Phenolic Compounds and Antioxidant Activity of Phalaenopsis Orchid Hybrids
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals
2.2. Plant Material
2.3. Sample Preparation
2.4. Preparation of Ethanol Crude Extracts (Free Phenolics)
2.5. Preparation of Ethyl Acetate Conjugate Crude Extracts (Conjugate Phenolics)
2.6. Estimation of Phenolic Contents
2.7. Estimation of Flavonoid Contents
2.8. Antioxidant Activity
2.8.1. DPPH Scavenging Assay
2.8.2. β-Carotene Bleaching Method
2.9. Quantification of Phenolic Compounds by HPLC
2.10. Statistical Analysis
3. Results
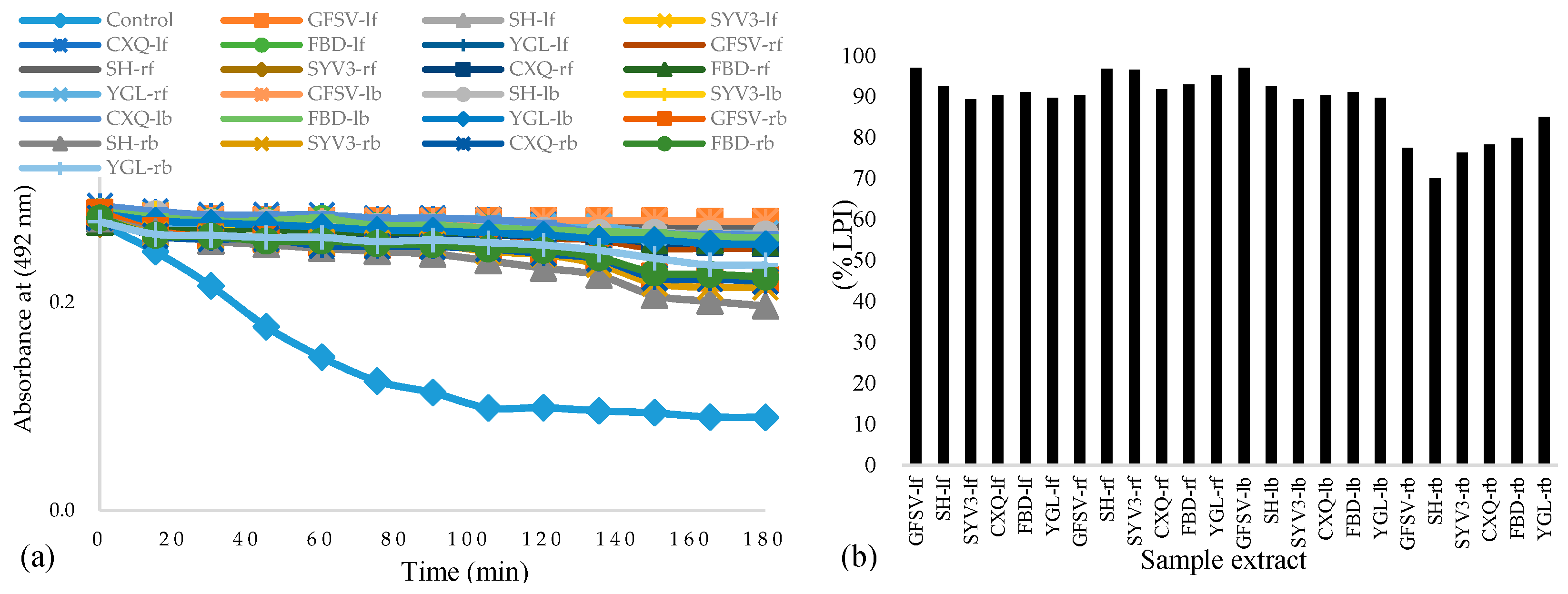
3.1. Antioxidant Activity, Phenolic Contents, and Lipid Peroxidation Inhibition of Phal. Hybrid Extracts
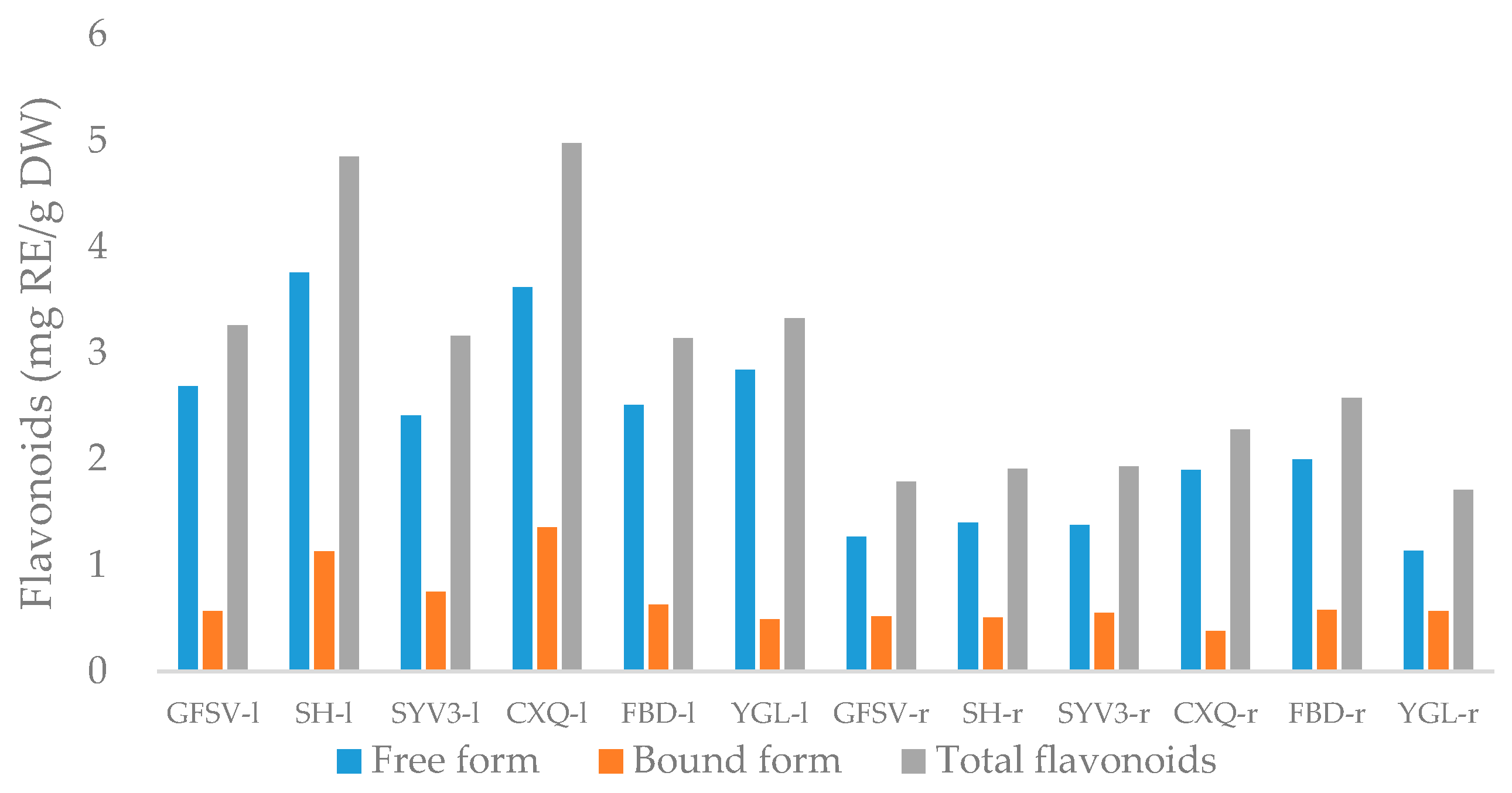
3.2. Total Flavonoids Contents
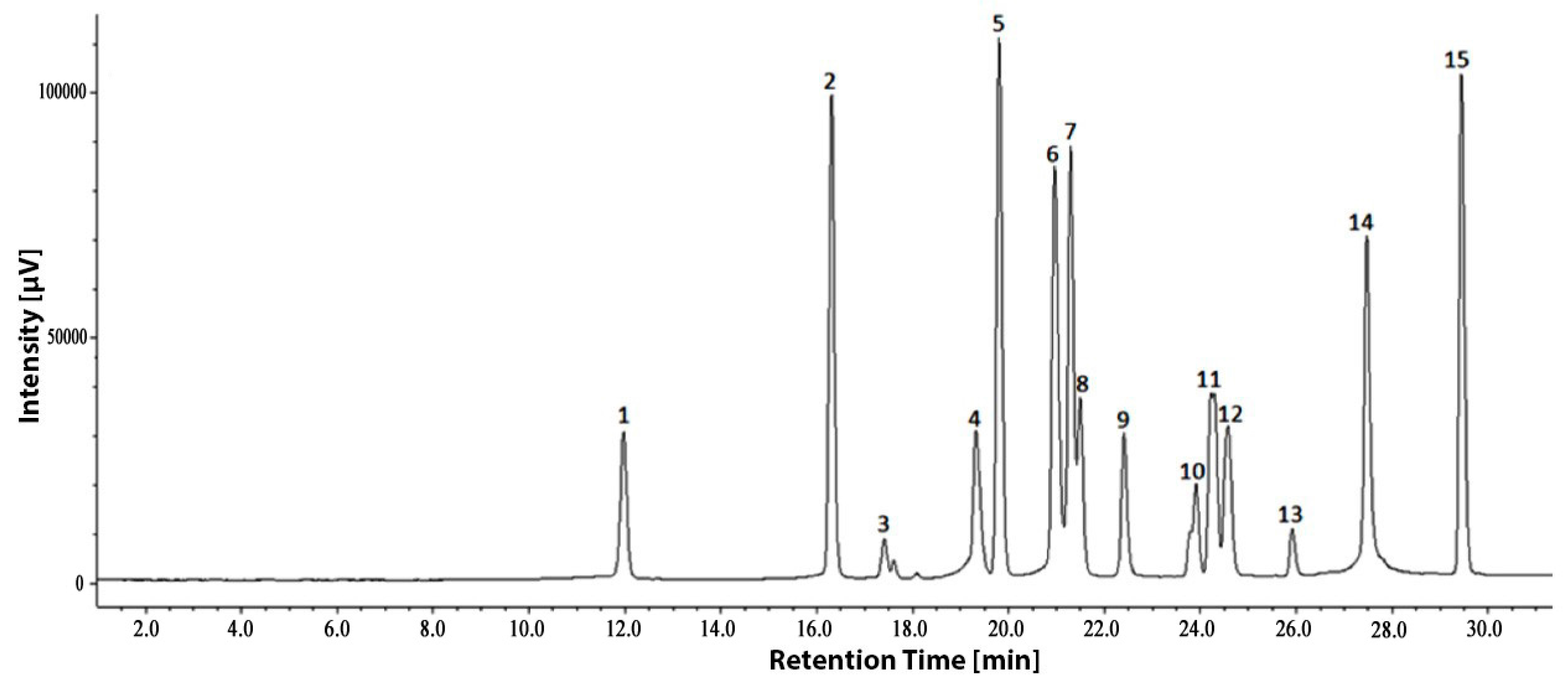
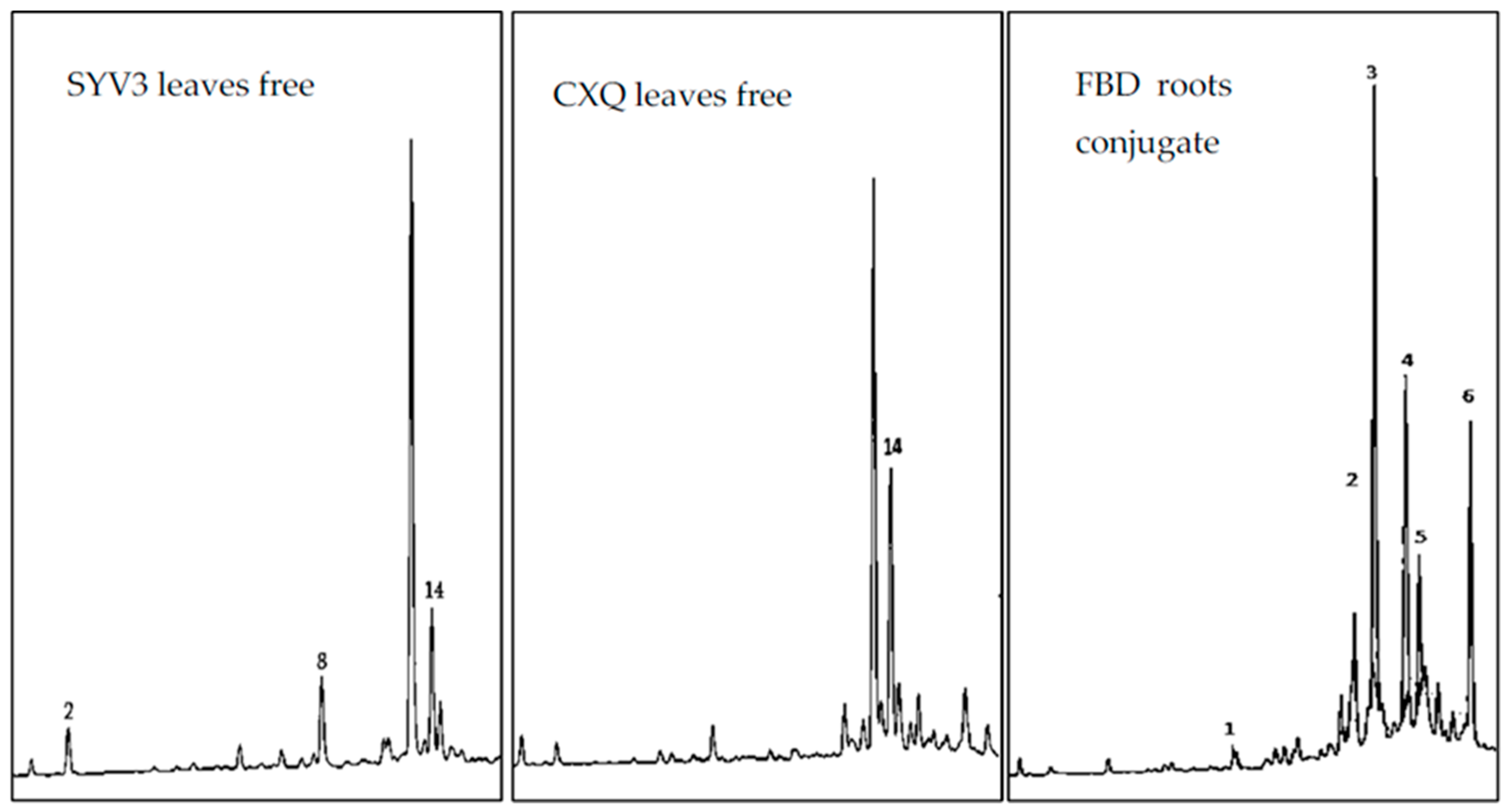
3.3. HPLC Quantification
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interests
References
- Ao, C.; Li, A.; Elzaawely, A.A.; Xuan, T.D.; Tawata, S. Evaluation of antioxidant and antibacterial activities of Ficus microcarpa L. fil. extract. Food Control. 2008, 19, 940–948. [Google Scholar] [CrossRef]
