Ponicidin Inhibits Lung Cancer Progression Through Coordinated Downregulation of Sulfhydryl Antioxidants and TrxR1
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Cell Culture
2.3. Cell Viability Assay
2.4. Cell Staining
2.5. Colony Formation Assay
2.6. Cell Apoptosis Analysis
2.7. ROS Assay
2.8. RT-qPCR
2.9. Western Blot Analysis
2.10. Mitochondrial Membrane Potential Assay
2.11. ER Tracker Assay and Ca2+ Concentration Detection
2.12. Immunofluorescence Staining for γ-H2AX
2.13. In Vivo Experiments
2.14. Statistical Analysis
3. Results
3.1. Ponicidin Suppresses Lung Cancer Cell Proliferation and Induces Cell Death
3.2. Ponicidin Inhibits Lung Cancer Cell Growth In Vivo
3.3. Ponicidin Increases ROS Level in Lung Cancer Cells
3.4. Ponicidin Induces the Reduction of Sulfhydryl Compounds and Depletion of Glutathione, Thereby Regulating Oxidative Stress in Lung Cancer Cells
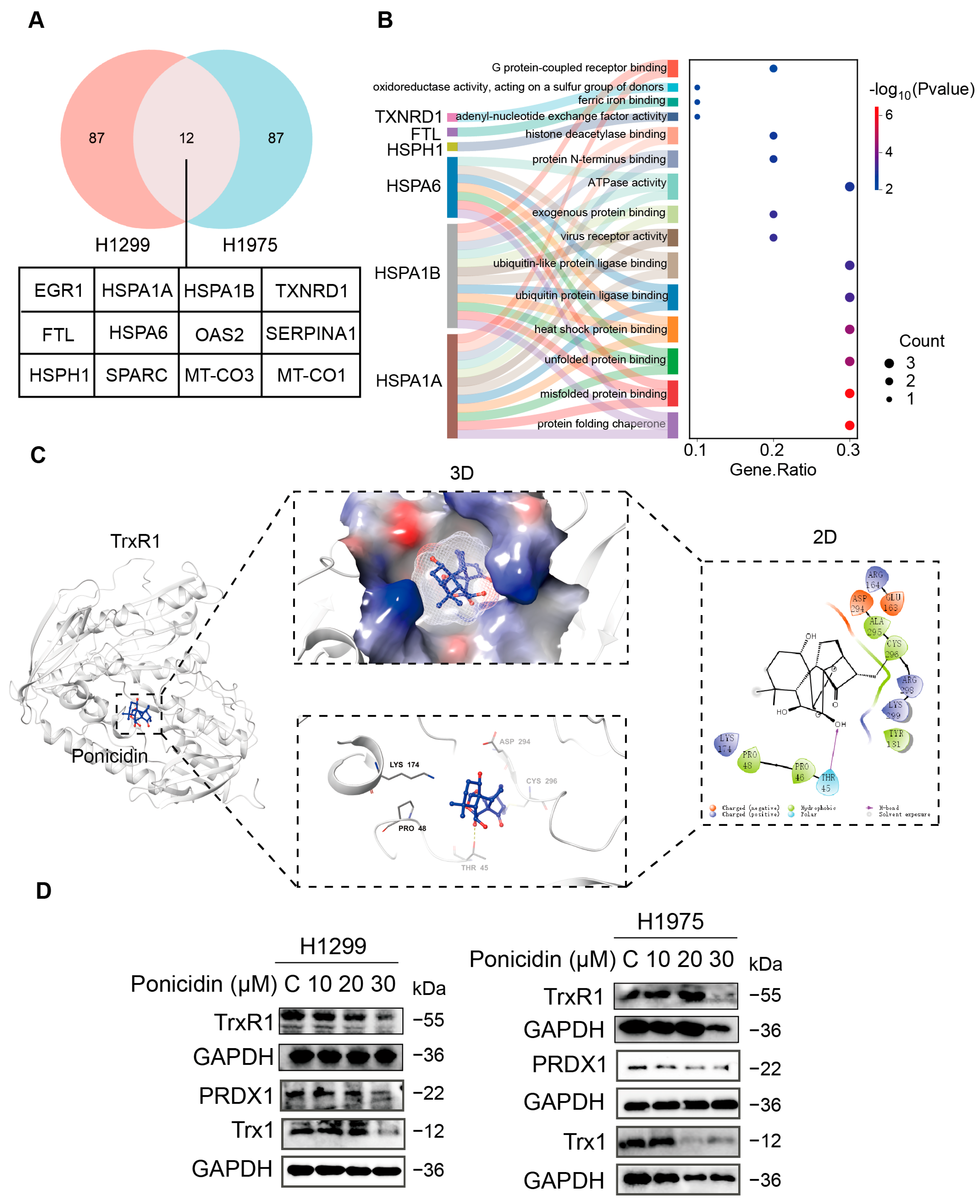
3.5. Ponicidin Downregulates TrxR1 in Lung Cancer Cells, a Molecule That Is Strongly Associated with Cellular Oxidative Stress
3.6. Ponicidin Reduces Mitochondrial Membrane Potential, Triggers ER Stress, and Induces DNA Damage in Lung Cancer Cells
3.7. NAC Attenuates Ponicidin-Induced Organellar Stress and DNA Damage in Lung Cancer Cells
3.8. Ponicidin Inhibits Lung Cancer Cell Growth by Regulating Oxidative Stress In Vivo
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Morgensztern, D.; Boshoff, C. The biology and management of non-small cell lung cancer. Nature 2018, 553, 446–454. [Google Scholar] [CrossRef]
- Ettinger, D.S.; Wood, D.E.; Aisner, D.L.; Akerley, W.; Bauman, J.R.; Bharat, A.; Bruno, D.S.; Chang, J.Y.; Chirieac, L.R.; D’Amico, T.A.; et al. NCCN Guidelines Insights: Non-Small Cell Lung Cancer, Version 2.2021. J. Natl. Compr. Cancer Netw. 2021, 19, 254–266. [Google Scholar] [CrossRef]
- Ettinger, D.S.; Wood, D.E.; Aisner, D.L.; Akerley, W.; Bauman, J.R.; Bharat, A.; Bruno, D.S.; Chang, J.Y.; Chirieac, L.R.; D’Amico, T.A.; et al. Non-Small Cell Lung Cancer, Version 3.2022, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2022, 20, 497–530. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Martinez, J.A.-O.; Guijosa, A.A.-O.; Flores-Estrada, D.; Cruz-Rico, G.; Turcott, J.A.-O.; Hernández-Pedro, N.A.-O.; Caballé-Perez, E.A.-O.; Cardona, A.A.-O.; Arrieta, O.A.-O. Real-World Survival Outcomes in Non-Small Cell Lung Cancer: The Impact of Genomic Testing and Targeted Therapies in a Latin American Middle-Income Country. JCO Glob. Oncol. 2024, 10, e2400338. [Google Scholar] [CrossRef]
- Duma, N.; Santana-Davila, R.; Molina, J.R. Non-Small Cell Lung Cancer: Epidemiology, Screening, Diagnosis, and Treatment. Mayo Clin. Proc. 2019, 94, 1623–1640. [Google Scholar] [CrossRef]
