Coenzyme Q10 and Dementia: A Systematic Review
Abstract
1. Introduction
2. Methods
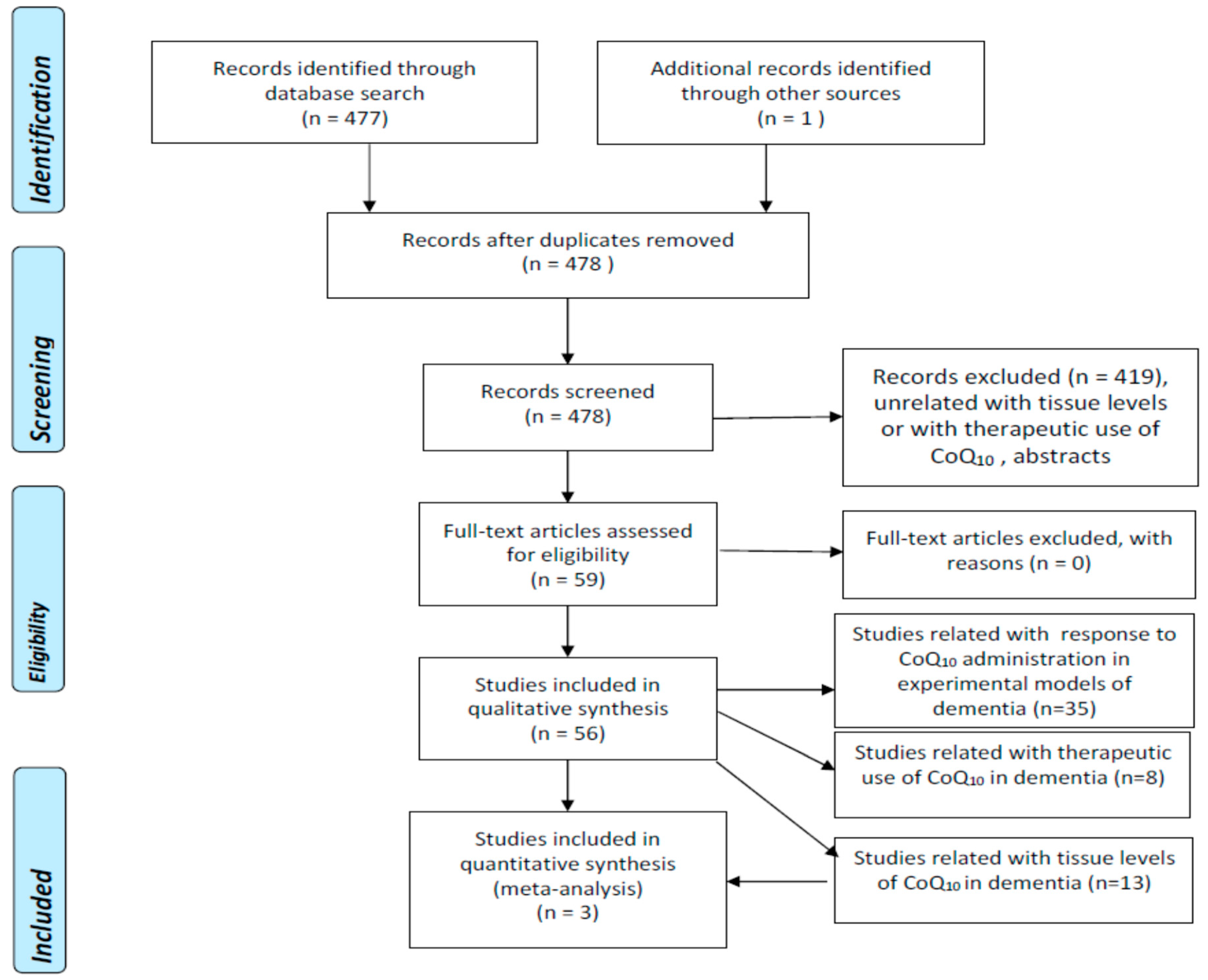
2.1. Search Strategy and Criteria for Eligibility of Studies
2.2. Selection of Studies and Methodology for the Meta-Analyses
3. Results
3.1. Studies Assessing Tissular CoQ10 Concentrations
3.1.1. Alzheimer’s Disease
3.1.2. Other Causes of Dementia
3.2. Studies Assessing Therapeutic Response to CoQ10 Administration in Experimental Models of AD and Other Dementias
3.3. Studies Assessing Therapeutic Response to CoQ10 Administration in Patients with Dementia
3.3.1. Alzheimer’s Disease
3.3.2. Vascular Dementia (VD)
3.3.3. Mild Cognitive Impairment and Normal Aging
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crane, F.L. Biochemical functions of coenzyme Q10. J. Am. Coll. Nutr. 2001, 20, 591–598. [Google Scholar] [CrossRef]
- Mantle, D.; Heaton, R.A.; Hargreaves, I.P. Coenzyme Q10, Ageing and the Nervous System: An Overview. Antioxidants 2021, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Jiménez, F.J.; Alonso-Navarro, H.; Ayuso-Peralta, L.; Jabbour-Wadih, T. Estrés oxidativo y enfermedad de Alzheimer. Rev. Neurol. 2006, 42, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Kumaran, K.R.; Yunusa, S.; Perimal, E.; Wahab, H.; Müller, C.P.; Hassan, Z. Insights into the Pathophysiology of Alzheimer’s Disease and Potential Therapeutic Targets: A Current Perspective. J. Alzheimers Dis. 2023, 91, 507–530. [Google Scholar] [CrossRef] [PubMed]
- Jurcau, A. Insights into the Pathogenesis of Neurodegenerative Diseases, Focus on Mitochondrial Dysfunction and Oxidative Stress. Int. J. Mol. Sci. 2021, 22, 11847. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses, the PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality If Nonrandomized Studies in Meta Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 3 January 2023).
