Cardiovascular and Metabolic Protection by Vitamin E: A Matter of Treatment Strategy?
Abstract
:1. Introduction
2. Vitamin E Characterization
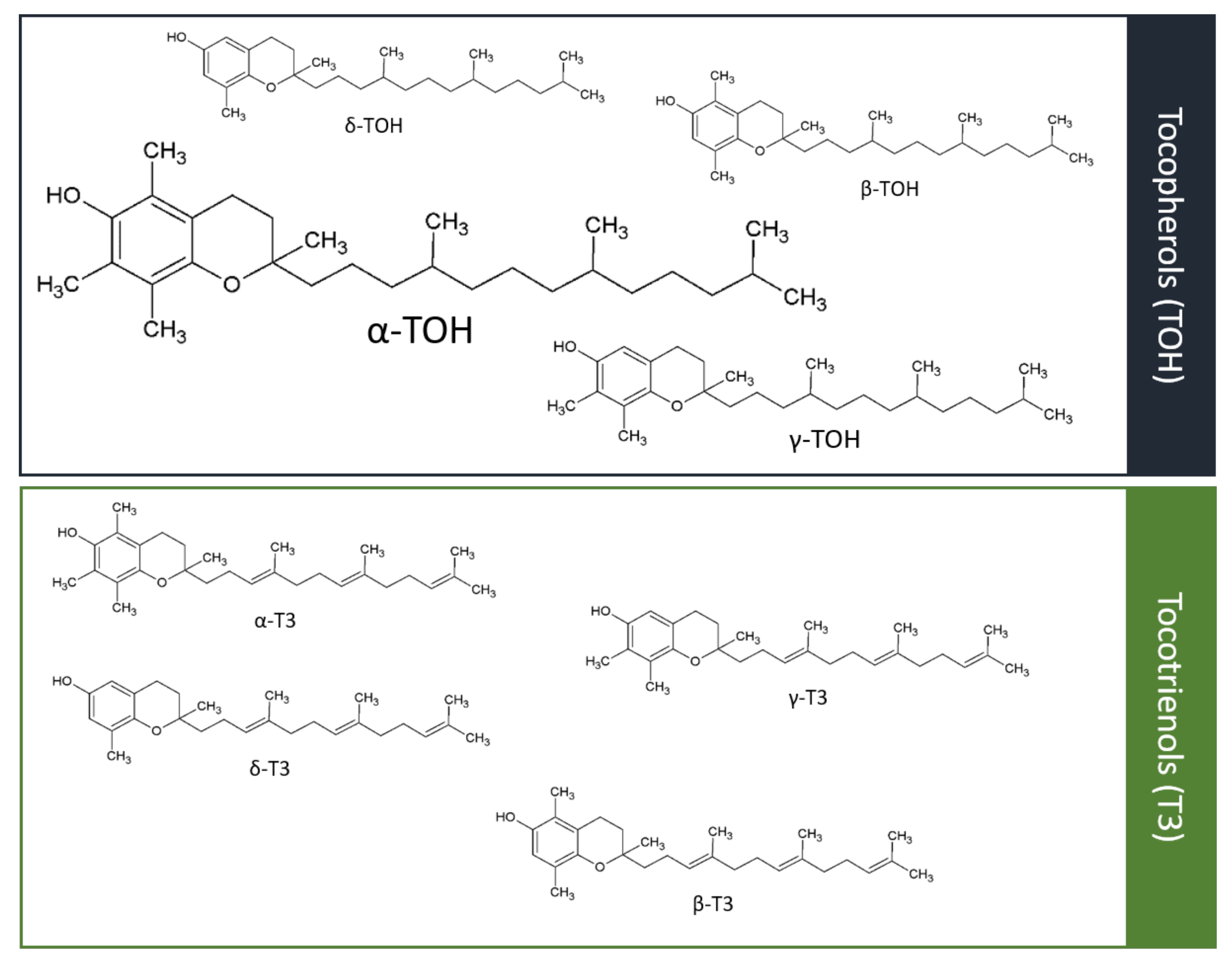
2.1. Different Forms of Vitamin E
2.2. Metabolism of Vitamin E
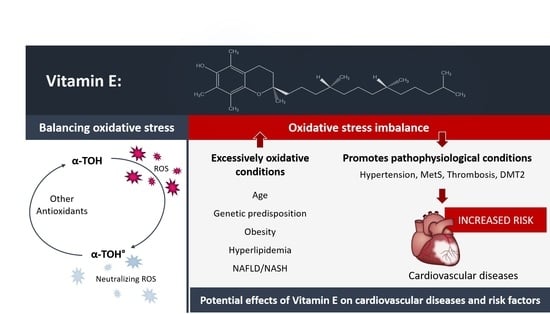
2.3. Relevance and Evidence of Vitamin E as an Antioxidant/Pro-Oxidant and Its Non-Antioxidative Properties
3. Cardiovascular Diseases
3.1. Atherogenesis, Atheroprogression, and Plaque Rupture
3.2. Cardiovascular Events
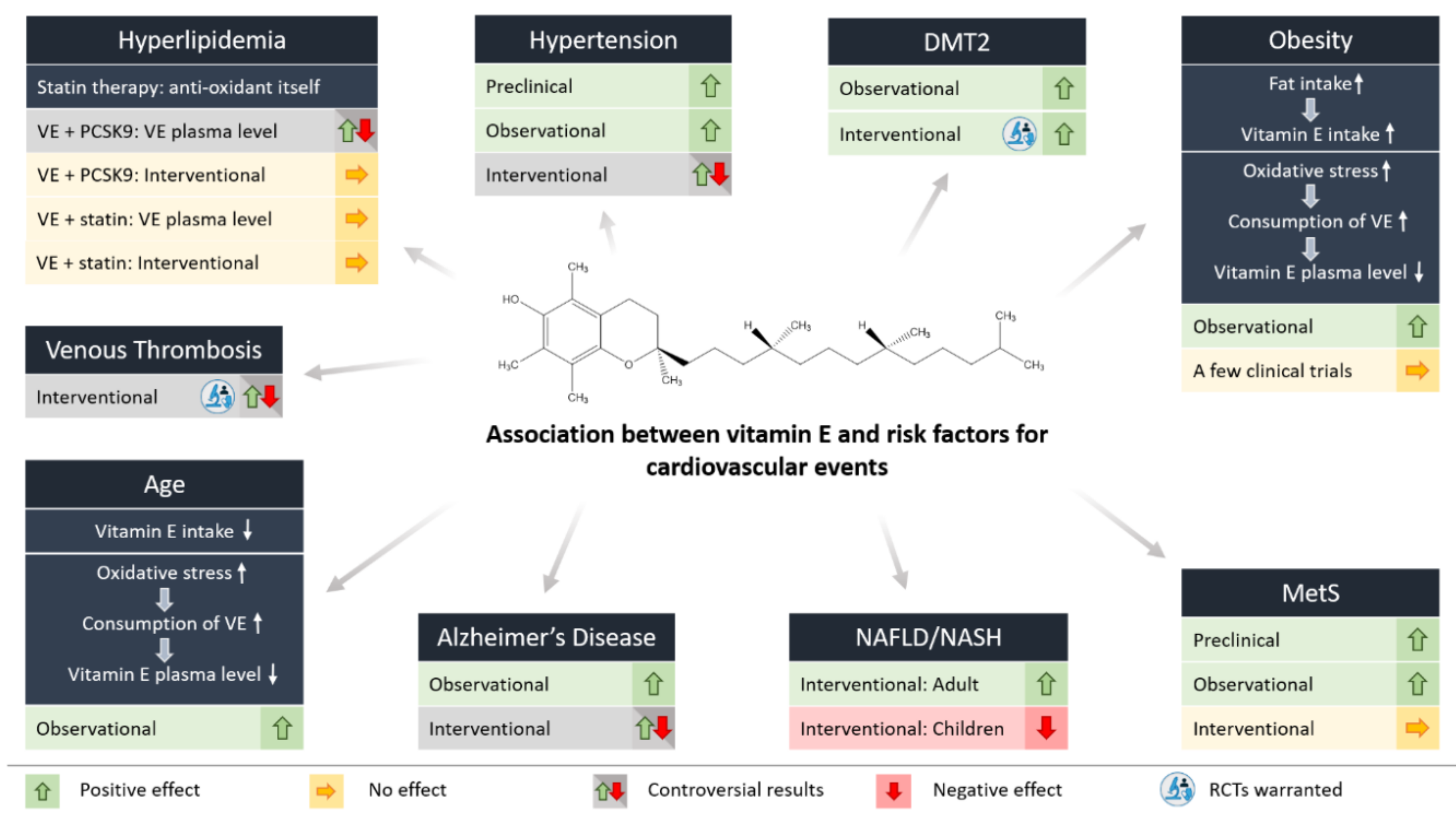
4. Vitamin E and Risk Factors for Cardiovascular Events
4.1. Hypertension
4.2. Hyperlipidemia
4.3. Thrombosis
4.4. Chronic Inflammation
4.4.1. Age
4.4.2. Obesity
4.4.3. Diabetes Mellitus Type 2
4.4.4. Fatty Liver Disease
4.4.5. Metabolic Syndrome
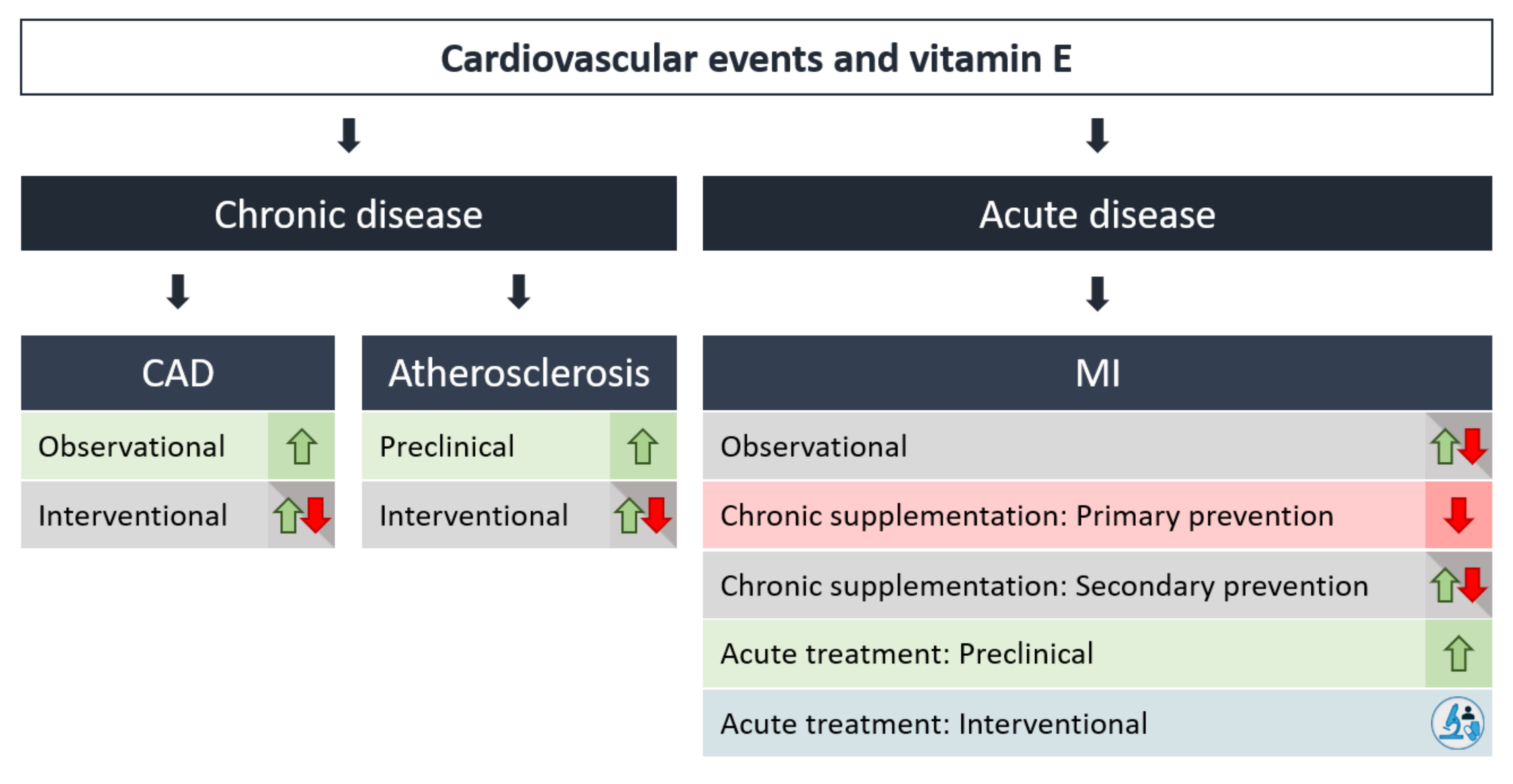
4.5. Cardiovascular Events, Particularly MI
5. Effects of Vitamin E in Chronic vs. Acute Events
5.1. Atherosclerosis/Plaque Formation/Stability and Primary/Secondary Prevention
5.2. Myocardial Infarction
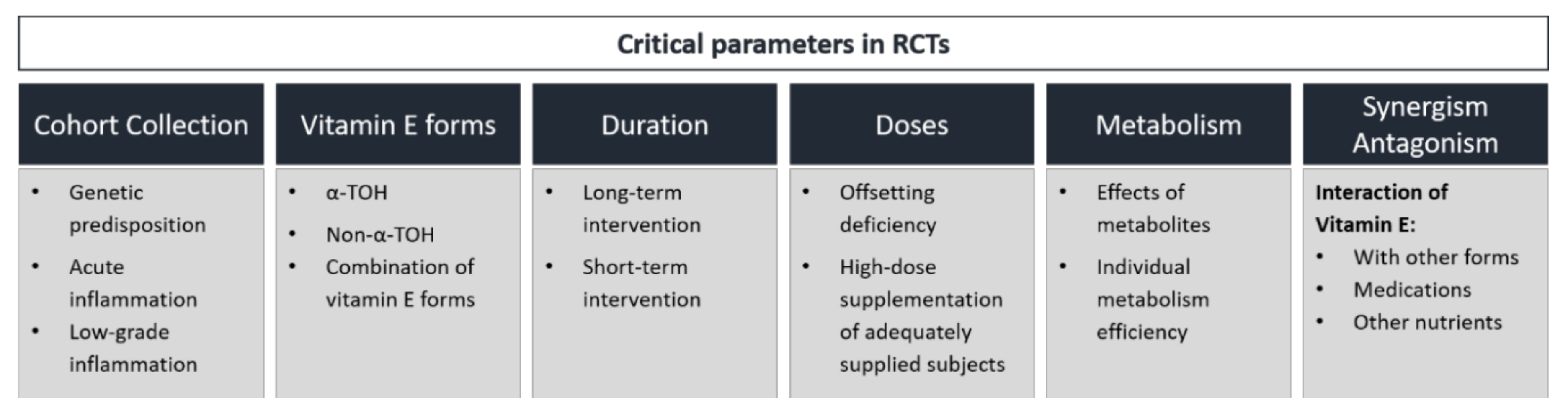
6. Controversial Outcome of RCTs—Explanatory Approaches
6.1. α-TOH vs Non-α-TOH Forms
6.2. Relevance of Metabolites
6.3. Comparison of Vitamin E and Anti-Inflammatory Therapies
6.4. Importance of Cohort Selection and Hp 2-2 Genotype
6.5. High-Dose Supplementation of Vitamin E vs. Balancing Vitamin E Deficiency
7. Merits and Limitations
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Libby, P.; Ridker, P.M.; Maseri, A. Inflammation and atherosclerosis. Circulation 2002, 105, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Marchio, P.; Guerra-Ojeda, S.; Vila, J.M.; Aldasoro, M.; Victor, V.M.; Mauricio, M.D. Targeting early atherosclerosis: A focus on oxidative stress and inflammation. Oxid. Med. Cell. Longev. 2019, 2019, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Baratchi, S.; Khoshmanesh, K.; Woodman, O.L.; Potocnik, S.; Peter, K.; McIntyre, P. Molecular sensors of blood flow in endothelial cells. Trends Mol. Med. 2017, 23, 850–868. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Biochemistry of oxidative stress. Biochem. Soc. Trans. 2007, 35, 1147–1150. [Google Scholar] [CrossRef]
- Murr, C.; Winklhofer-Roob, B.M.; Schroecksnadel, K.; Maritschnegg, M.; Mangge, H.; Böhm, B.O.; Winkelmann, B.R.; März, W.; Fuchs, D. Inverse association between serum concentrations of neopterin and antioxidants in patients with and without angiographic coronary artery disease. Atherosclerosis 2009, 202, 543–549. [Google Scholar] [CrossRef]
- Neuzil, J.; Kågedal, K.; Andera, L.; Weber, C.; Brunk, U.T. Vitamin E analogs: A new class of multiple action agents with anti-neoplastic and anti-atherogenic activity. Apoptosis 2002, 7, 179–187. [Google Scholar] [CrossRef]
- Burton, G.W.; Ingold, K.U. Vitamin E as an in vitro and in vivo antioxidant. Ann. N. Y. Acad. Sci. 1989, 570, 7–22. [Google Scholar] [CrossRef]
- Evans, H.M.; Bishop, K.S. On the existence of a hotherto unrecognized dietary factor essential for reproduction. Science 1922, 56, 650–651. [Google Scholar] [CrossRef] [Green Version]
- Azzi, A. Many tocopherols, one vitamin E. Mol. Asp. Med. 2018, 61, 92–103. [Google Scholar] [CrossRef]
- Traber, M.G.; Kayden, H.J. Preferential incorporation of alpha-tocopherol vs gamma-tocopherol in human lipoproteins. Am. J. Clin. Nutr. 1989, 49, 517–526. [Google Scholar] [CrossRef]
- Schmölz, L.; Birringer, M.; Lorkowski, S.; Wallert, M. Complexity of vitamin E metabolism. World J. Biol. Chem. 2016, 7, 14–43. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US) Panel on Dietary Antioxidants and Related Compounds. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press (US): Washington, DC, USA, 2000; ISBN 978-0-309-06949-6. [Google Scholar]
- Traber, M.G. Vitamin E inadequacy in humans: Causes and consequences. Adv. Nutr. 2014, 5, 503–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shils, M.; Shike, M.; Ross, A.; Caballero, B.; Cousins, R. Nutrition in Health and Disease, 11th ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2012; ISBN 1-60547-461-4. [Google Scholar]
- Grebenstein, N.; Schumacher, M.; Graeve, L.; Frank, J. α-Tocopherol transfer protein is not required for the discrimination against γ-tocopherol in vivo but protects it from side-chain degradation in vitro. Mol. Nutr. Food Res. 2014, 58, 1052–1060. [Google Scholar] [CrossRef] [PubMed]
- Zingg, J.-M.; Meydani, M.; Azzi, A. alpha-Tocopheryl phosphate--an active lipid mediator? Mol. Nutr. Food Res. 2010, 54, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Gianello, R.; Libinaki, R.; Azzi, A.; Gavin, P.D.; Negis, Y.; Zingg, J.-M.; Holt, P.; Keah, H.-H.; Griffey, A.; Smallridge, A.; et al. Alpha-tocopheryl phosphate: A novel, natural form of vitamin E. Free Radic. Biol. Med. 2005, 39, 970–976. [Google Scholar] [CrossRef]
- Traber, M.G.; Rudel, L.L.; Burton, G.W.; Hughes, L.; Ingold, K.U.; Kayden, H.J. Nascent VLDL from liver perfusions of cynomolgus monkeys are preferentially enriched in RRR- compared with SRR-alpha-tocopherol: Studies using deuterated tocopherols. J. Lipid Res. 1990, 31, 687–694. [Google Scholar]
- Parker, R.S.; Sontag, T.J.; Swanson, J.E. Cytochrome P4503A-dependent metabolism of tocopherols and inhibition by sesamin. Biochem. Biophys. Res. Commun. 2000, 277, 531–534. [Google Scholar] [CrossRef]
- Sontag, T.J.; Parker, R.S. Cytochrome P450 ω-hydroxylase pathway of tocopherol catabolism: Novel mechanism of regulation of vitamin E status. J. Biol. Chem. 2002, 277, 25290–25296. [Google Scholar] [CrossRef] [Green Version]
- Birringer, M.; Pfluger, P.; Kluth, D.; Landes, N.; Brigelius-Flohé, R. Identities and differences in the metabolism of tocotrienols and tocopherols in HepG2 Cells. J. Nutr. 2002, 132, 3113–3118. [Google Scholar] [CrossRef]
- Kiyose, C.; Saito, H.; Kaneko, K.; Hamamura, K.; Tomioka, M.; Ueda, T.; Igarashi, O. Alpha-tocopherol affects the urinary and biliary excretion of 2,7,8-trimethyl-2 (2′-carboxyethyl)-6-hydroxychroman, gamma-tocopherol metabolite, in rats. Lipids 2001, 36, 467–472. [Google Scholar] [CrossRef]
- Schultz, M.; Leist, M.; Petrzika, M.; Gassmann, B.; Brigelius-Flohé, R. Novel urinary metabolite of alpha-tocopherol, 2,5,7,8-tetramethyl-2(2′-carboxyethyl)-6-hydroxychroman, as an indicator of an adequate vitamin E supply? Am. J. Clin. Nutr. 1995, 62, 1527S–1534S. [Google Scholar] [CrossRef] [PubMed]
- Lebold, K.M.; Ang, A.; Traber, M.G.; Arab, L. Urinary α-carboxyethyl hydroxychroman can be used as a predictor of α-tocopherol adequacy, as demonstrated in the Energetics Study. Am. J. Clin. Nutr. 2012, 96, 801–809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Birringer, M.; EyTina, J.H.; Salvatore, B.A.; Neuzil, J. Vitamin E analogues as inducers of apoptosis: Structure-function relation. Br. J. Cancer 2003, 88, 1948–1955. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Traber, M.G.; Atkinson, J. Vitamin E, antioxidant and nothing more. Free Radic. Biol. Med. 2007, 43, 4–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brigelius-Flohé, R. Vitamin E: The shrew waiting to be tamed. Free Radic. Biol. Med. 2009, 46, 543–554. [Google Scholar] [CrossRef]
- Kontush, A.; Finckh, B.; Karten, B.; Kohlschütter, A.; Beisiegel, U. Antioxidant and prooxidant activity of alpha-tocopherol in human plasma and low density lipoprotein. J. Lipid Res. 1996, 37, 1436–1448. [Google Scholar]
- Miller, E.R.; Pastor-Barriuso, R.; Dalal, D.; Riemersma, R.A.; Appel, L.J.; Guallar, E. Meta-analysis: High-dosage vitamin E supplementation may increase all-cause mortality. Ann. Intern. Med. 2005, 142, 37–46. [Google Scholar] [CrossRef] [Green Version]
- Pearson, P.; Lewis, S.A.; Britton, J.; Young, I.S.; Fogarty, A. The pro-oxidant activity of high-dose vitamin E supplements in vivo. BioDrugs 2006, 20, 271–273. [Google Scholar] [CrossRef]
