Quercetin, a Flavonoid Antioxidant, Ameliorated Procarbazine-Induced Oxidative Damage to Murine Tissues
Abstract
:1. Introduction
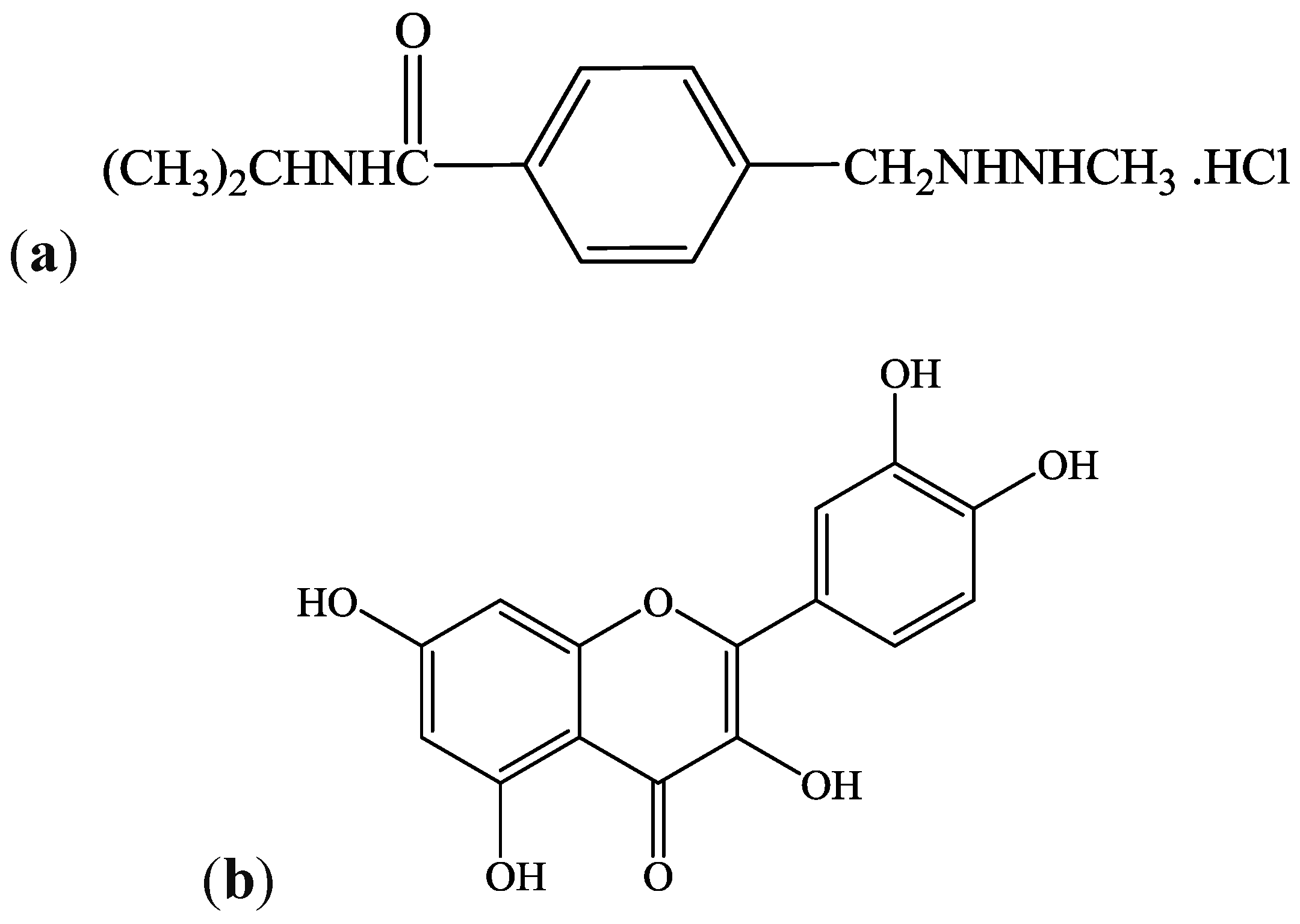
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Animal Selection and Care
2.3. Animal Grouping and Drug Treatments
| Treatment—Groups | Treatments—Duration | |
|---|---|---|
| Day 1–7 | Day 8–14 | |
| I (CTRL) | - | Control; distilled water |
| II (PCZ) | - | 2 mg/kg bw PCZ |
| III (PCZ + QCT-P) | 20 mg/kg bw QCT | 2 mg/kg bw PCZ |
| IV (PCZ + QCT-C) | - | 2 mg/kg bw PCZ + 20 mg/kg bw QCT |
| V (QCT-A) | - | 20 mg/kg bw QCT |
2.4. Animal Sacrifice, Collection of Blood and Liver Samples
2.5. Preparation of Plasma and Cytosolic Fractions
2.6. Determination of Plasma and Liver Protein Content
2.7. Assay of Biomarkers of Nephrotoxicity
2.8. Assay of Biomarkers of Hepatotoxicity
2.9. Assay for Non-Enzymatic Antioxidants in the Liver
2.9.1. Hepatic Reduced Glutathione Level (GSH)
2.9.2. Hepatic Ascorbic Acid Level (AA)
2.10. Assay of Hepatic Antioxidant Enzymes
2.10.1. Hepatic Glutathione S-Transferase (GST) Activity
2.10.2. Hepatic Superoxide Dismutase (SOD) Activity
2.10.3. Hepatic Catalase Activity
2.11. Assay of Hepatic Level of Lipid Peroxidation
2.12. Statistical Analysis
3. Results
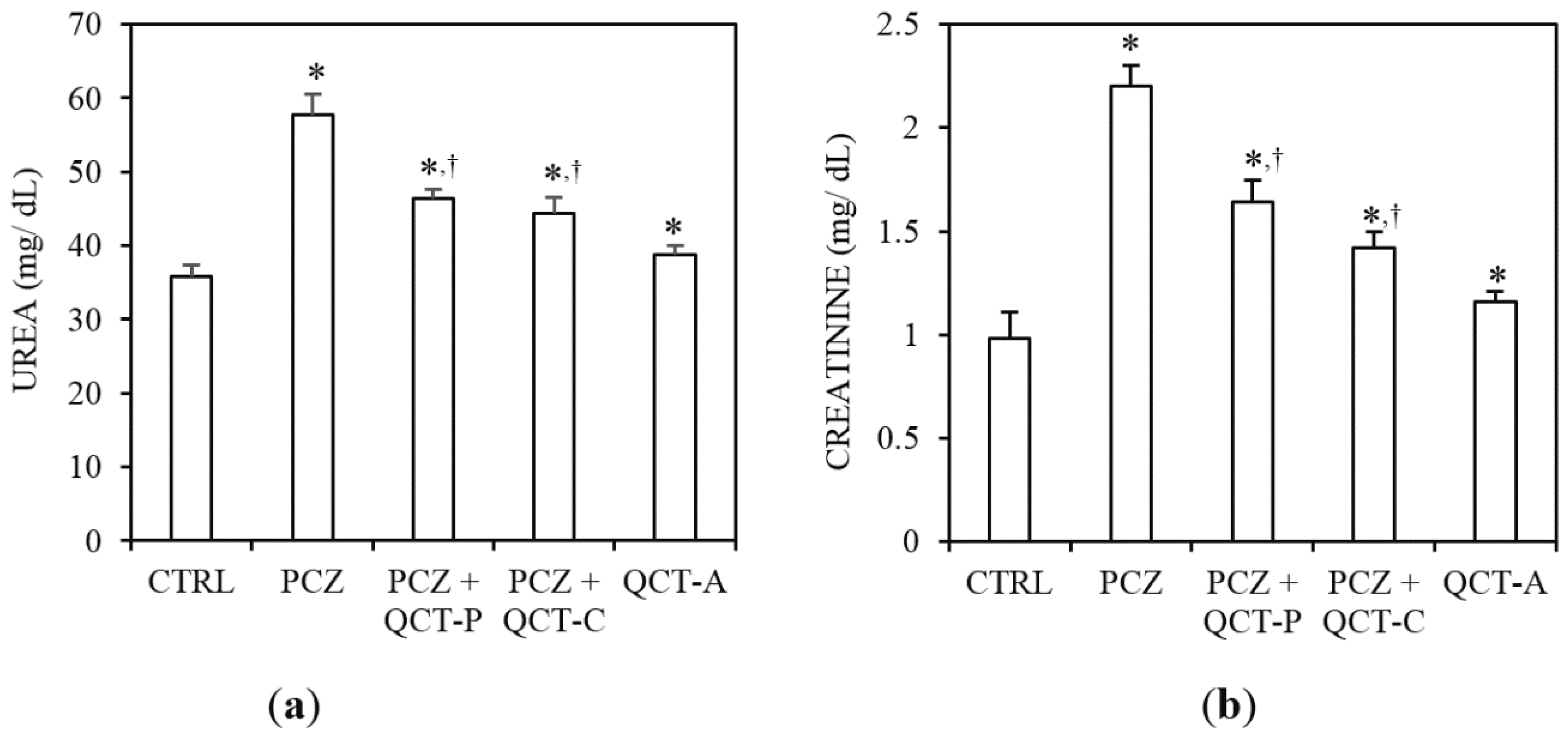
3.1. QCT Pre-Treatment and Co-Treatment Protects against PCZ-Induced Nephrotoxicity in Rats
3.2. QCT Pre-Treatment and Co-Treatment Protects against PCZ-Induced Hepatotoxicity in Rats

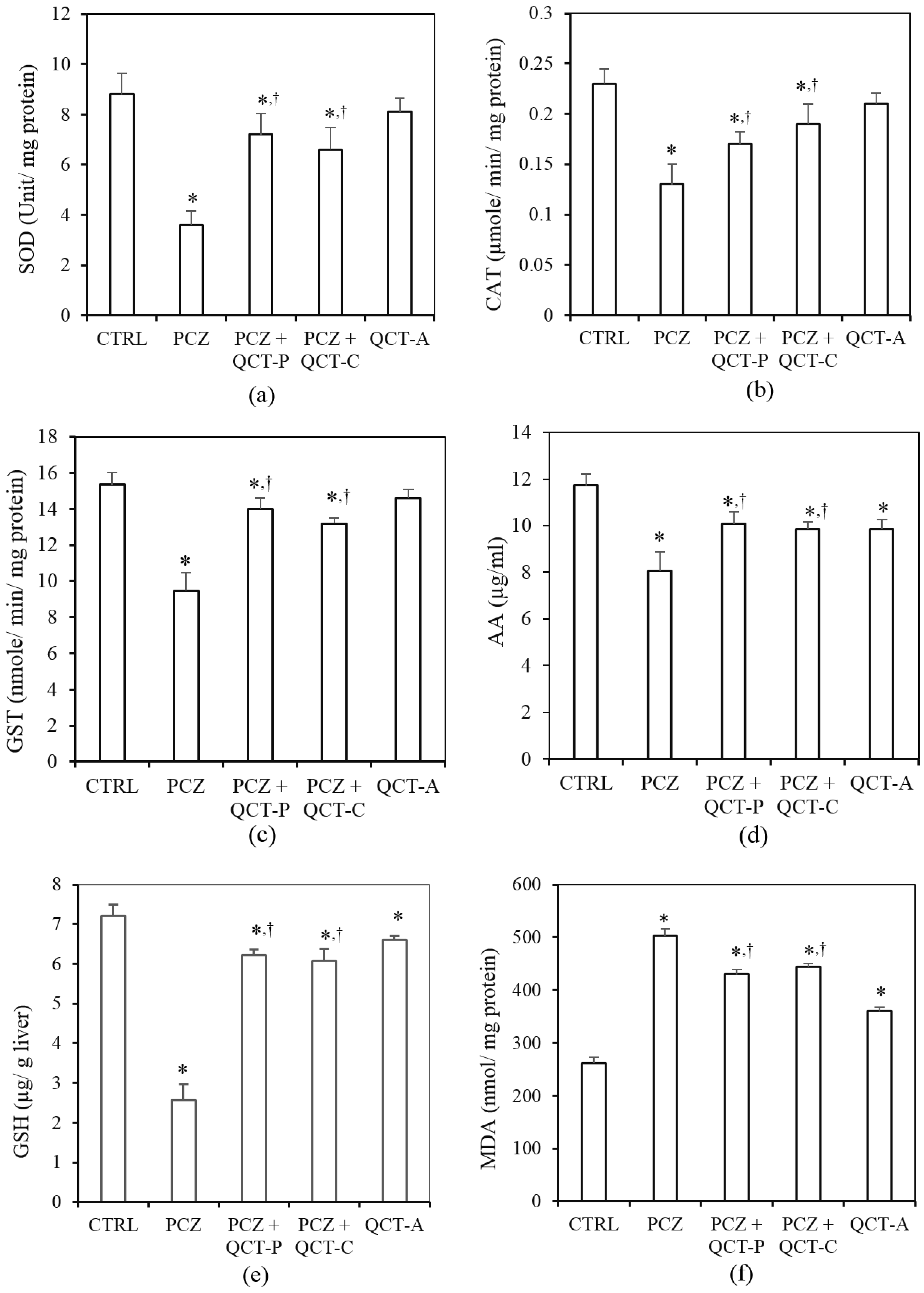
3.3. QCT Pre-Treatment and Co-Treatment Protects against PCZ-Induced Oxidative Stress in the Liver of Rats
4. Discussion
5. Conclusions
Abbreviations
| PCZ | Procarbazine (2 mg/ kg bw) |
| bw | Body weight |
| CTRL | Control |
| QCT | Quercetin |
| QCT-P | Quercetin pre-treated |
| QCT-C | Quercetin co-treated |
| QCT-A | Quercetin-alone |
| PMF | Post Mitochondrial Fraction |
| BSA | Bovine Serum Albumin |
| ALP | Alkaline phosphatase |
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| γ-GT | Gamma glutamyl transferase |
| SOD | Superoxide dismutase |
| CAT | Catalase |
| GST | Glutathione-S-transferase |
| AA | Ascorbic acid |
| GSH | Reduced Glutathione |
| LPO | Lipid peroxidation |
| MDA | Malondialdehyde |
| ROS | reactive oxygen species |
| H2O2 | hydrogen peroxide |
Author Contributions
Conflicts of Interest
References
