Effect of Resveratrol on Pregnancy, Prenatal Complications and Pregnancy-Associated Structure Alterations
Abstract
1. Introduction
2. Bioavailability, Toxicity, and Hormetic Effect of Resveratrol
3. Resveratrol’s Mechanism of Action during Pregnancy Complications
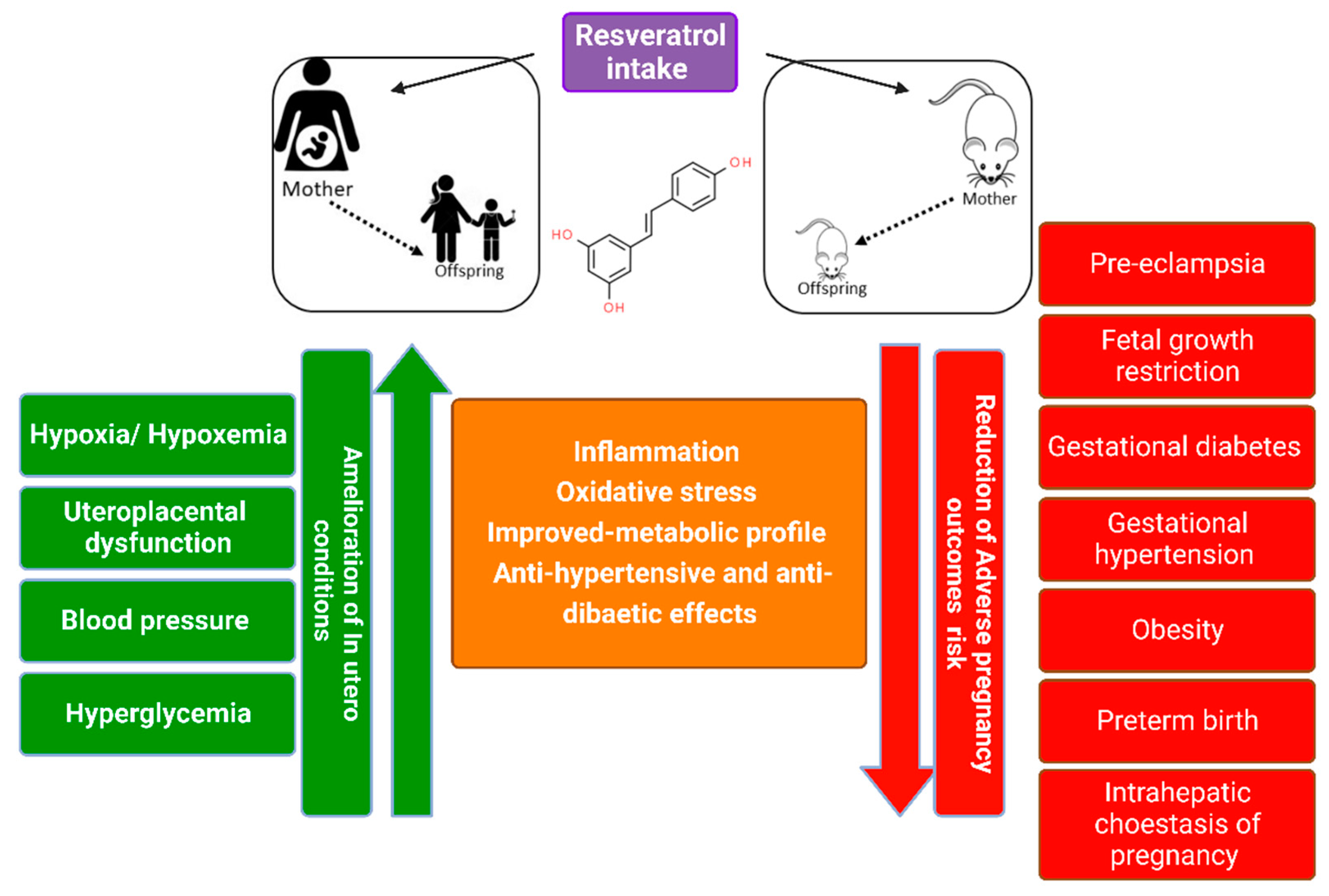
4. Maternal Pregnancy-Related Disorders
4.1. Resveratrol Effect in Pre-Eclampsia and Related Disorders
4.2. Resveratrol Effect in Gestational Diabetes and Related Metabolic Disorders
5. Fetus-Related Abnormalities and Resveratrol
5.1. Prenatal and Resveratrol
5.2. Placenta and Resveratrol
5.3. Preterm and Resveratrol
5.4. Fetus and Resveratrol
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jowell, A.R.; Sarma, A.A.; Gulati, M.; Michos, E.D.; Vaught, A.J.; Natarajan, P.; Powe, C.E.; Honigberg, M.C. Interventions to Mitigate Risk of Cardiovascular Disease After Adverse Pregnancy Outcomes: A Review. JAMA Cardiol. 2022, 7, 346–355. [Google Scholar] [CrossRef]
- Parikh, N.I.; Gonzalez, J.M.; Anderson, C.A.M.; Judd, S.E.; Rexrode, K.M.; Hlatky, M.A.; Gunderson, E.P.; Stuart, J.J.; Vaidya, D.; On behalf of the American Heart Association Council on Epidemiology and Prevention; et al. Adverse Pregnancy Outcomes and Cardiovascular Disease Risk: Unique Opportunities for Cardiovascular Disease Prevention in Women: A Scientific Statement From the American Heart Association. Circulation 2021, 143, e902–e916. [Google Scholar] [CrossRef]
- Darby, J.R.T.; Dollah, M.H.B.M.; Regnault, T.R.H.; Williams, M.T.; Morrison, J.L. Systematic review: Impact of resveratrol exposure during pregnancy on maternal and fetal outcomes in animal models of human pregnancy complications-Are we ready for the clinic? Pharmacol. Res. 2019, 144, 264–278. [Google Scholar] [CrossRef]
- Phu, H.T.; Thuan, D.T.; Nguyen, T.H.; Posadino, A.M.; Eid, A.H.; Pintus, G. Herbal medicine for slowing aging and aging-associated conditions: Efficacy, mechanisms and safety. Curr. Vasc. Pharmacol. 2020, 18, 369–393. [Google Scholar] [CrossRef]
- Hossain, R.; Quispe, C.; Khan, R.A.; Saikat, A.S.M.; Ray, P.; Ongalbek, D.; Yeskaliyeva, B.; Jain, D.; Smeriglio, A.; Trombetta, D. Propolis: An update on its chemistry and pharmacological applications. Chin. Med. 2022, 17, 100. [Google Scholar]
- Shaito, A.; Thuan, D.T.B.; Phu, H.T.; Nguyen, T.H.D.; Hasan, H.; Halabi, S.; Abdelhady, S.; Nasrallah, G.K.; Eid, A.H.; Pintus, G. Herbal medicine for cardiovascular diseases: Efficacy, mechanisms, and safety. Front. Pharmacol. 2020, 11, 422. [Google Scholar] [CrossRef]
- Maaliki, D.; Shaito, A.A.; Pintus, G.; El-Yazbi, A.; Eid, A.H. Flavonoids in hypertension: A brief review of the underlying mechanisms. Curr. Opin. Pharmacol. 2019, 45, 57–65. [Google Scholar] [CrossRef]
- Shaito, A.; Posadino, A.M.; Younes, N.; Hasan, H.; Halabi, S.; Alhababi, D.; Al-Mohannadi, A.; Abdel-Rahman, W.M.; Eid, A.H.; Nasrallah, G.K. Potential adverse effects of resveratrol: A literature review. Int. J. Mol. Sci. 2020, 21, 2084. [Google Scholar] [CrossRef]
- Giordo, R.; Cossu, A.; Porcu, M.C.; Cappuccinelli, R.; Biosa, G.; Sharifi-Rad, J.; Pretti, L.; Nasrallah, G.K.; Pintus, G.; Posadino, A.M. Cytoprotective, antioxidant, and anti-migratory activity of Pistacia lentiscus L. supercritical carbon dioxide extract on primary human endothelial cells. Nat. Prod. Res. 2022, 1–7. [Google Scholar] [CrossRef]
- Ramli, I.; Posadino, A.M.; Zerizer, S.; Spissu, Y.; Barberis, A.; Djeghim, H.; Azara, E.; Bensouici, C.; Kabouche, Z.; Rebbas, K. Low concentrations of Ambrosia maritima L. phenolic extract protect endothelial cells from oxidative cell death induced by H2O2 and sera from Crohn’s disease patients. J. Ethnopharmacol. 2023, 300, 115722. [Google Scholar] [CrossRef]
- Posadino, A.M.; Cossu, A.; Giordo, R.; Piscopo, A.; Abdel-Rahman, W.M.; Piga, A.; Pintus, G. Antioxidant properties of olive mill wastewater polyphenolic extracts on human endothelial and vascular smooth muscle cells. Foods 2021, 10, 800. [Google Scholar] [CrossRef] [PubMed]
- Posadino, A.M.; Phu, H.T.; Cossu, A.; Giordo, R.; Fois, M.; Thuan, D.T.B.; Piga, A.; Sotgia, S.; Zinellu, A.; Carru, C. Oxidative stress-induced Akt downregulation mediates green tea toxicity towards prostate cancer cells. Toxicol. Vitr. 2017, 42, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Giordo, R.; Zinellu, A.; Eid, A.H.; Pintus, G. Therapeutic potential of resveratrol in COVID-19-associated hemostatic disorders. Molecules 2021, 26, 856. [Google Scholar] [CrossRef] [PubMed]
- Koushki, M.; Amiri-Dashatan, N.; Ahmadi, N.; Abbaszadeh, H.A.; Rezaei-Tavirani, M. Resveratrol: A miraculous natural compound for diseases treatment. Food Sci. Nutr. 2018, 6, 2473–2490. [Google Scholar] [CrossRef]
- Posadino, A.M.; Giordo, R.; Cossu, A.; Nasrallah, G.K.; Shaito, A.; Abou-Saleh, H.; Eid, A.H.; Pintus, G. Flavin oxidase-induced ROS generation modulates PKC biphasic effect of resveratrol on endothelial cell survival. Biomolecules 2019, 9, 209. [Google Scholar] [CrossRef]
- Giordo, R.; Wehbe, Z.; Posadino, A.M.; Erre, G.L.; Eid, A.H.; Mangoni, A.A.; Pintus, G. Disease-associated regulation of non-coding RNAs by resveratrol: Molecular insights and therapeutic applications. Front. Cell Dev. Biol. 2022, 10, 894305. [Google Scholar] [CrossRef]
- Giordo, R.; Nasrallah, G.K.; Posadino, A.M.; Galimi, F.; Capobianco, G.; Eid, A.H.; Pintus, G. Resveratrol-elicited pkc inhibition counteracts nox-mediated endothelial to mesenchymal transition in human retinal endothelial cells exposed to high glucose. Antioxidants 2021, 10, 224. [Google Scholar] [CrossRef]
- Shaito, A.; Al-Mansoob, M.; Ahmad, S.M.; Haider, M.Z.; Eid, A.H.; Posadino, A.M.; Pintus, G.; Giordo, R. Resveratrol-mediated regulation of mitochondria biogenesis-associated pathways in neurodegenerative diseases: Molecular insights and potential therapeutic applications. Curr. Neuropharmacol. 2022. [Google Scholar] [CrossRef]
- Zou, T.; Chen, D.; Yang, Q.; Wang, B.; Zhu, M.-J.; Nathanielsz, P.W.; Du, M. Resveratrol supplementation of high-fat diet-fed pregnant mice promotes brown and beige adipocyte development and prevents obesity in male offspring. J. Physiol. 2017, 595, 1547–1562. [Google Scholar] [CrossRef]
- Lacerda, D.C.; Costa, P.C.T.; de Oliveira, Y.; de Brito Alves, J.L. The effect of resveratrol in cardio-metabolic disorders during pregnancy and offspring outcomes: A review. J. Dev. Orig. Health Dis. 2022, 14, 3–14. [Google Scholar] [CrossRef]
- Cottart, C.-H.; Nivet-Antoine, V.; Laguillier-Morizot, C.; Beaudeux, J.-L. Resveratrol bioavailability and toxicity in humans. Mol. Nutr. Food Res. 2010, 54, 7–16. [Google Scholar] [CrossRef]
- Singh, C.K.; Kumar, A.; LaVoie, H.A.; DiPette, D.J.; Singh, U.S. Diabetic complications in pregnancy: Is resveratrol a solution? Exp. Biol. Med. 2013, 238, 482–490. [Google Scholar] [CrossRef]
- Ding, J.; Kang, Y.; Fan, Y.; Chen, Q. Efficacy of resveratrol to supplement oral nifedipine treatment in pregnancy-induced preeclampsia. Endocr. Connect. 2017, 6, 595–600. [Google Scholar] [CrossRef]
- Böttner, M.; Christoffel, J.; Jarry, H.; Wuttke, W. Effects of long-term treatment with resveratrol and subcutaneous and oral estradiol administration on pituitary function in rats. J. Endocrinol. 2006, 189, 77–88. [Google Scholar] [CrossRef]
- Sale, S.; Verschoyle, R.D.; Boocock, D.; Jones, D.; Wilsher, N.; Ruparelia, K.C.; Potter, G.; Farmer, P.B.; Steward, W.P.; Gescher, A.J. Pharmacokinetics in mice and growth-inhibitory properties of the putative cancer chemopreventive agent resveratrol and the synthetic analogue trans 3, 4, 5, 4′-tetramethoxystilbene. Br. J. Cancer 2004, 90, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Bertelli, A.; Giovannini, L.; Stradi, R.; Bertelli, A.; Tillement, J. Plasma, urine and tissue levels of trans-and cis-resveratrol (3, 4′, 5-trihydroxystilbene) after short-term or prolonged administration of red wine to rats. Int. J. Tissue React. 1996, 18, 67–71. [Google Scholar]
