Polymeric Nanoparticles for Increasing Oral Bioavailability of Curcumin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Curcumin Quantification
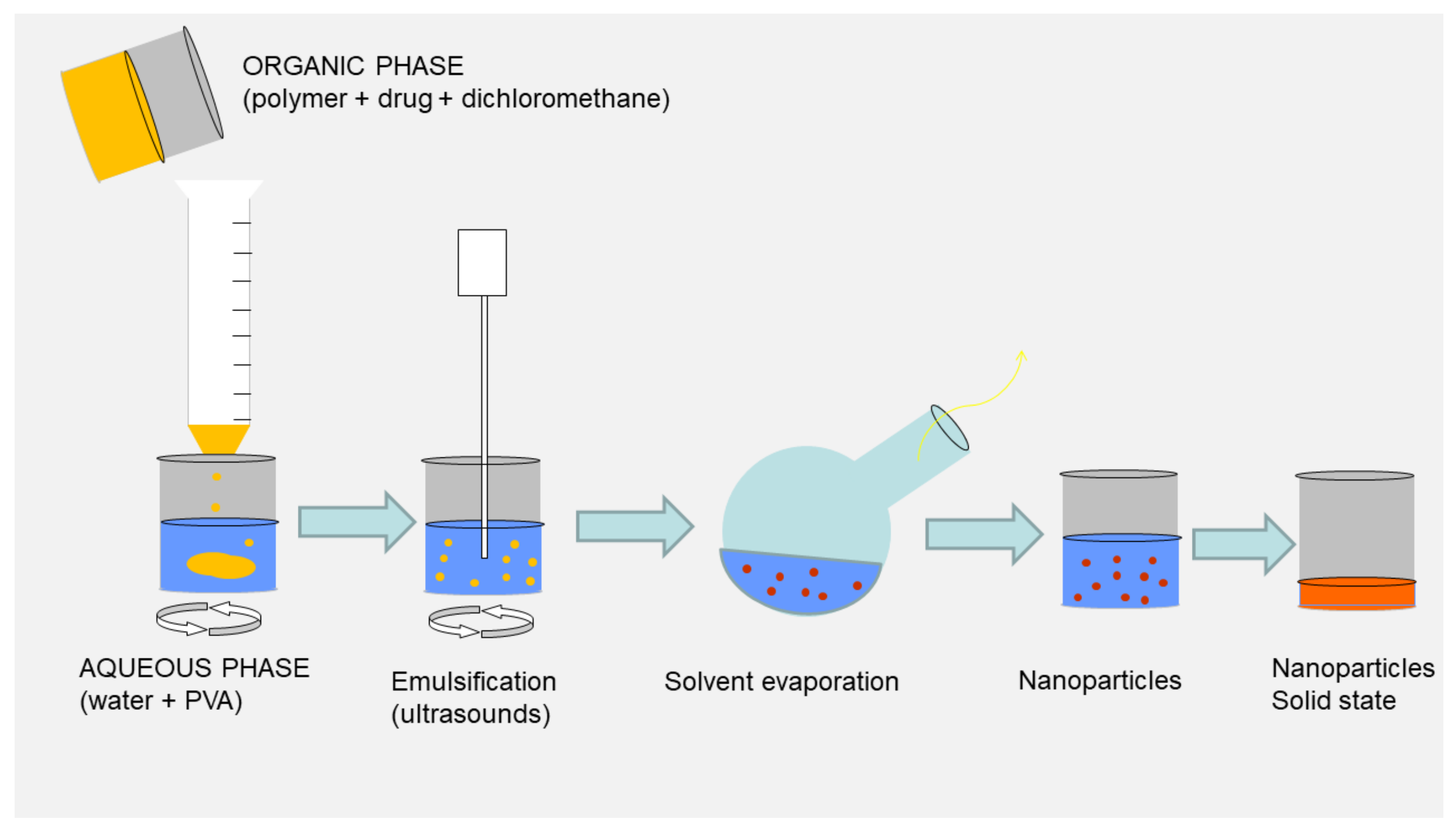
2.3. Preparation of Nanoparticles
2.4. Characterization of Nanoparticles
2.5. In Vitro Release of Curcumin from Nanoparticles
2.6. Intestinal Cells Cytocompatibility
2.7. Statistical Analysis
3. Results and Discussion
3.1. Emulsification Solvent Evaporation Method
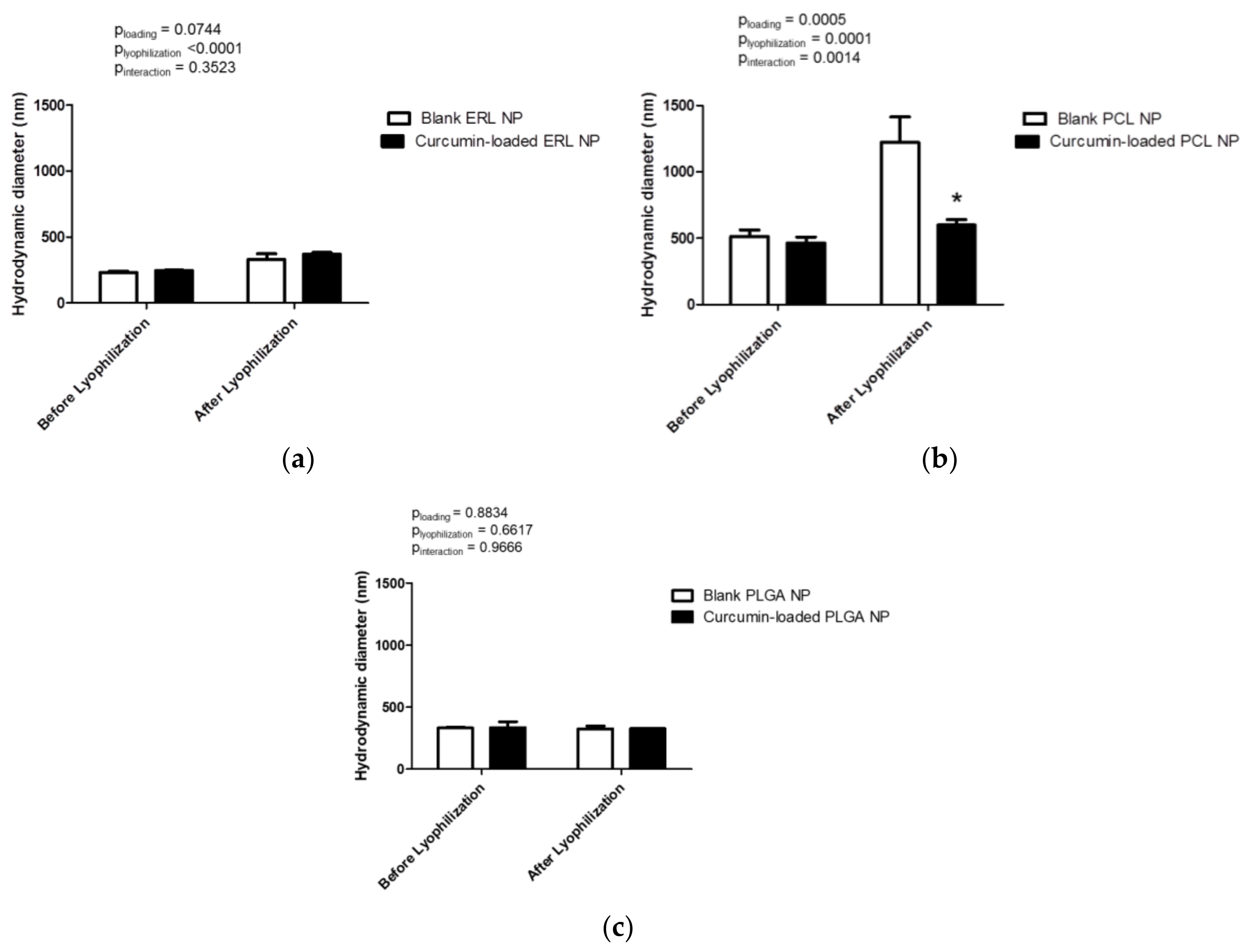
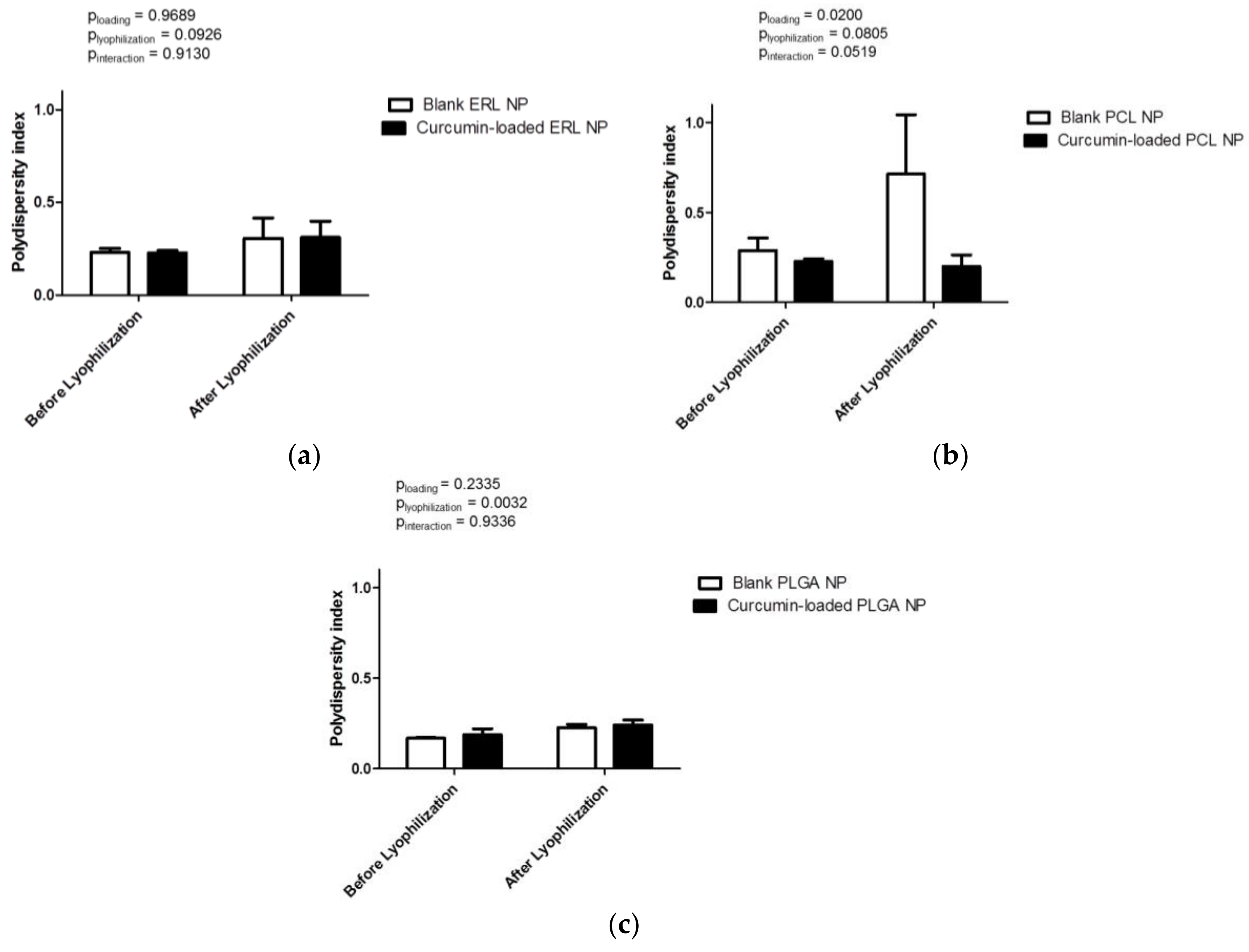
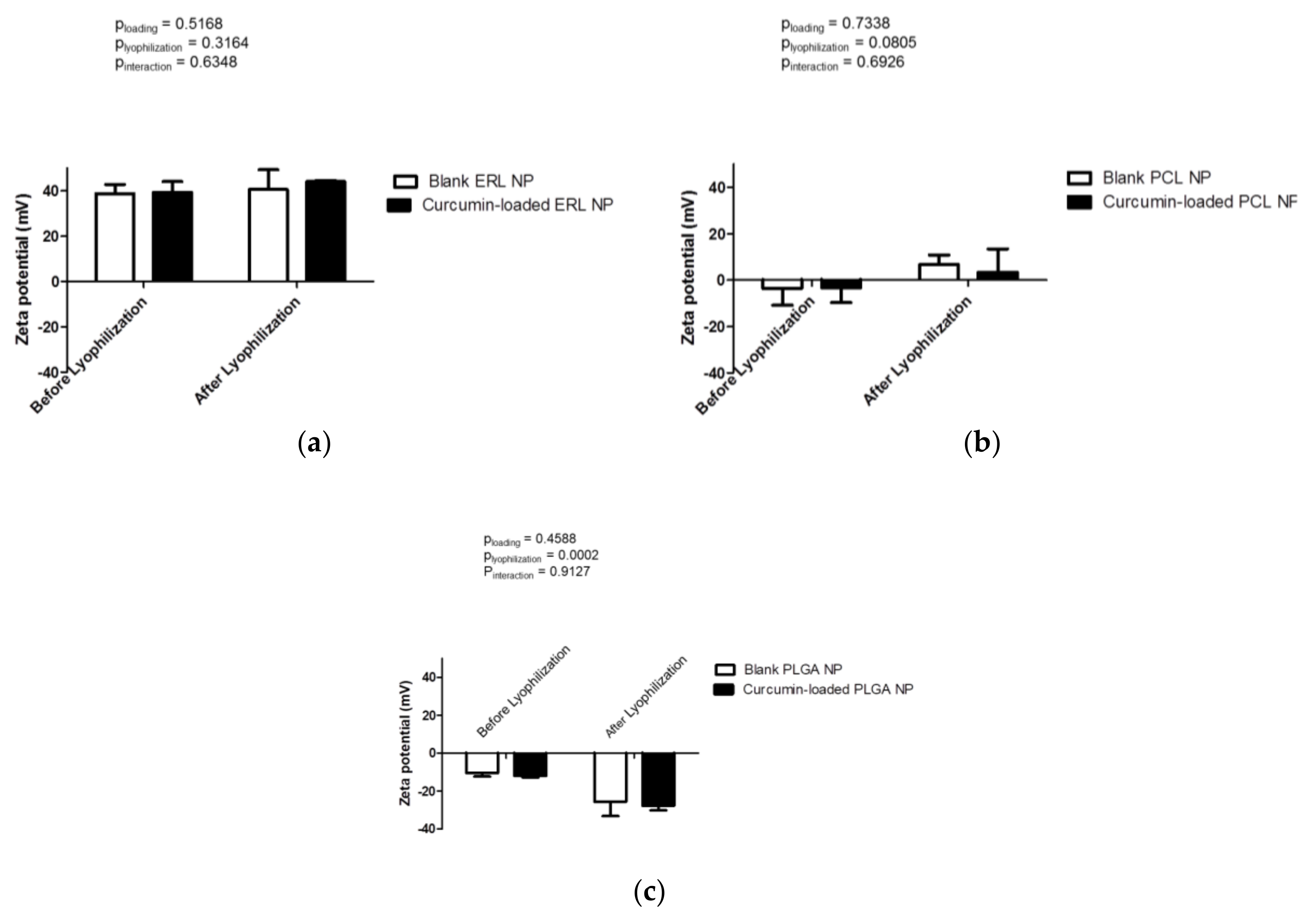
3.2. Formation and Characteristics of Blank and Drug-Loaded Polymeric Nanoparticles
3.3. Nanoparticle Recovery, Curcumin Loading and Encapsulation Efficiency
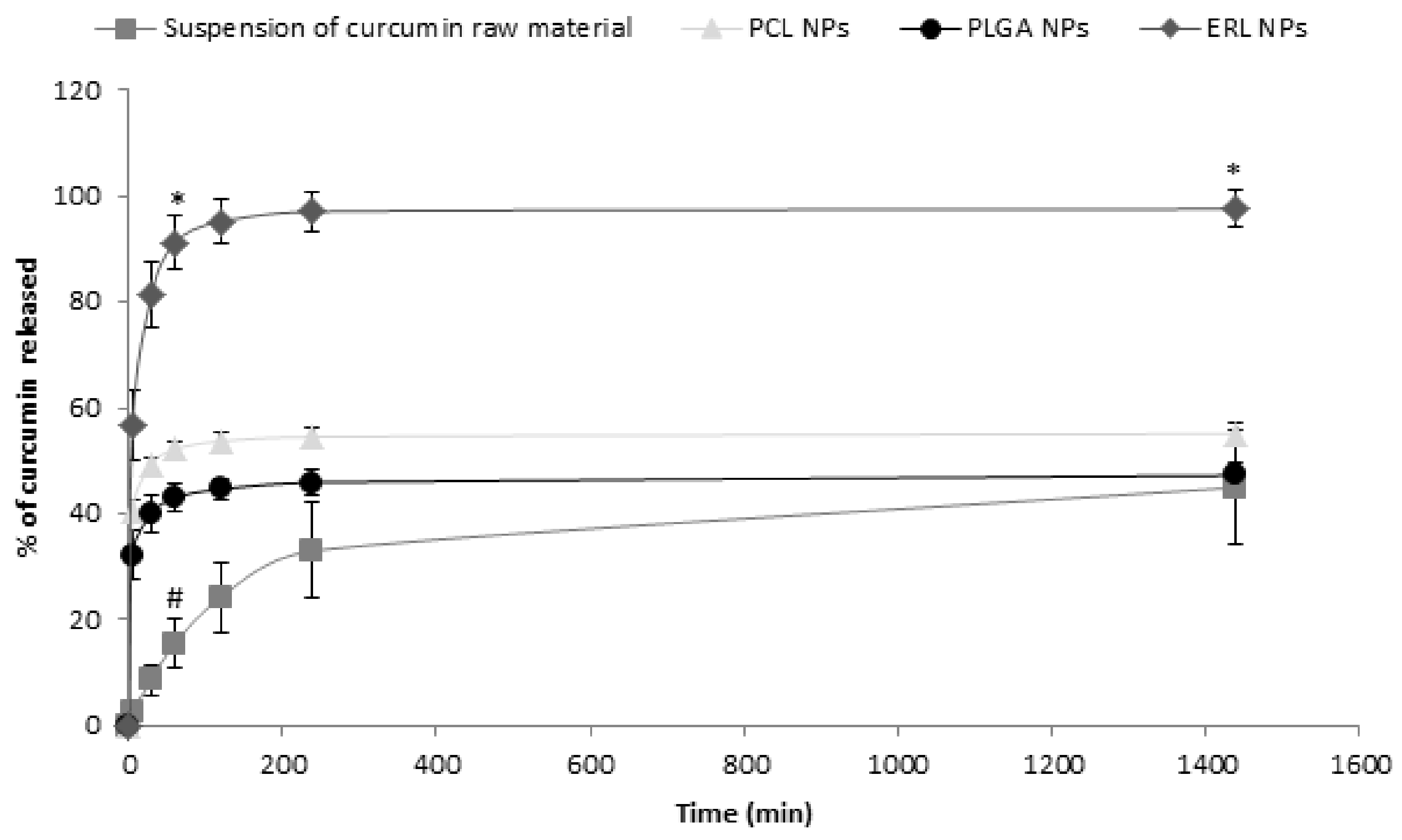
3.4. In Vitro Release Studies
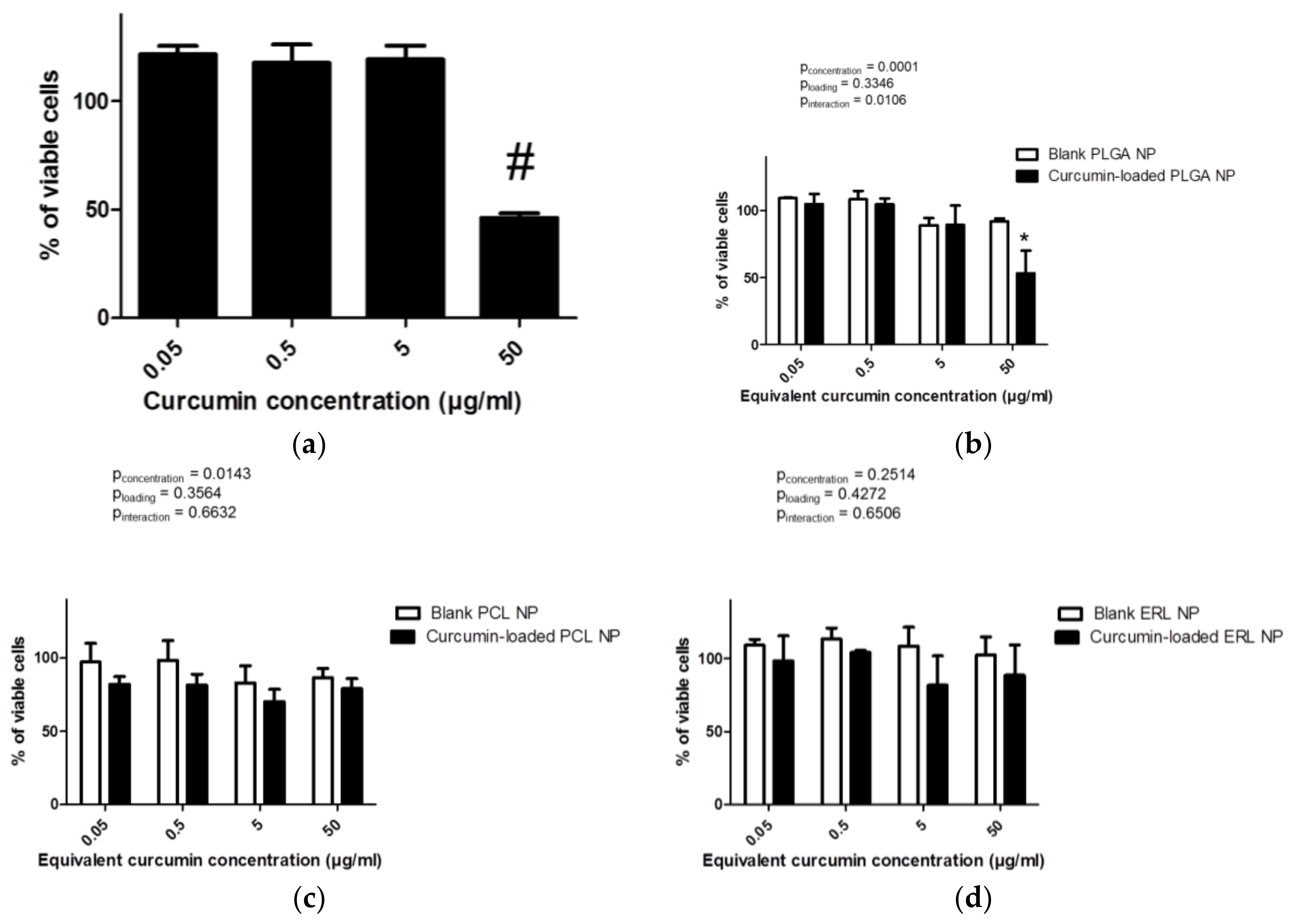
3.5. Cytocompatibility Studies
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Joe, B.; Vijaykumar, M.; Lokesh, B.R. Biological Properties of Curcumin-Cellular and Molecular Mechanisms of Action. Crit. Rev. Food Sci. Nutr. 2004, 44, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Esatbeyoglu, T.; Huebbe, P.; Ernst, I.M.A.; Chin, D.; Wagner, A.E.; Rimbach, G. Curcumin—From Molecule to Biological Function. Angew. Chem. Int. Ed. 2012, 51, 5308–5332. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, K.; Zia, K.M.; Zuber, M.; Salman, M.; Anjum, M.N. Recent development in curcumin and curcumin-based polymeric materials for biomedical applications: A review. Int. J. Biol. Macromol. 2015, 81, 877–890. [Google Scholar] [CrossRef] [PubMed]
- Joe, B.; Lokesh, B.R. Role of capsaicin, curcumin and dietary n-3 fatty acids in lowering the generation of reactive oxygen species in rat peritoneal macrophages. Biochim. Biophys. Acta 1994, 1224, 255–263. [Google Scholar] [CrossRef]
