Nitrates/Nitrites in Food—Risk for Nitrosative Stress and Benefits
Abstract
1. Introduction
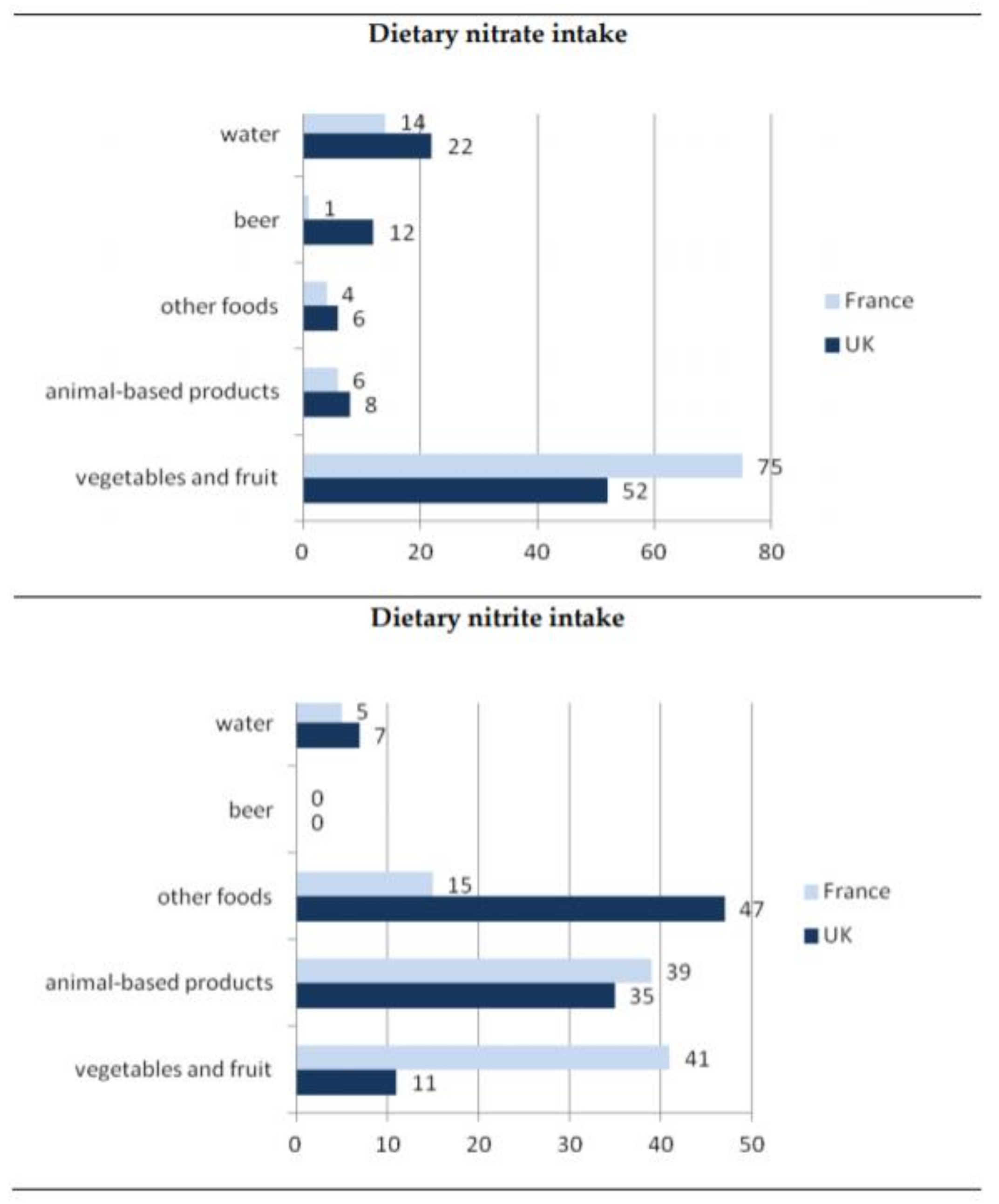
2. The Main Sources of Dietary Exposure to Nitrates and Nitrites
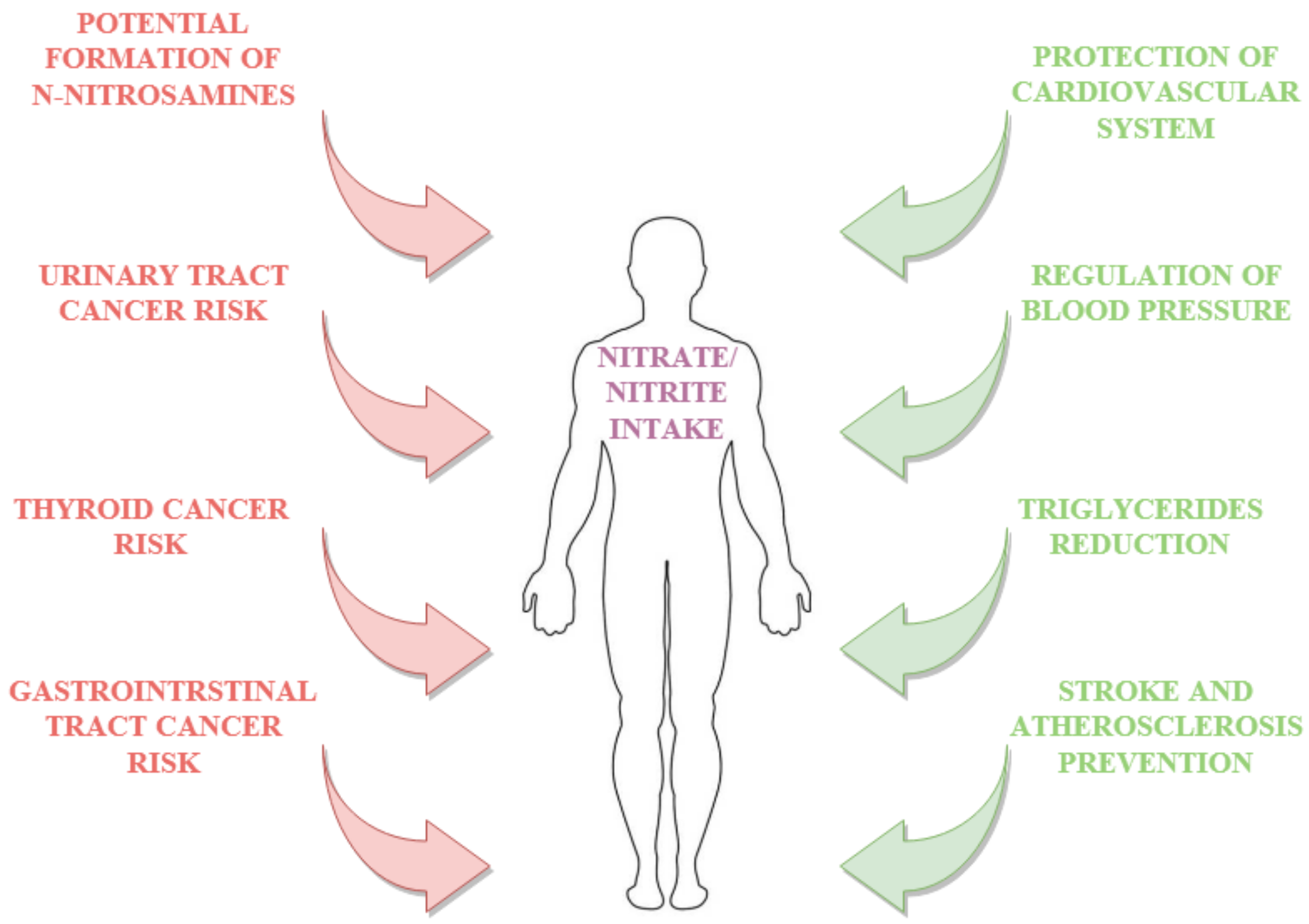
3. Health Concerns Related to Dietary Nitrites/Nitrates
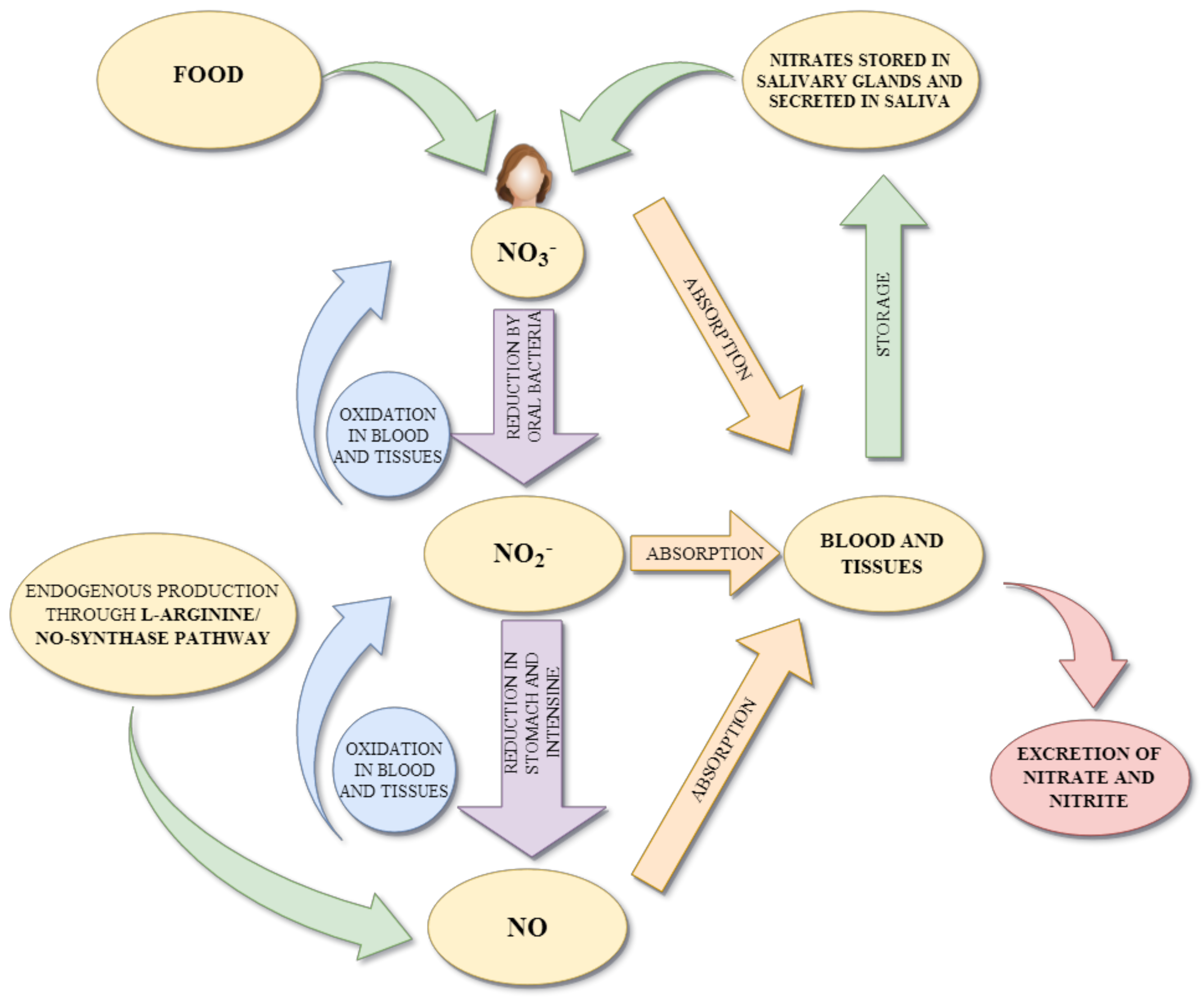
3.1. Metabolism of Nitrites/Nitrates
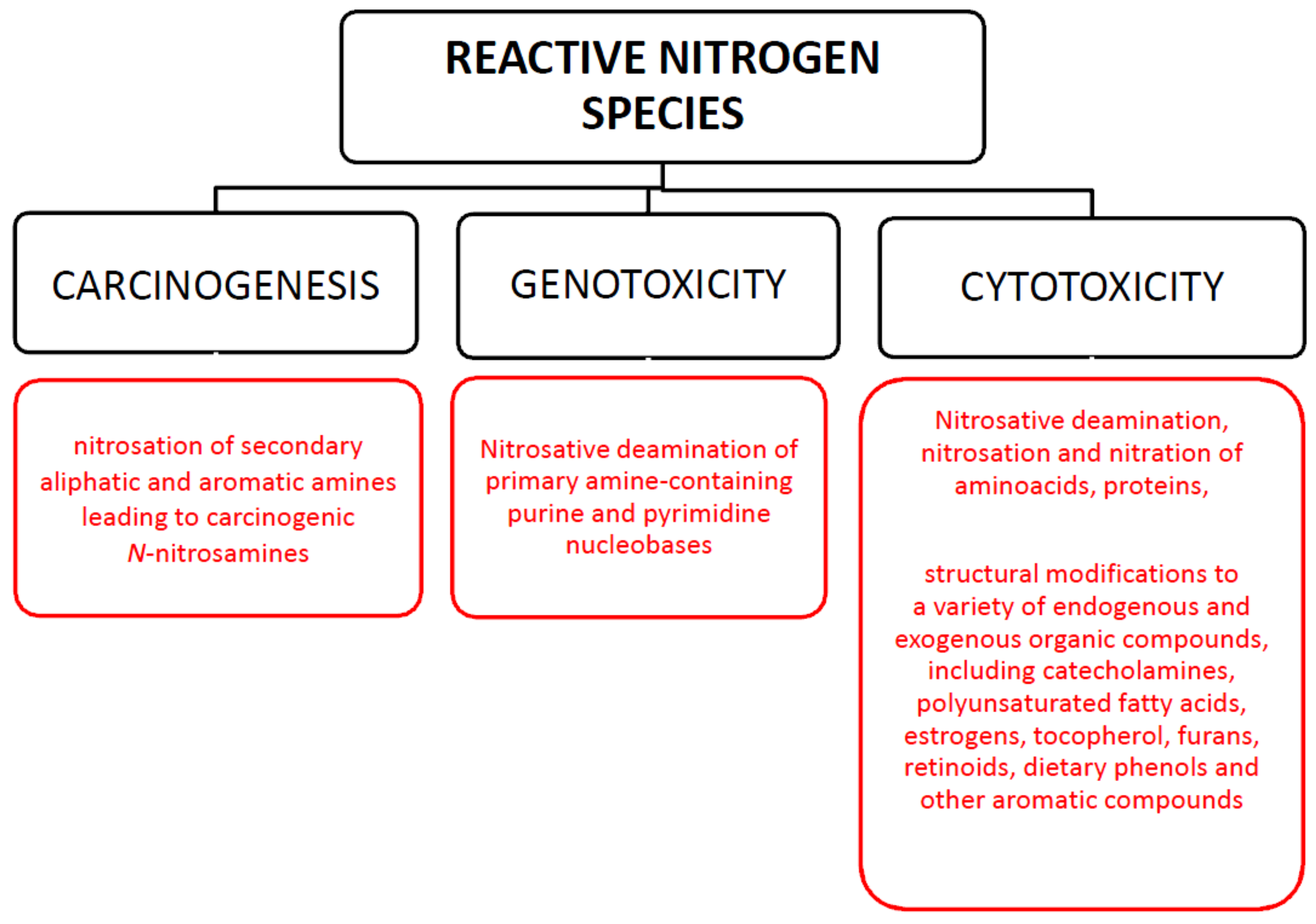
3.2. Effect on Nitrosative Stress
3.3. Benefits
4. The Use of Nitrites in Meat Processing
4.1. Role of Nitrites/Nitrates in Meat Products
4.2. Alternatives to Nitrites/Nitrates in Meat Products
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- WHO. Nitrate and Nitrite in Drinking-Water. Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Ward, M.H.; Jones, R.R.; Brender, J.D.; De Kok, T.M.; Weyer, P.J.; Nolan, B.T.; Villanueva, C.M.; Van Breda, S.G. Drinking Water Nitrate and Human Health: An Updated Review. Int. J. Environ. Res. Public Health 2018, 15, 1557. [Google Scholar]
- Larsson, K.; Darnerud, P.O.; Ilbäck, N.G.; Merino, L. Estimated dietary intake of nitrite and nitrate in Swedish children. Food Addit. Contam. Part A 2011, 28, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Temme, E.H.M.; Vandevijvere, S.; Vinkx, C.; Huybrechts, I.; Goeyens, L.; Van Oyen, H. Average daily nitrate and nitrite intake in the Belgian population older than 15 years. Food Addit. Contam. Part A 2011, 28, 1193–1204. [Google Scholar] [CrossRef] [PubMed]
- Tamme, T.; Reinik, M.; Roasto, M.; Juhkam, K.; Tenno, T.; Kiis, A. Nitrates and nitrites in vegetables and vegetable-based products and their intakes by the Estonian population. Food Addit. Contam. 2006, 23, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Johanningsmeier, S.D.; Price, R.; Reynolds, R.; Truong, V.-D.; Payton, S.C.; Breidt, F. Evolution of nitrate and nitrite content in pickled fruit and vegetable products. Food Control 2018, 90, 304–311. [Google Scholar] [CrossRef]
- Griesenbeck, J.S.; Steck, M.D.; Huber, J.C.; Sharkey, J.R.; Rene, A.A.; Brender, J.D. Development of estimates of dietary nitrates, nitrites, and nitrosamines for use with the Short Willet Food Frequency Questionnaire. Nutr. J. 2009, 8, 16. [Google Scholar] [CrossRef]
- EFSA. Nitrate in vegetables. Scientific opinion of the panel on contaminants in the food chain. EFSA J. 2008, 689, 1–79. [Google Scholar]
- Hord, N.G.; Tang, Y.; Bryan, N.S. Food sources of nitrates and nitrites: The physiologic context for potential health benefits. Am. J. Clin. Nutr. 2009, 90, 1–10. [Google Scholar] [CrossRef]
- Lucarini, M.; D’Evoli, L.; Tufi, S.; Gabrielli, P.; Paoletti, S.; Di Ferdinando, S.; Lombardi-Boccia, G. Influence of growing system on nitrate accumulation in two varieties of lettuce and red radicchio of Treviso. J. Sci. Food Agric. 2012, 92, 2796–2799. [Google Scholar] [CrossRef]
- Prasad, S.; Chetty, A.A. Flow injection assessment of nitrate contents in fresh and cooked fruits and vegetables grown in Fiji. J. Food Sci. 2011, 76, C1143–C1148. [Google Scholar] [CrossRef]
- World Health Organization. Nitrate and Nitrite in Drinking Water Development of WHO Guidelines for Drinking Water Quality; World Health Organization: Geneva, Switzerland, 2007; pp. 1–21. [Google Scholar]
- Nuñez de González, M.T.; Osburn, W.N.; Hardin, M.D.; Longnecker, M.; Garg, H.K.; Bryan, N.S.; Keeton, J.T. A Survey of nitrate and nitrite concentrations in conventional and organic-labeled raw vegetables at retail. J. Food Sci. 2015, 80, C942–C949. [Google Scholar] [CrossRef] [PubMed]
