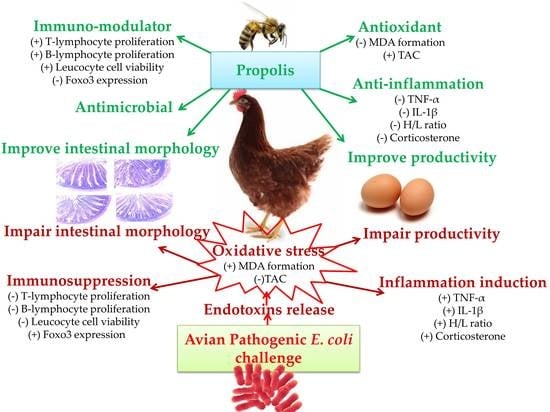
Modulating Laying Hens Productivity and Immune Performance in Response to Oxidative Stress Induced by E. coli Challenge Using Dietary Propolis Supplementation
Abstract
1. Introduction
2. Materials and Methods
2.1. Birds and Ethical Statement
2.2. Experimental Design and Birds Management
2.3. Blood Sample Collection and Preparation
2.4. Productive Performance
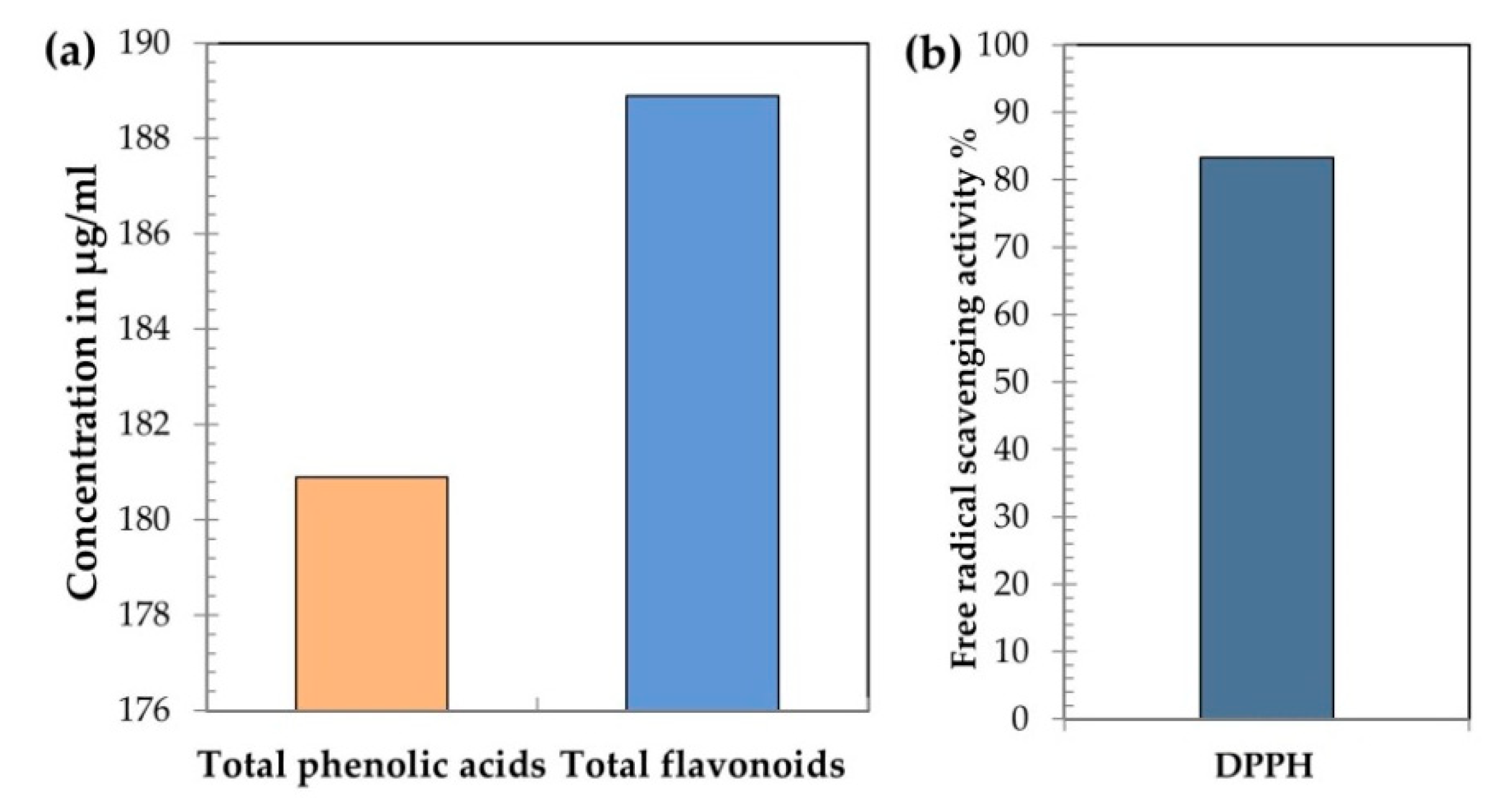
2.5. Propolis Phenolic Content and Free Radical Scavenging Activity
2.6. Physiological Parameters
2.6.1. Inflammation Markers and Antioxidant Parameters in PBMCs
2.6.2. Plasma Corticosterone Hormone Assay
2.7. Immunological Parameters
2.7.1. Wattle Thickness in Response to Phytohemagglutinin-P (PHA) Antigen Injection
2.7.2. Total WBC Count and Heterophile/Lymphocyte (H/L) Ratio Determination
2.7.3. Peripheral T- and B-Lymphocyte Proliferation
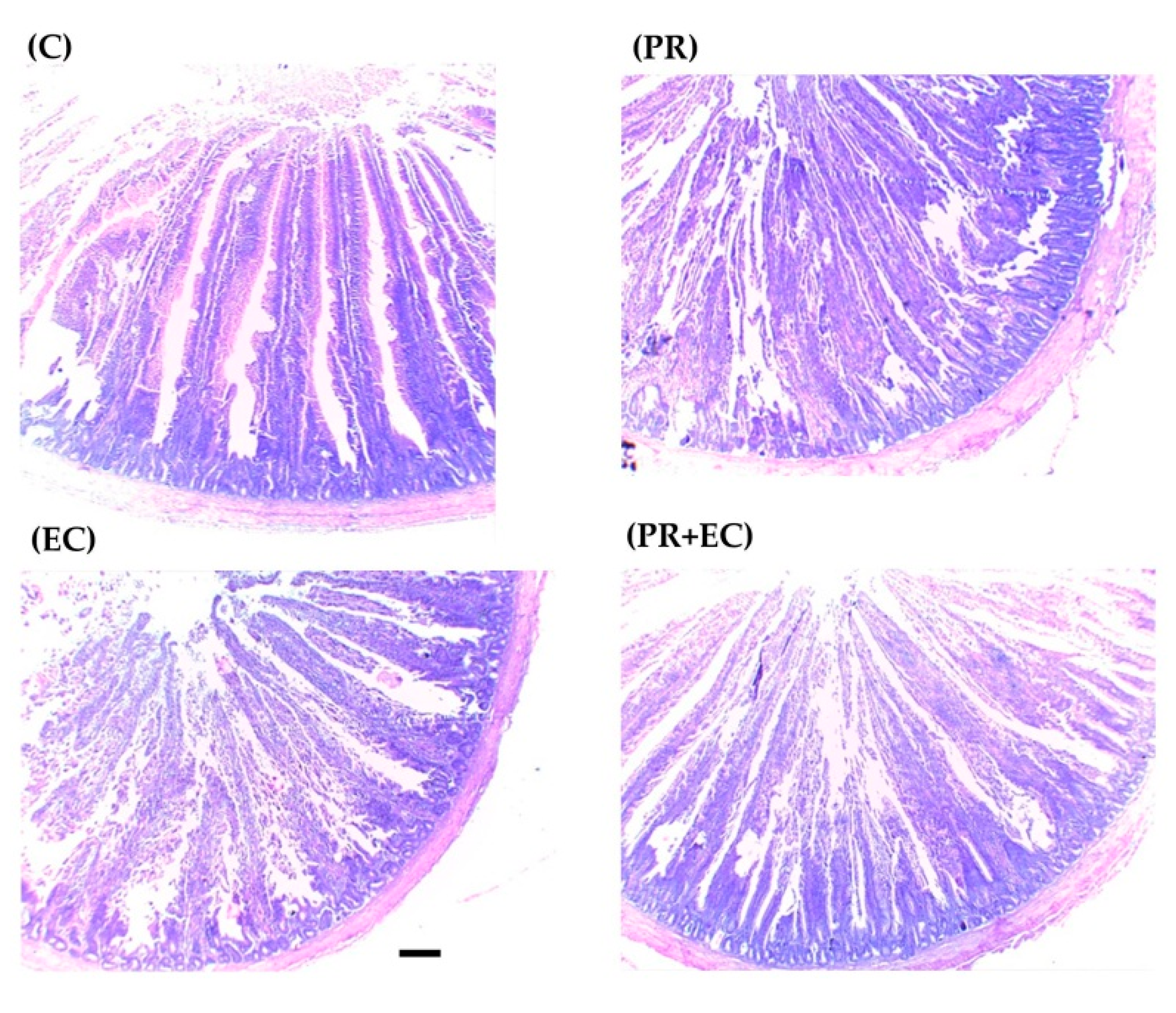
2.8. Small Intestine Histomorphology
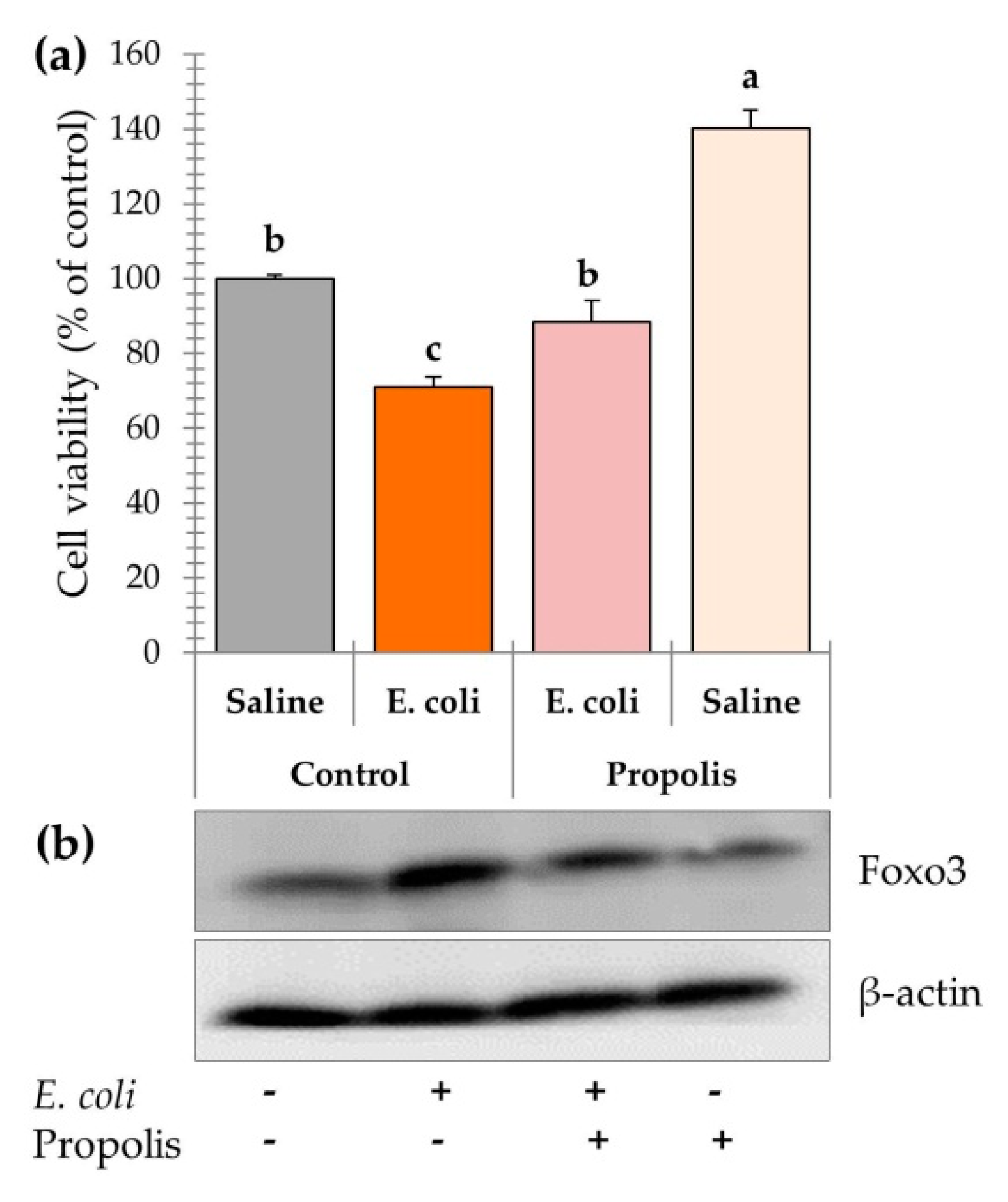
2.9. Leucocyte Cell Viability
2.10. Foxo3 Expression in PBMCs
2.11. Statistical Analysis
3. Results
3.1. Productive Performance
3.2. Inflammation Markers and Antioxidant Status
3.3. Immunological Performance
3.4. Small Intestines Histomorphology
3.5. Leucocyte Cell Viability and Foxo3 Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saiyasit, N.; Chunchai, T.; Apaijai, N.; Pratchayasakul, W.; Sripetchwandee, J.; Chattipakorn, N.; Chattipakorn, S.C. Chronic high-fat diet consumption induces an alteration in plasma/brain neurotensin signaling, metabolic disturbance, systemic inflammation/oxidative stress, brain apoptosis, and dendritic spine loss. Neuropeptides 2020, 82, 102047. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Tripathi, P.; Yadawa, A.K.; Singh, S. Promising Polyphenols in Parkinson’s Disease Therapeutics. Neurochem. Res. 2020, 45, 1731–1745. [Google Scholar] [CrossRef] [PubMed]
- Son, E.S.; Park, J.-W.; Kim, Y.J.; Jeong, S.H.; Hong, J.H.; Kim, S.-H.; Kyung, S.Y. Effects of antioxidants on oxidative stress and inflammatory responses of human bronchial epithelial cells exposed to particulate matter and cigarette smoke extract. Toxicol. In Vitro 2020, 67, 104883. [Google Scholar] [CrossRef] [PubMed]
- Petushkova, A.I.; Zamyatnin, J.A.A. Redox-Mediated Post-Translational Modifications of Proteolytic Enzymes and Their Role in Protease Functioning. Biomolecules 2020, 10, 650. [Google Scholar] [CrossRef]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxidative Med. Cell. Longev. 2017, 2017, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Raphael, I.; Nalawade, S.; Eagar, T.N.; Forsthuber, T.G. T cell subsets and their signature cytokines in autoimmune and inflammatory diseases. Cytokine 2014, 74, 5–17. [Google Scholar] [CrossRef]
- Enany, M.E.; Algammal, A.M.; Nasef, S.A.; Abo-Eillil, S.A.M.; Bin-Jumah, M.N.; Taha, A.E.; Allam, A.A. The occurrence of the multidrug resistance (MDR) and the prevalence of virulence genes and QACs resistance genes in E. coli isolated from environmental and avian sources. AMB Express 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Al Azad, M.A.R.; Rahman, M.; Amin, R.; Begum, M.I.A.; Fries, R.; Husna, A.; Khairalla, A.S.; Badruzzaman, A.; El Zowalaty, M.E.; Na Lampang, K.; et al. Susceptibility and Multidrug Resistance Patterns of Escherichia coli Isolated from Cloacal Swabs of Live Broiler Chickens in Bangladesh. Pathogens 2019, 8, 118. [Google Scholar] [CrossRef]