- Cho, J.W.; Lee, K.S.; Kim, C.V. Curcumin attenuates the expression of IL-1beta, IL-6, and TNF-alpha as well as cyclin E in TNF-alpha-treated HaCaT cells; NF-kappaB and MAPKs as potential upstream targets. Int. J. Mol. Med. 2007, 19, 469–474. [Google Scholar] [PubMed]
- Garcia-Nino, W.R.; Pedraza-Chaverri, J. Protective effect of curcumin against heavy metals-induced liver damage. Food Chem. Toxicol. 2014, 69, 182–201. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hang, Y.; Liu, J.; Hou, Y.; Wang, N.; Wang, M. Anticancer effect of curcumin inhibits cell growth through miR-21/PTEN/Akt pathway in breast cancer cell. Oncol. Lett. 2017, 13, 4825–4831. [Google Scholar] [CrossRef] [PubMed]
- Sreedhal, R.; Arumgam, S.; Thandavarayan, R.A.; Karuppagounder, V.; Watanabe, K. Curcumin as a therapeutic agent in the chemoprevention of inflammatory bowel disease. Drug Discov. Today 2016, 21, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Nelson, K.M.; Dahlin, J.L.; Bisson, J.; Graham, J.; Pauli, G.F.; Walters, M. The Essential Medicinal Chemistry of Curcumin. J. Med. Chem. 2017, 60, 1620–1637. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, D.M.; Takeuchi, K.P.; Geraldine, R.M.; Morua, C.J.; Torres, M.C.L. Production, solubility and antioxidant activity of curcumin nanosuspension. Food Sci. Technol. Camp. 2015, 35, 115–119. [Google Scholar] [CrossRef]
- Modasiya, M.K.; Patel, V.M. Studies on solubility of curcumin. Int. J. Pharm. Life Sci. 2012, 3, 1490–1497. [Google Scholar]
- Araiza-Calahorra, A.; Akhtar, M.; Sakar, A. Recent advances in emulsion-based delivery approaches for curcumin: From encapsulation to bioaccesibility. Trends Food Sci. Technol. 2018, 71, 155–169. [Google Scholar] [CrossRef]