- Westover, D.; Zugazagoitia, J.; Cho, B.C.; Lovly, C.M.; Paz-Ares, L. Mechanisms of acquired resistance to first- and second-generation EGFR tyrosine kinase inhibitors. Ann. Oncol. 2018, 29, i10–i19. [Google Scholar] [CrossRef]
- Lin, J.J.; Shaw, A.T. Resisting Resistance: Targeted Therapies in Lung Cancer. Trends Cancer 2016, 2, 350–364. [Google Scholar] [CrossRef]
- Passaro, A.; Brahmer, J.; Antonia, S.; Mok, T.; Peters, S. Managing Resistance to Immune Checkpoint Inhibitors in Lung Cancer: Treatment and Novel Strategies. J. Clin. Oncol. 2022, 40, 598–610. [Google Scholar] [CrossRef]
- Daylan, A.E.C.; Miao, E.; Tang, K.; Chiu, G.; Cheng, H. Lung Cancer in Never Smokers: Delving into Epidemiology, Genomic and Immune Landscape, Prognosis, Treatment, and Screening. Lung 2023, 201, 521–529. [Google Scholar] [CrossRef]
- Li, Z.; Feiyue, Z.; Gaofeng, L. Traditional Chinese medicine and lung cancer--From theory to practice. Biomed. Pharmacother. 2021, 137, 111381. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, L.S.; Shen, L.P.; Han, Z.F.; Jian, H.; Liu, J.X.; Xu, L.; Li, H.G.; Tian, J.H.; Mao, Z.J. Traditional Chinese Medicine treatment as maintenance therapy in advanced non-small-cell lung cancer: A randomized controlled trial. Complement. Ther. Med. 2016, 24, 55–62. [Google Scholar] [CrossRef]
- Jiang, Y.; Liu, L.S.; Shen, L.P.; Liu, J.X.; Jiang, G.N.; Gu, A.Q.; Li, H.C.; Li, Q.; Li, H.G.; Huang, P.X. Traditional Chinese Medicine Treatment as Adjuvant Therapy in Completely Resected Stage IB-IIIA Non-Small-Cell Lung Cancer: Study Protocol for a Multicenter, Double-Blind, Randomized, Placebo-Controlled Trial. Clin. Lung Cancer 2019, 20, e541–e547. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, J.; Chen, R.; Li, T.; Chen, M. Active Ingredients from Chinese Medicine for Combination Cancer Therapy. Int. J. Biol. Sci. 2023, 19, 3499–3525. [Google Scholar] [CrossRef] [PubMed]
- Sartippour, M.R.; Seeram, N.P.; Heber, D.; Hardy, M.; Norris, A.; Lu, Q.; Zhang, L.; Lu, M.; Rao, J.Y.; Brooks, M.N. Rabdosia rubescens inhibits breast cancer growth and angiogenesis. Int. J. Oncol. 2005, 26, 121–127. [Google Scholar] [CrossRef]
- Chen, S.; Liu, J.; Zhang, H. Efficacy of rabdosia rubescens in the treatment of gingivitis. J. Huazhong Univ. Sci. Technol. Med. Sci. 2009, 29, 659–663. [Google Scholar] [CrossRef] [PubMed]
- Meade-Tollin, L.C.; Wijeratne, E.M.; Cooper, D.; Guild, M.; Jon, E.; Fritz, A.; Zhou, G.X.; Whitesell, L.; Liang, J.Y.; Gunatilaka, A.A. Ponicidin and oridonin are responsible for the antiangiogenic activity of Rabdosia rubescens, a constituent of the herbal supplement PC SPES. J. Nat. Prod. 2004, 67, 2–4. [Google Scholar]
