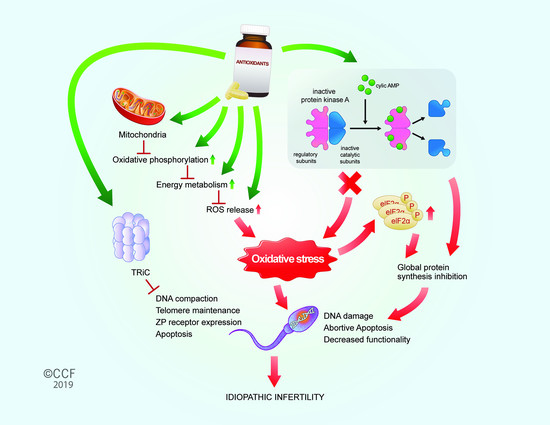
Effect of Antioxidant Supplementation on the Sperm Proteome of Idiopathic Infertile Men
Abstract
1. Introduction
2. Materials and Methods
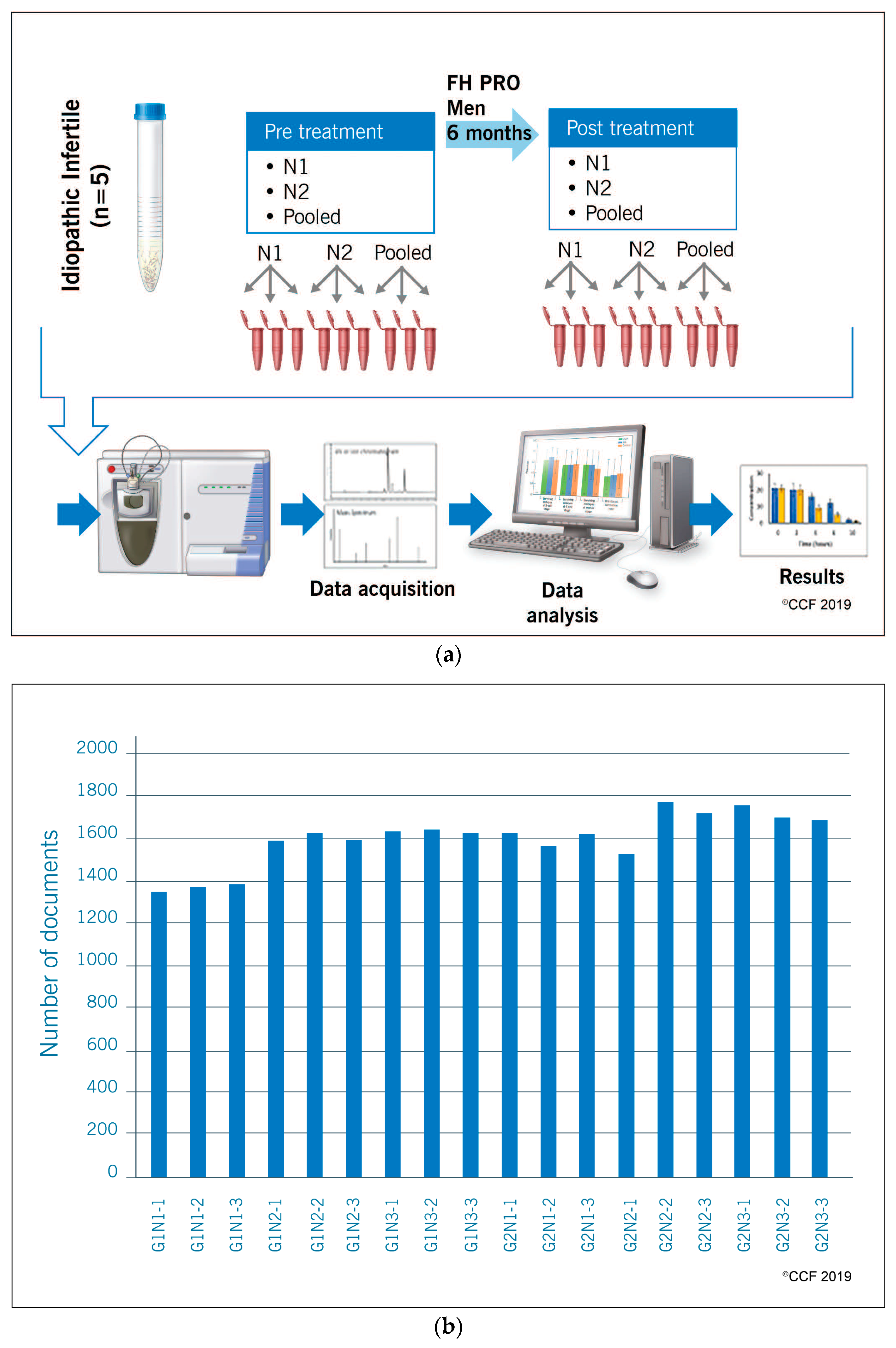
2.1. Study Design and Subjects
2.2. Semen Analysis and Processing of Semen Samples
2.3. Sperm Protein Extraction and Quantification
2.4. Liquid Chromatography-Tandem Mass Spectrometry
2.5. Bioinformatic Analysis
2.6. Protein Selection and Validation by Western Blot
3. Results
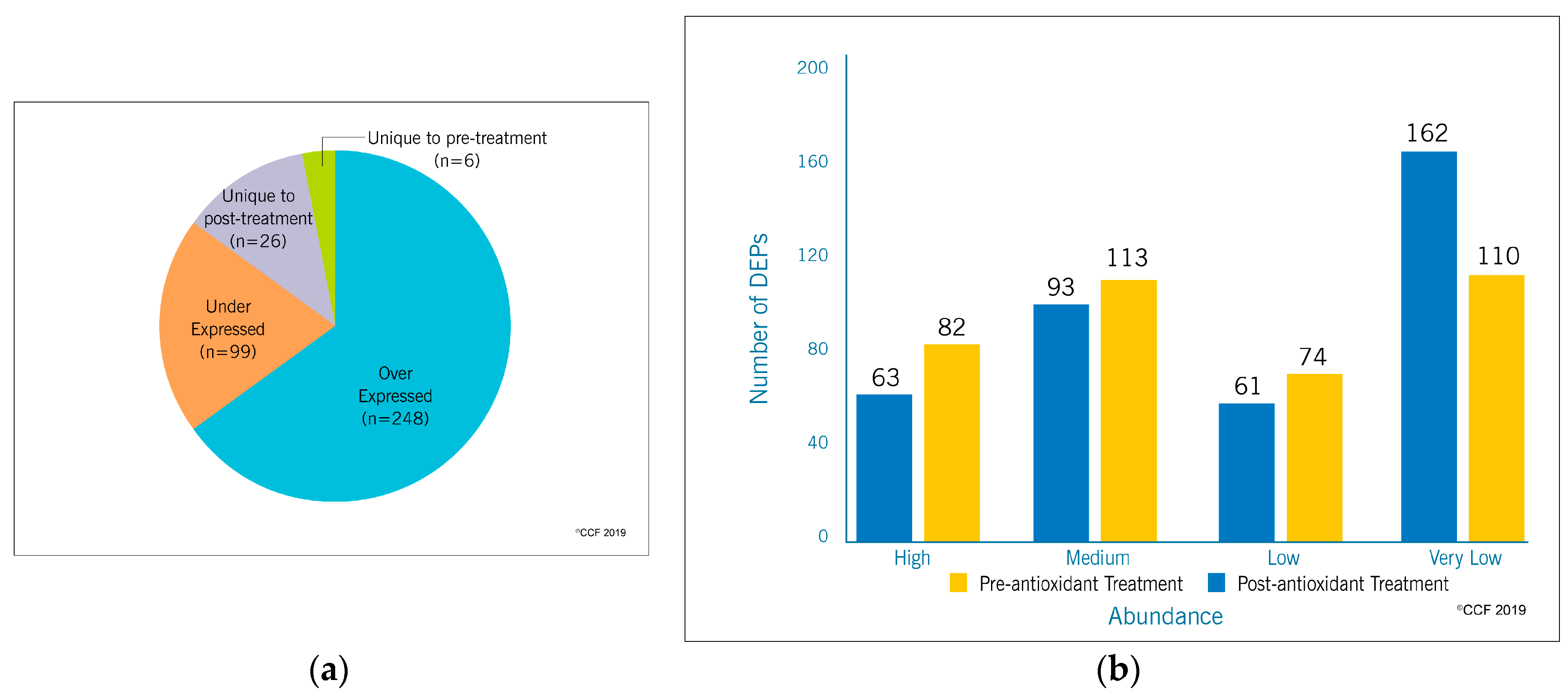
3.1. Proteomic Profile of Spermatozoa
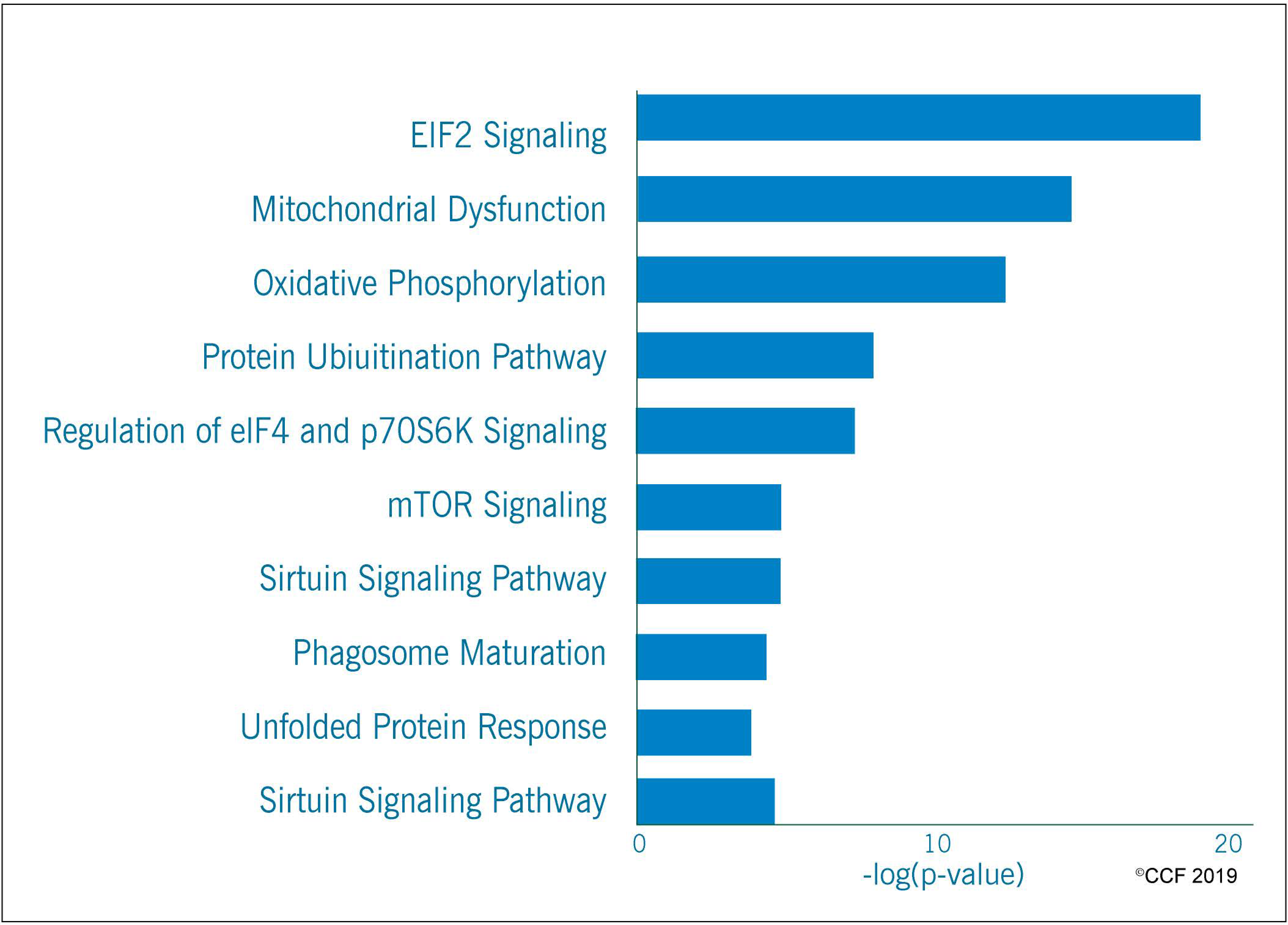
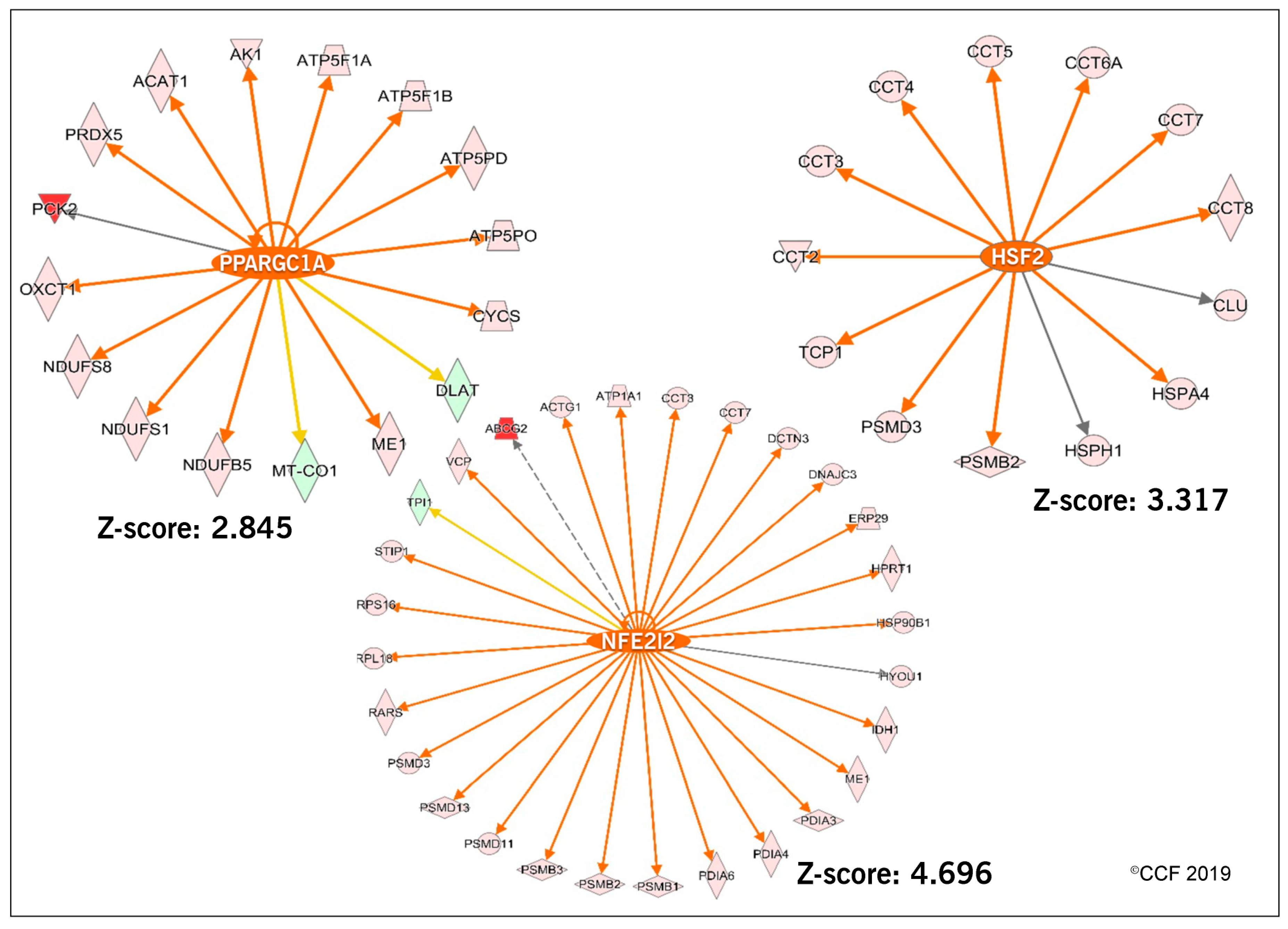
3.2. Functional Annotation and Pathway Analysis
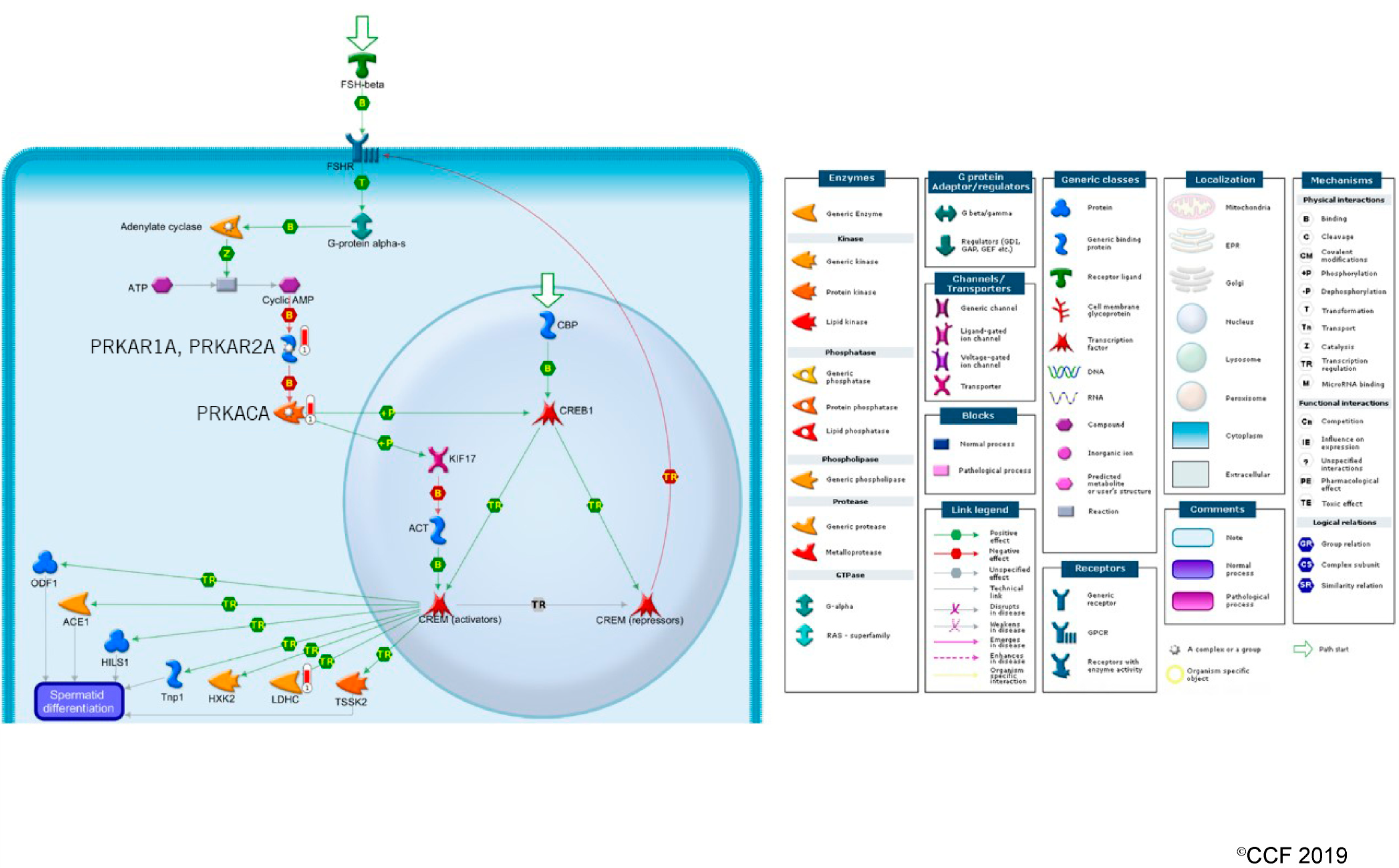
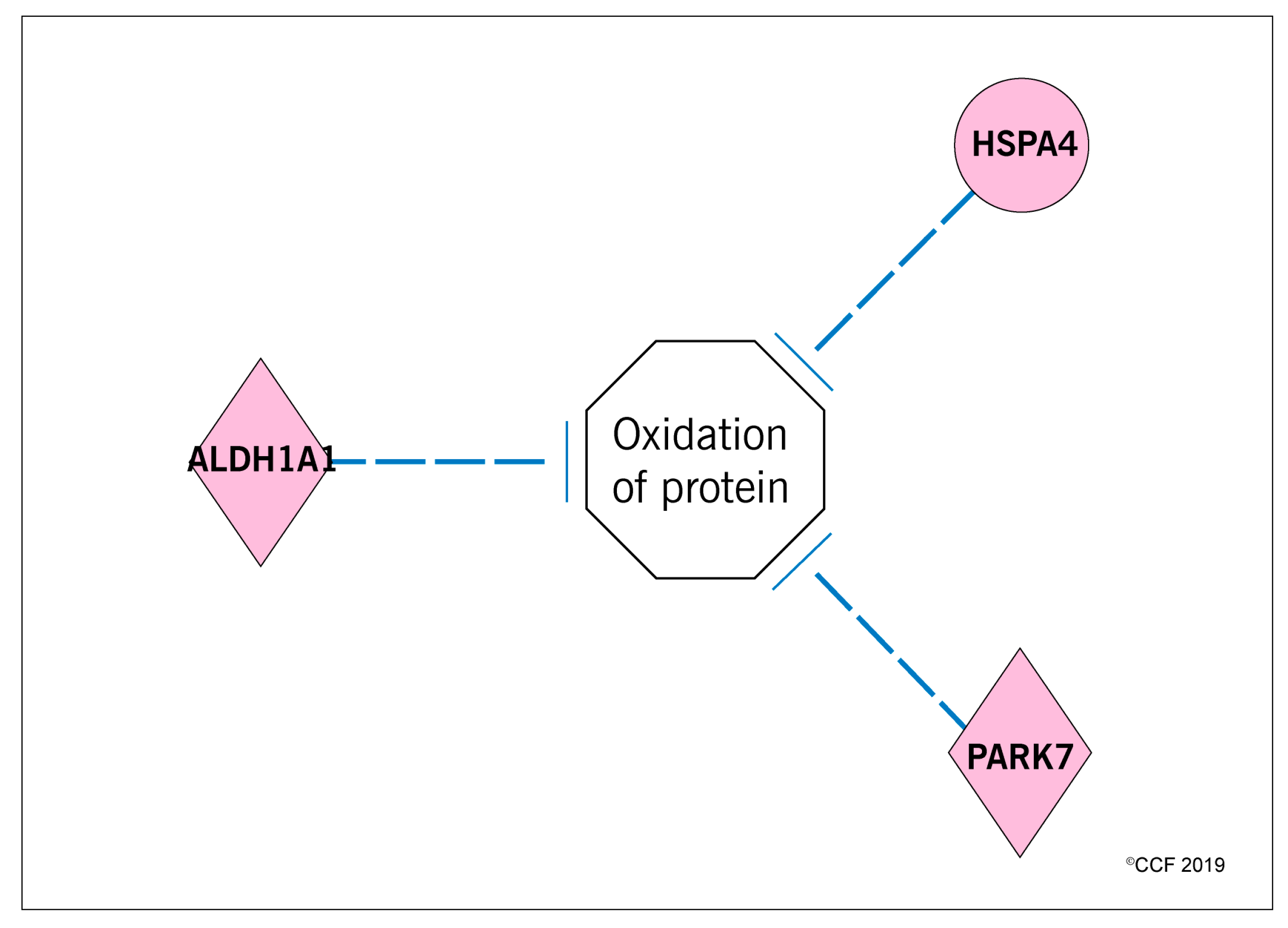
3.3. Effect of Antioxidant Treatment on Redox Regulation and Reproductive Function
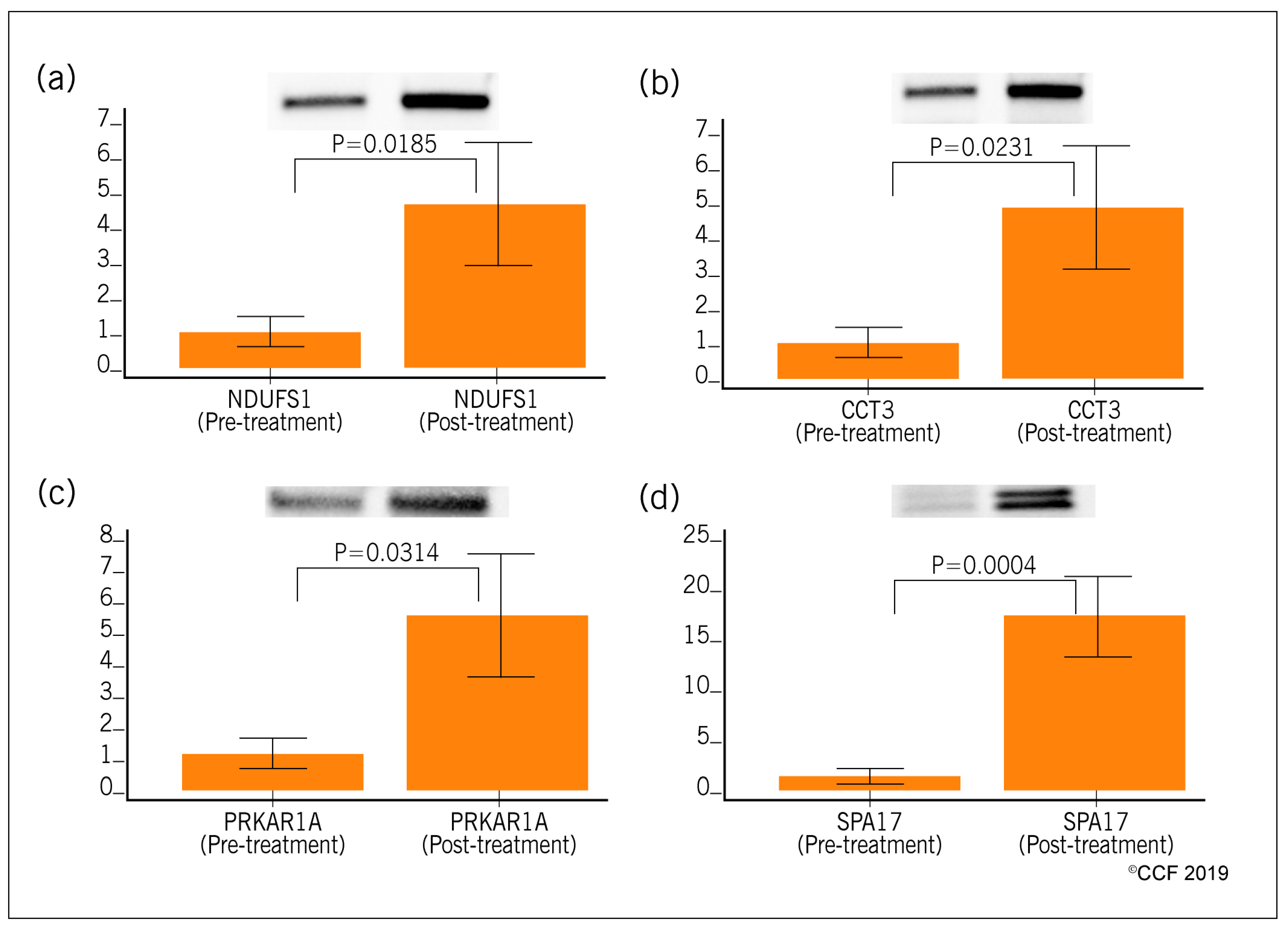
3.4. Selection and Validation of Expression Profile of Proteins by Western Blot
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Agarwal, A.; Parekh, N.; Panner Selvam, M.K.; Henkel, R.; Shah, R.; Homa, S.T.; Ramasamy, R.; Ko, E.; Tremellen, K.; Esteves, S.; et al. Male Oxidative Stress Infertility (MOSI): Proposed Terminology and Clinical Practice Guidelines for Management of Idiopathic Male Infertility. World J. Mens. Health 2019, 37, 296–312. [Google Scholar] [CrossRef] [PubMed]
- Bui, A.D.; Sharma, R.; Henkel, R.; Agarwal, A. Reactive oxygen species impact on sperm DNA and its role in male infertility. Andrologia 2018, 50, e13012. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.J.; De Iuliis, G.N.; Drevet, J.R. Chapter 2.2—Role of Oxidative Stress in the Etiology of Male Infertility and the Potential Therapeutic Value of Antioxidants. In Oxidants, Antioxidants and Impact of the Oxidative Status in Male Reproduction; Henkel, R., Samanta, L., Agarwal, A., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 91–100. [Google Scholar] [CrossRef]
- Tremellen, K. Oxidative stress and male infertility—A clinical perspective. Hum. Reprod. Update 2008, 14, 243–258. [Google Scholar] [CrossRef]
- Wagner, H.; Cheng, J.W.; Ko, E.Y. Role of reactive oxygen species in male infertility: An updated review of literature. Arab J. Urol. 2018, 16, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Sharma, R.; Roychoudhury, S.; Du Plessis, S.; Sabanegh, E. MiOXSYS: A novel method of measuring oxidation reduction potential in semen and seminal plasma. Fertil. Steril. 2016, 106, 566–573.e510. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Wang, S.M. Clinical Relevance of Oxidation-Reduction Potential in the Evaluation of Male Infertility. Urology 2017, 104, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Saleh, R.A.; Bedaiwy, M.A. Role of reactive oxygen species in the pathophysiology of human reproduction. Fertil. Steril. 2003, 79, 829–843. [Google Scholar] [CrossRef]
