Effect of the Phosphodiesterase 5 Inhibitor Sildenafil on Ischemia-Reperfusion-Induced Muscle Mitochondrial Dysfunction and Oxidative Stress
Abstract
1. Introduction
2. Material and Methods
2.1. Animals
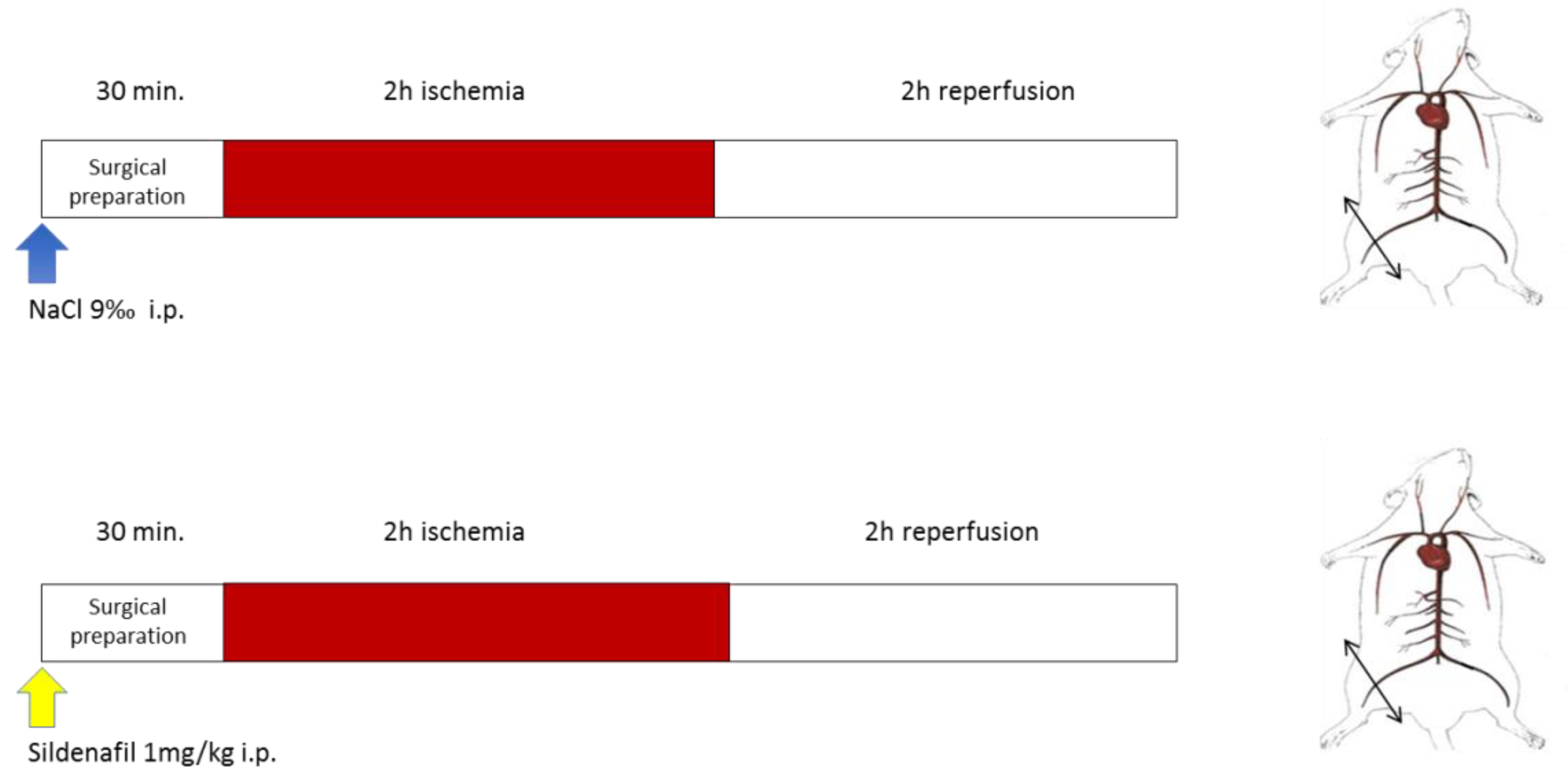
2.2. Experimental Design
2.3. Mitochondrial Respiratory Chain Complex Activities
2.4. Calcium Retention Capacity (CRC) Measurements in Gastrocnemius Ghost Fibers
2.5. Production of Reactive Oxygen Species Using Electron Paramagnetic Resonance
3. Statistical Analysis
4. Results
4.1. Lower-Limb Ischemia-Reperfusion Impaired Skeletal Muscle Mitochondrial Respiration and Calcium Retention Capacity and Tended to Increase ROS Production
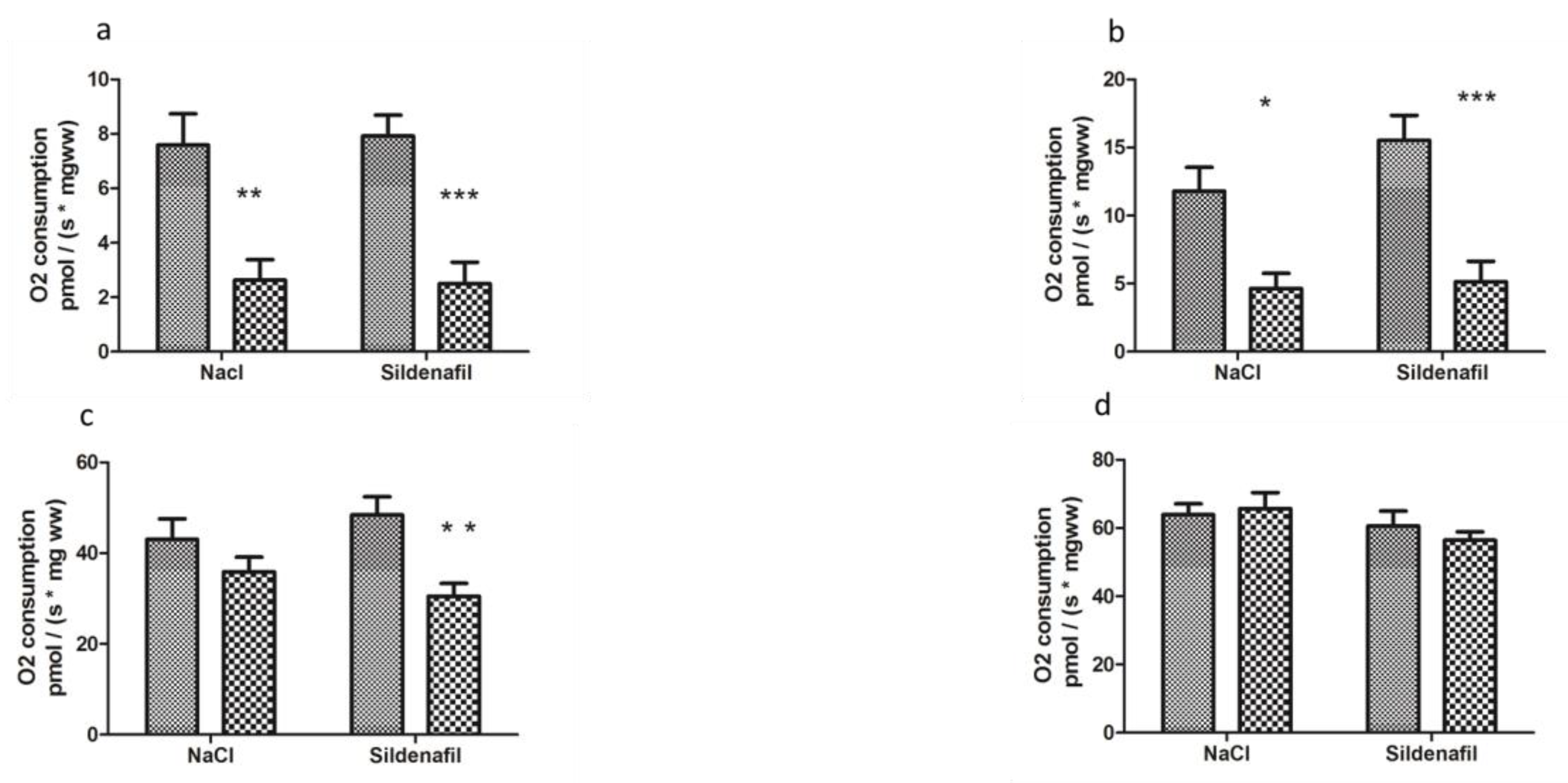
4.1.1. Mitochondrial Respiration
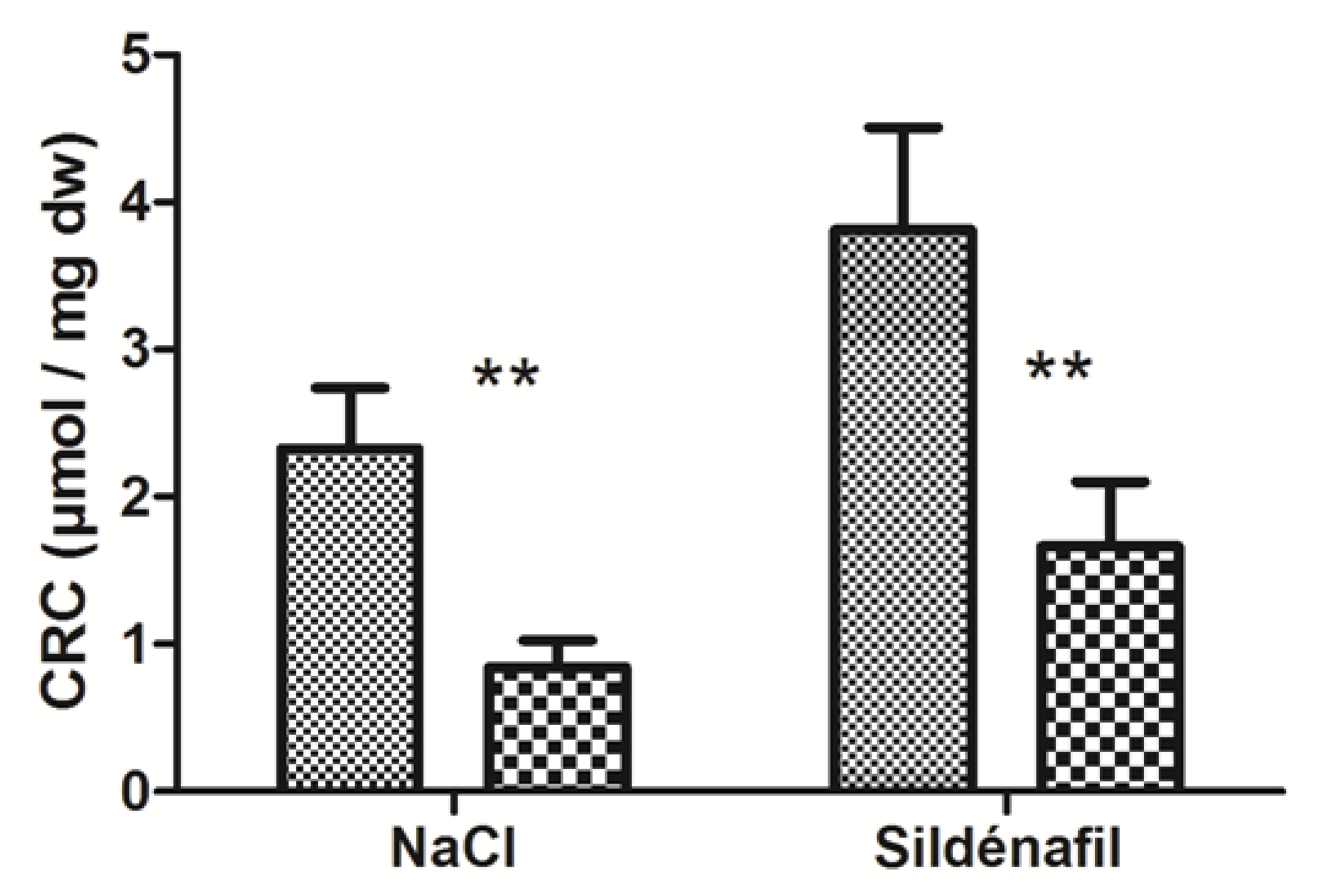
4.1.2. Calcium Retention Capacity (CRC)
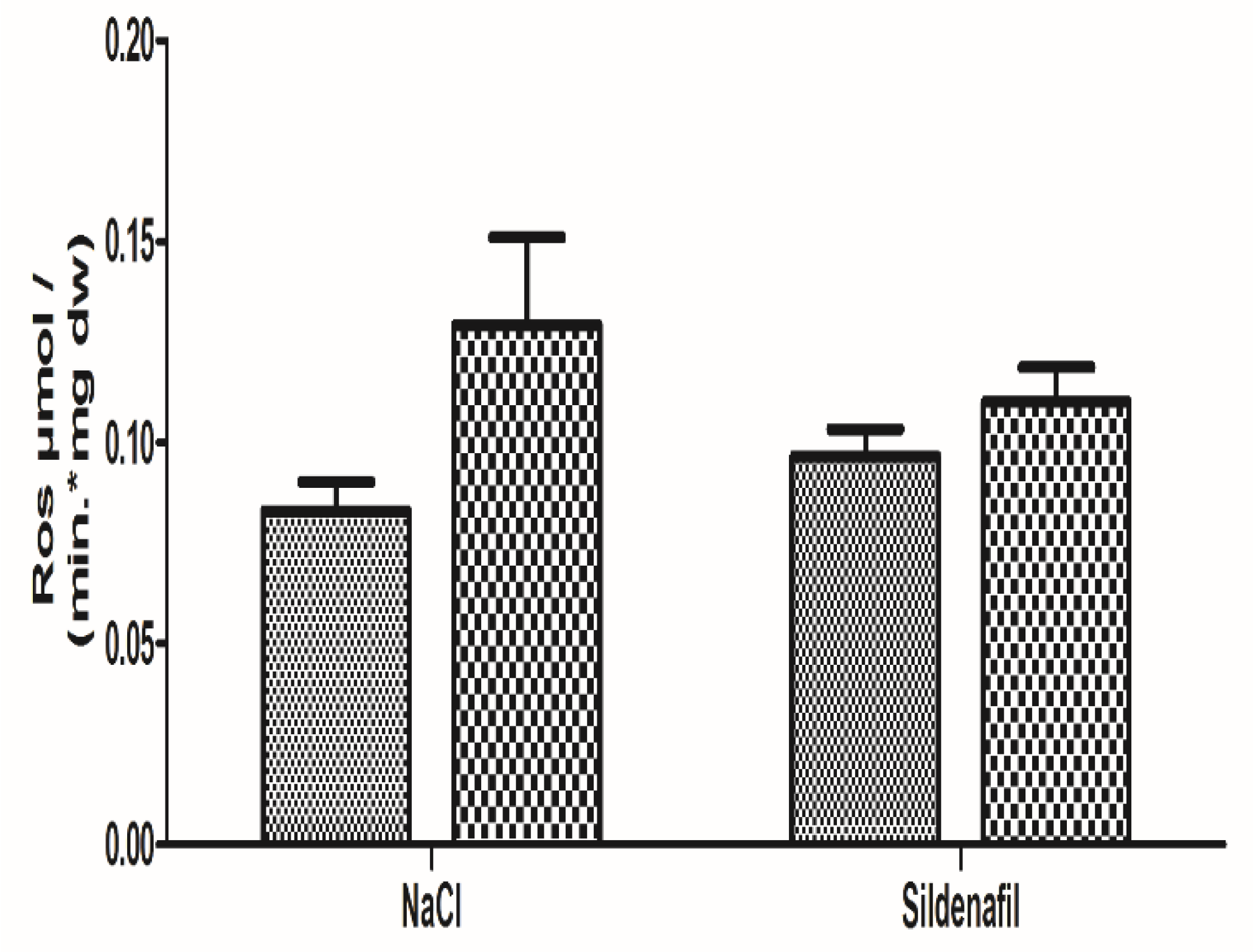
4.1.3. Oxidative Stress
4.2. Sildenafil Did Not Protect Skeletal Muscle Mitochondrial Respiration and Calcium Retention Capacity, Albeit Tending to Reduce ROS Production
4.2.1. Mitochondrial Respiration
4.2.2. Calcium Retention Capacity (CRC)
4.2.3. Oxidative Stress
5. Discussion
5.1. Effects of Ischemia-Reperfusion
5.2. Effects of Sildenafil
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Criqui, M.H.; Aboyans, V. Epidemiology of peripheral artery disease. Circ. Res. 2015, 116, 1509–1526. [Google Scholar] [CrossRef] [PubMed]
- Aboyans, V.; Ricco, J.B.; Bartelink, M.E.L.; Björck, M.; Brodmann, M.; Cohnert, T.; Collet, J.P.; Czerny, M.; De Carlo, M.; Debus, S.; et al. 2017 ESC guidelines on the diagnosis and treatment of peripheral arterial diseases, in collaboration with the european society for vascular surgery (ESVS). Eur. J. Vasc. Endovasc. Surg. 2018, 55, 305–368. [Google Scholar] [CrossRef] [PubMed]
- Dua, A.; Lee, C.J. Epidemiology of peripheral arterial disease and critical limb ischemia. Tech. Vasc. Interv. Radiol. 2016, 19, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Sud, K.; Shishehbor, M.H. Nationwide trends of hospital admission and outcomes among critical limb ischemia patients: From 2003–2011. J. Am. Coll. Cardiol. 2016, 67, 1901–1913. [Google Scholar] [CrossRef] [PubMed]