- Muloi, D.; Kiiru, J.; Ward, M.J.; Hassell, J.M.; Bettridge, J.M.; Robinson, T.P.; Van Bunnik, B.A.; Chase-Topping, M.; Robertson, G.; Pedersen, A.B.; et al. Epidemiology of antimicrobial-resistant Escherichia coli carriage in sympatric humans and livestock in a rapidly urbanizing city. Int. J. Antimicrob. Agents 2019, 54, 531–537. [Google Scholar] [CrossRef]
- Żbikowska, K.; Michalczuk, M.; Dolka, B. The Use of Bacteriophages in the Poultry Industry. Animals 2020, 10, 872. [Google Scholar] [CrossRef]
- Franklin, N.; Hope, K.; Glasgow, K.; Glass, K. Describing the Epidemiology of Foodborne Outbreaks in New South Wales from 2000 to 2017. Foodborne Pathog. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, R.A.; Cryer, T.L.; Lafi, S.; Abu-Basha, E.A.; Good, L.; Tarazi, Y.H. Identification of Escherichia coli from broiler chickens in Jordan, their antimicrobial resistance, gene characterization and the associated risk factors. BMC Veter. Res. 2019, 15, 159. [Google Scholar] [CrossRef]
- Yan, W.; Bai, R.; Wang, S.; Tian, X.; Li, Y.; Wang, S.; Yang, F.; Xiao, Y.; Lu, X.; Zhao, F. Antibiotic resistance genes are increased by combined exposure to sulfamethoxazole and naproxen but relieved by low-salinity. Environ. Int. 2020, 139, 105742. [Google Scholar] [CrossRef] [PubMed]
- Han, T.; Zhang, Q.; Liu, N.; Wang, J.; Li, Y.; Huang, X.; Liu, J.; Wang, J.; Qu, Z.; Qi, K. Changes in antibiotic resistance of Escherichia coli during the broiler feeding cycle. Poult. Sci. 2020. [Google Scholar] [CrossRef]
- Kumari, M.; Gupta, R.P.; Lather, D.; Bagri, P. Ameliorating effect of Withania somnifera root extract in Escherichia coli–infected broilers. Poult. Sci. 2020, 99, 1875–1887. [Google Scholar] [CrossRef] [PubMed]
- Daneshmand, A.; Kermanshahi, H.; Sekhavati, M.H.; Javadmanesh, A.; Ahmadian, M. Antimicrobial peptide, cLF36, affects performance and intestinal morphology, microflora, junctional proteins, and immune cells in broilers challenged with E. coli. Sci. Rep. 2019, 9, 14176–14179. [Google Scholar] [CrossRef]
- Ramadan, H.; Jackson, C.R.; Frye, J.G.; Hiott, L.M.; Samir, M.; Awad, A.; Woodley, T.A. Antimicrobial Resistance, Genetic Diversity and Multilocus Sequence Typing of Escherichia coli from Humans, Retail Chicken and Ground Beef in Egypt. Pathogens 2020, 9, 357. [Google Scholar] [CrossRef]
- Hazrati, S.; Rezaeipour, V.; Asadzadeh, S. Effects of phytogenic feed additives, probiotic and mannan-oligosaccharides on performance, blood metabolites, meat quality, intestinal morphology, and microbial population of Japanese quail. Br. Poult. Sci. 2019, 61, 132–139. [Google Scholar] [CrossRef]
- Dong, Y.; Li, R.; Liu, Y.; Ma, L.; Zha, J.; Qiao, X.; Chai, T.; Wu, B. Benefit of Dietary Supplementation with Bacillus subtilis BYS2 on Growth Performance, Immune Response, and Disease Resistance of Broilers. Probiot. Antimicrob. Proteins 2020. [Google Scholar] [CrossRef]
- Makled, M.N.; Abouelezz, K.; Gad-Elkareem, A.E.G.; Sayed, A.M. Comparative influence of dietary probiotic, yoghurt, and sodium butyrate on growth performance, intestinal microbiota, blood hematology, and immune response of meat-type chickens. Trop. Anim. Health Prod. 2019, 51, 2333–2342. [Google Scholar] [CrossRef]
