Antioxidant-Loaded Mesoporous Silica—An Evaluation of the Physicochemical Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Adsorption of Antioxidants onto MSMs
2.2.1. Adsorption of GA onto MCM-41, SBA-15, SBA-NH2, and SBA-SH
2.2.2. Adsorption of PCA, CGA, and 4-HBA onto SBA-NH2
2.3. Adsorption Efficiency—HPLC Analysis
2.4. Desorption of Antioxidants from MSMs
2.5. Antioxidant Capacity Assay
2.6. Physicochemical Analysis of Antioxidant-Loaded MSMs
2.6.1. SEM-EDX Analysis
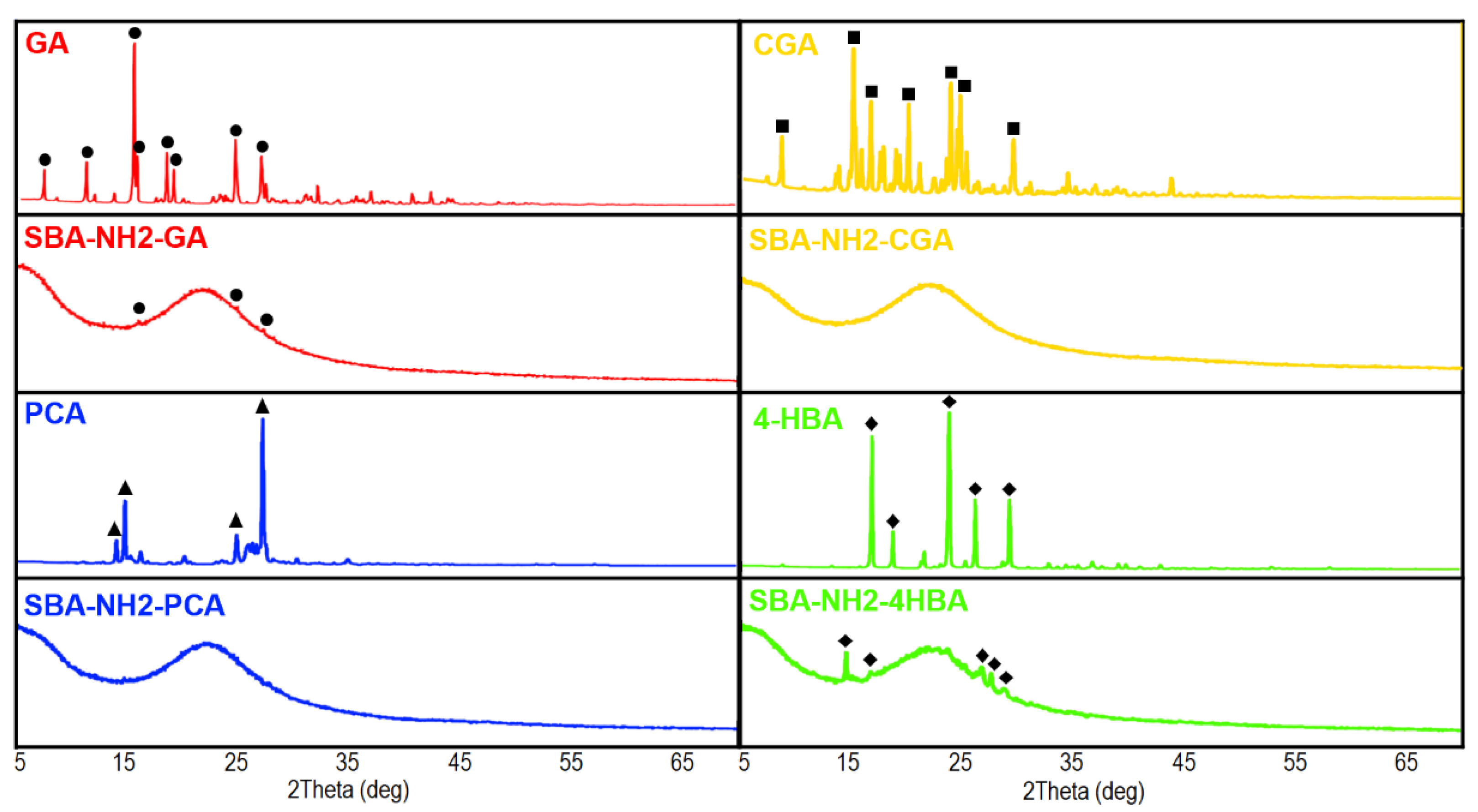
2.6.2. XRD Analysis
2.6.3. FTIR Analysis
2.6.4. Thermal Analyses
3. Results
3.1. Adsorption of Antioxidants onto MSMs
3.1.1. Adsorption of GA onto MCM-41, SBA-15, SBA-NH2, and SBA-SH
3.1.2. Adsorption of GA, PCA, CGA, 4-HBA, onto SBA-NH2
3.2. Desorption of Antioxidants and Antioxidant Capacity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
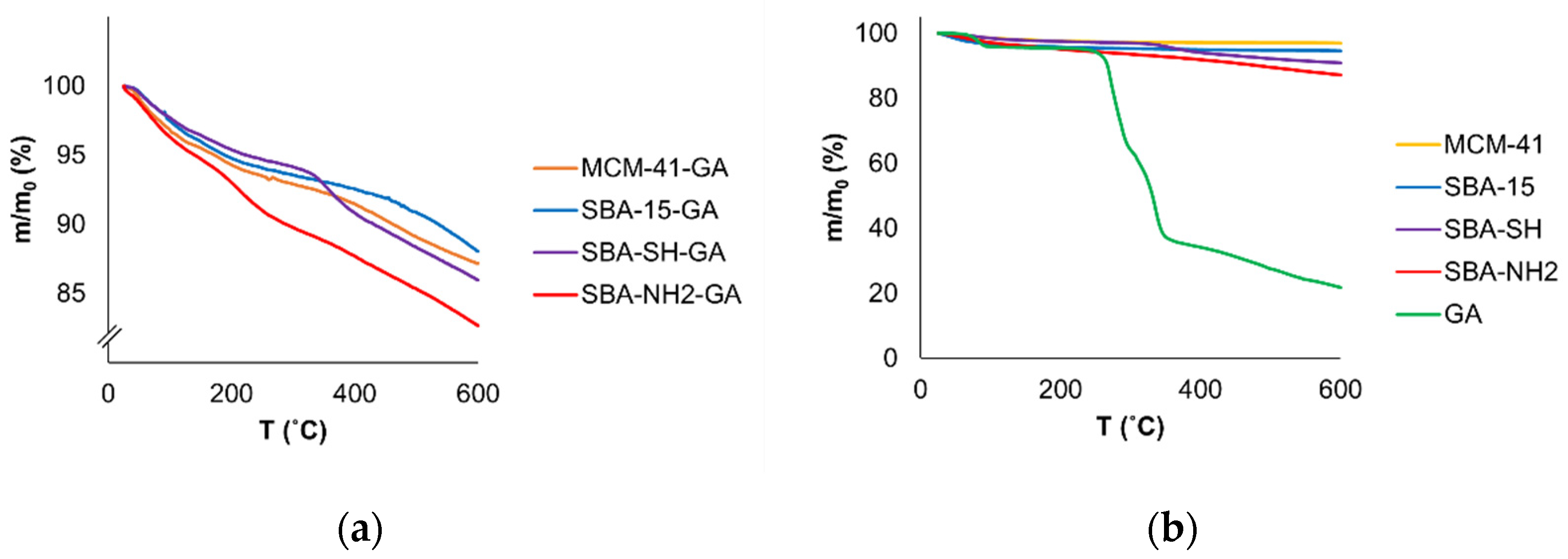
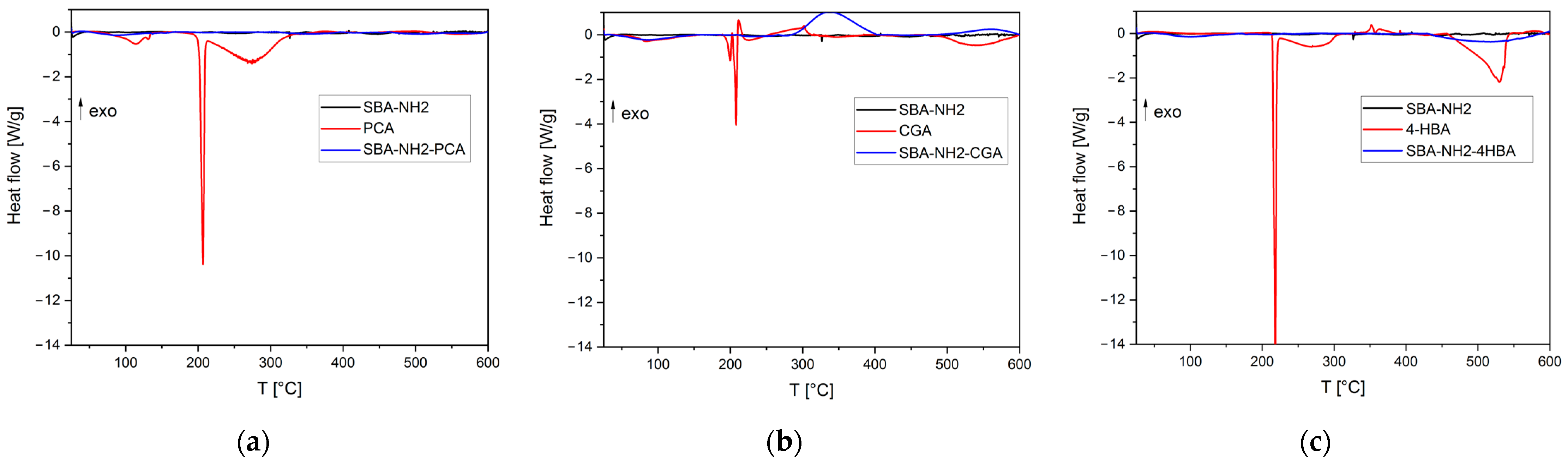
Appendix B
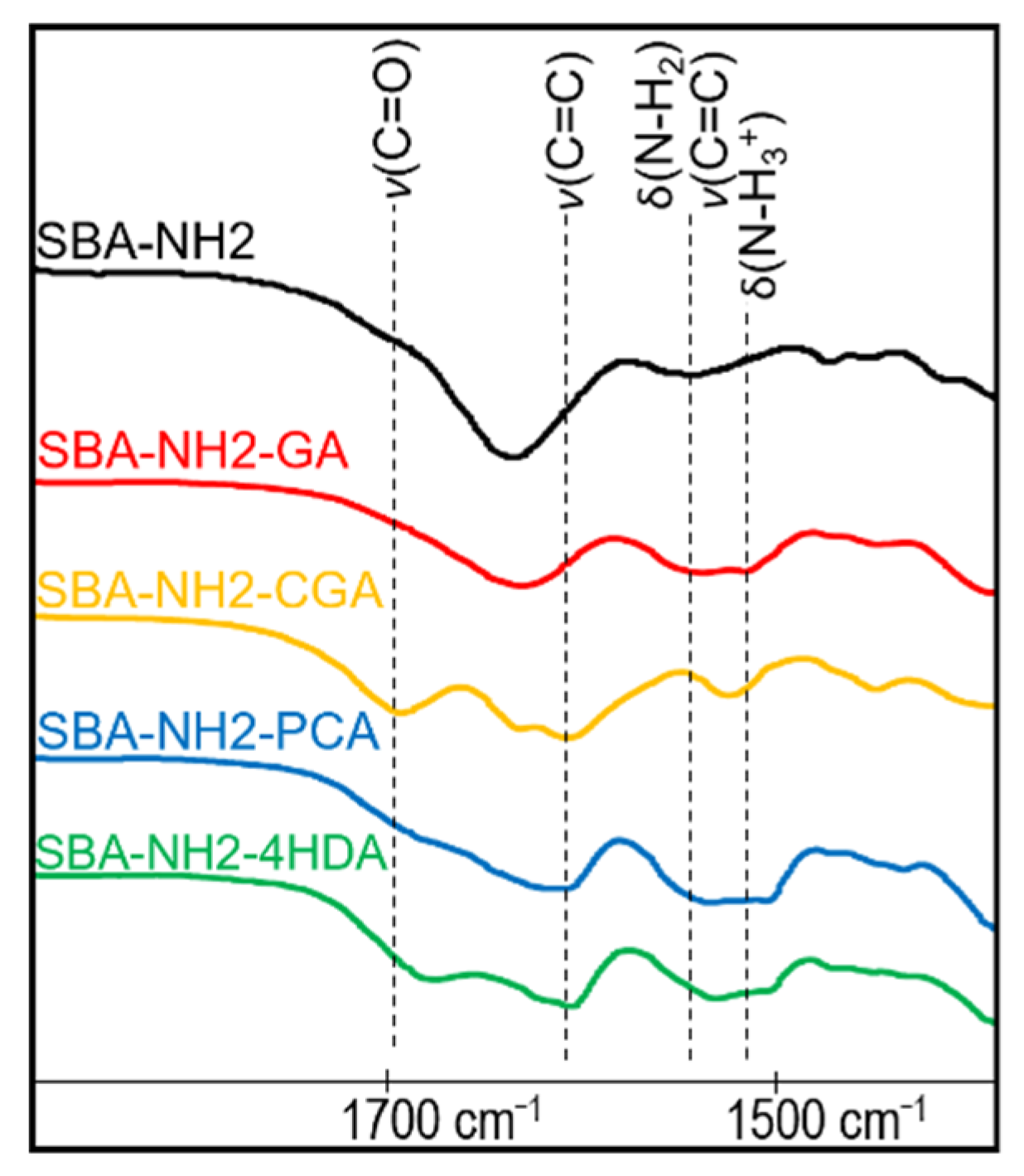
Appendix C
References
- Kisaoglu, A.; Borekci, B.; Yapca, O.E.; Bilen, H.; Suleyman, H. Tissue damage and oxidant/antioxidant balance. Eurasian J. Med. 2013, 45, 47–49. [Google Scholar] [CrossRef] [PubMed]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative stress: Harms and benefits for human health. Oxid. Med. Cell. Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef] [PubMed]
- Kibel, A.; Lukinac, A.M.; Dambic, V.; Juric, I.; Selthofer-Relatic, K. Oxidative stress in ischemic heart disease. Oxid. Med. Cell. Longev. 2020, 2020, 6627144. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T.; Tew, K.D. Oxidative stress in cancer. Cancer Cell 2020, 38, 167–197. [Google Scholar] [CrossRef]
