Factors Affecting Young Adults’ Decision Making to Undergo COVID-19 Vaccination: A Patient Preference Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Discrete Choice Experiment Design
2.2.1. Attributes and Levels
2.2.2. Survey Development
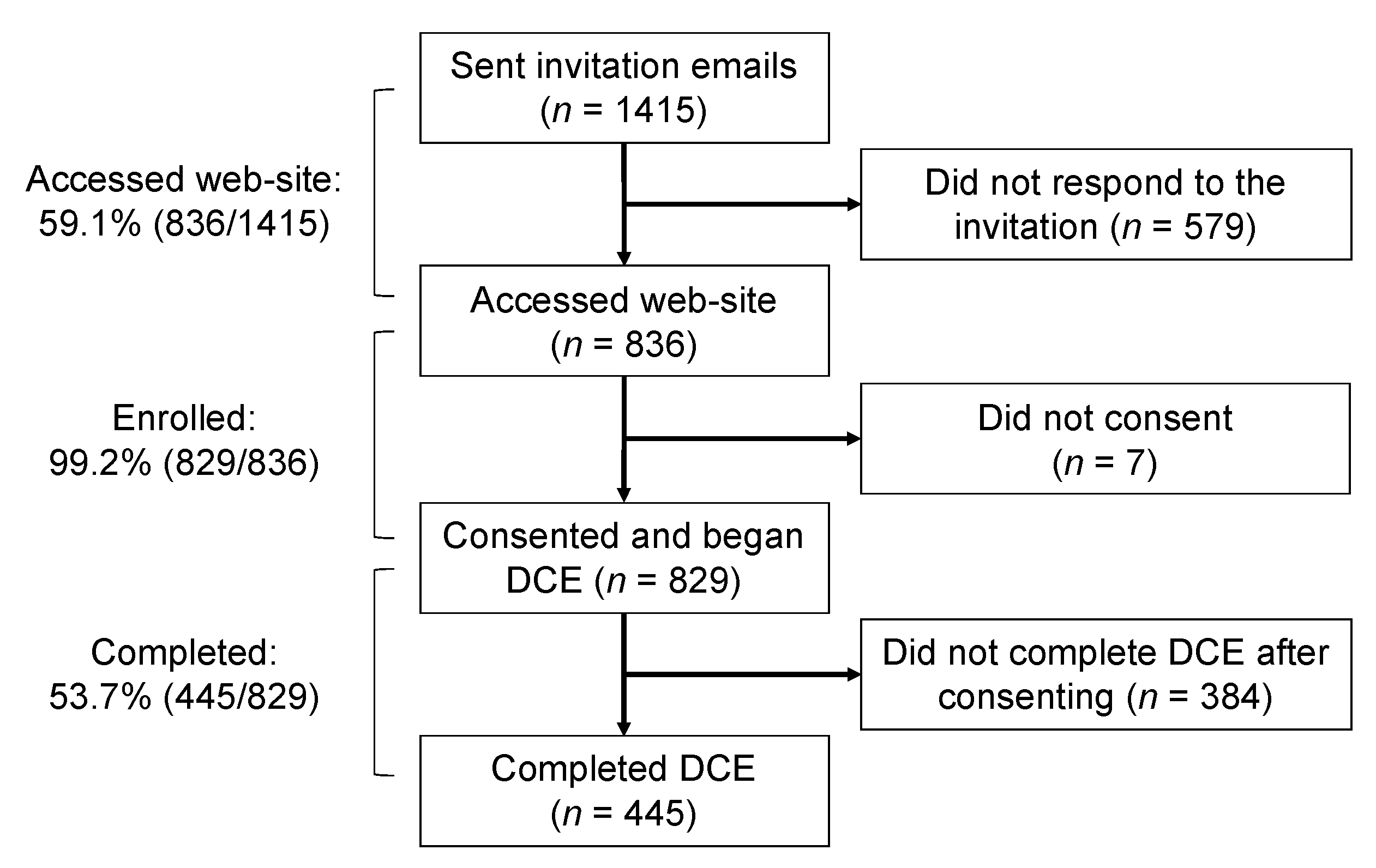
2.3. Setting, Participants, and Recruitment
2.4. Experimental Design and Variables
2.5. Analysis
2.5.1. Discrete Choice Experiment
2.5.2. Predictors of Vaccine Hesitancy
3. Results
3.1. Study Population
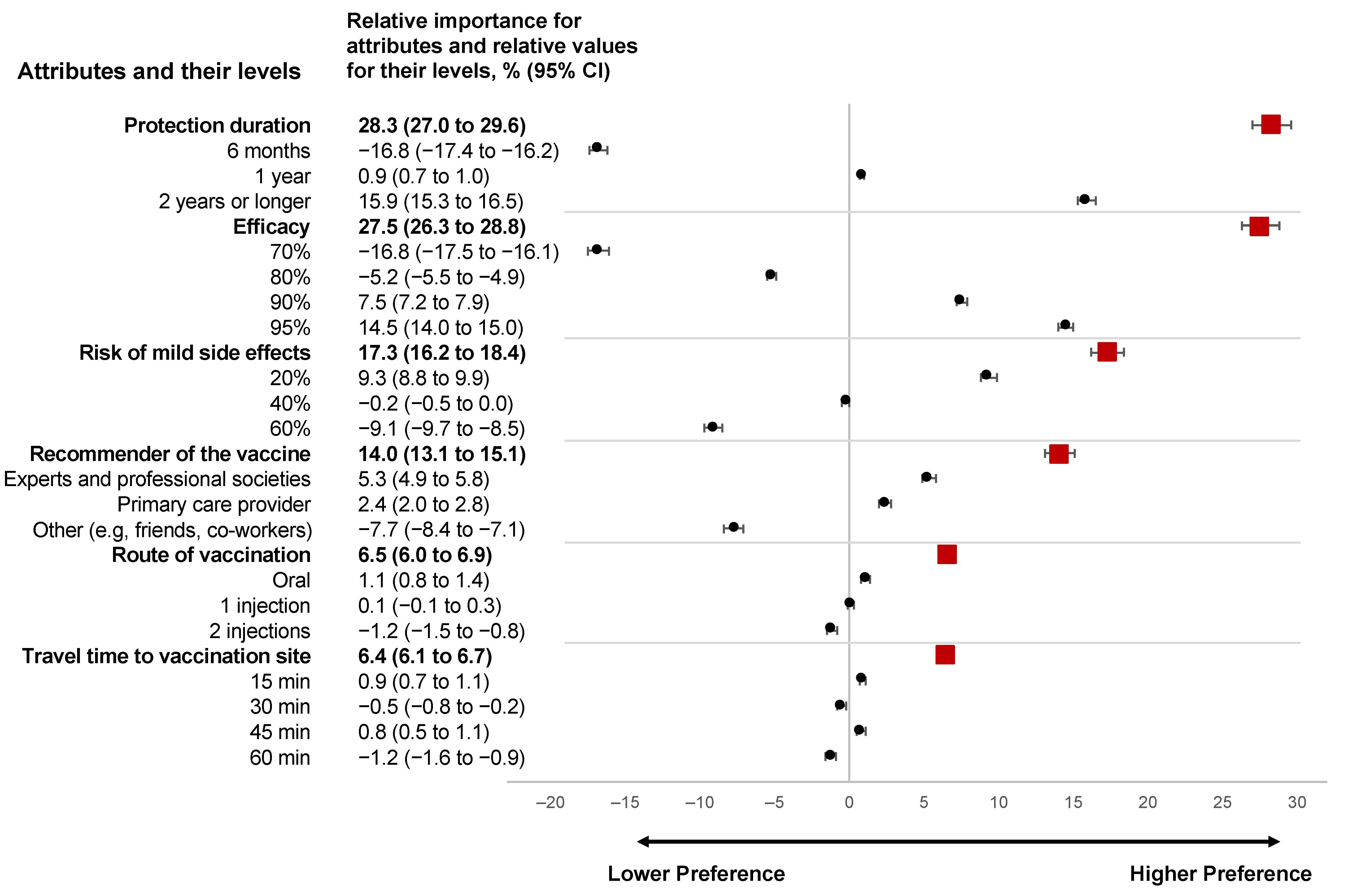
3.2. Discrete Choice Experiment
3.3. DCE Subgroup Analysis
3.4. Predictors of Vaccine Hesitancy
4. Discussion
4.1. Future Research
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Woolf, S.H.; Chapman, D.A.; Lee, J.H. COVID-19 as the Leading Cause of Death in the United States. JAMA 2020, 325, 123–124. [Google Scholar] [CrossRef]
- Panagiotou, O.A.; Kosar, C.M.; White, E.M.; Bantis, L.E.; Yang, X.; Santostefano, C.M.; Feifer, R.A.; Blackman, C.; Rudolph, J.L.; Gravenstein, S.; et al. Risk Factors Associated With All-Cause 30-Day Mortality in Nursing Home Residents With COVID-19. JAMA Intern. Med. 2021, 181, 439. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A.; Axfors, C.; Contopoulos-Ioannidis, D.G. Population-Level COVID-19 Mortality Risk for Non-elderly Individuals Overall and for Non-elderly Individuals without Underlying Diseases in Pandemic Epicenters. Environ. Res. 2020, 188, 109890. [Google Scholar] [CrossRef]
- Faust, J.S.; Krumholz, H.M.; Du, C.; Mayes, K.D.; Lin, Z.; Gilman, C.; Walensky, R.P. All-Cause Excess Mortality and COVID-19-Related Mortality Among US Adults Aged 25–44 Years, March–July 2020. JAMA 2021, 325, 785–787. [Google Scholar] [CrossRef]
- Shiels, M.S.; Almeida, J.S.; García-Closas, M.; Albert, P.S.; Freedman, N.D.; de González, A.B. Impact of Population Growth and Aging on Estimates of Excess U.S. Deaths During the COVID-19 Pandemic, March to August 2020. Ann. Intern. Med. 2021, 174, 437–443. [Google Scholar] [CrossRef]
- Vestergaard, L.S.; Nielsen, J.; Richter, L.; Schmid, D.; Bustos, N.; Braeye, T.; Denissov, G.; Veideman, T.; Luomala, O.; Möttönen, T.; et al. Excess All-Cause Mortality during the COVID-19 Pandemic in Europe—Preliminary Pooled Estimates from The EuroMOMO Network, March to April 2020. Eurosurveillance 2020, 25, 2001214. [Google Scholar] [CrossRef]
- Boehmer, T.K.; DeVies, J.; Caruso, E.; van Santen, K.L.; Tang, S.; Black, C.L.; Hartnett, K.P.; Kite-Powell, A.; Dietz, S.; Lozier, M.; et al. Changing Age Distribution of the COVID-19 Pandemic—United States, May–August 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 1404–1409. [Google Scholar] [CrossRef]
