Structural/Functional Matches and Divergences of Phytoprostanes and Phytofurans with Bioactive Human Oxylipins
Abstract
1. Introduction
2. Occurrence of Phytoprostanes and Phytofurans in Biological Systems. Physiological Significance
2.1. Phytoprostanes and Phytofurans in Vegetable Oils
2.2. Phytoprostanes and Phytofurans in Nuts and Seeds
2.3. Phytoprostanes and Phytofurans in Rice
2.4. Phytoprostanes and Phytofurans in Musts and Wines
2.5. Phytoprostanes and Phytofurans in Macroalgae
2.6. Phytoprostanes and Phytofurans in Foodstuffs
3. What do We Know on the Bioavailability of Phytoprostanes and Phytofurans?
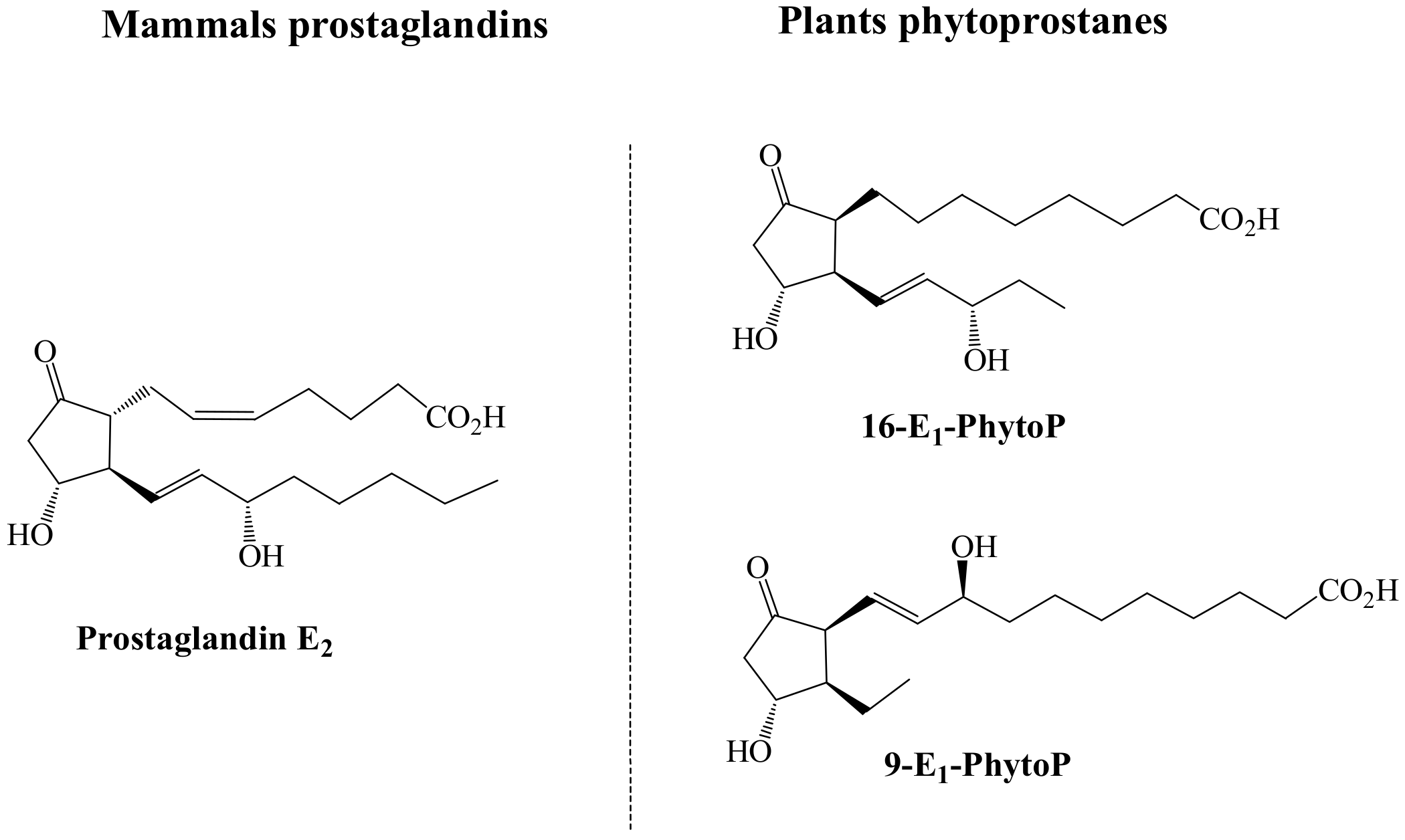
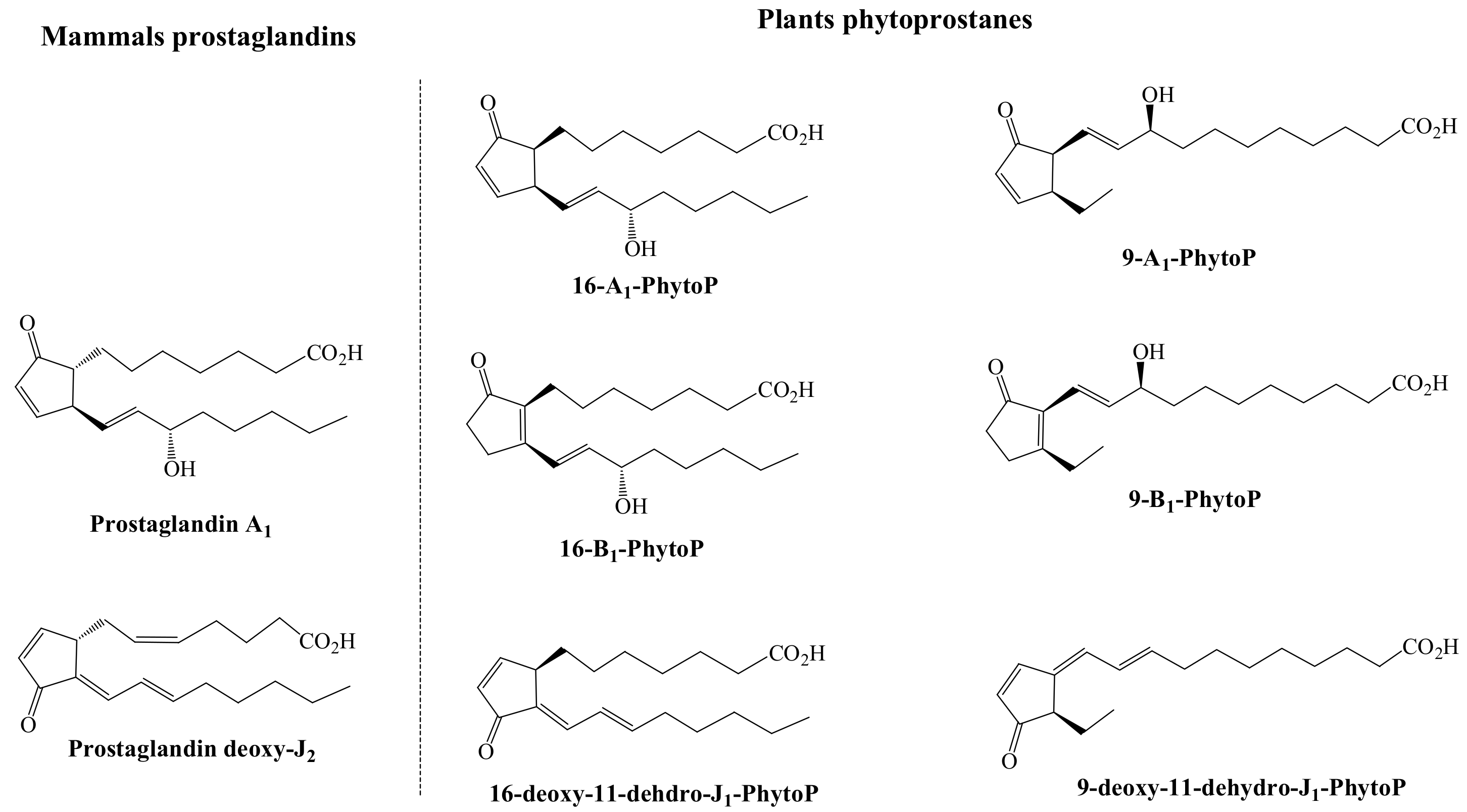
4. Comparison between Plant and Human Bioactive Oxylipins: Tentative Insights in the Structure-Activity Relationship
4.1. Biological Functions of Phytoprostanes in Higher Plants
4.2. Phytoprostanes as Immunomodulatory Molecules in Humans
4.3. Anti-Inflammatory Capacity of Phytoprostanes in Humans
4.4. Phytoprostanes vs. Mammals Oxylipins Regarding Antitumor Activity
4.5. Key Structural Features and Mechanism of Action of Oxylipins for Biological Functions
5. Conclusions and Future Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Jacinta, C.-G.; Thierry, D.; Federico, F.; Sonia, M.; Arturo, T.; Ángel, G.-I. Phytoprostanes. Lipid Technol. 2015, 27, 127–130. [Google Scholar]
- Parchmann, S.; Mueller, M.J. Evidence for the Formation of Dinor Isoprostanes E1from α-Linolenic Acid in Plants. J. Biol. Chem. 1998, 273, 32650–32655. [Google Scholar] [CrossRef] [PubMed]
- Cuyamendous, C.; Leung, K.S.; Durand, T.; Lee, J.C.; Oger, C.; Galano, J.M. Synthesis and discovery of phytofurans: Metabolites of alpha-linolenic acid peroxidation. Chem. Commun. 2015, 51, 15696–15699. [Google Scholar] [CrossRef] [PubMed]
- Cuyamendous, C.; Leung, K.S.; Bultel-Poncé, V.; Guy, A.; Durand, T.; Galano, J.M.; Lee, J.C.Y.; Oger, C. Total synthesis and in vivo quantitation of phytofurans derived from α-linolenic acid. Eur. J. Org. Chem. 2017, 2017, 2486–2490. [Google Scholar] [CrossRef]
- Borrego, E.J.; Kolomiets, M.V. Synthesis and Functions of Jasmonates in Maize. Plants 2016, 5, 41. [Google Scholar] [CrossRef] [PubMed]
- Imbusch, R.; Mueller, M.J. Analysis of oxidative stress and wound-inducible dinor isoprostanes F1 (phytoprostanes F1) in plants. Plant Physiol. 2000, 124, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Imbusch, R.; Mueller, M.J. Formation of isoprostane F2-like compounds (phytoprostanes F1) from α-linolenic acid in plants. Free Radic. Biol. Med. 2000, 28, 720–726. [Google Scholar] [CrossRef]
- Galano, J.M.; Lee, Y.Y.; Oger, C.; Vigor, C.; Vercauteren, J.; Durand, T.; Giera, M.; Lee, J.C. Isoprostanes, neuroprostanes and phytoprostanes: An overview of 25 years of research in chemistry and biology. Prog. Lipid Res. 2017, 68, 83–108. [Google Scholar] [CrossRef] [PubMed]
