Plant-Based Antioxidants for Prevention and Treatment of Neurodegenerative Diseases: Phytotherapeutic Potential of Laurus nobilis, Aronia melanocarpa, and Celastrol
Abstract
1. Introduction
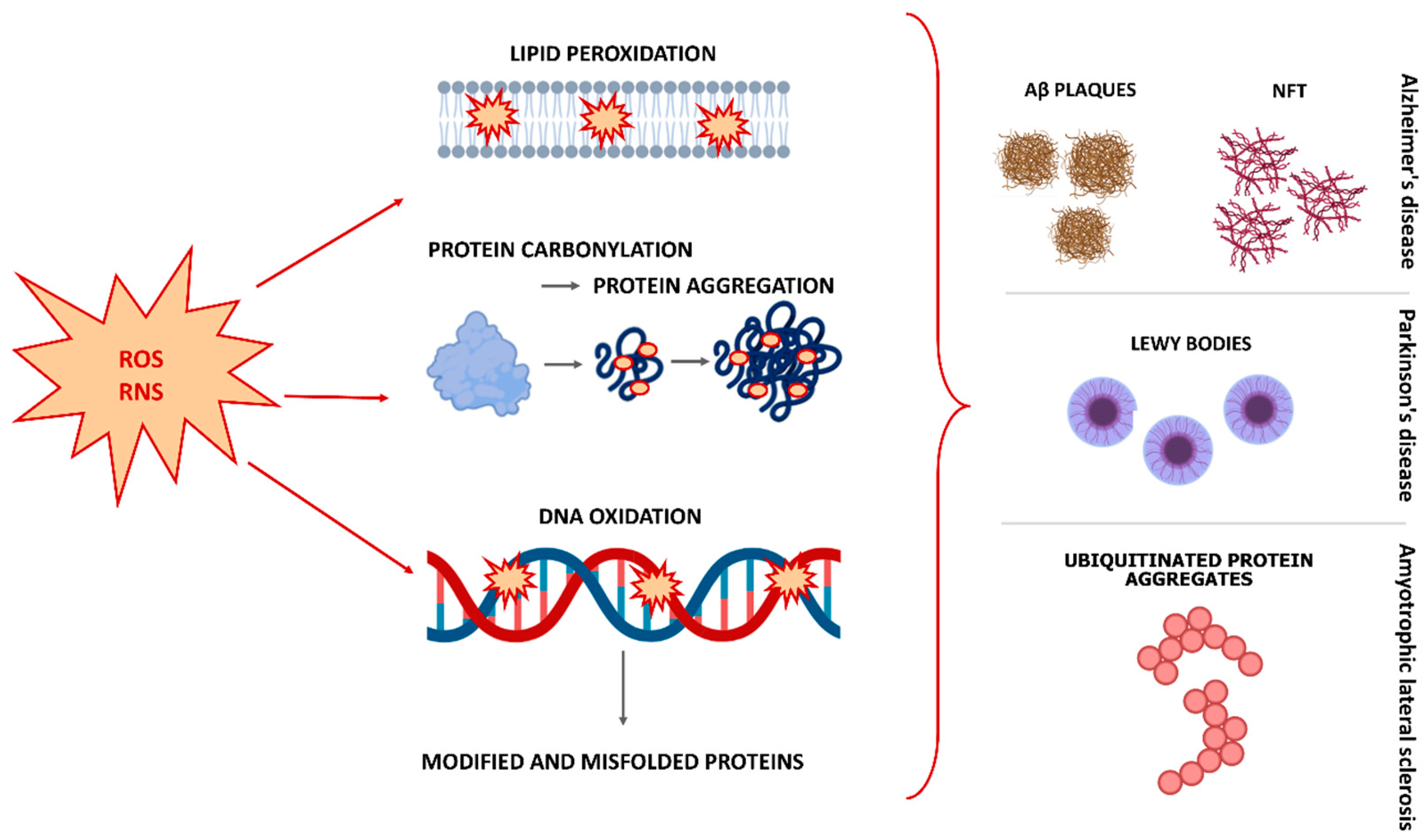
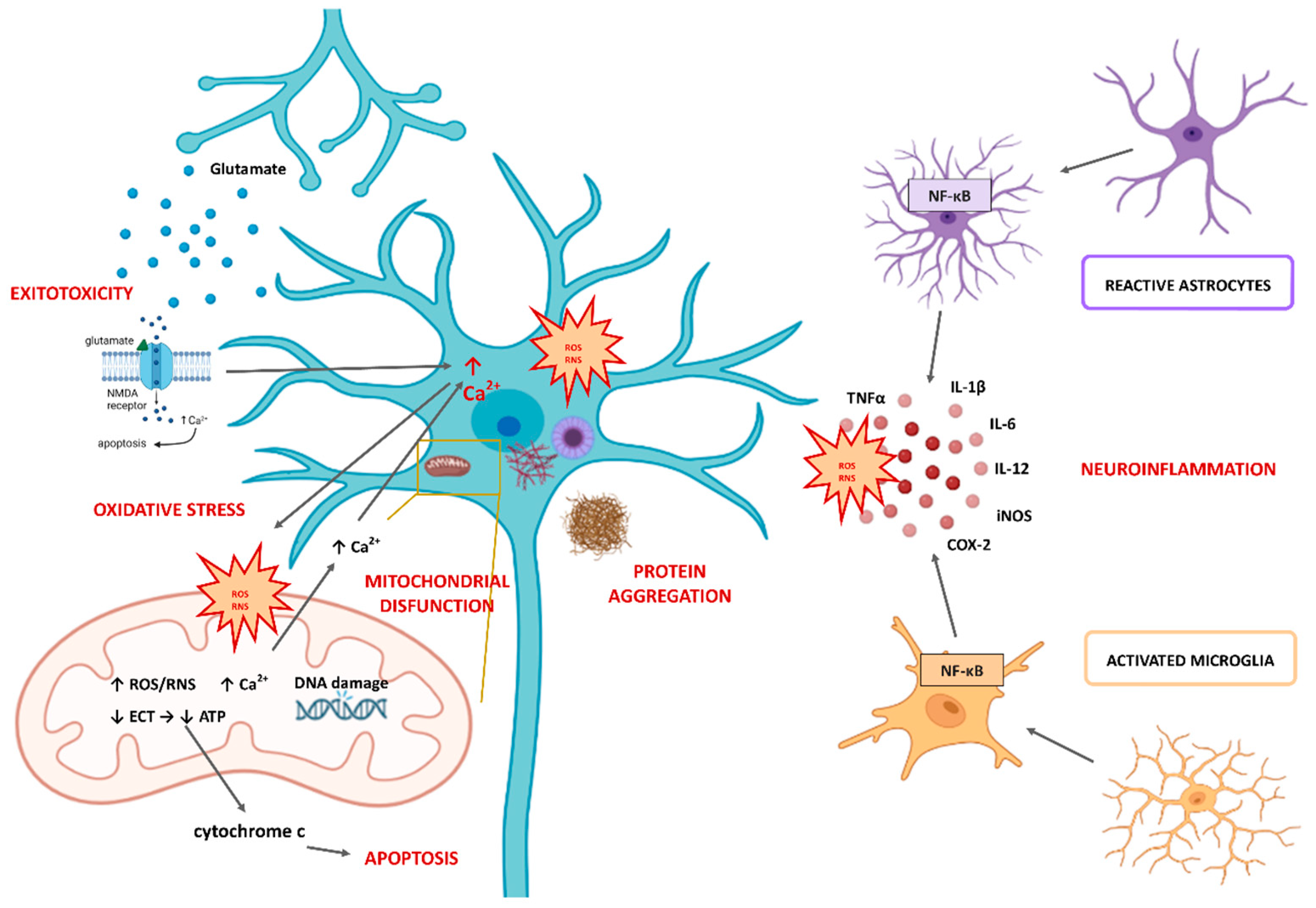
2. The Role of Oxidative Stress in the Pathophysiology of Neurodegenerative Diseases
3. Laurus nobilis (Lauraceae)
3.1. Therapeutic Potential of Laurus nobilis in Neurodegenerative Diseases
3.1.1. Laurus nobilis and Alzheimer’s Disease (Anti-Acetylcholinesterase Activity)
3.1.2. Laurus nobilis and Parkinson’s Disease
| Laurus nobilis Phytochemical Preparation or Compound | Action | Effect | Condition | References |
|---|---|---|---|---|
| Eucalyptol (1,8-cineol) | Regulation of NF-κB and Nrf2 | Anti-inflammatory | AD | [49] |
| Quercetin | Modulation of Nrf2 expression Suppression of NF-κB signal transducer Destabilization and enhance of the clearance of abnormal proteins | Anti-inflammatory Antioxidant | NP | [56] |
| Spirafolide | Inhibition of intracellular ROS generation Inhibition of apoptosis in DA neuroblastoma SH-SY5Y cells | Neuroprotective Antioxidant | AD | [54] |
| Reynosin | Protection of DA induced cell death in neuroblastoma SH-SY5Y cells and 6-OHDA-lesioned rats Up regulation of E6-AP expression Down regulation of ASYN expression | Neuroprotective | PD | [55] |
| Lindoldhamine | Antagonist ASIC1a AChE inhibition | Anti-inflammatory | Ischemic brain damage AD | [40,51,52,53] |
| Ethanolic extract | Oxidative damage protection | Neuroprotective | Neurotoxicity of heavy metals | [57] |
| Polyphenol enriched leaf extract | Oxidative damage protection | Neuroprotective Antioxidant | AD | [58] |
| Water extract | AChE inhibition | Antioxidant Neuroprotective | AD | [47] |
| Apolar extract | Cytotoxicity and apoptosis on human neuroblastoma cells and rat glioma cells | Cytotoxicity | Neuronal cancer | [40] |
| Essential oil | AChE inhibition Reduction in ADCY1 expression in SH-SY5Y cells | Antioxidant Neuroprotective | AD | [46,48,49] |
| Essential oil | Modulation of glutamatergic and GABAergic transmission | Anticonvulsant | Epilepsy | [59] |
3.2. Laurus nobilis and Other Neurological Conditions
4. Aronia melanocarpa (Rosaceae)
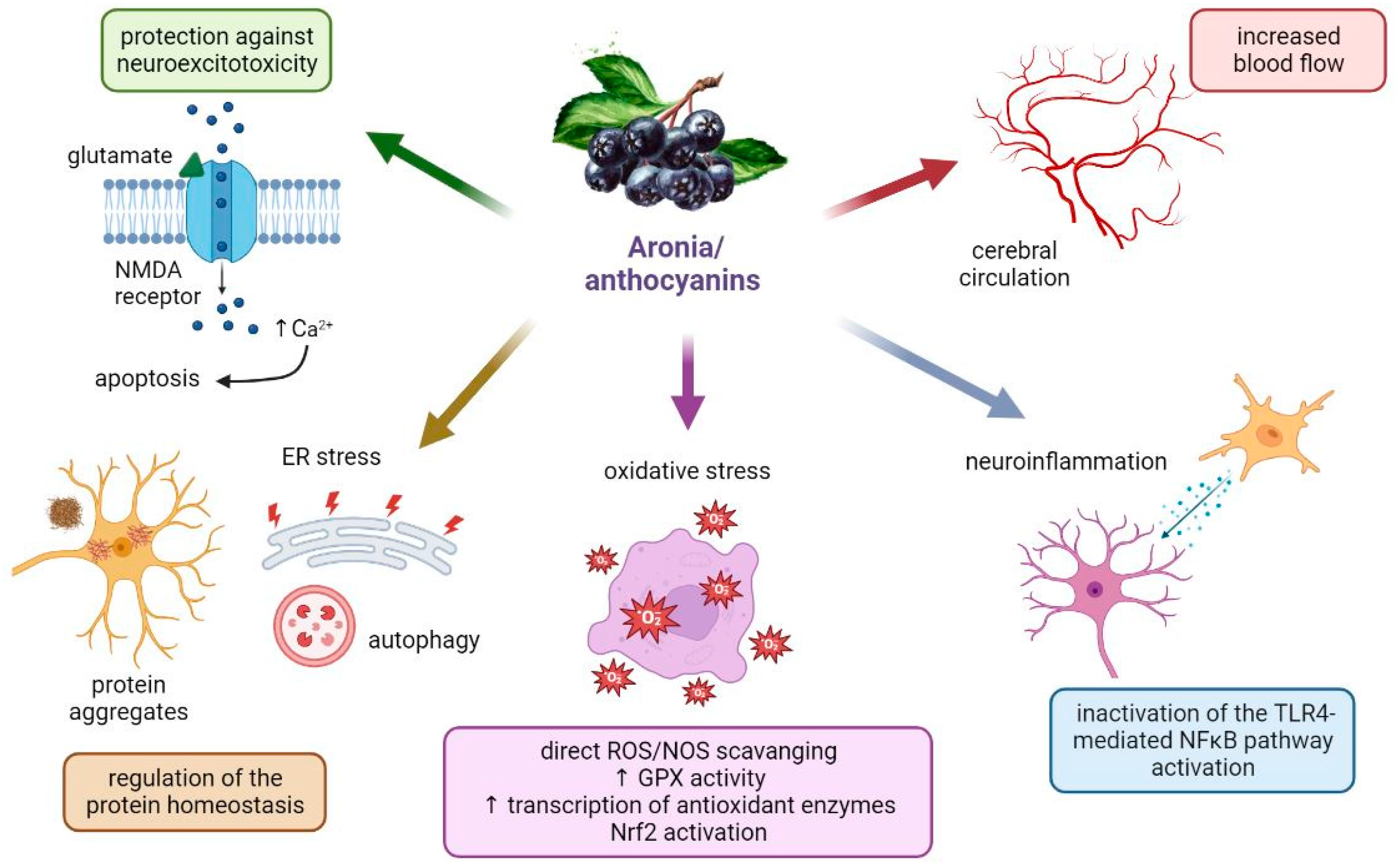
4.1. Aronia berries in Neurological Disorders: What Are the Potential Mechanisms for the Neuroprotective Actions?
4.2. Aronia melanocarpa and Its Extracts in Neurodegenerative Diseases
4.3. Aronia melanocarpa and Other Neurological Conditions
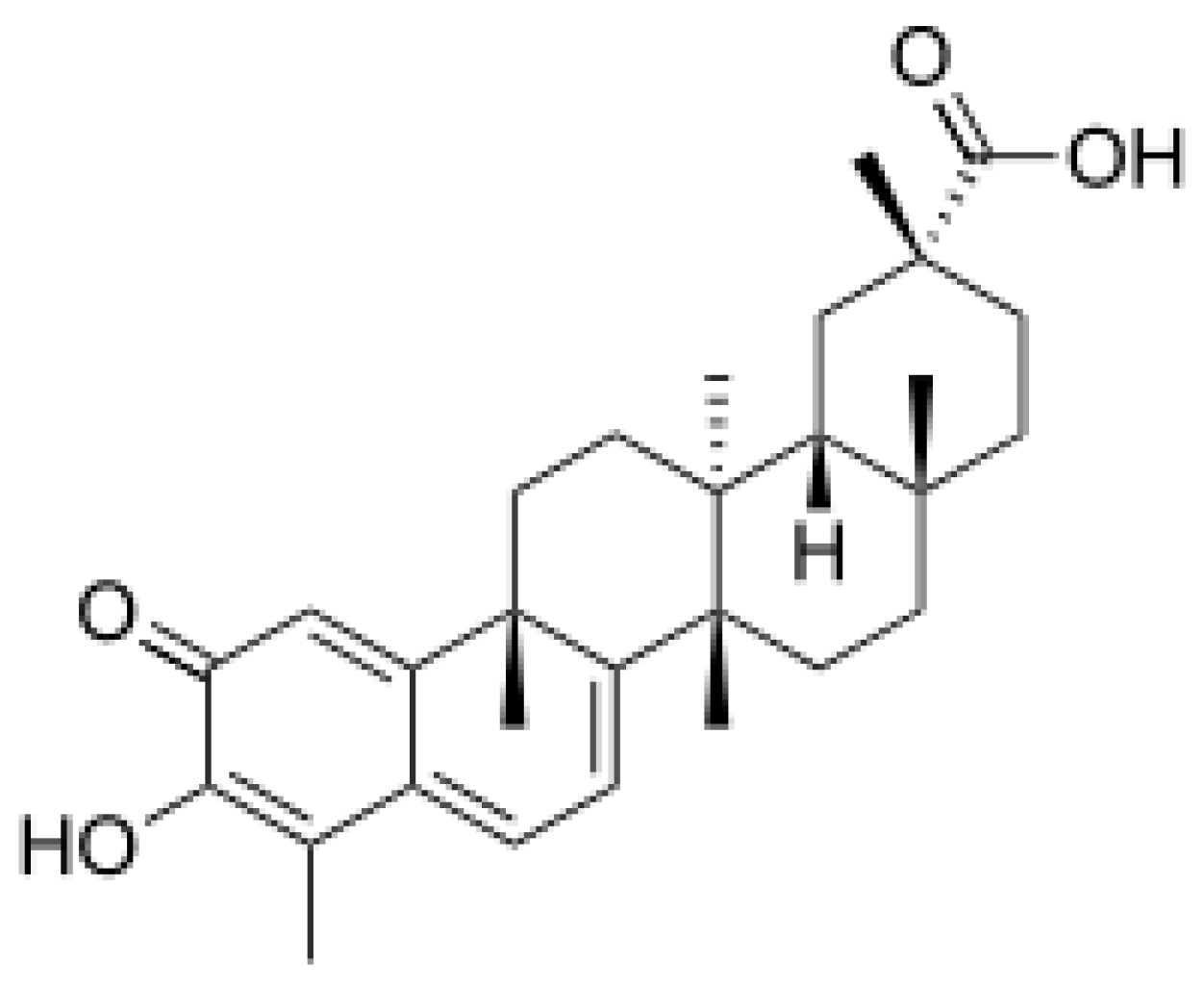
5. Celastrol
5.1. Therapeutic Potential of Celastrol in Neurodegenerative Diseases
5.2. Celastrol and Other Health Conditions
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Renaud, J.; Martinoli, M.-G. Considerations for the Use of Polyphenols as Therapies in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 1883. [Google Scholar] [CrossRef]
- Rekatsina, M.; Paladini, A.; Piroli, A.; Zis, P.; Pergolizzi, J.V.; Varrassi, G. Pathophysiology and Therapeutic Perspectives of Oxidative Stress and Neurodegenerative Diseases: A Narrative Review. Adv. Ther. 2020, 37, 113–139. [Google Scholar] [CrossRef]
- Boyd, R.J.; Avramopoulos, D.; Jantzie, L.L.; McCallion, A.S. Neuroinflammation represents a common theme amongst genetic and environmental risk factors for Alzheimer and Parkinson diseases. J. Neuroinflammation 2022, 19, 223. [Google Scholar] [CrossRef]
- Kim, G.H.; Kim, J.E.; Rhie, S.J.; Yoon, S. The Role of Oxidative Stress in Neurodegenerative Diseases. Exp. Neurobiol. 2015, 24, 325–340. [Google Scholar] [CrossRef] [PubMed]
- Si, Z.-Z.; Zou, C.-J.; Mei, X.; Li, X.-F.; Luo, H.; Shen, Y.; Hu, J.; Li, X.-X.; Wu, L.; Liu, Y. Targeting neuroinflammation in Alzheimer’s disease: From mechanisms to clinical applications. Neural Regen. Res. 2023, 18, 708. [Google Scholar] [CrossRef] [PubMed]
- Cenini, G.; Lloret, A.; Cascella, R. Oxidative Stress in Neurodegenerative Diseases: From a Mitochondrial Point of View. Oxidative Med. Cell. Longev. 2019, 2019, e2105607. [Google Scholar] [CrossRef]
- Niedzielska, E.; Smaga, I.; Gawlik, M.; Moniczewski, A.; Stankowicz, P.; Pera, J.; Filip, M. Oxidative Stress in Neurodegenerative Diseases. Mol. Neurobiol. 2016, 53, 4094–4125. [Google Scholar] [CrossRef]
- Chen, X.; Guo, C.; Kong, J. Oxidative stress in neurodegenerative diseases. Neural Regen. Res. 2012, 7, 376–385. [Google Scholar]
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R.T. Oxidative Stress and Neurodegenerative Diseases: A Review of Upstream and Downstream Antioxidant Therapeutic Options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Uddin, S.; Al Mamun, A.; Kabir, T.; Ahmad, J.; Jeandet, P.; Sarwar, S.; Ashraf, G.M.; Aleya, L. Neuroprotective role of polyphenols against oxidative stress-mediated neurodegeneration. Eur. J. Pharmacol. 2020, 886, 173412. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Panahi, Y.; Javadi, B.; Sahebkar, A. The Underlying Role of Oxidative Stress in Neurodegeneration: A Mechanistic Review. CNS Neurol. Disord.-Drug Targets 2018, 17, 207–215. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, T.; Ziegler, A.C.; Dimitrion, P.; Zuo, L. Oxidative Stress in Neurodegenerative Diseases: From Molecular Mechanisms to Clinical Applications. Oxid. Med. Cell. Longev. 2017, 2017, 2525967. [Google Scholar] [CrossRef] [PubMed]
