Investigation of the Antioxidant and Hepatoprotective Potential of Hypericum mysorense
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection and Identification of Plant Material
2.2. Preparation of Extracts
2.3. Preparation of Test and Standard Solutions
2.4. Chemicals
2.5. Selection and Maintenance of Animals
2.6. In Vitro Antioxidant Activity
2.7. In Vivo Antioxidant and Hepatoprotective Studies
2.7.1. Preparation of Extracts and Standard
2.7.2. Experimental Design
2.8. Histopathological Studies
2.9. HPLC Quantitation
2.10. Statistical Analysis
3. Results and Discussion
3.1. Preparation of Extracts
3.2. Estimation of Total Phenolic and Flavonoid Content
| Part of Plant | Total Flavonol Content * (mg/g of Extract) | Total Phenol Content * (mg/g of Extract) |
|---|---|---|
| HMF | 33.48 ± 2.1 | 38.6 ± 3.66 |
| HML | 28.76 ± 1.44 | 37.07 ± 2.90 |
| HMA | 27.52 ± 1.67 | 32.7 ± 1.23 |
| HMS | 26.54 ± 3.56 | 30.7 ± 3.14 |
| HMR | 15.84 ± 0.84 | 20.7 ± 1.11 |
3.3. In Vitro Antioxidant Activity
3.4. In Vivo Antioxidant and Hepatoprotective Studies
| Extracts/Standards | IC50 Values ± S.E.M. (µg/mL) * by Methods | Total Antioxidant Capacity | |||||||
|---|---|---|---|---|---|---|---|---|---|
| ABTS | DPPH | p-NDA | Hydrogen Peroxide | Lipid per Oxidation | Nitric Oxide | Alkaline DMSO | |||
| HMF | 1.53 ± 0.04 a,b | 3.65 ± 0.02 a,b | 690.15 ± 11.16 b,c | 57.56 ± 2.34 a,b | 13.68 ± 1.62 c,d | 208.35 ± 9.36 | 822.56 ± 18.93 a,b,c | 0.30 ± 0.01 d | |
| HML | 2.05 ± 0.04 a,b | 4.05 ± 0.03 a,b | >1000 | 68.34 ± 3.26 a,b | 18.47 ± 1.32 c,d | 250.53 ± 12.38 | 923.84 ± 22.45 a,b,c | 0.43 ± 0.01 d | |
| HMA | 2.97 ± 0.04 a | 5.23 ± 0.04 a,b | >1000 | 74.44 ± 3.32 a | 28.26 ± 1.76 c,d | 301.38 ± 12.57 | >1000 | 0.66 ± 0.02 d | |
| HMS | 3.63 ± 0.02 a | 5.95 ± 0.03 | >1000 | 80.50 ± 4.02 a | 33.32 ± 1.34 c,d | 365.64 ± 15.64 | >1000 | 0.96 ± 0.03 d | |
| HMR | 4.60 ± 0.04 a | 8.32 ± 0.06 | >1000 | 97.53 ± 2.98 a | 40.22 ± 2.13 c,d | 408.24 ± 14.75 | >1000 | 1.43 ± 0.06 d | |
| STANDARDS | |||||||||
| Ascorbic acid | 11.25 ± 0.49 | 2.69 ± 0.05 | - | 187.33 ± 3.93 | - | - | >1000 | - | |
| Rutin | 0.51± 0.01 | 3.91 ± 0.10 | >1000 | 36.66 ± 0.22 | - | 65.44 ± 2.56 | >1000 | - | |
