Effect of Marine Bacteria and Ulvan on the Activity of Antioxidant Defense Enzymes and the Bio-Protection of Papaya Fruit against Colletotrichum gloeosporioides
Abstract
1. Introduction
2. Materials and Methods
2.1. Colletotrichum Gloeosporioides
2.2. Marine Bacteria
2.3. Polysaccharide Ulvan
2.4. Effect of Ulvan on Spore Germination of C. gloeosporioides
2.5. In Vitro Antagonistic Activity of Marine Bacteria to C. gloeosporioides
2.5.1. Antifungal Activity
2.5.2. Inhibition of C. gloeosporioides for VOCs
2.5.3. Competition for Carbohydrates
2.6. Effect of B. amyloliquefaciens, S. rhizophila, and Ulvan on the Control of Papaya Fruit Anthracnose
2.6.1. Antioxidant Defense Enzymes
Sample Collection
Catalase (CAT) Activity
Superoxide Dismutase (SOD) Activity
Peroxidase (POD) Activity
2.7. Statistical Analysis
3. Results
3.1. Inhibition of C. gloeosporioides Spore Germination by Ulvan
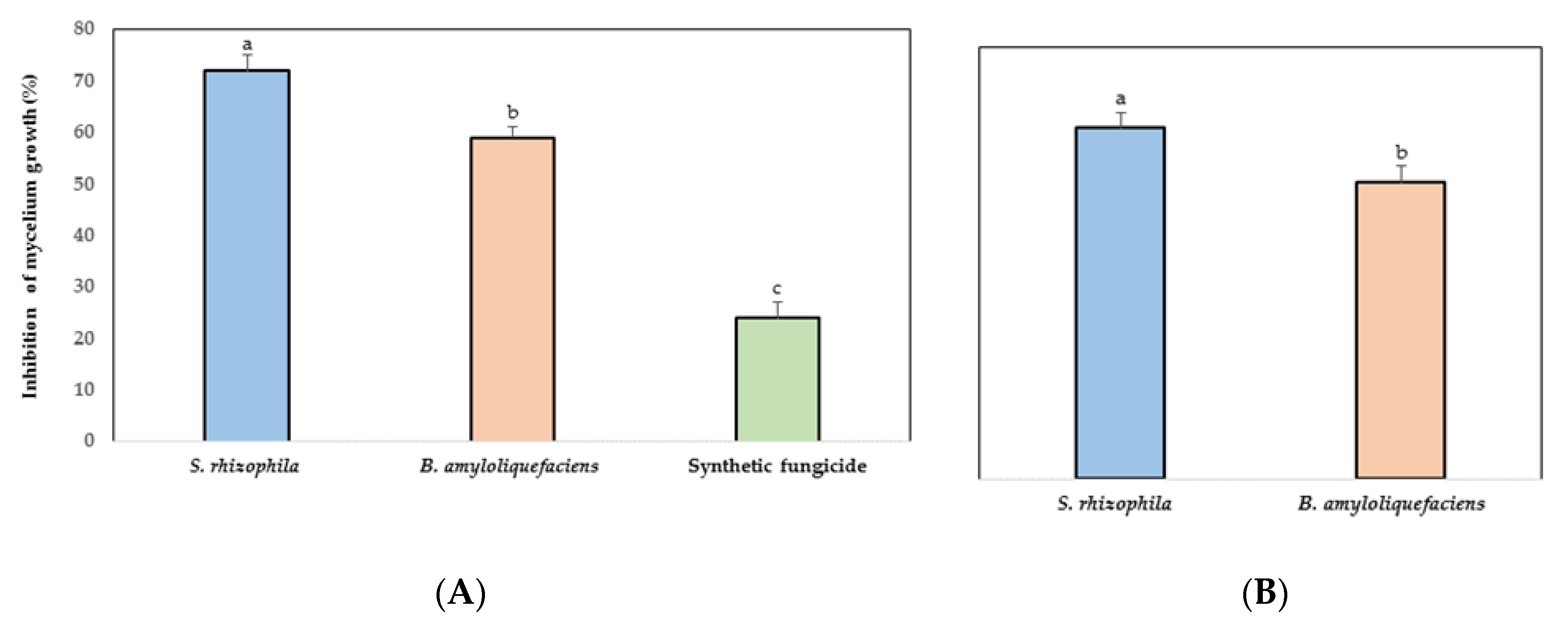
3.2. In Vitro Antifungal Activity of Marine Bacteria to Phytopathogen Fungus
3.3. Inhibition of C. gloeosporioides by VOCs
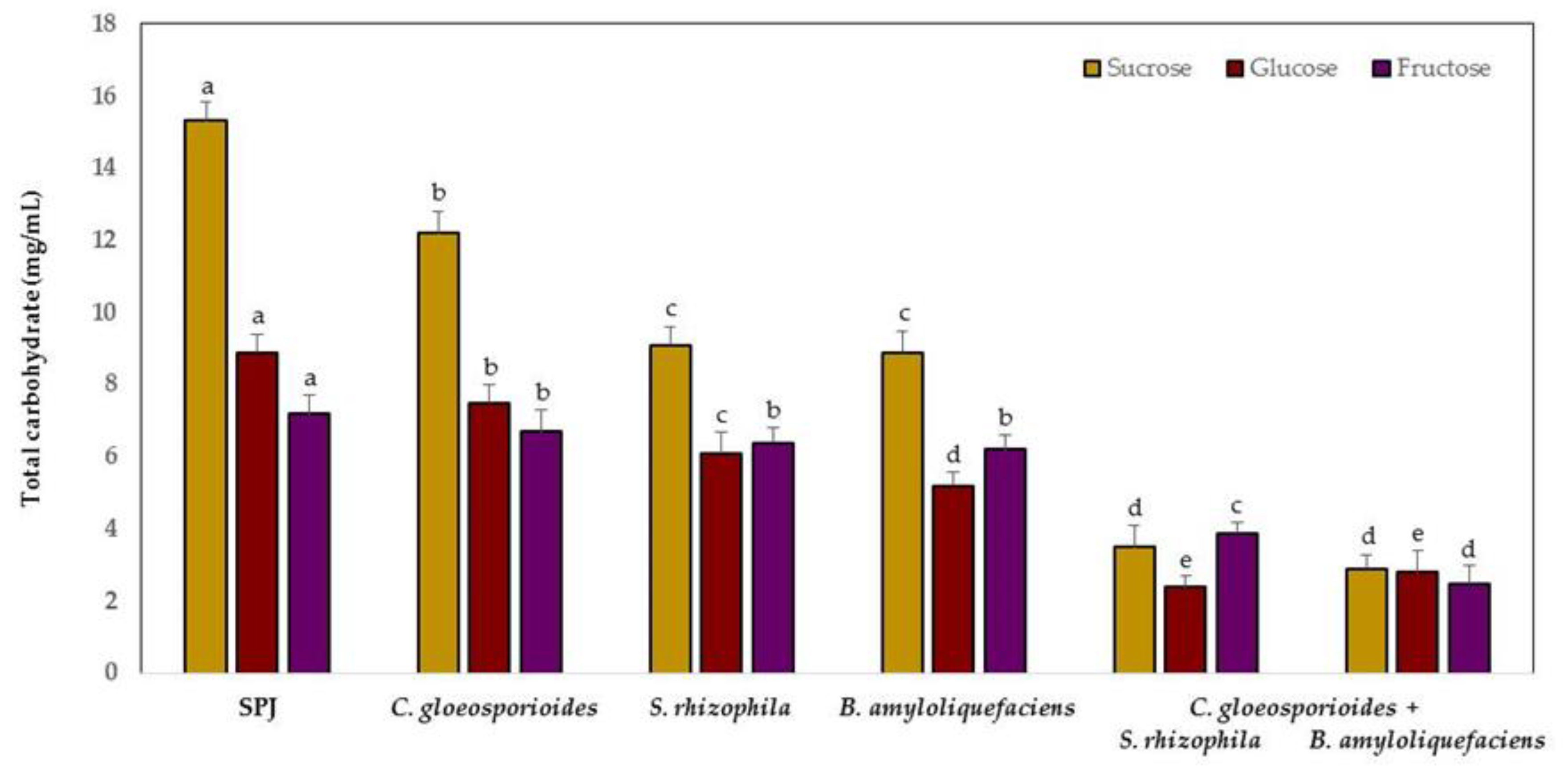
3.4. Carbohydrate Competition between Marine Bacteria and C. gloeosporioides
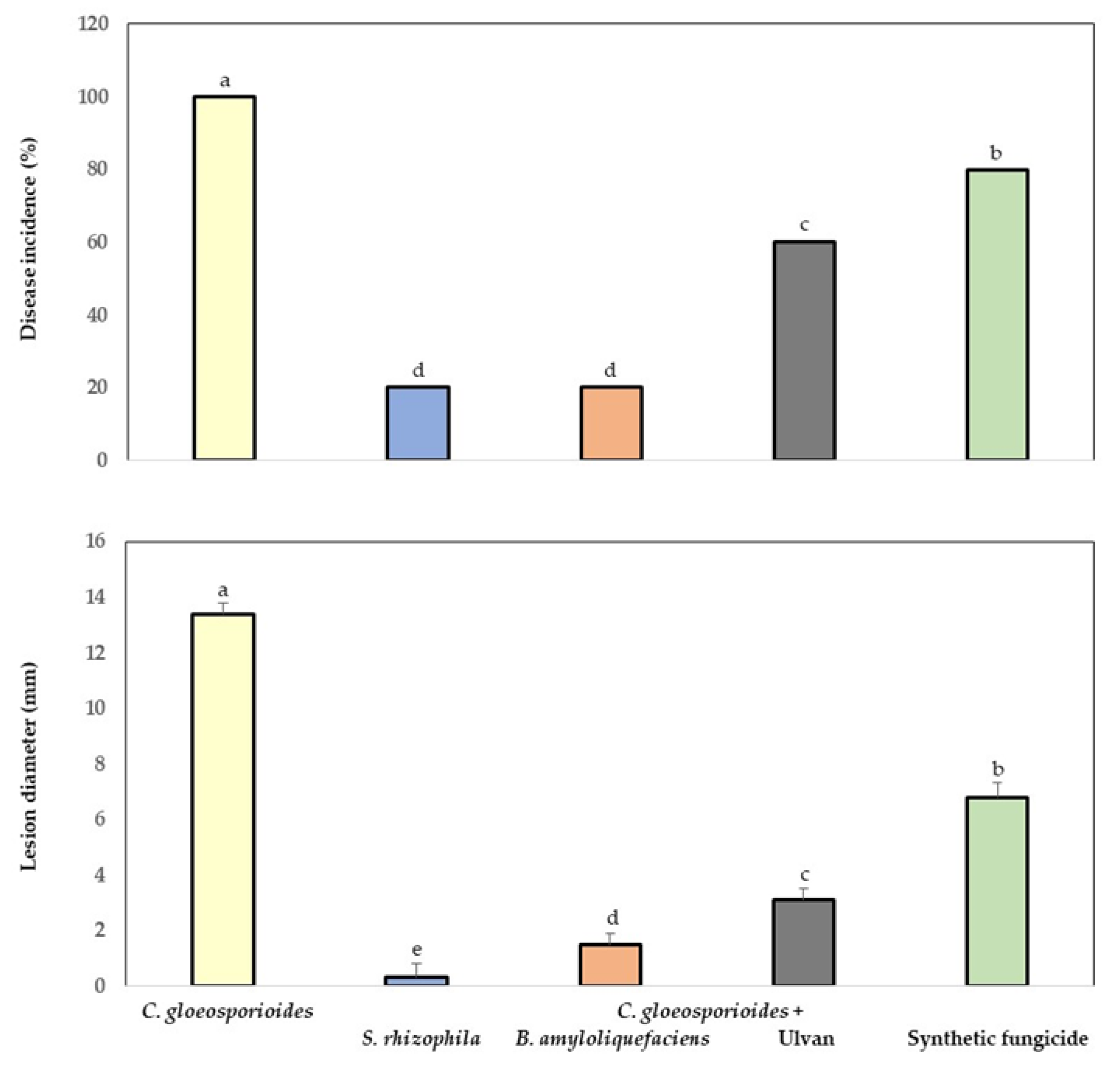
3.5. Bio-Protection of Papaya Fruit Inoculated with Marine Bacteria and Ulvan to Anthracnose
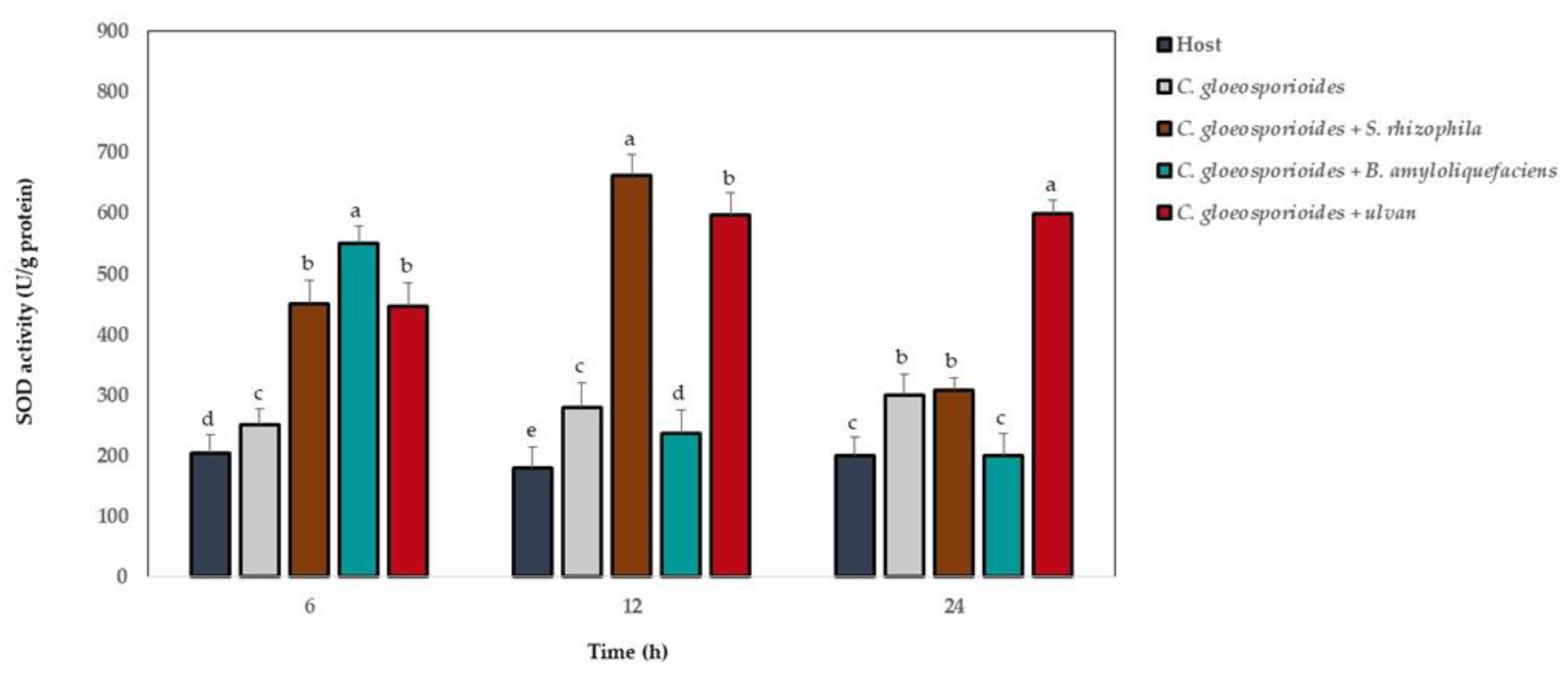
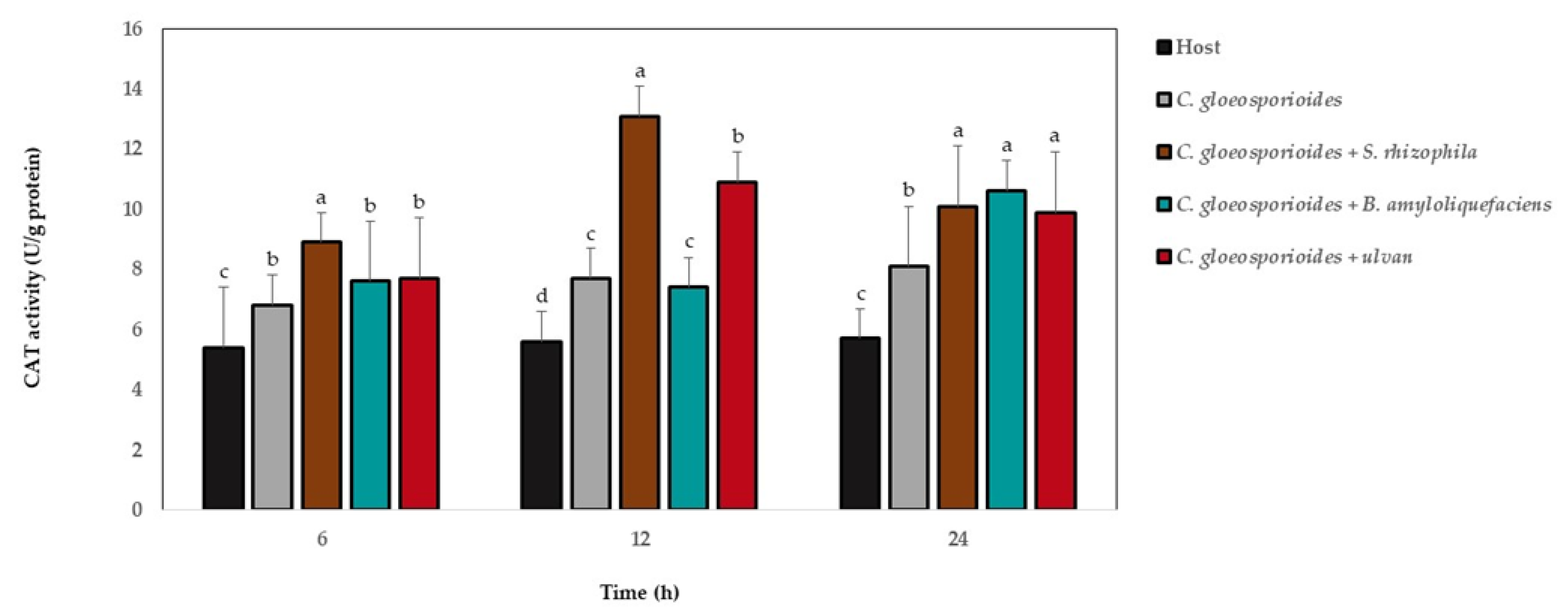
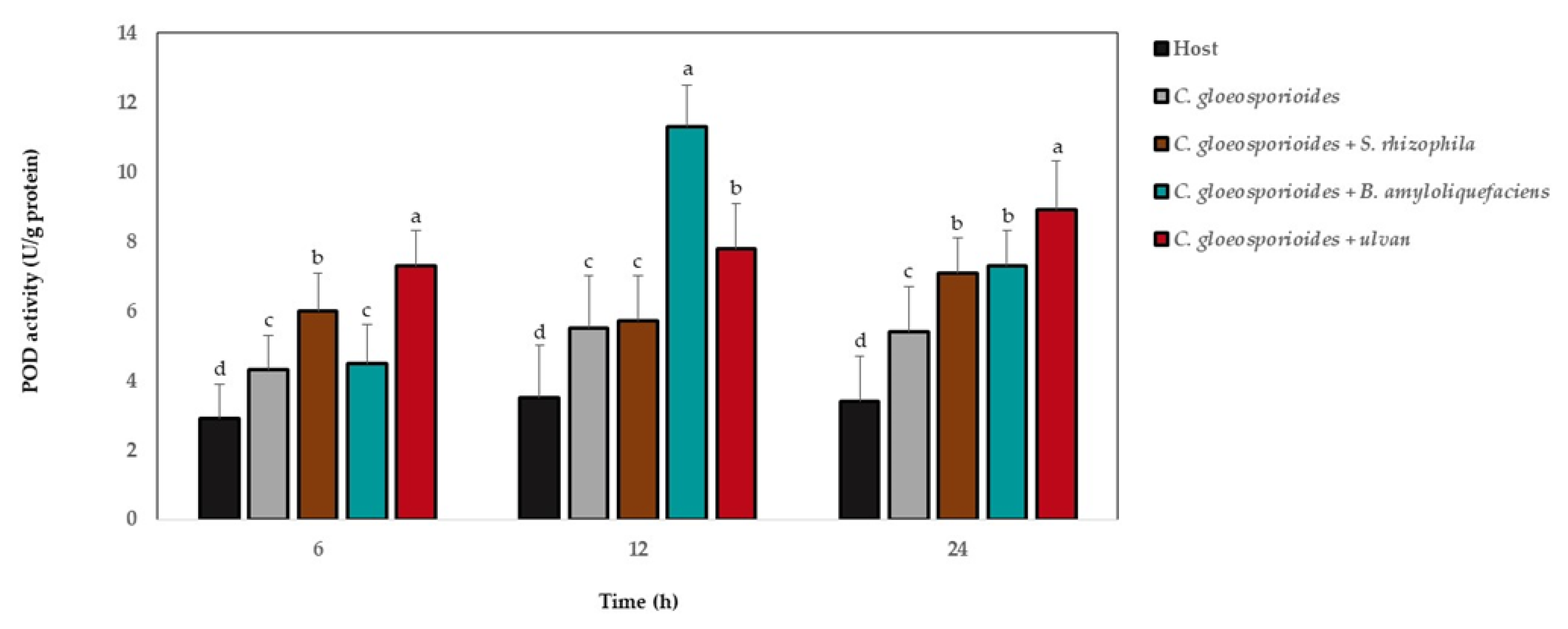
3.6. Antioxidant Defense Enzyme Activity in Papaya Fruit Inoculated with Marine Bacteria and Ulvan
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Landero, V.N.; Nieto, A.D.; Téliz, O.D.; Alatorre, R.R.; Ortiz, G.C.F.; Orozco, S.M. Biological control of anthracnose by postharvest application of Trichoderma spp., on Maradol papaya fruit. Biol. Control 2015, 91, 88–93. [Google Scholar] [CrossRef]
- Lima, G.; Sanzani, S.M.; Curtis, F.; Ippolito, A. Biological control of postharvest diseases. In Advances in Postharvest Fruit and Vegetables Technology; Wills, R.B.H., Golding, J.B., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 65–81. [Google Scholar]
- Hu, Y.; Chen, J.; Hu, G.; Yu, J.; Zhu, X.; Lin, Y.; Chen, S.; Yuan, J. Statistical research on the bioactivity of new marine natural products discovered during the 28 years from 1985 to 2012. Mar. Drugs 2015, 13, 202–221. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Noh, N.M.; Mustafa, M.A. Antimicrobial activity of chitosan enriched with lemongrass oil against anthracnose of bell pepper. Food Packag. Shelf Life 2015, 3, 56–61. [Google Scholar] [CrossRef]
