Impact of Pleurotus ostreatus β-Glucans on Oxidative Stability of Active Compounds Encapsulated in Powders during Storage and In Vitro Digestion
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. In Vitro Digestion
2.2.2. Oil Droplet Size Distribution
2.2.3. Accelerated Thermo-Oxidation
2.2.4. Oil Phase Extraction
2.2.5. Measurement of CD
2.2.6. Measurement of α-Toc Concentration
2.2.7. Statistical Analysis of Data
3. Results
3.1. Effect of In Vitro Digestion on Emulsion Structure
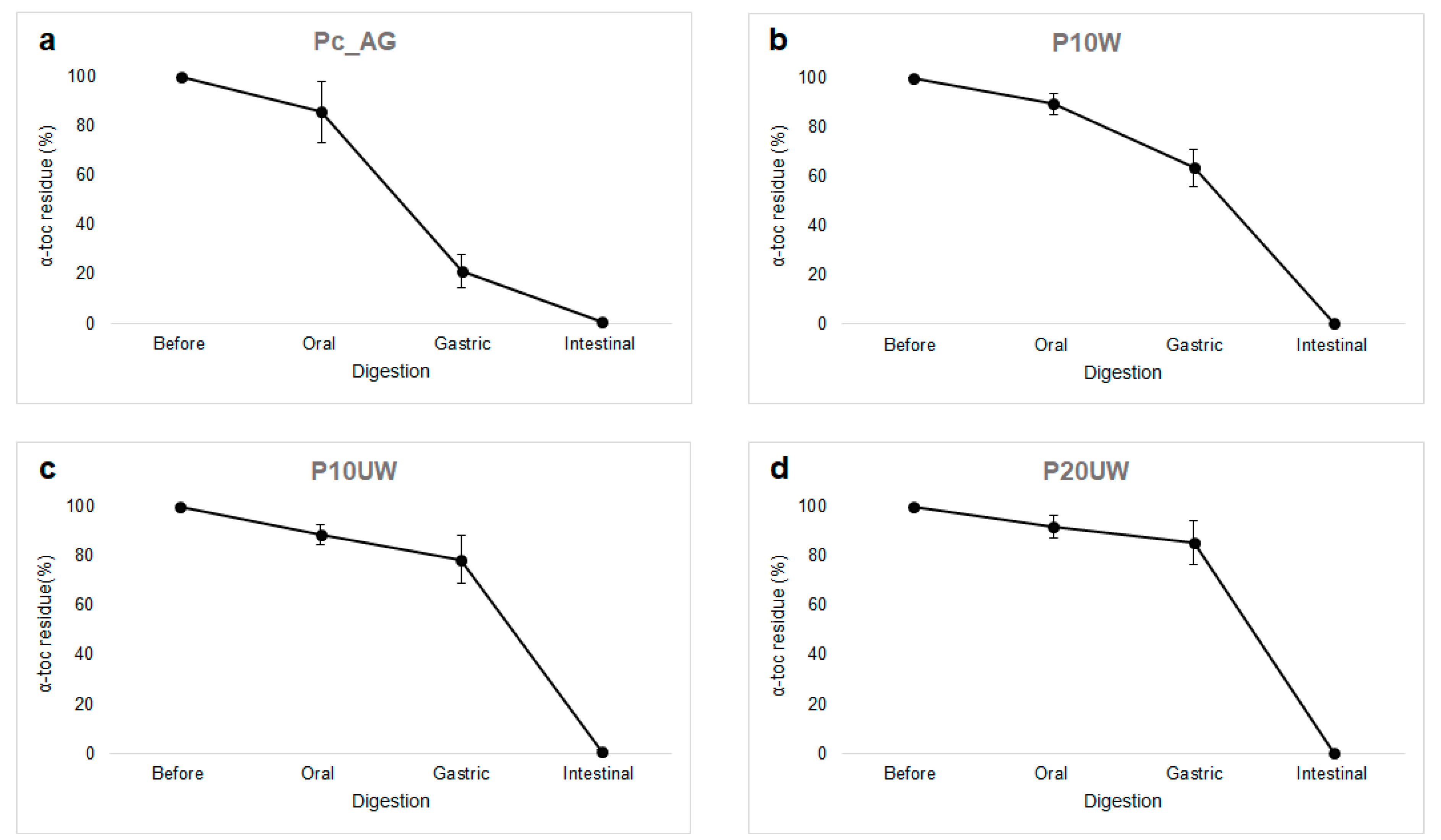
3.2. Effect of In Vitro Digestion on α-Toc Stability
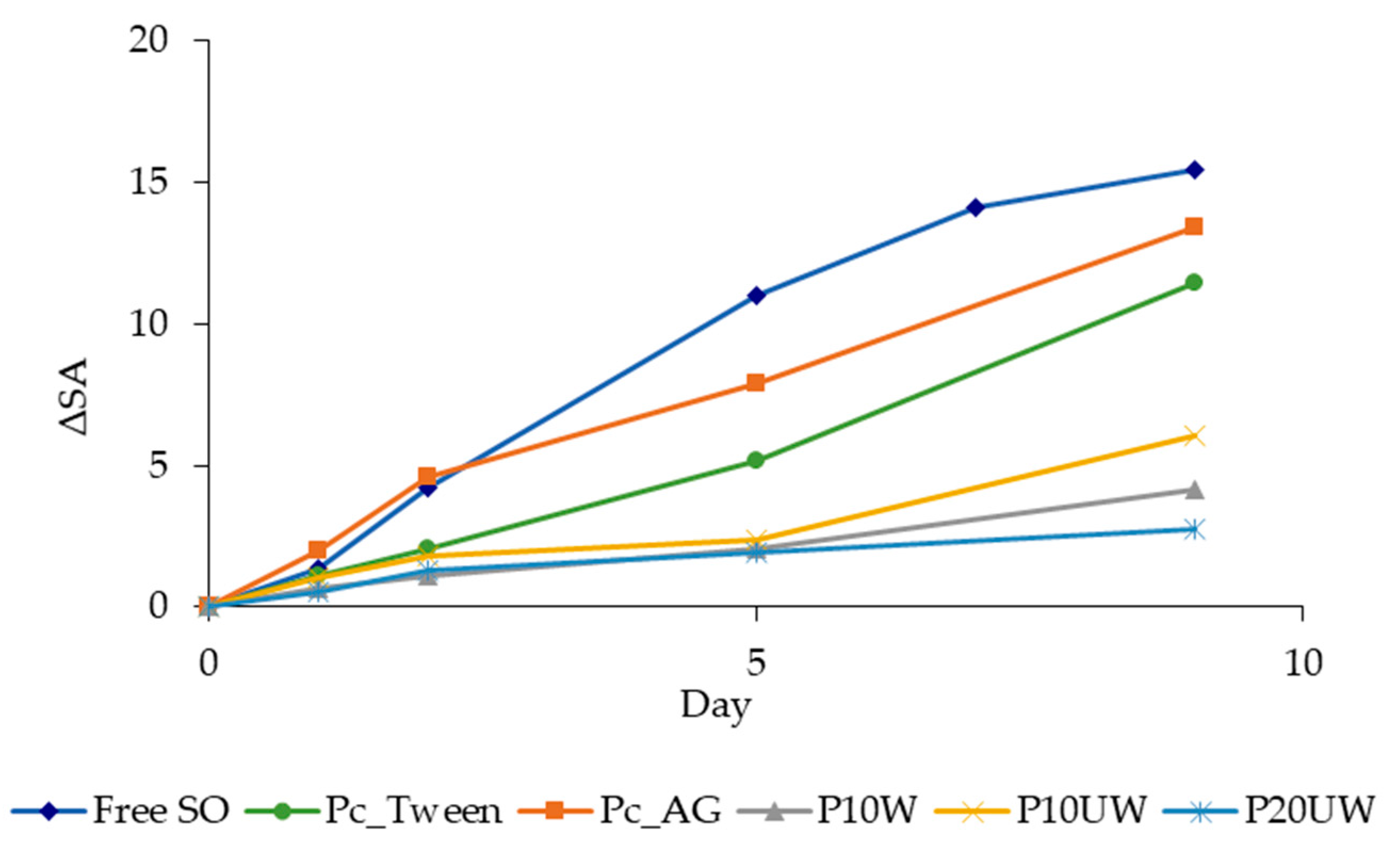
3.3. Oxidative Stability during Storage
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Hernandez Sanchez, M.R.; Cuvelier, M.-E.; Turchiuli, C. Effect of α-tocopherol on oxidative stability of oil during spray drying and storage of dried emulsions. Food Res. Int. 2016, 88, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Wiktorowska-Owczarek, A.; Berezińska, M.; Nowak, J.Z. PUFAs: Structures, Metabolism and Functions. Adv. Clin. Exp. Med. 2015, 24, 931–941. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, Y.; Choe, E. Temperature dependence of the autoxidation and antioxidants of soybean, sunflower, and olive oil. Eur. Food Res. Technol. 2007, 226, 239–246. [Google Scholar] [CrossRef]
- Lee, J.; Koo, N.; Min, D.B. Reactive oxygen species, aging, and antioxidative nutraceuticals. Compr. Rev. Food Sci. Food Saf. 2004, 3, 21–33. [Google Scholar] [CrossRef]
- Shahidi, F.; de Camargo, A.C. Tocopherols and Tocotrienols in Common and Emerging Dietary Sources: Occurrence, Applications, and Health Benefits. Int. J. Mol. Sci. 2016, 17, 1745. [Google Scholar] [CrossRef]
- Chen, B.; McClements, D.J.; Decker, E.A. Design of foods with bioactive lipids for improved health. Annu. Rev. Food Sci. Technol. 2013, 4, 35–56. [Google Scholar] [CrossRef]
- Gallotti, F.; Lavelli, V.; Turchiuli, C. Application of Pleurotus ostreatus β-glucans for oil–in–water emulsions encapsulation in powder. Food Hydrocoll. 2020, 105, 105841. [Google Scholar] [CrossRef]
