Coenzyme Q10: Clinical Applications in Cardiovascular Diseases
Abstract
1. Introduction
2. Methods
3. Results
3.1. CoQ10 and Cardiovascular Risk Factors
3.1.1. High Blood Pressure
3.1.2. Insulin-Resistance and Type 2 Diabetes
3.1.3. Dyslipidemias
3.1.4. Systemic Inflammation
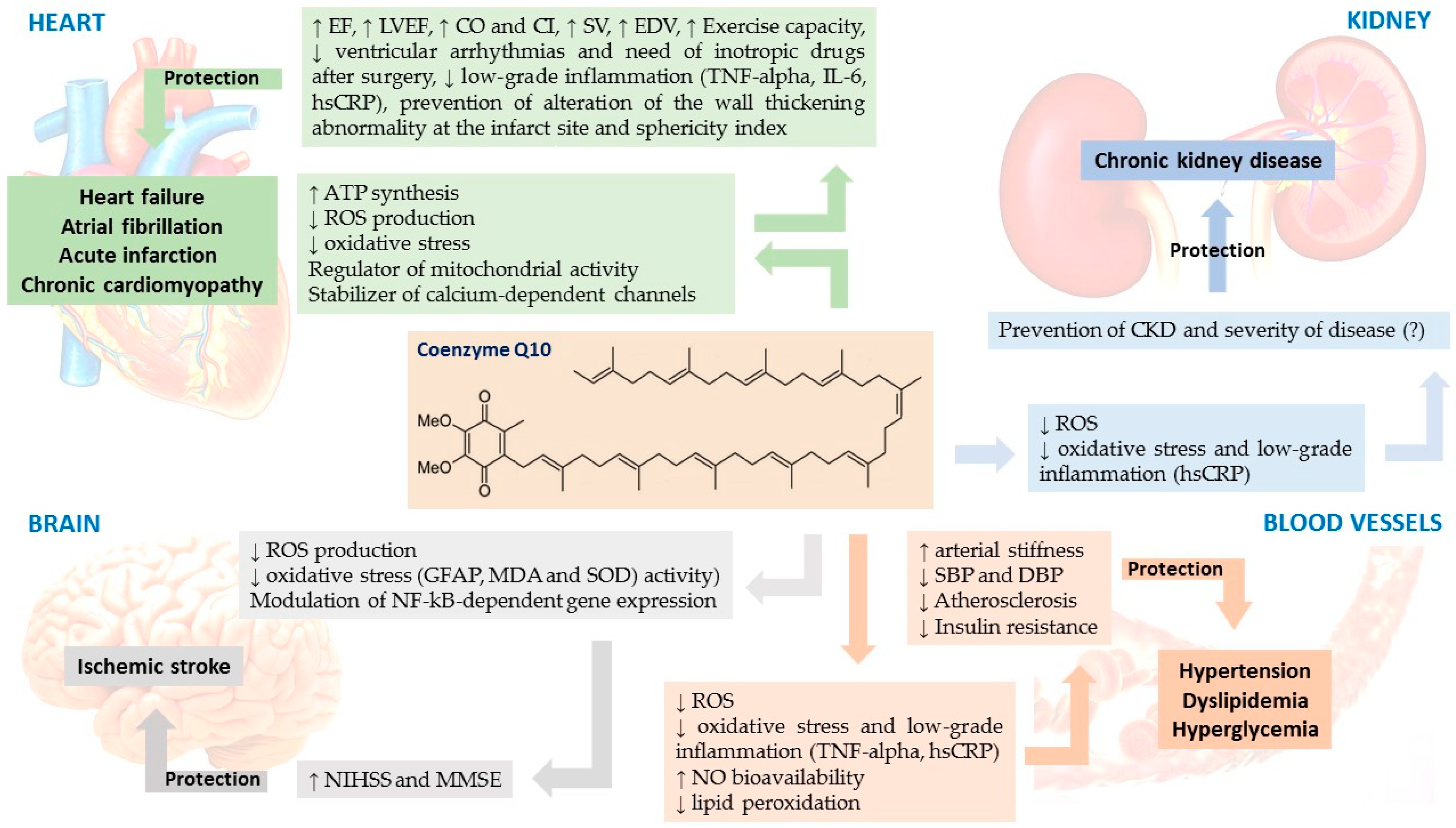
3.2. CoQ10 and Cardiovascular Disease
3.2.1. CoQ10 and Heart Failure (HF)
3.2.2. CoQ10 and Myocardial Infarction
3.2.3. CoQ10 and Atrial Fibrillation
3.2.4. CoQ10 and Nonischemic Cardiomyopathies
3.2.5. CoQ10 and Ischemic Stroke
3.3. Special Conditions
3.3.1. Chronic Kidney Disease
3.3.2. Migraine
3.3.3. Pre-Eclampsia
3.3.4. CoQ10 and Statin-Intolerance
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cicero, A.F.; Colletti, A. Nutraceuticals and Dietary Supplements to Improve Quality of Life and Outcomes in Heart Failure Patients. Curr. Pharm. Des. 2017, 23, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Bentinger, M.; Brismar, K.; Dallner, G. The antioxidant role of coenzyme Q. Mitochondrion 2007, 7, S41–S50. [Google Scholar] [CrossRef] [PubMed]
- Saini, R. Coenzyme Q10: The essential nutrient. J. Pharm. Bioallied Sci. 2011, 3, 466–467. [Google Scholar] [CrossRef] [PubMed]
- Aberg, F.; Appelkvist, E.L.; Dallner, G.; Ernster, L. Distribution and redox state of ubiquinones in rat and human tissues. Arch. Biochem. Biophys. 1992, 295, 230–234. [Google Scholar] [CrossRef]
- Miles, M.V.; Horn, P.S.; Morrison, J.A.; Tang, P.H.; DeGrauw, T.; Pesce, A.J. Plasma coenzyme Q10 reference intervals, but not redox status, are affected by gender and race in self-reported healthy adults. Clin. Chim. Acta 2003, 332, 123–132. [Google Scholar] [CrossRef]
- Hernández-Camacho, J.D.; Bernier, M.; López-Lluch, G.; Navas, P. Coenzyme Q10 Supplementation in Aging and Disease. Front. Physiol. 2018, 9, 44. [Google Scholar] [CrossRef]
- Garrido-Maraver, J.; Oropesa-Ávila, M.; Cordero, M.D.; Vega, A.F.; de la Mata, M.; Delgado, A.; de Miguel, M.; Calero, C.P.; Villanueva Paz, M.; Cotán, D.; et al. Coenzyme Q10 Therapy. Mol. Syndromol. 2014, 5, 187–197. [Google Scholar] [CrossRef]
- De Barcelos, I.P.; Haas, R.H. CoQ10 and Aging. Biology 2019, 8, 28. [Google Scholar] [CrossRef]
- Littarru, G.P.; Tiano, L. Bioenergetic and antioxidant properties of coenzyme Q10: Recent developments. Mol. Biotechnol. 2007, 37, 31–37. [Google Scholar] [CrossRef]
- Malekmohammad, K.; Sewell, R.D.E.; Rafieian-Kopaei, M. Antioxidants and Atherosclerosis: Mechanistic Aspects. Biomolecules 2019, 9, 301. [Google Scholar] [CrossRef]
- Stefely, J.A.; Pagliarini, D.J. Biochemistry of Mitochondrial Coenzyme Q Biosynthesis. Trends Biochem. Sci. 2017, 42, 824–843. [Google Scholar] [CrossRef] [PubMed]
- Szkopińska, A. Ubiquinone. Biosynthesis of quinone ring and its isoprenoid side chain. Intracellular localization. Acta Biochim. Pol. 2000, 47, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Turunen, M.; Olsson, J.; Dallner, G. Metabolism and function of coenzyme Q. Biochim. Biophys. Acta 2004, 1660, 171–199. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Aberg, F.; Appelkvist, E.L.; Dallner, G.; Ernster, L. Uptake of dietary coenzyme Q supplement is limited in rats. J. Nutr. 1995, 125, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Mariscal, F.M.; Yubero-Serrano, E.M.; Villalba, J.M.; Lopez-Miranda, J. Coenzyme Q10: From bench to clinic in aging diseases, a translational review. Crit. Rev. Food Sci. Nutr. 2018, 16, 1–18. [Google Scholar] [CrossRef]
- Boroujeni, M.B.; Khayat, Z.K.; Anbari, K.; Niapour, A.; Gholami, M.; Gharravi, A.M. Coenzyme Q10 Protects Skeletal Muscle From Ischemia-Reperfusion Through the NF-kappa B Pathway. Perfusion 2017, 32, 372–377. [Google Scholar] [CrossRef]
- Zhai, J.; Bo, Y.; Lu, Y.; Liu, C.; Zhang, L. Effects of Coenzyme Q10 on Markers of Inflammation: A Systematic Review and Meta-Analysis. PLoS ONE 2017, 12, e0170172. [Google Scholar] [CrossRef]
- Mantle, D.; Hargreaves, I. Coenzyme Q10 and Degenerative Disorders Affecting Longevity: An Overview. Antioxidants 2019, 8, 44. [Google Scholar] [CrossRef]
- Galasko, D.R.; Peskind, E.; Clark, C.M.; Quinn, J.F.; Ringman, J.M.; Jicha, G.A.; Cotman, C.; Cottrell, B.; Montine, T.J.; Thomas, R.G.; et al. Alzheimer’s Disease Cooperative Study Antioxidants for Alzheimer disease: A randomized clinical trial with cerebrospinal fluid biomarker measures. Arch. Neurol. 2012, 69, 836–841. [Google Scholar] [CrossRef]
- Müller, T.; Büttner, T.; Gholipour, A.F.; Kuhn, W. Coenzyme Q10 supplementation provides mild symptomatic benefit in patients with Parkinson’s disease. Neurosci. Lett. 2003, 341, 201–204. [Google Scholar] [CrossRef]
- Parkinson Study Group QE3 Investigators. A randomized clinical trial of high-dosage coenzyme Q10 in early Parkinson disease: No evidence of benefit. JAMA Neurol. 2014, 71, 543–552. [Google Scholar] [CrossRef] [PubMed]
- NCD Risk Factor Collaboration. Worldwide trends in blood pressure from 1975 to 2015: A pooled analysis of 1479 population-based measurement studies with 19.1 million participants. Lancet 2017, 389, 37–55. [Google Scholar] [CrossRef]
- Forouzanfar, M.H.; Liu, P.; Roth, G.A.; Ng, M.; Biryukov, S.; Marczak, L.; Alexander, L.; Estep, K.; Abate, K.A.; Akinyemiju, T.F.; et al. Global Burden of Hypertension and Systolic Blood Pressure of at Least 110 to 115 mm Hg, 1990–2015. JAMA 2017, 317, 165–182. [Google Scholar] [CrossRef] [PubMed]
- Hednerm, T.; Kjeldsen, S.E.; Narkiewicz, K. State of global health-hypertension burden and control. Blood Press 2012, 21, 12. [Google Scholar] [CrossRef]
- Digiesi, V.; Cantini, F.; Oradei, A.; Bisi, G.; Guarino, G.C.; Brocchi, A.; Bellandi, F.; Mancini, M.; Littarru, G.P. Coenzyme Q10 in essential hypertension. Mol. Aspects Med. 1994, 15, s257–s263. [Google Scholar] [CrossRef]
- Ignarro, L.J. Biological actions and properties of endothelium-derived nitric oxide formed and released from artery and vein. Circ. Res. 1989, 65, 1–21. [Google Scholar] [CrossRef]
- Fabre, L.F.; Banks, R.C., Jr.; McIsaac, W.M.; Farrell, G. Effects of ubiquinone and related substances on secretion of aldosterone and cortisol. Am. J. Physiol. 1965, 208, 1275–1280. [Google Scholar] [CrossRef]
- Langsjoen, P.; Willis, R.; Folkers, K. Treatment of essential hypertension with coenzyme Q10. Mol. Aspects Med. 1994, 15, S265–S272. [Google Scholar] [CrossRef]
- Zhang, P.; Yang, C.; Guo, H.; Wang, J.; Lin, S.; Li, H.; Yang, Y.; Ling, W. Treatment of coenzyme Q10 for 24 weeks improves lipid and glycemic profile in dyslipidemic individuals. J. Clin. Lipidol. 2018, 12, 417–427. [Google Scholar] [CrossRef]
- Ho, M.J.; Bellusci, A.; Wright, J.M. Blood pressure lowering efficacy of coenzyme Q10 for primary hypertension. Cochrane Database Syst. Rev. 2009, CD007435. [Google Scholar] [CrossRef]
