Antioxidant Biomaterials in Cutaneous Wound Healing and Tissue Regeneration: A Critical Review
Abstract
1. Introduction
2. Data Extraction Management
3. Oxidative Stress
3.1. Wound Healing
3.2. Importance of Redox Regulation in Normal Wound Healing
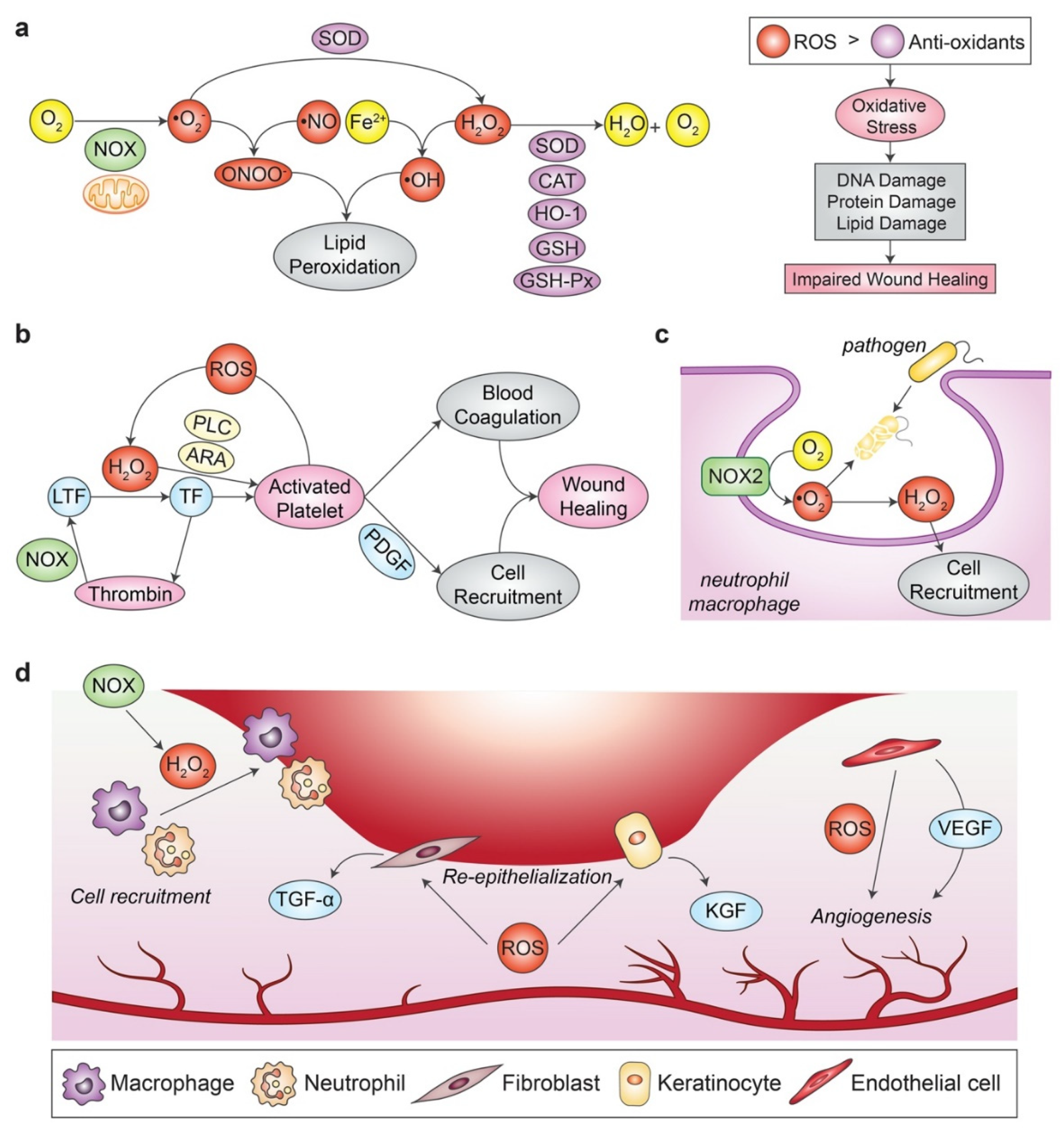
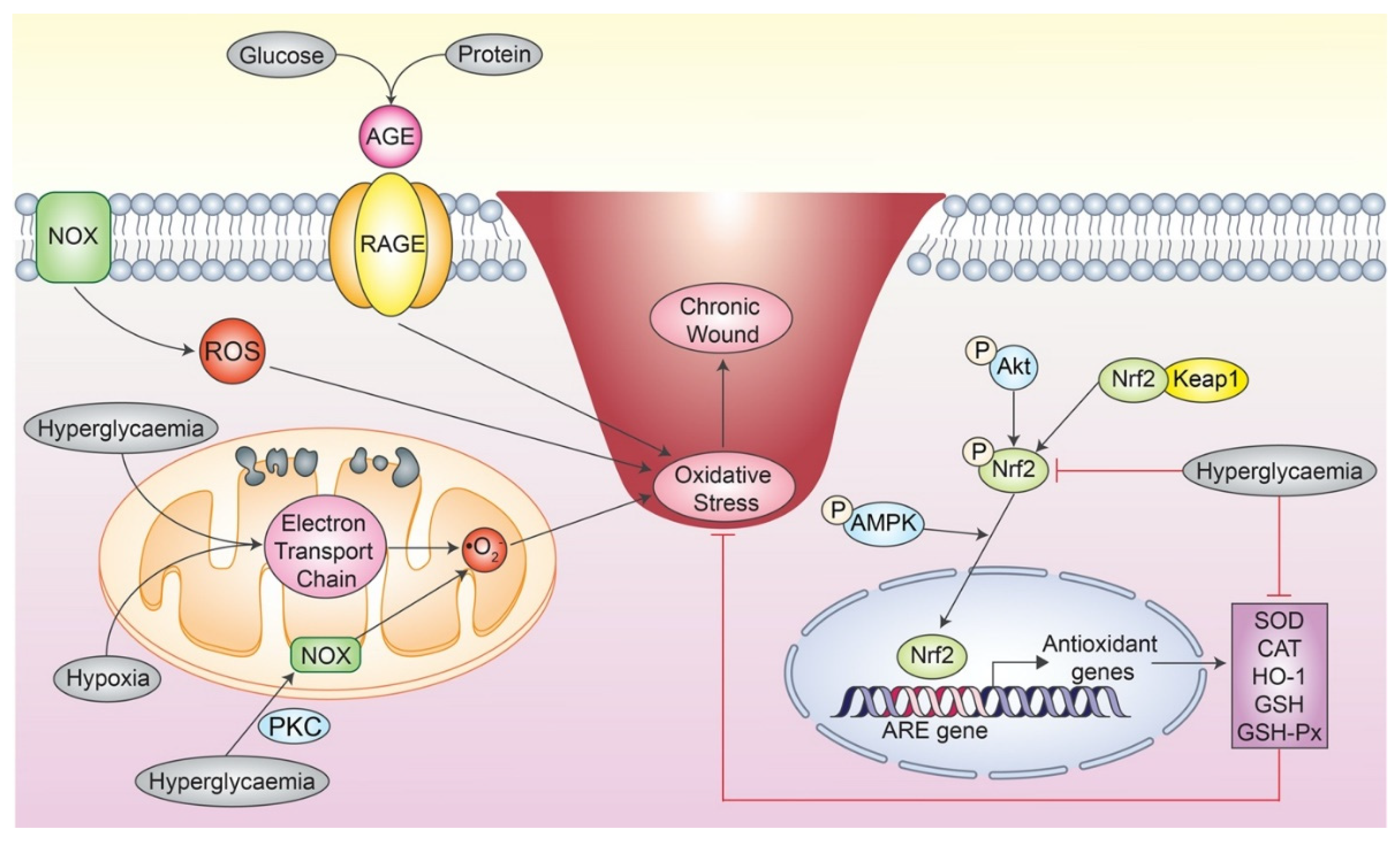
3.3. Impact of Oxidative Stress in Chronic Wound
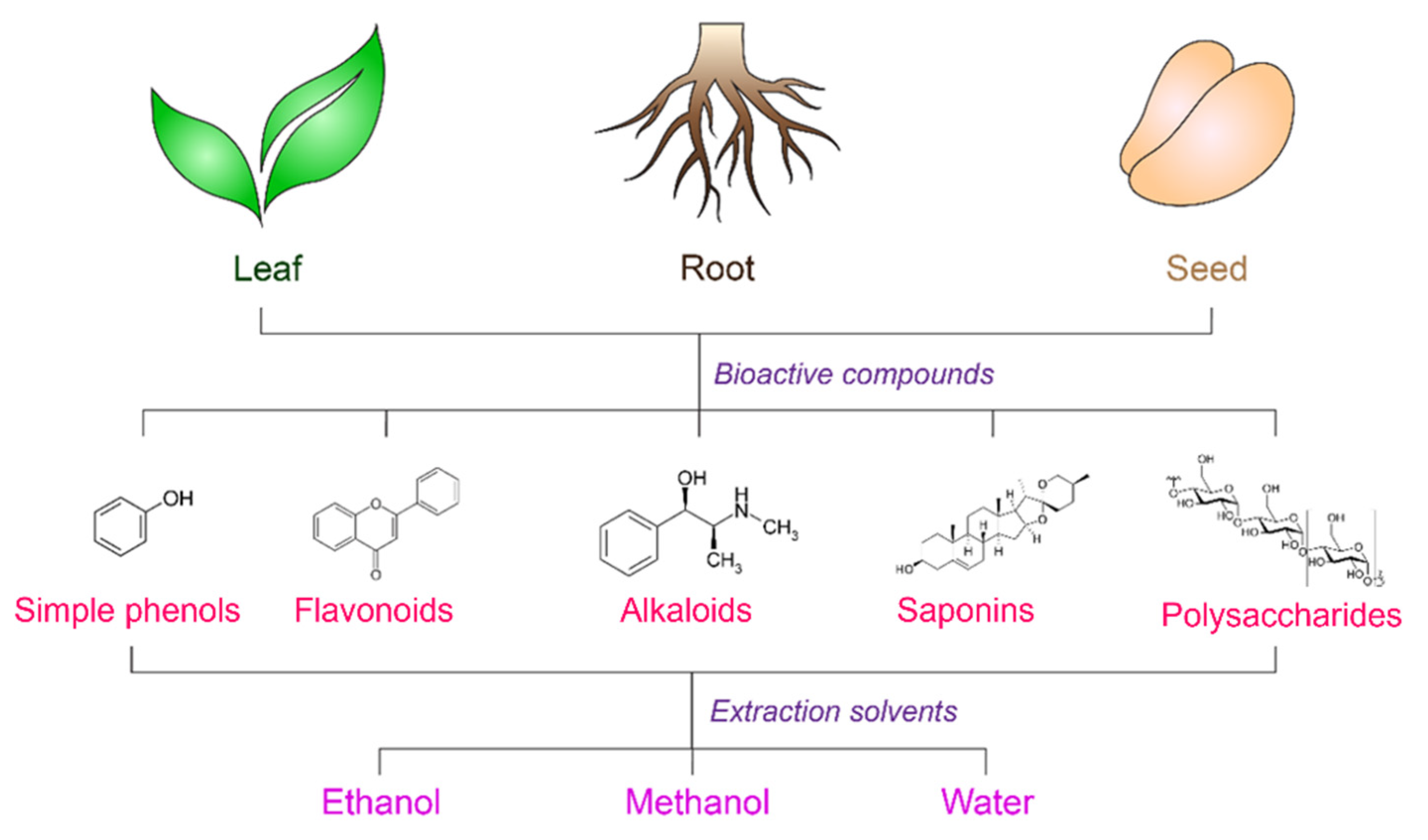
4. Natural Antioxidants
4.1. Extraction Methods
| Part of Plant | Compound(s) | Source(s) | Extraction Method | Biological Properties | Reference |
|---|---|---|---|---|---|
| Leaves | Saponins | Algerian urtica dioica (neetle) | Methanolic extract | Antibacterial and antioxidant | [71] |
| Marrubin, phenol and flavonoid | Marrubium vulgare | Methanolic extract; Microwave assisted solvent extraction (MASE) | Antihypertensive, analgesic, anti-inflammatory, hypoglycaemic, vasodilator, antidiabetic, and antibacterial | [61] | |
| Flavanoid (quercetin-3-O-rutinoside and catechin) | Zizyphus lotus | Methanolic extract | Antidiabetic, sedative and hypoglycaemic | [72] | |
| Phenol and flavonoid | Trigonella foenum-graecum (Fenugreek), Cassia acutifolia (Senna) and Rhazya stricta (Harmal) | Ethanolic extract | Antioxidant, anti-lipoxygenase and anticancer | [73] | |
| Tannin, saponin, flavonoid | Sarasinula marginata | Ethanolic extract | Antioxidant, responsible for slower release profile process | [74] | |
| Flavonoids, terpenoids and phenolic compounds | Melia azedarach | Water extract | Antibacterial, antidiabetic, and antioxidant | [75] | |
| Phenolic (caffeoylquinic acids) | Dittrichia viscosa | Ethanolic extract | Antiradical and antioxidant | [76] | |
| Retinol and alpha tocopherol | Rosemary and clove | Dried plants | Antioxidant and flavouring agent | [77] | |
| Vitamins, amino acids and anthraquinones, glucomannan | Aloe vera | Commercially available | Anti-inflammatory, antioxidant, antibacterial, control release, biocompatible, biomechanical stability, cell proliferation, attachment, re-epithelisation, angiogenesis, and high water uptake | [63,78,79,80,81] | |
| Epigallocatechin gallate (EGCG) | Green tea | Methane sulfonic acid and tetrahydrofuran | Anti-inflammation, and radical scavenger effects | [82] | |
| Alkaloids, polyphenols, phenolic acids, a range of flavonoids, and glusinolates | Moringa oleifera | water and methanol extract | Anti-inflammatory, antioxidant, and antimicrobial | [83,84] | |
| Coumarin compound named ostruthol | Peucedanum ostruthium | Ethanol and water extract | Anti-inflammatory, antibacterial | [64] | |
| Carotenoid, fucoxanthin, astaxanthin, and phenolic compounds such as tannins, flavonoid and phenolic acid | Eucheuma cottonii extract (red seaweed) | Water extract | Antioxidant, and high-gelling properties | [85] | |
| Fruit | Alkaloids | Berberine | Analytical grade | Biocompatible, proliferation, and antibacterial property | [86] |
| Lignin | Coconut husk | Ethanol and water extract | UV protective agent | [62] | |
| Flavonoids | Capparis spinosa fruit | Ethyl alcohol | Antioxidant | [87] | |
| Polyphenolic compounds, phenolic derivatives, flavonoids, and pectin, Vitamin C | Cydonia oblonga fruit (Quince fruit) | Ethanolic extract | Antibacterial, anti-inflammatory, anticancer, anti-bacteria and cardioprotective properties | [88] | |
| Seed | Tannic acid and picotannic acid, pyrogallic acid, gratanotannic acid, resin, and mucilage, alkaloids | Pomegranate seed | Ethanolic extract | Antibacterial, anti-inflammatory, and antioxidant | [65] |
| Fenugreek absolute (trigonelline and nicotinic acid) | Trigonella foenum graecum | Methanolic extract | Hypoglycaemic effect, hypocholesterolaemic activity and anti-ulcerogenic effects | [89] | |
| Silymarin | Milk thistle plant (Silybum marianum) | Commercially available | Antioxidant, antimicrobial | [90] | |
| Bixin | Seed | Ethanolic extract | Antioxidant, anti-inflammatory, and hypoglycaemic effects | [91] | |
| Fatty acid, tocopherol | Soybean | Analytical grade | Antibacterial, antioxidant, and anti-inflammatory | [92] |
4.2. Advantages of Natural Antioxidants Properties on Wound Healing
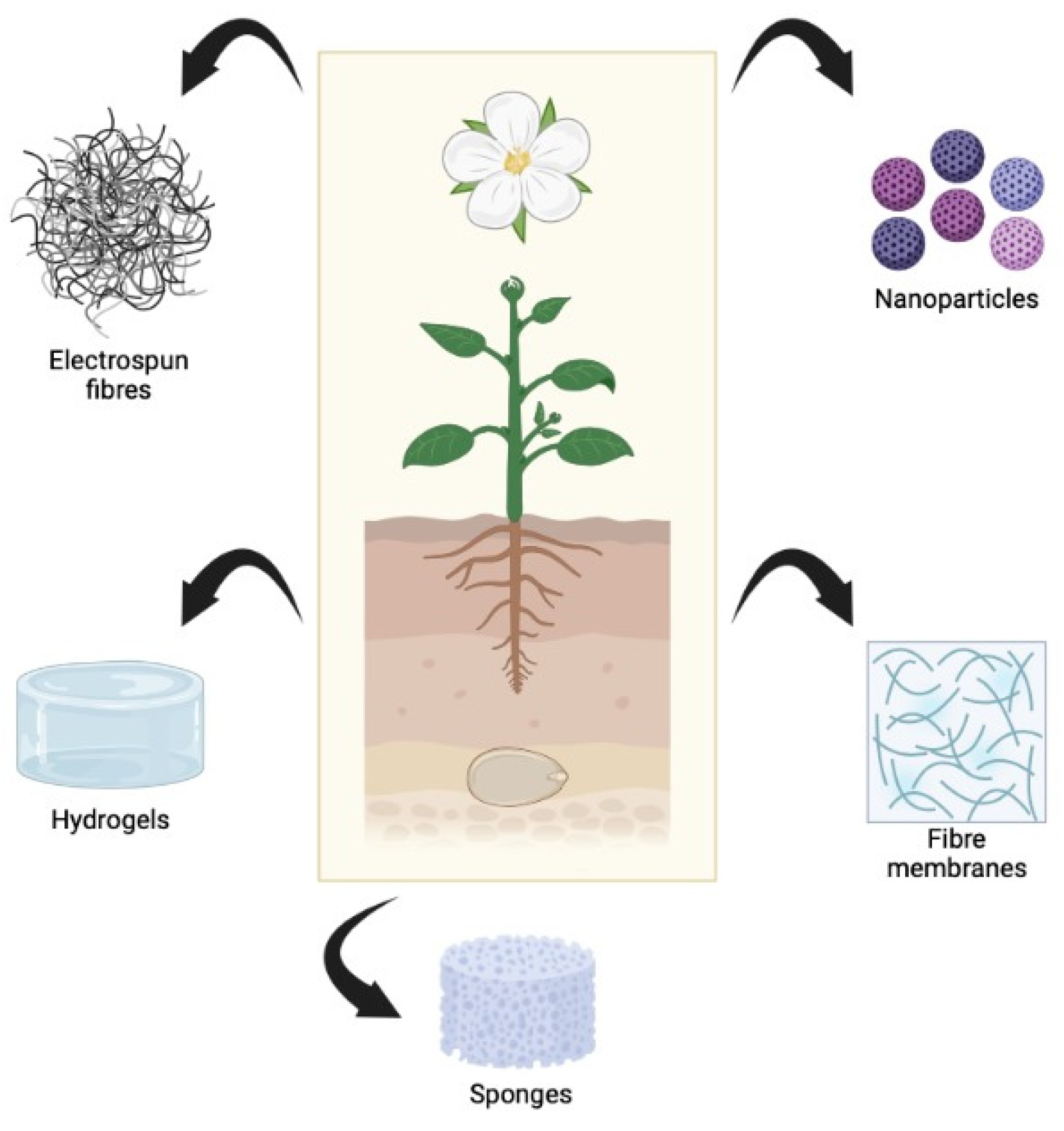
5. Incorporation of Natural Antioxidants into Biomaterials
5.1. Types of 3D-Biomaterials
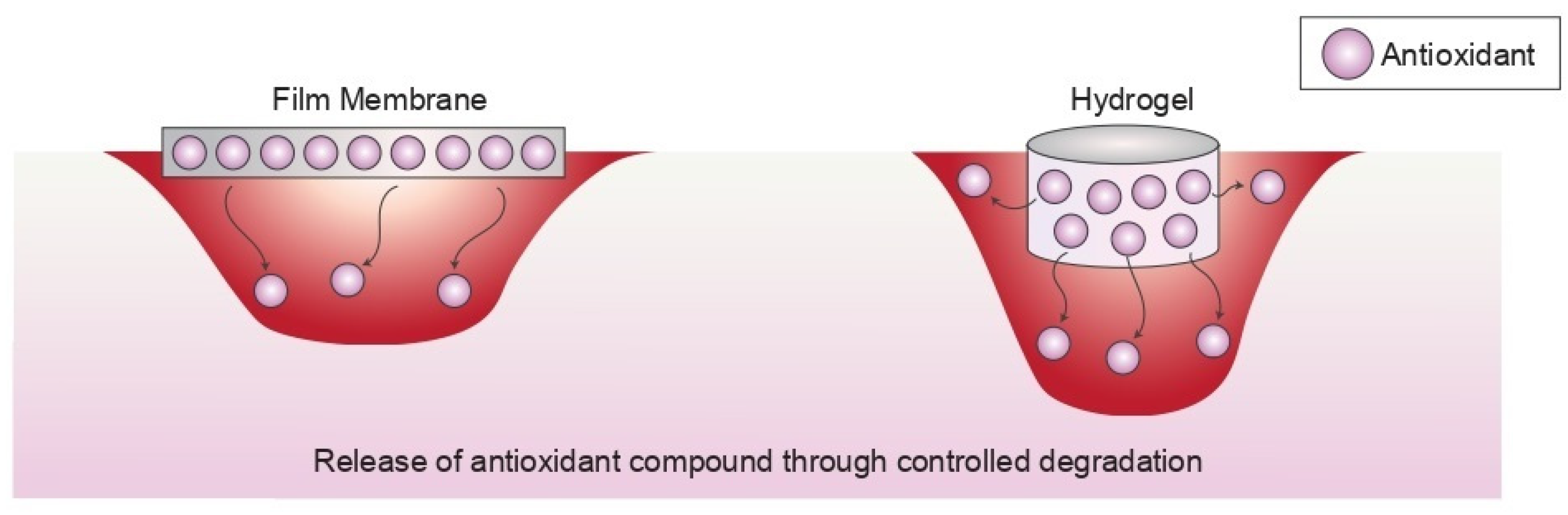
5.1.1. Film/Membrane
5.1.2. Hydrogels
5.1.3. Electrospinning Fibre
5.1.4. Sponge
5.1.5. Nanomaterial/Particles
| Biomaterial | Antioxidant Component | Biological Effect | Study Design | Reference |
|---|---|---|---|---|
| Hydrogel | Gallic Acid | Antioxidant | In vitro and in vivo | [122] |
| Chitosan Sodium alginate | Antioxidant, anti-inflammatory, and migration-promoting effects | In vitro and in vivo | [145] | |
| 5-hydroxymethylfurfural | Anti-inflammation and anti-bacterial | In vitro and in vivo | [123] | |
| Curcumin | Anti-bacterial | In vitro and in vivo | [146,147] | |
| Vitamin E | Antioxidant | In vitro and in vivo | [148] | |
| Acacia gum | Non-haemolytic, antioxidant and mucoadhesive | In vitro and in vivo | [125] | |
| Cannabis sativa | Anti-inflammation, analgesic effects, antioxidant and anti-bacterial | In vitro | [126] | |
| Humic acid | Anti-inflammation and antioxidant | In vitro | [149] | |
| Propenoic acid | Antioxidant and anti-microbial | In vitro and in vivo | [150] | |
| Olive leaves | Antioxidant, anti-inflammation and anti-microbial | In vitro and ex vivo | [151] | |
| Tannic acid | Antioxidant and anti-bacterial | In vitro and in vivo | [152] | |
| Nanogel | Lignin | Antioxidant | In vitro and in vivo | [62] |
| Films/Membrane | Quercetin | Antioxidant | In vitro and in vivo | [153] |
| Sarasinula marginata extract | Antioxidant | In vitro and in vivo | [74] | |
| Ferulic acid | Antioxidant and anti-bacterial | In vitro | [154] | |
| Plantago lanceolata | Antioxidant | In vitro and in vivo | [109] | |
| Tagates patula | Antioxidant and anti-microbial | In vitro and in vivo | [111] | |
| Symphytum officinale | ||||
| Calendula officinalis | ||||
| Geum urbanum | ||||
| Chitosan | ||||
| Copaiba oil | Antioxidant | In vitro and in vivo | [110] | |
| Clove essential oil | Antioxidant and anti-microbial | In vitro | [113] | |
| Papain | Antioxidant and anti-microbial | In vitro | [155] | |
| Curcumin | Anti-inflammation, antioxidant, and anticancer activity | In vitro | [112] | |
| Electrospun Fibre | Vitamin K3- Carnosine peptide | Antibacterial | In vitro and in vivo | [131] |
| Honey | Antioxidant and anti-bacterial | In vitro | [156] | |
| Curcumin | ||||
| Quercetin | Anti-bacterial | In vitro and in vivo | [157] | |
| Alkannin | Anti-inflammation, antioxidant, anti-mirobial and anti-tumor activity | In vitro | [158] | |
| Shikonin | ||||
| Quinone | Antioxidant | In vitro | [159] | |
| Konjac glucomannan EGCG | Anti-bacterial | In vitro and in vivo | [160] | |
| Nanoparticles | Catechol moiety | Antioxidant | In vitro and in vivo | [132] |
| Seed of Madhuca longifolia | Antioxidant and anti-microbial | In vitro and in vivo | [141] | |
| Star anise | Anti-inflammation and antioxidant | In vitro | [161] | |
| Gundelia tournefortii leaf | Antioxidant, anti-fungal, and anti-bacterial | In vitro and in vivo | [162] | |
| Aerogel | Wheat grass | Anti-microbial and angiogenic response | In vitro and in vivo | [163] |
| Hypericium perforatum oil | Antioxidant and anti-microbial | In vitro | [164] | |
| Bioactive glass | Curcumin | Antioxidant and anti-microbial | In vitro and in vivo | [165] |
5.2. Antioxidants Activity of Integrated-Biomaterials
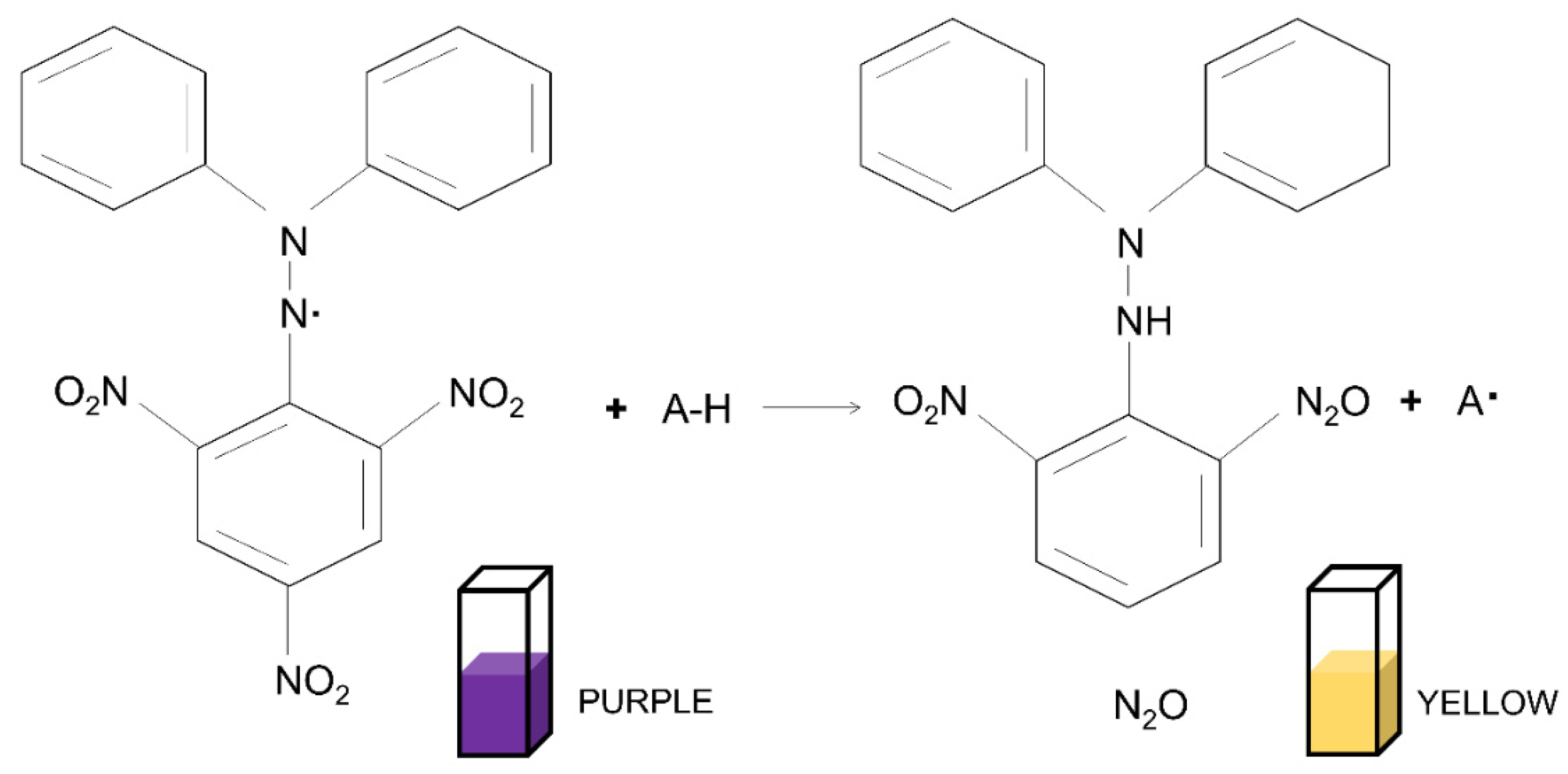
5.2.1. 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Assay
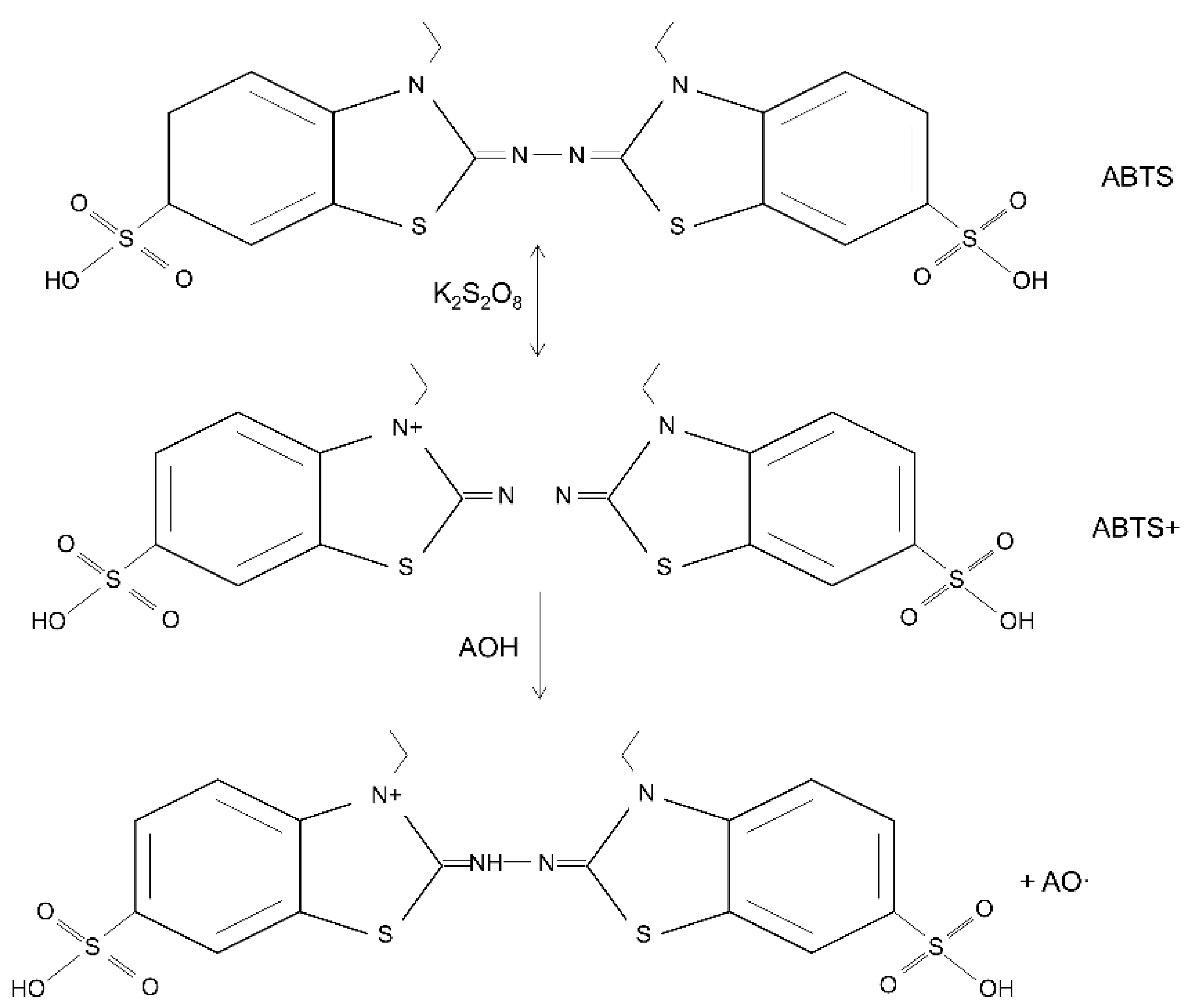
5.2.2. 2,2′-Azino-bis (3-ethylbenzothiazoline-6-sulfonic Acid) (ABTS) Assay
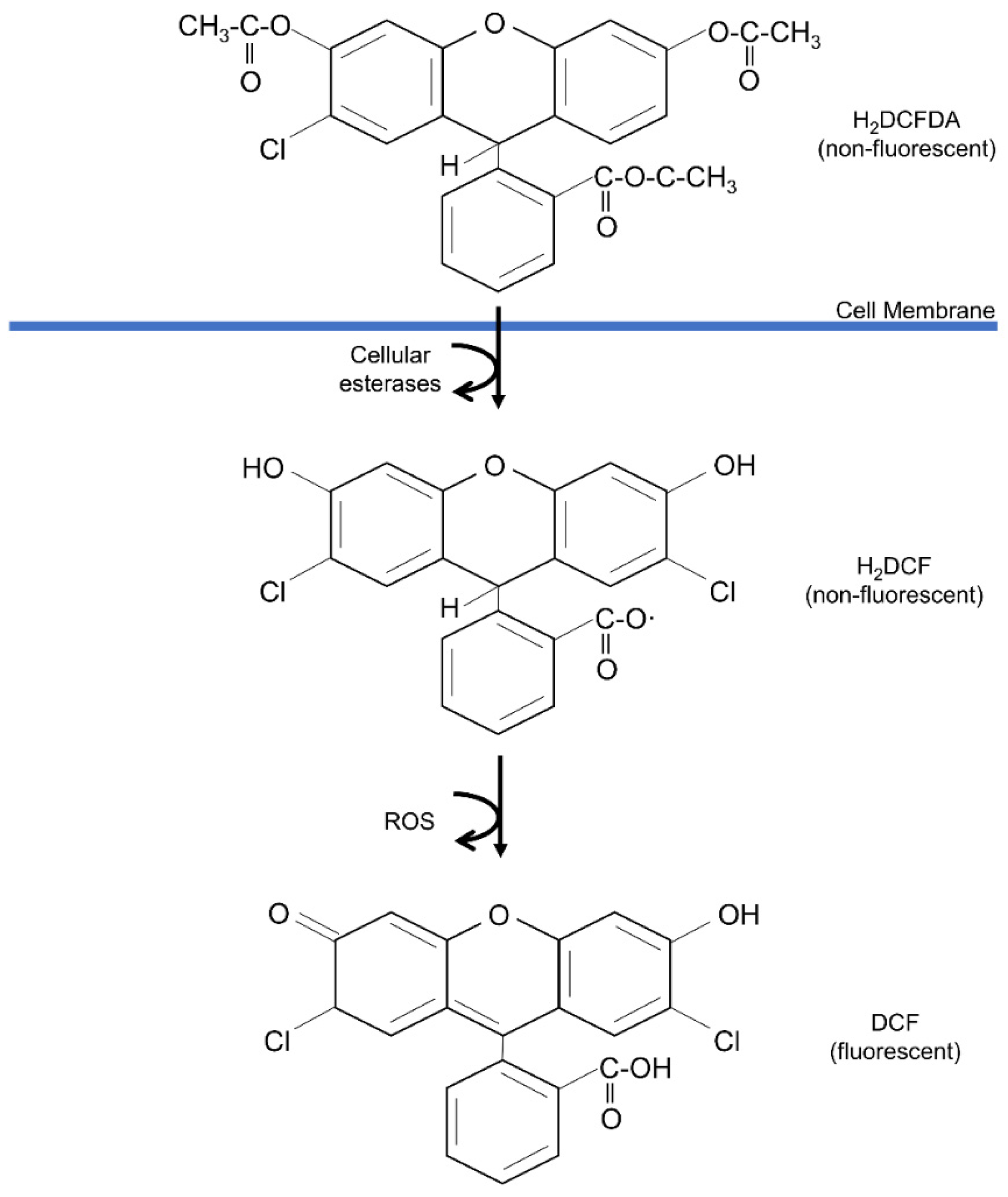
5.2.3. 2′,7′-Dichlorodihydrofluorescein Diacetate (DCFH-DA) Assay
6. Therapeutic Applications to Wound Healing
6.1. Pre-Clinical Models for Antioxidant Biomaterial Evaluation
6.1.1. In Vitro Analysis (Cell-Based Studies)
| Type of Cells | Antioxidant Compound | Biomaterial | Assay | Outcomes | References |
|---|---|---|---|---|---|
| NIH-3T3 fibroblast cells | Gallic acid | Hexanoyl glycol chitosan | Live/Dead assay | The cell viability is more than 90% when incorporated in the hydrogel | [122] |
| Chitosan | Hyaluronic acid | CCK-8 assay | The CCK-8 assay reported 100% cell viability after 24 h incubation and wound size decreased by 25.9% after 24 h | [145] | |