- Uccioli, L.; Meloni, M.; Izzo, V.; Giurato, L.; Merolla, S.; Gandini, R. Critical limb ischemia: Current challenges and future prospects. Vasc. Health Risk Manag. 2018, 14, 63–74. [Google Scholar] [CrossRef]
- Kropman, R.H.J.; Schrijver, A.M.; Kelder, J.C.; Moll, F.L.; de Vries, J.P.P.M. Clinical outcome of acute leg ischaemia due to thrombosed popliteal artery aneurysm: Systematic review of 895 cases. Eur. J. Vasc. Endovasc. Surg. 2010, 39, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Ismaeel, A.; Brumberg, R.S.; Kirk, J.S.; Papoutsi, E.; Farmer, P.J.; Bohannon, W.T.; Smith, R.S.; Eidson, J.L.; Sawicki, I.; Koutakis, P. Oxidative stress and arterial dysfunction in peripheral artery disease. Antioxidants 2018, 7, 145. [Google Scholar] [CrossRef]
- Duteil, D.; Chambon, C.; Ali, F.; Malivindi, R.; Zoll, J.; Kato, S.; Geny, B.; Chambon, P.; Metzger, D. The transcriptional coregulators TIF2 and SRC-1 regulate energy homeostasis by modulating mitochondrial respiration in skeletal muscles. Cell Metab. 2010, 12, 496–508. [Google Scholar] [CrossRef] [PubMed]
- Lejay, A.; Meyer, A.; Schlagowski, A.I.; Charles, A.L.; Singh, F.; Bouitbir, J.; Pottecher, J.; Chakfé, N.; Zoll, J.; Geny, B. Mitochondria: Mitochondrial participation in ischemia-reperfusion injury in skeletal muscle. Int. J. Biochem. Cell Biol. 2014, 50, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Paradis, S.; Charles, A.L.; Meyer, A.; Lejay, A.; Scholey, J.W.; Chakfé, N.; Zoll, J.; Geny, B. Chronology of mitochondrial and cellular events during skeletal muscle ischemia-reperfusion. Am. J. Physiol. Cell Physiol. 2016, 310, 968–982. [Google Scholar] [CrossRef]
- Guillot, M.; Charles, A.L.; Chamaraux-Tran, T.N.; Bouitbir, J.; Meyer, A.; Zoll, J.; Schneider, F.; Geny, B. Oxidative stress precedes skeletal muscle mitochondrial dysfunction during experimental aortic cross-clamping but is not associated with early lung, heart, brain, liver, or kidney mitochondrial impairment. J Vasc. Surg. 2014, 60, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Ali, Z.A.; Callaghan, C.J.; Lim, E.; Ali, A.A.; Nouraei, S.A.; Akthar, A.M.; Boyle, J.R.; Varty, K.; Kharbanda, R.K.; Dutka, D.P.; et al. Remote ischemic preconditioning reduces myocardial and renal injury after elective abdominal aortic aneurysm repair: A randomized controlled trial. Circulation 2007, 116, 98–105. [Google Scholar] [CrossRef]
- Mansour, Z.; Bouitbir, J.; Charles, A.L.; Talha, S.; Kindo, M.; Pottecher, J.; Zoll, J.; Geny, B. Remote and local ischemic preconditioning equivalently protects rat skeletal muscle mitochondrial function during experimental aortic cross-clamping. J. Vasc. Surg. 2012, 55, 497–505. [Google Scholar] [CrossRef][Green Version]
- Charles, A.L.; Guilbert, A.S.; Bouitbir, J.; Goette-Di Marco, P.; Enache, I.; Zoll, J.; Piquard, F.; Geny, B. Effect of postconditioning on mitochondrial dysfunction in experimental aortic cross-clamping. Br. J. Surg. 2011, 98, 511–516. [Google Scholar] [CrossRef]
- Mansour, Z.; Charles, A.L.; Bouitbir, J.; Pottecher, J.; Kindo, M.; Mazzucotelli, J.P.; Zoll, J.; Geny, B. Remote and local ischemic postconditioning further impaired skeletal muscle mitochondrial function after ischemia-reperfusion. J. Vasc. Surg. 2012, 56, 774–782. [Google Scholar] [CrossRef][Green Version]
- Pottecher, J.; Guillot, M.; Belaidi, E.; Charles, A.L.; Lejay, A.; Gharib, A.; Diemunsch, P.; Geny, B. Cyclosporine A normalizes mitochondrial coupling, reactive oxygen species production, and inflammation and partially restores skeletal muscle maximal oxidative capacity in experimental aortic cross-clamping. J. Vasc. Surg. 2013, 57, 1100–1108. [Google Scholar] [CrossRef] [PubMed]
- Pottecher, J.; Kindo, M.; Chamaraux-Tran, T.N.; Charles, A.L.; Lejay, A.; Kemmel, V.; Vogel, T.; Chakfe, N.; Zoll, J.; Diemunsch, P.; et al. Skeletal muscle ischemia-reperfusion injury and cyclosporine A in the aging rat. Fundam. Clin. Pharmacol. 2016, 30, 216–225. [Google Scholar] [CrossRef]