- Ariga, K.; Ji, Q.; McShane, M.J.; Lvov, Y.M.; Vinu, A.; Hill, J.P. Inorganic Nanoarchitectonics for Biological Applications. Chem. Mater. 2012, 24, 728–737. [Google Scholar] [CrossRef]
- Komiyama, M.; Yoshimoto, K.; Sisido, M.; Ariga, K. Chemistry Can Make Strict and Fuzzy Controls for Bio-Systems: DNA Nanoarchitectonics and Cell-Macromolecular Nanoarchitectonics. Bull. Chem. Soc. Jpn. 2017, 90, 967–1004. [Google Scholar] [CrossRef]
- Hartig, S.M.; Greene, R.R.; Dikov, M.M.; Prokop, A.; Davidson, J.M. Multifunctional nanoparticulate polyelectrolyte complexes. Pharm. Res. 2007, 24, 2353–2369. [Google Scholar] [CrossRef] [PubMed]
- Soppimath, K.S.; Aminabhavi, T.M.; Kulkarni, A.R.; Rudzinski, W.E. Biodegradable polymeric nanoparticles as drug delivery devices. J. Control. Release 2001, 70, 1–20. [Google Scholar] [CrossRef]
- Ikada, Y.; Tsuji, H. Biodegradable polyesters for medical and ecological applications. Macromol. Rapid Commun. 2000, 21, 117–132. [Google Scholar] [CrossRef]
- McCall, R.L.; Sirianni, R.W. PLGA Nanoparticles Formed by Single- or Double-emulsion with Vitamin ETPGS. J. Vis. Exp. 2013, 82, e51015. [Google Scholar] [CrossRef]
- Des Rieux, A.; Fievez, V.; Garinot, M.; Schneider, Y.-J.; Préat, V. Nanoparticles as potential oral delivery systems of proteins and vaccines: A mechanistic approach. J. Control. Release 2006, 116, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Kasinathan, N.; Amirthalingam, M.; Reddy, N.D.; Jagani, H.V.; Volety, S.M.; Rao, J.V. In-situ implant containing PCL-curcumin nanoparticles developed using design of experiments. Drug Deliv. 2016, 23, 1007–1015. [Google Scholar]
