Mycoredoxins Are Required for Redox Homeostasis and Intracellular Survival in the Actinobacterial Pathogen Rhodococcus equi
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains, Culture Conditions, and Genetic Engineering
2.2. Macrophage Infection Assays
2.3. In Vivo Quantification of Redox Homeostasis
2.4. Quantification of the Redox Potential
2.5. Sensitivity Assays to Oxidative Stressors
2.6. Statistical Analyses
3. Results and Discussion
3.1. Identification of R. equi Mycoredoxins
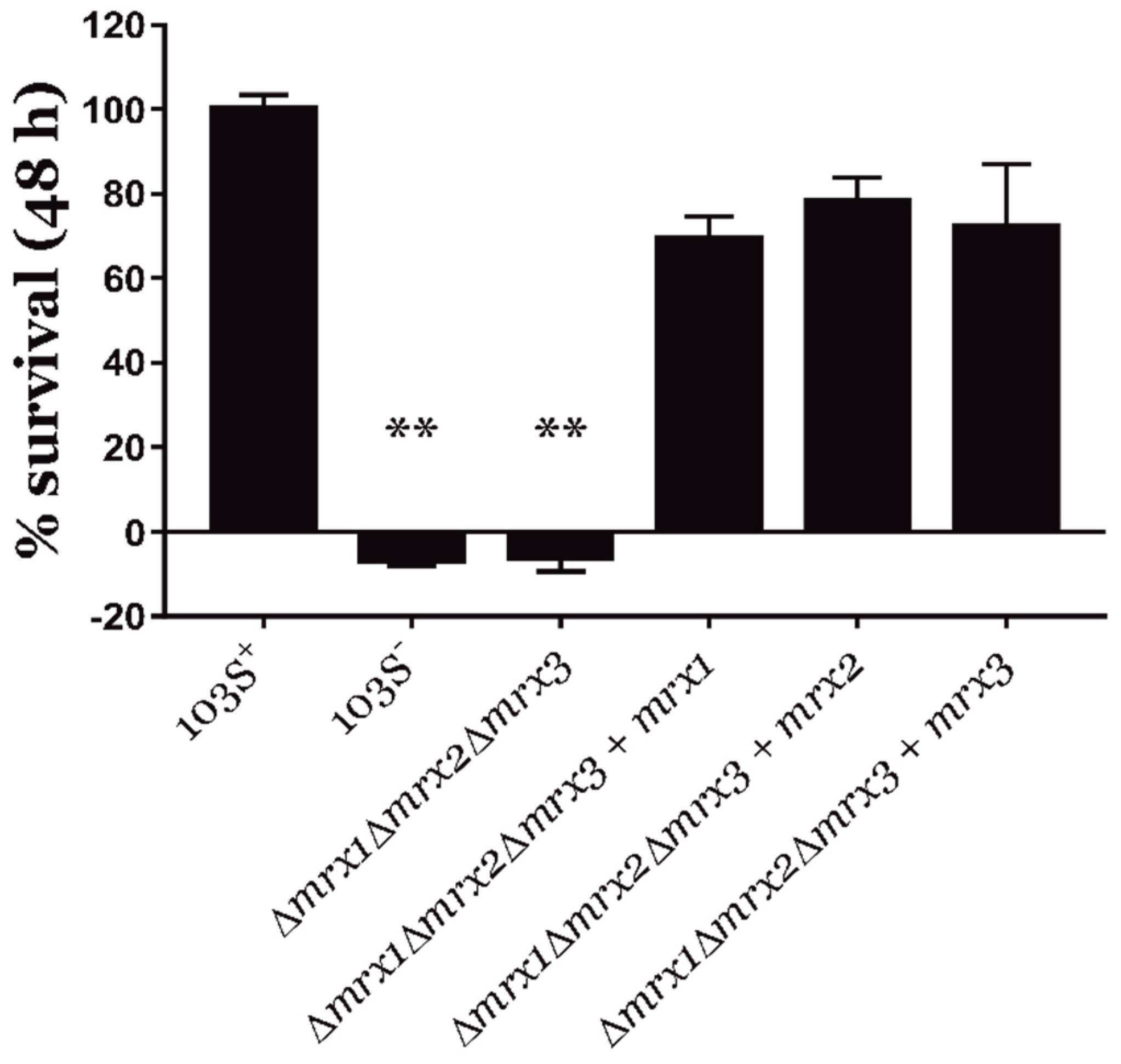
3.2. Mycoredoxins Are Essential for the Intracellular Survival of R. equi in Macrophages
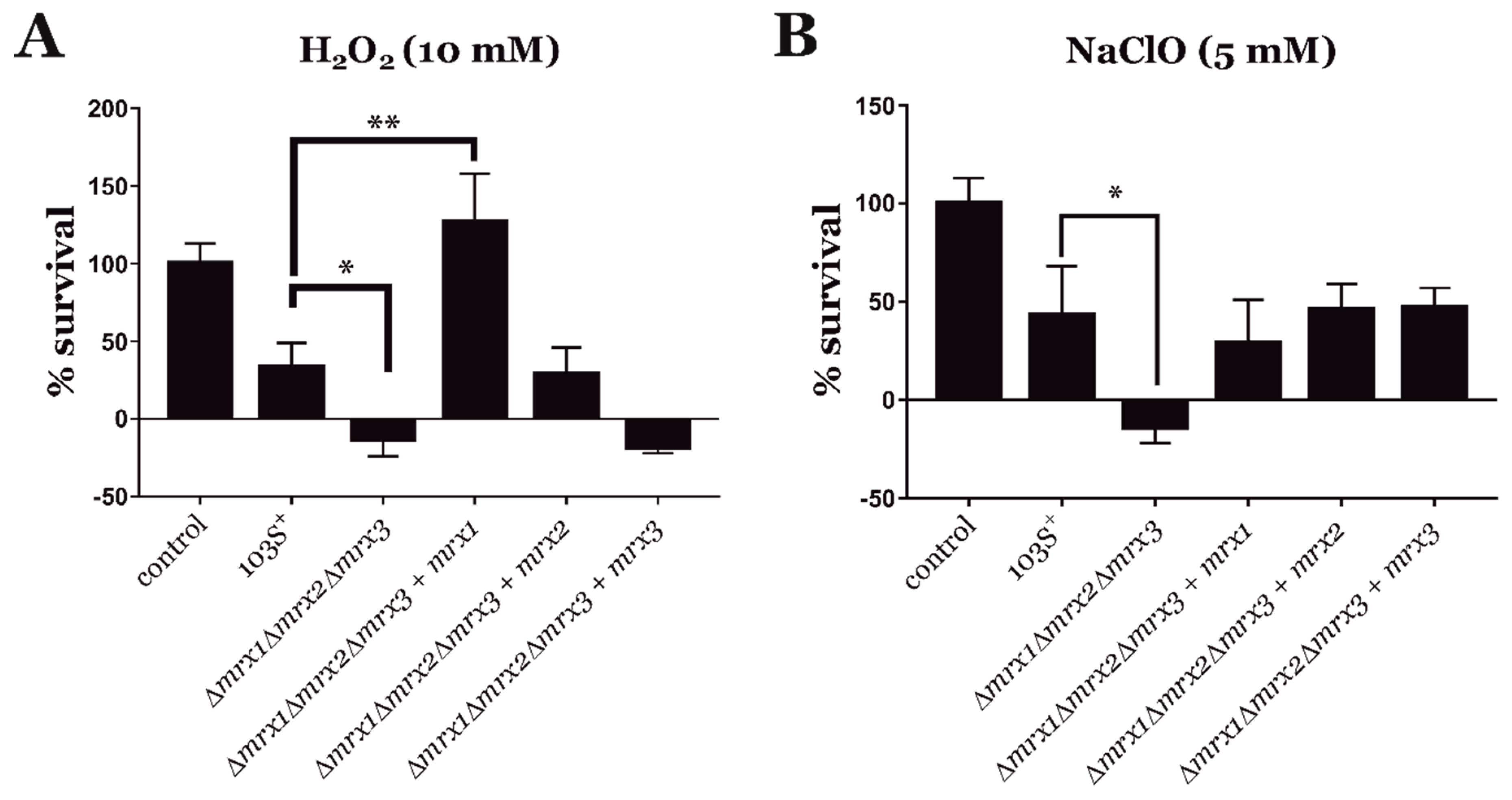
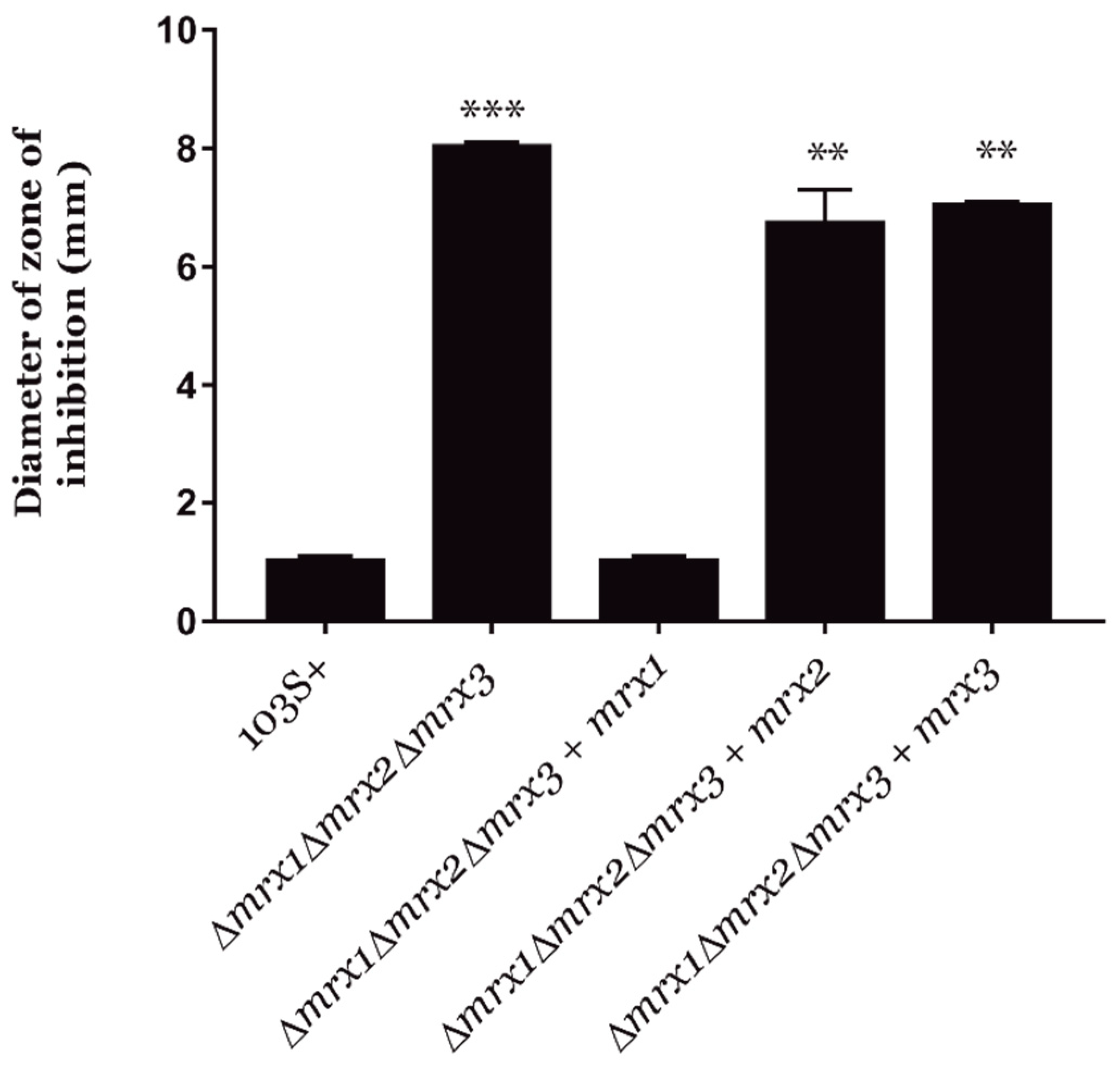
3.3. Mycoredoxins Are Essential for Redox Homeostasis in R. equi
3.4. Analysis of the Redox State of R. equi Mycoredoxins during Oxidative Stress and Host Cell Infection
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vázquez-Boland, J.A.; Meijer, W.G. The pathogenic actinobacterium Rhodococcus equi: What’s in a name? Mol. Microbiol. 2019, 112, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Boland, J.A.; Giguère, S.; Hapeshi, A.; MacArthur, I.; Anastasi, E.; Valero-Rello, A. Rhodococcus equi: The many facets of a pathogenic actinomycete. Vet. Microbiol. 2013, 167, 9–33. [Google Scholar] [CrossRef] [PubMed]
- Weinstock, D.M.; Brown, A.E. Rhodococcus equi: An emerging pathogen. Clin. Infect. Dis. 2002, 34, 1379–1385. [Google Scholar] [CrossRef] [PubMed]
- Tuon, F.F.; Siciliano, R.F.; Al-Musawi, T.; Rossi, F.; Capelozzi, V.L.; Gryschek, R.C.; Medeiros, E.A.S. Rhodococcus equi bacteremia with lung abscess misdiagnosed as Corynebacterium: A report of 2 cases. Clinics 2007, 62, 795–798. [Google Scholar] [CrossRef]
- Yamshchikov, A.V.; Schuetz, A.; Lyon, G.M. Rhodococcus equi infection. Lancet Infect. Dis. 2010, 10, 350–359. [Google Scholar] [CrossRef]
- Muscatello, G.; Anderson, G.A.; Gilkerson, J.R.; Browning, G.F. Associations between the ecology of virulent Rhodococcus equi and the epidemiology of R. equi pneumonia on Australian thoroughbred farms. Appl. Environ. Microbiol. 2006, 72, 6152–6160. [Google Scholar] [CrossRef]