- D’Souza, S.P.; Yellon, D.M.; Martin, C.; Schulz, R.; Heusch, G.; Onody, A.; Ferdinandy, P.; Baxter, G.F. B-type natriuretic peptide limits infarct size in rat isolated hearts via KATP channel opening. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H1592–H1600. [Google Scholar] [CrossRef]
- Talha, S.; Bouitbir, J.; Charles, A.; Zoll, J.; Goette-Di Marco, P.; Meziani, F.; Piquard, F.; Geny, B. Pretreatment with brain natriuretic peptide reduces skeletal muscle mitochondrial dysfunction and oxidative stress after ischemia-reperfusion. J. Appl. Physiol. 2013, 114, 172–179. [Google Scholar] [CrossRef]
- Keravis, T.; Lugnier, C. Cyclic nucleotide phosphodiesterase (PDE) isozymes as targets of the intracellular signalling network: Benefits of PDE inhibitors in various diseases and perspectives for future therapeutic developments. Br. J. Pharmacol. 2012, 165, 1288–1305. [Google Scholar] [CrossRef]
- Maurice, D.H.; Ke, H.; Ahmad, F.; Wang, Y.; Chung, J.; Manganiello, V.C. Advances in targeting cyclic nucleotide phosphodiesterases. Nat. Rev. Drug Discov. 2014, 13, 290–314. [Google Scholar] [CrossRef] [PubMed]
- Tetsi, L.; Charles, A.L.; Paradis, S.; Lejay, A.; Talha, S.; Geny, B.; Lugnier, C. Effects of cyclic nucleotide phosphodiesterases (PDEs) on mitochondrial skeletal muscle functions. Cell. Mol. Life Sci. 2017, 74, 1883–1893. [Google Scholar] [CrossRef]
- Andersson, K.E. PDE5 inhibitors—Pharmacology and clinical applications 20 years after sildenafil discovery. Br. J. Pharmacol. 2018, 75, 2554–2565. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, Q.; Yuan, W.; Wu, J.; Li, C. Sildenafil protects against myocardial ischemia-reperfusion injury following cardiac arrest in a porcine model: Possible role of the renin-angiotensin system. Int. J. Mol. Sci. 2015, 16, 27015–27031. [Google Scholar] [CrossRef]
- Korkmaz-Icöz, S.; Radovits, T.; Szabó, G. Targeting phosphodiesterase 5 as a therapeutic option against myocardial ischaemia/reperfusion injury and for treating heart failure. Br. J. Pharmacol. 