- Zhan, H.; Dong, X.; Li, L.; Zheng, Y.; Gong, Y.; Zou, X. Effects of dietary supplementation with Clostridium butyricum on laying performance, egg quality, serum parameters, and cecal microflora of laying hens in the late phase of production. Poult. Sci. 2019, 98, 896–903. [Google Scholar] [CrossRef]
- Simone-Finstrom, M.; Borba, R.S.; Wilson, M.; Spivak, M. Propolis Counteracts Some Threats to Honey Bee Health. Insects 2017, 8, 46. [Google Scholar] [CrossRef]
- Rivero-Cruz, J.F.; Granados-Pineda, J.; Pedraza-Chaverri, J.; Rojas, J.M.P.; Passari, A.K.; Díaz-Ruiz, G.; Rivero-Cruz, B.E. Phytochemical Constituents, Antioxidant, Cytotoxic, and Antimicrobial Activities of the Ethanolic Extract of Mexican Brown Propolis. Antioxidants 2020, 9, 70. [Google Scholar] [CrossRef] [PubMed]
- Galeotti, F.; Maccari, F.; Fachini, A.; Volpi, N. Chemical Composition and Antioxidant Activity of Propolis Prepared in Different Forms and in Different Solvents Useful for Finished Products. Foods 2018, 7, 41. [Google Scholar] [CrossRef]
- Woźniak, M.; Mrówczyńska, L.; Waśkiewicz, A.; Rogoziński, T.; Ratajczak, I. Phenolic Profile and Antioxidant Activity of Propolis Extracts from Poland. Nat. Prod. Commun. 2019, 14. [Google Scholar] [CrossRef]
- Osés, S.; Marcos, P.; Azofra, P.; De Pablo, A.; Fernández-Muíño, M.A.; Sancho, M.T. Phenolic Profile, Antioxidant Capacities and Enzymatic Inhibitory Activities of Propolis from Different Geographical Areas: Needs for Analytical Harmonization. Antioxidants 2020, 9, 75. [Google Scholar] [CrossRef]
- Kuo, Y.-H.; Chiang, H.-L.; Wu, P.-Y.; Chu, Y.; Chang, Q.-X.; Wen, K.-C.; Lin, C.-Y.; Chiang, H.-M. Protection against Ultraviolet A-Induced Skin Apoptosis and Carcinogenesis through the Oxidative Stress Reduction Effects of N-(4-bromophenethyl) Caffeamide, A Propolis Derivative. Antioxidants 2020, 9, 335. [Google Scholar] [CrossRef]
- Ramanauskienė, K.; Inkėnienė, A.M.; Petrikaitė, V.; Briedis, V. Total Phenolic Content and Antimicrobial Activity of Different Lithuanian Propolis Solutions. Evid. Based Complement. Altern. Med. 2013, 2013, 1–5. [Google Scholar] [CrossRef]
- Park, E.-H.; Kim, S.-H.; Park, S.-S. Anti-inflammatory activity of propolis. Arch. Pharm. Res. 1996, 19, 337–341. [Google Scholar] [CrossRef]
- Campos, J.F.; Dos Santos, E.L.; da Rocha, P.D.S.; Damião, M.J.; Balestieri, J.B.P.; Cardoso, C.A.L.; Paredes-Gamero, E.J.; Estevinho, L.M.; Souza, K.D.P.; Dos Santos, E.L. Antimicrobial, Antioxidant, Anti-Inflammatory, and Cytotoxic Activities of Propolis from the Stingless Bee Tetragonisca fiebrigi (Jataí). Evid. Based Complement. Altern. Med. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Abass, A.O.; Kamel, N.; Khalifa, W.; Gouda, G.F.; El-Manylawi, M.A.F.; Mehaisen, G.M.K.; Mashaly, M.M. Propolis supplementation attenuates the negative effects of oxidative stress induced by paraquat injection on productive performance and immune function in turkey poults. Poult. Sci. 2017, 96, 4419–4429. [Google Scholar] [CrossRef] [PubMed]
- Prakatur, I.; Miškulin, M.; Pavic, M.; Marjanovic, K.; Blazicevic, V.; Miškulin, M.; Domacinovic, M. Intestinal Morphology in Broiler Chickens Supplemented with Propolis and Bee Pollen. Animals 2019, 9, 301. [Google Scholar] [CrossRef] [PubMed]