- Rosenfeldt, F.L.; Haas, S.J.; Krum, H.; Hadj, A.; Leong, J.Y.; Watts, G.F. Coenzyme Q10 in the treatment of hypertension: A meta-analysis of the clinical trials. J. Hum. Hypertens. 2007, 21, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Tabrizi, R.; Akbari, M.; Sharifi, N.; Lankarani, K.B.; Kolahdooz, F.; Taghizadeh, M.; Asemi, Z. The Effects of Coenzyme Q10 Supplementation on Blood Pressures Among Patients with Metabolic Diseases: A Systematic Review and Meta-analysis of Randomized Controlled Trials. High Blood Press. Cardiovasc. Prev. 2018, 25, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.L.; Luk, T.H.; Yiu, K.H.; Wang, M.; Yip, P.M.C.; Lee, S.W.L.; Li, S.W.; Tam, S.; Fong, B.; Lau, C.P.; et al. Reversal of mitochondrial dysfunction by coenzyme Q10 supplement improves endothelial function in patients with ischaemic left ventricular systolic dysfunction: A randomized controlled trial. Atherosclerosis 2011, 216, 395–401. [Google Scholar] [CrossRef]
- Lim, S.C.; Lekshminarayanan, R.; Goh, S.K.; Ong, Y.Y.; Subramaniam, T.; Sum, C.F.; Ong, C.N.; Lee, B.L. The effect of coenzyme Q10 on microcirculatory endothelial function of subjects with type 2 diabetes mellitus. Atherosclerosis 2008, 196, 966–969. [Google Scholar] [CrossRef]
- Hamilton, S.J.; Chew, G.T.; Watts, G.F. Coenzyme Q10 improves endothelial dysfunction in statin-treated type 2 diabetic patients. Diabetes Care 2009, 32, 810–812. [Google Scholar] [CrossRef] [PubMed]
- Borghi, C.; Cicero, A.F.G. Nutraceuticals with a clinically detectable blood pressure-lowering effect: A review of available randomized clinical trials and their meta-analyses. Br. J. Clin. Pharmacol. 2017, 83, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Fogacci, F.; Colletti, A. Commentary to: “The Effects of Coenzyme Q10 Supplementation on Blood Pressures Among Patients with Metabolic Diseases: A Systematic Review and Meta-analysis of Randomized Controlled Trials”. Hig. Blood Press. Cardiovasc. Prev. 2018, 25, 51–52. [Google Scholar] [CrossRef]
- Paglialunga, S.; Ludzki, A.; Root-McCaig, J.; Holloway, G.P. In adipose tissue, increased mitochondrial emission of reactive oxygen species is important for short-term high-fat diet-induced insulin resistance in mice. Diabetologia 2015, 58, 1071–1080. [Google Scholar] [CrossRef]
- Anderson, E.J.; Lustig, M.E.; Boyle, K.E.; Woodlief, T.L.; Kane, D.A.; Lin, C.T.; Price, J.W.; Kang, L.; Rabinovitch, P.S.; Szeto, H.H.; et al. Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J. Clin. Investig. 2009, 119, 573–581. [Google Scholar] [CrossRef]
- Fazakerley, D.J.; Chaudhuri, R.; Yang, P.; Maghzal, G.J.; Thomas, K.C.; Krycer, J.R.; Humphrey, S.J.; Parker, B.L.; Fisher-Wellman, K.H.; Meoli, C.C.; et al. Mitochondrial CoQ deficiency is a common driver of mitochondrial oxidants and insulin resistance. ELife 2018, 7, e32111. [Google Scholar] [CrossRef]
- Raygan, F.; Rezavandi, Z.; Dadkhah Tehrani, S.; Farrokhian, A.; Asemi, Z. The effects of coenzyme Q10 administration on glucose homeostasis parameters, lipid profiles, biomarkers of inflammation and oxidative stress in patients with metabolic syndrome. Eur. J. Nutr. 2016, 55, 2357–2364. [Google Scholar] [CrossRef] [PubMed]
- Saboori, S.; Rad, E.Y.; Mardani, M.; Khosroshahi, M.Z.; Nouri, Y.; Falahi, E. Effect of Q10 supplementation on body weight and body mass index: A systematic review and meta-analysis of randomized controlled clinical trials. Diabetes Metab. Syndr. 2019, 13, 1179–1185. [Google Scholar] [CrossRef] [PubMed]
- Araújo, A.R.; Rosso, N.; Bedogni, G.; Tiribelli, C.; Bellentani, S. Global epidemiology of non-alcoholic fatty liver disease/non-alcoholic steatohepatitis: What we need in the future. Liver Int. 2018, 38, 47–51. [Google Scholar] [CrossRef]
- Buzzetti, E.; Pinzani, M.; Tsochatzis, E.A. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism 2016, 65, 1038–1048. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Tian, R.; She, Z.; Cai, J.; Li, H. Role of oxidative stress in the pathogenesis of nonalcoholic fatty liver disease. Free Radic. Biol. Med. 2020, 315–321. [Google Scholar] [CrossRef]
- Hernández-Alvarez, M.I.; Sebastian, D.; Vives, S.; Ivanova, S.; Bartoccioni, P.; Kakimoto, P.; Plana, N.; Veiga, S.R.; Hernández, V.; Vasconcelos, N.; et al. Deficient Endoplasmic Reticulum-Mitochondrial Phosphatidylserine Transfer Causes Liver Disease. Cell 2019, 177, 881–895. [Google Scholar] [CrossRef] [PubMed]
- Mourier, A.; Motori, E.; Brandt, T.; Lagouge, M.; Atanassov, I.; Galiner, A.; Rappl, G.; Brodesser, S.; Hultenby, K.; Dieterich, C.; et al. Mitofusin 2 Is Required to Maintain Mitochondrial Coenzyme Q Levels. J. Cell Biol. 2015, 208, 429–442. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Colletti, A.; Bellentani, S. Nutraceutical Approach to Non-Alcoholic Fatty Liver Disease (NAFLD): The Available Clinical Evidence. Nutrients 2018, 10, 1153. [Google Scholar] [CrossRef]
- Chen, K.; Chen, X.; Xue, H.; Zhang, P.; Fang, W.; Chen, X.; Ling, W. Coenzyme Q10 attenuates high-fat diet-induced non-alcoholic fatty liver disease through activation of the AMPK pathway. Food Funct. 2019, 10, 814–823. [Google Scholar] [CrossRef]
- Moazen, M.; Mazloom, Z.; Dabbaghmanesh, M.H.; Ahmadi, A. Effect of CoQ10 supplementation on blood pressure, inflammation, and lipid profile in type 2 diabetics. Iran. J. Nutr. Sci. Food Technol. 2013, 8, 145–153. [Google Scholar]
- Pala, R.; Orhan, C.; Tuzcu, M.; Sahin, N.; Ali, S.; Cinar, V.; Atalay, M.; Sahin, K. Coenzyme Q10 Supplementation Modulates NFκB and Nrf2 Pathways in Exercise Training. J. Sports Sci. Med. 2016, 15, 196–203. [Google Scholar] [PubMed]
- Tiefenbach, J.; Magomedova, L.; Liu, J.; Reunov, A.A.; Tsai, R.; Eappen, N.S.; Jockusch, R.A.; Nislow, C.; Cummins, C.L.; Krause, H.M. Idebenone and coenzyme Q10 are novel PPARα/γ ligands, with potential for treatment of fatty liver diseases. Dis. Model. Mech. 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Farsi, F.; Mohammadshahi, M.; Alavinejad, P.; Rezazadeh, A.; Zarei, M.; Engali, K.A. Functions of Coenzyme Q10 Supplementation on Liver Enzymes, Markers of Systemic Inflammation, and Adipokines in Patients Affected by Nonalcoholic Fatty Liver Disease: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial. J. Am. Coll. Nutr. 2016, 35, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.J.; Tseng, Y.F.; Yen, C.H.; Lin, P.T. Effects of coenzyme Q10 supplementation (300 mg/day) on antioxidation and anti-inflammation in coronary artery disease patients during statins therapy: A randomized, placebo-controlled trial. Nutr. J. 2013, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gokbel, H.; Gergerlioglu, H.S.; Okudan, N.; Belviranli, M. Effects of coenzyme Q10 supplementation on plasma adiponectin, interleukin-6, and tumor necrosis factor-alpha levels in men. J. Med. Food 2010, 13, 216–218. [Google Scholar] [CrossRef]
