Importance of the Use of Oxidative Stress Biomarkers and Inflammatory Profile in Aqueous and Vitreous Humor in Diabetic Retinopathy
Abstract
:1. Introduction
2. Diabetic Retinopathy
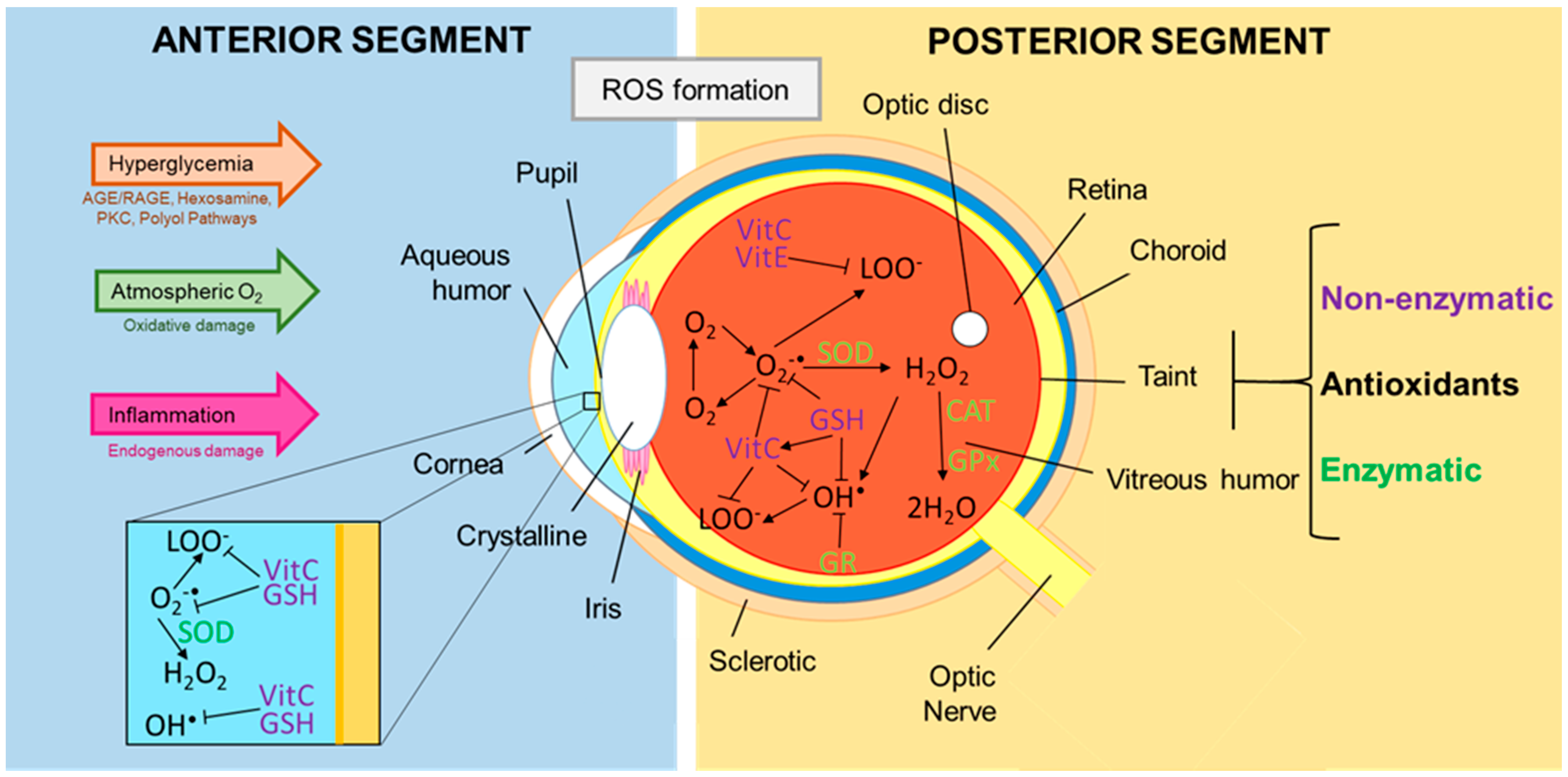
3. Role of Oxidants and Antioxidants in the Eye with Diabetic Retinopathy
3.1. Oxidative Stress and the Damage It Causes to the Eye
3.2. Antioxidants Present in the Tear Film, Aqueous Humor, and Vitreous Humor
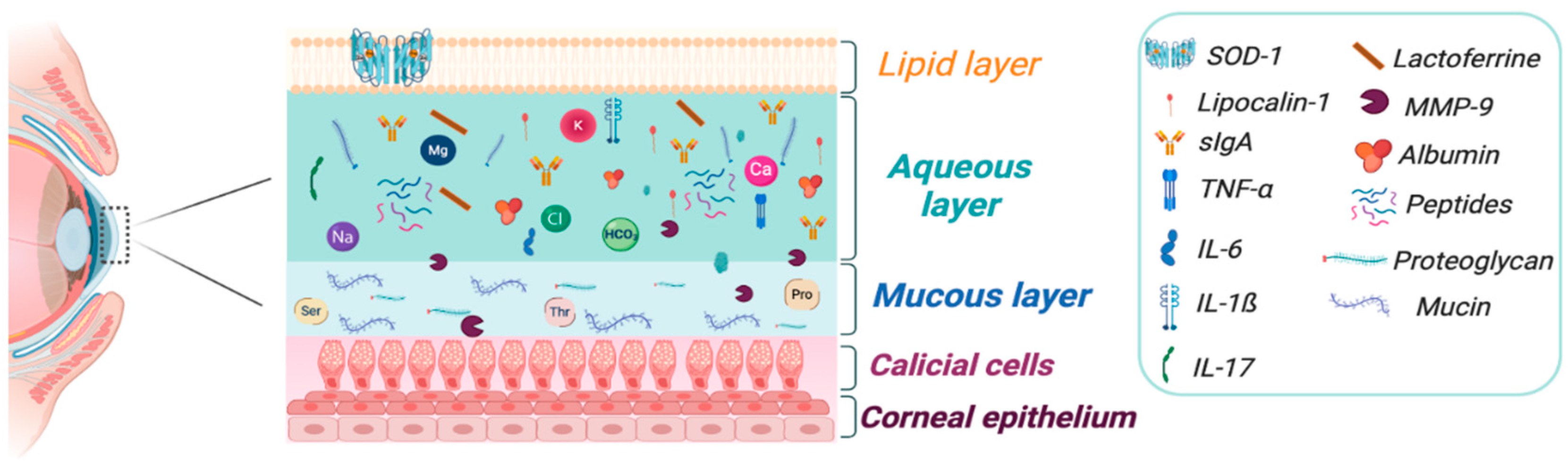
4. Ocular Matrices: Tears, Aqueous Humor, and Vitreous Humor
4.1. Tears
- Direct or aspiration: the collection is through microcapillary tubes (MCT) or micropipettes, the tip of the tube is placed in the cul-de-sac for 5 min, in non-stimulated tears (NST), until it forms a lake of tear, then for capillarity tears are absorbed and the sample (5.5–6.5 µL) is transferred immediately into a sterile tube with storage solution or buffer assay to produce a dilution 1:10 and storage at −80 °C. The main advantage is the amount of proteins and biomarkers obtained directly of ocular surface. A disadvantage is the loss of proteins may occur due to incomplete pouring into microvials [75,76].
- Indirect methods: These methods collect the samples of tears through absorbing papers like cellulose sponges or Schirmer test strips (STS), both are invasive techniques. The cellulose sponges are used frequently to analyze inflammatory markers like interleukins and MMP-9 and they are measured by enzyme-linked immunosorbent assay (ELISA) or Luminex technology using tears that were collected with Merocel, Pro-ophta, or Weck-Cel sponges. However, comparative studies with simultaneous measurements of cytokines have shown that Merocel is useful for clinical assess for cytokines/chemokines levels but have the limitation with measures of IL-7 and IL-4 due to protein stability problems with the extraction buffer [75,76,77]. In the case of STS, it may also be used for cytokine analysis assays. For collection of tears, the strip is placed on the inferior fornix of the eye and the patient should close their eyes for 5 min. After completed the time, the patient should open their eyes for remove carefully the strip and then it is collocated into a sterile 1.5 mL tube. Immediately transfer the tube to the laboratory to process later in a bead based multiplex assay or store at −80 °C. With this method the sample contains higher amounts of cellular proteins, lipids, and mucous compared with MCT and the analysis with multiplex provide high sensitivity for analyzing cytokines and other proteins [78].
4.2. Aqueous Humor
4.3. Vitreous Humor
5. Measurement of Biomarkers
5.1. Oxidative Stress: ROS and Relevance in DR
5.2. Inflammation: Cytokines and Relevance in DR
5.3. Apoptosis
6. Metabolic Memory of Oxidative Stress in Diabetic Retinopathy
6.1. DNA Methylation
6.2. Modification of Histones
6.3. Chromatin Remodeling
6.4. Non-Coding RNA
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Campochiaro, P.A. Molecular pathogenesis of retinal and choroidal vascular diseases. Prog. Retin. Eye Res. 2015, 49, 67–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahajan, N.; Arora, P.; Sandhir, R.A.-O. Perturbed Biochemical Pathways and Associated Oxidative Stress Lead to Vascular Dysfunctions in Diabetic Retinopathy. Oxid. Med. Cell. Longev. 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cunha-Vaz, J.; Bernardes, R.; Lobo, C. Blood-retinal barrier. Eur. J. Ophthalmol. 2011, 21, S3–S9. [Google Scholar] [CrossRef]
- Alder, V.A.; Su, E.N.; Yu, D.Y.; Cringle, S.; Yu, D.-Y. Overview of studies on metabolic and vascular regulatory changes in early diabetic retinopathy. Aust. N. Z. J. Ophthalmol. 1998, 26, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Pusparajah, P.; Lee, L.-H.; Kadir, K.A. Molecular Markers of Diabetic Retinopathy: Potential Screening Tool of the Future? Front. Physiol. 2016, 7, 200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aiello, L.P.; Cahill, M.T.; Wong, J.S. Systemic considerations in the management of diabetic retinopathy. Am. J. Ophthalmol. 2001, 132, 760–776. [Google Scholar] [CrossRef]
- Yau, J.W.; Rogers, S.L.; Kawasaki, R.; Lamoureux, E.L.; Kowalski, J.W.; Bek, T.; Chen, S.-J.; Dekker, J.M.; Fletcher, A.; Grauslund, J.; et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 2012, 35, 556–564. [Google Scholar] [CrossRef] [Green Version]
- Cecilia, O.-M.; Alberto, C.-G.J.; José, N.-P.; Germán, C.-M.E.; Karen, L.-C.A.; Miguel, R.-P.L.; Raúl, R.-R.R.; Daniel, R.-C.A. Oxidative Stress as the Main Target in Diabetic Retinopathy Pathophysiology. J. Diabetes Res. 2019, 2019, 8562408. [Google Scholar] [CrossRef] [Green Version]
- Zheng, Y.; He, M.; Congdon, N. The worldwide epidemic of diabetic retinopathy. Indian J. Ophthalmol. 2012, 60, 428–431. [Google Scholar] [CrossRef]
- Avidor, D.; Loewenstein, A.; Waisbourd, M.; Nutman, A. Cost-effectiveness of diabetic retinopathy screening programs using telemedicine: A systematic review. Cost Eff. Resour. Alloc. 2020, 18, 16. [Google Scholar] [CrossRef] [Green Version]
- Whitehead, M.; Wickremasinghe, S.; Osborne, A.; Wijngaarden, P.; Martin, K.R. Diabetic retinopathy: A complex pathophysiology requiring novel therapeutic strategies. Expert Opin. Biol. Ther. 2018, 18, 1257–1270. [Google Scholar] [CrossRef]
- Robles-Rivera, R.R.; Castellanos-González, J.A.; Olvera-Montaño, C.; Flores-Martin, R.A.; López-Contreras, A.K.; Arevalo-Simental, D.E.; Cardona-Muñoz, E.G.; Roman-Pintos, L.M.; Rodríguez-Carrizalez, A.D. Adjuvant Therapies in Diabetic Retinopathy as an Early Approach to Delay Its Progression: The Importance of Oxidative Stress and Inflammation. Oxid. Med. Cell. Longev. 2020, 2020, 3096470. [Google Scholar] [CrossRef] [PubMed]
- Santiago, A.R.; Boia, R.; Aires, I.D.; Ambrósio, A.F.; Fernandes, R. Sweet Stress: Coping With Vascular Dysfunction in Diabetic Retinopathy. Front. Physiol. 2018, 13, 820. [Google Scholar] [CrossRef] [PubMed]
- Flaxman, S.R.; Bourne, R.R.A.; Resnikoff, S.; Ackland, P.; Braithwaite, T.; Cicinelli, M.V.; Das, A.; Jonas, J.B.; Keeffe, J.; Kempen, J.H.; et al. Global causes of blindness and distance vision impairment 1990–2020: A systematic review and meta-analysis. Lancet Glob. Health 2017, 5, 1221–1234. [Google Scholar] [CrossRef] [Green Version]
- van der Giet, M.; Henkel, C.; Schuchardt, M.; Tolle, M. Anti-VEGF Drugs in Eye Diseases: Local Therapy with Potential Systemic Effects. Curr. Pharm. Des. 2015, 21, 3548–3556. [Google Scholar] [CrossRef] [PubMed]
- Saccà, S.C.; Roszkowska, A.M.; Izzotti, A. Environmental light and endogenous antioxidants as the main determinants of non-cancer ocular diseases. Mutat. Res. Rev. Mutat. Res. 2013, 752, 153–171. [Google Scholar] [CrossRef] [PubMed]
- Riley, P.A. Free Radicals in Biology: Oxidative Stress and the Effects of Ionizing Radiation. Int. J. Radiat. Biol. 1994, 65, 27–33. [Google Scholar] [CrossRef]
- Richdale, K.; Chao, C.; Hamilton, M. Eye care providers’ emerging roles in early detection of diabetes and management of diabetic changes to the ocular surface: A review. BMJ Open Diabetes Res. Care 2020, 8, e001094. [Google Scholar] [CrossRef]
- DeMill, D.L.; Hussain, M.; Pop-Busui, R.; Shtein, R.M. Ocular surface disease in patients with diabetic peripheral neuropathy. Br. J. Ophthalmol. 2016, 100, 924. [Google Scholar] [CrossRef] [Green Version]
- Yoon, K.C.; Im, S.K.; Seo, M.S. Changes of Tear Film and Ocular Surface in Diabetes Mellitus. Korean J. Ophthalmol. 2004, 18, 168–174. [Google Scholar] [CrossRef] [Green Version]
- Behl, T.; Kaur, I.; Kotwani, A. Implication of oxidative stress in progression of diabetic retinopathy. Surv. Ophthalmol. 2016, 61, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Johnson, F.; Giulivi, C. Superoxide dismutases and their impact upon human health. Mol. Asp. Med. 2005, 26, 340–352. [Google Scholar] [CrossRef] [PubMed]
- Leonard, S.S.; Harris, G.K.; Shi, X. Metal-induced oxidative stress and signal transduction. Free Radic. Biol. Med. 2004, 37, 1921–1942. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Mehta, G.; Vasiliou, V. Antioxidant defenses in the ocular surface. Ocul. Surf. 2009, 7, 176–185. [Google Scholar] [CrossRef] [Green Version]
- Hardy, P.; Beauchamp, M.; Sennlaub, F.; Gobeil, F.J.; Tremblay, L.; Mwaikambo, B.; Lachapelle, P.; Chemtob, S. New insights into the retinal circulation: Inflammatory lipid mediators in ischemic retinopathy. Prostaglandins Leukot. Essent. Fat. Acids 2005, 72, 301–325. [Google Scholar] [CrossRef]
- Bishop, P.N. The role of extracellular matrix in retinal vascular development and preretinal neovascularization. Exp. Eye Res. 2015, 133, 30–36. [Google Scholar] [CrossRef]
- Géhl, Z.; Bakondi, E.; Resch, M.D.; Hegedűs, C.; Kovács, K.; Lakatos, P.; Szabó, A.; Nagy, Z.; Virág, L. Diabetes-induced oxidative stress in the vitreous humor. Redox Biol. 2016, 9, 100–103. [Google Scholar] [CrossRef] [Green Version]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef] [Green Version]
- Ohia, S.E.; Opere, C.A.; LeDay, A.M. Pharmacological consequences of oxidative stress in ocular tissues. Mutat. Res. Fundam. Mol. Mech. Mutagenes. 2005, 579, 22–36. [Google Scholar] [CrossRef]
- Rose, R.C.; Richer, S.P.; Bode, A.M. Ocular Oxidants and Antioxidant Protection. Proc. Soc. Exp. Biol. Med. 1998, 217, 397–407. [Google Scholar] [CrossRef]
- Shang, F.; Lu, M.; Dudek, E.; Reddan, J.; Taylor, A. Vitamin C and vitamin E restore the resistance of GSH-depleted lens cells to H2O2. Free Radic. Biol. Med. 2003, 34, 521–530. [Google Scholar] [CrossRef]
- Shichi, H. Cataract formation and prevention. Expert Opin. Investig. Drugs 2004, 13, 691–701. [Google Scholar] [CrossRef] [PubMed]
- Beatty, S.; Koh, H.-H.; Phil, M.; Henson, D.; Boulton, M. The Role of Oxidative Stress in the Pathogenesis of Age-Related Macular Degeneration. Surv. Ophthalmol. 2000, 45, 115–134. [Google Scholar] [CrossRef] [Green Version]