- Mehaisen, G.M.K.; Ibrahim, R.M.; Desoky, A.A.; Safaa, H.; El-Sayed, O.A.; Abass, A.O. The importance of propolis in alleviating the negative physiological effects of heat stress in quail chicks. PLoS ONE 2017, 12, e0186907. [Google Scholar] [CrossRef] [PubMed]
- Mehaisen, G.M.K.; Desoky, A.A.; Sakr, O.G.; Sallam, W.; Abass, A.O. Propolis alleviates the negative effects of heat stress on egg production, egg quality, physiological and immunological aspects of laying Japanese quail. PLoS ONE 2019, 14, e0214839. [Google Scholar] [CrossRef] [PubMed]
- AlQarni, A.M.; Niwasabutra, K.; Sahlan, M.; Fearnley, H.; Ferro, V.; Watson, D.G. Propolis Exerts an Anti-Inflammatory Effect on PMA-Differentiated THP-1 Cells via Inhibition of Purine Nucleoside Phosphorylase. Metabolites 2019, 9, 75. [Google Scholar] [CrossRef]
- Mehaisen, G.M.K.; Eshak, M.G.; Elkaiaty, A.M.; Atta, A.M.; Mashaly, M.M.; Abass, A.O. Comprehensive growth performance, immune function, plasma biochemistry, gene expressions and cell death morphology responses to a daily corticosterone injection course in broiler chickens. PLoS ONE 2017, 12, e0172684. [Google Scholar] [CrossRef]
- Shimada, K.; Fujikawa, K.; Yahara, K.; Nakamura, T. Antioxidative properties of xanthan on the autoxidation of soybean oil in cyclodextrin emulsion. J. Agric. Food Chem. 1992, 40, 945–948. [Google Scholar] [CrossRef]
- Oktay, M.; Gülçin, I.; Küfrevioğlu, Ö.I. Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. LWT 2003, 36, 263–271. [Google Scholar] [CrossRef]
- Edelman, A.S.; Sánchez, P.L.; Robinson, M.E.; Hochwald, G.M.; Thorbecke, G. Primary and Secondary Wattle Swelling Response to Phytohemagglutinin as a Measure of Immunocompetence in Chickens. Avian Dis. 1986, 30, 105. [Google Scholar] [CrossRef]
- Gehad, A.E.; Mehaisen, G.M.K.; Abbas, A.O.; Mashaly, M.M. The Role of Light Program and Melatonin on Alleviation of Inflammation Induced by Lipopolysaccharide Injection in Broiler Chickens. Int. J. Poult. Sci. 2008, 7, 193–201. [Google Scholar] [CrossRef]
- Campbell, T.W. Hematology. In Avian Medicine: Principles and Application; Ritchie, B.W., Harrison, G.J., Harrison, L.R., Eds.; Winger’s Publishing Inc.: Lake Worth, FL, USA, 1994; pp. 176–198. [Google Scholar]
- Suvarna, S.K.; Layton, C.; Bancroft, J.D. Bancroft’s Theory and Practice of Histological Techniques, 8th ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2019; p. 584. [Google Scholar] [CrossRef]
- Reinhold, D.; Bank, U.; Bühling, F.; Neubert, K.; Mattern, T.; Ulmer, A.J.; Flad, H.-D.; Ansorge, S. Dipeptidyl Peptidase IV (CD26) on Human Lymphocytes. Synthetic Inhibitors of and Antibodies against Dipeptidyl Peptidase IV Suppress the Proliferation of Pokeweed Mitogen-Stimulated Peripheral Blood Mononuclear Cells, and IL-2 and IL-6 Production. Immunobiology 1993, 188, 403–414. [Google Scholar] [CrossRef]
- SAS Institute Inc. SAS/STATR 9.3 User’s Guide; SAS Institute Inc.: Cary, NC, USA, 2004. [Google Scholar]
- Kirikae, T.; Kirikae, F.; Saito, S.; Tominaga, K.; Tamura, H.; Uemura, Y.; Yokochi, T.; Nakano, M. Biological Characterization of Endotoxins Released from Antibiotic-Treated Pseudomonas aeruginosa and Escherichia coli. Antimicrob. Agents Chemother. 1998, 42, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, K.Z.; Edens, F. Influence of organic selenium on hsp70 response of heat-stressed and enteropathogenic Escherichia coli-challenged broiler chickens (Gallus gallus). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2005, 141, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, A.A.; Kariyawasam, S. An Experimental Infection Model for Escherichia coli Egg Peritonitis in Layer Chickens. Avian Dis. 2014, 58, 25–33. [Google Scholar] [CrossRef]
