRef]
- Leigh, J.A. Streptococcus uberis: A permanent barrier to the control of bovine mastitis? Vet. J. 1999, 157, 225–238. [Google Scholar] [CrossRef]
- Reiter, B.; Oram, J.D. Bacterial inhibitors in milk and other biological fluids. Nature 1967, 216, 328–330. [Google Scholar] [CrossRef]
- Wilson, A.T.; Rosenblum, H. The antistreptococcal property of milk. III. The role of lactenin in milk-borne epidemics; the in vivo action of lactenin. J. Exp. Med. 1952, 95, 51–59. [Google Scholar] [CrossRef]
- Rainard, P.; Riollet, C. Innate immunity of the bovine mammary gland. Vet. Res. 2006, 37, 369–400. [Google Scholar] [CrossRef]
- Wheeler, T.T.; Smolenski, G.A.; Harris, D.P.; Gupta, S.K.; Haigh, B.J.; Broadhurst, M.K.; Molenaar, A.J.; Stelwagen, K. Host-defence-related proteins in cows’ milk. Animal 2012, 6, 415–422. [Google Scholar] [CrossRef][Green Version]
- Gutzeit, C.; Magri, G.; Cerutti, A. Intestinal IgA production and its role in host-microbe interaction. Immunol. Rev. 2014, 260, 76–85. [Google Scholar] [CrossRef]
- Butler, J.E.; Rainard, P.; Lippolis, J.D.; Salmon, H.; Kacskovics, I. The mammary gland in mucosal and regional immunity. In Mucosal Immunology, 4th ed.; Mestecky, J., Strober, W., Russell, M., Cheroutre, H., Lambrecht, B.N., Kelsall, B.L., Eds.; Academic Press: Waltham, MA, USA, 2015; pp. 2269–2306. [Google Scholar]
- White, L.J.; Schukken, Y.H.; Dogan, B.; Green, L.; Dopfer, D.; Chappell, M.J.; Medley, G.F. Modelling the dynamics of intramammary E. coli infections in dairy cows: Understanding mechanisms that distinguish transient from persistent infections. Vet. Res. 2010, 41, 13. [Google Scholar] [CrossRef][Green Version]
- Linton, A.H.; Howe, K.; Sojka, W.J.; Wray, C. Range of Escherichia-Coli O-serotypes causing clinical bovine mastitis and their antibiotic-resistance spectra. J. Appl. Bacteriol. 1979, 46, 585–590. [Google Scholar] [CrossRef]
- Sanchez-Carlo, V.; Wilson, R.A.; McDonald, J.S.; Packer, R.A. Biochemical and serologic properties of Escherichia coli isolated from cows with acute mastitis. Am. J. Vet. Res. 1984, 45, 1771–1774. [Google Scholar]
- Lipman, L.J.; de Nijs, A.; Gaastra, W. Isolation and identification of fimbriae and toxin production by Escherichia coli strains from cows with clinical mastitis. Vet. Microbiol. 1995, 47, 1–7. [Google Scholar] [CrossRef]
- Pietrocola, G.; Arciola, C.R.; Rindi, S.; Montanaro, L.; Speziale, P. Streptococcus agalactiae non-pilus, cell wall-anchored proteins: Involvement in colonization and pathogenesis and potential as vaccine candidates. Front. Immunol. 2018, 9, 602. [Google Scholar] [CrossRef]
- Wolf, C.; Engelmann, S. Comparative genomic and proteomic analysis of Staphylococcus aureus mastitis isolates from bovine and human origin. Reprod. Domest. Anim. 2010, 45, 59. [Google Scholar]
- Germain, R.N. Vaccines and the future of human immunology. Immunity 2010, 33, 441–450. [Google Scholar] [CrossRef]
- Poutrel, B. Susceptibility to mastitis: A review of factors related to the cow. Ann. Rech. Vet. 1982, 13, 85–99. [Google Scholar] [PubMed]
- Rainard, P. Staphylococcus aureus leucotoxin LukM/F’ is secreted and stimulates neutralising antibody response in the course of intramammary infection. Vet. Res. 2007, 38, 685–696. [Google Scholar] [CrossRef] [PubMed]
- Doymaz, M.Z.; Sordillo, L.M.; Oliver, S.P.; Guidry, A.J. Effects of Staphylococcus aureus mastitis on bovine mammary gland plasma cell populations and immunoglobin concentrations in milk. Vet. Immunol. Immunopathol. 1988, 20, 87–93. [Google Scholar] [CrossRef]
- Hill, A.W.; Heneghan, D.J.; Field, T.R.; Williams, M.R. Increase in specific opsonic activity in bovine milk following experimental Escherichia coli mastitis. Res. Vet. Sci. 1983, 35, 222–226. [Google Scholar] [CrossRef]
- Williams, M.R.; Craven, N.; Hill, A.W. Therapy and natural defences in mastitis: I the phagocytic defence of the udder. In Antimicrobials and Agriculture: Proceedings of the 4th International Symposium on Antibiotics in Agriculture: Benefits and Malefits; Woodbine, M., Ed.; Elsevier: Amsterdam, The Netherlands, 1984; pp. 165–174. [Google Scholar]
- Leelahapongsathon, K.; Schukken, Y.H.; Srithanasuwan, A.; Suriyasathaporn, W. Molecular epidemiology of Streptococcus uberis intramammary infections: Persistent and transient patterns of infection in a dairy herd. J. Dairy Sci. 2020, 103, 3565–3576. [Google Scholar] [CrossRef]
- Van Werven, T.; Noordhuizen-Stassen, E.N.; Daemen, A.J.; Schukken, Y.H.; Brand, A.; Burvenich, C. Preinfection in vitro chemotaxis, phagocytosis, oxidative burst, and expression of CD11/CD18 receptors and their predictive capacity on the outcome of mastitis induced in dairy cows with Escherichia coli. J. Dairy Sci. 1997, 80, 67–74. [Google Scholar] [CrossRef]
- Cha, E.; Hertl, J.; Schukken, Y.; Tauer, L.; Welcome, F.; Grohn, Y. Evidence of no protection for a recurrent case of pathogen specific clinical mastitis from a previous case. J. Dairy Res. 2016, 83, 72–80. [Google Scholar] [CrossRef]
- Hill, A.W. Factors influencing the outcome of Escherichia coli mastitis in the dairy cow. Res. Vet. Sci. 1981, 31, 107–112. [Google Scholar] [CrossRef]