- Minghetti, L.; Salvi, R.; Lavinia Salvatori, M.; Ajmone-Cat, M.A.; De Nuccio, C.; Visentin, S.; Bultel-Ponce, V.; Oger, C.; Guy, A.; Galano, J.M.; et al. Nonenzymatic oxygenated metabolites of alpha-linolenic acid B1- and L1-phytoprostanes protect immature neurons from oxidant injury and promote differentiation of oligodendrocyte progenitors through PPAR-gamma activation. Free Radic. Biol. Med. 2014, 73, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Marhuenda, J.; Medina, S.; Díaz-Castro, A.; Martínez-Hernández, P.; Arina, S.; Zafrilla, P.; Mulero, J.; Oger, C.; Galano, J.-M.; Durand, T.; et al. Dependency of Phytoprostane Fingerprints of Must and Wine on Viticulture and Enological Processes. J. Agric. Food Chem. 2015, 63, 9022–9028. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, A.J.; Attar-Bashi, N.M.; Li, D. What is the role of alpha-linolenic acid for mammals? Lipids 2002, 37, 1113–1123. [Google Scholar] [CrossRef] [PubMed]
- Karg, K.; Karg, K.; Dirsch, V.M.; Karg, K.; Dirsch, V.M.; Vollmar, A.M.; Cracowski, J.-L.; Laporte, F.; Mueller, M.J. Biologically active oxidized lipids (phytoprostanes) in the plant diet and parenteral lipid nutrition. Free Radic. Res. 2007, 41, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Barden, A.E.; Croft, K.D.; Durand, T.; Guy, A.; Mueller, M.J.; Mori, T.A. Flaxseed oil supplementation increases plasma F1-phytoprostanes in healthy men. J. Nutr. 2009, 139, 1890–1895. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.; Schutz, A.L.; Galano, J.M.; Herrfurth, C.; Feussner, K.; Durand, T.; Brodhun, F.; Feussner, I. The Alphabet of Galactolipids in Arabidopsis thaliana. Front. Plant Sci. 2011, 2, 95. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Perles, R.; Abellan, A.; Leon, D.; Ferreres, F.; Guy, A.; Oger, C.; Galano, J.M.; Durand, T.; Gil-Izquierdo, A. Sorting out the phytoprostane and phytofuran profile in vegetable oils. Food Res. Int. 2018, 107, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Collado-González, J.; Medina, S.; Durand, T.; Guy, A.; Galano, J.-M.; Torrecillas, A.; Ferreres, F.; Gil-Izquierdo, A. New UHPLC–QqQ-MS/MS method for quantitative and qualitative determination of free phytoprostanes in foodstuffs of commercial olive and sunflower oils. Food Chem. 2015, 178, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Collado-González, J.; Pérez-López, D.; Memmi, H.; Gijón, M.C.; Medina, S.; Durand, T.; Guy, A.; Galano, J.M.; Fernández, D.J.; Carro, F. Effect of the season on the free phytoprostane content in Cornicabra extra virgin olive oil from deficit-irrigated olive trees. J. Sci. Food Agric. 2016, 96, 1585–1592. [Google Scholar] [CrossRef] [PubMed]
- Collado-González, J.; Moriana, A.; Girón, I.F.; Corell, M.; Medina, S.; Durand, T.; Guy, A.; Galano, J.-M.; Valero, E.; Garrigues, T.; et al. The phytoprostane content in green table olives is influenced by Spanish-style processing and regulated deficit irrigation. LWT-Food Sci. Technol. 2015, 64, 997–1003. [Google Scholar] [CrossRef]
- Barbosa, M.; Collado-Gonzalez, J.; Andrade, P.B.; Ferreres, F.; Valentao, P.; Galano, J.M.; Durand, T.; Gil-Izquierdo, A. Nonenzymatic alpha-Linolenic Acid Derivatives from the Sea: Macroalgae as Novel Sources of Phytoprostanes. J. Agric. Food Chem. 2015, 63, 6466–6474. [Google Scholar] [CrossRef] [PubMed]