- Gerss, J.; Köpcke, W. The questionable association of vitamin E supplementation and mortality--inconsistent results of different meta-analytic approaches. Cell. Mol. Biol. (Noisy-Le-Grand) 2009, 55 Suppl. 1, OL1111–OL1120. [Google Scholar]
- Meagher, E.A.; Barry, O.P.; Lawson, J.A.; Rokach, J.; FitzGerald, G.A. Effects of vitamin E on lipid peroxidation in healthy persons. JAMA 2001, 285, 1178–1182. [Google Scholar] [CrossRef]
- Roberts, L.J.; Oates, J.A.; Linton, M.F.; Fazio, S.; Meador, B.P.; Gross, M.D.; Shyr, Y.; Morrow, J.D. The relationship between dose of vitamin E and suppression of oxidative stress in humans. Free Radic. Biol. Med. 2007, 43, 1388–1393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davi, G.; Alessandrini, P.; Mezzetti, A.; Minotti, G.; Bucciarelli, T.; Costantini, F.; Cipollone, F.; Bon, G.B.; Ciabattoni, G.; Patrono, C. In vivo formation of 8-Epi-prostaglandin F2 alpha is increased in hypercholesterolemia. Arter. Thromb. Vasc. Biol. 1997, 17, 3230–3235. [Google Scholar] [CrossRef] [PubMed]
- Hathcock, J.N.; Azzi, A.; Blumberg, J.; Bray, T.; Dickinson, A.; Frei, B.; Jialal, I.; Johnston, C.S.; Kelly, F.J.; Kraemer, K.; et al. Vitamins E and C are safe across a broad range of intakes. Am. J. Clin. Nutr. 2005, 81, 736–745. [Google Scholar] [CrossRef]
- Hickman, K.C.D.; Harris, P.L. Tocopherol interrelationships. In Advances in Enzymology and Related Areas of Molecular Biology; Nord, F.F., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2006; pp. 469–524. ISBN 978-0-470-12251-8. [Google Scholar]
- Azzi, A. Molecular mechanism of alpha-tocopherol action. Free Radic. Biol. Med. 2007, 43, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Azzi, A.; Ricciarelli, R.; Zingg, J.M. Non-antioxidant molecular functions of alpha-tocopherol (vitamin E). FEBS Lett. 2002, 519, 8–10. [Google Scholar] [CrossRef] [Green Version]
- Schächinger, V.; Zeiher, A.M. Atherogenesis—Recent insights into basic mechanisms and their clinical impact. Nephrol. Dial. Transpl. 2002, 17, 2055–2064. [Google Scholar] [CrossRef] [Green Version]
- Weber, C.; Noels, H. Atherosclerosis: Current pathogenesis and therapeutic options. Nat. Med. 2011, 17, 1410–1422. [Google Scholar] [CrossRef]
- Htun, N.M.; Chen, Y.C.; Lim, B.; Schiller, T.; Maghzal, G.J.; Huang, A.L.; Elgass, K.D.; Rivera, J.; Schneider, H.G.; Wood, B.R.; et al. Near-infrared autofluorescence induced by intraplaque hemorrhage and heme degradation as marker for high-risk atherosclerotic plaques. Nat. Commun. 2017, 8, 75. [Google Scholar] [CrossRef] [Green Version]
- Rafieian-Kopaei, M.; Setorki, M.; Doudi, M.; Baradaran, A.; Nasri, H. Atherosclerosis: Process, indicators, risk factors and new hopes. Int. J. Prev. Med. 2014, 5, 927–946. [Google Scholar]
- Nording, H.M.; Seizer, P.; Langer, H.F. Platelets in Inflammation and Atherogenesis. Front. Immunol. 2015, 6. [Google Scholar] [CrossRef] [Green Version]
- Palasubramaniam, J.; Wang, X.; Peter, K. Myocardial infarction—From atherosclerosis to thrombosis. Arterioscler. Thromb. Vasc. Biol. 2019, 39, e176–e185. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das Sandeep, R.; et al. Heart disease and stroke statistics—2019 update: A report from the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.; Manmathan, G.; Wilkinson, P. Primary prevention of cardiovascular disease: A review of contemporary guidance and literature. JRSM Cardiovasc. Dis. 2017, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Batty, G.D.; Kivimäki, M.; Bell, S. Comparison of risk factors for coronary heart disease morbidity versus mortality. Eur. J. Prev. Cardiol. 2019, 2047487319882512. [Google Scholar] [CrossRef]
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). Eur. Heart J. 2020, 41, 407–477. [Google Scholar] [CrossRef]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; et al. Fourth universal definition of myocardial infarction (2018). Eur. Heart J. 2019, 40, 237–269. [Google Scholar] [CrossRef] [Green Version]
- Jennings, R.B.; Sommers, H.M.; Smyth, G.A.; Flack, H.A.; Linn, H. Myocardial necrosis induced by temporary occlusion of a coronary artery in the dog. Arch. Pathol. 1960, 70, 68–78. [Google Scholar]
- Ziegler, M.; Xu, X.; Yap, M.L.; Hu, H.; Zhang, J.; Peter, K. A self-assembled fluorescent nanoprobe for imaging and therapy of cardiac ischemia/reperfusion injury. Adv. Ther. 2019, 2, 1800133. [Google Scholar] [CrossRef]
- van Nieuwenhoven, F.A.; Turner, N.A. The role of cardiac fibroblasts in the transition from inflammation to fibrosis following myocardial infarction. Vasc. Pharm. 2013, 58, 182–188. [Google Scholar] [CrossRef] [Green Version]
- Kuwabara, A.; Nakade, M.; Tamai, H.; Tsuboyama-Kasaoka, N.; Tanaka, K. The association between vitamin E intake and hypertension: Results from the re-analysis of the National Health and Nutrition Survey. J. Nutr. Sci. Vitam. 2014, 60, 239–245. [Google Scholar] [CrossRef] [Green Version]
- Boshtam, M.; Rafiei, M.; Sadeghi, K.; Sarraf-Zadegan, N. Vitamin E can reduce blood pressure in mild hypertensives. Int. J. Vitam. Nutr. Res. 2002, 72, 309–314. [Google Scholar] [CrossRef]
- Santhoshakumari, T.M.J.; Mathivanan, J.; Jeyalakshmi, J. Role of vitamin C and vitamin E on hypertension. Asian J. Pharm. Clin. Res. 2019, 95–98. [Google Scholar] [CrossRef] [Green Version]
- Palumbo, G.; Avanzini, F.; Alli, C.; Roncaglioni, M.C.; Ronchi, E.; Cristofari, M.; Capra, A.; Rossi, S.; Nosotti, L.; Costantini, C.; et al. Effects of vitamin E on clinic and ambulatory blood pressure in treated hypertensive patients. Collaborative Group of the Primary Prevention Project (PPP)--Hypertension study. Am. J. Hypertens. 2000, 13, 564–567. [Google Scholar] [CrossRef] [Green Version]
- Mihalj, M.; Tadzic, R.; Vcev, A.; Rucevic, S.; Drenjancevic, I. Blood pressure reduction is associated with the changes in oxidative stress and endothelial activation in hypertension, regardless of antihypertensive therapy. KBR 2016, 41, 721–735. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M.; Dominguez, L.J.; Tagliamonte, M.R.; Resnick, L.M.; Paolisso, G. Effects of vitamin E and glutathione on glucose metabolism. Hypertension 1999, 34, 1002–1006. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cangemi, R.; Loffredo, L.; Carnevale, R.; Perri, L.; Patrizi, M.P.; Sanguigni, V.; Pignatelli, P.; Violi, F. Early decrease of oxidative stress by atorvastatin in hypercholesterolaemic patients: Effect on circulating vitamin E. Eur. Heart J. 2008, 29, 54–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, M.-J.; Chung, N.; Lee, J.H.; Jang, Y.; Park, E.; Jeon, K.-I.; Chung, J.H.; Seo, B.-Y. Effects of simvastatin on plasma antioxidant status and vitamins in hypercholesterolemic patients. Int. J. Cardiol. 2007, 118, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Blom, D.J.; Djedjos, C.S.; Monsalvo, M.L.; Bridges, I.; Wasserman, S.M.; Scott, R.; Roth, E. Effects of evolocumab on vitamin E and steroid hormone levels: Results from the 52-Week, phase 3, double-blind, randomized, placebo-controlled DESCARTES study. Circ. Res. 2015, 117, 731–741. [Google Scholar] [CrossRef]
- Liu, C.-S.; Lii, C.-K.; Chang, L.-L.; Kuo, C.-L.; Cheng, W.-L.; Su, S.-L.; Tsai, C.-W.; Chen, H.-W. Atorvastatin increases blood ratios of vitamin E/low-density lipoprotein cholesterol and coenzyme Q10/low-density lipoprotein cholesterol in hypercholesterolemic patients. Nutr. Res. 2010, 30, 118–124. [Google Scholar] [CrossRef]
- Leonard, S.W.; Joss, J.D.; Mustacich, D.J.; Blatt, D.H.; Lee, Y.S.; Traber, M.G. Effects of vitamin E on cholesterol levels of hypercholesterolemic patients receiving statins. Am. J. Health Syst. Pharm. 2007, 64, 2257–2266. [Google Scholar] [CrossRef]