- Hill, A.W. Protective effect of previous intramammary infection with Streptococcus uberis against subsequent clinical mastitis in the cow. Res. Vet. Sci. 1988, 44, 386–387. [Google Scholar] [CrossRef]
- Finch, J.M.; Winter, A.; Walton, A.W.; Leigh, J.A. Further studies on the efficacy of a live vaccine against mastitis caused by Streptococcus uberis. Vaccine 1997, 15, 1138–1143. [Google Scholar] [CrossRef]
- Gourlay, R.N.; Howard, C.J.; Brownlie, J. Localized immunity in experimental bovine mastitis caused by Mycoplasma dispar. Infect. Immun. 1975, 12, 947–950. [Google Scholar] [CrossRef]
- Reiter, B.; Bramley, A.J. Defence mechanisms of the udder and their relevance to mastitis control. Doc. Int. Dairy Fed. 1975, 85, 210–222. [Google Scholar]
- Watson, D.L. Staphylococcal mastitis vaccine. Vaccine 1992, 10, 359. [Google Scholar] [CrossRef]
- Kennedy, J.W.; Watson, D.L. Cellular basis for differences in humoral immune responses of sheep immunized with living or killed Staphylococcus aureus vaccines. Aust. J. Exp. Biol. Med. Sci. 1982, 60, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.K.K.; Reglinski, M.; Teo, D.; Reza, N.; Lamb, L.E.M.; Nageshwaran, V.; Turner, C.E.; Wikstrom, M.; Frick, I.M.; Bjorck, L.; et al. Vaccine-induced, but not natural immunity, against the Streptococcal inhibitor of complement protects against invasive disease. NPJ Vaccines 2021, 6, 62. [Google Scholar] [CrossRef] [PubMed]
- Vanderhaeghen, W.; Piepers, S.; Leroy, F.; Van Coillie, E.; Haesebrouck, F.; De Vliegher, S. Invited review: Effect, persistence, and virulence of coagulase-negative Staphylococcus species associated with ruminant udder health. J. Dairy Sci. 2014, 97, 5275–5293. [Google Scholar] [CrossRef] [PubMed]
- Burriel, A.R.; Dagnall, G.J. Leukotoxic factors produced by staphylococci of ovine origin. Microbiol. Res. 1997, 152, 247–250. [Google Scholar] [CrossRef]
- Piepers, S.; Opsomer, G.; Barkema, H.W.; de Kruif, A.; De Vliegher, S. Heifers infected with coagulase-negative staphylococci in early lactation have fewer cases of clinical mastitis and higher milk production in their first lactation than noninfected heifers. J. Dairy Sci. 2010, 93, 2014–2024. [Google Scholar] [CrossRef]
- Heikkila, A.M.; Liski, E.; Pyorala, S.; Taponen, S. Pathogen-specific production losses in bovine mastitis. J. Dairy Sci. 2018, 101, 9493–9504. [Google Scholar] [CrossRef]
- Jain, N.C.; Lasmanis, J.; Schalm, O.W. Influence of egg albumin-induced leukopenia on experimental Aerobacter aerogenes mastitis and on natural infection of mammary gland with coagulase-negative staphylococcus in a cow. Am. J. Vet. Res. 1967, 28, 1243–1250. [Google Scholar]
- Scali, F.; Camussone, C.; Calvinho, L.F.; Cipolla, M.; Zecconi, A. Which are important targets in development of S. aureus mastitis vaccine? Res. Vet. Sci. 2015, 100, 88–99. [Google Scholar] [CrossRef]
- Taponen, S.; Pyörälä, S. Coagulase-negative staphylococci as cause of bovine mastitis—Not so different from Staphylococcus aureus? Vet. Microbiol. 2009, 134, 29–36. [Google Scholar] [CrossRef]
- Atalla, H.; Gyles, C.; Mallard, B. Staphylococcus aureus small colony variants (SCVs) and their role in disease. Anim. Health Res. Rev. 2011, 12, 33–45. [Google Scholar] [CrossRef]
- Jensen, N.E. Experimental bovine group-B streptococcal mastitis induced by strains of human and bovine origin. Nord. Vet. Med. 1982, 34, 441–450. [Google Scholar] [PubMed]
- Günther, J.; Czabanska, A.; Bauer, I.; Leigh, J.A.; Holst, O.; Seyfert, H.M. Streptococcus uberis strains isolated from the bovine mammary gland evade immune recognition by mammary epithelial cells, but not of macrophages. Vet. Res. 2016, 47, 13. [Google Scholar] [CrossRef]
- Hossain, M.; Egan, S.A.; Coffey, T.; Ward, P.N.; Wilson, R.; Leigh, J.A.; Emes, R.D. Virulence related sequences; insights provided by comparative genomics of Streptococcus uberis of differing virulence. BMC Genom. 2015, 16, 334. [Google Scholar] [CrossRef] [PubMed]
- Kempf, F.; Slugocki, C.; Blum, S.E.; Leitner, G.; Germon, P. Genomic comparative study of bovine mastitis Escherichia coli. PLoS ONE 2016, 11, e0147954. [Google Scholar] [CrossRef] [PubMed]
- Blum, S.E.; Leitner, G. Genotyping and virulence factors assessment of bovine mastitis Escherichia coli. Vet. Microbiol. 2013, 163, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Herron-Olson, L.; Fitzgerald, J.R.; Musser, J.M.; Kapur, V. molecular correlates of host specialization in Staphylococcus aureus. PLoS ONE 2007, 2, e1120. [Google Scholar] [CrossRef] [PubMed]
- Marbach, H.; Mayer, K.; Vogl, C.; Lee, J.Y.H.; Monk, I.R.; Sordelli, D.O.; Buzzola, F.R.; Ehling-Schulz, M.; Grunert, T. Within-host evolution of bovine Staphylococcus aureus selects for a SigB-deficient pathotype characterized by reduced virulence but enhanced proteolytic activity and biofilm formation. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Tuffs, S.W.; Haeryfar, S.M.M.; McCormick, J.K. manipulation of innate and adaptive immunity by staphylococcal superantigens. Pathogens 2018, 7, 53. [Google Scholar] [CrossRef]
- Zielinski, C.E.; Mele, F.; Aschenbrenner, D.; Jarrossay, D.; Ronchi, F.; Gattorno, M.; Monticelli, S.; Lanzavecchia, A.; Sallusto, F. Pathogen-induced human T(H)17 cells produce IFN-gamma or IL-10 and are regulated by IL-1beta. Nature 2012, 484, 514–518. [Google Scholar] [CrossRef]
