Effects and Mechanisms of Tea for the Prevention and Management of Diabetes Mellitus and Diabetic Complications: An Updated Review
Abstract
1. Introduction
2. Epidemiological Investigations
3. Experimental Studies
3.1. Diabetes Mellitus
3.1.1. Type 1 Diabetes Mellitus (T1DM)
3.1.2. Type 2 Diabetes Mellitus (T2DM)
3.2. Diabetic Complications
3.2.1. Diabetic Nephropathy
3.2.2. Diabetic Cardiovascular Diseases
3.2.3. Diabetic Neuropathy
3.2.4. Diabetic Retinopathy
3.2.5. Diabetic Hepatopathy
3.2.6. Other Complications
3.3. Adjuvant Therapy
4. Clinical Trials
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Assoc, A.D. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37, S81–S90. [Google Scholar]
- Whiting, D.R.; Guariguata, L.; Weil, C.; Shaw, J. IDF diabetes atlas: Global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res. Clin. Pract. 2011, 94, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.E.; Sicree, R.A.; Zimmet, P.Z. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res. Clin. Pract. 2010, 87, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Guariguata, L.; Whiting, D.R.; Hambleton, I.; Beagley, J.; Linnenkamp, U.; Shaw, J.E. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res. Clin. Pract. 2014, 103, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Du, W.H.; Peng, S.M.; Liu, Z.H.; Shi, L.; Tan, L.F.; Zou, X.Q. Hypoglycemic effect of the water extract of Pu-erh tea. J. Agric. Food Chem. 2012, 60, 10126–10132. [Google Scholar] [CrossRef]
- Li, Y.; Wang, C.; Huai, Q.; Guo, F.; Liu, L.; Feng, R.; Sun, C. Effects of tea or tea extract on metabolic profiles in patients with type 2 diabetes mellitus: A meta-analysis of ten randomized controlled trials. Diabetes Res. Clin. Pract. 2016, 32, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Gan, R.Y.; Li, H.B.; Sui, Z.Q.; Corke, H. Absorption, metabolism, anti-cancer effect and molecular targets of epigallocatechin gallate (EGCG): An updated review. Crit. Rev. Food Sci. Nutr. 2018, 58, 924–941. [Google Scholar] [CrossRef]
- Li, F.; Li, S.; Li, H.-B.; Deng, G.-F.; Ling, W.-H.; Xu, X.-R. Antiproliferative activities of tea and herbal infusions. Food Funct. 2013, 4, 530–538. [Google Scholar] [CrossRef]
- Li, S.; Gan, L.-Q.; Li, S.-K.; Zheng, J.-C.; Xu, D.-P.; Li, H.-B. Effects of herbal infusions, tea and carbonated beverages on alcohol dehydrogenase and aldehyde dehydrogenase activity. Food Funct. 2014, 5, 42–49. [Google Scholar] [CrossRef]
- Li, Y.; Li, S.; Lin, S.-J.; Zhang, J.-J.; Zhao, C.-N.; Li, H.-B. Microwave-assisted extraction of natural antioxidants from the exotic gordonia axillaris fruit: Optimization and identification of phenolic compounds. Molecules 2017, 22, 1481. [Google Scholar] [CrossRef]
- Meng, X.; Li, S.; Li, Y.; Gan, R.Y.; Li, H.B. Gut microbiota’s relationship with liver disease and role in hepatoprotection by dietary natural products and probiotics. Nutrients 2018, 10, 1457. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Li, Y.; Li, S.; Gan, R.-Y.; Li, H.-B. Natural products for prevention and treatment of chemical-induced liver injuries. Compr. Rev. Food. Sci. Food Saf. 2018, 17, 472–495. [Google Scholar] [CrossRef]
- Tao, J.; Li, S.; Gan, R.-Y.; Zhao, C.-N.; Meng, X.; Li, H.-B. Targeting gut microbiota with dietary components on cancer: Effects and potential mechanisms of action. Crit. Rev. Food Sci. Nutr. 2019, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-Y.; Zhao, C.-N.; Cao, S.-Y.; Tang, G.-Y.; Gan, R.-Y.; Li, H.-B. Effects and mechanisms of tea for the prevention and management of cancers: An updated review. Crit. Rev. Food Sci. Nutr. 2019, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Teng, Y.; Li, D.X.; Guruvaiah, P.; Xu, N.; Xie, Z.W. Dietary supplement of large yellow tea ameliorates metabolic syndrome and attenuates hepatic steatosis in db/db Mice. Nutrients 2018, 10, 75. [Google Scholar] [CrossRef] [PubMed]
- Ning, J.M.; Li, D.X.; Luo, X.J.; Ding, D.; Song, Y.S.; Zhang, Z.Z.; Wan, X.C. Stepwise identification of six tea (camellia sinensis (L.)) categories based on catechins, caffeine, and theanine contents combined with fisher discriminant analysis. Food Anal. Meth. 2016, 9, 3242–3250. [Google Scholar] [CrossRef]
- Ramadan, G.; El-Beih, N.M.; El-Ghffar, E.A.A. Modulatory effects of black v. green tea aqueous extract on hyperglycaemia, hyperlipidaemia and liver dysfunction in diabetic and obese rat models. Br. J. Nutr. 2009, 102, 1611–1619. [Google Scholar] [CrossRef]
- Jing, Y.L.; Han, G.J.; Hu, Y.; Bi, Y.; Li, L.R.; Zhu, D.L. Tea consumption and risk of type 2 diabetes: A meta-analysis of cohort studies. J. Gen. Intern. Med. 2009, 24, 557–562. [Google Scholar] [CrossRef]
- Boggs, D.A.; Rosenberg, L.; Ruiz-Narvaez, E.A.; Palmer, J.R. Coffee, tea, and alcohol intake in relation to risk of type 2 diabetes in African American women. Am. J. Clin. Nutr. 2010, 92, 960–966. [Google Scholar] [CrossRef]
- Tang, W.P.; Li, S.M.; Liu, Y.; Huang, M.T.; Ho, C.T. Anti-diabetic activity of chemically profiled green tea and black tea extracts in a type 2 diabetes mice model via different mechanisms. J. Funct. Foods 2013, 5, 1784–1793. [Google Scholar] [CrossRef]
- Toolsee, N.A.; Aruoma, O.I.; Gunness, T.K.; Kowlessur, S.; Dambala, V.; Murad, F.; Googoolye, K.; Daus, D.; Indelicato, J.; Rondeau, P.; et al. Effectiveness of green tea in a randomized human cohort: Relevance to diabetes and its complications. BioMed Res. Int. 2013, 2013, 412379. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, F.; Haines, D.; Al-Ozairi, E.; Dashti, A. Effect of black tea consumption on intracellular cytokines, regulatory T cells and metabolic biomarkers in type 2 diabetes patients. Phytother. Res. 2016, 30, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Hinkle, S.N.; Laughon, S.K.; Catov, J.M.; Olsen, J.; Bech, B.H. First trimester coffee and tea intake and risk of gestational diabetes mellitus: A study within a national birth cohort. BJOG Int. J. Obstet. Gynaecol. 2015, 122, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, J.A.; Axen, K.V.; Schnoll, R.; Boozer, C.N. Coffee, tea and diabetes: The role of weight loss and caffeine. Int. J. Obes. 2005, 29, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Hamer, M.; Witte, D.R.; Mosdol, A.; Marmot, M.G.; Brunner, E.J. Prospective study of coffee and tea consumption in relation to risk of type 2 diabetes mellitus among men and women: The whitehall II study. Br. J. Nutr. 2008, 100, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Hirata, A.; Ohnaka, K.; Tashiro, N.; Wang, Z.; Kohno, M.; Kiyohara, C.; Kono, S.; Takayanagi, R. Effect modification of green tea on the association between rice intake and the risk of diabetes mellitus: A prospective study in Japanese men and women. Asia Pac. J. Clin. Nutr. 2017, 26, 545–555. [Google Scholar] [PubMed]
- Odegaard, A.O.; Pereira, M.A.; Koh, W.P.; Arakawa, K.; Lee, H.P.; Yu, M.C. Coffee, tea, and incident type 2 diabetes: The Singapore chinese health study. Am. J. Clin. Nutr. 2008, 88, 979–985. [Google Scholar] [CrossRef]