- Glynn, R.J.; Ridker, P.M.; Goldhaber, S.Z.; Zee Robert, Y.L.; Buring, J.E. Effects of random allocation to vitamin E supplementation on the occurrence of venous thromboembolism. Circulation 2007, 116, 1497–1503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vučković, B.A.; van Rein, N.; Cannegieter, S.C.; Rosendaal, F.R.; Lijfering, W.M. Vitamin supplementation on the risk of venous thrombosis: Results from the MEGA case-control study. Am. J. Clin. Nutr 2015, 101, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Ortega, R.M.; Requejo, A.M.; López-Sobaler, A.M.; Andrés, P.; Navia, B.; Perea, J.M.; Robles, F. Cognitive function in elderly people is influenced by vitamin E status. J. Nutr. 2002, 132, 2065–2068. [Google Scholar] [CrossRef] [Green Version]
- Vatassery, G.T.; Johnson, G.J.; Krezowski, A.M. Changes in vitamin E concentrations in human plasma and platelets with age. J. Am. Coll. Nutr. 1983, 2, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Capuron, L.; Moranis, A.; Combe, N.; Cousson-Gélie, F.; Fuchs, D.; De Smedt-Peyrusse, V.; Barberger-Gateau, P.; Layé, S. Vitamin E status and quality of life in the elderly: Influence of inflammatory processes. Br. J. Nutr. 2009, 102, 1390–1394. [Google Scholar] [CrossRef] [Green Version]
- Requejo, A.M.; Andrés, P.; Redondo, M.R.; Mena, M.C.; Navia, B.; Perea, J.M.; Lopez-Sobaler, A.M.; Ortega, R.M. Vitamin E status in a group of elderly people from Madrid. J. Nutr. Health Aging 2002, 6, 72–74. [Google Scholar]
- Rudman, D.; Abbasi, A.A.; Isaacson, K.; Karpiuk, E. Observations on the nutrient intakes of eating-dependent nursing home residents: Underutilization of micronutrient supplements. J. Am. Coll. Nutr. 1995, 14, 604–613. [Google Scholar] [CrossRef]
- De la Fuente, M.; Hernanz, A.; Guayerbas, N.; Victor, V.M.; Arnalich, F. Vitamin E ingestion improves several immune functions in elderly men and women. Free Radic. Res. 2008, 42, 272–280. [Google Scholar] [CrossRef]
- da Silva, I.T.; de Queiroz Mello, A.P.; Sanches, L.B.; Abdalla, D.S.P.; Damasceno, N.R.T. Is plasma alpha-tocopherol associated with electronegative LDL in obese adolescents? J. Nutr. Sci. Vitam. 2013, 59, 100–107. [Google Scholar] [CrossRef]
- Mehmetoglu, I.; Yerlikaya, F.H.; Kurban, S. Correlation between vitamin A, E, coenzyme Q(10) and degree of insulin resistance in obese and non-obese subjects. J. Clin. Biochem. Nutr. 2011, 49, 159–163. [Google Scholar] [CrossRef] [Green Version]
- Kuno, T.; Hozumi, M.; Morinobu, T.; Murata, T.; Mingci, Z.; Tamai, H. Antioxidant vitamin levels in plasma and low density lipoprotein of obese girls. Free Radic. Res. 1998, 28, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Strauss, R.S. Comparison of serum concentrations of α-tocopherol and β-carotene in a cross-sectional sample of obese and nonobese children (NHANES III). J. Pediatr. 1999, 134, 160–165. [Google Scholar] [CrossRef]
- Molnár, D.; Decsi, T.; Koletzko, B. Reduced antioxidant status in obese children with multimetabolic syndrome. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1197–1202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gunanti, I.R.; Marks, G.C.; Al-Mamun, A.; Long, K.Z. Low serum concentrations of carotenoids and vitamin E are associated with high adiposity in Mexican-American children. J. Nutr. 2014, 144, 489–495. [Google Scholar] [CrossRef] [Green Version]
- Schneider, M.; Verges, B.; Klein, A.; Miller, E.R.; Deckert, V.; Desrumaux, C.; Masson, D.; Gambert, P.; Brun, J.-M.; Fruchart-Najib, J.; et al. Alterations in plasma vitamin E distribution in type 2 diabeticpatients with elevated plasma phospholipid transfer protein Activity. Diabetes 2004, 53, 2633–2639. [Google Scholar] [CrossRef] [Green Version]
- Galvan, A.Q.; Muscelli, E.; Catalano, C.; Natali, A.; Sanna, G.; Masoni, A.; Bernardini, B.; Barsacchi, R.; Ferrannini, E. Insulin decreases circulating vitamin E levels in humans. Metab. Clin. Exp. 1996, 45, 998–1003. [Google Scholar] [CrossRef]
- Kollerits, B.; Lamina, C.; Huth, C.; Marques-Vidal, P.; Kiechl, S.; Seppälä, I.; Cooper, J.; Hunt, S.C.; Meisinger, C.; Herder, C.; et al. Plasma concentrations of afamin are associated with prevalent and incident type 2 diabetes: A pooled analysis in more than 20,000 individuals. Diabetes Care 2017, 40, 1386–1393. [Google Scholar] [CrossRef] [Green Version]
- Mayer-Davis, E.J.; Costacou, T.; King, I.; Zaccaro, D.J.; Bell, R.A. Plasma and dietary vitamin E in relation to incidence of type 2 diabetes: The insulin resistance and atherosclerosis study (IRAS). Diabetes Care 2002, 25, 2172–2177. [Google Scholar] [CrossRef] [Green Version]
- Škrha, J.; Prázný, M.; Hilgertová, J.; Weiserová, H. Serum α-tocopherol and ascorbic acid concentrations in Type 1 and Type 2 diabetic patients with and without angiopathy. Clin. Chim. Acta 2003, 329, 103–108. [Google Scholar] [CrossRef]
- Salonen, R.M.; Nyyssönen, K.; Kaikkonen, J.; Porkkala-Sarataho, E.; Voutilainen, S.; Rissanen, T.H.; Tuomainen, T.-P.; Valkonen, V.-P.; Ristonmaa, U.; Lakka, H.-M.; et al. Six-year effect of combined vitamin C and E supplementation on atherosclerotic progression: The antioxidant supplementation in atherosclerosis prevention (ASAP) study. Circulation 2003, 107, 947–953. [Google Scholar] [CrossRef]
- Eljaoudi, R.; Elomri, N.; Laamarti, M.; Cherrah, Y.; Amezyane, T.; Ghafir, D.; Ibrahimi, A. Antioxidants status in type 2 diabetic patients in Morocco. Turk. J. Med. Sci. 2017, 47, 782–788. [Google Scholar] [CrossRef]
- Nourooz-Zadeh, J.; Rahimi, A.; Tajaddini-Sarmadi, J.; Tritschler, H.; Rosen, P.; Halliwell, B.; Betteridge, D.J. Relationships between plasma measures of oxidative stress and metabolic control in NIDDM. Diabetologia 1997, 40, 647–653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rafighi, Z.; Shiva, A.; Arab, S.; Mohd Yousof, R. Association of dietary vitamin C and E intake and antioxidant enzymes in type 2 diabetes mellitus patients. GJHS 2013, 5, 183. [Google Scholar] [CrossRef] [PubMed]
- Manning, P.J.; Sutherland, W.H.F.; Walker, R.J.; Williams, S.M.; De Jong, S.A.; Ryalls, A.R.; Berry, E.A. Effect of high-dose vitamin E on insulin resistance and associated parameters in overweight subjects. Diabetes Care 2004, 27, 2166–2171. [Google Scholar] [CrossRef] [Green Version]
- Xu, R.; Zhang, S.; Tao, A.; Chen, G.; Zhang, M. Influence of vitamin E supplementation on glycaemic control: A meta-analysis of randomised controlled trials. PLoS ONE 2014, 9, e95008. [Google Scholar] [CrossRef]
- Erhardt, A.; Stahl, W.; Sies, H.; Lirussi, F.; Donner, A.; Häussinger, D. Plasma levels of vitamin E and carotenoids are decreased in patients with Nonalcoholic Steatohepatitis (NASH). Eur. J. Med. Res. 2011, 16, 76–78. [Google Scholar] [CrossRef]
- Machado, M.V.; Ravasco, P.; Jesus, L.; Marques-Vidal, P.; Oliveira, C.R.; Proença, T.; Baldeiras, I.; Camilo, M.E.; Cortez-Pinto, H. Blood oxidative stress markers in non-alcoholic steatohepatitis and how it correlates with diet. Scand. J. Gastroenterol. 2008, 43, 95–102. [Google Scholar] [CrossRef]
- Violet, P.-C.; Ebenuwa, I.C.; Wang, Y.; Niyyati, M.; Padayatty, S.J.; Head, B.; Wilkins, K.; Chung, S.; Thakur, V.; Ulatowski, L.; et al. Vitamin E sequestration by liver fat in humans. JCI Insight 2020, 5, e133309. [Google Scholar] [CrossRef]
- Sanyal, A.J.; Chalasani, N.; Kowdley, K.V.; McCullough, A.; Diehl, A.M.; Bass, N.M.; Neuschwander-Tetri, B.A.; Lavine, J.E.; Tonascia, J.; Unalp, A.; et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N. Engl. J. Med. 2010, 362, 1675–1685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lavine, J.E. Vitamin E treatment of nonalcoholic steatohepatitis in children: A pilot study. J. Pediatr. 2000, 136, 734–738. [Google Scholar] [CrossRef]
