Flavonoids Affect Host-Microbiota Crosstalk through TLR Modulation
Abstract
:1. Host-Microbiota Crosstalk and Dietary Factors
1.1. Interaction between Microbiota and the Host
1.2. Toll-Like Receptors
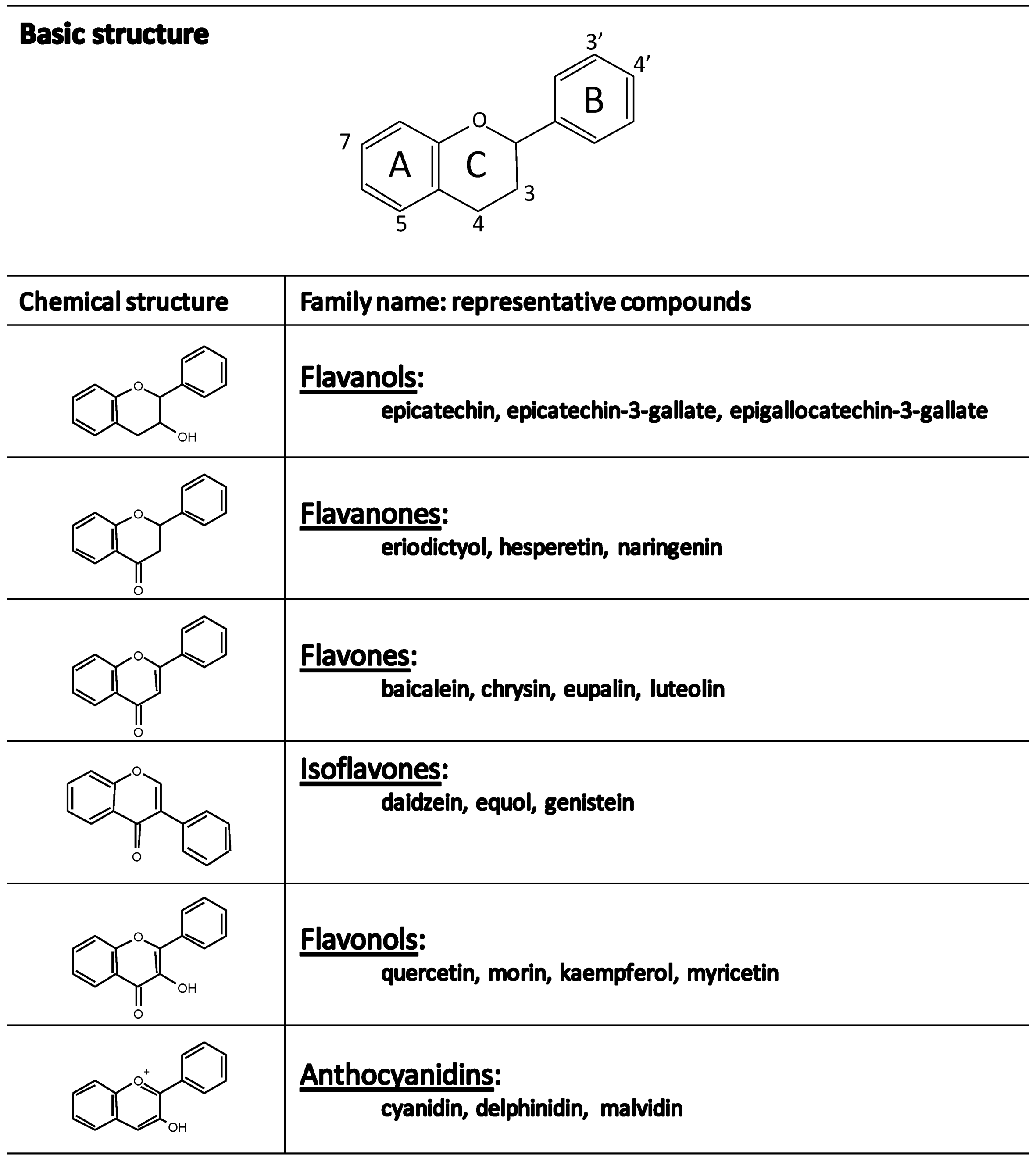
1.3. Flavonoids
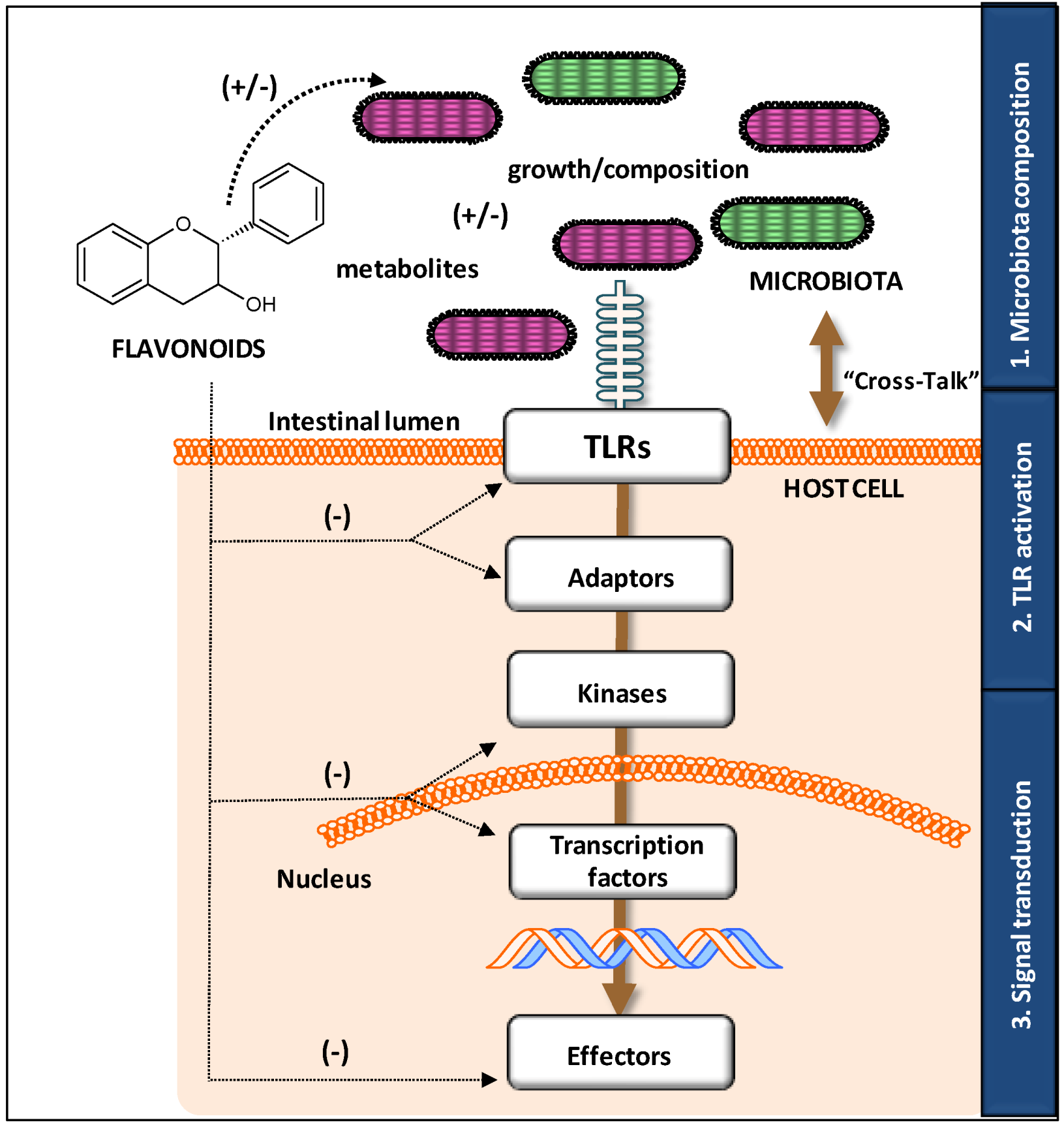
2. Objective
3. Mechanisms of Modifying “Cross-Talk” by Flavonoids
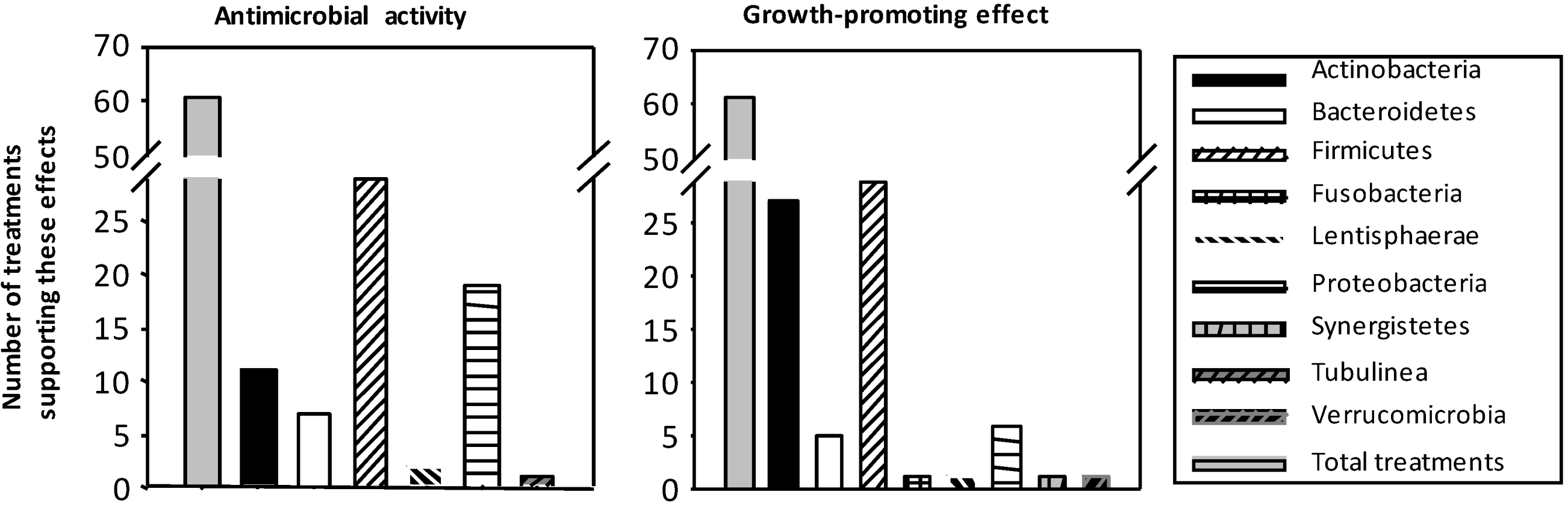
3.1. Influence on Growth and Composition of Microbiota
3.2. Modulatory Action on TLR Gene and Protein Expression
| Flavonoid | Dose | Duration 1 | TLR 2 | Studied Target | Cell (Challenge) | Reference |
|---|---|---|---|---|---|---|
| FLAVONOLS Engeletin | 10/50 μM | 2 h | ↓ TLR4 | mRNA | J774 macrophages (LPS) | [53] |
| Astilbin | ↓ TLR4 | mRNA | ||||
| Kaempferol-3-O-sophoroside | – | 6 h | ↓ TLR2 | Protein | Endothelial cells | [54] |
| ↓ TLR4 | ||||||
| Quercetin | 25 μM | 1 h | ↓ TLR2 | Protein | PBMC | [55] |
| ↓ TLR4 | ||||||
| FLAVANONES Naringenin | 1 μg/mL | 48 h | ↓ TLR2 | Protein/mRNA | J774 macrophages (C. trachomatis) | [56] |
| ↓ TLR4 | mRNA | |||||
| Naringenin | 100 μM | – | ↓ TLR2 | mRNA | 3T3L1 cells (adipocyte differentiation) | [57] |
| FLAVANOLS Epigallocatechin-3-gallate | 1 μM | 24 h | ↓ TLR4 | Protein/mRNA | RAW 264.7 macrophages (LPS) | [58] |
| Epigallocatechin-3-gallate | 10 μM | 24 h | ↔ TLR4 | mRNA | Murine bone marrow-derived DCs (unstimulated) | [59] |
| FLAVONES Baicalin | 40/80 μM | 24 + 3 h | ↔ TLR1-9 | mRNA | Human oral keraynocytes (LPS) | [60] |
| Baicalin | 5/10 μg/mL | 0.5–6 h | ↓ TLR2 | Protein/mRNA | PC12 and primary rat neurons (oxygen glucose deprivation) | [61] |
| ↓ TLR4 | Protein/mRNA |
| Flavonoid | Dose | Duration | TLR 1,2 | Studied Target | Cell | Reference |
|---|---|---|---|---|---|---|
| FLAVONES Luteolin | Daily dose of 10 and 25 mg/kg body weight | 24/78 h | ↓ TLR4 | Protein/mRNA | Cerebral cortex from. p.o 3 fed SD rats | [62] |
| ↓ TLR5 | ||||||
| Baicalin | One dose of 50 mg/kg body weight | 4 h | ↓ TLR2 | Protein/ | Mice hippocampus cells (carotid arteries ligation) | [61] |
| ↓ TLR4 | mRNA | |||||
| mRNA | ||||||
| FLAVANONES Naringenin | 1% included in food | 16 weeks | ↓ TLR2 | mRNA | Adipocytes C57Bl/6J mice after HFD | [57] |
| EXTRACTS Achyrocline satureoides (Quercetin and Luteolin) | One dose of 100 mg/kg | 1 h | ↓ TLR4 | Protein | Neutrophils from p.o fed Wistar rats | [63] |
| Cocoa (Procyanidins) | 5%/10% included in food | 3 weeks | ↔ , ↓ TLR2 | mRNA | Small intestine and mesenteric lymph nodes from p.o fed Wistar rats | [64] |
| ↔ , ↔ TLR4 | ||||||
| ↔ ,↓ TLR7 | ||||||
| ↔ , ↑ TLR9 | ||||||
| Cocoa (Procyanidins) | 10%included in food | 7 weeks | ↔ , ↓ , ↔ TLR2 | mRNA | Small intestine, Peyer’s patches and mesenteric lymph nodes from p.o fed Wistar rats | [65] |
| ↓ , ↑, ↔ TLR4 | ||||||
| ↔ , ↓ , ↔ TLR7 | ||||||
| ↔ , ↑ , ↑ TLR9 | ||||||
| Cocoa (Procyanidins) | 10% included in food | 6 weeks | ↔ TLR2 | mRNA | Large intestine from p.o fed Wistar rats | [30] |
| ↔ TLR4 | ||||||
| ↔ TLR7 | ||||||
| ↓ TLR9 | ||||||
| Orange juice (Flavanones) | One dose of 300 kcal drink of orange juice | 1/3/5 h | ↓ TLR2 | mRNA | Mononuclear cells from healthy subjects given high-fat high-carbohydrate meal | [66] |
| ↓ TLR4 | Protein | |||||
| Orange juice (Flavanones) | One dose of 300 kcal drink of orange juice | 1/3/5 h | ↔ TLR4 | mRNA | Mononuclear cells from healthy subjects given high-fat high-carbohydrate meal | [67] |
| Protein |
3.3. Modulatory Action in TLR Activation
3.4. Effect on Signal Transduction Molecules
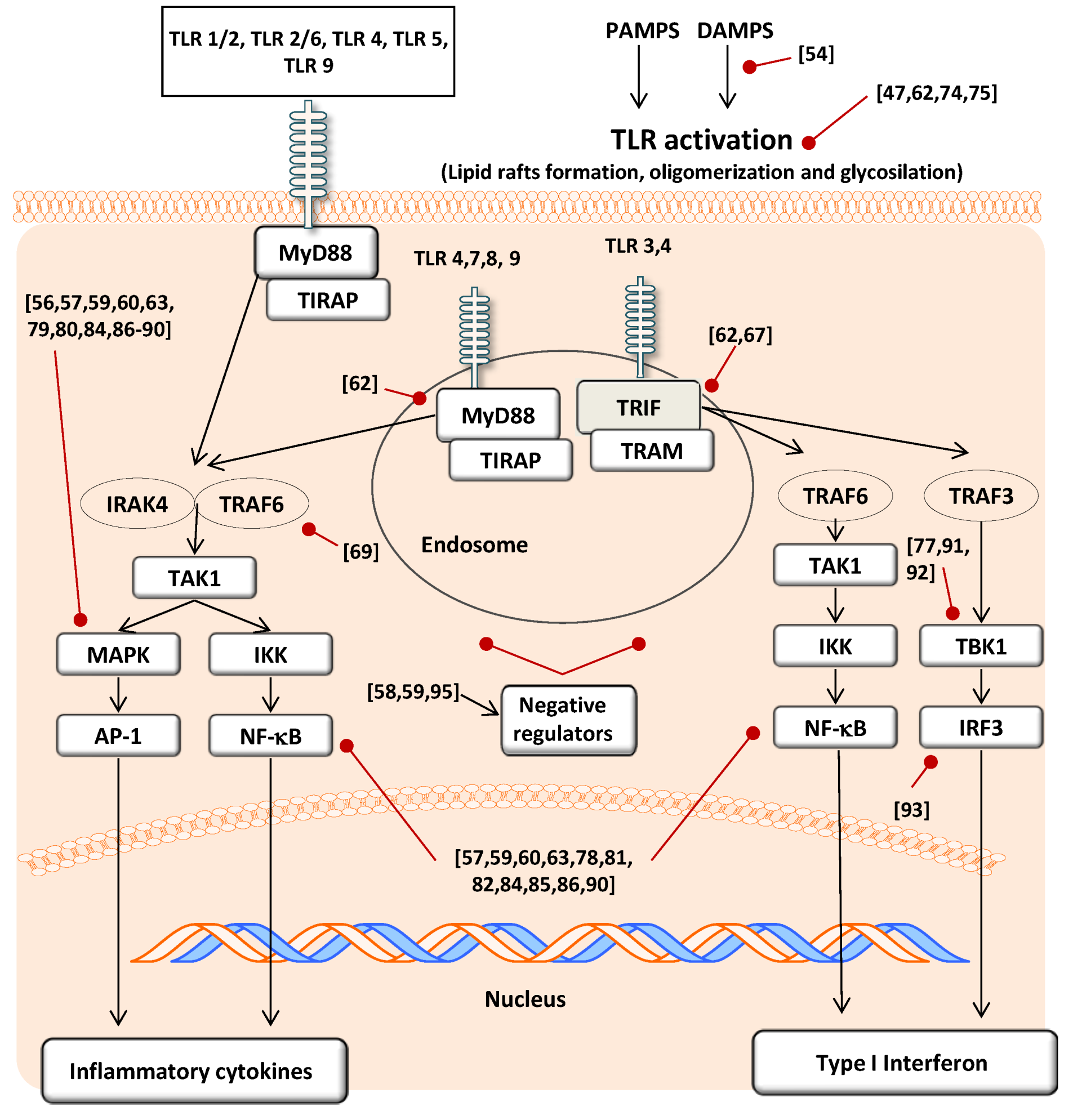