- Akl, M.A.; Kartal-Hodzic, A.; Oksanen, T.; Ismael, H.R.; Afouna, M.M.; Yliperttula, M.; Samy, A.M.; Viitala, T. Factorial design formulation optimization and in vitro characterization of curcumin-loaded PLGA nanoparticles for colon delivery. J. Drug Deliv. Sci. Technol. 2016, 32, 10–20. [Google Scholar] [CrossRef]
- Hoffart, V.; Lamprecht, A.; Maincent, P.; Lecompte, T.; Vigneron, C.; Ubrich, N. Oral bioavailability of a low molecular weight heparin using a polymeric delivery system. J. Control. Release 2006, 113, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Gaucher, C.; Diab, R.; Fries, I.; Xiao, Y.-L.; Hu, X.-M.; Maincent, P.; Sapin-Minet, A. Time lasting S-nitrosoglutathione polymeric nanoparticles delay cellular protein S-nitrosation. Eur. J. Pharm. Biopharm. 2015, 89, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Soltani, S.; Zakeri-Milani, P.; Barzegar-Jalali, M.; Jelvehgari, M. Design of eudragit RL nanoparticles by nanoemulsion method as carriers for ophthalmic drug delivery of ketotifen fumarate. Iran. J. Basic Med. Sci. 2016, 19, 850–860. [Google Scholar]
- Kim, S.R.; Ho, M.J.; Kim, S.H.; Cho, H.R.; Kim, H.S.; Choi, Y.S.; Choi, Y.W.; Kang, M.J. Increased localized delivery of piroxicam by cationic nanoparticles after intra-articular injection. Drug Des. Dev. Ther. 2016, 10, 3779–3787. [Google Scholar] [CrossRef] [PubMed]
- Corrigan, D.O.; Healy, A.M.; Corrigan, O.I. Preparation and release of salbutamol from chitosan and chitosan co-spray dried compacts and multiparticulates. Eur. J. Pharm. Biopharm. 2006, 62, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Umerska, A.; Paluch, K.J.; Santos-Martinez, M.J.; Medina, C.; Corrigan, O.I.; Tajber, L. Chondroitin-based nanoplexes as peptide delivery systems—Investigations into the self-assembly process, solid-state and extended release characteristics. Eur. J. Pharm. Biopharm. 