- Fesler, M.J.; Becpker-Koepke, S.; Di Biscegilie, A.M.; Petruska, P.J. Procarbazine-induced hepatotoxicity: Case report and review of the literature. Pharmacotherapy 2010, 30, 540–544. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Antona, C.; Ingelman-Sundberg, M. Cytochrome P450 pharmacogenetics and cancer. Oncogene 2006, 25, 1679–1691. [Google Scholar] [CrossRef] [PubMed]
- Dunn, D.L.; Lubet, R.A.; Prough, R.A. Oxidative Metabolism of N-lsopropyl-α-(2-methylhydrazino)-Ptoluamide Hydrochloride (Procarbazine) by Rat Liver Microsomes. Cancer Res. 1979, 39, 4555–4563. [Google Scholar] [PubMed]
- Swaffar, D.S.; Horstman, M.G.; Jaw, J.; Thrall, B.D.; Meadows, G.G.; Harker, W.G.; Yost, G.S. Methylazoxyprocarbazine, the Active Metabolite Responsible for the Anticancer Activity of Procarbazine against LI 210 Leukemia. Cancer Res. 1989, 49, 2442–2447. [Google Scholar] [PubMed]
- Sinha, B.K. Metabolic activation of procarbazine: Evidence for carbin-centered free radical intermediates. Biochem. Pharmacol. 1984, 33, 2777–2781. [Google Scholar] [CrossRef] [PubMed]
- Goria-Gatti, L.; Iannone, A.; Poli, G.; Albano, E. In vitro and in vivo evidence for the formation of methyl radical from procarbazine: A spin-trapping study. Carcinogenesis 1992, 13, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Sinha, B.K.; Mason, R.P. Biotransformation of Hydrazine Dervatives in the Mechanism of Toxicity. J. Drug Metab. Toxicol. 2014, 5, 168–171. [Google Scholar]
- Wang, J.; Yi, J. Cancer cell killing via ROS: To increase or decrease, that is the question. Cancer Biol. Ther. 2008, 7, 1875–1884. [Google Scholar] [CrossRef] [PubMed]
- Olayinka, E.T.; Ore, A.; Ola, O.S.; Adeyemo, O.A. Protective Effect of Quercetin on Melphalan-Induced Oxidative Stress and Impaired Renal and Hepatic Functions in Rat. Chemother. Res. Prac. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Fruehauf, J.P.; Meyskens, F.L., Jr. Reactive oxygen species: A breath of life or death? Clin. Cancer Res. 2007, 13, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Sikic, B.I. Antineoplastic agents. In Mordern Pharmacology with Clinical Applications, 6th ed.; Craig, R., Stitzel, R.E., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2004; p. 651. [Google Scholar]
- Price, P.; Thompson, H.; Bessell, E.M.; Bloom, H.J. Renal Impairment following the combined use of high-dose methotrexate and procarbazine. Cancer Chemother. Pharmacol. 1988, 21, 265–267. [Google Scholar] [CrossRef] [PubMed]
- McMaster, K.R.; Hennigar, G.R. Drug-induced granulomatous hepatitis. Lab. Investig. 1981, 44, 61–73. [Google Scholar] [PubMed]
- King, P.D.; Perry, M.C. Hepatotoxicity of Chemotherapy. The Oncol. 2001, 6, 162–176. [Google Scholar] [CrossRef]