- Aitken, R.J.; Koppers, A.J. Apoptosis and DNA damage in human spermatozoa. Asian J. Androl. 2011, 13, 36–42. [Google Scholar] [CrossRef]
- Dorostghoal, M.; Kazeminejad, S.R.; Shahbazian, N.; Pourmehdi, M.; Jabbari, A. Oxidative stress status and sperm DNA fragmentation in fertile and infertile men. Andrologia 2017, 49, e12762. [Google Scholar] [CrossRef]
- Balercia, G.; Regoli, F.; Armeni, T.; Koverech, A.; Mantero, F.; Boscaro, M. Placebo-controlled double-blind randomized trial on the use of l-carnitine, l-acetylcarnitine, or combined l-carnitine and l-acetylcarnitine in men with idiopathic asthenozoospermia. Fertil. Steril. 2005, 84, 662–671. [Google Scholar] [CrossRef]
- Safarinejad, M.R.; Safarinejad, S.; Shafiei, N.; Safarinejad, S. Effects of the Reduced Form of Coenzyme Q10 (Ubiquinol) on Semen Parameters in Men with Idiopathic Infertility: A Double-Blind, Placebo Controlled, Randomized Study. J. Urol. 2012, 188, 526–531. [Google Scholar] [CrossRef] [PubMed]
- ElSheikh, M.G.; Hosny, M.B.; Elshenoufy, A.; Elghamrawi, H.; Fayad, A.; Abdelrahman, S. Combination of vitamin E and clomiphene citrate in treating patients with idiopathic oligoasthenozoospermia: A prospective, randomized trial. Andrology 2015, 3, 864–867. [Google Scholar] [CrossRef] [PubMed]
- Majzoub, A.; Agarwal, A.; Esteves, S.C. Antioxidants for elevated sperm DNA fragmentation: A mini review. Transl. Androl. Urol. 2017, 6, S649–S653. [Google Scholar] [CrossRef] [PubMed]
- Micic, S.; Lalic, N.; Djordjevic, D.; Bojanic, N.; Bogavac-Stanojevic, N.; Busetto, G.M.; Virmani, A.; Agarwal, A. Double-blind, randomised, placebo-controlled trial on the effect of L-carnitine and L-acetylcarnitine on sperm parameters in men with idiopathic oligoasthenozoospermia. Andrologia 2019, 51, e13267. [Google Scholar] [CrossRef]
- Arafa, M.; Agarwal, A.; Majzoub, A.; Khalafalla, K.; Alsaid, S.; Elbardisi, H. Efficacy of antioxidant supplementation on conventional and advanced sperm function tests in patients with idiopathic male infertility. Fertil. Steril. 2019, 112, e362. [Google Scholar] [CrossRef]
- Gharagozloo, P.; Gutierrez-Adan, A.; Champroux, A.; Noblanc, A.; Kocer, A.; Calle, A.; Perez-Cerezales, S.; Pericuesta, E.; Polhemus, A.; Moazamian, A.; et al. A novel antioxidant formulation designed to treat male infertility associated with oxidative stress: Promising preclinical evidence from animal models. Hum. Reprod. 2016, 31, 252–262. [Google Scholar] [CrossRef]
- Panner Selvam, M.K.; Agarwal, A. Update on the proteomics of male infertility: A systematic review. Arab J. Urol. 2018, 16, 103–112. [Google Scholar] [CrossRef]