| Scratch assay | |||||
| Catechol moiety | Nanofibrous mat | MTT | The incorporation of catechol increases the cell adhesion and viability by 100% and complete wound closure after 24 h | [132] | |
| Calcein AM | |||||
| SEM | |||||
| Scratch assay | |||||
| Curcumin | Polyvinyl alcohol and alginate | MTT | The biomaterial shown to have more than 100% cell viability | [146] | |
| Vitamin K3-Carnosine peptide | Silk fibroin | Immunocytochemistry | Immunocytochemistry reported that the cells are actively proliferating when treated. The incorporation of bioactive molecules significantly increases the cell migration | [131] | |
| Scratch assay | |||||
| Live/Dead assay | |||||
| Honey | Ethylcellulose/gum tragacanth | MTT | The cell viability is more than 100% at day 7 for treated group and viability increased with increasing concentration of honey | [170] | |
| Phenolic acid | Polymer foams | Alamar blue | More than 79% of cell viable after 24 h incubation | [193] | |
| Propenoic acid | Gelatin | Live cell tracker | More than 90% reported in the cell viability and scratch area within 24 h. | [150] | |
| MTT | |||||
| Scratch assay | |||||
| Olives leaves Camellia sinensis extract | Carboxymethylcellulose | MTT | The cell viability decreases by 88% when the concentration increased to 100 µg/mL. The skin sensitization and irritation shown to have negative results | [151] | |
| h-CLAT assay | |||||
| OECD 439 assay | |||||
| Quinone | Silk-fibroin | MTT | The addition of quinone increased the cell viability by 100% after 6 days incubation and enhance the cell migration | [159] | |
| Scratch assay | |||||
| Human skin fibroblast cells | 5-Hydroxymethylfurfural | Polyvinyl alcohol and sodium alginate | CCK-8 assay | Integration of 5-HMF does not give significant results in cell proliferation. However, biomolecule affect the migration rate at concentration 50.4 and 75.6 µg/mL at 60% and 80% healing rate, respectively and slightly increase the production of collagen | [123] |
| CFSE | |||||
| Scratch assay | |||||
| Hydroxyproline assay (Collagen production) | |||||
| Curcumin | Polyvinyl alcohol and alginate | MTT | The biomaterial is non-toxic and able to increase the proliferation rate by 120% | [146] | |
| EA.hy926 keratinocyte cells | Catechol moiety | 3-4-dihydroxyphenylalanine (DOPA) | MTT | DOPA integration shows higher cell viability and cell adhesion through MTT as well as rigorous proliferation, adhesion and network formed shown via SEM and complete wound scratch closure within 24 h and accelerate tube formation at 72 h | [132] |
| Calcein AM | |||||
| SEM | |||||
| Scratch assay | |||||
| RNA isolation | |||||
| Immunocytochemistry | |||||
| Tube formation assay | |||||
| Wheat grass | Collagen | MTT | Increase proliferation rate by 120% in 24 h | [163] | |
| L929 fibroblast cells | Ferulic acid | Polyvinyl alcohol and chitosan | MTT | 6.25% integration of Ferulic acid showed 99% cell viability via MTT and 100% healed wound scratch by 24 h | [154] |
| Scratch assay | |||||
| Chitosan | Chitosan-sulfonamide | MTT | Retain cell viability higher than 70% after 72 h | [111] | |
| Humic acid | Alginate | MTT | Integration of humic acid show 8.18% higher cell viability of 100.80% compared to alginate alone | [149] | |
| Tannic acid | Hyaluronic acid | MTT | Low dose (250 µg mL−1) TA attain cell viability above 80%, as well for the love and dead assay, however higher dosage of AgNP (4 mM) incorporation showed significant reduce of cell viability (approx. 60%) | [152] | |
| Live/Dead assay | |||||
| Human umbilical vein endothelial cells (HUVEC) | Gundelia tournefortii | Silver nanoparticles | MTT | AgNP/GT IC50: 100 µg mL−1 AgNO3 IC50: 896 µg mL−1 | [162] |
| Quercetin | Polycaprolactone and gelatin | CCK-8 assay | CCK-8 report show P/Qu/Cup composites significantly enhanced cell proliferation whereas wound scratch assay show the composites are able to stimulate cell migration at 12 h. | [157] | |
| Scratch assay |
6.1.2. Animal Studies
6.1.3. Clinical Studies
| Compound | Carrier Type | Wound Type | Population | Results | Outcome | Reference |
|---|---|---|---|---|---|---|
| Chitosan | Dressing (not specified) | Chronic wounds (pressure ulcers, vascular ulcers, diabetic foot ulcers, and wounds with minor infections) | 90 patients | Wound area: Significantly reduced in the test group (65.97 ± 4.48%) than in the control group (39.95 ± 4.48%). Pain score: In the test group was 1.12 ± 0.23 and 2.30 ± 0.23 in the control group Wound depth: Lower in the test group (0.30 ± 0.48 cm) than in the control group (0.54 ± 0.86 cm) | 11 wounds had healed within 30 days | [201] |
| Film and hydrocolloid dressing | Superficial wound | 244 patients where 86 were treated with chitosan derivative film, and 84 with hydrocolloid dressing | Wound area: Day 13, the mean wound epithelialisation in the chitosan derivative film group was 99.17%, while 99.84% was in the hydrocolloid group. Pain score: Chitosan derivative film experienced more pain during the removal of the dressing, less exudate, and less odor than hydrocolloid group | No significant difference between the two groups; one, treated with a chitosan derivative film, and two, treated with hydrocolloid dressing | [205] | |
| Curcumin and galactomannan | Biofilm | Chronic wounds (type II diabetes) | 31 patients | Wound area: On the 12-weeks period, 16 over 31 wounds totally healed | Antioxidant wound treatment is a suitable therapeutic alternative and good performance for daily clinical practice. | [202] |
| Curcumin and N-Acetyl cysteine (NAC) | Dressing (not specified) | Acute and chronic wounds (venous leg ulcer, calciphylaxis ulcer, traumatic and dehisced wounds) | 31 patients | Wound area: On the 8-week period, 9 wounds (29%) completely healed, of which 7 were acute (77.8%) and 2 were chronic (22.2%) Other effect: The remaining wounds (22) presented a significant improvement after treatment | Treatment with the antioxidant dressing was more marked in the first 4 weeks, that the dressing works well both with acute and chronic wounds | [199] |
| Leptospermum honey | Gel | Partial-thickness burn wounds (face) | 7 patients | Wound area: Healing time ranged from 3 to 14 days (mean, 8.1 days) Other effect: Wound cultures revealed normal bacterial growth on days 1 and 7 for all patients | Active Leptospermum honey (ALH) improves outcomes in patients with partial-thickness burns by enhancing healing and re-epithelialisation rates, as well as by protecting against antibiotic-resistant microorganisms | [204] |
| Quercetin | Nano-hydrogel | Diabetic foot ulcer | 56 patients | Wound area: Nano-hydrogel embedded with quercetin and oleic acid demonstrated complete wound healing in 1 month Other effect: Only two patients of 56 (3.6%) were unresponsive to the treatment after 3 months. No local recurrence was observed during the follow-up period | No record on the development of other adverse drug reactions such as topical skin allergy, skin discoloration, or keloid scars. | [205] |
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, F.; Han, X.; Guo, C.; Yang, H.; Wang, J.; Wu, X. Fibrous aramid hydrogel supported antibacterial agents for accelerating bacterial-infected wound healing. Mater. Sci. Eng. C 2021, 121, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Naseri-Nosar, M.; Ziora, Z.M. Wound dressings from naturally-occurring polymers: A review on homopolysaccharide-based composites. Carbohydr. Polym. 2018, 189, 379–398. [Google Scholar] [CrossRef] [PubMed]
- Yousef, H.; Alhajj, M.; Sharma, S. Anatomy, Skin (Integument), Epidermis-StatPearls-NCBI Bookshelf; StatPearls Publishing: Treasure Island, FL, USA, 2020; pp. 1–12. [Google Scholar]
- Nike, D.U.; Md Fadilah, N.I.; Sallehuddin, N.; Azlan, A.Y.H.N.; Imran, F.H.; Maarof, M.; Fauzi, M.B. Genipin-Crosslinking Effects on Biomatrix Development for Cutaneous Wound Healing: A Concise Review. Front. Bioeng. Biotechnol. 2022, 10, 726. [Google Scholar]
- Boateng, J.; Catanzano, O. Advanced therapeutic dressings for effective wound healing—A review. J. Pharm. Sci. 2015, 104, 3653–3680. [Google Scholar] [CrossRef]
- Fadilah, N.I.M.; Ahmad, H.; Rahman, M.B.A.; Chia, S.L.; Ng, S.-F.; Leong, S.W. Synthesis and in vitro biological evaluations of novel tetrapeptide as therapeutic agent for wound treatment. J. Saudi Chem. Soc. 2020, 24, 606–619. [Google Scholar] [CrossRef]