- Iso, H.; Date, C.; Wakai, K.; Fukui, M.; Tamakoshi, A.; Grp, J.S. The relationship between green tea and total caffeine intake and risk for self-reported type 2 diabetes among Japanese adults. Ann. Intern. Med. 2006, 144, 554–562. [Google Scholar] [CrossRef]
- Nguyen, C.T.; Lee, A.H.; Pham, N.M.; Do, V.V.; Ngu, N.D.; Tran, B.Q.; Binns, C. Habitual tea drinking associated with a lower risk of type 2 diabetes in Vietnamese adults. Asia Pac. J. Clin. Nutr. 2018, 27, 701–706. [Google Scholar]
- Ma, Q.H.; Chen, D.D.; Sun, H.P.; Yan, N.; Xu, Y.; Pan, C.W. Regular Chinese green tea consumption is protective for diabetic retinopathy: A clinic-based case-control study. J. Diabetes Res. 2015, 2015, 231570. [Google Scholar] [CrossRef]
- Yi, D.Q.; Tan, X.R.; Zhao, Z.G.; Cai, Y.M.; Li, Y.M.; Lin, X.Y.; Lu, S.L.; Chen, Y.S.; Zhang, Q.Y. Reduced risk of dyslipidaemia with oolong tea consumption: A population-based study in southern China. Br. J. Nutr. 2014, 111, 1421–1429. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Lionis, C.; Zeimbekis, A.; Gelastopoulou, K.; Papairakleous, N.; Das, U.N.; Polychronopoulos, E. Long-term tea intake is associated with reduced prevalence of (type 2) diabetes mellitus among elderly people from mediterranean islands: MEDIS epidemiological study. Yonsei Med. J. 2009, 50, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Huxley, R.; Lee, C.M.Y.; Barzi, F.; Timmermeister, L.; Czernichow, S.; Perkovic, V.; Grobbee, D.E.; Batty, D.; Woodward, M. Coffee, Decaffeinated coffee, and tea consumption in relation to incident type 2 diabetes mellitus: A systematic review with meta-analysis. Arch. Intern. Med. 2009, 169, 2053–2063. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.S.; Wang, W.Y.; Fan, W.Y.; Deng, Q.; Wang, X. Tea consumption and risk of type 2 diabetes: A dose-response meta-analysis of cohort studies. Br. J. Nutr. 2014, 111, 1329–1339. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Mao, Q.X.; Xu, H.X.; Ma, X.; Zeng, C.Y. Tea consumption and risk of type 2 diabetes mellitus: A systematic review and meta-analysis update. BMJ Open 2014, 4, e005632. [Google Scholar] [CrossRef] [PubMed]
- Hayashino, Y.; Fukuhara, S.; Okamura, T.; Tanaka, T.; Ueshima, H.; Grp, H.-O.R. High oolong tea consumption predicts future risk of diabetes among Japanese male workers: A prospective cohort study. Diabetic Med. 2011, 28, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tian, J.; Jiang, J.; Li, L.; Ying, X.; Tian, H.; Nie, M. Effects of green tea or green tea extract on insulin sensitivity and glycaemic control in populations at risk of type 2 diabetes mellitus: A systematic review and meta-analysis of randomised controlled trials. J. Hum. Nutr. Diet. 2014, 27, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.Y.; Song, P.G.; Perry, R.; Penfold, C.; Cooper, A.R. The effectiveness of green tea or green tea extract on insulin resistance and glycemic control in type 2 diabetes mellitus: A meta-analysis. Diabetes Metab. J. 2017, 41, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Butacnum, A.; Chongsuwat, R.; Bumrungpert, A. Black tea consumption improves postprandial glycemic control in normal and pre-diabetic subjects: A randomized, double-blind, placebo-controlled crossover study. Asia Pac. J. Clin. Nutr. 2017, 26, 59–64. [Google Scholar]
- Van Woudenbergh, G.J.; Kuijsten, A.; Drogan, D.; Van Der, A.D.L.; Romaguera, D.; Ardanaz, E.; Amiano, P.; Barricarte, A.; Beulens, J.W.J.; Boeing, H.; et al. Tea consumption and incidence of type 2 diabetes in europe: The EPIC-interact case-cohort study. PLoS ONE 2012, 7, e36910. [Google Scholar]
- Borges, C.M.; Papadimitriou, A.; Duarte, D.A.; Lopes de Faria, J.M.; Lopes de Faria, J.B. The use of green tea polyphenols for treating residual albuminuria in diabetic nephropathy: A double-blind randomised clinical trial. Sci. Rep. 2016, 6, 28282. [Google Scholar] [CrossRef] [PubMed]
- Nunes, A.R.; Alves, M.G.; Moreira, P.I.; Oliveira, P.F.; Silva, B.M. Can tea consumption be a safe and effective therapy against diabetes mellitus-induced neurodegeneration? Curr. Neuropharmacol. 2014, 12, 475–489. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Choi, H. Green tea, anti-diabetic or diabetogenic: A dose response study. Biofactors 2007, 29, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.J.; Shi, S.S.; Bao, B.; Li, X.J.; Wang, S.C. Structure characterization of an arabinogalactan from green tea and its anti-diabetic effect. Carbohydr. Polym. 2015, 124, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Chemler, J.A.; Lock, L.T.; Koffas, M.A.; Tzanakakis, E.S. Standardized biosynthesis of flavan-3-ols with effects on pancreatic β-cell insulin secretion. Appl. Microbiol. Biotechnol. 2007, 77, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Pastoriza, S.; Mesias, M.; Cabrera, C.; Rufian-Henares, J.A. Healthy properties of green and white teas: An update. Food Funct. 2017, 8, 2650–2662. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Ding, Y.; Dai, X.; Wang, J.; Li, Y. Epigallocatechin-3-gallate protects pro-inflammatory cytokine induced injuries in insulin-producing cells through the mitochondrial pathway. Eur. J. Pharmacol. 2011, 670, 311–316. [Google Scholar] [CrossRef]
- Yan, J.Q.; Zhao, Y.; Suo, S.; Liu, Y.; Zhao, B.L. Green tea catechins ameliorate adipose insulin resistance by improving oxidative stress. Free Radic. Biol. Med. 2012, 52, 1648–1657. [Google Scholar] [CrossRef]
- Ortsater, H.; Grankvist, N.; Wolfram, S.; Kuehn, N.; Sjoholm, A. Diet supplementation with green tea extract epigallocatechin gallate prevents progression to glucose intolerance in db/db mice. Nutr. Metab. 2012, 9, 11. [Google Scholar] [CrossRef]
- Ma, X.; Tsuda, S.; Yang, X.; Gu, N.; Tanabe, H.; Oshima, R.; Matsushita, T.; Egawa, T.; Dong, A.J.; Zhu, B.W.; et al. Pu-erh tea hot-water extract activates Akt and induces insulin-independent glucose transport in rat skeletal muscle. J. Med. Food 2013, 16, 259–262. [Google Scholar] [CrossRef]
- Hameed, I.; Masoodi, S.R.; Mir, S.A.; Nabi, M.; Ghazanfar, K.; Ganai, B.A. Type 2 diabetes mellitus: From a metabolic disorder to an inflammatory condition. World J. Diabetes 2015, 6, 598–612. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.L.; Lin, J.K. Epigallocatechin gallate (EGCG) attenuates high glucose-induced insulin signaling blockade in human hepG2 hepatoma cells. Mol. Nutr. Food Res. 2008, 52, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.L.; Polansky, M.M.; Harry, D.; Anderson, R.A. Green tea polyphenols improve cardiac muscle mRNA and protein levels of signal pathways related to insulin and lipid metabolism and inflammation in insulin-resistant rats. Mol. Nutr. Food Res. 2010, 54, S14–S23. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, T.; Kukidome, D.; Sonoda, K.; Fujisawa, K.; Matsuhisa, T.; Motoshima, H.; Matsumura, T.; Araki, E. Impact of mitochondrial ROS production in the pathogenesis of insulin resistance. Diabetes Res. Clin. Pract. 2007, 77, S161–S164. [Google Scholar] [CrossRef] [PubMed]