- de Bustos, F.; Molina, J.A.; Jiménez-Jiménez, F.J.; García-Redondo, A.; Gómez-Escalonilla, C.; Porta-Etessam, J.; Berbel, A.; Zurdo, M.; Barcenilla, B.; Parrilla, G.; et al. Serum levels of coenzyme Q10 in patients with Alzheimer’s disease. J. Neural. Transm. 2000, 107, 233–239. [Google Scholar] [CrossRef]
- Battino, M.; Bompadre, S.; Leone, L.; Devecchi, E.; Degiuli, A.; D’Agostino, F.; Cambiè, G.; D’Agostino, M.; Faggi, L.; Colturani, G.; et al. Coenzyme Q, Vitamin E and Apo-E alleles in Alzheimer Disease. Biofactors 2003, 18, 277–281. [Google Scholar] [CrossRef]
- Giavarotti, L.; Simon, K.A.; Azzalis, L.A.; Fonseca, F.L.; Lima, A.F.; Freitas, M.C.; Brunialti, M.K.; Salomão, R.; Moscardi, A.A.; Montaño, M.B.; et al. Mild systemic oxidative stress in the subclinical stage of Alzheimer’s disease. Oxid. Med. Cell. Longev. 2013, 2013, 609019. [Google Scholar] [CrossRef]
- Isobe, C.; Abe, T.; Terayama, Y. Increase in the oxidized/total coenzyme Q-10 ratio in the cerebrospinal fluid of Alzheimer’s disease patients. Dement. Geriatr. Cogn. Disord. 2009, 28, 449–454. [Google Scholar] [CrossRef]
- Isobe, C.; Abe, T.; Terayama, Y. Levels of reduced and oxidized coenzyme Q-10 and 8-hydroxy-2’-deoxyguanosine in the CSF of patients with Alzheimer’s disease demonstrate that mitochondrial oxidative damage and/or oxidative DNA damage contributes to the neurodegenerative process. J. Neurol. 2010, 257, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Molina, J.A.; de Bustos, F.; Ortiz, S.; Del Ser, T.; Seijo, M.; Benito-Léon, J.; Oliva, J.M.; Pérez, S.; Manzanares, J. Serum levels of coenzyme Q in patients with Lewy body disease. J. Neural. Transm. 2002, 109, 1195–1201. [Google Scholar] [CrossRef]
- Yamagishi, K.; Ikeda, A.; Moriyama, Y.; Chei, C.L.; Noda, H.; Umesawa, M.; Cui, R.; Nagao, M.; Kitamura, A.; Yamamoto, Y.; et al. Serum coenzyme Q10 and risk of disabling dementia, the Circulatory Risk in Communities Study (CIRCS). Atherosclerosis 2014, 237, 400–403. [Google Scholar] [CrossRef]
- Chang, P.S.; Chou, H.H.; Lai, T.J.; Yen, C.H.; Pan, J.C.; Lin, P.T. Investigation of coenzyme Q10 status.; serum amyloid-β.; and tau protein in patients with dementia. Front. Aging Neurosci. 2022, 14, 910289. [Google Scholar] [CrossRef]
- Balduzzi, S.; Rücker, G.; Schwarzer, G. How to perform a meta-analysis with R: A practical tutorial. BMJ Ment. Health 2019, 22, 153–160. [Google Scholar] [CrossRef]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2009, 283, 2008–2012. [Google Scholar] [CrossRef]
- Der Simonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D. Confidence intervals for the between-study variance in random effects meta-analysis using generalised Cochran heterogeneity statistics. Res. Synth. Methods 2013, 4, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.V. Meta-Analysis. J. Educ. Stat. 1992, 17, 279–296. [Google Scholar] [CrossRef]
- Edlund, C.; Söderberg, M.; Kristensson, K.; Dallner, G. Ubiquinone, dolichol, and cholesterol metabolism in aging and Alzheimer’s disease. Biochem. Cell. Biol. 1992, 70, 422–428. [Google Scholar] [CrossRef]
- Kim, S.H.; Vlkolinsky, R.; Cairns, N.; Fountoulakis, M.; Lubec, G. The reduction of NADH ubiquinone oxidoreductase 24- and 75-kDa subunits in brains of patients with Down syndrome and Alzheimer’s disease. Life Sci. 2001, 68, 2741–2750. [Google Scholar] [CrossRef]
- Santa-Mara, I.; Santpere, G.; MacDonald, M.J.; Gomez de Barreda, E.; Hernandez, F.; Moreno, F.J.; Ferrer, I.; Avila, J. Coenzyme q induces tau aggregation.; tau filaments.; and Hirano bodies. J. Neuropathol. Exp. Neurol. 2008, 67, 428–434. [Google Scholar] [PubMed]
- McDonald, S.R.; Sohal, R.S.; Forster, M.J. Concurrent administration of coenzyme Q10 and alpha-tocopherol improves learning in aged mice. Free Radic. Biol. Med. 2005, 38, 729–736. [Google Scholar] [CrossRef]
- Wadsworth, T.L.; Bishop, J.A.; Pappu, A.S.; Woltjer, R.L.; Quinn, J.F. Evaluation of coenzyme Q as an antioxidant strategy for Alzheimer’s disease. J. Alzheimers Dis. 2008, 14, 225–234. [Google Scholar] [PubMed]
- Sumien, N.; Heinrich, K.R.; Shetty, R.A.; Sohal, R.S.; Forster, M.J. Prolonged intake of coenzyme Q10 impairs cognitive functions in mice. J. Nutr. 2009, 139, 1926–1932. [Google Scholar] [CrossRef]
- Shetty, R.A.; Forster, M.J.; Sumien, N. Coenzyme Q(10) supplementation reverses age-related impairments in spatial learning and lowers protein oxidation. Age 2013, 35, 1821–1834. [Google Scholar] [CrossRef] [PubMed]
- Shetty, R.A.; Ikonne, U.S.; Forster, M.J.; Sumien, N. Coenzyme Q10 and α-tocopherol reversed age-associated functional impairments in mice. Exp. Gerontol. 2014, 58, 208–218. [Google Scholar] [CrossRef]
- Ibrahim Fouad, G. Combination of Omega 3 and Coenzyme Q10 Exerts Neuroprotective Potential against Hypercholesterolemia-Induced Alzheimer’s-Like Disease in Rats. Neurochem. Res. 2020, 45, 1142–1155. [Google Scholar] [CrossRef]
- Ali, A.A.; Abo El-Ella, D.M.; El-Emam, S.Z.; Shahat, A.S.; El-Sayed, R.M. Physical & mental activities enhance the neuroprotective effect of vinpocetine & coenzyme Q10 combination against Alzheimer & bone remodeling in rats. Life Sci. 2019, 229, 21–35. [Google Scholar]
- Attia, H.; Albuhayri, S.; Alaraidh, S.; Alotaibi, A.; Yacoub, H.; Mohamad, R.; Al-Amin, M. Biotin, coenzyme Q10, and their combination ameliorate aluminium chloride-induced Alzheimer’s disease via attenuating neuroinflammation and improving brain insulin signaling. J. Biochem. Mol. Toxicol. 2020, 34, e22519. [Google Scholar] [CrossRef]