- Salvatore, P.P.; Sula, E.; Coyle, J.P.; Caruso, E.; Smith, A.R.; Levine, R.S.; Baack, B.N.; Mir, R.; Lockhart, E.R.; Tiwari, T.S.; et al. Recent Increase in COVID-19 Cases Reported Among Adults Aged 18–22 Years—United States, May 31–September 5, 2020. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 1419–1424. [Google Scholar] [CrossRef]
- Cunningham, J.W.; Vaduganathan, M.; Claggett, B.L.; Jering, K.S.; Bhatt, A.S.; Rosenthal, N.; Solomon, S.D. Clinical Outcomes in Young US Adults Hospitalized With COVID-19. JAMA Intern. Med. 2021, 181, 379. [Google Scholar] [CrossRef]
- Flasche, S.; Edmunds, W.J. The role of schools and schoolaged children in SARS-CoV-2 transmission. Lancet Infect. Dis. 2021, 21, 298–299. [Google Scholar] [CrossRef]
- Casini, L.; Roccetti, M. Reopening Italy’s schools in September 2020: A Bayesian estimation of the change in the growth rate of new SARS-CoV-2 cases. BMJ Open 2021, 11, e051458. [Google Scholar] [CrossRef]
- Daly, M.; Jones, A.; Robinson, E. Public Trust and Willingness to Vaccinate Against COVID-19 in the US From October 14, 2020, to March 29, 2021. JAMA 2021, 325, 2397–2399. [Google Scholar] [CrossRef]
- Szilagyi, P.G.; Thomas, K.; Shah, M.D.; Vizueta, N.; Cui, Y.; Vangala, S.; Kapteyn, A. Changes in COVID-19 Vaccine Intent From April/May to June/July 2021. JAMA 2021, 326, 1971–1974. [Google Scholar] [CrossRef]
- Mascola, J.R.; Graham, B.S.; Fauci, A.S. SARS-CoV-2 Viral Variants-Tackling a Moving Target. JAMA 2021, 325, 1261–1262. [Google Scholar] [CrossRef]
- Schaffer DeRoo, S.; Pudalov, N.J.; Fu, L.Y. Planning for a COVID-19 Vaccination Program. JAMA 2020, 323, 2458. [Google Scholar] [CrossRef]
- Lazarus, J.V.; Ratzan, S.C.; Palayew, A.; Gostin, L.O.; Larson, H.J.; Rabin, K.; Kimball, S.; El-Mohandes, A. A global survey of potential acceptance of a COVID-19 vaccine. Nat. Med. 2021, 27, 225–228. [Google Scholar] [CrossRef]
- Wong, L.P.; Alias, H.; Danaee, M.; Ahmed, J.; Lachyan, A.; Cai, C.Z.; Lin, Y.; Hu, Z.; Tan, S.Y.; Lu, Y.; et al. COVID-19 Vaccination Intention and Vaccine Characteristics Influencing Vaccination Acceptance: A Global Survey of 17 Countries. Infect. Dis. Poverty 2021, 10, 122. [Google Scholar] [CrossRef]
- Detoc, M.; Bruel, S.; Frappe, P.; Tardy, B.; Botelho-Nevers, E.; Gagneux-Brunon, A. Intention to Participate in a COVID-19 Vaccine Clinical Trial and to Get Vaccinated against COVID-19 in France during the Pandemic. Vaccine 2020, 38, 7002–7006. [Google Scholar] [CrossRef]