- Xu, W.; Hu, H.; Wang, Z.; Chen, X.; Yang, F.; Zhu, Z.; Fang, P.; Dai, J.; Wang, L.; Shi, H.; et al. Proteomic characteristics of spermatozoa in normozoospermic patients with infertility. J. Proteom. 2012, 75, 5426–5436. [Google Scholar] [CrossRef]
- Sharma, R.; Agarwal, A.; Mohanty, G.; Hamada, A.J.; Gopalan, B.; Willard, B.; Yadav, S.; du Plessis, S. Proteomic analysis of human spermatozoa proteins with oxidative stress. Reprod. Biol. Endocrinol. RBE 2013, 11, 48. [Google Scholar] [CrossRef]
- Agarwal, A.; Ayaz, A.; Samanta, L.; Sharma, R.; Assidi, M.; Abuzenadah, A.M.; Sabanegh, E. Comparative proteomic network signatures in seminal plasma of infertile men as a function of reactive oxygen species. Clin. Proteom. 2015, 12, 23. [Google Scholar] [CrossRef]
- Clough, T.; Thaminy, S.; Ragg, S.; Aebersold, R.; Vitek, O. Statistical protein quantification and significance analysis in label-free LC-MS experiments with complex designs. BMC Bioinform. 2012, 13 (Suppl. 16), S6. [Google Scholar] [CrossRef]
- WHO. WHO Laboratory Manual for the Examination and Processing of Human Semen; WHO: Geneva, Switzerland, 2010. [Google Scholar]
- Panner Selvam, M.K.; Agarwal, A.; Sharma, R.; Samanta, L. Treatment of semen samples with α-chymotrypsin alters the expression pattern of sperm functional proteins—A pilot study. Andrology 2018, 6, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Bartolomé, S.; Deutsch, E.W.; Binz, P.-A.; Jones, A.R.; Eisenacher, M.; Mayer, G.; Campos, A.; Canals, F.; Bech-Serra, J.-J.; Carrascal, M. Guidelines for reporting quantitative mass spectrometry based experiments in proteomics. J. Proteom. 2013, 95, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Sekhon, L.H. The role of antioxidant therapy in the treatment of male infertility. Hum. Fertil. 2010, 13, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Balercia, G.; Buldreghini, E.; Vignini, A.; Tiano, L.; Paggi, F.; Amoroso, S.; Ricciardo-Lamonica, G.; Boscaro, M.; Lenzi, A.; Littarru, G. Coenzyme Q10 treatment in infertile men with idiopathic asthenozoospermia: A placebo-controlled, double-blind randomized trial. Fertil. Steril. 2009, 91, 1785–1792. [Google Scholar] [CrossRef] [PubMed]
- Khalafalla, K.; Arafa, M.; Majzoub, A.; Elbardisi, H.; Alsaid, S.; Agarwal, A. Antioxidant combination therapy: A new hope for oligoathenoteratospermic patients. Fertil. Steril. 2019, 112, e365. [Google Scholar] [CrossRef]
- Ayaz, A.; Agarwal, A.; Sharma, R.; Arafa, M.; Elbardisi, H.; Cui, Z. Impact of precise modulation of reactive oxygen species levels on spermatozoa proteins in infertile men. Clin Proteom. 2015, 12, 4. [Google Scholar] [CrossRef]