2015, 93, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Quintanar-Guerrero, D.; Fessi, H.; Allémann, E.; Doelker, E. Influence of stabilizing agents and preparative variables on the formation of poly(d,l-lactic acid) nanoparticles by an emulsification-diffusion technique. Int. J. Pharm. 1996, 143, 133–141. [Google Scholar] [CrossRef]
- Padhye, S.; Chavan, D.; Pandey, S.; Deshpande, J.; Swamy, K.W.; Sarkar, F.H. Perspectives on Chemopreventive and Therapeutic Potential of Curcumin Analogs in Medicinal Chemistry. Mini Rev. Med. Chem. 2010, 10, 372–387. [Google Scholar] [CrossRef] [PubMed]
- Priyadarsini, K.I. The Chemistry of Curcumin: From Extraction to Therapeutic Agent. Molecules 2014, 19, 20091–20112. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B.; Deb, L.; Prasad, S. Curcumin Differs from Tetrahydrocurcumin for Molecular Targets, Signaling Pathways and Cellular Responses. Molecules 2015, 20, 185–205. [Google Scholar] [CrossRef] [PubMed]
- Pourreza, N.; Golmohammadi, H. Application of curcumin nanoparticles in a lab-on-paper device as a simple and green pH probe. Talanta 2015, 131, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Pikal, M. Freeze-drying of mannitol-trehalose-sodium chloride-based formulations: The impact of annealing on dry layer resistance to mass transfer and cake structure. Pharm. Dev. Technol. 2004, 9, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Zhi-Gang, F.; Ping, P.; Zhi-Qiang, Y.; Ya-Gen, C.; Jian-Kang, Z.; Miao, W.; Xue-Nong, Z.; Qiang, Z. Two novel freeze-dried pH-sensitive cyclosporine a nanoparticles: Preparation, in vitro drug release, and in vivo absorption enhancement effects. Curr. Nanosci. 2009, 5, 449–456. [Google Scholar] [CrossRef]