2018, 175, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Mohey, V.; Singh, M.; Puri, N.; Kaur, T.; Pathak, D.; Singh, A.P. Sildenafil obviates ischemia-reperfusion injury-induced acute kidney injury through peroxisome proliferator-activated receptor γ agonism in rats. Surg. Res. 2016, 201, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Guerra-Mora, J.R.; Perales-Caldera, E.; Aguilar-León, D.; Nava-Sanchez, C.; Díaz-Cruz, A.; Díaz-Martínez, N.E.; Santillán-Doherty, P.; Torres-Villalobos, G.; Bravo-Reyna, C.C. Effects of sildenafil and tadalafil on edema and reactive oxygen species production in an experimental model of lung ischemia-reperfusion injury. Transplant. Proc. 2017, 49, 1461–1466. [Google Scholar] [CrossRef] [PubMed]
- Savvanis, S.; Nastos, C.; Tasoulis, M.K.; Papoutsidakis, N.; Demonakou, M.; Karmaniolou, I.; Arkadopoulos, N.; Smyrniotis, V.; Theodoraki, K. Sildenafil attenuates hepatocellular injury after liver ischemia reperfusion in rats: A preliminary study. Oxid. Med. Cell. Longev. 2014, 2014, 161942. [Google Scholar] [CrossRef]
- Ozdegirmenci, O.; Kucukozkan, T.; Akdag, E.; Topal, T.; Haberal, A.; Kayir, H.; Oter, S.; Akyol, M.; Uzbay, T. Effects of sildenafil and tadalafil on ischemia/reperfusion injury in fetal rat brain. J. Matern. Fetal Neonatal Med. 2011, 24, 317–323. [Google Scholar] [CrossRef]
- Kulkarni, S.K.; Patil, C.S. Phosphodiesterase 5 enzyme and its inhibitors: Update on pharmacological and therapeutical aspects. Methods Find. Exp. Clin. Pharmacol. 2004, 26, 789–799. [Google Scholar] [CrossRef]
- Dussault, S.; Maingrette, F.; Ménard, C.; Michaud, S.E.; Haddad, P.; Groleau, J.; Turgeon, J.; Perez, G.; Rivard, A. Sildenafil increases endothelial progenitor cell function and improves ischemia-induced neovascularization in hypercholesterolemic apolipoprotein E-deficient mice. Hypertension 2009, 54, 1043–1049. [Google Scholar] [CrossRef]
- Valatsou, A.; Briasoulis, A.; Vogiatzi, G.; Pantopoulou, A.; Oikonomo, E.; Miliou, A.; Perrea, D.; Tousoulis, D. Beneficial effects of sildenafil on tissue perfusion and inflammation in a murine model of limb ischemia and atherosclerosis. Curr. Vasc. Pharmacol. 2017, 15, 282–287. [Google Scholar] [CrossRef]