- Ali, A.A.; Khalil, M.G.; Abd El-Latif, D.M.; Okda, T.; Abdelaziz, A.I.; Abu-Elfotuh, K.; Kamal, M.M.; Wahid, A. The influence of vinpocetine alone or in combination with Epigallocatechin-3-gallate, Coenzyme COQ10, Vitamin E and Selenium as a potential neuroprotective combination against aluminium-induced Alzheimer’s disease in Wistar Albino Rats. Arch. Gerontol. Geriatr. 2022, 98, 104557. [Google Scholar] [CrossRef] [PubMed]
- Nitta, A.; Murakami, Y.; Furukawa, Y.; Kawatsura, W.; Hayashi, K.; Yamada, K.; Hasegawa, T.; Nabeshima, T. Oral administration of idebenone induces nerve growth factor in the brain and improves learning and memory in basal forebrain-lesioned rats. Naunyn Schmiedebergs Arch. Pharmacol. 1994, 349, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Tanaka, T.; Han, D.; Senzaki, K.; Kameyama, T.; Nabeshima, T. Protective effects of idebenone and alpha-tocopherol on beta-amyloid-(1-42)-induced learning and memory deficits in rats: Implication of oxidative stress in beta-amyloid-induced neurotoxicity in vivo. Eur. J. Neurosci. 1999, 11, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Kumar, A. Microglial Inhibitory Mechanism of Coenzyme Q10 Against Aβ (1-42) Induced Cognitive Dysfunctions: Possible Behavioral, Biochemical, Cellular, and Histopathological Alterations. Front. Pharmacol. 2015, 6, 268. [Google Scholar] [CrossRef]
- Komaki, H.; Faraji, N.; Komaki, A.; Shahidi, S.; Etaee, F.; Raoufi, S.; Mirzaei, F. Investigation of protective effects of coenzyme Q10 on impaired synaptic plasticity in a male rat model of Alzheimer’s disease. Brain Res. Bull. 2019, 147, 14–21. [Google Scholar] [CrossRef]
- Ishrat, T.; Khan, M.B.; Hoda, M.N.; Yousuf, S.; Ahmad, M.; Ansari, M.A.; Ahmad, A.S.; Islam, F. Coenzyme Q10 modulates cognitive impairment against intracerebroventricular injection of streptozotocin in rats. Behav. Brain Res. 2006, 171, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Sheykhhasan, M.; Amini, R.; Soleimani Asl, S.; Saidijam, M.; Hashemi, S.M.; Najafi, R. Neuroprotective effects of coenzyme Q10-loaded exosomes obtained from adipose-derived stem cells in a rat model of Alzheimer’s disease. Biomed. Pharmacother. 2022, 152, 113224. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yang, Y.; Li, G.; Wang, J.; Yang, E.S. Coenzyme Q10 attenuates beta-amyloid pathology in the aged transgenic mice with Alzheimer presenilin 1 mutation. J. Mol. Neurosci. 2008, 34, 165–171. [Google Scholar] [CrossRef]
- Yang, X.; Dai, G.; Li, G.; Yang, E.S. Coenzyme Q10 reduces beta-amyloid plaque in an APP/PS1 transgenic mouse model of Alzheimer’s disease. J. Mol. Neurosci. 2010, 41, 110–113. [Google Scholar] [CrossRef]
- Dumont, M.; Kipiani, K.; Yu, F.; Wille, E.; Katz, M.; Calingasan, N.Y.; Gorras, G.K.; Lin, M.T.; Beal, M.F. Coenzyme Q10 decreases amyloid pathology and improves behavior in a transgenic mouse model of Alzheimer’s disease. J. Alzheimers Dis. 2011, 27, 211–223. [Google Scholar] [CrossRef]
- Elipenahli, C.; Snack, C.; Jainuddin, S.; Gerges, M.; Yang, L.; Starkov, A.; Beal, M.F.; Dumont, M. Behavioral improvement after chronic administration of coenzyme Q10 in P301S transgenic mice. J. Alzheimers Dis. 2012, 28, 173–182. [Google Scholar] [CrossRef]
- Muthukumaran, K.; Kanwar, A.; Vegh, C.; Marginean, A.; Elliott, A.; Guilbeault, N.; Badour, A.; Sikorska, M.; Cohen, J.; Pandey, S. Ubisol-Q10 (a Nanomicellar Water-Soluble Formulation of CoQ10) Treatment Inhibits Alzheimer-Type Behavioral and Pathological Symptoms in a Double Transgenic Mouse (TgAPEswe.; PSEN1dE9) Model of Alzheimer’s Disease. J. Alzheimers Dis. 2018, 61, 221–236. [Google Scholar] [CrossRef]
- Sui, X.; Ren, X.; Huang, P.; Li, S.; Ma, Q.; Ying, M.; Ni, J.; Liu, J.; Yang, X. Proteomic analysis of serum proteins in triple transgenic Alzheimer’s disease mice: Implications for identifying biomarkers for use to screen potential candidate therapeutic drugs for early Alzheimer’s disease. J Alzheimers Dis. 2014, 40, 575–586. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Stokes, K.; Mahngar, K.; Domazet-Damjanov, D.; Sikorska, M.; Pandey, S. Inhibition of stress induced premature senescence in presenilin-1 mutated cells with water soluble Coenzyme Q10. Mitochondrion 2014, 17, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Vegh, C.; Pupulin, S.; Wear, D.; Culmone, L.; Huggard, R.; Ma, D.; Pandey, S. Resumption of Autophagy by Ubisol-Q10 in Presenilin-1 Mutated Fibroblasts and Transgenic AD Mice, Implications for Inhibition of Senescence and Neuroprotection. Oxid. Med. Cell. Longev. 2019, 2019, 7404815. [Google Scholar] [CrossRef]
- Qi, X.L.; Xiu, J.; Shan, K.R.; Xiao, Y.; Gu, R.; Liu, R.Y.; Guan, Z.Z. Oxidative stress induced by beta-amyloid peptide(1-42) is involved in the altered composition of cellular membrane lipids and the decreased expression of nicotinic receptors in human SH-SY5Y neuroblastoma cells. Neurochem. Int. 2005, 46, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Durán-Prado, M.; Frontiñán, J.; Santiago-Mora, R.; Peinado, J.R.; Parrado-Fernández, C.; Gómez-Almagro, M.V.; Moreno, M.; López-Domínguez, J.A.; Villalba, J.M.; Alcaín, F.J. Coenzyme Q10 protects human endothelial cells from β-amyloid uptake and oxidative stress-induced injury. PLoS ONE 2014, 9, e109223. [Google Scholar] [CrossRef] [PubMed]