- Surai, P.; Kochish, I.I.; Fisinin, V.I.; Kidd, M. Antioxidant Defence Systems and Oxidative Stress in Poultry Biology: An Update. Antioxidants 2019, 8, 235. [Google Scholar] [CrossRef]
- Da Rosa, G.; Alba, D.F.; Silva, A.D.; Gris, A.; Mendes, R.E.; Mostardeiro, V.B.; Lopes, T.F.; Schetinger, M.R.C.; Stefani, L.M.; Lopes, M.T.; et al. Impact of Escherichia coli infection in broiler breeder chicks: The effect of oxidative stress on weight gain. Microb. Pathog. 2019, 139, 103861. [Google Scholar] [CrossRef]
- Asgharpour, F.; Moghadamnia, A.A.; Motallebnejad, M.; Nouri, H.R. Propolis attenuates lipopolysaccharide-induced inflammatory responses through intracellular ROS and NO levels along with downregulation of IL-1β and IL-6 expressions in murine RAW 264.7 macrophages. J. Food Biochem. 2019, 43, e12926. [Google Scholar] [CrossRef]
- Stepanović, S.; Antić, N.; Dakić, I.; Švabić-Vlahović, M. In vitro antimicrobial activity of propolis and synergism between propolis and antimicrobial drugs. Microbiol. Res. 2003, 158, 353–357. [Google Scholar] [CrossRef]
- Abdel-Kareem, A.A.A.; El-Sheikh, T.M. Impact of supplementing diets with propolis on productive performance, egg quality traits and some haematological variables of laying hens. J. Anim. Physiol. Anim. Nutr. 2015, 101, 441–448. [Google Scholar] [CrossRef]
- Seven, P.T. The Effects of Dietary Turkish Propolis and Vitamin C on Performance, Digestibility, Egg Production and Egg Quality in Laying Hens under Different Environmental Temperatures. Asian Australas. J. Anim. Sci. 2008, 21, 1164–1170. [Google Scholar] [CrossRef]
- Kačániová, M.; Rovná, K.; Arpášová, H.; Čuboň, J.; Hleba, L.; Pochop, J.; Kunová, S.; Haščík, P. In vitroandIn vivoantimicrobial activity of propolis on the microbiota from gastrointestinal tract of chickens. J. Environ. Sci. Health Part A 2012, 47, 1665–1671. [Google Scholar] [CrossRef]
- Mahmoud, U.T.; Cheng, H.; Applegate, T. Functions of propolis as a natural feed additive in poultry. Worlds Poult. Sci. J. 2016, 72, 37–48. [Google Scholar] [CrossRef]
- The Effect of Diet Propolis Supplementation on Ross Broiler Chicks Performance. Int. J. Poult. Sci. 2006, 5, 84–88. [CrossRef]
- Zhang, J.M.; An, J. Cytokines, Inflammation, and Pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef]
- Lauridsen, C. From oxidative stress to inflammation: Redox balance and immune system. Poult. Sci. 2019, 98, 4240–4246. [Google Scholar] [CrossRef]
- Mehaisen, G.M.K.; Eshak, M.G.; El Sabry, M.; Abass, A.O. Expression of Inflammatory and Cell Death Program Genes and Comet DNA Damage Assay Induced by Escherichia coli in Layer Hens. PLoS ONE 2016, 11, e0158314. [Google Scholar] [CrossRef]
- Turnbull, A.V.; Rivier, C. Regulation of the hypothalamic-pituitary-adrenal axis by cytokines: Actions and mechanisms of action. Physiol. Rev. 1999, 79, 1–71. [Google Scholar] [CrossRef]
- Løtvedt, P.; Fallahshahroudi, A.; Bektic, L.; Altimiras, J.; Jensen, P. Chicken domestication changes expression of stress-related genes in brain, pituitary and adrenals. Neurobiol. Stress 2017, 7, 113–121. [Google Scholar] [CrossRef]