- Vitrac, X.; Desmouliere, A.; Brouillaud, B.; Krisa, S.; Deffieux, G.; Barthe, N.; Rosenbaum, J.; Mérillon, J.-M. Distribution of [14C]-trans-resveratrol, a cancer chemopreventive polyphenol, in mouse tissues after oral administration. Life Sci. 2003, 72, 2219–2233. [Google Scholar] [CrossRef]
- Bourque, S.L.; Dolinsky, V.W.; Dyck, J.R.B.; Davidge, S.T. Maternal resveratrol treatment during pregnancy improves adverse fetal outcomes in a rat model of severe hypoxia. Placenta 2012, 33, 449–452. [Google Scholar] [CrossRef] [PubMed]
- Park, E.-J.; Pezzuto, J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2015, 1852, 1071–1113. [Google Scholar] [CrossRef]
- Wenzel, E.; Somoza, V. Metabolism and bioavailability of trans-resveratrol. Mol. Nutr. Food Res. 2005, 49, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Walle, T. Bioavailability of resveratrol. Ann. N. Y. Acad. Sci. 2011, 1215, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, E.; Soldo, T.; Erbersdobler, H.; Somoza, V. Bioactivity and metabolism of trans-resveratrol orally administered to Wistar rats. Mol. Nutr. Food Res. 2005, 49, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Kemper, C.; Behnam, D.; Brothers, S.; Wahlestedt, C.; Volmar, C.-H.; Bennett, D.; Hayward, M. Safety and pharmacokinetics of a highly bioavailable resveratrol preparation (JOTROL TM). AAPS Open 2022, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Howells, L.M.; Berry, D.P.; Elliott, P.J.; Jacobson, E.W.; Hoffmann, E.; Hegarty, B.; Brown, K.; Steward, W.; Gescher, A.J. Phase I randomized, double-blind pilot study of micronized resveratrol (SRT501) in patients with hepatic metastases—Safety, pharmacokinetics, and pharmacodynamics. Cancer Prev. Res. 2011, 4, 1419–1425. [Google Scholar] [CrossRef]
- Poulsen, M.M.; Vestergaard, P.F.; Clasen, B.F.; Radko, Y.; Christensen, L.P.; Stødkilde-Jørgensen, H.; Møller, N.; Jessen, N.; Pedersen, S.B.; Jørgensen, J.O.L. High-dose resveratrol supplementation in obese men: An investigator-initiated, randomized, placebo-controlled clinical trial of substrate metabolism, insulin sensitivity, and body composition. Diabetes 2013, 62, 1186–1195. [Google Scholar] [CrossRef]
- Moon, R.T.; Kohn, A.D.; Ferrari, G.V.D.; Kaykas, A. WNT and β-catenin signalling: Diseases and therapies. Nat. Rev. Genet. 2004, 5, 691–701. [Google Scholar] [CrossRef]
- Detampel, P.; Beck, M.; Krähenbühl, S.; Huwyler, J. Drug interaction potential of resveratrol. Drug Metab. Rev. 2012, 44, 253–265. [Google Scholar] [CrossRef]
- Ramírez-Garza, S.L.; Laveriano-Santos, E.P.; Marhuenda-Muñoz, M.; Storniolo, C.E.; Tresserra-Rimbau, A.; Vallverdú-Queralt, A.; Lamuela-Raventós, R.M. Health effects of resveratrol: Results from human intervention trials. Nutrients 2018, 10, 1892. [Google Scholar] [CrossRef]
- Roberts, V.H.; Pound, L.D.; Thorn, S.R.; Gillingham, M.B.; Thornburg, K.L.; Friedman, J.E.; Frias, A.E.; Grove, K.L. Beneficial and cautionary outcomes of resveratrol supplementation in pregnant nonhuman primates. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2014, 28, 2466–2477. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Mattson, M.P.; Calabrese, V. Resveratrol commonly displays hormesis: Occurrence and biomedical significance. Hum. Exp. Toxicol. 2010, 29, 980–1015. [Google Scholar] [CrossRef] [PubMed]
- Rocha, K.; Souza, G.; Ebaid, G.X.; Seiva, F.; Cataneo, A.; Novelli, E. Resveratrol toxicity: Effects on risk factors for atherosclerosis and hepatic oxidative stress in standard and high-fat diets. Food Chem. Toxicol. 2009, 47, 1362–1367. [Google Scholar] [CrossRef] [PubMed]
- Moraloglu, O.; Engin-Ustun, Y.; Tonguç, E.; Var, T.; Tapisiz, Ö.L.; Ergün, H.; Guvenc, T.; Gacar, A. The effect of resveratrol on blood pressure in a rat model of preeclampsia. J. Matern.-Fetal Neonatal Med. 2012, 25, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Quon, A.; Morton, J.S.; Davidge, S.T. Postnatal resveratrol supplementation improves cardiovascular function in male and female intrauterine growth restricted offspring. Physiol. Rep. 2017, 5, e13109. [Google Scholar] [CrossRef] [PubMed]
- Malvasi, A.; Kosmas, I.; Mynbaev, O.A.; Sparic, R.; Gustapane, S.; Guido, M.; Tinelli, A. Can trans resveratrol plus d-chiro-inositol and myo-inositol improve maternal metabolic profile in overweight pregnant patients? Clin. Ter. 2017, 168, e240–e247. [Google Scholar] [CrossRef] [PubMed]
- Costa-Silva, J.H.; Simões-Alves, A.C.; Fernandes, M.P. Developmental Origins of Cardiometabolic Diseases: Role of the Maternal Diet. Front. Physiol. 2016, 7, 504. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, A.S.; de Albuquerque, T.M.R.; de Brito Alves, J.L.; de Souza, E.L. Effects of Quercetin and Resveratrol on in vitro Properties Related to the Functionality of Potentially Probiotic Lactobacillus Strains. Front. Microbiol. 2019, 10, 2229. [Google Scholar] [CrossRef]
- Badran, A.; Nasser, S.A.; Mesmar, J.; El-Yazbi, A.F.; Bitto, A.; Fardoun, M.M.; Baydoun, E.; Eid, A.H. Reactive Oxygen Species: Modulators of Phenotypic Switch of Vascular Smooth Muscle Cells. Int. J. Mol. Sci. 2020, 21, 8764. [Google Scholar] [CrossRef] [PubMed]
- Shaito, A.; Aramouni, K.; Assaf, R.; Parenti, A.; Orekhov, A.; El Yazbi, A.; Pintus, G.; Eid, A.H. Oxidative Stress-Induced Endothelial Dysfunction in Cardiovascular Diseases. Front. Biosci.-Landmark 2022, 27, 105. [Google Scholar] [CrossRef] [PubMed]
- Mihu, D.; Sabau, L.; Costin, N.; Ciortea, R.; Malutan, A.; Mihu, C.M. Implications of maternal systemic oxidative stress in normal pregnancy and in pregnancy complicated by preeclampsia. J. Matern.-Fetal Neonatal Med. 2012, 25, 944–951. [Google Scholar] [CrossRef]
- Tan, C.; Wei, H.; Sun, H.; Ao, J.; Long, G.; Jiang, S.; Peng, J. Effects of Dietary Supplementation of Oregano Essential Oil to Sows on Oxidative Stress Status, Lactation Feed Intake of Sows, and Piglet Performance. BioMed Res. Int. 2015, 2015, 525218. [Google Scholar] [CrossRef]
- Pereira, A.C.; Martel, F. Oxidative stress in pregnancy and fertility pathologies. Cell Biol. Toxicol. 2014, 30, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Aranguren, L.C.; Prada, C.E.; Riano-Medina, C.E.; Lopez, M. Endothelial dysfunction and preeclampsia: Role of oxidative stress. Front. Physiol. 2014, 5, 372. [Google Scholar] [CrossRef] [PubMed]
- Vega, C.C.; Reyes-Castro, L.A.; Rodríguez-González, G.L.; Bautista, C.J.; Vázquez-Martínez, M.; Larrea, F.; Chamorro-Cevallos, G.A.; Nathanielsz, P.W.; Zambrano, E. Resveratrol partially prevents oxidative stress and metabolic dysfunction in pregnant rats fed a low protein diet and their offspring. J. Physiol. 2016, 594, 1483–1499. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.K.; Kumar, A.; Hitchcock, D.B.; Fan, D.; Goodwin, R.; LaVoie, H.A.; Nagarkatti, P.; DiPette, D.J.; Singh, U.S. Resveratrol prevents embryonic oxidative stress and apoptosis associated with diabetic embryopathy and improves glucose and lipid profile of diabetic dam. Mol. Nutr. Food Res. 2011, 55, 1186–1196. [Google Scholar] [CrossRef]
- Trejo-González, N.L.; Chirino-Galindo, G.; Palomar-Morales, M. Capacidad antiteratogénica del resveratrol en diabetes inducida por estreptozotocina en ratas. Rev. Peru. Med. Exp. Salud Pública 2015, 32, 457–463. [Google Scholar] [CrossRef]