- Fernandes, R.V.B.; Borges, S.V.; Botrel, D.A. Gum arabic/starch/maltodextrin/inulin as wall materials on the microencapsulation of rosemary essential oil. Carbohydr. Polym. 2014, 101, 524–532. [Google Scholar] [CrossRef]
- Gharsallaoui, A.; Roudaut, G.; Chambin, O.; Voilley, A.; Saurel, R. Applications of spray drying in microencapsulation of food ingredients: An overview. Food Res.Int. 2007, 40, 1107–1121. [Google Scholar] [CrossRef]
- Turchiuli, C.; Jimenez Munguia, M.T.; Hernandez Sanchez, M.R.; Cortes Ferre, H.; Dumoulin, E. Use of different supports for oil encapsulation in powder by spray drying. Powder Technol. 2014, 255, 103–108. [Google Scholar] [CrossRef]
- Hong, J.H.; Jung, H.K. Antioxidant and antitumor activities of β-glucan-rich exopolysaccharides with different molecular weight from Paenibacillus polymyxa JB115. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 105–112. [Google Scholar] [CrossRef]
- Jayakumar, T.; Thomas, P.A.; Sheu, J.R.; Geraldine, P. In vitro and in vivo antioxidant effects of the oyster mushroom Pleurotus ostreatus. Food. Res. Int. 2011, 44, 851–861. [Google Scholar] [CrossRef]
- Nieva-Echevarría, B.; Goicoechea, E.; Guillén, M.D. Food lipid oxidation under gastrointestinal digestion conditions: A review. Crit. Rev. Food Sci. Nutr. 2020, 60, 461–478. [Google Scholar] [CrossRef] [PubMed]
- Kenmogne-Domguia, H.B.; Moisan, S.; Viau, M.; Genot, C.; Meynier, A. The initial characteristics of marine oil emulsions and the composition of the media inflect lipid oxidation during in vitro gastrointestinal digestion. Food Chem. 2014, 152, 146–154. [Google Scholar] [CrossRef]
- Yang, Y.; McClements, D.J. Vitamin E bioaccessibility: Influence of carrier oil type on digestion and release of emulsified a-tocopherol acetate. Food Chem. 2013, 141, 473–481. [Google Scholar] [CrossRef]
- Jeanes, Y.M.; Hall, W.; Ellard, S.; Lee, E.; Lodge, J. The absorption of vitamin E is influenced by the amount of fat in a meal and the food matrix. Br. J. Nutr. 2004, 92, 575. [Google Scholar] [CrossRef]
- Ratnayake, W.M.N.; Galli, C. Fat and Fatty Acid Terminology, Methods of Analysis and Fat Digestion and Metabolism: A Background Review Paper. Ann. Nutr. Metab. 2009, 55, 8–43. [Google Scholar] [CrossRef]