- Ford, E.S.; Mokdad, A.H.; Giles, W.H.; Brown, D.W. The metabolic syndrome and antioxidant concentrations: Findings from the Third National Health and Nutrition Examination Survey. Diabetes 2003, 52, 2346–2352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beydoun, M.A.; Shroff, M.R.; Chen, X.; Beydoun, H.A.; Wang, Y.; Zonderman, A.B. Serum antioxidant status is associated with metabolic syndrome among U.S. adults in recent national surveys. J. Nutr. 2011, 141, 903–913. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.-H.; Yang, N.-C.; Lee, B.-J.; Lin, J.-Y.; Hsia, S.; Lin, P.-T. The antioxidant status and concentrations of coenzyme Q10 and vitamin E in metabolic syndrome. Sci. World J. 2013, 2013, 767968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mah, E.; Sapper, T.N.; Chitchumroonchokchai, C.; Failla, M.L.; Schill, K.E.; Clinton, S.K.; Bobe, G.; Traber, M.G.; Bruno, R.S. α-Tocopherol bioavailability is lower in adults with metabolic syndrome regardless of dairy fat co-ingestion: A randomized, double-blind, crossover trial. Am. J. Clin. Nutr. 2015, 102, 1070–1080. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E.; Collins, K.J.; Dennison, H.C.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 2018, 71, e13–e115. [Google Scholar] [CrossRef]
- Emami, M.R.; Safabakhsh, M.; Alizadeh, S.; Asbaghi, O.; Khosroshahi, M.Z. Effect of vitamin E supplementation on blood pressure: A systematic review and meta-analysis. J. Hum. Hypertens. 2019, 33, 499–507. [Google Scholar] [CrossRef]
- Hodgson, J.M.; Croft, K.D.; Woodman, R.J.; Puddey, I.B.; Bondonno, C.P.; Wu, J.H.Y.; Beilin, L.J.; Lukoshkova, E.V.; Head, G.A.; Ward, N.C. Effects of vitamin E, vitamin C and polyphenols on the rate of blood pressure variation: Results of two randomised controlled trials. Br. J. Nutr. 2014, 112, 1551–1561. [Google Scholar] [CrossRef] [Green Version]
- Mellyana, O.; Widajat, R.; Sekarwana, N. Combined supplementation with α-tocopherol and vitamin C improves the blood pressure of pediatric idiopathic nephrotic syndrome patients. Clin. Nutr. Exp. 2019, 23, 116–121. [Google Scholar] [CrossRef]
- Sorriento, D.; Luca, N.D.; Trimarco, B.; Iaccarino, G. The antioxidant therapy: New insights in the treatment of hypertension. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef] [Green Version]
- Cuevas, S.; Villar, V.A.M.; Jose, P.A. Genetic polymorphisms associated with reactive oxygen species and blood pressure regulation. Pharm. J. 2019, 19, 315–336. [Google Scholar] [CrossRef]
- Kizhakekuttu, T.J.; Widlansky, M.E. Natural antioxidants and hypertension: Promise and challenges. Cardiovasc. Ther. 2010, 28, e20–e32. [Google Scholar] [CrossRef] [PubMed]
- Gencer, B.; Mach, F. Lipid management in ACS: Should we go lower faster? Atherosclerosis 2018, 275, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Borén, J.; Chapman, M.J.; Krauss, R.M.; Packard, C.J.; Bentzon, J.F.; Binder, C.J.; Daemen, M.J.; Demer, L.L.; Hegele, R.A.; Nicholls, S.J.; et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease: Pathophysiological, genetic, and therapeutic insights: A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart J. 2020, 41, 2313–2330. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of blood cholesterol: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2019, 73, e285–e350. [Google Scholar] [CrossRef] [PubMed]
- Galli, F.; Iuliano, L. Do statins cause myopathy by lowering vitamin E levels? Med. Hypotheses 2010, 74, 707–709. [Google Scholar] [CrossRef]
- De Caterina, R.; Cipollone, F.; Filardo, F.P.; Zimarino, M.; Bernini, W.; Lazzerini, G.; Bucciarelli, T.; Falco, A.; Marchesani, P.; Muraro, R.; et al. Low-Density Lipoprotein level reduction by the 3-Hydroxy-3-Methylglutaryl Coenzyme-A inhibitor simvastatin is accompanied by a related reduction of F2-Isoprostane formation in hypercholesterolemic subjects: No further effect of vitamin E. Circulation 2002, 106, 2543–2549. [Google Scholar] [CrossRef] [Green Version]
- Gotto, A.M. Antioxidants, statins, and atherosclerosis. J. Am. Coll. Cardiol. 2003, 41, 1205–1210. [Google Scholar] [CrossRef] [Green Version]
- Qamar, A.; Bhatt, D.L. Effect of low cholesterol on steroid hormones and vitamin E levels: Just a theory or real concern? Circ. Res. 2015, 117, 662–664. [Google Scholar] [CrossRef] [Green Version]
- Violi, F.; Cangemi, R. Statin treatment as a confounding factor in human trials with vitamin E. J. Nutr. 2008, 138, 1179–1181. [Google Scholar] [CrossRef] [Green Version]
- Traber, M.G. Regulation of xenobiotic metabolism, the only signaling function of alpha-tocopherol? Mol. Nutr. Food Res. 2010, 54, 661–668. [Google Scholar] [CrossRef]
- Landes, N.; Pfluger, P.; Kluth, D.; Birringer, M.; Rühl, R.; Böl, G.-F.; Glatt, H.; Brigelius-Flohé, R. Vitamin E activates gene expression via the pregnane X receptor. Biochem. Pharm. 2003, 65, 269–273. [Google Scholar] [CrossRef]
- Masuyama, H.; Suwaki, N.; Tateishi, Y.; Nakatsukasa, H.; Segawa, T.; Hiramatsu, Y. The pregnane X receptor regulates gene expression in a ligand- and promoter-selective fashion. Mol. Endocrinol. 2005, 19, 1170–1180. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.M.; McKee, D.D.; Watson, M.A.; Willson, T.M.; Moore, J.T.; Kliewer, S.A. The human orphan nuclear receptor PXR is activated by compounds that regulate CYP3A4 gene expression and cause drug interactions. J. Clin. Investig. 1998, 102, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G. Vitamin E and K interactions--a 50-year-old problem. Nutr. Rev. 2008, 66, 624–629. [Google Scholar] [CrossRef] [Green Version]
- Ichikawa, T.; Horie-Inoue, K.; Ikeda, K.; Blumberg, B.; Inoue, S. Steroid and xenobiotic receptor SXR mediates vitamin K2-activated transcription of extracellular matrix-related genes and collagen accumulation in osteoblastic cells. J. Biol. Chem. 2006, 281, 16927–16934. [Google Scholar] [CrossRef] [Green Version]
- Ikegami, H.; Kawawa, R.; Ichi, I.; Ishikawa, T.; Koike, T.; Aoki, Y.; Fujiwara, Y. Excessive vitamin E intake does not cause bone loss in male or ovariectomized female mice fed normal or high-fat diets. J. Nutr. 2017, 147, 1932–1937. [Google Scholar] [CrossRef] [Green Version]
- Booth, S.L.; Golly, I.; Sacheck, J.M.; Roubenoff, R.; Dallal, G.E.; Hamada, K.; Blumberg, J.B. Effect of vitamin E supplementation on vitamin K status in adults with normal coagulation status. Am. J. Clin. Nutr. 2004, 80, 143–148. [Google Scholar] [CrossRef]
- Pastori, D.; Carnevale, R.; Cangemi, R.; Saliola, M.; Nocella, C.; Bartimoccia, S.; Vicario, T.; Farcomeni, A.; Violi, F.; Pignatelli, P. Vitamin E serum levels and bleeding risk in patients receiving oral anticoagulant therapy: A retrospective cohort study. J. Am. Heart Assoc. 2013, 2. [Google Scholar] [CrossRef] [Green Version]
- Campbell, D.; Bunker, V.W.; Thomas, A.J.; Clayton, B.E. Selenium and vitamin E status of healthy and institutionalized elderly subjects: Analysis of plasma, erythrocytes and platelets. Br. J. Nutr. 1989, 62, 221–227. [Google Scholar] [CrossRef] [Green Version]
- Meydani, S.N.; Han, S.N.; Hamer, D.H. Vitamin E and respiratory infection in the elderly. Ann. N. Y. Acad. Sci. 2004, 1031, 214–222. [Google Scholar] [CrossRef]
- Polidori, M.C.; Mecocci, P.; Stahl, W.; Parente, B.; Cecchetti, R.; Cherubini, A.; Cao, P.; Sies, H.; Senin, U. Plasma levels of lipophilic antioxidants in very old patients with type 2 diabetes. Diabetes Metab. Res. Rev. 2000, 16, 15–19. [Google Scholar] [CrossRef]
- Athinarayanan, S.; Wei, R.; Zhang, M.; Bai, S.; Traber, M.G.; Yates, K.; Cummings, O.W.; Molleston, J.; Liu, W.; Chalasani, N. Genetic polymorphism of cytochrome P450 4F2, vitamin E level and histological response in adults and children with nonalcoholic fatty liver disease who participated in PIVENS and TONIC clinical trials. PLoS ONE 2014, 9, e95366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meydani, S.N.; Lewis, E.D.; Wu, D. Perspective: Should vitamin E recommendations for older adults be increased? Adv. Nutr. 2018, 9, 533–543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meydani, M. Effect of functional food ingredients: Vitamin E modulation of cardiovascular diseases and immune status in the elderly. Am. J. Clin. Nutr. 2000, 71, 1665–1668, discussion 1674S–1675S. [Google Scholar] [CrossRef] [PubMed]
- Hales, C.; Carroll, M.; Fryar, C.; Ogden, C. Prevalence of Obesity and Severe Obesity among Adults: United States, 2017–2018; NCHS Data Brief, No 360; National Center for Health Statistics: Hyattsville, MD, USA, 2020.