- Wang, L.; Tang, J.; Wang, L.; Tan, F.; Song, H.; Zhou, J.; Li, F. Oxidative stress in oocyte aging and female reproduction. J. Cell. Physiol. 2021, 236, 7966–7983. [Google Scholar] [CrossRef] [PubMed]
- Homer, H.A. The role of oocyte quality in explaining “unexplained” infertility. Semin. Reprod. Med. 2020, 38, 021–028. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhang, M.; Jiang, Z.; Seli, E. Mitochondrial dysfunction and ovarian aging. Am. J. Reprod. Immunol. 2017, 77, e12651. [Google Scholar] [CrossRef]
- Liang, Q.-X.; Lin, Y.-H.; Zhang, C.-H.; Sun, H.-M.; Zhou, L.; Schatten, H.; Sun, Q.-Y.; Qian, W.-P. Resveratrol increases resistance of mouse oocytes to postovulatory aging in vivo. Aging 2018, 10, 1586–1596. [Google Scholar] [CrossRef] [PubMed]
- Takeo, S.; Sato, D.; Kimura, K.; Monji, Y.; Kuwayama, T.; Kawahara-Miki, R.; Iwata, H. Resveratrol Improves the Mitochondrial Function and Fertilization Outcome of Bovine Oocytes. J. Reprod. Dev. 2014, 60, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.-J.; Sun, A.-G.; Zhao, S.-G.; Liu, H.; Ma, S.-Y.; Li, M.; Huai, Y.-X.; Zhao, H.; Liu, H.-B. Resveratrol improves in vitro maturation of oocytes in aged mice and humans. Fertil. Steril. 2018, 109, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.; Juhn, K.-M.; Jung, E.-H.; Park, H.-J.; Yoon, S.-H.; Ko, Y.; Hur, C.-Y.; Lim, J.-H. Effects of resveratrol, granulocyte-macrophage colony-stimulating factor or dichloroacetic acid in the culture media on embryonic development and pregnancy rates in aged mice. Aging 2020, 12, 2659–2669. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Yin, Y.; Ye, X.; Zeng, M.; Zhao, Q.; Keefe, D.L.; Liu, L. Resveratrol protects against age-associated infertility in mice. Hum. Reprod. 2013, 28, 707–717. [Google Scholar] [CrossRef]
- Igarashi, H.; Takahashi, T.; Abe, H.; Nakano, H.; Nakajima, O.; Nagase, S. Poor embryo development in post-ovulatory in vivo-aged mouse oocytes is associated with mitochondrial dysfunction, but mitochondrial transfer from somatic cells is not sufficient for rejuvenation. Hum. Reprod. 2016, 31, 2331–2338. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chian, R.-C. Mitochondrial Dysfunction and Age-related Oocyte Quality. Reprod. Dev. Med. 2017, 1, 45–54. [Google Scholar] [CrossRef]
- Zheng, S.; Feng, Q.; Cheng, J.; Zheng, J. Maternal resveratrol consumption and its programming effects on metabolic health in offspring mechanisms and potential implications. Biosci. Rep. 2018, 38, BSR20171741. [Google Scholar] [CrossRef]
- Park, E.-Y.; Park, J.-B. High glucose-induced oxidative stress promotes autophagy through mitochondrial damage in rat notochordal cells. Int. Orthop. 2013, 37, 2507–2514. [Google Scholar] [CrossRef]
- Sebori, R.; Kuno, A.; Hosoda, R.; Hayashi, T.; Horio, Y. Resveratrol Decreases Oxidative Stress by Restoring Mitophagy and Improves the Pathophysiology of Dystrophin-Deficient mdx Mice. Oxidative Med. Cell. Longev. 2018, 2018, 9179270. [Google Scholar] [CrossRef]
- Ding, W.-X.; Yin, X.-M. Mitophagy: Mechanisms, pathophysiological roles, and analysis. Biol. Chem. 2012, 393, 547–564. [Google Scholar] [CrossRef]
- Zha, C.; Xiao, H.; Song, B.; Zheng, C.; Yang, X.; Wang, W.; Wang, L. Resveratrol promotes mammary cell proliferation and antioxidation capacity during pregnancy and lactation in mice. J. Appl. Microbiol. 2021, 130, 450–463. [Google Scholar] [CrossRef]
- Phoswa, W.N.; Khaliq, O.P. The Role of Oxidative Stress in Hypertensive Disorders of Pregnancy (Preeclampsia, Gestational Hypertension) and Metabolic Disorder of Pregnancy (Gestational Diabetes Mellitus). Oxidative Med. Cell. Longev. 2021, 2021, 5581570. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, R.; González, J.; Paoletto, F. The role of oxidative stress in the pathophysiology of hypertension. Hypertens. Res. 2011, 34, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Magee, L.A.; Kenny, L.; Ananth Karumanchi, S.; McCarthy, F.; Saito, S.; Hall, D.R.; Warren, C.E.; Adoyi, G.; Mohammed, S.I. TEMPORARY REMOVAL: The hypertensive disorders of pregnancy: ISSHP classification, diagnosis and management recommendations for international practice 2018. Pregnancy Hypertens. 2018, 72, 24–43. [Google Scholar] [CrossRef]
- Agarwal, P.; Morriseau, T.S.; Kereliuk, S.M.; Doucette, C.A.; Wicklow, B.A.; Dolinsky, V.W. Maternal obesity, diabetes during pregnancy and epigenetic mechanisms that influence the developmental origins of cardiometabolic disease in the offspring. Crit. Rev. Clin. Lab. Sci. 2018, 55, 71–101. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Auwerx, J. The Role of Sirtuins in the Control of Metabolic Homeostasis. Ann. N. Y. Acad. Sci. 2009, 1173, E10–E19. [Google Scholar] [CrossRef]
- Yu, J.; Auwerx, J. Protein deacetylation by SIRT1: An emerging key post-translational modification in metabolic regulation. Pharmacol. Res. 2010, 62, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-W.; Yu, H.-R.; Sheen, J.-M.; Tiao, M.-M.; Tain, Y.-L.; Lin, I.C.; Lin, Y.-J.; Chang, K.-A.; Tsai, C.-C.; Huang, L.-T. A maternal high-fat diet during pregnancy and lactation, in addition to a postnatal high-fat diet, leads to metabolic syndrome with spatial learning and memory deficits: Beneficial effects of resveratrol. Oncotarget 2017, 8, 111998. [Google Scholar] [CrossRef]
- Burton, G.J.; Jauniaux, E. Oxidative stress. Best Pract. Res. Clin. Obstet. Gynaecol. 2011, 25, 287–299. [Google Scholar] [CrossRef]
- Lambert, G.; Brichant, J.F.; Hartstein, G.; Bonhomme, V.; Dewandre, P.Y. Preeclampsia: An update. Acta Anaesthesiol. Belg. 2014, 65, 137–149. [Google Scholar]
- Sibai, B.; Dekker, G.; Kupferminc, M. Pre-eclampsia. Lancet 2005, 365, 785–799. [Google Scholar] [CrossRef]
- Pishko, A.M.; Levine, L.D.; Cines, D.B. Thrombocytopenia in pregnancy: Diagnosis and approach to management. Blood Rev. 2020, 40, 100638. [Google Scholar] [CrossRef] [PubMed]
- Silvestro, S.; Calcaterra, V.; Pelizzo, G.; Bramanti, P.; Mazzon, E. Prenatal Hypoxia and Placental Oxidative Stress: Insights from Animal Models to Clinical Evidences. Antioxidants 2020, 9, 414. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.-Q.; Zhang, L. Hypoxia and Mitochondrial Dysfunction in Pregnancy Complications. Antioxidants 2021, 10, 405. [Google Scholar] [CrossRef] [PubMed]
- Dolinsky, V.W.; Rueda-Clausen, C.F.; Morton, J.S.; Davidge, S.T.; Dyck, J.R. Continued postnatal administration of resveratrol prevents diet-induced metabolic syndrome in rat offspring born growth restricted. Diabetes 2011, 60, 2274–2284. [Google Scholar] [CrossRef] [PubMed]
- Baumwell, S.; Karumanchi, S.A. Pre-Eclampsia: Clinical Manifestations and Molecular Mechanisms. Nephron Clin. Pract. 2007, 106, c72–c81. [Google Scholar] [CrossRef] [PubMed]
- Rosser, M.L.; Katz, N.T. Preeclampsia: An Obstetrician’s Perspective. Adv. Chronic Kidney Dis. 2013, 20, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Eiland, E.; Nzerue, C.; Faulkner, M. Preeclampsia 2012. J. Pregnancy 2012, 2012, 586578. [Google Scholar] [CrossRef]
- Hansson, S.R.; Nääv, Å.; Erlandsson, L. Oxidative stress in preeclampsia and the role of free fetal hemoglobin. Front. Physiol. 2015, 5, 516. [Google Scholar] [CrossRef]
- Sagrillo-Fagundes, L.; Laurent, L.; Bienvenue-Pariseault, J.; Vaillancourt, C. In Vitro Induction of Hypoxia/Reoxygenation on Placental Cells: A Suitable Model for Understanding Placental Diseases. In Preeclampsia: Methods and Protocols; Murthi, P., Vaillancourt, C., Eds.; Springer: New York, NY, USA, 2018; pp. 277–283. [Google Scholar]