- Vigor, C.; Reversat, G.; Rocher, A.; Oger, C.; Galano, J.-M.; Vercauteren, J.; Durand, T.; Tonon, T.; Leblanc, C.; Potin, P. Isoprostanoids quantitative profiling of marine red and brown macroalgae. Food Chem. 2018, 268, 452–462. [Google Scholar] [CrossRef] [PubMed]
- Medina, S.; Collado-González, J.; Ferreres, F.; Londoño-Londoño, J.; Jiménez-Cartagena, C.; Guy, A.; Durand, T.; Galano, J.-M.; Gil-Izquierdo, A. Quantification of phytoprostanes–bioactive oxylipins–and phenolic compounds of Passiflora edulis Sims shell using UHPLC-QqQ-MS/MS and LC-IT-DAD-MS/MS. Food Chem. 2017, 229, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Medina, S.; Collado, J.; Ferreres, F.; Londoño, J.; Jiménez-Cartagena, C.; Guy, A.; Durand, T.; Galano, J.-M.; Gil-Izquierdo, A. Valorization strategy of banana passion fruit shell wastes: An innovative source of phytoprostanes and phenolic compounds and their potential use in pharmaceutical and cosmetic industries. J. Food Nutr. Res. 2017, 5, 801–808. [Google Scholar] [CrossRef]
- Medina, S.; Collado-González, J.; Ferreres, F.; Londoño-Londoño, J.; Jiménez-Cartagena, C.; Guy, A.; Durand, T.; Galano, J.M.; Gil-Izquierdo, A. Potential of Physalis peruviana calyces as a low-cost valuable resource of phytoprostanes and phenolic compounds. J. Sci. Food Agric. 2018. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Del Amor, A.M.; Aguayo, E.; Collado-González, J.; Guy, A.; Galano, J.-M.; Durand, T.; Gil-Izquierdo, Á. Impact of packaging atmosphere, storage and processing conditions on the generation of phytoprostanes as quality processing compounds in almond kernels. Food Chem. 2016, 211, 869–875. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Del Amor, A.; Collado-Gonzalez, J.; Aguayo, E.; Guy, A.; Galano, J.; Durand, T.; Gil-Izquierdo, A. Phytoprostanes in almonds: Identification, quantification, and impact of cultivar and type of cultivation. RSC Adv. 2015, 5, 51233–51241. [Google Scholar] [CrossRef]
- Carrasco-Del Amor, A.M.; Aguayo, E.; Collado-González, J.; Guy, A.; Galano, J.-M.; Durand, T.; Gil-Izquierdo, Á. Impact of processing conditions on the phytoprostanes profile of three types of nut kernels. Free Radic. Res. 2017, 51, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Pinciroli, M.; Domínguez-Perles, R.; Abellán, A.; Guy, A.; Durand, T.; Oger, C.; Galano, J.M.; Ferreres, F.; Gil-Izquierdo, A. Comparative Study of the Phytoprostane and Phytofuran Content of indica and japonica Rice (Oryza sativa L.) Flours. J. Agric. Food Chem. 2017, 65, 8938–8947. [Google Scholar] [CrossRef] [PubMed]
- Thoma, I.; Loeffler, C.; Sinha, A.K.; Gupta, M.; Krischke, M.; Steffan, B.; Roitsch, T.; Mueller, M.J. Cyclopentenone isoprostanes induced by reactive oxygen species trigger defense gene activation and phytoalexin accumulation in plants. Plant J. Cell Mol. Biol. 2003, 34, 363–375. [Google Scholar] [CrossRef]
- Yonny, M.E.; Rodríguez Torresi, A.; Cuyamendous, C.; Réversat, G.; Oger, C.; Galano, J.-M.; Durand, T.; Vigor, C.; Nazareno, M.A. Thermal Stress in Melon Plants: Phytoprostanes and Phytofurans as Oxidative Stress Biomarkers and the Effect of Antioxidant Supplementation. J. Agric. Food Chem. 2016, 64, 8296–8304. [Google Scholar] [CrossRef] [PubMed]