- Barreira, J.C.M.; Ferreira, I.C.F.R.; Oliveira, M.B.P.P.; Pereira, J.A. Antioxidant activities of the extracts from chestnut flower leaf skins and fruit. Food Chem. 2008, 107, 1106–1113. [Google Scholar] [CrossRef]
- Elzaawely, A.A.; Xuan, T.D.; Koyama, H.; Tawata, S. Antioxidant activity and contents of essential oil and phenolic compounds in flowers and seeds of Alpinia zerumbet (Pers.) B.L. Burtt. & R.M. Sm. Food Chem. 2007, 104, 1648–1653. [Google Scholar]
- Manian, R.; Anusuya, N.; Siddhuraju, P.; Manian, S. The antioxidant activity and free radical scavenging potential of two different solvent extracts of Camellia sinensis (L.) O. Kuntz, Ficus bengalensis L. and Ficus. racemosa L. Food Chem. 2008, 107, 1000–1007. [Google Scholar] [CrossRef]
- Biswas, N.; Balac, P.; Narlakanti, S.K.; Haque, M.D.E.; Mehedi, H.M.D. Identification of phenolic compounds in processed cranberries by HPLC method. J. Nutr. Food Sci. 2013, 3. [Google Scholar] [CrossRef]
- Indrianingsih, A.W.; Tachibana, S.; Dewi, R.T.; Itoh, K. Antioxidant and α-glucosidase inhibitor activities of natural compounds isolated from Quercus gilva Blume leaves. Asian Pac. J. Trop. Biomed. 2015, 5, 748–755. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Samoticha, J.; Eler, K.; Stampar, F.; Veberic, R. Traditional elderflower beverages: A rich source of phenolic compounds with high antioxidant activity. J. Agric. Food Chem. 2015, 63, 1477–1487. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Kumari, N. Comparative determination of phytochemicals and antioxidant activity from leaf and fruit of Sapindus mukorrossi Gaertn.—A valuable medicinal tree. Ind. Crops. Prod. 2015, 73, 1–8. [Google Scholar] [CrossRef]
- Wen, L.; Guo, X.; Liu, R.H.; You, L.; Abbasi, A.M.; Fu, X. Phenolic contents and cellular antioxidant activity of Chinese hawthorn “Crataegus Pinnatifida”. Food Chem. 2015, 186, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Do, Q.D.; Angkawijaya, A.E.; Tran-Nguyen, P.L.; Huynh, L.H.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.H. Effect of extraction solvent on total phenol content total flavonoid content and antioxidant activity of Limnophila aromatic. J. Food Drug Anal. 2014, 22, 296–302. [Google Scholar] [CrossRef]
- Bursal, E.; Köksal, E. Evaluation of reducing power and radical scavenging activities of water and ethanol extracts from sumac (Rhus coriaria L.). Food Res. Int. 2011, 44, 2217–2221. [Google Scholar] [CrossRef]
- Hsiao, Y.Y.; Pan, Z.J.; Hsu, C.C.; Yang, Y.P.; Hsu, Y.C.; Chuang, Y.C.; Shih, H.H.; Chen, W.H.; Tsai, W.C.; Chen, H.H. Research on orchid biology and biotechnology. Plant. Cell. Phy. 2011, 52, 1467–1486. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.F.; Subramaniam, S. Biochemical analyses of Phalaenopsis violacea orchid. Asian J. Biochem. 2007, 2, 237–246. [Google Scholar]
- Moretti, M.; Cossignani, L.; Messina, F.C.; Dominici, L.; Villarini, M.; Curini, M.; Marcotullio, M.C. Antigenotoxic effect composition and antioxidant activity of Dendrobium speciosum. Food Chem. 2012, 140, 660–665. [Google Scholar] [CrossRef] [PubMed]
- Lekawatana, S. Thai Orchid: Current situation. In Proceedings of Taiwan International Orchid Symposium, Tainan, Taiwan, 5 March 2010.