- Kowluru, R.; Kern, T.S.; Engerman, R.L. Abnormalities of retinal metabolism in diabetes or galactosemia II. Comparison of γ-glutamyl transpeptidase in retina and cerebral cortex, and effects of antioxidant therapy. Curr. Eye Res. 1994, 13, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Kuroki, M.; Amano, S.; Tolentino, M.; Keough, K.; Kim, I.; Bucala, R.; Adamis, A.P. Advanced glycation end products increase retinal vascular endothelial growth factor expression. J. Clin. Investig. 1998, 101, 1219–1224. [Google Scholar] [CrossRef]
- van Reyk, D.M.; Gillies, M.C.; Davies, M.J. The retina: Oxidative stress and diabetes. Redox Rep. 2003, 8, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Domènech, E.; Marfany, G. The Relevance of Oxidative Stress in the Pathogenesis and Therapy of Retinal Dystrophies. Antioxidants 2020, 9, 347. [Google Scholar] [CrossRef] [Green Version]
- Rashid, K.; Akhtar-Schaefer, I.; Langmann, T. Microglia in Retinal Degeneration. Front. Immunol. 2019, 10, 1975. [Google Scholar] [CrossRef] [Green Version]
- Maan-Yuh Lin, A.; Yang, C.-H.; Chai, C.-Y. Striatal Dopamine Dynamics Are Altered Following an Intranigral Infusion of Iron in Adult Rats. Free Radic. Biol. Med. 1998, 24, 988–993. [Google Scholar] [CrossRef]
- LeDay, A.M.; Ganguly, S.; Kulkarni, K.; Dash, A.; Opere, C.; Ohia, S. Effect of hydrogen peroxide on amino acid concentrations in bovine retina and vitreous humor, ex vivo. Methods Find Exp. Clin. Pharmacol. 2003, 25, 695–701. [Google Scholar] [CrossRef]
- Sharpe, M.A.; Robb, S.J.; Clark, J.B. Nitric oxide and Fenton/Haber–Weiss chemistry: Nitric oxide is a potent antioxidant at physiological concentrations. J. Neurochem. 2003, 87, 386–394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- LeDay, A.M.; Kulkarni, K.H.; Opere, C.A.; Ohia, S.E. Arachidonic acid metabolites and peroxide-induced inhibition of [3H]D-aspartate release from bovine isolated retinae. Curr. Eye Res. 2004, 28, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, J.M.C. The definition and measurement of antioxidants in biological systems. Free Radic. Biol. Med. 1995, 18, 125–126. [Google Scholar] [CrossRef]
- Ankamah, E.; Sebag, J.; Ng, E.; Nolan, J. Vitreous Antioxidants, Degeneration, and Vitreo-Retinopathy: Exploring the Links. Antioxidants 2019, 9, 7. [Google Scholar] [CrossRef] [Green Version]
- Behndig, A.; Svensson, B.; Marklund, S.L.; Karlsson, K. Superoxide dismutase isoenzymes in the human eye. Investig. Ophthalmol. Vis. Sci. 1998, 39, 471–475. [Google Scholar]
- Richer, S.P.; Rose, R.C. Water soluble antioxidants in mammalian aqueous humor: Interaction with UV B and hydrogen peroxide. Vis. Res. 1998, 38, 2881–2888. [Google Scholar] [CrossRef] [Green Version]
- Horwath-Winter, J.; Kirchengast, S.; Meinitzer, A.; Wachswender, C.; Faschinger, C.; Schmut, O. Determination of uric acid concentrations in human tear fluid, aqueous humour and serum. Acta Ophthalmol. 2009, 87, 188–192. [Google Scholar] [CrossRef]
- Milston, R.; Madigan, M.C.; Sebag, J. Vitreous floaters: Etiology, diagnostics, and management. Surv. Ophthalmol. 2016, 61, 211–227. [Google Scholar] [CrossRef]
- Filas, B.A.; Zhang, Q.; Okamoto, R.J.; Shui, Y.-B.; Beebe, D.C. Enzymatic degradation identifies components responsible for the structural properties of the vitreous body. Investig. Ophthalmol. Vis. Sci. 2014, 55, 55–63. [Google Scholar] [CrossRef]
- Kita, T.; Sakamoto, T.; Ishibashi, T. II.D. Hyalocytes: Essential Vitreous Cells in Vitreoretinal Health and Disease. In Vitreous: In Health and Disease; Sebag, J., Ed.; Springer: New York, NY, USA, 2014; pp. 151–164. [Google Scholar]
- Shui, Y.-B.; Holekamp, N.M.; Kramer, B.C.; Crowley, J.R.; Wilkins, M.A.; Chu, F.; Malone, P.E.; Mangers, S.J.; Hou, J.H.; Siegfried, C.J.; et al. The gel state of the vitreous and ascorbate-dependent oxygen consumption: Relationship to the etiology of nuclear cataracts. Arch. Ophthalmol. 2009, 127, 475–482. [Google Scholar] [CrossRef] [Green Version]
- Sa, A.; Elawadi, A.I. Liquefaction of the Vitreous Humor floaters is a Risk Factor for Lens Opacity and Retinal Dysfunction. J. Am. Sci. 2011, 7, 927–936. [Google Scholar]
- Mirończuk-Chodakowska, I.; Witkowska, A.M.; Zujko, M.E. Endogenous non-enzymatic antioxidants in the human body. Adv. Med Sci. 2018, 63, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. IJBS 2008, 4, 89–96. [Google Scholar]
- Park, S.W.; Ghim, W.; Oh, S.; Kim, Y.; Park, U.C.; Kang, J.; Yu, H.G. Association of vitreous vitamin C depletion with diabetic macular ischemia in proliferative diabetic retinopathy. PLoS ONE 2019, 14, e0218433. [Google Scholar] [CrossRef] [PubMed]
- Ashoori, M.; Saedisomeolia, A. Riboflavin (vitamin B2) and oxidative stress: A review. Br. J. Nutr. 2014, 111, 1985–1991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konerirajapuram, N.; Coral, K.; Punitham, R.; Sharma, T.; Kasinathan, N.; Sivaramakrishnan, R. Trace Elements Iron, Copper and Zinc in Vitreous of Patients with Various Vitreoretinal Diseases. Indian J. Ophthalmol. 2004, 52, 145–148. [Google Scholar]
- Sato, M.; Kondoh, M. Recent Studies on Metallothionein: Protection Against Toxicity of Heavy Metals and Oxygen Free Radicals. Tohoku J. Exp. Med. 2002, 196, 9–22. [Google Scholar] [CrossRef] [Green Version]
- Tinggi, U. Selenium: Its role as antioxidant in human health. Environ. Health Prev. Med. 2008, 13, 102–108. [Google Scholar] [CrossRef] [Green Version]
- Golbidi, S.; Laher, I. Antioxidant therapy in human endocrine disorders. Med. Sci. Monit. 2010, 16, RA9–RA24. [Google Scholar]
- Golbidi, S.; Badran, M.; Laher, I. Antioxidant and anti-inflammatory effects of exercise in diabetic patients. Exp. Diabetes Res. 2012, 2012, 941868. [Google Scholar] [CrossRef] [Green Version]
- Sunitha, K.; Suresh, P.; Santhosh, M.S.; Hemshekhar, M.; Thushara, R.; Marathe, G.K.; Thirunavukkarasu, C.; Kemparaju, K.; Kumar, M.S.; Girish, K. Inhibition of hyaluronidase by N-acetyl cysteine and glutathione: Role of thiol group in hyaluronan protection. Int. J. Biol. Macromol. 2013, 55, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.W.; Richa, D.C.; Hahn, P.; Green, W.R.; Dunaief, J.L. Iron toxicity as a potential factor in AMD. Retina 2007, 7, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Krizova, L.; Kalousova, M.; Kubena, A.; Benakova, H.; Zima, T.; Kovarik, Z.; Kalvoda, J.; Kalvodova, B. Increased Uric Acid and Glucose Concentrations in Vitreous and Serum of Patients with Diabetic Macular Oedema. Ophthalmic Res. 2011, 46, 73–79. [Google Scholar] [CrossRef]
- González de Vega, R.; Fernández-Sánchez, M.L.; González-Iglesias, H.; Prados, M.C.; Sanz-Medel, A. Quantitative selenium speciation by HPLC-ICP-MS(IDA) and simultaneous activity measurements in human vitreous humor. Anal. Bioanal. Chem. 2015, 407, 2405–2413. [Google Scholar] [CrossRef]
- Herbette, S.; Roeckel-Drevet, P.; Drevet, J.R. Seleno-independent glutathione peroxidases. FEBS J. 2007, 274, 2163–2180. [Google Scholar] [CrossRef]
- Wert, K.J.; Vélez, G.; Cross, M.R.; Wagner, B.A.; Teoh-Fitzgerald, M.L.; Buettner, G.R.; McAnany, J.J.; Olivier, A.; Tsang, S.H.; Harper, M.M.; et al. Extracellular superoxide dismutase (SOD3) regulates oxidative stress at the vitreoretinal interface. Free Radic. Biol. Med. 2018, 124, 408–419. [Google Scholar] [CrossRef] [Green Version]
- Yamane, K.; Minamoto, A.; Yamashita, H.; Takamura, H.; Miyamoto-Myoken, Y.; Yoshizato, K.; Nabetani, T.; Tsugita, A.; Mishima, H.K. Proteome Analysis of Human Vitreous Proteins. Mol. Cell. Proteom. 2003, 2, 1177. [Google Scholar] [CrossRef] [Green Version]
- Murthy, K.R.; Goel, R.; Subbannayya, Y.; Jacob, H.K.C.; Murthy, P.R.; Manda, S.S.; Patil, A.H.; Sharma, R.; Sahasrabuddhe, N.A.; Parashar, A.; et al. Proteomic analysis of human vitreous humor. Clin. Proteom. 2014, 11, 29. [Google Scholar] [CrossRef] [Green Version]
- Łukasik, M.; Szutowski, M.; Sołtyszewski, I.; Cieślak, P.A.; Małkowska, A. Postmortem Vitreous Humor Analysis for Xenobiotics and their Metabolites. Law Forensic Sci. 2018, 15, 1–8. [Google Scholar]
- Yadav, D.; Varma, L.T.; Yadav, K. Drug Delivery to Posterior Segment of the Eye: Conventional Delivery Strategies, Their Barriers, and Restrictions. In Drug Delivery for the Retina and Posterior Segment Disease; Springer: Cham, Switzerland, 2018; pp. 51–67. [Google Scholar] [CrossRef]
- Li, S.; Fu, X.-A.; Zhou, X.-F.; Chen, Y.-Y.; Chen, W.-Q. Angiogenesis-related cytokines in serum of proliferative diabetic retinopathy patients before and after vitrectomy. Int. J. Ophthalmol. 2012, 5, 726–730. [Google Scholar] [CrossRef]
- Tamhane, M.; Cabrera-Ghayouri, S.; Abelian, G.; Viswanath, V. Review of Biomarkers in Ocular Matrices: Challenges and Opportunities. Pharm. Res. 2019, 36, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fong, P.Y.; Shih, K.C.; Lam, P.Y.; Chan, T.C.Y.; Jhanji, V.; Tong, L. Role of tear film biomarkers in the diagnosis and management of dry eye disease. Taiwan J. Ophthalmol. 2019, 9, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Rentka, A.; Koroskenyi, K.; Hársfalvi, J.; Szekanecz, Z.; Szucs, G.; Szodoray, P.; Kemeny-Beke, A. Evaluation of commonly used tear sampling methods and their relevance in subsequent biochemical analysis. Ann. Clin. Biochem. 2017, 54, 521–529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Zazzo, A.; Micera, A.; De Piano, M.; Cortes, M.; Bonini, S. Tears and ocular surface disorders: Usefulness of biomarkers. J. Cell. Physiol. 2019, 234, 9982–9993. [Google Scholar] [CrossRef]
- Inic-Kanada, A.; Nussbaumer, A.; Montanaro, J.; Belij, S.; Schlacher, S.; Stein, E.; Bintner, N.; Merio, M.; Zlabinger, G.J.; Barisani-Asenbauer, T. Comparison of ophthalmic sponges and extraction buffers for quantifying cytokine profiles in tears using Luminex technology. Mol. Vis. 2012, 18, 2717–2725. [Google Scholar]
