Pyoderma Gangrenosum Induced by BNT162b2 COVID-19 Vaccine in a Healthy Adult
Abstract
:1. Background
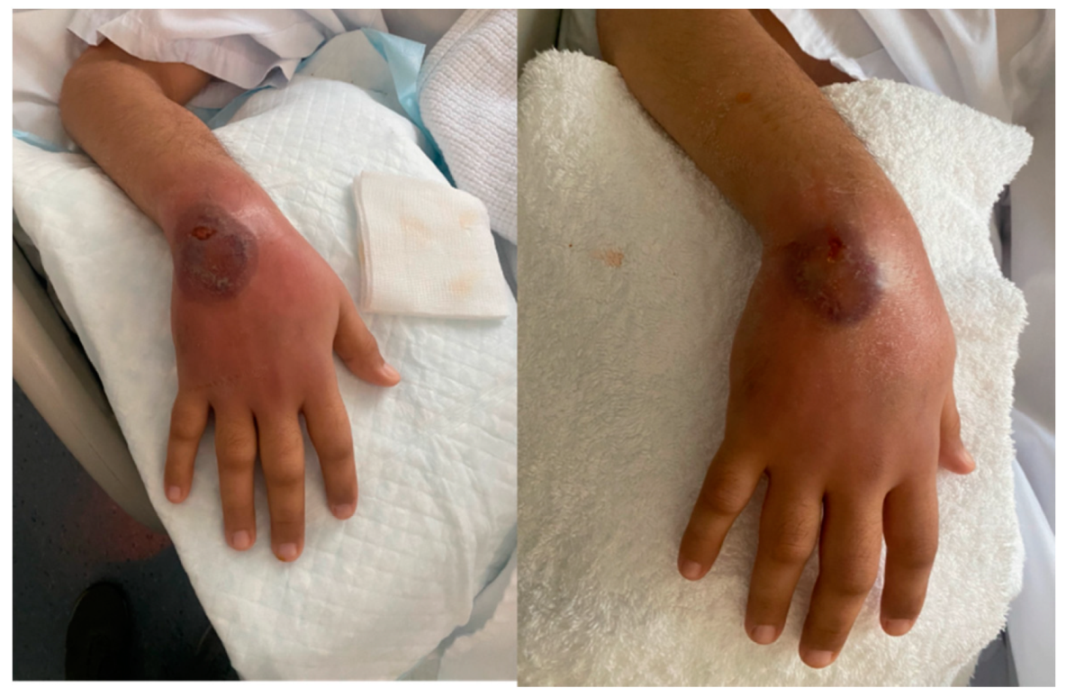
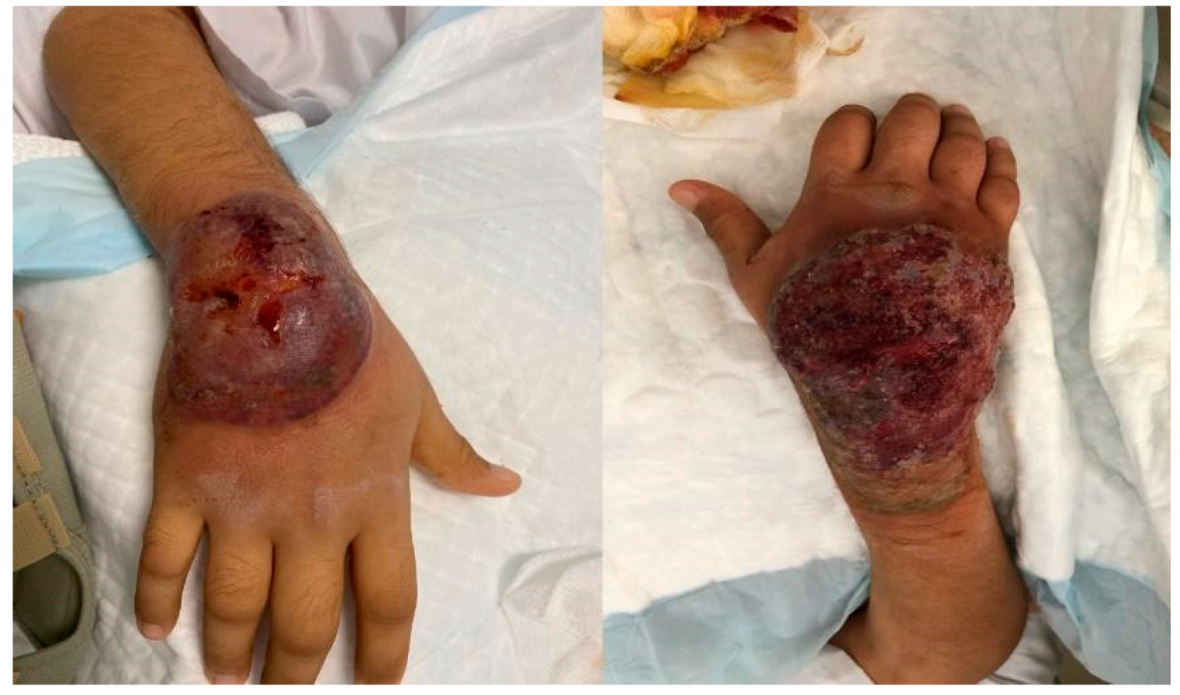
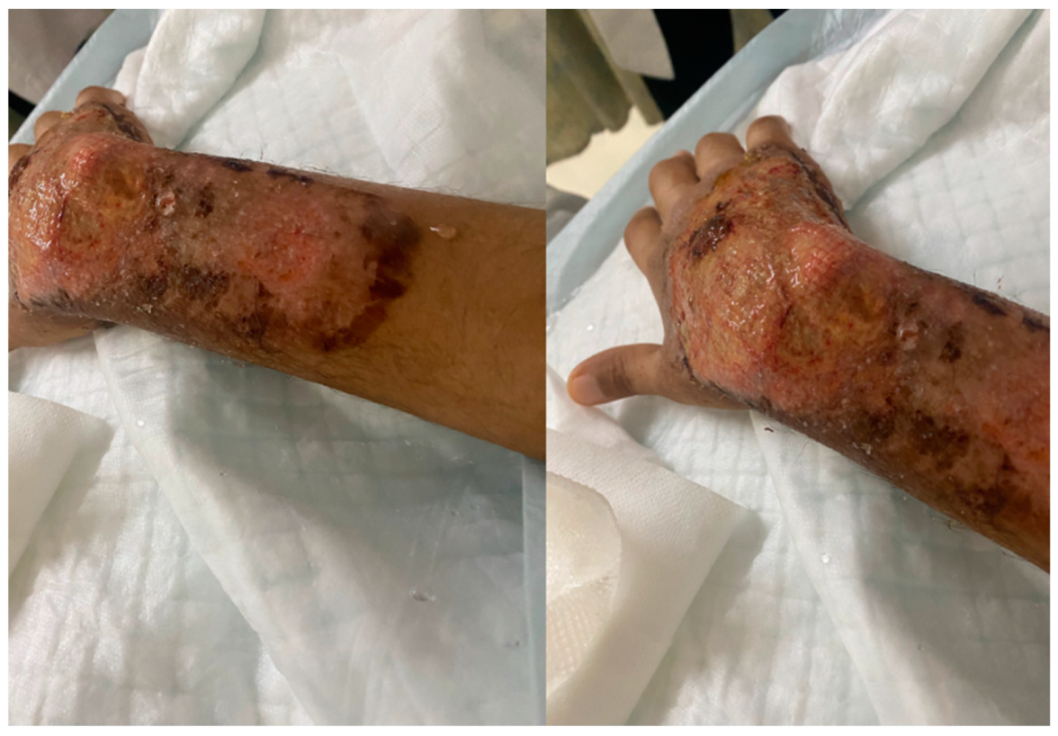
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PG | pyoderma gangrenosum |
| COVID-19 | coronavirus disease 2019 |
| mRNA | messenger ribonucleic acid |
| URTI | upper respiratory tract infection |
| MRI | magnetic resonance imaging |
| NPS | nasopharyngeal swab |
| RT-PCR | reverse transcription polymerase chain reaction |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
| H&E | hematoxylin and eosin |
| RAND | research and development |
| UCLA | University of California at Los Angeles |
References
- Simonsen, L.; Viboud, C. Mortality: A comprehensive look at the COVID-19 pandemic death toll. eLife 2021, 10, e71974. [Google Scholar] [CrossRef] [PubMed]
- Meo, S.A.; Bukhari, I.A.; Akram, J.; Meo, A.S.; Klonoff, D.C. COVID-19 vaccines: Comparison of biological, pharmacological characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 1663–1669. [Google Scholar] [PubMed]
- Magro, C.; Crowson, A.N.; Franks, L.; Schaffer, P.R.; Whelan, P.; Nuovo, G. The histologic and molecular correlates of COVID-19 vaccine-induced changes in the skin. Clin. Dermatol. 2021, 39, 966–984. [Google Scholar] [CrossRef] [PubMed]
- Maverakis, E.; Marzano, A.V.; Le, S.T.; Callen, J.P.; Brüggen, M.C.; Guenova, E.; Dissemond, J.; Shinkai, K.; Langan, S.M. Pyoderma gangrenosum. Nat. Rev. Dis. Primers 2020, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Maverakis, E.; Ma, C.; Shinkai, K.; Fiorentino, D.; Callen, J.P.; Wollina, U.; Marzano, A.V.; Wallach, D.; Kim, K.; Schadt, C.; et al. Diagnostic Criteria of Ulcerative Pyoderma Gangrenosum: A Delphi Consensus of International Experts. JAMA Dermatol. 