Normalization of Oxygen Levels Induces a Metabolic Reprogramming in Livers Exposed to Intermittent Hypoxia Mimicking Obstructive Sleep Apnea
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Polygraphic Study
2.3. Inclusion and Exclusion Criteria
2.4. Clinical and Laboratory Assessment
2.5. Serum Metabolomic Analysis
2.6. Animal Care and Intermittent Hypoxia Protocol Followed by Reoxygenation
2.7. Histopathological Evaluation
2.8. Assessment of Lipid Accumulation
2.9. IBA1 (Ionized Calcium-Binding Adapter Molecule 1) Immunohistochemistry
2.10. 4-HNE (4-Hydroxynonenal) Immunohistochemistry
2.11. Analysis of ORO Staining and IHQ Images
2.12. Fatty Acid Oxidation (FAO) Assay
2.13. Quantitative Real-Time PCR (RT-qPCR)
2.14. β-Hydroxybutyrate Assay Kit Assay
2.15. Statistical Analysis
3. Results
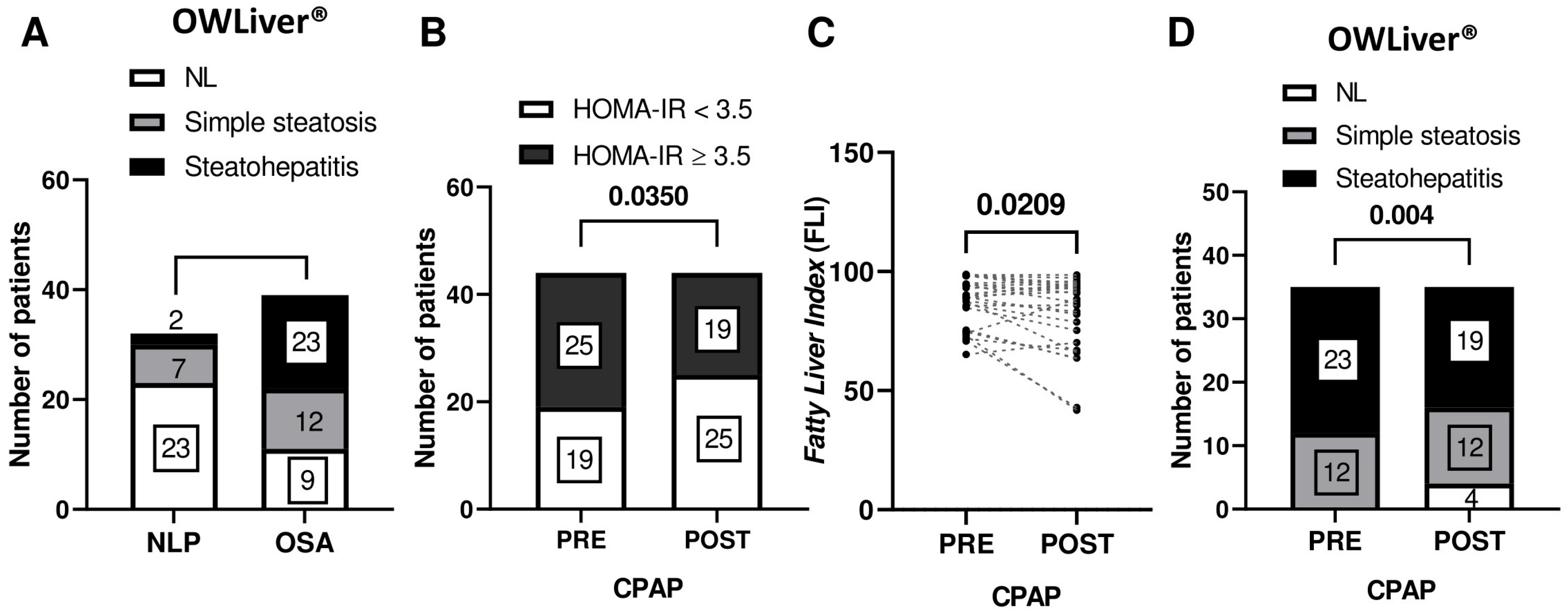
3.1. Characteristics of the Study Population and Prevalence of Metabolic Disorders
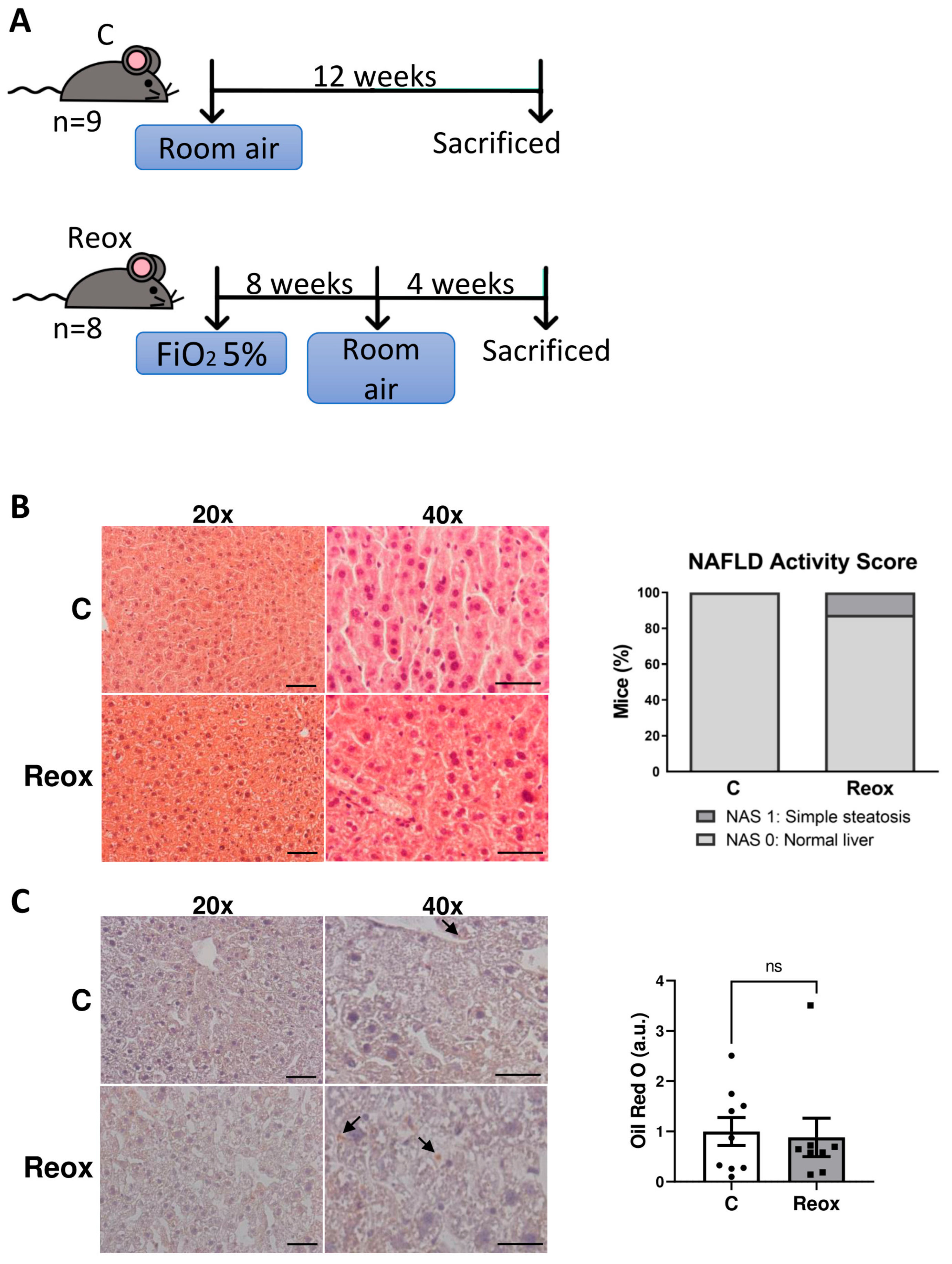
3.2. Normalization of Oxygen Levels Reverses Lipid Accumulation in Livers from Mice Exposed to IH
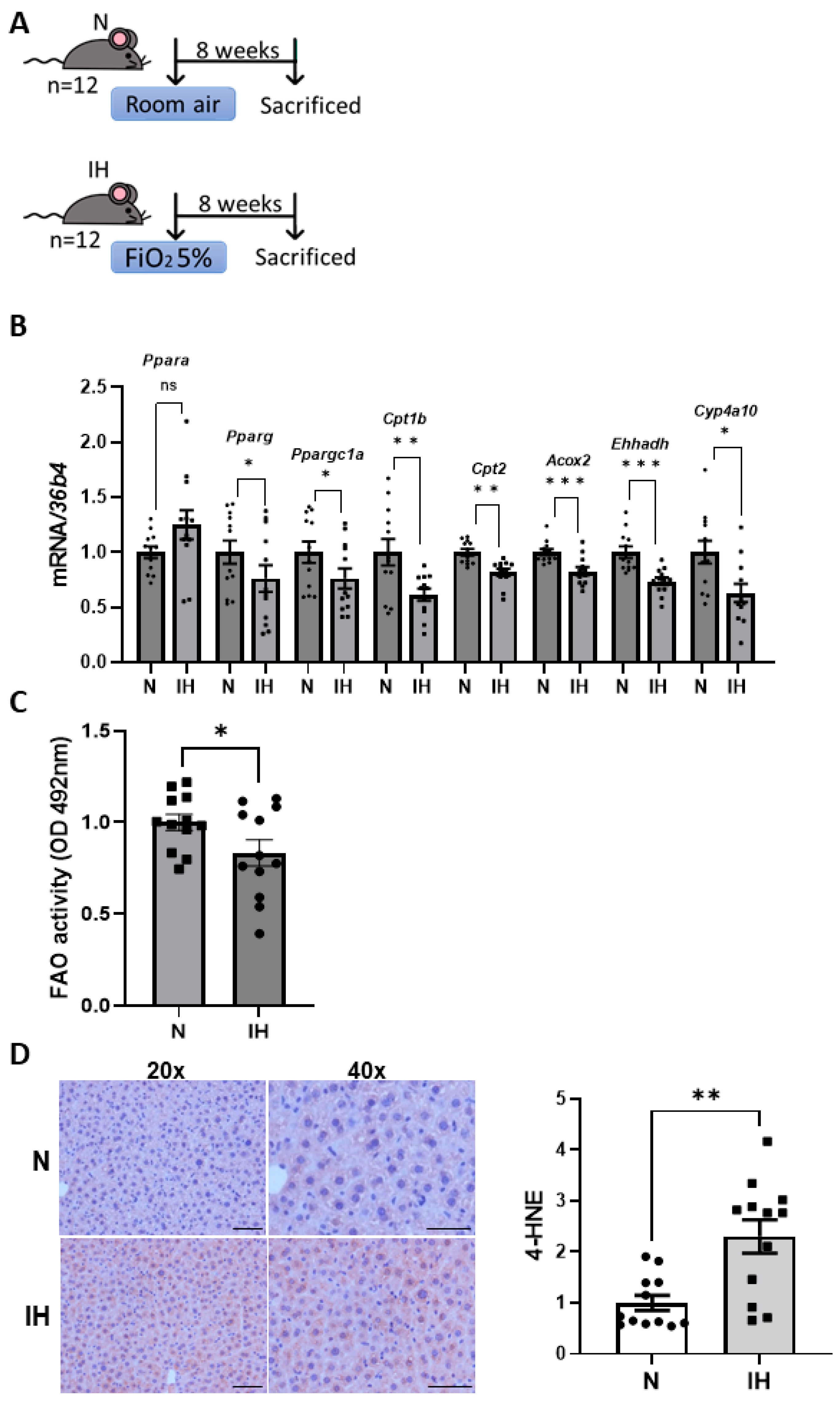
3.3. IH Impairs Liver FAO in Mice, Triggering an Increase in Hepatic Oxidative Stress
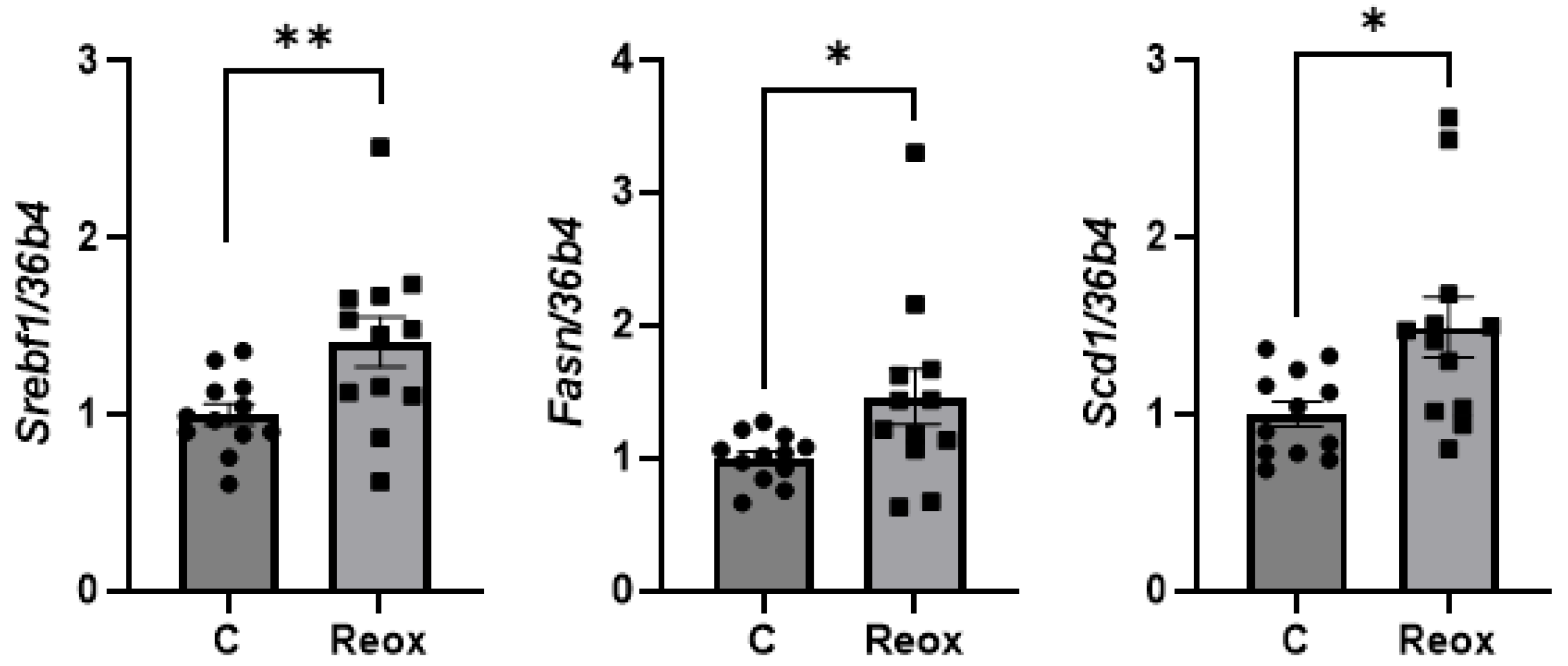
3.4. Reoxygenation Induces a Metabolic Reprogramming and Reduces Oxidative Stress in Livers from Mice Exposed to IH
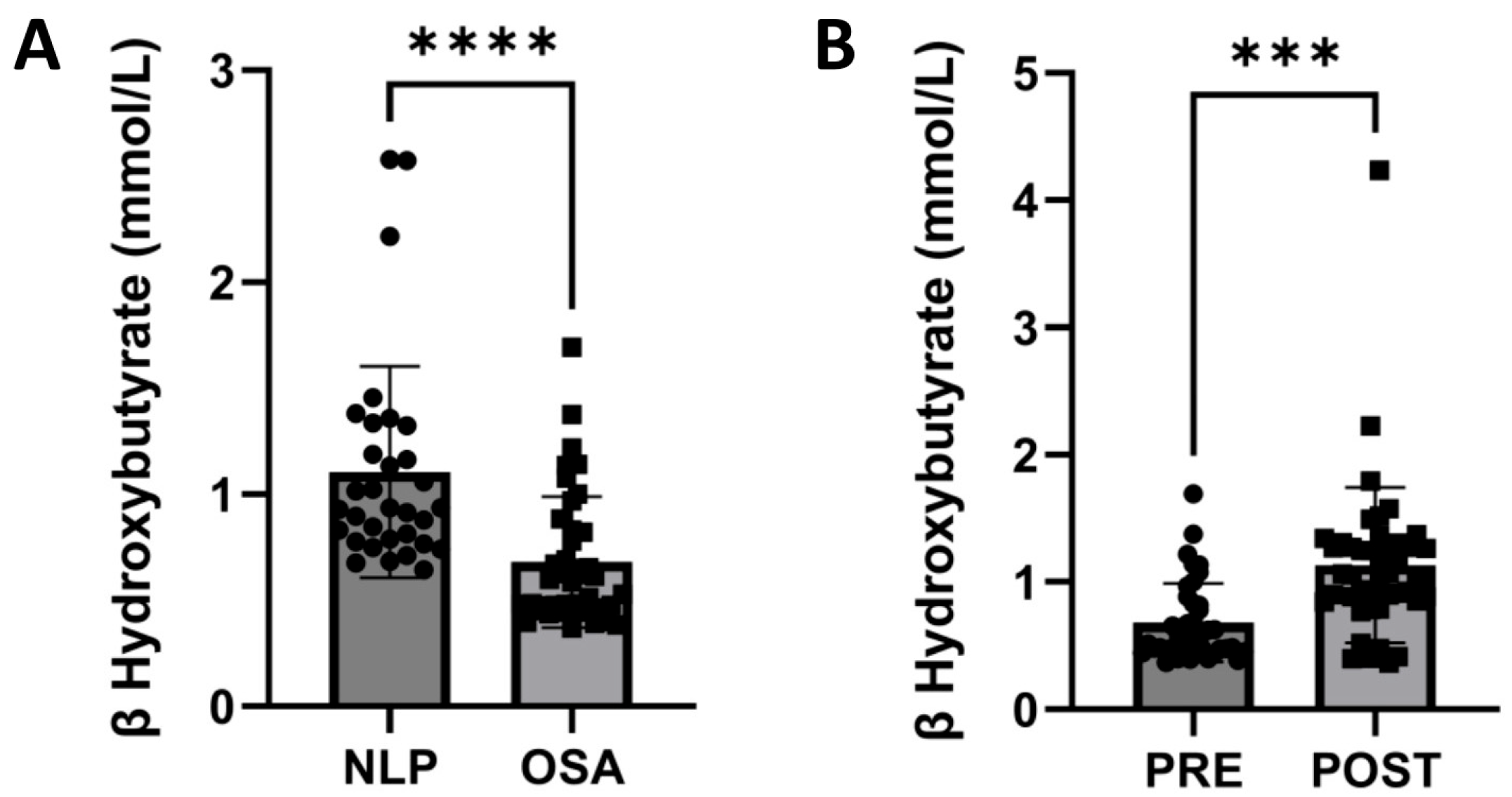
3.5. CPAP Treatment Normalizes Circulating Levels of β-Hydroxybutyrate, a Marker for Hepatic FAO, in OSA Patients
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Senaratna, C.V.; Perret, J.L.; Lodge, C.J.; Lowe, A.J.; Campbell, B.E.; Matheson, M.C.; Hamilton, G.S.; Dharmage, S.C. Prevalence of obstructive sleep apnea in the general population: A systematic review. Sleep Med. Rev. 2017, 34, 70–81. [Google Scholar] [CrossRef]
- Khalyfa, A.; Trzepizur, W.; Gileles-Hillel, A.; Qiao, Z.; Sanz-Rubio, D.; Marin, J.M.; Martinez-Garcia, M.A.; Campos-Rodriguez, F.; Almendros, I.; Farre, R.; et al. Heterogeneity of melanoma cell responses to sleep apnea-derived plasma exosomes and to intermittent hypoxia. Cancers 2021, 13, 4781. [Google Scholar] [CrossRef]