- Yasui, K.; Tanabe, H.; Okada, N.; Fukutomi, R.; Ishigami, Y.; Isemura, M. Effects of catechin-rich green tea on gene expression of gluconeogenic enzymes in rat hepatoma H4IIE cells. Biomed. Res. 2010, 31, 183–189. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shin, D.W.; Kim, S.N.; Lee, S.M.; Lee, W.; Song, M.J.; Park, S.M.; Lee, T.R.; Baik, J.-H.; Kim, H.K.; Hong, J.-H.; et al. (−)-Catechin promotes adipocyte differentiation in human bone marrow mesenchymal stem cells through PPARγ transactivation. Biochem. Pharmacol. 2009, 77, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.M.; Chang, Y.H.; Chao, Y.C.; Lin, J.A.; Wu, C.H.; Lai, C.Y.; Chan, K.C.; Tseng, S.T.; Yen, G.C. EGCG-rich green tea extract stimulates sRAGE secretion to inhibit S100A12-RAGE axis through ADAM10-mediated ectodomain shedding of extracellular RAGE in type 2 diabetes. Mol. Nutr. Food Res. 2013, 57, 2264–2268. [Google Scholar] [CrossRef]
- Neyestani, T.R.; Shariatzade, N.; Kalayi, A.; Gharavi, A.; Khalaji, N.; Dadkhah, M.; Zowghi, T.; Haidari, H.; Shab-Bidar, S. Regular daily intake of black tea improves oxidative stress biomarkers and decreases serum C-reactive protein levels in type 2 diabetic patients. Ann. Nutr. Metab. 2010, 57, 40–49. [Google Scholar] [CrossRef]
- Yamashita, Y.; Wang, L.Q.; Wang, L.H.; Tanaka, Y.; Zhang, T.S.; Ashida, H. Oolong, black and pu-erh tea suppresses adiposity in mice via activation of AMP-activated protein kinase. Food Funct. 2014, 5, 2420–2429. [Google Scholar] [CrossRef]
- Hilal, Y.; Engelhardt, U. Correction/Erratum characterisation of white tea-comparison to green and black tea. J. Verbrauch. Lebensm. 2009, 4, 218–220. [Google Scholar]
- Sanlier, N.; Atik, İ.; Atik, A. A minireview of effects of white tea consumption on diseases. Trends Food Sci. Technol. 2018, 82, 82–88. [Google Scholar] [CrossRef]
- Islam, M.S. Effects of the aqueous extract of white tea (Camellia sinensis) in a streptozotocin-induced diabetes model of rats. Phytomedicine 2011, 19, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Chu, J.; Wang, M.; Chen, L.; Zhang, L.; Xie, Z.W.; Zhang, J.S.; Ho, C.T.; Li, D.X.; Wan, X.C. Large yellow tea attenuates macrophage-related chronic inflammation and metabolic syndrome in high-fat diet treated mice. J. Agric. Food Chem. 2018, 66, 3823–3832. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, R.; Kobayashi, M.; Matsuda, Y.; Ojika, M.; Shigeoka, S.; Yamamoto, Y.; Tou, Y.; Inoue, T.; Katagiri, T.; Murai, A.; et al. Coffee and caffeine ameliorate hyperglycemia, fatty liver, and inflammatory adipocytokine expression in spontaneously diabetic KK-Ay Mice. J. Agric. Food Chem. 2010, 58, 5597–5603. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.Y.; Yu, Z.; Zhu, H.K.; Zhang, W.; Chen, Y.Q. In vitro α-glucosidase inhibitory activity of isolated fractions from water extract of qingzhuan dark tea. BMC Complement. Altern. Med. 2016, 16, 378. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.-T.; Lin-Shiau, S.-Y.; Shyur, L.-F.; Lin, J.-K. Pu-erh tea polysaccharides decrease blood sugar by inhibition of α-glucosidase activity in vitro and in mice. Food Funct. 2015, 6, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.Z.; Zheng, W.; Zhang, B.W.; Chen, X.J.; Zhang, J.; Pang, X.; Zhang, Y.; Jia, D.X.; Pei, S.R.; Dong, Y.S.; et al. Comparison of hypoglycemic effects of ripened pu-erh tea and raw pu-erh tea in streptozotocin-induced diabetic rats. RSC Adv. 2019, 9, 2967–2977. [Google Scholar] [CrossRef]
- Lin, H.-C.; Lee, C.-T.; Yen, Y.-Y.; Chu, C.-L.; Hsieh, Y.-P.; Yang, C.-S.; Lan, S.-J. Systematic review and meta-analysis of anti-hyperglycaemic effects of pu-erh tea. Int. J. Food Sci. Technol. 2019, 54, 516–525. [Google Scholar] [CrossRef]
- Du, H.; Wang, Q.; Yang, X. Fu Brick Tea alleviates chronic kidney disease of rats with high fat diet consumption through attenuating insulin resistance in skeletal muscle. J. Agric. Food Chem. 2019, 67, 2839–2847. [Google Scholar] [CrossRef]
- Renno, W.M.; Abdeen, S.; Alkhalaf, M.; Asfar, S. Effect of green tea on kidney tubules of diabetic rats. Br. J. Nutr. 2008, 100, 652–659. [Google Scholar] [CrossRef]
- Zhou, J.; Zhang, L.; Meng, Q.L.; Wang, Y.J.; Long, P.P.; Ho, C.T.; Cui, C.J.; Cao, L.T.; Li, D.X.; Wan, X.C. Roasting improves the hypoglycemic effects of a large-leaf yellow tea infusion by enhancing the levels of epimerized catechins that inhibit α-glucosidase. Food Funct. 2018, 9, 5162–5168. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Gonzalez, J.F.; Mora-Fernandez, C.; Muros de Fuentes, M.; Garcia-Perez, J. Inflammatory molecules and pathways in the pathogenesis of diabetic nephropathy. Nat. Rev. Nephrol. 2011, 7, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.J.; Wang, L.; Li, Z.; Zhu, D.N.; Guo, S.C.; Xin, W.F.; Yang, Y.F.; Cong, X.; Ma, T.; Shen, P.P.; et al. Inhibition of advanced glycation end product formation by pu-erh tea ameliorates progression of experimental diabetic nephropathy. J. Agric. Food Chem. 2012, 60, 4102–4110. [Google Scholar] [CrossRef] [PubMed]
- Hase, M.; Babazono, T.; Karibe, S.; Kinae, N.; Iwamoto, Y. Renoprotective effects of tea catechin in streptozotocin-induced diabetic rats. Int. Urol. Nephrol. 2006, 38, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Yaribeygi, H.; Atkin, S.L.; Sahebkar, A. Interleukin-18 and diabetic nephropathy: A review. J. Cell. Physiol. 2019, 234, 5674–5682. [Google Scholar] [CrossRef] [PubMed]
- Jeong, B.C.; Kim, B.S.; Kim, J.I.; Kim, H.H. Effects of green tea on urinary stone formation: An in vivo and in vitro study. J. Endourol. 2006, 20, 356–361. [Google Scholar] [CrossRef]
- Fiorino, P.; Evangelista, F.S.; Santos, F.; Magri, F.M.M.; Delorenzi, J.C.M.O.B.; Ginoza, M.; Farah, V. The effects of green tea consumption on cardiometabolic alterations induced by experimental diabetes. Exp. Diabetes Res. 2012, 2012, 309231. [Google Scholar] [CrossRef]
- Peixoto, E.B.; Papadimitriou, A.; Teixeira, D.A.T.; Montemurro, C.; Duarte, D.A.; Silva, K.C.; Joazeiro, P.P.; de Faria, J.M.L.; de Faria, J.B.L. Reduced LRP6 expression and increase in the interaction of GSK3β with p53 contribute to podocyte apoptosis in diabetes mellitus and are prevented by green tea. J. Nutr. Biochem. 2015, 26, 416–430. [Google Scholar] [CrossRef]
- Yokozawa, T.; Nakagawa, T.; Oya, T.; Okubo, T.; Juneja, L.R. Green tea polyphenols and dietary fibre protect against kidney damage in rats with diabetic nephropathy. J. Pharm. Pharmacol. 2005, 57, 773–780. [Google Scholar] [CrossRef]
- Zhu, D.N.; Wang, L.; Zhou, Q.L.; Yan, S.J.; Li, Z.; Sheng, J.; Zhang, W.S. (+)-Catechin ameliorates diabetic nephropathy by trapping methylglyoxal in type 2 diabetic mice. Mol. Nutr. Food. Res. 2014, 58, 2249–2260. [Google Scholar] [CrossRef]
- Choi, J.H.; Chai, Y.M.; Joo, G.J.; Rhee, I.K.; Lee, I.S.; Kim, K.R.; Choi, M.S.; Rhee, S.J. Effects of green tea catechin on polymorphonuclear leukocyte 5′-lipoxygenase activity, leukotriene B-4 synthesis, and renal damage in diabetic rats. Ann. Nutr. Metab. 2004, 48, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Mohabbulla Mohib, M.; Fazla Rabby, S.M.; Paran, T.Z.; Mehedee Hasan, M.; Ahmed, I.; Hasan, N.; Abu Taher Sagor, M.; Mohiuddin, S.; Hsu, T.-C. Protective role of green tea on diabetic nephropathy—A review. Cogent Biol. 2016, 2, 1248166. [Google Scholar] [CrossRef]