- Yost, G.S.; Horstman, M.G.; El Walily, A.F.; Gordon, W.P.; Nelson, S.D. Procarbazine spermatogenesis toxicity: Deuterium isotope effect point to regioselective metabolism in mice. Toxicol. Appl. Pharmacol. 1985, 80, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Alp, B.F.; Kesik, V.; Malkoc, E.; Yigit, N.; Saldir, M.; Babacan, O.; Akgul, E.O.; Poyrazoglu, Y.; Korkmazer, N.; Gulgun, M.; Erdem, O. The effect of melatonine on procarbazine induced testicular toxicity on rats. Syst. Biol. Reprod. Med. 2014, 60, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress induced cancer. Chem.Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Sikder, K.; Das, N.; Kesh, S.B.; Dey, S. Quercetin and B-sitosterol prevents high fat diet induced dyslipidemia and hepatotoxicity in swiss albino mice. Indian J. Exp. Biol. 2014, 52, 60–66. [Google Scholar] [PubMed]
- Magalingam, K.B.; Radhakrishnan, A.; Haleagrahara, N. Protective effects of flavonol isoquercitrin, against 6-hydroxy dopamine (6-OHDA)-induced toxicity in PC12 cells. BMC Res. Notes 2014, 7, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Kinker, B.; Comstock, A.T.; Sajjan, U.S. Quercetin: A Promising Treatment for the Common Cold. J. Anc. Dis. Prev. Remedies 2014, 2, 1–3. [Google Scholar]
- Maalik, A.; Khan, F.A.; Mumtaz, A.; Mehmood, A.; Azhar, S.; Atif, M.; Karim, S.; Altaf, Y.; Tariq, I. Pharmacological Applications of Quercetin and Its Derivatives: A Short Review. Trop. J. Pharm. Res. 2014, 13, 1561–1566. [Google Scholar] [CrossRef]
- Ahmad, W.; Shaikh, S.; Nazam, N.; Lone, M.I. Protective Effects of Quercetin against Dimethoate-Induced Cytotoxicity and Genotoxicity in Allium sativum Test. Int. Sch. Res. Not. 2014, 2014. [Google Scholar] [CrossRef]
- Qader, G.I.; Aziz, R.S.; Ahmed, Z.A.; Abdullah, Z.F.; Hussain, S.A. Protective Effects of Quercetin against Isoniazid and Rifampicin Induced Hepatotoxicity in Rats. Am. J. Pharmacol. Sci. 2014, 2, 56–60. [Google Scholar]
- Takashima, K.; Matsushima, M.; Hashimoto, K.; Nose, H.; Sato, M.; Hashimoto, N.; Hasegawa, Y.; Kawabe, T. Protective effects of intratracheally administered quercetin on lipopolysaccharide-induced acute lung injury. Respir. Res. 2014, 15, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Wang, J.; Feng, D.; Qin, H.; Wen, H.; Yin, Z.; Gao, G.; Li, C. Protective Effect of Quercetin against Oxidative Stress and Brain Edema in an Experimental Rat Model of Subarachnoid Hemorrhage. Int. J. Med. Sci. 2014, 11, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Charles, B.; Simone, M.D., II; Nicole, L.; Simone, M.D.; Victoria Simone, R.N.; Charles, B.; Simone, M.D. Antioxidants and other nutrients do not interfere with chemotherapy or radiation therapy and can increase kill and increase Survival, Part 1. Altern. Ther. 2007, 13, 22–28. [Google Scholar]