- Balne, P.K.; Au, V.B.; Tong, L.; Ghosh, A.; Agrawal, M.; Connolly, J. Bead Based Multiplex Assay for Analysis of Tear Cytokine Profiles. J. Vis. Exp. 2017. [Google Scholar] [CrossRef]
- Johnson, M.; McLaren, J.W.; Overby, D.R. Unconventional aqueous humor outflow: A review. Exp. Eye Res. 2017, 158, 94–111. [Google Scholar] [CrossRef] [Green Version]
- Chowdhury, U.R.; Madden, B.J.; Charlesworth, M.C.; Fautsch, M.P. Proteome analysis of human aqueous humor. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4921–4931. [Google Scholar] [CrossRef]
- Murthy, K.R.; Rajagopalan, P.; Pinto, S.M.; Advani, J.; Murthy, P.R.; Goel, R.; Subbannayya, Y.; Balakrishnan, L.; Dash, M.; Anil, A.K.; et al. Proteomics of Human Aqueous Humor. Omics J. Integr. Biol. 2015, 19, 283–293. [Google Scholar] [CrossRef]
- Goel, M.; Picciani, R.G.; Lee, R.K.; Bhattacharya, S.K. Aqueous humor dynamics: A review. Open Ophthalmol. J. 2010, 4, 52–59. [Google Scholar] [CrossRef] [Green Version]
- Rusnak, S.; Vrzalová, J.; Sobotova, M.; Hecová, L.; Ricarova, R.; Topolcan, O. The Measurement of Intraocular Biomarkers in Various Stages of Proliferative Diabetic Retinopathy Using Multiplex xMAP Technology. J. Ophthalmol. 2015, 2015, 424783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scott, J.E. The chemical morphology of the vitreous. Eye 1992, 6, 553–555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ulrich, J.N.; Spannagl, M.; Kampik, A.; Gandorfer, A. Components of the fibrinolytic system in the vitreous body in patients with vitreoretinal disorders. Clin. Exp. Ophthalmol. 2008, 36, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.W.; Sauter, J.L.; Johnson, P.K.; Chen, C.-D.; Olsen, T.W. Identification and localization of major soluble vitreous proteins in human ocular tissue. Am. J. Ophthalmol. 2004, 137, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Ghodasra, D.H.; Fante, R.; Gardner, T.W.; Langue, M.; Niziol, L.M.; Besirli, C.; Cohen, S.R.; Dedania, V.S.; Demirci, H.; Jain, N.; et al. Safety and Feasibility of Quantitative Multiplexed Cytokine Analysis From Office-Based Vitreous Aspiration. Investig. Ophthalmol. Vis. Sci. 2016, 57, 3017–3023. [Google Scholar] [CrossRef] [Green Version]
- Bergandi, L.; Skorokhod, O.A.; La Grotta, R.; Schwarzer, E.; Nuzzi, R. Oxidative Stress, Lipid Peroxidation, and Loss of Hyaluronic Acid in the Human Vitreous Affected by Synchysis Scintillans. J. Ophthalmol. 2019, 2019, 7231015. [Google Scholar] [CrossRef]
- Srividya, G.; Jain, M.; Mahalakshmi, K.; Gayathri, S.; Raman, R.; Angayarkanni, N. A novel and less invasive technique to assess cytokine profile of vitreous in patients of diabetic macular oedema. Eye 2018, 32, 820–829. [Google Scholar] [CrossRef] [Green Version]
- García-Ramírez, M.; Canals, F.; Hernandez, C.; Colomé, N.; Ferrer, C.; Carrasco, E.; García-Arumí, J.; Simó, R. Proteomic analysis of human vitreous fluid by fluorescence-based difference gel electrophoresis (DIGE): A new strategy for identifying potential candidates in the pathogenesis of proliferative diabetic retinopathy. Diabetologia 2007, 50, 1294–1303. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, A.J.; Joglekar, M.V.; Hardikar, A.A.; Keech, A.C.; O’Neal, D.N.; Januszewski, A.S. Biomarkers in Diabetic Retinopathy. Rev. Diabet. Stud. 2015, 12, 159–195. [Google Scholar] [CrossRef] [Green Version]
- FDANIH Biomarker Working Group. BEST (Biomarkers, EndpointS, and other Tools) Resource; Food and Drug Administration: Silver Spring, MD, USA, 2018; p. 61. [Google Scholar]
- Wecker, T.; Hoffmeier, K.; Plötner, A.; Grüning, B.; Horres, R.; Backofen, R.; Reinhard, T.; Schlunck, G. MicroRNA Profiling in Aqueous Humor of Individual Human Eyes by Next-Generation Sequencing. Investig. Ophthalmol. Vis. Sci. 2016, 57, 1706–1713. [Google Scholar] [CrossRef]
- Tanaka, Y.; Tsuda, S.; Kunikata, H.; Sato, J.; Kokubun, T.; Yasuda, M.; Nishiguchi, K.M.; Inada, T.; Nakazawa, T. Profiles of Extracellular miRNAs in the Aqueous Humor of Glaucoma Patients Assessed with a Microarray System. Sci. Rep. 2014, 4, 5089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kulaksızoglu, S.; Karalezli, A. Aqueous Humour and Serum Levels of Nitric Oxide, Malondialdehyde and Total Antioxidant Status in Patients with Type 2 Diabetes with Proliferative Diabetic Retinopathy and Nondiabetic Senile Cataracts. Can. J. Diabetes 2016, 40, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Nezzar, H.; Chiambaretta, F.; Marceau, G.; Blanchon, L.; Faye, B.; Dechelotte, P.; Rigal, D.; Sapin, V. Molecular and metabolic retinoid pathways in the human ocular surface. Mol. Vis. 2007, 13, 1641–1650. [Google Scholar] [PubMed]
- Goyal, A.; Srivastava, A.; Sihota, R.; Kaur, J. Evaluation of Oxidative Stress Markers in Aqueous Humor of Primary Open Angle Glaucoma and Primary Angle Closure Glaucoma Patients. Curr. Eye Res. 2014, 39, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Flohé, L.; Günzler, W.A. Assays of glutathione peroxidase. Methods Enzymol. 1984, 105, 114–120. [Google Scholar] [CrossRef]
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and Clinical Significance of Biomarkers of Oxidative Stress in Humans. Oxid. Med. Cell. Longev. 2017, 2017, 6501046. [Google Scholar] [CrossRef]
- Huang, W.; Koralewska-Makár, A.; Bauer, B.; Åkesson, B. Extracellular glutathione peroxidase and ascorbic acid in aqueous humor and serum of patients operated on for cataract. Clin. Chim. Acta 1997, 261, 117–130. [Google Scholar] [CrossRef]
- Ferreira, S.M.; Lerner, S.F.; Brunzini, R.; Evelson, P.; Llesuy, S. Oxidative stress markers in aqueous humor of glaucoma patients. Am. J. Ophthalmol. 2004, 137, 62–69. [Google Scholar] [CrossRef]
- Stadtman, E.R.; Oliver, C.N. Metal-catalyzed oxidation of proteins. Physiological consequences. J. Biol. Chem. 1991, 266, 2005–2008. [Google Scholar]
- Altomare, E.; Grattagliano, I.; Vendemaile, G.; Micelli-Ferrari, T.; Signorile, A.; Cardia, L. Oxidative protein damage in human diabetic eye: Evidence of a retinal participation. Eur. J. Clin. Investig. 1997, 27, 141–147. [Google Scholar] [CrossRef]
- Mancino, R.; Di Pierro, N.; Varesi, C.; Cerulli, A.; Feraco, A.; Cedrone, C.; Pinazo-Duran, M.D.; Coletta, M.; Nucci, C. Lipid peroxidation and total antioxidant capacity in vitreous, aqueous humor, and blood samples from patients with diabetic retinopathy. Mol. Vis. 2011, 17, 1298–1304. [Google Scholar]
- Brzović-Šarić, V.; Landeka, I.; Šarić, B.; Barberić, M.; Andrijašević, L.; Cerovski, B.; Oršolić, N.; Đikić, D. Levels of selected oxidative stress markers in the vitreous and serum of diabetic retinopathy patients. Mol. Vis. 2015, 21, 649–664. [Google Scholar] [PubMed]
- Georgakopoulos, C.D.; Lamari, F.N.; Karathanasopoulou, I.N.; Gartaganis, V.S.; Pharmakakis, N.M.; Karamanos, N. Tear analysis of ascorbic acid, uric acid and malondialdehyde with capillary electrophoresis. Biomed. Chromatogr. 2010, 24, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Martínez, F.J.; Piñas-García, P.; Lleó-Pérez, A.; Zanon-Moreno, V.C.; Bendala-Tufanisco, E.; García-Medina, J.; Vinuesa-Silva, I.; Pinazo-Durán, M.D. Biomarkers of lipid peroxidation in the aqueous humor of primary open-angle glaucoma patients. Arch. Soc. Esp. Oftalmol. 2016, 91, 357–362. [Google Scholar] [CrossRef]
- Behndig, A.; Karlsson, K.; Johansson, B.O.; Brännström, T.; Marklund, S.L. Superoxide dismutase isoenzymes in the normal and diseased human cornea. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2293–2296. [Google Scholar]
- Kim, E.B.; Kim, H.K.; Hyon, J.Y.; Wee, W.R.; Shin, Y.J. Oxidative Stress Levels in Aqueous Humor from High Myopic Patients. Korean J. Ophthalmol. 2016, 30, 172–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koliakos, G.G.; Konstas, A.G.P.; Schlötzer-Schrehardt, U.; Hollo, G.; Katsimbris, I.E.; Georgiadis, N.; Ritch, R. 8-Isoprostaglandin F2a and ascorbic acid concentration in the aqueous humour of patients with exfoliation syndrome. Br. J. Ophthalmol. 2003, 87, 353–356. [Google Scholar] [CrossRef] [Green Version]
- Rahim, A. 8-Isoprostaglandin F2a Levels in Aqueous Humor of Senile and Diabetic Cataract Patients. Iosr J. Dent. Med Sci. 2012, 2, 40–42. [Google Scholar] [CrossRef]
- Yilmaz, G.; Esser, P.; Kociek, N.; Aydin, P.; Heimann, K. Elevated vitreous nitric oxide levels in patients with proliferative diabetic retinopathy. Am. J. Ophthalmol. 2000, 130, 87–90. [Google Scholar] [CrossRef]
- Liu, J.; Shi, B.; He, S.; Yao, X.; Willcox, M.D.; Zhao, Z. Changes to tear cytokines of type 2 diabetic patients with or without retinopathy. Mol. Vis. 2010, 16, 2931–2938. [Google Scholar] [PubMed]
- Liu, R.; Gao, C.; Chen, H.; Li, Y.; Jin, Y.; Qi, H. Analysis of Th17-associated cytokines and clinical correlations in patients with dry eye disease. PLoS ONE 2017, 12, e0173301. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Zhang, X.; Liao, N.; Wen, F. Assessment of biomarkers using multiplex assays in aqueous humor of patients with diabetic retinopathy. BMC Ophthalmol. 2017, 17, 176. [Google Scholar] [CrossRef]
- Funatsu, H.; Yamashita, H.; Mimura, T.; Noma, H.; Nakamura, S.; Hori, S. Risk evaluation of outcome of vitreous surgery based on vitreous levels of cytokines. Eye 2007, 21, 377–382. [Google Scholar] [CrossRef]
- Tsai, T.; Kuehn, S.; Tsiampalis, N.; Vu, M.-K.; Kakkassery, V.; Stute, G.; Dick, H.B.; Joachim, S.C. Anti-inflammatory cytokine and angiogenic factors levels in vitreous samples of diabetic retinopathy patients. PLoS ONE 2018, 13, e0194603. [Google Scholar] [CrossRef] [Green Version]
- Chernykh, V.; Smirnov, E.; Varvarinsky, Y.; Chernykh, D.; Obukhova, O.; Trunov, A. IL-4, IL-6, IL-10, IL-17A and vascular endothelial growth factor in the vitreous of patients with proliferative diabetic retinopathy. Adv. Biosci. Biotechnol. 2015, 5, 184–187. [Google Scholar] [CrossRef] [Green Version]