- Weintraub, R.L.; Subramanian, L.; Karlage, A.; Ahmad, I.; Rosenberg, J. COVID-19 Vaccine To Vaccination: Why Leaders Must Invest In Delivery Strategies Now. Health Aff. 2021, 40, 33–41. [Google Scholar] [CrossRef]
- Malik, A.A.; McFadden, S.M.; Elharake, J.; Omer, S.B. Determinants of COVID-19 Vaccine Acceptance in the US. EClinicalMedicine 2020, 26, 100495. [Google Scholar] [CrossRef]
- Neumann-Böhme, S.; Varghese, N.E.; Sabat, I.; Barros, P.P.; Brouwer, W.; van Exel, J.; Schreyögg, J.; Stargardt, T. Once We Have It, Will We Use It? A European Survey on Willingness to Be Vaccinated against COVID-19. Eur. J. Heal. Econ. 2020, 21, 977–982. [Google Scholar] [CrossRef]
- Sallam, M. COVID-19 Vaccine Hesitancy Worldwide: A Concise Systematic Review of Vaccine Acceptance Rates. Vaccines 2021, 9, 160. [Google Scholar] [CrossRef]
- Fisher, K.A.; Bloomstone, S.J.; Walder, J.; Crawford, S.; Fouayzi, H.; Mazor, K.M. Attitudes Toward a Potential SARS-CoV-2 Vaccine. Ann. Intern. Med. 2020, 173, 964–973. [Google Scholar] [CrossRef]
- Ruiz, J.B.; Bell, R.A. Predictors of Intention to Vaccinate against COVID-19: Results of a Nationwide Survey. Vaccine 2021, 39, 1080–1086. [Google Scholar] [CrossRef]
- Lucia, V.C.; Kelekar, A.; Afonso, N.M. COVID-19 Vaccine Hesitancy among Medical Students. J. Public Health 2021, 43, 445–449. [Google Scholar] [CrossRef]
- Barello, S.; Nania, T.; Dellafiore, F.; Graffigna, G.; Caruso, R. ‘Vaccine Hesitancy’ among University Students in Italy during the COVID-19 Pandemic. Eur. J. Epidemiol. 2020, 35, 781–783. [Google Scholar] [CrossRef]
- Murphy, J.; Vallières, F.; Bentall, R.P.; Shevlin, M.; McBride, O.; Hartman, T.K.; McKay, R.; Bennett, K.; Mason, L.; Gibson-Miller, J.; et al. Psychological Characteristics Associated with COVID-19 Vaccine Hesitancy and Resistance in Ireland and the United Kingdom. Nat. Commun. 2021, 12, 29. [Google Scholar] [CrossRef]
- Paul, E.; Steptoe, A.; Fancourt, D. Attitudes towards Vaccines and Intention to Vaccinate against COVID-19: Implications for Public Health Communications. Lancet Reg. Heal. Eur. 2021, 1, 100012. [Google Scholar] [CrossRef]
- Bogart, L.M.; Ojikutu, B.O.; Tyagi, K.; Klein, D.J.; Mutchler, M.G.; Dong, L.; Lawrence, S.J.; Thomas, D.R.; Kellman, S. COVID-19 Related Medical Mistrust, Health Impacts, and Potential Vaccine Hesitancy Among Black Americans Living With HIV. JAIDS J. Acquir. Immune Defic. Syndr. 2021, 86, 200–207. [Google Scholar] [CrossRef]