- Sanchez-Gonzalez, P.D.; Lopez-Hernandez, F.J.; Perez-Barriocana, F.; Morales, A.I.; Lopez-Novoa, M. Quercetin reduces cisplatin nephrotoxicity in rats without compromising its anti-tumour activity. Nephrol. Dial. Transplant. 2011, 26, 3484–3495. [Google Scholar] [CrossRef] [PubMed]
- Sak, K. Chemotherapy and dietary phytochemical agents. Chemother. Res. Prac. 2012, 2012, 1–11. [Google Scholar] [CrossRef]
- Souliotis, V.L.; Valavanis, C.; Boussiotis, V.A.; Pangalis, G.A.; Kyrtopoulos, S.A. Comparative dosimetry of O6-methylguanine in humans and rodents treated with procarbazine. Carcinogenesis 1994, 15, 1675–1680. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Guide for the Care and Use of Laboratory Animals, 8th ed.; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Krishnappa, P.; Venkatarangaiah, K.; Kumar, V.S.; Rajanna, S.; Kashi, R.; Gupta, P. Antioxidant and prophylactic effects of Delonix elata L., stem bark extracts, and flavonoid isolated quercetin against carbon tetrachloride-induced hepatotoxicity in rats. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Gornall, A.C.; Bardwawill, C.J.; David, M.M. Determination of serum protein by means of the biuret reaction. J. Biol. Chem. 1949, 177, 751–756. [Google Scholar] [PubMed]
- Jaffe, E.R. Oxidative Hemolysis, or What Made the Red Cell Break? N. Engl. J. Med. 1972, 286, 156–157. [Google Scholar] [CrossRef] [PubMed]
- Tietz, N.W. Clinical Guide to Laboratory Tests, 3rd ed.; W.B. Saunders Company: Philadelphia, PA, USA, 1995. [Google Scholar]
- Tietz, N.W.; Pruden, E.L.; Siggaard-Andersen, O. Liver function. In Tietz Textbook of Clinical Chemistry; Burtis, A.C., Ashwood, E.R., Eds.; WB Saunders: London, UK, 1994; pp. 1354–1374. [Google Scholar]
- Reltman, S.; Frankel, S.A. Colorimetric method for the determination of serum ALT and AST. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar] [PubMed]
- Szasz, G. A kinetic photometric method for serum γ-glutamyl transpeptidase. Clin Chem. 1969, 15, 124–136. [Google Scholar] [PubMed]