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Brandtzaeg, P. Function of mucosa-associated lymphoid tissue in antibody formation. Immunol. Investing. 2010, 39, 303–355. [Google Scholar] [CrossRef]
- Pabst, O.; Mowat, A.M. Oral tolerance to food protein. Mucosal. Immunol. 2012, 5, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Marques, R.; Boneca, I.G. Expression and functional importance of innate immune receptors by intestinal epithelial cells. Cell. Mol. Life Sci. 2011, 68, 3661–3673. [Google Scholar] [CrossRef] [PubMed]
- Ospelt, C.; Gay, S. TLRs and chronic inflammation. Int. J. Biochem. Cell. Biol. 2010, 42, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Jeong, E.; Lee, J.Y. Intrinsic and extrinsic regulation of innate immune receptors. Yonsei Med. J. 2011, 52, 379–392. [Google Scholar] [CrossRef] [PubMed]
- Piccinini, A.M.; Midwood, K.S. Illustrating the interplay between the extracellular matrix and microRNAs. Int. J. Exp. Pathol. 2014, 95, 158–180. [Google Scholar] [CrossRef] [PubMed]
- McGettrick, A.F.; O’Neill, L.A.J. Localisation and trafficking of toll-like receptors: An important mode of regulation. Curr. Opin. Immunol. 2010, 22, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Allan, S.M.; Rothwell, N.J. Cytokines and acute neurodegeneration. Nat. Rev. Neurosci. 2001, 2, 734–744. [Google Scholar] [CrossRef] [PubMed]
- Thompson, W.L.; van Eldik, L.J. Inflammatory cytokines stimulate the chemokines CCL2/MCP-1 and CCL7/MCP-7 through NFκB and MAPK dependent pathways in rat astrocytes. Brain Res. 2009, 1287, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Dickens, M.; Raingeaud, J.; Davis, R.J.; Greenberg, M.E. Opposing effects of ERK and JNK-p38 MAP kinases on apoptosis. Science 1995, 270, 1326–1331. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Wang, L.; Chen, S. Endogenous toll-like receptor ligands and their biological significance. J. Cell. Mol. Med. 2010, 14, 2592–2603. [Google Scholar] [CrossRef] [PubMed]
- Newton, K.; Dixit, V.M. Signaling in innate immunity and inflammation. Cold Spring Harb. Perspect. Biol. 2012, 4. [Google Scholar] [CrossRef]
- Cao, Z.; Henzel, W.J.; Gao, X. IRAK: A kinase associated with the interleukin-1 receptor. Science 1996, 271, 1128–1131. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Akira, S. Microbial recognition by toll-like receptors. J. Dermatol. Sci. 2004, 34, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Akira, S. TLR signaling pathways. Semin. Immunol. 2004, 16, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Hoebe, K.; Du, X.; Georgel, P.; Janssen, E.; Tabeta, K.; Kim, S.O.; Goode, J.; Lin, P.; Mann, N.; Mudd, S.; et al. Identification of Lps2 as a key transducer of MyD88-independent TIR signaling. Nature 2003, 424, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Sato, S.; Hemmi, H.; Hoshino, K.; Kaisho, T.; Sanjo, H.; Takeuchi, O.; Sugiyama, M.; Okabe, M.; Takeda, K.; et al. Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science 2003, 301, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Pietta, P.G. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Corradini, E.; Foglia, P.; Giansanti, P.; Gubbiotti, R.; Samperi, R.; Lagana, A. Flavonoids: Chemical properties and analytical methodologies of identification and quantitation in foods and plants. Nat. Prod. Res. 2011, 25, 469–495. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Crozier, A.; Del Rio, D.; Clifford, M.N. Bioavailability of dietary flavonoids and phenolic compounds. Mol. Aspects Med. 2010, 31, 446–467. [Google Scholar] [CrossRef] [PubMed]