- Kanclerska, J.; Wickiewicz, M.; Nowacki, D.; Szymańska-Chabowska, A.; Poreba, R.; Mazur, G.; Martynowicz, H. Sleep architecture and vitamin D in hypertensives with obstructive sleep apnea: A polysomnographic study. Dent. Med. Probl. 2023, 61, 43–52. [Google Scholar] [CrossRef]
- Karuga, F.F.; Jaromirska, J.; Sochal, M.; Białasiewicz, P.; Gabryelska, A. Association between glucose metabolism, the circadian cycle and hypoxia: Evaluation of the NPAS2 and Rev-Erb-α protein serum levels in obstructive sleep apnea patients–A pilot study. Dent. Med. Probl. 2024, 61, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Stryelkina, M.; Nguyen, M.; Vincent, J.L. The Interplay between Sleep Disorders and MASLD: A Mini Review. J. Sleep Med. Disord. 2025, 9, 1145. [Google Scholar]
- Miao, L.; Targher, G.; Byrne, C.D.; Cao, Y.Y.; Zheng, M.H. Current status and future trends of the global burden of MASLD. Trends Endocrinol. Metab. 2024, 35, 697–707. [Google Scholar] [CrossRef]
- Hashimoto, E.; Taniai, M.; Tokushige, K. Characteristics and diagnosis of NAFLD/NASH. J. Gastroenterol. Hepatol. 2013, 28 (Suppl. S4), 64–70. [Google Scholar] [CrossRef]
- Goh, G.B.-B.; McCullough, A.J. Natural History of Nonalcoholic Fatty Liver Disease. Dig. Dis. Sci. 2016, 61, 1226–1233. [Google Scholar] [CrossRef] [PubMed]
- Badmus, O.O.; Hillhouse, S.A.; Anderson, C.D.; Hinds, T.D.; Stec, D.E. Molecular mechanisms of metabolic associated fatty liver disease (MAFLD): Functional analysis of lipid metabolism pathways. Clin. Sci. 2022, 136, 1347–1366. [Google Scholar] [CrossRef] [PubMed]
- Ipsen, D.H.; Lykkesfeldt, J.; Tveden-Nyborg, P. Molecular mechanisms of hepatic lipid accumulation in non-alcoholic fatty liver disease. Cell. Mol. Life Sci. 2018, 75, 3313–3327. [Google Scholar] [CrossRef]
- Yoon, M. The role of PPARα in lipid metabolism and obesity: Focusing on the effects of estrogen on PPARα actions. Pharmacol. Res. 2009, 60, 151–159. [Google Scholar] [CrossRef]
- Walczak, R.; Tontonoz, P. PPARadigms and PPARadoxes: Expanding roles for PPARγ in the control of lipid metabolism. J. Lipid Res. 2002, 43, 177–186. [Google Scholar] [CrossRef]
- Polotsky, V.Y.; Patil, S.P.; Savransky, V.; Laffan, A.; Fonti, S.; Frame, L.A.; Steele, K.E.; Schweizter, M.A.; Clark, J.M.; Torbenson, M.S.; et al. Obstructive sleep apnea, insulin resistance, and steatohepatitis in severe obesity. Am. J. Respir. Crit. Care Med. 2009, 179, 228–234. [Google Scholar] [CrossRef]