- Jollow, D.J.; Mitchell, J.R.; Zampaghone, N.; Gillete, J.R. Bromobenzene induced liver necrosis, protective role of glutathione and evidence for 3,4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacology 1974, 11, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Jagota, S.K.; Dani, H.M. A new colorimetric technique for the estimation of vitamin C using Folin phenol reagent. Anal. Biochem. 1982, 127, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Habig, W.A.; Pabst, M.J.; Jacoby, W.B. Glutathione trsansferases. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [PubMed]
- Misra, H.P.; Fridovich, I. The role of superoxide anion in the auto-oxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 1972, 247, 3170–3175. [Google Scholar] [PubMed]
- Singha, A.K. Colorimetric assay of catalase. Anal. Biochem. 1972, 47, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Varshney, R.; Kale, R.K. Effect of calmodulin antagonist on radiation induced lipid peroxidation in microsomes. Int. J. Radiat. Biol. 1990, 58, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Alrawaiq, N.S.; Abdullah, A. A Review of Flavonoid Quercetin: Metabolism, Bioactivity and Antioxidant Properties. Int. J. PharmTech Res. 2014, 6, 933–941. [Google Scholar]
- Kanadaswami, C.; Lee, L.; Lee, P.H.; Hwang, J.; Ke, F.; Huang, Y.; Lee, M. The Antitumor Activities of Flavonoids. In vivo 2005, 19, 895–910. [Google Scholar] [PubMed]
- Zhang, H.; Zhang, M.; Yu, L.; Zhao, Y.; He, N.; Yang, X. Antitumor activities of quercetin and quercetin-5′,8-disulfonate in human colon and breast cancer cell lines. Food Chem. Toxicol. 2012, 50, 1589–1599. [Google Scholar] [CrossRef] [PubMed]
- Priya, E.S.; Selvakumar, K.; Bavithra, S.; Elumalai, P.; Arunkumar, R.; Singh, P.R.; Mercy, A.B.; Arunakaran, J. Anti-cancer activity of quercetin in neuroblastoma: An in vitro approach. Neurol. Sci. 2014, 35, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, M.J.; Fiebig, J.; Winterhalter, H.H.; Berger, B.R.; Grunicke, H. Enhancement of the antiproliferative activity of cis-diamminedichloroplatinum (II) by quercetin. Int. J. Cancer 1990, 45, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Kang, J.H. Quercetin and trichostatin A cooperatively kill human leukemia cells. Pharmazie 2005, 60, 856–860. [Google Scholar] [PubMed]
- Chan, S.; Yang, N.; Huang, C.; Liao, J.; Yeh, S. Quercetin Enhances the Antitumor Activity of Trichostatin a through Upregulation of p53 Protein Expression In Vitro and In Vivo. PLoS ONE 2013, 8, 1–10. [Google Scholar] [CrossRef]
- Samuel, T.; Fadlalla, K.; Mosley, L.; Katkoori, V.; Turner, T.; Manne, U. Dual-Mode Interaction between Quercetin and DNA-Damaging Drugs in Cancer Cells. Anticancer Res. 2012, 32, 61–71. [Google Scholar] [PubMed]