- Sun, C.; Zhang, H.; Jiang, J.; Li, Y.; Nie, C.; Gu, J.; Luo, L.; Wang, Z. Angiogenic and inflammatory biomarker levels in aqueous humor and vitreous of neovascular glaucoma and proliferative diabetic retinopathy. Int. Ophthalmol. 2020, 40, 467–475. [Google Scholar] [CrossRef]
- Simsek, M.; Ozdal, P.C.; Akbiyik, F.; Citirik, M.; Berker, N.; Erol, Y.O.; Yilmazbas, P. Aqueous humor IL-8, IL-10, and VEGF levels in Fuchs’ uveitis syndrome and Behçet’s uveitis. Int. Ophthalmol. 2019, 39, 2629–2636. [Google Scholar] [CrossRef]
- Mao, C.; Yan, H. Roles of elevated intravitreal IL-1β and IL-10 levels in proliferative diabetic retinopathy. Indian J. Ophthalmol. 2014, 62, 699–701. [Google Scholar] [CrossRef]
- Costagliola, C.; Romano, V.; De Tollis, M.; Aceto, F.; Dell’Omo, R.; Romano, M.; Pedicino, C.; Semeraro, F. TNF-alpha levels in tears: A novel biomarker to assess the degree of diabetic retinopathy. Mediat. Inflamm. 2013, 2013, 629529. [Google Scholar] [CrossRef]
- Wu, H.; Hwang, D.-K.; Song, X.; Tao, Y. Association between Aqueous Cytokines and Diabetic Retinopathy Stage. J. Ophthalmol. 2017, 2017, 9402198. [Google Scholar] [CrossRef]
- Boss, J.D.; Singh, P.K.; Pandya, H.K.; Tosi, J.; Kim, C.; Tewari, A.; Juzych, M.S.; Abrams, G.W.; Kumar, A. Assessment of Neurotrophins and Inflammatory Mediators in Vitreous of Patients With Diabetic Retinopathy. Investig. Ophthalmol. Vis. Sci. 2017, 58, 5594–5603. [Google Scholar] [CrossRef]
- Canataroglu, H.; Varinli, I.; Ozcan, A.; Canataroglu, A.; Doran, F.; Varinli, S. Interleukin (IL)-6, Interleukin (IL)-8 Levels and Cellular Composition of the Vitreous Humor in Proliferative Diabetic Retinopathy, Proliferative Vitreoretinopathy, and Traumatic Proliferative Vitreoretinopathy. Ocul. Immunol. Inflamm. 2005, 13, 375–381. [Google Scholar] [CrossRef]
- Liu, R.; Ma, B.; Gao, Y.; Ma, B.; Liu, Y.; Qi, H. Tear Inflammatory Cytokines Analysis and Clinical Correlations in Diabetes and Nondiabetes With Dry Eye. Am. J. Ophthalmol. 2019, 200, 10–15. [Google Scholar] [CrossRef]
- Fu, R.; Klinngam, W.; Heur, M.; Edman, M.C.; Hamm-Alvarez, S.F. Tear Proteases and Protease Inhibitors: Potential Biomarkers and Disease Drivers in Ocular Surface Disease. Eye Contact Lenses 2020, 46 (Suppl. 2), S70–S83. [Google Scholar] [CrossRef]
- Kłysik, A.; Naduk-Kik, J.; Hrabec, Z.; Gos, R.; Hrabec, E. Intraocular matrix metalloproteinase 2 and 9 in patients with diabetes mellitus with and without diabetic retinopathy. Arch. Med Sci. AMS 2010, 6, 375–381. [Google Scholar] [CrossRef]
- Tuuminen, R.; Loukovaara, S. High intravitreal TGF-β1 and MMP-9 levels in eyes with retinal vein occlusion. Eye 2014, 28, 1095–1099. [Google Scholar] [CrossRef]
- Luo, L.; Li, D.Q.; Pflugfelder, S.C. Hyperosmolarity-induced apoptosis in human corneal epithelial cells is mediated by cytochrome c and MAPK pathways. Cornea 2007, 26, 452–460. [Google Scholar] [CrossRef]
- Peskin, A.V.; Winterbourn, C.C. Assay of superoxide dismutase activity in a plate assay using WST-1. Free Radic. Biol. Med. 2017, 103, 188–191. [Google Scholar] [CrossRef]
- Kernell, A.; Lundh, B.L.; Marklund, S.L.; Skoog, K.O.; Björkstén, B. Superoxide dismutase in the anterior chamber and the vitreous of diabetic patients. Investig. Ophthalmol. Vis. Sci. 1992, 33, 3131–3135. [Google Scholar]
- Sies, H. Total Antioxidant Capacity: Appraisal of a Concept. J. Nutr. 2007, 137, 1493–1495. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Ghiselli, A.; Serafini, M.; Natella, F.; Scaccini, C. Total antioxidant capacity as a tool to assess redox status: Critical view and experimental data. Free Radic. Biol. Med. 2000, 29, 1106–1114. [Google Scholar] [CrossRef]
- Saccà, S.C.; Pascotto, A.; Camicione, P.; Capris, P.; Izzotti, A. Oxidative DNA Damage in the Human Trabecular Meshwork: Clinical Correlation in Patients With Primary Open-Angle Glaucoma. Arch. Ophthalmol. 2005, 123, 458–463. [Google Scholar] [CrossRef] [Green Version]
- Izuta, H.; Matsunaga, N.; Shimazawa, M.; Sugiyama, T.; Ikeda, T.; Hara, H. Proliferative diabetic retinopathy and relations among antioxidant activity, oxidative stress, and VEGF in the vitreous body. Mol. Vis. 2010, 16, 130–136. [Google Scholar]
- Dizdaroglu, M.; Jaruga, P.; Birincioglu, M.; Rodriguez, H. Free radical-induced damage to DNA: Mechanisms and measurement. Free Radic. Biol. Med. 2002, 32, 1102–1115. [Google Scholar] [CrossRef]
- Cadet, J.; Douki, T.; Gasparutto, D.; Ravanat, J.L. Oxidative damage to DNA: Formation, measurement and biochemical features. Mutat. Res. Fundam. Mol. Mech. Mutagenesis 2003, 531, 5–23. [Google Scholar] [CrossRef]
- Cadet, J.; Wagner, J.R. Oxidatively generated base damage to cellular DNA by hydroxyl radical and one-electron oxidants: Similarities and differences. Arch. Biochem. Biophys. 2014, 557, 47–54. [Google Scholar] [CrossRef]
- Thiagarajan, R.; Manikandan, R. Antioxidants and cataract. Free Radic. Res. 2013, 47, 337–345. [Google Scholar] [CrossRef]
- Jia, Y.; Hu, D.-N.; Zhu, D.; Zhang, L.; Gu, P.; Fan, X.; Zhou, J.-B. MMP-2, MMP-3, TIMP-1, TIMP-2, and TIMP-3 Protein Levels in Human Aqueous Humor: Relationship With Axial Length. Investig. Ophthalmol. Vis. Sci. 2014, 55, 3922–3928. [Google Scholar] [CrossRef] [Green Version]
- Saxena, S.; Srivastava, P.; Khanna, V.K. Elevated lipid peroxides induced angiogenesis in proliferative diabetic retinopathy. J. Ocul. Biol. Dis. Inform. 2010, 3, 85–87. [Google Scholar] [CrossRef] [Green Version]
- Kersten, E.; Paun, C.C.; Schellevis, R.L.; Hoyng, C.B.; Delcourt, C.; Lengyel, I.; Peto, T.; Ueffing, M.; Klaver, C.C.W.; Dammeier, S.; et al. Systemic and ocular fluid compounds as potential biomarkers in age-related macular degeneration. Surv. Ophthalmol. 2018, 63, 9–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, J.; Nelson, K.C.; Wu, M.; Sternberg, P.; Jones, D.P. Oxidative damage and protection of the RPE. Prog. Retin. Eye Res. 2000, 19, 205–221. [Google Scholar] [CrossRef]
- Bergandi, L.; Skorokhod, O.A.; Franzone, F.; La Grotta, R.; Schwarzer, E.; Nuzzi, R. Induction of oxidative stress in human aqueous and vitreous humors by Nd:YAG laser posterior capsulotomy. Int. J. Ophthalmol. 2018, 11, 1145–1151. [Google Scholar] [CrossRef]
- Ethen, C.M.; Reilly, C.; Feng, X.; Olsen, T.W.; Ferrington, D.A. Age-Related Macular Degeneration and Retinal Protein Modification by 4-Hydroxy-2-nonenal. Investig. Ophthalmol. Vis. Sci. 2007, 48, 3469–3479. [Google Scholar] [CrossRef] [Green Version]
- Cipak, A.; Mrakovcic, L.; Ciz, M.; Lojek, A.; Mihaylova, B.; Goshev, I.; Jaganjac, M.; Cindric, M.; Sitic, S.; Margaritoni, M.; et al. Growth suppression of human breast carcinoma stem cells by lipid peroxidation product 4-hydroxy-2-nonenal and hydroxyl radicalmodified collagen. Acta Biochim. Pol. 2010, 57, 165–171. [Google Scholar] [CrossRef]
- Bishop, P.N.; Holmes, D.F.; Kadler, K.E.; McLeod, D.; Bos, K.J. Age-Related Changes on the Surface of Vitreous Collagen Fibrils. Investig. Ophthalmol. Vis. Sci. 2004, 45, 1041–1046. [Google Scholar] [CrossRef] [Green Version]
- Kamegawa, M.; Nakanishi-Ueda, T.; Iwai, S.; Ueda, T.; Kosuge, S.; Ogura, H.; Sasuga, K.; Inagaki, M.; Watanabe, M.; Oguchi, K.; et al. Effect of Lipid-Hydroperoxide-Induced Oxidative Stress on Vitamin E, Ascorbate and Glutathione in the Rabbit Retina. Ophthalmic Res. 2007, 39, 49–54. [Google Scholar] [CrossRef]
- Madsen-Bouterse, S.A.; Kowluru, R.A. Oxidative stress and diabetic retinopathy: Pathophysiological mechanisms and treatment perspectives. Rev. Endocr. Metab. Disord. 2008, 9, 315–327. [Google Scholar] [CrossRef]
- Smith, K.A.; Shepherd, J.; Wakil, A.; Kilpatrick, E.S. A comparison of methods for the measurement of 8-isoPGF2α: A marker of oxidative stress. Ann. Clin. Biochem. 2011, 48, 147–154. [Google Scholar] [CrossRef]
- Evereklioglu, C.; Er, H.; Doganay, S.; Cekmen, M.; Turkoz, Y.; Otlu, B.; Ozerol, E. Nitric oxide and lipid peroxidation are increased and associated with decreased antioxidant enzyme activities in patients with age-related macular degeneration. Doc. Ophthalmol. 2003, 106, 129–136. [Google Scholar] [CrossRef]
- Bhutto, I.A.; Baba, T.; Merges, C.; McLeod, D.S.; Lutty, G.A. Low nitric oxide synthases (NOSs) in eyes with age-related macular degeneration (AMD). Exp. Eye Res. 2010, 90, 155–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakravarthy, U.; Stitt, A.W.; McNally, J.; Bailie, J.R.; Hoey, E.M.; Duprex, P.; Duprex, W.P. Nitric oxide synthase activity and expression in retinal capillary endothelial cells and pericytes. Curr. Eye Res. 1995, 14, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Becquet, F.; Courtois, Y.; Goureau, O. Nitric oxide in the eye: Multifaceted roles and diverse outcomes. Surv. Ophthalmol. 1997, 42, 71–82. [Google Scholar] [CrossRef]
- Hattenbach, L.-O.; Allers, A.; Klais, C.; Koch, F.; Hecker, M. L-Arginine–Nitric Oxide Pathway–Related Metabolites in the Aqueous Humor of Diabetic Patients. Investig. Ophthalmol. Vis. Sci. 2000, 41, 213–217. [Google Scholar]
- Wiederholt, M.; Sturm, A.; Lepple-Wienhues, A. Relaxation of trabecular meshwork and ciliary muscle by release of nitric oxide. Investig. Ophthalmol. Vis. Sci. 1994, 35, 2515–2520. [Google Scholar]
- Bhattacharya, S.K.; Lee, R.K.; Grus, F.H. Molecular biomarkers in glaucoma. Investig. Ophthalmol. Vis. Sci. 2013, 54, 121–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinazo-Durán, M.D.; Gallego-Pinazo, R.; García-Medina, J.J.; Zanon-Moreno, V.C.; Nucci, C.; Dolz-Marco, R.; Martinez-Castillo, S.; Galbis-Estrada, C.; Marco-Ramírez, C.; López-Gálvez, M.I.; et al. Oxidative stress and its downstream signaling in aging eyes. Clin. Interv. Aging 2014, 9, 637–652. [Google Scholar] [CrossRef] [Green Version]
- Diederen, R.M.; La Heij, E.C.; Deutz, N.; Kessels, A.G.H.; Van Eijk, H.M.H.; Hendrikse, F. Increased nitric oxide (NO) pathway metabolites in the vitreous fluid of patients with rhegmatogenous retinal detachment or diabetic traction retinal detachment. Graefe Arch. Clin. Exp. Ophthalmol. 2006, 244, 683–688. [Google Scholar] [CrossRef] [PubMed]