- Patil, S.P.; Ayappa, I.A.; Caples, S.M.; Kimoff, R.J.; Patel, S.R.; Harrod, C.G. Treatment of adult obstructive sleep apnea with positive airway pressure: An American academy of sleep medicine clinical practice guideline. J. Clin. Sleep Med. 2019, 15, 335–343. [Google Scholar] [CrossRef]
- Tang, H.; Lv, F.; Zhang, P.; Liu, J.; Mao, J. The impact of obstructive sleep apnea on nonalcoholic fatty liver disease. Front. Endocrinol. 2023, 14, 1254459. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Bedogni, G.; Bellentani, S.; Miglioli, L.; Masutti, F.; Passalacqua, M.; Castiglione, A.; Tiribelli, C. The fatty liver index: A simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Ampuero, J.; Pais, R.; Aller, R.; Gallego-Durán, R.; Crespo, J.; García-Monzón, C.; Boursier, J.; Vilar, E.; Petta, S.; Zheng, M.-H.; et al. Development and Validation of Hepamet Fibrosis Scoring System–A Simple, Noninvasive Test to Identify Patients With Nonalcoholic Fatty Liver Disease With Advanced Fibrosis. Clin. Gastroenterol. Hepatol. 2020, 18, 216–225.e5. [Google Scholar] [CrossRef]
- Angulo, P.; Hui, J.M.; Marchesini, G.; Bugianesi, E.; George, J.; Farrell, G.C.; Enders, F.; Saksena, S.; Burt, A.D.; Bida, J.P.; et al. The NAFLD fibrosis score: A noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology 2007, 45, 846–854. [Google Scholar] [CrossRef]
- Iruzubieta, P.; Mayo, R.; Mincholé, I.; Martínez-Arranz, I.; Arias-Loste, M.T.; Ibañez-Samaniego, L.; Ampuero, J.; Abad, J.; Martín-Mateos, R.; Fernández-Laso, A.B.; et al. One-step non-invasive diagnosis of metabolic dysfunction-associated steatohepatitis and fibrosis in high-risk population. United Eur. Gastroenterol. J. 2024, 12, 919–929. [Google Scholar] [CrossRef]
- Almendros, I.; Montserrat, J.M.; Torres, M.; Bonsignore, M.R.; Chimenti, L.; Navajas, D.; Farré, R. Obesity and intermittent hypoxia increase tumor growth in a mouse model of sleep apnea. Sleep Med. 2012, 13, 1254–1260. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.-C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Rey, E.; Meléndez-Rodríguez, F.; Marañón, P.; Gil-Valle, M.; Carrasco, A.G.; Torres-Capelli, M.; Chávez, S.; del Pozo-Maroto, E.; de Cía, J.R.; Aragonés, J.; et al. Hypoxia-inducible factor 2α drives hepatosteatosis through the fatty acid translocase CD36. Liver Int. 2020, 40, 2553–2567. [Google Scholar] [CrossRef]