- Ferguson, M.A.; Waikar, S.S. Established and Emerging Markers of Kidney Function. Clin. Chem. 2012, 58, 4–14. [Google Scholar] [CrossRef]
- Simsek, A.; Tugcu, V.; Tasci, A.I. New Biomarkers for the Quick Detection of Acute Kidney Injury. ISRN Nephrology 2013, 2013. [Google Scholar] [CrossRef]
- George, G.S.; Wakasi, M.E.; Egoro, E. Creatinine and urea levels as critical markers in end-stage renal failure. Res. Rev.: J. Med. Health Sci. 2014, 3, 41–44. [Google Scholar]
- Behling, E.B.; Sendao, M.C.; Francescato, H.D.; Antunes, L.M.; Costa, R.S.; Bianchi Mde, L. Comparative study of multiple dosage of quercetin against cisplatin-induced nephrotoxicity and oxidative stress in rat kidneys. Pharmacol. Rep. 2006, 58, 526–532. [Google Scholar] [PubMed]
- Francescato, H.D.; Coimbra, T.M.; Costa, R.S.; Bianchi Mde, L. Protective effect of quercetin on the evolution of cisplatin-induced acute tubular necrosis. Kidney Blood Press Res. 2004, 27, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Boone, L.; Meyer, D.; Cusick, P.; Ennulat, D.; Bolliger, A.P.; Everds, N.; Meador, V.; Elliott, G.; Honor, D.; Bounous, D.; et al. Selection and interpretation of clinical pathology indicators of hepatic injury in preclinical studies. Vet. Clin. Pathol. 2005, 34, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Bhat, T.K.; Sharma, O.P. Clinical Biochemistry of Hepatotoxicity. J. Clin. Toxicol. 2011. [Google Scholar] [CrossRef]
- Ramaiah, S.K. A toxicologist guide to the diagnostic interpretation of hepatic biochemical parameters. Food Chem. Toxicol. 2007, 45, 1551–1557. [Google Scholar] [CrossRef] [PubMed]
- Olayinka, E.T.; Ore, A.; Fashiku, K.A. Kolaviron and l-Ascorbic acid Ameliorates Chlorambucil-Induced Hepatic and Renal Toxicity in Rat. Int. J. Toxicol. Appl. Pharmacol. 2014, 4, 23–32. [Google Scholar]
- Amacher, D.E. Serum Transaminase Elevations as Indicators of Hepatic Injury Following the Administration of Drugs. Regul. Toxicol. Pharmacol. 1998, 27, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Hozayen, W.G.; Abou-Seif, H.S.; Amin, S. Protective Effects of Ruitn and/or Hesperidin against Doxorubicin-Induced Hepatotoxicity. Int. J. Clin. Nutr. 2014, 2, 11–17. [Google Scholar]