| Butylated hydroxyl anisole | - | - | >1000 | 24.88 ± 0.16 | 112.66 ± 1.32 | - | >1000 | - | |
| α-Tocopherol | - | - | - | - | 91.66 ± 4.92 | - | - | 3.41 ± 0.47 | |
| Treatment | Dose (mg or mL/Kg b.w.) | ASAT (U/L) | ALAT (U/L) | ALP (U/L) | Total Protein (G/dL) | Total Bilirubin (mg/dL) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Liver | Serum | Liver | Serum | Liver | Serum | Liver | Serum | Liver | Serum | ||
| Normal | - | 101.52 ± 1.078 | 123.39 ± 1.244 | 51.02 ± 0.926 | 69.49 ± 1.435 | 197.83 ± 4.321 | 260.31 ± 6.622 | 6.917 ± 0.175 | 6.283 ± 0.207 | 0.516 ± 0.031 | 0.702 ± 0.036 |
| CCl4 intoxicated | 1 mL | 390.74 ± 1.254 a | 450.23 ± 1.630 a | 198.57 ± 1.708 a | 237.58 ± 1.931 a | 515.23 ± 11.746 a | 691.05 ± 15.282 a | 4.867 ± 0.088 a | 4.692 ± 0.129 a | 1.783 ± 0.047 a | 2.017 ± 0.127 a |
| CCl4 + Silymarin | 1 mL + 100 mg | 143.07 ± 1.633 b | 188.82 ± 1.493 b | 64.16 ± 1.071 b | 94.02 ± 1.317 b | 256.28 ± 9.387 b | 289.54 ± 7.784 b | 6.283 ± 0.105 b | 5.983 ± 0.194 b | 0.708 ± 0.063 b | 0.966 ± 0.042 b |
| CCl4 + HMF | 1 mL + 100 mg | 292.75 ± 1.414 b | 304.62 ± 2.139 b | 160.34 ± 1.838 b | 180.47 ± 2.072 b | 428.52 ± 11.455 b | 547.53 ± 11.846 b | 5.556 ± 0.131 c | 4.933 ± 0.185 | 1.310 ± 0.068 b | 1.517 ± 0.047 b |
| 1 mL + 200 mg | 152.73 ± 1.062 b | 219.79 ± 1.282 b | 66.19 ± 1.052 b | 107.73 ± 1.606 b | 282.37 ± 10.147 b | 328.59 ± 9.316 b | 6.023 ± 0.116 b | 5.758 ± 0.180 c | 0.753 ± 0.062 b | 0.903 ± 0.036 b | |
| CCl4 + HML | 1 mL + 100 mg | 315.24 ± 1.695 b | 331.32 ± 2.274 b | 171.93 ± 1.517 b | 194.76 ± 1.942 b | 469.83 ± 14.269 | 584.38 ± 16.682 b | 5.032 ± 0.066 | 4.796 ± 0.169 | 1.483 ± 0.054 c | 1.717 ± 0.063 d |
| 1 mL + 200 mg | 187.03 ± 1.732 b | 254.42 ± 1.904 b | 76.69 ± 1.217 b | 121.59 ± 1.585 b | 307.27 ± 9.665 b | 361.31 ± 11.926 b | 5.767 ± 0.168 b | 5.413 ± 0.171 | 0.883 ± 0.043 b | 1.220 ± 0.066 b | |
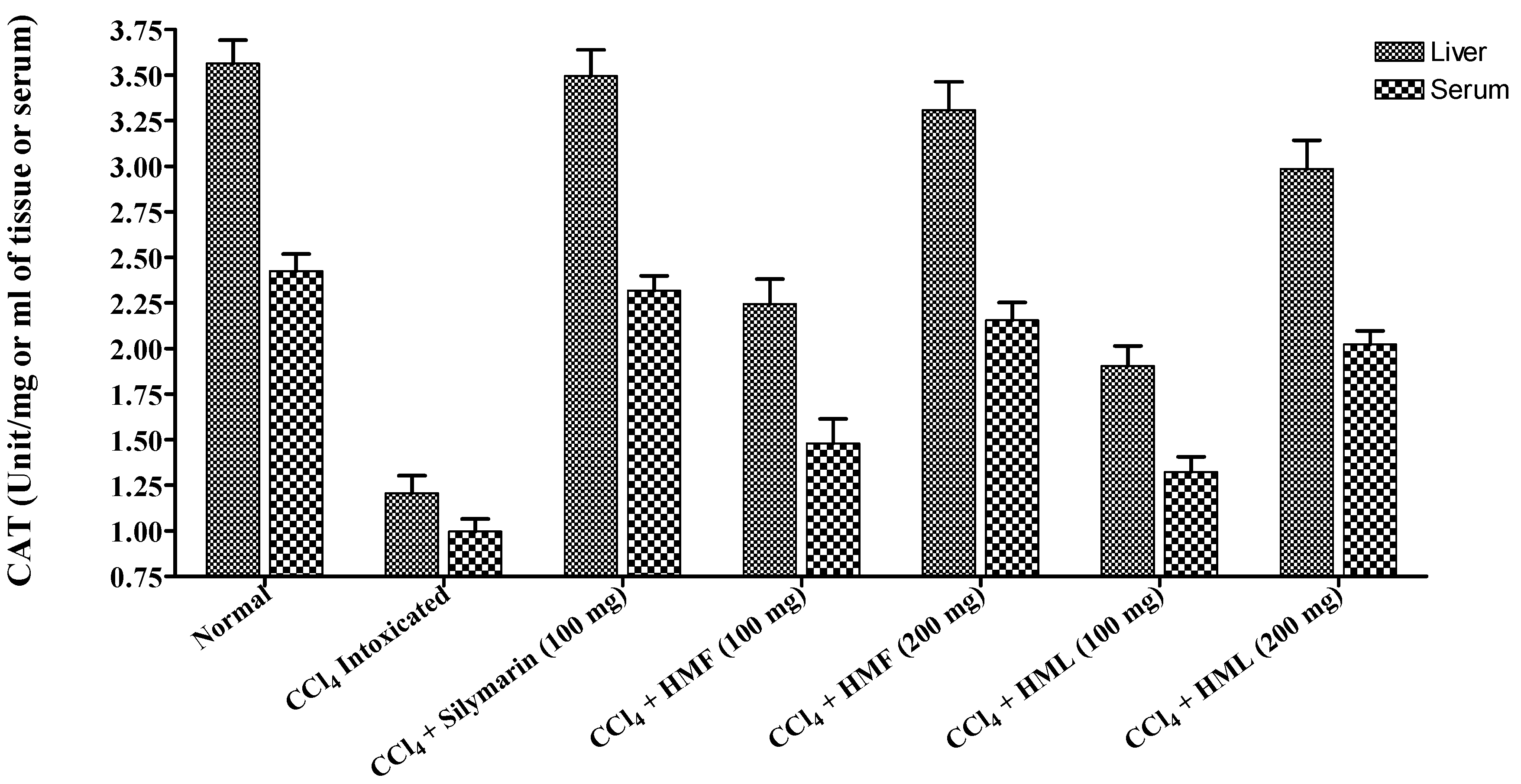
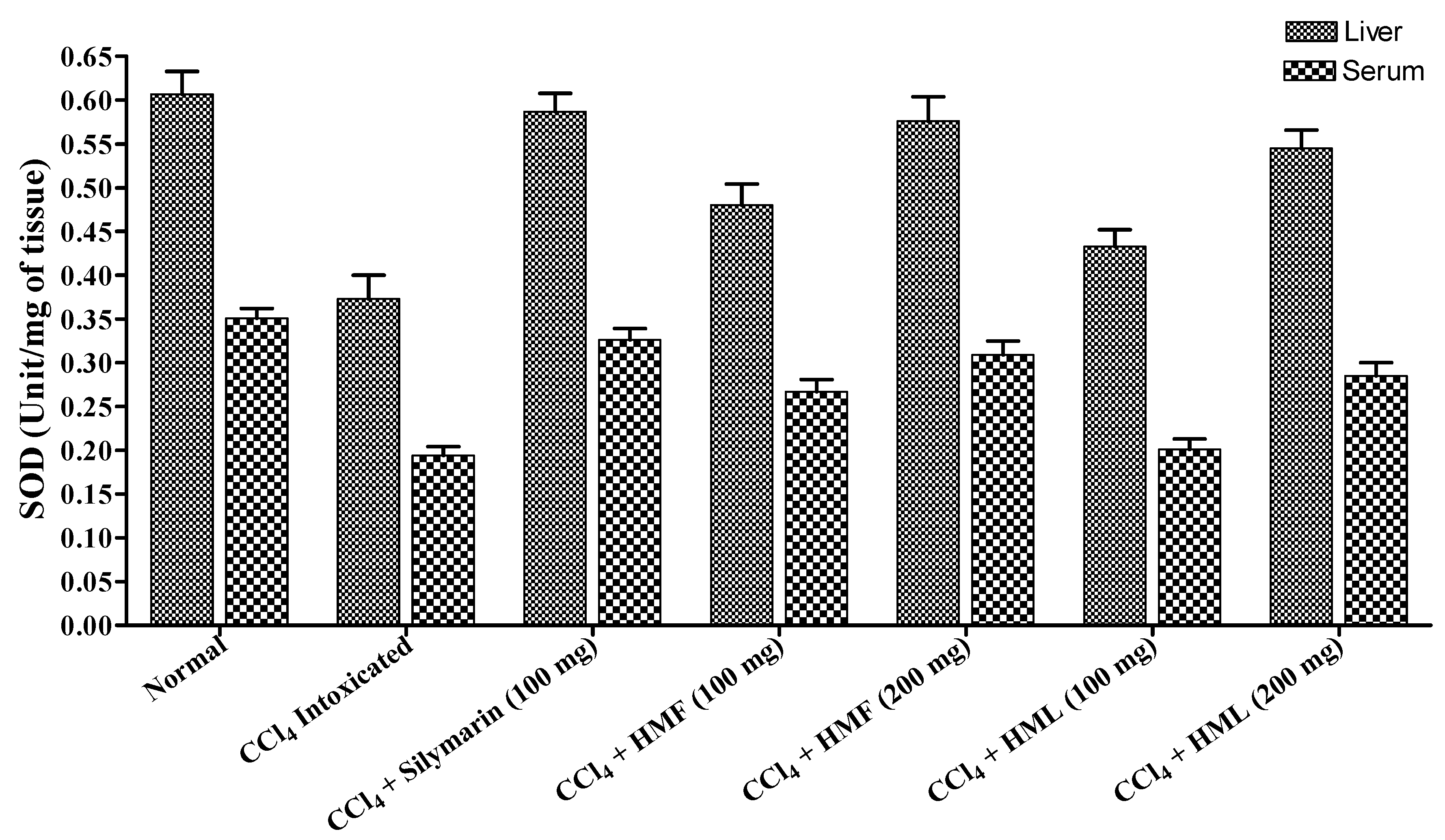
| Treatment | Dose (mg/kg Body Weight) | CAT (Unit/mg of Tissue) | SOD (Unit/mg of Tissue) | TBARS (n mole of MDA/mg of Protein) | |||
|---|---|---|---|---|---|---|---|
| Liver | Serum | Liver | Serum | Liver | Serum | ||