- Kreps, S.; Prasad, S.; Brownstein, J.S.; Hswen, Y.; Garibaldi, B.T.; Zhang, B.; Kriner, D.L. Factors Associated With US Adults’ Likelihood of Accepting COVID-19 Vaccination. JAMA Netw. Open 2020, 3, e2025594. [Google Scholar] [CrossRef]
- Lin, C.; Tu, P.; Beitsch, L.M. Confidence and Receptivity for COVID-19 Vaccines: A Rapid Systematic Review. Vaccines 2020, 9, 16. [Google Scholar] [CrossRef]
- Szmyd, B.; Bartoszek, A.; Karuga, F.F.; Staniecka, K.; Błaszczyk, M.; Radek, M. Medical Students and SARS-CoV-2 Vaccination: Attitude and Behaviors. Vaccines 2021, 9, 128. [Google Scholar] [CrossRef]
- Tuells, J.; Egoavil, C.M.; Pena Pardo, M.A.; Montagud, A.C.; Montagud, E.; Caballero, P.; Zapater, P.; Puig-Barberá, J.; Hurtado-Sanchez, J.A. Seroprevalence Study and Cross-Sectional Survey on COVID-19 for a Plan to Reopen the University of Alicante (Spain). Int. J. Environ. Res. Public Health 2021, 18, 1908. [Google Scholar] [CrossRef]
- Grüner, S.; Krüger, F. The Intention to Be Vaccinated against COVID-19: Stated Preferences before Vaccines Were Available. Appl. Econ. Lett. 2021, 28, 1847–1851. [Google Scholar] [CrossRef]
- Kose, S.; Mandiracioglu, A.; Sahin, S.; Kaynar, T.; Karbus, O.; Ozbel, Y. Vaccine Hesitancy of the COVID-19 by Health Care Personnel. Int. J. Clin. Pract. 2021, 75, e13917. [Google Scholar] [CrossRef]
- Marsh, K.; van Til, J.A.; Molsen-David, E.; Juhnke, C.; Hawken, N.; Oehrlein, E.M.; Choi, Y.C.; Duenas, A.; Greiner, W.; Haas, K.; et al. Health Preference Research in Europe: A Review of Its Use in Marketing Authorization, Reimbursement, and Pricing Decisions—Report of the ISPOR Stated Preference Research Special Interest Group. Value Heal. 2020, 23, 831–841. [Google Scholar] [CrossRef]
- Hoogink, J.; Verelst, F.; Kessels, R.; van Hoek, A.J.; Timen, A.; Willem, L.; Beutels, P.; Wallinga, J.; de Wit, G.A. Preferential Differences in Vaccination Decision-Making for Oneself or One’s Child in the Netherlands: A Discrete Choice Experiment. BMC Public Health 2020, 20, 828. [Google Scholar] [CrossRef]
- McPhedran, R.; Toombs, B. Efficacy or Delivery? An Online Discrete Choice Experiment to Explore Preferences for COVID-19 Vaccines in the UK. Econ. Lett. 2021, 200, 109747. [Google Scholar] [CrossRef]
- Motta, M. Can a COVID-19 Vaccine Live up to Americans’ Expectations? A Conjoint Analysis of How Vaccine Characteristics Influence Vaccination Intentions. Soc. Sci. Med. 2021, 272, 113642. [Google Scholar] [CrossRef]