- Etxeberria, U.; Fernández-Quintela, A.; Milagro, F.I.; Aguirre, L.; Martínez, J.A.; Portillo, M.P. Impact of polyphenols and polyphenol-rich dietary sources on gut microbiota composition. J. Agric. Food Chem. 2013, 61, 9517–9533. [Google Scholar] [PubMed]
- Pérez-Jiménez, J.; Díaz-Rubio, M.E.; Saura-Calixto, F. Non-extractable polyphenols, a major dietary antioxidant: Occurrence, metabolic fate and health effects. Nutr. Res. Rev. 2013, 26, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Tremaroli, V.; Bäckhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 2012, 489, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Duda-Chodak, A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. 2012, 63, 497–503. [Google Scholar] [PubMed]
- Parkar, S.G.; Trower, T.M.; Stevenson, D.E. Fecal microbial metabolism of polyphenols and its effects on human gut microbiota. Anaerobe 2013, 23, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, K.; Sugiyama, Y.; Sakano, T.; Ohigashi, H. Flavonols enhanced production of anti-inflammatory substance(s) by Bifidobacterium adolescentis: Prebiotic actions of galangin, quercetin, and fisetin. Biofactors 2013, 39, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Tzounis, X.; Rodriguez-Mateos, A.; Vulevic, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P.E. Prebiotic evaluation of cocoa-derived flavanols in healthy humans by using a randomized, controlled, double-blind, crossover intervention study. Am. J. Clin. Nutr. 2011, 93, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.C.; Jenner, A.M.; Low, C.S.; Lee, Y.K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006, 157, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Massot-Cladera, M.; Pérez-Berezo, T.; Franch, A.; Castell, M.; Pérez-Cano, F.J. Cocoa modulatory effect on rat faecal microbiota and colonic crosstalk. Arch. Biochem. Biophys. 2012, 527, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M. Overview of antibacterial, antitoxin, antiviral, and antifungal activities of tea flavonoids and teas. Mol. Nutr. Food Res. 2007, 51, 116–134. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.-S.; Touyama, M.; Hisada, T.; Benno, Y. Effects of green tea consumption on human fecal microbiota with special reference to Bifidobacterium species. Microbiol. Immunol. 2012, 56, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Kemperman, R.A.; Gross, G.; Mondot, S.; Possemiers, S.; Marzorati, M.; van de Wiele, T.; Doré, J.; Vaughan, E.E. Impact of polyphenols from black tea and red wine/grape juice on a gut model microbiome. Food Res. Int. 2013, 53, 659–669. [Google Scholar] [CrossRef]
- Queipo-Ortuño, M.I.; Boto-Ordóñez, M.; Murri, M.; Gomez-Zumaquero, J.M.; Clemente-Postigo, M.; Estruch, R.; Cardona Díaz, F.; Andrés-Lacueva, C.; Tinahones, F.J. Influence of red wine polyphenols and ethanol on the gut microbiota. Am. J. Clin. Nutr. 2012, 95, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Dolara, P.; Luceri, C.; de Filippo, C.; Femia, A.P.; Giovannelli, L.; Caderni, G.; Cecchini, C.; Silvi, S.; Orpianesi, C.; Cresci, A. Red wine polyphenols influence carcinogenesis, intestinal microflora, oxidative damage and gene expression profiles of colonic mucosa in F344 rats. Mutat. Res. 2005, 591, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Patán, F.; Cueva, C.; Monagas, M.; Walton, G.E.; Gibson, G.R.M.; Quintanilla-López, J.E.; Lebrón-Aguilar, R.; Martín-Álvarez, P.J.; Moreno-Arribas, M.V.; Bartolomé, B. In vitro fermentation of a red wine extract by human gut microbiota: Changes in microbial groups and formation of phenolic metabolites. J. Agric. Food Chem. 2012, 60, 2136–2147. [Google Scholar] [CrossRef] [PubMed]
