Management of Traumatic Brain Injury: From Present to Future
Abstract
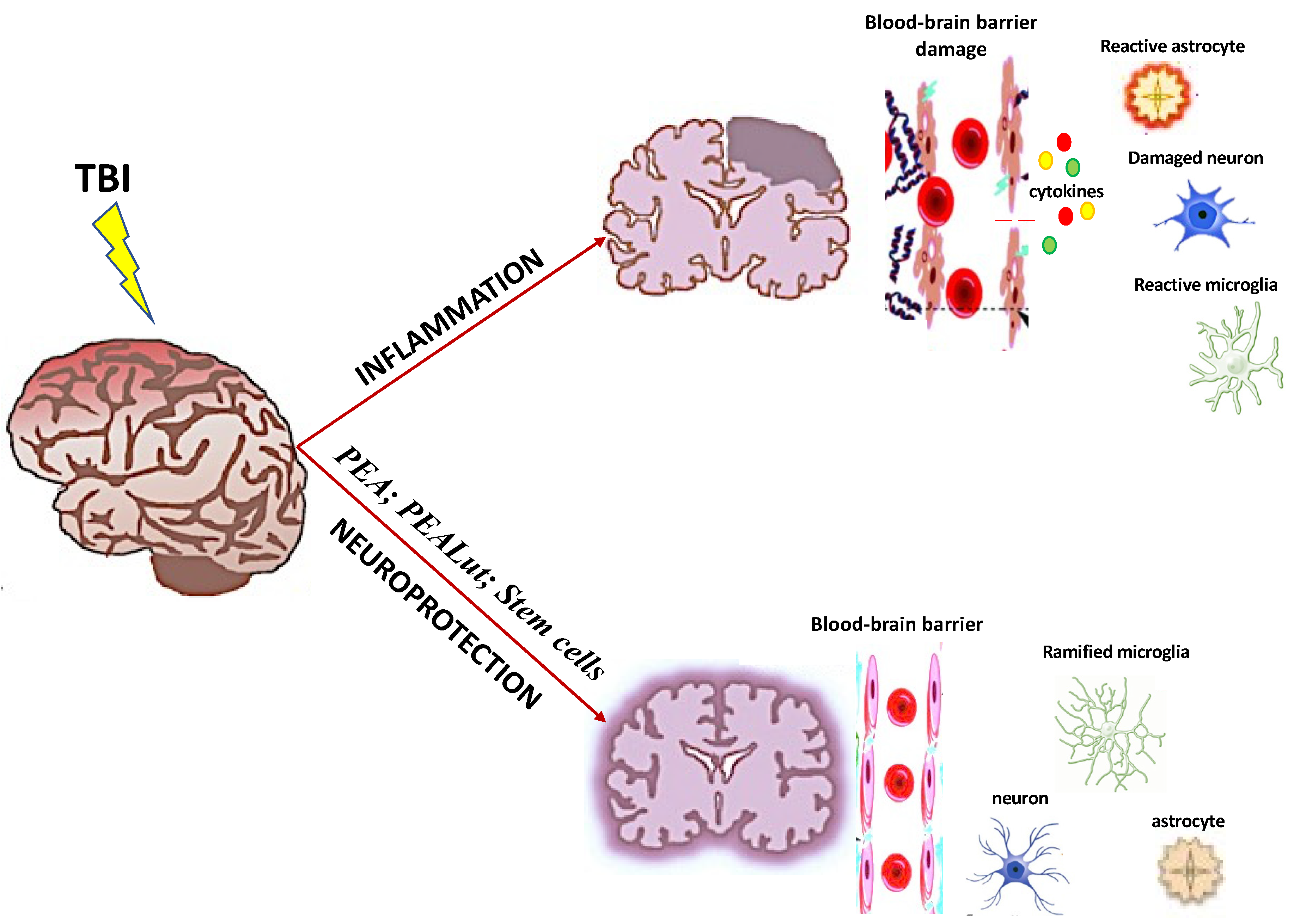
1. Introduction
2. The Pathophysiology of TBI
3. TBI and Neurotoxicity
4. TBI and Oxidative Stress
5. Biomarkers in TBI
6. Review of Existing Drug Interventions
6.1. Clinical Trials of Drugs with Anti-Inflammatory Effect
6.2. Therapeutic Strategies to manage Neuronal Recovery and Neurobehavioral Sequelae after Injury
7. New Therapeutic Strategies
8. Biologics
9. Neuropsychological Rehabilitation (NR) and Neurotherapy
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kline, A.E.; Leary, J.B.; Radabaugh, H.L.; Cheng, J.P.; Bondi, C.O. Combination therapies for neurobehavioral and cognitive recovery after experimental traumatic brain injury: Is more better? Prog. Neurobiol. 2016, 142, 45–67. [Google Scholar] [CrossRef] [PubMed]
- Logsdon, A.F.; Lucke-Wold, B.P.; Turner, R.C.; Huber, J.D.; Rosen, C.L.; Simpkins, J.W. Role of Microvascular Disruption in Brain Damage from Traumatic Brain Injury. Compr. Physiol. 2015, 5, 1147–1160. [Google Scholar] [CrossRef] [PubMed]
- Zibara, K.; Ballout, N.; Mondello, S.; Karnib, N.; Ramadan, N.; Omais, S.; Nabbouh, A.; Caliz, D.; Clavijo, A.; Hu, Z.; et al. Combination of drug and stem cells neurotherapy: Potential interventions in neurotrauma and traumatic brain injury. Neuropharmacology 2019, 145, 177–198. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Mahmood, A.; Chopp, M. Animal models of traumatic brain injury. Nat. Rev. Neurosci. 2013, 14, 128–142. [Google Scholar] [CrossRef]
- Kochanek, P.M.; Jackson, T.C.; Jha, R.; Clark, R.S.B.; Okonkwo, D.O.; Bayir, H.; Poloyac, S.M.; Wagner, A.M.D.; Empey, P.E.; Conley, Y.P.; et al. Paths to successful translation of new therapies for severe TBI in the golden age of traumatic brain injury research: A Pittsburgh vision. J. Neurotrauma 2018. [Google Scholar] [CrossRef]
- Greve, M.W.; Zink, B.J. Pathophysiology of traumatic brain injury. Mt. Sinai J. Med. 2009, 76, 97–104. [Google Scholar] [CrossRef]
- Chan, T.L.H.; Woldeamanuel, Y.W. Exploring naturally occurring clinical subgroups of post-traumatic headache. J. Headache Pain 2020, 21, 12. [Google Scholar] [CrossRef]
- Larsen, E.L.; Ashina, H.; Iljazi, A.; Al-Khazali, H.M.; Seem, K.; Ashina, M.; Ashina, S.; Schytz, H.W. Acute and preventive pharmacological treatment of post-traumatic headache: A systematic review. J. Headache Pain 2019, 20, 98. [Google Scholar] [CrossRef]
- Bedaso, A.; Geja, E.; Ayalew, M.; Oltaye, Z.; Duko, B. Post-concussion syndrome among patients experiencing head injury attending emergency department of Hawassa University Comprehensive specialized hospital, Hawassa, southern Ethiopia. J. Headache Pain 2018, 19, 112. [Google Scholar] [CrossRef]
- Niyonkuru, C.; Wagner, A.K.; Ozawa, H.; Amin, K.; Goyal, A.; Fabio, A. Group-based trajectory analysis applications for prognostic biomarker model development in severe TBI: A practical example. J. Neurotrauma 2013, 30, 938–945. [Google Scholar] [CrossRef]
- Wang, Y.; Qin, Z.H. Molecular and cellular mechanisms of excitotoxic neuronal death. Apoptosis Int. J. Program. Cell Death 2010, 15, 1382–1402. [Google Scholar] [CrossRef] [PubMed]
- Dasuri, K.; Zhang, L.; Keller, J.N. Oxidative stress, neurodegeneration, and the balance of protein degradation and protein synthesis. Free Radic. Biol. Med. 2013, 62, 170–185. [Google Scholar] [CrossRef] [PubMed]
- Mutinati, M.; Pantaleo, M.; Roncetti, M.; Piccinno, M.; Rizzo, A.; Sciorsci, R.L. Oxidative stress in neonatology: A review. Reprod. Domest. Anim. 2014, 49, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Fluiter, K.; Opperhuizen, A.L.; Morgan, B.P.; Baas, F.; Ramaglia, V. Inhibition of the membrane attack complex of the complement system reduces secondary neuroaxonal loss and promotes neurologic recovery after traumatic brain injury in mice. J. Immunol. 2014, 192, 2339–2348. [Google Scholar] [CrossRef]
- Wu, L.Y.; Bao, X.Q.; Sun, H.; Zhang, D. Scavenger receptor on astrocytes and its relationship with neuroinflammation. Acta Acad. Med. Sin. 2014, 36, 330–335. [Google Scholar] [CrossRef]
- Kumar, A.; Loane, D.J. Neuroinflammation after traumatic brain injury: Opportunities for therapeutic intervention. Brain Behav. Immun. 2012, 26, 1191–1201. [Google Scholar] [CrossRef]
- Margulies, S.; Hicks, R. Combination therapies for traumatic brain injury: Prospective considerations. J. Neurotrauma 2009, 26, 925–939. [Google Scholar] [CrossRef]
- Yang, Z.; Zhu, T.; Mondello, S.; Akel, M.; Wong, A.T.; Kothari, I.M.; Lin, F.; Shear, D.A.; Gilsdorf, J.S.; Leung, L.Y.; et al. Serum-Based Phospho-Neurofilament-Heavy Protein as Theranostic Biomarker in Three Models of Traumatic Brain Injury: An Operation Brain Trauma Therapy Study. J. Neurotrauma 2019, 36, 348–359. [Google Scholar] [CrossRef]
- Kim, H.J.; Tsao, J.W.; Stanfill, A.G. The current state of biomarkers of mild traumatic brain injury. JCI Insight 2018, 3. [Google Scholar] [CrossRef]
- Hagos, F.T.; Empey, P.E.; Wang, P.; Ma, X.; Poloyac, S.M.; Bayir, H.; Kochanek, P.M.; Bell, M.J.; Clark, R.S.B. Exploratory Application of Neuropharmacometabolomics in Severe Childhood Traumatic Brain Injury. Crit. Care Med. 2018, 46, 1471–1479. [Google Scholar] [CrossRef]
