Interaction of Polyphenols as Antioxidant and Anti-Inflammatory Compounds in Brain–Liver–Gut Axis
Abstract
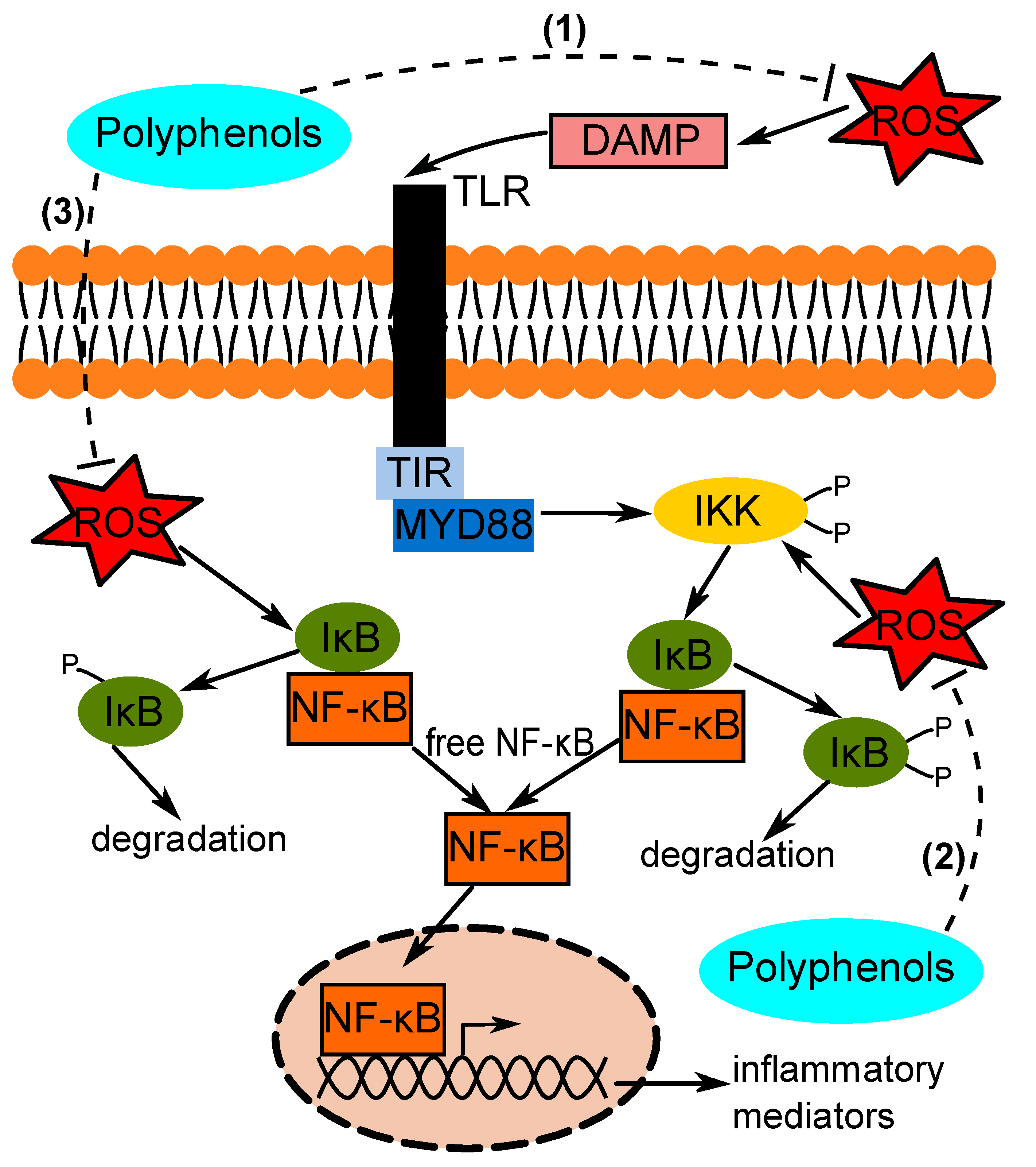
1. Introduction
2. Polyphenol Intervention in Brain Inflammation
2.1. In Vitro Models of Polyphenol Treatment in Brain Inflammation
2.2. In Vivo Models of Polyphenol Treatment in Brain Inflammation
3. Polyphenol Intervention in Liver Inflammation
3.1. In Vitro Models of Polyphenol Treatment in Liver Inflammation
3.2. In Vivo Models of Polyphenol Treatment in Liver Inflammation
4. Polyphenol Intervention in Gut Inflammation
4.1. In Vitro Models of Polyphenol Treatment in Gut Inflammation
4.2. In Vivo Models of Polyphenol Treatment in Gut Inflammation
5. Research Gap
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lugrin, J.; Rosenblatt-Velin, N.; Parapanov, R.; Liaudet, L. The role of oxidative stress during inflammatory processes. Biol. Chem. 2014, 395, 203–230. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.M.; An, J. Cytokines, inflammation and pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Song, D.H.; Lee, J.O. Sensing of microbial molecular patterns by Toll-like receptors. Immunol. Rev. 2012, 250, 216–229. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S. Oxidative stress, inflammation, and disease. In Oxidative Stress and Biomaterials; Dziubla, T., Butterfield, D.A., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 35–58. [Google Scholar]
- Powers, K.A.; Szászi, K.; Khadaroo, R.G.; Tawadros, P.S.; Marshall, J.C.; Kapus, A.; Rotstein, O.D. Oxidative stress generated by hemorrhagic shock recruits Toll-like receptor 4 to the plasma membrane in macrophages. J. Exp. Med. 2006, 203, 1951–1961. [Google Scholar] [CrossRef] [PubMed]
- Nakahira, K.; Kim, H.P.; Geng, X.H.; Nakao, A.; Wang, X.; Murase, N.; Drain, P.F.; Wang, X.; Sasidhar, M.; Nabel, E.G.; et al. Carbon monoxide differentially inhibits TLR signaling pathways by regulating ROS-induced trafficking of TLRs to lipid rafts. J. Exp. Med. 2006, 203, 2377–2389. [Google Scholar] [CrossRef]
- Lawrence, T. The nuclear factor NF-κB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef]
- Ramos-Tovar, E.; Muriel, P. Free radicals, antioxidants, nuclear factor-E2-related factor-2 and liver damage. J. Appl. Toxicol. 2020, 40, 151–168. [Google Scholar] [CrossRef]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. 2008, 4, 89–96. [Google Scholar]
- Magrone, T.; Magrone, M.; Russo, M.A.; Jirillo, E. Recent Advances on the Anti-Inflammatory and Antioxidant Properties of Red Grape Polyphenols: In Vitro and In Vivo Studies. Antioxidants 2020, 9, 35. [Google Scholar] [CrossRef]
- Winter, A.N.; Bickford, P.C. Anthocyanins and Their Metabolites as Therapeutic Agents for Neurodegenerative Disease. Antioxidants 2019, 8, 333. [Google Scholar] [CrossRef]
- Silva, R.F.; Pogačnik, L. Polyphenols from Food and Natural Products: Neuroprotection and Safety. Antioxidants 2020, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- Devassy, J.G.; Leng, S.; Gabbs, M.; Monirujjaman, M.; Aukema, H.M. Omega-3 polyunsaturated fatty acids and oxylipins in neuroinflammation and management of Alzheimer disease. Adv. Nutr. 2016, 7, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Gamba, P.; Badilli, U.; Gargiulo, S.; Maina, M.; Guina, T.; Calfapietra, S.; Biasi, F.; Cavalli, R.; Poli, G.; et al. Loading into nanoparticles improves quercetin’s efficacy in preventing neuroinflammation induced by oxysterols. PLoS ONE 2014, 9, e96795. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Shalini, V.; Helen, A. Quercetin regulates oxidized LDL induced inflammatory changes in human PBMCs by modulating the TLR-NF-κB signaling pathway. Immunobiology 2011, 216, 367–373. [Google Scholar] [CrossRef]
- Casedas, G.; Bennett, A.C.; Gonzalez-Burgos, E.; Gomez-Serranillos, M.P.; Lopez, V.; Smith, C. Polyphenol-associated oxidative stress and inflammation in a model of LPS-induced inflammation in glial cells: Do we know enough for responsible compounding? Inflammopharmacology 2019, 27, 189–197. [Google Scholar] [CrossRef]
- Carey, A.N.; Fisher, D.R.; Rimando, A.M.; Gomes, S.M.; Bielinski, D.F.; Shukitt-Hale, B. Stilbenes and anthocyanins reduce stress signaling in BV-2 mouse microglia. J. Agric. Food Chem. 2013, 61, 5979–5986. [Google Scholar] [CrossRef]
- Lau, F.C.; Bielinski, D.F.; Joseph, J.A. Inhibitory effects of blueberry extract on the production of inflammatory mediators in lipopolysaccharide-activated BV2 microglia. J. Neurosci. Res. 2007, 85, 1010–1017. [Google Scholar] [CrossRef]
- Lau, F.C.; Joseph, J.A.; McDonald, J.E.; Kalt, W. Attenuation of iNOS and COX2 by blueberry polyphenols is mediated through the suppression of NF-κB activation. J. Funct. Foods 2009, 1, 274–283. [Google Scholar] [CrossRef]