- Gandhi, A.; Jana, S.; Sen, K.K. In-vitro release of aciclovir loaded Eudragit RLPO® nanoparticles for sustained drug delivery. Int. J. Biol. Macromol. 2014, 67, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Oh, J.T.; Jang, M.H.; Chung, S.I. Quantitative analysis of polyvinyl alcohol on the surface of poly(d,l-lactide-co-glycolide) microparticles prepared by solvent evaporation method: Effect of particle size and PVA concentration. J. Control. Release 1999, 59, 123–132. [Google Scholar] [CrossRef]
- Boury, F.; Ivanova, T.; Panaiotov, I.; Proust, J.E.; Bois, A.; Richou, J. Dynamical properties of poly(d,l-lactide) and poly-(vinyl-alcohol) monolayers at the air/water and at dichloromethane/water interfaces. J. Colloid Interface Sci. 1995, 169, 380–392. [Google Scholar] [CrossRef]
- Patra, C.N.; Priya, R.; Swain, S.; Jena, G.K.; Panigrahi, K.C.; Ghose, D. Pharmaceutical significance of Eudragit: A review. Future J. Pharm. Sci. 2017, 3, 33–45. [Google Scholar] [CrossRef]
- Nehme, H.; Saulnier, P.; Ramadan, A.A.; Cassisa, V.; Guillet, C.; Eveillard, M.; Umerska, A. Antibacterial activity of antipsychotic agents, their association with lipid nanocapsules and its impact on the properties of the nanocarriers and on antibacterial activity. PLoS ONE 2018, 13, e0189950. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Shi, Y.; Li, J.H.; Gao, N.; Ji, J.; Nui, F.; Chen, Q.; Yang, X.; Wang, S. Enhancement of Oral Bioavailability of Curcumin by a Novel Solid Dispersion System. AAPS PharmSciTech 2015, 16, 1327–1334. [Google Scholar] [CrossRef] [PubMed]
- Khalil, N.M.; do Nascimento, T.C.F.; Casa, D.M.; Dalmolin, L.F.; de Mattos, A.C.; Hoss, I.; Romano, M.A.; Mainardes, R.M. Pharmacokinetics of curcumin-loaded PLGA and PLGA-PEG blend nanoparticles after oral administration in rats. Colloids Surf. B Biointerfaces 2013, 101, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Wahlang, B.; Pawar, Y.B.; Bansal, A.K. Identification of permeability-related hurdles in oral delivery of curcumin using the Caco-2 cell model. Eur. J. Pharm. Biopharm. 