- Leimbach, A.; Hacker, J.; Dobrindt, U.E. Coli as an all-rounder: The thin line between commensalism and pathogenicity. Curr. Top. Microbiol. Immunol. 2013, 358, 3–32. [Google Scholar] [CrossRef]
- Gunther, J.; Petzl, W.; Bauer, I.; Ponsuksili, S.; Zerbe, H.; Schuberth, H.J.; Brunner, R.M.; Seyfert, H.M. Differentiating Staphylococcus aureus from Escherichia coli mastitis: S. aureus triggers unbalanced immune-dampening and host cell invasion immediately after udder infection. Sci. Rep. 2017, 7, 4811. [Google Scholar] [CrossRef]
- Wu, J.M.; Ding, Y.L.; Wang, J.L.; Wang, F.L. Staphylococcus aureus induces TGF-beta(1) and bFGF expression through the activation of AP-1 and NF-kappa B transcription factors in bovine mammary epithelial cells. Microb. Pathog. 2018, 117, 276–284. [Google Scholar] [CrossRef]
- Belkaid, Y.; Harrison, O.J. Homeostatic Immunity and the Microbiota. Immunity 2017, 46, 562–576. [Google Scholar] [CrossRef]
- Rabe, H.; Nordstrom, I.; Andersson, K.; Lundell, A.C.; Rudin, A. Staphylococcus aureus convert neonatal conventional CD4(+) T cells into FOXP3(+) CD25(+) CD127(low) T cells via the PD-1/PD-L1 axis. Immunology 2014, 141, 467–481. [Google Scholar] [CrossRef]
- Chen, Z.; Han, Y.; Gu, Y.; Liu, Y.; Jiang, Z.; Zhang, M.; Cao, X. CD11c(high)CD8+ regulatory T cell feedback inhibits CD4 T cell immune response via Fas ligand-Fas pathway. J. Immunol. 2013, 190, 6145–6154. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.S.; Lee, S.U.; Park, Y.H.; Davis, W.C.; Fox, L.K.; Bohach, G.A. Long-term staphylococcal enterotoxin C1 exposure induces soluble factor-mediated immunosuppression by bovine CD4+ and CD8+ T cells. Infect. Immun. 2007, 75, 260–269. [Google Scholar] [CrossRef]
- Eberl, G. A new vision of immunity: Homeostasis of the superorganism. Mucosal Immunol. 2010, 3, 450–460. [Google Scholar] [CrossRef]
- Rainard, P.; Cunha, P.; Martins, R.P.; Gilbert, F.B.; Germon, P.; Foucras, G. Type 3 immunity: A perspective for the defense of the mammary gland against infections. Vet. Res. 2020, 51, 129. [Google Scholar] [CrossRef]
- Matzinger, P. The danger model: A renewed sense of self. Science 2002, 296, 301–305. [Google Scholar] [CrossRef]
- Porcherie, A.; Gilbert, F.B.; Germon, P.; Cunha, P.; Trotereau, A.; Rossignol, C.; Winter, N.; Berthon, P.; Rainard, P. IL-17A is an important effector of the immune response of the mammary gland to Escherichia coli infection. J. Immunol. 2016, 196, 803–812. [Google Scholar] [CrossRef]
- Pandiyan, P.; Conti, H.R.; Zheng, L.; Peterson, A.C.; Mathern, D.R.; Hernandez-Santos, N.; Edgerton, M.; Gaffen, S.L.; Lenardo, M.J. CD4(+)CD25(+)Foxp3(+) regulatory T cells promote Th17 cells in vitro and enhance host resistance in mouse Candida albicans Th17 cell infection model. Immunity 2011, 34, 422–434. [Google Scholar] [CrossRef] [PubMed]
- Broker, B.M.; Holtfreter, S.; Bekeredjian-Ding, I. Immune control of Staphylococcus aureus—Regulation and counter-regulation of the adaptive immune response. Int. J. Med. Microbiol. 2014, 304, 204–214. [Google Scholar] [CrossRef]
- Ziegler, C.; Goldmann, O.; Hobeika, E.; Geffers, R.; Peters, G.; Medina, E. The dynamics of T cells during persistent Staphylococcus aureus infection: From antigen-reactivity to in vivo anergy. EMBO Mol. Med. 2011, 3, 652–666. [Google Scholar] [CrossRef] [PubMed]
- Deplanche, M.; Alekseeva, L.; Semenovskaya, K.; Fu, C.L.; Dessauge, F.; Finot, L.; Petzl, W.; Zerbe, H.; Le Loir, Y.; Rainard, P.; et al. Staphylococcus aureus Phenol-soluble modulins impair interleukin expression in bovine mammary epithelial cells. Infect. Immun. 2016, 84, 1682–1692. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.H.; Fox, L.K.; Hamilton, M.J.; Davis, W.C. Suppression of proliferative response of BoCD4+ T lymphocytes by activated BoCD8+ T lymphocytes in the mammary gland of cows with Staphylococcus aureus mastitis. Vet. Immunol. Immunopathol. 1993, 36, 137–151. [Google Scholar] [CrossRef]
- Kehrli, M. Importance of functional mammary gland immunity during times of stress. In Proceedings of the 41st Annual Meeting, Orlando, FL, USA, 3–6 February 2002; pp. 11–21. [Google Scholar]
- Pulendran, B.; Ahmed, R. Immunological mechanisms of vaccination. Nat. Immunol. 2011, 131, 509–517. [Google Scholar] [CrossRef]
- Gonzalez, R.N.; Cullor, J.S.; Jasper, D.E.; Farver, T.B.; Bushnell, R.B.; Oliver, M.N. Prevention of clinical coliform mastitis in dairy cows by a mutant Escherichia coli vaccine. Can. J. Vet. Res. 1989, 53, 301–305. [Google Scholar]
- Hogan, J.S.; Smith, K.L.; Todhunter, D.A.; Schoenberger, P.S. Field trial to determine efficacy of an Escherichia coli J5 mastitis vaccine. J. Dairy Sci. 1992, 75, 78–84. [Google Scholar] [CrossRef]
- Hill, A.W. Vaccination of cows with rough Escherichia coli mutants fails to protect against experimental intramammary bacterial challenge. Vet. Res. Commun. 1991, 15, 7–16. [Google Scholar] [CrossRef]
- Vangroenweghe, F.; Duchateau, L.; Burvenich, C. Short communication: J-5 Escherichia coli vaccination does not influence severity of an Escherichia coli intramammary challenge in primiparous cows. J. Dairy Sci. 2020, 103, 6692–6697. [Google Scholar] [CrossRef]