- Hsieh, T.C.; Wijeratne, E.K.; Liang, J.Y.; Gunatilaka, A.L.; Wu, J.M. Differential control of growth, cell cycle progression, and expression of NF-kappaB in human breast cancer cells MCF-7, MCF-10A, and MDA-MB-231 by ponicidin and oridonin, diterpenoids from the chinese herb Rabdosia rubescens. Biochem. Biophys. Res. Commun. 2005, 337, 224–231. [Google Scholar] [CrossRef]
- Ren, K.K.; Wang, H.Z.; Xie, L.P.; Chen, D.W.; Liu, X.; Sun, J.; Nie, Y.C.; Zhang, R.Q. The effects of oridonin on cell growth, cell cycle, cell migration and differentiation in melanoma cells. J. Ethnopharmacol. 2006, 103, 176–180. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H.; Liang, B.; Qin, L.; Zhang, M.; Lv, X.; Hu, S.; Fan, X.; Xie, W.; Yang, H.; et al. Ponicidin promotes ferroptosis to enhance treatment sensitivity in Lenvatinib-resistant hepatocellular carcinoma cells through regulation of KEAP1/NRF2. Phytomedicine 2025, 143, 156824. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, J.; Wu, M.; Ren, D.; Chen, C.; Du, Z.; Li, Q.; Chang, J.; Pu, Q.; Liu, Z. Ponicidin triggered ferroptosis in esophageal squamous cell carcinoma by suppressing the SLC7A11/Glutathione/GPX4 signalling axis. Phytomedicine 2025, 143, 156925. [Google Scholar] [CrossRef]
- Liang, Z.; Ye, Y.; Deng, Z.; Lan, H.; Liu, C.; Xu, Y.; Fan, M.; Liu, Z.; Wu, P.; An, L.; et al. CHPF2 as a novel biomarker and ponicidin as a potential therapeutic agent in hepatocellular carcinoma. Pharmacol. Res. 2025, 215, 107698. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Liang, Z.; Zhang, L.; Jiang, L.; Xu, Y.; Zhang, Y.; Zhang, R.; Wang, C.; Liu, Z. Ponicidin Promotes Hepatocellular Carcinoma Mitochondrial Apoptosis by Stabilizing Keap1-PGAM5 Complex. Adv. Sci. 2024, 11, e2406080. [Google Scholar] [CrossRef]
- Zhao, C.; Zhang, J.; Yang, Z.Y.; Shi, L.Q.; Liu, S.L.; Pan, L.J.; Dong, P.; Zhang, Y.; Xiang, S.S.; Shu, Y.J.; et al. Ponicidin inhibited gallbladder cancer proliferation and metastasis by decreasing MAGEB2 expression through FOXO4. Phytomedicine 2023, 114, 154785. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zuo, J.; Li, B.; Chen, R.; Luo, K.; Xiang, X.; Lu, S.; Huang, C.; Liu, L.; Tang, J.; et al. Drug-induced oxidative stress in cancer treatments: Angel or devil? Redox Biol. 2023, 63, 102754. [Google Scholar] [CrossRef]
- Lennicke, C.; Cochemé, H.M. Redox metabolism: ROS as specific molecular regulators of cell signaling and function. Mol. Cell 2021, 81, 3691–3707. [Google Scholar] [CrossRef]
- Cheung, E.C.; Vousden, K.H. The role of ROS in tumour development and progression. Nat. Rev. Cancer 2022, 22, 280–297. [Google Scholar] [CrossRef]