- Codoñer-Franch, P.; Pons-Morales, S.; Boix-García, L.; Valls-Bellés, V. Oxidant/antioxidant status in obese children compared to pediatric patients with type 1 diabetes mellitus. Pediatr Diabetes 2010, 11, 251–257. [Google Scholar] [CrossRef]
- Mohn, A.; Catino, M.; Capanna, R.; Giannini, C.; Marcovecchio, M.; Chiarelli, F. Increased oxidative stress in prepubertal severely obese children: Effect of a dietary restriction-weight loss program. J. Clin. Endocrinol. Metab. 2005, 90, 2653–2658. [Google Scholar] [CrossRef] [Green Version]
- Morinobu, T.; Murata, T.; Takaya, R.; Tamai, H. Nutritional status of beta-carotene, alpha-tocopherol and retinol in obese children. Int. J. Vitam. Nutr. Res. 2002, 72, 119–123. [Google Scholar] [CrossRef]
- Switzer, B.R.; Atwood, J.R.; Stark, A.H.; Hatch, J.W.; Travis, R.; Ullrich, F.; Lyden, E.R.; Wu, X.; Chiu, Y.; Smith, J.L. Plasma carotenoid and vitamins a and e concentrations in older African American women after wheat bran supplementation: Effects of age, body mass and smoking history. J. Am. Coll. Nutr. 2005, 24, 217–226. [Google Scholar] [CrossRef]
- Verrotti, A.; Greco, R.; Latini, G.; De Simone, M.; Chiarelli, F. Obesity and plasma concentrations of alpha-tocopherol and beta-carotene in epileptic girls treated with valproate. Neuroendocrinology 2004, 79, 157–162. [Google Scholar] [CrossRef]
- Guerendiain, M.; Mayneris-Perxachs, J.; Montes, R.; López-Belmonte, G.; Martín-Matillas, M.; Castellote, A.I.; Martín-Bautista, E.; Martí, A.; Martínez, J.A.; Moreno, L.; et al. Relation between plasma antioxidant vitamin levels, adiposity and cardio-metabolic profile in adolescents: Effects of a multidisciplinary obesity programme. Clin. Nutr 2017, 36, 209–217. [Google Scholar] [CrossRef] [Green Version]
- Voegele, A.F.; Jerković, L.; Wellenzohn, B.; Eller, P.; Kronenberg, F.; Liedl, K.R.; Dieplinger, H. Characterization of the vitamin E-binding properties of human plasma afamin. Biochemistry 2002, 41, 14532–14538. [Google Scholar] [CrossRef] [PubMed]
- Salonen, J.T.; Nyyssönen, K.; Tuomainen, T.P.; Mäenpää, P.H.; Korpela, H.; Kaplan, G.A.; Lynch, J.; Helmrich, S.P.; Salonen, R. Increased risk of non-insulin dependent diabetes mellitus at low plasma vitamin E concentrations: A four year follow up study in men. BMJ 1995, 311, 1124–1127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Upritchard, J.E.; Sutherland, W.H.; Mann, J.I. Effect of supplementation with tomato juice, vitamin E, and vitamin C on LDL oxidation and products of inflammatory activity in type 2 diabetes. Diabetes Care 2000, 23, 733–738. [Google Scholar] [CrossRef] [Green Version]
- Devaraj, S.; Jialal, I. Alpha tocopherol supplementation decreases serum C-reactive protein and monocyte interleukin-6 levels in normal volunteers and type 2 diabetic patients. Free Radic. Biol. Med. 2000, 29, 790–792. [Google Scholar] [CrossRef]
- Szanto, K.; Li, J.; Cordero, P.; Oben, J. Ethnic differences and heterogeneity in genetic and metabolic makeup contributing to nonalcoholic fatty liver disease. DMSO 2019, 12, 357–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamada, S.; Kamada, N.; Amiya, T.; Nakamoto, N.; Nakaoka, T.; Kimura, M.; Saito, Y.; Ejima, C.; Kanai, T.; Saito, H. Gut microbiota-mediated generation of saturated fatty acids elicits inflammation in the liver in murine high-fat diet-induced steatohepatitis. BMC Gastroenterol. 2017, 17, 136. [Google Scholar] [CrossRef]
- Henao-Mejia, J.; Elinav, E.; Jin, C.; Hao, L.; Mehal, W.Z.; Strowig, T.; Thaiss, C.A.; Kau, A.L.; Eisenbarth, S.C.; Jurczak, M.J.; et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 2012, 482, 179–185. [Google Scholar] [CrossRef] [Green Version]
- Jie, Z.; Xia, H.; Zhong, S.-L.; Feng, Q.; Li, S.; Liang, S.; Zhong, H.; Liu, Z.; Gao, Y.; Zhao, H.; et al. The gut microbiome in atherosclerotic cardiovascular disease. Nat. Commun. 2017, 8, 845. [Google Scholar] [CrossRef] [Green Version]
- Barrington, W.T.; Lusis, A.J. Atherosclerosis: Association between the gut microbiome and atherosclerosis. Nat. Rev. Cardiol. 2017, 14, 699–700. [Google Scholar] [CrossRef]
- Wallert, M.; Börmel, L.; Lorkowski, S. Inflammatory diseases andvitamin E - what do we know and where do we go? Mol. Nutr Food Res. 2020, e2000097. [Google Scholar] [CrossRef]
- Adams, L.A.; Anstee, Q.M.; Tilg, H.; Targher, G. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut 2017, 66, 1138–1153. [Google Scholar] [CrossRef] [Green Version]
- Expert panel on detection, evaluation, and treatment of high blood cholesterol in adults executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA J. Am. Med. Assoc. 2001, 285, 2486–2497. [CrossRef]
- Godala, M.; Materek-Kuśmierkiewicz, I.; Moczulski, D.; Rutkowski, M.; Szatko, F.; Gaszyńska, E.; Tokarski, S.; Kowalski, J. The risk of plasma vitamin A, C, E and D deficiency in patients with metabolic syndrome: A case-control study. Adv. Clin. Exp. Med. 2017, 26, 581–586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitagawa, A.; Ohta, Y.; Ohashi, K.; Yashiro, K.; Fukuzawa, K. Effect of high fructose-induced metabolic syndrome on tissue vitamin E and lipid peroxide levels in rats. J. Nutr. Sci. Vitam. 2020, 66, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Mah, E.; Leonard, S.W.; Bobe, G.; Bruno, R.S. Metabolic syndrome increases dietary α-tocopherol requirements as assessed using urinary and plasma vitamin E catabolites: A double-blind, crossover clinical trial. Am. J. Clin. Nutr. 2017, 105, 571–579. [Google Scholar] [CrossRef] [Green Version]
- Wong, S.K.; Chin, K.-Y.; Suhaimi, F.H.; Ahmad, F.; Ima-Nirwana, S. The Effects of Vitamin E from Elaeis guineensis (Oil Palm) in a rat model of bone loss due to metabolic syndrome. Int. J. Environ. Res. Public Health 2018, 15, 1828. [Google Scholar] [CrossRef] [Green Version]
- Czernichow, S.; Vergnaud, A.-C.; Galan, P.; Arnaud, J.; Favier, A.; Faure, H.; Huxley, R.; Hercberg, S.; Ahluwalia, N. Effects of long-term antioxidant supplementation and association of serum antioxidant concentrations with risk of metabolic syndrome in adults. Am. J. Clin. Nutr. 2009, 90, 329–335. [Google Scholar] [CrossRef] [Green Version]
- Gey, K.F.; Puska, P.; Jordan, P.; Moser, U.K. Inverse correlation between plasma vitamin E and mortality from ischemic heart disease in cross-cultural epidemiology. Am. J. Clin. Nutr. 1991, 53, 326S–334S. [Google Scholar] [CrossRef]
- Riemersma, R.A.; Wood, D.A.; Macintyre, C.C.; Elton, R.A.; Gey, K.F.; Oliver, M.F. Risk of angina pectoris and plasma concentrations of vitamins A, C, and E and carotene. Lancet 1991, 337, 1–5. [Google Scholar] [CrossRef]
- Huang, J.; Weinstein, S.J.; Yu, K.; Männistö, S.; Albanes, D. Relationship between serum alpha-tocopherol and overall and cause-specific mortality. Circ. Res. 2019, 125, 29–40. [Google Scholar] [CrossRef]
- Rimm, E.B.; Stampfer, M.J.; Ascherio, A.; Giovannucci, E.; Colditz, G.A.; Willett, W.C. Vitamin E Consumption and the Risk of Coronary Heart Disease in Men. Available online: https://www.nejm.org/doi/10.1056/NEJM199305203282004?url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org&rfr_dat=cr_pub++0www.ncbi.nlm.nih.gov (accessed on 30 June 2020).
- Bolton-Smith, C.; Woodward, M.; Tunstall-Pedoe, H. The Scottish Heart Health Study. Dietary intake by food frequency questionnaire and odds ratios for coronary heart disease risk. II. The antioxidant vitamins and fibre. Eur. J. Clin. Nutr 1992, 46, 85–93. [Google Scholar] [PubMed]
- Knekt, P.; Reunanen, A.; Jävinen, R.; Seppänen, R.; Heliövaara, M.; Aromaa, A. Antioxidant vitamin intake and coronary mortality in a longitudinal population study. Am. J. Epidemiol. 1994, 139, 1180–1189. [Google Scholar] [CrossRef] [PubMed]
- Kushi, L.H.; Folsom, A.R.; Prineas, R.J.; Mink, P.J.; Wu, Y.; Bostick, R.M. Dietary Antioxidant Vitamins and Death from Coronary Heart Disease in Postmenopausal Women. Available online: https://www.nejm.org/doi/10.1056/NEJM199605023341803?url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org&rfr_dat=cr_pub++0www.ncbi.nlm.nih.gov (accessed on 30 June 2020).
- Meyer, F.; Bairati, I.; Dagenais, G.R. Lower ischemic heart disease incidence and mortality among vitamin supplement users. Can. J. Cardiol. 1996, 12, 930–934. [Google Scholar] [PubMed]
- Losonczy, K.G.; Harris, T.B.; Havlik, R.J. Vitamin E and vitamin C supplement use and risk of all-cause and coronary heart disease mortality in older persons: The Established Populations for Epidemiologic Studies of the Elderly. Am. J. Clin. Nutr. 1996, 64, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Stampfer, M.J.; Hennekens, C.H.; Manson, J.E.; Colditz, G.A.; Rosner, B.; Willett, W.C. Vitamin E Consumption and the Risk Of Coronary Disease in Women. Available online: https://www.nejm.org/doi/10.1056/NEJM199305203282003?url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org&rfr_dat=cr_pub++0www.ncbi.nlm.nih.gov (accessed on 1 July 2020).