- Guvvala, P.R.; Ravindra, J.P.; Selvaraju, S.; Arangasamy, A.; Venkata, K.M. Ellagic and ferulic acids protect arsenic-induced male reproductive toxicity via regulating Nfe2l2, Ppargc1a and StAR expressions in testis. Toxicology 2019, 413, 1–12. [Google Scholar] [CrossRef]
- Dias, T.R.; Samanta, L.; Agarwal, A.; Pushparaj, P.N.; Panner Selvam, M.K.; Sharma, R. Proteomic Signatures Reveal Differences in Stress Response, Antioxidant Defense and Proteasomal Activity in Fertile Men with High Seminal ROS Levels. Int. J. Mol. Sci. 2019, 20, 203. [Google Scholar] [CrossRef]
- Said, T.M.; Grunewald, S.; Paasch, U.; Rasch, M.; Agarwal, A.; Glander, H.-J. Effects of magnetic-activated cell sorting on sperm motility and cryosurvival rates. Fertil. Steril. 2005, 83, 1442–1446. [Google Scholar] [CrossRef]
- Dutta, S.; Majzoub, A.; Agarwal, A. Oxidative stress and sperm function: A systematic review on evaluation and management. Arab J. Urol. 2019, 17, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Verratti, V.; Di Giulio, C.; D’Angeli, A.; Tafuri, A.; Francavilla, S.; Pelliccione, F. Sperm forward motility is negatively affected by short-term exposure to altitude hypoxia. Andrologia 2016, 48, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, T.; Rashed, L.; Taymour, M. Seminal cyclooxygenase relationship with oxidative stress in infertile oligoasthenoteratozoospermic men with varicocele. Andrologia 2016, 48, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Perrotta, I.; Santoro, M.; Guido, C.; Avena, P.; Tripepi, S.; De Amicis, F.; Gervasi, M.C.; Aquila, S. Expression of cyclooxygenase-1 (COX-1) and COX-2 in human male gametes from normal patients, and those with varicocele and diabetes: A potential molecular marker for diagnosing male infertility disorders. J. Anat. 2012, 221, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, L.E.; Suzuki, J.; Aguila, L.; Meinsohn, M.-C.; Smith, O.E.; Protopapas, N.; Xu, W.; Sutovsky, P.; Oko, R. Sperm-borne glutathione-S-transferase omega 2 accelerates the nuclear decondensation of spermatozoa during fertilization in mice. Biol. Reprod. 2019, 101, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Schmuck, E.M.; Board, P.G.; Whitbread, A.K.; Tetlow, N.; Cavanaugh, J.A.; Blackburn, A.C.; Masoumi, A. Characterization of the monomethylarsonate reductase and dehydroascorbate reductase activities of Omega class glutathione transferase variants: Implications for arsenic metabolism and the age-at-onset of Alzheimer’s and Parkinson’s diseases. Pharm. Genom. 2005, 15, 493–501. [Google Scholar] [CrossRef]
- Fujii, J.; Imai, H. Redox reactions in mammalian spermatogenesis and the potential targets of reactive oxygen species under oxidative stress. Spermatogenesis 2014, 4, e979108. [Google Scholar] [CrossRef]