- Merelli, A.; Repetto, M.; Lazarowski, A.; Auzmendi, J. Hypoxia, Oxidative Stress, and Inflammation: Three Faces of Neurodegenerative Diseases. J. Alzheimer’s Dis. 2021, 82, S109–S126. [Google Scholar] [CrossRef]
- Mirończuk-Chodakowska, I.; Witkowska, A.M.; Zujko, M.E. Endogenous non-enzymatic antioxidants in the human body. Adv. Med. Sci. 2018, 63, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Mekinić, I.G.; Skroza, D.; Ljubenkov, I.; Katalinić, V.; Šimat, V. Antioxidant and Antimicrobial Potential of Phenolic Metabolites from Traditionally Used Mediterranean Herbs and Spices. Foods 2019, 8, 579. [Google Scholar] [CrossRef]
- Pohl, F.; Lin, P.K.T. The Potential Use of Plant Natural Products and Plant Extracts with Antioxidant Properties for the Prevention/Treatment of Neurodegenerative Diseases: In Vitro, In Vivo and Clinical Trials. Molecules 2018, 23, 3283. [Google Scholar] [CrossRef] [PubMed]
- Babazadeh, A.; Vahed, F.M.; Liu, Q.; Siddiqui, S.A.; Kharazmi, M.S.; Jafari, S.M. Natural Bioactive Molecules as Neuromedicines for the Treatment/Prevention of Neurodegenerative Diseases. ACS Omega 2023, 8, 3667–3683. [Google Scholar] [CrossRef]
- Elfawy, H.A.; Das, B. Crosstalk between mitochondrial dysfunction, oxidative stress, and age related neurodegenerative disease: Etiologies and therapeutic strategies. Life Sci. 2019, 218, 165–184. [Google Scholar] [CrossRef]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules 2019, 24, 1583. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B. Natural Antioxidants for Neurodegenerative Diseases. Mol. Neurobiol. 2005, 31, 283–293. [Google Scholar] [CrossRef]
- Ikram, M.; Saeed, K.; Khan, A.; Muhammad, T.; Khan, M.S.; Jo, M.G.; Rehman, S.U.; Kim, M.O. Natural Dietary Supplementation of Curcumin Protects Mice Brains against Ethanol-Induced Oxidative Stress-Mediated Neurodegeneration and Memory Impairment via Nrf2/TLR4/RAGE Signaling. Nutrients 2019, 11, 1082. [Google Scholar] [CrossRef]
- Ahmad, A.; Ali, T.; Rehman, S.U.; Kim, M.O. Phytomedicine-Based Potent Antioxidant, Fisetin Protects CNS-Insult LPS-Induced Oxidative Stress-Mediated Neurodegeneration and Memory Impairment. J. Clin. Med. 2019, 8, 850. [Google Scholar] [CrossRef] [PubMed]
- Gilgun-Sherki, Y.; Melamed, E.; Offen, D. Oxidative stress induced-neurodegenerative diseases: The need for antioxidants that penetrate the blood brain barrier. Neuropharmacology 2001, 40, 959–975. [Google Scholar] [CrossRef]
- Winter, A.N.; Bickford, P.C. Anthocyanins and Their Metabolites as Therapeutic Agents for Neurodegenerative Disease. Antioxidants 2019, 8, 333. [Google Scholar] [CrossRef]
- Korovesis, D.; Rubio-Tomás, T.; Tavernarakis, N. Oxidative Stress in Age-Related Neurodegenerative Diseases: An Overview of Recent Tools and Findings. Antioxidants 2023, 12, 131. [Google Scholar] [CrossRef] [PubMed]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxidative Med. Cell. Longev. 2014, 2014, 360438. [Google Scholar] [CrossRef]
- Winczura, A.; Zdżalik, D.; Tudek, B. Damage of DNA and proteins by major lipid peroxidation products in genome stability. Free. Radic. Res. 2012, 46, 442–459. [Google Scholar] [CrossRef] [PubMed]
- Reed, T.T. Lipid peroxidation and neurodegenerative disease. Free. Radic. Biol. Med. 2011, 51, 1302–1319. [Google Scholar] [CrossRef]
- García-Beltrán, O.; Urrutia, P.J.; Núñez, M.T. On the Chemical and Biological Characteristics of Multifunctional Compounds for the Treatment of Parkinson’s Disease. Antioxidants 2023, 12, 214. [Google Scholar] [CrossRef]
- Castelli, V.; Benedetti, E.; Antonosante, A.; Catanesi, M.; Pitari, G.; Ippoliti, R.; Cimini, A.; D’Angelo, M. Neuronal Cells Rearrangement During Aging and Neurodegenerative Disease: Metabolism, Oxidative Stress and Organelles Dynamic. Front. Mol. Neurosci. 2019, 12, 132. [Google Scholar] [CrossRef]
- Abramov, A.Y.; Berezhnov, A.V.; Fedotova, E.I.; Zinchenko, V.P.; Dolgacheva, L.P. Interaction of misfolded proteins and mitochondria in neurodegenerative disorders. Biochem. Soc. Trans. 2017, 45, 1025–1033. [Google Scholar] [CrossRef]
- Hou, W.; Hao, Y.; Sun, L.; Zhao, Y.; Zheng, X.; Song, L. The dual roles of autophagy and the GPCRs-mediating autophagy signaling pathway after cerebral ischemic stroke. Mol. Brain 2022, 15, 14. [Google Scholar] [CrossRef] [PubMed]
- Iannuzzi, C.; Liccardo, M.; Sirangelo, I. Overview of the Role of Vanillin in Neurodegenerative Diseases and Neuropathophysiological Conditions. Int. J. Mol. Sci. 2023, 24, 1817. [Google Scholar] [CrossRef] [PubMed]
- Simpson, D.S.A.; Oliver, P.L. ROS Generation in Microglia: Understanding Oxidative Stress and Inflammation in Neurodegenerative Disease. Antioxidants 2020, 9, 743. [Google Scholar] [CrossRef] [PubMed]
- Namazi, F.; Bordbar, E.; Bakhshaei, F.; Nazifi, S. The effect of Urtica dioica extract on oxidative stress, heat shock proteins, and brain histopathology in multiple sclerosis model. Physiol. Rep. 2022, 10, e15404. [Google Scholar] [CrossRef]