- Roila, R.; Branciari, R.; Staccini, B.; Ranucci, D.; Miraglia, D.; Altissimo, M.S.; Mercuri, M.L.; Haouet, N.M. Contribution of vegetables and cured meat to dietary nitrate and nitrite intake in Italian population: Safe level for cured meat and controversial role of vegetables. Italian J. Food Saf. 2018, 7, 7692. [Google Scholar] [CrossRef] [PubMed]
- Sindelar, J.J.; Milkowski, A.L. Human safety controversies surrounding nitrate and nitrite in the diet. Nitric Oxide 2012, 26, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Merino, L.; Darnerud, P.O.; Toldrá, F.; Ilbäck, N.-G. Time-dependent depletion of nitrite in pork/beef and chicken meat products and its effect on nitrite intake estimation. Food Additiv. Contam. 2016, 33, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Liu, X.; Sun, Q.; Fan, Z.; Xia, D.; Ding, G.; Qi, S. Sialin (SLC17A5) functions as a nitrate transporter in the plasma membrane. Proc. Natl. Acad. Sci. USA 2012, 109, 13434–13439. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.M.; Wu, Z.F.; Pang, B.X.; Jin, L.Y.; Qin, L.Z.; Wang, S.L. From nitrate to nitric oxide: The role of salivary glands and oral bacteria. J. Den. Res. 2016, 95, 1452–1456. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate–nitrite–nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Discov. 2008, 7, 156. [Google Scholar] [CrossRef]
- Leaf, C.D.; Wishnok, J.S.; Tannenbaum, S.R. L-arginine is a precursor for nitrate biosynthesis in humans. Bioch. Bioph. Res. Commun. 1989, 163, 1032–1037. [Google Scholar] [CrossRef]
- Van Faassen, E.E.; Bahrami, S.; Feelisch, M.; Hogg, N.; Kelm, M.; Kim-Shapiro, D.B.; Nohl, H. Nitrite as regulator of hypoxic signaling in mammalian physiology. Med. Res. Rev. 2009, 29, 683–741. [Google Scholar] [CrossRef]
- Bryan, N.S.; Ivy, J.L. Inorganic nitrite and nitrate: Evidence to support consideration as dietary nutrients. Nutr. Res. 2015, 35, 643–654. [Google Scholar] [CrossRef]
- Doel, J.J.; Benjamin, N.; Hector, M.P.; Rogers, M.; Allaker, R.P. Evaluation of bacterial nitrate reduction in the human oral cavity. Eur. J. Oral Sci. 2005, 113, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Hyde, E.R.; Andrade, F.; Vaksman, Z.; Parthasarathy, K.; Jiang, H.; Parthasarathy, D.K.; Bryan, N.S. Metagenomic analysis of nitrate-reducing bacteria in the oral cavity: Implications for nitric oxide homeostasis. PLoS ONE 2014, 9, e88645. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Govoni, M. Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Rad. Biol. Med. 2004, 37, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Tripatara, P.; Patel, N.S.; Webb, A.; Rathod, K.; Lecomte, F.M.; Mazzon, E.; Thiemermann, C. Nitrite-derived nitric oxide protects the rat kidney against ischemia/reperfusion injury in vivo: Role for xanthine oxidoreductase. J. Am. Soc. Nephrol. 2007, 18, 570–580. [Google Scholar] [CrossRef]
- Rassaf, T.; Flögel, U.; Drexhage, C.; Hendgen-Cotta, U.; Kelm, M.; Schrader, J. Nitrite reductase function of deoxymyoglobin: Oxygen sensor and regulator of cardiac energetics and function. Circ. Res. 2007, 100, 1749–1754. [Google Scholar] [CrossRef]
- Pereira, C.; Ferreira, N.R.; Rocha, B.S.; Barbosa, R.M.; Laranjinha, J. The redox interplay between nitrite and nitric oxide: From the gut to the brain. Redox Biol. 2013, 1, 276–284. [Google Scholar] [CrossRef]
- Ignarro, L.J.; Byrns, R.E.; Sumi, D.; de Nigris, F.; Napoli, C. Pomegranate juice protects nitric oxide against oxidative destruction and enhances the biological actions of nitric oxide. Nitric Oxide 2006, 15, 93–102. [Google Scholar] [CrossRef]
- Ma, L.; Hu, L.; Feng, X.; Wang, S. Nitrate and nitrite in health and disease. Aging Dis. 2018, 9, 938. [Google Scholar] [CrossRef]