- Frontiñán-Rubio, J.; Rabanal-Ruiz, Y.; Durán-Prado, M.; Alcain, F.J. The Protective Effect of Ubiquinone against the Amyloid Peptide in Endothelial Cells Is Isoprenoid Chain Length-Dependent. Antioxidants 2021, 10, 1806. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Park, H.H.; Koh, S.H.; Choi, N.Y.; Yu, H.J.; Park, J.; Lee, Y.J.; Lee, K.Y. Coenzyme Q10 protects against amyloid beta-induced neuronal cell death by inhibiting oxidative stress and activating the P13K pathway. Neurotoxicology 2012, 33, 85–90. [Google Scholar] [CrossRef]
- Wang, H.; Li, L.; Jia, K.; Wang, Q.; Sui, S.; Lin, Y.; He, Y. Idebenone protects mitochondrial function against amyloid beta toxicity in primary cultured cortical neurons. Neuroreport 2020, 31, 1104–1110. [Google Scholar] [CrossRef]
- Choi, H.; Park, H.H.; Lee, K.Y.; Choi, N.Y.; Yu, H.J.; Lee, Y.J.; Park, J.; Huh, Y.M.; Lee, S.H.; Koh, S.H. Coenzyme Q10 restores amyloid beta-inhibited proliferation of neural stem cells by activating the PI3K pathway. Stem Cells Dev. 2013, 22, 2112–2120. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Lian, N.; Yu, Y.; Wang, Y.; Xie, K.; Yu, Y. Coenzyme Q10 alleviates sevoflurane-induced neuroinflammation by regulating the levels of apolipoprotein E and phosphorylated tau protein in mouse hippocampal neurons. Mol. Med. Rep. 2020, 22, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Moreira, P.I.; Santos, M.S.; Sena, C.; Nunes, E.; Seiça, R.; Oliveira, C.R. CoQ10 therapy attenuates amyloid beta-peptide toxicity in brain mitochondria isolated from aged diabetic rats. Exp. Neurol. 2005, 196, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Xu, D.; Lin, J.; Zhang, D.; Wang, G.; Sui, L.; Ding, H.; Du, J. Coenzyme Q10 attenuated β-amyloid25-35-induced inflammatory responses in PC12 cells through regulation of the NF-κB signaling pathway. Brain Res Bull. 2017, 131, 192–198. [Google Scholar] [CrossRef]
- Imagawa, M.; Naruse, S.; Tsuji, S.; Fujioka, A.; Yamaguchi, H. Coenzyme Q10.; iron.; and vitamin B6 in genetically-confirmed Alzheimer’s disease. Lancet 1992, 340, 671. [Google Scholar] [CrossRef] [PubMed]
- Weyer, G.; Babej-Dölle, R.M.; Hadler, D.; Hofmann, S.; Herrmann, W.M. A controlled study of 2 doses of idebenone in the treatment of Alzheimer’s disease. Neuropsychobiology 1997, 36, 73–82. [Google Scholar] [CrossRef]
- Gutzmann, H.; Hadler, D. Sustained efficacy and safety of idebenone in the treatment of Alzheimer’s disease: Update on a 2-year double-blind multicentre study. J. Neural Transm. Suppl. 1998, 54, 301–310. [Google Scholar]
- Gutzmann, H.; Kühl, K.P.; Hadler, D.; Rapp, M.A. Safety and efficacy of idebenone versus tacrine in patients with Alzheimer’s disease: Results of a randomized, double-blind, parallel-group multicenter study. Pharmacopsychiatry 2002, 35, 12–18. [Google Scholar] [CrossRef]
- Thal, L.J.; Grundman, M.; Berg, J.; Ernstrom, K.; Margolin, R.; Pfeiffer, E.; Weiner, M.F.; Zamrini, E.; Thomas, R.G. Idebenone treatment fails to slow cognitive decline in Alzheimer’s disease. Neurology 2003, 61, 1498–1502. [Google Scholar] [CrossRef]
- Voronkova, K.V.; Meleshkov, M.N. Use of Noben (idebenone) in the treatment of dementia and memory impairments without dementia. Neurosci. Behav. Physiol. 2009, 39, 501–506. [Google Scholar] [CrossRef]
- Galasko, D.R.; Peskind, E.; Clark, C.M.; Quinn, J.F.; Ringman, J.M.; Jicha, G.A.; Cotman, C.; Cottrell, B.; Montine, T.J.; Thomas, R.G.; et al. Alzheimer’s Disease Cooperative Study. Antioxidants for Alzheimer disease: A randomized clinical trial with cerebrospinal fluid biomarker measures. Arch. Neurol. 2012, 69, 836–841. [Google Scholar] [CrossRef]
- Karakahya, R.H.; Özcan, T.Ş. Salvage of the retinal ganglion cells in transition phase in Alzheimer’s disease with topical coenzyme Q10, is it possible? Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 411–418. [Google Scholar] [CrossRef]
- Kawakami, M.; Itoh, T. Effects of idebenone on monoamine metabolites in cerebrospinal fluid of patients with cerebrovascular dementia. Arch. Gerontol. Geriatr. 1989, 8, 343–353. [Google Scholar] [CrossRef]
- Qi, F.X.; Hu, Y.; Kang, L.J.; Li, P.; Gao, T.C.; Zhang, X. Effects of Butyphthalide Combined with Idebenone on Inflammatory Cytokines and Vascular Endothelial Functions of Patients with Vascular Dementia. J. Coll. Physicians Surg. Pak. 2020, 30, 23–27. [Google Scholar] [CrossRef]
- García-Carpintero, S.; Domínguez-Bértalo, J.; Pedrero-Prieto, C.; Frontiñán-Rubio, J.; Amo-Salas, M.; Durán-Prado, M.; García-Pérez, E.; Vaamonde, J.; Alcain, F.J. Ubiquinol Supplementation Improves Gender-Dependent Cerebral Vasoreactivity and Ameliorates Chronic Inflammation and Endothelial Dysfunction in Patients with Mild Cognitive Impairment. Antioxidants 2021, 10, 143. [Google Scholar] [CrossRef]
- Stough, C.; Nankivell, M.; Camfield, D.A.; Perry, N.L.; Pipingas, A.; Macpherson, H.; Wesnes, K.; Ou, R.; Hare, D.; de Haan, J.; et al. CoQ10 and Cognition a Review and Study Protocol for a 90-Day Randomized Controlled Trial Investigating the Cognitive Effects of Ubiquinol in the Healthy Elderly. Front. Aging Neurosci. 2019, 11, 103. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Portero, C.; Amián, J.G.; Bella, R.; López-Lluch, G.; Alarcón, D. Coenzyme Q10 Levels Associated with Cognitive Functioning and Executive Function in Older Adults. J. Gerontol. A Biol. Sci. Med. Sci. 2023, 78, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Grundman, M.; Grundman, M.; Delaney, P. Antioxidant strategies for Alzheimer’s disease. Proc. Nutr. Soc. 2002, 61, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Beal, M.F. Mitochondrial dysfunction and oxidative damage in Alzheimer’s and Parkinson’s diseases and coenzyme Q10 as a potential treatment. J. Bioenerg. Biomembr. 2004, 36, 381–386. [Google Scholar] [CrossRef]