- Poulose, S.M.; Fisher, D.R.; Larson, J.; Bielinski, D.F.; Rimando, A.M.; Carey, A.N.; Schauss, A.G.; Shukitt-Hale, B. Anthocyanin-rich acai (Euterpe oleracea Mart.) fruit pulp fractions attenuate inflammatory stress signaling in mouse brain BV-2 microglial cells. J. Agric. Food Chem. 2012, 60, 1084–1093. [Google Scholar] [CrossRef]
- Jeong, J.W.; Lee, W.S.; Shin, S.C.; Kim, G.Y.; Choi, B.T.; Choi, Y.H. Anthocyanins downregulate lipopolysaccharide-induced inflammatory responses in BV2 microglial cells by suppressing the NF-kappaB and Akt/MAPKs signaling pathways. Int. J. Mol. Sci. 2013, 14, 1502–1515. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, S.; Liu, T.; Wang, X.; Huang, H.; Liu, W. Callistephin enhances the protective effects of isoflurane on microglial injury through downregulation of inflammation and apoptosis. Mol. Med. Rep. 2019, 20, 802–812. [Google Scholar] [CrossRef] [PubMed]
- Shukitt-Hale, B.; Kelly, M.E.; Bielinski, D.F.; Fisher, D.R. Tart Cherry Extracts Reduce Inflammatory and Oxidative Stress Signaling in Microglial Cells. Antioxidants 2016, 5, 33. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, S.M.; Azambuja, J.H.; De Carvalho, T.R.; Soares, M.S.P.; Oliveira, P.S.; Da Silveira, E.F.; Stefanello, F.M.; Braganhol, E.; Gutierres, J.M.; Spanevello, R.M. Glioprotective Effects of Lingonberry Extract Against Altered Cellular Viability, Acetylcholinesterase Activity, and Oxidative Stress in Lipopolysaccharide-Treated Astrocytes. Cell. Mol. Neurobiol. 2018, 38, 1107–1121. [Google Scholar] [CrossRef] [PubMed]
- Ben Youssef, S.; Brisson, G.; Doucet-Beaupré, H.; Castonguay, A.M.; Gora, C.; Amri, M.; Lévesque, M. Neuroprotective benefits of grape seed and skin extract in a mouse model of Parkinson’s disease. Nutr. Neurosci. 2019, 25, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Capiralla, H.; Vingtdeux, V.; Zhao, H.; Sankowski, R.; Al-Abed, Y.; Davies, P.; Marambaud, P. Resveratrol mitigates lipopolysaccharide- and Aβ-mediated microglial inflammation by inhibiting the TLR4/NF-κB/STAT signaling cascade. J. Neurochem. 2012, 120, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Seong, K.J.; Lee, H.G.; Kook, M.S.; Ko, H.M.; Jung, J.Y.; Kim, W.J. Epigallocatechin-3-gallate rescues LPS-impaired adult hippocampal neurogenesis through suppressing the TLR4-NF-κB signaling pathway in mice. Korean J. Physiol. Pharmacol. 2016, 20, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Ali, T.; Kim, M.W.; Jo, M.H.; Chung, J.I.; Kim, M.O. Anthocyanins Improve Hippocampus-Dependent Memory Function and Prevent Neurodegeneration via JNK/Akt/GSK3beta Signaling in LPS-Treated Adult Mice. Mol. Neurobiol. 2019, 56, 671–687. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, Y.; Lu, J.; Chen, G.; Wang, X.; Feng, J.; Ruan, J.; Sun, X.; Li, C.; Sun, Q. Purple sweet potato color suppresses lipopolysaccharide-induced acute inflammatory response in mouse brain. Neurochem. Int. 2010, 56, 424–430. [Google Scholar] [CrossRef]
- Khan, M.S.; Ali, T.; Kim, M.W.; Jo, M.H.; Jo, M.G.; Badshah, H.; Kim, M.O. Anthocyanins protect against LPS-induced oxidative stress-mediated neuroinflammation and neurodegeneration in the adult mouse cortex. Neurochem. Int. 2016, 100, 1–10. [Google Scholar] [CrossRef]
- Carvalho, F.B.; Gutierres, J.M.; Bueno, A.; Agostinho, P.; Zago, A.M.; Vieira, J.; Fruhauf, P.; Cechella, J.L.; Nogueira, C.W.; Oliveira, S.M.; et al. Anthocyanins control neuroinflammation and consequent memory dysfunction in mice exposed to lipopolysaccharide. Mol. Neurobiol. 2017, 54, 3350–3367. [Google Scholar] [CrossRef]
- Li, J.; Shi, Z.; Mi, Y. Purple sweet potato color attenuates high fat-induced neuroinflammation in mouse brain by inhibiting MAPK and NF-kappaB activation. Mol. Med. Rep. 2018, 17, 4823–4831. [Google Scholar] [PubMed]
- Pan, Z.; Cui, M.; Dai, G.; Yuan, T.; Li, Y.; Ji, T.; Pan, Y. Protective Effect of Anthocyanin on Neurovascular Unit in Cerebral Ischemia/Reperfusion Injury in Rats. Front. Neurosci. 2018, 12, 947. [Google Scholar] [CrossRef] [PubMed]
- Moussa, C.; Hebron, M.; Huang, X.; Ahn, J.; Rissman, R.A.; Aisen, P.S.; Turner, R.S. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J. Neuroinflamm. 2017, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Elvira-Torales, L.I.; García-Alonso, J.; Periago-Castón, M.J. Nutritional importance of carotenoids and their effect on liver health: A review. Antioxidants 2019, 8, 229. [Google Scholar] [CrossRef]
- Salomone, F.; Godos, J.; Zelber-Sagi, S. Natural antioxidants for non-alcoholic fatty liver disease: Molecular targets and clinical perspectives. Liver Int. 2016, 36, 5–20. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Tang, Y.; Kang, Q.; Feng, Y.; Chen, A. Curcumin inhibits gene expression of receptor for advanced glycation end-products (RAGE) in hepatic stellate cells in vitro by elevating PPARgamma activity and attenuating oxidative stress. Br. J. Pharmacol. 2012, 166, 2212–2227. [Google Scholar] [CrossRef]