- Leak, R.K. Heat shock proteins in neurodegenerative disorders and aging. J. Cell Commun. Signal. 2014, 8, 293–310. [Google Scholar] [CrossRef]
- Al-Mijalli, S.H.; Mrabti, H.N.; Ouassou, H.; Flouchi, R.; Abdallah, E.M.; Sheikh, R.A.; Alshahrani, M.M.; Awadh, A.A.A.; Harhar, H.; El Omari, N.; et al. Chemical Composition, Antioxidant, Anti-Diabetic, Anti-Acetylcholinesterase, Anti-Inflammatory, and Antimicrobial Properties of Arbutus unedo L. and Laurus nobilis L. Essential Oils. Life 2022, 12, 1876. [Google Scholar] [CrossRef]
- Grdinić, V.; Kremer, D. Botanički podaci. In Ljekovite biljke i ljekovite droge: Farmakoterapijski, botanički i farmaceutski podaci; Grdinić, V., Kremer, D., Eds.; Croatian Pharmaceutical Chamber: Zagreb, Croatia, 2009; Volume 4, pp. 349–350. [Google Scholar]
- Sayyah, M.; Valizadeh, J.; Kamalinejad, M. Anticonvulsant activity of the leaf essential oil of Laurus nobilis against pentylenetetrazole- and maximal electroshock-induced seizures. Phytomedicine 2002, 9, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, S.; Gallicchio, M.; Lorenz, P.; Potenza, N.; Galasso, S.; Marciano, S.; Fiorentino, A.; Stintzing, F.C.; Monaco, P. Apolar Laurus nobilis leaf extracts induce cytotoxicity and apoptosis towards three nervous system cell lines. Food Chem. Toxicol. 2013, 62, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Paparella, A.; Nawade, B.; Shaltiel-Harpaz, L.; Ibdah, M. A Review of the Botany, Volatile Composition, Biochemical and Molecular Aspects, and Traditional Uses of Laurus nobilis. Plants 2022, 11, 1209. [Google Scholar] [CrossRef]
- Mansour, O.; Darwish, M.; Ismail, G.; Eldair, K.S. Review Study on the Physiological Properties and Chemical Composition of the Laurus Nobilis. Pharm. Chem. J. 2018, 5, 225–231. [Google Scholar]
- Bhadra, S. Evidence-Based Validation of Herbal Medicine-2nd Edition. Available online: https://www.elsevier.com/books/evidence-based-validation-of-herbal-medicine/mukherjee/978-0-323-85542-6 (accessed on 9 February 2023).
- López, M.D.; Pascual-Villalobos, M. Mode of inhibition of acetylcholinesterase by monoterpenoids and implications for pest control. Ind. Crop. Prod. 2010, 31, 284–288. [Google Scholar] [CrossRef]
- Ingkaninan, K.; Temkitthawon, P.; Chuenchom, K.; Yuyaem, T.; Thongnoi, W. Screening for acetylcholinesterase inhibitory activity in plants used in Thai traditional rejuvenating and neurotonic remedies. J. Ethnopharmacol. 2003, 89, 261–264. [Google Scholar] [CrossRef]
- Yakoubi, R.; Megateli, S.; Sadok, T.H.; Bensouici, C.; Bağci, E. A synergistic interactions of Algerian essential oils of Laurus nobilis L., Lavandula stoechas L. and Mentha pulegium L. on anticholinesterase and antioxidant activities. Biocatal. Agric. Biotechnol. 2021, 31, 101891. [Google Scholar] [CrossRef]
- Duletić-Laušević, S.; Oalđe, M.; Alimpić-Aradski, A. In vitro evaluation of antioxidant, antineurodegenerative and antidiabetic activities of Ocimum basilicum L., Laurus nobilis L. leaves and Citrus reticulata Blanco peel extracts. Lek. sirovine 2019, 60–68. [Google Scholar] [CrossRef]
- Caputo, L.; Nazzaro, F.; Souza, L.F.; Aliberti, L.; De Martino, L.; Fratianni, F.; Coppola, R.; De Feo, V. Laurus nobilis: Composition of Essential Oil and Its Biological Activities. Molecules 2017, 22, 930. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.-M.; Peng, J.-Q.; Chen, Y.; Tao, L.; Zhang, Y.-Y.; Fu, L.-Y.; Long, Q.-D.; Shen, X.-C. 1,8-Cineole: A review of source, biological activities, and application. J. Asian Nat. Prod. Res. 2020, 23, 938–954. [Google Scholar] [CrossRef]
- Mendes, J.W.D.S.; Camilo, C.J.; de Carvalho, N.K.G.; Nonato, C.D.F.A.; de Lima, R.D.P.; Alves, D.R.; de Morais, S.M.; Silveira, E.R.; Coutinho, H.D.M.; Martins, N.; et al. In vitro antioxidant and acetylcholinesterase inhibitory properties of the alkaloid fraction of Cissampelos sympodialis Eichler. South Afr. J. Bot. 2021, 141, 99–104. [Google Scholar] [CrossRef]
- Osmakov, D.I.; Koshelev, S.G.; Palikov, V.A.; Palikova, Y.A.; Shaykhutdinova, E.R.; Dyachenko, I.A.; Andreev, Y.A.; Kozlov, S.A. Alkaloid Lindoldhamine Inhibits Acid-Sensing Ion Channel 1a and Reveals Anti-Inflammatory Properties. Toxins 2019, 11, 542. [Google Scholar] [CrossRef]
- Zhou, R.-P.; Liang, H.-Y.; Hu, W.-R.; Ding, J.; Li, S.-F.; Chen, Y.; Zhao, Y.-J.; Lu, C.; Chen, F.-H.; Hu, W. Modulators of ASIC1a and its potential as a therapeutic target for age-related diseases. Ageing Res. Rev. 2023, 83. [Google Scholar] [CrossRef]
- Bai, R.; Yao, C.; Zhong, Z.; Ge, J.; Bai, Z.; Ye, X.; Xie, T.; Xie, Y. Discovery of natural anti-inflammatory alkaloids: Potential leads for the drug discovery for the treatment of inflammation. Eur. J. Med. Chem. 2021, 213, 113165. [Google Scholar] [CrossRef] [PubMed]
- Ham, A.; Kim, B.; Koo, U.; Nam, K.-W.; Lee, S.-J.; Kim, K.H.; Shin, J.; Mar, W. Spirafolide from bay leaf (Laurus nobilis) prevents dopamine-induced apoptosis by decreasing reactive oxygen species production in human neuroblastoma SH-SY5Y cells. Arch. Pharmacal Res. 2010, 33, 1953–1958. [Google Scholar] [CrossRef] [PubMed]
