Nutrients for Prevention of Macular Degeneration and Eye-Related Diseases
Abstract
1. Introduction
2. Antioxidant Nutrients
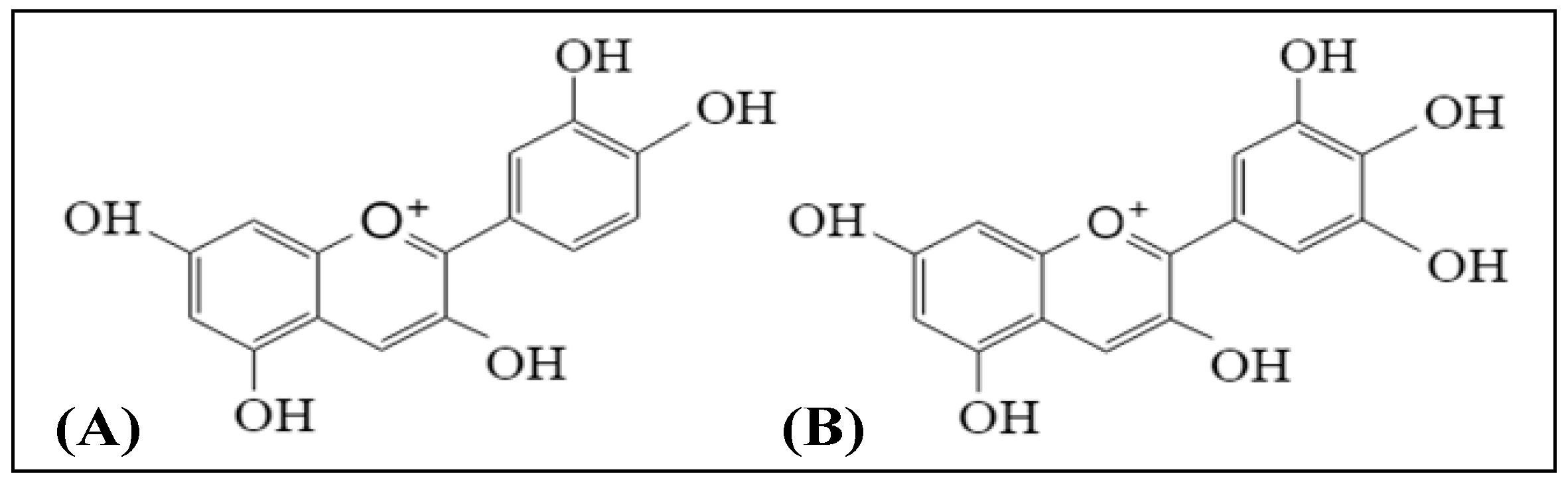
2.1. Anthocyanins
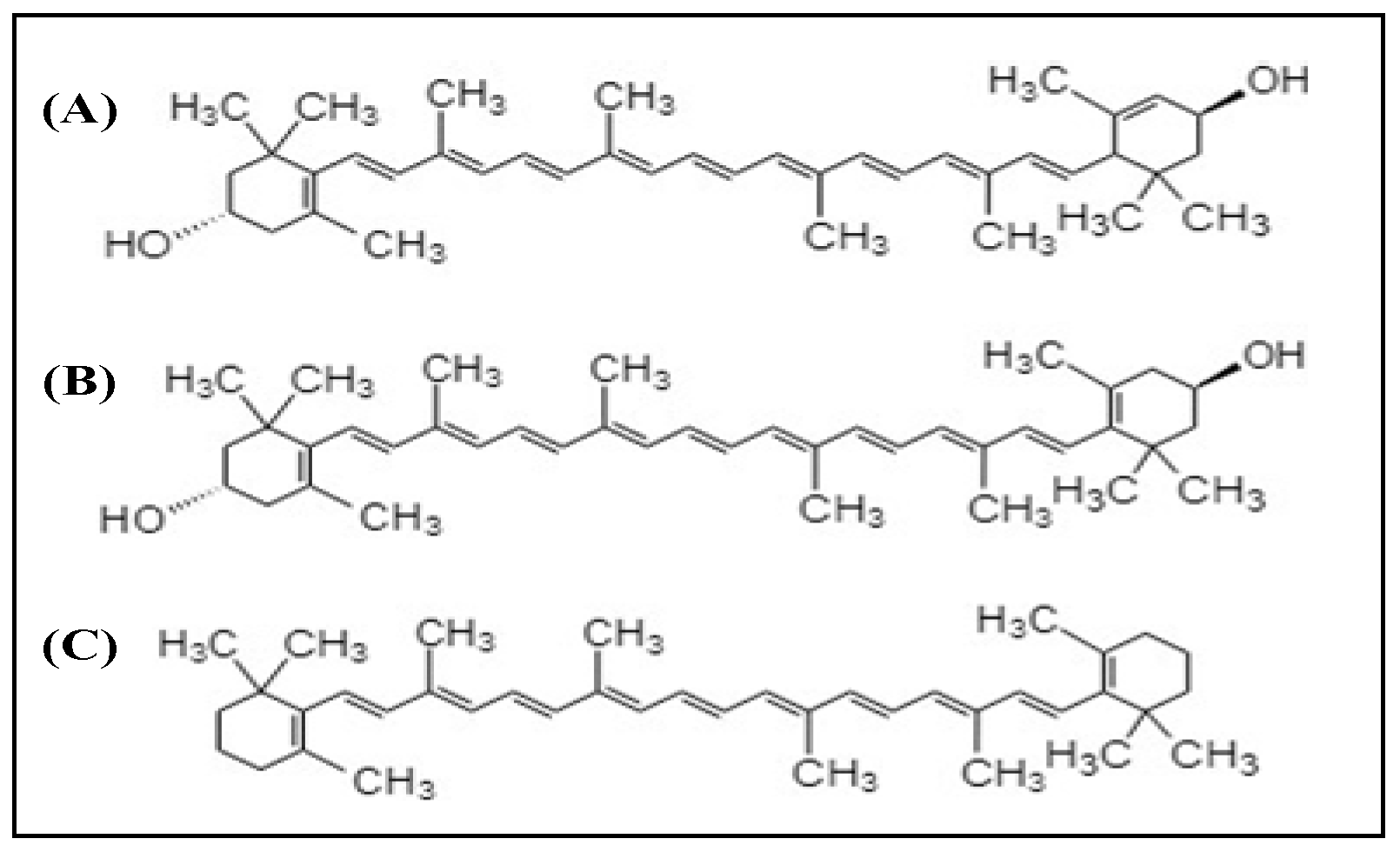
2.2. Xanthophylls
3. Vitamins
4. Minerals
5. Absorption and Bioavailability of Nutrients and Antioxidants
6. Adverse Effects of Antioxidants
7. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chew, E.Y. Nutrition effects on ocular diseases in the aging eye. Investig. Ophthalmol. Vis. Sci. 2013, 54, ORSF42–ORSF47. [Google Scholar] [CrossRef]
- Schleicher, M.; Weikel, K.; Garber, C.; Taylor, A. Diminishing risk for age-related macular degeneration with nutrition: A current view. Nutrients 2013, 5, 2405–2456. [Google Scholar] [CrossRef]
- Andreatta, W.; El-Sherbiny, S. Evidence-based nutritional advice for patients affected by age-related macular degeneration. Ophthalmologica 2014, 231, 185–190. [Google Scholar] [CrossRef]
- Gorusupudi, A.; Nelson, K.; Bernstein, P.S. The Age-Related Eye Disease 2 Study: Micronutrients in the treatment of macular degeneration. Adv. Nutr. 2017, 8, 40–53. [Google Scholar] [CrossRef]
- Bourne, R.R.; Jonas, J.B.; Bron, A.M.; Cicinelli, M.V.; Das, A.; Flaxman, S.R.; Friedman, D.S.; Keeffe, J.E.; Kempen, J.H.; Leasher, J.; et al. Prevalence and causes of vision loss in high-income countries and in Eastern and Central Europe in 2015: Magnitude, temporal trends and projections. Br. J. Ophthalmol. 2018, 102, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Hogg, R.; Chakravarthy, U. AMD and micronutrient antioxidants. Curr. Eye Res. 2004, 29, 387–401. [Google Scholar] [CrossRef]
- Deshpande, S. Role of anti-oxidants in prevention of age-related macular degeneration. J. Med. Nutr. Nutraceut. 2012, 1, 83–86. [Google Scholar] [CrossRef]
- Rasmussen, H.M.; Johnson, E.J. Nutrients for the aging eye. Clin. Interv. Aging 2013, 8, 741–748. [Google Scholar]
- Krinsky, N.I.; Landrum, J.T.; Bone, R.A. Biologic mechanisms of the protective role of lutein and zeaxanthin in the eye. Annu. Rev. Nutr. 2003, 23, 171–201. [Google Scholar] [CrossRef]
- Hammond, B.R.; Fletcher, L.M.; Roos, F.; Wittwer, J.; Schalch, W. A double-blind, placebo-controlled study on the effects of lutein and zeaxanthin on photostress recovery, glare disability, and chromatic contrast. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8583–8589. [Google Scholar] [CrossRef]
- Yu, B.; Wang, J.; Suter, P.M.; Russell, R.M.; Grusak, M.A.; Wang, Y.; Yin, S.; Tang, G. Spirulina is an effective dietary source of zeaxanthin to humans. Br. J. Nutr. 2012, 108, 611–619. [Google Scholar] [CrossRef]
- Nolan, J.M.; Power, R.; Stringham, J.; Dennison, J.; Stack, J.; Kelly, D.; Moran, R.; Akuffo, K.O.; Corcoran, L.; Beatty, S. Enrichment of macular pigment enhances contrast sensitivity in subjects free of retinal disease: Central Retinal Enrichment Supplementation Trials–Report 1. Investig. Ophthalmol. Vis. Sci. 2016, 57, 3429–3439. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study Research Group (AREDS). A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS Report No. 8. Arch. Ophthalmol. 2001, 119, 1417–1436. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef]
- Khoo, H.E.; Chew, L.Y.; Ismail, A.; Azlan, A. Anthocyanins in purple colored fruits. In Polyphenols: Chemistry, Dietary Sources and Health Benefits; Sun, J., Prasad, K.N., Ismail, A., Yang, B., You, X., Li, L., Eds.; Nova Science Publisher: New York, NY, USA, 2012; pp. 133–152. ISBN 978-1-62081-809-1. [Google Scholar]
- Sin, H.P.; Liu, D.T.; Lam, D.S. Lifestyle modification, nutritional and vitamins supplements for age-related macular degeneration. Acta Ophthalmol. 2013, 91, 6–11. [Google Scholar] [CrossRef]
- Madhavi, D.; Bomser, J.; Smith, M.; Singletary, K. Isolation of bioactive constituents from Vaccinium myrtillus (bilberry) fruits and cell cultures. Plant Sci. 1998, 131, 95–103. [Google Scholar] [CrossRef]
- Jang, Y.P.; Zhou, J.; Nakanishi, K.; Sparrow, J.R. Anthocyanins protect against A2E photooxidation and membrane permeabilization in retinal pigment epithelial cells. Photochem. Photobiol. 2005, 81, 529–536. [Google Scholar] [CrossRef]