- Aydın, S.; Benian, A.; Madazli, R.; Uludaǧ, S.; Uzun, H.; Kaya, S. Plasma malondialdehyde, superoxide dismutase, sE-selectin, fibronectin, endothelin-1 and nitric oxide levels in women with preeclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2004, 113, 21–25. [Google Scholar] [CrossRef]
- Gupta, S.; Agarwal, A.; Sharma, R.K. The Role of Placental Oxidative Stress and Lipid Peroxidation in Preeclampsia. Obstet. Gynecol. Surv. 2005, 60, 807–816. [Google Scholar] [CrossRef]
- Parsamanesh, N.; Asghari, A.; Sardari, S.; Tasbandi, A.; Jamialahmadi, T.; Xu, S.; Sahebkar, A. Resveratrol and endothelial function: A literature review. Pharmacol. Res. 2021, 170, 105725. [Google Scholar] [CrossRef] [PubMed]
- Mann, G.E.; Niehueser-Saran, J.; Watson, A.; Gao, L.; Ishii, T.; de Winter, P.; Siow, R.C. Nrf2/ARE regulated antioxidant gene expression in endothelial and smooth muscle cells in oxidative stress: Implications for atherosclerosis and preeclampsia. Acta Physiol. Sin.-Chin. Ed. 2007, 59, 117. [Google Scholar]
- Caldeira-Dias, M.; Viana-Mattioli, S.; de Souza Rangel Machado, J.; Carlström, M.; de Carvalho Cavalli, R.; Sandrim, V.C. Resveratrol and grape juice: Effects on redox status and nitric oxide production of endothelial cells in in vitro preeclampsia model. Pregnancy Hypertens. 2021, 23, 205–210. [Google Scholar] [CrossRef]
- Poudel, R.; Stanley, J.L.; Rueda-Clausen, C.F.; Andersson, I.J.; Sibley, C.P.; Davidge, S.T.; Baker, P.N. Effects of Resveratrol in Pregnancy Using Murine Models with Reduced Blood Supply to the Uterus. PLoS ONE 2013, 8, e64401. [Google Scholar] [CrossRef]
- Viana-Mattioli, S.; Cinegaglia, N.; Bertozzi-Matheus, M.; Bueno-Pereira, T.O.; Caldeira-Dias, M.; Cavalli, R.C.; Sandrim, V.C. SIRT1-dependent effects of resveratrol and grape juice in an in vitro model of preeclampsia. Biomed. Pharmacother. 2020, 131, 110659. [Google Scholar] [CrossRef] [PubMed]
- Khong, T.Y.; De Wolf, F.; Robertson, W.B.; Brosens, I. Inadequate maternal vascular response to placentation in pregnancies complicated by pre-eclampsia and by small-for-gestational age infants. BJOG Int. J. Obstet. Gynaecol. 1986, 93, 1049–1059. [Google Scholar] [CrossRef]
- Labarrere, C.A.; Althabe, O.H. Inadequate maternal vascular response to placentation in pregnancies complicated by preeclampsia and by small-for-gestational-age infants. BJOG Int. J. Obstet. Gynaecol. 1987, 94, 1113–1115. [Google Scholar] [CrossRef] [PubMed]
- Friedl, P.; Wolf, K. Tumour-cell invasion and migration: Diversity and escape mechanisms. Nat. Rev. Cancer 2003, 3, 362–374. [Google Scholar] [CrossRef]
- Mao, X.-W.; Xiao, J.-Q.; Li, Z.-Y.; Zheng, Y.-C.; Zhang, N. Effects of microRNA-135a on the epithelial–mesenchymal transition, migration and invasion of bladder cancer cells by targeting GSK3β through the Wnt/β-catenin signaling pathway. Exp. Mol. Med. 2018, 50, e429. [Google Scholar] [CrossRef]
- Kokkinos, M.I.; Murthi, P.; Wafai, R.; Thompson, E.W.; Newgreen, D.F. Cadherins in the human placenta—Epithelial–mesenchymal transition (EMT) and placental development. Placenta 2010, 31, 747–755. [Google Scholar] [CrossRef]
- Zou, Y.; Li, S.; Wu, D.; Xu, Y.; Wang, S.; Jiang, Y.; Liu, F.; Jiang, Z.; Qu, H.; Yu, X.; et al. Resveratrol promotes trophoblast invasion in pre-eclampsia by inducing epithelial-mesenchymal transition. J. Cell. Mol. Med. 2019, 23, 2702–2710. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, C.; Pei, J.; Li, M.; Gu, W. SIRT1: A Novel Protective Molecule in Pre-eclampsia. Int. J. Med. Sci. 2022, 19, 993–1002. [Google Scholar] [CrossRef]
- Romero, R.; Chaiworapongsa, T. Preeclampsia: A link between trophoblast dysregulation and an antiangiogenic state. J. Clin. Investig. 2013, 123, 2775–2777. [Google Scholar] [CrossRef] [PubMed]
- Phipps, E.A.; Thadhani, R.; Benzing, T.; Karumanchi, S.A. Pre-eclampsia: Pathogenesis, novel diagnostics and therapies. Nat. Rev. Nephrol. 2019, 15, 275–289. [Google Scholar] [CrossRef]
- Venkatesha, S.; Toporsian, M.; Lam, C.; Hanai, J.-i.; Mammoto, T.; Kim, Y.M.; Bdolah, Y.; Lim, K.-H.; Yuan, H.-T.; Libermann, T.A.; et al. Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat. Med. 2006, 12, 642–649. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Nien, J.K.; Espinoza, J.; Todem, D.; Fu, W.; Chung, H.; Kusanovic, J.P.; Gotsch, F.; Erez, O.; Mazaki-Tovi, S.; et al. A longitudinal study of angiogenic (placental growth factor) and anti-angiogenic (soluble endoglin and soluble vascular endothelial growth factor receptor-1) factors in normal pregnancy and patients destined to develop preeclampsia and deliver a small for gestational age neonate. J. Matern.-Fetal Neonatal Med. 2008, 21, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Hannan, N.J.; Brownfoot, F.C.; Cannon, P.; Deo, M.; Beard, S.; Nguyen, T.V.; Palmer, K.R.; Tong, S.; Kaitu’u-Lino, T.u.J. Resveratrol inhibits release of soluble fms-like tyrosine kinase (sFlt-1) and soluble endoglin and improves vascular dysfunction—Implications as a preeclampsia treatment. Sci. Rep. 2017, 7, 1819. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Zuo, Q.; Huang, S.; Yu, X.; Jiang, Z.; Zou, S.; Fan, M.; Sun, L. Resveratrol Inhibits Trophoblast Apoptosis through Oxidative Stress in Preeclampsia-Model Rats. Molecules 2014, 19, 20570–20579. [Google Scholar] [CrossRef]
- Jiao, X.C.; Xiao, M.; Gao, Z.X.; Xie, J.; Liu, Y.; Yin, M.J.; Wu, Y.; Tao, R.X.; Zhu, P. Effects of comorbid gestational diabetes mellitus and depression on glucose metabolism during pregnancy and neonatal morphological outcome. Zhonghua Yu Fang Yi Xue Za Zhi 2020, 54, 968–973. [Google Scholar] [CrossRef]
- Kim, J.; Piao, Y.; Pak, Y.K.; Chung, D.; Han, Y.M.; Hong, J.S.; Jun, E.J.; Shim, J.-Y.; Choi, J.; Kim, C.J. Umbilical Cord Mesenchymal Stromal Cells Affected by Gestational Diabetes Mellitus Display Premature Aging and Mitochondrial Dysfunction. Stem Cells Dev. 2014, 24, 575–586. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.-J.; Yoon, C.-H.; Lee, H.; Ahn, S.-Y.; Oh, K.J.; Park, H.-Y.; Lee, H.Y.; Cho, M.C.; Chung, I.-M.; Shin, M.-S.; et al. The Association of Family History of Premature Cardiovascular Disease or Diabetes Mellitus on the Occurrence of Gestational Hypertensive Disease and Diabetes. PLoS ONE 2016, 11, e0167528. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-M.; Gao, Y.; Eriksson, J.G.; Chen, W.; Chong, Y.S.; Tan, K.H.; Zhang, C.; Zhou, L.; Li, L.-J. Metabolic signatures in the conversion from gestational diabetes mellitus to postpartum abnormal glucose metabolism: A pilot study in Asian women. Sci. Rep. 2021, 11, 16435. [Google Scholar] [CrossRef] [PubMed]
- Su, M.; Zhao, W.; Xu, S.; Weng, J. Resveratrol in Treating Diabetes and Its Cardiovascular Complications: A Review of Its Mechanisms of Action. Antioxidants 2022, 11, 1085. [Google Scholar] [CrossRef]
- Do, G.-M.; Jung, U.J.; Park, H.-J.; Kwon, E.-Y.; Jeon, S.-M.; McGregor, R.A.; Choi, M.-S. Resveratrol ameliorates diabetes-related metabolic changes via activation of AMP-activated protein kinase and its downstream targets in db/db mice. Mol. Nutr. Food Res. 2012, 56, 1282–1291. [Google Scholar] [CrossRef]
- Zhang, G.; Wang, X.; Ren, B.; Zhao, Q.; Zhang, F. The Effect of Resveratrol on Blood Glucose and Blood Lipids in Rats with Gestational Diabetes Mellitus. Evid.-Based Complement. Altern. Med. 2021, 2021, 2956795. [Google Scholar] [CrossRef] [PubMed]
- Du, S.; Lv, Y.; Li, N.; Huang, X.; Liu, X.; Li, H.; Wang, C.; Jia, Y.-F. Biological investigations on therapeutic effect of chitosan encapsulated nano resveratrol against gestational diabetes mellitus rats induced by streptozotocin. Drug Deliv. 2020, 27, 953–963. [Google Scholar] [CrossRef]