- Higashida, T.; Kreipke, C.W.; Rafols, J.A.; Peng, C.; Schafer, S.; Schafer, P.; Ding, J.Y.; Dornbos, D., 3rd; Li, X.; Guthikonda, M.; et al. The role of hypoxia-inducible factor-1alpha, aquaporin-4, and matrix metalloproteinase-9 in blood-brain barrier disruption and brain edema after traumatic brain injury. J. Neurosurg. 2011, 114, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Pan, R.; Yu, K.; Weatherwax, T.; Zheng, H.; Liu, W.; Liu, K.J. Blood Occludin Level as a Potential Biomarker for Early Blood Brain Barrier Damage Following Ischemic Stroke. Sci. Rep. 2017, 7, 40331. [Google Scholar] [CrossRef] [PubMed]
- Zongo, D.; Ribereau-Gayon, R.; Masson, F.; Laborey, M.; Contrand, B.; Salmi, L.R.; Montaudon, D.; Beaudeux, J.L.; Meurin, A.; Dousset, V.; et al. S100-B protein as a screening tool for the early assessment of minor head injury. Ann. Emerg. Med. 2012, 59, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Neher, M.D.; Keene, C.N.; Rich, M.C.; Moore, H.B.; Stahel, P.F. Serum biomarkers for traumatic brain injury. South. Med. J. 2014, 107, 248–255. [Google Scholar] [CrossRef]
- Kochanek, P.M.; Dixon, C.E.; Shellington, D.K.; Shin, S.S.; Bayir, H.; Jackson, E.K.; Kagan, V.E.; Yan, H.Q.; Swauger, P.V.; Parks, S.A.; et al. Screening of biochemical and molecular mechanisms of secondary injury and repair in the brain after experimental blast-induced traumatic brain injury in rats. J. Neurotrauma 2013, 30, 920–937. [Google Scholar] [CrossRef]
- Pham, N.; Akonasu, H.; Shishkin, R.; Taghibiglou, C. Plasma soluble prion protein, a potential biomarker for sport-related concussions: A pilot study. PLoS ONE 2015, 10, e0117286. [Google Scholar] [CrossRef]
- Rubenstein, R.; Chang, B.; Yue, J.K.; Chiu, A.; Winkler, E.A.; Puccio, A.M.; Diaz-Arrastia, R.; Yuh, E.L.; Mukherjee, P.; Valadka, A.B.; et al. Comparing Plasma Phospho Tau, Total Tau, and Phospho Tau-Total Tau Ratio as Acute and Chronic Traumatic Brain Injury Biomarkers. JAMA Neurol. 2017, 74, 1063–1072. [Google Scholar] [CrossRef]
- Papa, L.; Lewis, L.M.; Silvestri, S.; Falk, J.L.; Giordano, P.; Brophy, G.M.; Demery, J.A.; Liu, M.C.; Mo, J.; Akinyi, L.; et al. Serum levels of ubiquitin C-terminal hydrolase distinguish mild traumatic brain injury from trauma controls and are elevated in mild and moderate traumatic brain injury patients with intracranial lesions and neurosurgical intervention. J. Trauma Acute Care Surg. 2012, 72, 1335–1344. [Google Scholar] [CrossRef]
- Mondello, S.; Linnet, A.; Buki, A.; Robicsek, S.; Gabrielli, A.; Tepas, J.; Papa, L.; Brophy, G.M.; Tortella, F.; Hayes, R.L.; et al. Clinical utility of serum levels of ubiquitin C-terminal hydrolase as a biomarker for severe traumatic brain injury. Neurosurgery 2012, 70, 666–675. [Google Scholar] [CrossRef]
- Puvenna, V.; Brennan, C.; Shaw, G.; Yang, C.; Marchi, N.; Bazarian, J.J.; Merchant-Borna, K.; Janigro, D. Significance of ubiquitin carboxy-terminal hydrolase L1 elevations in athletes after sub-concussive head hits. PLoS ONE 2014, 9, e96296. [Google Scholar] [CrossRef]
- Gatson, J.W.; Barillas, J.; Hynan, L.S.; Diaz-Arrastia, R.; Wolf, S.E.; Minei, J.P. Detection of neurofilament-H in serum as a diagnostic tool to predict injury severity in patients who have suffered mild traumatic brain injury. J. Neurosurg. 2014, 121, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- Rossi, D.; Volanti, P.; Brambilla, L.; Colletti, T.; Spataro, R.; La Bella, V. CSF neurofilament proteins as diagnostic and prognostic biomarkers for amyotrophic lateral sclerosis. J. Neurol. 2018, 265, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Bohmer, A.E.; Oses, J.P.; Schmidt, A.P.; Peron, C.S.; Krebs, C.L.; Oppitz, P.P.; D’Avila, T.T.; Souza, D.O.; Portela, L.V.; Stefani, M.A. Neuron-specific enolase, S100B, and glial fibrillary acidic protein levels as outcome predictors in patients with severe traumatic brain injury. Neurosurgery 2011, 68, 1624–1630. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.; Gao, G.; Feng, J.; Jin, Y.; Wang, C.; Mao, Q.; Jiang, J. Glial fibrillary acidic protein as a biomarker in severe traumatic brain injury patients: A prospective cohort study. Crit. Care 2015, 19, 362. [Google Scholar] [CrossRef]
- Berger, R.P.; Adelson, P.D.; Pierce, M.C.; Dulani, T.; Cassidy, L.D.; Kochanek, P.M. Serum neuron-specific enolase, S100B, and myelin basic protein concentrations after inflicted and noninflicted traumatic brain injury in children. J. Neurosurg. 2005, 103, 61–68. [Google Scholar] [CrossRef]
- Mondello, S.; Robicsek, S.A.; Gabrielli, A.; Brophy, G.M.; Papa, L.; Tepas, J.; Robertson, C.; Buki, A.; Scharf, D.; Jixiang, M.; et al. alphaII-spectrin breakdown products (SBDPs): Diagnosis and outcome in severe traumatic brain injury patients. J. Neurotrauma 2010, 27, 1203–1213. [Google Scholar] [CrossRef]
- Zhao, J.; Xi, G.; Wu, G.; Keep, R.F.; Hua, Y. Deferoxamine Attenuated the Upregulation of Lipocalin-2 Induced by Traumatic Brain Injury in Rats. Acta Neurochir. Suppl. 2016, 121, 291–294. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, H.; Zhang, M.; Zhang, Y.; Qian, C.; Liu, Y.; He, S.; Zou, Y.; Liu, H. Early expression of serum neutrophil gelatinase-associated lipocalin (NGAL) is associated with neurological severity immediately after traumatic brain injury. J. Neurol. Sci. 2016, 368, 392–398. [Google Scholar] [CrossRef]