- Müller, D.; Schantz, M.; Richling, E. High performance liquid chromatography analysis of anthocyanins in bilberries (Vaccinium myrtillus L.), blueberries (Vaccinium corymbosum L.), and corresponding juices. J. Food Sci. 2012, 77, C340–C345. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Jiang, Y.; Su, X.; Zhang, Z.; Shi, J. Antioxidant properties of anthocyanins extracted from litchi (Litchi chinenesis Sonn.) fruit pericarp tissues in relation to their role in the pericarp browning. Food Chem. 2007, 101, 1365–1371. [Google Scholar] [CrossRef]
- Matsumoto, H.; Nakamura, Y.; Tachibanaki, S.; Kawamura, S.; Hirayama, M. Stimulatory effect of cyanidin 3-glycosides on the regeneration of rhodopsin. J. Agric. Food Chem. 2003, 51, 3560–3563. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, L.; Lu, F.; Yang, X.; Deng, Q.; Ji, B.; Huang, F. Retinoprotective effects of bilberry anthocyanins via antioxidant, anti-inflammatory, and anti-apoptotic mechanisms in a visible light-induced retinal degeneration model in pigmented rabbits. Molecules 2015, 20, 22395–22410. [Google Scholar] [CrossRef] [PubMed]
- Silván, J.M.; Reguero, M.; de Pascual-Teresa, S. A protective effect of anthocyanins and xanthophylls on UVB-induced damage in retinal pigment epithelial cells. Food Funct. 2016, 7, 1067–1076. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, D.; Konishi, T. Anthocyanins and anthocyanin-rich extracts: Role in diabetes and eye function. Asia Pac. J. Clin. Nutr. 2007, 16, 200–208. [Google Scholar] [PubMed]
- Kalt, W.; Blumberg, J.B.; McDonald, J.E.; Vinqvist-Tymchuk, M.R.; Fillmore, S.A.; Graf, B.A.; O’Leary, J.M.; Milbury, P.E. Identification of anthocyanins in the liver, eye, and brain of blueberry-fed pigs. J. Agric. Food Chem. 2008, 56, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Olmedilla-Alonso, B.; Estévez-Santiago, R.; Silván, J.M.; Sánchez-Prieto, M.; de Pascual-Teresa, S. Effect of long-term xanthophyll and anthocyanin supplementation on lutein and zeaxanthin serum concentrations and macular pigment optical density in postmenopausal women. Nutrients 2018, 10, 959. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, P.S.; Li, B.; Vachali, P.P.; Gorusupudi, A.; Shyam, R.; Henriksen, B.S.; Nolan, J.M. Lutein, zeaxanthin, and meso-zeaxanthin: The basic and clinical science underlying carotenoid-based nutritional interventions against ocular disease. Prog. Retin. Eye Res. 2016, 50, 34–66. [Google Scholar] [CrossRef] [PubMed]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophy. 2018, 652, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Lin, X.M. Effects of lutein and zeaxanthin on aspects of eye health. J. Sci. Food Agric. 2010, 90, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Olmedilla, B.; Granado, F.; Blanco, I.; Vaquero, M. Lutein, but not alpha-tocopherol, supplementation improves visual function in patients with age-related cataracts: A 2-y doubleblind, placebo-controlled pilot study. Nutrition 2003, 19, 21–24. [Google Scholar] [CrossRef]
- Stringham, J.M.; Hammond, B.R. Macular pigment and visual performance under glare conditions. Optom. Vis. Sci. 2008, 85, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Sweet, B.V. Lutein and zeaxanthin for macular degeneration. Am. J. Health Syst. Pharm. 2008, 65, 1232–1238. [Google Scholar] [CrossRef]
- Nolan, J.M.; Stack, J.; O’ Donovan, O.; Loane, E.; Beatty, S. Risk factors for age-related maculopathy are associated with a relative lack of macular pigment. Exp. Eye Res. 2007, 84, 61–74. [Google Scholar] [CrossRef]
- Johnson, E.J.; Maras, J.E.; Rasmussen, H.M.; Tucker, K.L. Intake of lutein and zeaxanthin differ with age, sex, and ethnicity. J. Am. Diet. Assoc. 2010, 110, 1357–1362. [Google Scholar] [CrossRef]
- Chung, H.Y.; Rasmussen, H.M.; Johnson, E.J. Lutein bioavailability is higher from lutein-enriched eggs than from supplements and spinach in men. J. Nutr. 2004, 134, 1887–1893. [Google Scholar] [CrossRef]
- Chew, E.Y.; Clemons, T.E.; SanGiovanni, J.P.; Danis, R.P.; Ferris, F.L.; Elman, M.J.; Antoszyk, A.N.; Ruby, A.J.; Orth, D.; Bressler, S.B.; et al. Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: AREDS2 Report No. 3. JAMA Ophthalmol. 2014, 132, 142–149. [Google Scholar] [CrossRef]