- Farhangi, M.A.; Alipour, B.; Jafarvand, E.; Khoshbaten, M. Oral coenzyme Q10 supplementation in patients with nonalcoholic fatty liver disease: Effects on serum vaspin, chemerin, pentraxin 3, insulin resistance and oxidative stress. Arch. Med. Res. 2014, 45, 589–595. [Google Scholar] [CrossRef]
- Samimi, M.; Zarezade Mehrizi, M.; Foroozanfard, F.; Akbari, H.; Jamilian, M.; Asemi, Z. The effects of coenzyme Q10 supplementation on glucose metabolism and lipid profiles in women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled trial. Clin. Endocrinol. 2017, 86, 560–566. [Google Scholar] [CrossRef]
- Rahmani, E.; Jamilian, M.; Samimi, M.; Zarezade Mehrizi, M.; Aghadavod, E.; Akbari, E.; Tamtaji, O.R.; Asemi, Z. The effects of coenzyme Q10 supplementation on gene expression related to insulin, lipid and inflammation in patients with polycystic ovary syndrome. Gynecol. Endocrinol. 2018, 34, 217–222. [Google Scholar] [CrossRef]
- Izadi, A.; Shirazi, S.; Taghizadeh, S.; Gargari, B.P. Independent and Additive Effects of Coenzyme Q10 and Vitamin E on Cardiometabolic Outcomes and Visceral Adiposity in Women with Polycystic Ovary Syndrome. Arch. Med. Res. 2019, 50, 1–10. [Google Scholar] [CrossRef]
- Ates, O.; Bilen, H.; Keles, S.; Hakan Alp, H.; Keleş, M.S.; Yıldırım, K.; Ondaş, O.; Pınar, L.C.; Civelekler, C.; Baykal, O. Plasma coenzyme Q10 levels in type 2 diabetic patients with retinopathy. Int. J. Ophthalmol. 2013, 6, 675–679. [Google Scholar] [CrossRef]
- El-ghoroury, E.A.; Raslan, H.M.; Badawy, E.A.; El-Saaid, G.S.; Agybi, M.H.; Siam, I.; Salem, S.I. Malondialdehyde and coenzyme Q10 in platelets and serum in type 2 diabetes mellitus: Correlation with glycemic control. Blood Coagul. Fibrinolysis 2009, 20, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.; Yamamoto, Y. Simultaneous detection of ubiquinol and ubiquinone in human plasma as a marker of oxidative stress. Anal. Biochem. 1997, 250, 66–73. [Google Scholar] [CrossRef]
- Hasegawa, G.; Yamamoto, Y.; Zhi, J.G.; Yamasaki, M.; Yano, M.; Nakajima, T.; Fukui, M.; Yoshikawa, T.; Nakamura, N. Daily profile of plasma %CoQ10 level, a biomarker of oxidative stress, in patients with diabetes manifesting postprandial hyperglycaemia. Acta Diabetol. 2005, 42, 179–181. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Chi, H.; Liao, D.; Zou, Y. Effects of coenzyme Q10 on cardiovascular and metabolic biomarkers in overweight and obese patients with type 2 diabetes mellitus: A pooled analysis. Diabetes Metab. Syndr. Obes. 2018, 11, 875–886. [Google Scholar] [CrossRef] [PubMed]
- Maheshwari, R.A.; Balaraman, R.; Sen, A.K.; Seth, A.K. Effect of coenzyme Q10 alone and its combination with metformin on streptozotocin-nicotinamide-induced diabetic nephropathy in rats. Indian J. Pharmacol. 2014, 46, 627–632. [Google Scholar] [CrossRef] [PubMed]
- Maheshwari, R.; Balaraman, R.; Sen, A.K.; Shukla, D.; Seth, A. Effect of concomitant administration of coenzyme Q10 with sitagliptin on experimentally induced diabetic nephropathy in rats. Ren. Fail. 2017, 39, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.K.; Lee, J.O.; Kim, J.H.; Kim, N.; You, G.; Moon, J.W.; Sha, J.; Kim, S.J.; Lee, Y.W.; Kang, H.J.; et al. Coenzyme Q10 increases the fatty acid oxidation through AMPK-mediated PPARalpha induction in 3T3-L1 preadipocytes. Cell Signal. 2012, 24, 2329–2336. [Google Scholar] [CrossRef] [PubMed]
- Feige, J.N.; Gelman, L.; Michalik, L.; Desvergne, B.; Wahli, W. From molecular action to physiological outputs: Peroxisome proliferator activated receptors are nuclear receptors at the crossroads of key cellular functions. Prog. Lipid Res. 2006, 45, 120–159. [Google Scholar] [CrossRef]
- Tsai, K.L.; Chen, L.H.; Chiou, S.H.; Chiou, G.Y.; Chen, Y.C.; Chou, H.Y.; Chen, L.K.; Chen, H.Y.; Chiu, T.H.; Tsai, C.S.; et al. Coenzyme Q10 suppresses oxLDL-induced endothelial oxidative injuries by the modulation of LOX-1-mediated ROS generation via the AMPK/PKC/NADPH oxidase signaling pathway. Mol. Nutr. Food Res. 2011, 55, S227–S240. [Google Scholar] [CrossRef]
- Kaikkonen, J.; Nyyssönen, K.; Porkkala-Sarataho, E.; Poulsen, H.E.; Metsä-Ketelä, T.; Hayn, M.; Salonen, R.; Salonen, J.T. Effect of oral coenzyme Q10 supplementation on the oxidation resistance of human VLDL+LDL fraction: Absorption and antioxidative properties of oil and granule-based preparations. Free Radic. Biol. Med. 1997, 22, 1195–1202. [Google Scholar] [CrossRef]
- Sarmiento, A.; Diaz-Castro, J.; Pulido-Moran, M.; Moreno-Fernandez, J.; Kajarabille, N.; Chirosa, I.; Guisado, I.M.; Javier Chirosa, L.; Guisado, R.; Ochoa, J.J. Short-term ubiquinol supplementation reduces oxidative stress associated with strenuous exercise in healthy adults: A randomized trial. Biofactors 2016, 42, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, N.; Tabrizi, R.; Moosazadeh, M.; Mirhosseini, N.; Lankarani, K.B.; Akbari, M.; Chamani, M.; Kolahdooz, F.; Asemi, Z. The effects of coenzyme Q10 supplementation on lipid profiles among patients with metabolic diseases: A systematic review and meta-analysis of randomized controlled trials. Curr. Pharm. Des. 2018, 24, 2729–2742. [Google Scholar] [CrossRef]
- Sahebkar, A.; Simental-Mendıa, L.E.; Stefanutti, C.; Pirro, M. Supplementation with coenzyme Q10 reduces plasma lipoprotein(a) concentrations but not other lipid indices: A systematic review and meta-analysis. Pharmacol. Res. 2016, 105, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Geovanini, G.R.; Libby, P. Atherosclerosis and inflammation: Overview and updates. Clin. Sci. 2018, 132, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Farsi, F.; Heshmati, J.; Keshtkar, A.; Irandoost, P.; Meri, A.; Akbari, A.; Jannani, L.; Morshedzadeh, N.; Vafa, M. Can Coenzyme Q10 Supplementation Effectively Reduce Human Tumor Necrosis Factor-α and interleukin-6 Levels in Chronic Inflammatory Diseases? A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Pharmacol. Res. 2019, 148, 104290. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Feng, Y.; Chen, G.C.; Qin, L.Q.; Fu, C.L.; Chen, L.H. Effects of coenzyme Q10 supplementation on inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2017, 119, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, B. What Is New in Heart Failure Management in 2017? Update on ACC/AHA Heart Failure Guidelines. Curr. Cardiol. Rep. 2018, 20, 39. [Google Scholar] [CrossRef]
- Jessup, M.; Marwick, T.H.; Ponikowski, P.; Voors, A.A.; Yancy, C.W. 2016 ESC and ACC/AHA/HFSA heart failure guideline update—What is new and why is it important? Nat. Rev. Cardiol. 2016, 13, 623–628. [Google Scholar] [CrossRef]
- Liu, L.; Eisen, H.J. Epidemiology of heart failure and scope of the problem. Cardiol. Clin. 2014, 32, 1–8. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; de Ferranti, S.; Després, J.P.; Fullerton, H.J.; et al. American Heart Association Statistics Committee; Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2016 Update: A Report from the American Heart Association. Circulation 2016, 133, e38–e360. [Google Scholar]
