Oxidative Stress, NF-κB-Mediated Inflammation and Apoptosis in the Testes of Streptozotocin–Induced Diabetic Rats: Combined Protective Effects of Malaysian Propolis and Metformin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Extraction of Propolis
2.3. Laboratory Animals
2.4. Induction of Diabetes Mellitus
2.5. Experimental Design
2.6. Weight and Relative Weights of Reproductive Organs
2.7. Sample Collection and Preparation
2.8. Histopathology of the Testes and Epididymis
2.9. Testicular and Epididymal Oxidative Stress Status
2.10. Testicular mRNA Transcript Levels of Antioxidant, Inflammation and Apoptosis-Related Genes
2.10.1. RNA Extraction, Quality and Purity Determination
2.10.2. Real Time Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR)
2.11. Immunohistochemical Study in the Testes
2.12. Statistical Analysis
3. Results
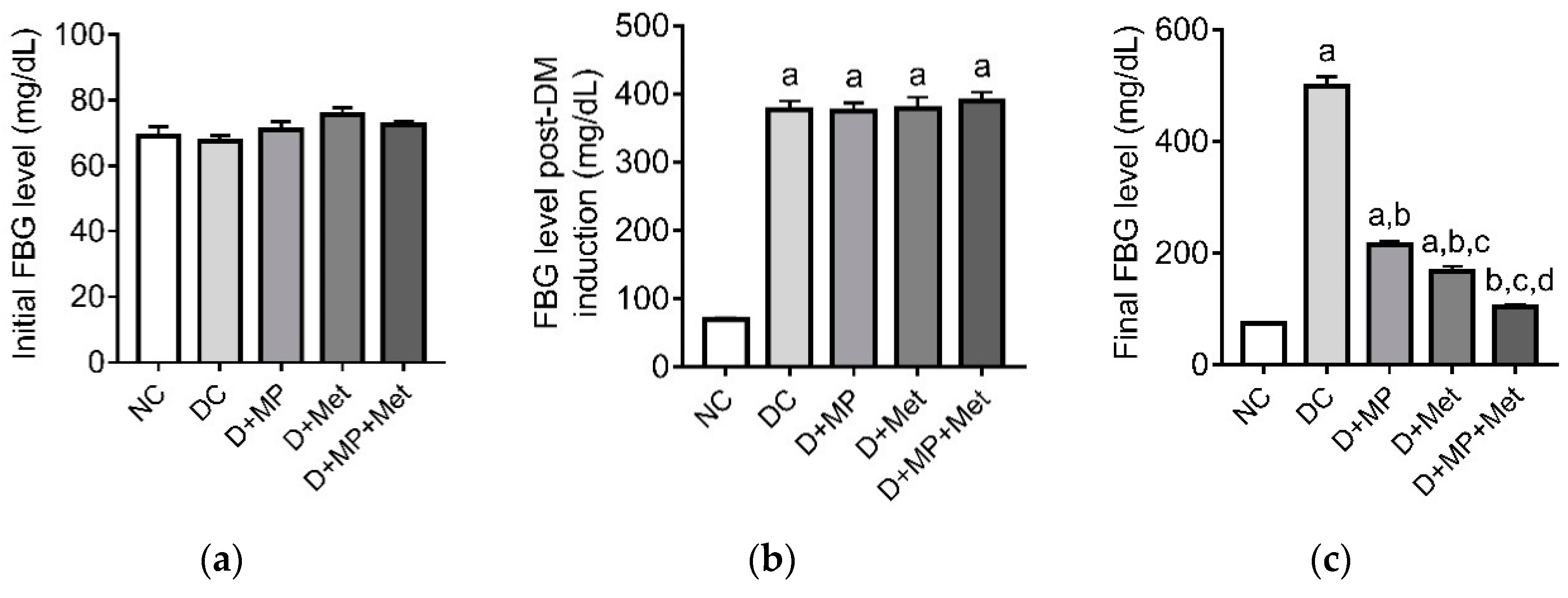
3.1. Blood Glucose Level
3.2. Weights and Relative Weights of Reproductive Organs
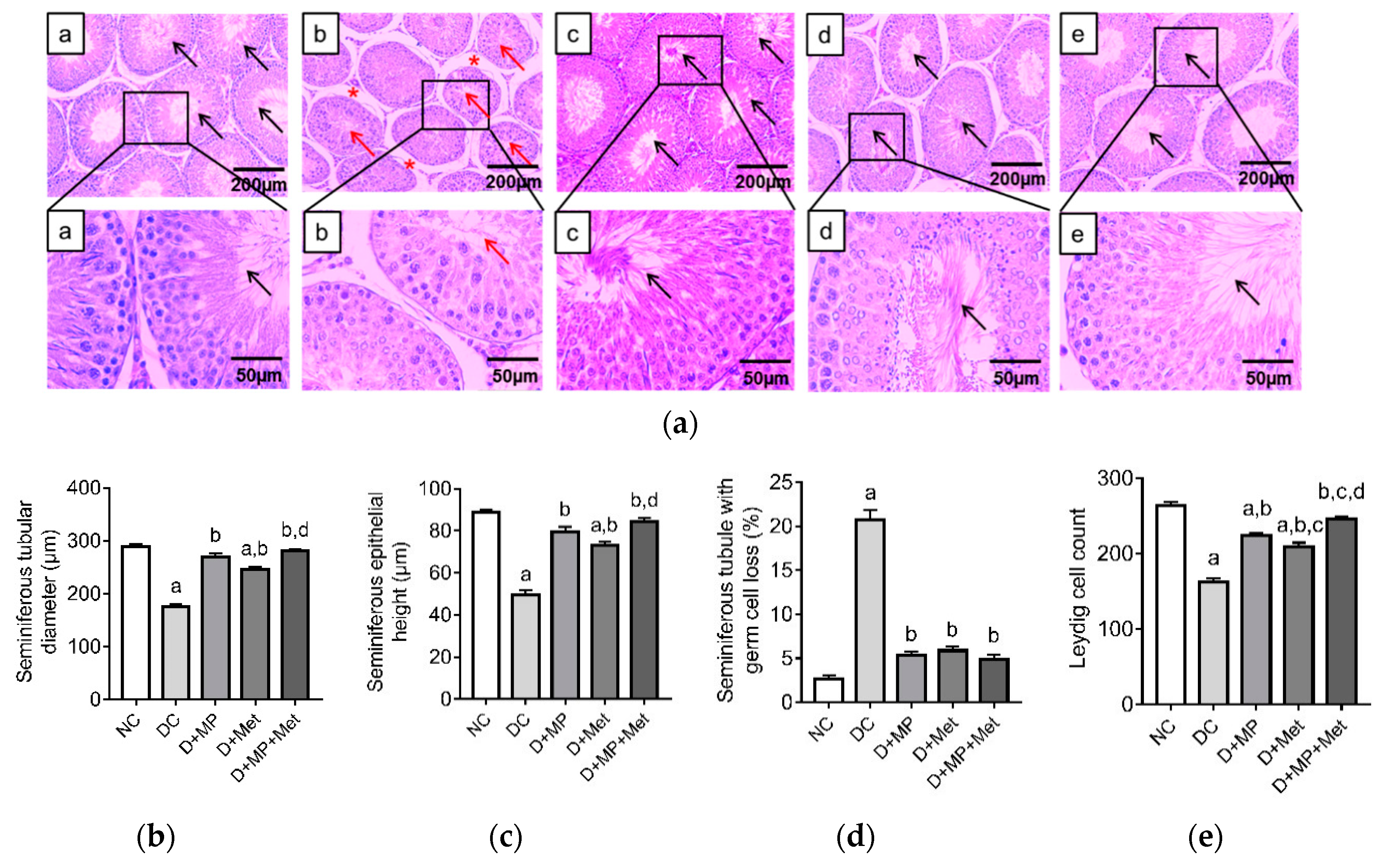
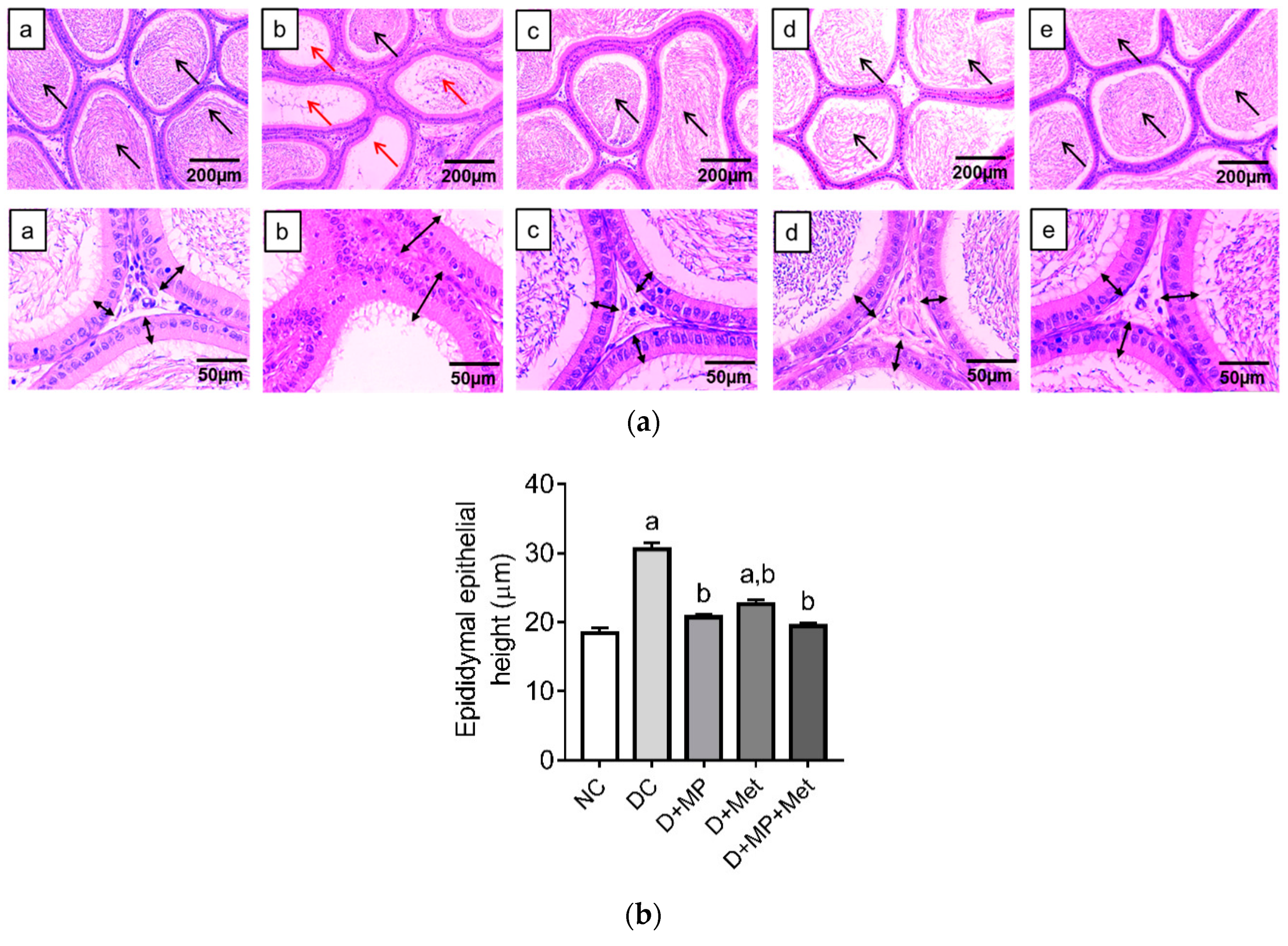
3.3. Histopathology of the Testes and Epididymis
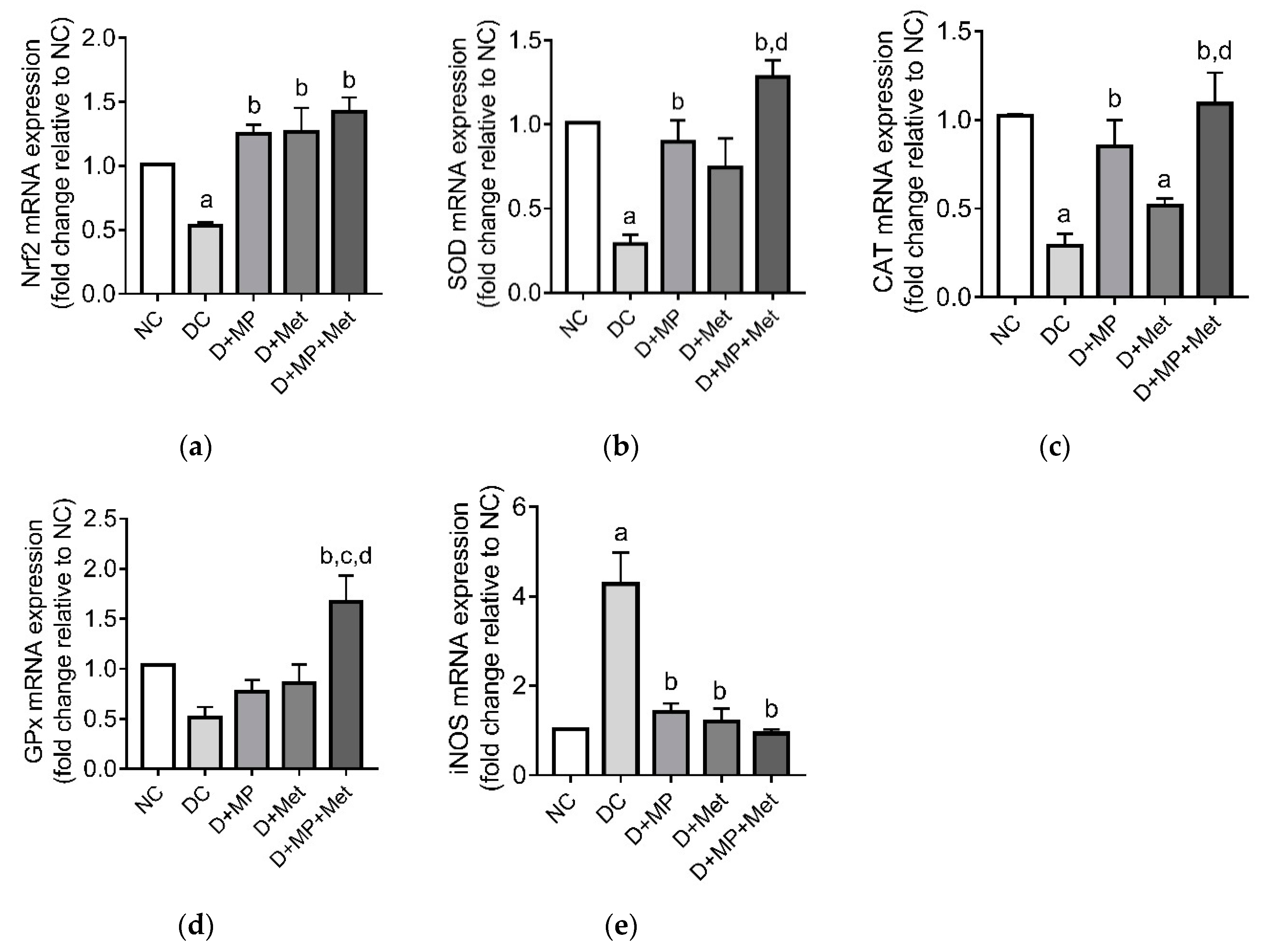
3.4. mRNA Transcript Levels of Antioxidant-Related Genes
3.5. Testicular Antioxidants and Oxidative Stress Status
3.6. Epididymal Antioxidants and Oxidative Stress Status
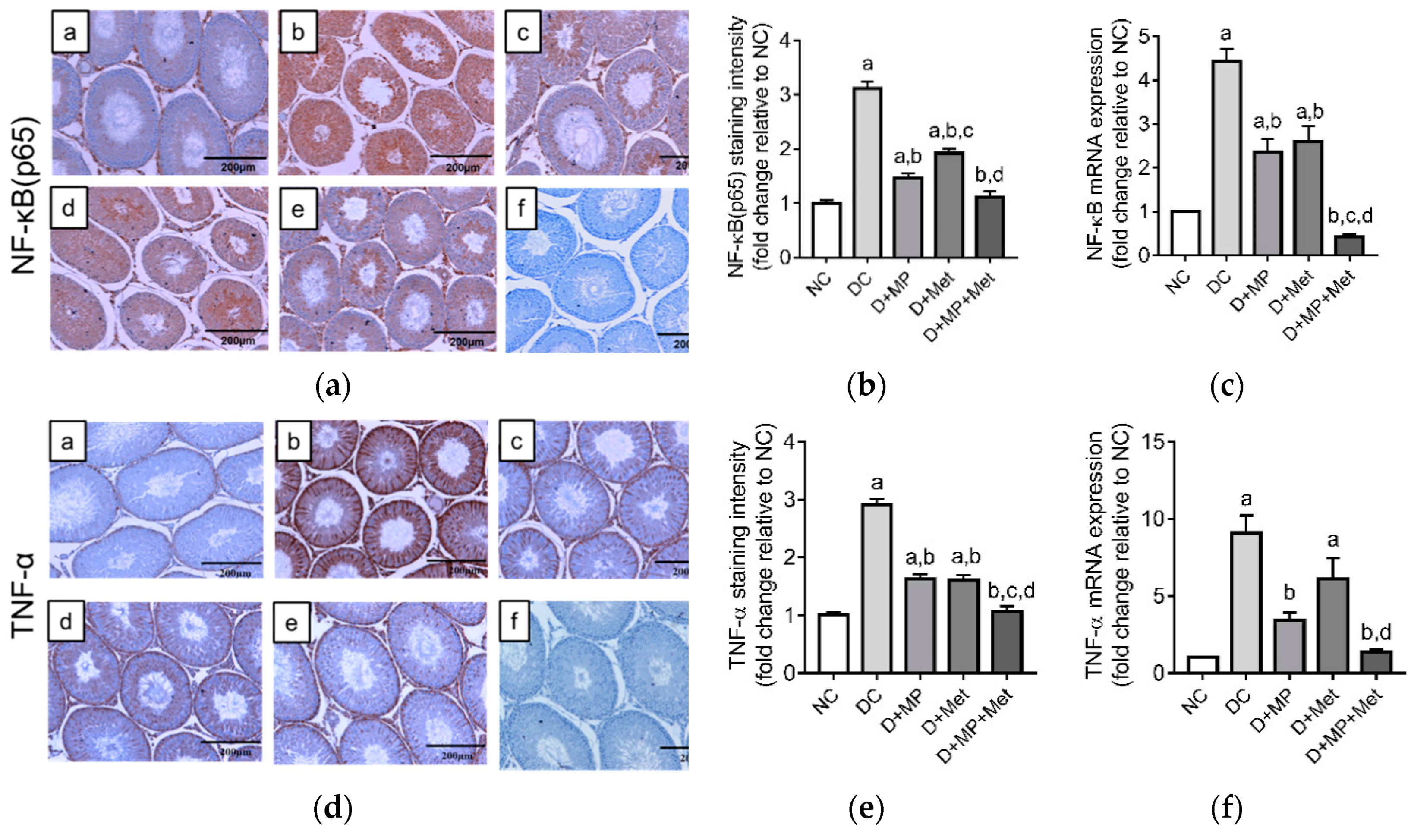
3.7. Testicular mRNA Transcript Levels and Immunoexpressions of Pro and Anti-Inflammatory Proteins
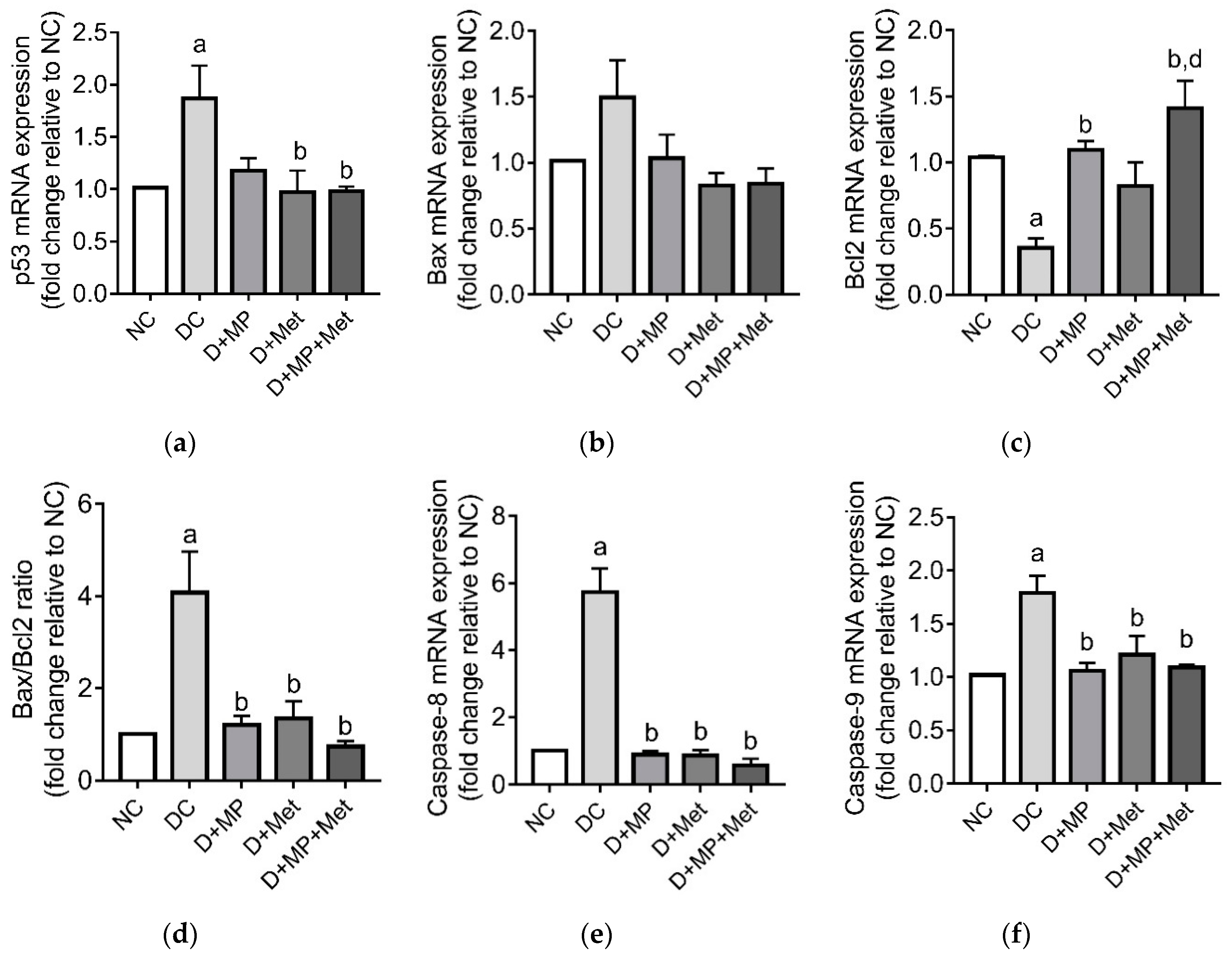
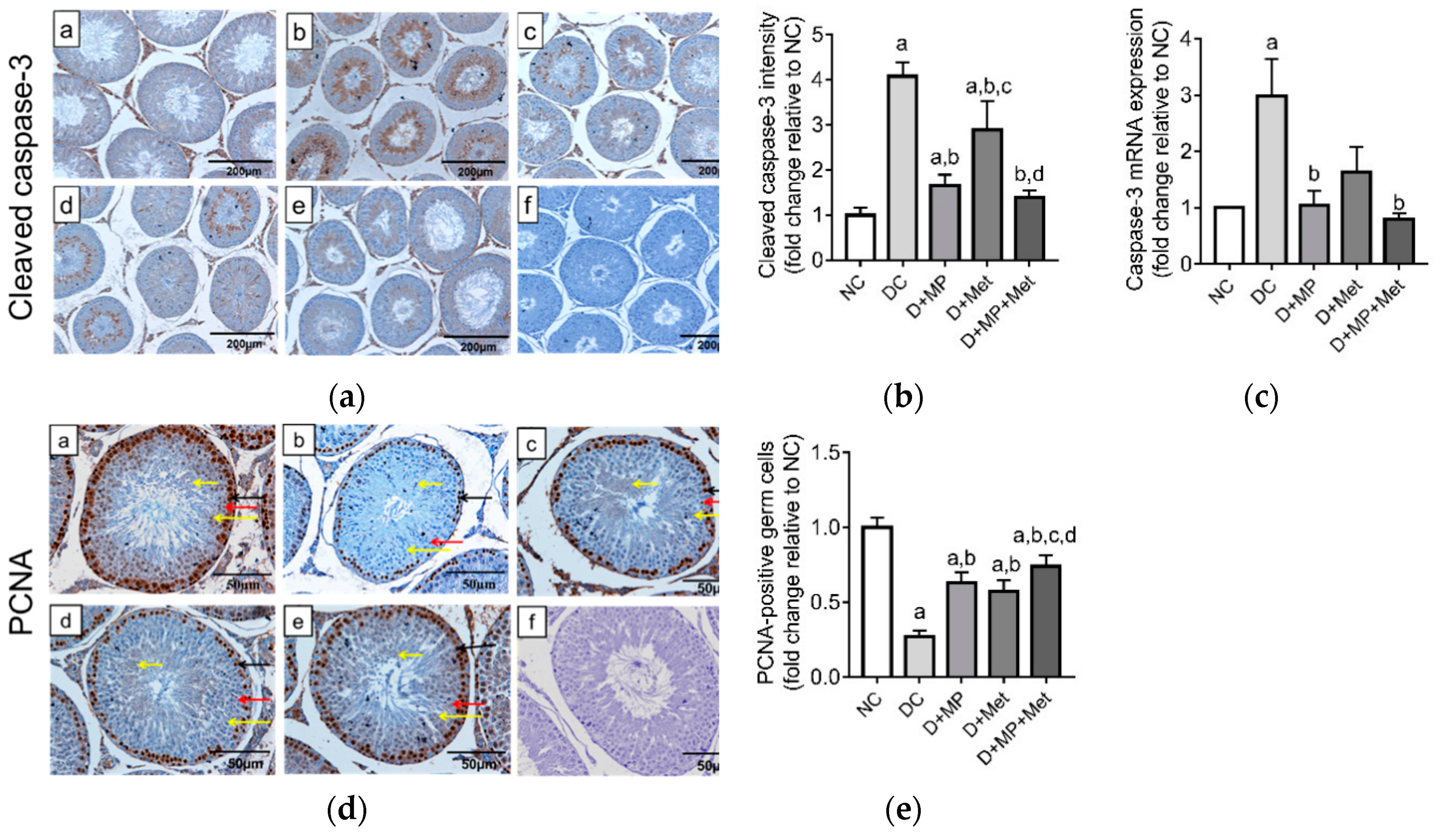
3.8. Testicular Germ Cell Apoptosis and Proliferation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, S.T.; Moley, K.H. Paternal effect on embryo quality in diabetic mice is related to poor sperm quality and associated with decreased glucose transporter expression. Reproduction 2008, 136, 313–322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nna, V.U.; Bakar, A.B.A.; Ahmad, A.; Mohamed, M. Down-regulation of steroidogenesis-related genes and its accompanying fertility decline in streptozotocin-induced diabetic male rats: Ameliorative effect of metformin. Andrology 2019, 7, 110–123. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Z.; Guo, W.; Sun, W.; Miao, X.; Wu, H.; Cong, X.; Wintergerst, K.A.; Kong, X.; Cai, L. Sulforaphane reduction of testicular apoptotic cell death in diabetic mice is associated with the upregulation of Nrf2 expression and function. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E14–E23. [Google Scholar] [CrossRef] [PubMed]
- Rashid, K.; Sil, P.C. Curcumin ameliorates testicular damage in diabetic rats by suppressing cellular stress-mediated mitochondria and endoplasmic reticulum-dependent apoptotic death. Biochim. Biophys. Acta 2015, 1852, 70–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bener, A.; Al-Ansari, A.A.; Zirie, M.; Al-Hamaq, A.O. Is male fertility associated with type 2 diabetes mellitus? Int. Urol. Nephrol. 2009, 41, 777. [Google Scholar] [CrossRef] [PubMed]
- Nna, V.U.; Bakar, A.B.A.; Ahmad, A.; Mohamed, M. Diabetes-induced testicular oxidative stress, inflammation, and caspase-dependent apoptosis: The protective role of metformin. Arch. Physiol Biochem. 2018, 2018. [Google Scholar] [CrossRef]