- Semple, B.D.; Bye, N.; Rancan, M.; Ziebell, J.M.; Morganti-Kossmann, M.C. Role of CCL2 (MCP-1) in traumatic brain injury (TBI): Evidence from severe TBI patients and CCL2-/- mice. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2010, 30, 769–782. [Google Scholar] [CrossRef]
- Buttram, S.D.; Wisniewski, S.R.; Jackson, E.K.; Adelson, P.D.; Feldman, K.; Bayir, H.; Berger, R.P.; Clark, R.S.; Kochanek, P.M. Multiplex assessment of cytokine and chemokine levels in cerebrospinal fluid following severe pediatric traumatic brain injury: Effects of moderate hypothermia. J. Neurotrauma 2007, 24, 1707–1717. [Google Scholar] [CrossRef]
- Berger, R.P.; Ta’asan, S.; Rand, A.; Lokshin, A.; Kochanek, P. Multiplex assessment of serum biomarker concentrations in well-appearing children with inflicted traumatic brain injury. Pediatric Res. 2009, 65, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Oliver, J.; Abbas, K.; Lightfoot, J.T.; Baskin, K.; Collins, B.; Wier, D.; Bramhall, J.P.; Huang, J.; Puschett, J.B. Comparison of Neurocognitive Testing and the Measurement of Marinobufagenin in Mild Traumatic Brain Injury: A Preliminary Report. J. Exp. Neurosci. 2015, 9, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Yeh, C.T.; Ou, J.C.; Ma, H.P.; Chen, K.Y.; Chang, C.F.; Lai, J.H.; Liao, K.H.; Lin, C.M.; Lin, S.Y.; et al. The Association of Apolipoprotein E Allele 4 Polymorphism with the Recovery of Sleep Disturbance after Mild Traumatic Brain Injury. Acta Neurol. Taiwanica 2017, 26, 13–19. [Google Scholar]
- Hayes, J.P.; Reagan, A.; Logue, M.W.; Hayes, S.M.; Sadeh, N.; Miller, D.R.; Verfaellie, M.; Wolf, E.J.; McGlinchey, R.E.; Milberg, W.P.; et al. BDNF genotype is associated with hippocampal volume in mild traumatic brain injury. Genes Brain Behav. 2018, 17, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, P.G.; Sebastian, A.H.; Hall, E.D. Therapeutic window analysis of the neuroprotective effects of cyclosporine A after traumatic brain injury. J. Neurotrauma 2011, 28, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Holmin, S.; Mathiesen, T. Dexamethasone and colchicine reduce inflammation and delayed oedema following experimental brain contusion. Acta Neurochir. 1996, 138, 418–424. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Z.; Artelt, M.; Burnet, M.; Schluesener, H.J. Dexamethasone attenuates early expression of three molecules associated with microglia/macrophages activation following rat traumatic brain injury. Acta Neuropathol. 2007, 113, 675–682. [Google Scholar] [CrossRef]
- Hakan, T.; Toklu, H.Z.; Biber, N.; Ozevren, H.; Solakoglu, S.; Demirturk, P.; Aker, F.V. Effect of COX-2 inhibitor meloxicam against traumatic brain injury-induced biochemical, histopathological changes and blood-brain barrier permeability. Neurol. Res. 2010, 32, 629–635. [Google Scholar] [CrossRef]
- Siopi, E.; Cho, A.H.; Homsi, S.; Croci, N.; Plotkine, M.; Marchand-Leroux, C.; Jafarian-Tehrani, M. Minocycline restores sAPPalpha levels and reduces the late histopathological consequences of traumatic brain injury in mice. J. Neurotrauma 2011, 28, 2135–2143. [Google Scholar] [CrossRef]
- Thau-Zuchman, O.; Shohami, E.; Alexandrovich, A.G.; Trembovler, V.; Leker, R.R. The anti-inflammatory drug carprofen improves long-term outcome and induces gliogenesis after traumatic brain injury. J. Neurotrauma 2012, 29, 375–384. [Google Scholar] [CrossRef]
- Chao, P.K.; Lu, K.T.; Jhu, J.Y.; Wo, Y.Y.; Huang, T.C.; Ro, L.S.; Yang, Y.L. Indomethacin protects rats from neuronal damage induced by traumatic brain injury and suppresses hippocampal IL-1beta release through the inhibition of Nogo-A expression. J. Neuroinflamm. 2012, 9, 121. [Google Scholar] [CrossRef] [PubMed]
- Girgis, H.; Palmier, B.; Croci, N.; Soustrat, M.; Plotkine, M.; Marchand-Leroux, C. Effects of selective and non-selective cyclooxygenase inhibition against neurological deficit and brain oedema following closed head injury in mice. Brain Res. 2013, 1491, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Clond, M.A.; Lee, B.S.; Yu, J.J.; Singer, M.B.; Amano, T.; Lamb, A.W.; Drazin, D.; Kateb, B.; Ley, E.J.; Yu, J.S. Reactive oxygen species-activated nanoprodrug of Ibuprofen for targeting traumatic brain injury in mice. PLoS ONE 2013, 8, e61819. [Google Scholar] [CrossRef] [PubMed]
- Cheong, C.U.; Chang, C.P.; Chao, C.M.; Cheng, B.C.; Yang, C.Z.; Chio, C.C. Etanercept attenuates traumatic brain injury in rats by reducing brain TNF- alpha contents and by stimulating newly formed neurogenesis. Mediat. Inflamm. 2013, 2013, 620837. [Google Scholar] [CrossRef]
- Campolo, M.; Ahmad, A.; Crupi, R.; Impellizzeri, D.; Morabito, R.; Esposito, E.; Cuzzocrea, S. Combination therapy with melatonin and dexamethasone in a mouse model of traumatic brain injury. J. Endocrinol. 2013, 217, 291–301. [Google Scholar] [CrossRef]
- Luo, C.L.; Li, Q.Q.; Chen, X.P.; Zhang, X.M.; Li, L.L.; Li, B.X.; Zhao, Z.Q.; Tao, L.Y. Lipoxin A4 attenuates brain damage and downregulates the production of pro-inflammatory cytokines and phosphorylated mitogen-activated protein kinases in a mouse model of traumatic brain injury. Brain Res. 2013, 1502, 1–10. [Google Scholar] [CrossRef]