- Thoma, I.; Krischke, M.; Loeffler, C.; Mueller, M.J. The isoprostanoid pathway in plants. Chem. Phys. Lipids 2004, 128, 135–148. [Google Scholar] [CrossRef] [PubMed]
- Loeffler, C.; Berger, S.; Guy, A.; Durand, T.; Bringmann, G.; Dreyer, M.; von Rad, U.; Durner, J.; Mueller, M.J. B(1)-Phytoprostanes Trigger Plant Defense and Detoxification Responses. Plant Physiol. 2005, 137, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Romero, A.; Verdaguer, X.; Riera, A. General approach to prostanes b1 by intermolecular pauson–khand reaction: Syntheses of methyl esters of prostaglandin b1 and phytoprostanes 16-b1-phytop and 9-l1-phytop. Eur. J. Org. Chem. 2009, 2013, 1716–1725. [Google Scholar] [CrossRef]
- Gutermuth, J.; Bewersdorff, M.; Traidl-Hoffmann, C.; Ring, J.; Mueller, M.J.; Behrendt, H.; Jakob, T. Immunomodulatory effects of aqueous birch pollen extracts and phytoprostanes on primary immune responses in vivo. J. Allergy Clin. Immunol. 2007, 120, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Collado-Gonzalez, J.; Perez-Lopez, D.; Memmi, H.; Gijon, M.C.; Medina, S.; Durand, T.; Guy, A.; Galano, J.M.; Ferreres, F.; Torrecillas, A.; et al. Water deficit during pit hardening enhances phytoprostanes content, a plant biomarker of oxidative stress, in extra virgin olive oil. J. Agric. Food Chem. 2015, 63, 3784–3792. [Google Scholar] [CrossRef] [PubMed]
- Farmer, E.E.; Almeras, E.; Krishnamurthy, V. Jasmonates and related oxylipins in plant responses to pathogenesis and herbivory. Curr. Opin. Plant Biol. 2003, 6, 372–378. [Google Scholar] [CrossRef]
- Vigor, C.; Bertrand-Michel, J.; Pinot, E.; Oger, C.; Vercauteren, J.; Le Faouder, P.; Galano, J.-M.; Lee, J.C.-Y.; Durand, T. Non-enzymatic lipid oxidation products in biological systems: Assessment of the metabolites from polyunsaturated fatty acids. J. Chromatogr. B 2014, 964, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Cuyamendous, C.; de la Torre, A.; Lee, Y.Y.; Leung, K.S.; Guy, A.; Bultel-Ponce, V.; Galano, J.M.; Lee, J.C.; Oger, C.; Durand, T. The novelty of phytofurans, isofurans, dihomo-isofurans and neurofurans: Discovery, synthesis and potential application. Biochimie 2016, 130, 49–62. [Google Scholar] [CrossRef] [PubMed]
- El Fangour, S.; Guy, A.; Despres, V.; Vidal, J.P.; Rossi, J.C.; Durand, T. Total synthesis of the eight diastereomers of the syn-anti-syn phytoprostanes F1 types I and II. J. Org. Chem. 2004, 69, 2498–2503. [Google Scholar] [CrossRef] [PubMed]
- El Fangour, S.; Guy, A.; Vidal, J.P.; Rossi, J.C.; Durand, T. A flexible synthesis of the phytoprostanes B1 type I and II. J. Org. Chem. 2005, 70, 989–997. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.; Evans, P.; Lledo, A.; Verdaguer, X.; Pericas, M.A.; Riera, A.; Loeffler, C.; Sinha, A.K.; Mueller, M.J. Total synthesis and biological activity of 13,14-dehydro-12-oxo-phytodienoic acids (deoxy-J1-phytoprostanes). ChemBioChem 2005, 6, 276–280. [Google Scholar] [CrossRef] [PubMed]