- Yang, X.; Li, Y.; Li, Y.; Ren, X.; Zhang, X.; Hu, D.; Gao, Y.; Xing, Y.; Shang, H. Oxidative stress-mediated atherosclerosis: Mechanisms and therapies. Front. Physiol. 2017, 8, 600. [Google Scholar] [CrossRef]
- Loperena, R.; Harrison, D.G. Oxidative Stress and Hypertensive Diseases. Med. Clin. N. 2017, 101, 169–193. [Google Scholar] [CrossRef]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef]
- Mirończuk-Chodakowska, I.; Witkowska, A.M.; Zujko, M.E. Endogenous non-enzymatic antioxidants in the human body. Adv. Med. Sci. 2018, 63, 68–78. [Google Scholar] [CrossRef]
- Nimse, S.B.; Pal, D. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015, 5, 27986–28006. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Kahkeshani, N.; Farzaei, F.; Fotouhi, M.; Alavi, S.S.; Bahramsoltani, R.; Naseri, R.; Momtaz, S.; Abbasabadi, Z.; Rahimi, R.; Farzaei, M.H.; et al. Pharmacological effects of gallic acid in health and diseases: A mechanistic review. Iran J. Basic Med. Sci. 2019, 22, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Kakkar, S.; Bais, S. A review on protocatechuic acid and its pharmacological potential. ISRN Pharmacol. 2014, 2014, 952943. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Tanaka, T.; Tanaka, M. Potential cancer chemopreventive activity of protocatechuic acid. J. Exp. Clin. Med. 2011, 3, 27–33. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Huang, C.-S.; Huang, C.-Y.; Yin, M.-C. Anticoagulatory, antiinflammatory, and antioxidative effects of protocatechuic acid in diabetic mice. J. Agric. Food Chem. 2009, 57, 6661–6667. [Google Scholar] [CrossRef] [PubMed]