- Lin, C.L.; Huang, H.C.; Lin, J.K. Theaflavins attenuate hepatic lipid accumulation through activating AMPK in human HepG2 cells. J. Lipid Res. 2007, 48, 2334–2343. [Google Scholar] [CrossRef]
- Zhu, W.; Jia, Q.; Wang, Y.; Zhang, Y.; Xia, M. The anthocyanin cyanidin-3-O-beta-glucoside, a flavonoid, increases hepatic glutathione synthesis and protects hepatocytes against reactive oxygen species during hyperglycemia: Involvement of a cAMP-PKA-dependent signaling pathway. Free. Radic. Biol. Med. 2012, 52, 314–327. [Google Scholar] [CrossRef]
- Gao, M.; Ma, Y.; Liu, D. Rutin suppresses palmitic acids- triggered inflammation in macrophages and blocks high fat diet-induced obesity and fatty liver in mice. Pharm. Res. 2013, 30, 2940–2950. [Google Scholar] [CrossRef]
- Kobori, M.; Masumoto, S.; Akimoto, Y.; Oike, H. Chronic dietary intake of quercetin alleviates hepatic fat accumulation associated with consumption of a Western-style diet in C57/BL6J mice. Mol. Nutr. Food Res. 2011, 55, 530–540. [Google Scholar] [CrossRef]
- Marcolin, E.; San-Miguel, B.; Vallejo, D.; Tieppo, J.; Marroni, N.; González-Gallego, J.; Tuñón, M.J. Quercetin treatment ameliorates inflammation and fibrosis in mice with nonalcoholic steatohepatitis. J. Nutr. 2012, 142, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-F.; Fan, S.-H.; Zheng, Y.-L.; Lu, J.; Wu, D.-M.; Shan, Q.; Hu, B. Troxerutin improves hepatic lipid homeostasis by restoring NAD(+)-depletion-mediated dysfunction of lipin 1 signaling in high-fat diet-treated mice. Biochem. Pharmacol. 2014, 91, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.Y.; Takahara, T.; Hou, J.; Kawai, K.; Sugiyama, T.; Tsukada, K.; Takemoto, M.; Takeuchi, M.; Zhong, L.; Li, X.-K. Theaflavin attenuates ischemia-reperfusion injury in a mouse fatty liver model. Biochem. Biophys. Res. Commun. 2012, 417, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Pu, P.; Wang, X.A.; Salim, M.; Zhu, L.-H.; Wang, L.; Chen, K.-J.; Xiao, J.-F.; Deng, W.; Shi, H.-W.; Jiang, H.; et al. Baicalein, a natural pro- duct, selectively activating AMPKalpha(2) and ameliorates metabolic disorder in diet-induced mice. Mol. Cell Endocrinol. 2012, 362, 128–138. [Google Scholar] [CrossRef]
- Jeon, B.T.; Jeong, E.A.; Shin, H.J.; Lee, Y.; Lee, N.H.; Kim, H.J.; Kang, S.S.; Cho, G.J.; Choi, W.S.; Roh, G.S. Resveratrol attenuates obesity-associated peripheral and central inflammation and improves memory deficit in mice fed a high-fat diet. Diabetes 2012, 61, 1444–1454. [Google Scholar] [CrossRef]
- Leclercq, I.A.; Farrell, G.C.; Sempoux, C.; Dela Pena, A.; Hors-mans, Y. Curcumin inhibits NF-kappaB activation and reduces the severity of experimental steatohepatitis in mice. J. Hepatol. 2004, 41, 926–934. [Google Scholar] [CrossRef]
- Vizzutti, F.; Provenzano, A.; Galastri, S.; Milani, S.; Delogu, W.; Novo, E.; Caligiuri, A.; Zamara, E.; Arena, U.; Laffi, G.; et al. Curcumin limits the fibrogenic evolution of experimental steatohepatitis. Lab. Investig. 2010, 90, 104–115. [Google Scholar] [CrossRef]
- Salamone, F.; Galvano, F.; Cappello, F.; Mangiameli, A.; Barbagallo, I.; Volti, G.L. Silibinin modulates lipid homeostasis and inhibits nuclear factor kappa B activation in experimental nonalcoholic steatohepatitis. Transl. Res. 2012, 159, 477–486. [Google Scholar] [CrossRef]
- Salamone, F.; Galvano, F.; Marino Gammazza, A.; Paternostro, C.; Tibullo, D.; Bucchieri, F.; Mangiameli, A.; Parola, M.; Bugianesi, E.; Volti, G.L. Silibinin improves hepatic and myocardial injury in mice with nonalcoholic steatohepatitis. Dig. Liver Dis. 2012, 44, 334–342. [Google Scholar] [CrossRef]
- Panchal, S.K.; Poudyal, H.; Brown, L. Quercetin ameliorates cardiovascular, hepatic, and metabolic changes in diet- induced metabolic syndrome in rats. J. Nutr. 2012, 142, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Panchal, S.K.; Poudyal, H.; Arumugam, T.V.; Brown, L. Rutin attenuates metabolic changes, nonalcoholic steatohepatitis, and cardiovascular remodeling in high-carbohydrate, high-fat diet-fed rats. J. Nutr. 2011, 141, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Kuzu, N.; Bahcecioglu, I.H.; Dagli, A.F.; Ozercan, I.H.; Ustundag, B.; Sahin, K. Epigallocatechin gallate attenuates experimental non-alcoholic steatohepatitis induced by high fat diet. J. Gastroenterol. Hepatol. 2008, 23, e465–e470. [Google Scholar] [CrossRef]
- Xiao, J.; Ho, C.T.; Liong, E.C.; Nanji, A.A.; Leung, T.M.; Lau, T.Y.H.; Fung, M.L.; Tipoe, G.L. Epigallocatechin gallate attenuates fibrosis, oxidative stress, and inflammation in non-alcoholic fatty liver disease rat model through TGF/ SMAD, PI3 K/Akt/FoxO1, and NF-kappa B pathways. Eur. J. Nutr. 2014, 53, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Yalniz, M.; Bahcecioglu, I.H.; Kuzu, N.; Poyrazoğlu, O.K.; Bulmus, Ö.; Celebi, S.; Ustundag, B.; Ozercan, I.H.; Sahin, K. Preventive role of genistein in an experimental non-alcoholic steatohepatitis model. J. Gastroenterol. Hepatol. 2007, 22, 2009–2014. [Google Scholar] [CrossRef] [PubMed]