- Rey, E.; del Pozo-Maroto, E.; Marañón, P.; Beeler, B.; García-García, Y.; Landete, P.; Isaza, S.C.; Farré, R.; García-Monzón, C.; Almendros, I.; et al. Intrahepatic Expression of Fatty Acid Translocase CD36 Is Increased in Obstructive Sleep Apnea. Front. Med. 2020, 7, 450. [Google Scholar] [CrossRef]
- Arroyave-Ospina, J.C.; Wu, Z.; Geng, Y.; Moshage, H. Role of Oxidative Stress in the Pathogenesis of Non-Alcoholic Fatty Liver Disease: Implications for Prevention and Therapy. Antioxidants 2021, 10, 174. [Google Scholar] [CrossRef]
- Tilg, H.; Adolph, T.E.; Dudek, M.; Knolle, P. Non-alcoholic fatty liver disease: The interplay between metabolism, microbes and immunity. Nat. Metab. 2021, 3, 1596–1607. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.P.; Shryack, G.; Alessi, I.; Wieschhaus, N.; Meers, G.M.; Johnson, S.A.; Wheeler, A.A.; Ibdah, J.A.; Parks, E.J.; Rector, R.S. Relationship between serum b-hydroxybutyrate and hepatic fatty acid oxidation in individuals with obesity and NAFLD. Am. J. Physiol. Endocrinol. Metab. 2024, 326, E493–E502. [Google Scholar] [CrossRef]
- Mesarwi, O.A.; Loomba, R.; Malhotra, A. Obstructive sleep apnea, hypoxia, and nonalcoholic fatty liver disease. Am. J. Respir. Crit. Care Med. 2019, 199, 830–841. [Google Scholar] [CrossRef]
- Jullian-Desayes, I.; Trzepizur, W.; Boursier, J.; Joyeux-Faure, M.; Bailly, S.; Benmerad, M.; Le Vaillant, M.; Jaffre, S.; Pigeanne, T.; Bizieux-Thaminy, A.; et al. Obstructive sleep apnea, chronic obstructive pulmonary disease and NAFLD: An individual participant data meta-analysis. Sleep Med. 2021, 77, 357–364. [Google Scholar] [CrossRef]
- Musso, G.; Cassader, M.; Olivetti, C.; Rosina, F.; Carbone, G.; Gambino, R. Association of obstructive sleep apnoea with the presence and severity of non-alcoholic fatty liver disease. A systematic review and meta-analysis. Obes. Rev. 2013, 14, 417–431. [Google Scholar] [CrossRef]
- Cattazzo, F.; Pengo, M.F.; Giontella, A.; Soranna, D.; Bilo, G.; Zambon, A.; Karalliedde, J.; Gnudi, L.; Martinez-Garcia, M.Á.; Minuz, P.; et al. Effect of Continuous Positive Airway Pressure on Glucose and Lipid Profiles in Patients With Obstructive Sleep Apnoea: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Arch. Bronconeumol. 2023, 59, 370–376. [Google Scholar] [CrossRef]