- Chien, K.W.; Agrawal, D.C.; Tsay, H.S.; Chang, C.A. Elimination of mixed “Odontoglossum ringspot” and “Cymbidium mosaic” viruses from Phalaenopsis hybrid “V3” through shoot-tip culture and protocorm-like body selection. Crop. Prot. 2015, 67, 1–6. [Google Scholar] [CrossRef]
- Chen, C. Application of growth models to evaluate the microenvironmental conditions using tissue culture plantlets of Phalaenopsis Sogo Yukidian “V3”. Scientia. Hort. 2015, 191, 25–30. [Google Scholar] [CrossRef]
- Elzaawely, A.A.; Xuan, T.D.; Tawata, S. Antioxidant and antibacterial activities of Rumex japonicus HOUTT Aerial parts. Biol. Pharm. Bull. 2005, 28, 2225–2230. [Google Scholar] [CrossRef] [PubMed]
- Xuan, T.D.; Tsuzuki, E.; Terao, H.; Matsuo, M.; Khanh, T.D. Correlation between growth inhibitory exhibition and suspected allelochemicals (phenolic compounds) in the extract of alfalfa (Medicago sativa L.). Plant. Prod. Sci. 2003, 6, 165–171. [Google Scholar] [CrossRef]
- Elzaawely, A.A.; Tawata, S. Antioxidant activity of phenolic rich fraction obtained from Convolvulus arvensis L. leaves grown in Egypt. J. Crop. Sci. 2012, 4, 32–40. [Google Scholar] [CrossRef]
- Djeridane, A.; Yousfi, M.; Nadjemi, B.; Boutassouna, D.; Stocker, P.; Vidal, N. Antioxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem. 2006, 97, 654–660. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Studies on antioxidant activities of mucuna seed (Mucuna pruriens var utilis) extract and various non-protein amino/imino acids through in vitro models. J. Sci. Food Agric. 2003, 83, 1517–1524. [Google Scholar] [CrossRef]
- Soares, A.A.; Marques de Souza, C.G.; Daniel, F.M.; Ferrari, G.P.; Gomes da Costa, S.M.; Peralta, R.M. Antioxidant activity and total phenolic content of Agaricus brasiliensis (Agaricus blazei Murril) in two stages of maturity. Food Chem. 2009, 112, 775–781. [Google Scholar] [CrossRef]
- Bhattacharyya, P.; Kumaria, S.; Diengdoh, R.; Tandon, P. Genetic stability and phytochemical analysis of the in vitro regenerated plants of Dendrobium nobile Lindl., an endangered medicinal orchid. Meta. Gene. 2014, 2, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Shiow, Y.W. Antioxidant activity and phenolic compounds in selected herbs. J. Agric. Food Chem. 2001, 49, 5165–5170. [Google Scholar]
- Kikuzaki, H.; Hisamoto, M.; Hirose, K.; Akiyama, K.; Taniguchi, H. Antioxidant properties of ferulic acid and its related compounds. J. Agric. Food Chem. 2002, 50, 2161–2168. [Google Scholar] [CrossRef] [PubMed]
- Han, D.H.; Lee, M.J.; Kim, J.H. Antioxidant and apoptosis-inducing activities of ellagic acid. Anticancer Res. 2006, 26, 3601–3606. [Google Scholar] [PubMed]
- Siger, A.; Czubinski, J.; Dwiecki, K.; Kachlicki, P.; Nogala-Kalucka, M. Identification and antioxidant activity of sinapic acid derivatives in Brassica napus L. seed meal extracts. Eur. J. Lipid Sci. Technol. 2013, 115, 1130–1138. [Google Scholar]
- Khang, D.T.; Dung, T.N.; Elzaawely, A.A.; Xuan, T.D. Phenolic profiles and antioxidant activity of germinated legumes. Foods 2016, 5. [Google Scholar] [CrossRef]
- Tokuhara, K.; Mii, M. Highly-efficient somatic embryogenesis from cell suspension cultures of Phalaenopsis orchids by adjusting carbohydrate sources. Cell. Dev. Biol. Plant. 2003, 39, 635–639. [Google Scholar] [CrossRef]
- Moin, S.; Sahaya, B.S.; Servin, P.W.; Chitra, B.D. Bioactive potential of Coelogyne stricta (D.Don) Schltr: An ornamental and medicinally important orchid. J. Phar. Res. 2012, 5, 2191–2196. [Google Scholar]
- Rokaya, M.B.; Uprety, Y.; Poudele, R.C.; Timsina, B.; Münzbergová, Z.; Asselind, H.; Tiwari, A.; Shresthac, S.S.; Sigdeli, S.R. Traditional uses of medicinal plants in gastrointestinal disorders in Nepal. J. Ethnophar. 2014, 158, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Shubha, J.; Chowdappa, S. Phytochemical analysis and Antibacterial activity of Cymbidium aloifolium L. a medicinal orchid from Western Ghats of Karnataka, India. Int. J. Adv. Sci. Res. Pub. 2016, 2, 19–23. [Google Scholar]
- Negi, A.; Sharma, N.; Pant, R.; Singh, M.F. Determination of total phenolic content of the stem bark of Bauhinia variegata Linn.; an approach to standardization. Phar. Res. 2013, 7, 16–22. [Google Scholar]
- Chen, B.; Trueman, S.J.; Li, J.; Li, Q.; Fan, H.; Zhang, J. Micropropagation of the endangered medicinal orchid, Dendrobium officinale. Life Sci. J. 2014, 11, 526–530. [Google Scholar]
- Dalara, A.; Guoa, Y.; Esimd, N.; Bengue, A.S.; Konczak, I. Health attributes of an endemic orchid from Eastern Anatolia, Dactylorhiza chuhensis Renz & Taub.—In vitro investigations. J. Herb. Med. 2015, 5, 77–85. [Google Scholar]
- Pujari, I.; Shetty, V.; Thomas, A.; Muthusamy, A.; and Babu, V.S. Callus induction and regeneration for moscatilin yield in Dendrobium ovatum, a threatened medicinal orchid. Nat. Prod. Chem. Res. 2015, 3, 6. [Google Scholar]