- Maggioni, A.P. Epidemiology of Heart Failure in Europe. Heart Fail. Clin. 2015, 11, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.; Brouwers, F.P.; Voors, A.A.; Hillege, H.L.; de Boer, R.A.; Gansevoort, R.T.; van der Harst, P.; Rienstra, M.; van Gelder, I.C.; van Veldhuisen, D.J.; et al. Sex differences in new-onset heart failure. Clin. Res. Cardiol. 2015, 104, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Kannel, W.B. Incidence and epidemiology of heart failure. Heart Fail. Rev. 2000, 5, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Florkowski, C.M.; Molyneux, S.L.; Young, J.M. Coenzyme Q10 and congestive heart failure: An evolving evidence base. Kardiol. Polska. 2015, 73, 73–79. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Folkers, K.; Vadhanavikit, S.; Mortensen, S.A. Biochemical rationale and myocardial tissue data on the effective therapy of cardiomyopathy with coenzyme Q10. Proc. Natl. Acad. Sci. USA 1985, 82, 901–904. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, N.; Yamaguchi, A.; Otaki, M.; Sawatani, O.; Minoji, T.; Tamura, H.; Atobe, M. Myocardial tissue level of coenzyme Q10 in patients with cardiac failure. Biomed. Clin. Asp. Coenzyme Q. 1984, 4, 221–229. [Google Scholar]
- Judy, W.V.; Stogsdill, W.W.; Folkers, K. Myocardial preservation by therapy with coenzyme Q10 during heart surgery. Clin. Investig. 1993, 71, 155–161. [Google Scholar] [CrossRef]
- Weber, C.; Bysted, A.; Hłlmer, G. The coenzyme Q10 content of the average Danish diet. Int. J. Vitam. Nutr. Res. 1997, 67, 123–129. [Google Scholar]
- Onur, S.; Niklowitz, P.; Jacobs, G.; Lieb, W.; Menke, T.; Döring, F. Association between serum level of ubiquinol and NT-proBNP, a marker for chronic heart failure, in healthy elderly subjects. Biofactors 2015, 41, 35–43. [Google Scholar] [CrossRef]
- Mortensen, S.A.; Rosenfeldt, F.; Kumar, A.; Dolliner, P.; Filipiak, K.J.; Pella, D.; Alehagen, U.; Steurer, G.; Littarru, G.P. Q-SYMBIO Study Investigators. The effect of coenzyme Q10 on morbidity and mortality in chronic heart failure: Results from Q-SYMBIO: A randomized double-blind trial. JACC Heart Fail. 2014, 2, 641–649. [Google Scholar] [CrossRef]
- Lei, L.; Liu, Y. Efficacy of coenzyme Q10 in patients with cardiac failure: A meta-analysis of clinical trials. BMC Cardiovasc. Disord. 2017, 17, 196. [Google Scholar] [CrossRef] [PubMed]
- Fotino, A.D.; Thompson-Paul, A.M.; Bazzano, L.A. Effect of coenzyme Q10 supplementation on heart failure: A meta-analysis. Am. J. Clin. Nutr. 2013, 97, 268–275. [Google Scholar] [CrossRef]
- Sander, S.; Coleman, C.I.; Patel, A.A.; Kluger, J.; White, C.M. The impact of coenzyme Q10 on systolic function in patients with chronic heart failure. J. Card Fail. 2006, 12, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Belardinelli, R.; Mucaj, A.; Lacalaprice, F.; Solenghi, M.; Principi, F.; Tiano, L.; Littarru, G.P. Coenzyme Q10 improves contractility of dysfunctional myocardium in chronic heart failure. Biofactors 2005, 25, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Munkholm, H.; Hansen, H.H.; Rasmussen, K. Coenzyme Q10 treatment in serious heart failure. Biofactors 1999, 9, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Keogh, A.; Fenton, S.; Leslie, C.; Aboyoun, C.; Macdonald, P.; Zhao, Y.C.; Bailey, M.; Rosenfeldt, F. Randomised double-blind, placebo-controlled trial of coenzyme Q, therapy in class II and III systolic heart failure. Heart Lung Circ. 2003, 12, 135–141. [Google Scholar] [CrossRef]
- Langsjoen, P.H. Lack of effect of coenzyme Q on left ventricular function in patients with congestive heart failure. J. Am. Coll. Cardiol. 2000, 35, 816–817. [Google Scholar] [CrossRef][Green Version]
- Soja, A.M.; Mortensen, S.A. Treatment of congestive heart failure with coenzyme Q10 illuminated by metaanalysis of clinical trials. Mol. Asp. Med. 1997, 18, 159–168. [Google Scholar] [CrossRef]
- Swedberg, K.; Hoffman-Bang, C.; Rehnqvist, N.; Astrom, H. Coenzyme Q10 as adjunctive in treatment of congestive heart failure. J. Card Fail. 1995, 1, 101–107. [Google Scholar] [CrossRef]
- Shi, H.; Noguchi, N.; Niki, E. Dynamics of antioxidant action of ubiquinol: A reappraisal. Biofactors 1999, 9, 141–148. [Google Scholar] [CrossRef]
- Morisco, C.; Trimuco, B.; Condorelh, M. Effect of coenzyme therapy in patients with congestive heart failure: A long term multicentre randomized study. Clin. Investig. 1993, 71, S134–S136. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, S.A.; Leth, A.; Agner, E.; Rohde, M. Coenzyme Q10: Clinical benefits with biochemical correlates suggesting a scientific breakthrough in the management of chronic heart failure. Int. J. Tissue React. 1990, 12, 155–162. [Google Scholar] [PubMed]
- Beyer, R. An analysis of coenzyme Q in free radical generation and as an antioxidant. Biochem. Cell Biol. 1992, 70, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Niibori, K.; Wroblewski, K.P.; Yokoyama, H.; Juan, A.; Crestanello, J.A.; Whitman, G.J.R. Bioenergetic effect of liposomal coenzyme Q10 on myocardial ischaemia reperfusion injury. Biofactors 1999, 9, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Cohn, J.N.; Ferrari, R.; Sharpe, N. Cardiac remodelling. Concepts and clinical implications: A consensus paper from an international forum on cardiac remodeling. Behalf of an International Forum on Cardiac Remodeling. J. Am. Coll. Cardiol. 2000, 35, 569–582. [Google Scholar] [CrossRef]
- Ulla, A.; Mohamed, M.K.; Sikder, B.; Rahman, A.T.; Sumi, F.A.; Hossain, M.; Mahmud, H.; Rahman, G.M.S.; Alam, M.A. Coenzyme Q10 prevents oxidative stress and fibrosis in isoprenaline induced cardiac remodeling in aged rats. BMC Pharmacol. Toxicol. 2017, 18. [Google Scholar] [CrossRef]
- Singh, R.B.; Niaz, M.A.; Rastogi, S.S.; Sharma, J.P.; Kumar, R.; Bishnoi, I.; Beegom, R. Plasma levels of antioxidant vitamins and oxidative stress in patients with suspected acute myocardial infarction. Acta Cardiol. 1994, 49, 411–452. [Google Scholar]
- Grech, E.D.; Jackson, M.; Ramsdale, D.R. Reperfusion injury after acute myocardial infarction. Br. Med. J. 1995, 310, 477–478. [Google Scholar] [CrossRef]
- Singh, R.B.; Fedacko, J.; Mojto, V.; Pella, D. Coenzyme Q10 Modulates Remodeling Possibly by Decreasing Angiotensin-Converting Enzyme in Patients with Acute Coronary Syndrome. Antioxidants 2018, 7, 99. [Google Scholar] [CrossRef]
- Dhalla, A.K.; Hill, M.; Singal, P.K. Role of oxidative stress in the transition of hypertrophy to heart failure. J. Am. Coll. Cardiol. 1996, 28, 506–514. [Google Scholar] [CrossRef]