- Ceylan, S. Propolis loaded and genipin-crosslinked PVA/chitosan membranes; characterization properties and cytocompatibility/genotoxicity response for wound dressing applications. Int. J. Biol. Macromol. 2021, 181, 1196–1206. [Google Scholar] [CrossRef]
- Fadilah, N.I.M.; Rahman, M.B.A.; Yusof, L.M.; Mustapha, N.M.; Ahmad, H. The Therapeutic Effect and In Vivo Assessment of Palmitoyl- GDPH on the Wound Healing Process. Pharmaceutics 2021, 13, 193. [Google Scholar] [CrossRef]
- Dhivya, S.; Padma, V.V.; Santhini, E. Wound dressings—A review. BioMedicine 2015, 5, 22. [Google Scholar] [CrossRef]
- Ren, Y.; Ailierken, A.; Zhao, L.; Lin, Z.; Jiang, J.; Balun, L.; Wang, J.; Hua, J.; Tu, Q. hUC-MSCs lyophilized powder loaded polysaccharide ulvan driven functional hydrogel for chronic diabetic wound healing. Carbohydr. Polym. 2022, 119404. [Google Scholar] [CrossRef]
- Lynch, R.I.; Lavelle, E.C. Immuno-modulatory biomaterials as anti-inflammatory therapeutics. Biochem. Pharmacol. 2022, 288, 114890. [Google Scholar] [CrossRef]
- Detsch, R.; Will, J.; Hum, J.; Roether, J.A.; Boccaccini, A.R. Biomaterials. In Cell Culture Technology; Kasper, C., Charwat, V., Lavrentieva, A., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 91–105. [Google Scholar]
- Fadilah, N.I.M.; Maarof, M.; Motta, A.; Tabata, Y.; Fauzi, M.B. The Discovery and Development of Natural-Based Biomaterials with Demonstrated Wound Healing Properties: A Reliable Approach in Clinical Trials. Biomedicines 2022, 10, 2226. [Google Scholar] [CrossRef] [PubMed]
- Busra, F.M.; Lokanathan, Y.; Nadzir, M.M.; Saim, A.; Idrus, R.B.H.; Chowdhury, S.R. Attachment, proliferation, and morphological properties of human dermal fibroblasts on ovine tendon collagen scaffolds: A comparative study. Malays. J. Med. Sci. MJMS 2017, 24, 33. [Google Scholar] [CrossRef]
- Sallehuddin, N.; Md Fadilah, N.I.; Hwei, N.M.; Wen, A.P.Y.; Yusop, S.M.; Rajab, N.F.; Hiraoka, Y.; Tabata, Y.; Fauzi, M.B. Characterization and Cytocompatibility of Collagen–Gelatin–Elastin (CollaGee) Acellular Skin Substitute towards Human Dermal Fibroblasts: In Vitro Assessment. Biomedicines 2022, 10, 1327. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shu, W.; Yu, Q.; Qu, W.; Wang, Y.; Li, R. Functional biomaterials for treatment of chronic wound. Front. Bioeng. Biotechnol. 2020, 8, 516. [Google Scholar] [CrossRef]
- Md Fadilah, N.I.; Jailani, M.S.M.A.K.; Hisham, M.A.I.B.; Sunthar Raj, N.; Shamsuddin, S.A.; Ng, M.H.; Fauzi, M.B.; Maarof, M. Cell secretomes for wound healing and tissue regeneration: Next generation acellular based tissue engineered products. J. Tissue Eng. 2022, 13, 20417314221114273. [Google Scholar] [CrossRef] [PubMed]
- Browne, S.; Pandit, A. Biomaterial-Mediated Modification of the Local Inflammatory Environment. Front. Bioeng. Biotechnol. 2015, 3, 67. [Google Scholar] [CrossRef]
- Aamodt, J.M.; Grainger, D.W. Extracellular matrix-based biomaterial scaffolds and the host response. Biomaterials 2016, 86, 68–82. [Google Scholar] [CrossRef]
- Shafiq, M.; Chen, Y.; Hashim, R.; He, C.; Mo, X.; Zhou, X. Reactive Oxygen Species-Based Biomaterials for Regenerative Medicine and Tissue Engineering Applications. Front. Bioeng. Biotechnol. 2021, 9, 821288. [Google Scholar] [CrossRef]
- Pedro, A.C.; Paniz, O.G.; Fernandes, I.d.A.A.; Bortolini, D.G.; Rubio, F.T.V.; Haminiuk, C.W.I.; Maciel, G.M.; Magalhães, W.L.E. The Importance of Antioxidant Biomaterials in Human Health and Technological Innovation: A Review. Antioxidants 2022, 11, 1644. [Google Scholar] [CrossRef]
- Kohen, R. Skin antioxidants: Their role in aging and in oxidative stress--new approaches for their evaluation. Biomed. Pharmacother. 1999, 53, 181–192. [Google Scholar] [CrossRef]
- Liu, J.; Chen, Z.; Wang, J.; Li, R.; Li, T.; Chang, M.; Yan, F.; Wang, Y. Encapsulation of curcumin nanoparticles with MMP9-responsive and thermos-sensitive hydrogel improves diabetic wound healing. ACS Appl. Mater. Interfaces 2018, 10, 16315–16326. [Google Scholar] [CrossRef] [PubMed]
- Bektas, N.; Şenel, B.; Yenilmez, E.; Özatik, O.; Arslan, R. Evaluation of wound healing effect of chitosan-based gel formulation containing vitexin. Saudi Pharm. J. 2020, 28, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.K.; Ajmal, G.; Upadhyay, S.N.; Mishra, P.K. Nano-fibrous scaffold with curcumin for anti-scar wound healing. Int. J. Pharm. 2020, 589, 119858. [Google Scholar] [CrossRef]
- Comino-Sanz, I.M.; López-Franco, M.D.; Castro, B.; Pancorbo-Hidalgo, P.L. The role of antioxidants on wound healing: A review of the current evidence. J. Clin. Med. 2021, 10, 3558. [Google Scholar] [CrossRef] [PubMed]
- e Silva, S.A.M.; Calixto, G.M.F.; Cajado, J.; De Carvalho, P.C.A.; Rodero, C.F.; Chorilli, M.; Leonardi, G.R. Gallic acid-loaded gel formulation combats skin oxidative stress: Development, characterization and ex vivo biological assays. Polymers 2017, 9, 391. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Sood, S.; Agarwal, S.; Saini, A.K.; Pathania, D. Antioxidant activity and controlled drug delivery potential of tragacanth gum-cl-poly (lactic acid-co-itaconic acid) hydrogel. Int. J. Biol. Macromol. 2018, 107, 2534–2543. [Google Scholar] [CrossRef] [PubMed]
- Marrazzo, P.; O’Leary, C. Repositioning Natural Antioxidants for Therapeutic Applications in Tissue Engineering. Bioengineering 2020, 7, 104. [Google Scholar] [CrossRef]
- Ou, Q.; Zhang, S.; Fu, C.; Yu, L.; Xin, P.; Gu, Z.; Cao, Z.; Wu, J.; Wang, Y. More natural more better: Triple natural anti-oxidant puerarin/ferulic acid/polydopamine incorporated hydrogel for wound healing. J. Nanobiotechnol. 2021, 19, 237. [Google Scholar] [CrossRef]
- Ren, Y.; Zhang, D.; He, Y.; Chang, R.; Guo, S.; Ma, S.; Yao, M.; Guan, F. Injectable and antioxidative HT/QGA hydrogel for potential application in wound healing. Gels 2021, 7, 204. [Google Scholar] [CrossRef]
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound Healing: A Cellular Perspective. Physiol. Rev. 2018, 99, 665–706. [Google Scholar] [CrossRef]
- Phang, S.J.; Arumugam, B.; Kuppusamy, U.R.; Fauzi, M.B.; Looi, M.L. A review of diabetic wound models-Novel insights into diabetic foot ulcer. J. Tissue Eng. Regen. Med. 2021, 15, 1051–1068. [Google Scholar] [CrossRef] [PubMed]
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D.; Georgopoulos, N.T. Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. Wound J. 2017, 14, 89–96. [Google Scholar] [CrossRef]
- André-Lévigne, D.; Modarressi, A.; Pepper, M.S.; Pittet-Cuénod, B. Reactive Oxygen Species and NOX Enzymes Are Emerging as Key Players in Cutaneous Wound Repair. Int. J. Mol. Sci. 2017, 18, 2149. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.C.; Lancel, S.; Boulanger, E.; Neviere, R. Targeting Oxidative Stress and Mitochondrial Dysfunction in the Treatment of Impaired Wound Healing: A Systematic Review. Antioxidants 2018, 7, 98. [Google Scholar] [CrossRef] [PubMed]
- Bretón-Romero, R.; Lamas, S. Hydrogen peroxide signaling in vascular endothelial cells. Redox Biol. 2014, 2, 529–534. [Google Scholar] [CrossRef]
- Görlach, A. Redox regulation of the coagulation cascade. Antioxid. Redox Signal 2005, 7, 1398–1404. [Google Scholar] [CrossRef]
- Hoffmann, M.H.; Griffiths, H.R. The dual role of Reactive Oxygen Species in autoimmune and inflammatory diseases: Evidence from preclinical models. Free Radic. Biol. Med. 2018, 125, 62–71. [Google Scholar] [CrossRef]
- Jiang, F.; Zhang, Y.; Dusting, G.J. NADPH oxidase-mediated redox signaling: Roles in cellular stress response, stress tolerance, and tissue repair. Pharmacol. Rev. 2011, 63, 218–242. [Google Scholar] [CrossRef]
- Kunkemoeller, B.; Kyriakides, T.R. Redox Signaling in Diabetic Wound Healing Regulates Extracellular Matrix Deposition. Antioxid. Redox Signal 2017, 27, 823–838. [Google Scholar] [CrossRef]
- Loo, A.E.; Wong, Y.T.; Ho, R.; Wasser, M.; Du, T.; Ng, W.T.; Halliwell, B. Effects of hydrogen peroxide on wound healing in mice in relation to oxidative damage. PLoS ONE 2012, 7, e49215. [Google Scholar] [CrossRef]
- Deng, L.; Du, C.; Song, P.; Chen, T.; Rui, S.; Armstrong, D.G.; Deng, W. The Role of Oxidative Stress and Antioxidants in Diabetic Wound Healing. Oxidative Med. Cell. Longev. 2021, 2021, 8852759. [Google Scholar] [CrossRef] [PubMed]
- Bitar, M.S.; Al-Mulla, F. A defect in Nrf2 signaling constitutes a mechanism for cellular stress hypersensitivity in a genetic rat model of type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E1119–E1129. [Google Scholar] [CrossRef] [PubMed]