- Brutsch, S.H.; Rademacher, M.; Roth, S.R.; Muller, K.; Eder, S.; Viertel, D.; Franz, C.; Kuhn, H.; Borchert, A. Male Subfertility Induced by Heterozygous Expression of Catalytically Inactive Glutathione Peroxidase 4 Is Rescued in Vivo by Systemic Inactivation of the Alox15 Gene. J. Biol. Chem. 2016, 291, 23578–23588. [Google Scholar] [CrossRef]
- Bromfield, E.G.; Walters, J.L.H.; Cafe, S.L.; Bernstein, I.R.; Stanger, S.J.; Anderson, A.L.; Aitken, R.J.; McLaughlin, E.A.; Dun, M.D.; Gadella, B.M.; et al. Differential cell death decisions in the testis: Evidence for an exclusive window of ferroptosis in round spermatids. Mol. Hum. Reprod. 2019, 25, 241–256. [Google Scholar] [CrossRef]
- O’Flaherty, C.; de Souza, A.R. Hydrogen peroxide modifies human sperm peroxiredoxins in a dose-dependent manner. Biol. Reprod. 2011, 84, 238–247. [Google Scholar] [CrossRef]
- Baker, M.A.; Nixon, B.; Naumovski, N.; Aitken, R.J. Proteomic insights into the maturation and capacitation of mammalian spermatozoa. Syst. Biol. Reprod. Med. 2012, 58, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Panner Selvam, M.K.; Agarwal, A.; Pushparaj, P.N.; Baskaran, S.; Bendou, H. Sperm Proteome Analysis and Identification of Fertility-Associated Biomarkers in Unexplained Male Infertility. Genes 2019, 10, 522. [Google Scholar] [CrossRef] [PubMed]
- Samanta, L.; Agarwal, A.; Swain, N.; Sharma, R.; Gopalan, B.; Esteves, S.C.; Durairajanayagam, D.; Sabanegh, E. Proteomic Signatures of Sperm Mitochondria in Varicocele: Clinical Use as Biomarkers of Varicocele Associated Infertility. J. Urol. 2018, 200, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Cui, Y.; Zhang, X.; Lou, J.; Zhou, J.; Bei, H.; Wei, R. Proteomic profile of human spermatozoa in healthy and asthenozoospermic individuals. Reprod. Biol. Endocrinol. 2018, 16, 16. [Google Scholar] [CrossRef] [PubMed]
- Dad, B.R.; Li-Jun, H. Posttranslational Modifications in Spermatozoa and Effects on Male Fertility and Sperm Viability. OMICS A J. Integr. Biol. 2017, 21, 245–256. [Google Scholar]
- Dun, M.D.; Smith, N.D.; Baker, M.A.; Lin, M.; Aitken, R.J.; Nixon, B. The Chaperonin Containing TCP1 Complex (CCT/TRiC) Is Involved in Mediating Sperm-Oocyte Interaction. J. Biol. Chem. 2011, 286, 36875–36887. [Google Scholar] [CrossRef] [PubMed]
- Kubota, H.; Hynes, G.M.; Kerr, S.M.; Willison, K.R. Tissue-specific subunit of the mouse cytosolic chaperonin-containing TCP-1 1. FEBS Lett. 1997, 402, 53–56. [Google Scholar] [CrossRef]
- Freund, A.; Zhong, F.L.; Venteicher, A.S.; Meng, Z.; Veenstra, T.D.; Frydman, J.; Artandi, S.E. Proteostatic control of telomerase function through TRiC-mediated folding of TCAB1. Cell 2014, 159, 1389–1403. [Google Scholar] [CrossRef]