- Lönnrot, K.; Metsä-Ketelä, T.; Molnár, G.; Ahonen, J.P.; Latvala, M.; Peltola, J.; Pietilä, T.; Alho, H. The effect of ascorbate and ubiquinone supplementation on plasma and CSF total antioxidant capacity. Free Radic. Biol. Med. 1996, 21, 211–217. [Google Scholar] [CrossRef]
- Shults, C.W.; Flint Beal, M.; Song, D.; Fontaine, D. Pilot trial of high dosages of coenzyme Q10 in patients with Parkinson’s disease. Exp. Neurol. 2004, 188, 491–494. [Google Scholar] [CrossRef] [PubMed]
- Nukui, K.; Yamagishi, T.; Miyawaki, H.; Kettawan, A.; Okamoto, T.; Belardinelli, R.; Tiano, L.; Littarru, G.P.; Sato, K. Blood CoQ10 levels and safety profile after single-dose or chronic administration of PureSorb-Q40, animal and human studies. Biofactors 2008, 32, 209–219. [Google Scholar] [CrossRef] [PubMed]

| Alzheimer’s Disease (AD) | |||||||
| Tissue | Author, Year [Ref] | Parameter | AD N | AD Mean ± SD | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
| Serum/plasma | De Bustos et al., 2000 [8] | Total CoQ10 (nmol/L) | 44 | 1262 ± 389 | 21 | 1209 ± 457 | 53.00 (−165.26 to 271.26); 0.629 |
| Battino et al., 2003 [9] | Total CoQ10 (nmol/L) | 18 | 1216 ± 69 | 53 | 1278 ± 104 | −62.00 (−114.55 to −9.45), 0.021 | |
| Giavarotti et al., 2013 [10] | Total CoQ10 (nmol/L) | 23 | 130 ± 30 | 42 | 115 ± 20 | 15.00 (2.57 to 27.43), 0.019 | |
| Total series | Total CoQ10 (nmol/L) | 85 | 945.95 ± 573.50 | 116 | 844.42 ± 588.71 | Random effects model p = 0.911 | |
| De Bustos et al., 2000 [8] | Total CoQ10 / cholesterol (µmol/mmol) | 44 | 0.24 ± 0.05 | 21 | 0.23 ± 0.05 | 0.01 (−0.02 to 0.04), 0.454 | |
| CSF | Isobe et al., 2009, 2010 [11,12] | Oxidized CoQ10 (nmol/L) | 30 | 5.2 ± 1.5 | 30 | 1.9 ± 1.3 | 3.30 (2.69 to 3.91), <0.0001 |
| Isobe et al., 2009, 2010 [11,12] | Reduced CoQ10 (nmol/L) | 30 | 1.4 ± 0.6 | 30 | 2.7 ± 0.7 | −1.30 (−1.64 to −0.96), <0.0001 | |
| Isobe et al., 2009, 2010 [11,12] | Total CoQ10 (nmol/L) | 30 | 6.6 ± 1.2 | 30 | 4.6 ± 1.0 | 2.00 (1.43 to 2.57), <0.0001 | |
| Isobe et al., 2009, 2010 [11,12] | Oxidized/total CoQ10 | 30 | 0.782 ± 0.188 | 30 | 0.413 ± 0.104 | 0.37 (0.29 to 0.45), <0.0001 | |
| Lewy Body Dementia (LBD) | |||||||
| Tissue | Author, Year [Ref] | Parameter | LBD N | LBD Mean ± SD | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
| Serum/plasma | Molina et al., 2002 [13] | Total CoQ10 (nmol/L) | 18 | 960.6 ± 359.1 | 20 | 1205.2 ± 362.2 | −244.60 (−482.30 to −6.90 to); 0.044 |
| Molina et al., 2002 [13] | Total CoQ10/cholesterol | 18 | 4.67 ± 1.75 | 20 | 5.05 ± 1.52 | −0.38 (−1.46 to 0.70); 0.478 | |
| Vascular Dementia (VD) | |||||||
| Tissue | Author, Year [Ref] | Parameter | VD N | VD Mean ± SD | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
| Serum/plasma | De Bustos et al., 2000 [8] | Total CoQ10 (nmol/L) | 17 | 1130 ± 452 | 21 | 1209 ± 457 | −79.00 (−379.92 to 221.92), 0.598 |
| De Bustos et al., 2000 [8] | Total CoQ10/cholesterol (µmol/mmol) | 44 | 0.22 ± 0.06 | 21 | 0.23 ± 0.05 | −0.01 (−0.04 to 0.02), 0.511 | |
| Dementia without Specific Etiologic Diagnosis (DEM) | |||||||
| Tissue | Author, Year [Ref] | Parameter | DEM N | DEM Mean ± SD | HC N | HC Mean ± SD | Difference in Means (95% C.I.), p |
| Serum/plasma | Yamagishi et al., 2014 [14] | Total CoQ10 (nmol/L) | 65 | 731 ± NA | 130 | 762 ± NA | p = 0.32 (according to the authors, SD not provided) |
| Serum/plasma | Chang et al., 2022 [15] | Total CoQ10 (nmol/L) | 80 | 410 ± 21 | NA | NA | 73% of patients showed low CoQ10 status using as reference values 500–1700 nmol/L |
| Serum/plasma | Yamagishi et al., 2014 [14] | Total CoQ10/cholesterol (µmol /mmol) | 65 | 0.14 ± NA | 130 | 0.15 ± NA | p = 0.15 (according to the authors, SD not provided) |
| Serum/plasma | Chang et al., 2022 [15] | Total CoQ10/cholesterol (µmol/mmol) | 80 | 0.09 ± 0.04 | NA | NA | 73% of patients showed low CoQ10 status using as reference values 500–1700 nmol/L |
| Experimental Model | Author, Year [Ref] | Main Findings |
|---|---|---|
| AGED RATS | McDonald et al., 2005 [24] | Coadministration of CoQ10 and alpha-tocopherol (but not administration of each of these compounds alone) improved learning and memory tasks (assessed by a test that required the mice to rapidly identify and remember the correct arm of a T-maze, and to respond preemptively in order to avoid an electric shock). |
| AGED MICE | Wadsworth et al., 2008 [25] | Administration of CoQ10 decreased protein carbonyls in the brain but had no effect on lipid peroxidation, brain ATP levels, and mitochondrial membrane potential. |
| Sumien et al., 2009 [26] | Intake of a low-CoQ10 diet did not change age-associated decrements in muscle strength, balance, coordinated running, or learning/memory, whereas intake of CoQ10 at a higher amount increased spontaneous activity, worsened age-related losses in acuity to auditory and shock stimuli, and impaired spatial learning/memory of old mice. | |
| Shetty et al., 2013 [27] | Intake of a low-CoQ10 diet did not change age-associated decrements in tests for spatial learning (Morris water maze), spontaneous locomotor activity, motor coordination, and startle reflex. However, intake of high-CoQ10 improved spatial learning and decreased protein oxidative damage in the heart, liver, skeletal muscle, and to a lesser extent, in the brain mitochondria. | |