- Lende, A.B.; Kshirsagar, A.D.; Deshpande, A.D.; Muley, M.M.; Patil, R.R.; Bafna, P.A.; Naik, S.R. Anti-inflammatory and analgesic activity of protocatechuic acid in rats and mice. Inflammopharmacol 2011, 19, 255–263. [Google Scholar] [CrossRef]
- Lu, H.; Tian, Z.; Cui, Y.; Liu, Z.; Ma, X. Chlorogenic acid: A comprehensive review of the dietary sources, processing effects, bioavailability, beneficial properties, mechanisms of action, and future directions. Rev. Food Sci. Food Saf. 2020, 19, 3130–3158. [Google Scholar] [CrossRef]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Huang, K.; Liang, X.C.; Zhong, Y.L.; He, W.Y.; Wang, Z. 5-Caffeoylquinic acid decreases diet-inducedobesity in rats by modulating PPARα and LXRα transcription. J. Sci. Food Agric. 2015, 95, 1903–1910. [Google Scholar] [CrossRef]
- Suzuki, A.; Yamamoto, N.; Jokura, H.; Yamamoto, M.; Fujii, A.; Tokimitsu, I.; Saito, I. Chlorogenic acid attenuates hypertension and improves endothelial function in spontaneously hypertensive rats. J. Hypertens. 2006, 24, 1065–1073. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Clifford, M.N. Dietary hydroxybenzoic acid derivatives—Nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1024–1032. [Google Scholar] [CrossRef]
- Velika, B.; Kron, I. Antioxidant properties of benzoic acid derivatives against superoxide radical. Free Radic. Antioxid. 2012, 2, 62–67. [Google Scholar] [CrossRef]
- Merkl, R.; Hrádková, I.; Filip, V.; Šmidrkal, J. Antimicrobial and antioxidant properties of phenolic acids alkyl esters. Czech J. Food Sci. 2010, 28, 275–279. [Google Scholar] [CrossRef]