- Kim, K.H.; Kabir, E.; Jahan, S.A. Exposure to pesticides and the associated human health effects. Sci. Total Environ. 2017, 575, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Spadaro, D.; Droby, S. Development of biocontrol products for postharvest diseases of fruit: The importance of elucidating the mechanisms of action of yeast antagonists. Trends Food Sci. Technol. 2016, 47, 39–49. [Google Scholar] [CrossRef]
- Torres, M.J.; Pérez, B.C.; Petroselli, G.; Erra-Balsells, R.; Audisio, M.C. Antagonistic effects of Bacillus subtilis subsp. subtilis and B. amyloliquefaciens against Macrophomina phaseolina: SEM study of fungal changes and UV-MALDI-TOF MS analysis of their bioactive compounds. Microbiol. Res. 2016, 182, 31–39. [Google Scholar] [CrossRef]
- Zhang, S.; Zheng, Q.; Xu, B.; Liu, J. Identification of the fungal pathogens of postharvest disease on peach fruits and the control mechanisms of Bacillus subtilis JK-14. Toxins 2019, 11, 322. [Google Scholar] [CrossRef]
- Teixeira, B.; Marques, A.; Ramos, C.; Neng, R.N.; Nogueira, M.F.J.; Saravia, A.J.; Nunes, L.M. Chemical compositions and antibacterial and antioxidant properties of comercial essential oils. Ind. Crops Prod. 2013, 43, 587–595. [Google Scholar] [CrossRef]
- Bibi, F.; Imran, N.M.; Mohamed, H.A.; Yasir, M.; Khalaf, G.A.A.; Ibrahin, A.E. LC-MS based identification of secondary metabolites from marine antagonistic endophytic bacteria. Genet. Mol. Res. 2017, 17, 1–14. [Google Scholar] [CrossRef]
- Bibi, F.; Imran, N.M.; Mohamed, H.A.; Yasir, M.; Khalaf, G.A.A.; Ibrahin, A.E. Diversity and antagonistic potential of bacteria isolated from marine grass Halodule uninervis. Biotechnology 2018, 8, 48. [Google Scholar] [CrossRef]
- Aiello, D.; Restuccia, C.; Stefani, E.; Vitale, A.; Cirvilleri, G. Postharvest biocontrol ability of Pseudomonas synxantha against Monilinia fructicola and Monilinia fructigena on stone fruit. Postharvest Biol. Technol. 2019, 149, 83–89. [Google Scholar] [CrossRef]
- Hernandez-Montiel, L.G.; Zulueta, R.R.; Angulo, C.; Rueda, P.E.O.; Quiñonez, A.E.E.; Galicia, R. Marine yeasts and bacteria as biological control agents against anthracnose on mango. J. Phytopathol. 2017, 165, 833–840. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Y.; Fu, X.; Li, Y.; Wang, Q. Isolation and characterization of Bacillus amyloliquefaciens PG12 for the biological control of apple ring rot. Postharvest Biol. Technol. 2016, 115, 113–121. [Google Scholar] [CrossRef]