| Shetty et al., 2014 [28] | Administration of α-tocopherol or α-tocopherol + CoQ10 diets improved coordinated running performance. The α -tocopherol + CoQ10 diet improved performance in a discriminated avoidance task (α-tocopherol and CoQ10 diets alone improved this task to a lesser degree). Both α-tocopherol and CoQ10 diets decreased protein damage, this effect being more marked with the α-tocopherol + CoQ10 combination. | |
| HYPERCHOLESTE-ROLEMIA-INDUCED AD IN RATS | Ibrahim Fouad, 2020 [29] | Treatment with omega-3 and CoQ10 alone or in combination decreased markers of brain oxidative stress and inflammation and serum Aβ levels, regulated cholinergic functioning, and enhanced the functional outcome. |
| ALUMINIUM-INDUCED AD IN RATS | Ali et al., 2019 [30] | Treatment with CoQ10 in combination with vinpocetine partially reversed the changes induced by aluminium chloride (AlCl3) by decreasing malonyl-dialdehyde (MDA), increasing superoxide dismutase (SOD) and total antioxidant total capacity, decreasing IL1β, TNFα, chitinase, β-secretase, Aβ, tau protein, acetyl-cholinesterase, increasing catecholamine and brain-derived neurotrophic factor (BDNF) levels in brain tissue. |
| Attia et al., 2020 [31] | Treatment with CoQ10 alone or in combination with biotin attenuated the changes induced by AlCl3 (impaired memory, a significant increase in Aβ, lipid peroxides, inflammatory markers—TNFα, IL6, IL1, nuclear factor κB-, caspase-3, and pSer-IRS-1, significant reduction in the antioxidants reduced glutathione and SOD-, pTyr-IRS-1, and p-Akt, reflecting Aβ-induced inflammation and defective insulin signaling, focal aggregations of inflammatory cells and neuronal degeneration). | |
| Ali et al., 2022 [32] | Treatment with CoQ10 reversed changes induced by aluminium by decreasing Aβ and acetylcholinesterase expression, increasing monoamine levels, restoring levels of total antioxidant capacity and superoxide-dismutase, and decreasing MDA, TNFα, and IL6. | |
| FOREBRAIN LESIONED RATS | Nitta et al., 1994 [33] | Administration of CoQ10 to forebrain lesioned rats caused an increase in nerve growth factor (NGF) protein and mRNA and in choline acetyltransferase activity, and improved memory tasks such as behavioral deficits in habituation, water maze, and passive avoidance tasks in these animals. |
| INTRACEREBRO-VENTRICULAR INFUSION OF Aβ(1-42) IN RATS | Yamada et al., 1999 [34] | Coadministration of CoQ10 prevented some learning and memory deficits (Y-maze and water maze, but not passive avoidance tasks) in this model without affecting lipid peroxide levels in the hippocampus and cerebral cortex. |
| INTRAHIPPO- CAMPAL INJECTION OF Aβ(1-42) IN RATS | Singh et al., 2015 [35] | Treatment with CoQ10 and minocycline alone improved cognitive performance (reduced transfer latency and increased time spent in the target quadrant in the Morris Water Maze), reduced acetyl–cholinesterase activity, decreased oxidative damage (by reducing lipoperoxide and nitrite level and restoring superoxide, catalase, and reduced glutathione levels), decreased TNFα level, and restored mitochondrial respiratory enzyme complex activities and histopathological alterations induced by Aβ(1-42) in a dose-dependent and synergistic manner. |
| Komaki et al., 2019 [36] | Treatment with CoQ10 reversed the decreased excitatory postsynaptic potential (EPSP) slope and population spike (PS) amplitude in the hippocampal dentate gyrus after induction of long-term potentiation (LTP) induced by injection of Aβ, reversed the decrease in serum MDA levels and total oxidant levels induced by injection of Aβ, and increased total antioxidant capacity levels. | |
| INTRACEREBRO-VENTRICULAR INFUSION OF STREPTOZOTOCIN IN RATS | Ishrat et al., 2006 [37] | Coadministration of CoQ10 prevented learning and memory deficits (loss of cognitive performance in Morris water maze and passive avoidance tests), the increase in markers of oxidative damage (thiobarbituric acid reactive substances, reduced glutathione, protein carbonyl, activities of glutathione peroxidase and glutathione reductase), the decline of ATP in the hippocampus and cerebral cortex, the decrease in choline-acetyl-transferase activity and the increase in acetyl-cholinesterase activity induced by this neurotoxin. |
| Sheykhhasan et al., 2022 [38] | Administration of CoQ10-loaded exosomes derived from adipose-derived stem cells improved memory impairment (assessed with the Morris water maze and passive avoidance task), increased BDNF expression, and increased cell density and the transcription factor SOX2 gene expression in comparison with the administration of CoQ10 exosomes derived from adipose-derived stem cells alone. | |
| TRANSGENIC MICE: AD PRESENILIN 1 MUTATION L235P | Yang et al., 2008 [39] | CoQ10 administration partially attenuated Abeta overproduction and intracellular Aβ deposit, partially decreased MDA increase, and up-regulated the decreased activity of SOD [24]. |
| Yang et al., 2010 [40] | CoQ10 administration reduced the burden of the amyloid plaques (assessed by immunohistochemistry and magnetic resonance imaging) | |
| TRANSGENIC MICE: TG19959 MUTATION | Dumont et al., 2011 [41] | CoQ10 administration improved cognitive performance during Morris water maze testing, decreased brain levels of protein carbonyls (a marker of oxidative stress), decreased brain Aβ42 levels and Aβ protein precursor (AβPP), β-carboxyterminal fragments, and decreased plaque area and number in the hippocampus and the overlying cortex (assessed by immunostained with an Aβ42-specific antibody). |