2011, 77, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Shen, Z.L.; Zhai, S.; Xu, J.L.; Liang, H.; Shen, Q.; Li, Q.Y. Transport of curcumin derivatives in Caco-2 cell monolayers. Eur. J. Pharm. Biopharm. 2017, 117, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Umerska, A.; Corrigan, O.I.; Tajber, L. Design of chondroitin sulfate-based polyelectrolyte nanoplexes: Formation of nanocarriers with chitosan and a case study of salmon calcitonin. Carbohydr. Polym. 2017, 156, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Umerska, A.; Paluch, K.J.; Inkielewicz-Stepniak, I.; Santos-Martinez, M.J.; Corrigan, O.I.; Medina, C.; Tajber, L. Exploring the assembly process and properties of novel crossliner-free hyaluronate-based polyelectrolyte complex nanocarriers. Int. J. Pharm. 2012, 436, 75–87. [Google Scholar] [CrossRef] [PubMed]

| Polymer | ERL | PLGA | PCL |
|---|---|---|---|
| Formula | 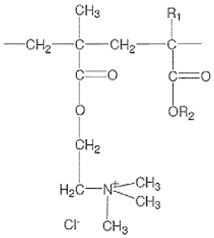 | 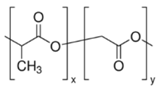 |  |
| Average molecular weight | Mw = 150,000 g/mol | Mw = 40,000 g/mol | Mn = 10,000 g/mol Mw = 14,000 g/mol |
| Viscosity | Max 15 mpa·s a | 0.22 dL/g b | 758 mPa·s c |
| Acid value | N/A | 9 mg KOH/g | 0.2 mg KOH/g |
| Alkali value | 23.9–32.3 mg KOH/g | N/A | 12.6 mg KOH/g |
| Ammonio methacrylate units on DS | 8.85–11.96% | N/R | N/R |
| % of d,l-lactide and glycolide units | N/R | 50 mol % of d,l-lactide and 50 mol % glycolide d | N/R |
| Formulation | NP Recovery | Encapsulation Efficiency | Drug Loading |
|---|---|---|---|
| Blank PLGA NPs | 66 ± 9% | N/A | N/A |
| Blank PCL NPs | 67 ± 5% | N/A | N/A |