- Carmona-Hernandez, S.; Reyes-Pérez, J.J.; Chiquito-Contreras, R.G.; Rincon-Enriquez, G.; Cerdan-Cabrera, C.R.; Hernandez-Montiel, L.G. Biocontrol of postharvest fruit fungal diseases by bacterial antagonists: A review. Agronomy 2019, 9, 121. [Google Scholar] [CrossRef]
- Kolsi, R.B.A.; Gargouri, B.; Sassi, S.; Frikha, D.; Lassoued, S.; Belghith, K. In vitro biological propierties and health benedits of a novel sulfated polysaccharide isolated from Cymodocea nodosa. Lipids Health Dis. 2017, 16, 1–11. [Google Scholar] [CrossRef]
- Stadnik, M.J.; Freitas, M.B. Algal polysaccharide as source of plant resistance inducers. Trop. Plant Pathol. 2014, 39, 111–118. [Google Scholar] [CrossRef]
- Pankiewicz, R.; Leska, B.; Messyasz, B.; Fabrowska, J.; Soloducha, M.; Pikosz, M. First isolation of polysaccharides ulvans from the cell walls of freshwater algae. Algal Res. 2016, 19, 348–354. [Google Scholar] [CrossRef]
- Berri, M.; Slugocki, C.; Oliver, M.; Helloin, E.; Jacques, I.; Salmon, H.; Le Goff, M.; Collen, P.N. Marine sulfated polysaccharides extract of Ulva armoricana green algae exhibits an antimicrobial activity and stimulates cytokine expression by intestinal epithelial cell. J. Appl. Phycol. 2016, 28, 2999–3008. [Google Scholar] [CrossRef]
- Gadenne, V.; Lebrun, L.; Jouenne, T.; Thebault, P. Antiadhesive activity of ulvan polysaccharides covalently immobilized onto titanium surface. Colloid Surf. B 2013, 112, 229–236. [Google Scholar] [CrossRef]
- Tuney, İ.; Cadirci, B.H.; Dilek, Ü.; Sukatar, A. Antimicrobial activities of the extracts of marine algae from the coast of Urla (Izmir, Turkey). Turk. J. Biol. 2006, 30, 171–175. [Google Scholar]
- Esquer-Miranda, E.; Nieves-Soto, M.; Rivas-Vega, M.E.; Miranda-Baeza, A.; Pi, P. Effects of methanolic macroalgae extracts from Caulerpa sertularioides and Ulva lactuca on Litopenaeus vannamei survival in the presence of Vibrio bacteria. Fish Shellfish Immunol. 2016, 51, 346–350. [Google Scholar] [CrossRef]
- Yaich, H.; Garna, H.; Bchir, B.; Besbes, S.; Paquot, M.; Richel, A.; Blecker, C.; Attia, H. Chemical composition and functional properties of dietary fibre extracted by Englyst and Prosky methods from the alga Ulva lactuca collected in Tunisia. Algal Res. 2015, 9, 65–73. [Google Scholar] [CrossRef]
- Spavieri, J.; Kaiser, M.; Casey, R.; Hingley-Wilson, S.; Lalvani, A.; Blunden, G.; Tasdemir, D. Antiprotozoal, antimycobacterial and cytotoxic potential of some British green algae. Phytother. Res. 2010, 24, 1095–1098. [Google Scholar] [CrossRef]
- Abbassy, M.A.; Marzouk, M.A.; Rabea, E.I.; Abd-Elnabi, A.D. Insecticidal and fungicidal activity of Ulva lactuca Linnaeus (Chlorophyta) extracts and their fractions. Annu. Res. Rev. Biol. 2014, 4, 2252–2262. [Google Scholar] [CrossRef]