- Ezenwali, M.O.; Njoku, O.U.; Okoli, C.O. Studies on the anti-diarrheal properties of seed extract of Monodora tenuifolia. Int. J. App. Res. Na. Pro. 2010, 2, 20–26. [Google Scholar]
- Bhattacharjee, B.; Islam, T.; Rahman, Z.; Islam, S.M.S. Antimicrobial activity and phytochemical screening of whole plant extracts of Vanda tessellata (Roxb.). W. J. Phar. Phar. Sci. 2014, 4, 72–83. [Google Scholar]
- Sahaya, B.S.; Sarmad, M.; Servin, P.W.; Chitra, B.D. Preliminary phytochemical screening, antibacterial and antioxidant activity of Eria pseudoclavicaulis Blatt.—An endemic orchid of Western Ghats. Am. J. Pharm. Tech. Res. 2012, 2, 519–525. [Google Scholar]
- Bhattacharjee, B.; Islam, S.M.S. Assessment of antibacterial and antifungal activity of the extract of Rhynchostylis retusa blume—A medicinal orchid. W. J. Phar. Phar. Sci. 2015, 4, 74–87. [Google Scholar]
- Maridassa, M.; Hussainb, M.I.Z.; Raju, G. Phytochemical survey of orchids in the Tirunelveli hills of South India. Ethnobotanical. Leaflets 2008, 12, 705–712. [Google Scholar]
- Johnson, M.; Janakiraman, N. Phytochemical and TLC studies on stem and leaves of the orchid Dendrobium panduratum subsp. villosum Gopalan & AN Henry. Indian J. Nat. Pro. Res. 2013, 4, 250–254. [Google Scholar]
- Ramos, P.; Colareda, G.A.; Rosella, M.A.; Debenedetti, S.L.; Spegazzini, E.D.; Consolini, A.E. Phytochemical profile and anti-inflammatory effect of the orchid Catasetum macroglossum. Latin Ame. J. Phar. 2012, 31, 62–67. [Google Scholar]
- Miyazawa, M.; Shimamura, H.; Nakamura, S.; Kameoka, H. Antimutagenic activity of gigantol from Dendrobium nobile. J. Agric. Food Chem. 1997, 45, 2849–2853. [Google Scholar] [CrossRef]
- Suzuki, M.; Hayakawa, Y.; Aoki, K.; Nagase, H.; Nakamura, H.; Yamada, K.; Hirata, Y. Stereochemistry of intermediates in the syntheses of Dendrobium alkaloids. Tetrahedron Lett. 1973, 14, 331–334. [Google Scholar] [CrossRef]
- Zhao, W.; Ye, Q.; Tan, X.; Jiang, H.; Li, X.; Chen, K.; Kinghorn, A.D. Three new sesquiterpene glycosides from Dendrobium nobile with immunomodulatory Activity. J. Nat. Prod. 2001, 64, 1196–1200. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Wang, H.J.; Murphy, P.A.; Hendrich, S. Soybean isoflavone extract suppresses early but not later promotion of hepatocarcinogenesis by phenobarbital in female rat liver. J. Nut. Can. 1995, 24, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Natsume, M.; Onaka, T.; Uchmaru, F.; Shimizu, M. The structure of dendramine (6-oxydendrobine) and 6-oxydendroxine. The fourth and fifth alkaloid from Dendrobium nobile. Chem. Pha. Bull. 1966, 14, 676–680. [Google Scholar] [CrossRef]
- Williams, C.A. The leaf flavonoids of the orchidaceae. Phytochemistry 1979, 18, 803–813. [Google Scholar] [CrossRef]
- Majumder, P.L.; Sen, R.C. Pendulin, a polyoxygenated phenanthrene derivative from the orchid Cymbidium pendulum. Phytochemistry 1991, 30, 2432–2434. [Google Scholar] [CrossRef]
- Majumder, P.L.; Lahiri, S.; Mukhoti, N. Four stilbenoids from the orchid Agrostophyllum khasiyanum. Phytochemistry 1996, 42, 1157–1161. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, Q.; Halaweish, F.; Shao, B.; Ye, Y.; Zhao, W. Copacamphane, picrotoxane, and alloaromadendrane sesquiterpene glycosides and phenolic glycosides from Dendrobium moniliforme. J. Nat. Prod. 2003, 66, 1140–1143. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhang, Y.; Liu, G.; Zhang, J. Dactylorhin B reduces toxic effects of β-amyloid fragment (25–35) on neuron cells and isolated rat brain mitochondria. Naunyn. Schmiedebergs. Arch. Pharmacol. 2006, 374, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Duggal, S. Medicinal orchids—An overview. Ethnobotanical. Leaflets 2009, 13, 399–412. [Google Scholar]
- Ghanaksh, A.; Kaushik, P. Antibacterial effect of Aerides multiflora Roxb: A study in vitro. J. Orchid Soc. India. 1999, 1, 65–68. [Google Scholar]
- Bijaya, P. Medicinal orchids and their uses: Tissue culture a potential alternative for conservation. Afr. J. Plant. Sci. 2013, 7, 448–467. [Google Scholar]
- Islam, M.S.; Mehraj, H.; Roni, M.Z.K.; Shimasaki, K.; Uddin, A.F.M.J. Correlation between cane growth and flowering behavior of Dendrobium orchid cultivars. J. Bangladesh Aca. Sci. 2013, 37, 205–209. [Google Scholar] [CrossRef]
- Luo, H.; Lin, S.; Ren, F.; Wu, L.; Chen, L.; Sun, Y. Antioxidant andantimicrobial capacity of Chinese medicinal herb extracts in rawsheep meat. J. Food Protect. 2007, 70, 1440–1445. [Google Scholar]
- Bulpitt, C.J. The uses and misuses of orchids in medicine. Q. J. Med. 2005, 98, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.F. Medicinal Plant Fact Sheet: Cypripedium: Lady’s Slipper Orchids. Available online: http://www.pollinator.org/Resources/Cypripedium.draft.pdf (accessed on 6 August 2016).