- Atta, M.S.; Almadaly, E.A.; El-Far, A.H.; Saleh, R.M.; Assar, D.H.; Jaouni, S.K.A.; Mousa, S.A. Thymoquinone defeats diabetes-induced testicular damage in rats targeting antioxidant, inflammatory and aromatase expression. Int. J. Mol. Sci. 2017, 18, 919. [Google Scholar] [CrossRef] [PubMed]
- Maremanda, K.P.; Khan, S.; Jena, G.B. Role of zinc supplementation in testicular and epididymal damages in diabetic rat: Involvement of Nrf2, SOD1, and GPX5. Biol. Trace Elem. Res. 2016, 173, 452–464. [Google Scholar] [CrossRef]
- Bonnefont-Rousselot, D.; Bastard, J.P.; Jaudon, M.C.; Delattre, J. Consequences of the diabetic status on the oxidant/antioxidant balance. Diabetes Metab. 2000, 26, 163–176. [Google Scholar]
- Huang, S.; Zhang, C.P.; Wang, K.; Li, G.Q.; Hu, F.L. Recent advances in the chemical composition of propolis. Molecules 2014, 19, 19610–19632. [Google Scholar] [CrossRef]
- Burdock, G.A. Review of the biological properties and toxicity of bee propolis (propolis). Food Chem. Toxicol. 1998, 36, 347–363. [Google Scholar] [CrossRef]
- Pietta, P.G.; Gardana, C.; Pietta, A.M. Analytical methods for quality control of propolis. Fitoterapia 2002, 73 (Suppl. 1), S7–S20. [Google Scholar] [CrossRef]
- Sforcin, J.M. Biological properties and therapeutic applications of propolis. Phytother. Res. 2016, 30, 894–905. [Google Scholar] [CrossRef] [PubMed]
- Velikova, M.; Bankova, V.; Sorkun, K.; Houcine, S.; Tsvetkova, I.; Kujumgiev, A. Propolis from the mediterranean region: Chemical composition and antimicrobial activity. Z. Nat. C 2000, 55, 790–793. [Google Scholar] [CrossRef] [PubMed]
- Athikomkulchai, S.; Awale, S.; Ruangrungsi, N.; Ruchirawat, S.; Kadota, S. Chemical constituents of Thai propolis. Fitoterapia 2013, 88, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Massaro, C.F.; Smyth, T.J.; Smyth, W.F.; Heard, T.; Leonhardt, S.D.; Katouli, M.; Wallace, H.M.; Brooks, P. Phloroglucinols from anti-microbial deposit-resins of australian stingless bees (Tetragonula carbonaria). Phytother. Res. 2015, 29, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Omar, R.M.; Igoli, J.; Gray, A.I.; Ebiloma, G.U.; Clements, C.; Fearnley, J.; Ebel, R.A.; Zhang, T.; de Koning, H.P.; Watson, D.G. Chemical characterisation of Nigerian red propolis and its biological activity against Trypanosoma brucei. Phytochem. Anal. 2016, 27, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Usman, U.Z.; Bakar, A.; Mohamed, M. Phytochemical screening and comparison of antioxidant activity of water and ethanol extract propolis from Malaysia. Int. J. Pharm. Pharm. Sci. 2016, 8, 413–415. [Google Scholar]
- Orsi, R.D.O.; Fernandes, A.; Bankova, V.; Sforcin, J. The effects of Brazilian and Bulgarian propolis in vitro against Salmonella typhi and their synergism with antibiotics acting on the ribosome. Nat. Prod. Res. 2012, 26, 430–437. [Google Scholar] [CrossRef]
- Nna, V.U.; Bakar, A.B.A.; Lazin, M.R.M.L.M.; Mohamed, M. Antioxidant, anti-inflammatory and synergistic anti-hyperglycemic effects of Malaysian propolis and metformin in streptozotocin-induced diabetic rats. Food Chem. Toxicol. 2018, 120, 305–320. [Google Scholar] [CrossRef]