- Petroski, M.D.; Deshaies, R. Effects of green tea on matrix metalloproteinases in streptozotocin-induced diabetic rats. J. Clin. Biochem. Nutr. 2005, 37, 77–85. [Google Scholar]
- Nagase, H.; Visse, R.; Murphy, G. Structure and function of matrix metalloproteinases and TIMPs. Cardiovasc. Res. 2006, 69, 562–573. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, J.; Nandy, S.K.; Chowdhury, A.; Chakraborti, T.; Chakraborti, S. Inhibition of MMP-9 by green tea catechins and prediction of their interaction by molecular docking analysis. Biomed. Pharmacother. 2016, 84, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.M.; Sun, L.L.; Lai, X.F.; Xiang, L.M.; Li, Q.H.; Zhang, W.J.; Zhang, L.Z.; Sun, S.L. Tea polypeptide ameliorates diabetic nephropathy through RAGE and NF-κB signaling pathway in type 2 diabetes mice. J. Agric. Food Chem. 2018, 66, 11957–11967. [Google Scholar] [CrossRef] [PubMed]
- Ni, D.; Chen, Y.; Song, C.; Xie, B.; Zhou, S. Effect of oolong tea polysaccharide on hepatic-nephritic antioxidation and histommorphology in the diabetic rats. J. Tea Sci. 2003, 23, 11–15. [Google Scholar]
- Alves, M.G.; Martins, A.D.; Teixeira, N.F.; Rato, L.; Oliveira, P.F.; Silva, B.M. White tea consumption improves cardiac glycolytic and oxidative profile of prediabetic rats. J. Funct. Foods 2015, 14, 102–110. [Google Scholar] [CrossRef]
- Lin, C.; Zhang, M.; Zhang, Y.; Yang, K.; Hu, J.; Si, R.; Zhang, G.; Gao, B.; Li, X.; Xu, C.; et al. Helix B surface peptide attenuates diabetic cardiomyopathy via AMPK-dependent autophagy. Biochem. Biophys. Res. Commun. 2017, 482, 665–671. [Google Scholar] [CrossRef]
- Zhou, H.; Chen, Y.; Huang, S.W.; Hu, P.F.; Tang, L.J. Regulation of autophagy by tea polyphenols in diabetic cardiomyopathy. J. Zhejiang Univ.-Sci. B 2018, 19, 333–341. [Google Scholar] [CrossRef]
- Fahie, K.; Zachara, N.E. Molecular functions of glycoconjugates in autophagy. J. Mol. Biol. 2016, 428, 3305–3324. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.C.; Bartlett, J.J.; Perez, L.J.; Brunt, K.R.; Legare, J.F.; Hassan, A.; Kienesberger, P.C.; Pulinilkunnil, T. Glucolipotoxicity diminishes cardiomyocyte TFEB and inhibits lysosomal autophagy during obesity and diabetes. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2016, 1861, 1893–1910. [Google Scholar] [CrossRef] [PubMed]
- Velazquez, A.P.; Graef, M. Autophagy regulation depends on ER homeostasis controlled by lipid droplets. Autophagy 2016, 12, 1409–1410. [Google Scholar] [CrossRef] [PubMed]
- Babu, P.V.A.; Sabitha, K.E.; Shyamaladevi, C.S. Green tea impedes dyslipidemia, lipid peroxidation, protein glycation and ameliorates Ca2+-ATPase and Na+/K+-ATPase activity in the heart of streptozotocin-diabetic rats. Chem.-Biol. Interact. 2006, 162, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Potenza, M.A.; Marasciulo, F.L.; Tarquinio, M.; Tiravanti, E.; Colantuono, G.; Federici, A.; Kim, J.A.; Quon, M.J.; Montagnani, M. EGCG, a green tea polyphenol, improves endothelial function and insulin sensitivity, reduces blood pressure, and protects against myocardial I/R injury in SHR. Am. J. Physiol.-Endocrinol. Metab. 2007, 292, E1378–E1387. [Google Scholar] [CrossRef]
- Mahmoud, M.F.; Hassan, N.A.; El Bassossy, H.M.; Fahmy, A. Quercetin protects against diabetes-induced exaggerated vasoconstriction in rats: Effect on low grade inflammation. PLoS ONE 2013, 8, e63784. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Ladomery, M.R.; Chedea, V.S.; Irimie, A.; Berindan-Neagoe, I. The relationship between the structure and biological actions of green tea catechins. Food Chem. 2013, 141, 3282–3289. [Google Scholar] [CrossRef]
- Babu, P.V.A.; Sabitha, K.E.; Shyamaladevi, C.S. Therapeutic effect of green tea extract on advanced glycation and cross-linking of collagen in the aorta of streptozotocin diabetic rats. Clin. Exp. Pharmacol. Physiol. 2006, 33, 351–357. [Google Scholar] [CrossRef]
- Minatti, J.; Wazlawik, E.; Hort, M.A.; Zaleski, F.L.; Ribeiro-do-Valle, R.M.; Maraschin, M.; da Silva, E.L. Green tea extract reverses endothelial dysfunction and reduces atherosclerosis progression in homozygous knockout low-density lipoprotein receptor mice. Nutr. Res. 2012, 32, 684–693. [Google Scholar] [CrossRef]
- Babu, P.V.A.; Sabitha, K.E.; Srinivasan, P.; Shyamaladevi, C.S. Green tea attenuates diabetes induced Maillard-type fluorescence and collagen cross-linking in the heart of streptozotocin diabetic rats. Pharmacol. Res. 2007, 55, 433–440. [Google Scholar] [CrossRef]
- Tijburg, L.M.B.; Mattern, T.; Folts, J.D.; Weisgerber, U.M.; Katan, M.B. Tea flavonoids and cardiovascular diseases: A review. Crit. Rev. Food Sci. Nutr. 1997, 37, 771–785. [Google Scholar] [CrossRef] [PubMed]
- Murase, T.; Misawa, K.; Haramizu, S.; Hase, T. Catechin-induced activation of the LKB1/AMP-activated protein kinase pathway. Biochem. Pharmacol. 2009, 78, 78–84. [Google Scholar] [CrossRef]
- Zhao, P.; Kuai, J.K.; Gao, J.J.; Sun, L.; Wang, Y.; Yao, L.N. Delta opioid receptor agonist attenuates lipopolysaccharide-induced myocardial injury by regulating autophagy. Biochem. Biophys. Res. Commun. 2017, 492, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Yaribeygi, H.; Atkin, S.L.; Pirro, M.; Sahebkar, A. A review of the anti-inflammatory properties of antidiabetic agents providing protective effects against vascular complications in diabetes. J. Cell. Physiol. 2019, 234, 8286–8294. [Google Scholar] [CrossRef] [PubMed]
- Anter, E.; Chen, K.; Shapira, O.M.; Karas, R.H.; Keaney, J.F. P38 Mitogen-activated protein kinase activates eNOS in endothelial cells by an estrogen receptor α-dependent pathway in response to black tea polyphenols. Circ. Res. 2005, 96, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Anter, E.; Thomas, S.R.; Schulz, E.; Shapira, O.M.; Vita, J.A.; Keaney, J.F. Activation of endothelial nitric-oxide synthase by the p38 MAPK in response to black tea polyphenols. J. Biol. Chem. 2004, 279, 46637–46643. [Google Scholar] [CrossRef] [PubMed]
- Rahma, A.; Martini, R.; Kusharto, C.M.; Damayanthi, E.; Rohdiana, D. White tea camellia sinensis and moringa oleifera as antihyperglycemic agent on streptozotocin-induced diabetic sprague dawley rats. Jurnal Gizi dan Pangan 2017, 12, 179–186. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Gachhui, R.; Sil, P.C. Effect of Kombucha, a fermented black tea in attenuating oxidative stress mediated tissue damage in alloxan induced diabetic rats. Food Chem. Toxicol. 2013, 60, 328–340. [Google Scholar] [CrossRef]
- Rodrigues, B.; Cam, M.C.; McNeill, J.H. Metabolic disturbances in diabetic cardiomyopathy. Mol. Cell. Biochem. 1998, 180, 53–57. [Google Scholar] [CrossRef]
- How, O.J.; Aasum, E.; Severson, D.L.; Chan, W.Y.A.; Essop, M.F.; Larsen, T.S. Increased myocardial oxygen consumption reduces cardiac efficiency in diabetic mice. Diabetes 2006, 55, 466–473. [Google Scholar] [CrossRef]