- Singh, H.; Ye, A.; Horne, D. Structuring food emulsions in the gastrointestinal tract to modify lipid digestion. Prog. Lipid Res. 2009, 48, 92–100. [Google Scholar] [CrossRef]
- Bakala N’Goma, J.-C.; Amara, S.; Dridi, K.; Jannin, V.; Carriere, F. Understanding the lipid-digestion processes in the GI tract before designing lipid-based drug-delivery systems. Ther. Delivery 2012, 3, 105–124. [Google Scholar] [CrossRef]
- Carriere, F.; Barrowman, J.A.; Verger, R.; Laugier, R. Secretion and contribution to lipolysis of gastric and pancreatic lipases during a test meal in humans. Gastroenterology 1993, 105, 876–888. [Google Scholar] [CrossRef]
- Brobst, D.F. Pancreatic Function. In Clinical Biochemistry of Domestic Animals, 3rd ed.; Kaneko, J.J., Ed.; Academic Press: Cambridge, MA, USA, 1980; pp. 259–281. [Google Scholar]
- Porter, C.J.H.; Trevaskis, N.L.; Charman, W.N. Lipids and lipid-based formulations: Optimizing the oral delivery of lipophilic drugs. Nat. Rev. Drug Discov. 2007, 6, 231–248. [Google Scholar] [CrossRef] [PubMed]
- Pouton, C.W.; Porter, C.J.H. Formulation of lipid-based delivery systems for oral administration: Materials, methods and strategies. Adv. Drug Deliv. Rev. 2008, 60, 625–637. [Google Scholar] [CrossRef] [PubMed]
- WHO/FAO. Available online: http://www.who.int/nutrition/publications/micronutrients/9241546123/en/ (accessed on 14 September 2020).
- Trucillo, P.; Campardelli, R.; Reverchon, E. Antioxidant loaded emulsions entrapped in liposomes produced using a supercritical assisted technique. J. Supercrit. Fluid 2019, 154, 104626. [Google Scholar] [CrossRef]
- Fang, Z.; Bhandari, B. Effect of spray drying and storage on the stability of bayberry polyphenols. Food Chem. 2011, 129, 1139–1147. [Google Scholar] [CrossRef]
- Ahmad, M.; Ashraf, B.; Gani, A.; Gani, A. Microencapsulation of saffron anthocyanins using β glucan and β cyclodextrin: Microcapsule characterization, release behaviour & antioxidant potential during in-vitro digestion. Int. J. Biol. Macromol. 2018, 109, 435–442. [Google Scholar]
- Guillén, M.D.; Cabo, N. Fourier transform infrared spectra data versus peroxide and anisidine values to determine oxidative stability of edible oils. Food Chem. 2002, 77, 503–510. [Google Scholar] [CrossRef]