- Harrison, J.L.; Rowe, R.K.; O’Hara, B.F.; Adelson, P.D.; Lifshitz, J. Acute over-the-counter pharmacological intervention does not adversely affect behavioral outcome following diffuse traumatic brain injury in the mouse. Exp. Brain Res. 2014, 232, 2709–2719. [Google Scholar] [CrossRef]
- Homsi, S.; Piaggio, T.; Croci, N.; Noble, F.; Plotkine, M.; Marchand-Leroux, C.; Jafarian-Tehrani, M. Blockade of acute microglial activation by minocycline promotes neuroprotection and reduces locomotor hyperactivity after closed head injury in mice: A twelve-week follow-up study. J. Neurotrauma 2010, 27, 911–921. [Google Scholar] [CrossRef]
- Bye, N.; Habgood, M.D.; Callaway, J.K.; Malakooti, N.; Potter, A.; Kossmann, T.; Morganti-Kossmann, M.C. Transient neuroprotection by minocycline following traumatic brain injury is associated with attenuated microglial activation but no changes in cell apoptosis or neutrophil infiltration. Exp. Neurol. 2007, 204, 220–233. [Google Scholar] [CrossRef]
- Besson, V.C.; Chen, X.R.; Plotkine, M.; Marchand-Verrecchia, C. Fenofibrate, a peroxisome proliferator-activated receptor alpha agonist, exerts neuroprotective effects in traumatic brain injury. Neurosci. Lett. 2005, 388, 7–12. [Google Scholar] [CrossRef]
- Sauerbeck, A.; Gao, J.; Readnower, R.; Liu, M.; Pauly, J.R.; Bing, G.; Sullivan, P.G. Pioglitazone attenuates mitochondrial dysfunction, cognitive impairment, cortical tissue loss, and inflammation following traumatic brain injury. Exp. Neurol. 2011, 227, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Shi, J.; Hu, Z.; Hang, C. Inhibitory effect on cerebral inflammatory response following traumatic brain injury in rats: A potential neuroprotective mechanism of N-acetylcysteine. Mediat. Inflamm. 2008, 2008, 716458. [Google Scholar] [CrossRef] [PubMed]
- Bergold, P.J. Treatment of traumatic brain injury with anti-inflammatory drugs. Exp. Neurol. 2016, 275 Pt 3, 367–380. [Google Scholar] [CrossRef]
- Chen, G.; Shi, J.X.; Hang, C.H.; Xie, W.; Liu, J.; Liu, X. Inhibitory effect on cerebral inflammatory agents that accompany traumatic brain injury in a rat model: A potential neuroprotective mechanism of recombinant human erythropoietin (rhEPO). Neurosci. Lett. 2007, 425, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Yu, Z.Y.; Ding, L.; Zheng, S.Y. Experimental studies of erythropoietin protection following traumatic brain injury in rats. Exp. Med. 2012, 4, 977–982. [Google Scholar] [CrossRef][Green Version]
- Chen, G.; Zhang, S.; Shi, J.; Ai, J.; Qi, M.; Hang, C. Simvastatin reduces secondary brain injury caused by cortical contusion in rats: Possible involvement of TLR4/NF-kappaB pathway. Exp. Neurol. 2009, 216, 398–406. [Google Scholar] [CrossRef]
- Li, B.; Mahmood, A.; Lu, D.; Wu, H.; Xiong, Y.; Qu, C.; Chopp, M. Simvastatin attenuates microglial cells and astrocyte activation and decreases interleukin-1beta level after traumatic brain injury. Neurosurgery 2009, 65, 179–185. [Google Scholar] [CrossRef]
- Si, D.; Li, J.; Liu, J.; Wang, X.; Wei, Z.; Tian, Q.; Wang, H.; Liu, G. Progesterone protects blood-brain barrier function and improves neurological outcome following traumatic brain injury in rats. Exp. Med. 2014, 8, 1010–1014. [Google Scholar] [CrossRef]
- Cutler, S.M.; Cekic, M.; Miller, D.M.; Wali, B.; VanLandingham, J.W.; Stein, D.G. Progesterone improves acute recovery after traumatic brain injury in the aged rat. J. Neurotrauma 2007, 24, 1475–1486. [Google Scholar] [CrossRef]
- Orsucci, D.; Calsolaro, V.; Mancuso, M.; Siciliano, G. Neuroprotective effects of tetracyclines: Molecular targets, animal models and human disease. CNS Neurol. Disord. Drug Targets 2009, 8, 222–231. [Google Scholar] [CrossRef]
- Villapol, S.; Yaszemski, A.K.; Logan, T.T.; Sanchez-Lemus, E.; Saavedra, J.M.; Symes, A.J. Candesartan, an angiotensin II AT(1)-receptor blocker and PPAR-gamma agonist, reduces lesion volume and improves motor and memory function after traumatic brain injury in mice. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2012, 37, 2817–2829. [Google Scholar] [CrossRef] [PubMed]
- Mandrekar-Colucci, S.; Sauerbeck, A.; Popovich, P.G.; McTigue, D.M. PPAR agonists as therapeutics for CNS trauma and neurological diseases. ASN Neuro 2013, 5, e00129. [Google Scholar] [CrossRef] [PubMed]
- Di Giovanni, S.; Movsesyan, V.; Ahmed, F.; Cernak, I.; Schinelli, S.; Stoica, B.; Faden, A.I. Cell cycle inhibition provides neuroprotection and reduces glial proliferation and scar formation after traumatic brain injury. Proc. Natl. Acad. Sci. USA 2005, 102, 8333–8338. [Google Scholar] [CrossRef] [PubMed]
- Kabadi, S.V.; Stoica, B.A.; Byrnes, K.R.; Hanscom, M.; Loane, D.J.; Faden, A.I. Selective CDK inhibitor limits neuroinflammation and progressive neurodegeneration after brain trauma. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2012, 32, 137–149. [Google Scholar] [CrossRef]
- Peng, W.; Xing, Z.; Yang, J.; Wang, Y.; Wang, W.; Huang, W. The efficacy of erythropoietin in treating experimental traumatic brain injury: A systematic review of controlled trials in animal models. J. Neurosurg. 2014, 121, 653–664. [Google Scholar] [CrossRef]