- Senior, R.; Basu, S.; Kinsey, C.; Schaeffer, S.; Lahiri, A. Carvidilol prevents remodeling in patients with left ventricular dysfunction after acute myocardial infarction. Am. Heart J. 1999, 137, 646–652. [Google Scholar] [CrossRef]
- Khaper, N.; Singal, P.K. Effects of after load reducing drugs on the pathogenesis of antioxidant changes and congestive heart failure in rats. J. Am. Coll. Cardiol. 1997, 219, 856–861. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Levy, D.; Vasan, R.S.; Leip, E.P.; Wolf, P.A.; D’Agostino, R.B.; Kannel, W.B.; Benjamin, E.J. Temporal relations of atrial fibrillation and congestive heart failure and their joint influence on mortality: The Framingham Heart Study. Circulation 2003, 107, 2920–2925. [Google Scholar] [CrossRef] [PubMed]
- Maisel, W.H.; Stevenson, L.W. Atrial fibrillation in heart failure: Epidemiology, pathophysiology, and rationale for therapy. Am. J. Cardiol. 2003, 91, 2D–8D. [Google Scholar] [CrossRef]
- Hynes, B.J.; Luck, J.C.; Wolbrette, D.L.; Bhatta, L.; Khan, L.; Samii, S.; Naccarelli, G.V. Atrial fibrillation in patients with heart failure. Curr. Opin. Cardiol. 2003, 18, 32–38. [Google Scholar] [CrossRef]
- Zozina, V.I.; Covantev, S.; Goroshko, O.A.; Krasnykh, L.M.; Kukes, V.G. Coenzyme Q10 in Cardiovascular and Metabolic Diseases: Current State of the Problem. Curr. Cardiol. Rev. 2018, 14, 164–174. [Google Scholar] [CrossRef]
- Kumar, A.; Kaur, H.; Devi, P.; Mohan, V. Role of coenzyme Q10 (CoQ10) in cardiac disease, hypertension and Meniere-like syndrome. Pharmacol. Ther. 2009, 124, 259–268. [Google Scholar] [CrossRef]
- de Frutos, F.; Gea, A.; Hernandez-Estefania, R.; Rabago, G. Prophylactic treatment with coenzyme Q10 in patients undergoing cardiac surgery: Could an antioxidant reduce complications? A systematic review and meta-analysis. Interact. Cardiovasc. Thorac. Surg. 2015, 20, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Kebbati, A.H.; Zhang, Y.; Tang, Y.; Okello, E.; Huang, C. Effect of coenzyme Q10 on the incidence of atrial fibrillation in patients with heart failure. J. Investig. Med. 2015, 63, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Senes, M.; Erbay, A.R.; Yilmaz, F.M.; Topkaya, C.; Zengi, O.; Dogan, M.; Yucel, D. Coenzyme Q10 and high-sensitivity C-reactive protein in ischemic and idiopathic dilated cardiomyopathy. Clin. Chem. Lab. Med. 2008, 46, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Manzoli, U.; Rossi, E.; Littarru, G.P.; Frustaci, A.; Lippa, S.; Oradei, A.; Aureli, V. Coenzyme Q10 in dilated cardiomyopathy. Int. J. Tissue React. 1990, 12, 173–178. [Google Scholar]
- Soongswang, J.; Sangtawesin, C.; Durongpisitkul, K. The effect of coenzyme Q10 on idiopathic chronic dilated cardiomyopathy in children. Pediatr. Cardiol. 2005, 26, 361–366. [Google Scholar] [CrossRef]
- Kocharian, A.; Shabanian, R.; Rafiei-Khorgami, M.; Kiani, A.; Heidari-Bateni, G. Coenzyme Q10 improves diastolic function in children with idiopathic dilated cardiomyopathy. Cardiol. Young 2009, 19, 501–506. [Google Scholar] [CrossRef]
- Langsjoen, P.H.; Langsjoen, A.; Willis, R.; Folkers, K. Treatment of hypertrophic cardiomyopathy with coenzyme Q10. Mol. Asp. Med. 1997, 18, S145–S151. [Google Scholar] [CrossRef]
- Adarsh, K.; Kaur, H.; Mohan, V. Coenzyme Q10 (CoQ10) in isolated diastolic heart failure in hypertrophic cardiomyopathy (HCM). Biofactors 2008, 32, 145–149. [Google Scholar] [CrossRef]
- Conklin, K.A. Coenzyme Q10 for prevention of anthracycline-induced cardiotoxicity. Integr. Cancer Ther. 2005, 4, 110–130. [Google Scholar] [CrossRef] [PubMed]
- Greenlee, H.; Shaw, J.; Lau, Y.I.; Naini, A.; Maurer, M. Lack of effect of coenzyme Q10 on doxorubicin cytotoxicity in breast cancer cell cultures. Integr. Cancer Ther. 2012, 11, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, H.N.; Hegazy, G.A.; Awdan, S.A.E.; AbdelBaset, M. Protective role of COQ10 or L-carnitine on the integrity of the myocardium in doxorubicin induced toxicity. Tissue Cell 2017, 49, 410–426. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, M.A.; Lo, E.H.; Iadecola, C. The science of stroke: Mechanisms in search of treatments. Neuron 2010, 67, 181–198. [Google Scholar] [CrossRef]
- Kleinig, T.J.; Vink, R. Suppression of inflammation in ischemic and hemorrhagic stroke: Therapeutic options. Curr. Opin. Neurol. 2009, 22, 294–301. [Google Scholar] [CrossRef]
- Simani, L.; Ryan, F.; Hashemifard, S.; Hooshmandi, E.; Madahi, M.; Sahraei, Z.; Razaei, O.; Heydari, K.; Ramezami, M. Serum Coenzyme Q10 is associated with clinical neurological outcomes in acute stroke patients. J. Mol. Neurosci. 2018, 31, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.; Bayraktutan, U. Oxidative stress and its role in the pathogenesis of ischaemic stroke. Int. J. Stroke 2009, 4, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Nasoohi, S.; Simani, L.; Khodagholi, F.; Nikseresht, S.; Faizi, M.; Naderi, N. Coenzyme Q10 supplementation improves acute outcomes of stroke in rats pretreated with atorvastatin. Nutr. Neurosci. 2017, 22, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Geng, J. Glial fibrillary acidic protein as a prognostic marker of acute ischemic stroke. Hum. Exp. Toxicol. 2018, 37, 1048–1053. [Google Scholar] [CrossRef]
- Sharpe, P.C.; Mulholland, C.; Trinick, T. Ascorbate and malondialdehyde in stroke patients. Ir. J. Med. Sci. 1994, 163, 488–491. [Google Scholar] [CrossRef]
- Milanlioglu, A.; Aslan, M.; Ozkol, H.; Çilingir, V.; Nuri Aydın, M.; Karadas, S. Serum antioxidant enzymes activities and oxidative stress levels in patients with acute ischemic stroke: Influence on neurological status and outcome. Wien. Klin Wochenschr. 2016, 128, 169–174. [Google Scholar] [CrossRef]
- Ramezani, M.; Sahraei, Z.; Simani, L.; Heydari, K.; Shahidi, F. Coenzyme Q10 supplementation in acute ischemic stroke: Is it beneficial in short-term administration? Nutr. Neurosci. 2018, 1–6. [Google Scholar] [CrossRef]
- Girndt, M.; Seibert, E. Premature cardiovascular disease in chronic renal failure (CRF): A model for an advanced ageing process. Exp. Gerontol. 2010, 45, 797–800. [Google Scholar] [CrossRef]
- Go, A.S.; Chertow, G.M.; Fan, D.; McCulloch, C.E.; Hsu, C.Y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef]
- Stenvinkel, P.; Diczfalusy, U.; Lindholm, B.; Heimburger, O. Phospholipid plasmalogen, a surrogate marker of oxidative stress, is associated with increased cardiovascular mortality in patients on renal replacement therapy. Nephrol. Dial. Transplant. 2004, 19, 972–976. [Google Scholar] [CrossRef] [PubMed]
- Himmelfarb, J.; McMenamin, M.E.; Loseto, G.; Heinecke, J.W. Myeloperoxidase-catalyzed 3-chlorotyrosine formation in dialysis patients. Free Radic. Biol. Med. 2001, 31, 1163–1169. [Google Scholar] [CrossRef]