- Margret, R.J.; Kumaresan, S.; Ravikumar, S. A preliminary study on the anti-inflammatory activity of methanol extract of Ulva lactuca in rat. J. Environ. Biol. 2009, 30, 899–902. [Google Scholar]
- Mao, W.; Zang, X.; Li, Y.; Zhang, H. Sulfated polysaccharides from marine green algae Ulva conglobata and their anticoagulant activity. J. Appl. Phycol. 2006, 18, 9–14. [Google Scholar] [CrossRef]
- Hernández-Herrera, R.M.; Santacruz-Ruvalcaba, F.; Zañudo-Hernández, J.; Hernández-Carmona, G. Activity of seaweed extracts and polysaccharide-enriched extracts from Ulva lactuca and Padina gymnospora as growth promoters of tomato and mung bean plants. J. Appl. Phycol. 2016, 28, 2549–2560. [Google Scholar] [CrossRef]
- Abdel-Latif, H.H.; Shams El-Din, N.G.; Ibrahim, H.A. Antimicrobial activity of the newly recorded red alga Grateloupia doryphora collected from the Eastern Harbor, Alexandria, Egypt. J. Appl. Microbiol. 2018, 125, 1321–1332. [Google Scholar] [CrossRef]
- Barot, M.; Kumar, N.J.I.; Kumar, R.N. Bioactive compounds and antifungal activity of three different seaweed species Ulva lactuca, Sargassum tenerrimum and Laurencia obtusa collected from Okha coast, Western India. J. Coast. Life Med. 2016, 4, 284–289. [Google Scholar] [CrossRef]
- Araújo, L.; Stadnik, M.J. Cultivar-specific and ulvan-induced resistance of appleplants to Glomerella leaf spot are associated with enhanced activity of peroxi-dases. Acta Sci. Agron. 2013, 35, 287–293. [Google Scholar] [CrossRef]
- Rivas-Garcia, T.; Murillo-Amador, B.; Nieto-Garibay, A.; Chiquito-Contreras, R.; Rincon-Enriquez, G.; Hernandez-Montiel, L.G. Effect of ulvan on the biocontrol activity of Debaryomyces hansenii and Stenotrophomonas rhizophila against fruit rot of Cucumis melo L. Agronomy 2018, 8, 273. [Google Scholar] [CrossRef]
- Cluzet, S.; Torregrosa, C.; Jacquet, C.; Lafitte, C.; Fournier, J.; Mercier, L.; Salamagne, S.; Briand, X.; Esquerre-Tugaye, M.T.; Dumas, B. Gene expression profiling and protection of Medicago truncatula against a fungal infection in response to an elicitor from green algae Ulva spp. Plant Cell Environ. 2004, 27, 917–928. [Google Scholar] [CrossRef]
- Derksen, H.; Rampitsch, C.; Daayf, F. Signaling cross-talk in plant disease resistance. Plant Sci. 2013, 207, 79–87. [Google Scholar] [CrossRef]
- Abouraïcha, E.; El Alaoui-Talibi, Z.; El Boutachfaiti, R.; Petit, E.; Courtois, B.; Courtois, J.; El Modafar, C. Induction of natural defense and protection against Penicillium expansum and Botrytis cinerea in apple fruit in response to bioelicitors isolated from green algae. Sci. Hortic. 2015, 181, 121–128. [Google Scholar] [CrossRef]
