Systematic Physical Exercise and Spirulina maxima Supplementation Improve Body Composition, Cardiorespiratory Fitness, and Blood Lipid Profile: Correlations of a Randomized Double-Blind Controlled Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Supplements Characteristics
2.2. Ethics Approval and Consent to Participate
2.3. Inclusion and Exclusion Criteria
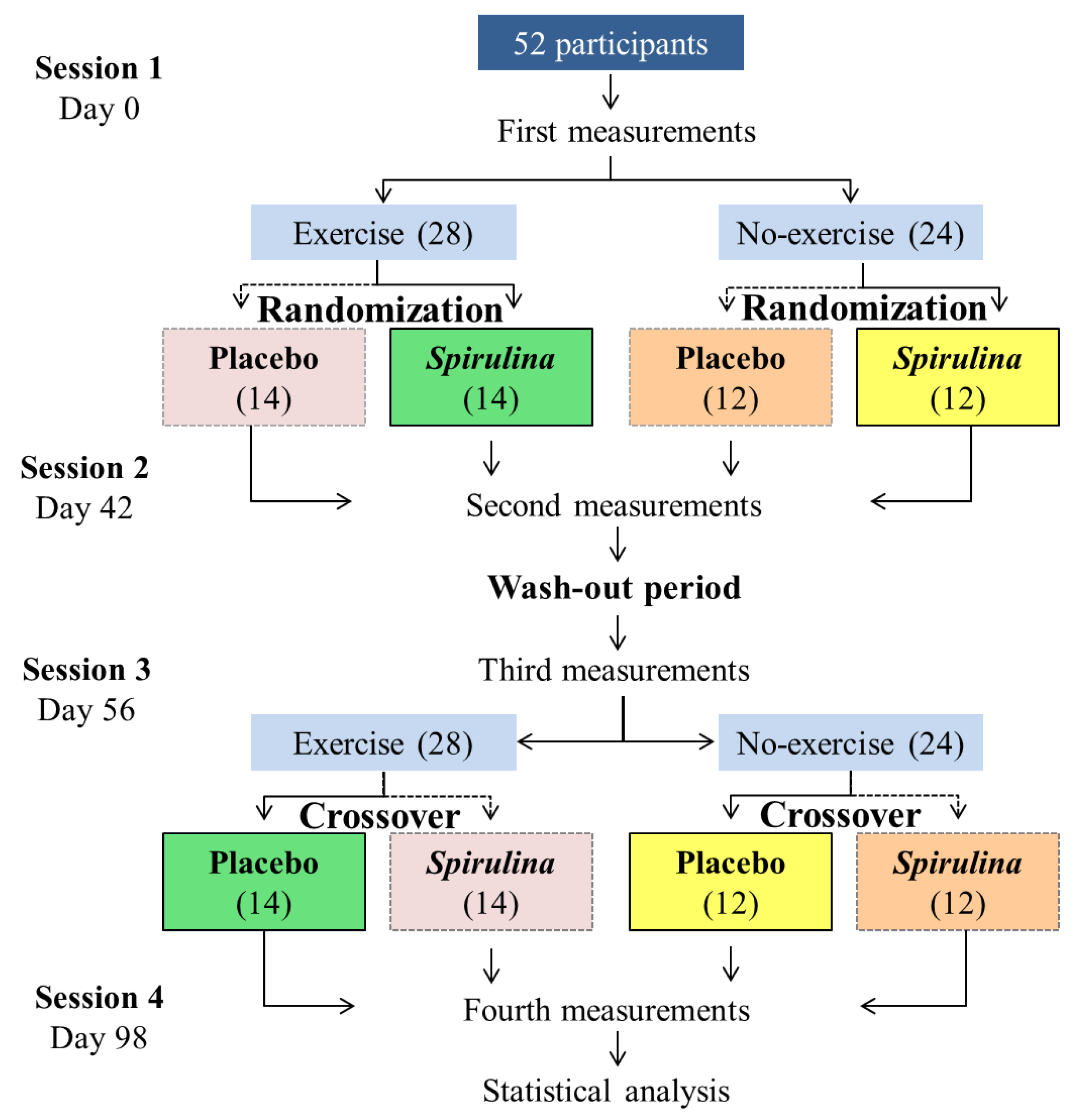
2.4. Participants and Study Design
2.5. Diet
2.6. Determination of Maximal Oxygen Uptake (VO2max)
2.7. Physical Exercise Program
2.8. Blood Sample Analysis
2.9. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smith, S.C.; Collins, A.; Ferrari, R.; Holmes, D.R.; Logstrup, S.; McGhie, D.V.; Ralston, J.; Sacco, R.L.; Stam, H.; Taubert, K.; et al. Our time: A call to save preventable death from cardiovascular disease (heart disease and stroke). J. Am. Coll. Cardiol. 2012, 60, 2343–2348. [Google Scholar] [CrossRef] [PubMed]
- Wall-Medrano, A.; Ramos-Jiménez, A.; Hernández-Torres, R.P.; Villalobos-Molina, R.; Tapia-Pancardo, D.C.; Jiménez-Flores, R.; Méndez-Cruz, A.R.; Murguía-Romero, M.; Gallardo-Ortíz, I.A.; Urquídez-Romero, R. Cardiometabolic risk in young adults from northern Mexico: Revisiting body mass index and waist-circumference as predictors. BMC Public Health 2016, 16, 236. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ávila, M.; Rivera-Dommarco, J.; Shamah-Levy, T.; Cuevas-Nasu, L.; Gómez-Acosta, L.M.; Gaona-Pineda, E.B.; Romero-Martínez, M.; Gómez-Humarán, I.M.; Saturno-Hernández, P.; Villalpando-Hernández, S.; et al. Encuesta Nacional de Salud y Nutrición de Medio Camino 2016 (ENSANUT 2016); Informe final de resultados; Instituto Nacional de Salud Pública: Cuernavaca, México, 2016; pp. 1–149. Available online: https://www.gob.mx/cms/uploads/attachment/file/209093/ENSANUT.pdf (accessed on 20 October 2019).
- Ramos-Jiménez, A.; Wall-Medrano, A.; Hernández-Lepe, M.A.; Chávez-Treviño, G.; Guereca-Arvizuo, J.; Hernández-Torres, R.P. Borg’s category ratio-scale (CR-10) is useful to predict the onset of blood lactate accumulation (OBLA) in young Mexican adults, regardless their body mass. Ciencia ergo-sum 2019, 26, e42. [Google Scholar] [CrossRef]