- Mehmetoglu, I.; Yerlikaya, F.H.; Kurban, S.; Erdem, S.S.; Tonbul, Z. Oxidative stress markers in hemodialysis and peritoneal dialysis patients, including coenzyme Q10 and ischemia-modified albumin. Int. J. Artif. Organs 2012, 35, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Bakhshayeshkaram, M.; Lankarani, K.B.; Mirhosseini, N.; Tabrizi, R.; Akbari, M.; Dabbaghmanesh, M.H.; Asemi, Z. The Effects of Coenzyme Q10 Supplementation on Metabolic Profiles of Patients with Chronic Kidney Disease: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Curr. Pharm. Des. 2018, 24, 3710–3723. [Google Scholar] [CrossRef] [PubMed]
- Gholnari, T.; Aghadavod, E.; Soleimani, A.; Hamidi, G.A.; Sharifi, N.; Asemi, Z. The Effects of coenzyme Q10 supplementation on glucose metabolism, lipid profiles, inflammation, and oxidative stress in patients with diabetic nephropathy: A randomized, double-blind, placebo-controlled trial. J. Am. Coll. Nutr. 2018, 37, 188–193. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, Z.; Liu, Q.; Quan, H.; Cheng, X. Effects of coenzyme Q10 intervention on diabetic kidney disease: A systematic review and meta-analysis. Medicine 2019, 98, e15850. [Google Scholar] [CrossRef]
- Shojaei, M.; Djalali, M.; Khatami, M.; Siassi, F.; Eshraghian, M. Effects of carnitine and coenzyme Q10 on lipid profile and serum levels of lipoprotein(a) in maintenance hemodialysis patients on statin therapy. Iran. J. Kidney Dis. 2011, 5, 114–118. [Google Scholar]
- Fallah, M.; Askari, G.; Soleimani, A.; Feizi, A.; Asemi, Z. Clinical trial of the effects of coenzyme Q10 supplementation on glycemic control and markers of lipid profiles in diabetic hemodialysis patients. Int. Urol. Nephrol. 2018, 50, 2073–2079. [Google Scholar] [CrossRef]
- Zahed, N.S.; Ghassami, M.; Nikbakht, H. Effects of coenzyme Q10 supplementation on C-reactive protein and homocysteine as the inflammatory markers in hemodialysis patients; a randomized clinical trial. J. Nephropathol. 2016, 5, 38–43. [Google Scholar] [CrossRef]
- Rivara, M.B.; Yeung, C.K.; Robinson-Cohen, C.; Phillips, B.R.; Ruzinski, J.; Rock, D.; Linke, L.; Shen, D.D.; Ikizler, T.A.; Himmelfarb, J. Effect of Coenzyme Q10 on Biomarkers of Oxidative Stress and Cardiac Function in Hemodialysis Patients: The CoQ10 Biomarker Trial. Am. J. Kidney Dis. 2017, 69, 389–399. [Google Scholar] [CrossRef]
- Starr, M.C.; Chang, I.J.; Finn, L.S.; Sun, A.; Larson, A.A.; Goebel, J.; Hanevold, C.; Thies, J.; Van Hove, J.L.K.; Hingorani, S.R.; et al. COQ2 nephropathy: A treatable cause of nephrotic syndrome in children. Pediatr. Nephrol. 2018, 33, 1257–1261. [Google Scholar] [CrossRef]
- Mahmoud, A.N.; Mentias, A.; Elgendy, A.Y.; Qazi, A.; Barakat, A.F.; Saad, M.; Mohsen, A.; Abuzaid, A.; Mansoor, H.; Mojadidi, M.K.; et al. Migraine and the risk of cardiovascular and cerebrovascular events: A meta-analysis of 16 cohort studies including 1152407 subjects. BMJ Open 2018, 8, e020498. [Google Scholar] [CrossRef] [PubMed]
- Hershey, A.D.; Powers, S.W.; Vockell, A.L.; Lecates, S.L.; Ellinor, P.L.; Segers, A.; Burdine, D.; Manning, P.; Kabbouche, M.A. Coenzyme Q10 deficiency and response to supplementation in pediatric and adolescent migraine. Headache 2007, 47, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Parohan, M.; Sarraf, P.; Javanbakht, M.H.; Ranji-Burachaloo, S.; Djalali, M. Effect of coenzyme Q10 supplementation on clinical features of migraine: A systematic review and dose-response meta-analysis of randomized controlled trials. Nutr. Neurosci. 2019, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Li, Y.; Lu, S.; Huang, W.; Di, W. Efficacy of CoQ10 as supplementation for migraine: A meta-analysis. Acta Neurol. Scand. 2019, 139, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, T.; Pringsheim, T. Nutraceuticals in Migraine: A Summary of Existing Guidelines for Use. Headache 2016, 56, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Dahri, M.; Tarighat-Esfanjani, A.; Asghari-Jafarabadi, M.; Hashemilar, M. Oral coenzyme Q10 supplementation in patients with migraine: Effects on clinical features and inflammatory markers. Nutr. Neurosci. 2018, 3, 1–9. [Google Scholar] [CrossRef]
- Abdollahzad, H.; Aghdashi, M.A.; Asghari Jafarabadi, M.; Alipour, B. Effects of Coenzyme Q10 Supplementation on Inflammatory Cytokines (TNF-α, IL-6) and Oxidative Stress in Rheumatoid Arthritis Patients: A Randomized Controlled Trial. Arch. Med. Res. 2015, 46, 527–533. [Google Scholar] [CrossRef]
- Gaul, C.; Diener, H.C.; Danesch, U.; Migravent® Study Group. Improvement of migraine symptoms with a proprietary supplement containing riboflavin, magnesium and Q10: A randomized, placebo-controlled, double-blind, multicenter trial. J. Headache Pain 2015, 16, 516. [Google Scholar] [CrossRef]
- Parohan, M.; Sarraf, P.; Javanbakht, M.H.; Foroushani, A.R.; Ranji-Burachaloo, S.; Djalali, M. The synergistic effects of nano-curcumin and coenzyme Q10 supplementation in migraine prophylaxis: A randomized, placebo-controlled, double-blind trial. Nutr. Neurosci. 2019, 26, 1–10. [Google Scholar] [CrossRef]
- Fogacci, S.; Fogacci, F.; Cicero, A.F.G. Nutraceuticals and Hypertensive Disorders in Pregnancy: The Available Clinical Evidence. Nutrients 2020, 12, 378. [Google Scholar] [CrossRef]
- WHO. Recommendations for Prevention and Treatment of Pre-Eclampsia and Eclampsia; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Rana, S.; Lemoine, E.; Granger, J.; Karumanchi, S.A. Preeclampsia: Pathophysiology, Challenges, and Perspectives. Circ. Res. 2019, 124, 1094–1112. [Google Scholar] [CrossRef] [PubMed]
- Teran, E.; Racines-Orbe, M.; Vivero, S.; Escudero, C.; Molina, G.; Calle, A. Preeclampsia is associated with a decrease in plasma coenzyme Q10 levels. Free Radic. Biol. Med. 2003, 35, 1453–1456. [Google Scholar] [CrossRef] [PubMed]
- Palan, P.R.; Shaban, D.W.; Martino, T.; Mikhail, M.S. Lipid-soluble antioxidants and pregnancy: Maternal serum levels of coenzyme Q10, alpha-tocopherol and gamma-tocopherol in preeclampsia and normal pregnancy. Gynecol. Obstet. Investig. 2004, 58, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Teran, E.; Hernandez, I.; Nieto, B.; Tavara, R.; Ocampo, J.E.; Calle, A. Coenzyme Q10 supplementation during pregnancy reduces the risk of pre-eclampsia. Int. J. Gynaecol. Obstet. 2009, 105, 43–45. [Google Scholar] [CrossRef]
- Tenório, M.B.; Ferreira, R.C.; Moura, F.A.; Bueno, N.B.; Goulart, M.O.F.; Oliveira, A.C.M. Oral antioxidant therapy for prevention and treatment of preeclampsia: Meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 865–876. [Google Scholar] [CrossRef] [PubMed]
- Corsini, A. Statin-Related Muscle Complaints: An Underestimated Risk. Cardiovasc. Drugs Ther. 2005, 19, 379–381. [Google Scholar] [CrossRef]
- Deichmann, R.; Lavie, C.; Andrews, S. Coenzyme Q10 and statin-induced mitochondrial dysfunction. Ochsner J. 2010, 10, 16–21. [Google Scholar]