| Blank ERL NPs | 34 ± 4% *,# | N/A | N/A |
| Curcumin-loaded PLGA NPs | 66 ± 16% | 90.0 ± 1.5% | 81.8 ± 1.4 mg/g |
| Curcumin-loaded PCL NPs | 64 ± 2% | 99.2 ± 0.2% | 90.2 ± 0.2 mg/g |
| Curcumin-loaded ERL NPs | 35 ± 6% *,# | 61.7 ± 4.7% *,# | 56.1 ± 4.3 mg/g *,# |
| Sample | K (h−1) | W∞ (µg/mg) | Goodness of Fit (R2) |
|---|---|---|---|
| Suspension of curcumin raw material | 0.0060 ± 0.0004 | 44.6 ± 10.8 | 0.8960 |
| PCL NPs | 0.288 ± 0.022 *,# | 47.8 ± 1.5 | 0.9854 |
| PLGA NPs | 0.264 ± 0.052 * | 36.1 ± 1.9 # | 0.9607 |
| ERL NPs | 0.185 ± 0.041 * | 52.0 ± 2.3 | 0.9646 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umerska, A.; Gaucher, C.; Oyarzun-Ampuero, F.; Fries-Raeth, I.; Colin, F.; Villamizar-Sarmiento, M.G.; Maincent, P.; Sapin-Minet, A. Polymeric Nanoparticles for Increasing Oral Bioavailability of Curcumin. Antioxidants 2018, 7, 46. https://doi.org/10.3390/antiox7040046
Umerska A, Gaucher C, Oyarzun-Ampuero F, Fries-Raeth I, Colin F, Villamizar-Sarmiento MG, Maincent P, Sapin-Minet A. Polymeric Nanoparticles for Increasing Oral Bioavailability of Curcumin. Antioxidants. 2018; 7(4):46. https://doi.org/10.3390/antiox7040046
Chicago/Turabian StyleUmerska, Anita, Caroline Gaucher, Felipe Oyarzun-Ampuero, Isabelle Fries-Raeth, Florence Colin, María Gabriela Villamizar-Sarmiento, Philippe Maincent, and Anne Sapin-Minet. 2018. "Polymeric Nanoparticles for Increasing Oral Bioavailability of Curcumin" Antioxidants 7, no. 4: 46. https://doi.org/10.3390/antiox7040046
APA StyleUmerska, A., Gaucher, C., Oyarzun-Ampuero, F., Fries-Raeth, I., Colin, F., Villamizar-Sarmiento, M. G., Maincent, P., & Sapin-Minet, A. (2018). Polymeric Nanoparticles for Increasing Oral Bioavailability of Curcumin. Antioxidants, 7(4), 46. https://doi.org/10.3390/antiox7040046