- Bradley, A.J.; Breen, J.E.; Payne, B.; White, V.; Green, M.J. An investigation of the efficacy of a polyvalent mastitis vaccine using different vaccination regimens under field conditions in the United Kingdom. J. Dairy Sci. 2015, 98, 1706–1720. [Google Scholar] [CrossRef]
- Lin, J.; Hogan, J.S.; Smith, K.L. Growth responses of coliform bacteria to purified immunoglobulin G from cows immunized with ferric enterobactin receptor FepA. J. Dairy Sci. 1999, 82, 86–92. [Google Scholar] [CrossRef]
- Takemura, K.; Hogan, J.S.; Lin, J.; Smith, K.L. Efficacy of immunization with ferric citrate receptor FecA from Escherichia coli on induced coliform mastitis. J. Dairy Sci. 2002, 85, 774–781. [Google Scholar] [CrossRef]
- Gorden, P.J.; Kleinhenz, M.D.; Ydstie, J.A.; Brick, T.A.; Slinden, L.M.; Peterson, M.P.; Straub, D.E.; Burkhardt, D.T. Efficacy of vaccination with a Klebsiella pneumoniae siderophore receptor protein vaccine for reduction of Klebsiella mastitis in lactating cattle. J. Dairy Sci. 2018, 101, 10398–10408. [Google Scholar] [CrossRef]
- Tomazi, T.; Tomazi, A.C.C.H.; Silva, J.C.C.; Bringhenti, L.; Bravo, M.L.M.C.; Rodrigues, M.X.; Bicalho, R.C. Immunization with a novel recombinant protein (YidR) reduced the risk of clinical mastitis caused by Klebsiella spp. and decreased milk losses and culling risk after Escherichia coli infections. J. Dairy Sci. 2021, 104, 4787–4802. [Google Scholar] [CrossRef]
- Middleton, J.R.; Ma, J.; Rinehart, C.L.; Taylor, V.N.; Luby, C.D.; Steevens, B.J. Efficacy of different Lysigin formulations in the prevention of Staphylococcus aureus intramammary infection in dairy heifers. J. Dairy Res. 2006, 73, s10–s19. [Google Scholar] [CrossRef]
- Middleton, J.R.; Luby, C.D.; Adams, D.S. Efficacy of vaccination against staphylococcal mastitis: A review and new data. Vet. Microbiol. 2009, 134, 192–198. [Google Scholar] [CrossRef]
- Pankey, J.W.; Boddie, N.T.; Watts, J.L.; Nickerson, S.C. Evaluation of protein A and a commercial bacterin as vaccines against Staphylococcus aureus mastitis by experimental challenge. J. Dairy Sci. 1985, 68, 726–731. [Google Scholar] [CrossRef]
- Shkreta, L.; Talbot, B.G.; Diarra, M.S.; Lacasse, P. Immune responses to a DNA/protein vaccination strategy against Staphylococcus aureus induced mastitis in dairy cows. Vaccine 2004, 23, 114–126. [Google Scholar] [CrossRef]
- Hill, A.W.; Finch, J.M.; Field, T.R.; Leigh, J.A. Immune modification of the pathogenesis of Streptococcus uberis mastitis in the dairy cow. FEMS Immunol. Med. Microbiol. 1994, 8, 109–117. [Google Scholar] [CrossRef]
- Siebert, L.; Headrick, S.; Lewis, M.; Gillespie, B.; Young, C.; Wojakiewicz, L.; Kerro-Dego, O.; Prado, M.E.; Almeida, R.; Oliver, S.P.; et al. Genetic variation in CXCR1 haplotypes linked to severity of Streptococcus uberis infection in an experimental challenge model. Vet. Immunol. Immunopathol. 2017, 190, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Collado, R.; Montbrau, C.; Sitja, M.; Prenafeta, A. Study of the efficacy of a Streptococcus uberis mastitis vaccine against an experimental intramammary infection with a heterologous strain in dairy cows. J. Dairy Sci. 2018, 101, 10290–10302. [Google Scholar] [CrossRef]
- Kehrli, M.E., Jr.; Harp, J.A. Immunity in the mammary gland. Vet. Clin. N. Am. Food Anim. Pract. 2001, 17, 495–516. [Google Scholar] [CrossRef]
- Hodgkinson, A.J.; Carpenter, E.A.; Smith, C.S.; Molan, P.C.; Prosser, C.G. Adhesion molecule expression in the bovine mammary gland. Vet. Immunol. Immunopathol. 2007, 115, 205–215. [Google Scholar] [CrossRef]
- Newby, T.J.; Bourne, J. The nature of the local immune system of the bovine mammary gland. J. Immunol. 1977, 118, 461–465. [Google Scholar] [PubMed]
- Hodgkinson, A.J.; Carpenter, E.A.; Smith, C.S.; Molan, P.C.; Prosser, C.G. Effects on adhesion molecule expression and lymphocytes in the bovine mammary gland following intra-mammary immunisation. Vet. Immunol. Immunopathol. 2009, 131, 110–116. [Google Scholar] [CrossRef]
- Masopust, D.; Choo, D.; Vezys, V.; Wherry, E.J.; Duraiswamy, J.; Akondy, R.; Wang, J.; Casey, K.A.; Barber, D.L.; Kawamura, K.S.; et al. Dynamic T cell migration program provides resident memory within intestinal epithelium. J. Exp. Med. 2010, 207, 553–564. [Google Scholar] [CrossRef]
- De Cueninck, B.J. Immune-mediated inflammation in the lumen of the bovine mammary gland. Int. Arch. Allergy Appl. Immunol. 1979, 59, 394–402. [Google Scholar] [CrossRef]
- Rainard, P.; Cunha, P.; Bougarn, S.; Fromageau, A.; Rossignol, C.; Gilbert, B.F.; Berthon, P. T helper 17-associated cytokines are produced during antigen-specific inflammation in the mammary gland. PLoS ONE 2013, 8, e63471. [Google Scholar] [CrossRef]
- Rainard, P.; Cunha, P.; Ledresseur, M.; Staub, C.; Touze, J.L.; Kempf, F.; Gilbert, F.B.; Foucras, G. Antigen-specific mammary inflammation depends on the production of IL-17A and IFN-gamma by Bovine CD4+ T lymphocytes. PLoS ONE 2015, 10, e0137755. [Google Scholar] [CrossRef]
- Cunha, P.; Vern, Y.L.; Gitton, C.; Germon, P.; Foucras, G.; Rainard, P. Expansion, isolation and first characterization of bovine Th17 lymphocytes. Sci. Rep. 2019, 9, 16115. [Google Scholar] [CrossRef] [PubMed]