- Lee, C.-H.; Chan, R.S.M.; Wan, H.Y.L.; Woo, Y.-C.; Cheung, C.Y.Y.; Fong, C.H.Y.; Cheung, B.M.Y.; Lam, T.-H.; Janus, E.; Woo, J.; et al. Dietary intake of anti-oxidant vitamins A, C, and E is inversely associated with adverse cardiovascular outcomes in chinese—A 22-years population-based prospective study. Nutrients 2018, 10, 1664. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Xu, L. Circulating vitamin E levels and risk of coronary artery disease and myocardial infarction: A mendelian randomization study. Nutrients 2019, 11, 2153. [Google Scholar] [CrossRef] [Green Version]
- Hak, A.E.; Stampfer, M.J.; Campos, H.; Sesso, H.D.; Gaziano, J.M.; Willett, W.; Ma, J. Plasma Carotenoids and tocopherols and risk of myocardial infarction in a low-risk population of US male physicians. Circulation 2003, 108, 802–807. [Google Scholar] [CrossRef]
- Hense, H.W.; Stender, M.; Borsc, W.; Keil, U. Lack of an association between serum vitamin E and myocardial infarction in a population with high vitamin E levels. Atherosclerosis 1993, 103, 21–28. [Google Scholar] [CrossRef]
- Bashar, T.; Akhter, N. Oxidative stress and antioxidant status in patients of acute myocardial infarction before and after regular treatment. Univ. Heart J. 2014, 10, 60–65. [Google Scholar] [CrossRef] [Green Version]
- Scragg, R.; Jackson, R.; Holdaway, I.; Woollard, G.; Woollard, D. Changes in plasma vitamin levels in the first 48 h after onset of acute myocardial infarction. Am. J. Cardiol. 1989, 64, 971–974. [Google Scholar] [CrossRef]
- Labadarios, D.; Brink, P.A.; Weich, H.F.; Visser, L.; Louw, M.E.; Shephard, G.S.; van Stuijvenberg, M.E. Plasma vitamin A, E, C and B6 levels in myocardial infarction. S. Afr. Med. J. 1987, 71, 561–563. [Google Scholar] [PubMed]
- Sood, R.; Narang, A.P.S.; Abraham, R.; Arora, U.; Calton, R.; Sood, N. Changes in vitamin C and vitamin E during oxidative stress in myocardial reperfusion. Indian J. Physiol. Pharm. 2007, 51, 165–169. [Google Scholar]
- Ricciarelli, R.; Zingg, J.M.; Azzi, A. Vitamin E: Protective role of a Janus molecule. FASEB J. 2001, 15, 2314–2325. [Google Scholar] [CrossRef]
- Nègre-Salvayre, A.; Mabile, L.; Delchambre, J.; Salvayre, R. alpha-Tocopherol, ascorbic acid, and rutin inhibit synergistically the copper-promoted LDL oxidation and the cytotoxicity of oxidized LDL to cultured endothelial cells. Biol Trace Elem. Res. 1995, 47, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Villacorta, L.; Azzi, A.; Zingg, J.-M. Regulatory role of vitamins E and C on extracellular matrix components of the vascular system. Mol. Asp. Med. 2007, 28, 507–537. [Google Scholar] [CrossRef] [PubMed]
- Ricciarelli, R.; Maroni, P.; Özer, N.; Zingg, J.-M.; Azzi, A. Age-dependent increase of collagenase expression can be reduced by α-tocopherol via protein kinase C inhibition. Free Radic. Biol. Med. 1999, 27, 729–737. [Google Scholar] [CrossRef]
- Boscoboinik, D.; Szewczyk, A.; Hensey, C.; Azzi, A. Inhibition of cell proliferation by alpha-tocopherol. Role of protein kinase C. J. Biol. Chem. 1991, 266, 6188–6194. [Google Scholar]
- Mahoney, C.W.; Azzi, A. Vitamin E inhibits protein kinase C activity. Biochem. Biophys. Res. Commun. 1988, 154, 694–697. [Google Scholar] [CrossRef]
- Tasinato, A.; Boscoboinik, D.; Bartoli, G.M.; Maroni, P.; Azzi, A. d-alpha-tocopherol inhibition of vascular smooth muscle cell proliferation occurs at physiological concentrations, correlates with protein kinase C inhibition, and is independent of its antioxidant properties. Proc. Natl. Acad. Sci. USA 1995, 92, 12190–12194. [Google Scholar] [CrossRef] [Green Version]
- Devaraj, S.; Jialal, I. The effects of alpha-tocopherol on critical cells in atherogenesis. Curr. Opin. Lipidol. 1998, 9, 11–15. [Google Scholar] [CrossRef]
- Cominacini, L.; Garbin, U.; Pasini, A.F.; Davoli, A.; Campagnola, M.; Contessi, G.B.; Pastorino, A.M.; Lo Cascio, V. Antioxidants inhibit the expression of intercellular cell adhesion molecule-1 and vascular cell adhesion molecule-1 induced by oxidized LDL on human umbilical vein endothelial cells. Free Radic. Biol. Med. 1997, 22, 117–127. [Google Scholar] [CrossRef]
- van Dam, B.; van Hinsbergh, V.W.M.; Stehouwer, C.D.A.; Versteilen, A.; Dekker, H.; Buytenhek, R.; Princen, H.M.; Schalkwijk, C.G. Vitamin E inhibits lipid peroxidation-induced adhesion molecule expression in endothelial cells and decreases soluble cell adhesion molecules in healthy subjects. Cardiovasc. Res. 2003, 57, 563–571. [Google Scholar] [CrossRef]
- Koga, T.; Kwan, P.; Zubik, L.; Ameho, C.; Smith, D.; Meydani, M. Vitamin E supplementation suppresses macrophage accumulation and endothelial cell expression of adhesion molecules in the aorta of hypercholesterolemic rabbits. Atherosclerosis 2004, 176, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Koga, T.; Claycombe, K.; Meydani, M. Homocysteine increases monocyte and T-cell adhesion to human aortic endothelial cells. Atherosclerosis 2002, 161, 365–374. [Google Scholar] [CrossRef]
- Orbe, J.; Rodríguez, J.A.; Arias, R.; Belzunce, M.; Nespereira, B.; Pérez-Ilzarbe, M.; Roncal, C.; Páramo, J.A. Antioxidant vitamins increase the collagen content and reduce MMP-1 in a porcine model of atherosclerosis: Implications for plaque stabilization. Atherosclerosis 2003, 167, 45–53. [Google Scholar] [CrossRef] [Green Version]
- Schwenke, D.C.; Rudel, L.L.; Sorci-Thomas, M.G.; Thomas, M.J. α-Tocopherol protects against diet induced atherosclerosis in New Zealand white rabbits. J. Lipid Res. 2002, 43, 1927–1938. [Google Scholar] [CrossRef] [Green Version]
- Hodis, H.N.; Mack, W.J.; LaBree, L.; Mahrer, P.R.; Sevanian, A.; Liu, C.-R.; Liu, C.-H.; Hwang, J.; Selzer, R.H.; Azen, S.P. Alpha-tocopherol supplementation in healthy individuals reduces low-density lipoprotein oxidation but not atherosclerosis. Circulation 2002, 106, 1453–1459. [Google Scholar] [CrossRef] [Green Version]
- McQuillan, B.M.; Hung, J.; Beilby, J.P.; Nidorf, M.; Thompson, P.L. Antioxidant vitamins and the risk of carotid atherosclerosis: The perth carotid ultrasound disease assessment study (CUDAS). J. Am. Coll. Cardiol. 2001, 38, 1788–1794. [Google Scholar] [CrossRef] [Green Version]
- Magliano, D.; McNeil, J.; Branley, P.; Shiel, L.; Demos, L.; Wolfe, R.; Kotsopoulos, D.; McGrath, B. The Melbourne Atherosclerosis Vitamin E Trial (MAVET): A study of high dose vitamin E in smokers. Eur. J. Cardiovasc. Prev. Rehabil. 2006, 13, 341–347. [Google Scholar] [CrossRef]
- Devaraj, S.; Tang, R.; Adams-Huet, B.; Harris, A.; Seenivasan, T.; de Lemos, J.A.; Jialal, I. Effect of high-dose α-tocopherol supplementation on biomarkers of oxidative stress and inflammation and carotid atherosclerosis in patients with coronary artery disease. Am. J. Clin. Nutr. 2007, 86, 1392–1398. [Google Scholar] [CrossRef] [Green Version]
- Lonn, E.M.; Yusuf, S.; Dzavik, V.; Doris, C.I.; Yi, Q.; Smith, S.; Moore-Cox, A.; Bosch, J.; Riley, W.A.; Teo, K.K. Effects of ramipril and vitamin E on atherosclerosis. Circulation 2001, 103, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Rapola, J.M.; Virtamo, J.; Haukka, J.K.; Heinonen, O.P.; Albanes, D.; Taylor, P.R.; Huttunen, J.K. Effect of vitamin E and beta carotene on the incidence of angina pectoris: A randomized, double-blind, controlled trial. JAMA 1996, 275, 693–698. [Google Scholar] [CrossRef] [PubMed]
- de Gaetano, G.; Collaborative Group of the Primary Prevention Project. Low-dose aspirin and vitamin E in people at cardiovascular risk: A randomised trial in general practice. Collaborative Group of the Primary Prevention Project. Lancet 2001, 357, 89–95. [Google Scholar] [PubMed]
- Stephens, N.G.; Parsons, A.; Schofield, P.M.; Kelly, F.; Cheeseman, K.; Mitchinson, M.J. Randomised controlled trial of vitamin E in patients with coronary disease: Cambridge Heart Antioxidant Study (CHAOS). Lancet 1996, 347, 781–786. [Google Scholar] [CrossRef]
- Boaz, M.; Smetana, S.; Weinstein, T.; Matas, Z.; Gafter, U.; Iaina, A.; Knecht, A.; Weissgarten, Y.; Brunner, D.; Fainaru, M.; et al. Secondary prevention with antioxidants of cardiovascular disease in endstage renal disease (SPACE): Randomised placebo-controlled trial. Lancet 2000, 356, 1213–1218. [Google Scholar] [CrossRef]
- Marchioli, R. Treatment with n-3 polyunsaturated fatty acids after myocardial infarction: Results of GISSI-prevenzione trial. Eur. Heart J. Suppl. 2001, 3, D85–D97. [Google Scholar] [CrossRef] [Green Version]
- Alkhenizan, A.H.; Al-Omran, M.A. The role of vitamin E in the prevention of coronary events and stroke. Meta-analysis of randomized controlled trials. Neurosciences (Riyadh) 2005, 10, 23–29. [Google Scholar]
- Wallert, M.; Ziegler, M.; Wang, X.; Maluenda, A.; Xu, X.; Yap, M.L.; Witt, R.; Giles, C.; Kluge, S.; Hortmann, M.; et al. α-Tocopherol preserves cardiac function by reducing oxidative stress and inflammation in ischemia/reperfusion injury. Redox Biol. 2019, 26, 101292. [Google Scholar] [CrossRef]
- Saleh, N.K.; Saleh, H.A. Protective effects of vitamin E against myocardial ischemia/reperfusion injury in rats. Saudi Med. J. 2010, 31, 142–147. [Google Scholar]
- Tripathi, Y.; Hegde, B.M. Effect of alpha-tocopherol pretreatment on infarct size following 90 min of ischemia and 4 h of reperfusion in dogs. Indian J. Physiol. Pharm. 1997, 41, 241–247. [Google Scholar]
- Sethi, R.; Takeda, N.; Nagano, M.; Dhalla, N.S. Beneficial effects of vitamin E treatment in acute myocardial infarction. J. Cardiovasc. Pharmacol. Ther. 2016. [Google Scholar] [CrossRef] [PubMed]
- Jaxa-Chamiec, T.; Bednarz, B.; Drozdowska, D.; Gessek, J.; Gniot, J.; Janik, K.; Kawka-Urbanek, T.; Maciejewski, P.; Ogórek, M.; Szpajer, M.; et al. Antioxidant effects of combined vitamins C and E in acute myocardial infarction. The randomized, double-blind, placebo controlled, multicenter pilot Myocardial Infarction and VITamins (MIVIT) trial. Kardiol. Pol. 2005, 62, 344–350. [Google Scholar] [PubMed]
- Singh, R.B.; Niaz, M.A.; Rastogi, S.S.; Rastogi, S. Usefulness of antioxidant vitamins in suspected acute myocardial infarction (the Indian experiment of infarct survival-3). Am. J. Cardiol. 1996, 77, 232–236. [Google Scholar] [CrossRef]
- Wallert, M.; Schmölz, L.; Galli, F.; Birringer, M.; Lorkowski, S. Regulatory metabolites of vitamin E and their putative relevance for atherogenesis. Redox Biol. 2014, 2, 495–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Döring, F.; Rimbach, G.; Lodge, J. In Silico Search for Single Nucleotide Polymorphisms in Genes Important in Vitamin E Homeostasis. IUBMB Life (Int. Union Biochem. Mol. Biol. Life) 2004, 56, 615–620. [Google Scholar] [CrossRef]
- Christen, S.; Woodall, A.A.; Shigenaga, M.K.; Southwell-Keely, P.T.; Duncan, M.W.; Ames, B.N. gamma-tocopherol traps mutagenic electrophiles such as NO(X) and complements alpha-tocopherol: Physiological implications. Proc. Natl. Acad. Sci. USA 1997, 94, 3217–3222. [Google Scholar] [CrossRef] [Green Version]
- Devaraj, S.; Leonard, S.; Traber, M.G.; Jialal, I. Gamma-tocopherol supplementation alone and in combination with alpha-tocopherol alters biomarkers of oxidative stress and inflammation in subjects with metabolic syndrome. Free Radic. Biol. Med. 2008, 44, 1203–1208. [Google Scholar] [CrossRef] [Green Version]
- Himmelfarb, J.; Kane, J.; McMonagle, E.; Zaltas, E.; Bobzin, S.; Boddupalli, S.; Phinney, S.; Miller, G. Alpha and gamma tocopherol metabolism in healthy subjects and patients with end-stage renal disease. Kidney Int. 2003, 64, 978–991. [Google Scholar] [CrossRef] [Green Version]
- Smith, K.S.; Lee, C.-L.; Ridlington, J.W.; Leonard, S.W.; Devaraj, S.; Traber, M.G. Vitamin E supplementation increases circulating vitamin E metabolites tenfold in end-stage renal disease patients. Lipids 2003, 38, 813–819. [Google Scholar] [CrossRef]