- Delcourt, C.; Cristol, J.-P.; Tessier, F.; Léger, C.L.; Descomps, B.; Papoz, L. Age-related macular degeneration and antioxidant status in the POLA study. Arch. Ophthalmol. 1999, 117, 1384–1390. [Google Scholar] [CrossRef]
- Chong, E.W.; Wong, T.Y.; Kreis, A.J.; Simpson, J.A.; Guymer, R.H. Dietary antioxidants and primary prevention of age related macular degeneration: Systematic review and meta-analysis. BMJ 2007, 335, 755. [Google Scholar] [CrossRef]
- Goldberg, J.; Flowerdew, G.; Smith, E.; Brody, J.A.; Tso, M.O.M. Factors associated with age-related macular degeneration: An analysis of data from the First National Health and Nutrition Examination Survey. Am. J. Epidemiol. 1988, 128, 700–710. [Google Scholar] [CrossRef]
- De Koning-Backus, A.P.; Buitendijk, G.H.; Kiefte-de Jong, J.C.; Colijn, J.M.; Hofman, A.; Vingerling, J.R.; Haverkort, E.B.; Franco, O.H.; Klaver, C.C. Intake of vegetables, fruit, and fish is beneficial for Age-related Macular Degeneration. Am. J. Ophthalmol. 2019, 198, 70–79. [Google Scholar] [CrossRef]
- Seddon, J.M.; Ajani, U.A.; Sperduto, R.D.; Hiller, R.; Blair, N.; Burton, T.C.; Farber, M.D.; Gragoudas, E.S.; Haller, J.; Miller, D.T.; et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. JAMA 1994, 272, 1413–1420. [Google Scholar] [CrossRef]
- Zampatti, S.; Ricci, F.; Cusumano, A.; Marsella, L.T.; Novelli, G.; Giardina, E. Review of nutrient actions on age-related macular degeneration. Nutr. Res. 2014, 34, 95–105. [Google Scholar] [CrossRef]
- SanGiovanni, J.P.; Chew, E.Y.; Clemons, T.E.; Ferris, F.L., II; Gensler, G.; Lindblad, A.S.; Milton, R.C.; Seddon, J.M.; Sperduto, R.D. The relationship of dietary carotenoid and vitamin A, E, and C intake with age-related macular degeneration in a case-control study. Arch Ophthalmol. 2007, 125, 1225–1232. [Google Scholar]
- Christen, W.G.; Ajani, U.A.; Glynn, R.J.; Manson, J.E.; Schaumberg, D.A.; Chew, E.C.; Buring, J.E.; Hennekens, C.H. Prospective cohort study of antioxidant vitamin supplement use and the risk of age-related maculopathy. Am. J. Epidemiol. 1999, 149, 476–484. [Google Scholar] [CrossRef]
- Klein, M.L.; Francis, P.J.; Rosner, B.; Reynolds, R.; Hamon, S.C.; Schultz, D.W.; Ott, J.; Seddon, J.M. CFH and LOC387715/ARMS2 genotypes and treatment with antioxidants and zinc for age-related macular degeneration. Ophthalmology 2008, 115, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.R.; Lawrenson, J.G. Antioxidant vitamin and mineral supplements for preventing age-related macular degeneration. Cochrane Database Syst. Rev. 2017, 7, CD000253. [Google Scholar] [CrossRef] [PubMed]
- Eye Disease Case-Control Study Group. Antioxidant status and neovascular age-related macular degeneration. Arch. Ophthalmol. 1993, 111, 104–109. [Google Scholar] [CrossRef]
- Robison, W.G.; Kuwabara, T.; Bieri, J.G. The roles of vitamin E and unsaturated fatty acids in the visual process. Retina 1982, 2, 263–281. [Google Scholar] [CrossRef] [PubMed]
- Tanito, M.; Yoshida, Y.; Kaidzu, S.; Chen, Z.-H.; Cynshi, O.; Jishage, K.-I.; Niki, E.; Ohira, A. Acceleration of age-related changes in the retina in a-tocopherol transfer protein null mice fed a vitamin E–deficient diet. Investig. Ophthalmol. Vis. Sci. 2007, 48, 396–404. [Google Scholar] [CrossRef]
- Katz, M.L.; Eldred, G.E. Failure of vitamin E to protect the retina against damage resulting from bright cyclic light exposure. Investig. Ophthalmol. Vis. Sci. 1989, 30, 29–36. [Google Scholar]
- Infante, J.P. Vitamin E and selenium participation in fatty acid desaturation. A proposal for an enzymatic function of these nutrients. Mol. Cell. Biochem. 1986, 69, 93–108. [Google Scholar] [CrossRef]
- Handelman, G.J.; Machlin, L.J.; Fitch, K.; Weiter, J.J.; Dratz, E.A. Oral α-tocopherol supplements decrease plasma γ-tocopherol levels in humans. J. Nutr. 1985, 115, 807–813. [Google Scholar] [CrossRef]
- Alvarez, R.; Liou, F.; Fong, S. Levels of alpha-, and gamma-tocopherol in human eyes: Evaluation of the possible role of IRBP in intraocular alpha-tocopherol transport. Am. J. Clin. Nutr. 1987, 46, 481–487. [Google Scholar] [CrossRef]
- Belda, J.I.; Romá, J.; Vilela, C.; Puertas, F.J.; Díaz-Llopis, M.; Bosch-Morell, F.; Romero, F.J. Serum vitamin E levels negatively correlate with severity of age-related macular degeneration. Mech. Ageing Dev. 1999, 107, 159–164. [Google Scholar] [CrossRef]