- Fransway, A.F.; Fransway, P.J.; Belsito, D.V.; Warshaw, E.M.; Sasseville, D.; Fowler, J.F., Jr.; DeKoven, J.G.; Pratt, M.D.; Maibach, H.I.; Taylor, J.S.; et al. Parabens. Dermatitis 2019, 30, 3–31. [Google Scholar] [CrossRef]
- Lafay, S.; Gil-Izquierdo, A. Bioavailability of phenolic acids. Phytochem. Rev. 2008, 7, 301–311. [Google Scholar] [CrossRef]
- Brglez Mojzer, E.; Knez Hrnčič, M.; Škerget, M.; Knez, Ž.; Bren, U. Polyphenols: Extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules 2016, 21, 901. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Jürgens, H.S. Effect of pH on the stability of plant phenolic compounds. J. Agric. Food Chem. 2000, 48, 2101–2110. [Google Scholar] [CrossRef] [PubMed]
- Volf, I.; Ignat, I.; Neamtu, M.; Popa, V. Thermal stability, antioxidant activity, and photo-oxidation of natural polyphenols. Chem. Pap. 2014, 68, 121–129. [Google Scholar] [CrossRef]
- Cao, H.; Saroglu, O.; Karadag, A.; Diaconeasa, Z.; Zoccatelli, G.; Conte-Junior, C.A.; Gonzalez-Aguilar, G.A.; Ou, J.; Bai, W.; Zamarioli, C.M.; et al. Available technologies on improving the stability of polyphenols in food processing. Food Front. 2021, 2, 109–139. [Google Scholar] [CrossRef]
- Guo, Y.; Sun, Q.; Wu, F.-G.; Dai, Y.; Chen, X. Polyphenol-containing nanoparticles: Synthesis, properties, and therapeutic delivery. Adv. Mater. 2021, 33, 2007356. [Google Scholar] [CrossRef]
- Yang, B.; Dong, Y.; Wang, F.; Zhang, Y. Nanoformulations to enhance the bioavailability and physiological functions of polyphenols. Molecules 2020, 25, 4613. [Google Scholar] [CrossRef]