- Milne, G.L.; Yin, H.; Morrow, J.D. Human biochemistry of the isoprostane pathway. J. Biol. Chem. 2008, 283, 15533–15537. [Google Scholar] [CrossRef] [PubMed]
- Lands, B. Highly unsaturated fatty acids (HUFA) mediate and monitor food’s impact on health. Prostaglandins Other Lipid Mediat. 2017, 133, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Montuschi, P.; Barnes, P.J.; Roberts, L.J., II. Isoprostanes: Markers and mediators of oxidative stress. FASEB J. 2004, 18, 1791–1800. [Google Scholar] [CrossRef] [PubMed]
- Gilles, S.; Mariani, V.; Bryce, M.; Mueller, M.J.; Ring, J.; Jakob, T.; Pastore, S.; Behrendt, H.; Traidl-Hoffmann, C. Pollen-Derived E1-Phytoprostanes Signal via PPAR-γ and NF-κB-Dependent Mechanisms. J. Immunol. 2009, 182, 6653–6658. [Google Scholar] [CrossRef] [PubMed]
- Traidl-Hoffmann, C.; Mariani, V.; Hochrein, H.; Karg, K.; Wagner, H.; Ring, J.; Mueller, M.J.; Jakob, T.; Behrendt, H. Pollen-associated phytoprostanes inhibit dendritic cell interleukin-12 production and augment T helper type 2 cell polarization. J. Exp. Med. 2005, 201, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Straus, D.S.; Glass, C.K. Cyclopentenone prostaglandins: New insights on biological activities and cellular targets. Med. Res. Rev. 2001, 21, 185–210. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, F.A.; Wynalda, M.A. Albumin-lipid interactions: Prostaglandin stability as a probe for characterizing binding sites on vertebrate albumins. Biochemistry 1981, 20, 6129–6134. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.; Hilbert, B.; Dueckershoff, K.; Roitsch, T.; Krischke, M.; Mueller, M.J.; Berger, S. General detoxification and stress responses are mediated by oxidized lipids through TGA transcription factors in Arabidopsis. Plant Cell 2008, 20, 768–785. [Google Scholar] [CrossRef] [PubMed]
- Mariani, V.; Gilles, S.; Jakob, T.; Thiel, M.; Mueller, M.J.; Ring, J.; Behrendt, H.; Traidl-Hoffmann, C. Immunomodulatory mediators from pollen enhance the migratory capacity of dendritic cells and license them for Th2 attraction. J. Immunol. 2007, 178, 7623–7631. [Google Scholar] [CrossRef] [PubMed]
- Musiek, E.S.; Gao, L.; Milne, G.L.; Han, W.; Everhart, M.B.; Wang, D.; Backlund, M.G.; DuBois, R.N.; Zanoni, G.; Vidari, G.; et al. Cyclopentenone isoprostanes inhibit the inflammatory response in macrophages. J. Biol. Chem. 2005, 280, 35562–35570. [Google Scholar] [CrossRef] [PubMed]
- Kondo, M.; Shibata, T.; Kumagai, T.; Osawa, T.; Shibata, N.; Kobayashi, M.; Sasaki, S.; Iwata, M.; Noguchi, N.; Uchida, K. 15-Deoxy-Δ(12,14)-prostaglandin J(2): The endogenous electrophile that induces neuronal apoptosis. Proc. Natl. Acad. Sci. USA 2002, 99, 7367–7372. [Google Scholar] [CrossRef] [PubMed]
- Zernecke, A.; Erl, W.; Fraemohs, L.; Lietz, M.; Weber, C. Suppression of endothelial adhesion molecule up-regulation with cyclopentenone prostaglandins is dissociated from IkappaB-alpha kinase inhibition and cell death induction. FASEB J. 2003, 17, 1099–1101. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Pajares, J.; Oger, C.; Galano, J.M.; Durand, T.; Chevalier, S.; Frank, P. 627-Oxidized products of α-linolenic acid negatively affect cell survival and motility of breast cancer cells. Eur. J. Cancer 2016, 61, S140. [Google Scholar] [CrossRef]