- und Hohenstein-Blaul NV, T.; Funke, S.; Grus, F.H. Tears as a source of biomarkers for ocular and systemic diseases. Exp. Eye Res. 2013, 117, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Amil-Bangsa, N.H.; Mohd-Ali, B.; Ishak, B.; Abdul-Aziz, C.N.N.; Ngah, N.F.; Hashim, H.; Ghazali, A.R. Total Protein Concentration and Tumor Necrosis Factor α in Tears of Nonproliferative Diabetic Retinopathy. Optom. Vis. Sci. 2019, 96, 934–939. [Google Scholar] [CrossRef]
- Aveleira, C.A.; Lin, C.-M.; Abcouwer, S.F.; Ambrósio, A.F.; Antonetti, D.A. TNF-α signals through PKCζ/NF-κB to alter the tight junction complex and increase retinal endothelial cell permeability. Diabetes 2010, 59, 2872–2882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arita, R.; Nakao, S.; Kita, T.; Kawahara, S.; Asato, R.; Yoshida, S.; Enaida, H.; Hafezi-Moghadam, A.; Ishibashi, T. A Key Role for ROCK in TNF-α–Mediated Diabetic Microvascular Damage. Investig. Ophthalmol. Vis. Sci. 2013, 54, 2373–2383. [Google Scholar] [CrossRef] [PubMed]
- Adamiec-Mroczek, J.; Oficjalska-Młyńczak, J. Assessment of selected adhesion molecule and proinflammatory cytokine levels in the vitreous body of patients with type 2 diabetes—Role of the inflammatory-immune process in the pathogenesis of proliferative diabetic retinopathy. Graefe Arch. Clin. Exp. Ophthalmol. Albrecht Graefes Arch. Klin. Exp. Ophthalmol. 2008, 246, 1665–1670. [Google Scholar] [CrossRef] [PubMed]
- McArthur, K.; Feng, B.; Wu, Y.; Chen, S.; Chakrabarti, S. MicroRNA-200b regulates vascular endothelial growth factor-mediated alterations in diabetic retinopathy. Diabetes 2011, 60, 1314–1323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomaa, A.R.; Elsayed, E.T.; Moftah, R.F. MicroRNA-200b Expression in the Vitreous Humor of Patients with Proliferative Diabetic Retinopathy. Ophthalmic Res. 2017, 58, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Qiu, F.; Zhou, K.; Matlock, H.G.; Takahashi, Y.; Rajala, R.V.; Yang, Y.; Moran, E.; Ma, J.-X. Pathogenic Role of microRNA-21 in Diabetic Retinopathy Through Downregulation of PPARα. Diabetes 2017, 66, 1671–1682. [Google Scholar] [CrossRef] [Green Version]
- Usui-Ouchi, A.; Ouchi, Y.; Kiyokawa, M.; Sakuma, T.; Ito, R.; Ebihara, N. Upregulation of Mir-21 Levels in the Vitreous Humor Is Associated with Development of Proliferative Vitreoretinal Disease. PLoS ONE 2016, 11, e0158043. [Google Scholar] [CrossRef] [Green Version]
- Besnier, M.; Shantikumar, S.; Anwar, M.; Dixit, P.; Chamorro-Jorganes, A.; Sweaad, W.; Sala-Newby, G.; Madeddu, P.; Thomas, A.C.; Howard, L.; et al. miR-15a/-16 Inhibit Angiogenesis by Targeting the Tie2 Coding Sequence: Therapeutic Potential of a miR-15a/16 Decoy System in Limb Ischemia. Molecular therapy. Nucleic Acids 2019, 17, 49–62. [Google Scholar] [CrossRef] [Green Version]
- Hirota, K.; Keino, H.; Inoue, M.; Ishida, H.; Hirakata, A. Comparisons of microRNA expression profiles in vitreous humor between eyes with macular hole and eyes with proliferative diabetic retinopathy. Graefe Arch. Clin. Exp. Ophthalmol. 2015, 253, 335–342. [Google Scholar] [CrossRef]
- Ye, E.-A.; Steinle, J.J. Regulatory role of microRNA on inflammatory responses of diabetic retinopathy. Neural Regen. Res. 2017, 12, 580–581. [Google Scholar] [CrossRef]
- Cho, H.; Hwang, M.; Hong, E.H.; Yu, H.; Park, H.; Koh, S.; Shin, Y.U. Micro-RNAs in the aqueous humour of patients with diabetic macular oedema. Clin. Exp. Ophthalmol. 2020, 48, 624–635. [Google Scholar] [CrossRef] [PubMed]
- Feng, B.; Chakrabarti, S. miR-320 Regulates Glucose-Induced Gene Expression in Diabetes. Isrn Endocrinol. 2012, 2012, 549875. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zampetaki, A.; Willeit, P.; Burr, S.; Yin, X.; Langley, S.R.; Kiechl, S.; Klein, R.; Rossing, P.; Chaturvedi, N.; Mayr, M. Angiogenic microRNAs Linked to Incidence and Progression of Diabetic Retinopathy in Type 1. Diabetes 2016, 65, 216–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Zhou, J.; Li, M.-Q.; Xu, J.; Zhang, J.-P.; Jin, L.-P. MicroRNA-184 promotes apoptosis of trophoblast cells via targeting WIG1 and induces early spontaneous abortion. Cell Death Dis. 2019, 10, 223. [Google Scholar] [CrossRef]
- Chen, S.; Yuan, M.; Liu, Y.; Zhao, X.; Lian, P.; Chen, Y.; Liu, B.; Lu, L. Landscape of microRNA in the aqueous humour of proliferative diabetic retinopathy as assessed by next-generation sequencing. Clin. Exp. Ophthalmol. 2019, 47, 925–936. [Google Scholar] [CrossRef]
- Fang, L.; Deng, Z.; Shatseva, T.; Yang, J.; Peng, C.; Du, W.W.; Yee, A.J.; Ang, L.C.; He, C.; Shan, S.W.; et al. MicroRNA miR-93 promotes tumor growth and angiogenesis by targeting integrin-β8. Oncogene 2011, 30, 806–821. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.-Q.; Cui, H.; Wang, L.; Fang, X.; Su, S. Role of microRNA-29a in the development of diabetic retinopathy by targeting AGT gene in a rat model. Exp. Mol. Pathol. 2017, 102, 296–302. [Google Scholar] [CrossRef]
- Qu, Y.; Liu, H.; Lv, X.; Liu, Y.; Wang, X.; Zhang, M.; Zhang, X.; Li, Y.; Lou, Q.; Li, S.; et al. MicroRNA-16-5p overexpression suppresses proliferation and invasion as well as triggers apoptosis by targeting VEGFA expression in breast carcinoma. Oncotarget 2017, 8, 72400–72410. [Google Scholar] [CrossRef]
- Luo, Z.; Feng, X.; Wang, H.; Xu, W.; Zhao, Y.; Ma, W.; Jiang, S.; Liu, D.; Huang, J.; Songyang, Z. Mir-23a induces telomere dysfunction and cellular senescence by inhibiting TRF2 expression. Aging Cell 2015, 14, 391–399. [Google Scholar] [CrossRef]
- Su, J.L.; Chen, P.S.; Johansson, G.; Kuo, M.L. Function and regulation of let-7 family microRNAs. Microrna 2012, 1, 34–39. [Google Scholar] [CrossRef]
- Li, T.; Pan, H.; Li, R. The dual regulatory role of miR-204 in cancer. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2016, 37, 11667. [Google Scholar] [CrossRef] [Green Version]
- Jackson, D.C.; Zeng, W.; Wong, C.Y.; Mifsud, E.J.; Williamson, N.A.; Ang, C.-S.; Vingrys, A.J.; Downie, L.E. Tear Interferon-Gamma as a Biomarker for Evaporative Dry Eye Disease. Investig. Ophthalmol. Vis. Sci. 2016, 57, 4824–4830. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Chen, X.; Qin, G.; Xie, H.; Lv, P. Tear film function in type 2 diabetic patients with retinopathy. Ophthalmologica 2008, 222, 284–291. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, Y.; Ru, Y.-S.; Wang, X.-W.; Yang, J.-Z.; Li, C.-H.; Wang, H.-X.; Li, X.-R.; Li, B. Ocular surface changes in type II diabetic patients with proliferative diabetic retinopathy. Int. J. Ophthalmol. 2015, 8, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Csősz, É.; Boross, P.; Csutak, A.; Berta, A.; Toth, F.; Poliska, S.; Török, Z.; Tőzsér, J. Quantitative analysis of proteins in the tear fluid of patients with diabetic retinopathy. J. Proteom. 2012, 75, 2196–2204. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A.; Kastin, A.J.; Gutierrez, E.G. Penetration of interleukin-6 across the murine blood-brain barrier. Neurosci. Lett. 1994, 179, 53–56. [Google Scholar] [CrossRef]
- Moriarty, A.; Spalton, D.; Moriarty, B.; Shilling, J.; Ffytche, T.; Bulsara, M. Studies of the Blood-Aqueous Barrier in Diabetes Mellitus. Am. J. Ophthalmol. 1994, 117, 768–771. [Google Scholar] [CrossRef]
- Holzinger, C.; Weissinger, E.; Zuckermann, A.; Imhof, M.; Kink, F.; Schöllhammer, A.; Kopp, C.; Wolner, E. Effects of interleukin-1, -2, -4, -6, interferon-gamma and granulocyte/macrophage colony stimulating factor on human vascular endothelial cells. Immunol. Lett. 1993, 35, 109–117. [Google Scholar] [CrossRef]
- Sharma, R.K.; Rogojina, A.T.; Chalam, K.V. Multiplex immunoassay analysis of biomarkers in clinically accessible quantities of human aqueous humor. Mol. Vis. 2009, 15, 60–69. [Google Scholar]
- Globočnik Petrovič, M.; Korošec, P.; Košnik, M.; Hawlina, M. Vitreous Levels of Interleukin-8 in Patients With Proliferative Diabetic Retinopathy. Am. J. Ophthalmol. 2007, 143, 175–176. [Google Scholar] [CrossRef]
- Chono, I.; Miyazaki, D.; Miyake, H.; Komatsu, N.; Ehara, F.; Nagase, D.; Kawamoto, Y.; Shimizu, Y.; Ideta, R.; Inoue, Y. High interleukin-8 level in aqueous humor is associated with poor prognosis in eyes with open angle glaucoma and neovascular glaucoma. Sci. Rep. 2018, 8, 14533. [Google Scholar] [CrossRef] [PubMed]
- Paine, S.K.; Sen, A.; Choudhuri, S.; Mondal, L.K.; Chowdhury, I.H.; Basu, A.; Mukherjee, A.; Bhattacharya, B. Association of tumor necrosis factor α, interleukin 6, and interleukin 10 promoter polymorphism with proliferative diabetic retinopathy in type 2 diabetic subjects. Retina 2012, 32. [Google Scholar] [CrossRef] [PubMed]
- Hernández, C.; Segura, R.M.; Fonollosa, A.; Carrasco, E.; Francisco, G.; Simo, R. Interleukin-8, monocyte chemoattractant protein-1 and IL-10 in the vitreous fluid of patients with proliferative diabetic retinopathy. Diabet. Med. 2005, 22, 719–722. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Perez, J.M.; Morillas-Ruiz, J.M. A review: Inflammatory process in Alzheimer’s disease, role of cytokines. Sci. World J. 2012, 2012, 756357. [Google Scholar] [CrossRef]
- Bigda, J.; Beletsky, I.; Brakebusch, C.; Varfolomeev, Y.; Engelmann, H.; Holtmann, H.; Wallach, D. Dual role of the p75 tumor necrosis factor (TNF) receptor in TNF cytotoxicity. J. Exp. Med. 1994, 180, 445–460. [Google Scholar] [CrossRef]
- Ozturk, B.T.; Bozkurt, B.; Kerimoglu, H.; Okka, M.; Kamis, U.; Gunduz, K. Effect of serum cytokines and VEGF levels on diabetic retinopathy and macular thickness. Mol. Vis. 2009, 15, 1906–1914. [Google Scholar]
- Zorena, K.; Raczyńska, D.; Raczyńska, K. Biomarkers in Diabetic Retinopathy and the Therapeutic Implications. Mediat. Inflamm. 2013, 2013, 193604. [Google Scholar] [CrossRef] [Green Version]
- Ferrara, N.; Gerber, H.-P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef]