- De Boever, P.; Deplancke, B.; Verstraete, W. Fermentation by gut microbiota cultured in a simulator of the human intestinal microbial ecosystem is improved by supplementing a soygerm powder. J. Nutr. 2000, 130, 2599–2606. [Google Scholar] [PubMed]
- Bedani, R.; Pauly-Silveira, N.D.; Roselino, M.N.; de Valdez, G.F.; Rossi, E.A. Effect of fermented soy product on the fecal microbiota of rats fed on a beef-based animal diet. J. Sci. Food Agric. 2010, 90, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Cavallini, D.C.U.; Suzuki, J.Y.; Abdalla, D.S.P.; Vendramini, R.C.; Pauly-Silveira, N.D.; Roselino, M.N.; Pinto, R.A.; Rossi, E.A. Influence of a probiotic soy product on fecal microbiota and its association with cardiovascular risk factors in an animal model. Lipids Health Dis. 2011, 10, 126. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Raudales, D.; Hoeflinger, J.L.; Bringe, N.A.; Cox, S.B.; Dowd, S.E.; Miller, M.J.; de Mejía, E.G. Consumption of different soymilk formulations differentially affects the gut microbiomes of overweight and obese men. Gut Microbes 2012, 3, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Sembries, S.; Dongowski, G.; Mehrländer, K.; Will, F.; Dietrich, H. Physiological effects of extraction juices from apple, grape, and red beet pomaces in rats. J. Agric. Food Chem. 2006, 54, 10269–10280. [Google Scholar] [CrossRef] [PubMed]
- Bialonska, D.; Kasimsetty, S.G.; Schrader, K.K.; Ferreira, D. The effect of pomegranate (Punica granatum L.) byproducts and ellagitannins on the growth of human gut bacteria. J. Agric. Food Chem. 2009, 57, 8344–8349. [Google Scholar] [CrossRef] [PubMed]
- Vendrame, S.; Guglielmetti, S.; Riso, P.; Arioli, S.; Klimis-Zacas, D.; Porrini, M. Six-week consumption of a wild blueberry powder drink increases bifidobacteria in the human gut. J. Agric. Food Chem. 2011, 59, 12815–12820. [Google Scholar] [CrossRef] [PubMed]
- Neyrinck, A.M.; van Hée, V.F.; Bindels, L.B.; de Backer, F.; Cani, P.D.; Delzenne, N.M. Polyphenol-rich extract of pomegranate peel alleviates tissue inflammation and hypercholesterolaemia in high-fat diet-induced obese mice: Potential implication of the gut microbiota. Br. J. Nutr. 2013, 109, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Selma, M.V.; Espín, J.C.; Tomás-Barberán, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef] [PubMed]
- Monagas, M.; Urpi-Sarda, M.; Sánchez-Patán, F.; Llorach, R.; Garrido, I.; Gómez-Cordovés, C.; Andrés-Lacueva, C.; Bartolomé, B. Insights into the metabolism and microbial biotransformation of dietary flavan-3-ols and the bioactivity of their metabolites. Food Funct. 2010, 1, 233–253. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-M.; Yeo, M.; Choue, J.-S.; Jin, J.-H.; Park, S.-J.; Cheong, J.-Y.; Lee, K.-J.; Kim, J.-H.; Hahm, K.-B. Protective mechanism of epigallocatechin-3-gallate against Helicobacter pylori-induced gastric epithelial cytotoxicity via the blockage of TLR-4 signaling. Helicobacter 2004, 9, 632–642. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N. Diet-derived phenols in plasma and tissues and their implications for health. Planta Med. 2004, 70, 1103–1114. [Google Scholar] [CrossRef] [PubMed]
- Tzounis, X.; Vulevic, J.; Kuhnle, G.G.C.; George, T.; Leonczak, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P. Flavanol monomer-induced changes to the human faecal microflora. Br. J. Nutr. 2008, 99, 782–792. [Google Scholar] [CrossRef] [PubMed]