- Choe, E.; Min, D.B. Mechanisms and factors for edible oil oxidation. Compr. Rev. Food Sci. Food Saf. 2006, 5, 169–186. [Google Scholar] [CrossRef]
- Taneja, A.; Singh, H. Challenges for the delivery of long-chain n-3 fatty acids in functional foods. Annu. Rev. Food Sci. Technol. 2012, 3, 105–123. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Corredig, M.; Dupont, D.; et al. A standardised static in vitro digestion method suitable for food—An international consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef]
- Turchiuli, C.; Gallotti, F.; Hernandez Sanchez, M.R.; Cuvelier, M.-E. Improvement of oxidative stability of dry emulsion containing antioxidants by modifying process conditions. Chem. Eng. Trans. 2017, 57, 1915–1920. [Google Scholar]
- Gallotti, F.; Turchiuli, C.; Lavelli, V. Production of stable emulsions using β-glucans extracted from Pleurotus ostreatus to encapsulate oxidisable compounds. In Proceedings of the 4th I.C. FABE, Agia Pelagia, Greece, 31 May–2 June 2019; Petrotos, K., Leontopoulos, S., Eds.; University of Thessaly: Volos, Greece, 2019; pp. 201–208. [Google Scholar]
- Christensen, K.L.; Pedersen, G.P.; Kristensen, H.G. Preparation of redispersible dry emulsions by spray drying. Int. J. Pharm. 2001, 212, 187–194. [Google Scholar] [CrossRef]
- Helbig, A.; Silletti, E.; Timmerman, E.; Hamer, R.J.; Gruppen, H. In vitro study of intestinal lipolysis using pH-stat and gas chromatography. Food Hydrocoll. 2012, 28, 10–19. [Google Scholar] [CrossRef]
- Tan, Y. Factors impacting lipid digestion and nutraceutical bioaccessibility assessed by standardized gastrointestinal model (INFOGEST): Emulsifier type. Food Res. Int. 2020, 137, 109739. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, E. Hydrocolloids at interfaces and the influence on the properties of dispersed systems. Food Hydrocoll. 2003, 17, 25–39. [Google Scholar] [CrossRef]
- Nakauma, M.; Funami, T.; Noda, S.; Ishihara, S.; Al-Assaf, S.; Nishinari, K.; Phillips, G.O. Comparison of sugar beet pectin, soybean soluble polysaccharide, and gum arabic as food emulsifiers. 1. Effect of concentration, pH, and salts on the emulsifying properties. Food Hydrocoll. 2008, 22, 1254–1267. [Google Scholar] [CrossRef]