- Xu, J.Y.; Wang, M.; Zhao, J.P.; Wang, Y.H.; Tang, Q.; Khan, I.A. Yellow tea (Camellia sinensis L.), a promising Chinese tea: Processing, chemical constituents and health benefits. Food Res. Int. 2018, 107, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Han, M.M.; Zhao, G.S.; Wang, Y.J.; Wang, D.X.; Sun, F.; Ning, J.M.; Wan, X.C.; Zhang, J.S. Safety and anti-hyperglycemic efficacy of various tea types in mice. Sci. Rep. 2016, 6, 31703. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.Y.; Hackman, R.M.; Ensunsa, J.L.; Holt, R.R.; Keen, C.L. Antioxidative activities of oolong tea. J. Agric. Food Chem. 2002, 50, 6929–6934. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.B.; Hayashi, S.; Fang, C.Y.; Hao, S.M.; Wang, X.J.; Nishiguchi, S.; Tsutsui, H.; Sheng, J. Pu-erh tea extract-mediated protection against hepatosteatosis and insulin resistance in mice with diet-induced obesity is associated with the induction of de novo lipogenesis in visceral adipose tissue. J. Gastroenterol. 2017, 52, 1240–1251. [Google Scholar] [CrossRef] [PubMed]
- Shammas, M.A.; Neri, P.; Koley, H.; Batchu, R.B.; Bertheau, R.C.; Munshi, V.; Prabhala, R.; Fulciniti, M.; Tai, Y.; Treon, S.P.; et al. Specific killing of multiple myeloma cells by (-)-epigaflocatechin-3-gallate extracted from green tea: Biologic activity and therapeutic implications. Blood 2006, 108, 2804–2810. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Bae, J.H.; Lee, S.R. Protective effect of green tea polyphenol EGCG against neuronal damage and brain edema after unilateral cerebral ischemia in gerbils. J. Neurosci. Res. 2004, 77, 892–900. [Google Scholar] [CrossRef] [PubMed]
- Singal, A.; Anjaneyulu, M.; Chopra, K. Modulatory role of green tea extract on antinociceptive effect of morphine in diabetic mice. J. Med. Food 2005, 8, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.X.; Xu, C.; Reece, E.A.; Yang, P.X. The green tea polyphenol EGCG alleviates maternal diabetes-induced neural tube defects by inhibiting DNA hypermethylation. Am. J. Obstet. Gynecol. 2016, 215, 368. [Google Scholar] [CrossRef] [PubMed]
- Barber, A.J. A new view of diabetic retinopathy: A neurodegenerative disease of the eye. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2003, 27, 283–290. [Google Scholar] [CrossRef]
- Rahimi-Madiseh, M.; Malekpour-Tehrani, A.; Bahmani, M.; Rafieian-Kopaei, M. The research and development on the antioxidants in prevention of diabetic complications. Asian Pac. J. Trop. Med. 2016, 9, 825–831. [Google Scholar] [CrossRef]
- Mustata, G.T.; Rosca, M.; Biemel, K.M.; Reihl, O.; Smith, M.A.; Viswanathan, A.; Strauch, C.; Du, Y.; Tang, J.; Kern, T.S.; et al. Paradoxical effects of green tea (Camellia Sinensis) and antioxidant vitamins in diabetic rats: Improved retinopathy and renal mitochondrial defects but deterioration of collagen matrix glycoxidation and cross-linking. Diabetes 2005, 54, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Silva, K.C.; Rosales, M.A.B.; Hamassaki, D.E.; Saito, K.C.; Faria, A.M.; Ribeiro, P.A.O.; de Faria, J.B.L.; de Faria, J.M.L. Green tea is neuroprotective in diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2013, 54, 1325–1336. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Gupta, S.K.; Nag, T.C.; Srivastava, S.; Saxena, R. Green tea prevents hyperglycemia-induced retinal oxidative stress and inflammation in streptozotocin-induced diabetic rats. Ophthalmic Res. 2012, 47, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.A.; Zhang, J. Black and green teas equally inhibit diabetic cataracts in a streptozotocin-induced rat model of diabetes. J. Agric. Food Chem. 2005, 53, 3710–3713. [Google Scholar] [CrossRef]
- Sook, S.J.; Quan, Z.-J. Effect of oolong tea extracts on plasma glucose level and antioxidant system in diabetic rats. J. Community Nutr. 2006, 8, 208–214. [Google Scholar]
- Al-Hussaini, A.A.; Sulaiman, N.M.; AlZahrani, M.D.; Alenizi, A.S.; Khan, M. Prevalence of hepatopathy in type 1 diabetic children. BMC Pediatr. 2012, 12, 160. [Google Scholar] [CrossRef] [PubMed]
- Akbar, A.A.; Daryoush, M.; Ali, R.; Mehrdad, N. Green tea attenuates hepatic tissue injury in STZ-streptozotocin-induced diabetic rats. J. Anim. Vet. Adv. 2012, 11, 2081–2090. [Google Scholar] [CrossRef][Green Version]
- Xiao, J.; Lu, R.; Shen, X.; Wu, M. Green tea extracts protected against carbon tetrachloride-induced chronic liver damage and cirrhosis. Chin. J. Prev. Med. 2002, 36, 243–246. [Google Scholar]
- Li, Y.-M.; Zhang, X.-G.; Zhou, H.-L.; Chen, S.-H.; Zhang, Y.; Yu, C.-H. Effects of tea polyphenols on hepatic fibrosis in rats with alcoholic liver disease. Hepatobil. Pancreat. Dis. Int. 2004, 3, 577–579. [Google Scholar]
- Thomson, M.; Al-Qattan, K.; Mansour, M.H.; Ali, A. Green tea attenuates oxidative stress and downregulates the expression of angiotensin II AT1 receptor in renal and hepatic tissues of streptozotocin-induced diabetic rats. Evid.-Based Complement. Altern. Med. 2012, 2012, 409047. [Google Scholar] [CrossRef]
- Gennaro, G.; Claudino, M.; Cestari, T.M.; Ceolin, D.; Germino, P.; Garlet, G.P.; de Assis, G.F. Green tea modulates cytokine expression in the periodontium and attenuates alveolar bone resorption in type 1 diabetic rats. PLoS ONE 2015, 10, e0134784. [Google Scholar] [CrossRef] [PubMed]
- Ko, C.H.; Lau, K.M.; Choy, W.Y.; Leung, P.C. Effects of tea catechins, epigallocatechin, gallocatechin, and gallocatechin gallate, on bone metabolism. J. Agric. Food Chem. 2009, 57, 7293–7297. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shen, X.P. Effect of rosiglitazone on inflammatory cytokines and oxidative stress after intensive insulin therapy in patients with newly diagnosed type 2 diabetes. Diabetol. Metab. Syndr. 2019, 11, 35. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; Igarashi, M.; Yamada, S.; Takahashi, N.; Watanabe, K. Inhibitory effect of black tea and its combination with acarbose on small intestinal α-glucosidase activity. J. Ethnopharmacol. 2015, 161, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.J.; Xu, P.; Wang, Y.F.; Wang, Y.Q.; Hochstetter, D. Combined Effects of Green Tea Extracts, Green Tea Polyphenols or Epigallocatechin Gallate with Acarbose on Inhibition against α-Amylase and alpha-Glucosidase in Vitro. Molecules 2013, 18, 11614–11623. [Google Scholar] [CrossRef]
- Huang, H.; Jin, C.Y.; Bi, X.K.; Zhao, Y.B.; Xu, S.J.; Wang, M.H.; Yu, L.; Sun, Y.X.; Hu, D. Green tea polyphenol epigallocatechin-3-gallate promotes reendothelialization in carotid artery of diabetic rabbits by reactivating Akt/eNOS pathway. Front. Pharmacol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.S.; Liou, S.Y.; Kuo, C.H.; Pan, L.F.; Yeh, Y.L.; Liou, J.; Padma, V.V.; Yao, C.H.; Kuo, W.W.; Huang, C.Y. Green tea epigallocatechin gallate enhances cardiac function restoration through survival signaling expression in diabetes mellitus rats with autologous adipose tissue-derived stem cells. J. Appl. Physiol. 2017, 123, 1081–1091. [Google Scholar] [CrossRef]
- Itoh, T.; Imano, M.; Nishida, S.; Tsubaki, M.; Hashimoto, S.; Ito, A.; Satou, T. (-)-Epigallocatechin-3-gallate protects against neuronal cell death and improves cerebral eunction after traumatic brain injury in rats. Neuromol. Med. 2011, 13, 300–309. [Google Scholar] [CrossRef]