- Araby, B.E.; Ahmed, D.F.I.; Zahkouk, S.A. Effect of Foeniculum vulgare and propolis on liver in alloxan diabetic rats. Adv. Biol. Res. 2017, 11, 311–318. [Google Scholar]
- Granados-Pineda, J.; Uribe-Uribe, N.; Garcia-Lopez, P.; Ramos-Godinez, M.D.P.; Rivero-Cruz, J.F.; Perez-Rojas, J.M. Effect of pinocembrin isolated from Mexican brown propolis on diabetic nephropathy. Molecules 2018, 23, 852. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, R.; Tanvir, E.M.; Hossen, M.S.; Afroz, R.; Ahmmed, I.; Rumpa, N.E.; Paul, S.; Gan, S.H.; Sulaiman, S.A.; Khalil, M.I. Antioxidant properties and cardioprotective mechanism of Malaysian propolis in rats. Evid. Based Complement. Alternat. Med. 2017, 2017, 5370545. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.H.; Chien, Y.W.; Chang, M.L.; Hou, C.C.; Chan, C.H.; Tang, H.W.; Huang, H.Y. Taiwanese green propolis ethanol extract delays the progression of type 2 diabetes mellitus in rats treated with streptozotocin/high-fat diet. Nutrients 2018, 10, 503. [Google Scholar] [CrossRef] [PubMed]
- Wali, A.F.; Avula, B.; Ali, Z.; Khan, I.A.; Mushtaq, A.; Rehman, M.U.; Akbar, S.; Masoodi, M.H. Antioxidant, hepatoprotective potential and chemical profiling of propolis ethanolic extract from Kashmir Himalaya region using UHPLC-DAD-QTOF-MS. Biomed. Res. Int. 2015, 2015, 393462. [Google Scholar] [CrossRef] [PubMed]
- Nna, V.U.; Bakar, A.B.A.; Mohamed, M. Malaysian propolis, metformin and their combination, exert hepatoprotective effect in streptozotocin-induced diabetic rats. Life Sci. 2018, 211, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Zakerkish, M.; Jenabi, M.; Zaeemzadeh, N.; Hemmati, A.A.; Neisi, N. The effect of Iranian propolis on glucose metabolism, lipid profile, insulin resistance, renal function and inflammatory biomarkers in patients with type 2 diabetes mellitus: A randomized double-blind clinical trial. Sci. Rep. 2019, 9, 7289. [Google Scholar] [CrossRef]
- Afsharpour, F.; Javadi, M.; Hashemipour, S.; Koushan, Y. Propolis supplementation improves glycemic and antioxidant status in patients with type 2 diabetes: A randomized, double-blind, placebo-controlled study. Complement. Ther. Med. 2019, 43, 283–288. [Google Scholar] [CrossRef]
- Gao, W.; Pu, L.; Wei, J.; Yao, Z.; Wang, Y.; Shi, T.; Zhao, L.; Jiao, C.; Guo, C. Serum antioxidant parameters are significantly increased in patients with type 2 diabetes mellitus after consumption of Chinese propolis: A randomized controlled trial based on fasting serum glucose level. Diabetes Ther. 2018, 9, 101–111. [Google Scholar] [CrossRef]
- Fukuda, T.; Fukui, M.; Tanaka, M.; Senmaru, T.; Iwase, H.; Yamazaki, M.; Aoi, W.; Inui, T.; Nakamura, N.; Marunaka, Y. Effect of Brazilian green propolis in patients with type 2 diabetes: A double-blind randomized placebo-controlled study. Biomed. Rep. 2015, 3, 355–360. [Google Scholar] [CrossRef]
- Usman, U.Z.; Bakar, A.B.A.; Zin, A.A.M.; Mohamed, M. LC-MS analysis and effects of Malaysian propolis on insulin, glucagon, pancreas and oxidative stress status in streptozotocin-induced diabetic rats. J. Med. Biomed. Res. 2017, 16, 15–27. [Google Scholar]
- Usman, U.Z.; Mohamed, M. Analysis of phytochemical compounds in water and ethanol extracts of Malaysian propolis. Int. J. Pharma Bio. Sci. 2015, 6, P374–P380. [Google Scholar]