- Long, M.; de la Vega, M.R.; Wen, Q.; Bharara, M.; Jiang, T.; Zhang, R.; Zhou, S.; Wong, P.K.; Wondrak, G.T.; Zheng, H.; et al. An Essential Role of NRF2 in Diabetic Wound Healing. Diabetes 2016, 65, 780–793. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, A.; Rojo, A.I.; Wells, G.; Hayes, J.D.; Cousin, S.P.; Rumsey, W.L.; Attucks, O.C.; Franklin, S.; Levonen, A.-L.; Kensler, T.W.; et al. Therapeutic targeting of the NRF2 and KEAP1 partnership in chronic diseases. Nat. Rev. Drug Discov. 2019, 18, 295–317. [Google Scholar] [CrossRef]
- Joo, M.S.; Kim, W.D.; Lee, K.Y.; Kim, J.H.; Koo, J.H.; Kim, S.G. AMPK Facilitates Nuclear Accumulation of Nrf2 by Phosphorylating at Serine 550. Mol. Cell. Biol. 2016, 36, 1931–1942. [Google Scholar] [CrossRef]
- Kurahashi, T.; Fujii, J. Roles of antioxidative enzymes in wound healing. J. Dev. Biol. 2015, 3, 57–70. [Google Scholar] [CrossRef]
- Sen, C.K.; Roy, S. Redox signals in wound healing. Biochim. Biophys. Acta (BBA) Gen. Subj. 2008, 1780, 1348–1361. [Google Scholar] [CrossRef]
- Arya, A.K.; Pokharia, D.; Tripathi, K. Relationship between oxidative stress and apoptotic markers in lymphocytes of diabetic patients with chronic non healing wound. Diabetes Res. Clin. Pract. 2011, 94, 377–384. [Google Scholar] [CrossRef]
- Vairamon, S.J.; Babu, M.; Viswanathan, V. Oxidative stress markers regulating the healing of foot ulcers in patients with type 2 diabetes. Wounds 2009, 21, 273–279. [Google Scholar]
- Zhang, W.; Chen, L.; Xiong, Y.; Panayi, A.C.; Abududilibaier, A.; Hu, Y.; Yu, C.; Zhou, W.; Sun, Y.; Liu, M.; et al. Antioxidant Therapy and Antioxidant-Related Bionanomaterials in Diabetic Wound Healing. Front. Bioeng. Biotechnol. 2021, 9, 707479. [Google Scholar] [CrossRef]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Phang, S.J.; Teh, H.X.; Looi, M.L.; Arumugam, B.; Fauzi, M.B.; Kuppusamy, U.R. Phlorotannins from brown algae: A review on their antioxidant mechanisms and applications in oxidative stress-mediated diseases. J. Appl. Phycol. 2023, 1–26. [Google Scholar] [CrossRef]
- Xu, D.-P.; Li, Y.; Meng, X.; Zhou, T.; Zhou, Y.; Zheng, J.; Zhang, J.-J.; Li, H.-B. Natural antioxidants in foods and medicinal plants: Extraction, assessment and resources. Int. J. Mol. Sci. 2017, 18, 96. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, S.C.; Moldão-Martins, M.; Alves, V.D. Antioxidants of natural plant origins: From sources to food industry applications. Molecules 2019, 24, 4132. [Google Scholar] [CrossRef]
- Shah, M.A.; Bosco, S.J.D.; Mir, S.A. Plant extracts as natural antioxidants in meat and meat products. Meat Sci. 2014, 98, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, E.d.S.; Paranhos, S.B.; da Paz, S.P.A.; Canelas, C.A.d.A.; Nascimento, L.A.S.d.; Passos, M.F.; da Silva, A.C.R.; Monteiro, S.N.; Paula, M.V.d.S.; Candido, V.S. Synthesis and Characterization of Natural Polymeric Membranes Composed of Chitosan, Green Banana Peel Extract and Andiroba Oil. Polymers 2022, 14, 1105. [Google Scholar] [CrossRef]
- Amri, B.; Martino, E.; Vitulo, F.; Corana, F.; Ben-Kaâb, L.B.; Rui, M.; Rossi, D.; Mori, M.; Rossi, S.; Collina, S. Marrubium vulgare L. leave extract: Phytochemical composition, antioxidant and wound healing properties. Molecules 2017, 22, 1851. [Google Scholar] [CrossRef]
- Xu, J.; Xu, J.J.; Lin, Q.; Jiang, L.; Zhang, D.; Li, Z.; Ma, B.; Zhang, C.; Li, L.; Kai, D. Lignin-incorporated nanogel serving as an antioxidant biomaterial for wound healing. ACS Appl. Bio Mater. 2020, 4, 3–13. [Google Scholar] [CrossRef]
- Ezhilarasu, H.; Ramalingam, R.; Dhand, C.; Lakshminarayanan, R.; Sadiq, A.; Gandhimathi, C.; Ramakrishna, S.; Bay, B.H.; Venugopal, J.R.; Srinivasan, D.K. Biocompatible aloe vera and tetracycline hydrochloride loaded hybrid nanofibrous scaffolds for skin tissue engineering. Int. J. Mol. Sci. 2019, 20, 5174. [Google Scholar] [CrossRef]
- Danna, C.; Bazzicalupo, M.; Ingegneri, M.; Smeriglio, A.; Trombetta, D.; Burlando, B.; Cornara, L. Anti-Inflammatory and Wound Healing Properties of Leaf and Rhizome Extracts from the Medicinal Plant Peucedanumostruthium (L.) WDJ Koch. Molecules 2022, 27, 4271. [Google Scholar] [CrossRef]
- Hemmati, A.A.; Rashidi, I.; Dahanzadeh, S.; Moeini, M. Investigating the healing effect of the hydroalcoholic extract of pomegranate seed (Punica granatum) on the full thickness wound in rabbit. J. Contemp. Med. Sci. 2020, 6, 66–72. [Google Scholar] [CrossRef]
- Belwal, T.; Dhyani, P.; Bhatt, I.D.; Rawal, R.S.; Pande, V. Optimization extraction conditions for improving phenolic content and antioxidant activity in Berberis asiatica fruits using response surface methodology (RSM). Food Chem. 2016, 207, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.T.; Pham, H.N.T.; Bowyer, M.C.; van Altena, I.A.; Scarlett, C.J. Influence of solvents and novel extraction methods on bioactive compounds and antioxidant capacity of Phyllanthus amarus. Chem. Pap. 2016, 70, 556–566. [Google Scholar] [CrossRef]
- Tiwari, P.K.; Kaur, M.; Kaur, H. Phytochemical screening and Extraction: A Review. Int. Pharm. Sci. 2011, 1, 98–106. [Google Scholar]
- Sun, C.; Wu, Z.; Wang, Z.; Zhang, H. Effect of Ethanol/Water Solvents on Phenolic Profiles and Antioxidant Properties of Beijing Propolis Extracts. Evid. Based Complement. Altern. Med. 2015, 2015, 595393. [Google Scholar] [CrossRef]
- Ajila, C.; Brar, S.; Verma, M.; Tyagi, R.; Godbout, S.; Valéro, J. Extraction and analysis of polyphenols: Recent trends. Crit. Rev. Biotechnol. 2011, 31, 227–249. [Google Scholar] [CrossRef]
- Razika, L.; Thanina, A.C.; Nadjiba, C.-M.; Narimen, B.; Mahdi, D.M.; Karim, A. Antioxidant and wound healing potential of saponins extracted from the leaves of Algerian Urtica dioica L. Pak. J. Pharm. Sci. 2017, 30, 1023–1029. [Google Scholar]
- Saiah, H.; Mokhtar, M.; Saiah, W.; Aichouni, A.; EL Kebir, F.Z.; Allem, R. In vivo wound healing potential and HPLC-PDA-ESI-MS profiling of Zizyphus lotus L.(Desf.) leaves methanol extract. J. Food Biochem. 2018, 42, e12570. [Google Scholar] [CrossRef]
- Al-Dabbagh, B.; Elhaty, I.A.; Al Hrout, A.; Al Sakkaf, R.; El-Awady, R.; Ashraf, S.S.; Amin, A. Antioxidant and anticancer activities of Trigonella foenum-graecum, Cassia acutifolia and Rhazya stricta. BMC Complement. Altern. Med. 2018, 18, 240. [Google Scholar] [CrossRef]
- De Barros, N.R.; Heredia-Vieira, S.C.; Borges, F.A.; Benites, N.M.; Dos Reis, C.E.; Miranda, M.C.R.; Cardoso, C.A.L.; Herculano, R.D. Natural rubber latex biodevice as controlled release system for chronic wounds healing. Biomed. Phys. Eng. Express 2018, 4, 035026. [Google Scholar] [CrossRef]
- Chinnasamy, G.; Chandrasekharan, S.; Bhatnagar, S. Biosynthesis of silver nanoparticles from Melia azedarach: Enhancement of antibacterial, wound healing, antidiabetic and antioxidant activities. Int. J. Nanomed. 2019, 14, 9823. [Google Scholar] [CrossRef] [PubMed]
- Rhimi, W.; Hlel, R.; Salem, I.B.; Boulila, A.; Rejeb, A.; Saidi, M. Dittrichia viscosa L. Ethanolic Extract Based Ointment with Antiradical, Antioxidant, and Healing Wound Activities. BioMed Res. Int. 2019, 2019, 4081253. [Google Scholar] [CrossRef] [PubMed]
- Maiza, A.; Nzekoue, F.K.; Ghazouani, T.; Afif, M.; Caprioli, G.; Fiorini, D.; Vittori, S.; Maggi, F.; Buccioni, M.; Navia, A.M. Butter oil (ghee) enrichment with aromatic plants: Chemical characterization and effects on fibroblast migration in an in-vitro wound healing model. Arab. J. Chem. 2020, 13, 8909–8919. [Google Scholar] [CrossRef]
- Oryan, A.; Alemzadeh, E.; Mohammadi, A.A.; Moshiri, A. Healing potential of injectable Aloe vera hydrogel loaded by adipose-derived stem cell in skin tissue-engineering in a rat burn wound model. Cell Tissue Res. 2019, 377, 215–227. [Google Scholar] [CrossRef]
- Garcia-Orue, I.; Gainza, G.; Garcia-Garcia, P.; Gutierrez, F.B.; Aguirre, J.J.; Hernandez, R.M.; Delgado, A.; Igartua, M. Composite nanofibrous membranes of PLGA/Aloe vera containing lipid nanoparticles for wound dressing applications. Int. J. Pharm. 2019, 556, 320–329. [Google Scholar] [CrossRef]
- Rahman, M.S.; Islam, R.; Rana, M.M.; Spitzhorn, L.-S.; Rahman, M.S.; Adjaye, J.; Asaduzzaman, S.M. Characterization of burn wound healing gel prepared from human amniotic membrane and Aloe vera extract. BMC Complement. Altern. Med. 2019, 19, 115. [Google Scholar] [CrossRef]