- Hernandez-Montiel, L.G.; Gutierrez-Perez, E.D.; Murillo-Amador, B.; Vero, S.; Chiquito-Contreras, R.G.; Rincon-Enriquez, G. Mechanisms employed by Debaryomyces hansenii in biological control of anthracnose diseases on papaya fruit. Postharvest Biol. Technol. 2018, 139, 31–37. [Google Scholar] [CrossRef]
- Ezziyyani, M.; Pérez-Sánchez, C.; Requena, M.E.; Rubio, L.; Candela, M.E. Biocontrol por Streptomyces rochei-Ziyani-, de la podredumbre del pimiento (Capsicum annuum L.) causada por Phytophthora capsici. Anal. Biol. 2004, 26, 69–78. [Google Scholar]
- Rouissi, W.; Ugolini, L.; Martini, C.; Lazzeri, L.; Mari, M. Control of postharvest fungal pathogens by antifungal compounds from Penicillium expansum. J. Food Prot. 2013, 76, 1879–1886. [Google Scholar] [CrossRef]
- Hedge, J.E.; Hofreiter, B.T. Carbohydrate Chemistry; Whistler, R.L., Be Miller, J.N., Eds.; Academic Press: New York, NY, USA, 1962. [Google Scholar]
- Bruner, L.R. Determination of reducing value: 3,5 Dinitrosalicylic acid method. In Methods in Carbohydrate Chemistry; Whistler, R.L., Smith, R.J., BeMiller, J.N., Eds.; Academic Press: New York, NY, USA, 1964; Volume 4, p. 6771. [Google Scholar]
- Barham, D.; Trinder, P. An improved colour reagent for the determination of blood glucose by the oxidase system. Analyst 1972, 97, 142–145. [Google Scholar] [CrossRef]
- Taylor, K.A. A colorimetric fructose assay. Appl. Biochem. Biotechnol. 1995, 53, 215–227. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of micro-gram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Wang, Y.S.; Tian, S.P.; Xu, Y.; Qin, G.Z.; Yao, H.J. Changes in the activities of protein and antioxidant enzymes in peach fruit inoculated with Cryptococcus laurentii or Penicillium expansum at 0 or 20 C. Postharvest Biol. Technol. 2004, 34, 21–28. [Google Scholar] [CrossRef]
- Reverberi, M.; Fabbri, A.A.; Zjalic, S.; Ricelli, A.; Punelli, F.; Fanelli, C. Antioxidant enzymes stimulation in Aspergillus parasiticus by Lentinula edodes inhibits aflatoxin production. Appl. Microbiol. Biotechnol. 2005, 69, 207–215. [Google Scholar] [CrossRef]
- Yao, H.J.; Tian, S.P. Effect of pre- and post-harvest application of salicylic acid or methyl jasmonate on inducing disease resistance of sweet cherry fruit in storage. Postharvest Biol. Technol. 2005, 35, 253–262. [Google Scholar] [CrossRef]