- Gallier, S.; Cui, J.; Olson, T.D.; Rutherfurd, S.M.; Ye, A.; Moughan, P.J.; Singh, H. In vivo digestion of bovine milk fat globules: Effect of processing and interfacial structural changes. I. Gastric digestion. Food Chem. 2013, 141, 3273–3281. [Google Scholar] [CrossRef]
- Giang, T.M.; Le Feunteun, S.; Gaucel, S.; Brestaz, P.; Anton, M.; Meynier, A.; Trelea, I.C. Dynamic modeling highlights the major impact of droplet coalescence on the in vitro digestion kinetics of a whey protein stabilized submicron emulsion. Food Hydrocoll. 2015, 43, 66–72. [Google Scholar] [CrossRef]
- Frascareli, E.C.; Silva, V.M.; Tonon, R.V.; Hubinger, M.D. Effect of process conditions on the microencapsulation of coffee oil by spray drying. Food Bioprod. Process. 2012, 90, 413–424. [Google Scholar] [CrossRef]
- Lv, S.; Zhang, Y.; Tan, H.; Zhang, R.; McClements, D.J. Vitamin E encapsulation within oil-in-water emulsions: Impact of emulsifier type on physicochemical stability and bioaccessibility. J. Agric. Food Chem. 2019, 67, 1521–1529. [Google Scholar] [CrossRef]
- Somchue, W.; Sermsri, W.; Shiowatana, J.; Siripinyanond, A. Encapsulation of a-tocopherol in protein-based delivery particles. Food Res. Int. 2009, 42, 909–914. [Google Scholar] [CrossRef]
- Verkempinck, S.H.E.; Salvia-Trujillo, L.; Denis, S.; Van Loey, A.M.; Hendrickx, M.E.; Grauwet, T. Pectin influences the kinetics of in vitro lipid digestion in oil-in-water emulsions. Food Chem. 2018, 262, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Talón, E.; Vargas, M.; Chiralt, A.; González-Martínez, C. Antioxidant starch-based films with encapsulated eugenol. Application to sunflower oil preservation. LWT 2019, 133, 108290. [Google Scholar] [CrossRef]
- Tan, C.P.; Nakajima, M. b-Carotene nanodispersions: Preparation, characterization and stability evaluation. Food Chem. 2005, 92, 661–671. [Google Scholar] [CrossRef]

| Powder Code | Powder Composition | Extract d.m. Composition | |||||
|---|---|---|---|---|---|---|---|
| Sunflower Oil (SO) (%) | Tween® 20 (%) | AG (%) | MD (%) | Extract d.m. (%) | β-Glucans (%) | Proteins (%) | |