- Greene, N.P.; Martin, S.E.; Crouse, S.F. Acute exercise and training alter blood lipid and lipoprotein profiles differently in overweight and obese men and women. Obesity 2012, 20, 1618–1627. [Google Scholar] [CrossRef]
- Chen, G.; Wang, H.; Zhang, X.; Yang, S.-T. Nutraceuticals and functional foods in the management of hyperlipidemia. Crit. Rev. Food Sci. Nutr. 2014, 54, 1180–1201. [Google Scholar] [CrossRef]
- Serban, M.-C.; Sahebkar, A.; Dragan, S.; Stoichescu-Hogea, G.; Ursoniu, S.; Andrica, F.; Banach, M. A systematic review and meta-analysis of the impact of Spirulina supplementation on plasma lipid concentrations. Clin. Nutr. 2016, 35, 842–851. [Google Scholar] [CrossRef]
- Huang, H.; Liao, D.; Pu, R.; Cui, Y. Quantifying the effects of spirulina supplementation on plasma lipid and glucose concentrations, body weight, and blood pressure. Diab. Metab. Synd. Obes. Target Ther. 2018, 11, 729–742. [Google Scholar] [CrossRef]
- Hernández-Lepe, M.A.; Wall-Medrano, A.; Juárez-Oropeza, M.A.; Ramos-Jiménez, A.; Hernández-Torres, R.P. Spirulina y su efecto hipolipemiante y antioxidante en humanos: Una revisión sistemática. Nutr. Hosp. 2015, 32, 494–500. [Google Scholar] [CrossRef]
- Moura, L.P.; Puga, G.M.; Beck, W.R.; Teixeira, I.P.; Ghezzi, A.C.; Silva, G.A.; Mello, M.A.R. Exercise and spirulina control non-alcoholic hepatic steatosis and lipid profile in diabetic Wistar rats. Lipids Health. Dis. 2011, 10, 77. [Google Scholar] [CrossRef]
- Mazzola, D.; Fornari, F.; Vigano, G.; Oro, T.; Costa, J.A.V.; Bertolin, T.E. Spirulina platensis enhances the beneficial effect of exercise on oxidative stress and the lipid profile in rats. Braz. Arch. Biol. Techn. 2015, 6, 961–969. [Google Scholar] [CrossRef]
- Kata, F.S.; Athbi, A.M.; Manwar, E.Q.; Al-Ashoor, A.; Abdel-Daim, M.M.; Aleya, L. Therapeutic effect of the alkaloid extract of the cyanobacterium Spirulina platensis on the lipid profile of hypercholesterolemic male rabbits. Environ. Sci. Pollut. Res. 2018, 25, 19635. [Google Scholar] [CrossRef] [PubMed]
- Memije-Lazaro, I.N.; Blas-Valdivia, V.; Franco-Colín, M.; Cano-Europa, E. Arthrospira maxima (Spirulina) and C-phycocyanin prevent the progression of chronic kidney disease and its cardiovascular complications. J. Funct. Food 2018, 43, 37–43. [Google Scholar] [CrossRef]