- Wu, J.H.Y.; Ward, N.C.; Indrawan, A.P.; Almeida, C.-A.; Hodgson, J.M.; Proudfoot, J.M.; Puddey, I.B.; Croft, K.D. Effects of alpha-tocopherol and mixed tocopherol supplementation on markers of oxidative stress and inflammation in type 2 diabetes. Clin. Chem. 2007, 53, 511–519. [Google Scholar] [CrossRef] [Green Version]
- Ward, N.C.; Wu, J.H.; Clarke, M.W.; Puddey, I.B.; Burke, V.; Croft, K.D.; Hodgson, J.M. The effect of vitamin E on blood pressure in individuals with type 2 diabetes: A randomized, double-blind, placebo-controlled trial. J. Hypertens. 2007, 25, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Mathur, P.; Ding, Z.; Saldeen, T.; Mehta, J.L. Tocopherols in the prevention and treatment of atherosclerosis and related cardiovascular disease. Clin. Cardiol 2015, 38, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q. Natural forms of vitamin E: Metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free Radic. Biol. Med. 2014, 72, 76–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qureshi, A.A.; Qureshi, N.; Wright, J.J.K.; Shen, Z.; Kramer, G.; Gapor, A.; Chong, Y.H.; DeWitt, G.; Ong, A.S.H.; Peterson, D.M.; et al. Lowering of serum cholesterol in hypercholesterolemic humans by tocotrienols (palmvitee). Am. J. Clin. Nutr. 1991, 53, 1021S–1026S. [Google Scholar] [CrossRef]
- Qureshi, A.A.; Bradlow, B.A.; Brace, L.; Manganello, J.; Peterson, D.M.; Pearce, B.C.; Wright, J.J.; Gapor, A.; Elson, C.E. Response of hypercholesterolemic subjects to administration of tocotrienols. Lipids 1995, 30, 1171–1177. [Google Scholar] [CrossRef]
- Yuen, K.H.; Wong, J.W.; Lim, A.B.; Ng, B.H.; Choy, W.P. Effect of Mixed-Tocotrienols in Hypercholesterolemic Subjects. FFHD 2011, 1, 106. [Google Scholar] [CrossRef]
- Rasool, A.H.G.; Rahman, A.R.A.; Yuen, K.H.; Wong, A.R. Arterial compliance and vitamin E blood levels with a self emulsifying preparation of tocotrienol rich vitamin E. Arch. Pharm. Res. 2008, 31, 1212–1217. [Google Scholar] [CrossRef]
- Wallert, M.; Kluge, S.; Schubert, M.; Koeberle, A.; Werz, O.; Birringer, M.; Lorkowski, S. Diversity of chromanol and chromenol structures and functions: An emerging class of anti-inflammatory and anti-carcinogenic agents. Front. Pharm. 2020, 11, 362. [Google Scholar] [CrossRef]
- Birringer, M.; Siems, K.; Maxones, A.; Frank, J.; Lorkowski, S. Natural 6-hydroxy-chromanols and -chromenols: Structural diversity, biosynthetic pathways and health implications. RSC Adv. 2018, 8, 4803–4841. [Google Scholar] [CrossRef] [Green Version]
- Wallert, M.; Schmölz, L.; Koeberle, A.; Krauth, V.; Glei, M.; Galli, F.; Werz, O.; Birringer, M.; Lorkowski, S. α-Tocopherol long-chain metabolite α-13′-COOH affects the inflammatory response of lipopolysaccharide-activated murine RAW264.7 macrophages. Mol. Nutr. Food Res. 2015, 59, 1524–1534. [Google Scholar] [CrossRef]
- Ciffolilli, S.; Wallert, M.; Bartolini, D.; Krauth, V.; Werz, O.; Piroddi, M.; Sebastiani, B.; Torquato, P.; Lorkowski, S.; Birringer, M.; et al. Human serum determination and in vitro anti-inflammatory activity of the vitamin E metabolite α-(13′-hydroxy)-6-hydroxychroman. Free Radic. Biol. Med. 2015, 89, 952–962. [Google Scholar] [CrossRef] [PubMed]
- Wallert, M.; Bauer, J.; Kluge, S.; Schmölz, L.; Chen, Y.-C.; Ziegler, M.; Searle, A.K.; Maxones, A.; Schubert, M.; Thürmer, M.; et al. The vitamin E derivative garcinoic acid from Garcinia kola nut seeds attenuates the inflammatory response. Redox Biol. 2019, 24, 101166. [Google Scholar] [CrossRef] [PubMed]
- Pein, H.; Ville, A.; Pace, S.; Temml, V.; Garscha, U.; Raasch, M.; Alsabil, K.; Viault, G.; Dinh, C.-P.; Guilet, D.; et al. Endogenous metabolites of vitamin E limit inflammation by targeting 5-lipoxygenase. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallert, M.; Mosig, S.; Rennert, K.; Funke, H.; Ristow, M.; Pellegrino, R.M.; Cruciani, G.; Galli, F.; Lorkowski, S.; Birringer, M. Long-chain metabolites of α-tocopherol occur in human serum and inhibit macrophage foam cell formation in vitro. Free Radic. Biol. Med. 2014, 68, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Schmölz, L.; Schubert, M.; Kirschner, J.; Kluge, S.; Galli, F.; Birringer, M.; Wallert, M.; Lorkowski, S. Long-chain metabolites of vitamin E: Interference with lipotoxicity via lipid droplet associated protein PLIN2. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2018, 1863, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Birringer, M.; Lorkowski, S. Vitamin E: Regulatory role of metabolites: Vitamin E Metabolites. IUBMB Life 2019, 71, 479–486. [Google Scholar] [CrossRef]
- Mangan, M.S.J.; Olhava, E.J.; Roush, W.R.; Seidel, H.M.; Glick, G.D.; Latz, E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat. Rev. Drug Discov. 2018. [Google Scholar] [CrossRef]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef]
- Ridker, P.M. High-sensitivity c-reactive protein: Potential adjunct for global risk assessment in the primary prevention of cardiovascular disease. Circulation 2001, 103, 1813–1818. [Google Scholar] [CrossRef] [Green Version]
- Ridker, P.M.; MacFadyen, J.G.; Thuren, T.; Libby, P. Residual inflammatory risk associated with interleukin-18 and interleukin-6 after successful interleukin-1β inhibition with canakinumab: Further rationale for the development of targeted anti-cytokine therapies for the treatment of atherothrombosis. Eur. Heart J. 2020, 41, 2153–2163. [Google Scholar] [CrossRef] [Green Version]
- Bedoui, Y.; Guillot, X.; Sélambarom, J.; Guiraud, P.; Giry, C.; Jaffar-Bandjee, M.C.; Ralandison, S.; Gasque, P. Methotrexate an old drug with new tricks. Int. J. Mol. Sci 2019, 20, 5023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westlake, S.L.; Colebatch, A.N.; Baird, J.; Kiely, P.; Quinn, M.; Choy, E.; Ostor, A.J.K.; Edwards, C.J. The effect of methotrexate on cardiovascular disease in patients with rheumatoid arthritis: A systematic literature review. Rheumatology 2010, 49, 295–307. [Google Scholar] [CrossRef] [Green Version]
- Micha, R.; Imamura, F.; Wyler von Ballmoos, M.; Solomon, D.H.; Hernán, M.A.; Ridker, P.M.; Mozaffarian, D. Systematic review and meta-analysis of methotrexate use and risk of cardiovascular disease. Am. J. Cardiol. 2011, 108, 1362–1370. [Google Scholar] [CrossRef] [Green Version]
- Ridker, P.M.; Everett, B.M.; Pradhan, A.; MacFadyen, J.G.; Solomon, D.H.; Zaharris, E.; Mam, V.; Hasan, A.; Rosenberg, Y.; Iturriaga, E.; et al. Low-dose methotrexate for the prevention of atherosclerotic events. N. Engl. J. Med. 2019, 380, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Pope, R.M.; Tschopp, J. The role of interleukin-1 and the inflammasome in gout: Implications for therapy. Arthritis Rheum. 2007, 56, 3183–3188. [Google Scholar] [CrossRef] [PubMed]
- Nidorf, S.M.; Thompson, P.L. Why colchicine should be considered for secondary prevention of atherosclerosis: An overview. Clin. Ther. 2019, 41, 41–48. [Google Scholar] [CrossRef] [Green Version]
- Nidorf, S.M.; Eikelboom, J.W.; Budgeon, C.A.; Thompson, P.L. Low-dose colchicine for secondary prevention of cardiovascular disease. J. Am. Coll. Cardiol. 2013, 61, 404–410. [Google Scholar] [CrossRef] [Green Version]
- Tardif, J.-C.; Kouz, S.; Waters, D.D.; Bertrand, O.F.; Diaz, R.; Maggioni, A.P.; Pinto, F.J.; Ibrahim, R.; Gamra, H.; Kiwan, G.S.; et al. Efficacy and safety of low-dose colchicine after myocardial infarction. N. Engl. J. Med. 2019, 381, 2497–2505. [Google Scholar] [CrossRef]
- Melamed-Frank, M.; Lache, O.; Enav, B.I.; Szafranek, T.; Levy, N.S.; Ricklis, R.M.; Levy, A.P. Structure-function analysis of the antioxidant properties of haptoglobin. Blood 2001, 98, 3693–3698. [Google Scholar] [CrossRef] [Green Version]
- Beeri, M.S.; Lin, H.-M.; Sano, M.; Ravona-Springer, R.; Liu, X.; Bendlin, B.B.; Gleason, C.E.; Guerrero-Berroa, E.; Soleimani, L.; Launer, L.J.; et al. Association of the haptoglobin gene polymorphism with cognitive function and decline in elderly African American adults with type 2 diabetes: Findings from the Action to Control Cardiovascular Risk in Diabetes–Memory in Diabetes (ACCORD-MIND) Study. JAMA Netw. Open 2018, 1, e184458. [Google Scholar] [CrossRef] [Green Version]
- Dalan, R.; Liew, H.; Goh, L.L.; Gao, X.; Chew, D.E.; Boehm, B.O.; Leow, M.K.S. The haptoglobin 2-2 genotype is associated with inflammation and carotid artery intima-media thickness. Diab. Vasc. Dis. Res. 2016, 13, 373–376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levy, A.P.; Hochberg, I.; Jablonski, K.; Resnick, H.E.; Lee, E.T.; Best, L.; Howard, B.V. Haptoglobin phenotype is an independent risk factor for cardiovascular disease in individuals with diabetes. J. Am. Coll. Cardiol. 2002, 40, 1984–1990. [Google Scholar] [CrossRef] [Green Version]
- Adams, J.N.; Cox, A.J.; Freedman, B.I.; Langefeld, C.D.; Carr, J.J.; Bowden, D.W. Genetic analysis of haptoglobin polymorphisms with cardiovascular disease and type 2 diabetes in the Diabetes Heart Study. Cardiovasc. Diabetol. 2013, 12, 31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Bacquer, D.; De Backer, G.; Langlois, M.; Delanghe, J.; Kesteloot, H.; Kornitzer, M. Haptoglobin polymorphism as a risk factor for coronary heart disease mortality. Atherosclerosis 2001, 157, 161–166. [Google Scholar] [CrossRef]
- Orchard, T.J.; Backlund, J.-Y.C.; Costacou, T.; Cleary, P.; Lopes-Virella, M.; Levy, A.P.; Lachin, J.M. DCCT/EDIC Research Group Haptoglobin 2-2 genotype and the risk of coronary artery disease in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications study (DCCT/EDIC). J. Diabetes Complicat. 2016, 30, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Blum, S.; Vardi, M.; Brown, J.B.; Russell, A.; Milman, U.; Shapira, C.; Levy, N.S.; Miller-Lotan, R.; Asleh, R.; Levy, A.P. Vitamin E reduces cardiovascular disease in individuals with diabetes mellitus and the haptoglobin 2-2 genotype. Pharmacogenomics 2010, 11, 675–684. [Google Scholar] [CrossRef] [Green Version]
- Vardi, M.; Blum, S.; Levy, A.P. Haptoglobin genotype and cardiovascular outcomes in diabetes mellitus—Natural history of the disease and the effect of vitamin E treatment. Meta-analysis of the medical literature. Eur. J. Intern. Med. 2012, 23, 628–632. [Google Scholar] [CrossRef] [Green Version]
- Asleh, R.; Briasoulis, A.; Berinstein, E.; Wiener, J.; Palla, M.; Kushwaha, S.; Levy, A. Meta-analysis of the association of the haptoglobin genotype with cardiovascular outcomes and the pharmacogenomic interactions with vitamin E supplementation. PGPM 2018, 11, 71–82. [Google Scholar] [CrossRef] [Green Version]
- Belitskaya-Lévy, I.; Dysken, M.; Guarino, P.; Sano, M.; Asthana, S.; Vertrees, J.E.; Pallaki, M.; Llorente, M.; Love, S.; Schellenberg, G. Impact of apolipoprotein E genotypes on vitamin E and memantine treatment outcomes in Alzheimer’s disease. Alzheimers Dement. (N.Y.) 2018, 4, 344–349. [Google Scholar] [CrossRef]
- Yuan, L.; Liu, J.; Dong, L.; Cai, C.; Wang, S.; Wang, B.; Xiao, R. Effects of APOE rs429358, rs7412 and GSTM1/GSTT1 polymorphism on plasma and erythrocyte antioxidant parameters and cognition in old chinese adults. Nutrients 2015, 7, 8261–8273. [Google Scholar] [CrossRef] [Green Version]
- Huebbe, P.; Lodge, J.K.; Rimbach, G. Implications of apolipoprotein E genotype on inflammation and vitamin E status. Mol. Nutr. Food Res. 2010, 54, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Jarvik, G.P.; Tsai, N.T.; McKinstry, L.A.; Wani, R.; Brophy, V.H.; Richter, R.J.; Schellenberg, G.D.; Heagerty, P.J.; Hatsukami, T.S.; Furlong, C.E. Vitamin C and Eintake is associated with increased paraoxonase activity. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1329–1333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lou-Bonafonte, J.M.; Gabás-Rivera, C.; Navarro, M.A.; Osada, J. The search for dietary supplements to elevate or activate circulating paraoxonases. Int. J. Mol. Sci. 2017, 18, 416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lassnigg, A.; Punz, A.; Barker, R.; Keznickl, P.; Manhart, N.; Roth, E.; Hiesmayr, M. Influence of intravenous vitamin E supplementation in cardiac surgery on oxidative stress: A double-blinded, randomized, controlled study. Br. J. Anaesth. 2003, 90, 148–154. [Google Scholar] [CrossRef] [Green Version]