- Ohshima, H.; Bartsch, H. Quantitative estimation of endogenous nitrosation in humans by monitoring N-nitrosoproline excreted in the urine. Canc. Res. 1981, 41, 3658–3662. [Google Scholar]
- Shephard, S.E.; Schlatter, C.H.; Lutz, W.K. Assessment of the risk of formation of carcinogenic N-nitroso compounds from dietary precursors in the stomach. Food Chem. Toxic. 1987, 25, 91–108. [Google Scholar] [CrossRef]
- Tricker, A.R. N-nitroso compounds and man: Sources of exposure, endogenous formation and occurrence in body fluids. Eur. J. Cancer Prev. Off. J. Eur. Canc. Prev. Org. 1997, 6, 226–268. [Google Scholar] [CrossRef]
- De La Pomélie, D.; Santé-Lhoutellier, V.; Gatellier, P. Mechanisms and kinetics of tryptophan N-nitrosation in a gastro-intestinal model. Food Chem. 2017, 218, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, H.; Pignatelli, B.; Calmels, S.; Ohshima, H. Inhibition of nitrosation. In Antimutagenesis and Anticarcinogenesis Mechanisms III; Springer: Boston, MA, USA, 1993. [Google Scholar]
- Lunn, J.C.; Kuhnle, G.; Mai, V.; Frankenfeld, C.; Shuker, D.E.G.; Glen, R.C.; Bingham, S.A. The effect of haem in red and processed meat on the endogenous formation of N-nitroso compounds in the upper gastrointestinal tract. Carcinogenesis 2007, 28, 685–690. [Google Scholar] [CrossRef] [PubMed]
- D’Ischia, M.; Napolitano, A.; Manini, P.; Panzella, L. Secondary targets of nitrite-derived reactive nitrogen species: Nitrosation/nitration pathways, antioxidant defense mechanisms and toxicological implications. Chem. Res. Toxicol. 2011, 24, 2071–2092. [Google Scholar] [CrossRef] [PubMed]
- Dalle-Donne, I.; Rossi, R.; Colombo, R.; Giustarini, D.; Milzani, A. Biomarkers of oxidative damage in human disease. Clin. Chem. 2006, 52, 601–623. [Google Scholar] [CrossRef] [PubMed]
- Calcerrada, P.; Peluffo, G.; Radi, R. Nitric oxide-derived oxidants with a focus on peroxynitrite: Molecular targets, cellular responses and therapeutic implications. Curr. Pharm. Des. 2011, 17, 3905–3932. [Google Scholar] [CrossRef]
- Li, X.; Tao, R.R.; Hong, L.J.; Cheng, J.; Jiang, Q.; Lu, Y.M.; Hu, Y.Z. Visualizing peroxynitrite fluxes in endothelial cells reveals the dynamic progression of brain vascular injury. J. Am. Chem. Soc. 2015, 137, 12296–12303. [Google Scholar] [CrossRef]
- Alhasawi, A.; Legendre, F.; Jagadeesan, S.; Appanna, V.; Appanna, V. Chapter 10-Biochemical strategies to counter nitrosative stress: Nanofactories for value-added products. In Microbial Diversity in the Genomic Era; Academic Press: Cambridge, MA, USA, 2019; pp. 153–169. [Google Scholar]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric oxide and peroxynitrite in health and disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef]
- Trostchansky, A.; Rubbo, H. Nitrated fatty acids: Mechanisms of formation, chemical characterization, and biological properties. Free Rad. Biol. Med. 2008, 44, 1887–1896. [Google Scholar] [CrossRef]
- White, P.J.; Charbonneau, A.; Cooney, G.J.; Marette, A. Nitrosative modifications of protein and lipid signaling molecules by reactive nitrogen species. Am. J. Physiol. 2010, 299, E868–E878. [Google Scholar] [CrossRef]