- Hernández-Lepe, M.A.; López-Díaz, J.A.; de la Rosa, L.A.; Hernández-Torres, R.P.; Wall-Medrano, A.; Juárez-Oropeza, M.A.; Pedraza-Chaverri, J.; Urquídez-Romero, R.; Ramos-Jiménez, A. Double-blind randomized controlled trial of the independent and synergistic effect of Spirulina maxima with exercise (ISESE) on general fitness, lipid profile and redox status in overweight and obese subjects: Study protocol. BMJ Open 2017, 7, e013744. [Google Scholar] [CrossRef] [PubMed]
- Riebe, D.; Ehrman, J.K.; Liguori, G.; Magal., M. American College of Sports Medicine. In ACSM’s Guidelines for Exercise Testing and Prescription, 10th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2016; pp. 111–142. [Google Scholar]
- Kalafati, M.; Jamurtas, A.Z.; Nikolaidis, M.G.; Paschalis, V.; Theodorou, A.A.; Sakellariou, G.K.; Koutedakis, Y.; Kouretas, D. Ergogenic and antioxidant effects of spirulina supplementation in humans. Med. Sci. Sports Exerc. 2010, 1, 142–151. [Google Scholar] [CrossRef]
- Iwata, K.; Inayama, T.; Kato, T. Effects of Spirulina platensis on plasma lipoprotein lipase activity in fructose-induced hyperlipidemic rats. J. Nutr. Sci. Vitaminol. 1990, 2, 165–171. [Google Scholar] [CrossRef]
- Moor, V.J.A.; Biapa, P.C.N.; Njinkio, B.L.N.; Moukette, B.M.; Sando, Z.; Kenfack, C.; Ateba, B.; Ngo-Matip, M.E.; Pieme, C.A.; Ngogang, J. Hypolipidemic effect and activation of Lecithin Cholesterol Acyl Transferase (LCAT) by aqueous extract of Spirulina platensis during toxicological investigation. BMC Nutr. 2017, 3, 25. [Google Scholar] [CrossRef]
- Merry, T.L.; Ristow, M. Do antioxidant supplements interfere with skeletal muscle adaptation to exercise training? J. Physiol. 2016, 18, 5135–5147. [Google Scholar] [CrossRef]
- Ristow, M.; Zarse, K.; Oberbach, A.; Klöting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Blüher, M. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc. Natl. Acad. Sci. USA 2009, 21, 8665–8670. [Google Scholar] [CrossRef]
- Yfanti, C.; Akerström, T.; Nielsen, S.; Nielsen, A.R.; Mounier, R.; Mortensen, O.H.; Lykkesfeldt, J.; Rose, A.J.; Fischer, C.; Pedersen, B.K. Antioxidant supplementation does not alter endurance training adaptation. Med. Sci. Sports Exerc. 2010, 7, 1388–1395. [Google Scholar] [CrossRef]