- Ham, A.; Kim, D.-W.; Kim, K.H.; Lee, S.-J.; Oh, K.-B.; Shin, J.; Mar, W. Reynosin protects against neuronal toxicity in dopamine-induced SH-SY5Y cells and 6-hydroxydopamine-lesioned rats as models of Parkinson’s disease: Reciprocal up-regulation of E6-AP and down-regulation of α-synuclein. Brain Res. 2013, 1524, 54–61. [Google Scholar] [CrossRef]
- Suganthy, N.; Devi, K.P.; Nabavi, S.F.; Braidy, N.; Nabavi, S.M. Bioactive effects of quercetin in the central nervous system: Focusing on the mechanisms of actions. Biomed. Pharmacother. 2016, 84, 892–908. [Google Scholar] [CrossRef]
- Gazwi, H.S.S.; Yassien, E.E.; Hassan, H.M. Mitigation of lead neurotoxicity by the ethanolic extract of Laurus leaf in rats. Ecotoxicol. Environ. Saf. 2020, 192, 110297. [Google Scholar] [CrossRef]
- Pacifico, S.; Gallicchio, M.; Lorenz, P.; Duckstein, S.M.; Potenza, N.; Galasso, S.; Marciano, S.; Fiorentino, A.; Stintzing, F.C.; Monaco, P. Neuroprotective Potential of Laurus nobilis Antioxidant Polyphenol-Enriched Leaf Extracts. Chem. Res. Toxicol. 2014, 27, 611–626. [Google Scholar] [CrossRef]
- Faheem, M.; Ameer, S.; Khan, A.W.; Haseeb, M.; Raza, Q.; Ali Shah, F.; Khusro, A.; Aarti, C.; Umar Khayam Sahibzada, M.; El-Saber Batiha, G.; et al. A Comprehensive Review on Antiepileptic Properties of Medicinal Plants. Arab. J. Chem. 2022, 15, 103478. [Google Scholar] [CrossRef]
- Cho, E.-Y.; Lee, S.-J.; Nam, K.-W.; Shin, J.; Oh, K.; Kim, K.H.; Mar, W. Amelioration of Oxygen and Glucose Deprivation-Induced Neuronal Death by Chloroform Fraction of Bay Leaves (Laurus nobilis). Biosci. Biotechnol. Biochem. 2010, 74, 2029–2035. [Google Scholar] [CrossRef] [PubMed]
- Koo, U.; Nam, K.-W.; Ham, A.; Lyu, D.; Kim, B.; Lee, S.-J.; Kim, K.H.; Oh, K.-B.; Mar, W.; Shin, J. Neuroprotective Effects of 3α-Acetoxyeudesma-1,4(15),11(13)-trien-12,6α-olide Against Dopamine-Induced Apoptosis in the Human Neuroblastoma SH-SY5Y Cell Line. Neurochem. Res. 2011, 36, 1991–2001. [Google Scholar] [CrossRef]
- Dobroslavić, E.; Garofulić, I.E.; Zorić, Z.; Pedisić, S.; Dragović-Uzelac, V. Polyphenolic Characterization and Antioxidant Capacity of Laurus nobilis L. Leaf Extracts Obtained by Green and Conventional Extraction Techniques. Processes 2021, 9, 1840. [Google Scholar] [CrossRef]
- Testa, G.; Gamba, P.; Badilli, U.; Gargiulo, S.; Maina, M.; Guina, T.; Calfapietra, S.; Biasi, F.; Cavalli, R.; Poli, G.; et al. Loading into Nanoparticles Improves Quercetin’s Efficacy in Preventing Neuroinflammation Induced by Oxysterols. PLoS ONE 2014, 9, e96795. [Google Scholar] [CrossRef] [PubMed]
- Eftimov, M.; Dobreva, C.; Velkova, D.; Valcheva-Kuzmanova, S.; Eftimov, M. Effect of aronia melanocarpa fruit juice on reserpine-induced hypokinesia and oxydative stress in rats. Scr. Sci. Pharm. 2014, 2, 29–34. [Google Scholar] [CrossRef]
- Jurendić, T.; Ščetar, M. Aronia melanocarpa Products and By-Products for Health and Nutrition: A Review. Antioxidants 2021, 10, 1052. [Google Scholar] [CrossRef] [PubMed]
- Dufour, C.; Villa-Rodriguez, J.A.; Furger, C.; Lessard-Lord, J.; Gironde, C.; Rigal, M.; Badr, A.; Desjardins, Y.; Guyonnet, D. Cellular Antioxidant Effect of an Aronia Extract and Its Polyphenolic Fractions Enriched in Proanthocyanidins, Phenolic Acids, and Anthocyanins. Antioxidants 2022, 11, 1561. [Google Scholar] [CrossRef] [PubMed]
- Denev, P.N.; Kratchanov, C.G.; CizČíž, M.; Lojek, A.; Kratchanova, M.G. Bioavailability and Antioxidant Activity of Black Chokeberry (Aronia melanocarpa) Polyphenols: In vitro and in vivo Evidences and Possible Mechanisms of Action: A Review. Compr. Rev. Food Sci. Food Saf. 2012, 11, 471–489. [Google Scholar] [CrossRef]
- Dini, C.; Zaro, M.J.; Viña, S.Z. Bioactivity and Functionality of Anthocyanins: A Review. Curr. Bioact. Compd. 2019, 15, 507–523. [Google Scholar] [CrossRef]
- Ren, Y.; Frank, T.; Meyer, G.; Lei, J.; Grebenc, J.R.; Slaughter, R.; Gao, Y.G.; Kinghorn, A.D. Potential Benefits of Black Chokeberry (Aronia melanocarpa) Fruits and Their Constituents in Improving Human Health. Molecules 2022, 27, 7823. [Google Scholar] [CrossRef]
- King, E.S.; Bolling, B.W. Composition, polyphenol bioavailability, and health benefits of aronia berry: A review. J. Food Bioact. 2020, 11, 13–30. [Google Scholar] [CrossRef]
- Jeon, Y.-D.; Kang, S.-H.; Moon, K.-H.; Lee, J.-H.; Kim, D.-G.; Kim, W.; Kim, J.-S.; Ahn, B.-Y.; Jin, J.-S. The Effect of Aronia Berry on Type 1 Diabetes In Vivo and In Vitro. J. Med. Food 2018, 21, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Jurikova, T.; Mlcek, J.; Skrovankova, S.; Sumczynski, D.; Sochor, J.; Hlavacova, I.; Snopek, L.; Orsavová, J. Fruits of Black Chokeberry Aronia melanocarpa in the Prevention of Chronic Diseases. Molecules 2017, 22, 944. [Google Scholar] [CrossRef]
- Daskalova, E.; Delchev, S.; Topolov, M.; Dimitrova, S.; Uzunova, Y.; Valcheva-Kuzmanova, S.; Kratchanova, M.; Vladimirova-Kitova, L.; Denev, P. Aronia melanocarpa (Michx.) Elliot fruit juice reveals neuroprotective effect and improves cognitive and locomotor functions of aged rats. Food Chem. Toxicol. 2019, 132, 110674. [Google Scholar] [CrossRef]