- Niu, L.; Li, Z.; Fan, W.; Zhong, X.; Peng, M.; Liu, Z. Nano-strategies for enhancing the bioavailability of tea polyphenols: Preparation, applications, and challenges. Foods 2022, 11, 387. [Google Scholar] [CrossRef] [PubMed]
- Milinčić, D.D.; Popović, D.A.; Lević, S.M.; Kostić, A.Ž.; Tešić, Ž.L.; Nedović, V.A.; Pešić, M.B. Application of polyphenol-loaded nanoparticles in food industry. Nanomaterials 2019, 9, 1629. [Google Scholar] [CrossRef] [PubMed]
- Brezoiu, A.-M.; Bajenaru, L.; Berger, D.; Mitran, R.-A.; Deaconu, M.; Lincu, D.; Stoica Guzun, A.; Matei, C.; Moisescu, M.G.; Negreanu-Pirjol, T. Effect of nanoconfinement of polyphenolic extract from grape pomace into functionalized mesoporous silica on its biocompatibility and radical scavenging activity. Antioxidants 2020, 9, 696. [Google Scholar] [CrossRef] [PubMed]
- Ianni, F.; Gagliardi, A.; Taticchi, A.; Servili, M.; Pinna, N.; Schoubben, A.; Sardella, R.; Bruscoli, S. Exploiting food-grade mesoporous silica to preserve the antioxidant properties of fresh olive mill wastewaters phenolic extracts. Antioxidants 2021, 10, 1361. [Google Scholar] [CrossRef]
- Narayan, R.; Nayak, U.Y.; Raichur, A.M.; Garg, S. Mesoporous silica nanoparticles: A comprehensive review on synthesis and recent advances. Pharmaceutics 2018, 10, 118. [Google Scholar] [CrossRef]
- Yang, P.; Gaib, S.; Lin, J. Functionalized mesoporous silica materials for controlled drug delivery. Chem. Soc. Rev. 2012, 41, 3679–3698. [Google Scholar] [CrossRef]
- Park, J.H.; Gu, L.; von Maltzahn, G.; Ruoslahti, E.; Bhatia, S.N.; Sailor, M.J. Biodegradable luminescent porous silicon nanoparticles for in vivo applications. Nat. Mater. 2009, 8, 331–336. [Google Scholar] [CrossRef]