- Gowda, S.; Desai, P.B.; Hull, V.V.; Math, A.A.K.; Vernekar, S.N.; Kulkarni, S.S. A review on laboratory liver function tests. Pan Afr. Med. J. 2009, 3, 17. [Google Scholar]
- Friščić, T.; Perčić, M.; Vidović, D.; Štajduhar, A.; Galić, E. Impact of CPAP Therapy on New Inflammation Biomarkers. J. Clin. Med. 2022, 11, 6113. [Google Scholar] [CrossRef]
- Aron-Wisnewsky, J.; Minville, C.; Tordjman, J.; Lévy, P.; Bouillot, J.-L.; Basdevant, A.; Bedossa, P.; Clément, K.; Pépin, J.-L. Chronic intermittent hypoxia is a major trigger for non-alcoholic fatty liver disease in morbid obese. J. Hepatol. 2012, 56, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Bettini, S.; Serra, R.; Fabris, R.; Prà, C.D.; Favaretto, F.; Dassie, F.; Duso, C.; Vettor, R.; Busetto, L. Association of obstructive sleep apnea with non-alcoholic fatty liver disease in patients with obesity: An observational study. Eat. Weight. Disord. 2022, 27, 335–343. [Google Scholar] [CrossRef]
- Ng, S.S.S.; Wong, V.W.S.; Wong, G.L.H.; Chu, W.C.W.; Chan, T.-O.; To, K.-W.; Ko, F.W.S.; Chan, K.-P.; Hui, D.S. Continuous positive airway pressure does not improve nonalcoholic fatty liver disease in patients with obstructive sleep apnea: A randomized clinical trial. Am. J. Respir. Crit. Care Med. 2021, 203, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Grigoryev, D.N.; Ye, S.Q.; Thorne, L.; Schwartz, A.R.; Smith, P.L.; O’DOnnell, C.P.; Polotsky, V.Y. Chronic intermittent hypoxia upregulates genes of lipid biosynthesis in obese mice. J. Appl. Physiol. 2005, 99, 1643–1648. [Google Scholar] [CrossRef] [PubMed]
- Quintero, M.; Gonzalez-Martin, M.d.C.; Vega-Agapito, V.; Gonzalez, C.; Obeso, A.; Farré, R.; Agapito, T.; Yubero, S. The effects of intermittent hypoxia on redox status, NF-κB activation, and plasma lipid levels are dependent on the lowest oxygen saturation. Free Radic. Biol. Med. 2013, 65, 1143–1154. [Google Scholar] [CrossRef]
- Gaucher, J.; Vial, G.; Montellier, E.; Guellerin, M.; Bouyon, S.; Lemarie, E.; Pelloux, V.; Bertrand, A.; Pernet-Gallay, K.; Lamarche, F.; et al. Intermittent Hypoxia Rewires the Liver Transcriptome and Fires up Fatty Acids Usage for Mitochondrial Respiration. Front. Med. 2022, 9, 829979. [Google Scholar] [CrossRef]