- Bridges, J.F.P.; Hauber, A.B.; Marshall, D.; Lloyd, A.; Prosser, L.A.; Regier, D.A.; Johnson, F.R.; Mauskopf, J. Conjoint Analysis Applications in Health—A Checklist: A Report of the ISPOR Good Research Practices for Conjoint Analysis Task Force. Value Health 2011, 14, 403–413. [Google Scholar] [CrossRef] [Green Version]
- Dong, D.; Xu, R.H.; Wong, E.L.; Hung, C.; Feng, D.; Feng, Z.; Yeoh, E.; Wong, S.Y. Public Preference for COVID-19 Vaccines in China: A Discrete Choice Experiment. Heal. Expect. 2020, 23, 1543–1578. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and Efficacy of the chadox1 Ncov-19 Vaccine (AZD1222) Against SARS-CoV-2: An Interim Analysis of Four Random-Ised Controlled Trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Walsh, E.E.; Frenck, R.W.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R.; et al. Safety and Immunogenicity of Two RNA-Based COVID-19 Vaccine Candidates. N. Engl. J. Med. 2020, 383, 2439–2450. [Google Scholar] [CrossRef]
- Knoll, M.D.; Wonodi, C. Oxford–AstraZeneca COVID-19 Vaccine Efficacy. Lancet 2021, 397, 72–74. [Google Scholar] [CrossRef]
- Pfizer and BioNTech Conclude Phase 3 Study of COVID-19 Vaccine Candidate, Meeting All Primary Efficacy Endpoints. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-conclude-phase-3-study-covid-19-vaccine (accessed on 24 August 2021).
- AstraZeneca AZD1222 Vaccine Met Primary Efficacy Endpoint in Preventing COVID-19. Available online: https://www.astrazeneca.com/content/astraz/media-centre/press-releases/2020/azd1222hlr.html (accessed on 24 August 2021).
- Moderna Moderna’s COVID-19 Vaccine Candidate Meets its Primary Efficacy Endpoint in the First Interim Analysis of the Phase 3 COVE Study. Available online: https://investors.modernatx.com/news-releases/news-release-details/modernas-covid-19-vaccine-candidate-meets-its-primary-efficacy (accessed on 24 August 2021).
- US Food and Drug Administration. Moderna COVID-19 Vaccine EUA Fact Sheet for Vaccination Providers. Available online: https://www.fda.gov/media/144637/download (accessed on 24 August 2021).
- US Food and Drug Administration. Pfizer-BioNTech COVID-19 Vaccine EUA Fact Sheet for Vaccination Providers. Available online: https://www.fda.gov/media/144413/download (accessed on 24 August 2021).
- US Food and Drug Administration. Janssen COVID-19 Vaccine EUA Fact Sheet for Vaccination Providers. Available online: https://www.fda.gov/media/146304/download (accessed on 24 August 2021).
- World Health Organization. COVID-19 Vaccine Tracker and Landscape. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 24 August 2021).