- Cebron, N.; Maman, S.; Walachowski, S.; Gausseres, B.; Cunha, P.; Rainard, P.; Foucras, G. Mammary Th17-related immunity, but not high systemic Th1 response is associated with protection against E. coli mastitis. NPJ Vaccines 2020, 5, 108. [Google Scholar] [CrossRef]
- Soltys, J.; Quinn, M.T. Selective recruitment of T-cell subsets to the udder during staphylococcal and streptococcal mastitis: Analysis of lymphocyte subsets and adhesion molecule expression. Infect. Immun. 1999, 67, 6293–6302. [Google Scholar] [CrossRef]
- Asai, K.; Yamaguchi, T.; Kuroishi, T.; Komine, Y.; Kai, K.; Komine, K.; Kumagai, K. Differential gene expression of cytokine and cell surface molecules in T cell subpopulation derived from mammary gland secretion of cows. Am. J. Reprod. Immunol. 2003, 50, 453–462. [Google Scholar] [CrossRef]
- Taylor, B.C.; Dellinger, J.D.; Cullor, J.S.; Stott, J.L. Bovine milk lymphocytes display the phenotype of memory T cells and are predominantly CD8+. Cell. Immunol. 1994, 156, 245–253. [Google Scholar] [CrossRef]
- Entrican, G.; Lunney, J.K.; Rutten, V.P.; Baldwin, C.L. A current perspective on availability of tools, resources and networks for veterinary immunology. Vet. Immunol. Immunopathol. 2009, 128, 24–29. [Google Scholar] [CrossRef]
- Wattegedera, S.R.; Corripio-Miyar, Y.; Pang, Y.; Frew, D.; McNeilly, T.N.; Palarea-Albaladejo, J.; McInnes, C.J.; Hope, J.C.; Glass, E.J.; Entrican, G. Enhancing the toolbox to study IL-17A in cattle and sheep. Vet. Res. 2017, 48, 20. [Google Scholar] [CrossRef]
- Thomas, L.H.; Haider, W.; Hill, A.W.; Cook, R.S. Pathologic findings of experimentally induced Streptococcus uberis infection in the mammary gland of cows. Am. J. Vet. Res. 1994, 55, 1723–1728. [Google Scholar]
- Schalm, O.W.; Lasmanis, J.; Carroll, E.J. Pathogenesis of experimental coliform (Aerobacter Aerogenes) mastitis in cattle. Am. J. Vet. Res. 1964, 25, 75–82. [Google Scholar]
- Coliform Subcommittee of The Research Committee of The National Mastitis Council. Coliform mastitis—A review. J. Dairy Sci. 1979, 62, 1–22. [Google Scholar] [CrossRef]
- Paape, M.J.; Shafer-Weaver, K.; Capuco, A.V.; Van Oostveldt, K.; Burvenich, C. Immune surveillance of mammary tissue by phagocytic cells. Adv. Exp. Med. Biol. 2000, 480, 259–277. [Google Scholar] [CrossRef] [PubMed]
- Annunziato, F.; Romagnani, C.; Romagnani, S. The 3 major types of innate and adaptive cell-mediated effector immunity. J. Allergy Clin. Immunol. 2015, 135, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Tassi, R.; McNeilly, T.N.; Fitzpatrick, J.L.; Fontaine, M.C.; Reddick, D.; Ramage, C.; Lutton, M.; Schukken, Y.H.; Zadoks, R.N. Strain-specific pathogenicity of putative host-adapted and nonadapted strains of Streptococcus uberis in dairy cattle. J. Dairy Sci. 2013, 96, 5129–5145. [Google Scholar] [CrossRef]
- Herry, V.; Gitton, C.; Tabouret, G.; Reperant, M.; Forge, L.; Tasca, C.; Gilbert, F.B.; Guitton, E.; Barc, C.; Staub, C.; et al. Local immunization impacts the response of dairy cows to Escherichia coli mastitis. Sci. Rep. 2017, 7, 3441. [Google Scholar] [CrossRef]
- Tao, W.; Mallard, B. Differentially expressed genes associated with Staphylococcus aureus mastitis of Canadian Holstein cows. Vet. Immunol. Immunopathol. 2007, 120, 201–211. [Google Scholar] [CrossRef]
- Rainard, P.; Gitton, C.; Chaumeil, T.; Fassier, T.; Huau, C.; Riou, M.; Tosser-Klopp, G.; Krupova, Z.; Chaize, A.; Gilbert, F.B.; et al. Host factors determine the evolution of infection with Staphylococcus aureus to gangrenous mastitis in goats. Vet. Res. 2018, 49, 72. [Google Scholar] [CrossRef] [PubMed]
- Roussel, P.; Cunha, P.; Porcherie, A.; Petzl, W.; Gilbert, F.B.; Riollet, C.; Zerbe, H.; Rainard, P.; Germon, P. Investigating the contribution of IL-17A and IL-17F to the host response during Escherichia coli mastitis. Vet. Res. 2015, 46, 56. [Google Scholar] [CrossRef]
- Blum, S.E.; Heller, E.D.; Jacoby, S.; Krifucks, O.; Leitner, G. Comparison of the immune responses associated with experimental bovine mastitis caused by different strains of Escherichia coli. J. Dairy Res. 2017, 84, 190–197. [Google Scholar] [CrossRef]
- Andreotti, C.S.; Baravalle, C.; Sacco, S.C.; Lovato, M.; Pereyra, E.A.L.; Renna, M.S.; Ortega, H.H.; Calvinho, L.F.; Dallard, B.E. Characterization of immune response in Staphylococcus aureus chronically infected bovine mammary glands during active involution. Comp. Immunol. Microbiol. Infect. Dis. 2017, 54, 51–60. [Google Scholar] [CrossRef]
- Bruno, D.R.; Rossitto, P.V.; Bruno, R.G.; Blanchard, M.T.; Sitt, T.; Yeargan, B.V.; Smith, W.L.; Cullor, J.S.; Stott, J.L. Differential levels of mRNA transcripts encoding immunologic mediators in mammary gland secretions from dairy cows with subclinical environmental Streptococci infections. Vet. Immunol. Immunopathol. 2010, 138, 15–24. [Google Scholar] [CrossRef]
- Dawson, C.A.; Pal, B.; Vaillant, F.; Gandolfo, L.C.; Liu, Z.Y.; Bleriot, C.; Ginhoux, F.; Smyth, G.K.; Lindeman, G.J.; Mueller, S.N.; et al. Tissue-resident ductal macrophages survey the mammary epithelium and facilitate tissue remodelling. Nat. Cell Biol. 2020, 22, 546–558. [Google Scholar] [CrossRef] [PubMed]