- Valcheva-Kuzmanova, S.; Zhelyazkova-Savova, M. Anxiolytic-like effect of Aronia melanocarpa fruit juice in rats. Methods Find. Exp. Clin. Pharmacol. 2009, 31, 651–654. [Google Scholar] [CrossRef]
- Tomić, M.; Ignjatović, D.; Tovilović-Kovačević, G.; Krstić-Milošević, D.; Ranković, S.; Popović, T.; Glibetić, M. Reduction of anxiety-like and depression-like behaviors in rats after one month of drinking Aronia melanocarpa berry juice. Food Funct. 2016, 7, 3111–3120. [Google Scholar] [CrossRef] [PubMed]
- Jo, A.R.; Imm, J.-Y. Effects of aronia extract on lifespan and age-related oxidative stress in Drosophila melanogaster. Food Sci. Biotechnol. 2017, 26, 1399–1406. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Zhang, G.; Zhang, X.; Xu, D.; Gao, J.; Fan, J.; Zhou, Z. Anthocyanins from Black Chokeberry (Aroniamelanocarpa Elliot) Delayed Aging-Related Degenerative Changes of Brain. J. Agric. Food Chem. 2017, 65, 5973–5984. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Zhang, J.; Qin, M. Protective effect of cyanidin 3-O-glucoside on beta-amyloid peptide-induced cognitive impairment in rats. Neurosci. Lett. 2013, 534, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Min, J.; Yu, S.-W.; Baek, S.-H.; Nair, K.M.; Bae, O.-N.; Bhatt, A.; Kassab, M.; Nair, M.G.; Majid, A. Neuroprotective effect of cyanidin-3-O-glucoside anthocyanin in mice with focal cerebral ischemia. Neurosci. Lett. 2011, 500, 157–161. [Google Scholar] [CrossRef]
- Hornedo-Ortega, R.; Álvarez-Fernández, M.A.; Cerezo, A.B.; Richard, T.; Troncoso, A.M.; Garcia-Parrilla, M.C. Protocatechuic Acid: Inhibition of Fibril Formation, Destabilization of Preformed Fibrils of Amyloid-β and α-Synuclein, and Neuroprotection. J. Agric. Food Chem. 2016, 64, 7722–7732. [Google Scholar] [CrossRef]
- Ahles, S.; Stevens, Y.; Joris, P.; Vauzour, D.; Adam, J.; de Groot, E.; Plat, J. The Effect of Long-Term Aronia melanocarpa Extract Supplementation on Cognitive Performance, Mood, and Vascular Function: A Randomized Controlled Trial in Healthy, Middle-Aged Individuals. Nutrients 2020, 12, 2475. [Google Scholar] [CrossRef]
- Ahles, S.; Stevens, Y.R.; Joris, P.J.; Vauzour, D.; Adam, J.; de Groot, E.; Plat, J. Protective Influence of Natural Anthocyanins of Aronia Melanocarpa on Selected Parameters of Antioxidative Status in Experimental Intoxication with Sulphide2-Chloroethyl-3-Chloropropyl. Pol. J. Environ. Stud. 2004, 13, 339–341. [Google Scholar]
- Dietrich-Muszalska, A.; Kopka, J.; Kontek, B. Polyphenols from Berries of Aronia melanocarpa Reduce the Plasma Lipid Peroxidation Induced by Ziprasidone. Schizophr. Res. Treat. 2014, 2014, 602390. [Google Scholar] [CrossRef]
- Lee, H.Y.; Weon, J.B.; Ryu, G.; Yang, W.S.; Kim, N.Y.; Kim, M.K.; Ma, C.J. Neuroprotective effect of Aronia melanocarpa extract against glutamate-induced oxidative stress in HT22 cells. BMC Complement. Altern. Med. 2017, 17, 207. [Google Scholar] [CrossRef]
- Banach, M.; Wiloch, M.; Zawada, K.; Cyplik, W.; Kujawski, W. Evaluation of Antioxidant and Anti-Inflammatory Activity of Anthocyanin-Rich Water-Soluble Aronia Dry Extracts. Molecules 2020, 25, 4055. [Google Scholar] [CrossRef] [PubMed]
- Ohgami, K.; Ilieva, I.; Shiratori, K.; Koyama, Y.; Jin, X.-H.; Yoshida, K.; Kase, S.; Kitaichi, N.; Suzuki, Y.; Tanaka, T.; et al. Anti-inflammatory Effects of Aronia Extract on Rat Endotoxin-Induced Uveitis. Investig. Opthalmology Vis. Sci. 2005, 46, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Zapolska-Downar, D.; Bryk, D.; Małecki, M.; Hajdukiewicz, K.; Sitkiewicz, D. Aronia melanocarpa fruit extract exhibits anti-inflammatory activity in human aortic endothelial cells. Eur. J. Nutr. 2012, 51, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Jang, B.-K.; Lee, J.-W.; Choi, H.; Yim, S.-V. Aronia melanocarpa Fruit Bioactive Fraction Attenuates LPS-Induced Inflammatory Response in Human Bronchial Epithelial Cells. Antioxidants 2020, 9, 816. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Auger, C.; Kurita, I.; Anselm, E.; Rivoarilala, L.O.; Lee, H.J.; Lee, K.W.; Schini-Kerth, V.B. Aronia melanocarpa juice, a rich source of polyphenols, induces endothelium-dependent relaxations in porcine coronary arteries via the redox-sensitive activation of endothelial nitric oxide synthase. Nitric Oxide 2013, 35, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Varela, C.E.; Fromentin, E.; Roller, M.; Villarreal, F.; Ramirez-Sanchez, I. Effects of a Natural Extract of Aronia melanocarpa Berry on Endothelial Cell Nitric Oxide Production. J. Food Biochem. 2016, 40, 404–410. [Google Scholar] [CrossRef]
- Ullah, I.; Park, H.Y.; Kim, M.O. Anthocyanins Protect against Kainic Acid-induced Excitotoxicity and Apoptosis via ROS-activated AMPK Pathway in Hippocampal Neurons. CNS Neurosci. Ther. 2014, 20, 327–338. [Google Scholar] [CrossRef]
- Tarozzi, A.; Morroni, F.; Merlicco, A.; Bolondi, C.; Teti, G.; Falconi, M.; Cantelli-Forti, G.; Hrelia, P. Neuroprotective effects of cyanidin 3-O-glucopyranoside on amyloid beta (25–35) oligomer-induced toxicity. Neurosci. Lett. 2010, 473, 72–76. [Google Scholar] [CrossRef]