| Pc_Tween | 10 | 0.3 | --- | 89.7 | --- | --- | --- |
| Pc_AG | 10 | --- | 36 | 54 | --- | --- | --- |
| P10W | 10 | --- | --- | 89 | 1 | 0.21 | 0.21 |
| P10UW | 10 | --- | --- | 89 | 1 | 0.22 | 0.14 |
| P20UW | 20 | --- | --- | 78 | 2 | 0.43 | 0.29 |
| Emulsion Reconstituted | Digestion Stage | Particle Size | |||||
|---|---|---|---|---|---|---|---|
| D (3,2) | D (4,3) | ||||||
| Ec_AG | Before | 0.81 ± 0.01 | A | a | 1.15 ± 0.01 | A | a |
| Oral | 0.75 ± 0.05 | A | a | 1.13 ± 0.03 | A | a | |
| Gastric | 0.77 ± 0.06 | A | a | 1.13 ± 0.03 | A | a | |
| Intestinal | 0.68 ± 0.15 | A | a | 299.15 ± 100.12 | B | a | |
| E10W | Before | 1.76 ± 0.01 | A | b | 2.58 ± 0.36 | A | a,b |
| Oral | 1.67 ± 0.04 | A | b | 3.46 ± 0.43 | A | a | |
| Gastric | 1.53 ± 0.24 | A | b | 4.19 ± 1.58 | A | a,b | |
| Intestinal | 0.80 ± 0.10 | B | a | 305.13 ± 83.79 | B | a | |
| E10UW | Before | 2.23 ± 0.02 | A | c | 4.16 ± 0.90 | A | b,c |
| Oral | 1.88 ± 0.31 | A | b | 5.34 ± 3.60 | A | a | |
| Gastric | 2.01 ± 0.33 | A | b | 17.28 ± 1.46 | B | c | |
| Intestinal | 1.83 ± 0.56 | A | a | 407.13 ± 37.51 | C | a | |
| E20UW | Before | 2.64 ± 0.05 | A | d | 4.37 ± 0.38 | A | c |
| Oral | 2.45 ± 0.22 | A | c | 16.81 ± 6.63 | B | b | |
| Gastric | 2.68 ± 1.10 | A | c | 9.53 ± 3.08 | B | b | |
| Intestinal | 8.39 ± 3.05 | B | b | 233.05 ± 27.77 | C | a | |
| Free SO | Pc_Tween | Pc_AG | P10W | P10UW | P20UW | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Day | CD (ΔSA) | |||||||||||||||||
| 0 | 0.0 ± 0.0 | A | a | 0.0 ± 0.0 | A | a | 0.0 ± 0.0 | B,C | a | 0.0 ± 0.0 | A | a | 0.0 ± 0.0 | A | a | 0.0 ± 0.0 | A | a |
| 1 | 1.4 ± 0.2 | A | a | 1.1 ± 0.1 | A,B | a | 2.0 ± 0.9 | B,C | a | 0.6 ± 0.6 | A | a | 1.0 ± 0.7 | A,B | a | 0.6 ± 0.4 | A | a |
| 2 | 4.2 ± 0.6 | B | b,c | 2.1 ± 0.2 | B | a,b | 4.6 ± 1.5 | C | c | 1.2 ± 0.5 | A | a | 1.8 ± 0.7 | A,B | a,b | 1.4 ± 0.7 | A,B | a |
| 5 | 11.0 ± 1.2 | C | c | 5.4 ± 0.4 | C | a,b | 7.9 ± 1.5 | C,D | b,c | 2.1 ± 0.5 | A | a | 2.4 ± 0.3 | B | a | 2.0 ± 0.2 | A,B | a |
| 9 | 15.5 ± 0.5 | D | c | 11.5 ± 0.3 | D | a,b,c | 13.4 ± 5.9 | D | b,c | 4.2 ± 1.1 | B | a | 6.1 ± 0.9 | C | a,b | 2.8 ± 0.2 | A,B | a |