- Qu, H.; Meng, Y.Y.; Chai, H.; Liang, F.; Zhang, J.Y.; Gao, Z.Y.; Shi, D.Z. The effect of statin treatment on circulating coenzyme Q10 concentrations: An updated meta-analysis of randomized controlled trials. Eur. J. Med. Res. 2018, 23, 57. [Google Scholar] [CrossRef]
- Qu, H.; Guo, M.; Chai, H.; Wang, W.T.; Gao, Z.Y.; Shi, D.Z. Effects of Coenzyme Q10 on Statin-Induced Myopathy: An Updated Meta-Analysis of Randomized Controlled Trials. J. Am. Heart Assoc. 2018, 7, e009835. [Google Scholar] [CrossRef]
- Mizuno, K.; Tanaka, M.; Nozaki, S.; Mizuma, M.; Ataka, S.; Tahara, T.; Sugino, T.; Shirai, T.; Kajimoto, Y.; Kuratsune, H.; et al. Antifatigue effects of coenzyme Q10 during physical fatigue. Nutrition 2008, 24, 293–299. [Google Scholar] [CrossRef]
- Lee, Y.J.; Cho, W.J.; Kim, J.K.; Lee, D.C. Effects of coenzyme Q10 on arterial stiffness, metabolic parameters, and fatigue in obese subjects: A double-blind randomized controlled study. J. Med. Food 2011, 14, 386–390. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; Cano-García, F.J.; Alcocer-Gómez, E.; de Miguel, M.; Sànchez-Alcàzar, J.A. Oxidative stress correlates with headache symptoms in fibromyalgia: Coenzyme Q10 effect on clinical improvement. PLoS ONE 2012, 7, e35677. [Google Scholar] [CrossRef] [PubMed]
- Di Pierro, F.; Rossi, A.; Consensi, A.; Giacomelli, C.; Bazzichi, L. Role for a water-soluble form of CoQ10 in female subjects affected by fibromyalgia. A preliminary study. Clin. Exp. Rheumatol. 2017, 35, 20–27. [Google Scholar]
- Kikkawa, K.; Takehara, I.; Miyakoshi, T.; Miyawaki, H. Safety of High Dose Supplementation of Coenzyme Q10 in Healthy Human Adults. Jpn. J. Food Chem. 2007, 14, 76–81. [Google Scholar]
- Bentinger, M.; Dallner, G.; Choknacki, T.; Swiezewska, E. Distrinution and breakdown of labeled coenzyme Q10 in rats. Free Radic. Biol. Med. 2003, 34, 563–575. [Google Scholar] [CrossRef]
- Nukui, K.; Yamagishi, T.; Miyawaki, H.; Kettawan, A.; Okamoto, T.; Sato, K. Comparison of uptake between PureSorb-Q40 and regular hydrophobic coenzyme Q10 in rats and humans after single oral intake. J. Nutr. Sci. Vitaminol. 2007, 53, 187–190. [Google Scholar] [CrossRef][Green Version]
- Bhagavan, H.N.; Chopra, R.K. Coenzyme Q10: Absorption, tissue uptake, metabolism and pharmacokinetics. Free Radic. Res. 2006, 40, 445–453. [Google Scholar] [CrossRef]
- Bhagavan, H.N.; Chopra, R.K. Plasma coenzyme Q10 response to oral ingestion of coenzyme Q10 formulations. Mitochondrion 2007, 7, S78–S88. [Google Scholar] [CrossRef]
- Tomono, Y.; Hasegawa, J.; Seki, T.; Motegi, K.; Morishita, N. Pharmacokinetic study of deuterium-labeled Coenzyme-Q10 in man. Int. J. Clin. Pharmacol. Ther. 1986, 24, 536–541. [Google Scholar]
- Weis, M.; Mortensen, S.A.; Rassing, M.R.; Møller-Sonnergaard, J.; Poulsen, G.; Rasmussen, S.N. Bioavailability of four oral coenzyme Q10 formulations in healthy volunteers. Mol. Asp. Med. 1994, 15, 273–280. [Google Scholar] [CrossRef]
- Miles, M.V.; Horn, P.; Miles, L.; Tang, P.; Steele, P.; De Grauw, T. Bioequivalence of coenzyme Q10 from over-the-counter supplements. Nutr. Res. 2002, 22, 919–929. [Google Scholar] [CrossRef]
- Kumar, S.; Rao, R.; Kumar, A.; Mahant, S.; Nanda, S. Novel Carriers for Coenzyme Q10 Delivery. Curr. Drug Deliv. 2016, 13, 1184–1204. [Google Scholar] [CrossRef] [PubMed]
- Pravst, I.; Zmitek, K. The coenzyme Q10 content of food supplements. J. Verbrauch. Lebensm. J. Consum. Prot. Food Saf. 2011, 6, 457–463. [Google Scholar] [CrossRef]
- Pravst, I.; Prosek, M.; Wondra, A.G.; Zmitek, K.; Zmitek, J. The Stability of Coenzyme Q10 in Fortified Foods. Acta Chim. Slov. 2009, 56, 953–958. [Google Scholar]
- Evans, M.; Baisley, J.; Barss, S.; Guthrie, N. A randomized, double-blind trial on the bioavailability of two CoQ10 formulations. J. Funct. Foods 2009, 1, 65–73. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, J.; Chen, X.Q.; Chen, C.Y.O. Ubiquinol is superior to ubiquinone to enhance Coenzyme Q10 status in older men. Food Funct. 2018, 9, 5653–5659. [Google Scholar] [CrossRef] [PubMed]
- Zmitek, J.; Smidovnik, A.; Fir, M.; Prosek, M.; Zmitek, K.; Walczak, J.; Pravst, I. Relative bioavailability of two forms of a novel water soluble Coenzyme Q10. Ann. Nutr. Metab. 2008, 52, 281–287. [Google Scholar] [CrossRef]
- López-Lluch, G.; Del Pozo-Cruz, J.; Sánchez-Cuesta, A.; Cortés-Rodríguez, A.B.; Navas, P. Bioavailability of coenzyme Q10 supplements depends on carrier lipids and solubilization. Nutrition 2019, 57, 133–140. [Google Scholar] [CrossRef]
- Weber, C.; Bysted, A.; Hølmer, G. Coenzyme Q10 in the diet-daily intake and relative bioavailability. Mol. Asp. Med. 1997, 18, S251–S254. [Google Scholar] [CrossRef]
- Wajda, R.; Zirkel, J.; Schaffer, T. Increase of bioavailability of coenzyme Q10 and vitamin E. J. Med. Food 2007, 10, 731–734. [Google Scholar] [CrossRef]
- Martinefski, M.; Samassa, P.; Buontempo, F.; Höcht, C.; Lucangioli, S.; Tripodi, V. Relative bioavailability of coenzyme Q10 formulation for paediatric individualized therapy. J. Pharm. Pharmacol. 2017, 69, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Constantinescu, R.; McDermott, M.P.; Dicenzo, R. A randomized study of the bioavailability of different formulations of coenzyme Q10 (ubiquinone). J. Clin. Pharmacol. 2007, 47, 1580–1586. [Google Scholar] [CrossRef] [PubMed]
- Lucker, P.W.; Wetzelsberger, N.; Hennings, G.; Rehn, D. Pharmacokinetics of coenzyme ubidecarenone in healthy volunteers. In Biomedical and Clinical Aspects of Coenzyme Q; Elsevier: Amsterdam, The Netherlands, 1984; pp. 141–151. [Google Scholar]
- Hosoe, K.; Kitano, M.; Kishida, H.; Kubo, H.; Fujii, K.; Kitahara, M. Study on safety and bioavailability of ubiquinol (Kaneka QH) after single and 4-week multiple oral administration to healthy volunteers. Regul. Toxicol. Pharmacol. 2007, 47, 19–28. [Google Scholar] [CrossRef]
- Molyneux, S.; Florkowski, C.; Lever, M.; George, P. The bioavailability of coenzyme Q10 supplements available in New Zealand differs markedly. N. Z. Med. J. 2004, 117, U1108. [Google Scholar]
- Young, J.M.; Molyneux, S.L.; Florkowski, C.M.; Frampton, C.M.; George, P.M.; Scott, R.S. Pharmacokinetic comparison of a generic coenzyme Q10 solubilizate and a formulation with soybean phytosterols. Phytother. Res. 2012, 26, 1092–1096. [Google Scholar] [CrossRef]
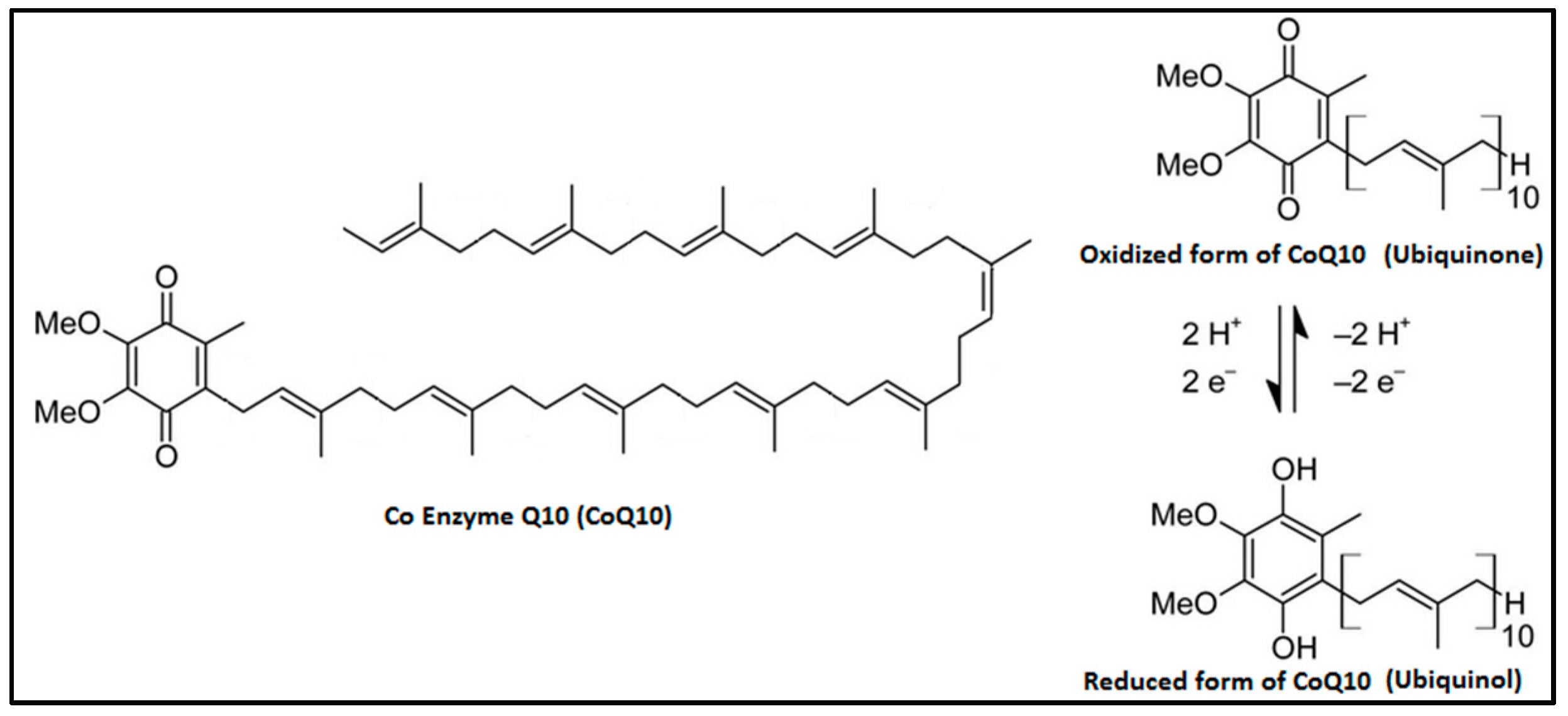
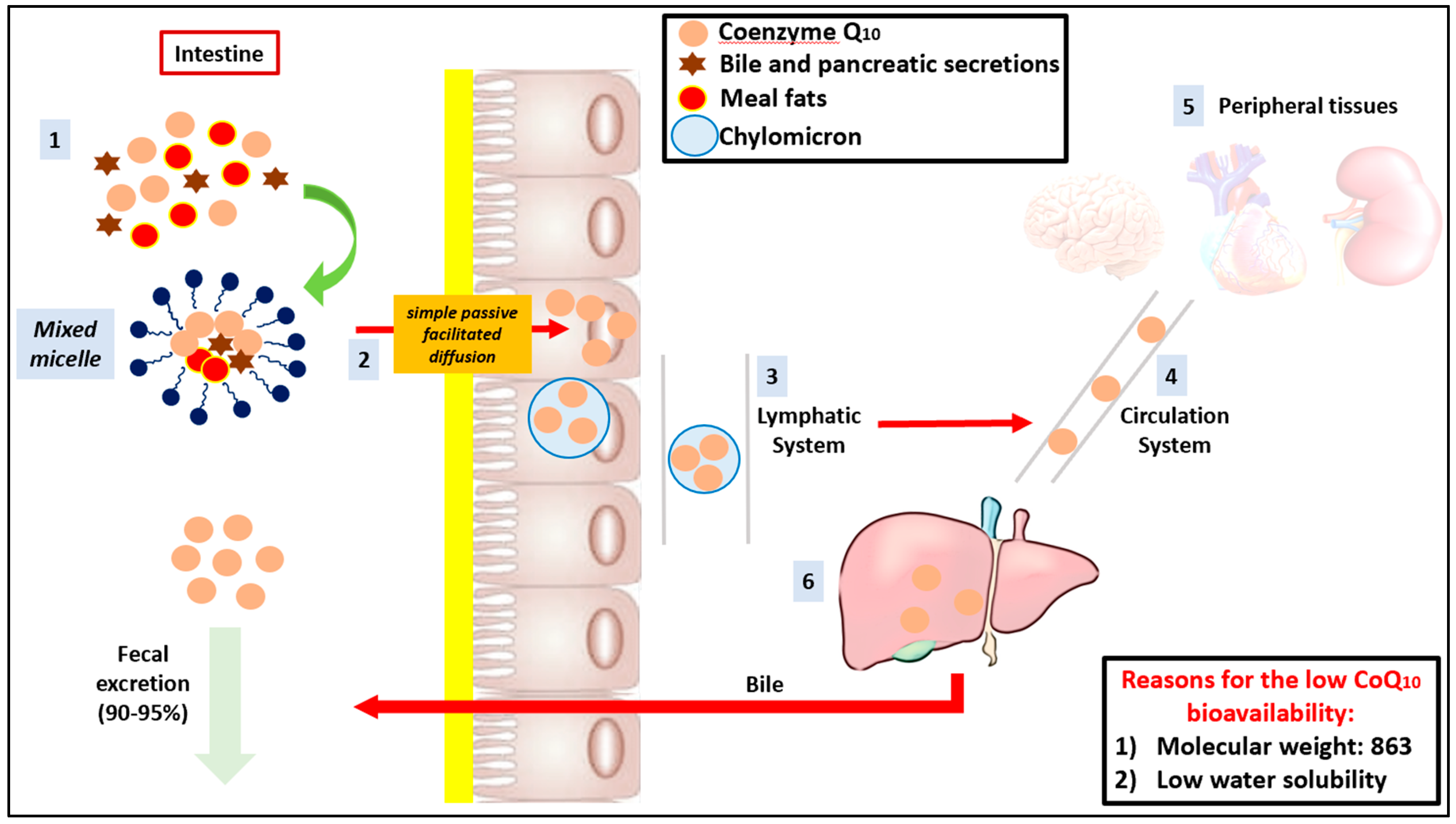
| Organ | Ubiquinone Concentration (µg/g) | Ubiquinol Concentration (µg/g) |
|---|---|---|
| Heart | 132.0 | 61.0 |
| Kidneys | 77.0 | 75.0 |
| Liver | 63.6 | 95.0 |
| Muscle | 39.7 | 65.0 |
| Brain | 13.4 | 23.0 |
| Pancreas | 32.7 | |
| Spleen | 24.6 | |
| Lung | 7.9 | 25.0 |
| Thyroid | 24.7 | |
| Testis | 10.5 | |
| Intestine | 11.5 | 95.0 |
| Colon | 10.7 | |
| Ventricle | 11.8 | |
| Plasma (µmol/mL) | 1.1 | 96.0 |
| Level of Evidence | Active Daily Doses | Effects on Symptoms and/or Grade of Disease | Effects on Lab or Instrumental Parameters | Effects on Hard Outcomes | |