- Srivirojana, N.; Theptepa, T.; Punpuing, S.; Guest, P.; Tun, K.; Chankham, O.; Suvansrual, A. Population pressure, utilization of chemicals in agriculture, health outcomes and solid waste management. In Proceedings of the International Conference on Integrated Solid Waste Management in Southeast Asian Cities, Siem Reap, Cambodia, 5–7 July 2005.
- Johnson, M.K.; Alexander, K.E.; Lindquist, N.; Loo, G. A phenolic antioxidant from the freshwater orchid, Habenaria repens. Comp. Biochem. Phys. C Pharmacol. Toxicol. Endocrinol. 1998, 122, 211–214. [Google Scholar] [CrossRef]
- Qasem, J.R.; Foy, C.L. Weed allelopathy, its ecological impact and future prospects. J. Crop. Prod. 2001, 4, 43–119. [Google Scholar] [CrossRef]
| Name | Abbreviation |
|---|---|
| Green Field Sweet Valentine “Montclair” | GFSV |
| Sakura Hime | SH |
| Sogo Yukidian “V3” | SYV3 |
| Chian Xen Queen | CXQ |
| Fusheng‘s Bridal Dress “Meidarland” | FBD |
| Younghome Golden Leopard “Peachy” | YGL |
| Sample | DPPH IC50 (mg/mL) | Phenolics (mg GAE/g DW) | ||
|---|---|---|---|---|
| Free Form | Conjugate Form | Free Form | Conjugate Form | |
| Leaves | ||||
| GFSV | > 4 | 0.534 ± 0.004 b | 1.65 ± 0.06 c,d | 4.57 ± 0.09 g |
| SH | > 4 | 0.494 ± 0.016 c | 1.98 ± 0.03 b,c | 6.96 ± 0.10 d |
| SYV3 | > 4 | 0.430 ± 0.004 d | 2.29 ± 0.05 b,c | 5.50 ± 0.12 f |
| CXQ | > 4 | 0.435 ± 0.008 d | 2.97 ± 0.45 b,c | 8.55 ± 0.02 a |
| FBD | > 4 | 0.536 ± 0.000 b | 1.91 ± 0.12 b,c | 5.15 ± 0.06 f |
| YGL | > 4 | 0.645 ± 0.003 a | 0.55 ± 0.00 d | 4.76 ± 0.14 d |
| Roots | ||||
| GFSV | 0.362 ± 0.021 c | 0.087 ± 0.000 e | 1.55 ± 0.32 c,d | 5.87 ± 0.02 e |
| GFSV | 0.427 ± 0.008 b | 0.092 ± 0.000 e | 2.34 ± 0.08 b,c | 6.91 ± 0.00 d |
| SYV3 | 0.348 ± 0.001 c | 0.089 ± 0.000 e | 3.78 ± 0.44 a | 7.55 ± 0.10 b |
| CXQ | 0.715 ± 0.022 a | 0.100 ± 0.000 e | 1.44 ± 0.03 c,d | 7.14 ± 0.10 c,d |
| FBD | 0.364 ± 0.000 c | 0.098 ± 0.000 e | 4.01 ± 0.76 a | 6.91 ± 0.13 d |
| YGL | 0.379 ± 0.010 b,c | 0.092 ± 0.000 e | 3.61 ± 0.29 a | 7.42 ± 0.09 b,c |
| BHT | 0.019 ± 0.001 | - | ||
| Sample | Contents of Phenolic Compounds (µg/g DW) | ||||||
|---|---|---|---|---|---|---|---|
| PA | SYA | FA | SI | p-CA | BA | EA | |
| Leaves | |||||||
| GFSV | 133.65 ± 0.08 b | - | - | - | - | - | 38.34 ± 1.10 c |
| SH | - | - | - | 274.24 ± 9.85 | - | 178.60 ± 2.49 | 40.07 ± 1.89 c |
| SYV3 | 147.09 ± 0.12 a | 99.18 ± 0.67 | - | - | - | - | 119.21 ± 0.54 b |
| CXQ | - | - | - | - | - | - | 346.30 ± 14.38 a |
| FBD | - | - | 216.05 ± 0.19 | - | - | - | - |
| YGL | - | - | - | - | - | - | 57.16 ± 1.15 c |
| Roots | |||||||
| GFSV | - | - | - | - | - | - | - |
| SH | - | - | - | - | - | - | 118.20 ± 2.24 b |
| SYV3 | - | - | - | - | 272.00 ± 2.83 | - | - |
| CXQ | - | - | - | - | - | - | - |
| FBD | - | - | - | - | - | - | - |
| YGL | - | - | - | - | 236.94 ± 19.62 | - | - |
| ns | |||||||
| Sample | Contents of Phenolic Compounds (µg/g DW) | |||||
|---|---|---|---|---|---|---|
| PA | p-HBA | VA | CA | SYA | V | |