| Normal | 0.5 mL Sodium CMC | 3.564 ± 0.128 | 2.425 ± 0.094 | 0. 607 ± 0.026 | 0.351 ± 0.011 | 4.872 ± 0.178 | 3.845 ± 0.065 |
| Control (CCl4) | 1 mL | 1.207 ± 0.096 a | 0.997 ± 0.069 a | 0.373 ± 0.027 c | 0.194 ± 0.010 a | 8.425 ± 0.149 a | 6.583 ± 0.176 a |
| Silymarin + CCl4 | 100 | 3.495 ± 0.145 b | 2.318 ± 0.082 b | 0.587 ± 0.021 b | 0.326 ± 0.013 b | 5.102 ± 0.170 b | 4.207 ± 0.123 b |
| HMF + CCl4 | 100 | 2.245 ± 0.137 d | 1.481 ± 0.133 b | 0.480 ± 0.024 c | 0.267 ± 0.014 d | 7.214 ± 0.243 b | 5.512 ± 0.114 b |
| 200 | 3.310 ± 0.154 b | 2.156 ± 0.098 b | 0.576 ± 0.028 b | 0.309 ± 0.016 b | 5.456 ± 0.162 b | 4.417 ± 0.107 b | |
| HML + CCl4 | 100 | 1.906 ± 0.109 | 1.323 ± 0.084 d | 0.433 ± 0.019 | 0.201 ± 0.012 | 7.676 ± 0.196 | 5.878 ± 0.134 c |
| 200 | 2.987 ± 0.156 b | 2.023 ± 0.075 b | 0.545 ± 0.021 d | 0.285 ± 0.015 b | 5.873 ± 0.112 b | 4.765 ± 0.124 b | |
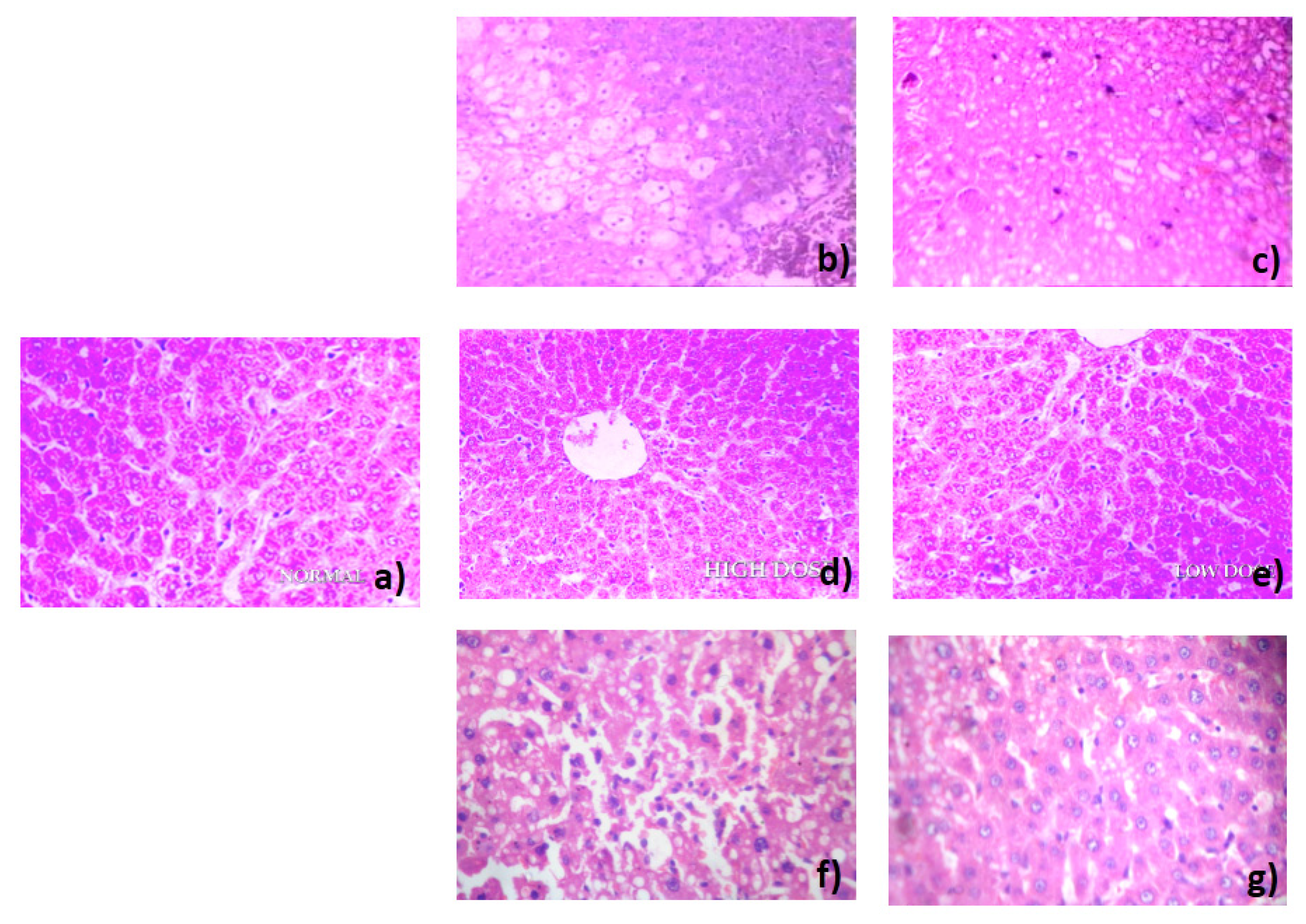
3.5. Histopathology
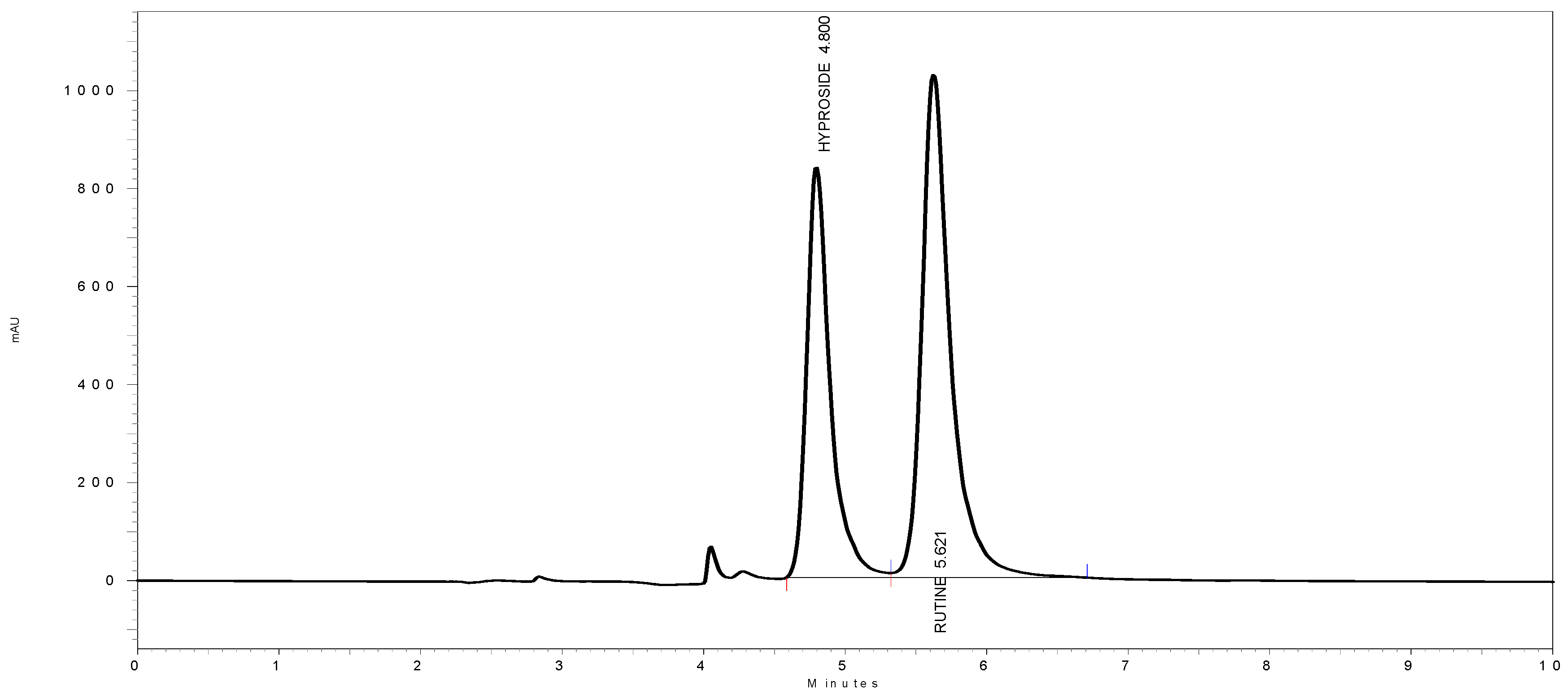
3.6. HPLC Quantitation
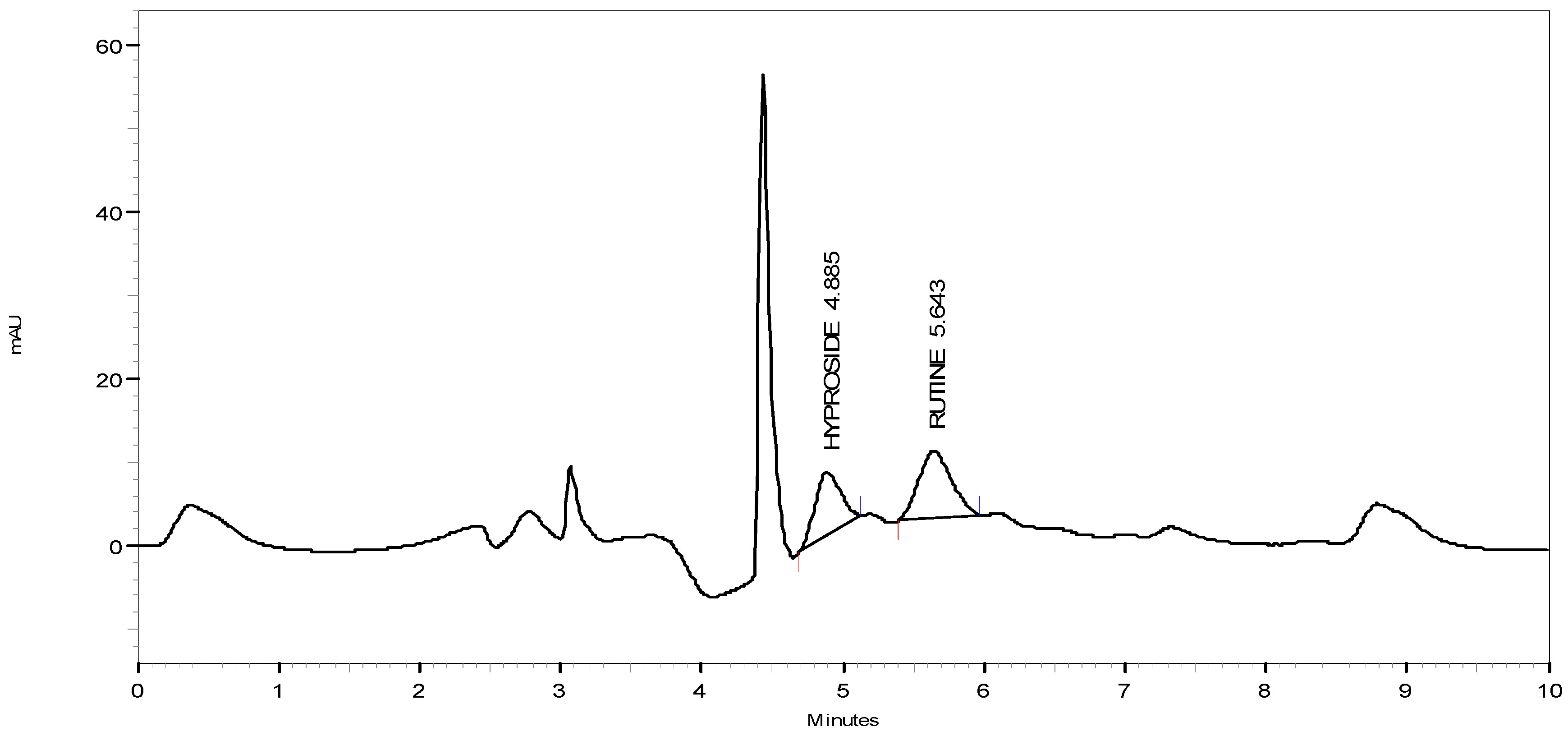
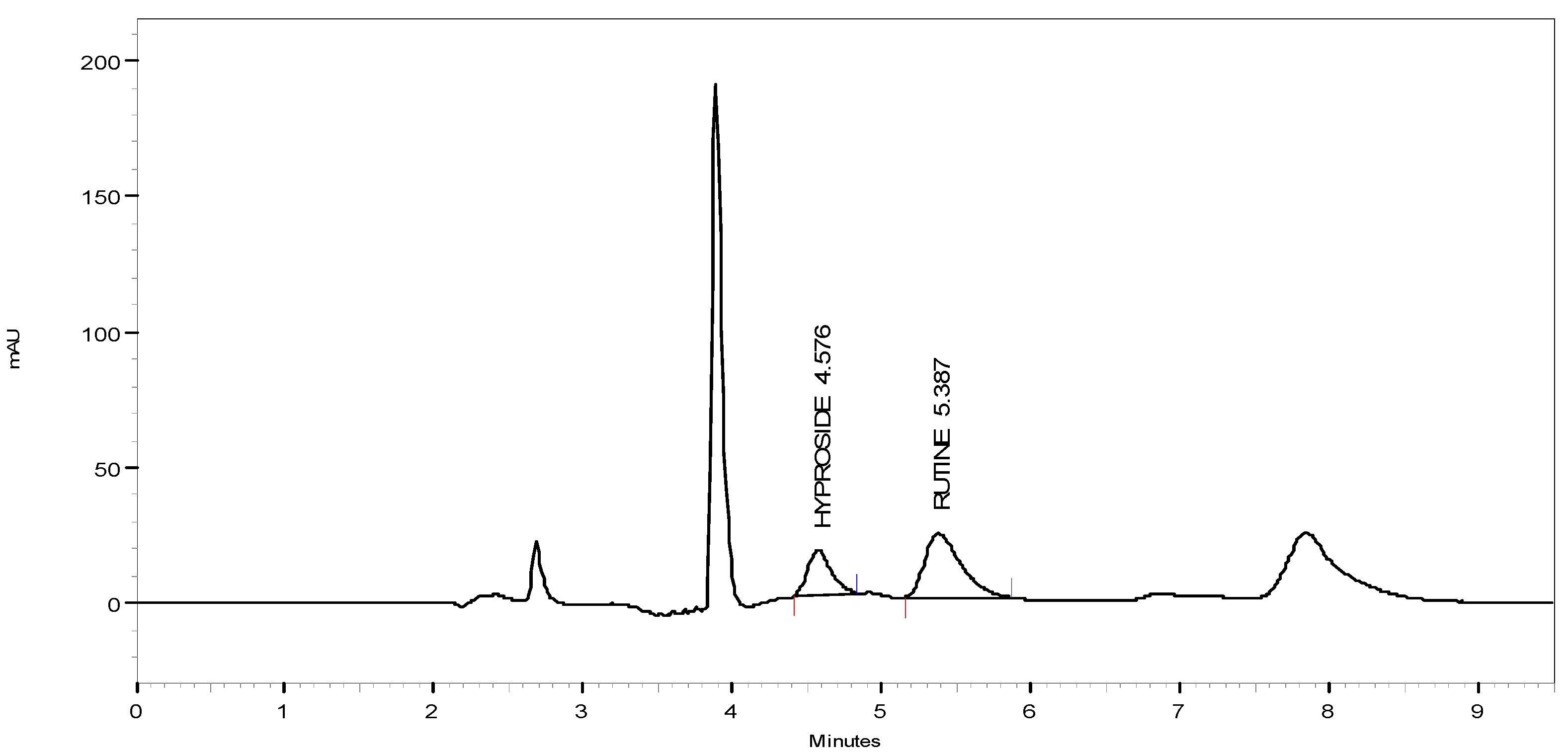
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Ben-Yoseph, O.; Boxer, P.A.; Ross, B.D. Assessment of the role of the glutathione and pentose phosphate pathways in the protection of primary cerebrocortical cultures from oxidative stress. J. Neurochem. 1996, 66, 2329–2337. [Google Scholar] [PubMed]
- Harman, D. Free radical theory of aging: Origin of life, evolution, and aging. Age 1980, 3, 100–102. [Google Scholar] [CrossRef]
- Moskovitz, J.; Yim, K.A.; Choke, P.B. Free radicals and disease. Arch. Biochem. Biophys. 2002, 397, 354–359. [Google Scholar] [CrossRef]
- Hollman, P.C.H.; Katan, M.B. Absorption, metabolism, and bioavailability of flavonoids. In Flavonoids in Health and Disease; Rice-Evans, C.A., Packer, L., Eds.; Marcel Dekker: New York, NY, York, 1996; pp. 483–522. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure antioxidant activities of flavonoids as bioactive components of food. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Bors, W.; Michel, C. Antioxidant capacity of flavonols and gallate esters: Pulse radiolysis studies. Free Radic. Biol. Med. 1999, 27, 1413–1426. [Google Scholar] [CrossRef] [PubMed]