- Kurutas, E.B. The importance of antioxidants which play the role in cellular response of against oxidative/nitrosative stress: Current state. Nutr. J. 2016, 15, 71. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Zhang, X.M.; Tarnawski, L.; Peleli, M.; Zhuge, Z.; Terrando, N.; Lundberg, J.O. Dietary nitrate attenuates renal ischemia-reperfusion injuries by modulation of immune responses and reduction of oxidative stress. Redox Biol. 2017, 13, 320–330. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lee, S.S.; Choi, B.Y.; Kim, M.K. Nitrate intake relative to antioxidant vitamin intake affects gastric cancer risk: A case-control study in Korea. Nutr. Canc. 2007, 59, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.H.; Heineman, E.F.; Markin, R.S.; Weisenburger, D.D. Adenocarcinoma of the stomach and esophagus and drinking water and dietary sources of nitrate and nitrite. Int. J. Occup. Environ. Health 2008, 14, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Keszei, A.P.; Goldbohm, R.A.; Schouten, L.J.; Jakszyn, P.; van den Brandt, P.A. Dietary N-nitroso compounds, endogenous nitrosation, and the risk of esophageal and gastric cancer subtypes in the Netherlands Cohort Study. Am. J. Clin. Nutr. 2012, 97, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Weyer, P.J.; Cerhan, J.R.; Kross, B.C.; Hallberg, G.R.; Kantamneni, J.; Breuer, G.; Lynch, C.F. Municipal drinking water nitrate level and cancer risk in older women: The Iowa Women’s Health Study. Epidemiology 2001, 12, 327–338. [Google Scholar] [CrossRef] [PubMed]
- DellaValle, C.T.; Xiao, Q.; Yang, G.; Shu, X.O.; Aschebrook-Kilfoy, B.; Zheng, W.; Gao, Y.T. Dietary nitrate and nitrite intake and risk of colorectal cancer in the Shanghai Women’s Health Study. Int. J. Cancer 2014, 134, 2917–2926. [Google Scholar] [CrossRef]
- Grieb, S.M.D.; Theis, R.P.; Burr, D.; Benardot, D.; Siddiqui, T.; Asal, N.R. Food groups and renal cell carcinoma: Results from a case-control study. J. Am. Diet. Assoc. 2009, 109, 656–667. [Google Scholar] [CrossRef]
- Dubrow, R.; Darefsky, A.S.; Park, Y.; Mayne, S.T.; Moore, S.C.; Kilfoy, B.; Ward, M.H. Dietary components related to N-nitroso compound formation: A prospective study of adult glioma. Cancer Epidemiol. Prev. Biomark. 2010, 19, 1709–1722. [Google Scholar] [CrossRef]
- De Roos, A.J.; Ray, R.M.; Gao, D.L.; Wernli, K.J.; Fitzgibbons, E.D.; Ziding, F.; Checkoway, H. Colorectal cancer incidence among female textile workers in Shanghai, China: A case-cohort analysis of occupational exposures. Cancer Causes Control 2005, 16, 1177–1188. [Google Scholar] [CrossRef]
- Cross, A.J.; Ferrucci, L.M.; Risch, A.; Graubard, B.I.; Ward, M.H.; Park, Y.; Sinha, R. A large prospective study of meat consumption and colorectal cancer risk: An investigation of potential mechanisms underlying this association. Cancer Res. 2010, 70, 2406–2414. [Google Scholar] [CrossRef] [PubMed]
- Bahadoran, Z.; Mirmiran, P.; Ghasemi, A.; Kabir, A.; Azizi, F.; Hadaegh, F. Is dietary nitrate/nitrite exposure a risk factor for development of thyroid abnormality? A systematic review and meta-analysis. Nitric Oxide 2015, 47, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Wu, L.; Guan, W. Dietary nitrates, nitrites, and nitrosamines intake and the risk of gastric cancer: A meta-analysis. Nutrients 2015, 7, 9872–9895. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Mao, Y.; White, K. Diet and vitamin or mineral supplements and risk of renal cell carcinoma in Canada. Cancer Causes Control 2003, 14, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Aschebrook-Kilfoy, B.; Shu, X.O.; Gao, Y.T.; Ji, B.T.; Yang, G.; Li, H.L.; Ward, M.H. Thyroid cancer risk and dietary nitrate and nitrite intake in the Shanghai women’s health study. Int. J. Cancer 2013, 132, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Van Hecke, T.; Vossen, E.; Hemeryck, L.Y.; Bussche, J.V.; Vanhaecke, L.; De Smet, S. Increased oxidative and nitrosative reactions during digestion could contribute to the association between well-done red meat consumption and colorectal cancer. Food Chem. 2015, 187, 29–36. [Google Scholar] [CrossRef]