| Leaves | ||||||
| GFSV | - | - | - | - | - | 5.07 ± 1.07 c |
| SH | 122.31 ± 0.24 b | 77.44 ± 0.10 | 6.78 ± 1.72 d | - | 93.73 ± 3.47 c | - |
| SYV3 | - | - | - | - | - | - |
| CXQ | - | - | 18.93 ± 3.78 d | - | - | - |
| FBD | - | - | - | - | - | - |
| YGL | - | - | - | - | 104.97 ± 1.04 b,c | - |
| Roots | ||||||
| GFSV | - | 86.50 ± 4.22 | 85.25 ± 19.29 a,b,c | - | 125.54 ± 5.30 a | 119.15 ± 27.48 b |
| SH | - | - | 69.53 ± 0.21 c | 123.62 ± 23.66 | - | - |
| SYV3 | - | - | - | - | - | - |
| CXQ | - | - | 107.24 ± 8.93 a,b | - | 106.18 ± 3.93 b,c | 113.31 ± 3.13 b |
| FBD | - | - | 77.99 ± 1.95 b,c | - | 109.81 ± 2.67 b | 97.58 ± 7.15 b |
| YGL | 184.24 ± 1.24 a | - | 117.84 ± 1.06 a | - | 117.08 ± 1.82 a,b | 185.23 ± 1.52 a |
| Sample | Contents of Phenolic Compounds (µg/g DW) | ||||
|---|---|---|---|---|---|
| FA | SIA | p-CA | BA | EA | |
| Leaves | |||||
| GFSV | 383.63 ± 22.21 a | - | - | - | - |
| SH | 414.31 ± 17.69 a | 1141.65 ± 67.00 c | - | - | 53.80 ± 6.49 a,b |
| SYV3 | 414.95 ± 12.08 a | 709.15 ± 15.71 d | 232.31 ± 9.32 c | - | 31.51 ± 0.21 c |
| CXQ | 384.75 ± 6.19 a | - | - | - | - |
| FBD | - | - | 422.94 ± 70.13 b | - | - |
| YGL | 432.68 ± 19.03 a | - | 313.29 ± 28.47 b,c | - | 60.71 ± 7.37 a |
| Roots | |||||
| GFSV | 173.77 ± 4.67 c | - | - | 340.54 ± 106.00 | - |
| SH | 192.41 ± 3.44b c | - | 767.81 ± 20.67 a | - | 35.01 ± 5.56 b,c |
| SYV3 | 221.61 ± 3.30b c | 2232.81 ± 29.71 a | 288.02 ± 9.31 c | 242.47 ± 10.65 | 46.99 ± 0.29 a,b,c |
| CXQ | 224.14 ± 13.29 b | 1639.24 ± 42.70 b | 298.20 ± 11.29 c | 268.62 ± 41.34 | - |
| FBD | - | - | 289.51 ± 0.64 c | 139.86 ± 50.62 | 36.23 ± 4.63 b,c |
| YGL | 227.80 ± 4.73 b | 1800.92 ± 39.45 b | 274.31 ± 2.12 c | 262.22 ± 13.36 | - |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Minh, T.N.; Khang, D.T.; Tuyen, P.T.; Minh, L.T.; Anh, L.H.; Quan, N.V.; Ha, P.T.T.; Quan, N.T.; Toan, N.P.; Elzaawely, A.A.; et al. Phenolic Compounds and Antioxidant Activity of Phalaenopsis Orchid Hybrids. Antioxidants 2016, 5, 31. https://doi.org/10.3390/antiox5030031
Minh TN, Khang DT, Tuyen PT, Minh LT, Anh LH, Quan NV, Ha PTT, Quan NT, Toan NP, Elzaawely AA, et al. Phenolic Compounds and Antioxidant Activity of Phalaenopsis Orchid Hybrids. Antioxidants. 2016; 5(3):31. https://doi.org/10.3390/antiox5030031
Chicago/Turabian StyleMinh, Truong Ngoc, Do Tan Khang, Phung Thi Tuyen, Luong The Minh, La Hoang Anh, Nguyen Van Quan, Pham Thi Thu Ha, Nguyen Thanh Quan, Nguyen Phu Toan, Abdelnaser Abdelghany Elzaawely, and et al. 2016. "Phenolic Compounds and Antioxidant Activity of Phalaenopsis Orchid Hybrids" Antioxidants 5, no. 3: 31. https://doi.org/10.3390/antiox5030031
APA StyleMinh, T. N., Khang, D. T., Tuyen, P. T., Minh, L. T., Anh, L. H., Quan, N. V., Ha, P. T. T., Quan, N. T., Toan, N. P., Elzaawely, A. A., & Xuan, T. D. (2016). Phenolic Compounds and Antioxidant Activity of Phalaenopsis Orchid Hybrids. Antioxidants, 5(3), 31. https://doi.org/10.3390/antiox5030031