- Taylor, H.R.; Tikellis, G.; Robman, L.D.; McCarty, C.A.; McNeil, J.J. Vitamin E supplementation and macular degeneration: Randomised controlled trial. Br. Med. J. 2002, 325, 11. [Google Scholar] [CrossRef]
- Thurnham, D.I. Macular zeaxanthins and lutein—A review of dietary sources and bioavailability and some relationships with macular pigment optical density and age-related macular disease. Nutr. Res. Rev. 2007, 20, 163–179. [Google Scholar] [CrossRef]
- Christen, W.G.; Glynn, R.J.; Gaziano, J.M.; Darke, A.K.; Crowley, J.J.; Goodman, P.J.; Lippman, S.M.; Lad, T.E.; Bearden, J.D.; Goodman, G.E.; et al. Age-related cataract in men in the selenium and vitamin E cancer prevention trial eye endpoints study: A randomized clinical trial. JAMA Ophthalmol. 2015, 133, 17–24. [Google Scholar] [CrossRef]
- Aoki, A.; Inoue, M.; Nguyen, E.; Obata, R.; Kadonosono, K.; Shinkai, S.; Hashimoto, H.; Sasaki, S.; Yanagi, Y. Dietary n-3 fatty acid, α-tocopherol, zinc, vitamin D, vitamin C, and β-carotene are associated with age-related macular degeneration in Japan. Sci. Rep. 2016, 6, 20723. [Google Scholar] [CrossRef]
- Newsome, D.A.; Swartz, M.; Leone, N.C.; Elston, R.C.; Miller, E. Oral zinc in macular degeneration. Arch. Ophthalmol. 1988, 106, 192–198. [Google Scholar] [CrossRef]
- Newsome, D.A. A randomized, prospective, placebo-controlled clinical trial of a novel zinc-monocysteine compound in age-related macular degeneration. Curr. Eye Res. 2008, 33, 591–598. [Google Scholar] [CrossRef]
- Stur, M.; Tittl, M.; Reitner, A.; Meisinger, V. Oral zinc and the second eye in age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 1996, 37, 1225–1235. [Google Scholar]
- Grahn, B.H.; Paterson, P.G.; Gottschall-Pass, K.T.; Zhang, Z. Zinc and the eye. J. Am. Coll. Nutr. 2001, 20, 106–118. [Google Scholar] [CrossRef]
- King, J.C. Zinc: An essential but elusive nutrient. Am. J. Clin. Nutr. 2011, 94, 679S–684S. [Google Scholar] [CrossRef]
- Ugarte, M.; Osborne, N.N. Zinc in the retina. Prog. Neurobiol. 2001, 64, 219–249. [Google Scholar] [CrossRef]
- Vishwanathan, R.; Chung, M.; Johnson, E.J. A systematic review on zinc for the prevention and treatment of age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2013, 54, 3985–3998. [Google Scholar] [CrossRef]
- Assel, M.J.; Li, F.; Wang, Y.; Allen, A.S.; Baggerly, K.A.; Vickers, A.J. Genetic polymorphisms of CFH and ARMS2 do not predict response to antioxidants and zinc in patients with age-related macular degeneration: Independent statistical evaluations of data from the Age-Related Eye Disease Study. Ophthalmol. 2018, 125, 391–397. [Google Scholar] [CrossRef]
- Ursini, F.; Bindoli, A. The role of selenium peroxidases in the protection against oxidative damage of membranes. Chem. Phys. Lipids 1987, 44, 255–276. [Google Scholar] [CrossRef]
- Tsang, N.C.; Penfold, P.L.; Snitch, P.J.; Billson, F. Serum levels of antioxidants and age-related macular degeneration. Doc. Ophthalmol. 1992, 81, 387–400. [Google Scholar] [CrossRef]
- Farnsworth, C.C.; Stone, W.L.; Dratz, E.A. Effects of vitamin E and selenium deficiency on the fatty acid composition of rat retinal tissues. Biochim. Biophys. Acta 1979, 552, 281–293. [Google Scholar] [CrossRef]
- La Frano, M.R.; de Moura, F.F.; Boy, E.; Lönnerdal, B.; Burri, B.J. Bioavailability of iron, zinc, and provitamin A carotenoids in biofortified staple crops. Nutr. Rev. 2014, 72, 289–307. [Google Scholar] [CrossRef]
- Parada, J.; Aguilera, J.M. Food microstructure affects the bioavailability of several nutrients. J. Food Sci. 2007, 72, R21–R32. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Uptake of lycopene and its geometrical isomers is greater from heat-processed than from unprocessed tomato juice in humans. J. Nutr. 1992, 122, 2161–2166. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Guerra, L.H.; Ruiz, C.A. Lutein and zeaxanthin dietary supplements raise macular pigment density and serum concentrations of these carotenoids in humans. J. Nutr. 2003, 133, 992–998. [Google Scholar] [CrossRef]
- West, J.W. Nutritional strategies for managing the heat-stressed dairy cow. J. Anim. Sci. 1999, 77, 21–35. [Google Scholar] [CrossRef]