- Bastide, N.M.; Chenni, F.; Audebert, M.; Santarelli, R.L.; Taché, S.; Naud, N.; Kuhnle, G.G. A central role for heme iron in colon carcinogenesis associated with red meat intake. Cancer Res. 2015, 75, 870–879. [Google Scholar] [CrossRef]
- Zheng, J.; Stuff, J.; Tang, H.; Hassan, M.M.; Daniel, C.R.; Li, D. Dietary N-nitroso compounds and risk of pancreatic cancer: Results from a large case–control study. Carcinogenesis 2018, 40, 254–262. [Google Scholar] [CrossRef]
- Chan, T.Y. Vegetable-borne nitrate and nitrite and the risk of methaemoglobinaemia. Toxicol. Lett. 2011, 200, 107–108. [Google Scholar] [CrossRef]
- Weitzberg, E.; Lundberg, J.O. Novel aspects of dietary nitrate and human health. Ann. Rev. Nutr. 2013, 33, 129–159. [Google Scholar] [CrossRef]
- Kapil, V.; Khambata, R.S.; Robertson, A.; Caulfield, M.J.; Ahluwalia, A. Dietary nitrate provides sustained blood pressure lowering in hypertensive patients: A randomized, phase 2, double-blind, placebo-controlled study. Hypertension 2015, 65, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Berry, M.J.; Justus, N.W.; Hauser, J.I.; Case, A.H.; Helms, C.C.; Basu, S.; Miller, G.D. Dietary nitrate supplementation improves exercise performance and decreases blood pressure in COPD patients. Nitric Oxide 2015, 48, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, A.; Mitchell, K.; Blackwell, J.R.; Vanhatalo, A.; Jones, A.M. High-nitrate vegetable diet increases plasma nitrate and nitrite concentrations and reduces blood pressure in healthy women. Public Health Nutr. 2015, 18, 2669–2678. [Google Scholar] [CrossRef] [PubMed]
- Montenegro, M.F.; Amaral, J.H.; Pinheiro, L.C.; Sakamoto, E.K.; Ferreira, G.C.; Reis, R.I.; Tanus-Santos, J.E. Sodium nitrite downregulates vascular NADPH oxidase and exerts antihypertensive effects in hypertension. Free Rad. Biol. Med. 2011, 51, 144–152. [Google Scholar] [CrossRef]
- Amaral, J.H.; Ferreira, G.C.; Pinheiro, L.C.; Montenegro, M.F.; Tanus-Santos, J.E. Consistent antioxidant and antihypertensive effects of oral sodium nitrite in DOCA-salt hypertension. Redox Biol. 2015, 5, 340–346. [Google Scholar] [CrossRef]
- Ling, W.C.; Mustafa, M.R.; Vanhoutte, P.M.; Murugan, D.D. Chronic administration of sodium nitrite prevents hypertension and protects arterial endothelial function by reducing oxidative stress in angiotensin II-infused mice. Vasc. Pharmacol. 2018, 102, 11–20. [Google Scholar] [CrossRef]
- Siervo, M.; Lara, J.; Ogbonmwan, I.; Mathers, J.C. Inorganic nitrate and beetroot juice supplementation reduces blood pressure in adults: A systematic review and meta-analysis. J. Nutr. 2013, 143, 818–826. [Google Scholar] [CrossRef]
- Ghasemi, A.; Gheibi, S. Effect of oral nitrate administration on glucose metabolism and inflammation in obese type 2 diabetic rats. In 19th European Congress of Endocrinology; BioScientifica: Bristol, UK, 2017. [Google Scholar]
- Khalifi, S.; Rahimipour, A.; Jeddi, S.; Ghanbari, M.; Kazerouni, F.; Ghasemi, A. Dietary nitrate improves glucose tolerance and lipid profile in an animal model of hyperglycemia. Nitric Oxide 2015, 44, 24–30. [Google Scholar] [CrossRef]
- Gilchrist, M.; Winyard, P.G.; Fulford, J.; Anning, C.; Shore, A.C.; Benjamin, N. Dietary nitrate supplementation improves reaction time in type 2 diabetes: Development and application of a novel nitrate-depleted beetroot juice placebo. Nitric Oxide 2014, 40, 67–74. [Google Scholar] [CrossRef]