- Annuk, M.; Zilmer, M.; Lind, L.; Linde, T.; Fellstrom, B. Oxidative stress and endothelial function in chronic renal failure. J. Am. Soc. Nephrol. 2001, 12, 2747–2752. [Google Scholar] [PubMed]
- Sun, Y.; Oberley, L.W.; Li, Y. A simple method for clinical assay of superoxide dismutase. Clin. Chem. 1988, 34, 497–500. [Google Scholar] [PubMed]
- Goth, L. A simple method for determination of serum catalase activity and revision of reference range. Clin. Chim. Acta 1991, 196, 143–151. [Google Scholar] [CrossRef]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione s-transferases. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar]
- Carlberg, I.; Mannervik, B. Glutathione reductase. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1985; Volume 113, pp. 484–490. [Google Scholar]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Koracevic, D.; Koracevic, G.; Djordjevic, V.; Andrejevic, S.; Cosic, V. Method for the measurement of antioxidant activity in human fluids. J. Clin. Pathol. 2001, 54, 356–361. [Google Scholar] [CrossRef] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Faid, I.; Al-Hussaini, H.; Kilarkaje, N. Resveratrol alleviates diabetes-induced testicular dysfunction by inhibiting oxidative stress and c-Jun N-terminal kinase signaling in rats. Toxicol. Appl. Pharmacol. 2015, 289, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Khavarimehr, M.; Nejati, V.; Razi, M.; Najafi, G. Ameliorative effect of omega-3 on spermatogenesis, testicular antioxidant status and preimplantation embryo development in streptozotocin-induced diabetes in rats. Int. Urol. Nephrol. 2017, 49, 1545–1560. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, N.A.; Ahmed, O.M.; Hozayen, W.G.; Ahmed, M.A. Ameliorative effects of bee pollen and date palm pollen on the glycemic state and male sexual dysfunctions in streptozotocin-induced diabetic wistar rats. Biomed. Pharmacother. 2018, 97, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.M.; Swamy, V.; Gopkumar, P.; Dhanapal, R. Anti-diabetic activity of Terminalia catappa Linn. leaf extracts in alloxan-induced diabetic rats. Iran. J. Pharmacol. Ther. 2005, 4, 36-0. [Google Scholar]
- Abo-Salem, O.M.; El-Edel, R.H.; Harisa, G.; El-Halawany, N.; Ghonaim, M.M. Experimental diabetic nephropathy can be prevented by propolis: Effect on metabolic disturbances and renal oxidative parameters. Pak. J. Pharm. Sci. 2009, 22, 205–210. [Google Scholar] [PubMed]
- Sameni, H.R.; Ramhormozi, P.; Bandegi, A.R.; Taherian, A.A.; Mirmohammadkhani, M.; Safari, M. Effects of ethanol extract of propolis on histopathological changes and anti-oxidant defense of kidney in a rat model for type 1 diabetes mellitus. J. Diabetes Investig. 2016, 7, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Chen, S.; Evans, T.; Deng, D.X.; Mukherjee, K.; Chakrabarti, S. Apoptotic germ-cell death and testicular damage in experimental diabetes: Prevention by endothelin antagonism. Urol. Res. 2000, 28, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Cameron, D.F.; Murray, F.T.; Drylie, D.D. Interstitial compartment pathology and spermatogenic disruption in testes from impotent diabetic men. Anat. Rec. 1985, 213, 53–62. [Google Scholar] [CrossRef]