- Nidhi, B.; Mamatha, B.S.; Baskaran, V. Olive oil improves the intestinal absorption and bioavailability of lutein in lutein-deficient mice. Eur. J. Nutr. 2014, 53, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Gleize, B.; Tourniaire, F.; Depezay, L.; Bott, R.; Nowicki, M.; Albino, L.; Lairon, D.; Kesse-Guyot, E.; Galan, P.; Hercberg, S.; et al. Effect of type of TAG fatty acids on lutein and zeaxanthin bioavailability. Br. J. Nutr. 2013, 110, 1–10. [Google Scholar] [CrossRef]
- Solomons, N.W.; Jacob, R.A. Studies on the bioavailability of zinc in humans: Effects of heme and nonheme iron on the absorption of zinc. Am. J. Clin. Nutr. 1981, 34, 475–482. [Google Scholar] [CrossRef]
- Vishwanathan, R.; Goodrow-Kotyla, E.F.; Wooten, B.R.; Wilson, T.A.; Nicolosi, R.J. Consumption of 2 and 4 egg yolks/d for 5 wk increases macular pigment concentrations in older adults with low macular pigment taking cholesterol-lowering statins. Am. J. Clin. Nutr. 2009, 90, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Vassileva, G.; Huwyler, L.; Poirier, K.; Agellon, L.B.; Toth, M.J. The intestinal fatty acid binding protein is not essential for dietary fat absorption in mice. FASEB J. 2000, 14, 2040–2046. [Google Scholar] [CrossRef]
- Hollander, D.; Ruble, P.E., Jr. Beta-carotene intestinal absorption: Bile, fatty acid, pH, and flow rate effects on transport. Am. J. Physiol. Endocrinol. Metab. 1978, 235, E686–E691. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.S.; Bailey, K.B.; Gibbs, M.; Ferguson, E.L. A review of phytate, iron, zinc, and calcium concentrations in plant-based complementary foods used in low-income countries and implications for bioavailability. Food Nutr. Bull. 2010, 31, S134–S146. [Google Scholar] [CrossRef]
- Van het Hof, K.H.; Brouwer, I.A.; West, C.E.; Haddeman, E.; Steegers-Theunissen, R.P.; van Dusseldorp, M.; Weststrate, J.A.; Eskes, T.K.; Hautvast, J.G. Bioavailability of lutein from vegetables is 5 times higher than that of β-carotene. Am. J. Clin. Nutr. 1999, 70, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Roodenburg, A.J.; Leenen, R.; van het Hof, K.H.; Weststrate, J.A.; Tijburg, L.B. Amount of fat in the diet affects bioavailability of lutein esters but not of α-carotene, β-carotene, and vitamin E in humans. Am. J. Clin. Nutr. 2000, 71, 1187–1193. [Google Scholar] [CrossRef] [PubMed]
- Kruger, C.L.; Murphy, M.; DeFreitas, Z.; Pfannkuch, F.; Heimbach, J. An innovative approach to the determination of safety for a dietary ingredient derived from a new source: Case study using a crystalline lutein product. Food Chem. Toxicol. 2002, 40, 1535–1549. [Google Scholar] [CrossRef]
- Lakshminarayana, R.; Raju, M.; Krishnakantha, T.P.; Baskaran, V. Lutein and zeaxanthin in leafy greens and their bioavailability: Olive oil influences the absorption of dietary lutein and its accumulation in adult rats. J. Agric. Food Chem. 2007, 55, 6395–6400. [Google Scholar] [CrossRef] [PubMed]
- Satyanarayana, U.; Kumar, A.N.; Naidu, J.N.; Prasad, D.K.V. Antioxidant supplementation for health—A boon or a bane? J. Dr. NTR Univ. Health Sci. 2014, 3, 221–230. [Google Scholar] [CrossRef]
- Hercberg, S.; Ezzedine, K.; Guinot, C.; Preziosi, P.; Galan, P.; Bertrais, S.; Estaquio, C.; Briançon, S.; Favier, A.; Latreille, J.; et al. Antioxidant supplementation increases the risk of skin cancers in women but not in men. J. Nutr. 2007, 137, 2098–2105. [Google Scholar] [CrossRef]
- Scientific Committee on Food. Tolerable Upper Intake Levels for Vitamins and Minerals; European Food Safety Authority: Parma, Italy, 2006; Available online: http://www.efsa.europa.eu/sites/default/files/efsa_rep/blobserver_assets/ndatolerableuil.pdf (accessed on 8 September 2018).
- Wallace, T.C.; Giusti, M.M. Anthocyanins. Adv. Nutr. 2015, 6, 620–622. [Google Scholar] [CrossRef]
- Ravikrishnan, R.; Rusia, S.; Ilamurugan, G.; Salunkhe, U.; Deshpande, J.; Shankaranarayanan, J.; Shankaranarayana, M.L.; Soni, M.G. Safety assessment of lutein and zeaxanthin (Lutemax™ 2020): Subchronic toxicity and mutagenicity studies. Food Chem. Toxicol. 2011, 49, 2841–2848. [Google Scholar] [CrossRef] [PubMed]
- Dagnelie, G.; Zorge, I.S.; McDonald, T.M. Lutein improves visual function in some patients with retinal degeneration: A pilot study via the Internet. Optometry 2000, 71, 147–164. [Google Scholar] [PubMed]