- Cermak, N.M.; Hansen, D.; Kouw, I.W.; van Dijk, J.W.; Blackwell, J.R.; Jones, A.M.; van Loon, L.J. A single dose of sodium nitrate does not improve oral glucose tolerance in patients with type 2 diabetes mellitus. Nutr. Res. 2015, 35, 674–680. [Google Scholar] [CrossRef]
- Zand, J.; Lanza, F.; Garg, H.K.; Bryan, N.S. All-natural nitrite and nitrate containing dietary supplement promotes nitric oxide production and reduces triglycerides in humans. Nutr. Res. 2011, 31, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Rammos, C.; Hendgen-Cotta, U.B.; Sobierajski, J.; Bernard, A.; Kelm, M.; Rassaf, T. Dietary nitrate reverses vascular dysfunction in older adults with moderately increased cardiovascular risk. J. Am. Coll. Cardiol. 2014, 63, 1584–1585. [Google Scholar] [CrossRef] [PubMed]
- Bondonno, C.P.; Blekkenhorst, L.C.; Prince, R.L.; Ivey, K.L.; Lewis, J.R.; Devine, A.; Hodgson, J.M. Association of vegetable nitrate intake with carotid atherosclerosis and ischemic cerebrovascular disease in older women. Stroke 2017, 48, 1724–1729. [Google Scholar] [CrossRef] [PubMed]
- Burnley-Hall, N.; Abdul, F.; Androshchuk, V.; Morris, K.; Ossei-Gerning, N.; Anderson, R.; James, P.E. Dietary nitrate supplementation reduces circulating platelet-derived extracellular vesicles in coronary artery disease patients on clopidogrel therapy: A randomised, double-blind, placebo-controlled study. Thromb. Haemost. 2018, 118, 112–122. [Google Scholar] [CrossRef]
- Lefer, D.J.; Bryan, N.S.; Organ, C.L. Nitrite and Nitrate in Ischemia–Reperfusion Injury. In Nitrite and Nitrate in Human Health and Disease; Humana Press: Totowa, NJ, USA, 2017; pp. 217–234. [Google Scholar]
- Coggan, A.R.; Leibowitz, J.L.; Spearie, C.A.; Kadkhodayan, A.; Thomas, D.P.; Ramamurthy, S.; Peterson, L.R. Acute dietary nitrate intake improves muscle contractile function in patients with heart failure: A double-blind, placebo-controlled, randomized trial. Circ. Heart Fail. 2015, 8, 914–920. [Google Scholar] [CrossRef]
- Coggan, A.R.; Leibowitz, J.L.; Kadkhodayan, A.; Thomas, D.P.; Ramamurthy, S.; Spearie, C.A.; Peterson, L.R. Effect of acute dietary nitrate intake on maximal knee extensor speed and power in healthy men and women. Nitric Oxide 2015, 48, 16–21. [Google Scholar] [CrossRef]
- Xie, L.; Mo, M.; Jia, H.X.; Liang, F.; Yuan, J.; Zhu, J. Association between dietary nitrate and nitrite intake and site-specific cancer risk: Evidence from observational studies. Oncotarget 2016, 7, 56915. [Google Scholar] [CrossRef]
- Cantwell, M.; Elliott, C. Nitrates, Nitrites and Nitrosamines from Processed Meat Intake and Colorectal Cancer Risk. J. Clin. Nutr. Diet. 2017, 3, 27. [Google Scholar] [CrossRef]
- Honikel, K.O. Chemical analysis of specific components curing agents. In Encyclopedia of Meat Sciences, 2nd ed.; Devine, C., Ed.; Academic Press: Oxford, UK, 2014; pp. 200–205. [Google Scholar]
- Menard, C.; Heraud, F.; Volatier, J.L.; Leblanc, J.C. Assessment of dietary exposure of nitrate and nitrite in France. Food Addit. Contam. 2008, 25, 971–988. [Google Scholar] [CrossRef]
- Herrmann, S.S.; Duedahl-Olesen, L.; Granby, K. Occurrence of volatile and nonvolatile N-nitrosamines in processed meat products and the role of heat treatment. Food Control 2015, 48, 163–169. [Google Scholar] [CrossRef]
- Bedale, W.; Sindelar, J.J.; Milkowski, A.L. Dietary nitrate and nitrite: Benefits, risks, and evolving perceptions. Meat Sci. 2016, 120, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Alahakoon, A.U.; Jayasena, D.D.; Ramachandra, S.; Jo, C. Alternatives to nitrite in processed meat: Up to date. Trends Food Sci. Technol. 2015, 45, 37–40. [Google Scholar] [CrossRef]