| TRANSGENIC MICE: P301 TAU MUTATION (FRONTO- TEMPORAL DEMENTIA) | Elipenahli et al., 2012 [42] | CoQ10 administration improved survival and behavioral deficits (it increased locomotor activity and anxiety in open field testing), caused a modest reduction in phosphorylated tau, a significant increase in complex I activity and protein levels, and a reduction in lipid peroxidation in the cortex. |
| DOUBLE TRANSGENIC MICE: MUTATIONS TGAPESWE AND PSEN1DE9 | Muthukumaran et al., 2018 [43] | Administration of ubisol-Q10 (a water-soluble form of coenzyme Q10) improved long-term memory, preserved working spatial memory, and inhibited Aβ plaque formation in 18-month-old transgenic mice compared to an untreated transgenic group. |
| TRIPLE TRANSGENIC MICE: MUTATIONS PS1M146V, APPSWE, AND TAUP301L | Sui et al., 2014 [44] | The administration of CoQ10 altered changes in the differentially expressed serum proteins in the transgenic compared with wild-type mice by up-regulating 10 proteins and down-regulating another 10 proteins. Among the proteins modulated by CoQ10, clusterin and α-2-macroglobulin were validated via ELISA assay. |
| CELL CULTURES: HUMAN SKIN FIBROBLASTS FROM PS1 MUTATED FAMILIAL AD | Ma et al., 2014 [45] | CoQ10 treatment decreased reactive oxygen species generation, increased population doublings, and postponed stress-induced premature senescence. CoQ10 treatment increased proliferating cell nuclear antigen expression, and decreased levels of manganese-SOD (MnSOD), p21, p16Ink4A and cell cycle regulatory protein retinoblastoma (suggesting a resumption of autophagy). |
| Vegh et al., 2019 [46] | Administration of ubisol-Q10 caused enhanced expression autophagy-related genes such as beclin-1 (a major autophagy regulator) and mitogen-activated protein kinase 8 (MAPK8/JNK1, a major activator of beclin-1) avoiding resumption of premature senescence. Withdrawal of ubisol-Q10 treatment led to the return of the senescence phenotype in AD fibroblasts. | |
| CELL CULTURES: HUMAN SH-SY5Y NEUROBLASTOMA CELLS | Qi et al., 2005 [47] | Exposure of these cells to Aβ(1-42) caused, among other effects, enhanced lipid peroxidation and protein oxidation and significant reductions in the total contents of phospholipids, ubiquinone-10, and alpha3 and alpha7 subunit proteins of nicotinic acetylcholine receptors. |
| CELL CULTURES: HUMAN MC65 NEUROBLASTOMA CELLS | Wadsworth et al., 2008 [25] | Administration of CoQ10 showed a neuroprotective effect on the neurotoxic effects induced by the Aβ protein precursor C-terminal fragment (APP CTF). |
| CELL CULTURES: HUMAN UMBILICAL VEIN ENDOTHELIAL CELLS (HUVECS) | Durán-Prado et al., 2014 [48] | CoQ10 pretreatment delayed Aβ incorporation into the plasma membrane and mitochondria, reduced the influx of extracellular Ca2+ and Ca2+ release from mitochondria due to opening the mitochondrial transition pore after Aβ administration, decreasing O2− and hydrogen peroxide (H2O2) levels, prevented Aβ-induced necrosis and apoptosis, and restored the ability to proliferate, migrate and form tube-like structures in vitro. |
| CELL CULTURES: RAT BRAIN ENDOTHELIAL CELLS | Frontiñán-Rubio et al., 2021 [49] | CoQ10 pretreatment protected endothelial brain cells from Aβ(25–35)-induced damage, preventing nicotinamide adenine dinucleotide phosphate (NADPH) oxidase activity and reducing both reactive oxygen species generation and increase in free cytosolic Ca2+ induced by Aβ(25–35) (this prevented apoptosis and necrosis). |
| CELL CULTURES: PRIMARY CULTURED RAT CORTICAL NEURONS | Choi et al., 2012 [50] | CoQ10 protected neuronal cells against Aβ(25–35)-induced neurotoxicity in a concentration-dependent manner by increasing the expression levels of proteins related to neuronal cell survival (p85aPI3K, phosphorylated protein kinase B–Akt-, phosphorylated glycogen synthase kinase-3β, and heat shock transcription factor), and decreasing the levels of proteins associated with neuronal death (cytosolic cytochrome c and cleaved caspase-3). This protective effect was blocked by a phosphatidylinositol 3-kinase (PI3K) inhibitor. |
| Wang et al., 2020 [51] | CoQ10 pretreatment significantly prevented neurons from Aβ-induced collapse of mitochondrial bioenergetics and perturbations of the protein kinase A (PKA)/cAMP response element-binding protein (CREB) signaling. | |
| CELL CULTURES: CULTURED NEURAL STEM CELLS | Choi et al., 2013 [52] | Co-administration of CoQ10 restored the Aβ(25–35) oligomer-inhibited proliferation of neural stem cells by increasing the expression levels of proteins related to the PI3K pathway (p85α PI3K, phosphorylated Akt-Ser473-, phosphorylated glycogen synthase kinase-3β-Ser9-, and heat shock transcription factor). This protective effect was blocked by a phosphatidylinositol 3-kinase (PI3K) inhibitor. |
| CELL CULTURES: PRIMARY CULTURED HIPPOCAMPAL NEURONS FROM FETAL MICE | Yang et al., 2020 [53] | Administration of CoQ10 reversed all the effects induced by sevoflurane anesthesia (decrease in ATP and SOD levels, increase in apolipoprotein E (ApoE) mRNA, total ApoE protein, full-length ApoE, and ApoE fragments, increase in phosphorylated tau and neuroinflammatory factor (TNFα, IL6, and IL1β) expression levels. |