- Muscatello, G.; Gerbaud, S.; Kennedy, C.; Gilkerson, J.R.; Buckley, T.; Klay, M.; Leadon, D.P.; Browning, G.F. Comparison of concentrations of Rhodococcus equi and virulent R. equi in air of stables and paddocks on horse breeding farms in a temperate climate. Equine Vet. J. 2006, 38, 263–265. [Google Scholar] [CrossRef]
- Giguère, S.; Berghaus, L.J.; Willingham-Lane, J.N. Antimicrobial resistance in Rhodococcus equi. Microbiol. Spectrum. 2017, 5. [Google Scholar] [CrossRef]
- Cisek, A.A.; Rzewuska, M.; Witkowski, L.; Binek, M. Antimicrobial resistance in Rhodococcus equi. Acta Biochim. Pol. 2014, 61, 633–638. [Google Scholar] [CrossRef]
- Duchesne, R.; Castagnet, S.; Maillard, K.; Petry, S.; Cattoir, V.; Giard, J.-C.; Leon, A. In vitro antimicrobial susceptibility of equine clinical isolates from France, 2006–2016. J. Glob. Antimicrob. Resist. 2019, 19, 144–153. [Google Scholar] [CrossRef]
- Huber, L.; Giguère, S.; Cohen, N.D.; Slovis, N.M.; Berghaus, L.; Greiter, M.; Hart, K.A. Identification of macrolide- and rifampicin-resistant Rhodococcus equi in environmental samples from equine breeding farms in central Kentucky during 2018. Vet. Microbiol. 2019, 232, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Valero-Rello, A.; Hapeshi, A.; Anastasi, E.; Alvarez, S.; Scortti, M.; Meijer, W.G.; MacArthur, I.; Vázquez-Boland, J.A. An invertron-like linear plasmid mediates intracellular survival and virulence in bovine isolates of Rhodococcus equi. Infect. Immun. 2015, 83, 2725–2737. [Google Scholar] [CrossRef] [PubMed]
- Letek, M.; Ocampo-Sosa, A.A.; Sanders, M.; Fogarty, U.; Buckley, T.; Leadon, D.P.; González, P.; Scortti, M.; Meijer, W.G.; Parkhill, J.; et al. Evolution of the Rhodococcus equi vap pathogenicity island seen through comparison of host-associated vapA and vapB virulence plasmids. J. Bacteriol. 2008, 190, 5797–5805. [Google Scholar] [CrossRef] [PubMed]
- von Bargen, K.; Scraba, M.; Krämer, I.; Ketterer, M.; Nehls, C.; Krokowski, S.; Repnik, U.; Wittlich, M.; Maaser, A.; Zapka, P.; et al. Virulence-associated protein A from Rhodococcus equi is an intercompartmental pH-neutralising virulence factor. Cell. Microbiol. 2019, 21, e12958. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.T.; Herron, L.L.; Kapur, V.; Meijer, W.G.; Byrne, B.A.; Ren, J.; Nicholson, V.M.; Prescott, J.F. Partial genome sequencing of Rhodococcus equi ATCC 33701. Vet. Microbiol. 2003, 94, 143–158. [Google Scholar] [CrossRef]