- Chtourou, Y.; Fetoui, H.; Jemai, R.; Ben Slima, A.; Makni, M.; Gdoura, R. Naringenin reduces cholesterol-induced hepatic inflammation in rats by modulating matrix metalloproteinases-2, 9 via inhibition of nuclear factor kappaB pathway. Eur. J. Pharmacol. 2015, 746, 96–105. [Google Scholar] [CrossRef]
- Vitaglione, P.; Morisco, F.; Mazzone, G.; Amoruso, D.C.; Ribecco, M.T.S.; Romano, A.; Fogliano, V.; Caporaso, N.; D’Argenio, G. Coffee reduces liver damage in a rat model of steatohepatitis: The under- lying mechanisms and the role of polyphenols and melanoidins. Hepatology 2010, 52, 1652–1661. [Google Scholar] [CrossRef] [PubMed]
- Salomone, F.; Volti, G.L.; Vitaglione, P.; Morisco, F.; Fogliano, V.; Zappalà, A.; Palmigiano, A.; Garozzo, D.; Caporaso, N.; D’Argenio, G.; et al. Coffee enhances the expression of chaperones and antioxidant proteins in rats with nonalcoholic fatty liver disease. Transl. Res. 2014, 163, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Bagul, P.K.; Middela, H.; Matapally, S.; Padiya, R.; Bastia, T.; Madhusudana, K.; Reddy, B.R.; Chakravarty, S.; Banerjee, S.K. Attenuation of insulin resistance, metabolic syndrome and hepatic oxidative stress by resveratrol in fructose-fed rats. Pharmacol. Res. 2012, 66, 260–268. [Google Scholar] [CrossRef]
- Sakata, R.; Nakamura, T.; Torimura, T.; Ueno, T.; Sata, M. Green tea with high-density catechins improves liver function and fat infiltration in non-alcoholic fatty liver disease (NAFLD) patients: A double-blind placebo-controlled study. Int. J. Mol. Med. 2013, 32, 989–994. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, X.; Ran, L.; Wan, J.; Wang, X.; Qin, Y.; Shu, F.; Gao, Y.; Yuan, L.; Zhang, Q.; et al. Resveratrol improves insulin resistance, glucose and lipid metabolism in patients with non-alcoholic fatty liver disease: A randomized controlled trial. Dig. Liver Dis. 2015, 47, 226–232. [Google Scholar] [CrossRef]
- Faghihzadeh, F.; Adibi, P.; Rafiei, R.; Hekmatdoost, A. Resveratrol supplementation improves inflammatory biomarkers in patients with nonalcoholic fatty liver disease. Nutr. Res. 2014, 34, 837–843. [Google Scholar] [CrossRef]
- Stiuso, P.; Scognamiglio, I.; Murolo, M.; Ferranti, P.; De Simone, C.; Rizzo, M.R.; Tuccillo, C.; Caraglia, M.; Loguercio, C.; Federico, A. Serum oxidative stress markers and lipidomic profile to detect NASH patients responsive to an antioxidant treatment: A pilot study. Oxid. Med. Cell Longev. 2014, 2014, 169216. [Google Scholar] [CrossRef] [PubMed]
- Chachay, V.S.; Macdonald, G.A.; Martin, J.H.; Whitehead, J.; O’Moore–Sullivan, T.M.; Lee, P.; Franklin, M.; Klein, K.; Taylor, P.J.; Ferguson, M.; et al. Resveratrol does not benefit patients with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 2014, 12, 2092–2103. [Google Scholar] [CrossRef] [PubMed]
- Lavefve, L.; Howard, L.R.; Carbonero, F. Berry polyphenols metabolism and impact on human gut microbiota and health. Food Funct. 2020, 11, 45–65. [Google Scholar] [CrossRef]
- Nunes, S.; Danesi, F.; Del Rio, D.; Silva, P. Resveratrol and inflammatory bowel disease: The evidence so far. Nutr. Res. Rev. 2018, 31, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Triebel, S.; Trieu, H.L.; Richling, E. Modulation of inflammatory gene expression by a bilberry (Vaccinium myrtillus L.) extract and single anthocyanins considering their limited stability under cell culture conditions. J. Agric. Food Chem. 2012, 60, 8902–8910. [Google Scholar] [CrossRef]
- Roth, S.; Spalinger, M.R.; Gottier, C.; Biedermann, L.; Zeitz, J.; Lang, S.; Weber, A.; Rogler, G.; Scharl, M. Bilberry-derived anthocyanins modulate cytokine expression in the intestine of patients with ulcerative colitis. PLoS ONE 2016, 11, e0154817. [Google Scholar] [CrossRef]
- Cianciulli, A.; Calvello, R.; Cavallo, P.; Dragone, T.; Carofiglio, V.; Panaro, M.A. Modulation of NF-κB activation by resveratrol in LPS treated human intestinal cells results in downregulation of PGE2 production and COX-2 expression. Toxicol. In Vitro 2012, 26, 1122–1128. [Google Scholar] [CrossRef]
- Panaro, M.A.; Carofiglio, V.; Acquafredda, A.; Cavallo, P.; Cianciulli, A. Anti- inflammatory effects of resveratrol occur via inhibition of lipopolysaccharide-induced NF-κB activation in Caco-2 and SW480 human colon cancer cells. Br. J. Nutr. 2012, 108, 1623–1632. [Google Scholar] [CrossRef]
- Romier, B.; Van De Walle, J.; During, A.; Larondelle, Y.; Schneider, Y.-J. Modulation of signalling nuclear factor-κB activation pathway by polyphenols in human intestinal Caco-2 cells. Br. J. Nutr. 2008, 100, 542–551. [Google Scholar] [CrossRef]