- Winter, A.N.; Brenner, M.C.; Punessen, N.; Snodgrass, M.; Byars, C.; Arora, Y.; Linseman, D.A. Comparison of the Neuroprotective and Anti-Inflammatory Effects of the Anthocyanin Metabolites, Protocatechuic Acid and 4-Hydroxybenzoic Acid. Oxidative Med. Cell. Longev. 2017, 2017, 6297080. [Google Scholar] [CrossRef]
- An, L.J.; Guan, S.; Shi, G.F.; Bao, Y.M.; Duan, Y.L.; Jiang, B. Protocatechuic acid from Alpinia oxyphylla against MPP+-induced neurotoxicity in PC12 cells. Food Chem. Toxicol. 2006, 44, 436–443. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, G.; Szeto, S.S.W.; Chong, C.M.; Quan, Q.; Huang, C.; Cui, W.; Guo, B.; Wang, Y.; Han, Y.; et al. Examining the neuroprotective effects of protocatechuic acid and chrysin on in vitro and in vivo models of Parkinson disease. Free. Radic. Biol. Med. 2015, 84, 331–343. [Google Scholar] [CrossRef]
- Gallardo-Fernández, M.; Hornedo-Ortega, R.; Cerezo, A.B.; Troncoso, A.M.; García-Parrilla, M.C. Melatonin, protocatechuic acid and hydroxytyrosol effects on vitagenes system against alpha-synuclein toxicity. Food Chem. Toxicol. 2019, 134, 110817. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-N.; An, C.-N.; Zhang, H.-N.; Pu, X.-P. Protocatechuic acid inhibits neurotoxicity induced by MPTP in vivo. Neurosci. Lett. 2010, 474, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Yangqian, K.; Bais, S. Neuroprotective Effect of Protocatechuic Acid Through MAO-B Inhibition in Aluminium Chloride Induced Dementia of Alzheimer’s Type in Rats. Int. J. Pharmacol. 2018, 14, 879–888. [Google Scholar] [CrossRef]
- Maya, S.; Prakash, T.; Goli, D. Evaluation of neuroprotective effects of wedelolactone and gallic acid on aluminium-induced neurodegeneration: Relevance to sporadic amyotrophic lateral sclerosis. Eur. J. Pharmacol. 2018, 835, 41–51. [Google Scholar] [CrossRef]
- Chen, B.-H.; Inbaraj, B.S. Nanoemulsion and Nanoliposome Based Strategies for Improving Anthocyanin Stability and Bioavailability. Nutrients 2019, 11, 1052. [Google Scholar] [CrossRef]
- Kay, C.D.; Mazza, G.; Holub, B.J.; Wang, J. Anthocyanin metabolites in human urine and serum. Br. J. Nutr. 2004, 91, 933–942. [Google Scholar] [CrossRef]
- Lee, H.Y.; Weon, J.B.; Jung, Y.S.; Kim, N.Y.; Kim, M.K.; Ma, C.J. Cognitive-Enhancing Effect of Aronia melanocarpa Extract against Memory Impairment Induced by Scopolamine in Mice. Evid. -Based Complement. Altern. Med. 2016, 2016, 1–7. [Google Scholar] [CrossRef]
- Wen, H.; Tian, H.; Liu, C.; Zhang, X.; Peng, Y.; Yang, X.; Chen, F.; Li, J. Metformin and cyanidin 3-O-galactoside from Aronia melanocarpa synergistically alleviate cognitive impairment in SAMP8 mice. Food Funct. 2021, 12, 10994–11008. [Google Scholar] [CrossRef]
- Fan, Z.; Wen, H.; Zhang, X.; Li, J.; Zang, J. Cyanidin 3-O-β-Galactoside Alleviated Cognitive Impairment in Mice by Regulating Brain Energy Metabolism During Aging. J. Agric. Food Chem. 2022, 70, 1111–1121. [Google Scholar] [CrossRef]
- Meng, L.; Xin, G.; Li, B.; Li, D.; Sun, X.; Yan, T.; Li, L.; Shi, L.; Cao, S.; Meng, X. Anthocyanins Extracted from Aronia melanocarpa Protect SH-SY5Y Cells against Amyloid-beta (1–42)-Induced Apoptosis by Regulating Ca2+ Homeostasis and Inhibiting Mitochondrial Dysfunction. J. Agric. Food Chem. 2018, 66, 12967–12977. [Google Scholar] [CrossRef] [PubMed]
- Márta, K.; Afrodité, S. Investigation of the Anti-Amyloidogenic Effect of Black Chokeberry Juice. Curr. Top. Pept. Protein Res. 2020, 21, 31–36. [Google Scholar]
- Kim, J.; Lee, K.P.; Beak, S.; Kang, H.R.; Kim, Y.K.; Lim, K. Effect of black chokeberry on skeletal muscle damage and neuronal cell death. J. Exerc. Nutr. Biochem. 2019, 23, 26–31. [Google Scholar] [CrossRef]
- Case, A.J.; Agraz, D.; Ahmad, I.M.; Zimmerman, M.C. Low-DoseAronia melanocarpaConcentrate Attenuates Paraquat-Induced Neurotoxicity. Oxidative Med. Cell. Longev. 2015, 2016, 5296271. [Google Scholar] [CrossRef]
- Gao, X.; Cassidy, A.; Schwarzschild, M.A.; Rimm, E.B.; Ascherio, A. Habitual intake of dietary flavonoids and risk of Parkinson disease. Neurology 2012, 78, 1138–1145. [Google Scholar] [CrossRef]
- Fan, D.; Alamri, Y.; Liu, K.; MacAskill, M.; Harris, P.; Brimble, M.; Dalrymple-Alford, J.; Prickett, T.; Menzies, O.; Laurenson, A.; et al. Supplementation of Blackcurrant Anthocyanins Increased Cyclic Glycine-Proline in the Cerebrospinal Fluid of Parkinson Patients: Potential Treatment to Improve Insulin-Like Growth Factor-1 Function. Nutrients 2018, 10, 714. [Google Scholar] [CrossRef]
- Zhang, X.; Molsberry, S.A.; Yeh, T.-S.; Cassidy, A.; Schwarzschild, M.A.; Ascherio, A.; Gao, X. Intake of Flavonoids and Flavonoid-Rich Foods and Mortality Risk Among Individuals With Parkinson Disease. Neurology 2022, 98, e1064–e10762022. [Google Scholar] [CrossRef]
- Strathearn, K.E.; Yousef, G.G.; Grace, M.H.; Roy, S.L.; Tambe, M.A.; Ferruzzi, M.G.; Wu, Q.-L.; Simon, J.E.; Lila, M.A.; Rochet, J.-C. Neuroprotective effects of anthocyanin- and proanthocyanidin-rich extracts in cellular models of Parkinson׳s disease. Brain Res. 2014, 1555, 60–77. [Google Scholar] [CrossRef] [PubMed]
- Allison, A.C.; Cacabelos, R.; Lombardi, V.R.M.; Álvarez, X.A.; Vigo, C. Central Nervous System Effects of Celastrol, a Potent Antioxidant and Antiinflammatory Agent. CNS Drug Rev. 2000, 6, 45–62. [Google Scholar] [CrossRef]