- Constantinescu, I.M.; Bolfa, P.; Constantinescu, D.; Mironiuc, A.I.; Gherman, C.D. Treatment with sildenafil and donepezil improves angiogenesis in experimentally induced critical limb ischemia. Biomed. Res. Int. 2017, 2017, 9532381. [Google Scholar] [CrossRef] [PubMed]
- Thaveau, F.; Zoll, J.; Rouyer, O.; Chafke, N.; Kretz, J.G.; Piquard, F.; Geny, B. Ischemic preconditioning specifically restores complexes I and II activities of the mitochondrial respiratory chain in ischemic skeletal muscle. J. Vasc. Surg. 2007, 46, 541–547. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Charles, A.L.; Guilbert, A.S.; Guillot, M.; Talha, S.; Lejay, A.; Meyer, A.; Kindo, M.; Wolff, V.; Bouitbir, J.; Zoll, J.; et al. Muscles susceptibility to ischemia-reperfusion injuries depends on fiber type specific antioxidant level. Front. Physiol. 2017, 6, 52. [Google Scholar] [CrossRef] [PubMed]
- Flück, M.; von Allmen, R.S.; Ferrié, C.; Tevaearai, H.; Dick, F. Protective effect of focal adhesion kinase against skeletal muscle reperfusion injury after acute limb ischemia. Eur. J. Vasc. Endovasc. Surg. 2015, 49, 306–313. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tran, T.P.; Tu, H.; Liu, J.; Muelleman, R.L.; Li, Y.-L. Mitochondria-derived superoxide links to tourniquet-induced apoptosis in mouse skeletal muscle. PLoS ONE 2012, 7, e43410. [Google Scholar] [CrossRef][Green Version]
- Miura, S.; Saitoh, S.I.; Kokubun, T.; Owada, T.; Yamauchi, H.; Machii, H.; Takeishi, Y. Mitochondrial-targeted antioxidant maintains blood flow, mitochondrial function, and redox balance in old mice following prolonged limb ischemia. Int. J. Mol. Sci. 2017, 18, 1897. [Google Scholar] [CrossRef]
- Koutakis, P.; Weiss, D.J.; Miserlis, D.; Shostrom, V.K.; Papoutsi, E.; Ha, D.M.; Carpenter, L.A.; McComb, R.D.; Casale, G.P.; Pipinos, I.I. Oxidative damage in the gastrocnemius of patients with peripheral artery disease is myofiber type selective. Redox Biol. 2014, 2, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Frankenreiter, S.; Groneberg, D.; Kuret, A.; Krieg, T.; Ruth, P.; Friebe, A.; Lukowski, R. Cardioprotection by ischemic postconditioning and cyclic