- Robertson, C.S.; Hannay, H.J.; Yamal, J.M.; Gopinath, S.; Goodman, J.C.; Tilley, B.C.; Epo Severe, T.B.I.T.I.; Baldwin, A.; Rivera Lara, L.; Saucedo-Crespo, H.; et al. Effect of erythropoietin and transfusion threshold on neurological recovery after traumatic brain injury: A randomized clinical trial. JAMA 2014, 312, 36–47. [Google Scholar] [CrossRef]
- Xiong, Y.; Zhang, Y.; Mahmood, A.; Chopp, M. Investigational agents for treatment of traumatic brain injury. Expert Opin. Investig. Drugs 2015, 24, 743–760. [Google Scholar] [CrossRef]
- Meythaler, J.; Fath, J.; Fuerst, D.; Zokary, H.; Freese, K.; Martin, H.B.; Reineke, J.; Peduzzi-Nelson, J.; Roskos, P.T. Safety and feasibility of minocycline in treatment of acute traumatic brain injury. Brain Inj. 2019, 33, 679–689. [Google Scholar] [CrossRef]
- Lu, D.; Goussev, A.; Chen, J.; Pannu, P.; Li, Y.; Mahmood, A.; Chopp, M. Atorvastatin reduces neurological deficit and increases synaptogenesis, angiogenesis, and neuronal survival in rats subjected to traumatic brain injury. J. Neurotrauma 2004, 21, 21–32. [Google Scholar] [CrossRef]
- Wu, H.; Lu, D.; Jiang, H.; Xiong, Y.; Qu, C.; Li, B.; Mahmood, A.; Zhou, D.; Chopp, M. Increase in phosphorylation of Akt and its downstream signaling targets and suppression of apoptosis by simvastatin after traumatic brain injury. J. Neurosurg. 2008, 109, 691–698. [Google Scholar] [CrossRef]
- Wu, H.; Lu, D.; Jiang, H.; Xiong, Y.; Qu, C.; Li, B.; Mahmood, A.; Zhou, D.; Chopp, M. Simvastatin-mediated upregulation of VEGF and BDNF, activation of the PI3K/Akt pathway, and increase of neurogenesis are associated with therapeutic improvement after traumatic brain injury. J. Neurotrauma 2008, 25, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, A.; Goussev, A.; Kazmi, H.; Qu, C.; Lu, D.; Chopp, M. Long-term benefits after treatment of traumatic brain injury with simvastatin in rats. Neurosurgery 2009, 65, 187–191. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Head, B.P.; Patel, H.H.; Insel, P.A. Interaction of membrane/lipid rafts with the cytoskeleton: Impact on signaling and function: Membrane/lipid rafts, mediators of cytoskeletal arrangement and cell signaling. Biochim. Biophys. Acta 2014, 1838, 532–545. [Google Scholar] [CrossRef] [PubMed]
- Tobinick, E. Perispinal etanercept: A new therapeutic paradigm in neurology. Expert Rev. Neurother. 2010, 10, 985–1002. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tobinick, E.; Kim, N.M.; Reyzin, G.; Rodriguez-Romanacce, H.; DePuy, V. Selective TNF inhibition for chronic stroke and traumatic brain injury: An observational study involving 629 consecutive patients treated with perispinal etanercept. CNS Drugs 2012, 26, 1051–1070. [Google Scholar] [CrossRef]
- Tobinick, E.; Rodriguez-Romanacce, H.; Levine, A.; Ignatowski, T.A.; Spengler, R.N. Immediate neurological recovery following perispinal etanercept years after brain injury. Clin. Drug Investig. 2014, 34, 361–366. [Google Scholar] [CrossRef]
- Hoffer, M.E.; Balaban, C.; Slade, M.D.; Tsao, J.W.; Hoffer, B. Amelioration of acute sequelae of blast induced mild traumatic brain injury by N-acetyl cysteine: A double-blind, placebo controlled study. PLoS ONE 2013, 8, e54163. [Google Scholar] [CrossRef]
- Clark, R.S.B.; Empey, P.E.; Bayir, H.; Rosario, B.L.; Poloyac, S.M.; Kochanek, P.M.; Nolin, T.D.; Au, A.K.; Horvat, C.M.; Wisniewski, S.R.; et al. Phase I randomized clinical trial of N-acetylcysteine in combination with an adjuvant probenecid for treatment of severe traumatic brain injury in children. PLoS ONE 2017, 12, e0180280. [Google Scholar] [CrossRef]
- Stein, D.G. Embracing failure: What the Phase III progesterone studies can teach about TBI clinical trials. Brain Inj. 2015, 29, 1259–1272. [Google Scholar] [CrossRef]
- Atkins, C.M.; Oliva, A.A., Jr.; Alonso, O.F.; Pearse, D.D.; Bramlett, H.M.; Dietrich, W.D. Modulation of the cAMP signaling pathway after traumatic brain injury. Exp. Neurol. 2007, 208, 145–158. [Google Scholar] [CrossRef]
- Ceyhan, O.; Birsoy, K.; Hoffman, C.S. Identification of biologically active PDE11-selective inhibitors using a yeast-based high-throughput screen. Chem. Biol. 2012, 19, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Kaminska, K.; Golembiowska, K.; Rogoz, Z. Effect of risperidone on the fluoxetine-induced changes in extracellular dopamine, serotonin and noradrenaline in the rat frontal cortex. Pharmacol. Rep. Pr. 2013, 65, 1144–1151. [Google Scholar] [CrossRef]
- Huot, P.; Johnston, T.H.; Lewis, K.D.; Koprich, J.B.; Reyes, M.G.; Fox, S.H.; Piggott, M.J.; Brotchie, J.M. UWA-121, a mixed dopamine and serotonin re-uptake inhibitor, enhances L-DOPA anti-parkinsonian action without worsening dyskinesia or psychosis-like behaviours in the MPTP-lesioned common marmoset. Neuropharmacology 2014, 82, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Chew, E.; Zafonte, R.D. Pharmacological management of neurobehavioral disorders following traumatic brain injury--a state-of-the-art review. J. Rehabil. Res. Dev. 2009, 46, 851–879. [Google Scholar] [CrossRef] [PubMed]
- Buckley, C.D.; Gilroy, D.W.; Serhan, C.N.; Stockinger, B.; Tak, P.P. The resolution of inflammation. Nat. Rev. Immunol. 2013, 13, 59–66. [Google Scholar] [CrossRef]