- Torrel, J.; Cillard, J.; Cillard, P. Antioxidant activity of flavonoids and reactivity with peroxyl radical. Phytochemistry 1986, 25, 383–385. [Google Scholar] [CrossRef]
- Van Acker, S.A.B.E.; van den Berg, D.J.; Tromp, M.N.J.L.; Griffioen, D.H.; van Bennekom, W.P.; van der Vijgh, W.J.F.; Bast, A. Structural aspect of antioxidant activity of flavonoids. Free Radic. Biol. Med. 1996, 20, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Yazaki, K.; Okada, T. Hypericum erectum Thunb (St. John’s wort): In vitro culture and the production of procynadins. Biotechnol. Agric. For. 1994, 26, 167–178. [Google Scholar]
- Hu, L.H.; Sim, K.Y.; Sampsoniones, A.-M. A unique family of caged pilyprenylated benzoylphloroglucinol derivatives, from Hypericum sampsonii. Tetrahedron 2000, 56, 1379–1386. [Google Scholar] [CrossRef]
- Decosterd, L.; Hoffmann, E.; Kyburz, R.; Bray, D.; Jostettmann, K. A new phloroglucinol derivative from Hypericum calycinum with antifungal and in vitro antimalarial activity. Planta Med. 1991, 57, 548–551. [Google Scholar] [CrossRef]
- Ishiguro, K.; Yamaki, M.; Kashihara, M.; Takagi, S.; Isoi, K. Srothraling: A new antimicrobial compound from Hypericum japonicum. Planta Med. 1990, 56, 274–276. [Google Scholar] [CrossRef]
- Hudson, J.B.; Lopez-Bazzocchi, I.; Towers, G.H.N. Antiviral activities of hypericin. Antivir. Res. 1991, 15, 101–112. [Google Scholar] [PubMed]
- Agostinins, P.; Vantieghem, A.; Merlevede, W.; de Witte, P.A.M. Hypericin in cancer treatment: More light on the way. Int. J. Biochem. Cell Biol. 2002, 34, 221–241. [Google Scholar] [CrossRef] [PubMed]
- Veronika, B.; Guido, J.; Adolf, N.; Hilke, W. Flavonoids from Hypericum perforaturm show antidepressant activity in the forced swimming test. Planta Med. 2000, 66, 3–6. [Google Scholar] [CrossRef]
- Luo, L.; Sun, Q.; Mao, Y.Y.; Lu, Y.H.; Tan, R.X. Inhibitory effects of flavonoids from Hypericum perforatum on nitric oxide synthesis. J. Ethnopharmacol. 2004, 93, 221–225. [Google Scholar] [CrossRef]
- Sara, C.; Franco, O.R.; Aleessandro, R.; Nicola, M.; Antonella, C.; Piero, M.; Grancesca, B.A.; Mauro, P. Interaction with Type II estrogen binding sites and antiproloferateve activity of tamoxifen and quercetin in human non-small-cell lung cancer. Am. J. Respir. Cell. Mol. Biol. 1997, 17, 51–59. [Google Scholar] [CrossRef]