| 12 | n.d. | 15.1 ± 0.6 | E | d | −5.7 ± 0.4 | A | a | 7.0 ± 0.2 | C | c | 7.0 ± 0.8 | C | c | 4.0 ± 0.4 | B,C | b | ||
| 15 | n.d. | 15.4 ± 0.8 | E | d | −3.3 ± 1.3 | A | a | 9.6 ± 0.1 | D | c | 11.3 ± 0.8 | D | c | 6.6 ± 0.1 | C | b | ||
| Day | Degradation of α-Toc (%) | |||||||||||||||||
| 0 | 0 ± 0 | A | a | 0 ± 0 | A | a | 0 ± 0 | A | a | 0 ± 0 | A | a | 0 ± 0 | A | a | 0 ± 0 | A | a |
| 1 | 8 ± 15 | A | a,b | 1 ± 8 | A | a | 27 ± 6 | B | b | 3 ± 1 | A | a | 10 ± 5 | A,B | a,b | 7 ± 4 | A | a,b |
| 2 | 12 ± 11 | A | a | 22 ± 9 | B | a | 64 ± 3 | C | b | 8 ± 4 | A | a | 12 ± 4 | A,B | a | 16 ± 5 | A | a |
| 5 | 42 ± 11 | B | b | 50 ± 2 | C | b | 87 ± 2 | D | c | 17 ± 5 | B | a | 18 ± 3 | B,C | a | 17 ± 7 | A | a |
| 9 | 99 ± 0 | C | c | 67 ± 5 | C | b | 95 ± 1 | D | c | 18 ± 3 | B | a | 20 ± 5 | B,C | a | 25 ± 6 | A | a |
| 12 | n.d. | 95 ± 1 | D | b | 97 ± 1 | D | b | 20 ± 2 | B | a | 28 ± 3 | C,D | a | 28 ± 3 | A | a | ||
| 15 | n.d. | 98 ± 1 | D | b | 97± 1 | D | b | 27 ± 4 | B | a | 37 ± 3 | D | a | 29 ± 7 | A | a | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallotti, F.; Lavoisier, A.; Turchiuli, C.; Lavelli, V. Impact of Pleurotus ostreatus β-Glucans on Oxidative Stability of Active Compounds Encapsulated in Powders during Storage and In Vitro Digestion. Antioxidants 2020, 9, 1219. https://doi.org/10.3390/antiox9121219
Gallotti F, Lavoisier A, Turchiuli C, Lavelli V. Impact of Pleurotus ostreatus β-Glucans on Oxidative Stability of Active Compounds Encapsulated in Powders during Storage and In Vitro Digestion. Antioxidants. 2020; 9(12):1219. https://doi.org/10.3390/antiox9121219
Chicago/Turabian StyleGallotti, Francesca, Anaïs Lavoisier, Christelle Turchiuli, and Vera Lavelli. 2020. "Impact of Pleurotus ostreatus β-Glucans on Oxidative Stability of Active Compounds Encapsulated in Powders during Storage and In Vitro Digestion" Antioxidants 9, no. 12: 1219. https://doi.org/10.3390/antiox9121219
APA StyleGallotti, F., Lavoisier, A., Turchiuli, C., & Lavelli, V. (2020). Impact of Pleurotus ostreatus β-Glucans on Oxidative Stability of Active Compounds Encapsulated in Powders during Storage and In Vitro Digestion. Antioxidants, 9(12), 1219. https://doi.org/10.3390/antiox9121219