- Upasani, C.; Balaraman, R. Protective effect of Spirulina on lead induced deleterious changes in the lipid peroxidation and endogenous antioxidants in rats. Phytother. Res. 2003, 17, 330e4. [Google Scholar] [CrossRef] [PubMed]
- Torres-Duran, P.V.; Ferreira-Hermosillo, A.; Ramos-Jiménez, A.; Hernández-Torres, R.P.; Juárez-Oropeza, M.A. Effect of Spirulina maxima on postprandial lipemia in young runners: A preliminary report. J. Med. Food 2012, 15, 753–757. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, S.; Shimizu, K.; Kaneko, H.; Shibayama, F.; Morikawa, K.; Kanamaru, Y.; Otsuka, A.; Hirahashi, T.; Kato, T. A novel protein C-phycocyanin plays a crucial role in the hypocholesterolemic action of Spirulina platensis concentrate in rats. J. Nutr. 2005, 10, 2425–2430. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.J.; Gao, S.S.; Li, H. The Effect of Spirulin’s Complex Prescriptions on Cardiac Muscle, Skeletal Muscle and Liver Tissue’s Morphologies and Protein Expression of HSP70 in These Tissues of Mice During High-Intensity Increasing Exercise. J. Tianjin Inst. Phys. Ed. 2005, 3, 11. [Google Scholar]
| Total | Overweight | Obesity | |
|---|---|---|---|
| N | 52 | 27 | 25 |
| Age (y) | 26 ± 5 | 26 ± 4 | 27 ± 6 |
| Bodyweight (kg) | 90 ± 13 | 81 ± 8 | 100 ± 12 |
| Height (m) | 1.72 ± 0.1 | 1.72 ± 0.1 | 1.73 ± 0.1 |
| BMI (kg·m−2) | 30.2 ± 4 | 27.4 ± 1.2 | 33.3 ± 3.8 |
| Body fat (%) | 28.8 ± 7.2 | 24.8 ± 5.9 | 33.2 ± 6.1 |
| Energy intake (kcal·day−1) | 2054 ± 104 | 1977 ± 139 | 2054 ± 151 |
| VO2max (mL·min−1·kg−1) | 35.4 ± 6.9 | 39.6 ± 5.1 | 30.8 ± 5.6 |
| Total cholesterol (mg·dL−1) | 196 ± 36 | 177 ± 28 | 218 ± 30 |
| Triacylglycerols (mg·dL−1) | 142 ± 41 | 136 ± 35 | 150 ± 46 |
| LDL-C (mg·dL−1) | 134 ± 36 | 115 ± 27 | 158 ± 31 |
| HDL-C (mg·dL−1) | 34 ± 9.6 | 35.3 ± 8.2 | 32.5 ± 10.9 |
| Variable | Basal | Final | p-Value |
|---|---|---|---|
| TC (mg·dL−1) | 218 ± 30 | 184 ± 33 | 0.000 |
| TAG (mg·dL−1) | 150 ± 46 | 127 ± 35 | 0.007 |
| LDL-C (mg·dL−1) | 158 ± 31 | 122 ± 34 | 0.000 |
| HDL-C (mg·dL−1) | 32.5 ± 10.9 | 38.6 ± 9.6 | 0.004 |
| BMI (kg·m−2) | 33.3 ± 3.8 | 30.1 ± 4.9 | 0.003 |
| VO2max (mL·min−1 kg−1) | 30.8 ± 5.6 | 34.7 ± 6.2 | 0.003 |