- Venkatesha, S.H.; Moudgil, K.D. Celastrol and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016, 928, 267–289. [Google Scholar] [CrossRef]
- Schiavone, S.; Morgese, M.G.; Tucci, P.; Trabace, L. The Therapeutic Potential of Celastrol in Central Nervous System Disorders: Highlights from In Vitro and In Vivo Approaches. Molecules 2021, 26, 4700. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.D.; Liu, Y.-G.; Kim, T.; Bobbala, S.; Yi, S.; Zhang, X.; Choi, J.; Scott, E.A. Celastrol-loaded PEG-b-PPS nanocarriers as an anti-inflammatory treatment for atherosclerosis. Biomater. Sci. 2019, 7, 657–668. [Google Scholar] [CrossRef]
- Der Sarkissian, S.; Cailhier, J.-F.; Borie, M.; Stevens, L.-M.; Gaboury, L.; Mansour, S.; Hamet, P.; Noiseux, N. Celastrol protects ischaemic myocardium through a heat shock response with up-regulation of haeme oxygenase-Br. J. Pharmacol. 2014, 171, 5265–5279. [Google Scholar] [CrossRef]
- Boridy, S.; Soliman, G.M.; Maysinger, D. Modulation of inflammatory signaling and cytokine release from microglia by celastrol incorporated into dendrimer nanocarriers. Nanomedicine 2012, 7, 1149–1165. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, Q.; Luo, P.; Gu, L.; Shen, S.; Tang, H.; Zhang, Y.; Lyu, M.; Shi, Q.; Yang, C.; et al. Neuroprotective Effects of Celastrol in Neurodegenerative Diseases-Unscramble Its Major Mechanisms of Action and Targets. Aging Dis. 2022, 13, 815–836. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Fu, R.-J.; Zhang, S.; Yue, S.-J.; Chen, Y.-Y.; Xu, D.-Q.; Tang, Y.-P. Potential medicinal value of celastrol and its synthesized analogues for central nervous system diseases. Biomed. Pharmacother. 2021, 139, 111551. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, H.; Lobo, N.; Guo, X.; Gentleman, S.M.; Ma, D. Celastrol Enhances Cell Viability and Inhibits Amyloid-β Production Induced by Lipopolysaccharide In Vitro. J. Alzheimer’s Dis. 2014, 41, 835–844. [Google Scholar] [CrossRef]
- Allison, A.C.; Cacabelos, R.; Lombardi, V.R.M.; Álvarez, X.A.; Vigo, C. Celastrol, a potent antioxidant and anti-inflammatory drug, as a possible treatment for Alzheimer’s disease. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2001, 25, 1341–1357. [Google Scholar] [CrossRef] [PubMed]
- Paris, D.; Ganey, N.J.; Laporte, V.; Patel, N.S.; Beaulieu-Abdelahad, D.; Bachmeier, C.; March, A.; Ait-Ghezala, G.; Mullan, M.J. Reduction of β-amyloid pathology by celastrol in a transgenic mouse model of Alzheimer’s disease. J. Neuroinflammation 2010, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Su, C.; Iyaswamy, A.; Krishnamoorthi, S.K.; Zhu, Z.; Yang, S.; Tong, B.C.; Liu, J.; Sreenivasmurthy, S.G.; Guan, X.; et al. Celastrol enhances transcription factor EB (TFEB)-mediated autophagy and mitigates Tau pathology: Implications for Alzheimer’s disease therapy. Acta Pharm. Sin. B 2022, 12, 1707–1722. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Jiang, X.; Feng, J. The therapeutic potential of triptolide and celastrol in neurological diseases. Front. Pharmacol. 2022, 13, 1024955. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Fang, M.; Wang, J.; Yu, H.; Hu, Z.; Yew, D.T.; Chen, W. Triptolide Down-regulates COX-2 Expression and PGE2 Release by Suppressing the Activity of NF-κB and MAP kinases in Lipopolysaccharide-treated PC12 Cells. Phytotherapy Res. 2011, 26, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.-S.; Kim, H.; Lee, H.J.; Sapkota, K.; Park, S.E.; Kim, S.; Kim, S.-J. Celastrol from ‘Thunder God Vine’ Protects SH-SY5Y Cells Through the Preservation of Mitochondrial Function and Inhibition of p38 MAPK in a Rotenone Model of Parkinson’s Disease. Neurochem. Res. 2014, 39, 84–96. [Google Scholar] [CrossRef]
- Li, Y.-H.; Liu, S.-B.; Zhang, H.-Y.; Zhou, F.-H.; Liu, Y.-X.; Lu, Q.; Yang, L. Antioxidant effects of celastrol against hydrogen peroxide-induced oxidative stress in the cell model of amyotrophic lateral sclerosis. Sheng Li Xue Bao. 2017, 69, 751–758. [Google Scholar]
- Zhang, B.; Zhong, Q.; Chen, X.; Wu, X.; Sha, R.; Song, G.; Zhang, C.; Chen, X. Neuroprotective Effects of Celastrol on Transient Global Cerebral Ischemia Rats via Regulating HMGB1/NF-κB Signaling Pathway. Front. Neurosci. 2020, 14, 847. [Google Scholar] [CrossRef]
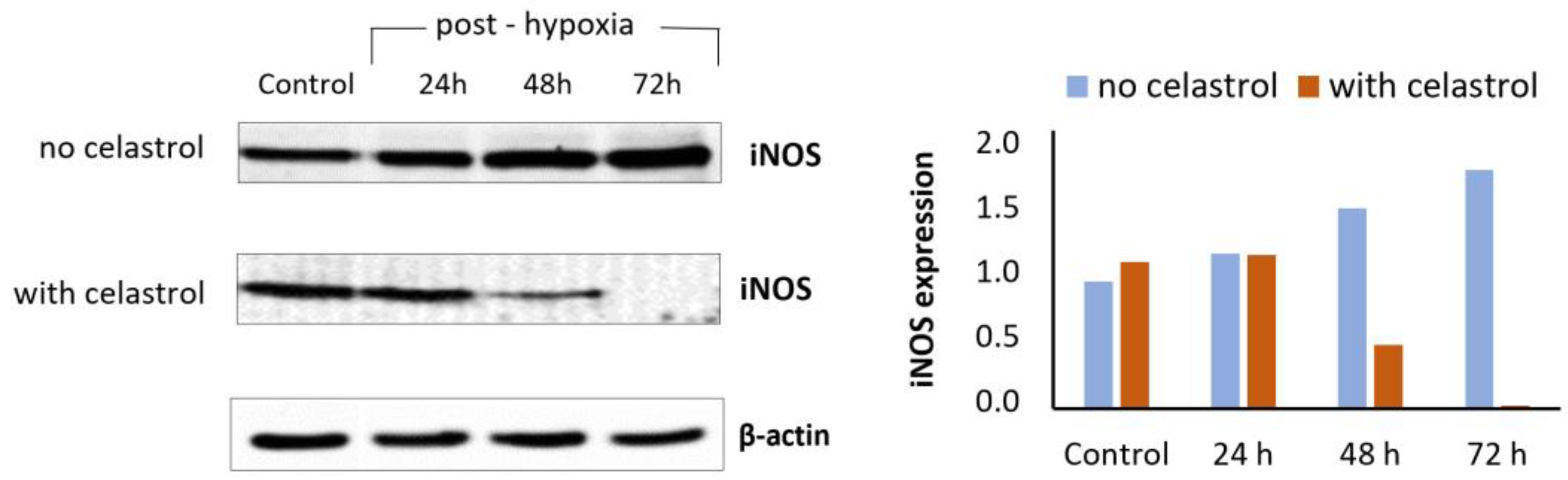
- Rački, V.; Marcelić, M.; Mršić-Pelčić, J.; Kučić, N. Effects of Celastrol on Bv-2 Microglial Cells Exposed to in Vitro Hypoxia. Clin. Ther. 2016, 38, e21. [Google Scholar] [CrossRef]
- Jiang, M.; Liu, X.; Zhang, D.; Wang, Y.; Hu, X.; Xu, F.; Jin, M.; Cao, F.; Xu, L. Celastrol treatment protects against acute ischemic stroke-induced brain injury by promoting an IL-33/ST2 axis-mediated microglia/macrophage M2 polarization. J. Neuroinflammation 2018, 15, 78. [Google Scholar] [CrossRef]
- Hong, Z.; Cao, J.; Liu, D.; Liu, M.; Chen, M.; Zeng, F.; Qin, Z.; Wang, J.; Tao, T. Celastrol targeting Nedd4 reduces Nrf2-mediated oxidative stress in astrocytes after ischemic stroke. J. Pharm. Anal. 2023. [Google Scholar] [CrossRef]