- Peto, R.; Doll, R.; Buckley, J.D.; Sporn, M.B. Can dietary beta-carotene materially reduce human cancer rates? Nature 1981, 290, 201–208. [Google Scholar] [CrossRef]
- Diplock, A.T. Safety of antioxidant vitamins and beta-carotene. Am. J. Clin. Nutr. 1995, 62, 1510S–1516S. [Google Scholar] [CrossRef] [PubMed]
- Kong, K.W.; Khoo, H.E.; Prasad, K.N.; Ismail, A.; Tan, C.P.; Rajab, N.F. Revealing the power of the natural red pigment lycopene. Molecules 2010, 15, 959–987. [Google Scholar] [CrossRef]
- Bánhegyi, G. Lycopene—A natural antioxidant. Orvosi Hetilap 2005, 146, 1621–1624. [Google Scholar] [PubMed]
- Penniston, K.L.; Tanumihardjo, S.A. The acute and chronic toxic effects of vitamin A. Am. J. Clin. Nutr. 2006, 83, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Hathcock, J.N.; Hattan, D.G.; Jenkins, M.Y.; McDonald, J.T.; Sundaresan, P.R.; Wilkening, V.L. Evaluation of vitamin A toxicity. Am. J. Clin. Nutr. 1990, 52, 183–202. [Google Scholar] [CrossRef]
- Khoo, H.E.; Prasad, K.N.; Kong, K.W.; Jiang, Y.; Ismail, A. Carotenoids and their isomers: Color pigments in fruits and vegetables. Molecules 2011, 16, 1710–1738. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS). Statement on the safety of β-carotene use in heavy smokers. EFSA J. 2012, 10, 2953. [Google Scholar]
- Miller, D.R.; Hayes, K.C. Vitamin excess and toxicity. In Nutritional Toxicology; Hathcock, J., Ed.; Academic Press: New York, NY, USA, 1982; Volume 1, pp. 81–133. [Google Scholar]
- Braun, L. Vitamin C and kidney stones. J. Complement. Med. 2009, 8, 42–44. [Google Scholar]
- Osredkar, J.; Sustar, N. Copper and zinc, biological role and significance of copper/zinc imbalance. J. Clin. Toxicol. 2011, S3. [Google Scholar] [CrossRef]
- Sandstead, H.H. Understanding zinc: Recent observations and interpretations. J. Lab. Clin. Med. 1994, 124, 322–327. [Google Scholar]
- Lewis, M.R.; Kokan, L. Zinc gluconate: Acute ingestion. J. Toxicol. 1998, 36, 99–101. [Google Scholar] [CrossRef]
- Hooper, P.L.; Visconti, L.; Garry, P.J.; Johnson, G.E. Zinc lowers high-density lipoprotein-cholesterol levels. JAMA 1980, 244, 1960–1961. [Google Scholar] [CrossRef]
- Vinceti, M.; Wei, E.T.; Malagoli, C.; Bergomi, M.; Vivoli, G. Adverse Health Effects of Selenium in Humans. Rev. Environ. Health 2001, 16, 233–252. [Google Scholar] [CrossRef] [PubMed]
- Forceville, X.; Laviolle, B.; Annane, D.; Vitoux, D.; Bleichner, G.; Korach, J.M.; Cantais, E.; Georges, H.; Soubirou, J.L.; Combes, A.; et al. Effects of high doses of selenium, as sodium selenite, in septic shock: A placebo-controlled, randomized, double-blind, phase II study. Crit. Care 2007, 11, R73. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
| Compounds | Study Design | Doses | Outcomes | Ref. |
|---|---|---|---|---|
| Anthocyanins | ||||
| Cyanidin 3-glucoside, cyanidin 3-rutinoside, delphinidin 3-glucoside, and delphinidin 3-rutinoside | In vitro bioassays: Rod outer segment and opsin membranes of frog | 10–50 µM | Positive outcomes: Cyanidin 3-glucoside and cyanidin 3-rutinoside stimulated regeneration of rhodopsin | [21] |
| Cyanidin 3-glucoside and delphinidin 3-glucoside | Cell culture: ARPE-19 cells (human retinal pigment epithelial cell line) | 5 μM | Positive outcome: Anthocyanins pre-treatment attenuated apoptosis of ARPE-19 cells induced by UVB irradiation. | [23] |
| Bilberry anthocyanin extract | In vivo study: Retinal degeneration model in pigmented rabbits (seven days) | 250 and 500 mg/kg/day | Positive outcomes: Attenuated changes caused by light to Bax, Bcl-2, and caspase-3. Increased the levels of superoxide dismutase, glutathione peroxidase, catalase, and total antioxidant capacity. Decreased malondialdehyde level in the retinal cells. Inhibited light-induced elevation in the levels of pro-inflammatory cytokines and angiogenic parameters (IL-1β and VEGF). | [22] |
| Anthocyanin supplement | Randomized, parallel study. Postmenopausal, one woman (eight months) | 60 mg/day | Negative outcome (compared to baseline): No significant increase in macular pigment optical density | [26] |
| Carotenoids | ||||