guanosine monophosphate-elevating agents involves cardiomyocyte nitric oxide-sensitive guanylyl cyclase. Cardiovasc. Res. 2018, 114, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Veres, G.; Hagenhoff, M.; Schmidt, H.; Radovits, T.; Loganathan, S.; Bai, Y.; Korkmaz-Icöz, S.; Brlecic, P.; Sayour, A.A.; Karck, M.; et al. Targeting phosphodiesterase-5 by vardenafil improves vascular graft function. Eur. J. Vasc. Endovasc. Surg. 2018, 56, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Bloom, T.J. Cyclic nucleotide phosphodiesterase isozymes expressed in mouse skeletal muscle. Can. J. Physiol. Pharmacol. 2002, 80, 1132–1135. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, M.A.; Marques, R.J.; Vicente, J.A.; Santos, M.S.; Monteiro, P.; Moreno, A.J.; Custódio, J.B. Sildenafil citrate concentrations not affecting oxidative phosphorylation depress H2O2 generation by rat heart mitochondria. Mol. Cell. Biochem. 2008, 309, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Aksu, V.; Yüksel, V.; Chousein, S.; Taştekin, E.; İşcan, Ş.; Sağiroğlu, G.; Canbaz, S.; Sunar, H. The effects of sildenafil and n-acetylcysteine on ischemia and reperfusion injury in gastrocnemius muscle and femoral artery endothelium. Vascular 2015, 23, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Karamatsoukis, S.L.; Trigka, E.A.; Stasinopoulou, M.; Stavridou, A.; Zacharioudaki, A.; Tsarea, K.; Karamperi, M.; Pittaras, T.; Papadopoulos, O.; Patsouris, E.; et al. Beneficial Effect of U-74389 G and sildenafil in an experimental model of flap ischemia/reperfusion injury in swine. Histological and biochemical evaluation of the model. J. Investig. Surg. 2018, 30, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.M.; Armstrong Ada, C.; Figueiredo, R.C.; Florentino, J.E.; Saad, P.F.; Fox-Talbot, K.; Halushka, M.K.; Berkowitz, D.E.; Taha, M.O.; Fagundes, D.J. Sildenafil citrate protects skeletal muscle of ischemia-reperfusion injury: Immunohistochemical study in rat model. Acta Cir. Bras. 2013, 28, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Walker, D.K.; Ackland, M.J.; James, G.C.; Muirhead, G.J.; Rance, D.J.; Wastall, P.; Wright, P.A. Pharmacokinetics