- Anastasi, E.; MacArthur, I.; Scortti, M.; Alvarez, S.; Giguére, S.; Vázquez-Boland, J.A. Pangenome and phylogenomic analysis of the pathogenic actinobacterium Rhodococcus equi. Genome Biol. Evol. 2016, 8, 3140–3148. [Google Scholar] [CrossRef]
- Letek, M.; González, P.; MacArthur, I.; Rodríguez, H.; Freeman, T.C.; Valero-Rello, A.; Blanco, M.; Buckley, T.; Cherevach, I.; Fahey, R.; et al. The genome of a pathogenic Rhodococcus: Cooptive virulence underpinned by key gene acquisitions. PLoS Genet. 2010, 6, e1001145. [Google Scholar] [CrossRef]
- Miranda-CasoLuengo, R.; Coulson, G.B.; Miranda-CasoLuengo, A.; Vázquez-Boland, J.A.; Hondalus, M.K.; Meijer, W.G. The hydroxamate siderophore rhequichelin is required for virulence of the pathogenic actinomycete Rhodococcus equi. Infect. Immun. 2012, 80, 4106–4114. [Google Scholar] [CrossRef]
- Van Der Geize, R.; Grommen, A.W.F.; Hessels, G.I.; Jacobs, A.A.C.; Dijkhuizen, L. The steroid catabolic pathway of the intracellular pathogen Rhodococcus equi is important for pathogenesis and a target for vaccine development. PLoS Pathog. 2011, 7, e1002181. [Google Scholar] [CrossRef]
- Darrah, P.A.; Hondalus, M.K.; Chen, Q.; Ischiropoulos, H.; Mosser, D.M. Cooperation between reactive oxygen and nitrogen intermediates in killing of Rhodococcus equi by activated macrophages. Infect. Immun. 2000, 68, 3587–3593. [Google Scholar] [CrossRef]
- Fang, F.C. Antimicrobial reactive oxygen and nitrogen species: Concepts and controversies. Nat. Rev. Microbiol. 2004, 2, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Benoit, S.; Benachour, A.; Taouji, S.; Auffray, Y.; Hartke, A. H2O2, which causes macrophage-related stress, triggers induction of expression of virulence-associated plasmid determinants in Rhodococcus equi. Infect. Immun. 2002, 70, 3768–3776. [Google Scholar] [CrossRef] [PubMed]
- Haas, A. The phagosome: Compartment with a license to kill. Traffic 2007, 8, 311–330. [Google Scholar] [CrossRef] [PubMed]
