Food Inhibits the Oral Bioavailability of the Major Green Tea Antioxidant Epigallocatechin Gallate in Humans
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Reagents
Preparation and Quality Control of EGCG Products for Oral Delivery
| Ingredient | Weight (g) | % (w/w) |
|---|---|---|
| Strawberries (Creative Gourmet Pty Ltd., Silverwater, NSW, Australia) | 250 | 76.63 |
| Caster Sugar (CSR, Yarraville, VIC, Australia) | 45 | 13.79 |
| Strawberry Flavoured WPI (Vital Strength Nutraceuticals, Marrickville, NSW, Australia) | 30 | 9.20 |
| Carboxyl methyl cellulose (Sigma-Aldrich, Castle Hill, NSW, Australia) | 0.40 | 0.12 |
| EGCG (RejuvaCare International, Sydney, NSW, Australia) | 0.84 | 0.25 |
| Total Weight | 326.24 | 100 |
2.2. Treatments
2.2.1. Ethics
2.2.2. Participants and Selection Criteria
2.2.3. Study Clinics
2.3. Blood Collection and Handling
2.4. Determination of Free EGCG in Plasma Samples
2.4.1. Preparation and Extraction of EGCG from Plasma Samples
2.4.2. Equipment and Chromatographic Conditions
2.4.3. Tuning and Setting Parameters of the ESI-MS
2.5. Statistical Analysis
3. Results
3.1. Recovery of EGCG in Strawberry Sorbet
| n | EGCG Added to Strawberry Sorbet (mg/g) | EGCG Extracted from Strawberry Sorbet (mg/g) † | EGCG Recovery (%) † |
|---|---|---|---|
| 5 | 2.50 | 2.44 ± 0.03 | 97.4 ± 1.3 |
3.2. Measurements of EGCG in Human Plasma
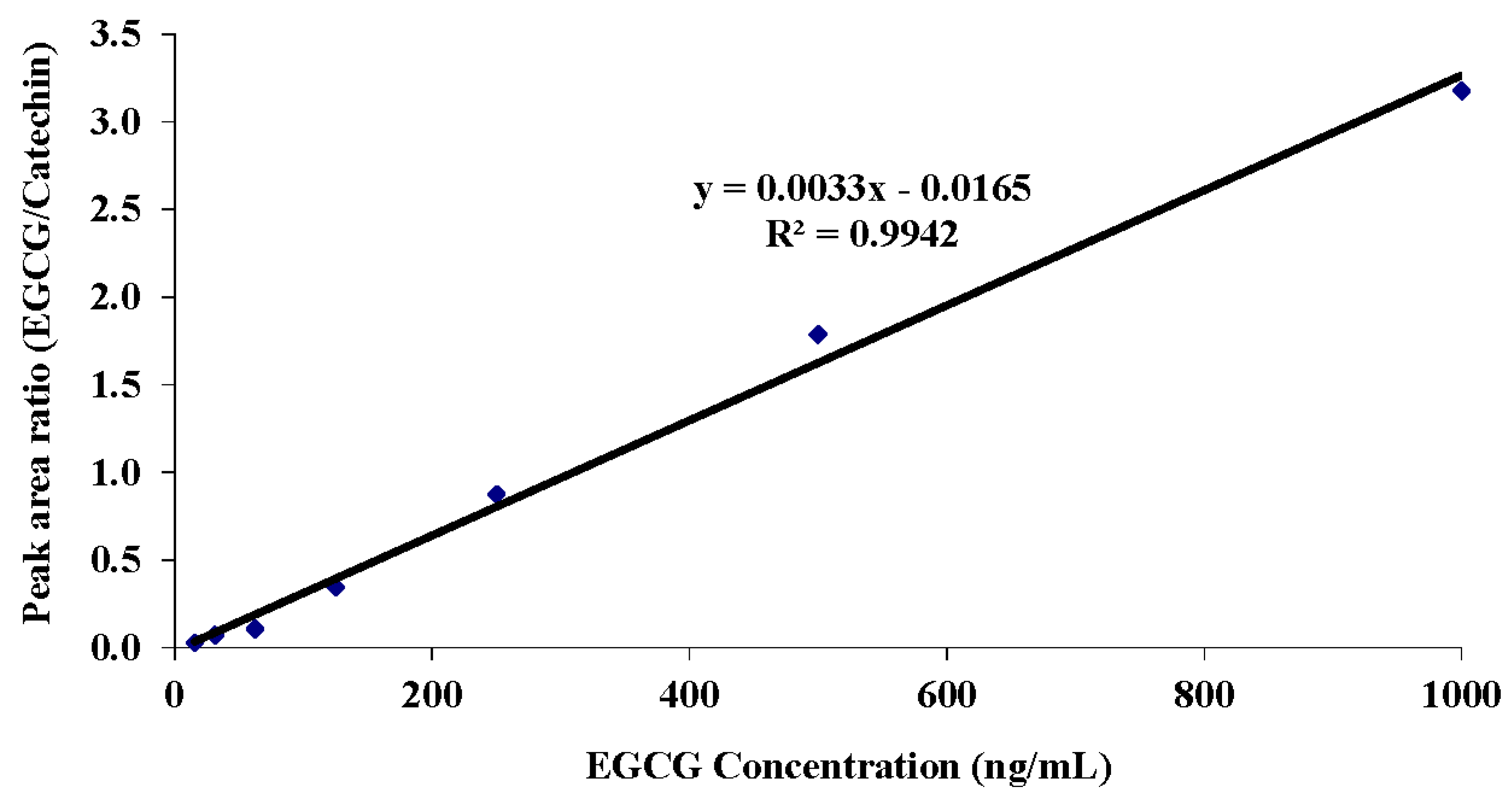
| EGCG Concentration (ng/mL) | n | Mean Peak Area Ratio (EGCG/Catechin) | SD Peak Area Ratio | CV (%) |
|---|---|---|---|---|
| 1000 | 5 | 3.18 | 0.004 | 1.21 |
| 500 | 5 | 1.79 | 0.041 | 2.32 |
| 250 | 5 | 0.88 | 0.023 | 2.67 |
| 125 | 5 | 0.35 | 0.014 | 4.01 |
| 62.5 | 5 | 0.11 | 0.003 | 2.82 |
| 31.25 † | 5 | 0.07 | 0.004 | 4.78 |
| 15.63 * | 5 | 0.03 | 0.003 | 8.77 |
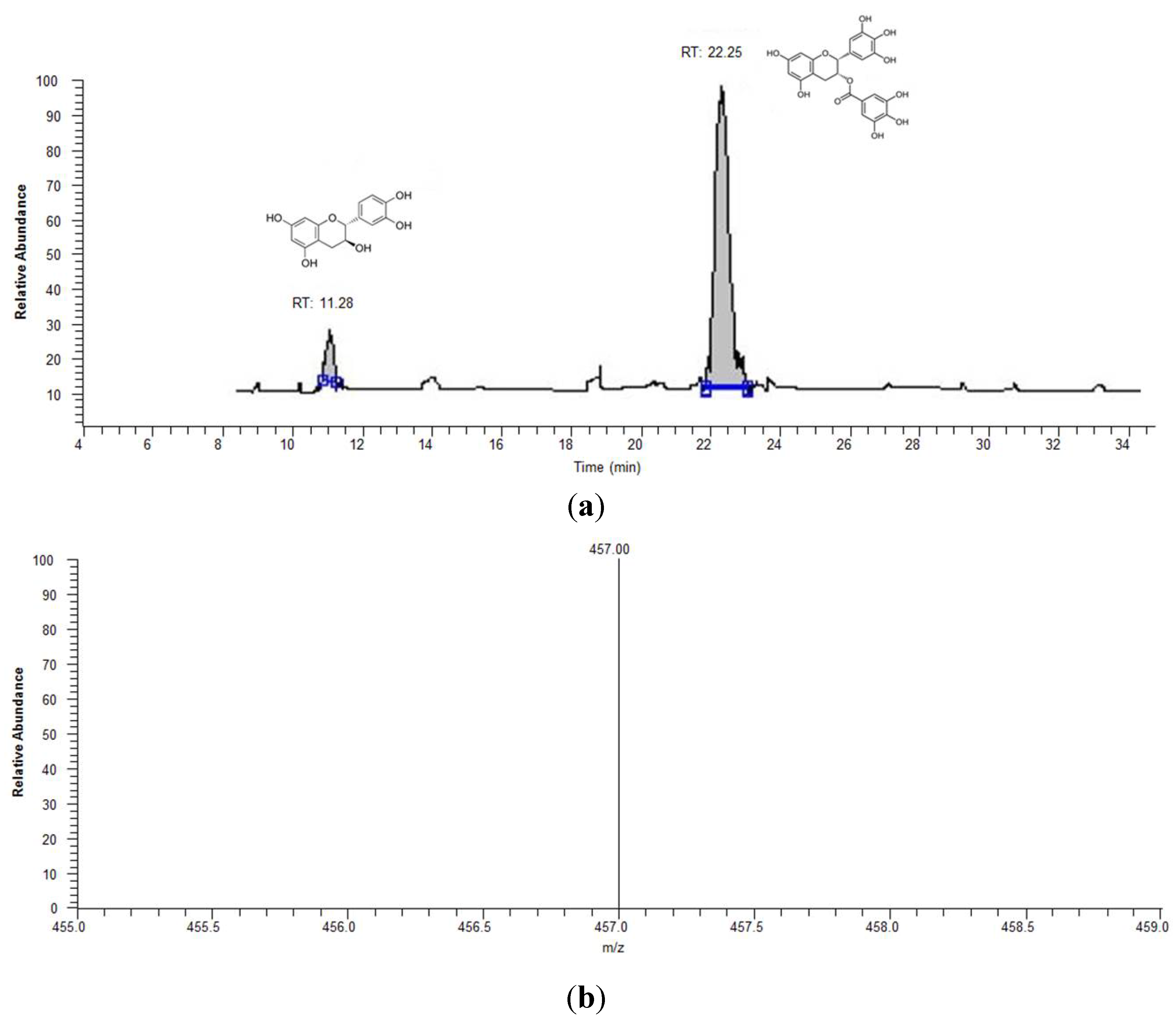
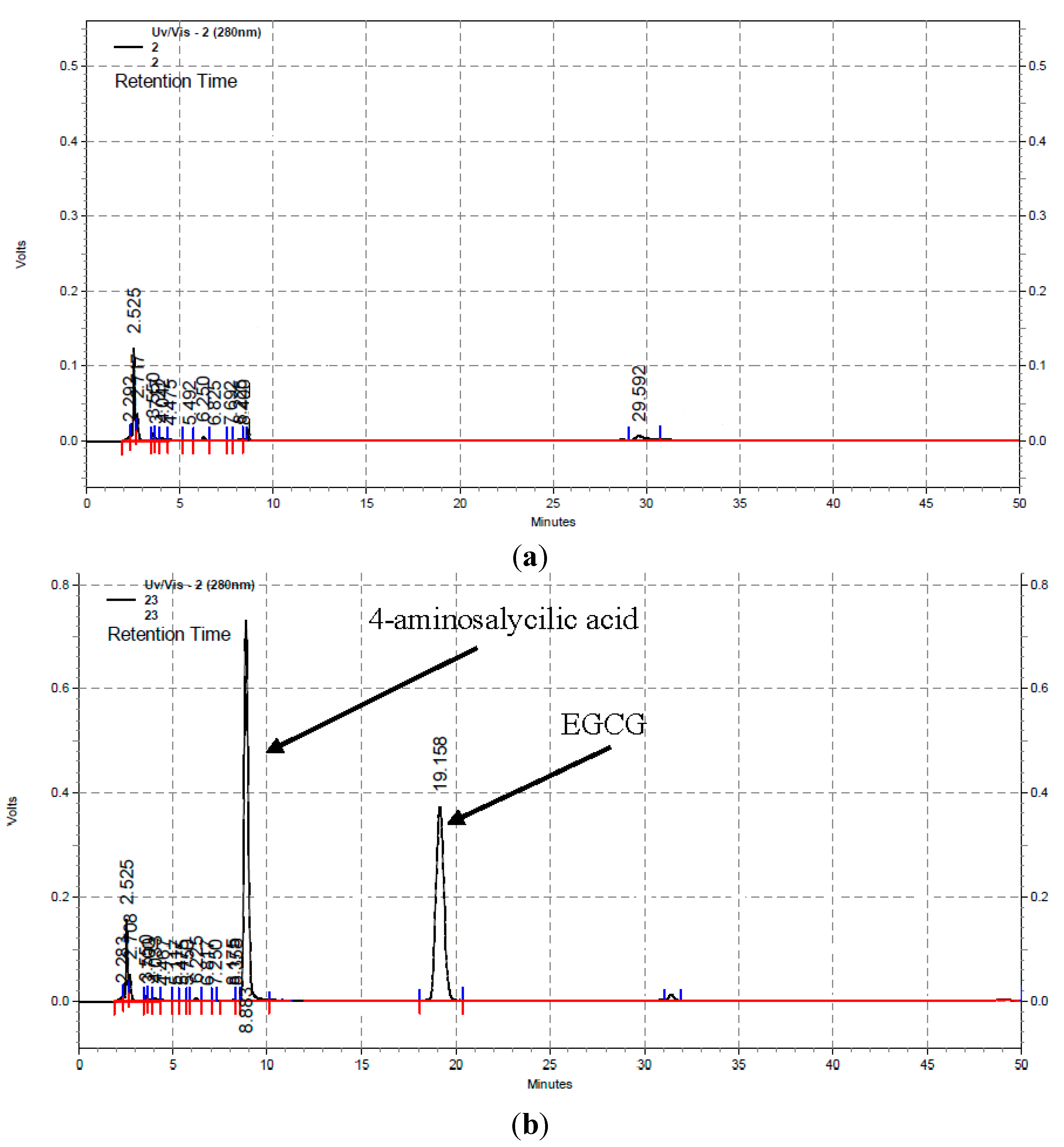
3.3. Participants’ Anthropometric and Blood Pressure Data
| Clinic 1 | Clinic 2 | Clinic 3 | |
|---|---|---|---|
| Weight (kg) | 70.5 ± 10.7 | 70.7 ± 10.8 | 70.5 ± 10.8 |
| Height (m) | 1.75 ± 0.08 | 1.75 ± 0.08 | 1.75 ± 0.08 |
| BMI (kg/m2) | 23.1 ± 3.5 | 23.2 ± 3.6 | 23.1 ± 3.5 |
| SBP (mmHg) | 112.5 ± 5.0 | 115.3 ± 6.1 | 112.5 ± 5.0 |