- Yamamoto, T.; Kikkawa, R.; Yamada, H.; Horii, I. Identification of oxidative stress-related proteins for predictive screening of hepatotoxicity using a proteomic approach. J. Toxicol. Sci. 2005, 30, 213–227. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.M.; Jung, Y.S.; Jeon, B.S.; Yoon, B.I.; Lee, K.S.; Kim, B.H.; Oh, S.J.; Kim, S.K. Evaluation of hepatotoxicity and oxidative stress in rats treated with tert-butyl hydroperoxide. Food Chem. Toxicol. 2012, 50, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncola, J.; Cronin, M.T.D.; Mazura, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Sherratt, P.J.; Hayes, J.D. Glutathione S-transferases. In Enzyme Systems that Metabolise Drugs and other Xenobiotics; Ioannides, C., Ed.; John Wiley & Sons: Chichester, UK, 2002; pp. 219–252. [Google Scholar]
- Morgenstern, R.; Zhang, J.; Johansson, K. Microsomal glutathione transferase: Mechanism and functional roles. Drug Metab. Rev. 2011, 43, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Devasagayam, T.P.A.; Tilak, J.C.; Boloor, K.K.; Sane, K.S.; Ghaskadbi, S.S.; Lele, R.D. Free Radicals and Antioxidants in Human Health: Current Status and Future Prospects. JAPI 2004, 52, 794–804. [Google Scholar] [PubMed]
- Kojo, S. Vitamin C: Basic metabolism and its function as an index of oxidative stress. Curr. Med. Chem. 2004, 11, 1041–1064. [Google Scholar] [CrossRef] [PubMed]
- Masella, R.; di Benedetto, R.; Var, R.; Filesi, C.; Giovannini, C. Novel mechanisms of natural antioxidant compounds in biological systems: Involvement of glutathione and glutathionerelated enzymes. J. Nutr. Biochem. 2005, 16, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Gautam, A.; Vijayaraghavan, R.; Pant, S.C.; Kumar, O.; Singh, S.; Kumar, H.T.S. Protective Effect of Quercetin against Sulphur Mustard-Induced Oxidative Stress in Mice. Def. Sci. J. 2007, 57, 707–720. [Google Scholar] [CrossRef]
- Hollman, P.C.H.; Trip, J.M.P.V.; Baysman, M.N.C.P.; Cagg, M.S.V.D.; Mengelers, M.J.B.; Vries, J.H.M.D.; Katan, M.B. Relative bioavailability of the antioxidant flavonoid quercetin from various food in man. FEBS Lett. 1997, 418, 152–156. [Google Scholar] [CrossRef] [PubMed]
- El-Beltagi, H.S.; Mohamed, H.I. Reactive Oxygen Species, Lipid Peroxidation and Antioxidative Defense Mechanism. Not. Bot. Horti Agrobo. 2013, 41, 44–57. [Google Scholar]
- Guéraud, F.; Atalay, M.; Bresgen, N.; Cipak, A.; Eckl, P.M.; Huc, L.; Jouanin, I.; Siems, W.; Uchida, K. Chemistry and biochemistry of lipid peroxidation products. Free Rad. Res. 2010, 44, 1098–1124. [Google Scholar] [CrossRef]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxid. Med. Cell. Longev. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Catalá, A. Lipid peroxidation modifies the assembly of biological membranes “The Lipid Whisker Model”. Front. Physiol. 2015, 5. [Google Scholar] [CrossRef]