- Chandramani-Shivalingappa, P.; Bhandari, M.; Wiechert, S.; AGilbertie, J.; Jones, D.E.; Sponseller, B. Induction of reactive intermediates and autophagy-related proteins upon infection of macrophages with Rhodococcus equi. Scientifica 2017, 2017, 8135737. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Holmgren, A. The thioredoxin antioxidant system. Free Radic. Biol. Med. 2014, 66, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Ordóñez, E.; Van Belle, K.; Roos, G.; De Galan, S.; Letek, M.; Gil, J.A.; Wyns, L.; Mateos, L.M.; Messens, J. Arsenate reductase, mycothiol, and mycoredoxin concert thiol/disulfide exchange. J. Biol. Chem. 2009, 284, 15107–15116. [Google Scholar] [CrossRef]
- Rosado, L.A.; Wahni, K.; Degiacomi, G.; Pedre, B.; Young, D.; de la Rubia, A.G.; Boldrin, F.; Martens, E.; Marcos-Pascual, L.; Sancho-Vaello, E.; et al. The antibacterial prodrug activator Rv2466c is a mycothiol-dependent reductase in the oxidative stress response of Mycobacterium tuberculosis. J. Biol. Chem. 2017, 292, 13097–13110. [Google Scholar] [CrossRef]
- Tung, Q.N.; Loi, V.V.; Busche, T.; Nerlich, A.; Mieth, M.; Milse, J.; Kalinowski, J.; Hocke, A.C.; Antelmann, H. Stable integration of the Mrx1-roGFP2 biosensor to monitor dynamic changes of the mycothiol redox potential in Corynebacterium glutamicum. Redox Biol. 2019, 20, 514–525. [Google Scholar] [CrossRef]
- Van Laer, K.; Buts, L.; Foloppe, N.; Vertommen, D.; Van Belle, K.; Wahni, K.; Roos, G.; Nilsson, L.; Mateos, L.M.; Rawat, M.; et al. Mycoredoxin-1 is one of the missing links in the oxidative stress defence mechanism of Mycobacteria. Mol. Microbiol. 2012, 86, 787–804. [Google Scholar] [CrossRef]
- Hugo, M.; Van Laer, K.; Reyes, A.M.; Vertommen, D.; Messens, J.; Radi, R.; Trujillo, M. Mycothiol/mycoredoxin 1-dependent reduction of the peroxiredoxin AhpE from Mycobacterium tuberculosis. J. Biol. Chem. 2014, 289, 5228–5239. [Google Scholar] [CrossRef]
- Bhaskar, A.; Chawla, M.; Mehta, M.; Parikh, P.; Chandra, P.; Bhave, D.; Kumar, D.; Carroll, K.S.; Singh, A. Reengineering Redox sensitive GFP to measure mycothiol Redox potential of Mycobacterium tuberculosis during infection. PLoS Pathog. 2014, 10, e1003902. [Google Scholar] [CrossRef] [PubMed]