| BF (%) | 33.2 ± 6.0 | 30.5 ± 7.1 | 0.050 |
| Variable | Systematic Physical Exercise Program (N = 28) | |||||
|---|---|---|---|---|---|---|
| S. maxima Supplementation | Placebo Supplementation | |||||
| Basal | Final | p-Value | Basal | Final | p-Value | |
| BMI (kg·m−2) | 29.6 ± 3 | 28.9 ± 3 | 0.359 | 29.7 ± 3 | 29.4 ± 3 | 0.755 |
| Body fat (%) | 26.4 ± 7 | 25.2 ± 6 | 0.493 | 27.0 ± 7 | 26.0 ± 7 | 0.592 |
| TC (mg·dL−1) | 196.3 ± 35 | 162.6 ± 33 | 0.001 | 197.0 ± 38 | 177.2 ± 36 | 0.053 |
| TAG (mg·dL−1) | 157.3 ± 47 | 134.8 ± 37 | 0.070 | 139.3 ± 44 | 124.2 ± 42 | 0.212 |
| LDL-C (mg·dL−1) | 127.9 ± 36 | 93.3 ± 34 | 0.001 | 137.4 ± 38 | 115.4 ± 36 | 0.046 |
| HDL-C (mg·dL−1) | 34.4 ± 9 | 41.7 ± 10 | 0.005 | 31.4 ± 9 | 36.2 ± 9 | 0.059 |
| VO2max (mL·min−1 kg−1) | 37.0 ± 7 | 39.5 ± 7 | 0.212 | 36.6 ± 8 | 38.2 ± 7 | 0.486 |
| Variable | No exercise program (N = 24) | |||||
| S. maxima Supplementation | Placebo Supplementation | |||||
| Basal | Final | p-Value | Basal | Final | p-Value | |
| BMI (kg·m−2) | 30.8 ± 5 | 30.2 ± 5 | 0.709 | 30.9 ± 5 | 30.8 ± 5 | 0.945 |
| Body fat (%) | 31.3 ± 7 | 30.0 ± 7 | 0.551 | 31.2 ± 7 | 30.9 ± 7 | 0.917 |
| TC (mg·dL−1) | 201.2 ± 39 | 183.0 ± 37 | 0.103 | 190.2 ± 31 | 185.7 ± 31 | 0.619 |
| TAG (mg·dL−1) | 141.4 ± 37 | 127.3 ± 34 | 0.176 | 131.2 ± 31 | 125.1 ± 32 | 0.517 |
| LDL-C (mg·dL−1) | 138.0 ± 37 | 117.2 ± 38 | 0.062 | 131.3 ± 32 | 127.4 ± 30 | 0.680 |
| HDL-C (mg·dL−1) | 34.9 ± 10 | 40.3 ± 11 | 0.071 | 35.3 ± 12 | 36.7 ± 8 | 0.655 |
| VO2max (mL·min−1 kg−1) | 33.6 ± 6 | 35.4 ± 7 | 0.399 | 33.7 ± 6 | 34.2 ± 6 | 0.796 |
| Exercise and Spirulina maxima Supplementation | ||||||
|---|---|---|---|---|---|---|
| ΔBMI | ΔBF | ΔVO2max | ΔTC | ΔTAG | ΔLDL-C | |
| Δ%BF | 0.097 | 1 | ||||
| ΔVO2max | −0.492 * | −0.217 | 1 | |||
| ΔTC | 0.348 | −0.164 | 0.091 | 1 | ||
| ΔTAG | 0.162 | 0.101 | −0.157 | 0.094 | 1 | |
| ΔLDL-C | 0.255 | −0.164 | 0.035 | 0.798 * | −0.184 | 1 |
| ΔHDL-C | −0.137 | 0.038 | 0.159 | −0.2282 | 0.066 | −0.690 * |
| Exercise and placebo Supplementation | ||||||
| ΔBMI | ΔBF | ΔVO2max | ΔTC | ΔTAG | ΔLDL-C | |
| Δ%BF | −0.094 | 1 | ||||
| ΔVO2max | −0.151 | −0.056 | 1 | |||
| ΔTC | 0.329 | −0.401 * | −0.246 | 1 | ||
| ΔTAG | 0.030 | −0.155 | 0.075 | 0.461 * | 1 | |