- Baird, D.M.; Britt-Compton, B.; Rowson, J.; Amso, N.N.; Gregory, L.; Kipling, D. Telomere instability in the male germline. Hum. Mol. Genet. 2006, 15, 45–51. [Google Scholar] [CrossRef]
- Thilagavathi, J.; Venkatesh, S.; Dada, R. Telomere length in reproduction. Andrologia 2013, 45, 289–304. [Google Scholar] [CrossRef]
- Ioannou, D.; Griffin, D.K. Male fertility, chromosome abnormalities, and nuclear organization. Cytogenet. Genome Res. 2011, 133, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Nakada, K.; Sato, A.; Yoshida, K.; Morita, T.; Tanaka, H.; Inoue, S.-I.; Yonekawa, H.; Hayashi, J.-I. Mitochondria-related male infertility. Proc. Natl. Acad. Sci. USA 2006, 103, 15148–15153. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sharma, R.K.; Gupta, A.; George, V.; Thomas, A.J., Jr.; Falcone, T.; Agarwal, A. Alterations in mitochondria membrane potential and oxidative stress in infertile men: A prospective observational study. Fertil. Steril. 2003, 80, 844–850. [Google Scholar] [CrossRef]
- Lemasters, J.J.; Qian, T.; Bradham, C.A.; Brenner, D.A.; Cascio, W.E.; Trost, L.C.; Nishimura, Y.; Nieminen, A.-L.; Herman, B. Mitochondrial Dysfunction in the Pathogenesis of Necrotic and Apoptotic Cell Death. J. Bioenerg. Biomembr. 1999, 31, 305–319. [Google Scholar] [CrossRef]
- Wang, C.; Youle, R.J. The role of mitochondria in apoptosis. Annu. Rev. Genet. 2009, 43, 95–118. [Google Scholar] [CrossRef]
- Nowicka-Bauer, K.; Lepczynski, A.; Ozgo, M.; Kamieniczna, M.; Fraczek, M.; Stanski, L.; Olszewska, M.; Malcher, A.; Skrzypczak, W.; Kurpisz, M. Sperm mitochondrial dysfunction and oxidative stress as possible reasons for isolated asthenozoospermia. J. Physiol. Pharm. 2018, 69. [Google Scholar] [CrossRef]
- Agarwal, A.; Sharma, R.; Samanta, L.; Durairajanayagam, D.; Sabanegh, E. Proteomic signatures of infertile men with clinical varicocele and their validation studies reveal mitochondrial dysfunction leading to infertility. Asian J. Androl. 2016, 18, 282–291. [Google Scholar] [CrossRef]
- Kato, T.; Hirano, A.; Manaka, H.; Sasaki, H.; Katagiri, T.; Kawanami, T.; Shikama, Y.; Seino, T.; Sasaki, H. Calcitonin gene-related peptide immunoreactivity in familial amyotrophic lateral sclerosis. Neurosci. Lett. 1991, 133, 163–167. [Google Scholar] [CrossRef]
- Hernansanz-Agustin, P.; Ramos, E.; Navarro, E.; Parada, E.; Sanchez-Lopez, N.; Pelaez-Aguado, L.; Cabrera-Garcia, J.D.; Tello, D.; Buendia, I.; Marina, A.; et al. Mitochondrial complex I deactivation is related to superoxide production in acute hypoxia. Redox Biol. 2017, 12, 1040–1051. [Google Scholar] [CrossRef]
- Durairajanayagam, D.; Rengan, A.K.; Sharma, R.K.; Agarwal, A. Sperm Biology from Production to Ejaculation. In Unexplained Infertility: Pathophysiology, Evaluation and Treatment; Schattman, G.L., Esteves, S.C., Agarwal, A., Eds.; Springer: New York, NY, USA, 2015; pp. 29–42. [Google Scholar] [CrossRef]