- Negri, A.; Javidnia, P.; Mu, R.; Zhang, X.; Vendome, J.; Gold, B.; Roberts, J.; Barman, D.; Ioerger, T.; Sacchettini JCJiang, X.; et al. Identification of a mycothiol-dependent nitroreductase from Mycobacterium tuberculosis. ACS Infect. Dis. 2018, 4, 771–787. [Google Scholar] [CrossRef] [PubMed]
- Van Der Geize, R.; De Jong, W.; Hessels, G.I.; Grommen, A.W.F.; Jacobs, A.A.C.; Dijkhuizen, L. A novel method to generate unmarked gene deletions in the intracellular pathogen Rhodococcus equi using 5-fluorocytosine conditional lethality. Nucleic Acids Res. 2008, 36, e151. [Google Scholar] [CrossRef]
- Sekizaki, T.; Tanoue, T.; Osaki, M.; Shimoji, Y.; Tsubaki, S.; Takai, S. Improved electroporation of Rhodococcus equi. J. Vet. Med. Sci. 1998, 60, 277–279. [Google Scholar] [CrossRef][Green Version]
- Hong, Y.; Hondalus, M.K. Site-specific integration of Streptomyces ϕC31 integrase-based vectors in the chromosome of Rhodococcus equi. FEMS Microbiol. Lett. 2008, 287, 63–68. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hanson, G.T.; Aggeler, R.; Oglesbee, D.; Cannon, M.; Capaldi, R.A.; Tsien, R.Y.; Remington, S.J. Investigating mitochondrial redox potential with redox-sensitive Green Fluorescent Protein indicators. J. Biol. Chem. 2004, 279, 13044–13053. [Google Scholar] [CrossRef]
- Gutscher, M.; Pauleau, A.L.; Marty, L.; Brach, T.; Wabnitz, G.H.; Samstag, Y.; Meyer, A.J.; Dick, T.P. Real-time imaging of the intracellular glutathione redox potential. Nat. Methods 2008, 5, 553–559. [Google Scholar] [CrossRef]
- Adham, S.A.; Honrubia, P.; Díaz, M.; Fernández-Ábalos, J.M.; Santamaría, R.I.; Gil, J.A. Expression of the genes coding for the xylanase Xys1 and the cellulase Cel1 from the straw-decomposing Streptomyces halstedii JM8 cloned into the amino-acid producer Brevibacterium lactofermentum ATCC13869. Arch. Microbiol. 2001, 177, 91–97. [Google Scholar] [CrossRef]
- Bauer, A.W.; Kirby, W.M.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef]
- Carver, T.J.; Rutherford, K.M.; Berriman, M.; Rajandream, M.A.; Barrell, B.G.; Parkhill, J. ACT: The Artemis comparison tool. Bioinformatics 2005, 21, 3422–3423. [Google Scholar] [CrossRef]