- Jira, W. Chemical reaction of curing and smoking-Part 1: Curing. Fleischwirtstchaft 2004, 84, 235–239. [Google Scholar]
- Hammes, P.W. Metabolism of nitrate in fermented meats: The characteristic feature of a specific group of fermented foods. Food Microbiol. 2012, 29, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Sindelar, J.J.; Cordray, J.C.; Sebranek, J.G.; Love, J.A.; Ahn, D.U. Effects of varying levels of vegetable juice powder and incubation time on color, residual nitrate and nitrite, pigment, pH, and trained sensory attributes of ready-to-eat uncured ham. J. Food Sci. 2007, 72, S388–S395. [Google Scholar] [CrossRef] [PubMed]
- Kononiuk, A.; Karwowska, M. Physicochemical, proteolytic and sensory changes during long-term storage of dry-fermented sausages made from fallow deer meat in comparison to beef. Food Technol. Sci. Qual. 2019, 26, 137–154. [Google Scholar]
- Kononiuk, A.; Karwowska, M. Comparison of selected parameters related to food safety of fallow deer and beef uncured fermented sausages with freeze-dried acid whey addition. Meat Sci. 2020, 161, 108015. [Google Scholar] [CrossRef]
- Sucu, C.; Turp, G.Y. The investigation of the use of beetroot powder in Turkish fermented beef sausage (sucuk) as nitrite alternative. Meat Sci. 2018, 140, 158–166. [Google Scholar] [CrossRef]
- Bianchin, M.; Pereira, D.; Dos Reis, A.S.; De Florio Almeida, J.; Dangui, L.; Da Silva, C.D.M.; Carpes, S.T. Research Article Rosemary Essential Oil and Lyophilized Extract as Natural Antioxidant Source to Prevent Lipid Oxidation in Pork Sausage. Adv. J. Food Sci. Technol. 2017, 13, 210–217. [Google Scholar] [CrossRef]
- Karwowska, M.; Dolatowski, Z.J. Effect of acid whey and freeze-dried cranberries on lipid oxidation and fatty acid composition of nitrite-/nitrate-free fermented sausage made from deer meat. Asian-Austral. J. Anim. Sci. 2017, 30, 85. [Google Scholar] [CrossRef]
- Kim, T.K.; Hwang, K.E.; Lee, M.A.; Paik, H.D.; Kim, Y.B.; Choi, Y.S. Quality characteristics of pork loin cured with green nitrite source and some organic acids. Meat Sci. 2019, 152, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Gajowiecki, L.; Kotowicz, M.; Lachowicz, K.; Dabrowski, W.; Koronkiewicz, A.; Zochowska-Kujawska, J.; Zych, A. Zastosowanie mleczanow do produkcji wyrobow drobiowych niezawierajacych dodatku azotanu [III] sodu. Food Technol. Sci. Qual. 2006, 13, 86–99. [Google Scholar]
- Huang, L.; Zeng, X.; Sun, Z.; Wu, A.; He, J.; Dang, Y.; Pan, D. Production of a safe cured meat with low residual nitrite using nitrite substitutes. Meat Sci. 2019, 162, 108027. [Google Scholar] [CrossRef] [PubMed]
- Zarringhalami, S.; Sahari, M.A.; Hamidi-Esfehani, Z. Partial replacement of nitrite by annatto as a colour additive in sausage. Meat Sci. 2009, 81, 281–284. [Google Scholar] [CrossRef] [PubMed]
| Source | Total Nitrate Content (mg kg−1) | References |
|---|---|---|
| Spinach | 1066 | EFSA [8] |
| 2036 | Roila et al. [14] | |
| 2333 | Sidelar and Milkowski [15] | |
| Rucola | 4677 | EFSA [8] |
| Radish | 1297 | EFSA [8] |
| Celery | 1103 | EFSA [8] |
| 1544 | Sidelar and Milkowski [15] | |
| 1495 | Nuñez de González et al. [13] | |
| Rhubarb | 2943 | EFSA [8] |
| Lettuce | 1324 | EFSA [8] |
| 1079 | Roila et al. [14] | |
| 786 | Sidelar and Milkowski [15] | |
| Chard | 1690 | EFSA [8] |
| 1728 | Roila et al. [14] | |
| Beets | 2756 | Sidelar and Milkowski [15] |
| 1446 | Tamme et al. [5] | |
| Beetroot | 1379 | EFSA [8] |
| Carrot | 238 296 | Roila et al. [14] EFSA [8] |
| Potato | 168 | EFSA [8] |
| Adverse Effect | Reference | Benefits | Reference |