- Rajender, S.; Avery, K.; Agarwal, A. Epigenetics, spermatogenesis and male infertility. Mutat. Res. Rev. Mutat. Res. 2011, 727, 62–71. [Google Scholar] [CrossRef]
- Kothandaraman, N.; Agarwal, A.; Abu-Elmagd, M.; Al-Qahtani, M.H. Pathogenic landscape of idiopathic male infertility: New insight towards its regulatory networks. NPJ Genom. Med. 2016, 1, 16023. [Google Scholar] [CrossRef] [PubMed]
- Bracke, A.; Peeters, K.; Punjabi, U.; Hoogewijs, D.; Dewilde, S. A search for molecular mechanisms underlying male idiopathic infertility. Reprod. Biomed. Online 2018, 36, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.Y.; Sha, J.H. Proteomics of spermatogenesis: From protein lists to understanding the regulation of male fertility and infertility. Asian J. Androl. 2011, 13, 18–23. [Google Scholar] [CrossRef] [PubMed]
- O’rand, M.G.; Richardson, R.T.; Yamasaki, N. Expression of the rabbit sperm protein Sp17 in cos cells and interaction of recombinant Sp17 with the rabbit zona pellucida. Mol. Reprod. Dev. 1995, 40, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Grizzi, F.; Chiriva-Internati, M.; Franceschini, B.; Hermonat, P.L.; Soda, G.; Lim, S.H.; Dioguardi, N. Immunolocalization of Sperm Protein 17 in Human Testis and Ejaculated Spermatozoa. J. Histochem. Cytochem. 2003, 51, 1245–1248. [Google Scholar] [CrossRef]
- Intasqui, P.; Agarwal, A.; Sharma, R.; Samanta, L.; Bertolla, R.P. Towards the identification of reliable sperm biomarkers for male infertility: A sperm proteomic approach. Andrologia 2018, 50, e12919. [Google Scholar] [CrossRef]
- Humphries, K.M.; Deal, M.S.; Taylor, S.S. Enhanced dephosphorylation of cAMP-dependent protein kinase by oxidation and thiol modification. J. Biol. Chem. 2005, 280, 2750–2758. [Google Scholar] [CrossRef]
- Burton, K.A.; McDermott, D.A.; Wilkes, D.; Poulsen, M.N.; Nolan, M.A.; Goldstein, M.; Basson, C.T.; McKnight, G.S. Haploinsufficiency at the protein kinase A RI alpha gene locus leads to fertility defects in male mice and men. Mol. Endocrinol. 2006, 20, 2504–2513. [Google Scholar] [CrossRef]
- Hogeveen, K.N.; Sassone-Corsi, P. Regulation of gene expression in post-meiotic male germ cells: CREM-signalling pathways and male fertility. Hum. Fertil. 2006, 9, 73–79. [Google Scholar] [CrossRef]
- Monaco, L.; Kotaja, N.; Fienga, G.; Hogeveen, K.; Kolthur, U.S.; Kimmins, S.; Brancorsini, S.; Macho, B.; Sassone-Corsi, P. Specialized rules of gene transcription in male germ cells: The CREM paradigm. Int. J. Androl. 2004, 27, 322–327. [Google Scholar] [CrossRef]