- Vignali, D.A.A. Multiplexed particle-based flow cytometric assays. J. Immunol. Methods 2000, 243, 243–255. [Google Scholar] [CrossRef]
- Murugeswari, P.; Shukla, D.; Rajendran, A.; Kim, R.; Namperumalsamy, P.; Muthukkaruppan, V. Proinflammatory cytokines and angiogenic and anti-angiogenic factors in vitreous of patients with proliferative diabetic retinopathy and eales’ disease. RETINA 2008, 28. [Google Scholar] [CrossRef]
- Yoshimura, T.; Sonoda, K.-H.; Sugahara, M.; Mochizuki, Y.; Enaida, H.; Oshima, Y.; Ueno, A.; Hata, Y.; Yoshida, H.; Ishibashi, T. Comprehensive Analysis of Inflammatory Immune Mediators in Vitreoretinal Diseases. PLoS ONE 2009, 4, e8158. [Google Scholar] [CrossRef] [PubMed]
- Mocan, M.C.; Kadayifcilar, S.; Eldem, B. Elevated intravitreal interleukin-6 levels in patients with proliferative diabetic retinopathy. Can. J. Ophthalmol. 2006, 41, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Garbutcheon-Singh, K.B.; Carnt, N.; Pattamatta, U.; Samarawickrama, C.; White, A.; Calder, V. A Review of the Cytokine IL-17 in Ocular Surface and Corneal Disease. Curr. Eye Res. 2019, 44, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Zhou, Y. Interleukin-17: The Role for Pathological Angiogenesis in Ocular Neovascular Diseases. Tohoku J. Exp. Med. 2019, 247, 87–98. [Google Scholar] [CrossRef] [Green Version]
- Feenstra, D.J.; Yego, E.C.; Mohr, S. Modes of Retinal Cell Death in Diabetic Retinopathy. J. Clin. Exp. Ophthalmol. 2013, 4, 298. [Google Scholar] [CrossRef] [Green Version]
- Devi, T.S.; Hosoya, K.-I.; Terasaki, T.; Singh, L.P. Critical role of TXNIP in oxidative stress, DNA damage and retinal pericyte apoptosis under high glucose: Implications for diabetic retinopathy. Exp. Cell Res. 2013, 319, 1001–1012. [Google Scholar] [CrossRef] [Green Version]
- Dartt, D.A. Tear lipocalin: Structure and function. Ocul. Surf. 2011, 9, 126–138. [Google Scholar] [CrossRef] [Green Version]
- Yusifov, T.N.; Abduragimov, A.R.; Narsinh, K.; Gasymov, O.K.; Glasgow, B.J. Tear lipocalin is the major endonuclease in tears. Mol. Vis. 2008, 14, 180–188. [Google Scholar]
- Zhao, H.; He, Y.; Ren, Y.-R.; Chen, B.-H. Corneal alteration and pathogenesis in diabetes mellitus. Int. J. Ophthalmol. 2019, 12, 1939–1950. [Google Scholar] [CrossRef]
- Kim, J.; Kim, C.-S.; Sohn, E.; Jeong, I.-H.; Kim, H.; Kim, J.S. Involvement of advanced glycation end products, oxidative stress and nuclear factor-kappaB in the development of diabetic keratopathy. Graefe Arch. Clin. Exp. Ophthalmol. 2011, 4, 529–536. [Google Scholar] [CrossRef]
- Hrabec, E.; Naduk, J.; Strek, M.; Hrabec, Z. Type IV collagenases (MMP-2 and MMP-9) and their substrates--intracellular proteins, hormones, cytokines, chemokines and their receptors. Postepy Biochem. 2007, 53, 37–45. [Google Scholar] [PubMed]
- Notari, L.; Miller, A.; Martínez, A.; Amaral, J.; Ju, M.; Robinson, G.; Smith, L.E.H.; Becerra, S.P. Pigment Epithelium–Derived Factor Is a Substrate for Matrix Metalloproteinase Type 2 and Type 9: Implications for Downregulation in Hypoxia. Investig. Ophthalmol. Vis. Sci. 2005, 46, 2736–2747. [Google Scholar] [CrossRef] [Green Version]
- Giebel, S.J.; Menicucci, G.; McGuire, P.G.; Das, A. Matrix metalloproteinases in early diabetic retinopathy and their role in alteration of the blood–retinal barrier. Lab. Investig. 2005, 85, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Zucker, S.; Mirza, H.; Conner, C.E.; Lorenz, A.F.; Drews, M.D.; Bahou, W.F.; Jesty, J. Vascular endothelial groth factor induces tissue factor and matrix metalloproteinase production in endothelial cells: Conversion of prothrombin to thrombin results in progelatininase a activation and cell proliferation. Int. J. Cancer 1998, 75, 780–786. [Google Scholar] [CrossRef]
- Descamps, F.J.; Martens, E.; Kangave, D.; Struyf, S.; Geboes, K.; Van Damme, J.; Opdenakker, G.; Abu El-Asrar, A.M. The activated form of gelatinase B/matrix metalloproteinase-9 is associated with diabetic vitreous hemorrhage. Exp. Eye Res. 2006, 83, 401–407. [Google Scholar] [CrossRef]
- Noda, K.; Ishida, S.; Inoue, M.; Obata, K.-I.; Oguchi, Y.; Okada, Y.; Ikeda, E. Production and Activation of Matrix Metalloproteinase-2 in Proliferative Diabetic Retinopathy. Investig. Ophthalmol. Vis. Sci. 2003, 44, 2163–2170. [Google Scholar] [CrossRef] [Green Version]
- Ow, Y.-L.P.; Green, D.R.; Hao, Z.; Mak, T.W. Cytochrome c: Functions beyond respiration. Nat. Rev. Mol. Cell Biol. 2008, 9, 532–542. [Google Scholar] [CrossRef]
- Chertkova, R.V.; Brazhe, N.A.; Bryantseva, T.V.; Nekrasov, A.N.; Dolgikh, D.A.; Yusipovich, A.; Sosnovtseva, O.V.; Maksimov, G.V.; Rubin, A.B.; Kirpichnikov, M.P. New insight into the mechanism of mitochondrial cytochrome c function. PLoS ONE 2017, 12, e0178280. [Google Scholar] [CrossRef]
- Mizutani, M.; Kern, T.S.; Lorenzi, M. Accelerated death of retinal microvascular cells in human and experimental diabetic retinopathy. J. Clin. Investig. 1996, 97, 2883–2890. [Google Scholar] [CrossRef]
- Mohr, S.; Xi, X.; Tang, J.; Kern, T.S. Caspase Activation in Retinas of Diabetic and Galactosemic Mice and Diabetic Patients. Diabetes 2002, 51, 1172. [Google Scholar] [CrossRef] [Green Version]
- Chapple, C.E.; Robisson, B.; Spinelli, L.; Guien, C.; Becker, E.; Brun, C. Extreme multifunctional proteins identified from a human protein interaction network. Nat. Commun. 2015, 6, 7412. [Google Scholar] [CrossRef]
- Santucci, R.; Sinibaldi, F.; Cozza, P.; Polticelli, F.; Fiorucci, L. Cytochrome c: An extreme multifunctional protein with a key role in cell fate. Int. J. Biol. Macromol. 2019, 136, 1237–1246. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Tang, J.; Kern, T.S. Abnormalities of Retinal Metabolism in Diabetes and Experimental Galactosemia. Diabetes 2001, 50, 1938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKinnon, S.J.; Lehman, D.M.; Kerrigan-Baumrind, L.A.; Merges, C.A.; Pease, M.E.; Kerrigan, D.F.; Ransom, N.L.; Tahzib, N.G.; A Reitsamer, H.; Levkovitch-Verbin, H.; et al. Caspase Activation and Amyloid Precursor Protein Cleavage in Rat Ocular Hypertension. Investig. Ophthalmol. Vis. Sci. 2002, 43, 1077–1087. [Google Scholar]
- Tang, J.; Kern, T.S. Inflammation in diabetic retinopathy. Prog. Retin. Eye Res. 2011, 30, 343–358. [Google Scholar] [CrossRef] [Green Version]
- Jiang, N.; Chen, X.-L.; Yang, H.-W.; Ma, Y.-R. Effects of nuclear factor κB expression on retinal neovascularization and apoptosis in a diabetic retinopathy rat model. Int. J. Ophthalmol. 2015, 8, 448–452. [Google Scholar] [CrossRef]
- Hammes, H.-P.; Lin, J.; Renner, O.; Shani, M.; Lundqvist, A.; Betsholtz, C.; Brownlee, M.; Deutsch, U. Pericytes and the Pathogenesis of Diabetic Retinopathy. Diabetes 2002, 51, 3107. [Google Scholar] [CrossRef] [Green Version]
- Busik, J.V.; Mohr, S.; Grant, M.B. Hyperglycemia-induced reactive oxygen species toxicity to endothelial cells is dependent on paracrine mediators. Diabetes 2008, 57, 1952–1965. [Google Scholar] [CrossRef] [Green Version]
- Kowluru, R.A.; Koppolu, P.; Chakrabarti, S.; Chen, S. Diabetes-induced Activation of Nuclear Transcriptional Factor in the Retina, and its Inhibition by Antioxidants. Free Radic. Res. 2003, 37, 1169–1180. [Google Scholar] [CrossRef]
- Yuuki, T.; Kanda, T.; Kimura, Y.; Kotajima, N.; Tamura, J.; Kobayashi, I.; Kishi, S. Inflammatory cytokines in vitreous fluid and serum of patients with diabetic vitreoretinopathy. J. Diabetes Complicat. 2001, 15, 257–259. [Google Scholar] [CrossRef]
- Kowluru, R.A.; Shan, Y.; Mishra, M. Dynamic DNA methylation of matrix metalloproteinase-9 in the development of diabetic retinopathy. Lab. Investig. J. Technol. Methods Pathol. 2016, 96, 1040–1049. [Google Scholar] [CrossRef] [Green Version]
- Coco, C.; Sgarra, L.; Potenza, M.A.; Nacci, C.; Ms, B.P.; Barbano, R.; Parrella, P.; Montagnani, M. Can Epigenetics of Endothelial Dysfunction Represent the Key to Precision Medicine in Type 2 Diabetes Mellitus? Int. J. Mol. Sci. 2019, 20, 2949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanda, A.; Noda, K.; Saito, W.; Ishida, S. Vitreous renin activity correlates with vascular endothelial growth factor in proliferative diabetic retinopathy. Br. J. Ophthalmol. 2013, 97, 666–668. [Google Scholar] [CrossRef] [PubMed]
- Maghbooli, Z.; Hossein-Nezhad, A.; Larijani, B.; Amini, M.; Keshtkar, A. Global DNA methylation as a possible biomarker for diabetic retinopathy. Diabetes Metab. Res. Rev. 2015, 31, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Q.; Kowluru, R.A. Regulation of matrix metalloproteinase-9 by epigenetic modifications and the development of diabetic retinopathy. Diabetes Care 2013, 62, 2559–2568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tewari, S.; Zhong, Q.; Santos, J.M.; Kowluru, A. Mitochondria DNA replication and DNA methylation in the metabolic memory associated with continued progression of diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2012, 53, 4881–4888. [Google Scholar] [CrossRef]
- Mishra, M.; Kowluru, R.A. The Role of DNA Methylation in the Metabolic Memory Phenomenon Associated With the Continued Progression of Diabetic Retinopathy. Investig. Ophthalmol. Vis. Sci. 2016, 57, 5748–5757. [Google Scholar] [CrossRef] [Green Version]
- Shafabakhsh, R.; Aghadavod, E.; Ghayour-Mobarhan, M.; Ferns, G.; Asemi, Z. Role of histone modification and DNA methylation in signaling pathways involved in diabetic retinopathy. J. Cell. Physiol. 2019, 234, 7839–7846. [Google Scholar] [CrossRef]
- Mishra, M.; Kowluru, R.A. DNA Methylation—A Potential Source of Mitochondria DNA Base Mismatch in the Development of Diabetic Retinopathy. Mol. Neurobiol. 2019, 56, 88–101. [Google Scholar] [CrossRef]
- Mohammed, S.A.; Ambrosini, S.; Lüscher, T.; Paneni, F.; Costantino, S. Epigenetic Control of Mitochondrial Function in the Vasculature. Front. Cardiovasc. Med. 2020, 7, 28. [Google Scholar] [CrossRef] [Green Version]