- Serra, D.; Rufino, A.T.; Mendes, A.F.; Almeida, L.M.; Dinis, T.C.P. Resveratrol modulates cytokine-induced Jak/STAT activation more efficiently than 5-aminosalicylic acid: An in vitro approach. PLoS ONE 2014, 9, e109048. [Google Scholar] [CrossRef] [PubMed]
- Serra, D.; Almeida, L.M.; Dinis, T.C. Anti-inflammatory protection afforded by cyanidin-3-glucoside and resveratrol in human intestinal cells via Nrf2 and PPAR-γ: Comparison with 5-aminosalicylic acid. Chem. Biol. Interact. 2016, 260, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.H.; Xu, Z.L.; Dong, D.; He, S.A.; Yu, H. Protective effect of anthocyanins extract from blueberry on TNBS-induced IBD model of mice. Evid. Based Complementary Altern. Med. 2011, 2011, 525462. [Google Scholar] [CrossRef] [PubMed]
- Pervin, M.; Hasnat, M.A.; Lim, J.H.; Lee, Y.M.; Kim, E.O.; Um, B.H.; Lim, B.O. Preventive and therapeutic effects of blueberry (Vaccinium corymbosum) extract against DSS-induced ulcerative colitis by regulation of antioxidant and inflammatory mediators. J. Nutr. Biochem. 2016, 28, 103–113. [Google Scholar] [CrossRef]
- Montrose, D.C.; Horelik, N.A.; Madigan, J.P.; Stoner, G.D.; Wang, L.S.; Bruno, R.S.; Park, H.J.; Giardina, C.; Rosenberg, D.W. Anti-inflammatory effects of freeze-dried black raspberry powder in ulcerative colitis. Carcinogenesis 2011, 32, 343–350. [Google Scholar] [CrossRef]
- Piberger, H.; Oehme, A.; Hofmann, C.; Dreiseitel, A.; Sand, P.G.; Obermeier, F.; Schoelmerich, J.; Schreier, P.; Krammer, G.; Rogler, G. Bilberries and their anthocyanins ameliorate experimental colitis. Mol. Nutr. Food Res. 2011, 55, 1724–1729. [Google Scholar] [CrossRef]
- Wang, L.S.; Kuo, C.T.; Stoner, K.; Yearsley, M.; Oshima, K.; Yu, J.; Huang, T.H.; Rosenberg, D.; Peiffer, D.; Stoner, G.; et al. Dietary black raspberries modulate DNA methylation in dextran sodium sulfate (DSS)-induced ulcerative colitis. Carcinogenesis 2013, 34, 2842–2850. [Google Scholar] [CrossRef]
- Xiao, X.; Kim, J.; Sun, Q.; Kim, D.; Park, C.S.; Lu, T.S.; Park, Y. Preventive effects of cranberry products on experimental colitis induced by dextran sulphate sodium in mice. Food Chem. 2015, 167, 438–446. [Google Scholar] [CrossRef]
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E.; et al. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 2015, 64, 872–883. [Google Scholar] [CrossRef]
- Cui, X.; Jin, Y.; Hofseth, A.B.; Pena, E.; Habiger, J.; Chumanevich, A.; Poudyal, D.; Nagarkatti, M.; Nagarkatti, P.S.; Singh, U.P.; et al. Resveratrol suppresses colitis and colon cancer associated with colitis. Cancer Prev. Res. 2010, 3, 549–559. [Google Scholar] [CrossRef]
- Sánchez-Fidalgo, S.; Cárdeno, A.; Villegas, I.; Talero, E.; Alarcón, C. Dietary supplementation of resveratrol attenuates chronic colonic inflammation in mice. Eur. J. Pharmacol. 2010, 633, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Singh, U.P.; Singh, N.P.; Singh, B.; Hofseth, L.J.; Price, R.L.; Nagarkatti, M.; Nagarkatti, P.S. Resveratrol (trans-3,5,4ʹ-trihydroxystilbene) induces silent mating type information regulation-1 and down-regulates nuclear transcription factor-κB activation to abrogate dextran sulfate sodium-induced colitis. J. Pharmacol. Exp. Ther. 2010, 332, 829–839. [Google Scholar] [CrossRef] [PubMed]
- Wagnerova, A.; Babickova, J.; Liptak, R.; Vlkova, B.; Celec, P.; Gardlik, R. Sex differences in the effect of resveratrol on DSS-induced colitis in mice. Gastroenterol. Res. Pract. 2017, 2017, 8051870. [Google Scholar] [CrossRef]
- Yao, J.; Wang, J.Y.; Liu, L.; Li, Y.-X.; Xun, A.-Y.; Zeng, W.-S.; Jia, C.-H.; Wei, X.-X.; Feng, J.-L.; Zhao, L.; et al. Antioxidant effects of resveratrol on mice with DSS-induced ulcerative colitis. Arch. Med. Res. 2010, 41, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; Tomé-Carneiro, J.; Yáñez-Gascón, M.J.; Alcántara, D.; Selma, M.V.; Beltrán, D.; Garcia-Conesa, M.T.; Urbán, C.; Lucas, R.; Tomás-Barberán, F.A.; et al. Preventive oral treatment with resveratrol pro-prodrugs drastically reduce colon inflammation in rodents. J. Med. Chem. 2010, 53, 7365–7376. [Google Scholar] [CrossRef] [PubMed]
- Youn, J.; Lee, J.-S.; Na, H.-K.; Kundu, J.K.; Dong, Z. Resveratrol and piceatannol inhibit iNOS expression and NF-κB activation in dextran sulfate sodium-induced mouse colitis. Nutr. Cancer 2009, 61, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Altamemi, I.; Murphy, E.A.; Catroppo, J.F.; Zumbrun, E.E.; Zhang, J.; McClellan, J.L.; Singh, U.P.; Nagarkatti, P.S.; Nagarkatti, M. Role of microRNAs in resveratrol-mediated mitigation of colitis-associated tumorigenesis in ApcMin/+ mice. J. Pharmacol. Exp. Ther. 2014, 350, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Singh, U.P.; Singh, N.P.; Singh, B.; Hofseth, L.J.; Taub, D.D.; Price, R.L.; Nagarkatti, P.S.; Nagarkatti, M. Role of resveratrol-induced CD11b+ Gr-1+ myeloid derived sup-pressor cells (MDSCs) in the reduction of CXCR3+ T cells and amelioration of chronic colitis in IL-10(–/–) mice. Brain Behav. Immun. 2012, 26, 72–82. [Google Scholar] [CrossRef]