| DBP (mmHg) | 85.0 ± 4.1 | 85.5 ± 3.3 | 82.5 ± 5 |
3.4. Plasma EGCG Concentration-Time Results
3.5. Pharmacokinetic Parameters of Plasma EGCG
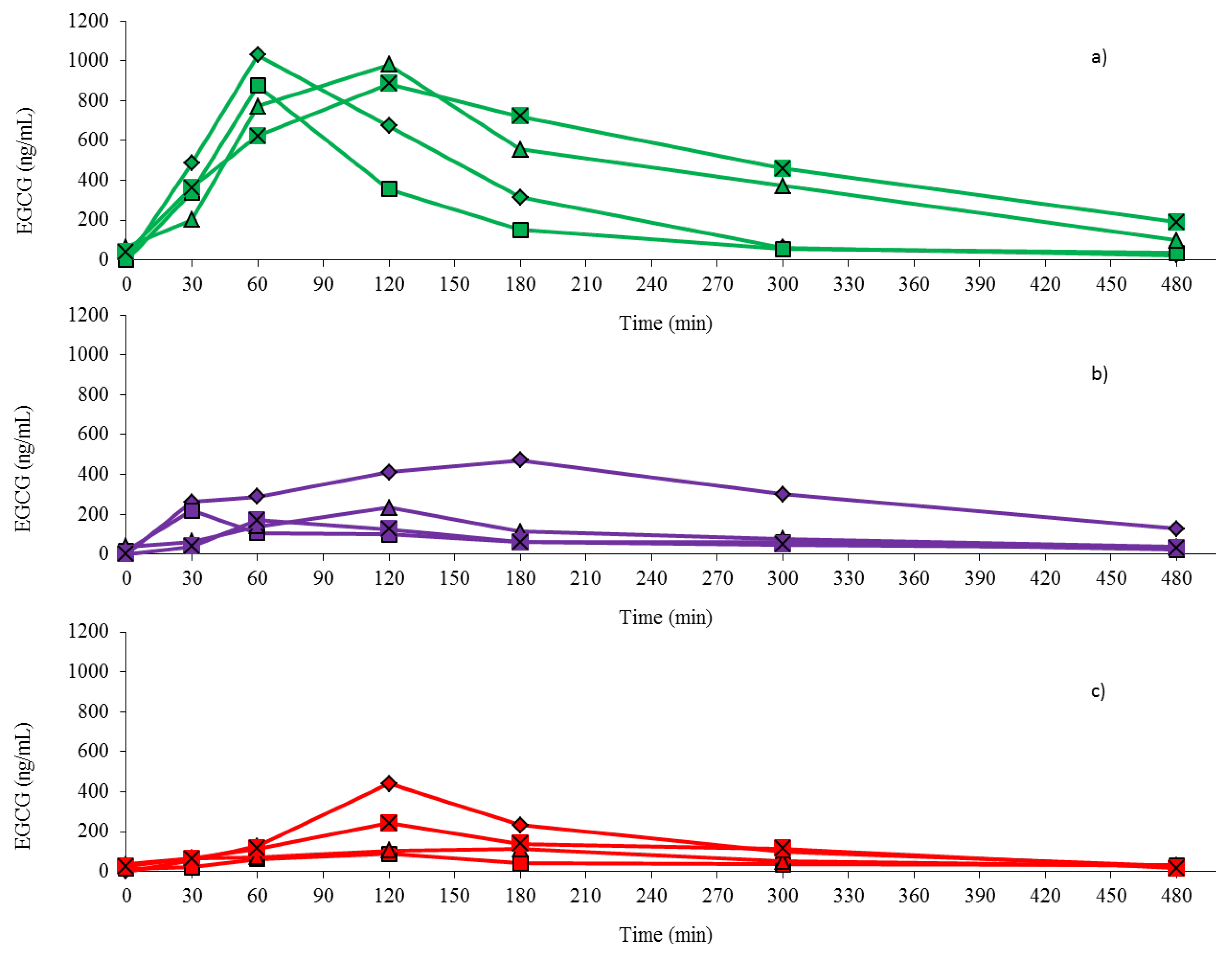
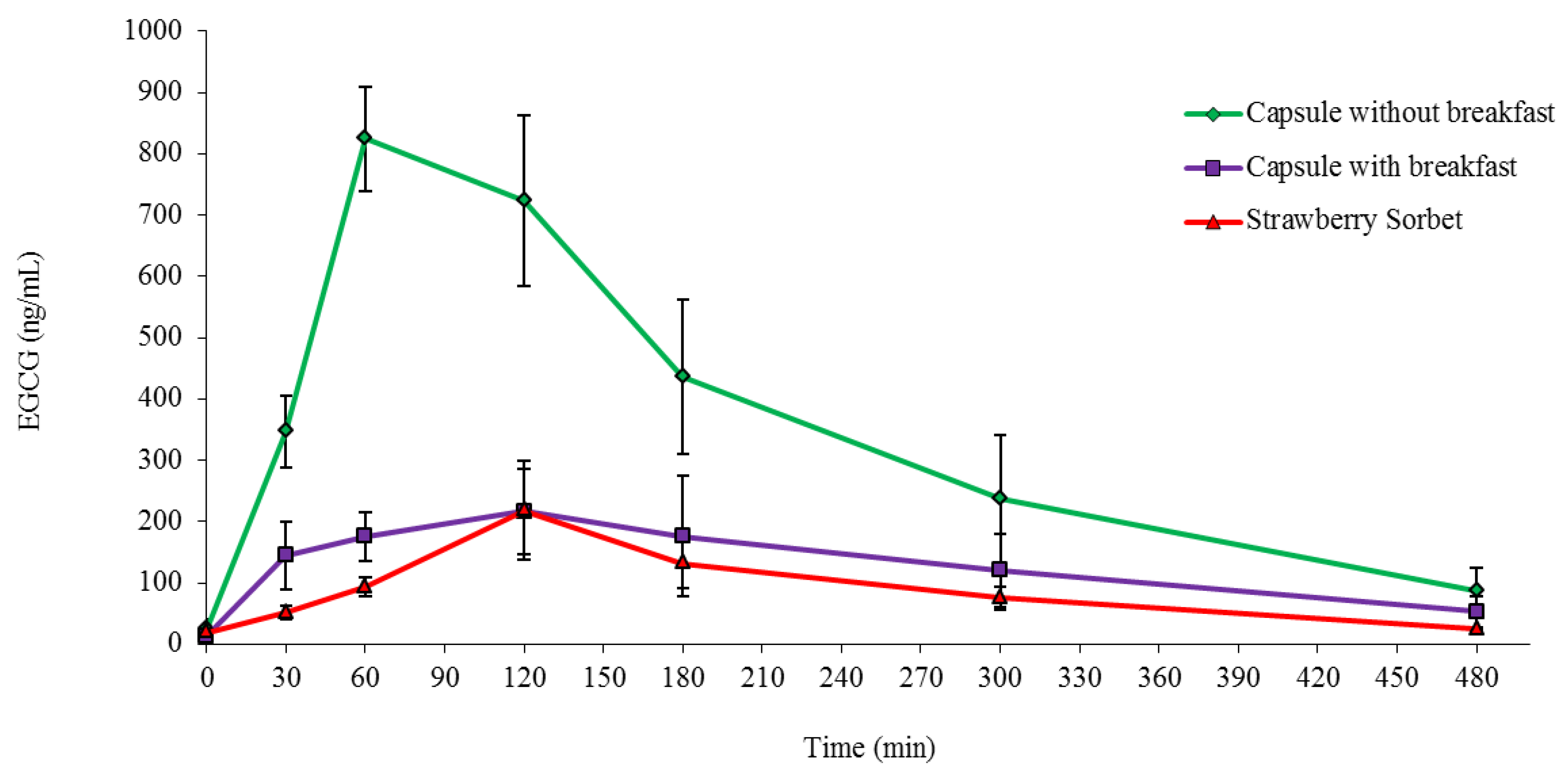
| EGCG Capsules Without Breakfast | EGCG Capsules With Breakfast | EGCG in Strawberry Sorbet | |
|---|---|---|---|
| AUC0–8 (μg/mL/8 h) | 173.8 ± 67.6 * | 64.1 ± 53.7 | 44.5 ± 22.9 |
| Cmax (ng/mL) | 824.2 ± 75.1 ** | 231.8 ± 134.3 | 218.0 ± 160.0 |
| Cav (ng/mL) | 382.6 ± 92.5 * | 92.0 ± 46.0 | 87.6 ± 43.0 |
| Cmin (ng/mL) | 86.9 ± 75.2 | 49.3 ± 24.6 | 25.0 ± 5.8 |
| T1/2 (min) | 78.6 ± 94.2 | 136.2 ± 85.2 | 228.6 ± 97.2 |
| Tmax (min) | 60 ± 34.6 ** | 120 ± 34.6 | 120 ± 34.6 |
| DF | 1.93 ± 0.77 | 1.39 ± 0.46 | 2.20 ± 0.76 |
| Swing | 8.49 ± 17.19 | 3.36 ± 3.04 | 7.70 ± 6.61 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gundimeda, U.; McNeill, T.H.; Barseghian, B.A.; Tzeng, W.; Rayudu, D.; Cadenas, E.; Gopalakrishna, R. Polyphenols from green tea prevent antineuritogenic action of Nogo-A via 67-kDa laminin receptor and hydrogen peroxide. J. Neurochem. 2015, 132, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Narotzki, B.; Reznick, A.Z.; Mitki, T.; Aizenbud, D.; Levy, Y. Green tea drinking improves erythrocytes and saliva oxidative status in the elderly. Adv. Exp. Med. Biol. 2015, 832, 25–33. [Google Scholar] [PubMed]
- Zhao, C.; Li, C.; Liu, S.; Yang, L. The galloyl catechins contributing to main antioxidant capacity of tea made from Camellia sinensis in China. Sci. World J. 2014, 2014, 1–11. [Google Scholar]
- Erba, D.; Riso, P.; Bordoni, A.; Foti, P.; Biagi, P.L.; Testolin, G. Effectiveness of moderate green tea consumption on antioxidative status and plasma lipid profile in humans. J. Nutr. Biochem. 2005, 16, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Kuriyama, S.; Shimazu, T.; Ohmori, K.; Kikuchi, N.; Nakaya, N.; Nishino, Y.; Tsubono, Y.; Tsuji, I. Green tea consumption and mortality due to cardiovascular disease, cancer, and all causes in Japan: The Ohsaki study. JAMA 2006, 296, 1255–1265. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, S.K.; O’Keefe, J.H.; Lavie, C.J. Coffee and tea: Perks for health and longevity? Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 688–697. [Google Scholar] [CrossRef] [PubMed]