|---|---|---|---|---|---|
| Heart Failure (HF) | Meta-analysis of RCTs | 100–300 mg | ↑ self-perceived quality of life and improvement in NYHA class | ↑ EF (if >30%), ↑ LVEF, ↑ CO and CI, ↑ SV, ↑ EDV, ↑ exercise capacity, ↓ ventricular arrhythmias after surgery and need of inotropic drugs (after cardiac surgery), and ↓ low-grade inflammation (TNF-alpha, IL-6, and hsCRP) | ↓ MACE, total mortality, and incidence of hospital stays for HF |
| Acute Myocardial Infarction (AMI) | RCTs | 120 mg | Not investigated | Prevention of alteration of the wall thickening abnormality at the infarct site and sphericity index and ↓ wall thickness opposite the site of infarction | Not investigated |
| Ischemic Stroke (IS) | RCTs | 300 mg | ↑ NIHSS and MMSE | Reduction of oxidative stress (?) | Not investigated |
| Atrial Fibrillation (AF) | Meta-analysis of RCTs | 100–300 mg | Improvement in NYHA class, reduction of risk to develop ventricular arrhythmias, and use of inotropic drugs after surgery | Reduction of malondialdehyde and oxidative stress | Not investigated |
| Cardiomyopathy | RCTs | 200–300 mg | Improvement of fatigue and dyspnea | Improvement of mean interventricular septal thickness, mean posterior wall thickness, diastolic function, and mean score for the index of cardiac failure | Not investigated |
| Cardiotoxicity | RCTs | 200–300 mg | Improvement of heart’s functions (in association with L-carnitine) | Reduction of oxidative stress (nitric oxide and malondialdehyde) and ↓IL-1, TNF-α Troponin-l and Troponin-T levels (in association with L-carnitine) | Not investigated |
| Hypertension | Meta-analysis of RCTs | 100–300 mg | Not reported | ↑ Exercise capacity and arterial stiffness, ↑ NO bioavailability, and ↓ SBP and DBP (only in prehypertensive or hypertensive patients) | Not investigated |
| Diabetes type II, Metabolic syndrome (MetS) | RCTs | 100–300 mg | Not reported | ↓ Lipid peroxidation, FPG, triglycerides, and low-grade inflammation (TNF-alpha, IL-6, and hsCRP) and ↑ insulin sensitivity | Not investigated |
| Dyslipidemia | RCTs | 100–300 mg | ↑ self-perceived quality of life (reduction side effects of lipid-lowering drugs) | ↑ Exercise capacity and arterial stiffness; ↓ lipid peroxidation, TC*, LDL-C*, TG*, BP*, FPG*, and low-grade inflammation (TNF-alpha, IL-6, and hsCRP); and ↑ insulin sensitivity *If >12 weeks of treatment | Not investigated |
| Non-Alcoholic Fatty Liver Disease (NAFLD) | Meta-analysis of RCTs | 100–300 mg | Improvement in NAFLD grade | ↑ Adiponectin (?) and leptin levels; ↓ AST, GGT, hsCRP, and TNF-alpha levels; and ↓ WC and lipid peroxidation | Not investigated |
| Chronic Kidney Disease (CKD) | Meta-analysis of RCTs | 100–300 mg | Not investigated | ↓ Lipid peroxidation, TC (?), LDL-C (?), Lp(a) (?), triglycerides (?), fasting plasma glucose (?), HbA1c (?), inflammation, and oxidative stress biomarkers (hsCRP (?) and malondialdehyde) and ↑ insulin sensitivity | Not investigated |
| Type of Formulation | Subjects | Tested Dosage | ΔCmax | Reference |
|---|---|---|---|---|
| Myoquinon (softgel) | Both gender (10 M, 4 F), age 18–30 | 100 mg | 1.069 | [189] |
| KOJ, CoQ10 (softgel) | Both gender (10 M, 4 F), age 18–30 | 100 mg | 0.238 | |
| ICT, CoQ10 (softgel) | Both gender (10 M, 4 F), age 18–30 | 100 mg | 0.351 | |
| ERG, CoQ10 (softgel) | Both gender (10 M, 4 F), age 18–30 | 100 mg | 0.258 | |
| Ubquinol QH (softgel) | Both gender (10 M, 4 F), age 18–30 | 100 mg | 0.473 | |
| NYD CoQ10 (hard gel) | Both gender (10 M, 4 F), age 18–30 | 100 mg | 0.381 | |
| SMF CoQ10 | Both gender (10 M, 4 F), age 18–30 | 100 mg | 0.181 | |
| Capsule CoQ10 | 9 M, age 18–30 | 30 mg | 0.31 | [190] |
| Gelatin capsule CoQ10 + vitamin E | Both gender (12 M, 12 F) | 100 mg | 0.025 | [191] |
| NanoSolve (purified phospholipids) capsule CoQ10 + vitamin E | Both gender (12 M, 12 F) | 100 mg | 0.103 | |
| Capsule CoQ10 (powder-filled hard-shell gelatine capsule) | Both gender (3 M, 3 F), age 18–40 | 250 mg | 0.490 | [192] |
| Liquid (O/W liquid emulsion (20 mg/mL)) | Both gender (3 M, 3 F), age 18–40 | 250 mg | 0.980 | |
| Chewable wafer | Both gender (15 M, 10 F), elderly people | 600 mg | 0.770 | [193] |
| Chewable wafer + 300 IU vitamin E | Both gender (15 M, 10 F), elderly people | 600 mg | 0.660 | |
| Softgel capsules (Mega Q-Gel “100”) CoQ10 solubilized in an oil-based vehicle + 900 IU d-alpha tocopherol | Both gender (15 M, 10 F), elderly people | 600 mg | 0.690 | |
| Hard gelatin capsule | Both gender (15 M, 10 F), elderly people | 600 mg | 0.660 | |
| Powder | 333 mg | 0.980 | [194] | |
| Kaneka QH, ubiquinol (softgel capsules) | Both gender (5 M, 5 F) | 150 mg | 1.061 | [195] |
| 5 M | 300 mg | 2.506 | ||
| Chewable tablets | 10 M, age 21–28 | 150 mg | 0.120 | [196] |
| Capsule liquid | 10 M, age 21–28 | 150 mg | 0.149 | |
| Capsule liquid | 10 M, age 21–28 | 150 mg | 0.152 | |
| Capsule powder | 10 M, age 21–28 | 150 mg | 0.175 | |
| Capsule liquid | 10 M, age 21–28 | 150 mg | 0.197 | |
| Softgel | 10 M, age 21–28 | 150 mg | 0.277 | |
| Q-gel (CoQ10 solubilized in an oil-based vehicle + vitamin E) softgel | 10 M, age 21–28 | 150 mg | 0.506 | |
| Q-gel (CoQ10 solubilized in an oil-based vehicle + vitamin E) softgel | 8 M, age 20–26 | 60 mg 150 mg 300 mg | 0.267 0.802 1.010 | |
| Softgel CoQ10 | 36 M, age 18–40 | 100 mg | 0.259 | [197] |
| Softgel CoQ10 + sterols | 36 M, age 18–40 | 100 mg | 0.189 | |
| Hardgel (CoQ10 + 400 mg 400 mg of Emcompress) | Both gender (5 M, 5 F), age 24–30 | 100 mg | 0.775 | [180] |
| Softgel Bioqinon (CoQ10 + 400 mg of soybean oil) | Both gender (5 M, 5 F), age 24–30 | 100 mg | 1.454 | |
| Softgel (CoQ10 + 20 mg of polysorbate 80, 100 mg of lecithin + 280 mg of soybean oil) | Both gender (5 M, 5 F), age 24–30 | 100 mg | 0.837 | |
| Softgel (CoQ10 + 20 mg of polysorbate 80 + 380 mg of soybean oil) | Both gender (5 M, 5 F), age 24–30 | 100 mg | 0.883 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martelli, A.; Testai, L.; Colletti, A.; Cicero, A.F.G. Coenzyme Q10: Clinical Applications in Cardiovascular Diseases. Antioxidants 2020, 9, 341. https://doi.org/10.3390/antiox9040341
Martelli A, Testai L, Colletti A, Cicero AFG. Coenzyme Q10: Clinical Applications in Cardiovascular Diseases. Antioxidants. 2020; 9(4):341. https://doi.org/10.3390/antiox9040341
Chicago/Turabian StyleMartelli, Alma, Lara Testai, Alessandro Colletti, and Arrigo F. G. Cicero. 2020. "Coenzyme Q10: Clinical Applications in Cardiovascular Diseases" Antioxidants 9, no. 4: 341. https://doi.org/10.3390/antiox9040341
APA StyleMartelli, A., Testai, L., Colletti, A., & Cicero, A. F. G. (2020). Coenzyme Q10: Clinical Applications in Cardiovascular Diseases. Antioxidants, 9(4), 341. https://doi.org/10.3390/antiox9040341