| ΔLDL-C | 0.398 * | −0.280 | −0.184 | 0.718 * | 0.189 | 1 |
| ΔHDL-C | 0.021 | 0.028 | −0.164 | 0.288 | 0.184 | −0.286 |
| Spirulina maxima supplementation without exercise | ||||||
| ΔBMI | ΔBF | ΔVO2max | ΔTC | ΔTAG | ΔLDL-C | |
| Δ%BF | 0.210 | 1 | ||||
| ΔVO2max | −0.526 * | −0.497 * | 1 | |||
| ΔTC | 0370 | 0.160 | −0.314 | 1 | ||
| ΔTAG | 0.425 * | 0.299 | −0.103 | 0.243 | 1 | |
| ΔLDL-C | 0.135 | 0.237 | −0.161 | 0.673 * | 0.143 | 1 |
| ΔHDL-C | 0.131 | −0.180 | −0.059 | 0.297 | 0.041 | −0.371 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Lepe, M.A.; Olivas-Aguirre, F.J.; Gómez-Miranda, L.M.; Hernández-Torres, R.P.; Manríquez-Torres, J.d.J.; Ramos-Jiménez, A. Systematic Physical Exercise and Spirulina maxima Supplementation Improve Body Composition, Cardiorespiratory Fitness, and Blood Lipid Profile: Correlations of a Randomized Double-Blind Controlled Trial. Antioxidants 2019, 8, 507. https://doi.org/10.3390/antiox8110507
Hernández-Lepe MA, Olivas-Aguirre FJ, Gómez-Miranda LM, Hernández-Torres RP, Manríquez-Torres JdJ, Ramos-Jiménez A. Systematic Physical Exercise and Spirulina maxima Supplementation Improve Body Composition, Cardiorespiratory Fitness, and Blood Lipid Profile: Correlations of a Randomized Double-Blind Controlled Trial. Antioxidants. 2019; 8(11):507. https://doi.org/10.3390/antiox8110507
Chicago/Turabian StyleHernández-Lepe, Marco Antonio, Francisco Javier Olivas-Aguirre, Luis Mario Gómez-Miranda, Rosa Patricia Hernández-Torres, José de Jesús Manríquez-Torres, and Arnulfo Ramos-Jiménez. 2019. "Systematic Physical Exercise and Spirulina maxima Supplementation Improve Body Composition, Cardiorespiratory Fitness, and Blood Lipid Profile: Correlations of a Randomized Double-Blind Controlled Trial" Antioxidants 8, no. 11: 507. https://doi.org/10.3390/antiox8110507
APA StyleHernández-Lepe, M. A., Olivas-Aguirre, F. J., Gómez-Miranda, L. M., Hernández-Torres, R. P., Manríquez-Torres, J. d. J., & Ramos-Jiménez, A. (2019). Systematic Physical Exercise and Spirulina maxima Supplementation Improve Body Composition, Cardiorespiratory Fitness, and Blood Lipid Profile: Correlations of a Randomized Double-Blind Controlled Trial. Antioxidants, 8(11), 507. https://doi.org/10.3390/antiox8110507