- Mineharu, Y.; Koizumi, A.; Wada, Y.; Iso, H.; Watanabe, Y.; Date, C.; Yamamoto, A.; Kikuchi, S.; Inaba, Y.; Toyoshima, H.; et al. Coffee, green tea, black tea and oolong tea consumption and risk of mortality from cardiovascular disease in Japanese men and women. J. Epidemiol. Community Health 2011, 65, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.M.; Daglia, M.; Moghaddam, A.H.; Nabavi, S.F.; Curti, V. Tea consumption and risk of ischemic stroke: A brief review of the literature. Curr. Pharm. Biotechnol. 2014, 15, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, E.; Yorifuji, T.; Takao, S.; Komatsu, H.; Sugiyama, M.; Ohta, T.; Ishikawa-Takata, K.; Doi, H. Green tea consumption and mortality among Japanese elderly people: The prospective Shizuoka elderly cohort. Ann. Epidemiol. 2009, 19, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Sano, J.; Inami, S.; Seimiya, K.; Ohba, T.; Sakai, S.; Takano, T.; Mizuno, K. Effects of green tea intake on the development of coronary artery disease. Circ. J. 2004, 68, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Tokunaga, S.; White, I.R.; Frost, C.; Tanaka, K.; Kono, S.; Tokudome, S.; Akamatsu, T.; Moriyama, T.; Zakouji, H. Green tea consumption and serum lipids and lipoproteins in a population of healthy workers in Japan. Ann. Epidemiol. 2002, 12, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Mielgo-Ayuso, J.; Barrenechea, L.; Alcorta, P.; Larrarte, E.; Margareto, J.; Labayen, I. Effects of dietary supplementation with epigallocatechin-3-gallate on weight loss, energy homeostasis, cardiometabolic risk factors and liver function in obese women: Randomised, double-blind, placebo-controlled clinical trial. Br. J. Nutr. 2014, 111, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, Y.; Suzuki, T.; Mochizuki, K.; Goda, T. Dietary supplementation with (−)-epigallocatechin-3-gallate reduces inflammatory response in adipose tissue of non-obese type 2 diabetic Goto-Kakizaki (Gk) rats. J. Agric. Food Chem. 2013, 61, 11410–11417. [Google Scholar] [CrossRef] [PubMed]