| Lutein and zeaxanthin | Cell culture: ARPE-19 cells | 5 μM | Positive outcome: Anthocyanins pre-treatment attenuated apoptosis of ARPE-19 cells induced by UVB irradiation. | [23] |
| Lutein and zeaxanthin | Prospective, randomized, double-blind, placebo-controlled human study (12 months) | 10 mg/day lutein and 2 mg/day zeaxanthin | Positive outcomes: Significantly increased macular pigment optical density for treatment group compared to placebo. Significantly increased levels of serum lutein and zeaxanthin. Significantly improved chromatic contrast and photo stress recovery time for treatment group compared to placebo. | [10] |
| Zeaxanthin-containing spirulina (4–5 g) | Human feeding trials (45 days) | 2.6–3.7 mg zeaxanthin | Positive outcome: Increased mean serum zeaxanthin concentration from 0.06 to 0.15 μmol/L. | [11] |
| Lutein, zeaxanthin, and meso-zeaxanthin in sunflower oil suspension | Double-blind, placebo-controlled, block-randomized human trial (12 months) | 10 mg lutein, 10 mg meso-zeaxanthin, and 2 mg zeaxanthin | Positive outcomes: Significantly improved contrast sensitivity of the visual function after 12 months supplementation compared to baseline. Treatment group had significant increase in serum concentrations of the xanthophylls in retina and macular pigment optical density compared to placebo. | [12] |
| Lutein vs. α-tocopherol | Randomized, double-blind, placebo-controlled supplementation study (24 months) | 12 mg lutein mixtures and 100 mg α-tocopherol | Positive outcomes: Significantly increased serum concentration of lutein. Increased visual performance (visual acuity and glare sensitivity) in lutein group only. No toxic effect found—no significant changes in hematological and biochemical profiles. | [30] |
| Oral total daily supplementation of antioxidants (mixture of β-carotene with other vitamins) | Randomized, placebo-controlled clinical trial (followed up for up to 10 years) | 15 mg β-carotene | Positive primary outcome (compared to baseline): Reduced risk of visual acuity lost. Negative secondary outcomes: No significant differences for all the secondary outcomes between the treatment group and placebo. | [31] |
| Nutrient intake (β-carotene, β-cryptoxathin, lutein, zeaxanthin, and lycopene) | Epidemiological study (Self-report data) | - | Positive outcome: Participants with the highest self-reported dietary intake of lutein and zeaxanthin were inversely associated with advancedage-related macular degeneration (AMD). | [37] |
| Total carotenoids (lutein/zeaxanthin, α-carotene, β-carotene, cryptoxanthin, and lycopene | Eye Disease Case-Control Study | - | Positive outcome: Serum carotenoid level significantly associated with the risk of AMD | [38] |
| Xanthophyll supplement | Randomized, parallel study. Postmenopausal women (8 months) | 6 mg lutein and 2 mg zeaxanthin daily) | Positive outcome: Dietary supplementation of lutein and zeaxanthin significantly increased the serum lutein and zeaxanthin levels. Negative outcome (compared to baseline): No significant increase in macular pigment optical density | [26] |
| Compounds | Study Design | Doses | Outcomes | Ref. |
|---|---|---|---|---|
| Vitamins | ||||
| Mixture of vitamin C and vitamin E with provitamin A carotenoid | Randomized, placebo-controlled clinical trials (followed-up for up to 10 years) | Vitamin C (500 mg) and vitamin E (400 IU) daily | Positive primary outcomes (compared to baseline): Increase in nuclear, cortical, or posterior subcapsular opacity grades or cataract surgery. Moderate visual acuity lost (≥15 letters). Negative secondary outcomes: No significant differences for all the secondary outcomes between the treatment group and placebo. | [11] |
| Provitamin A β-carotene, vitamin C, and vitamin E | Age-Related Eye Disease Study | - | Positive outcomes: Increased intake of β-carotene, vitamin C, and vitamin E associated with a reduced risk of neovascular AMD. | [42] |
| Vitamin A, vitamin C, and vitamin E | Systematic review and meta-analysis | - | Positive outcomes: Dietary intake of a mixture of vitamin A, vitamin C, and vitamin E had a larger effect on the reduction of AMD risk than the individual vitamin. | [43] |