| CELL CULTURES: BRAIN MITOCHON-DRIA ISOLATED FROM AGED DIABETIC RATS | Moreira et al., 2005 [54] | CoQ10 treatment attenuated the decrease in oxidative phosphorylation efficiency and avoided the increase in H2O2 production induced by Aβ1-40. |
| CELL CULTURES: RAT PHEOCHROMO-CYTOMA (PC12) CELL LINE | Li et al., 2017 [55] | CoQ10 treatment suppressed the protein expression of COX-2 and the level of PGE2 in Aβ(25–35)-injured PC12 cells (this effect was correlated with the suppression of NF-κB activation by CoQ10, attenuating neuroinflammation). |
| Authors, Year [Ref] | Study Setting | Type of Study | Main Findings | Level of Evidence (Quality Score) |
|---|---|---|---|---|
| Imagawa et al., 1992 [56] | Combined therapy with CoQ10, iron, and vitamin B6 in 27 AD patients. | Open-label study |
| II (NA) |
| Weyer et al., 1997 [57] | Three hundred patients with mild to moderate degree DAT were prescribed idebenone 30 mg t.i.d. (n = 100), idebenone 90 t.i.d. (n = 100), or placebo (n = 100). Evaluation at baseline, 1, 3, and 6 months including a total score of the ADAS-Total, ADAS cognitive (ADAS-Cog) and noncognitive scores (ADAS-Noncog), CGI-I, MMSE, Digit Symbol Substitution test (DSS) and several scales for the assessment of daily activities (the self- and observer-rating scales NAA and NAB of the Nuremberg Age Inventory NAI and Greene’s Assessment). | Multicenter, randomized, double-blind, placebo-controlled, dosage-ranging trial |
| I (>50%) |
| Gutzmann and Hadler D, 1998 [58] | Four hundred and fifty patients with mild to moderate degree DAT were prescribed placebo for 12 months, followed by idebenone 90 mg for another 12 months (n = 153) or idebenone 90 mg tid for 24 months (n = 148) or 120 mg ti for 24 months (n = 149). Evaluation included a total score of the Alzheimer’s Disease Assessment Scale (ADAS-Total), ADAS cognitive (ADAS-Cog) and noncognitive scores (ADAS-Noncog), CGI-Improvement), the SKT neuropsychological test battery, and the Nurses’ Observation Scale for Geriatric Patients (NOSGER-Total and IADL subscale). | Prospective, randomized, double-blind multicentre study in three parallel groups |
| I (>50%) |
| Gutzmann et al., 2002 [59] | Two hundred and three patients with mild to moderate degree DAT were prescribed idebenone 360 mg/day (n = 104) or tacrine up to 160 mg/day (n = 99) for 60 weeks. Evaluation included the Efficacy Index Score (EIS, a combination of improvement in cognitive function, activities of daily living, and global function), the ADAS-Cog score, the NOSGER-IADL score, and the CGI-I. | Prospective, randomized, double-blind, parallel-group multicenter study |
| I (>50%) |
| Thal et al., 2003 [60] | Five hundred and thirty-six patients diagnosed with probable AD aged over 50 with MMSE scores between 12 and 25 were prescribed idebenone 120, 240, or 360 mg, or placebo (n = 136, 138, 133, and 126, respectively) during 1 year. Evaluation included ADAS-Cog, CGIC (primary outcome measures), and measurements of ADL, Behavioral Pathology in Alzheimer’s Disease Rating Scale, and MMSE (secondary outcomes). | Multicenter, double-blind, placebo-controlled, randomized trial |
| I (>50%) |
| Voronkova and Meleshkov, 2009 [61] | Thirty-five patients were diagnosed with AD (n = 9), mixed dementia (n = 21), or memory impairment not reaching dementia (n = 5). Treatment with CoQ10 120 mg/day for 6 months. Assessment with the Luriya method (memory and especially auditory-speech memory), Clinical Dementia Rating scale (CDRS), CGIC, and MMSE. | Open-label study |
| II (NA) |
| Galasko et al., 2012 [62] | Seventy-eight patients with mild to moderate AD (66 of them provided serial CSF specimens adequate for biochemical analyses). Random assignment to treatment for 16 weeks with 800 IU/d of vitamin E (α-tocopherol) plus 500 mg/d of vitamin C plus 900 mg/d of α-lipoic acid (E/C/ALA); 400 mg of coenzyme Q10 3 times/day; or placebo (26 to each group; 24, 20, and 12, respectively, provided CSF). Evaluation at baseline and 16 weeks of MMSE and ADCS-ADL scale, and CSF biomarkers related to AD. | Monocenter, randomized, placebo-controlled, double-blind clinical trial |
| I (>50%) |
| Karakahya and Özcan, 2020 [63] | Sixty-two patients diagnosed with AD (31 randomized to the treatment group and 31 to the observational group), and 31 healthy controls. The treatment group received topical application of CoQ10 on the retina and choroids. Assessment of CMT, RNFL thickness, GCIPL thickness, and SCT with OCT at baseline and after 6 months. | Monocenter, randomized clinical trial |
| I (>50%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiménez-Jiménez, F.J.; Alonso-Navarro, H.; García-Martín, E.; Agúndez, J.A.G. Coenzyme Q10 and Dementia: A Systematic Review. Antioxidants 2023, 12, 533. https://doi.org/10.3390/antiox12020533
Jiménez-Jiménez FJ, Alonso-Navarro H, García-Martín E, Agúndez JAG. Coenzyme Q10 and Dementia: A Systematic Review. Antioxidants. 2023; 12(2):533. https://doi.org/10.3390/antiox12020533
Chicago/Turabian StyleJiménez-Jiménez, Félix Javier, Hortensia Alonso-Navarro, Elena García-Martín, and José A. G. Agúndez. 2023. "Coenzyme Q10 and Dementia: A Systematic Review" Antioxidants 12, no. 2: 533. https://doi.org/10.3390/antiox12020533
APA StyleJiménez-Jiménez, F. J., Alonso-Navarro, H., García-Martín, E., & Agúndez, J. A. G. (2023). Coenzyme Q10 and Dementia: A Systematic Review. Antioxidants, 12(2), 533. https://doi.org/10.3390/antiox12020533