- Jiang, C.H.; Liao, M.J.; Wang, H.K.; Zheng, M.Z.; Xu, J.J.; Guo, J.H. Bacillus velezensis, a potential and efficient biocontrol agent in control of pepper gray mold caused by Botrytis cinerea. Biol. Control 2018, 126, 147–157. [Google Scholar] [CrossRef]
- Jakobi, M.; Winkelmann, G.; Kaiser, D.; Kempter, C.; Jung, G.; Berg, G.; Bahl, H. Maltophilin, a new antifungal compound produced by Stenotrophomonas maltophilia R3089. J. Antibiot. 1996, 49, 1101–1104. [Google Scholar] [CrossRef]
- Hashidoko, Y.; Nakayama, T.; Homma, Y.; Tahara, S. Structure elucidation of xanthobaccin A, a new antibiotic produced from Stenotrophomonas sp. strain SB-K88. Tetrahedron Lett. 1999, 40, 2957–2960. [Google Scholar] [CrossRef]
- Nakayama, T.; Homma, Y.; Hashidoko, Y.; Mizutani, J.; Tahara, S. Possible Role of Xanthobaccins produced by Stenotrophomonas sp. strain SB-K88 in suppression of sugar beet damping-off disease. Appl. Environ. Microbiol. 1999, 65, 4334–4339. [Google Scholar]
- Reyes-Perez, J.J.; Hernandez-Montiel, L.G.; Vero, S.; Noa-Carrazana, J.C.; Quiñones-Aguilar, E.E.; Rincón-Enríquez, G. Postharvest biocontrol of Colletotrichum gloeosporioides on mango using the marine bacterium Stenotrophomonas rhizophila and its possible mechanisms of action. J. Food Sci. Technol. 2019, 56, 4992–4999. [Google Scholar] [CrossRef]
- Greenshields, L.D.; Guosheng, L.; Feng, J.; Selvaraj, G.; Wei, Y. The siderophore biosynthetic gene SID1, but not the ferroxidase gene FET3, is required for full Fusarium graminearum virulence. Mol. Plant Pathol. 2007, 8, 411–421. [Google Scholar] [CrossRef]
- Chandra, H.; Bishnoi, P.; Yadav, A.; Patni, B.; Mishra, A.; Nautiyal, A. Antimicrobial resistance and the alternative resources with special emphasis on plant-based antimicrobials-a review. Plants 2017, 6, 16. [Google Scholar] [CrossRef]
- Shen, L.; Wang, F.; Liu, Y.; Qian, Y.; Yang, J.; Sun, H. Suppression of tobacco mosaic virus by Bacillus amyloliquefaciens strain Ba33. J. Phytopathol. 2013, 161, 293–294. [Google Scholar] [CrossRef]
- Yang, P.; Sun, Z.X.; Liu, S.Y.; Lu, H.X.; Zhou, Y.; Sun, M. Combining antagonistic endophytic bacteria in different growth stages of cotton for control of Verticillium wilt. Crop Prot. 2013, 47, 17–23. [Google Scholar] [CrossRef]
- Blom, D.; Fabbri, C.; Connor, E.C.; Schiestl, F.P.; Klauser, D.R.; Boller, T.; Eberl, L.; Weisskopf, L. Production of plant growth modulating volatiles is widespread among rhizosphere bacteria and strongly depends on culture conditions. Environ. Microbiol. 2011, 13, 3047–3058. [Google Scholar] [CrossRef] [PubMed]
- Garbeva, P.; Hordijk, C.; Gerards, S.; de Boer, W. Volatile mediated interactions between phylogenetically different soil bacteria. Front. Microbiol. 2014, 5, 289. [Google Scholar] [CrossRef]