- Zhang, Q.; Xiang, S.; Liu, Q.; Gu, T.; Yao, Y.; Lu, X. PPAR antagonizes hypoxia-induced activation of hepatic stellate cell through cross mediating PI3K/AKT and cGMP/PKG signaling. PPAR Res. 2018, 2018, 6970407. [Google Scholar] [CrossRef]
- Knight, B.L.; Hebbachi, A.; Hauton, D.; Brown, A.-M.; Wiggins, D.; Patel, D.D.; Gibbons, G.F. A role for PPARα in the control of SREBP activity and lipid synthesis in the liver. Biochem. J. 2005, 389, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, N.; Friedman, S.L.; Goossens, N.; Hoshida, Y. Risk factors and prevention of hepatocellular carcinoma in the era of precision medicine. J. Hepatol. 2018, 68, 526–549. [Google Scholar] [CrossRef] [PubMed]
- Enooku, K.; Nakagawa, H.; Fujiwara, N.; Kondo, M.; Minami, T.; Hoshida, Y.; Shibahara, J.; Tateishi, R.; Koike, K. Altered serum acylcarnitine profile is associated with the status of nonalcoholic fatty liver disease (NAFLD) and NAFLD-related hepatocellular carcinoma. Sci. Rep. 2019, 9, 10663. [Google Scholar] [CrossRef] [PubMed]

| Gene | Accession Number | Forward (5′ → 3′) | Reverse (5′ → 3′) |
|---|---|---|---|
| m-36b4 | NM_007475.5 | AGATGCAGCAGATCCGCAT | GTTCTTGCCATCAGCACC |
| m-Srebf1 | NM_011480.4 | GGCCGAGATGTGCGAACT | AGCTGGAGCATGTCTTCGATG |
| m-Fasn | NM_007988.3 | CCCTTGATGAAGAGGGATCA | ACTCCACAGGTGGGAACAAG |
| m-Scd1 | NM_009127.4 | TTCTTGCGATACACTCTGGTGC | CGGGATTGAATGTTCTTGTCGT |
| m-Ppara | NM_011144.6 | AGAGCCCCATCTGTCCTCTC | ACTGGTAGTCTGCAAAACCAAA |
| m-Pparg | NM_001308354.2 | TCGCTGATGCACTGCCTATG | GAGAGGTCCACAGAGCTGATT |
| m-Ppargc1a | NM_008904.3 | AAGTGTGGAACTCTCTGGAACTG | GGGTTATCTTGGTTGGCTTTATG |
| m-Cpt1b | NM_009948.2 | CTCCATGGGACTGGTCGATT | CCAAAGTGGCCATACCTTTCC |
| m-Cpt2 | NM_009949.3 | GAATGACAGCCAGTTCAGGAAGA | CCTTCCCAATGCCGTTCTC |
| m-Acox2 | NM_053115.2 | TTGATGGAGGCATCCCAAA | CACGTTGGATGAGGCTTTCA |
| m-Ehhadh | NM_023737.3 | CCCACTCCCTGGCTATGATC | CAGTTGGACTGATGGCATTGA |
| m-Cyp4a10 | NM_010011.3 | CACTGGTTCTTTGGGCATGA | TCTCTATGCACGACACAATTTCCT |
| Control (N = 32) | OSA (N = 44) | p-Value | |
|---|---|---|---|
| Age (years) | 54.38 ± 8.53 | 59.36 ± 9.04 | 0.0174 |
| Male sex, n (%) | 12 (37.5) | 30 (68.2) | 0.0079 |