- Pacher, P.; Batkai, S.; Kunos, G. The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol. Rev. 2006, 58, 389–462. [Google Scholar] [CrossRef]
- Skaper, S.D.; Facci, L.; Giusti, P. Mast cells, glia and neuroinflammation: Partners in crime? Immunology 2014, 141, 314–327. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Bruschetta, G.; Cordaro, M.; Crupi, R.; Siracusa, R.; Esposito, E.; Cuzzocrea, S. Micronized/ultramicronized palmitoylethanolamide displays superior oral efficacy compared to nonmicronized palmitoylethanolamide in a rat model of inflammatory pain. J. Neuroinflamm. 2014, 11, 136. [Google Scholar] [CrossRef]
- Boccella, S.; Marabese, I.; Iannotta, M.; Belardo, C.; Neugebauer, V.; Mazzitelli, M.; Pieretti, G.; Maione, S.; Palazzo, E. Metabotropic Glutamate Receptor 5 and 8 Modulate the Ameliorative Effect of Ultramicronized Palmitoylethanolamide on Cognitive Decline Associated with Neuropathic Pain. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef]
- Scuderi, C.; Bronzuoli, M.R.; Facchinetti, R.; Pace, L.; Ferraro, L.; Broad, K.D.; Serviddio, G.; Bellanti, F.; Palombelli, G.; Carpinelli, G.; et al. Ultramicronized palmitoylethanolamide rescues learning and memory impairments in a triple transgenic mouse model of Alzheimer’s disease by exerting anti-inflammatory and neuroprotective effects. Transl. Psychiatry 2018, 8, 32. [Google Scholar] [CrossRef]
- Fusco, R.; Gugliandolo, E.; Campolo, M.; Evangelista, M.; Di Paola, R.; Cuzzocrea, S. Effect of a new formulation of micronized and ultramicronized N-palmitoylethanolamine in a tibia fracture mouse model of complex regional pain syndrome. PLoS ONE 2017, 12, e0178553. [Google Scholar] [CrossRef] [PubMed]
- Impellizzeri, D.; Peritore, A.F.; Cordaro, M.; Gugliandolo, E.; Siracusa, R.; Crupi, R.; D’Amico, R.; Fusco, R.; Evangelista, M.; Cuzzocrea, S.; et al. The neuroprotective effects of micronized PEA (PEA-m) formulation on diabetic peripheral neuropathy in mice. FASEB J. 2019, 33, 11364–11380. [Google Scholar] [CrossRef] [PubMed]
- Passavanti, M.B.; Fiore, M.; Sansone, P.; Aurilio, C.; Pota, V.; Barbarisi, M.; Fierro, D.; Pace, M.C. The beneficial use of ultramicronized palmitoylethanolamide as add-on therapy to Tapentadol in the treatment of low back pain: A pilot study comparing prospective and retrospective observational arms. BMC Anesth. 2017, 17, 171. [Google Scholar] [CrossRef] [PubMed]
- Schweiger, V.; Martini, A.; Bellamoli, P.; Donadello, K.; Schievano, C.; Balzo, G.D.; Sarzi-Puttini, P.; Parolini, M.; Polati, E. Ultramicronized Palmitoylethanolamide (um-PEA) as Add-on Treatment in Fibromyalgia Syndrome (FMS): Retrospective Observational Study on 407 Patients. CNS Neurol. Disord. Drug Targets 2019, 18, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Crupi, R.; Paterniti, I.; Ahmad, A.; Campolo, M.; Esposito, E.; Cuzzocrea, S. Effects of palmitoylethanolamide and luteolin in an animal model of anxiety/depression. CNS Neurol. Disord. Drug Targets 2013, 12, 989–1001. [Google Scholar] [CrossRef] [PubMed]
- Bertolino, B.; Crupi, R.; Impellizzeri, D.; Bruschetta, G.; Cordaro, M.; Siracusa, R.; Esposito, E.; Cuzzocrea, S. Beneficial Effects of Co-Ultramicronized Palmitoylethanolamide/Luteolin in a Mouse Model of Autism and in a Case Report of Autism. CNS Neurosci. Ther. 2017, 23, 87–98. [Google Scholar] [CrossRef]
- Crupi, R.; Impellizzeri, D.; Bruschetta, G.; Cordaro, M.; Paterniti, I.; Siracusa, R.; Cuzzocrea, S.; Esposito, E. Co-Ultramicronized Palmitoylethanolamide/Luteolin Promotes Neuronal Regeneration after Spinal Cord Injury. Front. Pharmacol. 2016, 7, 47. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Esposito, E.; Di Paola, R.; Ahmad, A.; Campolo, M.; Peli, A.; Morittu, V.M.; Britti, D.; Cuzzocrea, S. Palmitoylethanolamide and luteolin ameliorate development of arthritis caused by injection of collagen type II in mice. Arthritis Res. Ther. 2013, 15, R192. [Google Scholar] [CrossRef]
- Paterniti, I.; Cordaro, M.; Campolo, M.; Siracusa, R.; Cornelius, C.; Navarra, M.; Cuzzocrea, S.; Esposito, E. Neuroprotection by association of palmitoylethanolamide with luteolin in experimental Alzheimer’s disease models: The control of neuroinflammation. CNS Neurol. Disord. Drug Targets 2014, 13, 1530–1541. [Google Scholar] [CrossRef]
- Paterniti, I.; Impellizzeri, D.; Di Paola, R.; Navarra, M.; Cuzzocrea, S.; Esposito, E. A new co-ultramicronized composite including palmitoylethanolamide and luteolin to prevent neuroinflammation in spinal cord injury. J. Neuroinflamm. 2013, 10, 91. [Google Scholar] [CrossRef]
- Siracusa, R.; Paterniti, I.; Impellizzeri, D.; Cordaro, M.; Crupi, R.; Navarra, M.; Cuzzocrea, S.; Esposito, E. The Association of Palmitoylethanolamide with Luteolin Decreases Neuroinflammation and Stimulates Autophagy in Parkinson’s Disease Model. CNS Neurol. Disord. Drug Targets 2015, 14, 1350–1365. [Google Scholar] [CrossRef] [PubMed]
- Cordaro, M.; Impellizzeri, D.; Paterniti, I.; Bruschetta, G.; Siracusa, R.; De Stefano, D.; Cuzzocrea, S.; Esposito, E. Neuroprotective Effects of Co-UltraPEALut on Secondary Inflammatory Process and Autophagy Involved in Traumatic Brain Injury. J. Neurotrauma 2016, 33, 132–146. [Google Scholar] [CrossRef] [PubMed]