2018, 154, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Ramessur, R.; Saffar, N.; Czako, B.; Agarwal, A.; Batta, K. Cutaneous thrombosis associated with skin necrosis following Oxford-AstraZeneca COVID-19 vaccination. Clin. Exp. Dermatol. 2021, 46, 1610–1612. [Google Scholar] [CrossRef] [PubMed]
- McMahon, D.E.; Amerson, E.; Rosenbach, M.; Lipoff, J.B.; Moustafa, D.; Tyagi, A.; Desai, S.R.; French, L.E.; Lim, H.W.; Thiers, B.H.; et al. Cutaneous reactions reported after Moderna and Pfizer COVID-19 vaccination: A registry-based study of 414 cases. J. Am. Acad. Dermatol. 2021, 85, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Baffa, M.E.; Maglie, R.; Giovannozzi, N.; Montefusco, F.; Senatore, S.; Massi, D.; Antiga, E. Sweet Syndrome Following SARS-CoV2 Vaccination. Vaccines 2021, 9, 1212. [Google Scholar] [CrossRef] [PubMed]
- Vashisht, P.; Bansal, P.; Goyal, A.; Holmes, M.P.H. Sweet Syndrome. In StatPearls; StatPearls Publishing: Rochester, MN, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK431050/ (accessed on 14 September 2021).
- Reusch, N.; De Domenico, E.; Bonaguro, L.; Schulte-Schrepping, J.; Baßler, K.; Schultze, J.L.; Aschenbrenner, A.C. Neutrophils in COVID-19. Front. Immunol. 2021, 12, 652470. [Google Scholar] [CrossRef] [PubMed]
- Hazeldine, J.; Lord, J.M. Neutrophils and COVID-19: Active Participants and Rational Therapeutic Targets. Front. Immunol. 2021, 12, 680134. [Google Scholar] [CrossRef] [PubMed]
- Teijaro, J.R.; Farber, D.L. COVID-19 vaccines: Modes of immune activation and future challenges. Nat. Rev. Immunol. 2021, 21, 195–197. [Google Scholar] [CrossRef] [PubMed]
- Syed, K.; Chaudhary, H.; Balu, B. Pyoderma gangrenosum following COVID-19 infection. J. Community Hosp. Intern. Med. Perspect. 2021, 11, 601–603. [Google Scholar] [CrossRef] [PubMed]