- Cameron, D.F.; Rountree, J.; Schultz, R.E.; Repetta, D.; Murray, F.T. Sustained hyperglycemia results in testicular dysfunction and reduced fertility potential in BBWOR diabetic rats. Am. J. Physiol. 1990, 259, E881–E889. [Google Scholar] [CrossRef]
- Soudamani, S.; Malini, T.; Balasubramanian, K. Effects of streptozotocin-diabetes and insulin replacement on the epididymis of prepubertal rats: Histological and histomorphometric studies. Endocr. Res. 2005, 31, 81–98. [Google Scholar] [CrossRef]
- Fernandes, G.S.; Fernandez, C.D.; Campos, K.E.; Damasceno, D.C.; Anselmo-Franci, J.A.; Kempinas, W.D. Vitamin C partially attenuates male reproductive deficits in hyperglycemic rats. Reprod. Biol. Endocrinol. 2011, 9, 100. [Google Scholar] [CrossRef] [PubMed]
- Rizk, S.M.; Zaki, H.F.; Mina, M.A. Propolis attenuates doxorubicin-induced testicular toxicity in rats. Food Chem. Toxicol. 2014, 67, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Çilenk, K.T.; Öztürk, İ.; Sönmez, M.F. Ameliorative effect of propolis on the cadmium-induced reproductive toxicity in male albino rats. Exp. Mol. Pathol. 2016, 101, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Sönmez, M.F.; Çilenk, K.T.; Karabulut, D.; Ünalmış, S.; Deligönül, E.; Öztürk, İ.; Kaymak, E. Protective effects of propolis on methotrexate-induced testis injury in rat. Biomed. Pharmacother. 2016, 79, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S.; Nayak, G.; Lukose, S.T.; Kalthur, S.G.; Bhat, N.; Hegde, A.R.; Mutalik, S.; Kalthur, G.; Adiga, S.K. Indian propolis ameliorates the mitomycin C-induced testicular toxicity by reducing DNA damage and elevating the antioxidant activity. Biomed. Pharmacother. 2017, 95, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Kanter, M.; Aktas, C.; Erboga, M. Curcumin attenuates testicular damage, apoptotic germ cell death, and oxidative stress in streptozotocin-induced diabetic rats. Mol. Nutr. Food Res. 2013, 57, 1578–1585. [Google Scholar] [CrossRef] [PubMed]
- Pitocco, D.; Tesauro, M.; Alessandro, R.; Ghirlanda, G.; Cardillo, C. Oxidative stress in diabetes: Implications for vascular and other complications. Int. J. Mol. Sci. 2013, 14, 21525–21550. [Google Scholar] [CrossRef] [PubMed]
- Moazamian, R.; Polhemus, A.; Connaughton, H.; Fraser, B.; Whiting, S.; Gharagozloo, P.; Aitken, R.J. Oxidative stress and human spermatozoa: Diagnostic and functional significance of aldehydes generated as a result of lipid peroxidation. MHR Basic Sci. Reprod. Med. 2015, 21, 502–515. [Google Scholar] [CrossRef]
- Jiang, X.; Bai, Y.; Zhang, Z.; Xin, Y.; Cai, L. Protection by sulforaphane from type 1 diabetes-induced testicular apoptosis is associated with the up-regulation of Nrf2 expression and function. Toxicol. Appl. Pharmacol. 2014, 279, 198–210. [Google Scholar] [CrossRef]
- Artimani, T.; Amiri, I.; Asl, S.S.; Saidijam, M.; Hasanvand, D.; Afshar, S. Amelioration of diabetes-induced testicular and sperm damage in rats by cerium oxide nanoparticle treatment. Andrologia 2018, 50, e13089. [Google Scholar] [CrossRef]