- Siracusa, R.; Impellizzeri, D.; Cordaro, M.; Crupi, R.; Esposito, E.; Petrosino, S.; Cuzzocrea, S. Anti-Inflammatory and Neuroprotective Effects of Co-UltraPEALut in a Mouse Model of Vascular Dementia. Front. Neurol. 2017, 8, 233. [Google Scholar] [CrossRef] [PubMed]
- Caltagirone, C.; Cisari, C.; Schievano, C.; Di Paola, R.; Cordaro, M.; Bruschetta, G.; Esposito, E.; Cuzzocrea, S.; Stroke Study Group. Co-ultramicronized Palmitoylethanolamide/Luteolin in the Treatment of Cerebral Ischemia: From Rodent to Man. Transl. Stroke Res. 2016, 7, 54–69. [Google Scholar] [CrossRef] [PubMed]
- Impellizzeri, D.; Cordaro, M.; Bruschetta, G.; Crupi, R.; Pascali, J.; Alfonsi, D.; Marcolongo, G.; Cuzzocrea, S. 2-pentadecyl-2-oxazoline: Identification in coffee, synthesis and activity in a rat model of carrageenan-induced hindpaw inflammation. Pharmacol. Res. 2016, 108, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Petrosino, S.; Campolo, M.; Impellizzeri, D.; Paterniti, I.; Allara, M.; Gugliandolo, E.; D’Amico, R.; Siracusa, R.; Cordaro, M.; Esposito, E.; et al. 2-Pentadecyl-2-Oxazoline, the Oxazoline of Pea, Modulates Carrageenan-Induced Acute Inflammation. Front. Pharmacol. 2017, 8, 308. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Cordaro, M.; Bruschetta, G.; Siracusa, R.; Crupi, R.; Esposito, E.; Cuzzocrea, S. N-Palmitoylethanolamine-Oxazoline as a New Therapeutic Strategy to Control Neuroinflammation: Neuroprotective Effects in Experimental Models of Spinal Cord and Brain Injury. J. Neurotrauma. 2017, 34, 2609–2623. [Google Scholar] [CrossRef]
- Mouhieddine, T.H.; Kobeissy, F.H.; Itani, M.; Nokkari, A.; Wang, K.K. Stem cells in neuroinjury and neurodegenerative disorders: Challenges and future neurotherapeutic prospects. Neural Regen. Res. 2014, 9, 901–906. [Google Scholar] [CrossRef]
- Liao, G.P.; Harting, M.T.; Hetz, R.A.; Walker, P.A.; Shah, S.K.; Corkins, C.J.; Hughes, T.G.; Jimenez, F.; Kosmach, S.C.; Day, M.C.; et al. Autologous bone marrow mononuclear cells reduce therapeutic intensity for severe traumatic brain injury in children. Pediatr. Crit. Care Med. J. Soc. Crit. Care Med. World Fed. Pediatr. Intensive Crit. Care Soc. 2015, 16, 245–255. [Google Scholar] [CrossRef]
- Blaya, M.O.; Tsoulfas, P.; Bramlett, H.M.; Dietrich, W.D. Neural progenitor cell transplantation promotes neuroprotection, enhances hippocampal neurogenesis, and improves cognitive outcomes after traumatic brain injury. Exp. Neurol. 2015, 264, 67–81. [Google Scholar] [CrossRef]
- Thau-Zuchman, O.; Shohami, E.; Alexandrovich, A.G.; Leker, R.R. Combination of vascular endothelial and fibroblast growth factor 2 for induction of neurogenesis and angiogenesis after traumatic brain injury. J. Mol. Neurosci. 2012, 47, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Lv, Q.; Fan, X.; Xu, G.; Liu, Q.; Tian, L.; Cai, X.; Sun, W.; Wang, X.; Cai, Q.; Bao, Y.; et al. Intranasal delivery of nerve growth factor attenuates aquaporins-4-induced edema following traumatic brain injury in rats. Brain Res. 2013, 1493, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Guo, R.; Yue, X.; Lv, Q.; Ye, X.; Wang, Z.; Chen, Z.; Wu, B.; Xu, G.; Liu, X. Intranasal administration of nerve growth factor ameliorate beta-amyloid deposition after traumatic brain injury in rats. Brain Res. 2012, 1440, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Murlidharan, G.; Samulski, R.J.; Asokan, A. Biology of adeno-associated viral vectors in the central nervous system. Front. Mol. Neurosci. 2014, 7, 76. [Google Scholar] [CrossRef]
- Das, M.; Wang, C.; Bedi, R.; Mohapatra, S.S.; Mohapatra, S. Magnetic micelles for DNA delivery to rat brains after mild traumatic brain injury. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 1539–1548. [Google Scholar] [CrossRef]
- Harmon, B.T.; Aly, A.E.; Padegimas, L.; Sesenoglu-Laird, O.; Cooper, M.J.; Waszczak, B.L. Intranasal administration of plasmid DNA nanoparticles yields successful transfection and expression of a reporter protein in rat brain. Gene Ther. 2014, 21, 514–521. [Google Scholar] [CrossRef]
- Chantsoulis, M.; Mirski, A.; Rasmus, A.; Kropotov, J.D.; Pachalska, M. Neuropsychological rehabilitation for traumatic brain injury patients. Ann. Agric. Environ. Med. AAEM 2015, 22, 368–379. [Google Scholar] [CrossRef]
- Rostami, R.; Salamati, P.; Yarandi, K.K.; Khoshnevisan, A.; Saadat, S.; Kamali, Z.S.; Ghiasi, S.; Zaryabi, A.; Ghazi Mir Saeid, S.S.; Arjipour, M.; et al. Effects of neurofeedback on the short-term memory and continuous attention of patients with moderate traumatic brain injury: A preliminary randomized controlled clinical trial. Chin. J. Traumatol. 2017, 20, 278–282. [Google Scholar] [CrossRef]
| Model | Injury |
|---|---|
| FPI | Focal/diffuse |
| Lateral | Mixed |
| Middle | Mixed |
| CCI | Primarily focal |
| PBBI | Primarily focal |
| Blast | Primarily diffuse |
| Weight Drop | Focal/diffuse |
| Repeated Mild | Primarily diffuse |
| Biomarkers | Injury Field | Models | References |
|---|---|---|---|
| CSF/serum albumin ratio | BBB dysfunction | patients with severe TBI | [20] |
| Tight junction proteins | mTBI in rats ischemic stroke in rats | [21,22] | |