- Bierman, M.; Logan, R.; O’Brien, K.; Seno, E.T.; Nagaraja Rao, R.; Schoner, B.E. Plasmid cloning vectors for the conjugal transfer of DNA from Escherichia coli to Streptomyces spp. Gene 1992, 116, 43–49. [Google Scholar] [CrossRef]
- Kasozi, D.; Mohring, F.; Rahlfs, S.; Meyer, A.J.; Becker, K. Real-time imaging of the intracellular glutathione redox potential in the malaria parasite Plasmodium falciparum. PLoS Pathog. 2013, 9, e1003782. [Google Scholar] [CrossRef] [PubMed]
- Schuh, A.K.; Rahbari, M.; Heimsch, K.C.; Mohring, F.; Gabryszewski, S.J.; Weder, S.; Buchholz, K.; Rahlfs, S.; Fidock, D.A.; Becker, K. Stable integration and comparison of hGrx1-roGFP2 and sfroGFP2 redox probes in the malaria parasite Plasmodium falciparum. ACS Infect. Dis. 2018, 4, 1601–1612. [Google Scholar] [CrossRef] [PubMed]
- Gutscher, M.; Sobotta, M.C.; Wabnitz, G.H.; Ballikaya, S.; Meyer, A.J.; Samstag, Y.; Dick, T.P. Proximity-based protein thiol oxidation by H2O2-scavenging peroxidases. J. Biol. Chem. 2009, 284, 31532–31540. [Google Scholar] [CrossRef] [PubMed]
- Morgan, B.; Van Laer, K.; Owusu, T.N.E.; Ezerina, D.; Pastor-Flores, D.; Amponsah, P.S.; Tursch, A.; Dick, T.P. Real-time monitoring of basal H2O2 levels with peroxiredoxin-based probes. Nat. Chem. Biol. 2016, 12, 437–443. [Google Scholar] [CrossRef]
- Kim, J.S.; Holmes, R.K. Characterization of OxyR as a Negative transcriptional regulator that represses catalase production in Corynebacterium diphtheriae. PLoS ONE 2012, 7, e31709. [Google Scholar] [CrossRef]
- Bidaud, P.; Hérbert, L.; Barbey, C.; Appourchaux, A.-C.; Torelli, B.; Sanguinetti, M.; Laugier, C.; Petry, S. Rhodococcus equi’s extreme resistance to hydrogen peroxide is mainly conferred by one of its four catalase genes. PLoS ONE 2012, 7, e42396. [Google Scholar] [CrossRef]
- Teramoto, H.; Inui, M.; Yukawa, H. OxyR acts as a transcriptional repressor of hydrogen peroxide-inducible antioxidant genes in Corynebacterium glutamicum R. FEBS J. 2013, 280, 3298–3312. [Google Scholar] [CrossRef]
- Bollela, V.R.; Namburete, E.I.; Feliciano, C.S.; Macheque, D.; Harrison, L.H.; Caminero, J.A. Detection of katG and inhA mutations to guide isoniazid and ethionamide use for drug-resistant tuberculosis. Int. J. Tuberc. Lung Dis. 2016, 20, 1099–1104. [Google Scholar] [CrossRef]