- Iqbal, M.; Evans, P. Conjugate addition–Peterson olefination reactions: Expedient routes to cross conjugated dienones. Tetrahedron Lett. 2003, 44, 5741–5745. [Google Scholar] [CrossRef]
- Forman, B.M.; Tontonoz, P.; Chen, J.; Brun, R.P.; Spiegelman, B.M.; Evans, R.M. 15-Deoxy-delta 12, 14-prostaglandin J2 is a ligand for the adipocyte determination factor PPAR gamma. Cell 1995, 83, 803–812. [Google Scholar] [CrossRef]
- Kliewer, S.A.; Lenhard, J.M.; Willson, T.M.; Patel, I.; Morris, D.C.; Lehmann, J.M. A prostaglandin J2 metabolite binds peroxisome proliferator-activated receptor gamma and promotes adipocyte differentiation. Cell 1995, 83, 813–819. [Google Scholar] [CrossRef]
- Narumiya, S.; Sugimoto, Y.; Ushikubi, F. Prostanoid receptors: Structures, properties, and functions. Physiol. Rev. 1999, 79, 113–1226. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Ting, A.T.; Seed, B. PPAR-gamma agonists inhibit production of monocyte inflammatory cytokines. Nature 1998, 391, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Ricote, M.; Li, A.C.; Willson, T.M.; Kelly, C.J.; Glass, C.K. The peroxisome proliferator-activated receptor-gamma is a negative regulator of macrophage activation. Nature 1998, 391, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and inflammation. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 986–1000. [Google Scholar] [CrossRef] [PubMed]
- Noyori, R.; Suzuki, M. Organic synthesis of prostaglandins: Advancing biology. Science 1993, 259, 44–45. [Google Scholar] [CrossRef] [PubMed]
- Atsmon, J.; Sweetman, B.J.; Baertschi, S.W.; Harris, T.M.; Roberts, L.J. Formation of thiol conjugates of 9-deoxy-D9 D12(E)-prostaglandin D2 and D12(E)-prostaglandin D2. Biochemistry 1990, 29, 3760–3765. [Google Scholar] [CrossRef] [PubMed]
- Van Iersel, M.L.; Cnubben, N.H.; Smink, N.; Koeman, J.H.; van Bladeren, P.J. Interactions of prostaglandin A2 with the glutathione-mediated biotransformation system. Biochem. Pharmacol. 1999, 57, 1383–1390. [Google Scholar] [CrossRef]
- Fukushima, M. Prostaglandin J2-anti-tumour and anti-viral activities and the mechanisms involved. Eicosanoids 1990, 3, 189–199. [Google Scholar] [PubMed]
- Bui, T.; Straus, D.S. Effects of cyclopentenone prostaglandins and related compounds on insulin-like growth factor-I and Waf1 gene expression. Biochim. Biophys. Acta 1998, 1397, 31–42. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, J.H.; Kim, E.H.; Na, H.K.; Cha, Y.N.; Chung, J.H.; Surh, Y.J. 15-Deoxy-Delta12,14-prostaglandin J2 upregulates the expression of heme oxygenase-1 and subsequently matrix metalloproteinase-1 in human breast cancer cells: Possible roles of iron and ROS. Carcinogenesis 2009, 30, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Elia, G.; Santoro, M.G. 2-Cyclopenten-1-one, a new inducer of heat shock protein 70 with antiviral activity. J. Biol. Chem. 1996, 271, 32192–32196. [Google Scholar] [CrossRef] [PubMed]