| S100B | patients with minor head injury patients with extracranial pathology | [23,24] | |
| Plasma-soluble prion protein PrPc | rat model of concussion concussed athletes | [25,26] | |
| Tau proteins | Axonal injury | patients with acute TBI | [27] |
| UCHL1 | patients with mild or moderate TBI patients with severe TBI concussed athletes | [28,29,30] | |
| Neurofilaments (NFs) | rat models of TBI patients with mTBI patients with amyotrophic lateral sclerosis | [18,31,32] | |
| NSE | Patients with severe TBI | [33] | |
| GFAP | Rat TBI models Patients with severe TBI | [18,34] | |
| MBP | Children with TBI | [35] | |
| αII and βII-Spectrin breakdown products | Patients with severe TBI | [36] | |
| NGAL | Rat model of TBI Patients with severe TBI | [37,38] | |
| IL-6, IL-8, IL-10 | Neuroinflammation | Animal and clinical models of TBI | [39,40] |
| MMP | mTBI patients | [41] | |
| MBG | [42] | ||
| APOE | Genetic variations | mTBI patients | [43] |
| BDNF | [44] |
| Drug | Route of Administration | Preclinical Model | Inflammatory Events | References |
|---|---|---|---|---|
| Dexamethasone | I.P. | WD | ⇓ Microglia (CD68, MHC II) ⇓ Microglia (Endothelial-Monocyte Activating Polypeptide II, P2X4 Receptor, Iba-1) | [46,47] |
| Meloxicam | I.P. | M-WD | ⇓ Lipid Peroxidation GSSH Nakatpase | [48] |
| Etazolate | I.P. | WD | ⇓ IL-1β ⇓ Microglia (CD11b) | [49] |
| Carpofen | S.C. | WD | ⇓ Microglia (Iba-1) ⇓ IL-1β, ⇓ IL-6 ⇔ IL-4, ⇔ IL-10 | [50] |
| Indomethacin | I.P. | M-WD | ⇓ IL-1β, ⇓ 6-Keto PGF1α | [51,52] |
| I.P. | WD | |||
| Nimesulide | I.P. | WD | ⇓ 6-Keto PGF1a | [53] |
| Celecoxib | I.P. | WD | ⇓ Il-1β, ⇔ IL-10 | [52] |
| Meloxicam | I.P | WD | ⇓ 6-Keto PGF1a | [52] |
| Etanercept | I.P. | FPI | ⇓ TNF-α | [54] |
| Dexamethasone Melatonin | I.P. | CCI | ⇓ MMP-2, ⇓ MMP-9, ⇓ Inos | [55] |
| Lipoxin A4 | I.C.V. | WD | ⇓ IL-1β, ⇓ IL-6, ⇓ TNFα, ⇓ GFAP ⇓ Microglia (CD11b) | [56] |
| Ibuprofen | I.P. | FPI | ⇔ IL-4, ⇔ IL-10 ⇔ TNFα ⇔ IL-1α⇔ IL-6 | [57] |
| Minocycline | I.P. | WD | ⇓ microglia (CD11b) | [58] |
| I.P. | WD | ⇓ microglia, ⇓ IL-1β | [59] | |
| Fenofibrate | P.O. | LFP | ⇓ cerebral oedema ⇓ ICAM-1, ⇓ brain lesion | [60] |
| Pioglitazone and Rosiglitazone | I.P. | CCI | ⇓ activated microglia (OX-42) | [61] |
| N-acetylcysteine | I.P. | WD | ⇓ NF-kB, ⇓ IL-1β ⇓ IL-6, ⇓ TNF-α | [62] |
| Flavopiridol | I.P. | LFP | ⇓ GFAP, ⇓ microglia | [63] |
| Roscovitine | I.C.V. | CCI | ⇓ microglia (Iba-1) | [63] |
| Erythropoietin | I.P. | CCI | ⇓ NF-kB, ⇓ ICAM-1, ⇓ IL-1β ⇓ TNF-α, ⇔ IL-6 | [64] |
| I.P. | WD | ⇓ CCL-2 ⇓ microglia (CD-68) | [65] | |
| Simvastatin | P.O. | CCI | ⇓ TLR4, ⇓ NF-κB ⇓ IL-1β, ⇓ TNFα ⇓ Il-6, ⇓ ICAM-1 | [66] |
| ⇓ Il-1β, ⇓ GFAP ⇓ IL-6, ⇓ TNF-α microglia (CD68) | [67] | |||
| Progesterone | I.P. | WD | COX-2, ⇓ PGE2, ⇓ NF-κB | [68] |
| I.P./S.C. | CCI | ⇓ IL-6, ⇓ COX-2, ⇓ NF-κB | [69] |
| Class | Drug | Mechanism | Effect |
|---|---|---|---|
| CNS stimulants | Methylphenidate | acts as an NDRI | amplified the speed of information processing in many neuropsychological tests |
| CNS stimulants | Modafinil | unknown | raised alertness by the modulation of both noradrenergic and dopaminergic systems |
| Atypical antidepressant | Agomelatine | a melatonin receptor agonist and serotonin 5-HT2C and 5-HT2B | led to better sleep efficacy |
| Antiviral | Amantadine | reflect a growth in synthesis and discharge of dopamine | decreased the incidence and gravity of irritability |
| Antidepressant of the selective SNRI class | Venlafaxine | acts as an SNDRI | increased obsessive behaviors, irritability, and sadness symptoms |
| Anticonvulsant | Valproate | blockade of voltage-gated sodium channels and increased brain levels of GABA | had benign neuropsychological effects, and is a safe drug to control recognized seizures or stabilize mood |
| Acetylcholinesterase inhibitor | Rivastigmine | inhibits butyrylcholinesterase and acetylcholinesterase | encouraging in the subgroup of patients with moderate/severe memory weakening |
| Cholinesterase inhibitor | Galantamine | allosteric potentiating ligand of human nicotinic acetylcholine receptors (nAChRs) α4β2, α3β4, and α6β4 and chicken/mouse nAChRs α7/5-HT3 in some brain areas | refined fatigue, memory, attention, and initiative |
| Cholinesterase inhibitor | Donepezil | develops cholinergic function | indorsed clinical improvement and metabolism |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crupi, R.; Cordaro, M.; Cuzzocrea, S.; Impellizzeri, D. Management of Traumatic Brain Injury: From Present to Future. Antioxidants 2020, 9, 297. https://doi.org/10.3390/antiox9040297
Crupi R, Cordaro M, Cuzzocrea S, Impellizzeri D. Management of Traumatic Brain Injury: From Present to Future. Antioxidants. 2020; 9(4):297. https://doi.org/10.3390/antiox9040297
Chicago/Turabian StyleCrupi, Rosalia, Marika Cordaro, Salvatore Cuzzocrea, and Daniela Impellizzeri. 2020. "Management of Traumatic Brain Injury: From Present to Future" Antioxidants 9, no. 4: 297. https://doi.org/10.3390/antiox9040297
APA StyleCrupi, R., Cordaro, M., Cuzzocrea, S., & Impellizzeri, D. (2020). Management of Traumatic Brain Injury: From Present to Future. Antioxidants, 9(4), 297. https://doi.org/10.3390/antiox9040297