- Tian, C.; Ye, X.L.; Zhang, R.; Long, J.; Ren, W.Y.; Ding, S.B.; Liao, D.; Jin, X.; Wu, H.M.; Xu, S.Q.; et al. Green tea polyphenols reduced fat deposits in high fat-fed rats via erk1/2-PPAR gamma-adiponectin pathway. PLoS ONE 2013, 8, e53796. [Google Scholar] [CrossRef]
- Babu, P.V.A.; Sabitha, K.E.; Shyamaladevi, C.S. Green tea extract impedes dyslipidaemia and development of cardiac dysfunction in streptozotocin-diabetic rats. Clin. Exp. Pharmacol. Physiol. 2006, 33, 1184–1189. [Google Scholar] [CrossRef]
- Abolfathi, A.A.; Mohajeri, D.; Rezaie, A.; Nazeri, M. Protective effects of green tea extract against hepatic tissue injury in streptozotocin-induced diabetic rats. Evid.-Based Complement. Altern. Med. 2012, 2012, 740671. [Google Scholar] [CrossRef] [PubMed]
- Renno, W.M.; Alkhalaf, M.; Afsari, Z.; Abd-El-Basset, E.; Mousa, A. Consumption of green tea alters glial fibriliary acidic protein immunoreactivity in the spinal cord astrocytes of STZ-diabetic rats. Nutr. Neurosci. 2008, 11, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Manikandan, R.; Sundaram, R.; Thiagarajan, R.; Sivakumar, M.R.; Meiyalagan, V.; Arumugam, M. Effect of black tea on histological and immunohistochemical changes in pancreatic tissues of normal and streptozotocin-induced diabetic mice (Mus musculus). Microsc. Res. Tech. 2009, 72, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Rizvi, S.I. Black tea extract improves anti-oxidant profile in experimental diabetic rats. Arch. Physiol. Biochem. 2015, 121, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Stiuso, P.; Campiglia, P.; Novellino, E. In vitro hypoglycaemic and hypolipidemic potential of white tea polyphenols. Food Chem. 2013, 141, 2379–2384. [Google Scholar] [CrossRef] [PubMed]
- Dias, T.R.; Alves, M.G.; Rato, L.; Casal, S.; Silva, B.M.; Oliveira, P.F. White tea intake prevents prediabetes-induced metabolic dysfunctions in testis and epididymis preserving sperm quality. J. Nutr. Biochem. 2016, 37, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.F.; Chen, S.H.; Chen, H.; Wang, Y.F.; Wang, Y.Q.; Hochstetter, D.; Xu, P. Studies on the bioactivity of aqueous extract of pu-erh tea and its fractions: In vitro antioxidant activity and alpha-glycosidase inhibitory property, and their effect on postprandial hyperglycemia in diabetic mice. Food Chem. Toxicol. 2013, 53, 75–83. [Google Scholar] [CrossRef]
- Xu, P.; Chen, H.; Wang, Y.Q.; Hochstetter, D.; Zhou, T.; Wang, Y.F. Oral administration of pu-erh tea polysaccharides lowers blood glucose levels and enhances antioxidant status in alloxan-induced diabetic mice. J. Food Sci. 2012, 77, H246–H252. [Google Scholar] [CrossRef]
- Yuqiong, C.; Zhi, Y.U.; Yun, Z.; Dejiang, N.I.; Bijun, X.I.E.; Jirong, Z. Influence of oolong tea polysaccharides on immunomodulatory function of diabetic mice and rats. Acta Nutr. Sin. 2006, 28, 156–159. [Google Scholar]
- Song, E.K.; Hur, H.; Han, M.K. Epigallocatechin gallate prevents autoimmune diabetes induced by multiple low doses of streptozotocin in mice. Arch. Pharm. Res. 2003, 26, 559–563. [Google Scholar] [CrossRef]
- Rhee, S.J.; Kim, M.J.; Kwag, O.G. Effects of green tea catechin on prostaglandin synthesis of renal glomerular and renal dysfunction in streptozotocin-induced diabetic rats. Asia Pac. J. Clin. Nutr. 2002, 11, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.J.; Choi, J.H.; Park, M.R. Green tea catechin improves microsomal phospholipase A2 activity and the arachidonic acid cascade system in the kidney of diabetic rats. Asia Pac. J. Clin. Nutr. 2002, 11, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Nogueira de Amorim, L.M.N.; Vaz, S.R.; Cesario, G.; Coelho, A.S.G.; Botelho, P.B. Effect of green tea extract on bone mass and body composition in individuals with diabetes. J. Funct. Foods 2018, 40, 589–594. [Google Scholar] [CrossRef]
- Mirzaei, K.; Hossein-Nezhad, A.; Karimi, M.; Hosseinzadeh-Attar, M.J.; Jafari, N.; Najmafshar, A.; Larijani, B. Effect of green tea extract on bone turnover markers in type 2 diabetic patients; a double-blind, placebo-controlled clinical trial study. DARU J. Pharm. Sci. 2009, 17, 38–44. [Google Scholar]
- Mousavi, A.; Vafa, M.; Neyestani, T.; Khamseh, M.; Hoseini, F. The effects of green tea consumption on metabolic and anthropometric indices in patients with Type 2 diabetes. J. Res. Med. Sci. 2013, 18, 1080–1086. [Google Scholar] [PubMed]
- Liu, C.Y.; Huang, C.J.; Huang, L.H.; Chen, I.J.; Chiu, J.P.; Hsu, C.H. Effects of green tea extract on insulin resistance and glucagon-like peptide 1 in patients with type 2 diabetes and lipid abnormalities: A randomized, double-blinded, and placebo-controlled trial. PLoS ONE 2014, 9, e91163. [Google Scholar] [CrossRef] [PubMed]
- Hosoda, K.; Wang, M.F.; Liao, M.L.; Chuang, C.K.; Iha, M.; Clevidence, B.; Yamamoto, S. Antihyperglycentic effect of oolong tea in type 2 diabetes. Diabetes Care 2003, 26, 1714–1718. [Google Scholar] [CrossRef] [PubMed]
- Vaz, S.R.; de Amorim, L.M.N.; de Nascimento, P.V.F.; Veloso, V.S.P.; Nogueira, M.S.; Castro, I.A.; Mota, J.F.; Botelho, P.B. Effects of green tea extract on oxidative stress and renal function in diabetic individuals: A randomized, double-blinded, controlled trial. J. Funct. Foods 2018, 46, 195–201. [Google Scholar] [CrossRef]
- MacKenzie, T.; Leary, L.; Brooks, W.B. The effect of an extract of green and black tea on glucose control in adults with type 2 diabetes mellitus: Double-blind randomized study. Metabolism 2007, 56, 1340–1344. [Google Scholar] [CrossRef]
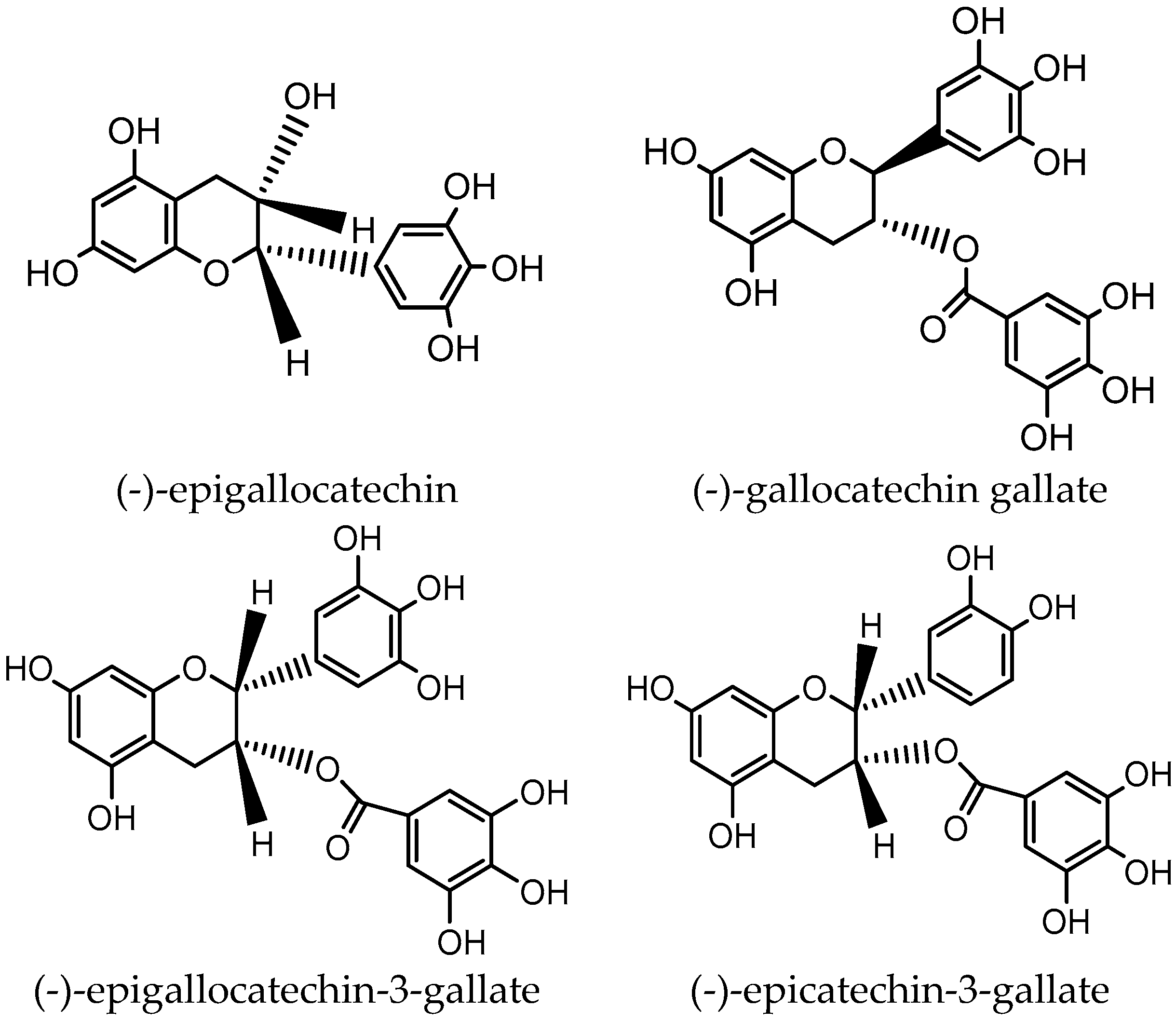
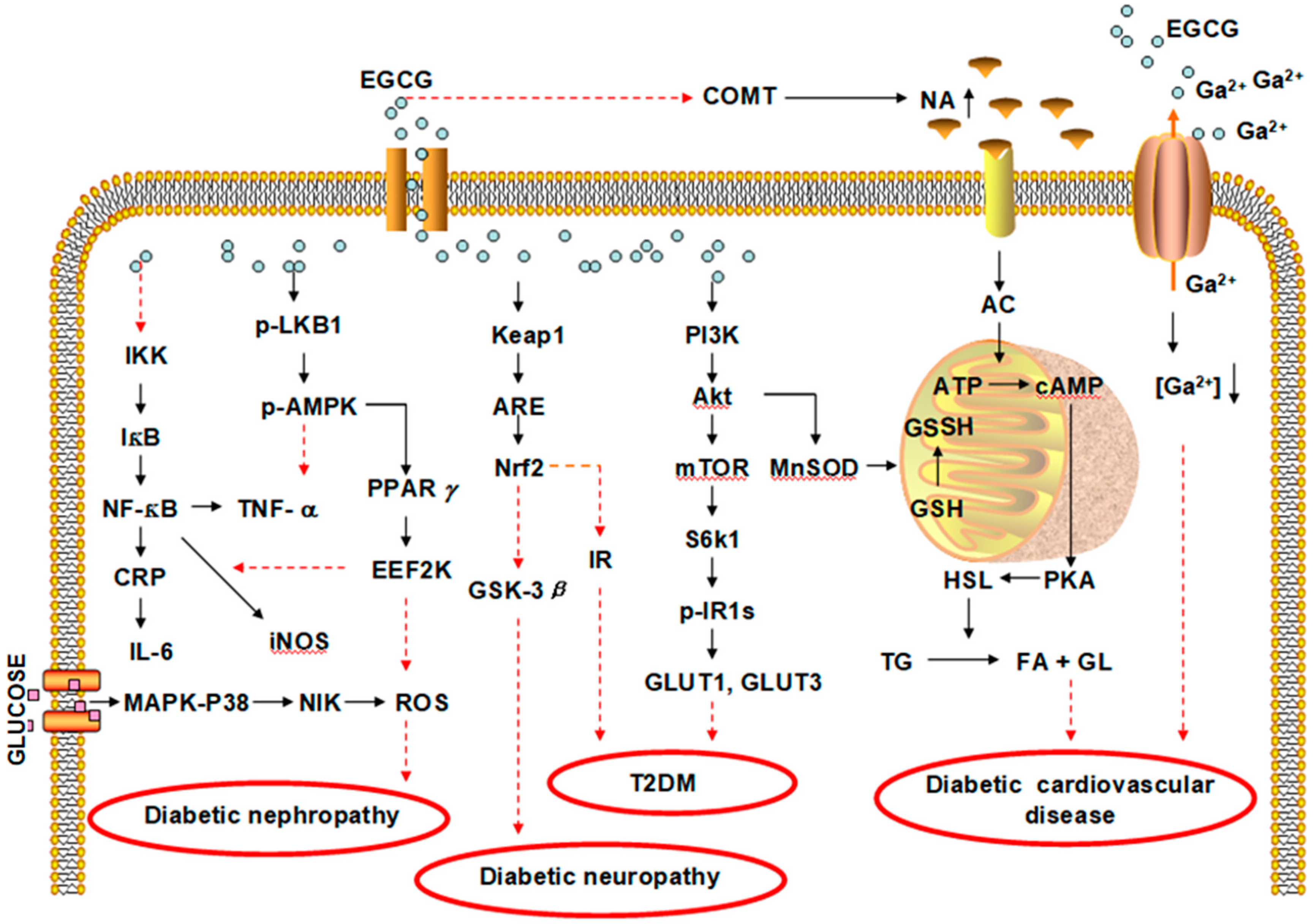
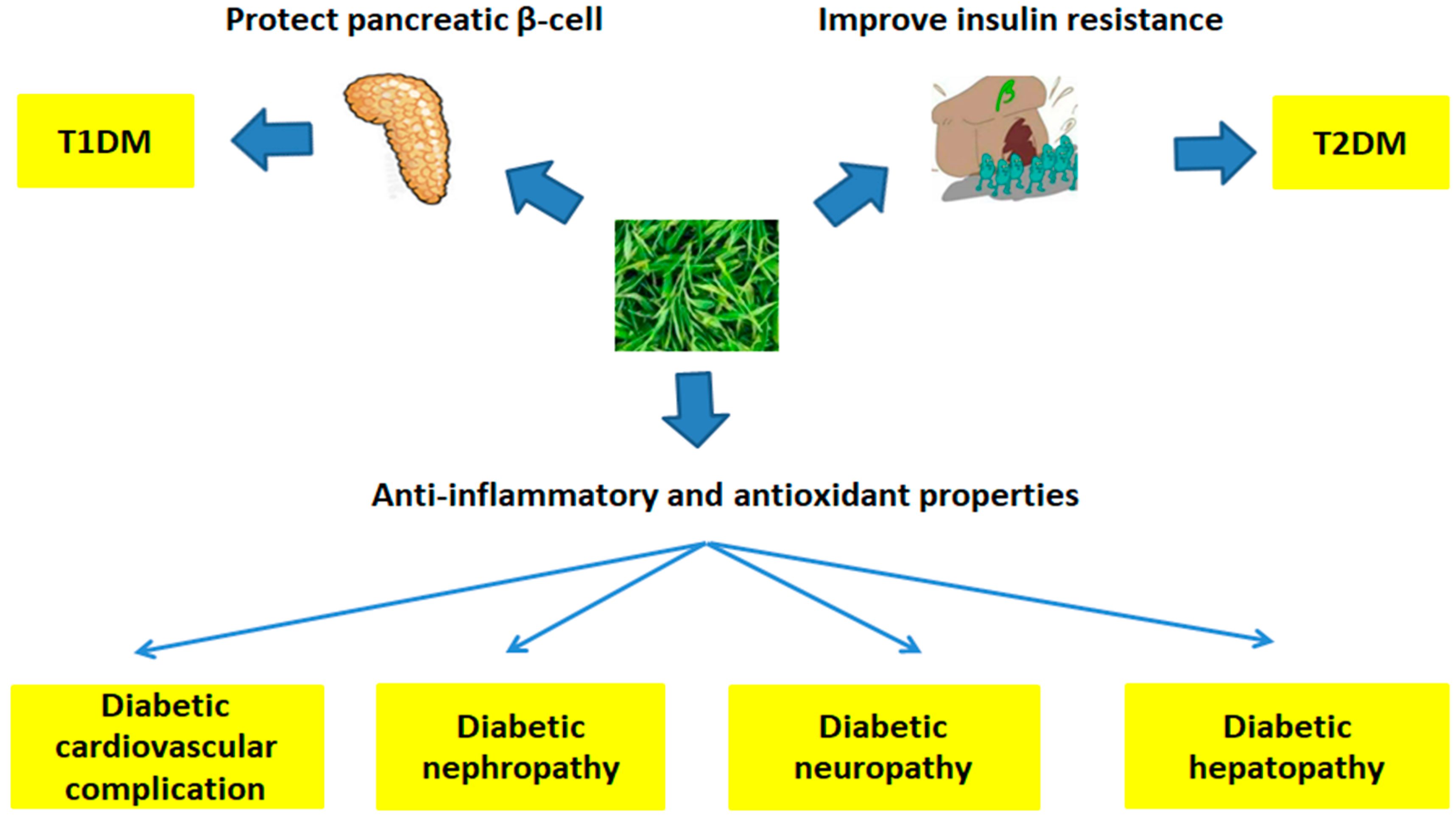
| Diseases | Tea Type | Study Type | Participants | Dose | Results | Ref. |
|---|---|---|---|---|---|---|
| Diabetes mellitus | Tea | Prospective cohort study | Individuals (N = 7006) aged 32–88 without diabetes mellitus | N/A | The consumption of tea showed an decreased risk of diabetes mellitus for nonelderly adults who had previously lost weight. | [24] |
| Diabetes mellitus | Green tea | Cohort study | Elderly Japanese men and women (N = 11,717) | N/A | Women with a higher intake of green tea had a lower risk of diabetes mellitus. | [26] |
| Diabetes mellitus | Black Tea | placebo-controlled study | Total participants (N = 24) aged 20–60 | N/A | Drinking black tea could decrease postprandial blood sugar. | [39] |
| T2DM | Tea | Population-based cohort study | Danish non-diabetic women with singleton pregnancies (N = 71,239) | 8 cups per day | The consumption of tea showed protective effects against T2DM (RR = 0.55, 95% CI (0.55, 1.08)). | [23] |
| T2DM | Tea | Prospective Cohort study. | African American women (N = 46,906) | N/A | The consumption of tea wasn’t associated with T2DM. | [19] |
| T2DM | Tea | Prospective cohort study. | British men (N = 4055) and women (N = 1768) | N/A | Tea intake was beneficial for DM (HR = 0.66, 95% CI (0.61, 1.22)), p < 0.05. | [25] |
| T2DM | Tea | Case-Control study | Newly diagnosed diabetic cases (N = 599) Hospital-based controls (N = 599) | 2 cups per day | Habitual drinking tea could reduce the risk of T2DM (OR = 0.66, 95% CI (0.49–0.89)). | [29] |