- Cakir, A.; Mavi, A.; Yildirim, A.; Duru, M.E.; Harmandar, M.; Kazaz, C. Isolation and characterization of antioxidant phenolic compounds from the aerial part of Hypericum hyssopifolium L. by activity-guided fractionation. J. Ethnopharmacol. 2003, 87, 73–83. [Google Scholar] [CrossRef]
- Gamble, J.S. Flora of the Presidency of Madras; Bishen Singh & Mahendra Pal Singh: Dehradun, India, 1984; Volume 1, pp. 69–71. [Google Scholar]
- Asolkar, L.V.; Kakkar, K.K.; Chakre, O.J. Second Supplement of Glossary of Indian Medicinal Plants with Active Principles; Publication and Information Directorate, CSIR: New Delhi, India, 1992; Volume 1, pp. 138–139. [Google Scholar]
- Vijayan, P.; Raghu, C.; Ashok, G.; Dhanaraj, S.A.; Suresh, B. Antiviral activity of medicinal plants of Nilgiris. Indian J. Med. Res. 2004, 120, 24–29. [Google Scholar] [PubMed]
- Vijayan, P.; Vinod Kumar, S.; Dhanaraj, S.A.; Mukherjee, P.K.; Suresh, B. In vitro cytotoxicity and antitumour properties of Hypericum mysorense and Hypericum patulum. Phytother. Res. 2003, 17, 952–956. [Google Scholar] [CrossRef]
- Raghu Chandrashekhar, H.; Venkatesh, P.; Arumugam, M.; Vijayan, P. Estimation of total phenols with special emphasis to antioxidant potentials of few hypericum species. Pharmacologyonline 1 2009, 1, 680–687. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Hwang, B.Y.; Kim, H.S.; Lee, J.H.; Hong, Y.S.; Lee, K.S.; Lee, J.J. Antioxidant benzoylated flavan-3-ol glycoside from Celastrus orbiculatus. J. Nat. Prod. 2001, 64, 82–84. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C.; Aruoma, O.I. The deoxyribose method: A simple “test-tube” assay for determination de-oxyribose of rate constants for reactions of hydroxyl radicals. Anal. Biochem. 1987, 165, 215–219. [Google Scholar] [CrossRef]
- Bors, W.; Michel, C.; Saran, M. On the nature of biochemically generated hydroxyl radicals studies using the bleaching of p-nitrosodimethylaniline as a direct assay method. Eur. J. Biochem. 1979, 95, 621–627. [Google Scholar] [CrossRef]