- Brawerman, G.M.; Kereliuk, S.M.; Brar, N.; Cole, L.K.; Seshadri, N.; Pereira, T.J.; Xiang, B.; Hunt, K.L.; Fonseca, M.A.; Hatch, G.M.; et al. Maternal resveratrol administration protects against gestational diabetes-induced glucose intolerance and islet dysfunction in the rat offspring. J. Physiol. 2019, 597, 4175–4192. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, Z.; Ke, L.; Li, Z.; Li, W.; Zhang, Z.; Zhou, Y.; Feng, X.; Zhu, W. Resveratrol improves glucose uptake in insulin-resistant adipocytes via Sirt1. J. Nutr. Biochem. 2018, 55, 209–218. [Google Scholar] [CrossRef]
- Yi, L.; Chen, C.Y.; Jin, X.; Mi, M.T.; Yu, B.; Chang, H.; Ling, W.H.; Zhang, T. Structural requirements of anthocyanins in relation to inhibition of endothelial injury induced by oxidized low-density lipoprotein and correlation with radical scavenging activity. Febs. Lett. 2010, 584, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Shu, L.; Zhao, H.A.-O.; Huang, W.; Hou, G.; Song, G.; Ma, H. Resveratrol Upregulates mmu-miR-363-3p via the PI3K-Akt Pathway to Improve Insulin Resistance Induced by a High-Fat Diet in Mice. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 391–403. [Google Scholar] [CrossRef]
- Ferrante, M.; Conti, O.G. Environment and Neurodegenerative Diseases: An Update on miRNA Role. MicroRNA 2017, 6, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Yadav, T.; Rani, V. Exploring miRNA based approaches in cancer diagnostics and therapeutics. Crit. Rev. Oncol./Hematol. 2016, 98, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, A.; Mukherjee, B.; Dixit, M. MicroRNA Key to Angiogenesis Regulation: MiRNA Biology and Therapy. Curr. Cancer Drug Targets 2018, 18, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Giordo, R.; Ahmed, Y.M.; Allam, H.; Abusnana, S.; Pappalardo, L.; Nasrallah, G.K.; Mangoni, A.A.; Pintus, G. EndMT regulation by small RNAs in diabetes-associated fibrotic conditions: Potential link with oxidative stress. Front. Cell Dev. Biol. 2021, 9, 683594. [Google Scholar] [CrossRef]
- Wehbe, N.; Nasser, S.A.; Pintus, G.; Badran, A.; Eid, A.H.; Baydoun, E. MicroRNAs in cardiac hypertrophy. Int. J. Mol. Sci. 2019, 20, 4714. [Google Scholar] [CrossRef]
- Lozano-Bartolomé, J.; Llauradó, G.; Portero-Otin, M.; Altuna-Coy, A.; Rojo-Martínez, G.; Vendrell, J.; Jorba, R.; Rodríguez-Gallego, E.; Chacón, M.R. Altered Expression of miR-181a-5p and miR-23a-3p Is Associated With Obesity and TNFα-Induced Insulin Resistance. J. Clin. Endocrinol. Metab. 2018, 103, 1447–1458. [Google Scholar] [CrossRef]
- Zheng, T.; Chen, H. Resveratrol ameliorates the glucose uptake and lipid metabolism in gestational diabetes mellitus mice and insulin-resistant adipocytes via miR-23a-3p/NOV axis. Mol. Immunol. 2021, 137, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Downs, D.S. Obesity in Special Populations: Pregnancy. Prim. Care Clin. Off. Pract. 2016, 43, 109–120. [Google Scholar] [CrossRef]
- Reichetzeder, C. Overweight and obesity in pregnancy: Their impact on epigenetics. Eur. J. Clin. Nutr. 2021, 75, 1710–1722. [Google Scholar] [CrossRef]
- Snelgrove-Clarke, E.; Macdonald, D.; Helwig, M.; Alsius, A. Women’s experiences of living with obesity during pregnancy, birthing, and postpartum: A qualitative systematic review protocol. JBI Evid. Synth. 2021, 19, 3183–3189. [Google Scholar] [CrossRef]
- Álvarez, D.; Muñoz, Y.; Ortiz, M.; Maliqueo, M.; Chouinard-Watkins, R.; Valenzuela, R. Impact of Maternal Obesity on the Metabolism and Bioavailability of Polyunsaturated Fatty Acids during Pregnancy and Breastfeeding. Nutrients 2021, 13, 19. [Google Scholar] [CrossRef] [PubMed]
- Mission, J.F.; Marshall, N.E.; Caughey, A.B. Pregnancy Risks Associated with Obesity. Obstet. Gynecol. Clin. North Am. 2015, 42, 335–353. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-González, G.L.; Vargas-Hernández, L.; Reyes-Castro, L.A.; Ibáñez, C.A.; Bautista, C.J.; Lomas-Soria, C.; Itani, N.; Estrada-Gutierrez, G.; Espejel-Nuñez, A.; Flores-Pliego, A.; et al. Resveratrol Supplementation in Obese Pregnant Rats Improves Maternal Metabolism and Prevents Increased Placental Oxidative Stress. Antioxidants 2022, 11, 1871. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.-R.; Sheen, J.-M.; Tiao, M.-M.; Tain, Y.-L.; Chen, C.-C.; Lin, I.C.; Lai, Y.-J.; Tsai, C.-C.; Lin, Y.-J.; Tsai, C.-C.; et al. Resveratrol Treatment Ameliorates Leptin Resistance and Adiposity Programed by the Combined Effect of Maternal and Post-Weaning High-Fat Diet. Mol. Nutr. Food Res. 2019, 63, 1801385. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Huang, L.-T.; Sheen, J.-M.; Hou, C.-Y.; Yeh, Y.-T.; Chiang, C.-P.; Lin, I.C.; Tiao, M.-M.; Tsai, C.-C.; Lin, Y.-J.; et al. Resveratrol treatment improves the altered metabolism and related dysbiosis of gut programed by prenatal high-fat diet and postnatal high-fat diet exposure. J. Nutr. Biochem. 2020, 75, 108260. [Google Scholar] [CrossRef]
- Ros, P.; Díaz, F.; Freire-Regatillo, A.; Argente-Arizón, P.; Barrios, V.; Argente, J.; Chowen, J.A. Resveratrol Intake During Pregnancy and Lactation Modulates the Early Metabolic Effects of Maternal Nutrition Differently in Male and Female Offspring. Endocrinology 2017, 159, 810–825. [Google Scholar] [CrossRef]
- Sun, S.; Meng, Q.; Luo, Z.; Shi, B.; Bi, C.; Shan, A. Effects of dietary resveratrol supplementation during gestation and lactation of sows on milk composition of sows and fat metabolism of sucking piglets. J. Anim. Physiol. Anim. Nutr. 2019, 103, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Liao, E.; Li, Z.; Shao, Y. Resveratrol regulates the silent information regulator 1–nuclear factor-κB signaling pathway in intrahepatic cholestasis of pregnancy. Hepatol. Res. 2018, 48, 1031–1044. [Google Scholar] [CrossRef]
- Chen, Z.; Hu, L.; Lu, M.; Shen, Z. Resveratrol reduces matrix metalloproteinases and alleviates intrahepatic cholestasis of pregnancy in rats. Can. J. Physiol. Pharmacol. 2015, 94, 402–407. [Google Scholar] [CrossRef]
- Herrera, E.; Ortega-Senovilla, H. Disturbances in lipid metabolism in diabetic pregnancy—Are these the cause of the problem? Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 515–525. [Google Scholar] [CrossRef]
- Cho, I.J.; Ahn, J.Y.; Kim, S.; Choi, M.S.; Ha, T.Y. Resveratrol attenuates the expression of HMG-CoA reductase mRNA in hamsters. Biochem. Biophys. Res. Commun. 2008, 367, 190–194. [Google Scholar] [CrossRef] [PubMed]
- Ofori, B.; Rey, E.; Bérard, A. Risk of congenital anomalies in pregnant users of statin drugs. Br. J. Clin. Pharmacol. 2007, 64, 496–509. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Wan, J.; Li, H.; Ding, J.; Wang, Y.; Wang, X.; Li, M. Resveratrol relieves gestational diabetes mellitus in mice through activating AMPK. Reprod. Biol. Endocrinol. 2015, 13, 118. [Google Scholar] [CrossRef] [PubMed]
- Bellentani, S. The epidemiology of non-alcoholic fatty liver disease. Liver Int. Off. J. Int. Assoc. Study Liver 2017, 37 (Suppl. S1), 81–84. [Google Scholar] [CrossRef]
- Seferovic, M.D.; Goodspeed, D.M.; Chu, D.M.; Krannich, L.A.; Gonzalez-Rodriguez, P.J.; Cox, J.E.; Aagaard, K.M. Heritable IUGR and adult metabolic syndrome are reversible and associated with alterations in the metabolome following dietary supplementation of 1-carbon intermediates. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2015, 29, 2640–2652. [Google Scholar] [CrossRef]
- Shen, L.; Liu, Z.; Gong, J.; Zhang, L.; Wang, L.; Magdalou, J.; Chen, L.; Wang, H. Prenatal ethanol exposure programs an increased susceptibility of non-alcoholic fatty liver disease in female adult offspring rats. Toxicol. Appl. Pharmacol. 2014, 274, 263–273. [Google Scholar] [CrossRef]