| Body mass index (kg/m2) | 28.91 ± 5.54 | 30.23 ± 6.14 | 0.3374 |
| AHI (h−1) | 2.12 ± 1.31 | 41.11 ± 19.71 | <0.0001 |
| Glucose (mg/dL) | 97.38 ± 13.38 | 103.40 ± 16.09 | 0.0902 |
| HbA1c (%) | 5.62 ± 0.53 | 5.71 ± 0.48 | 0.4435 |
| HOMA-IR | 2.93 ± 2.28 | 4.76 ± 3.52 | 0.0120 |
| Triglycerides (mg/dL) | 106.47 ± 66.57 | 129.80 ± 75.76 | 0.0399 |
| Total cholesterol (mg/dL) | 196.81 ± 39.17 | 188.50 ± 38.21 | 0.3585 |
| HDL (mg/dL) | 57.53 ± 12.96 | 53.93 ± 19.13 | 0.0617 |
| LDL (mg/dL) | 115.22 ± 30.75 | 108.30 ± 35.51 | 0.3780 |
| VLDL (mg/dL) | 21.25 ± 13.25 | 26.20 ± 15.04 | 0.0339 |
| ALT (IU/L) | 18.78 ± 7.34 | 23.52 ± 9.85 | 0.0116 |
| AST (IU/L) | 20.28 ± 5.61 | 22.00 ± 7.21 | 0.2650 |
| GGT (IU/L) | 22.19 ± 13.03 | 30.20 ± 16.23 | 0.0240 |
| LDH (IU/L) | 182.72 ± 36.12 | 182.20 ± 34.82 | 0.9478 |
| ALP (IU/L) | 69.63 ± 22.69 | 68.05 ± 20.64 | 0.7530 |
| Total bilirubin (IU/L) | 0.57 ± 0.48 | 0.57 ± 0.32 | 0.340 |
| Albumin (g/dL) | 4.38 ± 0.27 | 4.41 ± 0.25 | 0.5677 |
| Platelets (109/L) | 226.31 ± 71.27 | 234.10 ± 51.73 | 0.5828 |
| Iron (µg/dL) | 78.47 ± 33.50 | 87.66 ± 33.22 | 0.2391 |
| C-Reactive Protein (mg/L) | 0.42 ± 0.59 | 0.31 ± 0.24 | 0.9979 |
| FLI | 46.33 ± 28.65 | 66.26 ± 29.25 | 0.0042 |
| NAFLD FS | −1.43 ± 1.29 | −1.14 ± 1.03 | 0.2706 |
| HFS | 0.06 ± 0.07 | 0.07 ± 0.10 | 0.4614 |
| Features | OSA (N = 44) | p-Value | |
|---|---|---|---|
| Pre-CPAP (N = 44) | Post-CPAP (N = 44) | ||
| Age (years) | 59.36 ± 9.04 | 61.20 ± 9.19 | <0.0001 |
| Male sex, n (%) | 30 (68.2) | 30 (68.2) | 1.0000 |
| Body mass index (kg/m2) | 30.23 ± 6.14 | 30.12 ± 5.25 | 0.7755 |
| AHI (h−1) | 41.11 ± 19.71 | 7.12 ± 15.73 | <0.0001 |
| Glucose (mg/dL) | 103.40 ± 16.09 | 105.30 ± 18.40 | 0.3246 |
| HbA1c (%) | 5.71 ± 0.48 | 5.64 ± 0.47 | 0.1613 |
| HOMA-IR | 4.76 ± 3.52 | 4.86 ± 4.08 | 0.7838 |
| Triglycerides (mg/dL) | 129.80 ± 75.76 | 112.20 ± 56.74 | 0.0656 |
| Total cholesterol (mg/dL) | 188.50 ± 38.21 | 175.30 ± 36.08 | 0.0044 |
| HDL (mg/dL) | 53.93 ± 19.13 | 53.64 ± 13.16 | 0.0663 |
| LDL (mg/dL) | 108.30 ± 35.51 | 97.66 ± 34.61 | 0.0799 |
| VLDL (mg/dL) | 26.20 ± 15.04 | 22.45 ± 11.31 | 0.0304 |
| ALT (IU/L) | 23.52 ± 9.85 | 23.11 ± 10.27 | 0.3311 |
| AST (IU/L) | 22.00 ± 7.21 | 19.68 ± 6.35 | 0.0088 |