- Lan, G.; Zhang, J.; Ye, W.; Yang, F.; Li, A.; He, W.; Zhang, W.-D. Celastrol as a tool for the study of the biological events of metabolic diseases. Sci. China Chem. 2019, 62, 409–416. [Google Scholar] [CrossRef]
- Xu, S.; Feng, Y.; He, W.; Xu, W.; Xu, W.; Yang, H.; Li, X. Celastrol in metabolic diseases: Progress and application prospects. Pharmacol. Res. 2021, 167, 105572. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Shi, C.; Yang, X.; Yang, M.; Sun, H.; Wang, C. Celastrol suppresses obesity process via increasing antioxidant capacity and improving lipid metabolism. Eur. J. Pharmacol. 2014, 744, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, J.; Duan, X.; Zhao, G.; Zhang, M. Celastrol: A Promising Agent Fighting against Cardiovascular Diseases. Antioxidants 2022, 11, 1597. [Google Scholar] [CrossRef]

| Experimental Model | Treatment | Outcomes | Reference |
|---|---|---|---|
| In vivo studies | |||
| amyloid-β-induced memory damage in rats | isolated and purified anthocyanins | higher free radical scavenging abilities than the crude anthocyanins extract, improved spatial memory in a Morris water maze test, protection of the cells in the hippocampus against Aβ toxicity | [103] |
| D-galactose model of accelerated aging in mice | anthocyanins isolated as pure compounds (cyanidin 3-arabinoside, cyanidin 3-galactoside, cyanidin 3-xyloside, and cyanidin 3-glucoside), low (15 mg/kg) and high (30 mg/kg) dose | anthocyanins blocked age-associate cognitive decline and response capacity, improved redox system balance, decreased inflammatory cytokines’ levels, decreased DNA damage | [77] |
| amyloid-β-intracerebral injection induced cognitive impairment in rats | cyanidin 3-O-glucoside | protective against the Aβ-induced impairment, but no effect on normal learning and memory; attenuated Aβ-induced tau hyperphosphorylation and GSK-3β hyperactivation | [78] |
| experimental dementia model in rats by p.o.aluminium chloride application | protocatechuic acid | treatment significantly modulated memory deficits, via anti-cholinesterase, anti-oxidative and anti-inflammatory activities | [98] |
| senescence accelerated (SAMP8) aging mice | intragastric cyanidin 3-O-β-galactoside from black chokeberry (25 and 50 mg/kg/day) for 8 weeks | alleviated decline in brain glucose uptake, neuronal damage in the hippocampus and cortex, reduced β-amyloid load in the brain and significantly increased the crossing-platform number in the Morris water maze test | [104] |
| In vitro studies | |||
| PC12 cells exposed to amyloid-β and α-synuclein | protocatechuic acid | inhibition of fibril formation, destabilization of preformed fibrils of amyloid-β and α-synuclein | [80] |
| amyloid-β-induced apoptosis of SH-SY5Y cells | A. melanocarpa anthocyanins pretreatment | inhibited apoptosis, regulated Ca2+ homeostasis and apoptosis-related genes and inhibited mitochondrial dysfunction | [105] |
| α-chymotrypsin amyloid like fibrils formation model | chokeberry juice | inhibited aggregation of α-chymotrypsin | [106] |
| primary neuronal cells exposed to hydrogen peroxide or amyloid-β | ethanolic extract from black chokeberry fruit | reduced amyloid-β-induced neuronal cell death by modulating the inflammation-related signaling pathways | [107] |
| MPTP model of PD in mice | protocatechuic acid | improved rotarod test, ameliorated SN pathology and DA levels | [97] |
| PC12 cells exposed to α-synuclein | protocatechuic acid, hydroxytyrosol | decreased toxicity of α-synuclein, increased the expression vitagenes system proteins | [96] |
| neuroblastoma cells exposed to paraquat | aronia berry concentrate | protection of neurons from cell death, attenuated increase in superoxide, hydrogen peroxide, and oxidized glutathione levels | [108] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pilipović, K.; Jurišić Grubešić, R.; Dolenec, P.; Kučić, N.; Juretić, L.; Mršić-Pelčić, J. Plant-Based Antioxidants for Prevention and Treatment of Neurodegenerative Diseases: Phytotherapeutic Potential of Laurus nobilis, Aronia melanocarpa, and Celastrol. Antioxidants 2023, 12, 746. https://doi.org/10.3390/antiox12030746
Pilipović K, Jurišić Grubešić R, Dolenec P, Kučić N, Juretić L, Mršić-Pelčić J. Plant-Based Antioxidants for Prevention and Treatment of Neurodegenerative Diseases: Phytotherapeutic Potential of Laurus nobilis, Aronia melanocarpa, and Celastrol. Antioxidants. 2023; 12(3):746. https://doi.org/10.3390/antiox12030746
Chicago/Turabian StylePilipović, Kristina, Renata Jurišić Grubešić, Petra Dolenec, Natalia Kučić, Lea Juretić, and Jasenka Mršić-Pelčić. 2023. "Plant-Based Antioxidants for Prevention and Treatment of Neurodegenerative Diseases: Phytotherapeutic Potential of Laurus nobilis, Aronia melanocarpa, and Celastrol" Antioxidants 12, no. 3: 746. https://doi.org/10.3390/antiox12030746
APA StylePilipović, K., Jurišić Grubešić, R., Dolenec, P., Kučić, N., Juretić, L., & Mršić-Pelčić, J. (2023). Plant-Based Antioxidants for Prevention and Treatment of Neurodegenerative Diseases: Phytotherapeutic Potential of Laurus nobilis, Aronia melanocarpa, and Celastrol. Antioxidants, 12(3), 746. https://doi.org/10.3390/antiox12030746