- Hassel, C.; Gausserès, B.; Guzylack-Piriou, L.; Foucras, G. Ductal macrophages predominate in the immune landscape of the lactating mammary gland. Front. Immunol. 2021, 12, 754661. [Google Scholar] [CrossRef]
- Revu, S.; Wu, J.; Henkel, M.; Rittenhouse, N.; Menk, A.; Delgoffe, G.M.; Poholek, A.C.; McGeachy, M.J. IL-23 and IL-1beta drive human Th17 cell differentiation and metabolic reprogramming in absence of CD28 costimulation. Cell Rep. 2018, 22, 2642–2653. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Pancari, G.; Cope, L.; Bowman, E.P.; Cua, D.; Proctor, R.A.; McNeely, T. Immunization with Staphylococcus aureus iron regulated surface determinant B (IsdB) confers protection via Th17/IL17 pathway in a murine sepsis model. Hum. Vacc. Immunother. 2012, 8, 336–346. [Google Scholar] [CrossRef]
- Narita, K.; Hu, D.L.; Mori, F.; Wakabayashi, K.; Iwakura, Y.; Nakane, A. Role of interleukin-17A in cell-mediated protection against Staphylococcus aureus infection in mice immunized with the fibrinogen-binding domain of clumping factor A. Infect. Immun. 2010, 78, 4234–4242. [Google Scholar] [CrossRef]
- Rainard, P.; Cunha, P.; Gilbert, F.B. Innate and Adaptive Immunity Synergize to Trigger Inflammation in the Mammary Gland. PLoS ONE 2016, 11, e0154172. [Google Scholar] [CrossRef]
- Denis, M.; Lacy-Hulbert, S.J.; Buddle, B.M.; Williamson, J.H.; Wedlock, D.N. Streptococcus uberis-specific T cells are present in mammary gland secretions of cows and can be activated to kill. Vet. Res. Commun. 2011, 35, 145–156. [Google Scholar] [CrossRef]
- Denis, M.; Wedlock, D.N.; Lacy-Hulbert, S.J.; Hillerton, J.E.; Buddle, B.M. Vaccines against bovine mastitis in the New Zealand context: What is the best way forward? N. Z. Vet. J. 2009, 57, 132–140. [Google Scholar] [CrossRef]
- Joffre, O.P.; Segura, E.; Savina, A.; Amigorena, S. Cross-presentation by dendritic cells. Nat. Rev. Immunol. 2012, 12, 557–569. [Google Scholar] [CrossRef]
- Halle, S.; Halle, O.; Forster, R. Mechanisms and dynamics of t cell-mediated cytotoxicity in vivo. Trends Immunol. 2017, 38, 432–443. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Hiratsuka, M.; Asai, K.; Kai, K.; Kumagai, K. Differential distribution of T lymphocyte subpopulations in the bovine mammary gland during lactation. J. Dairy Sci. 1999, 82, 1459–1464. [Google Scholar] [CrossRef]
- Tatarczuch, L.; Philip, C.; Bischof, R.; Lee, C.S. Leucocyte phenotypes in involuting and fully involuted mammary glandular tissues and secretions of sheep. J. Anat. 2000, 196 Pt 3, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Cheroutre, H.; Husain, M.M. CD4 CTL: Living up to the challenge. Semin. Immunol. 2013, 25, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; McAleer, J.P.; Lin, Y.; Paterson, D.L.; Zheng, M.; Alcorn, J.F.; Weaver, C.T.; Kolls, J.K. Th17 cells mediate clade-specific, serotype-independent mucosal immunity. Immunity 2011, 35, 997–1009. [Google Scholar] [CrossRef]
- Kumar, P.; Chen, K.; Kolls, J.K. Th17 cell based vaccines in mucosal immunity. Curr. Opin. Immunol. 2013, 25, 373–380. [Google Scholar] [CrossRef]
- Robinson, H.L.; Amara, R.R. T cell vaccines for microbial infections. Nat. Med. 2005, 11, S25–S32. [Google Scholar] [CrossRef]
- Burchill, M.A.; Tamburini, B.A.; Pennock, N.D.; White, J.T.; Kurche, J.S.; Kedl, R.M. T cell vaccinology: Exploring the known unknowns. Vaccine 2013, 31, 297–305. [Google Scholar] [CrossRef]
- Côté-Gravel, J.; Brouillette, E.; Obradovic, N.; Ster, C.; Talbot, B.G.; Malouin, F. Characterization of a vraG mutant in a genetically stable Staphylococcus aureus small-colony variant and preliminary assessment for use as a live-attenuated vaccine against intrammamary infections. PLoS ONE 2016, 11, e0166621. [Google Scholar] [CrossRef]
- Qin, L.; Gilbert, P.B.; Corey, L.; McElrath, M.J.; Self, S.G. A framework for assessing immunological correlates of protection in vaccine trials. J. Infect. Dis. 2007, 196, 1304–1312. [Google Scholar] [CrossRef]
- Sadoff, J.C.; Wittes, J. Correlates, surrogates, and vaccines. J. Infect. Dis. 2007, 196, 1279–1281. [Google Scholar] [CrossRef]
- Zinkernagel, R.M.; Hengartner, H. Protective “immunity” by pre-existent neutralizing antibody titers and preactivated T cells but not by so-called “immunological memory”. Immunol. Rev. 2006, 211, 310–319. [Google Scholar] [CrossRef]
- Erskine, R.J. Vaccination strategies for mastitis. Vet. Clin. N. Am. Food Anim. Pract. 2012, 28, 257–270. [Google Scholar] [CrossRef]
- Rainard, P.; Reperant-Ferter, M.; Gitton, C.; Germon, P. Shielding Effect of Escherichia coli O-Antigen Polysaccharide on J5-Induced Cross-Reactive Antibodies. mSphere 2021, 6, e01227-20. [Google Scholar] [CrossRef]
- Thompson-Crispi, K.A.; Miglior, F.; Mallard, B.A. Genetic parameters for natural antibodies and associations with specific antibody and mastitis in Canadian Holsteins. J. Dairy Sci. 2013, 96, 3965–3972. [Google Scholar] [CrossRef]
- Abdellrazeq, G.S.; Elnaggar, M.M.; Bannantine, J.P.; Schneider, D.A.; Souza, C.D.; Hwang, J.; Mahmoud, A.H.A.; Hulubei, V.; Fry, L.M.; Park, K.T.; et al. A peptide-based vaccine for Mycobacterium avium subspecies paratuberculosis. Vaccine 2019, 37, 2783–2790. [Google Scholar] [CrossRef]