- Nagle, D.G.; Ferreira, D.; Zhou, Y.D. Epigallocatechin-3-gallate (EGCG): Chemical and biomedical perspectives. Phytochemistry 2006, 67, 1849–1855. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.S.; Butt, M.S.; Huma, N.; Sultan, M.T. Green tea catechins based functional drink (Green cool) improves the antioxidant status of SD rats fed on high cholesterol and sucrose diets. Pak. J. Pharm. Sci. 2013, 26, 721–726. [Google Scholar] [PubMed]
- Munir, K.M.; Chandrasekaran, S.; Gao, F.; Quon, M.J. Mechanisms for food polyphenols to ameliorate insulin resistance and endothelial dysfunction: Therapeutic implications for diabetes and its cardiovascular complications. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E679–E686. [Google Scholar] [CrossRef] [PubMed]
- Raederstorff, D.G.; Schlachter, M.F.; Elste, V.; Weber, P. Effect of EGCG on lipid absorption and plasma lipid levels in rats. J. Nutr. Biochem. 2003, 14, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Koo, S.I.; Noh, S.K. Green tea as inhibitor of the intestinal absorption of lipids: Potential mechanism for its lipid-lowering effect. J. Nutr. Biochem. 2007, 18, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.A.; Roach, P.D. A green tea catechin extract upregulates the hepatic low-density lipoprotein receptor in rats. Lipids 2007, 42, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.A.; Roach, P.D. Modulation of cholesterol metabolism by the green tea polyphenol (−)-epigallocatechin gallate in cultured human liver (HepG2) cells. J. Agric. Food. Chem. 2006, 54, 1621–1626. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.A.; Abbey, M.; Roach, P.D. A green tea extract lowers plasma cholesterol by inhibiting cholesterol synthesis and upregulating the LDL receptor in the cholesterol-fed rabbit. Atherosclerosis 2007, 193, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Bursill, C.; Roach, P.D.; Bottema, C.D.; Pal, S. Green tea upregulates the low-density lipoprotein receptor through the sterol-regulated element binding Protein in HepG2 liver cells. J. Agric. Food Chem. 2001, 49, 5639–5645. [Google Scholar] [CrossRef] [PubMed]