- Neilson, A.P.; Ferruzzi, M.G. Influence of formulation and processing on absorption and metabolism of flavan-3-ols from tea and cocoa. Annu. Rev. Food Sci. Technol. 2011, 2, 125–151. [Google Scholar] [CrossRef] [PubMed]
- Tuohy, K.M.; Conterno, L.; Gasperotti, M.; Viola, R. Up-regulating the human intestinal microbiome using whole plant foods, polyphenols, and/or fiber. J. Agric. Food Chem. 2012, 60, 8776–8782. [Google Scholar] [CrossRef] [PubMed]
- De Kivit, S.; Tobin, M.C.; Forsyth, C.B.; Keshavarzian, A.; Landay, A.L. Regulation of intestinal immune responses through TLR activation: Implications for pro- and prebiotics. Front. Immunol. 2014, 5, 60. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Cheng, Z.; Shi, H.; Xin, W.; Wang, T.T.Y.; Yu, L.L. Isolation and characterization of two flavonoids, engeletin and astilbin, from the leaves of Engelhardia roxburghiana and their potential anti-inflammatory properties. J. Agric. Food Chem. 2011, 59, 4562–4569. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Ku, S.-K.; Bae, J.-S. Inhibitory effects of kaempferol-3-O-sophoroside on HMGB1-mediated proinflammatory responses. Food Chem. Toxicol. 2012, 50, 1118–1123. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Shalini, V.; Helen, A. Quercetin regulates oxidized LDL induced inflammatory changes in human PBMCs by modulating the TLR-NF-κB signaling pathway. Immunobiology 2011, 216, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Yilma, A.N.; Singh, S.R.; Morici, L.; Dennis, V.A. Flavonoid naringenin: A potential immunomodulator for Chlamydia trachomatis inflammation. Mediators Inflamm. 2013, 2013. [Google Scholar] [CrossRef]
- Yoshida, H.; Watanabe, W.; Oomagari, H.; Tsuruta, E.; Shida, M.; Kurokawa, M. Citrus flavonoid naringenin inhibits TLR2 expression in adipocytes. J. Nutr. Biochem. 2013, 24, 1276–1284. [Google Scholar] [CrossRef] [PubMed]
- Byun, E.H.; Fujimura, Y.; Yamada, K.; Tachibana, H. TLR4 signaling inhibitory pathway induced by green tea polyphenol epigallocatechin-3-gallate through 67-kDa laminin receptor. J. Immunol. 2010, 185, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Byun, E.-B.; Choi, H.-G.; Sung, N.-Y.; Byun, E.-H. Green tea polyphenol epigallocatechin-3-gallate inhibits TLR4 signaling through the 67-kDa laminin receptor on lipopolysaccharide-stimulated dendritic cells. Biochem. Biophys. Res. Commun. 2012, 426, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Wang, C.-Y.; Jin, L. Baicalin downregulates Porphyromonas gingivalis lipopolysaccharide-upregulated IL-6 and IL-8 expression in human oral keratinocytes by negative regulation of TLR signaling. PLoS One 2012, 7, e51008. [Google Scholar] [CrossRef]
- Li, H.-Y.; Yuan, Z.-Y.; Wang, Y.-G.; Wan, H.-J.; Hu, J.; Chai, Y.-S.; Lei, F.; Xing, D.-M.; Du, L.-J. Role of baicalin in regulating toll-like receptor 2/4 after ischemic neuronal injury. Chin. Med. J. 2012, 125, 1586–1593. [Google Scholar] [PubMed]
- Qiao, H.; Zhang, X.; Zhu, C.; Dong, L.; Wang, L.; Zhang, X.; Xing, Y.; Wang, C.; Ji, Y.; Cao, X. Luteolin downregulates TLR4, TLR5, NF-κB and p-p38MAPK expression, upregulates the p-ERK expression, and protects rat brains against focal ischemia. Brain Res. 2012, 1448, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Barioni, E.D.; Santin, J.R.; Machado, I.D.; de Paula Rodrigues, S.F.; Ferraz-de-Paula, V.; Wagner, T.M.; Cogliati, B.; Corrêa Dos Santos, M.; Machado Mda, S.; de Andrade, S.F.; et al. Achyrocline satureioides (Lam.) D.C. hydroalcoholic extract inhibits neutrophil functions related to innate host defense. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Pérez-Berezo, T.; Franch, A.; Ramos-Romero, S.; Castellote, C.; Pérez-Cano, F.J.; Castell, M. Cocoa-enriched diets modulate intestinal and systemic humoral immune response in young adult rats. Mol. Nutr. Food Res. 2011, 55, S56–S66. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Berezo, T.; Franch, A.; Castellote, C.; Castell, M.; Pérez-Cano, F.J. Mechanisms involved in down-regulation of intestinal IgA in rats by high cocoa intake. J. Nutr. Biochem. 2012, 23, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Ghanim, H.; Sia, C.L.; Upadhyay, M.; Korzeniewski, K.; Viswanathan, P.; Abuaysheh, S.; Mohanty, P.; Dandona, P. Orange juice neutralizes the proinflammatory effect of a high-fat, high-carbohydrate meal and prevents endotoxin increase and toll-like receptor expression. Am. J. Clin. Nutr. 2010, 91, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Deopurkar, R.; Ghanim, H.; Friedman, J.; Abuaysheh, S.; Sia, C.L.; Mohanty, P.; Viswanathan, P.; Chaudhuri, A.; Dandona, P. Differential effects of cream, glucose, and orange juice on inflammation, endotoxin, and the expression of toll-like receptor-4 and suppressor of cytokine signaling-3. Diabetes Care 2010, 33, 991–997. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-J.; Youn, H.-S. Suppression of homodimerization of toll-like receptor 4 by isoliquiritigenin. Phytochemistry 2010, 71, 1736–1740. [Google Scholar] [CrossRef] [PubMed]
- Haque, A.; Koide, N.; Iftakhar-E-Khuda, I.; Noman, A.S.M.; Odkhuu, E.; Badamtseren, B.; Naiki, Y.; Komatsu, T.; Yoshida, T.; Yokochi, T. Flavopiridol inhibits lipopolysaccharide-induced TNF-α production through inactivation of nuclear factor-κB and mitogen-activated protein kinases in the MyD88-dependent pathway. Microbiol. Immunol. 2011, 55, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Shuto, T.; Ono, T.; Ohira, Y.; Shimasaki, S.; Mizunoe, S.; Watanabe, K.; Suico, M.A.; Koga, T.; Sato, T.; Morino, S.; et al. Curcumin decreases toll-like receptor-2 gene expression and function in human monocytes and neutrophils. Biochem. Biophys. Res. Commun. 2010, 398, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Ghanim, H.; Sia, C.L.; Korzeniewski, K.; Lohano, T.; Abuaysheh, S.; Marumganti, A.; Chaudhuri, A.; Dandona, P. A resveratrol and polyphenol preparation suppresses oxidative and inflammatory stress response to a high-fat, high-carbohydrate meal. J. Clin. Endocrinol. Metab. 2011, 96, 1409–1414. [Google Scholar] [CrossRef] [PubMed]
- Silver, A.C.; Arjona, A.; Walker, W.E.; Fikrig, E. The circadian clock controls toll-like receptor 9-mediated innate and adaptive immunity. Immunity 2012, 36, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Froy, O.; Chapnik, N. Circadian oscillation of innate immunity components in mouse small intestine. Mol. Immunol. 2007, 44, 1954–1960. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.S.; Saitoh, S.I.; Miyake, K.; Hwang, D.H. Inhibition of homodimerization of toll-like receptor 4 by curcumin. Biochem. Pharmacol. 2006, 72, 62–69. [Google Scholar] [CrossRef]
- Kaneko, M.; Takimoto, H.; Sugiyama, T.; Seki, Y.; Kawaguchi, K.; Kumazawa, Y. Suppressive effects of the flavonoids quercetin and luteolin on the accumulation of lipid rafts after signal transduction via receptors. Immunopharmacol. Immunotoxicol. 2008, 30, 867–882. [Google Scholar] [CrossRef] [PubMed]