- Peng, L.; Matthijs, M.G.; Haagsman, H.P.; Veldhuizen, E.J. Avian pathogenic Escherichia coli-induced activation of chicken macrophage HD11 cells. Dev. Comp. Immunol. 2018, 87, 75–83. [Google Scholar] [CrossRef]
- Mol, N.; Peng, L.; Esnault, E.; Quéré, P.; Haagsman, H.P.; Veldhuizen, E.J. Avian pathogenic Escherichia coli infection of a chicken lung epithelial cell line. Veter. Immunol. Immunopathol. 2019, 210, 55–59. [Google Scholar] [CrossRef]
- Kittana, H.; Neto, J.C.G.; Heck, K.; Geis, A.L.; Muñoz, R.R.S.; Cody, L.A.; Schmaltz, R.J.; Bindels, L.B.; Sinha, R.; Hostetter, J.M.; et al. Commensal Escherichia coli Strains Can Promote Intestinal Inflammation via Differential Interleukin-6 Production. Front. Immunol. 2018, 9, 2318. [Google Scholar] [CrossRef] [PubMed]
- Ansorge, S.; Reinhold, D.; Lendeckel, U. Propolis and Some of its Constituents Down-Regulate DNA Synthesis and Inflammatory Cytokine Production but Induce TGF-β1 Production of Human Immune Cells. Z. Nat. C 2003, 58, 580–589. [Google Scholar] [CrossRef] [PubMed]
- Seven, P.T.; Yilmaz, S.; Seven, I.; Kelestemur, G.T. The Effects of Propolis in Animals Exposed Oxidative Stress. In Oxidative Stress—Environmental Induction and Dietary Antioxidants; Lushchak, V.I., Ed.; InTech: London, UK, 2012; pp. 267–288. [Google Scholar] [CrossRef][Green Version]
- Ayala, A.; Muñoz, M.F.; Arguelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxidative Med. Cell. Longev. 2014, 2014, 1–31. [Google Scholar] [CrossRef]
- Halawa, A.A.; El-Adl, M.A.; Hamed, M.F.; Balboula, A.Z.; Elmetwally, M. Lipopolysaccharide Prompts Oxidative Stress and Apoptosis in Rats’ Testicular Tissue. J. Veter. Health 2018, 1, 20–31. [Google Scholar] [CrossRef]
- Yangi, B.; Ustuner, M.C.; Dinçer, M.; Özbayer, C.; Tekin, N.; Üstüner, D.; Colak, E.; Kolac, U.K.; Entok, E. Propolis Protects Endotoxin Induced Acute Lung and Liver Inflammation Through Attenuating Inflammatory Responses and Oxidative Stress. J. Med. Food 2018, 21, 1096–1105. [Google Scholar] [CrossRef]
- Kocot, J.; Kiełczykowska, M.; Luchowska-Kocot, D.; Kurzepa, J.; Musik, I. Antioxidant Potential of Propolis, Bee Pollen, and Royal Jelly: Possible Medical Application. Oxidative Med. Cell. Longev. 2018, 2018, 1–29. [Google Scholar] [CrossRef]
- Wang, S.; Peng, Q.; Jia, H.M.; Zeng, X.F.; Zhu, J.L.; Hou, C.L.; Liu, X.T.; Yang, F.J.; Qiao, S.Y. Prevention of Escherichia coli infection in broiler chickens with Lactobacillus plantarum B1. Poult. Sci. 2017, 96, 2576–2586. [Google Scholar] [CrossRef]
- Wang, K.; Jin, X.; Chen, Y.; Song, Z.; Jiang, X.; Hu, F.-L.; Conlon, M.A.; Topping, D.L. Polyphenol-rich propolis extracts strengthen intestinal barrier function by activating AMPK and ERK signaling. Nutrients 2016, 8, 272. [Google Scholar] [CrossRef]
- Xue, M.; Liu, Y.; Xu, H.; Zhou, Z.; Ma, Y.; Sun, T.; Liu, M.; Zhang, H.-Q.; Liang, H. Propolis modulates the gut microbiota and improves the intestinal mucosal barrier function in diabetic rats. Biomed. Pharmacother. 2019, 118, 109393. [Google Scholar] [CrossRef]
- Marshall, J.C.; Christou, N.V.; Meakins, J.L. Small-bowel bacterial overgrowth and systemic immunosuppression in experimental peritonitis. Surgery 1988, 104, 404–411. [Google Scholar]