- Lin, C.L.; Yu, C.I.; Lee, T.H.; Chuang, J.M.; Han, K.F.; Lin, C.S.; Huang, W.P.; Chen, J.Y.; Chen, C.Y.; Lin, M.Y.; et al. Plumbagin induces the apoptosis of drug-resistant oral cancer in vitro and in vivo through ROS-mediated endoplasmic reticulum stress and mitochondrial dysfunction. Phytomedicine 2023, 111, 154655. [Google Scholar] [CrossRef] [PubMed]
- You, J.H.; Lee, J.; Roh, J.L. PGRMC1-dependent lipophagy promotes ferroptosis in paclitaxel-tolerant persister cancer cells. J. Exp. Clin. Cancer Res. 2021, 40, 350. [Google Scholar] [CrossRef]
- Ni, M.; Zhou, J.; Zhu, Z.; Xu, Q.; Yin, Z.; Wang, Y.; Zheng, Z.; Zhao, H. Shikonin and cisplatin synergistically overcome cisplatin resistance of ovarian cancer by inducing ferroptosis via upregulation of HMOX1 to promote Fe2+ accumulation. Phytomedicine 2023, 112, 154701. [Google Scholar] [CrossRef]
- Dong, S.; Liang, S.; Cheng, Z.; Zhang, X.; Luo, L.; Li, L.; Zhang, W.; Li, S.; Xu, Q.; Zhong, M.; et al. ROS/PI3K/Akt and Wnt/β-catenin signalings activate HIF-1α-induced metabolic reprogramming to impart 5-fluorouracil resistance in colorectal cancer. J. Exp. Clin. Cancer Res. 2022, 41, 15. [Google Scholar] [CrossRef]
- Chayapong, J.; Madhyastha, H.; Madhyastha, R.; Nurrahmah, Q.I.; Nakajima, Y.; Choijookhuu, N.; Hishikawa, Y.; Maruyama, M. Arsenic trioxide induces ROS activity and DNA damage, leading to G0/G1 extension in skin fibroblasts through the ATM-ATR-associated Chk pathway. Environ. Sci. Pollut. Res. Int. 2017, 24, 5316–5325. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, X.; Han, X.; Liu, R.; Fang, J. Targeting the Thioredoxin System for Cancer Therapy. Trends Pharmacol. Sci. 2017, 38, 794–808. [Google Scholar] [CrossRef]
- Ron, D.; Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat. Rev. Mol. Cell Biol. 2007, 8, 519–529. [Google Scholar] [CrossRef]
- Scott, T.L.; Rangaswamy, S.; Wicker, C.A.; Izumi, T. Repair of oxidative DNA damage and cancer: Recent progress in DNA base excision repair. Antioxid. Redox Signal 2014, 20, 708–726. [Google Scholar] [CrossRef]
- Qi, J.; Li, M.; Wang, L.; Hu, Y.; Liu, W.; Long, Z.; Zhou, Z.; Yin, P.; Zhou, M. National and subnational trends in cancer burden in China, 2005–2020: An analysis of national mortality surveillance data. Lancet Public Health 2023, 8, e943–e955. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Guo, S.; Cui, X.; Jiang, M.; Bai, L.; Tian, X.; Guo, T.; Liu, Q.; Zhang, L.; Ho, C.T.; Bai, N. Simultaneous characterization and quantification of 17 main compounds in Rabdosia rubescens by high performance liquid chromatography. J. Food Drug Anal. 2017, 25, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Xu, J.; Liu, B.; Chen, F.; Li, J.; Liu, Y.; Zhu, J.; Shen, C. Ponicidin inhibits pro-inflammatory cytokine TNF-α-induced epithelial-mesenchymal transition and metastasis of colorectal cancer cells via suppressing the AKT/GSK-3β/Snail pathway. Inflammopharmacology 2019, 27, 627–638. [Google Scholar] [CrossRef]