- Neelamegam, M.; Looi, I.; Ng, K.S.; Malavade, S.S. Vitamin E supplementation for preventing recurrent stroke and other vascular events in patients with stroke or transient ischaemic attack. Cochrane Database Syst Rev. 2017. [Google Scholar] [CrossRef]
- Hankey, G.J. Vitamin supplementation and stroke prevention. Stroke 2012, 43, 2814–2818. [Google Scholar] [CrossRef]
- Cheng, P.; Wang, L.; Ning, S.; Liu, Z.; Lin, H.; Chen, S.; Zhu, J. Vitamin E intake and risk of stroke: A meta-analysis. Br. J. Nutr. 2018, 120, 1181–1188. [Google Scholar] [CrossRef] [Green Version]
- Bin, Q.; Hu, X.; Cao, Y.; Gao, F. The role of vitamin E (tocopherol) supplementation in the prevention of stroke. A meta-analysis of 13 randomised controlled trials. Thromb. Haemost. 2011, 105, 579–585. [Google Scholar] [CrossRef] [Green Version]
- Schürks, M.; Glynn, R.J.; Rist, P.M.; Tzourio, C.; Kurth, T. Effects of vitamin E on stroke subtypes: Meta-analysis of randomised controlled trials. BMJ 2010, 341. [Google Scholar] [CrossRef] [Green Version]
| Risk Factor | Type of Study | Author | Participants | Endpoints | Vitamin E Dosage |
|---|---|---|---|---|---|
| Hypertension | |||||
| SBP (systolic blood pressure) DPB (diastolic blood pressure) | Observational | Kuwabara et al. [53] | n = 3507 | higher vitamin E intake is associated with a lower percentage of subjects with hypertension | |
| Interventional | Boshtam et al. [54] | n = 70 mild hypertensive patients | Significant decrease in SBP and DBP (mainly in SBP) | 134 mg per day (200 IU) for 27 weeks | |
| Interventional | Tmj et al. [55] | n = 60 mild hypertensive subjects | decrease in blood pressure | 134 mg (200 IU) per day for 12 weeks | |
| Interventional | Palumbo et al. [56] | n = 142 treated hypertensive patients | no clinically relevant effect on blood pressure | 300 mg per day for 12 weeks | |
| Interventional | Mihalj et al. [57] | n = 57 treated hypertensive patients | no further effect of vitamin E/C supplementation | 720 mg vitamin E and 25 mg vitamin C per day for 8 weeks | |
| Interventional | Barbagallo et al. [58] | n = 12 hypertensive patients | no effect of vitamin E treatment on SBP or DBP | 600 mg vitamin E per day for 4 weeks | |
| Hyperlipidemia | |||||
| Hypercholesterolemic (HC) | Observational Interventional | Cangemi et al. [59] | n = 30 HC patients n = 20 healthy subjects n = 30 HC patients | lower vitamin E plasma level in HC patients vs. healthy subjects administration of atorvastatin restored vitamin E/TC plasma level | 10 mg atorvastatin per day for 30 days |
| Observational | Shin et al. [60] | n = 76 HC patients | increased α-TOH/lipid plasma level in HC patients | 20–40 mg simvastatin per day for 8 weeks | |
| Observational | Blom et al. [61] | n = 738 HC patients | increased vitamin E/TC plasma level in evolocumab (anti- PCSK9 antibody)-treated patients from baseline to week 52 | 10 or 80 mg atorvastatin per day or 80 mg atorvastatin plus 10 mg ezetimibe per day for 52 weeks, 420 mg Evolocumab or placebo for 8 weeks | |
| Interventional | Lui et al. [62] | n = 19 HC patients | increased vitamin E/LDL-C plasma level in atorvastatin-treated HC patients | 10 mg atorvastatin per day for 5 months | |
| Interventional | Leonard et al. [63] | n = 44 HC patients | vitamin E supplementation did not alter cholesterol levels under statin therapy | 268 mg (400 IU) vitamin E per day or placebo for 12 weeks | |
| Thrombosis | |||||
| Interventional | Glynn et al. [64] | n = 39,876 women aged 45 and older | women taking vitamin E were 21% less likely to suffer a venous thromboembolism | Vitamin E (540 mg) or a placebo on alternate days over a 10-year period. | |
| Interventional | Vuckovic et al. [65] | 2506 patients with venous thrombosis, 2506 partner controls, and 2684 random-digit-dialing (RDD) controls n = 96 patients supplemented with vitamin E | No association of vitamin E supplementation with a reduced venous thrombosis risk | No information was obtained on the dosage of vitamin E intake | |
| Age | |||||
| Observational | Ortega et al. [66] | n= 120 aged subjects (65–91 years) | Lower vitamin E intake and α-TOH/TC plasma level correlates with cognitive impairment in elderly | - | |
| Observational | Vatassery et al. [67] | 48 healthy male volunteers aged 24–91 years | α-TOH plasma level remained unchanged, decreased α-TOH level in platelets of elderly subjects | - | |
| Observational | Capuron et al. [68] | n = 69 aged subjects (73–86 years) | Lower α-TOH plasma level in subjects with poor physical and mental health status | - | |
| Observational | Requejo et al. [69] | n = 120 aged subjects (65–91 years) | 95.2% are below recommendations of a-TOH intake | - | |
| Observational | Rudman et al. [70] | n = 34 eating-dependent nursing home residents | The vast majority did not receive micronutrient supplements | - | |
| Interventional | De la Fuente et al. [71] | n = 33 aged subjects (65–75 years) n= 30 controls (25–35 years) | α-TOH improves immune functions and therefore health in aged people | 200 mg α-TOH per day for 3 months | |
| Obesity | |||||
| Observational | Silva et al. [72] | n = 33 overweight adolescents n = 42 obese adolescents n = 75 healthy adolescents (10–15 years) | Crude and energy-adjusted intake of vitamin E positively correlate with BMI, but not with plasma level of vitamin E; α-TOH/LDL-C and α-TOH/TC decrease in obese and overweight adolescents | - | |
| Observational | Mehmetoglu et al. [73] | n = 98 obese patients n = 78 healthy subjects (18–65 years) | decreased α-TOH/TC + TG plasma level in obese subjects | - | |
| Observational | Kljno et al. [74] | n = 17 obese girls n = 7 healthy girls (8–15 years) | α-TOH/total lipids decreased in plasma and in LDL in obese subjects | - | |
| Observational | Strauss et al. [75] | n = 6139 children (6–19 years) enrolled in the NHANES III | decreased α-TOH/TC + TG plasma level in obese subjects | - | |
| Observational | Molnar et al. [76] | n = 15 obese adolescents n = 16 healthy adolescents (13–16 years) | α-TOH/TC + TG plasma level remained unchanged in obese subjects | - | |
| Observational | Gunanti et al. [77] | 6139 children (8–15 years) enrolled in the 2001–2004 NHANES | Adequate plasma level of α-TOH/TC are associated with reduced probability of overweight | - | |
| DMT2 | |||||
| DMT2 (Diabetes mellitus type 2) | Observational | Schneider et al. [78] | n = 31 DMT2 patients (46–79 years) n = 31 control subjects (38–63 years) | VLDLs and LDLs of DMT2 patients contained fewer vitamin E molecules compared to controls due to PLPT | - |
| Observational | Galvan et al. [79] | n = 12 male DMT2 patients (49–54 years) n = 19 control subjects (29–34 years) | Insulin infusion decreased α-TOH/LDL-C plasma level | - | |
| Observational (meta-analysis) | Kollerits et al. [80] | n = 20,136 subjects | Vitamin E-binding protein afamin is an independent predictor for DMT2 incidence, increase in afamin is associated with prevalence DMT2 | - | |
| Observational/ Interventional | Mayer-Davis et al. [81] | n = 895 non-diabetic adults (45–65 years) (n = 318 non-supplement users and n = 577 supplement users) | α-TOH plasma level is decreased in DMT2 patients and correlates with diabetes incidence, but not the nutritional intake/use of supplements | -/not defined | |
| Observational | Škrha et al. [82] | n = 62 DMT2 patients (49–64 years) n = 20 controls subjects | decreased α-TOH/TC, TG serum level in diabetic patients with macroangiopathy versus without vascular changes | - | |
| Observational | Salonen et al. [83] | n = 944 male healthy subjects (42–60 years) | decreased α-TOH plasma levels associated with increase diabetes risk | - | |
| Observational | Eljaoudi et al. [84] | n = 60 DMT2 patients n = 40 healthy subjects (31–76 years) | decreased α-TOH plasma level in DMT2 | - | |
| Observational | Nourooz-Zadeh et al. [85] | n = 87 DMT2 patients n = 41 healthy subjects (17–86 years) | decreased α-TOH/TC plasma level in DMT2 | - | |
| Observational | Mehmetoglu et al. [73] | n= 98 obese subjects n = 78 healthy subjects (18–65 years) | no correlation of α-TOH/TC + TG plasma level and insulin resistance in obese subjects | - | |
| Interventional | Rafighi et al. [86] | n = 170 DMT2 patients (30–60 years) | Vitamin E supplementation decreased blood glucose level, antioxidative capacity, (increased SOD and GSH enzyme activity), oxidative stress and insulin resistance | 200 mg (300 IU) vitamin E (/day) and 267 mg vitamin C per day for 3 months | |
| Interventional | Manning et al. [87] | n = 80 healthy subjects (38–57 years) | Vitamin E supplementation decreased inflammatory processes, fasting plasma glucose and improved insulin sensitivity in overweight subjects | 537 mg (800 IU) vitamin E per day or placebo for 3 months | |
| Interventional (Meta-analysis) | Xu et al. [88] | n = 714 subjects | vitamin E supplementation did not change glycemic control (HbA1c, fasting glucose, fasting insulin) | 134–1074 mg (200–1600 IU) per day for 6–27 weeks | |
| Fatty Liver Disease | |||||
| Observational | Erhardt et al. [89] | n = 50 NASH patients n = 40 healthy controls (35–67 years) | decreased α-TOH plasma levels in NASH patients | - | |
| Observational | Machado et al. [90] | n = 43 NASH patients n = 33 healthy controls (27–68 years) | increased α-TOH plasma levels in NASH patients | - | |
| Interventional | Violet et al. [91] | n = 6 female NASH patients(33–53 years) n = 10 female healthy controls (19–35 years) | alteration of α-TOH kinetics in women with obesity-associated hepatosteatosis compared to healthy controls, decreased release of α-TOH from the liver, lower α-TOH plasma level | 2 mg α-TOH once | |
| Interventional | Sanyal et al. [92] | n = 167 NASH patients (n = 83 placebo, n = 84 α-TOH, 34–59 years) | α-TOH supplementation improves ALT, AST, lobular inflammation and NASH compared to placebo treated group | 537 mg (800 IU) α-TOH per day or placebo for 2 years | |
| Interventional | Lavine et al. [93] | n = 11 NASH patients (<16 years) | α-TOH supplementation decreased ALT, AST, ALP | 268–805 mg (400–1200 IU) α-TOH for 4–10 months | |
| MetS | |||||
| Observational | Ford et al. [94] | MetS patients and healthy controls from NHANES III (≥20 years) | lower α-TOH plasma level in MetS patients | - | |
| Observational | Beydoun et al. [95] | n = 3008–9099 participants from NHANES 2001–2006 (20–85 years) | higher α-TOH plasma level in MetS patients | - | |
| Observational | Yen et al. [96] | n = 72 MetS patients n = 105 healthy controls | α-TOH/TG plasma level remained unchanged | - | |
| Interventional | Mah et al. [97] | n = 10 MetS patients n = 10 healthy controls | MetS patients have lower α-TOH /lipid plasma level and lower α-TOH absorption and impaired hepatic trafficking compared to healthy subjects | 15 mg α-TOH once |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ziegler, M.; Wallert, M.; Lorkowski, S.; Peter, K. Cardiovascular and Metabolic Protection by Vitamin E: A Matter of Treatment Strategy? Antioxidants 2020, 9, 935. https://doi.org/10.3390/antiox9100935
Ziegler M, Wallert M, Lorkowski S, Peter K. Cardiovascular and Metabolic Protection by Vitamin E: A Matter of Treatment Strategy? Antioxidants. 2020; 9(10):935. https://doi.org/10.3390/antiox9100935
Chicago/Turabian StyleZiegler, Melanie, Maria Wallert, Stefan Lorkowski, and Karlheinz Peter. 2020. "Cardiovascular and Metabolic Protection by Vitamin E: A Matter of Treatment Strategy?" Antioxidants 9, no. 10: 935. https://doi.org/10.3390/antiox9100935