| T2DM | Tea | Case-Cohort Study | Total participants (N = 16,835) | ≥1 cups per day | The consumption of tea has negative relation with T2DM 1 cup/day (HR = 0.84, 95% CI (0.71, 1.00)) ≥2 cups/day (HR = 0.93, 95% CI (0.81, 1.05)) | [40] |
| T2DM | Tea | Meta-analysis | Total participants (N = 545,517); Cases with T2DM (N = 37,445) | N/A | The consumption of tea has negative relation with T2DM (p = 0.02). | [34] |
| T2DM | Tea | Meta-analysis | N/A | N/A | Drinking tea daily (≥3 cups/day) is associated with a lower risk of T2DM (RR = 0.84, 95% CI (0.73, 0.97)) | [35] |
| T2DM | Tea | Meta-analysis | Total participants (N = 324,141); Cases with T2DM (N = 11,400) | N/A | Tea consumption a ≥4 cups per day may lower the risk of T2DM. | [18] |
| T2DM | Tea | Meta-analysis | Total participants (N = 457,922) | N/A | The consumption of tea was associated with reduced risk of diabetes mellitus. | [33] |
| T2DM | Tea | Descriptive study | Total participants (N = 940) | N/A | Long-term tea intake had effects on the prevention and treatment of diabetes mellitus. | [32] |
| T2DM | Green tea | Meta-analysis | N/A | N/A | The consumption of tea wasn’t associated with T2DM. | [38] |
| T2DM | Green tea | Meta-analysis | N/A | N/A | Tea or tea extract could maintain stable fasting insulin level in patients with T2DM. | [6] |
| T2DM | Green tea | Meta-analysis | Total participants (N = 510) | N/A | Green tea had no effect on insulin sensitivity and blood glucose control. | [37] |
| T2DM | Black tea | Cohort study. | Total participants (N = 36,908) | ≥1 cups per day | Black tea had association with T2DM (RR = 0.86, 95% CI (0.74, 1.00)). | [27] |
| T2DM | Oolong tea | Prospective cohort study. | Japanese male workers (N = 4975) | ≥1 cups per day | Long-term consumption of oolong tea may be a predictive factor for new onset diabetes mellitus. 1 cup/day (HR = 1.00, 95% CI (0.67–1.49)). ≥2cups /day (HR = 1.64, 95% CI (1.11–2.40)) | [36] |
| Diabetic nephropathy | Green tea | Cohort study | Diabetic patients (N = 42) | N/A | Green tea extract could reduce proteinuria in diabetic patients. | [41] |
| Diabetic Retinopathy | Green tea | Case-Control Study | Cases with diabetic retinopathy (N = 100) and diabetic patients without retinopathy (N = 100) | N/A | Long-term drinking green tea had preventive effects on diabetic retinopathy (OR = 0.49, 95% CI (0.26–0.90)). | [30] |
| Tea Types | Constituents | Diseases Types | Study Types | Models | Dose | Effects | Mechanisms | Ref. |
|---|---|---|---|---|---|---|---|---|
| Green tea | EGCG | Diabetic cardiovascular disease | In vivo | Alloxan-induced diabetic rabbits | 50 mg/kg/day | Improved late endothelial progenitor cells(L-EPCs); Promoted reendothelialization. | Activated Akt/eNOS pathway | [136] |
| EGCG | Diabetic cardiomyopathy | In vivo | Wistar rats | 50 mg/kg/day | Enhanced cardiac function; Increased ADSC repair capability; | ↑ Insulin-like growth factor 1 ↑ H9c2 cell cycle | [137] | |
| EGCG | diabetic neuropathy | In vivo | Male Wistar rats | 0.1% (w/v) | Improved cerebral function. | ↓ Neuronal degeneration ↓ Apoptotic cell death | [138] | |
| Polyphenols | Diabetic Retinopathy | In vivo | Wistar-Kyoto rats | 5.7 g/kg/day | Protected the retina against glutamate toxicity. | ↓ ROS | [122] | |
| Polyphenols | Diabetic cardiovascular disease | In vivo | Male Wistar rats | 0.8, 1.6, and 3.2 g/L | Reduced fat deposit; Ameliorated hypoadiponectinemia in HF-fed rats; Relieved high glucose-induced adiponectin decrease. | ↓ Extracellular signal regulated kinase 1/2 phosphorylation ↑ PPARγ ↓ Adiponectin decrease | [139] | |
| Polyphenols | Diabetic cardiovascular disease | In vitro | Cardiac muscle of rats | 200 mg/kg | Ameliorated the effects of high-fructose diet on insulin signaling, lipid metabolism and inflammation. | ↑ PI3k, Akt1 ↑ Glut1, Glut4, glycogen synthase 1 ↑ Anti-inflammatory protein ↓ GSK-3β, TNF, IL-1B and IL-6 | [53] | |
| Diabetic cardiovascular disease | In vivo | STZ-induced rats | 300 mg/kg/day | Protected rat heart. | ↓ [Ca2+] and [Na+] ↑ Activities of Ca2+-ATPase and Na+/K+-ATPase | [94] | ||
| Diabetic cardiovascular disease | In vivo | STZ-induced rats | 300 mg/kg/day | Reduced the risk of diabetic cardiovascular disease. | ↓ Cholesterol, triglyceride ↓ Free fatty acid and LDL-C ↑ HDL-C | [140] | ||
| Diabetic cardiomyopathy | In vivo | Diabetic rats | 300 mg/kg/day | Treated diabetic cardiomyopathy. | ↓ AGEs ↓ Ollagen cross-linking | [100] | ||
| diabetic retinopathy | In vivo | Rats | 200 mg/kg/day | Prevented and treated diabetic retinopathy. | ↓ SOD and catalase enzyme | [123] | ||
| Diabetic hepatopathy | In vivo | Male Wistar rats | 1.5% (w/v) | Prevented diabetic tissue injury. | ↑ GSH-Px, SOD, catalase | [126] | ||
| Diabetic hepatopathy | In vivo | Male Wistar rats | 1.5% (w/v) | Pretected tissue. | ↑ GSH-Px, SOD, catalase ↓ MDA, alkaline phosphatase | [141] | ||
| Diabetic nephropathy and hepatopathy | In vivo | Male Sprague-Dawley rats | 0.1% (w/v) | Protected renal and hepatic tissues from injury. | ↑ Total antioxidant levels ↓ Malonyldialdehyde (MDA) ↓ Angiotensin II AT1 receptor | [130] | ||
| Diabetes mellitus-induced periodontitis | In vivo | STZ-induced rats | N/A | Treated diabetes mellitus-induced periodontitis. | ↓ TNF-α and RANKL ↑ RUNX-2, OPG ↑ Interleukin-10 (IL-10) | [131] | ||
| diabetic spinal cord | In vivo | STZ-induced rats | N/A | Improved diabetic spinal cord. | ↑ GFAP | [142] | ||
| Black tea | T1DM | In vivo | Female CD-1 mice | 0.01% (w/v) | Promoted insulin secretion and regenerated damaged pancreas and protected pancreatic β- cells. | ↓ Nitrosative stressRUNX-2, OPG↓ ROS | [20] | |
| Diabetes mellitus | In vivo | STZ-induced rats | 0.5 mL/day | Regenerated damaged pancreas and protected pancreatic β-cells. | ↓ Nitrosative stress | [143] | ||
| T2DM | In vivo | STZ-induced rats | 0.01 mL/g/day | Ameliorated diabetes mellitus associated oxidative stress. | ↑ GSH | [144] | ||
| Diabetic complication | In vivo | Diabetic animals | 50 mg/mL | Attenuated oxidative stress mediated tissue damage. | ↓ DNA fragmentation ↓ Activation of caspase-3 ↑ Oxidative stress related parameters | [108] | ||
| Diabetic tissue injury | In vivo | Adult male Wistar albino rat | 50 and 100 mg/kg/day | Protected the liver | ↑ Cellular antioxidant capacity ↓ Membrane lipid peroxidation ↓ Oxidative stress | [17] | ||
| EGC, GC, GCG | bone metabolism | In vitro | Cultured rat osteoblast-like osteosarcoma cell line UMR-106 | N/A | Improved bone metabolism | ↑ Osteoblast activity ↓ Osteoclast differentiation | [132] | |
| White tea | T2DM | In vivo | Male Sprague-Dawley rats | 0.5% (w/v) | Lowered blood sugar levels. | ↑ Insulin sensitivity ↑ The synthesis of liver glycogen | [62] | |
| Diabetic cardiovascular diseases | In vivo | Male Wistar rats | 0.01 mg/mL | Prevented cardiovascular diseases. | ↑ Insulin sensitivity ↑ Cardiac acetate and alanine contents and protein oxidation | [88] | ||
| Diabetes mellitus | In vitro | human hepatocellular carcinoma (HepG2) cell | 25 mg/mL | Improved glucose and lipid metabolism. | ↓ Glucose uptake and transport | [145] | ||