- Shastri, M.D.; Shukla, S.D.; Chong, W.C.; Dua, K.; Peterson, G.M.; Patel, R.P.; Hansbro, P.M.; Eri, R.; O’Toole, R.F. Role of oxidative stress in the pathology and management of human tuberculosis. Oxid. Med. Cell. Longev. 2018, 11, 7695364. [Google Scholar] [CrossRef]
- Gort, A.S.; Imlay, J.A. Balance between endogenous superoxide stress and antioxidant defenses. J. Bacteriol. 1998, 180, 1402–1410. [Google Scholar] [PubMed]
- Pedre, B.; Van Molle, I.; Villadangos, A.F.; Wahni, K.; Vertommen, D.; Turell, L.; Erdogan, H.; Mateos, L.M.; Messens, J. The Corynebacterium glutamicum mycothiol peroxidase is a reactive oxygen species-scavenging enzyme that shows promiscuity in thiol redox control. Mol. Microbiol. 2015, 96, 1176–1191. [Google Scholar] [CrossRef] [PubMed]
- Pedre, B.; Young, D.; Charlier, D.; Mourenza, Á.; Rosado, L.A.; Marcos-Pascual, L.; Wahni, K.; Gonzalo de la Rubia, A.; Belousov, V.; Mateos, L.M. Structural snapshots of OxyR reveal the peroxidatic mechanism of H2O2 sensing. Proc. Nat. Acad. Sci. USA 2018, 115, E11623–E11632. [Google Scholar] [CrossRef]
- Si, M.; Tauslf, M.; Chudhry, T.; Ding, W.; Xu, Y.; Chen, C.; Akbar, A.; Shen, X.; Liu, S.-J. Corynebacterium glutamicum methionine sulfoxide reductase A uses both mycoredoxin and thioredoxin for regeneration and oxidative stress resistance. Appl. Environ. Microbiol. 2015, 81, 2781–2796. [Google Scholar] [CrossRef] [PubMed]
- Tossounian, M.A.; Pedre, B.; Wahni, K.; Erdogan, H.; Vertommen, D.; Van Molle, I.; Messens, J. Corynebacterium diphtheriae methionine sulfoxide reductase A exploits a unique mycothiol redox relay mechanism. J. Biol. Chem. 2015, 18, 11365–11375. [Google Scholar] [CrossRef] [PubMed]
- Sanjuan, M.A.; Dillon, C.P.; Tait, S.W.G.; Moshiach, S.; Dorsey, F.; Connell, S.; Komatsu, M.; Tanaka, K.; Cleveland, J.L.; Withoff, S.; et al. Toll-like receptor signalling in macrophages links the autophagy pathway to phagocytosis. Nature 2007, 450, 1253–1257. [Google Scholar] [CrossRef]
- Fernandez-Mora, E.; Polidori, M.; Lührmann, A.; Schaible, U.E.; Haas, A. Maturation of Rhodococcus equi-containing vacuoles is arrested after completion of the early endosome stage. Traffic 2005, 6, 635–653. [Google Scholar] [CrossRef]