- Thompson, Z.; Rahman, S.; Yarmolenko, S.; Sankar, J.; Kumar, D.; Bhattarai, N. Fabrication and characterization of magnesium ferrite-based PCL/Aloe vera nanofibers. Materials 2017, 10, 937. [Google Scholar] [CrossRef]
- Kim, S.-H.; Kim, K.; Kim, B.S.; An, Y.-H.; Lee, U.-J.; Lee, S.-H.; Kim, S.L.; Kim, B.-G.; Hwang, N.S. Fabrication of polyphenol-incorporated anti-inflammatory hydrogel via high-affinity enzymatic crosslinking for wet tissue adhesion. Biomaterials 2020, 242, 119905. [Google Scholar] [CrossRef]
- Rubio-Elizalde, I.; Bernáldez-Sarabia, J.; Moreno-Ulloa, A.; Vilanova, C.; Juárez, P.; Licea-Navarro, A.; Castro-Ceseña, A.B. Scaffolds based on alginate-PEG methyl ether methacrylate-Moringa oleifera-Aloe vera for wound healing applications. Carbohydr. Polym. 2019, 206, 455–467. [Google Scholar] [CrossRef]
- Al-Ghanayem, A.A.; Alhussaini, M.S.; Asad, M.; Joseph, B. Effect of Moringa oleifera Leaf Extract on Excision Wound Infections in Rats: Antioxidant, Antimicrobial, and Gene Expression Analysis. Molecules 2022, 27, 4481. [Google Scholar] [CrossRef]
- Teo, B.S.X.; Gan, R.Y.; Aziz, S.A.; Sirirak, T.; Asmani, M.F.M.; Yusuf, E. In vitro evaluation of antioxidant and antibacterial activities of Eucheuma cottonii extract and its in vivo evaluation of the wound-healing activity in mice. J. Cosmet. Dermatol. 2021, 20, 993–1001. [Google Scholar] [CrossRef] [PubMed]
- Samadian, H.; Zamiri, S.; Ehterami, A.; Farzamfar, S.; Vaez, A.; Khastar, H.; Alam, M.; Ai, A.; Derakhshankhah, H.; Allahyari, Z. Electrospun cellulose acetate/gelatin nanofibrous wound dressing containing berberine for diabetic foot ulcer healing: In vitro and in vivo studies. Sci. Rep. 2020, 10, 8312. [Google Scholar] [CrossRef] [PubMed]
- Alwan, M.H.; Ghani, B.A. Histological Evaluation of Local Application of Flavonoid Extract of Capparis Spinosa on Wound Healing in Alloxan-Induced Diabetic Rats. Diyala J. Med. 2020, 18, 1–13. [Google Scholar] [CrossRef]
- Najeeb, V.D.; Abdulaziz, S.M.; Kurtas, P.T. Determination of Antibacterial and Wound Healing Potentials of Ethanolic Extract of Cydonia oblonga Miller Fruit. Diyala J. Med. 2020, 18, 44–54. [Google Scholar] [CrossRef]
- Selvaraj, S.; Fathima, N.N. Fenugreek Incorporated Silk Fibroin Nanofibers A Potential Antioxidant Scaffold for Enhanced Wound Healing. ACS Appl. Mater. Interfaces 2017, 9, 5916–5926. [Google Scholar] [CrossRef]
- Shaik, M.M.; Dapkekar, A.; Rajwade, J.M.; Jadhav, S.H.; Kowshik, M. Antioxidant-antibacterial containing bi-layer scaffolds as potential candidates for management of oxidative stress and infections in wound healing. J. Mater. Sci. Mater. Med. 2019, 30, 13. [Google Scholar] [CrossRef]
- Pinzón-García, A.D.; Cassini-Vieira, P.; Ribeiro, C.C.; de Matos Jensen, C.E.; Barcelos, L.S.; Cortes, M.E.; Sinisterra, R.D. Efficient cutaneous wound healing using bixin-loaded PCL nanofibers in diabetic mice. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 1938–1949. [Google Scholar] [CrossRef]
- Dias, F.T.G.; Ingracio, A.R.; Nicoletti, N.F.; Menezes, F.C.; Agnol, L.D.; Marinowic, D.R.; Soares, R.M.D.; da Costa, J.C.; Falavigna, A.; Bianchi, O. Soybean-modified polyamide-6 mats as a long-term cutaneous wound covering. Mater. Sci. Eng. C 2019, 99, 957–968. [Google Scholar] [CrossRef]
- Zulaikhah, S.T. The role of antioxidant to prevent free radicals in the body. Sains Med. 2017, 8, 39–45. [Google Scholar] [CrossRef]
- Parham, S.; Kharazi, A.Z.; Bakhsheshi-Rad, H.R.; Nur, H.; Ismail, A.F.; Sharif, S.; RamaKrishna, S.; Berto, F. Antioxidant, antimicrobial and antiviral properties of herbal materials. Antioxidants 2020, 9, 1309. [Google Scholar] [CrossRef]
- Bereksi, M.S.; Hassaïne, H.; Bekhechi, C.; Abdelouahid, D.E. Evaluation of antibacterial activity of some medicinal plants extracts commonly used in Algerian traditional medicine against some pathogenic bacteria. Pharmacogn. J. 2018, 10, 507–512. [Google Scholar] [CrossRef]
- Labib, R.M.; Ayoub, I.M.; Michel, H.E.; Mehanny, M.; Kamil, V.; Hany, M.; Magdy, M.; Moataz, A.; Maged, B.; Mohamed, A. Appraisal on the wound healing potential of Melaleuca alternifolia and Rosmarinus officinalis L. essential oil-loaded chitosan topical preparations. PLoS ONE 2019, 14, e0219561. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Mohsen, A.; Frankova, J.; Abdel-Rahman, R.M.; Salem, A.; Sahffie, N.; Kubena, I.; Jancar, J. Chitosan-glucan complex hollow fibers reinforced collagen wound dressing embedded with aloe vera. II. Multifunctional properties to promote cutaneous wound healing. Int. J. Pharm. 2020, 582, 119349. [Google Scholar] [CrossRef] [PubMed]
- Hajialyani, M.; Tewari, D.; Sobarzo-Sanchez, E.; Nabavi, S.M.; Farzaei, M.H.; Abdollahi, M. Natural product-based nanomedicines for wound healing purposes: Therapeutic targets and drug delivery systems. Int. J. Nanomed. 2018, 13, 5023. [Google Scholar] [CrossRef] [PubMed]
- Dev, S.K.; Choudhury, P.; Srivastava, R.; Sharma, M. Antimicrobial, anti-inflammatory and wound healing activity of polyherbal formulation. Biomed. Pharmacother. 2019, 111, 555–567. [Google Scholar] [CrossRef] [PubMed]
- Murray, R.Z.; West, Z.E.; Cowin, A.J.; Farrugia, B.L. Development and use of biomaterials as wound healing therapies. Burns Trauma 2019, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Liang, Y.; Zhang, H.; Guo, B. Antibacterial biomaterials for skin wound dressing. Asian J. Pharm. Sci. 2022, 17, 353–384. [Google Scholar] [CrossRef]
- Raut, H.K.; Das, R.; Liu, Z.; Liu, X.; Ramakrishna, S. Biocompatibility of biomaterials for tissue regeneration or replacement. Biotechnol. J. 2020, 15, 2000160. [Google Scholar] [CrossRef]
- Chen, F.M.; Liu, X. Advancing biomaterials of human origin for tissue engineering. Prog. Polym. Sci. 2016, 53, 86–168. [Google Scholar] [CrossRef]
- Sarv, V.; Venskutonis, P.R.; Bhat, R. The Sorbus spp.—Underutilised Plants for Foods and Nutraceuticals: Review on Polyphenolic Phytochemicals and Antioxidant Potential. Antioxidants 2020, 9, 813. [Google Scholar] [CrossRef]
- Md Fadilah, N.I.; Hassan, A.R. Preparation, Characterization and Performance Studies of Active PVDF Ultrafiltration-Surfactants Membranes Containing PVP as Additive. In Advanced Materials Research; Trans Tech Publications Ltd.: Wollerau, Switzerland, 2016; pp. 44–49. [Google Scholar]
- Fauzi, M.; Lokanathan, Y.; Aminuddin, B.; Ruszymah, B.; Chowdhury, S. Ovine tendon collagen: Extraction, characterisation and fabrication of thin films for tissue engineering applications. Mater. Sci. Eng. C 2016, 68, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.; Zhang, H.; Guo, B. Conductive biomaterials as bioactive wound dressing for wound healing and skin tissue engineering. Nano Micro Lett. 2022, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Vinklárková, L.; Masteiková, R.; Vetchý, D.; Doležel, P.; Bernatonienė, J. Formulation of novel layered sodium carboxymethylcellulose film wound dressings with ibuprofen for alleviating wound pain. BioMed Res. Int. 2015, 2015, 892671. [Google Scholar] [CrossRef] [PubMed]
- Colobatiu, L.; Gavan, A.; Potarniche, A.-V.; Rus, V.; Diaconeasa, Z.; Mocan, A.; Tomuta, I.; Mirel, S.; Mihaiu, M. Evaluation of bioactive compounds-loaded chitosan films as a novel and potential diabetic wound dressing material. React. Funct. Polym. 2019, 145, 104369. [Google Scholar] [CrossRef]
- Paranhos, S.B.; Ferreira, E.d.S.; Canelas, C.A.d.A.; da Paz, S.P.A.; Passos, M.F.; da Costa, C.E.F.; da Silva, A.C.R.; Monteiro, S.N.; Candido, V.S. Chitosan Membrane Containing Copaiba Oil (Copaifera spp.) for Skin Wound Treatment. Polymers 2021, 14, 35. [Google Scholar] [CrossRef]
- Dragostin, O.M.; Samal, S.K.; Dash, M.; Lupascu, F.; Pânzariu, A.; Tuchilus, C.; Ghetu, N.; Danciu, M.; Dubruel, P.; Pieptu, D. New antimicrobial chitosan derivatives for wound dressing applications. Carbohydr. Polym. 2016, 141, 28–40. [Google Scholar] [CrossRef]
- Mythili Gnanamangai, B.; Suganya, M.; Sabarinathan, R.; Ponmurugan, P. Fabrication of chitosan-alginate microencapsulated curcumin coated scaffold to develop novel cotton crepe bandage. Indian J. Fibre Text. Res. (IJFTR) 2019, 44, 271–278. [Google Scholar]
- Guilherme, E.d.O.; de Souza, C.W.; Bernardo, M.P.; Zenke, M.; Mattoso, L.H.; Moreira, F.K. Antimicrobially active gelatin/[Mg-Al-CO3]-LDH composite films based on clove essential oil for skin wound healing. Mater. Today Commun. 2021, 27, 102169. [Google Scholar]