- Trevena, L.J.; Zikmund-Fisher, B.J.; Edwards, A.; Gaissmaier, W.; Galesic, M.; Han, P.K.; King, J.; Lawson, M.L.; Linder, S.K.; Lipkus, I.; et al. Presenting Quantitative Information about Decision Outcomes: A Risk Communication Primer for Patient Decision Aid Developers. BMC Med. Inform. Decis. Mak. 2013, 13, S7. [Google Scholar] [CrossRef] [Green Version]
- McCaffery, K.J.; Holmes-Rovner, M.; Smith, S.K.; Rovner, D.; Nutbeam, D.; Clayman, M.L.; Kelly-Blake, K.; Wolf, M.S.; Sheridan, S.L. Addressing Health Literacy in Patient Decision Aids. BMC Med. Inform. Decis. Mak. 2013, 13, S10. [Google Scholar] [CrossRef] [Green Version]
- Veldwijk, J.; Lambooij, M.S.; van Til, J.A.; Groothuis-Oudshoorn, C.G.M.; Smit, H.A.; de Wit, G.A. Words or graphics to present a Discrete Choice Experiment: Does it matter? Patient Educ. Couns. 2015, 98, 1376–1384. [Google Scholar] [CrossRef] [Green Version]
- Orme, B.K. Sample Size Issues for Conjoint Analysis. In Getting Started with Conjoint Analysis: Strategies for Product Design and Pricing Research; Research Publishers: Madison, WI, USA, 2020; pp. 57–66. ISBN 978-1-60147-115-4. [Google Scholar]
- de Bekker-Grob, E.W.; Donkers, B.; Jonker, M.F.; Stolk, E.A. Sample Size Requirements for Discrete-Choice Experiments in Healthcare: A Practical Guide. Patient 2015, 8, 373–384. [Google Scholar] [CrossRef] [Green Version]
- Conjoint.ly. Technical Points on DCE with Conjoint.ly. Available online: https://conjointly.com/guides/conjoint-technical-notes/ (accessed on 24 August 2021).
- U.S. Centers for Disease Control and Prevention Underlying Medical Conditions Associated with Higher Risk for Severe COVID-19: Information for Healthcare Providers. Available online: https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-care/underlyingconditions.html (accessed on 24 March 2021).
- McFadden, D. Conditional logit analysis of qualitative choice behavior. In Frontiers in Economics; Zarembka, P., Ed.; Academic Press: New York, NY, USA, 1974; pp. 105–142. [Google Scholar]
- Hughes, R.A.; Heron, J.; Sterne, J.A.C.; Tilling, K. Accounting for missing data in statistical analyses: Multiple imputation is not always the answer. Int. J. Epidemiol. 2019, 48, 1294–1304. [Google Scholar] [CrossRef]
- White, I.R.; Carlin, J.B. Bias and efficiency of multiple imputation compared with complete-case analysis for missing covariate values. Stat. Med. 2010, 29, 2920–2931. [Google Scholar] [CrossRef]
- Diesel, J.; Sterrett, N.; Dasgupta, S.; Kriss, J.L.; Barry, V.; Esschert, K.V.; Whiteman, A.; Cadwell, B.L.; Weller, D.; Qualters, J.R.; et al. COVID-19 Vaccination Coverage Among Adults—United States, December 14, 2020–May 22, 2021. MMWR Recomm. Rep. 2021, 70, 922–927. [Google Scholar] [CrossRef] [PubMed]
- Eshun-Wilson, I.; Mody, A.; Tram, K.H.; Bradley, C.; Sheve, A.; Fox, B.; Thompson, V.; Geng, E.H. Preferences for COVID-19 vaccine distribution strategies in the US: A discrete choice survey. PLoS ONE 2021, 16, e0256394. [Google Scholar] [CrossRef] [PubMed]
- Kelly, B.J.; Southwell, B.G.; McCormack, L.A.; Bann, C.M.; MacDonald, P.D.M.; Frasier, A.M.; Bevc, C.A.; Brewer, N.T.; Squiers, L.B. Correction to: Predictors of Willingness to Get a COVID-19 vaccine in the U.S. BMC Infect. Dis. 2021, 21, 383. [Google Scholar] [CrossRef] [PubMed]
- Štěpánek, L.; Janošíková, M.; Nakládalová, M.; Štěpánek, L.; Boriková, A.; Vildová, H. Motivation to COVID-19 Vaccination and Reasons for Hesitancy in Employees of a Czech Tertiary Care Hospital: A Cross-Sectional Survey. Vaccines 2021, 9, 863. [Google Scholar] [CrossRef] [PubMed]