- Hasanvand, D.; Amiri, I.; Asl, S.S.; Saidijam, M.; Shabab, N.; Artimani, T. Effects of CeO2 nanoparticles on the HO-1, NQO1, and GCLc expression in the testes of diabetic rats. Can. J. Physiol. Pharmacol. 2018, 96, 963–969. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Song, W.; Wang, Z.; Wang, Z.; Jin, X.; Xu, J.; Bai, L.; Li, Y.; Cui, J.; Cai, L. Resveratrol attenuates testicular apoptosis in type 1 diabetic mice: Role of Akt-mediated Nrf2 activation and p62-dependent Keap1 degradation. Redox Biol. 2018, 14, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Ross, D.; Siegel, D. Functions of NQO1 in cellular protection and CoQ10 metabolism and its potential role as a redox sensitive molecular switch. Front. Physiol. 2017, 8, 595. [Google Scholar] [CrossRef] [PubMed]
- Niture, S.K.; Kaspar, J.W.; Shen, J.; Jaiswal, A.K. Nrf2 signaling and cell survival. Toxicol. Appl. Pharmacol. 2010, 244, 37–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, B.N.; Lawson, G.; Chan, J.Y.; Banuelos, J.; Cortes, M.M.; Hoang, Y.D.; Ortiz, L.; Rau, B.A.; Luderer, U. Knockout of the transcription factor Nrf2 disrupts spermatogenesis in an age-dependent manner. Free Radic. Biol. Med. 2010, 49, 1368–1379. [Google Scholar] [CrossRef]
- Afifi, M.; Almaghrabi, O.A.; Kadasa, N.M. Ameliorative effect of zinc oxide nanoparticles on antioxidants and sperm characteristics in streptozotocin-induced diabetic rat testes. Biomed. Res. Int. 2015, 2015, 153573. [Google Scholar] [CrossRef]
- Sekhar, K.R.; Rachakonda, G.; Freeman, M.L. Cysteine-based regulation of the CUL3 adaptor protein Keap1. Toxicol. Appl. Pharmacol. 2010, 244, 21–26. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.-L.; Chang, J.-C.; Lin, W.-Y.; Li, C.-C.; Hsieh, M.; Chen, H.-W.; Wang, T.-S.; Wu, W.-T.; Liu, C.-S.; Liu, K.-L. Caffeic acid and resveratrol ameliorate cellular damage in cell and drosophila models of spinocerebellar ataxia type 3 through upregulation of Nrf2 pathway. Free Radic. Biol. Med. 2018, 115, 309–317. [Google Scholar] [CrossRef]
- Liu, Y.-W.; Liu, X.-L.; Kong, L.; Zhang, M.-Y.; Chen, Y.-J.; Zhu, X.; Hao, Y.-C. Neuroprotection of quercetin on central neurons against chronic high glucose through enhancement of Nrf2/ARE/glyoxalase-1 pathway mediated by phosphorylation regulation. Biomed. Pharmacother. 2019, 109, 2145–2154. [Google Scholar] [CrossRef]
- Aprioku, J.S. Pharmacology of free radicals and the impact of reactive oxygen species on the testis. J. Reprod. Infertil. 2013, 14, 158–172. [Google Scholar]
- Reddy, K.P.; Rao, M.N.; Murthy, J.S.R.; Reddy, P.S. Lead aggravates the diabetic-induced reproductive toxicity in male wistar rats. Toxicol. Res. 2016, 5, 1465–1476. [Google Scholar] [CrossRef] [PubMed]
- Somogyi, A.; Rosta, K.; Pusztai, P.; Tulassay, Z.; Nagy, G. Antioxidant measurements. Physiol. Meas. 2007, 28, R41–R55. [Google Scholar] [CrossRef]
- Usman, U.Z.; Bakar, A.B.A.; Mohamed, M. Propolis improves pregnancy outcomes and placental oxidative stress status in streptozotocin-induced diabetic rats. BMC Complement. Altern. Med. 2018, 18, 324. [Google Scholar] [CrossRef] [PubMed]