- Jayaprakasha, G.K.; Jaganmohan Rao, L.; Sakariah, K.K. Antioxidant activity of flavidin in different in vitro model systems. Bioorg. Med. Chem. 2004, 12, 5141–5147. [Google Scholar] [CrossRef]
- Duh, P.D.; Yen, G.C.; Yen, W.J.; Chang, L.W. Antioxidant effects of water extracts from barley (Hordeum vulgare) prepared under different roasting temperatures. J. Agric. Food Chem. 2001, 49, 1455–1463. [Google Scholar] [CrossRef]
- Fukuzawa, K.; Tokumura, A.; Ouchi, S.; Tsukatani, H. Antioxidant activities of tocopherols on Fe2+-ascorbate-induced lipid peroxidation in lecithin liposomes. Lipids 1982, 17, 511–513. [Google Scholar] [CrossRef] [PubMed]
- Marcocci, L.; Packer, L.; Droy-Lefaix, M.T.; Sekaki, A.; Gardes-Albert, M. Antioxidant action of Ginko biloba extracts EGB 761. Methods Enzymol. 1994, 234, 462–475. [Google Scholar] [CrossRef]
- Garrat, D.C. The Quantitative Analysis of Drugs. Quant. Anal. Drugs 1964. [Google Scholar] [CrossRef]
- Elizabeth, K.; Rao, M.N.A. Oxygen radical scavenging activity of curcumin. Int. J. Pharm. 1990, 58, 237–240. [Google Scholar] [CrossRef]
- Prieto, P.; Pineda, M.; Aquilar, M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef]
- Sadasivam, S.; Manikam, A. Biochemical Methods for Agricultural Sciences; Wiley Eastern Limited: New Delhi, India, 1992. [Google Scholar]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Evans, W.C. Trease and Evans’ Pharmacognosy; Elsevier Health Sciences: Philadelphia, PA, USA, 2009. [Google Scholar]
- Trease, G.E.; Evans, W.C. A Textbook of Pharmacognosy; Bailliere Tindall: London, UK, 1989; p. 388. [Google Scholar]
- Bacon, B.R.; Tavill, A.S.; Brittenham, G.M.; Park, C.H.R.; Recknagel, O. Hepatic lipid peroxidation in vivo in rats with chronic iron overload. J. Clin. Investig. 1983, 71, 429–439. [Google Scholar] [CrossRef]
- Castro, J.A.; DeFerreyra, E.C.; DeCastro, C.R.; Fenoes, O.M.; Sasame, H.; Gillette, J.R. Prevention of carbon tetrachloride induced necrosis by inhibitors of drug metabolism—Further studies on their mechanism of action. Biochem. Pharmacol. 1974, 23, 295–302. [Google Scholar] [CrossRef]