- Ding, R.B.; Bao, J.; Deng, C.X. Emerging roles of SIRT1 in fatty liver diseases. Int. J. Biol. Sci. 2017, 13, 852–867. [Google Scholar] [CrossRef]
- Shen, L.; Xie, L.; Chen, L.; Liu, L.; Shi, X.; Wang, X.; Chen, D.; Wang, H.; Quan, S.; Wang, Z. Resveratrol reverses the programmed high-susceptibility to non-alcoholic fatty liver disease by targeting the hepatic SIRT1-SREBP1c pathway in prenatal ethanol-exposed rat offspring. J. Toxicol. Sci. 2021, 46, 413–423. [Google Scholar] [CrossRef]
- Chen, H.E.; Lin, Y.J.; Lin, I.C.; Yu, H.R.; Sheen, J.M.; Tsai, C.C.; Huang, L.T.; Tain, Y.L. Resveratrol prevents combined prenatal N(G)-nitro-L-arginine-methyl ester (L-NAME) treatment plus postnatal high-fat diet induced programmed hypertension in adult rat offspring: Interplay between nutrient-sensing signals, oxidative stress and gut microbiota. J. Nutr. Biochem. 2019, 70, 28–37. [Google Scholar] [CrossRef]
- Giussani, D.A.; Davidge, S.T. Developmental programming of cardiovascular disease by prenatal hypoxia. J. Dev. Orig. Health Dis 2013, 4, 328–337. [Google Scholar] [CrossRef]
- Demicheva, E.; Crispi, F. Long-term follow-up of intrauterine growth restriction: Cardiovascular disorders. Fetal Diagn. Ther. 2014, 36, 143–153. [Google Scholar] [CrossRef]
- Barker, D.J. Fetal origins of coronary heart disease. BMJ 1995, 311, 171–174. [Google Scholar] [CrossRef]
- Bjarnegard, N.; Morsing, E.; Cinthio, M.; Lanne, T.; Brodszki, J. Cardiovascular function in adulthood following intrauterine growth restriction with abnormal fetal blood flow. Ultrasound Obstet. Gynecol. 2013, 41, 177–184. [Google Scholar] [CrossRef]
- Jaquet, D.; Gaboriau, A.; Czernichow, P.; Levy-Marchal, C. Insulin resistance early in adulthood in subjects born with intrauterine growth retardation. J. Clin. Endocrinol. Metab. 2000, 85, 1401–1406. [Google Scholar] [CrossRef]
- Rueda-Clausen, C.F.; Morton, J.S.; Davidge, S.T. Effects of hypoxia-induced intrauterine growth restriction on cardiopulmonary structure and function during adulthood. Cardiovasc. Res. 2009, 81, 713–722. [Google Scholar] [CrossRef]
- Xue, Q.; Zhang, L. Prenatal hypoxia causes a sex-dependent increase in heart susceptibility to ischemia and reperfusion injury in adult male offspring: Role of protein kinase C epsilon. J. Pharmacol. Exp. Ther. 2009, 330, 624–632. [Google Scholar] [CrossRef]
- Shah, A.; Reyes, L.M.; Morton, J.S.; Fung, D.; Schneider, J.; Davidge, S.T. Effect of resveratrol on metabolic and cardiovascular function in male and female adult offspring exposed to prenatal hypoxia and a high-fat diet. J. Physiol. 2016, 594, 1465–1482. [Google Scholar] [CrossRef]
- Madhyastha, S.; Sahu, S.S.; Rao, G. Resveratrol for prenatal-stress-induced oxidative damage in growing brain and its consequences on survival of neurons. J. Basic Clin. Physiol. Pharmacol. 2014, 25, 63–72. [Google Scholar] [CrossRef]
- Sahu, S.S.; Madhyastha, S.; Rao, G.M. Neuroprotective effect of resveratrol against prenatal stress induced cognitive impairment and possible involvement of Na+, K+-ATPase activity. Pharmacol. Biochem. Behav. 2013, 103, 520–525. [Google Scholar] [CrossRef]
- Abrahams, B.S.; Geschwind, D.H. Advances in autism genetics: On the threshold of a new neurobiology. Nat. Rev. Genet. 2008, 9, 341–355. [Google Scholar] [CrossRef]
- Strifert, K. An epigenetic basis for autism spectrum disorder risk and oral contraceptive use. Med. Hypotheses 2015, 85, 1006–1011. [Google Scholar] [CrossRef]
- Zou, Y.; Lu, Q.; Zheng, D.; Chu, Z.; Liu, Z.; Chen, H.; Ruan, Q.; Ge, X.; Zhang, Z.; Wang, X.; et al. Prenatal levonorgestrel exposure induces autism-like behavior in offspring through ERbeta suppression in the amygdala. Mol. Autism 2017, 8, 46. [Google Scholar] [CrossRef] [PubMed]
- Willing, J.; Wagner, C.K. Exposure to the Synthetic Progestin, 17alpha-Hydroxyprogesterone Caproate During Development Impairs Cognitive Flexibility in Adulthood. Endocrinology 2016, 157, 77–82. [Google Scholar] [CrossRef]
- Aguirre, C.; Jayaraman, A.; Pike, C.; Baudry, M. Progesterone inhibits estrogen-mediated neuroprotection against excitotoxicity by down-regulating estrogen receptor-beta. J. Neurochem. 2010, 115, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Ge, X.; Li, L.; Yao, A.; Wang, X.; Li, M.; Gong, X.; Chu, Z.; Lu, Z.; Huang, X.; et al. Resveratrol ameliorates prenatal progestin exposure-induced autism-like behavior through ERbeta activation. Mol. Autism 2018, 9, 43. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Guo, T.; Li, G.; Sun, S.; He, S.; Cheng, B.; Shi, B.; Shan, A. Dietary resveratrol improves antioxidant status of sows and piglets and regulates antioxidant gene expression in placenta by Keap1-Nrf2 pathway and Sirt1. J. Anim. Sci. Biotechnol. 2018, 9, 34. [Google Scholar] [CrossRef]
- Burton, G.J.; Jauniaux, E. What is the placenta? Am. J. Obstet. Gynecol. 2015, 213, S6.e1–S6.e4. [Google Scholar] [CrossRef]
- Wu, F.; Tian, F.J.; Lin, Y.; Xu, W.M. Oxidative Stress: Placenta Function and Dysfunction. Am. J. Reprod. Immunol. 2016, 76, 258–271. [Google Scholar] [CrossRef]
- Lappas, M. Activation of inflammasomes in adipose tissue of women with gestational diabetes. Mol. Cell. Endocrinol. 2014, 382, 74–83. [Google Scholar] [CrossRef]
- Liong, S.; Lappas, M. Activation of AMPK improves inflammation and insulin resistance in adipose tissue and skeletal muscle from pregnant women. J. Physiol. Biochem. 2015, 71, 703–717. [Google Scholar] [CrossRef]
- Lappas, M.; Yee, K.; Permezel, M.; Rice, G.E. Release and regulation of leptin, resistin and adiponectin from human placenta, fetal membranes, and maternal adipose tissue and skeletal muscle from normal and gestational diabetes mellitus-complicated pregnancies. J. Endocrinol. 2005, 186, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.T.; Liong, S.; Lim, R.; Barker, G.; Lappas, M. Resveratrol ameliorates the chemical and microbial induction of inflammation and insulin resistance in human placenta, adipose tissue and skeletal muscle. PLoS ONE 2017, 12, e0173373. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef]
- Bonnefont-Rousselot, D. Resveratrol and Cardiovascular Diseases. Nutrients 2016, 8, 250. [Google Scholar] [CrossRef]
- Lykkesfeldt, J.; Svendsen, O. Oxidants and antioxidants in disease: Oxidative stress in farm animals. Vet. J. 2007, 173, 502–511. [Google Scholar] [CrossRef]
- Wang, W.; Liu, G.; Jiang, X.; Wu, G. Resveratrol ameliorates toxic effects of cadmium on placental development in mouse placenta and human trophoblast cells. Birth Defects Res. 2021, 113, 1470–1483. [Google Scholar] [CrossRef]
- Lizarraga, D.; Garcia-Gasca, A. The Placenta as a Target of Epigenetic Alterations in Women with Gestational Diabetes Mellitus and Potential Implications for the Offspring. Epigenomes 2021, 5, 13. [Google Scholar] [CrossRef]
- Şanlı, E.; Kabaran, S. Maternal obesity, maternal overnutrition and fetal programming: Effects of epigenetic mechanisms on the development of metabolic disorders. Curr. Genom. 2019, 20, 419–427. [Google Scholar] [CrossRef]
- Cudmore, M.J.; Ramma, W.; Cai, M.; Fujisawa, T.; Ahmad, S.; Al-Ani, B.; Ahmed, A. Resveratrol inhibits the release of soluble fms-like tyrosine kinase (sFlt-1) from human placenta. Am. J. Obstet. Gynecol. 2012, 206, 253.e10–253.e15. [Google Scholar] [CrossRef]
- Joshi, R.O.; Chellappan, S.; Kukshal, P. Exploring the role of maternal nutritional epigenetics in congenital heart disease. Curr. Dev. Nutr. 2020, 4, nzaa166. [Google Scholar] [CrossRef]
- Samuel, T.M.; Sakwinska, O.; Makinen, K.; Burdge, G.C.; Godfrey, K.M.; Silva-Zolezzi, I. Preterm Birth: A Narrative Review of the Current Evidence on Nutritional and Bioactive Solutions for Risk Reduction. Nutrients 2019, 11, 1811. [Google Scholar] [CrossRef]