- Ehrt, S.; Schnappinger, D. Mycobacterial survival strategies in the phagosome: Defence against host stresses. Cell. Microbiol. 2009, 11, 1170–1178. [Google Scholar] [CrossRef]
- Mehta, M.; Singh, A. Mycobacterium tuberculosis WhiB3 maintains redox homeostasis and survival in response to reactive oxygen and nitrogen species. Free Radic. Biol. Med. 2019, 131, 50–58. [Google Scholar] [CrossRef]
- Giles, C.; Vanniasinkam, T.; Ndi, S.; Barton, M.D. Rhodococcus equi (Prescottella equi) vaccines; the future of vaccine development. Equine Vet. J. 2015, 47, 510–518. [Google Scholar] [CrossRef]
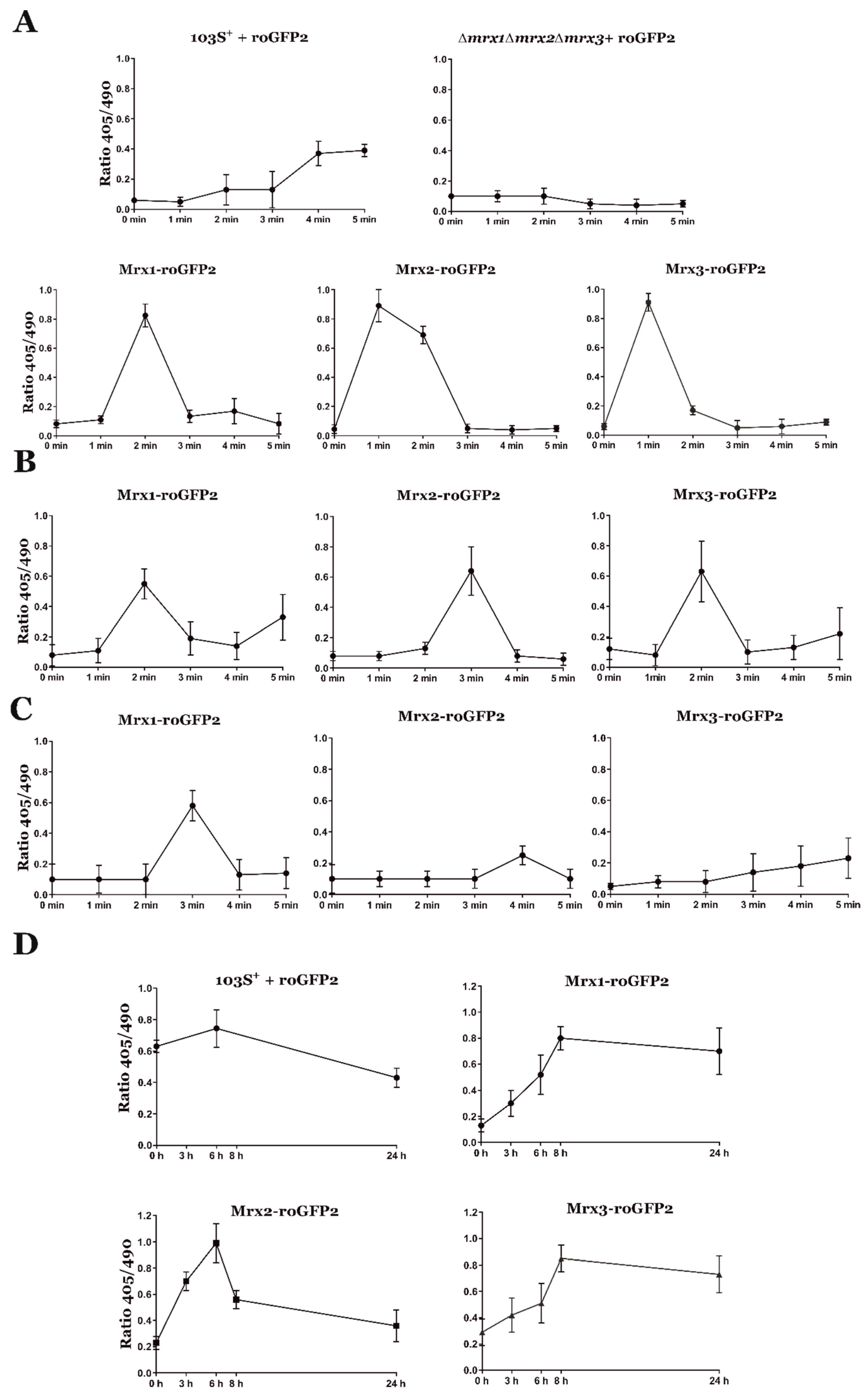
| EroGFP2 mV | Basal | H2O2 | NaClO | NO | Macrophages |
|---|---|---|---|---|---|
| Mrx1-roGFP2 | −290 mV | −264 mV | −278 mV | −274 mV | −260 mV |
| Mrx2-roGFP2 | −295 mV | −264 mV | −270 mV | −296 mV | −258 mV |
| Mrx3-roGFP2 | −310 mV | −264 mV | −271 mV | −295 mV | −254 mV |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mourenza, Á.; Bravo-Santano, N.; Pradal, I.; Gil, J.A.; Mateos, L.M.; Letek, M. Mycoredoxins Are Required for Redox Homeostasis and Intracellular Survival in the Actinobacterial Pathogen Rhodococcus equi. Antioxidants 2019, 8, 558. https://doi.org/10.3390/antiox8110558
Mourenza Á, Bravo-Santano N, Pradal I, Gil JA, Mateos LM, Letek M. Mycoredoxins Are Required for Redox Homeostasis and Intracellular Survival in the Actinobacterial Pathogen Rhodococcus equi. Antioxidants. 2019; 8(11):558. https://doi.org/10.3390/antiox8110558
Chicago/Turabian StyleMourenza, Álvaro, Natalia Bravo-Santano, Inés Pradal, Jose A. Gil, Luis M. Mateos, and Michal Letek. 2019. "Mycoredoxins Are Required for Redox Homeostasis and Intracellular Survival in the Actinobacterial Pathogen Rhodococcus equi" Antioxidants 8, no. 11: 558. https://doi.org/10.3390/antiox8110558
APA StyleMourenza, Á., Bravo-Santano, N., Pradal, I., Gil, J. A., Mateos, L. M., & Letek, M. (2019). Mycoredoxins Are Required for Redox Homeostasis and Intracellular Survival in the Actinobacterial Pathogen Rhodococcus equi. Antioxidants, 8(11), 558. https://doi.org/10.3390/antiox8110558