- Heipieper, H.-J.; Keweloh, H.; Rehm, H.-J. Influence of phenols on growth and membrane permeability of free and immobilized Escherichia coli. Appl. Environ. Microbiol. 1991, 57, 1213–1217. [Google Scholar] [CrossRef]
- Campos, F.; Couto, J.; Figueiredo, A.; Tóth, I.; Rangel, A.O.; Hogg, T. Cell membrane damage induced by phenolic acids on wine lactic acid bacteria. Int. J. Food Microbiol. 2009, 135, 144–151. [Google Scholar] [CrossRef]
- Yang, J.; Shen, M.; Luo, Y.; Wu, T.; Chen, X.; Wang, Y.; Xie, J. Advanced applications of chitosan-based hydrogels: From biosensors to intelligent food packaging system. Trends Food Sci. Technol. 2021, 110, 822–832. [Google Scholar] [CrossRef]
- Huang, C.; Dong, L.; Zhao, B.; Lu, Y.; Huang, S.; Yuan, Z.; Luo, G.; Xu, Y.; Qian, W. Anti-inflammatory hydrogel dressings and skin wound healing. Clin. Transl. Med. 2022, 12, e1094. [Google Scholar] [CrossRef] [PubMed]
- Bartkowiakb, G.; Frydrych, I. 23—Superabsorbents and Their Medical Applications. In Handbook of Medical Textiles; Bartels, V.T., Ed.; Woodhead Publishing: Sawston, UK, 2011; pp. 505–546. [Google Scholar]
- Weller, C. 4—Interactive Dressings and Their Role in Moist Wound Management. In Advanced Textiles for Wound Care; Rajendran, S., Ed.; Woodhead Publishing: Sawston, UK, 2009; pp. 97–113. [Google Scholar]
- Firlar, I.; Altunbek, M.; McCarthy, C.; Ramalingam, M.; Camci-Unal, G. Functional Hydrogels for Treatment of Chronic Wounds. Gels 2022, 8, 127. [Google Scholar] [CrossRef] [PubMed]
- Zawani, M.; Fauzi, M.B. Injectable Hydrogels for Chronic Skin Wound Management: A Concise Review. Biomedicines 2021, 9, 527. [Google Scholar] [CrossRef] [PubMed]
- Park, S.G.; Li, M.-X.; Cho, W.K.; Joung, Y.K.; Huh, K.M. Thermosensitive gallic acid-conjugated hexanoyl glycol chitosan as a novel wound healing biomaterial. Carbohydr. Polym. 2021, 260, 117808. [Google Scholar] [CrossRef]
- Kong, F.; Fan, C.; Yang, Y.; Lee, B.H.; Wei, K. 5-hydroxymethylfurfural-embedded poly (vinyl alcohol)/sodium alginate hybrid hydrogels accelerate wound healing. Int. J. Biol. Macromol. 2019, 138, 933–949. [Google Scholar] [CrossRef]
- Deng, Y.; Chen, J.; Huang, J.; Yang, X.; Zhang, X.; Yuan, S.; Liao, W. Preparation and characterization of cellulose/flaxseed gum composite hydrogel and its hemostatic and wound healing functions evaluation. Cellulose 2020, 27, 3971–3988. [Google Scholar] [CrossRef]
- Singh, B.; Sharma, S.; Dhiman, A. Acacia gum polysaccharide based hydrogel wound dressings: Synthesis, characterization, drug delivery and biomedical properties. Carbohydr. Polym. 2017, 165, 294–303. [Google Scholar] [CrossRef]
- Antezana, P.E.; Municoy, S.; Pérez, C.J.; Desimone, M.F. Collagen Hydrogels Loaded with Silver Nanoparticles and Cannabis Sativa Oil. Antibiotics 2021, 10, 1420. [Google Scholar] [CrossRef]
- Fadilah, N.I.M.; Ahmad, H.; Abdrahman, M.F.; Rahman, N.A. Electrospinning of Poly (vinyl) Alcohol Nanofibers Containing Mesoporous Silica Nanoparticles. In Proceedings of the 5th International Symposium on Applied Engineering and Sciences (SAES2017), Serdang, Malaysia, 14–15 November 2017. [Google Scholar]
- Xu, S.; Deng, L.; Zhang, J.; Yin, L.; Dong, A. Composites of electrospun-fibers and hydrogels: A potential solution to current challenges in biological and biomedical field. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 640–656. [Google Scholar] [CrossRef]
- Yuan, T.T.; DiGeorge Foushee, A.M.; Johnson, M.C.; Jockheck-Clark, A.R.; Stahl, J.M. Development of Electrospun Chitosan-Polyethylene Oxide/Fibrinogen Biocomposite for Potential Wound Healing Applications. Nanoscale Res. Lett. 2018, 13, 88. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, H.; Zhang, M.; Yu, D.-G. Electrospun Medicated Nanofibers for Wound Healing: Review. Membranes 2021, 11, 770. [Google Scholar] [CrossRef] [PubMed]
- Kandhasamy, S.; Liang, B.; Yang, D.-P.; Zeng, Y. Antibacterial vitamin K3 carnosine peptide-laden silk fibroin electrospun fibers for improvement of skin wound healing in diabetic rats. ACS Appl. Bio Mater. 2021, 4, 4769–4788. [Google Scholar] [CrossRef] [PubMed]
- Augustine, G.; Aarthy, M.; Thiagarajan, H.; Selvaraj, S.; Kamini, N.R.; Shanmugam, G.; Ayyadurai, N. Self-Assembly and Mechanical Properties of Engineered Protein Based Multifunctional Nanofiber for Accelerated Wound Healing. Adv. Healthc. Mater. 2021, 10, 2001832. [Google Scholar] [CrossRef] [PubMed]
- Fereshteh, Z. Functional 3D Tissue Engineering Scaffolds; Elsevier: Amsterdam, The Netherlands, 2018; Volume 24, pp. 3115–3123. [Google Scholar]
- Wahid, F.; Khan, T.; Hussain, Z.; Ullah, H. Nanocomposite Scaffolds for Tissue Engineering; Properties, Preparation and Applications. In Applications of Nanocomposite Materials in Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2018; pp. 701–735. [Google Scholar]
- Cao, S.; Xu, G.; Li, Q.; Zhang, S.; Yang, Y.; Chen, J. Double crosslinking chitosan sponge with antibacterial and hemostatic properties for accelerating wound repair. Compos. Part B Eng. 2022, 234, 109746. [Google Scholar] [CrossRef]
- Tamer, T.M.; Alsehli, M.H.; Omer, A.M.; Afifi, T.H.; Sabet, M.M.; Mohy-Eldin, M.S.; Hassan, M.A. Development of Polyvinyl Alcohol/Kaolin Sponges Stimulated by Marjoram as Hemostatic, Antibacterial, and Antioxidant Dressings for Wound Healing Promotion. Int. J. Mol. Sci. 2021, 22, 13050. [Google Scholar] [CrossRef]
- Mihai, M.M.; Dima, M.B.; Dima, B.; Holban, A.M. Nanomaterials for Wound Healing and Infection Control. Materials 2019, 12, 2176. [Google Scholar] [CrossRef]
- Niska, K.; Zielinska, E.; Radomski, M.W.; Inkielewicz-Stepniak, I. Metal nanoparticles in dermatology and cosmetology: Interactions with human skin cells. Chem. Biol. Interact. 2018, 295, 38–51. [Google Scholar] [CrossRef]
- Fadilah, N.I.M.; Isa, I.L.M.; Zaman, W.S.W.K.; Tabata, Y.; Fauzi, M.B. The Effect of Nanoparticle-Incorporated Natural-Based Biomaterials towards Cells on Activated Pathways: A Systematic Review. Polymers 2022, 14, 476. [Google Scholar] [CrossRef]
- Kushwaha, A.; Goswami, L.; Kim, B.S. Nanomaterial-Based Therapy for Wound Healing. Nanomaterials 2022, 12, 618. [Google Scholar] [CrossRef]
- Sharma, M.; Yadav, S.; Ganesh, N.; Srivastava, M.M.; Srivastava, S. Biofabrication and characterization of flavonoid-loaded Ag, Au, Au–Ag bimetallic nanoparticles using seed extract of the plant Madhuca longifolia for the enhancement in wound healing bio-efficacy. Prog. Biomater. 2019, 8, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Liang, J.; Liu, H.; Zhang, K.; Li, J. Nanomaterials for angiogenesis in skin tissue engineering. Tissue Eng. Part B Rev. 2020, 26, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Lau, P.; Bidin, N.; Islam, S.; Shukri, W.N.B.W.M.; Zakaria, N.; Musa, N.; Krishnan, G. Influence of gold nanoparticles on wound healing treatment in rat model: Photobiomodulation therapy. Lasers Surg. Med. 2017, 49, 380–386. [Google Scholar] [CrossRef]
- Nosrati, H.; Khouy, R.A.; Nosrati, A.; Khodaei, M.; Banitalebi-Dehkordi, M.; Ashrafi-Dehkordi, K.; Sanami, S.; Alizadeh, Z. Nanocomposite scaffolds for accelerating chronic wound healing by enhancing angiogenesis. J. Nanobiotechnol. 2021, 19, 1–21. [Google Scholar] [CrossRef]
- Hu, B.; Gao, M.; Boakye-Yiadom, K.O.; Ho, W.; Yu, W.; Xu, X.; Zhang, X.-Q. An intrinsically bioactive hydrogel with on-demand drug release behaviors for diabetic wound healing. Bioact. Mater. 2021, 6, 4592–4606. [Google Scholar] [CrossRef]
- Niranjan, R.; Kaushik, M.; Selvi, R.T.; Prakash, J.; Venkataprasanna, K.; Prema, D.; Pannerselvam, B.; Venkatasubbu, G.D. PVA/SA/TiO2-CUR patch for enhanced wound healing application: In vitro and in vivo analysis. Int. J. Biol. Macromol. 2019, 138, 704–717. [Google Scholar] [CrossRef] [PubMed]
- Dang, L.H.; Huynh, N.T.; Pham, N.O.; Nguyen, C.T.; Vu, M.T.; Dinh, V.T.; Le, V.T.; Tran, N.Q. Injectable nanocurcumin-dispersed gelatin–pluronic nanocomposite hydrogel platform for burn wound treatment. Bull. Mater. Sci. 2019, 42, 71. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, B.; Sun, Y.; Wang, C.; Mukherjee, S.; Yang, C.; Chen, Y. Injectable Enzyme-Based Hydrogel Matrix with Precisely Oxidative Stress Defense for Promoting Dermal Repair of Burn Wound. Macromol. Biosci. 2020, 20, 2000036. [Google Scholar] [CrossRef] [PubMed]
- Yalman, V.; Laçin, N.T. Development of humic acid and alginate-based wound dressing and evaluation on inflammation. Mater. Technol. 2019, 34, 705–717. [Google Scholar] [CrossRef]