- Osman, N.; Adawi, D.; Ahrné, S.; Jeppsson, B.; Molin, G. Probiotics and blueberry attenuate the severity of dextran sulfate sodium (DSS)-induced colitis. Digest. Dis. Sci. 2008, 53, 2464–2473. [Google Scholar] [CrossRef]
- Abdallah, D.M.; Ismael, N.R. Resveratrol abrogates adhesion molecules and protects against TNBS-induced ulcerative colitis in rats. Can. J. Physiol. Pharmacol. 2011, 89, 811–818. [Google Scholar]
- Lozano-Pérez, A.A.; Rodriguez-Nogales, A.; Ortiz-Cullera, V.; Algieri, F.; Zorrilla, P.; Rodríguez-Cabezas, M.E.; Mesa, N.G.; Utrilla, M.P.; De Matteis, L.; Mesa, J.G.; et al. Silk fibroin nanoparticles constitute a vector for controlled release of resveratrol in an experimental model of inflammatory bowel disease in rats. Int. J. Nanomed. 2014, 9, 4507–4520. [Google Scholar]
- Martín, A.R.; Villegas, I.; La Casa, C.; De La Lastra, A. Resveratrol, a polyphenol found in grapes, suppresses oxidative damage and stimulates apoptosis during early colonic inflammation in rats. Biochem. Pharmacol. 2004, 67, 1399–1410. [Google Scholar] [PubMed]
- Martín, A.R.; Villegas, I.; Sánchez-Hidalgo, M.; Alarcón, C. The effects of resveratrol, a phytoalexin derived from red wines, on chronic inflammation induced in an experimentally induced colitis model. Br. J. Pharmacol. 2006, 147, 873–885. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, G.; Yildiz, Y.; Ulutas, P.A.; Yaylali, A.; Ural, M. Resveratrol pretreatment ameliorates TNBS colitis in rats. Recent Pat. Endocr. Metab. Immune Drug Discov. 2015, 9, 134–140. [Google Scholar] [CrossRef]
- Arslan, A.; Ozcicek, F.; Cimen, F.K.; Altuner, D.; Yarali, O.; Kurt, N.; Tumkaya, L.; Ozturk, C.; Suleyman, H. Protective effect of resveratrol against methotrexate- induced oxidative stress in the small intestinal tissues of rats. Int. J. Clin. Exp. Med. 2015, 8, 10491–10500. [Google Scholar] [PubMed]
- Abdin, A.A. Targeting sphingosine kinase 1 (SphK1) and apoptosis by colon-specific delivery formula of resveratrol in treatment of experimental ulcerative colitis in rats. Eur. J. Pharmacol. 2013, 718, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; Yañéz-Gascón, M.J.; Selma, M.V.; González-Sarrías, A.; Toti, S.; Cerón, J.; Tomás-Barberán, F.A.; Dolara, P.; Espín, J.C. Effect of a low dose of dietary resveratrol on colon microbiota, inflammation and tissue damage in a DSS-induced colitis rat model. J. Agric. Food Chem. 2009, 57, 2211–2220. [Google Scholar] [CrossRef]
- Rahal, K.; Schmiedlin-Ren, P.; Adler, J.; Dhanani, M.; Sultani, V.; Rittershaus, A.C.; Reingold, L.; Zhu, J.; McKenna, B.J.; Christman, G.M.; et al. Resveratrol has antiinflammatory and antifibrotic effects in the peptidoglycan-polysaccharide rat model of Crohn’s disease. Inflamm. Bowel Dis. 2012, 18, 613–623. [Google Scholar] [CrossRef]
- Biedermann, L.; Mwinyi, J.; Scharl, M.; Frei, P.; Zeitz, J.; Kullak-Ublick, G.A.; Vavricka, S.R.; Fried, M.; Weber, A.; Humpf, H.U.; et al. Bilberry ingestion improves disease activity in mild to moderate ulcerative colitis—An open pilot study. J. Crohns Colitis 2013, 7, 271–279. [Google Scholar] [CrossRef]
- Samsami-Kor, M.; Daryani, N.E.; Asl, P.R.; Hekmatdoost, A. Anti-inflammatory effects of resveratrol in patients with ulcerative colitis: A randomized, double-blind, placebo-controlled pilot study. Arch. Med. Res. 2015, 46, 280–285. [Google Scholar] [CrossRef]
- Samsami-Kor, M.; Daryani, N.E.; Asl, P.R.; Hekmatdoost, A. Resveratrol supplementation and oxidative/anti-oxidative status in patients with ulcerative colitis: A randomized, double-blind, placebo-controlled pilot study. Arch. Med. Res. 2016, 47, 304–309. [Google Scholar] [CrossRef] [PubMed]
| Model of Study | Agent | Effects | Reference |
|---|---|---|---|
| In vitro | |||
| Human neuroblastoma SH-SY5Y cells (oxysterol induced) | Quercetin | ↓ TLR4 signaling | [14] |
| Human PBMC (oxLDL-induced) | Quercetin | ↓ TLR2 and TLR4 expressions, NF-κB activation, inflammatory enzymes activity | [15] |
| Human astrocytes (LPS-induced) | Anthocyanins | ↓ IL-6 secretion (low LPS and anthocyanin dose); ↑ IL-6 secretion (high anthocyanin dose in LPS absence) | [16] |
| Mouse BV2 microglial cells (LPS-induced) | Blueberry extract | ↓ NO and TNF-α release, iNOS and COX-2 expressions, NF-κB nuclear translocation | [17,18,19] |
| Mouse BV2 microglial cells (LPS-induced) | Anthocyanins | ↓ NO, PGE2, TNF-α and IL-1β release, iNOS and COX-2 expressions, NF-κB nuclear translocation | [20,21] |
| Mouse microglial cells (LPS/IFN-γ-induced) | Anthocyanins | ↓ NO and TNF-α release, iNOS expression | [22] |