- Capelluto, D.G.S. Tollip: A multitasking protein in innate immunity and protein trafficking. Microbes Infect. 2012, 14, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.S.; Lee, J.Y.; Saitoh, S.I.; Miyake, K.; Kang, K.W.; Choi, Y.J.; Hwang, D.H. Suppression of MyD88- and TRIF-dependent signaling pathways of toll-like receptor by (−)-epigallocatechin-3-gallate, a polyphenol component of green tea. Biochem. Pharmacol. 2006, 72, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.-T.; Chen, T.-L.; Loon, W.-S.; Tai, Y.-T.; Cherng, Y.-G.; Chen, R.-M. Lipopolysaccharide stimulates syntheses of toll-like receptor 2 and surfactant protein-A in human alveolar epithelial A549 cells through upregulating phosphorylation of MEK1 and ERK1/2 and sequential activation of NF-κB. Cytokine 2011, 55, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.J.; Spencer, J.P.E.; Rice-Evans, C. Flavonoids: Antioxidants or signaling molecules? Free Radic. Biol. Med. 2004, 36, 838–849. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, K.S.; Rupasinghe, H.P.V. Polyphenols: Multipotent therapeutic agents in neurodegenerative diseases. Oxid. Med. Cell. Longev. 2013, 2013. [Google Scholar] [CrossRef]
- Park, Y.C.; Rimbach, G.; Saliou, C.; Valacchi, G.; Packer, L. Activity of monomeric, dimeric, and trimeric flavonoids on NO production, TNF-α secretion, and NF-κB-dependent gene expression in RAW 264.7 macrophages. FEBS Lett. 2000, 465, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Comalada, M.; Camuesco, D.; Sierra, S.; Ballester, I.; Xaus, J.; Gálvez, J.; Zarzuelo, A. In vivo quercitrin anti-inflammatory effect involves release of quercetin, which inhibits inflammation through down-regulation of the NF-κB pathway. Eur. J. Immunol. 2005, 35, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Pantano, C.; Reynaert, N.L.; van der Vliet, A.; Janssen-Heininger, Y.M.W. Redox-sensitive kinases of the nuclear factor-κB signaling pathway. Antioxid. Redox Signal. 2006, 8, 1791–1806. [Google Scholar] [CrossRef] [PubMed]
- Hirao, K.; Yumoto, H.; Nakanishi, T.; Mukai, K.; Takahashi, K.; Takegawa, D.; Matsuo, T. Tea catechins reduce inflammatory reactions via mitogen-activated protein kinase pathways in toll-like receptor 2 ligand-stimulated dental pulp cells. Life Sci. 2010, 86, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-C.; Chow, M.-P.; Huang, W.-C.; Lin, Y.-C.; Chang, Y.-J. Flavonoids inhibit tumor necrosis factor-alpha-induced up-regulation of intercellular adhesion molecule-1 (ICAM-1) in respiratory epithelial cells through activator protein-1 and nuclear factor-kappaB: Structure-activity relationships. Mol. Pharmacol. 2004, 66, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-Y.; Hu, J.; Zhao, S.; Yuan, Z.-Y.; Wan, H.-J.; Lei, F.; Ding, Y.; Xing, D.-M.; Du, L.-J. Comparative study of the effect of baicalin and its natural analogs on neurons with oxygen and glucose deprivation involving innate immune reaction of TLR2/TNFα. J. Biomed. Biotechnol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.J.; Lee, K.W.; Lee, D.E.; Rogozin, E.A.; Bode, A.M.; Lee, H.J.; Dong, Z. Cocoa procyanidins suppress transformation by inhibiting mitogen-activated protein kinase kinase. J. Biol. Chem. 2008, 283, 20664–20673. [Google Scholar] [CrossRef] [PubMed]
- Ramiro-Puig, E.; Casadesús, G.; Lee, H.; Zhu, X.; McShea, A.; Perry, G.; Pérez-Cano, F.J.; Smith, M.A.; Castell, M. Neuroprotective effect of cocoa flavonoids on in vitro oxidative stress. Eur. J. Nutr. 2008, 48, 61. [Google Scholar] [CrossRef]
- Mackenzie, G.G.; Carrasquedo, F.; Delfino, J.M.; Keen, C.L.; Fraga, C.G.; Oteiza, P.I. Epicatechin, catechin, and dimeric procyanidins inhibit PMA-induced NF-κB activation at multiple steps in Jurkat T cells. FASEB J. 2004, 18, 167–169. [Google Scholar] [PubMed]