- Raja, S.T.K.; Thiruselvi, T.; Aravindhan, R.; Mandal, A.B.; Gnanamani, A. In vitro and in vivo assessments of a 3-(3,4-dihydroxyphenyl)-2-propenoic acid bioconjugated gelatin-based injectable hydrogel for biomedical applications. J. Mater. Chem. B 2015, 3, 1230–1244. [Google Scholar] [CrossRef]
- Ruffo, M.; Parisi, O.I.; Dattilo, M.; Patitucci, F.; Malivindi, R.; Pezzi, V.; Tzanov, T.; Puoci, F. Synthesis and evaluation of wound healing properties of hydro-diab hydrogel loaded with green-synthetized AGNPS: In vitro and in ex vivo studies. Drug Deliv. Transl. Res. 2022, 12, 1881–1894. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Kong, Y.; Su, Y.; Kuss, M.A.; Jiang, X.; Li, X.; Xie, J.; Duan, B. Tannic acid-inspired, self-healing, and dual stimuli responsive dynamic hydrogel with potent antibacterial and anti-oxidative properties. J. Mater. Chem. B 2021, 9, 7182–7195. [Google Scholar] [CrossRef] [PubMed]
- Gomathi, K.; Gopinath, D.; Ahmed, M.R.; Jayakumar, R. Quercetin incorporated collagen matrices for dermal wound healing processes in rat. Biomaterials 2003, 24, 2767–2772. [Google Scholar] [CrossRef] [PubMed]
- Balasubramaniam, M.P.; Murugan, P.; Chenthamara, D.; Ramakrishnan, S.G.; Salim, A.; Lin, F.-H.; Robert, B.; Subramaniam, S. Synthesis of chitosan-ferulic acid conjugated poly (vinyl alcohol) polymer film for an improved wound healing. Mater. Today Commun. 2020, 25, 101510. [Google Scholar] [CrossRef]
- Nayak, K.K.; Gupta, P. Study of the keratin-based therapeutic dermal patches for the delivery of bioactive molecules for wound treatment. Mater. Sci. Eng. C 2017, 77, 1088–1097. [Google Scholar] [CrossRef] [PubMed]
- Gaydhane, M.K.; Kanuganti, J.S.; Sharma, C.S. Honey and curcumin loaded multilayered polyvinylalcohol/cellulose acetate electrospun nanofibrous mat for wound healing. J. Mater. Res. 2020, 35, 600–609. [Google Scholar] [CrossRef]
- Zhang, Z.; Dai, Q.; Zhang, Y.; Zhuang, H.; Wang, E.; Xu, Q.; Ma, L.; Wu, C.; Huan, Z.; Guo, F. Design of a multifunctional biomaterial inspired by ancient Chinese medicine for hair regeneration in burned skin. ACS Appl. Mater. Interfaces 2020, 12, 12489–12499. [Google Scholar] [CrossRef]
- Kontogiannopoulos, K.N.; Assimopoulou, A.N.; Tsivintzelis, I.; Panayiotou, C.; Papageorgiou, V.P. Electrospun fiber mats containing shikonin and derivatives with potential biomedical applications. Int. J. Pharm. 2011, 409, 216–228. [Google Scholar] [CrossRef]
- Kandhasamy, S.; Arthi, N.; Arun, R.P.; Verma, R.S. Synthesis and fabrication of novel quinone-based chromenopyrazole antioxidant-laden silk fibroin nanofibers scaffold for tissue engineering applications. Mater. Sci. Eng. C 2019, 102, 773–787. [Google Scholar] [CrossRef]
- Ni, Y.; Lin, W.; Mu, R.; Wu, C.; Lin, Z.; Chen, S.; Pang, J. Facile fabrication of novel konjac glucomannan films with antibacterial properties via microfluidic spinning strategy. Carbohydr. Polym. 2019, 208, 469–476. [Google Scholar] [CrossRef]
- Prabhahar, M.; Kannayiram, G.; Prakash, S.; Saravanakumar, M.; Krishnamoorthi, S.; Sendilvelan, S.; Senthan, P.R.; Ashikmon, T.; Karthik, M.; Lenin, H. Physicochemical Characterization of Star Anise Silver Nanoparticles Incorporated Chitosan Biomaterial for Absorb Water and Cure Wounds. Adsorpt. Sci. Technol. 2022, 2022, 7522512. [Google Scholar] [CrossRef]
- Han, S.; Ahmeda, A.; Jalalvand, A.R.; Lu, W.; Zangeneh, M.M.; Zangeneh, A. Application of silver nanoparticles containing Gundelia tournefortii L. leaf aqueous extract in the treatment of microbial diseases and cutaneous wound healing. Appl. Organomet. Chem. 2020, 36, e5491. [Google Scholar] [CrossRef]
- Govindarajan, D.; Duraipandy, N.; Srivatsan, K.V.; Lakra, R.; Korapatti, P.S.; Jayavel, R.; Kiran, M.S. Fabrication of hybrid collagen aerogels reinforced with wheat grass bioactives as instructive scaffolds for collagen turnover and angiogenesis for wound healing applications. ACS Appl. Mater. Interfaces 2017, 9, 16939–16950. [Google Scholar] [CrossRef] [PubMed]
- Bölgen, N.; Demir, D.; Yalçın, M.S.; Özdemir, S. Development of Hypericum perforatum oil incorporated antimicrobial and antioxidant chitosan cryogel as a wound dressing material. Int. J. Biol. Macromol. 2020, 161, 1581–1590. [Google Scholar] [CrossRef] [PubMed]
- Jebahi, S.; Saoudi, M.; Farhat, L.; Oudadesse, H.; Rebai, T.; Kabir, A.; El Feki, A.; Keskes, H. Effect of novel curcumin-encapsulated chitosan–bioglass drug on bone and skin repair after gamma radiation: Experimental study on a Wistar rat model. Cell Biochem. Funct. 2015, 33, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Zawani, M.; Fauzi, M.B. Epigallocatechin Gallate: The Emerging Wound Healing Potential of Multifunctional Biomaterials for Future Precision Medicine Treatment Strategies. Polymers 2021, 13, 3656. [Google Scholar] [CrossRef] [PubMed]
- Wasim, L.; Chopra, M. Synergistic anticancer effect of panobinostat and topoisomerase inhibitors through ROS generation and intrinsic apoptotic pathway induction in cervical cancer cells. Cell. Oncol. 2018, 41, 201–212. [Google Scholar] [CrossRef]
- Viaña-Mendieta, P.; Sánchez, M.L.; Benavides, J. Rational selection of bioactive principles for wound healing applications: Growth factors and antioxidants. Int. Wound J. 2022, 19, 100–113. [Google Scholar] [CrossRef]
- Son, Y.J.; John, W.T.; Zhou, Y.; Mao, W.; Yim, E.K.; Yoo, H.S. Biomaterials and controlled release strategy for epithelial wound healing. Biomater. Sci. 2019, 7, 4444–4471. [Google Scholar] [CrossRef]
- Ghorbani, M.; Ramezani, S.; Rashidi, M.-R. Fabrication of honey-loaded ethylcellulose/gum tragacanth nanofibers as an effective antibacterial wound dressing. Colloids Surf. A Physicochem. Eng. Asp. 2021, 621, 126615. [Google Scholar] [CrossRef]
- Garcia, E.J.; Oldoni, T.L.C.; Alencar, S.M.d.; Reis, A.; Loguercio, A.D.; Grande, R.H.M. Antioxidant activity by DPPH assay of potential solutions to be applied on bleached teeth. Braz. Dent. J. 2012, 23, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Marjoni, M.; Zulfisa, A. Antioxidant activity of methanol extract/fractions of senggani leaves (Melastoma candidum D. Don). Pharm. Anal. Acta 2017, 8, 1–6. [Google Scholar]
- Ilyasov, I.R.; Beloborodov, V.L.; Selivanova, I.A.; Terekhov, R.P. ABTS/PP decolorization assay of antioxidant capacity reaction pathways. Int. J. Mol. Sci. 2020, 21, 1131. [Google Scholar] [CrossRef] [PubMed]
- Bedlovičová, Z.; Strapáč, I.; Baláž, M.; Salayová, A. A brief overview on antioxidant activity determination of silver nanoparticles. Molecules 2020, 25, 3191. [Google Scholar] [CrossRef] [PubMed]
- Posselt, J.R.; Grodsky, E. Graduate education and social stratification. Annu. Rev. Sociol. 2017, 43, 353. [Google Scholar] [CrossRef]
- Nova, Z.; Skovierova, H.; Strnadel, J.; Halasova, E.; Calkovska, A. Short-term versus long-term culture of A549 cells for evaluating the effects of lipopolysaccharide on oxidative stress, surfactant proteins and cathelicidin LL-37. Int. J. Mol. Sci. 2020, 21, 1148. [Google Scholar] [CrossRef]
- Hirsch, T.; Rothoeft, T.; Teig, N.; Bauer, J.W.; Pellegrini, G.; De Rosa, L.; Scaglione, D.; Reichelt, J.; Klausegger, A.; Kneisz, D. Regeneration of the entire human epidermis using transgenic stem cells. Nature 2017, 551, 327–332. [Google Scholar] [CrossRef]
- Rama, P.; Matuska, S.; Paganoni, G.; Spinelli, A.; De Luca, M.; Pellegrini, G. Limbal stem-cell therapy and long-term corneal regeneration. N. Engl. J. Med. 2010, 363, 147–155. [Google Scholar] [CrossRef]
- Meerloo, J.V.; Kaspers, G.J.; Cloos, J. Cell sensitivity assay: The MTT assay. In Cancer Cell Culture; Springer: Cham, Switzerland, 2011; pp. 237–245. [Google Scholar]
- Zawani, M.; Maarof, M.; Tabata, Y.; Motta, A.; Fauzi, M.B. Quercetin-Embedded Gelastin Injectable Hydrogel as Provisional Biotemplate for Future Cutaneous Application: Optimization and In Vitro Evaluation. Gels 2022, 8, 623. [Google Scholar] [CrossRef]
- Pastar, I.; Liang, L.; Sawaya, A.P.; Wikramanayake, T.C.; Glinos, G.D.; Drakulich, S.; Chen, V.; Stojadinovic, O.; Davis, S.C.; Tomic-Canic, M. Preclinical Models for Wound-Healing Studies. In Skin Tissue Models; Elsevier: Amsterdam, The Netherlands, 2018; pp. 223–253. [Google Scholar]
- Novus Biologicals, Immunocytochemistry (ICC) Handbook. Bio Techne 2015, 1–24. Available online: https://www.novusbio.com/support/immunocytochemistry-icc-handbook (accessed on 1 December 2022).