- Nantz, M.P.; Rowe, C.A.; Bukowski, J.F.; Percival, S.S. Standardized capsule of Camellia sinensis lowers cardiovascular risk factors in a randomized, double-blind, placebo-controlled study. Nutrition 2009, 25, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A.; Troufflard, S.; Serafini, M.; Crozier, A. Absorption, metabolism and excretion of Choladi green tea flavan-3-ols by humans. Mol. Nutr. Food Res. 2009, 53 (Suppl. S1), S44–S53. [Google Scholar] [PubMed]
- Fu, T.; Liang, J.; Han, G.; Lv, L.; Li, N. Simultaneous determination o fthe major active components of tea polyphenols in rat plasma by a simple and specific HPLC assay. J. Chromatogr. B 2008, 875, 363–367. [Google Scholar] [CrossRef]
- Ullmann, U.; Haller, J.; Bakker, G.C.; Brink, E.J.; Weber, P. Epigallocatechin gallate (EGCG) (TEAVIGO) does not impair nonhaem-iron absorption in man. Phytomedicine 2005, 12, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Ullmann, U.; Haller, J.; Decourt, J.P.; Girault, N.; Girault, J.; Richard-Caudron, A.S.; Pineau, B.; Weber, P. A single ascending dose study of epigallocatechin gallate in healthy volunteers. J. Int. Med. Res. 2003, 31, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Wolfram, S.; Raederstorff, D.; Wang, Y.; Teixeira, S.R.; Elste, V.; Weber, P. TEAVIGO (epigallocatechin gallate) supplementation prevents obesity in rodents by reducing adipose tissue mass. Ann. Nutr. Metab. 2005, 49, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Roach, P.D.; Le, V.H.; Naumovski, N.; Blades, B. Despite its instability, epigallocatechin gallate lowers serum cholesterol in the hypercholesterolaemic rabbit. Atheroscler. Suppl. 2006, 7, 441. [Google Scholar] [CrossRef]
- Wang, R.; Zhou, W. Stability of tea catechins in the breadmaking process. J. Agric. Food Chem. 2004, 52, 8224–8229. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhou, W.; Jiang, X. Reaction kinetics of degradation and epimerization of epigallocatechin gallate (EGCG) in aqueous system over a wide temperature range. J. Agric. Food Chem. 2008, 56, 2694–2701. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhou, W.; Wen, R.A. Kinetic study of the thermal stability of tea catechins in aqueous systems using a microwave reactor. J. Agric. Food Chem. 2006, 54, 5924–5932. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhu, Q.Y.; Tsang, D.; Huang, Y. Degradation of green tea catechins in tea drinks. J. Agric. Food Chem. 2001, 49, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Proniuk, S.; Liederer, B.M.; Blanchard, J. Preformulation study of epigallocatechin gallate, a promising antioxidant for topical skin cancer prevention. J. Pharm. Sci. 2002, 91, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Dube, A.; Ng, K.; Nicolazzo, J.A.; Larson, I. Effective use of reducing agents and nanoparticle encapsulation in stabilizing catechins in alkaline solution. Food Chem. 2010, 122, 662–667. [Google Scholar] [CrossRef]
- Laparra, J.M.; Sanz, Y. Interactions of gut microbiota with functional food components and nutraceuticals. Pharmacol. Res. 2010, 61, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Hirun, S.; Roach, P. A study of stability of (−)-epigallocatechin gallate (EGCG) from green tea in a frozen product. Int. Food Res. J. 2011, 18, 1261–1264. [Google Scholar]
- Green, R.J.; Murphy, A.S.; Schulz, B.; Watkins, B.A.; Ferruzzi, M.G. Common tea formulations modulate in vitro digestive recovery of green tea catechins. Mol. Nutr. Food Res. 2007, 51, 1152–1162. [Google Scholar] [CrossRef] [PubMed]
- Mereles, D.; Hunstein, W. Epigallocatechin-3-gallate (EGCG) for clinical trials: More pitfalls than promises? Int. J.Mol. Sci. 2011, 12, 5592–5603. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Maliakal, P.; Chen, L.; Meng, X.; Bondoc, F.Y.; Prabhu, S.; Lambert, G.; Mohr, S.; Yang, C.S. Pharmacokinetics of tea catechins after ingestion of green tea and (−)-epigallocatechin-3-gallate by humans: Formation of different metabolites and individual variability. Cancer Epidemiol. Biomark. Prev. 2002, 11, 1025–1032. [Google Scholar]
- Chow, H.H.; Cai, Y.; Hakim, I.A.; Crowell, J.A.; Shahi, F.; Brooks, C.A.; Dorr, R.T.; Hara, Y.; Alberts, D.S. Pharmacokinetics and safety of green tea polyphenols after multiple-dose administration of epigallocatechin gallate and polyphenon E in healthy individuals. Clin. Cancer Res. 2003, 9, 3312–3319. [Google Scholar] [PubMed]
- Chow, H.H.; Hakim, I.A.; Vining, D.R.; Crowell, J.A.; Ranger-Moore, J.; Chew, W.M.; Celaya, C.A.; Rodney, S.R.; Hara, Y.; Alberts, D.S. Effects of dosing condition on the oral bioavailability of green tea catechins after single-dose administration of polyphenon E in healthy individuals. Clin. Cancer Res. 2005, 11, 4627–4633. [Google Scholar] [CrossRef] [PubMed]