- Straus, D.S.; Pascual, G.; Li, M.; Welch, J.S.; Ricote, M.; Hsiang, C.-H.; Sengchanthalangsy, L.L.; Ghosh, G.; Glass, C.K. 15-Deoxy-Δ(12,14)-prostaglandin J(2) inhibits multiple steps in the NF-κB signaling pathway. Proc. Natl. Acad. Sci. USA 2000, 97, 4844–4849. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Kapahi, P.; Natoli, G.; Takahashi, T.; Chen, Y.; Karin, M.; Santoro, M.G. Anti-inflammatory cyclopentenone prostaglandins are direct inhibitors of IkappaB kinase. Nature 2000, 403, 103–108. [Google Scholar] [CrossRef] [PubMed]
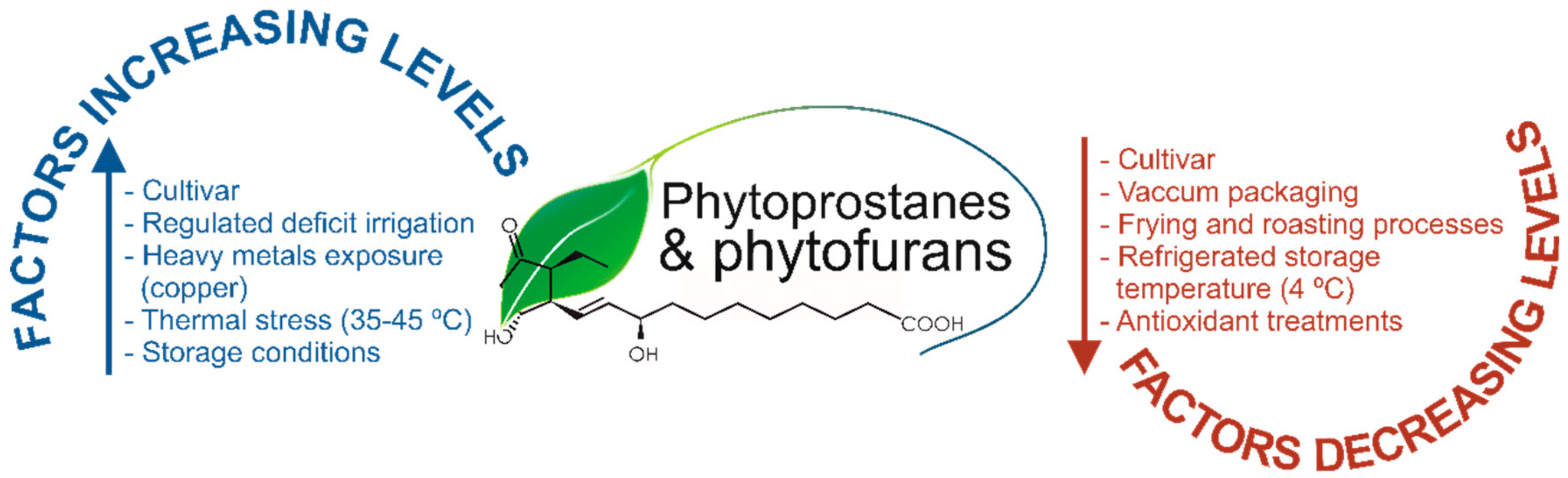
| Matrix/Plant Specie | Analytical Technique | Concentration [Reference] |
|---|---|---|
| Vegetable oils | UHPLC-QqQ-MS/MS | 119,150 ng mL−1 (flax oil) [16] |
| 19,420 ng mL−1 (sesame/safflower oil) [16] | ||
| 16,310 ng mL−1 (EVOO) [16] | ||
| <1900 ng mL−1 (argan, grapeseed, palm oil) [16] | ||
| GC-MS | 90–99,000 ng mL−1 (linseed, soy bean, olive, rapeseed, walnut and grapeseed oils) [13] | |
| UHPLC-QqQ-MS/MS | 14.97 ng mL−1 (EVOO) [17] | |
| 39.35 ng mL−1 (OO) [17] | ||
| 297.45 ng mL−1 (SO) [17] | ||
| UHPLC-QqQ-MS/MS | 31.92 ng mL−1 (EVOO from irrigated 100% trees) [18] | |
| 67.87 ng mL−1 (EVOO from deficit-irrigated trees) [18] | ||
| Olives | UHPLC-QqQ-MS/MS | 0.58–10 ng g−1 (raw flesh) [19] |
| 58–87 ng g−1 (treated flesh) [19] | ||
| Macroalgae | UHPLC-QqQ-MS/MS | 0.06–13.81 ng g−1 DW [20] |
| Brown macroalgae | Micro-LC-MS/MS | 310 ng g−1 FW (Ectocarpus siliculosus) [21] |
| 298.46 ng g−1 FW (Laminaria digitata) [21] | ||
| 78.51 ng g−1 FW (Pelvetia canaliculata) [21] | ||
| 28.91 ng g−1 FW (Fucus spiralis) [21] | ||
| Red macroalgae | Micro-LC-MS/MS | 12.77 ng g−1 FW (Osmundea pinnatifida) [21] |
| 36.97 ng g−1 FW (Grateloupia turuturu) [21] | ||
| Passion fruits | UHPLC-QqQ-MS/MS | 1.30–67.60 ng g−1 DW (Passiflora edulis Sims shell) [22] |
| 2.30–19,786 ng g−1 DW (Passiflora tripartita var. mollisima shell) [23] | ||
| 1.60–21,659.80 ng g−1 DW (Physalis peruviana calyx) [24] | ||
| Almond | UHPLC-QqQ-MS/MS | 77 ng g−1 DW (raw Largueta cultivar) [25] |
| 9.30 ng g−1 DW (fried salt Largueta cultivar) [25] | ||
| 3.50 ng g−1 DW (roasted Largueta cultivar) [25] | ||
| 107 ng g−1 DW (raw Marcona cultivar) [25] | ||
| 18.85 ng g−1 DW (fried salt Marcona cultivar) [25] | ||
| 7.20 ng g−1 DW (roasted Marcona cultivar) [25] | ||
| 40–238 ng g−1 DW [26] | ||
| Nut kernels | UHPLC-QqQ-MS/MS | 5 ng g−1 DW (raw walnut) [27] |
| 6 ng g−1 DW (raw Macadamia) [27] | ||
| 34 ng g−1 DW (fried Macadamia) [27] | ||
| 8 ng g−1 DW (raw Pecan) [27] | ||
| 19 ng g−1 DW (fried Pecan) [27] | ||
| Rice | UHPLC-QqQ-MS/MS | 22.20–106.67 ng g−1 DW (rice bran) [28] |
| 10.14–22.53 ng g−1 DW (brown grain flour) [28] | ||
| 2.55–22.47 ng g−1 DW (white grain flour) [28] | ||
| Betula pendula | GC-MS | 14.7 ng g−1 DW (leaves) [29] |
| Nicotiana tabacum | GC-MS | 31.6 ng g−1 DW (cell culture) [3] |
| 53.2 ng g−1 DW (leaves) [29] | ||
| Glycine max | GC-MS | 10.3 ng g−1 DW (cell culture) [29] |
| Rauvolfia serpentina | GC-MS | 60.9 ng g−1 DW (cell culture) [3] |
| Agrostis tenuis | GC-MS | 4.5 ng g−1 DW (cell culture) [3] |