- Cui, W.; Zhang, J.; Wu, D.; Zhang, J.; Zhou, H.; Rong, Y.; Liu, F.; Wei, B.; Xu, X. Ponicidin suppresses pancreatic cancer growth by inducing ferroptosis: Insight gained by mass spectrometry-based metabolomics. Phytomedicine 2022, 98, 153943. [Google Scholar] [CrossRef]
- Liu, Y.F.; Lu, Y.M.; Qu, G.Q.; Liu, Y.; Chen, W.X.; Liao, X.H.; Kong, W.M. Ponicidin induces apoptosis via JAK2 and STAT3 signaling pathways in gastric carcinoma. Int. J. Mol. Sci. 2015, 16, 1576–1589. [Google Scholar] [CrossRef]
- Tretter, V.; Hochreiter, B.; Zach, M.L.; Krenn, K.; Klein, K.U. Understanding Cellular Redox Homeostasis: A Challenge for Precision Medicine. Int. J. Mol. Sci. 2021, 23, 106. [Google Scholar] [CrossRef]
- Kitzman, J.O.; Snyder, M.W.; Ventura, M.; Lewis, A.P.; Qiu, R.; Simmons, L.E.; Gammill, H.S.; Rubens, C.E.; Santillan, D.A.; Murray, J.C.; et al. Noninvasive whole-genome sequencing of a human fetus. Sci. Transl. Med. 2012, 4, 137ra176. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.; Bae, J.S. ROS homeostasis and metabolism: A critical liaison for cancer therapy. Exp. Mol. Med. 2016, 48, e269. [Google Scholar] [CrossRef]
- Slika, H.; Mansour, H.; Wehbe, N.; Nasser, S.A.; Iratni, R.; Nasrallah, G.; Shaito, A.; Ghaddar, T.; Kobeissy, F.; Eid, A.H. Therapeutic potential of flavonoids in cancer: ROS-mediated mechanisms. Biomed. Pharmacother. 2022, 146, 112442. [Google Scholar] [CrossRef]
- Niu, B.; Liao, K.; Zhou, Y.; Wen, T.; Quan, G.; Pan, X.; Wu, C. Application of glutathione depletion in cancer therapy: Enhanced ROS-based therapy, ferroptosis, and chemotherapy. Biomaterials 2021, 277, 121110. [Google Scholar] [CrossRef]
- Wu, J.; Ning, P.; Gao, R.; Feng, Q.; Shen, Y.; Zhang, Y.; Li, Y.; Xu, C.; Qin, Y.; Plaza, G.R.; et al. Programmable ROS-Mediated Cancer Therapy via Magneto-Inductions. Adv. Sci. 2020, 7, 1902933. [Google Scholar]
- Daviu, N.; Portilla, Y.; Gómez de Cedrón, M.; Ramírez de Molina, A.; Barber, D.F. DMSA-coated IONPs trigger oxidative stress, mitochondrial metabolic reprograming and changes in mitochondrial disposition, hindering cell cycle progression of cancer cells. Biomaterials 2024, 304, 122409. [Google Scholar] [PubMed]
- Zhu, X.Y.; Wang, T.Y.; Jia, H.R.; Wu, S.Y.; Gao, C.Z.; Li, Y.H.; Zhang, X.; Shan, B.H.; Wu, F.G. A ferroptosis-reinforced nanocatalyst enhances chemodynamic therapy through dual H2O2 production and oxidative stress amplification. J. Control Release 2024, 367, 892–904. [Google Scholar] [PubMed]