- Asari, S.; Matzén, S.; Petersen, M.A.; Bejai, S.; Meijer, J. Multiple effects of Bacillus amyloliquefaciens volatile compounds: Plant growth promotion and growth inhibition of phytopathogens. FEMS Microbiol. Ecol. 2016, 92, 1–11. [Google Scholar] [CrossRef]
- Syed-Ab-Rahman, S.F.; Omar, D. Development of bio-formulations of Piper sarmentosum extracts against bacterial rice diseases. Curr. Biotechnol. 2018, 7, 453–463. [Google Scholar] [CrossRef]
- Ongena, M.; Jacques, P. Bacillus lipopeptides: Versatile weapons for plant disease biocontrol. Trends Microbiol. 2008, 16, 115–125. [Google Scholar] [CrossRef]
- Dietel, K.; Beator, B.; Budiharjo, A.; Fan, B.; Borriss, R. Bacterial traits involved in colonization of Arabidopsis thaliana roots by Bacillus amyloliquefaciens FZB42. Plant Pathol. J. 2013, 29, 59–66. [Google Scholar] [CrossRef]
- Debois, D.; Jourdan, E.; Smargiasso, N.; Thonart, P.; De Pauw, E.; Ongena, M. Spatiotemporal monitoring of the antibiome secreted by Bacillus biofilms on plant roots using MALDI mass spectrometry imaging. Anal. Chem. 2014, 86, 4431–4438. [Google Scholar] [CrossRef]
- Liu, J.; Sui, Y.; Wisniewski, M.; Droby, S.; Liu, Y. Review: Utilization of antagonistic yeast to manage postharvest fungal. Int. J. Food Microbiol. 2013, 167, 153–160. [Google Scholar] [CrossRef]
- Côté, F.; Ham, K.S.; Hahn, M.G.; Bergmann, C.W. Oligosaccharide elicitors in host–pathogen interactions. Generation, perception, and signal transduction. In Subcellular Biochemistry; Plenum Publishing Corp: New York, NY, USA, 1998; pp. 385–432. [Google Scholar]
- Campo, S.; Carrascal, M.; Coca, M.; Abián, J.; San Segundo, B. The defense response of germinating maize embryos against fungal infection: A proteomics approach. Proteomics 2004, 4, 383–396. [Google Scholar] [CrossRef]
- Xu, X.B.; Tian, S.P. Reducing oxidative stress in sweet cherry fruit by Pichia membranaefaciens: A possible mode of action against Penicillium expansum. J. Appl. Microbiol. 2008, 105, 1170–1177. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 5th ed.; Oxford University Press: London, UK, 1999; p. 905. [Google Scholar]
- Hernandez, J.A.; Ferrer, M.A.; Jimenez, A.; Barcelo, A.R.; Sevilla, F. Antioxidant systems and O2−⁄H2O2 production in the apoplast of pea leaves. Its relation with salt-induced necrotic lesions in minor veins. Plant Physiol. 2001, 127, 827–831. [Google Scholar] [CrossRef]
- Asada, K. Ascorbate peroxidase: A hydrogen peroxidescavenging enzyme in plants. Plant Physiol. 1992, 85, 235–241. [Google Scholar] [CrossRef]