| Rat HAPI microglial cells (LPS-induced) | Tart cherry extract | ↓ NO and TNF-α release, COX-2 expression; ↔ iNOS expression | [23] |
| Rat astrocytes (LPS-induced) | Lingonberry extract | ↓ ROS production | [24] |
| Animal | |||
| Mouse model (PD) | GSSE | ↓ ROS production, inflammatory markers | [25] |
| Mouse model (LPS and Aβ-induced microglia neuroinflammation) | RES | ↓ TLR4, NF-κB and cytokine secretion | [26] |
| Mouse model (LPS-impaired adult hippocampal neurogenesis) | EGCG | ↓ TLR4 signaling | [27] |
| Mouse model (LPS-treated) | Anthocyanins | ↓ NF-kB, TNF-α, and IL-1β levels | [28] |
| Mouse model (LPS-treated) | PSPC | ↓ TNF-α, IL-6 and IL-1β overproduction, NF-kB activation | [29] |
| Mouse model (LPS-treated) | Anthocyanins | ↓ ROS production, NF-kB activation, TNF-α, and IL-1β levels | [30] |
| Mouse model (LPS-treated) | Anthocyanins | ↓ TNF-α, and IL-1β increase; ↑ IL-10 expression | [31] |
| Mouse model (high-fat diet) | PSPC | ↓ iNOS, COX-2, TNF-α, IL-1β and IL-6 expressions, p38 MAPK and NF-kB activation; ↑ IL-10 levels | [32] |
| Rat model (MCAO/R) | Anthocyanins | ↓ TNF-α, IL-6 and IL-1β levels, NF-kB and NLRP3 expressions | [33] |
| Human | |||
| Subjects with AD | RES | ↓ plasma pro-inflammatory markers | [34] |
| Model of Study | Agent | Effects | Reference |
|---|---|---|---|
| In vitro | |||
| Hepatic stellate cells (glucose-induced) | Curcumin | ↓ ROS production; ↑ GCL activity, GSH level | [37] |
| Human HepG2 cells (fatty acid-induced) | Theaflavins | ↓ ROS production | [38] |
| Human HepG2 cells (glucose-induced) | C3G | ↓ ROS production; ↑ GCL activity, GSH level | [39] |
| Mouse macrophage cells (palmitic acid-induced) | Rutin | ↓ ROS production, MCP-1, TNF-α, IL-6, IFN-γ, IL-1β genes expressions | [40] |
| Animal | |||
| Mouse model (Western diet) | Quercetin | ↓ TBARS, TG and TNF-α levels; ↑ GPx and CAT levels | [41] |
| Mouse model (MCD) | Quercetin | ↓ TLR4 protein concentration, TNF-α, IL-6 and COX-2 mRNA expressions | [42] |
| Mouse model (HFD) | Rutin | ↓ TNF-α and Mcp1 gene expressions | [40] |
| Mouse model (HFD) | Troxerutin | ↓ ROS levels; ↑ GPx, GSH and SOD levels | [43] |
| Mouse model (MCDHFD) | Theaflavins | ↓ TBARS level, ROS production, TNF-α expressions | [44] |
| Mouse model (HFD) | Baicalein | ↓ MCP-1 and TNF-α levels | [45] |
| Mouse model (HFD) | RES | ↓ macrophage infiltration | [46] |
| Mouse model (MCD) | Curcumin | ↓ ICAM-1, COX-2 and MCP-1 expressions, NF-κB signalling | [47] |
| Mouse model (MCD) | Curcumin | ↓ ROS production | [48] |
| Mouse model (MCD) | Silibinin | ↓ ROS production, iNOS expression, NF-κB activation | [49] |
| Mouse model (MCD) | Silibinin | ↓ Il-6 and TNF-α expressions; ↑ GSH level | [50] |
| Rat model (HFD) | Quercetin | ↓ NF-κB expression; ↑ Nrf2 and HO-1 expressions | [51] |
| Rat model (HFD) | Rutin | ↓ plasma MDA; ↑ GPx expression | [52] |
| Rat model (HFD) | EGCG | ↓ plasma and liver MDA; ↑ GSH level | [53] |
| Rat model (HFD) | EGCG | ↓ iNOS, COX-2 and TNF-α expressions; ↑ GPx and CAT activity | [54] |
| Rat model (HFD) | Genistein | ↓ TNF-α and plasma and liver MDA levels | [55] |
| Rat model (HCD) | Naringenin | ↓ ROS production, TNF-α, IL-6, IL-1β and iNOS expressions | [56] |
| Rat model (HFD) | Coffee polyphenols | ↓ TNF-α, IFN-γ, IL-4 and IL-10 expressions; ↑ GSH/GSSG ratio, | [57] |
| Rat model (HFD) | Coffee polyphenols | ↑ GST expression | [58] |
| Rat model (fructose-fed) | RES | ↓ TBARS level; ↑ SOD activity, Nrf2 and GSH levels | [59] |
| Human | |||
| Subjects with NAFLD | Catechins | ↓ urinary F2t-isoprostane excretion (high dose) | [60] |
| Subjects with NAFLD | RES | ↓ inflammatory markers (TNF-α, CK-18, FGF-21) | [61] |
| Subjects with NAFLD | RES | ↓ inflammatory markers (IL-6, hs-CRP, NF-κB) | [62] |
| Subjects with NAFLD | Silymarin | ↓ NASH score and serum oxidative stress | [63] |
| Subjects with NAFLD | RES | ↑ ALT and AST levels (high dose) | [64] |
| Model of Study | Agent | Effects | Reference |
|---|---|---|---|
| In vitro | |||
| Human colon epithelial cells (cytokine-treated) | Anthocyanins | ↓ IP-10 and TNF-α expression | [67] |
| Human monocytic THP-1 cells (IFN-γ-treated) | Anthocyanins | ↓ IFN-γ receptor 2 expression | [68] |
| Human intestinal Caco-2 cells (LPS-treated) | RES | ↓ COX-2 expression, PGE2 release, NF-κB activation | [69] |
| Human intestinal Caco-2 cells (LPS-treated) | RES | ↓ iNOS and TLR4 expression, NF-κB activation, NO release (high dose) | [70] |
| Human intestinal Caco-2 cells (IL-1β-treated) | RES | ↑ NF-κB activation, p-IκB/IκB ratio, IL-8 production | [71] |
| Human intestinal Caco-2 cells (TNF-α-treated) | RES | ↑ NF-κB activation | [71] |
| Human colon epithelial HT-29 cells (cytokine-treated) | RES | ↓ ROS production, iNOS and COX-2 expression, NO and PGE2 release; ↔ NF-κB activation | [72] |