- Eberhardt, J.; Ling, J. Predicting COVID-19 Vaccination Intention Using Protection Motivation Theory and Conspiracy Beliefs. Vaccine 2021, 39, 6269–6275. [Google Scholar] [CrossRef]

| Attributes | Levels |
|---|---|
| Efficacy in preventing COVID-19 | 70%; 80%; 90%; 95% |
| Risk of mild side effects | 20%; 40%; 60% |
| Protection duration | 6 months; 1 year; 2 years |
| Vaccine administration route | Oral; 1 injection; 2 injections |
| Recommender of the vaccine | Experts and professional societies; primary care provider; family or friends |
| Expected travel time to the vaccination site | 15 min; 30 min; 45 min; 60 min |
| Characteristics | All Participants, n (%) n = 445 | Reluctant to Undergo Vaccination for COVID-19, n (%) a n = 94 | Inclined to Undergo Vaccination for COVID-19, n (%) b n = 233 |
|---|---|---|---|
| Age, years | |||
| 18–24 | 305 (68.5) | 71 (75.5) | 177 (76.0) |
| 25–34 | 83 (18.7) | 16 (17.0) | 41 (17.6) |
| ≥35 | 28 (6.3) | 7 (7.4) | 15 (6.4) |
| PNTS or no answer | 29 (6.5) | 0 (0.0) | 0 (0.0) |
| Gender | |||
| Woman | 272 (61.1) | 64 (68.1) | 151 (64.8) |
| Man | 142 (31.9) | 30 (31.9) | 79 (33.9) |
| PNTS or no answer | 31 (7.0) | 0 (0.0) | 3 (1.3) |
| Ethnicity c | |||
| Czech or multiple, including Czech | 370 (80.6) | 79 (80.6) | 212 (88.7) |
| Moravian | 17 (3.7) | 4 (4.1) | 7 (2.9) |
| Slovak | 19 (4.1) | 4 (4.1) | 11 (4.6) |
| Not listed | 12 (2.7) | 5 (5.1) | 4 (1.7) |
| PNTS or no answer | 41 (8.9) | 6 (6.1) | 5 (2.1) |
| Education background | |||
| Primary education | 1 (0.2) | 0 (0.0) | 1 (0.4) |
| Secondary education | 258 (58.0) | 62 (66.0) | 157 (67.4) |
| Post-secondary education | 7 (1.6) | 3 (3.2) | 3 (1.3) |
| Bachelor or equivalent | 137 (30.8) | 27 (28.7) | 63 (27.0) |
| Master’s or equivalent | 8 (1.8) | 2 (2.1) | 4 (1.7) |
| PhD or another doctorate | 5 (1.1) | 0 (0.0) | 4 (1.7) |
| PNTS or no answer | 29 (6.5) | 0 (0.0) | 1 (0.4) |
| Biomedical background | |||
| Yes | 264 (59.3) | 56 (59.6) | 134 (57.5) |
| No | 139 (31.2) | 35 (37.2) | 88 (37.8) |
| PNTS or no answer | 42 (9.4) | 3 (3.2) | 11 (4.7) |
| Employment status | |||
| Student | 307 (69.0) | 74 (78.7) | 188 (80.7) |
| Employee | 87 (19.6) | 15 (16.0) | 31 (13.3) |
| Self-employed/Entrepreneur | 7 (1.6) | 2 (2.1) | 5 (2.1) |
| Unemployed | 7 (1.6) | 2 (2.1) | 5 (2.1) |
| Other | 5 (1.1) | 0 (0.0) | 3 (1.3) |
| PNTS or no answer | 32 (7.2) | 1 (1.1) | 1 (0.4) |
| Annual gross household income, EUR d | |||
| <6900 | 67 (15.1) | 19 (20.2) | 36 (15.5) |
| 6900–13,900 | 54 (12.1) | 11 (11.7) | 28 (12.0) |
| 13,900–20,800 | 66 (14.8) | 12 (12.8) | 36 (15.5) |
| 20,800–30,100 | 54 (12.1) | 11 (11.7) | 28 (12.0) |
| 30,100–37,000 | 44 (9.9) | 7 (7.4) | 33 (14.2) |
| 37,000–41,700 | 14 (3.1) | 3 (3.2) | 5 (2.1) |
| 41,700–46,300 | 9 (2.0) | 2 (2.1) | 4 (1.7) |
| >46,300 | 19 (4.3) | 4 (4.3) | 12 (5.2) |
| PNTS or no answer | 118 (26.5) | 25 (26.6) | 51 (21.9) |
| Socioeconomic index (SEI) e | |||
| <33rd percentile (lowest SEI) | 115 (25.8) | 61 (26.2) | 30 (31.9) |
| ≥33rd percentile (highest SEI) | 200 (44.9) | 114 (48.9) | 37 (39.4) |
| No answer | 130 (29.2) | 58 (24.9) | 27 (28.7) |
| Pregnancy (percent of women) | |||
| Yes | 2 (0.7) | 1 (1.6) | 1 (0.7) |
| Planning within one year | 9 (3.3) | 5 (7.8) | 2 (1.3) |
| No | 260 (95.6) | 58 (90.6) | 147 (97.4) |
| No answer | 1 (0.4) | 0 (0.0) | 1 (0.7) |
| Risk factors for severe COVID-19 f | |||