- Lin, C.C.; Yen, M.H.; Lo, T.S.; Lin, J.M. Evaluation of the hepatoprotective and antioxidant activity of Boehmeria nivea Var. nivea and B. nivea Var, tenacissma. J. Ethnopharmacol. 1998, 60, 9–17. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Fujii, T. Toxicological correlation between changes in blood biochemical parameters and liver histopathological findings. J. Toxicol. Sci. 1997, 22, 161–183. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, P.; Vinod Kumar, S.; Shrishailappa, B.; Mukherjee, P.K.; Dhanaraj, S.A.; Suresh, B. Selective in vitro cytotoxicity of Hypericum hookerianum towards cancer cell lines. Orient. Pharm. Exp. Med. 2003, 3, 141–146. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Suresh, B. Studies on in vivo wound healing activity of leaf extract of Hypericum mysorense with different wound model in rats. Nat. Prod. Sci. 2000, 6, 73–78. [Google Scholar]
- Mukherjee, P.K.; Saritha, G.S.; Suresh, B. Antimicrobial potential of two different Hypericum species available in India. Phytother. Res. 2002, 16, 692–695. [Google Scholar] [CrossRef]
- Selvam, R.; Subramonian, L.; Gayathri, R.; Angayarkanni, N. The Antioxidant activity of turmeric (Curcuma longa). J. Ethnopharmacol. 1995, 47, 59–67. [Google Scholar] [CrossRef]
- Lin, J.M.; Lin, C.C.; Chen, M.F.; Ujiie, T.; Takada, T. Radical scavenger and antihepatotoxic activity of Ganoderma formosanum, Ganoderma lucidum and Ganodermaneo japonicum. J. Ethnopharmacol. 1995, 47, 33–41. [Google Scholar] [CrossRef]
- Sultana, S.; Pervaiz, S.; Iqbal, M.; Athar, M. Crude extracts of hepatoprotective plants, Solanum nigrum and Cichorum intybus inhibits free radical-mediated DNA Damage. J. Ethnopharmacol. 1995, 45, 189–192. [Google Scholar] [CrossRef]
- Torres-Duran, P.V.; Miranda Zamora, R.; Paredes-Carbajal, M.C. Studies on the preventive effect of Spirulina maxima on fatty liver development induced by carbon tetrachloride, in the rat. J. Ethnopharmacol. 1999, 64, 141–147. [Google Scholar] [CrossRef]
- Clauson, G.A. Mechanism of carbon tetrachloride hepatotoxicity. Pathol. Immunopathol. Res. 1989, 8, 104–112. [Google Scholar] [CrossRef]
- Rubinstein, D. Epinephrine release and liver glycogen levels after carbon tetrachloride administration. Am. J. Physiol. 1962, 203, 1033–1037. [Google Scholar] [PubMed]