and metabolism of sildenafil in mouse, rat, rabbit, dog and man. Xenobiotica 1999, 29, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Nio, Y.; Tanaka, M.; Hirozane, Y.; Muraki, Y.; Okawara, M.; Hazama, M.; Matsuo, T. Phosphodiesterase 4 inhibitor and phosphodiesterase 5 inhibitor combination therapy has antifibrotic and anti-inflammatory effects in mdx mice with Duchenne muscular dystrophy. FASEB J. 2017, 31, 5307–5320. [Google Scholar] [CrossRef] [PubMed]
- Broome, S.C.; Woodhead, J.S.T.; Merry, T.L. Mitochondria-targeted antioxidants and skeletal muscle function. Antioxidants 2018, 8, 7. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tetsi, L.; Charles, A.-L.; Georg, I.; Goupilleau, F.; Lejay, A.; Talha, S.; Maumy-Bertrand, M.; Lugnier, C.; Geny, B. Effect of the Phosphodiesterase 5 Inhibitor Sildenafil on Ischemia-Reperfusion-Induced Muscle Mitochondrial Dysfunction and Oxidative Stress. Antioxidants 2019, 8, 93. https://doi.org/10.3390/antiox8040093
Tetsi L, Charles A-L, Georg I, Goupilleau F, Lejay A, Talha S, Maumy-Bertrand M, Lugnier C, Geny B. Effect of the Phosphodiesterase 5 Inhibitor Sildenafil on Ischemia-Reperfusion-Induced Muscle Mitochondrial Dysfunction and Oxidative Stress. Antioxidants. 2019; 8(4):93. https://doi.org/10.3390/antiox8040093
Chicago/Turabian StyleTetsi, Liliane, Anne-Laure Charles, Isabelle Georg, Fabienne Goupilleau, Anne Lejay, Samy Talha, Myriam Maumy-Bertrand, Claire Lugnier, and Bernard Geny. 2019. "Effect of the Phosphodiesterase 5 Inhibitor Sildenafil on Ischemia-Reperfusion-Induced Muscle Mitochondrial Dysfunction and Oxidative Stress" Antioxidants 8, no. 4: 93. https://doi.org/10.3390/antiox8040093
APA StyleTetsi, L., Charles, A.-L., Georg, I., Goupilleau, F., Lejay, A., Talha, S., Maumy-Bertrand, M., Lugnier, C., & Geny, B. (2019). Effect of the Phosphodiesterase 5 Inhibitor Sildenafil on Ischemia-Reperfusion-Induced Muscle Mitochondrial Dysfunction and Oxidative Stress. Antioxidants, 8(4), 93. https://doi.org/10.3390/antiox8040093