- Janjua, T.I.; Cao, Y.; Yu, C.; Popat, A. Clinical translation of silica nanoparticles. Nat. Rev. Mater. 2021, 6, 1072–1074. [Google Scholar] [CrossRef]
- Casado, N.; Morante-Zarcero, S.; Pérez-Quintanilla, D.; Câmara, J.S.; Sierra, I. Two novel strategies in food sample preparation for the analysis of dietary polyphenols: Micro-extraction techniques and new silica-based sorbent materials. Trends Food Sci. Technol. 2020, 98, 167–180. [Google Scholar] [CrossRef]
- Cotea, V.V.; Luchian, C.E.; Bilba, N.; Niculaua, M. Mesoporous silica SBA-15, a new adsorbent for bioactive polyphenols from red wine. Anal. Chim. Acta 2012, 732, 180–185. [Google Scholar] [CrossRef]
- Ioniţă, S.; Lincu, D.; Mitran, R.-A.; Ziko, L.; Sedky, N.K.; Deaconu, M.; Brezoiu, A.-M.; Matei, C.; Berger, D. Resveratrol encapsulation and release from pristine and functionalized mesoporous silica carriers. Pharmaceutics 2022, 14, 203. [Google Scholar] [CrossRef] [PubMed]
- Buda, V.; Brezoiu, A.M.; Berger, D.; Pavel, I.Z.; Muntean, D.; Minda, D.; Dehelean, C.A.; Soica, C.; Diaconeasa, Z.; Folescu, R.; et al. Biological evaluation of black chokeberry extract free and embedded in two mesoporous silica-type matrices. Pharmaceutics 2020, 12, 838. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, L.; Vasheghani-Farahani, E.; Rostami, K.; Ganji, F.; Fallahpour, M. Mesoporous silica nanoparticles with different pore sizes for delivery of pH-sensitive gallic acid. Asia-Pac. J. Chem. Eng. 2014, 9, 845–853. [Google Scholar] [CrossRef]
- Lewandowski, D.; Ruszkowski, P.; Pińska, A.; Schroeder, G.; Kurczewska, J. SBA-15 mesoporous silica modified with gallic acid and evaluation of its cytotoxic activity. PLoS ONE 2015, 10, e0132541. [Google Scholar] [CrossRef] [PubMed]