| Arabidopsis thaliana | GC-MS | 131.3 ng g−1 DW (leaves) [29] |
| Lycopersicon esculentum | GC-MS | 16.9 ng g−1 DW (leaves) [29] |
| Salix alba | GC-MS | 10.6 ng g−1 DW (leaves) [29] |
| Rauvolfia serpentina | GC-MS | 20.5 ng g−1 DW (cell culture) [29] |
| Wine and must | UHPLC-QqQ-MS/MS | 48.7 ng mL−1 (CMM) [11] |
| 20.4 ng mL−1 (AM) [11] | ||
| 430.9 ng mL−1 (HEM) [11] | ||
| 131.8 ng mL−1 (CMW) [11] | ||
| 213 ng mL−1 (AW) [11] | ||
| 199.8 ng mL−1 (HEW) [11] | ||
| Cucumis melo L. | Micro-HPLC-QTRAP | 109–1146 ng g−1 (leaves) [30] |
| Valeriana officinalis | GC-MS | 43 ng g−1 DW (root) [8] |
| Mentha piperita | GC-MS | 76 ng g−1 DW (leaves) [8] |
| GC-MS | 76 ng g−1 DW (leaves) [7] | |
| Tilia cordata/platyphyllos | GC-MS | 135 ng g−1 DW (flowers) [8] |
| Tilia cordata | GC-MS | 18.7 ng g−1 DW (leaves) [29] |
| Hypericum perforatum | GC-MS | 211 ng g−1 DW [8] |
| Betula pendula/pubescens | GC-MS | 1380 ng g−1 DW (leaves) [8] |
| 32,440 ng g−1 DW (pollen) [8] |
| Matrix/Plant Specie | Methodology | Concentration [Reference] |
|---|---|---|
| Vegetables oils | UHPLC-QqQ-MS/MS | 37,920 ng mL−1 (Flax oil) [16] |
| 8850 ng mL−1 (EVOO) [16] | ||
| 30–260 ng mL−1 (sesame, safflower, argan, grapeseed, palm oil) [16] | ||
| Brown macroalgae | Micro-LC-MS/MS | 490 ng g−1 FW (Ectocarpus siliculosus) [21] |
| 93.14 ng g−1 FW (Laminaria digitata) [21] | ||
| 70.03 ng g−1 FW (Pelvetia canaliculata) [21] | ||
| 41.14 ng g−1 FW (Fucus spiralis) [21] | ||
| Red macroalgae | Micro-LC-MS/MS | 11.13 ng g−1 FW (Osmundea pinnatifida) [21] |
| 5.97 ng g−1 FW (Grateloupia turuturu) [21] | ||
| Rice | UHPLC-QqQ-MS/MS | 1.59–27.74 ng g−1 DW (rice bran) [28] |
| 9.51–24.83 ng g−1 DW (brown grain flour) [28] | ||
| 0.06–1.79 ng g−1 DW (white grain flour) [28] | ||
| Cucumis melo L. | Micro-HPLC-QTRAP | 130–4400 ng g−1 (leaves) [30] |
| Nuts and seeds | LC- MS/MS | 0.30 ng g−1 DW (nuts) [4] |
| 0.70 ng g−1 DW (flax seed) [4] | ||
| 6.0 ng g−1 DW (chia seed) [4] | ||
| 9.0 ng g−1 DW (walnuts) [4] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Medina, S.; Gil-Izquierdo, Á.; Durand, T.; Ferreres, F.; Domínguez-Perles, R. Structural/Functional Matches and Divergences of Phytoprostanes and Phytofurans with Bioactive Human Oxylipins. Antioxidants 2018, 7, 165. https://doi.org/10.3390/antiox7110165
Medina S, Gil-Izquierdo Á, Durand T, Ferreres F, Domínguez-Perles R. Structural/Functional Matches and Divergences of Phytoprostanes and Phytofurans with Bioactive Human Oxylipins. Antioxidants. 2018; 7(11):165. https://doi.org/10.3390/antiox7110165
Chicago/Turabian StyleMedina, Sonia, Ángel Gil-Izquierdo, Thierry Durand, Federico Ferreres, and Raúl Domínguez-Perles. 2018. "Structural/Functional Matches and Divergences of Phytoprostanes and Phytofurans with Bioactive Human Oxylipins" Antioxidants 7, no. 11: 165. https://doi.org/10.3390/antiox7110165
APA StyleMedina, S., Gil-Izquierdo, Á., Durand, T., Ferreres, F., & Domínguez-Perles, R. (2018). Structural/Functional Matches and Divergences of Phytoprostanes and Phytofurans with Bioactive Human Oxylipins. Antioxidants, 7(11), 165. https://doi.org/10.3390/antiox7110165