- Kanter, M.; Aktas, C.; Erboga, M. Protective effects of quercetin against apoptosis and oxidative stress in streptozotocin-induced diabetic rat testis. Food Chem. Toxicol. 2012, 50, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Rochette, L.; Zeller, M.; Cottin, Y.; Vergely, C. Diabetes, oxidative stress and therapeutic strategies. Biochim. Biophys. Acta 2014, 1840, 2709–2729. [Google Scholar] [CrossRef] [PubMed]
- Adela, R.; Nethi, S.K.; Bagul, P.K.; Barui, A.K.; Mattapally, S.; Kuncha, M.; Patra, C.R.; Reddy, P.N.; Banerjee, S.K. Hyperglycaemia enhances nitric oxide production in diabetes: A study from South Indian patients. PLoS ONE 2015, 10, e0125270. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Gu, Q.; Xiang, L.; Dong, X.; Li, H.; Ni, J.; Wan, L.; Cai, G.; Chen, G. Curcumin inhibits apoptosis by modulating Bax/Bcl-2 expression and alleviates oxidative stress in testes of streptozotocin-induced diabetic rats. Ther. Clin. Risk Manag. 2017, 13, 1099–1105. [Google Scholar] [CrossRef]
- Zhao, Y.; Tan, Y.; Dai, J.; Li, B.; Guo, L.; Cui, J.; Wang, G.; Shi, X.; Zhang, X.; Mellen, N. Exacerbation of diabetes-induced testicular apoptosis by zinc deficiency is most likely associated with oxidative stress, p38 MAPK activation, and p53 activation in mice. Toxicol. Lett. 2011, 200, 100–106. [Google Scholar] [CrossRef]
- Li, G.Y.; Xie, P.; Li, H.Y.; Hao, L.; Xiong, Q.; Qiu, T. Involment of p53, Bax, and Bcl-2 pathway in microcystins-induced apoptosis in rat testis. Environ. Toxicol. 2011, 26, 111–117. [Google Scholar] [CrossRef]
- Liu, G.; Gong, P.; Bernstein, L.R.; Bi, Y.; Gong, S.; Cai, L. Apoptotic cell death induced by low-dose radiation in male germ cells: Hormesis and adaptation. Crit. Rev. Toxicol. 2007, 37, 587–605. [Google Scholar] [CrossRef]
- Martineau, L.C. Large enhancement of skeletal muscle cell glucose uptake and suppression of hepatocyte glucose-6-phosphatase activity by weak uncouplers of oxidative phosphorylation. Biochim. Biophys. Acta (BBA) Gen. Subj. 2012, 1820, 133–150. [Google Scholar] [CrossRef] [PubMed]

| Parameter | NC | DC | D+MP | D+Met | D+MP+Met | |
|---|---|---|---|---|---|---|
| Testes | AW (g) | 3.64 ± 0.39 | 1.49 ± 0.36 a | 3.31 ± 0.40 b | 2.86 ± 0.20 a,b | 3.37 ± 0.37 b,d |
| RW (%) | 0.99 ± 0.18 | 0.75 ± 0.18 a | 1.03 ± 0.15 b | 0.91 ± 0.11 | 0.97 ± 0.16 | |
| Epididymis | AW (g) | 1.30 ± 0.17 | 0.62 ± 0.14 a | 1.05 ± 0.04 a,b | 0.93 ± 0.16 a,b | 1.17 ± 0.10 b,d |
| RW (%) | 0.35 ± 0.06 | 0.32 ± 0.09 | 0.33 ± 0.04 | 0.30 ± 0.06 | 0.34 ± 0.06 | |
| Prostate | AW (g) | 0.59 ± 0.13 | 0.21 ± 0.05 a | 0.52 ± 0.11 b | 0.36 ± 0.08 a | 0.65 ± 0.21 b,d |
| RW (%) | 0.17 ± 0.04 | 0.11 ± 0.03 a | 0.16 ± 0.04 b | 0.12 ± 0.03 a | 0.18 ± 0.05 b,d | |
| Seminal vesicle | AW (g) | 1.92 ± 0.27 | 0.34 ± 0.11 a | 1.25 ± 0.34 a,b | 1.01 ± 0.26 a,b | 1.92 ± 0.28 b,c,d |
| RW (%) | 0.52 ± 0.10 | 0.17 ± 0.06 a | 0.39 ± 0.11 a,b | 0.32 ± 0.08 a,b | 0.55 ± 0.07 b,c,d | |
| Groups | NC | DC | D+MP | D+Met | D+MP+Met |
|---|---|---|---|---|---|
| SOD activity (unit/mg protein) | 2.21 ± 0.08 | 1.32 ± 0.08 a | 2.06 ± 0.19 b | 1.98 ± 0.12 a,b | 2.23 ± 0.13 b,d |
| CAT activity (unit/mg protein) | 38.88 ± 4.85 | 11.51 ± 2.98 a | 32.36 ± 4.42 b | 27.24 ± 3.53 a,b | 38.49 ± 7.24 b,d |
| GPx activity (unit/mg protein) | 35.83 ± 3.17 | 10.46 ± 1.84 a | 27.51 ± 2.71 a,b | 22.87 ± 3.21 a,b | 30.41 ± 2.39 a,b,d |