- Vuong, Q.V.; Golding, J.B.; Nguyen, M.; Roach, P.D. Extraction and isolation of catechins from tea. J. Sep. Sci. 2010, 33, 3415–3428. [Google Scholar] [CrossRef] [PubMed]
- Vuong, Q.V.; Golding, J.B.; Stathopoulos, C.E.; Nguyen, M.H.; Roach, P.D. Optimizing conditions for the extraction of catechins from green tea using hot water. J. Sep. Sci. 2011, 34, 3099–3106. [Google Scholar] [CrossRef] [PubMed]
- FDA. Bioavailability and bioequivalence studies for orally administered drug products–General considerations. In Guidance for Industry, 1st ed.; Food and Drug Administration, Ed.; U.S. Department of Health and Human Services: Rockville, MD, USA, 2003; p. 23. [Google Scholar]
- Maron, D.J.; Lu, G.P.; Cai, N.S.; Wu, Z.G.; Li, Y.H.; Chen, H.; Zhu, J.Q.; Jin, X.J.; Woulters, B.C.; Zhao, J. Cholesterol-lowering effect of a theaflin-enriched green tea extract. Arch. Intern. Med. 2003, 163, 1448–1453. [Google Scholar] [CrossRef] [PubMed]
- Adiyaman, A.; Verhoeff, R.; Lenders, J.W.; Deinum, J.; Thien, T. The position of the arm during blood pressure measurement in sitting position. Blood Pressure Monit. 2006, 11, 309–313. [Google Scholar] [CrossRef]
- Sauter, R.; Steinijans, V.W.; Diletti, E.; Bohm, A.; Schulz, H.U. Presentation of results from bioequivalence studies. Int. J. Clin. Pharm. 1992, 30, 233–256. [Google Scholar]
- Welling, P.G. Effects of food on drug absorption. Pharmacol. Ther. 1989, 43, 425–441. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.P.; Schroeter, H.; Rechner, A.R.; Rice-Evans, C. Bioavailability of flavan-3-ols and procyanidins: Gastrointestinal tract influences and their relevance to bioactive forms in vivo. Antioxid. Redox Signal. 2001, 3, 1023–1039. [Google Scholar] [CrossRef] [PubMed]
- Holladay, J.W. Biofarmaceutics of orally ingested products. In Handbook of Food-Drug Interactions; McCabe, B.J., Wolfe, J.J., Frankel, E.H., Eds.; CRC Press LLC: Boca Raton, FL, USA, 2003. [Google Scholar]
- Record, I.R.; Lane, J.M. Simulated intestinal digestion of green and black teas. Food Chem. 2001, 73, 481–486. [Google Scholar] [CrossRef]