- Borsani, O.; Valpuesta, V.; Botella, M.A. Evidence for a role of salicylic acid in the oxidative damage generated by NaCl and osmotic stress in Arabidopsis seedlings. Plant Physiol. 2001, 126, 1024–1030. [Google Scholar] [CrossRef]
- Foyer, C.H.; Trebst, A.; Noctor, G. Signaling and integration of defense functions of tocopherol, ascorbate and glutathione. In Photoprotection, Photoinhibition, Gene Regulation, and Environment; Demmig-Adams, B., Adams, W.W., Mattoo, A.K., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 241–268. [Google Scholar]
- Zhao, P.; Li, P.; Wu, S.; Zhou, M.; Zhi, R.; Gao, H. Volatile organic compounds (VOCs) from Bacillus subtilis CF-3 reduce anthracnose and elicit active defense responses in harvested litchi fruits. AMB Express 2019, 9, 119. [Google Scholar] [CrossRef]
- Wang, X.; Wang, L.; Wang, J.; Jin, P.; Liu, H.; Zheng, Y. Bacillus cereus AR156-induced resistance to Colletotrichum acutatum is associated with priming of defense responses in loquat fruit. PLoS ONE 2014, 9, e112494. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiquito-Contreras, R.G.; Murillo-Amador, B.; Carmona-Hernandez, S.; Chiquito-Contreras, C.J.; Hernandez-Montiel, L.G. Effect of Marine Bacteria and Ulvan on the Activity of Antioxidant Defense Enzymes and the Bio-Protection of Papaya Fruit against Colletotrichum gloeosporioides. Antioxidants 2019, 8, 580. https://doi.org/10.3390/antiox8120580
Chiquito-Contreras RG, Murillo-Amador B, Carmona-Hernandez S, Chiquito-Contreras CJ, Hernandez-Montiel LG. Effect of Marine Bacteria and Ulvan on the Activity of Antioxidant Defense Enzymes and the Bio-Protection of Papaya Fruit against Colletotrichum gloeosporioides. Antioxidants. 2019; 8(12):580. https://doi.org/10.3390/antiox8120580
Chicago/Turabian StyleChiquito-Contreras, Roberto G., Bernardo Murillo-Amador, Saul Carmona-Hernandez, Cesar J. Chiquito-Contreras, and Luis G. Hernandez-Montiel. 2019. "Effect of Marine Bacteria and Ulvan on the Activity of Antioxidant Defense Enzymes and the Bio-Protection of Papaya Fruit against Colletotrichum gloeosporioides" Antioxidants 8, no. 12: 580. https://doi.org/10.3390/antiox8120580
APA StyleChiquito-Contreras, R. G., Murillo-Amador, B., Carmona-Hernandez, S., Chiquito-Contreras, C. J., & Hernandez-Montiel, L. G. (2019). Effect of Marine Bacteria and Ulvan on the Activity of Antioxidant Defense Enzymes and the Bio-Protection of Papaya Fruit against Colletotrichum gloeosporioides. Antioxidants, 8(12), 580. https://doi.org/10.3390/antiox8120580