| GST activity (unit/mg protein) | 167.90 ± 7.52 | 119.50 ± 4.84 a | 161.90 ± 5.16 b | 156.30 ± 5.23 a,b | 167.50 ± 4.24 b,d |
| GR activity (unit/mg protein) | 23.52 ± 2.36 | 11.01 ± 1.38 a | 17.76 ± 2.04 a,b | 16.19 ± 1.01 a,b | 20.85 ± 2.31 b,c,d |
| GSH level (nmol GSH Eq./mg protein) | 2.96 ± 0.48 | 1.15 ± 0.19 a | 2.35 ± 0.28 a,b | 2.01 ± 0.24 a,b | 2.61 ± 0.41 b,d |
| TAC (nmol uric acid Eq./mg protein) | 173.70 ± 8.60 | 77.46 ± 9.37 a | 158.00 ± 9.58 a,b | 141.50 ± 9.50 a,b,c | 183.50 ± 4.80 b,c,d |
| TBARS level (nmol/mg protein) | 1.03 ± 0.15 | 3.53 ± 0.26 a | 1.57 ± 0.29 a,b | 2.40 ± 0.31 a,b,c | 1.38 ± 0.27 b,d |
| Nitric oxide level (µmol/g tissue) | 2.66 ± 0.93 | 14.03 ± 2.41 a | 3.62 ± 0.98 b | 6.68 ± 1.57 a,b,c | 3.62 ± 0.52 b,d |
| Groups | NC | DC | D+MP | D+Met | D+MP+Met |
|---|---|---|---|---|---|
| SOD activity (unit/mg protein) | 2.89 ± 0.38 | 1.22 ± 0.26 a | 2.43 ± 0.22 b | 2.21 ± 0.31 a,b | 2.76 ± 0.40 b,d |
| CAT activity (unit/mg protein) | 28.32 ± 3.02 | 15.18 ± 3.00 a | 24.24 ± 2.89 a,b | 23.27 ± 2.50 a,b | 26.46 ± 2.03 b |
| GPx activity (unit/mg protein) | 34.08 ± 4.89 | 16.77 ± 3.06 a | 30.06 ± 3.34 b | 27.77 ± 2.27 a,b | 35.33 ± 3.22 b,c,d |
| GST activity (unit/mg protein) | 140.40 ± 13.57 | 91.15 ± 2.77 a | 104.80 ± 13.26 a | 108.50 ± 6.26 a,b | 127.00 ± 9.02 b,c,d |
| GR activity (unit/mg protein) | 22.29 ± 1.39 | 10.31 ± 1.29 a | 18.41 ± 1.26 a,b | 16.64 ± 1.59 a,b | 18.75 ± 1.41 b,c,d |
| GSH level (nmol GSH Eq./mg protein) | 2.28 ± 0.32 | 0.91 ± 0.12 a | 2.01 ± 0.25 b | 1.93 ± 0.25 b | 2.15 ± 0.29 b |
| TBARS level (nmol/mg protein) | 1.61 ± 0.19 | 7.05 ± 0.74 a | 3.73 ± 0.59 a,b | 4.45 ± 0.80 a,b | 2.32 ± 0.47 b,c,d |
| TAC (nmol uric acid Eq/mg protein) | 249.60 ± 17.79 | 95.57 ± 11.15 a | 197.40 ± 17.19 a,b | 153.20 ± 18.56 a,b,c | 224.10 ± 15.52 a,b,c,d |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nna, V.U.; Abu Bakar, A.B.; Ahmad, A.; Eleazu, C.O.; Mohamed, M. Oxidative Stress, NF-κB-Mediated Inflammation and Apoptosis in the Testes of Streptozotocin–Induced Diabetic Rats: Combined Protective Effects of Malaysian Propolis and Metformin. Antioxidants 2019, 8, 465. https://doi.org/10.3390/antiox8100465
Nna VU, Abu Bakar AB, Ahmad A, Eleazu CO, Mohamed M. Oxidative Stress, NF-κB-Mediated Inflammation and Apoptosis in the Testes of Streptozotocin–Induced Diabetic Rats: Combined Protective Effects of Malaysian Propolis and Metformin. Antioxidants. 2019; 8(10):465. https://doi.org/10.3390/antiox8100465
Chicago/Turabian StyleNna, Victor Udo, Ainul Bahiyah Abu Bakar, Azlina Ahmad, Chinedum Ogbonnaya Eleazu, and Mahaneem Mohamed. 2019. "Oxidative Stress, NF-κB-Mediated Inflammation and Apoptosis in the Testes of Streptozotocin–Induced Diabetic Rats: Combined Protective Effects of Malaysian Propolis and Metformin" Antioxidants 8, no. 10: 465. https://doi.org/10.3390/antiox8100465
APA StyleNna, V. U., Abu Bakar, A. B., Ahmad, A., Eleazu, C. O., & Mohamed, M. (2019). Oxidative Stress, NF-κB-Mediated Inflammation and Apoptosis in the Testes of Streptozotocin–Induced Diabetic Rats: Combined Protective Effects of Malaysian Propolis and Metformin. Antioxidants, 8(10), 465. https://doi.org/10.3390/antiox8100465