- Bariani, M.V.; Correa, F.; Leishman, E.; Domínguez Rubio, A.P.; Arias, A.; Stern, A.; Bradshaw, H.B.; Franchi, A.M. Resveratrol protects from lipopolysaccharide-induced inflammation in the uterus and prevents experimental preterm birth. Mol. Hum. Reprod. 2017, 23, 571–581. [Google Scholar] [CrossRef]
- Romero, R.; Espinoza, J.; Gonçalves, L.F.; Kusanovic, J.P.; Friel, L.A.; Nien, J.K. Inflammation in preterm and term labour and delivery. Semin. Fetal Neonatal Med. 2006, 11, 317–326. [Google Scholar] [CrossRef]
- Cella, M.; Farina, M.G.; Dominguez Rubio, A.P.; Di Girolamo, G.; Ribeiro, M.L.; Franchi, A.M. Dual effect of nitric oxide on uterine prostaglandin synthesis in a murine model of preterm labour. Br. J. Pharmacol. 2010, 161, 844–855. [Google Scholar] [CrossRef]
- Burdet, J.; Sacerdoti, F.; Cella, M.; Franchi, A.M.; Ibarra, C. Role of TNF-α in the mechanisms responsible for preterm delivery induced by Stx2 in rats. Br. J. Pharmacol. 2013, 168, 946–953. [Google Scholar] [CrossRef]
- Domínguez Rubio, A.P.; Sordelli, M.S.; Salazar, A.I.; Aisemberg, J.; Bariani, M.V.; Cella, M.; Rosenstein, R.E.; Franchi, A.M. Melatonin prevents experimental preterm labor and increases offspring survival. J. Pineal Res. 2014, 56, 154–162. [Google Scholar] [CrossRef]
- Törnblom, S.A.; Maul, H.; Klimaviciute, A.; Garfield, R.E.; Byström, B.; Malmström, A.; Ekman-Ordeberg, G. mRNA expression and localization of bNOS, eNOS and iNOS in human cervix at preterm and term labour. Reprod. Biol. Endocrinol. 2005, 3, 33. [Google Scholar] [CrossRef]
- Kozakiewicz, M.L.; Grotegut, C.A.; Howlett, A.C. Endocannabinoid System in Pregnancy Maintenance and Labor: A Mini-Review. Front. Endocrinol. 2021, 12, 699951. [Google Scholar] [CrossRef]
- Carta, G.; Poddighe, L.; Serra, M.P.; Boi, M.; Melis, T.; Lisai, S.; Murru, E.; Muredda, L.; Collu, M.; Banni, S.; et al. Preventive Effects of Resveratrol on Endocannabinoid System and Synaptic Protein Modifications in Rat Cerebral Cortex Challenged by Bilateral Common Carotid Artery Occlusion and Reperfusion. Int. J. Mol. Sci. 2018, 19, 426. [Google Scholar] [CrossRef]
- Denison, R. Pregnancy & perinatal transmission update. WORLD 1998, 89, 6–7. [Google Scholar]
- Osman, I.; Young, A.; Ledingham, M.A.; Thomson, A.J.; Jordan, F.; Greer, I.A.; Norman, J.E. Leukocyte density and pro-inflammatory cytokine expression in human fetal membranes, decidua, cervix and myometrium before and during labour at term. Mol. Hum. Reprod. 2003, 9, 41–45. [Google Scholar] [CrossRef]
- Sennström, M.B.; Ekman, G.; Westergren-Thorsson, G.; Malmström, A.; Byström, B.; Endrésen, U.; Mlambo, N.; Norman, M.; Ståbi, B.; Brauner, A. Human cervical ripening, an inflammatory process mediated by cytokines. Mol. Hum. Reprod. 2000, 6, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Young, A.; Thomson, A.J.; Ledingham, M.; Jordan, F.; Greer, I.A.; Norman, J.E. Immunolocalization of Proinflammatory Cytokines in Myometrium, Cervix, and Fetal Membranes During Human Parturition at Term1. Biol. Reprod. 2002, 66, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Olson, D.M. The role of prostaglandins in the initiation of parturition. Best Pract. Res. Clin. Obstet. Gynaecol. 2003, 17, 717–730. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Alfaidy, N.; Challis, J.R.G. Expression of Matrix Metalloproteinase (MMP)-2 and MMP-9 in Human Placenta and Fetal Membranes in Relation to Preterm and Term Labor. J. Clin. Endocrinol. Metab. 2002, 87, 1353–1361. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Furuya, H.; Taguchi, A.; Kawana, K.; Yamashita, A.; Inoue, E.; Yoshida, M.; Nakamura, H.; Fujimoto, A.; Inoue, T.; Sato, M.; et al. Resveratrol Protects Against Pathological Preterm Birth by Suppression of Macrophage-Mediated Inflammation. Reprod. Sci. 2015, 22, 1561–1568. [Google Scholar] [CrossRef]
- Yang, X.; Dong, W.-B.; Lei, X.-P.; Li, Q.-P.; Zhang, L.-Y.; Zhang, L.-P. Resveratrol suppresses hyperoxia-induced nucleocytoplasmic shuttling of SIRT1 and ROS production in PBMC from preterm infants in vitro. J. Matern.-Fetal Neonatal Med. 2018, 31, 1142–1150. [Google Scholar] [CrossRef]
- Skogen, J.C.; Øverland, S. The fetal origins of adult disease: A narrative review of the epidemiological literature. JRSM Short Rep. 2012, 3, 59. [Google Scholar] [CrossRef]
- Heindel, J.J. The fetal basis of adult disease: Role of environmental exposures—Introduction. Birth Defects Res. Part A Clin. Mol. Teratol. 2005, 73, 131–132. [Google Scholar] [CrossRef]
- Camacho, I.A.; Nagarkatti, M.; Nagarkatti, P.S. Effect of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) on maternal immune response during pregnancy. Arch. Toxicol. 2004, 78, 290–300. [Google Scholar] [CrossRef] [PubMed]
- Marlowe, J.L.; Puga, A. Aryl hydrocarbon receptor, cell cycle regulation, toxicity, and tumorigenesis. J. Cell. Biochem. 2005, 96, 1174–1184. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.P.; Singh, U.S.; Nagarkatti, M.; Nagarkatti, P.S. Resveratrol (3,5,4′-trihydroxystilbene) protects pregnant mother and fetus from the immunotoxic effects of 2,3,7,8-tetrachlorodibenzo-p-dioxin. Mol. Nutr. Food Res. 2011, 55, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Singh, C.K.; LaVoie, H.A.; DiPette, D.J.; Singh, U.S. Resveratrol Restores Nrf2 Level and Prevents Ethanol-Induced Toxic Effects in the Cerebellum of a Rodent Model of Fetal Alcohol Spectrum Disorders. Mol. Pharmacol. 2011, 80, 446–457. [Google Scholar] [CrossRef]
- Darby, J.R.T.; Saini, B.S.; Soo, J.Y.; Lock, M.C.; Holman, S.L.; Bradshaw, E.L.; McInnes, S.J.P.; Voelcker, N.H.; Macgowan, C.K.; Seed, M.; et al. Subcutaneous maternal resveratrol treatment increases uterine artery blood flow in the pregnant ewe and increases fetal but not cardiac growth. J. Physiol. 2019, 597, 5063–5077. [Google Scholar] [CrossRef]
- Aujla, T.; Darby, J.R.T.; Saini, B.S.; Lock, M.C.; Holman, S.L.; Bradshaw, E.L.; Perumal, S.R.; McInnes, S.J.P.; Voelcker, N.H.; Wiese, M.D.; et al. Impact of resveratrol-mediated increase in uterine artery blood flow on fetal haemodynamics, blood pressure and oxygenation in sheep. Exp. Physiol. 2021, 106, 1166–1180. [Google Scholar] [CrossRef]
- Mukherjee, M.; Rahaman, M.; Ray, S.K.; Shukla, P.C.; Dolai, T.K.; Chakravorty, N. Revisiting fetal hemoglobin inducers in beta-hemoglobinopathies: A review of natural products, conventional and combinatorial therapies. Mol. Biol. Rep. 2022, 49, 2359–2373. [Google Scholar] [CrossRef]
- Fibach, E.; Prus, E.; Bianchi, N.; Zuccato, C.; Breveglieri, G.; Salvatori, F.; Finotti, A.; Lipucci di Paola, M.; Brognara, E.; Lampronti, I.; et al. Resveratrol: Antioxidant activity and induction of fetal hemoglobin in erythroid cells from normal donors and beta-thalassemia patients. Int. J. Mol. Med. 2012, 29, 974–982. [Google Scholar] [CrossRef]
- Theodorou, A.; Phylactides, M.; Forti, L.; Cramarossa, M.R.; Spyrou, P.; Gambari, R.; Thein, S.L.; Kleanthous, M. The investigation of resveratrol and analogs as potential inducers of fetal hemoglobin. Blood Cell Mol. Dis. 2016, 58, 6–12. [Google Scholar] [CrossRef]
- Bosquesi, P.L.; Melchior, A.C.B.; Pavan, A.R.; Lanaro, C.; de Souza, C.M.; Rusinova, R.; Chelucci, R.C.; Barbieri, K.P.; Fernandes, G.F.D.; Carlos, I.Z.; et al. Synthesis and evaluation of resveratrol derivatives as fetal hemoglobin inducers. Bioorg. Chem. 2020, 100, 103948. [Google Scholar] [CrossRef]