| GGT (IU/L) | 30.20 ± 16.23 | 29.84 ± 16.86 | 0.6819 |
| LDH (IU/L) | 182.20 ± 34.82 | 169.40 ± 34.55 | 0.0002 |
| ALP (IU/L) | 68.05 ± 20.64 | 74.93 ± 24.97 | 0.0011 |
| Total bilirubin (IU/L) | 0.57 ± 0.32 | 0.57 ± 0.29 | 0.8502 |
| Albumin (g/dL) | 4.41 ± 0.25 | 4.61 ± 0.21 | <0.0001 |
| Platelets (109/L) | 234.10 ± 51.73 | 230.30 ± 52.06 | 0.4296 |
| Iron (µg/dL) | 87.66 ± 33.22 | 93.73 ± 29.10 | 0.2265 |
| C-Reactive Protein (mg/L) | 0.31 ± 0.24 | 0.24 ± 0.19 | 0.0599 |
| FLI | 66.26 ± 29.25 | 64.62 ± 28.14 | 0.3828 |
| NAFLD FS | −1.14 ± 1.03 | −1.22 ± 1.20 | 0.4821 |
| Hepamet FS | 0.07 ± 0.10 | 0.07 ± 0.09 | 0.7298 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-García, M.Á.; Aldave-Orzáiz, B.; Fernández-García, C.E.; Fuertes-Yebra, E.; Rey, E.; Berlana, Á.; Farré, R.; García-Monzón, C.; Almendros, I.; Landete, P.; et al. Normalization of Oxygen Levels Induces a Metabolic Reprogramming in Livers Exposed to Intermittent Hypoxia Mimicking Obstructive Sleep Apnea. Antioxidants 2025, 14, 971. https://doi.org/10.3390/antiox14080971
Hernández-García MÁ, Aldave-Orzáiz B, Fernández-García CE, Fuertes-Yebra E, Rey E, Berlana Á, Farré R, García-Monzón C, Almendros I, Landete P, et al. Normalization of Oxygen Levels Induces a Metabolic Reprogramming in Livers Exposed to Intermittent Hypoxia Mimicking Obstructive Sleep Apnea. Antioxidants. 2025; 14(8):971. https://doi.org/10.3390/antiox14080971
Chicago/Turabian StyleHernández-García, Miguel Á., Beatriz Aldave-Orzáiz, Carlos Ernesto Fernández-García, Esther Fuertes-Yebra, Esther Rey, Ángela Berlana, Ramón Farré, Carmelo García-Monzón, Isaac Almendros, Pedro Landete, and et al. 2025. "Normalization of Oxygen Levels Induces a Metabolic Reprogramming in Livers Exposed to Intermittent Hypoxia Mimicking Obstructive Sleep Apnea" Antioxidants 14, no. 8: 971. https://doi.org/10.3390/antiox14080971
APA StyleHernández-García, M. Á., Aldave-Orzáiz, B., Fernández-García, C. E., Fuertes-Yebra, E., Rey, E., Berlana, Á., Farré, R., García-Monzón, C., Almendros, I., Landete, P., & González-Rodríguez, Á. (2025). Normalization of Oxygen Levels Induces a Metabolic Reprogramming in Livers Exposed to Intermittent Hypoxia Mimicking Obstructive Sleep Apnea. Antioxidants, 14(8), 971. https://doi.org/10.3390/antiox14080971