| Diabetic reproductive dysfunction | In vivo | STZ-induced prediabetic rat model | 10 mg/mL | Improved epididymal sperm motility and restored sperm viability. | ↓ GLUT3 protein ↑ Lactate dehydrogenase ↑ Lactate content. | [146] | ||
| Dark tea | EGCG, ECG | Diabetes mellitus | In vitro | N/A | 50 mg/mL | Treated diabetes mellitus. | ↓ α-glucosidase | [65] |
| TP,TPS | Diabetes mellitus | In vivo | Diabetic rats | 50 mg/kg | Reduced postprandial blood sugar. | ↓ α-glucosidase | [147] | |
| Polysaccharides | T2DM | In vivo | Male ICR mice | 40 mg/kg | Lowered the blood glucose levels and reversed oxidative stress. | ↑ SOD activity ↑ Malondialdehyde contents ↑ GSH-Px | [148] | |
| T2DM | In vivo | Male ICR mice | 1 and 5 mg/kg | Improved insulin resistance. | ↓ α-glucosidase Maintain α-amylase | [66] | ||
| T2DM | In vitro In vivo | HepG2 cells db/db mice | 100, 200, and 400 mg/kg/day | Improved insulin resistance and maintained glucose homeostasis. | ↑ Glucose uptake ↓ Intestinal sucrase, maltase, and porcine pancreatic amylase activity | [5] | ||
| T2DM | In vivo | Male Sprague−Dawley rats | 400 mg/kg/day | Alleviated insulin resistance and chronic kidney disease. | ↓ SIRP-α ↑ PI3K/Akt ↑ Nrf2 expression in kidney ↓ GSK-3β phosphorylation Activated Akt/GLUT4, FoxO1 and mTOR/S6k1 pathways | [69] | ||
| diabetic nephropathy | In vivo | db/db mice and db/m mice | 1 g/kg/day | Attenuated the increases in urinary albumin, serum creatinine, and mesangial matrix. | ↓ AGEs ↓ Receptor for AGE expression in glomeruli ↓ Carbonyl compounds | [73] | ||
| Onloog tea | Polysaccharide | diabetic tissue and kidney | In vivo | STZ-induced diabetic diabetic mice | 50, 100, and 200 mg/kg | Prevented diabetic tissue and kidney diseases. | ↑ SOD and GSH-PX activity ↓ MDA | [87] |
| Polysaccharide | Diabetic immune disease | In vivo | STZ-induced diabetic mice | 100, 300, and 600 mg/kg in mice 50, 100, and 200 mg/kg in rats | Improved immunomodulatory function. | ↑ The activity of NK cellsIntensify DTH ↑ Phagocytotic function of peritoneal macrophage | [149] | |
| Yellow tea | EGCGGCG | Diabetes mellitus | In vitro | N/A | 1% (w/v) | CGC reduced postprandial blood sugar more effectively. | ↓ α-glucosidase | [71] |
| Diabetic complications | In vivo | db/db mice | N/A | Lowered the serum total and low-density lipoprotein cholesterol and triglyceride levels. Increased glucose tolerance. | ↓ The lipid synthesis ↓ SRET fator1, SREP 1 ↓ Acetyl-CoA carboxylase α, ↓ Fatty acid synthase | [15] | ||
| Tea | EGCG | T1DM | In vitro | RINm5F cells | 20-40 uM | Protected pro-inflammatory cytokine and induced injuries in insulin-producing cells. | ↓ iNOS and NO | [47] |
| T1DM | in vivo | C57BL/KsJ mice | 100 mg/kg/day | Protected pancreatic islets. | ↓ iNOS | [150] | ||
| EGCG | T2DM | In vivo | Diabetic patients | 300, 600, and 900 mg/day | Decreased pathogenesis of proinflammation and improved diabetes mellitus. | ↓ Free radicals ↓ S100A12-RAGE axis by stimulating sRAGE | [57] | |
| Catechins | T2DM | In vivo In vitro | Male obese KK-ay and C57BL/6J mice; 3T3-L1 adipocytes | 20 mg/kg/day | Decreased glucose levels and increased glucose tolerance in animals. | ↓ ROS ↓ JNK phosphorylation ↑ GLUT-4 translocation | [48] | |
| EGCG | T2DM | In vitro | Human HepG2 cells | N/A | Attenuated insulin signaling blockade. | ↓ Phosphorylation of IRS-1 ↑ 5′AMPK | [52] | |
| EGCG | T2DM | In vivo | Sprague-Dawley rats | 1-100 uM | Improved endothelial dysfunction and insulin resistance and protected against myocardial I/R injury. | ↑ NO via PI3k pathway ↑ Plasma adiponectin | [95] | |
| diabetic nephropathy | In vivo | Diabetic SHR rats | 5.7 g/kg/day | Reduced podocyte apoptosis, foot process effacement and albuminuria. | ↓ GSK3-p53 ↑ LRP6 | [78] | ||
| diabetic nephropathy | In vivo | STZ-induced diabetic rats | 5% (w/v) | Improved diabetic nephropathy. | ↓ MMP-9, TIMP-1 ↑ MMP-2 ,TIMP-2 | [83] | ||
| diabetic nephropathy | In vivo | Male Sprague-Dawley rats | 0.25% and 0.5% (w/w) | Reduced renal oxidative damage and inflammatory reactions. | ↑ Activity of 5′-lipoxygenase ↓ Ieukotriene B-4 | [81] | ||
| Catechins | diabetic nephropathy | In vivo | Sprague-Dawley rats | 0.25% and 0.5% (w/w) | Improved kidney function. | ↓ Thromboxane A(2) synthesis ↑ Prostacyclin synthesis | [151,152] |
| Tea Types | Diseases Types | Study Types | Participants | Dose and Duration | Results | Ref. |
|---|---|---|---|---|---|---|
| Green tea | Diabetes mellitus | RCT | Patients with T2DM (N = 63) | 0, 2, 4 cups per day | ↓ Body weight, body mass index, waist circumference and systolic blood pressure. | [155] |
| Green tea | Diabetes mellitus and diabetic nephropathy | RCT | Patients with diabetes mellitus (N = 60) | 2 capsules containing 1120 mg polyphenols per day for 20 weeks. | No significant effect on diabetes mellitus and diabetic nephropathy. | [158] |
| Green tea | T2DM and diabetic cardiomyopathy | RCT | Subjects with T2DM and lipid abnormalities (N = 92) | 500 mg per day | ↓ Triglyceride ↑ High density lipoprotein cholesterol ↑ Glucagon-like peptide 1 in the therapeutic arm | [156] |
| Green tea | Diabetic osteoporosis | RCT | Patients with diabetes mellitus (N = 35) | 1120 mg polyphenols per day | ↑ Bone mineral content ↓ PTH | [153] |
| Green tea | Bone turnover induced by diabetes mellitus | RCT | Patients with T2DM (N = 72) | 500 mg per day | ↓ Fasting serum osteocalcin ↓ FBG ↓ HbA1C | [154] |
| Black tea | T2DM and diabetic cardiovascular | N/A | Patients with T2DM (N = 46) | 150, 300, 450, and 600 mL black tea during the weeks 1, 2, 3 and 4. | ↓ Serum malondialdehyde ↓ Serum C-reactive protein ↑ Glutathione | [58] |
| Oolong tea | T2DM | N/A | Patients with T2DM | 1500 mL per day | ↓ Concentrations of plasma glucose and fructosamine | [73] |
| Green and black tea | T2DM | RCT | White persons (N = 49) | 0, 375, or 750 mg per day for 3 months | No significant effect on T2DM. | [159] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, J.-M.; Cao, S.-Y.; Wei, X.-L.; Gan, R.-Y.; Wang, Y.-F.; Cai, S.-X.; Xu, X.-Y.; Zhang, P.-Z.; Li, H.-B. Effects and Mechanisms of Tea for the Prevention and Management of Diabetes Mellitus and Diabetic Complications: An Updated Review. Antioxidants 2019, 8, 170. https://doi.org/10.3390/antiox8060170
Meng J-M, Cao S-Y, Wei X-L, Gan R-Y, Wang Y-F, Cai S-X, Xu X-Y, Zhang P-Z, Li H-B. Effects and Mechanisms of Tea for the Prevention and Management of Diabetes Mellitus and Diabetic Complications: An Updated Review. Antioxidants. 2019; 8(6):170. https://doi.org/10.3390/antiox8060170
Chicago/Turabian StyleMeng, Jin-Ming, Shi-Yu Cao, Xin-Lin Wei, Ren-You Gan, Yuan-Feng Wang, Shu-Xian Cai, Xiao-Yu Xu, Pang-Zhen Zhang, and Hua-Bin Li. 2019. "Effects and Mechanisms of Tea for the Prevention and Management of Diabetes Mellitus and Diabetic Complications: An Updated Review" Antioxidants 8, no. 6: 170. https://doi.org/10.3390/antiox8060170
APA StyleMeng, J.-M., Cao, S.-Y., Wei, X.-L., Gan, R.-Y., Wang, Y.-F., Cai, S.-X., Xu, X.-Y., Zhang, P.-Z., & Li, H.-B. (2019). Effects and Mechanisms of Tea for the Prevention and Management of Diabetes Mellitus and Diabetic Complications: An Updated Review. Antioxidants, 8(6), 170. https://doi.org/10.3390/antiox8060170