- Hope, J.C.; Thom, M.L.; McAulay, M.; Mead, E.; Vordermeier, H.M.; Clifford, D.; Hewinson, R.G.; Villarreal-Ramos, B. Identification of surrogates and correlates of protection in protective immunity against mycobacterium bovis infection induced in neonatal calves by vaccination with M. bovis BCG Pasteur and M. bovis BCG Danish. Clin. Vaccine Immunol. 2011, 18, 373–379. [Google Scholar] [CrossRef]
- Kitano, H. Systems biology: A brief overview. Science 2002, 295, 1662–1664. [Google Scholar] [CrossRef]
- Querec, T.D.; Akondy, R.S.; Lee, E.K.; Cao, W.; Nakaya, H.I.; Teuwen, D.; Pirani, A.; Gernert, K.; Deng, J.; Marzolf, B.; et al. Systems biology approach predicts immunogenicity of the yellow fever vaccine in humans. Nat. Immunol. 2009, 10, 116–125. [Google Scholar] [CrossRef]
- Chaussabel, D.; Baldwin, N. Democratizing systems immunology with modular transcriptional repertoire analyses. Nat. Rev. Immunol. 2014, 14, 271–280. [Google Scholar] [CrossRef]
- Sharifi, S.; Pakdel, A.; Ebrahimie, E.; Aryan, Y.; Ghaderi Zefrehee, M.; Reecy, J.M. Prediction of key regulators and downstream targets of E. coli induced mastitis. J. Appl. Genet. 2019, 60, 367–373. [Google Scholar] [CrossRef]
- Thomas, F.C.; Mullen, W.; Tassi, R.; Ramirez-Torres, A.; Mudaliar, M.; McNeilly, T.N.; Zadoks, R.N.; Burchmore, R.; Eckersall, P.D. Mastitomics, the integrated omics of bovine milk in an experimental model of Streptococcus uberis mastitis: 1. High abundance proteins, acute phase proteins and peptidomics. Mol. Biosyst. 2016, 12, 2735–2747. [Google Scholar] [CrossRef]
- Awate, S.; Babiuk, L.A.; Mutwiri, G. Mechanisms of action of adjuvants. Front. Immunol. 2013, 4, 114. [Google Scholar] [CrossRef]
- Blander, J.M.; Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 2006, 440, 808–812. [Google Scholar] [CrossRef]
- Coffman, R.L.; Sher, A.; Seder, R.A. Vaccine adjuvants: Putting innate immunity to work. Immunity 2010, 33, 492–503. [Google Scholar] [CrossRef]
- Frodermann, V.; Chau, T.A.; Sayedyahossein, S.; Toth, J.M.; Heinrichs, D.E.; Madrenas, J. A Modulatory Interleukin-10 Response to Staphylococcal Peptidoglycan Prevents Th1/Th17 Adaptive Immunity to Staphylococcus aureus. J. Infect. Dis. 2011, 204, 253–262. [Google Scholar] [CrossRef]
- Zielinski, C.E. Microbe driven T-helper cell differentiation: Lessons from Candida albicans and Staphylococcus aureus. Exp. Dermatol. 2014, 23, 795–798. [Google Scholar] [CrossRef]
- Côté-Gravel, J.; Brouillette, E.; Malouin, F. Vaccination with a live-attenuated small-colony variant improves the humoral and cell-mediated responses against Staphylococcus aureus. PLoS ONE 2019, 14, e0227109. [Google Scholar] [CrossRef]
- Bagnoli, F.; Bertholet, S.; Grandi, G. Inferring reasons for the failure of Staphylococcus aureus vaccines in clinical trials. Front. Cell. Infect. Microbiol. 2012, 2, 16. [Google Scholar] [CrossRef]
- De Cueninck, B.J. Expression of cell-mediated hypersensitivity in the lumen of the mammary gland in guinea pigs. Am. J. Vet. Res. 1982, 43, 1696–1700. [Google Scholar] [PubMed]
- Agrawal, S.; Gupta, S.; Agrawal, A. Human dendritic cells activated via dectin-1 are efficient at priming Th17, cytotoxic CD8 T and B cell responses. PLoS ONE 2010, 5, e13418. [Google Scholar] [CrossRef]
- Thakur, A.; Andrea, A.; Mikkelsen, H.; Woodworth, J.S.; Andersen, P.; Jungersen, G.; Aagaard, C. Targeting the Mincle and TLR3 receptor using the dual agonist cationic adjuvant formulation 9 (CAF09) induces humoral and polyfunctional memory T cell responses in calves. PLoS ONE 2018, 13, e0201253. [Google Scholar] [CrossRef] [PubMed]
- Didierlaurent, A.M.; Morel, S.; Lockman, L.; Giannini, S.L.; Bisteau, M.; Carlsen, H.; Kielland, A.; Vosters, O.; Vanderheyde, N.; Schiavetti, F.; et al. AS04, an aluminum salt- and TLR4 agonist-based adjuvant system, induces a transient localized innate immune response leading to enhanced adaptive immunity. J. Immunol. 2009, 183, 6186–6197. [Google Scholar] [CrossRef] [PubMed]
- Souza, C.D.; Bannantine, J.P.; Brown, W.C.; Norton, M.G.; Davis, W.C.; Hwang, J.K.; Ziaei, P.; Abdellrazeq, G.S.; Eren, M.V.; Deringer, J.R.; et al. A nano particle vector comprised of poly lactic-co-glycolic acid and monophosphoryl lipid A and recombinant Mycobacterium avium subsp paratuberculosis peptides stimulate a pro-immune profile in bovine macrophages. J. Appl. Microbiol. 2017, 123, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, S.; Takagi, R.; Hashimoto, K.; Higashi, T. Qualitative evaluation of adjuvant activities and its application to Th2/17 diseases. Int. Arch. Allergy Immunol. 2011, 155 (Suppl. 1), 2–5. [Google Scholar] [CrossRef]
- Vautier, S.; Sousa, M.G.; Brown, G.D. C-type lectins, fungi and Th17 responses. Cytokine Growth Factor Rev. 2010, 21, 405–412. [Google Scholar] [CrossRef]
- Mutwiri, G.; Gerdts, V.; van Drunen Littel-van den Hurk, S.; Auray, G.; Eng, N.; Garlapati, S.; Babiuk, L.A.; Potter, A. Combination adjuvants: The next generation of adjuvants? Expert Rev. Vaccines 2011, 10, 95–107. [Google Scholar] [CrossRef]
- Lascelles, A.K. The immune system of the ruminant mammary gland and its role in the control of mastitis. J. Dairy Sci. 1979, 62, 154–167. [Google Scholar] [CrossRef]