- Molina, M.F.; Sanchez-Reus, I.; Iglesias, I.; Benedi, J. Quercetin, a flavonoid antioxidant, prevents and protects against ethanol-induced oxidative stress in mouse liver. Biol. Pharm. Bull. 2003, 26, 1398–1402. [Google Scholar] [CrossRef] [PubMed]
- Mendes, V.; Costa, V.; Mateus, N. Involvement of the modulation of cancer cell redox status in the anti-tumoral effect of phenolic compounds. RSC Adv. 2015, 5, 1–9. [Google Scholar] [CrossRef]
- Jimenez, R.; Lopez-Sepulveda, R.; Romero, M.; Toral, M.; Cogolludo, A.; Perez-Vizcaino, F.; Duarte, J. Quercetin and its metabolites inhibit the membrane NADPH oxidase activity in vascular smooth muscle cells from normotensive and spontaneously hypertensive rats. Food Funct. 2015, 6, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Verstraeten, S.V.; Frag, C.G.; Oteiza, P.I. Interactions of flavan-3-ols and procyanidins with membranes: Mechanisms and the physiological relevance. Food Funct. 2015, 6, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, Y.; Yang, L.; Zu, Y.; Lu, Q. Effects of rosmarinic acid on liver and kidney antioxidant enzymes, lipid peroxidation and tissue ultrastructure in aging mice. Food Funct. 2015, 6, 927–731. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Rajanikant, G.K. Curcumin Stimulates the Antioxidant Mechanisms in Mouse Skin Exposed to Fractionated γ-Irradiation. Antioxidants 2015, 4, 25–41. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olayinka, E.T.; Ore, A.; Adeyemo, O.A.; Ola, O.S.; Olotu, O.O.; Echebiri, R.C. Quercetin, a Flavonoid Antioxidant, Ameliorated Procarbazine-Induced Oxidative Damage to Murine Tissues. Antioxidants 2015, 4, 304-321. https://doi.org/10.3390/antiox4020304
Olayinka ET, Ore A, Adeyemo OA, Ola OS, Olotu OO, Echebiri RC. Quercetin, a Flavonoid Antioxidant, Ameliorated Procarbazine-Induced Oxidative Damage to Murine Tissues. Antioxidants. 2015; 4(2):304-321. https://doi.org/10.3390/antiox4020304
Chicago/Turabian StyleOlayinka, Ebenezer Tunde, Ayokanmi Ore, Oluwatobi Adewumi Adeyemo, Olaniyi Solomon Ola, Olaoluwa Oluwaseun Olotu, and Roseline Chinonye Echebiri. 2015. "Quercetin, a Flavonoid Antioxidant, Ameliorated Procarbazine-Induced Oxidative Damage to Murine Tissues" Antioxidants 4, no. 2: 304-321. https://doi.org/10.3390/antiox4020304
APA StyleOlayinka, E. T., Ore, A., Adeyemo, O. A., Ola, O. S., Olotu, O. O., & Echebiri, R. C. (2015). Quercetin, a Flavonoid Antioxidant, Ameliorated Procarbazine-Induced Oxidative Damage to Murine Tissues. Antioxidants, 4(2), 304-321. https://doi.org/10.3390/antiox4020304