- Wolf, R.; Barzilai, A.; Davidovici, B. Neutrophilic dermatosis of the hands after influenza vaccination. Int. J. Dermatol. 2009, 48, 66–68. [Google Scholar] [CrossRef] [PubMed]

| Type of Pyoderma Gangrenosum Diagnostic Criteria | Description | Patient Fulfillment |
|---|---|---|
| Major criterion | Biopsy of ulcer edge demonstrating neutrophilic infiltrate | Yes |
| Minor criterion | Exclusion of infection | Yes |
| Minor criterion | Pathergy | Yes |
| Minor criteria | History of inflammatory bowel disease or inflammatory arthritis | No |
| Minor criteria | History of papule, pustule, or vesicle ulcerating within 4 days of appearing | Yes |
| Minor criteria | Peripheral erythema, undermining border, and tenderness at ulceration site | Yes |
| Minor criteria | multiple ulcerations, at least 1 on an anterior lower leg | Yes |
| Minor criteria | Cribriform or “wrinkled paper” scar(s) at healed ulcer sites | Yes |
| Minor criteria | Decreased ulcer size within 1 month of initiating immunosuppressive medication(s) | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barry, M.; AlRajhi, A.; Aljerian, K. Pyoderma Gangrenosum Induced by BNT162b2 COVID-19 Vaccine in a Healthy Adult. Vaccines 2022, 10, 87. https://doi.org/10.3390/vaccines10010087
Barry M, AlRajhi A, Aljerian K. Pyoderma Gangrenosum Induced by BNT162b2 COVID-19 Vaccine in a Healthy Adult. Vaccines. 2022; 10(1):87. https://doi.org/10.3390/vaccines10010087
Chicago/Turabian StyleBarry, Mazin, Abdulaziz AlRajhi, and Khaldoon Aljerian. 2022. "Pyoderma Gangrenosum Induced by BNT162b2 COVID-19 Vaccine in a Healthy Adult" Vaccines 10, no. 1: 87. https://doi.org/10.3390/vaccines10010087
APA StyleBarry, M., AlRajhi, A., & Aljerian, K. (2022). Pyoderma Gangrenosum Induced by BNT162b2 COVID-19 Vaccine in a Healthy Adult. Vaccines, 10(1), 87. https://doi.org/10.3390/vaccines10010087