- Iacobazzi, V.; Castegna, A.; Infantino, V.; Andria, G. Mitochondrial DNA methylation as a next-generation biomarker and diagnostic tool. Mol. Genet. Metab. 2013, 110, 25–34. [Google Scholar] [CrossRef]
- Lanza, M.; Benincasa, G.; Costa, D.; Napoli, C. Clinical Role of Epigenetics and Network Analysis in Eye Diseases: A Translational Science Review. J. Ophthalmol. 2019, 2019, 2424956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duraisamy, A.J.; Mishra, M.; Kowluru, A.; Kowluru, A. Epigenetics and Regulation of Oxidative Stress in Diabetic Retinopathy. Investig. Ophthalmol. Vis. Sci. 2018, 59, 4831–4840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duraisamy, A.J.; Radhakrishnan, R.; Seyoum, B.; Abrams, G.W.; Kowluru, A. Epigenetic Modifications in Peripheral Blood as Potential Noninvasive Biomarker of Diabetic Retinopathy. Transl. Vis. Sci. Technol. 2019, 8, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, C.; Wu, Y.; Liu, G.; Liu, X.; Wang, F.; Yu, J. Relationship between homocysteine level and diabetic retinopathy: A systematic review and meta-analysis. Diagn. Pathol. 2014, 9, 167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tawfik, A.; Mohamed, R.; Elsherbiny, N.M.; DeAngelis, M.M.; Bartoli, M.; Al-Shabrawey, M. Homocysteine: A Potential Biomarker for Diabetic Retinopathy. J. Clin. Med. 2019, 8, 121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elsherbiny, N.M.; Sharma, I.; Kira, D.; Alhusban, S.; Samra, Y.A.; Jadeja, R.; Martin, P.; Al-Shabrawey, M.; Tawfik, A. Homocysteine Induces Inflammation in Retina and Brain. Biomolecules 2020, 10, 393. [Google Scholar] [CrossRef] [Green Version]
- Kowluru, R.A.; Mishra, M. Therapeutic targets for altering mitochondrial dysfunction associated with diabetic retinopathy. Expert Opin. Ther. Targets 2018, 22, 233–245. [Google Scholar] [CrossRef]
- Rodríguez, M.L.; Pérez, S.; Mena-Mollá, S.; Desco, M.C.; Ortega, Á.L. Oxidative Stress and Microvascular Alterations in Diabetic Retinopathy: Future Therapies. Oxid. Med. Cell. Longev. 2019, 2019, 4940825. [Google Scholar] [CrossRef] [Green Version]
- Sinclair, S.H.; Schwartz, S.S. Diabetic Retinopathy-An Underdiagnosed and Undertreated Inflammatory, Neuro-Vascular Complication of Diabetes. Front. Endocrinol. 2019, 10, 843. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.W.; Zhao, H.; Chen, B.H. Reactive oxygen species mediates a metabolic memory of high glucose stress signaling in bovine retinal pericytes. Int. J. Ophthalmol. 2019, 12, 1067–1074. [Google Scholar] [CrossRef]
- Kowluru, R.A. Mitochondrial Stability in Diabetic Retinopathy: Lessons Learned From Epigenetics. Diabetes 2019, 68, 241–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khullar, M.; Cheema, B.S.; Raut, S.K. Emerging Evidence of Epigenetic Modifications in Vascular Complication of Diabetes. Front. Endocrinol. 2017, 8, 237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Bao, S.; Lai, D.; Rapkins, R.W.; Gillies, M.C. Intravitreal triamcinolone acetonide inhibits breakdown of the blood-retinal barrier through differential regulation of VEGF-A and its receptors in early diabetic rat retinas. Diabetes 2008, 57, 1026–1033. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Zhao, L.; Hambly, B.D.; Bao, S.; Wang, K. Diabetic retinopathy: Reversibility of epigenetic modifications and new therapeutic targets. Cell Biosci. 2017, 15, 42. [Google Scholar] [CrossRef] [Green Version]
- Kaspar, J.W.; Niture, S.K.; Jaiswal, A.K. Nrf2:INrf2 (Keap1) signaling in oxidative stress. Free Radic. Biol. Med. 2009, 47, 1304–1309. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Q.; Kowluru, R.A. Epigenetic modification of Sod2 in the development of diabetic retinopathy and in the metabolic memory: Role of histone methylation. Investig. Ophthalmol. Vis. Sci. 2013, 54, 244–250. [Google Scholar] [CrossRef] [Green Version]
- Santos, J.M.; Tewari, S.; Goldberg, A.F.X.; Kowluru, A. Mitochondrial biogenesis and the development of diabetic retinopathy. Free Radic Biol Med. 2011, 51, 1849–1860. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.W.; Xia, H.; Han, Q.; Chen, B. Effects of antioxidant gene therapy on the development of diabetic retinopathy and the metabolic memory phenomenon. Graefe Arch. Clin. Exp. Ophthalmol. 2015, 253, 249–259. [Google Scholar] [CrossRef]
- Sundar, I.K.; Yao, H.; Rahman, I. Oxidative stress and chromatin remodeling in chronic obstructive pulmonary disease and smoking-related diseases. Antioxid. Redox Signal. 2013, 18, 1956–1971. [Google Scholar] [CrossRef]
- Aydin, Ö.Z.; Vermeulen, W.; Lans, H. ISWI chromatin remodeling complexes in the DNA damage response. Cell Cycle 2014, 13, 3016–3025. [Google Scholar] [CrossRef] [Green Version]
- Matilainen, O.; Sleiman, M.S.B.; Quirós, P.M.; Garcia, S.M.D.A.; Auwerx, J. The chromatin remodeling factor ISW-1 integrates organismal responses against nuclear and mitochondrial stress. Nat. Commun. 2017, 8, 1818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perrone, L.; Devi, T.S.; Hosoya, K.-I.; Terasaki, T.; Singh, L.P. Inhibition of TXNIP expression in vivo blocks early pathologies of diabetic retinopathy. Cell Death Dis. 2010, 1, e65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reddy, M.A.; Zhang, E.; Natarajan, R. Epigenetic mechanisms in diabetic complications and metabolic memory. Diabetologia 2015, 58, 443–455. [Google Scholar] [CrossRef] [Green Version]
- Kreuz, S.; Fischle, W. Oxidative stress signaling to chromatin in health and disease. Epigenomics 2016, 8, 843–862. [Google Scholar] [CrossRef] [Green Version]
- Moodie, F.M.; Marwick, J.A.; Anderson, C.S.; Szulakowski, P.; Biswas, S.K.; Bauter, M.R.; Kilty, I.; Rahman, I. Oxidative stress and cigarette smoke alter chromatin remodeling but differentially regulate NF-kappaB activation and proinflammatory cytokine release in alveolar epithelial cells. FASEB J. 2004, 18, 1897–1899. [Google Scholar] [CrossRef]
- Gozani, O.; Karuman, P.; Jones, D.R.; Ivanov, D.; Cha, J.; Lugovskoy, A.A.; Baird, C.L.; Zhu, H.; Field, S.J.; Lessnick, S.L.; et al. The PHD finger of the chromatin-associated protein ING2 functions as a nuclear phosphoinositide receptor. Cell 2003, 114, 99–111. [Google Scholar] [CrossRef]
- Bua, D.J.; Martin, G.M.; Binda, O.; Gozani, O. Nuclear phosphatidylinositol-5-phosphate regulates ING2 stability at discrete chromatin targets in response to DNA damage. Sci. Rep. 2013, 3, 2137. [Google Scholar] [CrossRef] [Green Version]
- Saeidi, L.; Ghaedi, H.; Sadatamini, M.; Vahabpour, R.; Rahimipour, A.; Shanaki, M.; Mansoori, Z.; Kazerouni, F. Long non-coding RNA LY86-AS1 and HCG27_201 expression in type 2 diabetes mellitus. Mol. Biol. Rep. 2018, 45, 2601–2608. [Google Scholar] [CrossRef]
- Carthew, R.W.; Sontheimer, E.J. Origins and Mechanisms of miRNAs and siRNAs. Cell 2009, 136, 642–655. [Google Scholar] [CrossRef] [Green Version]
- Xue, M.; Zhuo, Y.; Shan, B. MicroRNAs, Long Noncoding RNAs, and Their Functions in Human Disease. Methods Mol. Biol. 2017, 1617, 1–25. [Google Scholar] [CrossRef]
- Ha, T.-Y. MicroRNAs in Human Diseases: From Cancer to Cardiovascular Disease. Immune Netw. 2011, 11, 135–154. [Google Scholar] [CrossRef] [Green Version]
- Khalil, A.M.; Guttman, M.; Huarte, M.; Garber, M.; Raj, A.; Morales, D.R.; Thomas, K.; Presser, A.; Bernstein, B.E.; Van Oudenaarden, A.; et al. Many human large intergenic noncoding RNAs associate with chromatin-modifying complexes and affect gene expression. Proc. Natl. Acad. Sci. USA 2009, 106, 11667–11672. [Google Scholar] [CrossRef] [Green Version]
- Panchapakesan, U.; Pollock, C. Long non-coding RNAs-towards precision medicine in diabetic kidney disease? Clin. Sci. 2016, 130, 1599–1602. [Google Scholar] [CrossRef]
- Kovacs, B.; Lumayag, S.; Cowan, C.; Xu, S. MicroRNAs in early diabetic retinopathy in streptozotocin-induced diabetic rats. Investig. Ophthalmol. Vis. Sci. 2011, 52, 4402–4409. [Google Scholar] [CrossRef]
- Xu, W.; Li, F.; Liu, Z.; Xu, Z.; Sun, B.; Cao, J.; Liu, Y. MicroRNA-27b inhibition promotes Nrf2/ARE pathway activation and alleviates intracerebral hemorrhage-induced brain injury. Oncotarget 2017, 8, 70669–70684. [Google Scholar] [CrossRef] [Green Version]
- Veliceasa, D.; Biyashev, D.; Qin, G.; Misener, S.; Mackie, A.R.; Kishore, R.; Volpert, O.V. Therapeutic manipulation of angiogenesis with miR-27b. Vasc. Cell 2015, 7, 6. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.N.; Cao, N.; Li, X.; Qian, W.; Chen, X.-L. Serum microRNA-211 as a biomarker for diabetic retinopathy via modulating Sirtuin 1. Biochem. Biophys. Res. Commun. 2018, 505, 1236–1243. [Google Scholar] [CrossRef]
- Mishra, M.; Duraisamy, A.J.; Kowluru, R.A. Sirt1: A Guardian of the Development of Diabetic Retinopathy. Diabetes 2018, 67, 745–754. [Google Scholar] [CrossRef] [Green Version]
- Liang, Z.; Gao, K.P.; Wang, Y.X.; Liu, Z.C.; Tian, L.; Yang, X.Z.; Ding, J.; Wu, W.T.; Yang, W.H.; Li, Y.L.; et al. RNA sequencing identified specific circulating miRNA biomarkers for early detection of diabetes retinopathy. Am. J. Physiol. Endocrinol. Metab. 2018, 315, 374–385. [Google Scholar] [CrossRef]
- Desmettre, T. Epigenetics in age-related macular degeneration (AMD)—French translation of the article. J. Fr. Ophtalmol. 2018, 41, 981–990. [Google Scholar] [CrossRef]
- Leung, A.K.; Calabrese, J.M.; Sharp, P.A. Quantitative analysis of Argonaute protein reveals microRNA-dependent localization to stress granules. Proc. Natl. Acad. Sci. USA 2006, 103, 18125–18130. [Google Scholar] [CrossRef] [Green Version]
- Shen, J.; Xia, W.; Khotskaya, Y.B.; Huo, L.; Nakanishi, K.; Lim, S.-O.; Du, Y.; Wang, Y.; Chang, W.-C.; Chen, C.-H.; et al. EGFR modulates microRNA maturation in response to hypoxia through phosphorylation of AGO2. Nature 2013, 497, 383–387. [Google Scholar] [CrossRef]
- Qin, L.L.; An, M.-X.; Liu, Y.-L.; Xu, H.-C.; Lu, Z.-Q. MicroRNA-126: A promising novel biomarker in peripheral blood for diabetic retinopathy. Int. J. Ophthalmol. 2017, 10, 530–534. [Google Scholar] [CrossRef]