- Lim, E.-K.; Mitchell, P.J.; Brown, N.; Drummond, R.A.; Brown, G.D.; Kaye, P.M.; Bowles, D.J. Regiospecific methylation of a dietary flavonoid scaffold selectively enhances IL-1β production following toll-like receptor 2 stimulation in THP-1 monocytes. J. Biol. Chem. 2013, 288, 21126–21135. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.K.; Kim, S.Y.; Kim, Y.S.; Lee, W.-H.; Hwang, D.H.; Lee, J.Y. Suppression of the TRIF-dependent signaling pathway of toll-like receptors by luteolin. Biochem. Pharmacol. 2009, 77, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.Y.; Sung, G.-H.; Kim, J.H.; Yoon, J.Y.; Yang, Y.; Park, J.-G.; Kim, S.-H.; Yi, Y.-S.; Yang, W.-S.; Yong, D.-H.; et al. Syk and Src are major pharmacological targets of a Cerbera manghas methanol extract with kaempferol-based anti-inflammatory activity. J. Ethnopharmacol. 2014, 151, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-J.; Song, H.-Y.; Youn, H.-S. Suppression of the TRIF-dependent signaling pathway of toll-like receptors by isoliquiritigenin in RAW264.7 macrophages. Mol. Cells 2009, 28, 365–368. [Google Scholar] [CrossRef] [PubMed]
- Liew, F.Y.; Xu, D.; Brint, E.K.; O’Neill, L.A.J. Negative regulation of toll-like receptor-mediated immune responses. Nat. Rev. Immunol. 2005, 5, 446–458. [Google Scholar] [CrossRef] [PubMed]
- Sung, N.-Y.; Yang, M.-S.; Song, D.-S.; Kim, J.-K.; Park, J.-H.; Song, B.-S.; Park, S.-H.; Lee, J.-W.; Park, H.-J.; Kim, J.-H.; et al. Procyanidin dimer B2-mediated IRAK-M induction negatively regulates TLR4 signaling in macrophages. Biochem. Biophys. Res. Commun. 2013, 438, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Renz, H.; Brandtzaeg, P.; Hornef, M. The impact of perinatal immune development on mucosal homeostasis and chronic inflammation. Nat. Rev. Immunol. 2012, 12, 9–23. [Google Scholar]
- Pérez-Cano, F.J.; Massot-Cladera, M.; Franch, A.; Castellote, C.; Castell, M. The effects of cocoa on the immune system. Front. Pharmacol. 2013, 4, 71. [Google Scholar] [CrossRef] [PubMed]
- Castell, M.; Pérez-Cano, F.J.; Abril-Gil, M.; Franch, À. Flavonoids on allergy. Curr. Pharm. Des. 2014, 20, 973–987. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Lee, H. Advances in nutritional research on regulatory T-cells. Nutrients 2013, 5, 4305–4315. [Google Scholar] [CrossRef] [PubMed]
- Magrone, T.; Jirillo, E. The interplay between the gut immune system and microbiota in health and disease: Nutraceutical intervention for restoring intestinal homeostasis. Curr. Pharm. Des. 2013, 19, 1329–1342. [Google Scholar] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Cano, F.J.; Massot-Cladera, M.; Rodríguez-Lagunas, M.J.; Castell, M. Flavonoids Affect Host-Microbiota Crosstalk through TLR Modulation. Antioxidants 2014, 3, 649-670. https://doi.org/10.3390/antiox3040649
Pérez-Cano FJ, Massot-Cladera M, Rodríguez-Lagunas MJ, Castell M. Flavonoids Affect Host-Microbiota Crosstalk through TLR Modulation. Antioxidants. 2014; 3(4):649-670. https://doi.org/10.3390/antiox3040649
Chicago/Turabian StylePérez-Cano, Francisco J., Malen Massot-Cladera, Maria J. Rodríguez-Lagunas, and Margarida Castell. 2014. "Flavonoids Affect Host-Microbiota Crosstalk through TLR Modulation" Antioxidants 3, no. 4: 649-670. https://doi.org/10.3390/antiox3040649
APA StylePérez-Cano, F. J., Massot-Cladera, M., Rodríguez-Lagunas, M. J., & Castell, M. (2014). Flavonoids Affect Host-Microbiota Crosstalk through TLR Modulation. Antioxidants, 3(4), 649-670. https://doi.org/10.3390/antiox3040649