- Giordo, R.; Cossu, A.; Pasciu, V.; Hoa, P.T.; Posadino, A.M.; Pintus, G. Different redox response elicited by naturally occurring antioxidants in human endothelial cells. Open Biochem. J. 2013, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Pasciu, V.; Posadino, A.M.; Cossu, A.; Sanna, B.; Tadolini, B.; Gaspa, L.; Marchisio, A.; Dessole, S.; Capobianco, G.; Pintus, G. Akt downregulation by flavin oxidase—Induced ROS generation mediates dose-dependent endothelial cell damage elicited by natural antioxidants. Toxicol. Sci. 2010, 114, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Posadino, A.M.; Cossu, A.; Giordo, R.; Zinellu, A.; Sotgia, S.; Vardeu, A.; Hoa, P.T.; Carru, C.; Pintus, G. Resveratrol alters human endothelial cells redox state and causes mitochondrial-dependent cell death. Food Chem. Toxicol. 2015, 78, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Mishra, A.P.; Nigam, M.; Sener, B.; Kilic, M.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Sharifi-Rad, J. Resveratrol: A double-edged sword in health benefits. Biomedicines 2018, 6, 91. [Google Scholar] [CrossRef]
- Savchuk, I.; Morvan, M.L.; Søeborg, T.; Antignac, J.P.; Gemzell-Danielsson, K.; Le Bizec, B.; Söder, O.; Svechnikov, K. Resveratrol inhibits steroidogenesis in human fetal adrenocortical cells at the end of first trimester. Mol. Nutr. Food Res. 2017, 61, 1600522. [Google Scholar] [CrossRef]
- Quispe, C.; Cruz-Martins, N.; Manca, M.L.; Manconi, M.; Sytar, O.; Hudz, N.; Shanaida, M.; Kumar, M.; Taheri, Y.; Martorell, M. Nanoderived Therapeutic Formulations with Curcumin in Inflammation-Related Diseases. Oxidative Med. Cell. Longev. 2021, 2021, 3149223. [Google Scholar] [CrossRef]
- Sanna, V.; Roggio, A.M.; Posadino, A.M.; Cossu, A.; Marceddu, S.; Mariani, A.; Alzari, V.; Uzzau, S.; Pintus, G.; Sechi, M. Novel docetaxel-loaded nanoparticles based on poly (lactide-co-caprolactone) and poly (lactide-co-glycolide-co-caprolactone) for prostate cancer treatment: Formulation, characterization, and cytotoxicity studies. Nanoscale Res. Lett. 2011, 6, 260. [Google Scholar] [CrossRef]
- Sanna, V.; Pintus, G.; Roggio, A.M.; Punzoni, S.; Posadino, A.M.; Arca, A.; Marceddu, S.; Bandiera, P.; Uzzau, S.; Sechi, M. Targeted biocompatible nanoparticles for the delivery of (−)-epigallocatechin 3-gallate to prostate cancer cells. J. Med. Chem. 2011, 54, 1321–1332. [Google Scholar] [CrossRef]
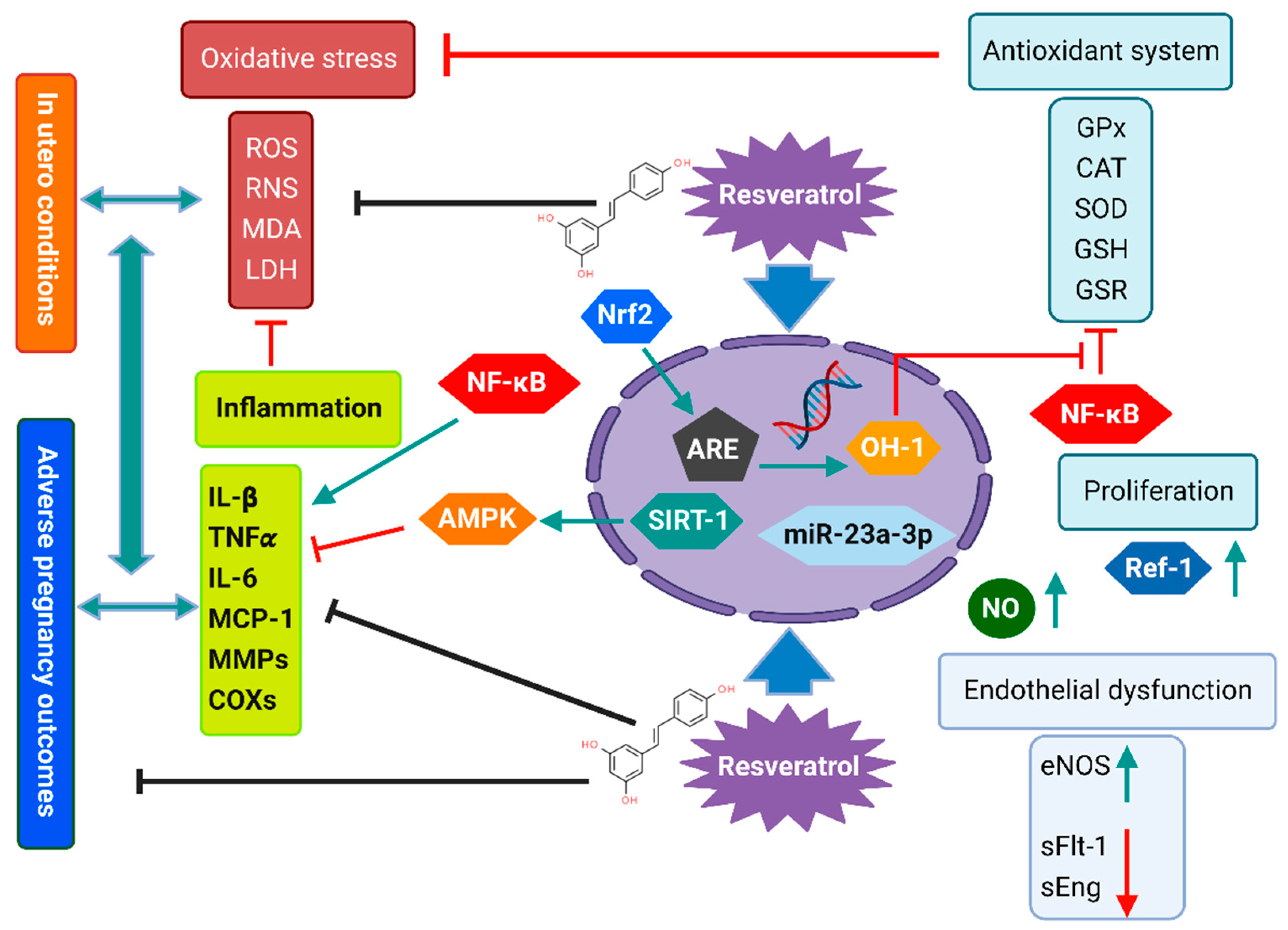
| Reference | Type of the Study | PE Experimental Model | Dose | Mechanism of Action | Outcomes of Supplementation in the Experimental Model |
|---|---|---|---|---|---|
| [94] | In vitro/Clinical trial | -ECs -Human | 200 mL of polyphenol rich-grape fruit/1 µM of trans-resveratrol | NO, HO-1, miRNA expression, GSH, ARE, ROS levels | ↑ ARE, NO, HO-1, GSH, ARE ↓ ROS, oxidative stress No studied effect in offspring |
| [96] | In vitro/Clinical trial | -ECs -Human | 200 mL of polyphenol rich-grape fruit/1µM of trans-resveratrol | SIRT1 expression | No significant effect No studied effect in offspring |
| [102] | In vitro/In vivo | -HTR-8/SVeno cell culture/ -NG-nitro-l-arginine methyl ester mice model (L-NAME) | 100 µmol/L resveratrol 20 mg/kg/day | Expression of genes regulating migration, invasion, angiogenesis and EMT-related factors in trophoblasts Hypertension and proteinuria measurement. Endothelial dysfunction/ injury | ↑ E-cadherin, β-catenin, N-cadherin, and vimentin expression, ↓ Alteration of WNT-related gene expression, including WNT1, WNT3 and WNT5B. ↓ Hypertension and proteinuria No studied effect in offspring |
| [108] | In vitro | primary trophoblasts and HUVECs | 0–100 μM | -Angiogenesis activation (sFlt-1) (sEng) secretion Anti-inflammatory effect -NFκB , IL-6 and IL-1β -Antioxidant effect HO-1, NQO1, GCLC, (TXN) Endothelial dysfunction, VCAM Endothelin-1 eNOS | ↓ sFlt-1, sEng ↓ IL-6, IL-1β and TNF-α.‘ ↑, NQO1, GCLC TXN ↓ HO-1 protein in trophoblast. ↑ VCAM-1 ↓ Endotelin-1, ↑eNOS No studied effect in offspring |
| [109] | In vivo | NG-nitro-l-arginine methyl ester mice model (L-NAME) | 20 mg/kg/day | -Antioxidant effect, Apoptosis SOD, MDA | ↑ SOD, MDA No studied effect in offspring |
| Reference | Type of the Study | GDM Experimental Model | Dose | Mechanism of Action | Outcomes of Supplementation in the Experimental Model |
|---|---|---|---|---|---|
| [116] | In vivo | Streptozotocin GDM pregnant rats model | 120 and 240 mg/kg | Amelioration of blood glucose and blood lipids levels | ↑ Insulin levels, ↓ blood glucose levels Amelioration of lipidic profile No studied effect in offspring |
| [117] | In vivo | Streptozotocin GDM pregnant rats model | 500 μg/mL (CS-ZnO-RS) | Anti-diabetic effect Anti-inflammatory effect | ↓ blood glucose levels lipid content reduced the level of ↓ IL-6 and MCP-1, GRP78, p-IRE1α, p-eIF2α, and p-PERK Inhibition of α-glucosidase and α-amylase No studied effect in offspring |
| [22] | In vivo | Streptozotocin GDM pregnant rats model | 120 and 240 mg/kg | Amelioration of the lipids metabolic profile | ↑ HDL-C and adiponectin ↓ leptin, resistin, TNF-α, IL-6 levels, the body weight, TC, TG, and LDL-C No studied effect in offspring |
| [142] | In vivo | high-fat fed hamster model | 0.025% | Amelioration of the lipids metabolic profile | -HMG-CoA reductase expression No studied effect in offspring |
| [128] | In vivo | high-fat diet GDM mice model IR adipocyte model was established by dexamethasone-inducing | 0.2% | Amelioration of glucose and lipids metabolic profile Amelioration of IR in adipocytes | ↓ The bodyweight, serum glucose ↑ serum insulin Upregulations of miR-23a-3p/NOV axis ↑ Adiponectin, Leptin, p-PI3K, and p-Akt No studied effect in offspring |
| [118] | In vivo | female Sprague-Dawley rat model, fed with a high-fat and sucrose diet | 147 mg/kg/day | Protection against gestational diabetes-induced glucose intolerance and islet dysfunction | -Restored glucose tolerance, normoglycaemia and improved insulin secretion in offspring -Attenuation of hepatic steatosis, insulin resistance, glucose intolerance and dysregulated gluconeogenesis in offspring -Downregulation of lpl, ppara, g6p genes |
| [144] | In vivo | C57BL/KsJ-Lep (db/+) (db/+) genetic GDM pregnant mouse model | 10 mg/kg/day | Amelioration of glucose metabolic profile via SIRT1/AMPK pathway Amelioration of IR | ↑ Glucose metabolism, insulin tolerance and reproductive outcome of the pregnant db/+ females AMPK activation ↓ glucose-6-phosphatase in both pregnant db/+ females and their offspring |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramli, I.; Posadino, A.M.; Giordo, R.; Fenu, G.; Fardoun, M.; Iratni, R.; Eid, A.H.; Zayed, H.; Pintus, G. Effect of Resveratrol on Pregnancy, Prenatal Complications and Pregnancy-Associated Structure Alterations. Antioxidants 2023, 12, 341. https://doi.org/10.3390/antiox12020341
Ramli I, Posadino AM, Giordo R, Fenu G, Fardoun M, Iratni R, Eid AH, Zayed H, Pintus G. Effect of Resveratrol on Pregnancy, Prenatal Complications and Pregnancy-Associated Structure Alterations. Antioxidants. 2023; 12(2):341. https://doi.org/10.3390/antiox12020341
Chicago/Turabian StyleRamli, Iman, Anna Maria Posadino, Roberta Giordo, Grazia Fenu, Manal Fardoun, Rabah Iratni, Ali H. Eid, Hatem Zayed, and Gianfranco Pintus. 2023. "Effect of Resveratrol on Pregnancy, Prenatal Complications and Pregnancy-Associated Structure Alterations" Antioxidants 12, no. 2: 341. https://doi.org/10.3390/antiox12020341
APA StyleRamli, I., Posadino, A. M., Giordo, R., Fenu, G., Fardoun, M., Iratni, R., Eid, A. H., Zayed, H., & Pintus, G. (2023). Effect of Resveratrol on Pregnancy, Prenatal Complications and Pregnancy-Associated Structure Alterations. Antioxidants, 12(2), 341. https://doi.org/10.3390/antiox12020341