- Thomas, A.A.; Biswas, S.; Feng, B.; Chen, S.; Gonder, J.; Chakrabarti, S. lncRNA H19 prevents endothelial-mesenchymal transition in diabetic retinopathy. Diabetologia 2019, 62, 517–530. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Shi, E.; Yang, L.; Fu, W.; Hu, F.; Zhou, X. LncRNA AK077216 is downregulated in diabetic retinopathy and inhibited the apoptosis of retinal pigment epithelial cells by downregulating miR-383. Endocr. J. 2019, 66, 1011–1016. [Google Scholar] [CrossRef] [Green Version]
- Yin, L.; Sun, Z.; Ren, Q.; Su, X.; Zhang, D. Long Non-Coding RNA BANCR Is Overexpressed in Patients with Diabetic Retinopathy and Promotes Apoptosis of Retinal Pigment Epithelial Cells. Med. Sci. Monit. 2019, 25, 2845–2851. [Google Scholar] [CrossRef]
- Zhang, X.; Zou, X.; Li, Y.; Wang, Y. Downregulation of lncRNA BANCR participates in the development of retinopathy among diabetic patients. Exp. Ther. Med. 2019, 17, 4132–4138. [Google Scholar] [CrossRef] [Green Version]
- Perrone, L.; Matrone, C.; Singh, L.P. Epigenetic modifications and potential new treatment targets in diabetic retinopathy. J. Ophthalmol. 2014, 2014, 789120. [Google Scholar] [CrossRef] [Green Version]
- El-Osta, A. Redox mediating epigenetic changes confer metabolic memories. Circ. Res. 2012, 262–264. [Google Scholar] [CrossRef] [Green Version]
- Santos, J.M.; Kowluru, R.A. Role of mitochondria biogenesis in the metabolic memory associated with the continued progression of diabetic retinopathy and its regulation by lipoic acid. Investig. Ophthalmol. Vis. Sci. 2011, 52, 8791–8798. [Google Scholar] [CrossRef] [Green Version]
- Kowluru, R.A. Diabetic retinopathy, metabolic memory and epigenetic modifications. Vis. Res. 2017, 139, 30–38. [Google Scholar] [CrossRef] [PubMed]
| Profile | Biomarker | Matrix | Pathology | Levels | Method | References |
|---|---|---|---|---|---|---|
| Oxidative Stress | TAC | Aqueous | PDR | 0.55 ± 0.28 µmol Trolox/g * | Radical absorbance capacity assay | [104] |
| Vitreous | PDR | 0.19 ± 0.10 µmol Trolox/g * | Radical absorbance capacity assay | [104] | ||
| LPO | Vitreous | PDR | Male: 145.8 ± 6.3 µM * | Colorimetric assay | [105] | |
| Female: 135.6 ± 10.9 µM * | ||||||
| MDA | Tears | DR | 95 µM | Capillary electrophoresis | [106] | |
| Aqueous humor | Cataract | 0.1 ± 0.1 µmol/L * | TBARS method | [107] | ||
| Vitreous | PDR | Male: 101.3 ± 7.6 nmol/mL * | Colorimetric assay | [105] | ||
| Female: 87.6 ± 18.4 nmol/mL * | ||||||
| GSH | Tears | DR | 107 µM | Chromatography electrochemical | [24] | |
| GPx | Aqueous | Glaucoma | 18.4 ± 2.5 U/mL * | - | [101] | |
| Aqueous | Cataract | 6.1 ± 0.6 U/mL * | - | [101] | ||
| SOD | Tears | DR | 1–32 U/mg | Spectrophotometry direct | [45,108] | |
| Vitreous | PDR | Male: 30.5 ± 2.5 U/mL * | Colorimetric Enzyme assay | [105] | ||
| Female: 28.5 ± 3.8 U/mL * | ||||||
| 8-OHdG | Aqueous | Cataract | 311.6 ± 127.7 µg/mL * | ELISA | [109] | |
| Aqueous | Myopic | 212.5 ± 103.2 µg/mL * | ELISA | [109] | ||
| 8-IPGF | Aqueous | Exfoliation syndrome | 2429 ± 2940 pg/mL * | Immunoassay | [110] | |
| Aqueous | Cataract | 529.1 ± 226.8 pg/mL * | Immunoassay | [110] | ||
| Aqueous | Diabetic cataract | 624 ± 95.7 pg/mL * | ELISA | [111] | ||
| ON | Aqueous humor | PDR | 19.43 ± 8.75 µM * | Colorimetric assay | [95] | |
| Vitreous humor | PDR | 0.524 ± 0.27 µM * | spectrophotometric | [112] | ||
| T1DM | Griess reaction | |||||
| Vitreous humor | PDR | 0.383 ± 0.17 µM * | spectrophotometric | [112] | ||
| T2DM | Griess reaction | |||||
| L-tyrosine | Tears | DR | 45 µM | Chromatography electrochemical | [24] | |
| L-cysteine | Tears | DR | 48 µM | Chromatography electrochemical | [24] | |
| Ascorbic acid | Tears | DR | 665 µM | Chromatography electrochemical | [24] | |
| Uric acid | Tears | DR | 328 µM | Chromatography electrochemical | [24] | |
| Inflammatory | IL-1β | Tears | DR | 16.7 ± 3.2 pg/mL * | Multiplex assay Bio-Plex system | [113] |
| IL-6 | Tears | DR | 63.3 ± 12.3 pg/mL * | Multiplex assay | [113] | |
| Bio-Plex system | ||||||
| Tears | DED | 26.25 ± 5.20 pg/mL * | Multiplex bead assay | [114] | ||
| Aqueous humor | DR | 40.64 ± 16.52 pg/mL * | Multiplex bead immunoassay | [115] | ||
| Aqueous humor | PDR | 37.19 pg/mL (3.992–4577.38) ** | Immunology Multiplex Assay | [83] | ||
| Vitreous fluid | PDR progression | 347.2 pg/mL (26.2–758.6) ** | ELISA | [116] | ||
| Vitreous fluid | DR | 42.29 ± 10.94 pg/mL * | ELISA | [117] | ||
| Vitreous | DR | 64.2 ± 10.4 pg/mL * | Immunoassay | [118] | ||
| IL-8 | Aqueous humor | DR | 42.20 ± 33.03 pg/mL * | Multiplex bead immunoassay | [115] | |
| Aqueous humor | PDR | 25.28 pg/mL (13.21–184.62) ** | Immunology Multiplex Assay | [83] | ||
| Aqueous humor | PDR | 76.55 ± 10.88 pg/mL * | ELISA | [119] | ||
| Vitreous humor | PDR | 63.55 ± 10.74 pg/mL * | ELISA | [119] | ||
| IL-10 | Aqueous humor | DR | 0.24 ± 0.16 pg/mL * | Multiplex bead immunoassay | [115] | |
| Aqueous humor | Fuchs’ uveitis | 11.70 ± 6.60 pg/mL * | ELISA | [120] | ||
| Aqueous humor | Behcet’s uveitis | 7.23 ± 1.73 pg/mL * | ELISA | [120] | ||
| Vitreous humor | PDR | 224.789 ± 43.801 pg/mL * | ELISA | [121] | ||
| Vitreous | DR | 4.43 ± 0.4 pg/mL * | Immunoassay | [118] | ||
| IL-17A | Tears | DED | 454.67 ± 37.70 pg/mL * | Multiplex bead analysis | [114] | |
| TNF-α | Tears | NPDR | 1.2–5.5 pg/mL | ELISA | [122] | |
| Tears | PDR | 9.2–21.7 pg/mL | ELISA | [122] | ||
| Aqueous humor | DR | 4.04 ± 1.83 pg/mL * | Multiplex bead immunoassay | [115] | ||
| Aqueous humor | PDR | 84.35 ± 30.82 pg/mL * | CBA technique | [123] | ||
| Vitreous fluid | DR | 155.8 ± 82.0 pg/mL * | ELISA | [124] | ||
| IFN-y | Tears | DR | 1957.50 ± 166.1 pg/mL * | Multiplex assay Bio-Plex system | [113] | |
| VEGF-A | Aqueous humor | DR | 357.02 ± 84.25 pg/mL * | Multiplex bead immunoassay | [115] | |
| VEGF | Tears | DR | 270.7 ± 40.2 pg/mL * | Multiplex assay Bio-Plex system | [113] | |
| Aqueous humor | PDR | 211.62 pg/mL (48.10–1990.98) ** | Immunology Multiplex Assay | [83] | ||
| Vitreous fluid | PDR progression | 1789.2 pg/mL (198.5–3436.8) ** | ELISA | [116] | ||
| Vitreous | DR | 731.20 ± 222.72 pg/mL * | ELISA | [117] | ||
| Vitreous | DR | 1491.0 ± 183.1 pg/mL * | ELISA | [125] | ||
| EGF | Tears | DED | 1318.9 ± 6 835.0 pg/mL * | Milliplex bead assay | [126] | |
| Apoptosis | MMP-9 | Tears | DED | 40 ng/mL | Immunoplex | [74,127] |
| Aqueous | PDR | 160.3 ± 39.5 AU/mL * | Zymographic analysis | [128] | ||
| Vitreous | Macular hole | 113.9 ± 229.7 AU/mL * | Zymographic analysis | [129] | ||
| Cytochrome-C | Tears/Cell culture | DED | - | Cell culture | [130] |
| MiRNA | Role | Matrix | Comparison | Result | Author(s) |
|---|---|---|---|---|---|
| miR-200b | Angiogenesis promotion [167] | Vitreous humor | PDR vs. NDM | Higher | Gomaa A, 2017 [168] |
| miR-21 | Fibrosis and inflammation promotion [169] | Vitreous humor | PVD vs. MH | Higher | Usui-Ouchi A, 2016 [170] |
| miR-15a | Angiogenesis inhibition [171] | Vitreous humor | PDR vs. MH | Higher | Hirota K, 2015 [172] |
| Pro-inflammatory signaling inhibition [173] | Aqueous humor + | DME + | + Cho Heeyoon,2020 [174] | ||
| miR-320 | Apoptosis regulation and angiogenesis repression [175] | Vitreous humor | PVD vs. MH | Higher | Usui-Ouchi A, 2016 [170] |
| miR-320a miR 320b | Angiogenesis repression [176] | Vitreous humor | PDR vs. MH | Higher | Hirota K, 2015 [172] |
| miR-184 | Apoptosis promotion [177] | Aqueous humor | PDR vs. Cataract | Higher | Chen S, 2019 [178] |
| miR-93 miR-93-5p | Proliferation and angiogenesis promotion [179] | Vitreous humor | PDR vs. NDM | Higher | Hirota K, 2015 [172] |
| Aqueous humor | PDR vs. Cataract | Chen S, 2019 [178] | |||
| miR-29a | Angiogenesis inhibition [180] | Vitreous humor | PDR vs. MH | Higher | Hirota, K 2015 [172] |
| Aqueous humor + | Cataract + | + Wecker T, 2016 [93] | |||
| miR-16 miR-16-5p | Tumor suppression [181] | Vitreous humor | PVD vs. MH | Higher | Usui-Ouchi A, 2016 [170] |
| Aqueous humor + | Cataract + | Wecker T, 2016 [93] | |||
| miR-23a | Senescence promotion [182] | Vitreous humor | PDR vs. MH | Higher | Hirota K, 2015 [172] |
| miR-126 | HMGB1 and VCAM-1 regulation [173] | Aqueous humor | PDR vs. Cataract | Lower | Chen S, 2019 [178] |
| Let-7e | Proliferation inhibition [183] | Vitreous humor | PVD vs. MH | Lower | Usui-Ouchi A, 2016 [170] |
| miR-204 | Apoptosis promotion [184] | Vitreous humor | PVD vs. macular hole | Lower | Usui-Ouchi A, 2016 [170] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Contreras, A.K.; Martínez-Ruiz, M.G.; Olvera-Montaño, C.; Robles-Rivera, R.R.; Arévalo-Simental, D.E.; Castellanos-González, J.A.; Hernández-Chávez, A.; Huerta-Olvera, S.G.; Cardona-Muñoz, E.G.; Rodríguez-Carrizalez, A.D. Importance of the Use of Oxidative Stress Biomarkers and Inflammatory Profile in Aqueous and Vitreous Humor in Diabetic Retinopathy. Antioxidants 2020, 9, 891. https://doi.org/10.3390/antiox9090891
López-Contreras AK, Martínez-Ruiz MG, Olvera-Montaño C, Robles-Rivera RR, Arévalo-Simental DE, Castellanos-González JA, Hernández-Chávez A, Huerta-Olvera SG, Cardona-Muñoz EG, Rodríguez-Carrizalez AD. Importance of the Use of Oxidative Stress Biomarkers and Inflammatory Profile in Aqueous and Vitreous Humor in Diabetic Retinopathy. Antioxidants. 2020; 9(9):891. https://doi.org/10.3390/antiox9090891
Chicago/Turabian StyleLópez-Contreras, Ana Karen, María Guadalupe Martínez-Ruiz, Cecilia Olvera-Montaño, Ricardo Raúl Robles-Rivera, Diana Esperanza Arévalo-Simental, José Alberto Castellanos-González, Abel Hernández-Chávez, Selene Guadalupe Huerta-Olvera, Ernesto German Cardona-Muñoz, and Adolfo Daniel Rodríguez-Carrizalez. 2020. "Importance of the Use of Oxidative Stress Biomarkers and Inflammatory Profile in Aqueous and Vitreous Humor in Diabetic Retinopathy" Antioxidants 9, no. 9: 891. https://doi.org/10.3390/antiox9090891