| One or more | 77 (17.3) | 13 (13.8) | 44 (18.9) |
| None | 334 (75.1) | 78 (83.0) | 187 (80.3) |
| PNTS or no answer | 34 (7.6) | 3 (3.2) | 2 (0.9) |
| Rejected any recommended vaccine in the past | |||
| Yes | 22 (4.9) | 12 (12.8) | 10 (4.3) |
| No | 387 (87.0) | 79 (84.0) | 221 (94.8) |
| PNTS or no answer | 36 (8.1) | 3 (3.2) | 2 (0.9) |
| Adverse events with any vaccine (not against COVID-19) c | |||
| Allergic reaction | 12 (2.7) | 5 (5.2) | 5 (2.1) |
| Other adverse events | 67 (14.9) | 17 (17.5) | 28 (11.9) |
| No or not sure | 339 (75.3) | 73 (75.3) | 201 (85.5) |
| PNTS or no answer | 32 (7.1) | 2 (2.1) | 1 (0.4) |
| History of COVID-19 c | |||
| Personal | 96 (16.4) | 21 (17.5) | 52 (16.6) |
| Relatives | 172 (29.4) | 34 (28.3) | 102 (32.5) |
| Other (e.g., friends, coworkers) | 203 (34.6) | 38 (31.7) | 113 (36.0) |
| No or not sure | 84 (14.3) | 24 (20.0) | 46 (14.6) |
| PNTS or no answer | 31 (5.3) | 3 (2.5) | 1 (0.3) |
| Vaccinated against COVID-19 | |||
| Yes, first dose or fully | 86 (19.3) | Not Applicable | Not Applicable |
| No | 331 (74.4) | 94 (100) | 233 (100) |
| PNTS or no answer | 28 (6.3) | Not Applicable | Not Applicable |
| Agreement with the statement “I want to be vaccinated against COVID-19 as soon as the vaccine is available for me.” g | |||
| Strongly agree | 140 (42.3) | Not Applicable | 140 (60.1) |
| Agree | 93 (28.1) | Not Applicable | 93 (39.9) |
| Not sure | 54 (16.3) | 54 (57.4) | Not Applicable |
| Disagree | 23 (6.9) | 23 (24.5) | Not Applicable |
| Strongly disagree | 17 (5.1) | 17 (18.1) | Not Applicable |
| Participants’ Characteristics | Reluctance to Undergo Vaccination | |
|---|---|---|
| Adjusted Odds Ratio (95% CI) | p | |
| Age, years | ||
| 18–24 | 0.99 (0.43–2.28) | 0.975 |
| ≥25 | Referent | |
| Gender | ||
| Man | 1.06 (0.55–2.03) | 0.852 |
| Woman | Referent | |
| Biomedical background | ||
| No | 1.65 (0.89–3.10) | 0.114 |
| Yes, any | Referent | |
| Risk factors for severe COVID-19 a | ||
| ≥1 | 1.56 (0.72–3.65) | 0.282 |
| None | Referent | |
| Adverse events with any vaccine (not against COVID-19) | ||
| Yes | 1.62 (0.72–3.52) | 0.233 |
| No | Referent | |
| Rejected any recommended vaccination in the past (not against COVID-19) | ||
| Yes | 3.30 (1.06–10.31) | 0.037 |
| No | Referent | |
| Socioeconomic index (SEI) | ||
| <33rd percentile (lowest SEI) | 1.51 (0.81–2.80) | 0.191 |
| ≥33rd percentile (highest SEI) | Referent | |
| Education | ||
| Bachelor degree or higher | 0.75 (0.35–1.56) | 0.446 |
| Post-secondary or lower | Referent | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donin, G.; Erfányuková, A.; Ivlev, I. Factors Affecting Young Adults’ Decision Making to Undergo COVID-19 Vaccination: A Patient Preference Study. Vaccines 2022, 10, 265. https://doi.org/10.3390/vaccines10020265
Donin G, Erfányuková A, Ivlev I. Factors Affecting Young Adults’ Decision Making to Undergo COVID-19 Vaccination: A Patient Preference Study. Vaccines. 2022; 10(2):265. https://doi.org/10.3390/vaccines10020265
Chicago/Turabian StyleDonin, Gleb, Anna Erfányuková, and Ilya Ivlev. 2022. "Factors Affecting Young Adults’ Decision Making to Undergo COVID-19 Vaccination: A Patient Preference Study" Vaccines 10, no. 2: 265. https://doi.org/10.3390/vaccines10020265
APA StyleDonin, G., Erfányuková, A., & Ivlev, I. (2022). Factors Affecting Young Adults’ Decision Making to Undergo COVID-19 Vaccination: A Patient Preference Study. Vaccines, 10(2), 265. https://doi.org/10.3390/vaccines10020265