- Fleisher, D.; Li, C.; Zhou, Y.; Pao, L.H.; Karim, A. Drug, meal and formulation interactions influencing drug absorption after oral administration. Clinical implications. Clin. Pharmacokinet. 1999, 36, 233–254. [Google Scholar] [CrossRef]
- Chen, L.; Lee, M.J.; Li, H.; Yang, C.S. Absorption, distribution, elimination of tea polyphenols in rats. Drug Metab. Dispos. 1997, 25, 1045–1050. [Google Scholar] [PubMed]
- Spencer, J.P. Metabolism of tea flavonoids in the gastrointestinal tract. J. Nutr. 2003, 133, 3255S–3261S. [Google Scholar] [PubMed]
- Ru, Q.; Yu, H.; Huang, Q. Encapsulation of epigallocatechin-3-gallate (EGCG) using oil-in-water (O/W) submicrometer emulsions stabilized by ι-carrageenan and β-lactoglobulin. J. Agric. Food Chem. 2010, 58, 10373–10381. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.Y.; Hwang, T.L.; Huang, Y.L.; Fang, C.L. Enhancement of the transdermal delivery of catechins by liposomes incorporating anionic surfactants and ethanol. Int. J. Pharm. 2006, 310, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Shutava, T.G.; Balkundi, S.S.; Lvov, Y.M. (−)-Epigallocatechin gallate/gelatin layer-by-layer assembled films and microcapsules. J. Colloid Interface Sci. 2009, 330, 276–283. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naumovski, N.; Blades, B.L.; Roach, P.D. Food Inhibits the Oral Bioavailability of the Major Green Tea Antioxidant Epigallocatechin Gallate in Humans. Antioxidants 2015, 4, 373-393. https://doi.org/10.3390/antiox4020373
Naumovski N, Blades BL, Roach PD. Food Inhibits the Oral Bioavailability of the Major Green Tea Antioxidant Epigallocatechin Gallate in Humans. Antioxidants. 2015; 4(2):373-393. https://doi.org/10.3390/antiox4020373
Chicago/Turabian StyleNaumovski, Nenad, Barbara L. Blades, and Paul D. Roach. 2015. "Food Inhibits the Oral Bioavailability of the Major Green Tea Antioxidant Epigallocatechin Gallate in Humans" Antioxidants 4, no. 2: 373-393. https://doi.org/10.3390/antiox4020373
APA StyleNaumovski, N., Blades, B. L., & Roach, P. D. (2015). Food Inhibits the Oral Bioavailability of the Major Green Tea Antioxidant Epigallocatechin Gallate in Humans. Antioxidants, 4(2), 373-393. https://doi.org/10.3390/antiox4020373