| Vitamin A, vitamin C, and vitamin E | Case-control study | - | Positive outcomes: Low dietary intake of vitamin C and vitamin E was associated with neovascular AMD. Negative outcome: Dietary vitamin A showed no association with neovascular AMD. | [58] |
| Vitamin E | Randomized controlled trial (four years) | 500 IU daily | Negative outcomes: Failed to prevent the development and progression of AMD. | [55] |
| Vitamin E | Randomized placebo controlled 4-arm trial (follow-up of 5.6 ± 1.2 years) | 400 IU daily (DL-α-tocopherol acetate) | Negative outcome: Vitamin supplementation showed no protective effect against cataracts among the participants (elderly men). | [57] |
| Vitamin A, vitamin C, and vitamin E | Multicenter eye disease case-control study (Epidemiological study) | - | Negative outcomes: Vitamins A, C, and E consumptions were not associated with the reduced risk of AMD. | [39] |
| Vitamin A (retinol), vitamin C (ascorbic acid), and vitamin E (α-tocopherol) | POLA (Pathologies Oculaires Liées à l’Age) study | - | Negative outcomes: Plasma vitamin A and vitamin C showed no association with reduction in macular degeneration risk. Plasma vitamin E was negatively associated with early signs of AMD and late AMD. | [40] |
| Vitamin C | Cochrane Review | - | Negative outcomes: Vitamin C supplementation did not prevent any AMD or late AMD. | [46] |
| Vitamin C and vitamin E | Eye Disease Case-Control Study | - | Negative outcome: No statistically significant overall association was found between serum vitamin status and neovascular AMD. | [47] |
| Minerals | ||||
| Zinc | Case-control study | - | Positive outcome: Low dietary intake of zinc was associated with neovascular AMD. | [58] |
| Zinc | Randomized, placebo-controlled clinical trials (followed-up for up to 10 years) | Zinc oxide (80 mg daily) | Positive outcome: Significantly reduced the risk of developing advanced AMD. | [11] |
| Zinc | Randomized double-blinded, placebo-controlled trials (2 years intervention) | Zinc sulfate (200 mg daily) | Positive outcome: Significantly reduced visual loss in treatment group compared to placebo. | [59] |
| Zinc | Randomized, prospective, placebo-controlled clinical trial (three and six months intervention) | Zinc monocysteine (25 mg daily) | Positive outcomes: Significantly improved visual acuity and contrast sensitivity. Significantly shortened macular light flash recovery time both at three months and at six months. | [60] |
| Zinc | Randomized, double-blinded, placebo-controlled study (two years intervention) | Zinc sulfate (200 mg daily) | Positive outcome: Significantly increased serum zinc. Negative outcome: No significant improvement of eye conditions for patients with AMD. | [61] |
| Selenium | Randomized, placebo-controlled, 4-arm trial (follow-up of 5.6 ± 1.2 years) | 200 μg daily (from l-selenomethionine) | Negative outcome: Selenium supplementation did not show significant effect in reducing risk of cataracts among the participants (elderly men). | [57] |
| Nutrients | Recommended Daily Intake | UL |
|---|---|---|
| Vitamins | ||
| Vitamin A | 700 μg RE/day for men and 600 μg RE/day for women | 3000 μg RE/day |
| Vitamin C | <1000 mg/day | 1000 mg/day |
| Vitamin E | 300 mg α-tocopherol equivalents (450 IU) | 300 mg α-tocopherol equivalents (450 IU) |
| Minerals | ||
| Selenium | 26–55 µg/day | 300 μg/day |
| Zinc | 11 mg/day for men and 8 mg/day for women | 25 mg/day |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khoo, H.E.; Ng, H.S.; Yap, W.-S.; Goh, H.J.H.; Yim, H.S. Nutrients for Prevention of Macular Degeneration and Eye-Related Diseases. Antioxidants 2019, 8, 85. https://doi.org/10.3390/antiox8040085
Khoo HE, Ng HS, Yap W-S, Goh HJH, Yim HS. Nutrients for Prevention of Macular Degeneration and Eye-Related Diseases. Antioxidants. 2019; 8(4):85. https://doi.org/10.3390/antiox8040085
Chicago/Turabian StyleKhoo, Hock Eng, Hui Suan Ng, Wai-Sum Yap, Henri Ji Hang Goh, and Hip Seng Yim. 2019. "Nutrients for Prevention of Macular Degeneration and Eye-Related Diseases" Antioxidants 8, no. 4: 85. https://doi.org/10.3390/antiox8040085
APA StyleKhoo, H. E., Ng, H. S., Yap, W.-S., Goh, H. J. H., & Yim, H. S. (2019). Nutrients for Prevention of Macular Degeneration and Eye-Related Diseases. Antioxidants, 8(4), 85. https://doi.org/10.3390/antiox8040085