- Weinbauer, G.F.; Behr, R.; Bergmann, M.; Nieschlag, E. Testicular cAMP responsive element modulator (CREM) protein is expressed in round spermatids but is absent or reduced in men with round spermatid maturation arrest. Mol. Hum. Reprod. 1998, 4, 9–15. [Google Scholar] [CrossRef] [PubMed]
| Function | Proteins (Expression Fold Change) |
|---|---|
| Binding of sperm | CCT2 (2.23), CCT3 (1.93), CCT4 (2.38), CCT5 (2.49), CCT6A (2.92), CCT7 (1.94), CCT8 (2.28), SPA17 (8.81), TCP1 (2.30) |
| Fertilization | LDHC (3.12), PARK7 (2.69), PRKACA (2.53), SPA17 (8.81), SPESP1 (3.89) |
| Maturation of sperm | CLU (2.36), TPP2 (5.30) |
| Spermatogenesis | CCT6B (2.64), DPCD (2.72), HSPA4 (4.08), HSPA4L (3.80), IMMP2L (3.03), NPEPPS (4.16), PDILT (3.69), SPA17 (8.81), TCP1 (2.30) |
| Cellular Pathways | # of DEPs | Activation State | Z-Score |
|---|---|---|---|
| Cell death | 142 | Decreased | −4.786 |
| Necrosis | 124 | Decreased | −4.877 |
| Apoptosis | 89 | Decreased | −3.748 |
| Function/Pathways | Proteins (Expression Fold Change) |
|---|---|
| Mitochondrial function | ACO2 (1.51), ATP5F1A (1.81), ATP5F1B (2.76), ATP5F1C (2.12), ATP5F1D (8.05), ATP5MG (2.57), ATP5PD (2.82), ATP5PO (2.19), CYCS (2.89), HSD17B10 (1.70), NDUFA6 (2.70), NDUFA9 (2.49), NDUFA13 (3.30), NDUFB5 (2.99), NDUFS1 (1.83), NDUFS8 (3.70), PARK7 (2.69), PRDX5 (2.78), UQCRB (2.04) |
| Stabilization of proteins | APOA1 (4.88), CALR (1.73), CCT2 (2.23), CCT3 (1.93), CCT4 (2.38), CCT5 (2.49), CCT6A (2.92), CCT7 (1.94), CCT8 (2.28), CLU (2.36), HSPD1 (3.26), PARK7 (2.69), PHB (3.82), PHB2 (3.32), RPL11 (2.66), RPL5 (2.59), RPS7 (6.00), STX12 (2.84), TCP1 (2.30) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agarwal, A.; Panner Selvam, M.K.; Samanta, L.; Vij, S.C.; Parekh, N.; Sabanegh, E.; Tadros, N.N.; Arafa, M.; Sharma, R. Effect of Antioxidant Supplementation on the Sperm Proteome of Idiopathic Infertile Men. Antioxidants 2019, 8, 488. https://doi.org/10.3390/antiox8100488
Agarwal A, Panner Selvam MK, Samanta L, Vij SC, Parekh N, Sabanegh E, Tadros NN, Arafa M, Sharma R. Effect of Antioxidant Supplementation on the Sperm Proteome of Idiopathic Infertile Men. Antioxidants. 2019; 8(10):488. https://doi.org/10.3390/antiox8100488
Chicago/Turabian StyleAgarwal, Ashok, Manesh Kumar Panner Selvam, Luna Samanta, Sarah C. Vij, Neel Parekh, Edmund Sabanegh, Nicholas N. Tadros, Mohamed Arafa, and Rakesh Sharma. 2019. "Effect of Antioxidant Supplementation on the Sperm Proteome of Idiopathic Infertile Men" Antioxidants 8, no. 10: 488. https://doi.org/10.3390/antiox8100488
APA StyleAgarwal, A., Panner Selvam, M. K., Samanta, L., Vij, S. C., Parekh, N., Sabanegh, E., Tadros, N. N., Arafa, M., & Sharma, R. (2019). Effect of Antioxidant Supplementation on the Sperm Proteome of Idiopathic Infertile Men. Antioxidants, 8(10), 488. https://doi.org/10.3390/antiox8100488