- Chen, X.; Song, M.; Zhang, B.; Zhang, Y. Reactive Oxygen Species Regulate T Cell Immune Response in the Tumor Microenvironment. Oxidative Med. Cell. Longev. 2016, 2016, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Larbi, A.; Kempf, J.; Pawelec, G. Oxidative stress modulation and T cell activation. Exp. Gerontol. 2007, 42, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Moro-García, M.A.; Mayo, J.C.; Sainz, R.M.; Alonso-Arias, R. Influence of Inflammation in the Process of T Lymphocyte Differentiation: Proliferative, Metabolic, and Oxidative Changes. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef]
- Colitti, M.; Stefanon, B.; Gabai, G.; Gelain, M.E.; Bonsembiante, F. Oxidative Stress and Nutraceuticals in the Modulation of the Immune Function: Current Knowledge in Animals of Veterinary Interest. Antioxidants 2019, 8, 28. [Google Scholar] [CrossRef]
- Tohyama, Y.; Takano, T.; Yamamura, H. B cell responses to oxidative stress. Curr. Pharm. Des. 2004, 10, 835–839. [Google Scholar] [CrossRef]
- Wagner, A.; Garner-Spitzer, E.; Jasińska, J.; Kollaritsch, H.; Stiasny, K.; Kundi, M.; Wiedermann, U. Age-related differences in humoral and cellular immune responses after primary immunisation: Indications for stratified vaccination schedules. Sci. Rep. 2018, 8, 9825. [Google Scholar] [CrossRef]
- Búfalo, M.C.; Ferreira, I.; Costa, G.; Francisco, V.L.G.; Liberal, J.; Cruz, M.T.; Lopes, M.; Batista, M.T.; Sforcin, J.M. Propolis and its constituent caffeic acid suppress LPS-stimulated pro-inflammatory response by blocking NF-κB and MAPK activation in macrophages. J. Ethnopharmacol. 2013, 149, 84–92. [Google Scholar] [CrossRef]
- Stefanetti, R.J.; Voisin, S.; Russell, A.P.; Lamon, S. Recent advances in understanding the role of FOXO3. F1000Research 2018, 7, 1372. [Google Scholar] [CrossRef]
- Cui, C.; Han, S.; Yin, H.; Luo, B.; Shen, X.; Yang, F.; Liu, Z.; Zhu, Q.; Li, D.; Wang, Y. FOXO3 Is Expressed in Ovarian Tissues and Acts as an Apoptosis Initiator in Granulosa Cells of Chickens. BioMed Res. Int. 2019, 2019, 6902906. [Google Scholar] [CrossRef]
- Wang, P.; Geng, J.; Gao, J.; Zhao, H.; Li, J.; Shi, Y.; Yang, B.; Xiao, C.; Linghu, Y.; Sun, X.; et al. Macrophage achieves self-protection against oxidative stress-induced ageing through the Mst-Nrf2 axis. Nat. Commun. 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Peng, S.L. Foxo in the immune system. Oncogene 2008, 27, 2337–2344. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Ortega, A.; Feinberg, D.; Liang, Y.; Rossa, J.C.; Graves, D.T. The Role of Forkhead Box 1 (FOXO1) in the Immune System: Dendritic Cells, T Cells, B Cells, and Hematopoietic Stem Cells. Crit. Rev. Immunol. 2017, 37, 1–13. [Google Scholar] [CrossRef]
- Bi, Y.; Xu, L.; Qiu, L.; Wang, S.; Liu, X.; Zhang, Y.; Chen, Y.; Xu, Q.; Chang, G.; Chen, G.; et al. Reticuloendotheliosis Virus Inhibits the Immune Response Acting on Lymphocytes from Peripheral Blood of Chicken. Front. Physiol. 2018, 9, 1–9. [Google Scholar] [CrossRef]
- Du, W.W.; Fang, L.; Yang, W.; Wu, N.; Awan, F.M.; Yang, Z.; Yang, B.B. Induction of tumor apoptosis through a circular RNA enhancing Foxo3 activity. Cell Death Differ. 2016, 24, 357–370. [Google Scholar] [CrossRef]
| Parameters | BD 1 | BD + PR 2 | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| C 3 | EC 4 | PR + EC 5 | PR 6 | PR | EC | PR × EC | ||