- Bai, D.; Zhou, C.; Du, J.; Zhao, J.; Gu, C.; Wang, Y.; Zhang, L.; Lu, N.; Zhao, Y. TrxR1 is involved in the activation of Caspase-11 by regulating the oxidative-reductive status of Trx-1. Redox Biol. 2024, 75, 103277. [Google Scholar] [CrossRef]
- Bjørklund, G.; Zou, L.; Wang, J.; Chasapis, C.T.; Peana, M. Thioredoxin reductase as a pharmacological target. Pharmacol. Res. 2021, 174, 105854. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, B.; Arnér, E.S.J. Thioredoxin-related protein of 14 kDa as a modulator of redox signalling pathways. Br. J. Pharmacol. 2019, 176, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Guo, Y.; Zhang, W.; Chai, H.; Xing, H.; Xing, M. Neurotoxicity induced by arsenic in Gallus Gallus: Regulation of oxidative stress and heat shock protein response. Chemosphere 2017, 166, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Bhandary, B.; Marahatta, A.; Kim, H.R.; Chae, H.J. An involvement of oxidative stress in endoplasmic reticulum stress and its associated diseases. Int. J. Mol. Sci. 2012, 14, 434–456. [Google Scholar] [CrossRef]







| Gene | Accession Number | Forward Primer | Reverse Primer |
|---|---|---|---|
| BIP | ID: 3309 | CATCACGCCGTCCTATGTCG | CGTCAAAGACCGTGTTCTCG |
| CHOP | ID: 1649 | GGAAACAGAGTGGTCATTCCC | CTGCTTGAGCCGTTCATTCTC |
| PERK | ID: 9451 | TGTCGCCAATGGGATAGTGACGAA | AATCCGGCTCTCGTTTCCATGTCT |
| ATF4 | ID: 468 | CTCCGGGACAGATTGGATGTT | GGCTGCTTATTAGTCTCCTGGAC |
| ATF6 | ID: 22926 | TCCTCGGTCAGTGGACTCTTA | CTTGGGCTGAATTGAAGGTTTTG |
| GAPDH | ID: 2597 | GGAGCGAGATCCCTCCAAAAT | GGCTGTTGTCATACTTCTCATGG |
| IRE1α | ID: 2081 | CACAGTGACGCTTCCTGAAAC | GCCATCATTAGGATCTGGGAGA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Huang, Y.; Liu, Y.; Liao, Z.; Zhang, R.; Sui, X.; Sun, X. Ponicidin Inhibits Lung Cancer Progression Through Coordinated Downregulation of Sulfhydryl Antioxidants and TrxR1. Antioxidants 2026, 15, 100. https://doi.org/10.3390/antiox15010100
Huang Y, Liu Y, Liao Z, Zhang R, Sui X, Sun X. Ponicidin Inhibits Lung Cancer Progression Through Coordinated Downregulation of Sulfhydryl Antioxidants and TrxR1. Antioxidants. 2026; 15(1):100. https://doi.org/10.3390/antiox15010100
Chicago/Turabian StyleHuang, Yufei, Yanfen Liu, Zehua Liao, Ruonan Zhang, Xinbing Sui, and Xueni Sun. 2026. "Ponicidin Inhibits Lung Cancer Progression Through Coordinated Downregulation of Sulfhydryl Antioxidants and TrxR1" Antioxidants 15, no. 1: 100. https://doi.org/10.3390/antiox15010100
APA StyleHuang, Y., Liu, Y., Liao, Z., Zhang, R., Sui, X., & Sun, X. (2026). Ponicidin Inhibits Lung Cancer Progression Through Coordinated Downregulation of Sulfhydryl Antioxidants and TrxR1. Antioxidants, 15(1), 100. https://doi.org/10.3390/antiox15010100