- Nashar, T.O.; Stokes, C.R.; Cripps, P.J. Immune responses to intramammary infusion with soluble (ovalbumin) and particulate (S. uberis) antigens in the preparturient bovine udder. Res. Vet. Sci. 1991, 50, 145–151. [Google Scholar] [CrossRef]
- Misra, N.; Wines, T.F.; Knopp, C.L.; Hermann, R.; Bond, L.; Mitchell, B.; McGuire, M.A.; Tinker, J.K. Immunogenicity of a Staphylococcus aureus-cholera toxin A(2)/B vaccine for bovine mastitis. Vaccine 2018, 36, 3513–3521. [Google Scholar] [CrossRef]
- Nagasawa, Y.; Kiku, Y.; Sugawara, K.; Hirose, A.; Kai, C.; Kitano, N.; Takahashi, T.; Nochi, T.; Aso, H.; Sawada, S.I.; et al. Staphylococcus aureus-specific IgA antibody in milk suppresses the multiplication of S. aureus in infected bovine udder. BMC Vet. Res. 2019, 15, 286. [Google Scholar] [CrossRef]
- Alabdullah, H.A.; Overgaard, E.; Scarbrough, D.; Williams, J.E.; Mohammad Mousa, O.; Dunn, G.; Bond, L.; McGuire, M.A.; Tinker, J.K. Evaluation of the efficacy of a cholera-toxin-based Staphylococcus aureus vaccine against bovine intramammary challenge. Vaccines 2021, 9, 6. [Google Scholar] [CrossRef]
- Chang, C.C.; Winter, A.J.; Norcross, N.L. Immune response in the bovine mammary gland after intestinal, local, and systemic immunization. Infect. Immun. 1981, 31, 650–659. [Google Scholar] [CrossRef]
- Guidry, A.J.; Berning, L.M.; Hambleton, C.N. Opsonization of Staphylococcus aureus by bovine immunoglobulin isotypes. J. Dairy Sci. 1993, 76, 1285–1289. [Google Scholar] [CrossRef]
- Bachmann, M.F.; Jennings, G.T. Vaccine delivery: A matter of size, geometry, kinetics and molecular patterns. Nat. Rev. Immunol. 2010, 10, 787–796. [Google Scholar] [CrossRef]
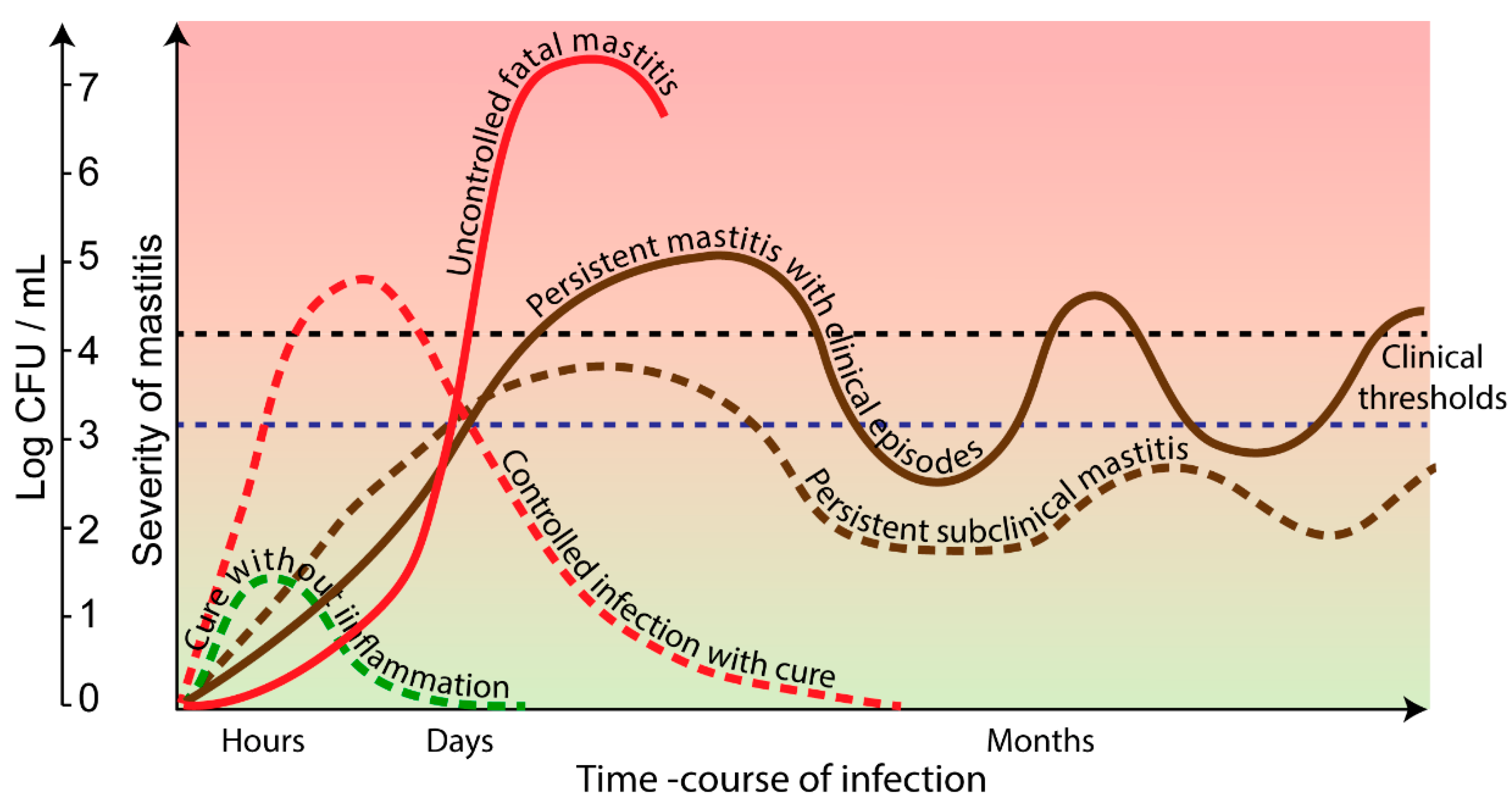
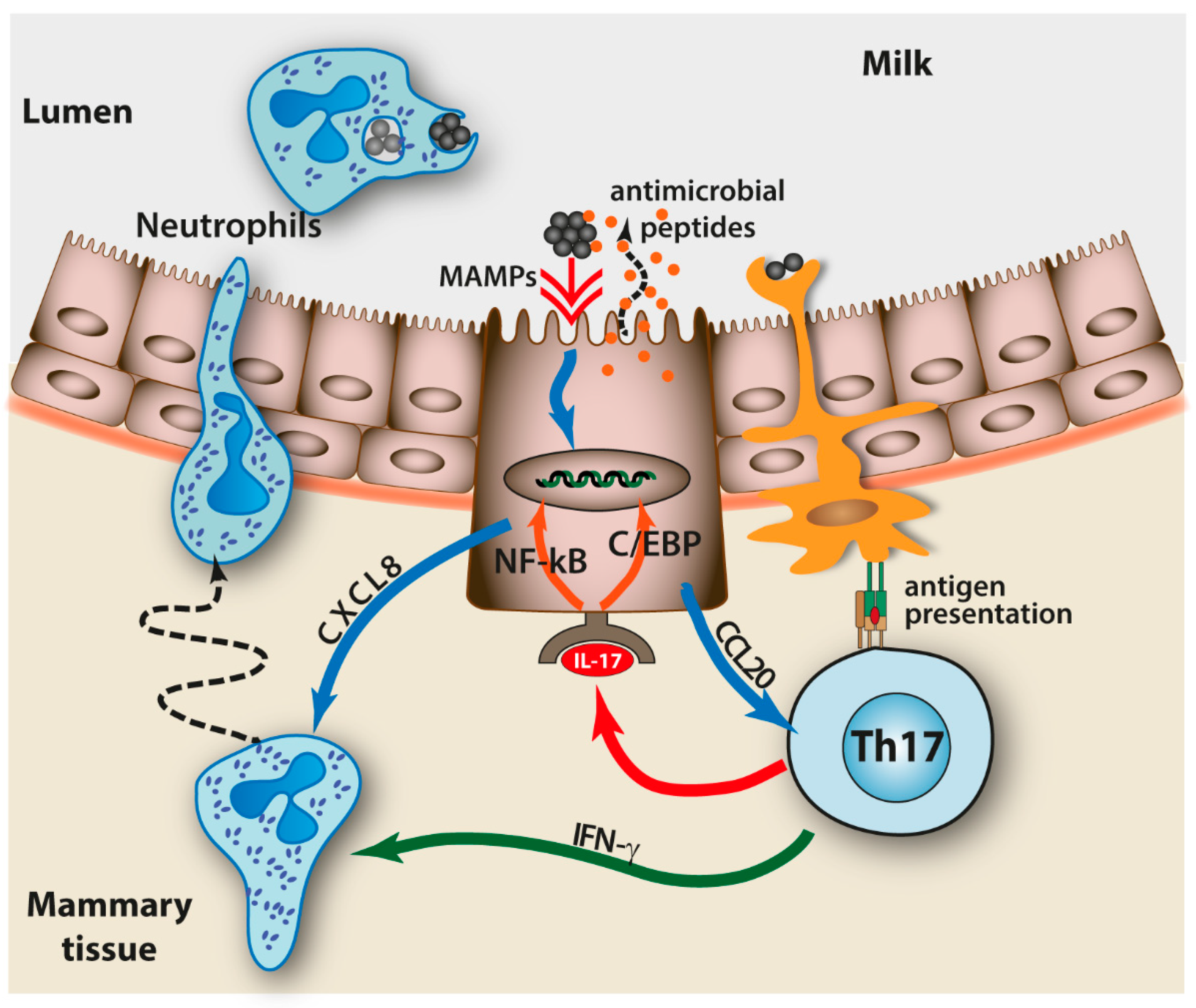
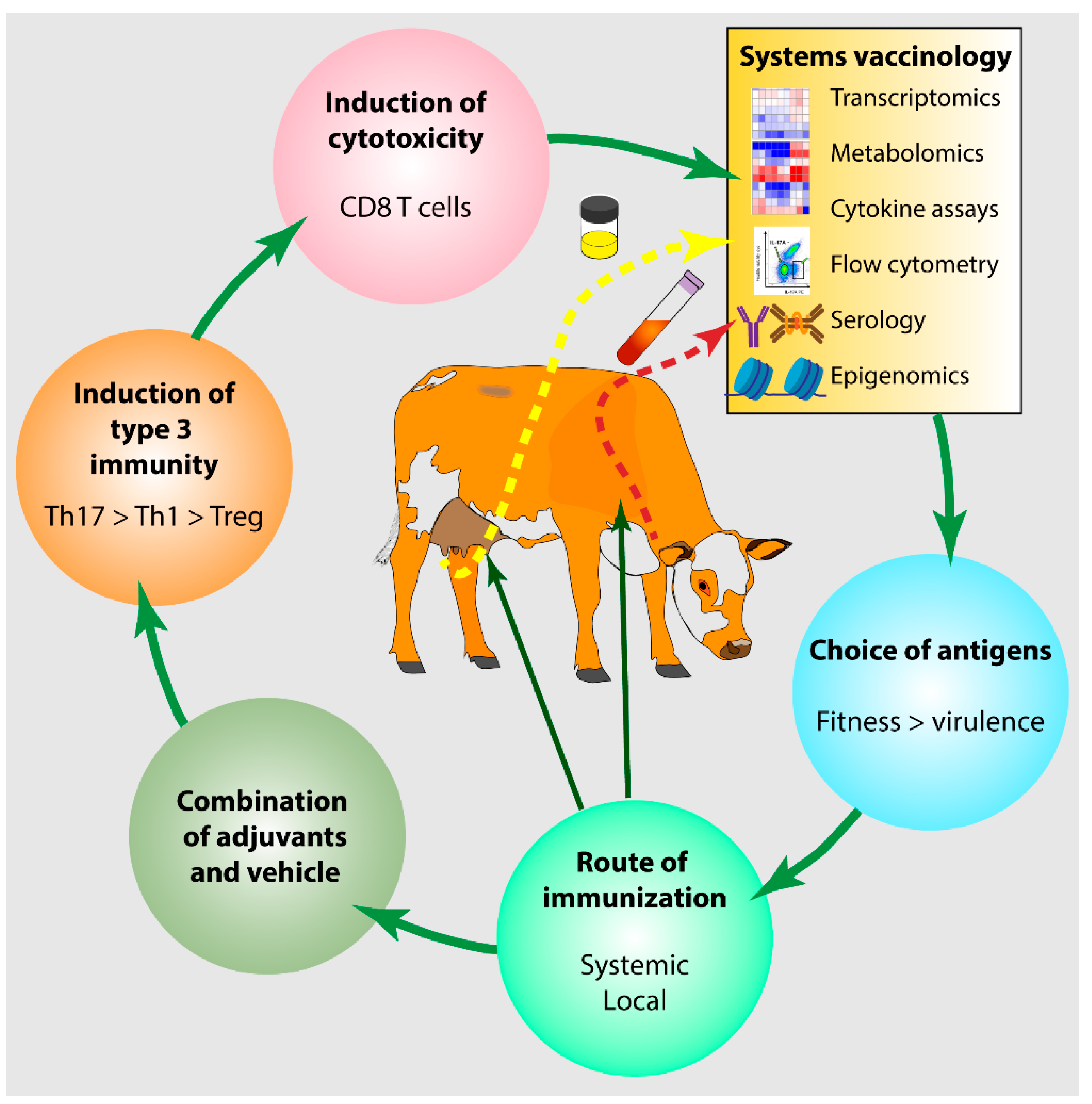
| Mammary Gland Features | Consequence for Defense Efficiency |
|---|---|
| Milk is a rich growth medium for many bacteria | Potentially high bacterial load |
| Dilution of antimicrobial defenses in milk | Reduced efficacy of antimicrobial agents |
| Absence of mucus barrier | Reduced efficiency of antibodies (sIgA) 1 and AMPs 2 |
| Impediments to phagocytes (casein, fat globules, low oxygen tension) | Reduced phagocytic efficiency, need for massive leucocyte recruitment |
| Quenching of ROS 3 and AMPs 1 by milk | Blunting of antimicrobial activity |
| No need for specific virulence factors | No main target for the immune response other than fitness factors |
| Adaptation to the MG niche | Immune evasion |
| No protection following infection | Frequent recurrences |
| Vaccine Antigens | Efficacy | Shortcomings | References |
|---|---|---|---|
| E. coliJ5 bacterins | Decreased severity of coliform mastitis in field experiments, little effect in experimental infections | Little effect on incidence of cases, variable among herds and experiments. Unknown mechanism | [80,81,82,83] |
| E. coliJ5 bacterin with killed S. aureus (StartVac®, Hipra) | Decreased mastitis severity in field studies | No effect on incidence of cases. Unknown mechanism | [84] |
| E. colienterobactin FepA or siderophore receptor FecA | Reduction of bacterial growth in vitro | Not tested in vivo (FepA) or not effective in challenge experiment (FecA) | [85,86] |
| Klebsiellasiderophore receptors and porin proteins (KlebVax™) | Little reduction in risk of coliform mastitis, some increase in milk yield | Administration with a J5 vaccine confounding the interpretation. Effect variable depending on experiments | [87,88] |
| S. aureusbacterins and toxoid or bacterial lysate | Some reduction in severity and incidence of mastitis | Variable results, little prevention of chronic infections | [89,90] |
| S. aureusprotein A | Increased spontaneous cure after experimental challenge | Not tested in field conditions Mechanism not identified | [91] |
| S. aureusFnBP and ClfA | Increased spontaneous cure after experimental challenge | Not tested in field conditions Mechanism not identified | [92] |
| S. uberislive bacteria and surface extract | Reduction in bacterial shedding in milk and local inflammation | Not tested in field conditions Mechanism not identified | [93] |
| S. uberisSUAM | Not reported | All cows developed mastitis. Not tested in field conditions | [94] |
| S. uberisslime preparation (UBAC®, Hipra) | Reduction in milk production losses and incidence of clinical mastitis cases | Few published field experiments Little confirmed effect on the prevalence of infections. Mechanisms not identified | [95] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rainard, P.; Gilbert, F.B.; Martins, R.P.; Germon, P.; Foucras, G. Progress towards the Elusive Mastitis Vaccines. Vaccines 2022, 10, 296. https://doi.org/10.3390/vaccines10020296
Rainard P, Gilbert FB, Martins RP, Germon P, Foucras G. Progress towards the Elusive Mastitis Vaccines. Vaccines. 2022; 10(2):296. https://doi.org/10.3390/vaccines10020296
Chicago/Turabian StyleRainard, Pascal, Florence B. Gilbert, Rodrigo P. Martins, Pierre Germon, and Gilles Foucras. 2022. "Progress towards the Elusive Mastitis Vaccines" Vaccines 10, no. 2: 296. https://doi.org/10.3390/vaccines10020296
APA StyleRainard, P., Gilbert, F. B., Martins, R. P., Germon, P., & Foucras, G. (2022). Progress towards the Elusive Mastitis Vaccines. Vaccines, 10(2), 296. https://doi.org/10.3390/vaccines10020296