| Egg Number/hen/7days | 6.36 a | 4.08 c | 4.90 b | 6.76 a | 0.13 | 0.004 | <0.0001 | 0.267 |
| Egg weight, g | 61.30 a | 53.60 c | 57.40 b | 63.20 a | 0.59 | 0.003 | <0.0001 | 0.264 |
| Feed intake, g/d | 117.60 a | 88.60 c | 104.40 b | 115.80 a | 2.11 | 0.005 | <0.0001 | 0.008 |
| Feed conversion, kg/kg | 2.11 b | 2.86 a | 2.63 a | 1.90 b | 0.08 | 0.073 | <0.0001 | 0.953 |
| Parameters | BD 1 | BD + PR 2 | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| C 3 | EC 4 | PR + EC 5 | PR 6 | PR | EC | PR × EC | ||
| TNF-α, pg/mL | 94.68 c | 178.00 a | 155.50 b | 93.66 c | 4.79 | 0.026 | <0.0001 | 0.039 |
| IL-1β, ng/mL | 0.29 c | 0.83 a | 0.52 b | 0.26 c | 0.03 | <0.0001 | <0.0001 | 0.0002 |
| Cort, pg/mL | 5.44 c | 14.86 a | 9.26 b | 3.28 c | 0.79 | 0.0002 | <0.0001 | 0.044 |
| MDA, μM/mL | 2.00 b | 4.35 a | 2.36 b | 1.60 b | 0.43 | 0.014 | 0.002 | 0.086 |
| TAC, μM/mL | 3.97 b | 2.63 c | 3.31 bc | 7.29 a | 0.42 | 0.0002 | <0.0001 | 0.006 |
| SOD, μM/mL | 355.6 b | 355.0 b | 386.0 ab | 425.0 a | 18.0 | 0.013 | 0.288 | 0.302 |
| Parameters | BD 1 | BD + PR 2 | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| C 3 | EC 4 | PR + EC 5 | PR 6 | PR | EC | PR × EC | ||
| TWBC, ×103/mL | 56.86 b | 36.70 d | 47.70 c | 64.20 a | 2.34 | 0.001 | <0.0001 | 0.445 |
| H/L ratio | 0.38 c | 0.98 a | 0.67 b | 0.32 c | 0.04 | 0.0007 | <0.0001 | 0.009 |
| SI T-lymphocytes | 2.78 b | 1.02 d | 1.96 c | 3.64 a | 0.20 | 0.0004 | <0.0001 | 0.845 |
| SI B-lymphocytes | 1.90 b | 0.94 c | 1.60 b | 2.80 a | 0.17 | 0.0003 | <0.0001 | 0.487 |
| Wattle thickness, mm | 0.45 b | 0.36 c | 0.45 b | 0.54 a | 0.02 | 0.0006 | 0.0005 | 0.964 |
| Parameters | BD 1 | BD + PR 2 | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| C 3 | EC 4 | PR + EC 5 | PR 6 | PR | EC | PR × EC | ||
| Villi height, μm | 1960 c | 1905 c | 2083 b | 2272 a | 34.9 | <0.0001 | 0.003 | 0.073 |
| Crypt depth, μm | 385 bc | 366 c | 406 b | 444 a | 10.8 | 0.0003 | 0.018 | 0.384 |
| Villi/Crypts ratio | 5.12 | 5.22 | 5.13 | 5.14 | 0.18 | 0.851 | 0.800 | 0.766 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbas, A.O.; Alaqil, A.A.; El-Beltagi, H.S.; Abd El-Atty, H.K.; Kamel, N.N. Modulating Laying Hens Productivity and Immune Performance in Response to Oxidative Stress Induced by E. coli Challenge Using Dietary Propolis Supplementation. Antioxidants 2020, 9, 893. https://doi.org/10.3390/antiox9090893
Abbas AO, Alaqil AA, El-Beltagi HS, Abd El-Atty HK, Kamel NN. Modulating Laying Hens Productivity and Immune Performance in Response to Oxidative Stress Induced by E. coli Challenge Using Dietary Propolis Supplementation. Antioxidants. 2020; 9(9):893. https://doi.org/10.3390/antiox9090893
Chicago/Turabian StyleAbbas, Ahmed O., Abdulaziz A. Alaqil, Hossam S. El-Beltagi, Hanaa K. Abd El-Atty, and Nancy N. Kamel. 2020. "Modulating Laying Hens Productivity and Immune Performance in Response to Oxidative Stress Induced by E. coli Challenge Using Dietary Propolis Supplementation" Antioxidants 9, no. 9: 893. https://doi.org/10.3390/antiox9090893
APA StyleAbbas, A. O., Alaqil, A. A., El-Beltagi, H. S., Abd El-Atty, H. K., & Kamel, N. N. (2020). Modulating Laying Hens Productivity and Immune Performance in Response to Oxidative Stress Induced by E. coli Challenge Using Dietary Propolis Supplementation. Antioxidants, 9(9), 893. https://doi.org/10.3390/antiox9090893