|---|---|---|---|
| Gastric cancer | Kim et al. [47] Ward et al. [48] Keszei et al. [49] Xie et al. [83] Song et al. [57] Zheng et al. [62] | Adult glioma | Dubrow et al. [53]; Xie et al. [83] |
| Colorectal cancer | De Roos et al. [54]; Cross et al. [55]; DellaValle [51]; Cantwell et al. [84] Xie et al. [83]; Van Hecke et al. [60] Bestide et al. [61] | Reduction of blood pressure | Kapil et al. [65]; Berry et al. [66]; Ashworth et al. [67]; Amaral et al. [69]; Ling et al. [70]; Sievro et al. [71] |
| Esophageal cancer | Cross et al. [55]; Keszei et al. [49]; Xie et al. [83] | Atherosclerosis prevention | Bondonno et al. [78]; Burnley-Hall et al. [79] |
| Thyroid cancer | Ward et al. [48]; Aschebrook-Kilfoy et al. [59]; Xie et al. [83]; Bahadoran et al. [56] | Protection against ischemia- reperfusion | Yang et al. [46]; Lefer et al. [80] |
| Renal cell carcinoma | Weyer et al. [50]; DellaVale et al. [51]; Grieb [52]; Xie et al. [83]; Hu et al. [58] | Exercise capacity | Coggan et al. [81] Coggan et al. [82] |
| Methemoglobinemia | Chan et al. [63] | Stroke prevention | Bondonno et al. [78]; Burnley-Hall et al. [79] |
| Hypothyroidism | Ward et al. [48]; Xie et al. [83] | Insulin resistance, glucose tolerance | Ghasemi, Ghebi [72]; Khalifi et al. [73]; Gilchrist et al. [74]; Cermak et al. [75] |
| Breast cancer | Yang et al. [46]; Xie et al. [83] | Reduction of triglycerides | Zand et al. [76] |
| Nitrosative stress | D’Ischia et al. [37] |
| Additives | Effects | Type of Product | Reference |
|---|---|---|---|
| beetroot powder | control lipid oxidation and residual nitrite contents | fermented beef sausage | Sucu andTurp [95] |
| rosemary essential oil /lyophilized extract | lipid oxidation inhibition higher antioxidant activity | pork sausages | Bianchin et al. [96] |
| celery juice powder + starter cultures | control lipid oxidation color development | ready-to-eat ham | Sindelar et al. [92] |
| freeze-dried cranberry | control lipid oxidation | fallow-deer fermented sausage | Karwowska, Dolatowski [97] |
| fermented spinach powder | control lipid oxidation | cured pork loins | Kim et al. [98] |
| acid whey (liquid and freeze-dried) | proteolysis changes food safety related parameters | dry-fermented sausages made of beef dry-fermented sausages made of fallow-deer meat | Kononiuk, Karwowska [93] Kononiuk, Karwowska [94] |
| lactates | antibacterial activity, color development, lower cooking loss | pasteurized canned poultry products | Gajowiecki et al. [99] |
| Lactobacillus fermentum RC4 and Lactobacillus plantarum B6 -starters, beetroot and Monascus – coloring agents, nisin as antibiotic | antibacterial activity, control lipid oxidation, color development | cured pork meat | Huang et al. [100] |
| annatto powder | color development control of bacterial growth | cooked sausages | Zarringhalami et al. [101] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karwowska, M.; Kononiuk, A. Nitrates/Nitrites in Food—Risk for Nitrosative Stress and Benefits. Antioxidants 2020, 9, 241. https://doi.org/10.3390/antiox9030241
Karwowska M, Kononiuk A. Nitrates/Nitrites in Food—Risk for Nitrosative Stress and Benefits. Antioxidants. 2020; 9(3):241. https://doi.org/10.3390/antiox9030241
Chicago/Turabian StyleKarwowska, Małgorzata, and Anna Kononiuk. 2020. "Nitrates/Nitrites in Food—Risk for Nitrosative Stress and Benefits" Antioxidants 9, no. 3: 241. https://doi.org/10.3390/antiox9030241
APA StyleKarwowska, M., & Kononiuk, A. (2020). Nitrates/Nitrites in Food—Risk for Nitrosative Stress and Benefits. Antioxidants, 9(3), 241. https://doi.org/10.3390/antiox9030241