- Bishayee, A.; Sarkar, A.; Chatterjee, M. The hepatoprotective activity of carrot (Daucas carota L.) against carbon tetrachloride intoxication in mouse liver. J. Ethnopharmacol. 1995, 47, 69–74. [Google Scholar] [CrossRef]
- Mahendale, H.M.; Gupta, P.K.; Shalunkhe, D.K. Hepatic Toxicity; Metropolitan Books: New Delhi, India, 1985; Volume 1, p. 225. [Google Scholar]
- Ramellini, G.; Meldolesi, J. Liver protection by silymarin. In vitro effect on dissociated rat hepatocytes. Arzneim. Forsch. 1976, 26, 69–73. [Google Scholar]
- Morazzoni, P.; Bombardelli, E. Silybum marianum (Carduus marianus). Fitoterapia 1995, 66, 3–42. [Google Scholar]
- Venukumar, M.R.; Latha, M.S. Antioxidant activity of Curculigo orchioides in carbon tetrachloride induced hepatopathy in rats. Indian J. Clin. Biochem. 2002, 17, 80–87. [Google Scholar] [CrossRef]
- Bandyopadhyay, U.; Das, D.; Ranajit Banerjee, K. Reactive oxygen species: Oxidative damage and pathogenesis. Curr. Sci. 1999, 77, 658–665. [Google Scholar]
- Shenoy, K.A.; Somayaji, S.N.; Bairy, K.L. Evaluation of hepatoprotective activity of Gingo biloba in rats. Indian J. Pharmacol. 2002, 46, 167–174. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hariharapura, R.C.; Srinivasan, R.; Ashok, G.; Dongre, S.H.; Jagani, H.V.; Vijayan, P. Investigation of the Antioxidant and Hepatoprotective Potential of Hypericum mysorense. Antioxidants 2014, 3, 526-543. https://doi.org/10.3390/antiox3030526
Hariharapura RC, Srinivasan R, Ashok G, Dongre SH, Jagani HV, Vijayan P. Investigation of the Antioxidant and Hepatoprotective Potential of Hypericum mysorense. Antioxidants. 2014; 3(3):526-543. https://doi.org/10.3390/antiox3030526
Chicago/Turabian StyleHariharapura, Raghu C., Ramamurthy Srinivasan, Godavarthi Ashok, Santoshkumar H. Dongre, Hitesh V. Jagani, and Pottekkad Vijayan. 2014. "Investigation of the Antioxidant and Hepatoprotective Potential of Hypericum mysorense" Antioxidants 3, no. 3: 526-543. https://doi.org/10.3390/antiox3030526
APA StyleHariharapura, R. C., Srinivasan, R., Ashok, G., Dongre, S. H., Jagani, H. V., & Vijayan, P. (2014). Investigation of the Antioxidant and Hepatoprotective Potential of Hypericum mysorense. Antioxidants, 3(3), 526-543. https://doi.org/10.3390/antiox3030526