| Human colon epithelial HT-29 cells (cytokine-treated) | RES | ↑ HO-1 and GCL expression, Nrf2 activation, GSH:GSSG ratio | [73] |
| Human colon SW480 cells (LPS-treated) | RES | ↓ iNOS and TLR4 expression, NF-κB activation, and NO release (high dose) | [70] |
| Animal | |||
| Mouse model (TNBS-induced colitis) | Anthocyanins | ↓ MPO activity, IL-12, TNF-α and IFN-γ increase, NO production; ↑ IL-10 expression | [74] |
| Mouse model (DSS-induced colitis) | Blueberry extract | ↓ COX-2, iNOS, IFN-γ and IL-1β expression, NF-κB activation, neutrophil infiltration, MDA and serum PGE2 levels; ↑ CAT and SOD activity | [75] |
| Mouse model (DSS-induced colitis) | Black raspberry powder | ↓ TNF-α and IL-1β expression, NF-κB and COX-2 activity; ↔ RNS and MDA levels, inflammatory cells infiltration | [76] |
| Mouse model (DSS-induced colitis) | Anthocyanins | ↓ TNF-α and IFN-γ secretion | [77] |
| Mouse model (DSS-induced colitis) | Black raspberry powder | ↓ macrophages and neutrophils infiltration, NF-κB translocation | [78] |
| Mouse model (DSS-induced colitis) | Cranberry extract or dried cranberries | ↓ MPO activity, TNF-α and IL-1β expression | [79] |
| Mouse model (high fat diet) | Cranberry extract | ↓ COX-2 and TNF-α expression, LPS level; ↔ MDA and SOD levels | [80] |
| Mouse model (DSS-induced colitis) | RES | ↓ iNOS, COX-2 and TNF-α levels | [81] |
| Mouse model (DSS-induced colitis) | RES | ↓ iNOS and COX-2 expression, TNF-α and IL-1β levels; ↑ IL-10 level | [82] |
| Mouse model (DSS-induced colitis) | RES | ↓ IL-6, TNF-α, IFN-γ and IL-1β levels, COX-1 and COX-2 expression | [83] |
| Mouse model (DSS-induced colitis) | RES | ↔ MPO activity and TNF-α level | [84] |
| Mouse model (DSS-induced colitis) | RES | ↓ MPO, SOD and GPx activity, MDA level, TNF-α, IFN-γ and IL-8 expression | [85] |
| Mouse model (DSS-induced colitis) | RES | ↔ MPO activity, PGE2, IL-6 and IL-10 levels | [86] |
| Mouse model (DSS-induced colitis) | RES | ↓ iNOS level, NF-κB and IκB activation | [87] |
| Mouse model (DSS-induced colitis) | RES | ↓ TNF-α level, COX-2 and IL-6 expression | [88] |
| Mouse model (Spontaneous chronic colitis) | RES | ↓ IL-6, IL-12, TNF-α, IFN-γ and IL-1β levels | [89] |
| Rat model (DSS-induced colitis) | Blueberry powder | ↓ MPO activity, MDA concentration; ↔ MCP-1 and GRO/CINC-1 levels | [90] |
| Rat model (TNBS-induced colitis) | RES | ↓ MPO activity, VCAM-1, ICAM-1, MDA, NO and GSH levels | [91] |
| Rat model (TNBS-induced colitis) | RES | ↓ MPO activity, GSH level, ICAM-1, MCP-1, CINC-1, TNF-α, IL-1β, IL-6 and IL-12 expression | [92] |
| Rat model (TNBS-induced colitis) | RES | ↓ MPO activity, IL-1β, PGE2 and PGD2 levels | [93] |
| Rat model (TNBS-induced colitis) | RES | ↓ MPO activity, TNF-α level, COX-1, COX-2 and NF-κB p65 expression; ↑ PGE2 level; ↔ PGD2 level | [94] |
| Rat model (TNBS-induced colitis) | RES | ↓ MDA level; ↑ GPx activity; ↔ MPO, SOD, CAT activities | [95] |
| Rat model (Methotrexate-induced colitis) | RES | ↓ MDA and GSH levels, MPO expression | [96] |
| Rat model (Oxazolone-induced colitis) | RES | ↓ MPO activity | [97] |
| Rat model (DSS-induced colitis) | RES | ↓ COX-2, PGE2 and NO levels; ↔ TBARS level | [98] |
| Rat model (PG-PS-induced colitis) | RES | ↓ IL-6, TNF-α and IL-1β expression | [99] |
| Human | |||
| Subjects with UC | Anthocyanins | ↓ TNF-α, IFN-γ and MCP-1 levels, NF-κB activation; ↑ IL-22, IL-10 and IL-17A levels | [68] |
| Subjects with mild to moderate UC | Anthocyanins | ↓ faecal calprotectin level and Riley Index | [100] |
| Subjects with UC | RES | ↓ hs-CRP, TNF-α levels, PBMC NF-κB activation; ↑ IBDQ-9 score; ↔ SCCAI score | [101] |
| Subjects with mild to moderate UC | RES | ↓ MDA level; ↑ SOD activity, IBDQ-9 score; ↔ SCCAI score | [102] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, A.; Yau, Y.F.; Leung, K.S.; El-Nezami, H.; Lee, J.C.-Y. Interaction of Polyphenols as Antioxidant and Anti-Inflammatory Compounds in Brain–Liver–Gut Axis. Antioxidants 2020, 9, 669. https://doi.org/10.3390/antiox9080669
Singh A, Yau YF, Leung KS, El-Nezami H, Lee JC-Y. Interaction of Polyphenols as Antioxidant and Anti-Inflammatory Compounds in Brain–Liver–Gut Axis. Antioxidants. 2020; 9(8):669. https://doi.org/10.3390/antiox9080669
Chicago/Turabian StyleSingh, Amritpal, Yu Fung Yau, Kin Sum Leung, Hani El-Nezami, and Jetty Chung-Yung Lee. 2020. "Interaction of Polyphenols as Antioxidant and Anti-Inflammatory Compounds in Brain–Liver–Gut Axis" Antioxidants 9, no. 8: 669. https://doi.org/10.3390/antiox9080669
APA StyleSingh, A., Yau, Y. F., Leung, K. S., El-Nezami, H., & Lee, J. C.-Y. (2020). Interaction of Polyphenols as Antioxidant and Anti-Inflammatory Compounds in Brain–Liver–Gut Axis. Antioxidants, 9(8), 669. https://doi.org/10.3390/antiox9080669