- Yousef, H.; Alhajj, M.; Sharma, S. Anatomy, Skin (Integument), Epidermis. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Wang, Y.; Graves, D.T. Keratinocyte Function in Normal and Diabetic Wounds and Modulation by FOXO1. J. Diabetes Res. 2020, 2020, 3714704. [Google Scholar] [CrossRef]
- Warinhomhoun, S.; Muangnoi, C.; Buranasudja, V.; Mekboonsonglarp, W.; Rojsitthisak, P.; Likhitwitayawuid, K.; Sritularak, B. Antioxidant Activities and Protective Effects of Dendropachol, a New Bisbibenzyl Compound from Dendrobium pachyglossum, on Hydrogen Peroxide-Induced Oxidative Stress in HaCaT Keratinocytes. Antioxidants 2021, 10, 252. [Google Scholar] [CrossRef] [PubMed]
- Mehrabani, D.; Manafi, N. Role of cultured skin fibroblasts in aesthetic and plastic surgery. World J. Plast. Surg. 2013, 2, 2–5. [Google Scholar] [PubMed]
- Merecz-Sadowska, A.; Sitarek, P.; Kucharska, E.; Kowalczyk, T.; Zajdel, K.; Cegliński, T.; Zajdel, R. Antioxidant Properties of Plant-Derived Phenolic Compounds and Their Effect on Skin Fibroblast Cells. Antioxidants 2021, 10, 726. [Google Scholar] [CrossRef] [PubMed]
- Zenin, V.; Ivanova, J.; Pugovkina, N.; Shatrova, A.; Aksenov, N.; Tyuryaeva, I.; Kirpichnikova, K.; Kuneev, I.; Zhuravlev, A.; Osyaeva, E.; et al. Resistance to H2O2-induced oxidative stress in human cells of different phenotypes. Redox Biol. 2022, 50, 102245. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Cheng, W.; Wang, Q.; Zhang, J.; Wang, C.; Li, M.; Zhao, D.; Wang, D.; An, Q. Exploring the Protective and Reparative Mechanisms of G. lucidum Polysaccharides against H2O2-Induced Oxidative Stress in Human Skin Fibroblasts. Clin. Cosmet. Investig. Dermatol. 2021, 14, 1481–1496. [Google Scholar] [CrossRef]
- O’Toole, E.A.; Goel, M.; Woodley, D.T. Hydrogen peroxide inhibits human keratinocyte migration. Dermatol. Surg. 1996, 22, 525–529. [Google Scholar] [CrossRef]
- Svobodová, A.; Zdarilová, A.; Walterová, D.; Vostálová, J. Flavonolignans from Silybum marianum moderate UVA-induced oxidative damage to HaCaT keratinocytes. J. Dermatol. Sci. 2007, 48, 213–224. [Google Scholar] [CrossRef]
- Vostálová, J.; Zdarilová, A.; Svobodová, A. Prunella vulgaris extract and rosmarinic acid prevent UVB-induced DNA damage and oxidative stress in HaCaT keratinocytes. Arch. Dermatol. Res. 2010, 302, 171–181. [Google Scholar] [CrossRef]
- Du, C.; Liu, J.; Fikhman, D.A.; Dong, K.S.; Monroe, M.B.B. Shape Memory Polymer Foams with Phenolic Acid-Based Antioxidant and Antimicrobial Properties for Traumatic Wound Healing. Front. Bioeng. Biotechnol. 2022, 10, 809361. [Google Scholar] [CrossRef]
- Hickman, D.; Johnson, J.; Vemulapalli, T.; Crisler, J.; Shepherd, R. Commonly used animal models. Princ. Anim. Res. Grad. Undergrad. Stud. 2017, 117–175. [Google Scholar] [CrossRef]
- Ribitsch, I.; Baptista, P.M.; Lange-Consiglio, A.; Melotti, L.; Patruno, M.; Jenner, F.; Schnabl-Feichter, E.; Dutton, L.C.; Connolly, D.J.; van Steenbeek, F.G.; et al. Large Animal Models in Regenerative Medicine and Tissue Engineering: To Do or Not to Do. Front. Bioeng. Biotechnol. 2020, 8, 972. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Yang, F.; Zhou, W.; Xiao, N.; Luo, M.; Tang, Z. The initiation of oxidative stress and therapeutic strategies in wound healing. Biomed. Pharmacother. 2023, 157, 114004. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, T.; Shaiful Hadi, N.; Sulaiman, S.; Fauzi, M.B.; Idrus, R.B.H.; Chowdhury, S.R.; Law, J.X.; Maarof, M. Comparison of three different skin substitutes in promoting wound healing in an ovine model. Burns 2022, 48, 1198–1208. [Google Scholar] [CrossRef]
- Castro, B.; Palomares, T.; Azcoitia, I.; Bastida, F.; del Olmo, M.; Soldevilla, J.J.; Alonso-Varona, A. Development and preclinical evaluation of a new galactomannan-based dressing with antioxidant properties for wound healing. Histol. Histopathol. 2015, 30, 1499–1512. [Google Scholar] [CrossRef] [PubMed]
- Castro, B.; Bastida, F.D.; Segovia, T.; Casanova, P.L.; Soldevilla, J.; Verdú-Soriano, J. The use of an antioxidant dressing on hard-to-heal wounds: A multicentre, prospective case series. J. Wound Care 2017, 26, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Przekora, A. A concise review on tissue engineered artificial skin grafts for chronic wound treatment: Can we reconstruct functional skin tissue in vitro? Cells 2020, 9, 1622. [Google Scholar] [CrossRef]
- Mo, X.; Cen, J.; Gibson, E.; Wang, R.; Percival, S.L. An open multicenter comparative randomized clinical study on chitosan. Wound Repair Regen. 2015, 23, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-García, J.F.; Aguilera-Manrique, G.; Parra-Anguita, L.; Torra-Bou, J.E.; Villegas-Tripiana, I.; García-Fernández, F.P. An antioxidant approach directed to non-healing wounds activation—A case report study. Clin. Case Rep. Rev. 2021, 7, 1–6. [Google Scholar] [CrossRef]
- Fitzmaurice, S.; Sivamani, R.K.; Isseroff, R.R. Antioxidant therapies for wound healing: A clinical guide to currently commercially available products. Ski. Pharmacol. Physiol. 2011, 24, 113–126. [Google Scholar] [CrossRef]
- Darwin, E.; Tomic-Canic, M. Healing chronic wounds: Current challenges and potential solutions. Curr. Dermatol. Rep. 2018, 7, 296–302. [Google Scholar] [CrossRef]
- Halim, A.S.; Nor, F.M.; Saad, A.Z.M.; Nasir, N.A.M.; Norsa’adah, B.; Ujang, Z. Efficacy of chitosan derivative films versus hydrocolloid dressing on superficial wounds. J. Taibah Univ. Med. Sci. 2018, 13, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Duncan, C.L.; Enlow, P.T.; Szabo, M.M.; Tolchin, E.; Kelly, R.W.; Castanon, L.; Aballay, A.M. A Pilot Study of the Efficacy of Active Leptospermum Honey for the Treatment of Partial-Thickness Facial Burns. Adv. Ski. Wound Care 2016, 29, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Gallelli, G.; Cione, E.; Serra, R.; Leo, A.; Citraro, R.; Matricardi, P.; Di Meo, C.; Bisceglia, F.; Caroleo, M.C.; Basile, S.; et al. Nano-hydrogel embedded with quercetin and oleic acid as a new formulation in the treatment of diabetic foot ulcer: A pilot study. Int. Wound J. 2020, 17, 485–490. [Google Scholar] [CrossRef] [PubMed]
| Antioxidant Assay | Antioxidant Component | Biomaterial | Type of Scaffold | Outcomes | References |
|---|---|---|---|---|---|
| DPPH | Sarasinula marginata extract | Natural rubber latex | Membrane | 8.53 µg mL−1 Sarasinula marginata extract able to achieve more than 50% inhibition of radical when compared to ascorbic acid. | [74] |
| Chitosan Curcumin | Hyaluronic acid and chitosan | Hydrogel | Concentration of 1–100 µg of curcumin gradually increase the scavenging activity by 33.6% to 84.6%. | [145] | |
| Catechol moiety | Not stated | Nanofibre | The bioactive nanofibrous mat manage to scavenge 50% of DPPH while the native construct only achieves 12.5% and 5%. | [132] | |
| Ferulic acid | Polyvinyl alcohol and chitosan | Film | Increasing concentration of bioactive molecules, increase the free radical scavenging activity with maximum value of 97.2% for 500 µL ferulic acid | [154] | |
| Plantago lanceolata Tagates patula Symphytum officinale Calendula officinalis Geum urbanum | Chitosan | Film | The evaluation of DPPH shown that bioactive compound is 1.953 mg TE/g | [109] | |
| Honey Curcumin | Polyvinyl alcohol and cellulose acetate | Nanofibrous mat | The honey-curcumin integrated scaffolds able to achieve 93% free radical activity | [156] | |
| Acacia gum | Acacia gum polysaccharide | Hydrogel | The bioactive film exhibits 51.35% of DPPH free radical inhibition | [125] | |
| Star anise | Chitosan | Not stated | Star anise able to reach 100% inhibition of DPPH at concentration of 25 µg/mL | [161] | |
| Honey | Ethylcellulose/gum tragacanth | Nanofibre | Electrospun fibre incorporated with 20% honey have more than 60% at 9 h incubation | [170] | |
| Cannabis sativa | Collagen | Hydrogel | The highest scavenging activity is 67 mg/g of cannabis sativa which value at 47.20% | [126] | |
| Gundelia tournefortii | Not stated | Ointment | The scavenging activity of bioactive compound is similar with AgNP which is 376 µg/mL | [162] | |
| Papain Alginate | Keratin/alginate, Keratin/agar, and Keratin/gellan | Patches | Bioactive compound able to decrease the DPPH concentration by 30.55% | [155] | |
| Propenoic acid | Gelatin | Hydrogel | The radical scavenging activity of biomaterials at 50% with concentration of 175 µg and 350 µg | [150] | |
| Curcumin | Chitosan-alginate | Nanofibre | Curcumin encapsulated biomaterial able to inhibit 41.37% DPPH at 60 µg/mL | [112] | |
| Olive leaves Camellia sinensis extract | Carboxymethylcellulose | Hydrogel | Incorporation of bioactive compound in hydrogel exhibit 83% radical scavenging activity | [151] | |
| Tannic acid | Hyaluronic acid | Hydrogel | The presence of tannic acid able to inhibit 80% of DPPH | [152] | |
| Hypericum perforatum oil | Chitosan | Cryogel | The scavenging activity increased to 53.2% when bioactive compound integrated into bioscaffold | [164] | |
| Quinone | Silk-fibroin | Nanofibre | The antioxidant activity resulted in increased IC50% at 5.5 µg of quinone incorporated in nanofibre | [159] | |
| ABTS | Ferulic acid | Chitosan-alginate | Nanofibre | The presence of ferulic acid able to reduce ABTS more than 1.5 time of native biofilm | [112] |
| Plantago lanceolata Tagates patula Symphytum officinale Calendula officinalis Geum urbanum | Chitosan | Film | The bio-composite result in ABTS value of 1.745 mg TE/g | [109] | |
| Olive leaves Camellia sinensis extract | Carboxymethylcellulose | Hydrogel | The compound result in 83% ABTS radical | [151] | |
| DCFH-DA | Lignin | Lignin | Nanogel | Lignin able to reduce the intensity of DCF fluorescence signal indicate reduction of intracellular ROS | [62] |
| Curcumin Chitosan | Hyaluronic acid and chitosan | Hydrogel | The treatment group decrease the fluorescence intensity with concentration as low as 1 µg | [145] | |
| Vitamin E | Not stated | Hydrogel | Significant decrease in DCF fluorescence intensity with treatment group of 600 µg/mL | [148] | |
| Tannic acid | Phenylboronic acid-modified hyaluronic acid | Hydrogel | Treatment group is significantly low DCF fluorescence intensity | [152] | |
| Total antioxidant status kit | Humic acid Alginate | Alginate | Hydrogel | Dual compounds show higher antioxidant capacity, which is 0.25 nM compared to alginate composite, 0.22 nM | [149] |
| Lipid peroxidation | Quercetin | Collagen | Film | The absorbance of AIBN increases when quercetin was added | [153] |
| Wound Type | Antioxidant Biomaterial | Parameter | Animal Model | Outcomes | References |
|---|---|---|---|---|---|
| Burn wound | Lignin nanogel | Wound closure, H&E, Ki67 | Balb/c mice | Wound closure: Accelerate recovery. Ki67: protein increased and accelerate cell proliferation were observed. H&E: Low inflammatory cells in the treated wound, hair follicles and epithelium regeneration were observed. | [62] |
| Chitosan films | Wound closure, H&E | Wistar rats | Wound closure: vascularisation and improved wound, formation of new epithelial layer on day 14. H&E: complete re-epithelialisation, mature epidermal, reduction of inflammatory infiltrate and congestion. | [154] | |
| Full-thickness wound | Quercetin films | Wound closure, hydroxyproline, uronic acid, total protein, superoxide dismutase, catalase | Albino Wistar rats | Wound closure: 20% wound contraction; increase hydroxy proline (1.836 mg/100 mg tissue) and protein (76 mg/g tissue) content compared to control. | [153] |
| Gallic acid hydrogel | Wound closure, Growth factor expression, H&E | Balb/c mice | H&E: 20% higher wound contraction (Day 10); wound completely heal (Day 15); 60 μm thicker granulation tissue Growth factor expression: High expression of TGF-β 13.6-; EGF 5.5-; VEGF fold on day 5, day 10 (6.3-fold) and day 15 (2.7-fold) | [122] | |
| 5-hydroxymethylfurfural hydrogel | Wound closure, H&E, IHC | Sprague Dawley rats | Wound closure: Higher rate of wound closure H&E: Higher collagen disposition, wound tissue structure almost similar to healthy tissue. IHC: Significantly high VEGF expression and blood vessel formation compared to other groups. | [123] | |
| Catechol moiety nanofibre | Wound closure, H&E, IHC | Wistar rats | Wound closure: Complete wound closure (day 20) compared to control (70%). Significantly low granulation tissue and proliferation of fibroblastic. H&E: Show uniform collagen bundles, complete re-epithelialisation and expression of cytokeratin on day 20 | [132] | |
| Curcumin hydrogels | Wound closure, Hydroxyproline, H&E, SEM | Wistar albino rats | Wound closure: wound shrinkage, 50% wound closure at day 16 H&E: No visible inflammation or fibrinoid. Show re-epithelialisation and rapid hair growth SEM: Show newly formed collagen and fibroblast in the epidermal | [146] | |
| Flaxseed gum hydrogels | Wound closure | C57BL/6 mice | Wound closure: (Day 10) Capillaries formation, differentiated collagen fibres, clear tissue structure, rapid hair follicles proliferation. | [124] | |
| Madhuca longifolia seed nanoparticles | Wound closure, H&E | Swiss albino mice | Wound closure: Faster wound closure rate (80.33%) and wound epithelisation (18 days) compared to the controls. H&E: Stratum corneum recover and rapid hair growth | [124] | |
| Acacia gum hydrogel | H&E | Balb/c mice | H&E: (Day 12) High collagen production, vessel formation and negligible inflammation | [125] | |
| Wheat grass aerogel | Wound closure | Wistar rats | Wound closure: (DAY 9) 75% wound reduction, significantly high rate of wound size reduction compared to other groups. Healed wound were observed on day 18. | [163] | |
| Gundelia tournefortii nanoparticles | Wound closure, H&E | Albino mice | Wound closure: Significantly decreased wound area compared to other groups. H&E: significantly reduced neutrophils and lymphocytes, high formation of blood vessels, fibrocytes, fibroblast, hydroxyproline, hexosamine and hexuronic acid compared to other groups. | [162] | |
| Konjac glucomannan- EGCG films | Wound closure, H&E | Sprague Dawley rats | Wound closure: Accelerate wound closure and healed wound on day 13. H&E: formation of keratin and epidermis layer, neovascularisation, blood vessels formation and mature hair follicles were observed. | [160] | |
| Propenoic acid hydrogels | Wound closure, H&E | Wistar rats | Wound closure: (Day 16) Complete wound closure. H&E: Significant wound closure and hair follicle growth were observed. | [150] | |
| Curcumin bioactive glass | Wound closure, H&E, Hydroxyproline | Wistar rats | Wound closure: (Day 14) complete epidermis re-epithelialisation. Hydroxyproline: Increase hydroxyproline content (33.5%) compared to other groups. | [165] | |
| Diabetic wound | Chitosan-hyaluronic acid hydrogels | Wound closure, H&E | C57BL/6 mice | Wound closure: Healing rate of 54.5%, (Day 10) 88.3% wound closure were observed. H&E: Abundant tissue granulation, compact and layered. | [145] |
| Plantago lanceolata, Tagates patula, Symphytum officinale, Calendula officinalis, Geum urbanum loaded chitosan films | Wound closure, H&E | Wistar rats | Wound closure: (Day 14) treated wound 97.47% healed H&E: rapid blood vessels proliferation, collagen fibres formation and fibroblast proliferation were observed. | [109] | |
| Vitamin K3- Carnosine peptide electrospun fibre | Wound closure, H&E | Sprague Dawley rats | Wound closure: wound closure rate (Day 3–9) 55.05 ± 10.2, 78.12 ± 14.5, 100% H&E: low number of inflammatory cells observed in the treated wound. | [131] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fadilah, N.I.M.; Phang, S.J.; Kamaruzaman, N.; Salleh, A.; Zawani, M.; Sanyal, A.; Maarof, M.; Fauzi, M.B. Antioxidant Biomaterials in Cutaneous Wound Healing and Tissue Regeneration: A Critical Review. Antioxidants 2023, 12, 787. https://doi.org/10.3390/antiox12040787
Fadilah NIM, Phang SJ, Kamaruzaman N, Salleh A, Zawani M, Sanyal A, Maarof M, Fauzi MB. Antioxidant Biomaterials in Cutaneous Wound Healing and Tissue Regeneration: A Critical Review. Antioxidants. 2023; 12(4):787. https://doi.org/10.3390/antiox12040787
Chicago/Turabian StyleFadilah, Nur Izzah Md, Shou Jin Phang, Nurkhuzaiah Kamaruzaman, Atiqah Salleh, Mazlan Zawani, Arka Sanyal, Manira Maarof, and Mh Busra Fauzi. 2023. "Antioxidant Biomaterials in Cutaneous Wound Healing and Tissue Regeneration: A Critical Review" Antioxidants 12, no. 4: 787. https://doi.org/10.3390/antiox12040787
APA StyleFadilah, N. I. M., Phang, S. J., Kamaruzaman, N., Salleh, A., Zawani, M., Sanyal, A., Maarof, M., & Fauzi, M. B. (2023). Antioxidant Biomaterials in Cutaneous Wound Healing and Tissue Regeneration: A Critical Review. Antioxidants, 12(4), 787. https://doi.org/10.3390/antiox12040787