- Iraji, S.; Ganji, F.; Rashidi, L. Surface modified mesoporous silica nanoparticles as sustained-release gallic acid nano-carriers. J. Drug Deliv. Sci. Technol. 2018, 47, 468–476. [Google Scholar] [CrossRef]
- Rashidi, L.; Vasheghani-Farahani, E.; Soleimani, M.; Atashi, A.; Rostami, K.; Gangi, F.; Fallahpour, M.; Tahouri, M.T. A cellular uptake and cytotoxicity properties study of gallic acid-loaded mesoporous silica nanoparticles on Caco-2 cells. J. Nanopart. Res. 2014, 16, 2285. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 18, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Majda, D.; Zimowska, M.; Tarach, K.; Góra-Marek, K.; Napruszewska, B.D.; Michalik-Zym, A. Water thermoporosimetry as a tool of characterization of the textural parameters of mesoporous materials. J. Therm. Anal. Calorim. 2017, 127, 207–220. [Google Scholar] [CrossRef]
- Araujo, P.Z.; Morando, P.J.; Blesa, M.A. Interaction of catechol and gallic acid with titanium dioxide in aqueous suspensions. 1. equilibrium studies. Langmuir 2005, 21, 3470–3474. [Google Scholar] [CrossRef]
- Rocha, J.E.; Guedes, T.T.A.M.; Bezerra, C.F.; Costa, M.S.; Campina, F.F.; de Freitas, T.S.; Souza, A.K.; Souza, C.E.S.; de Matos, Y.M.L.S.; Pereira-Junior, F.N.; et al. Identification of the gallic acid mechanism of action on mercury chloride toxicity reduction using infrared spectroscopy and antioxidant assays. Int. Biodeterior. Biodegrad. 2019, 141, 24–29. [Google Scholar] [CrossRef]
- Rabiei, M.; Sabahi, H.; Rezayan, A.H. Gallic acid-loaded montmorillonite nanostructure as a new controlled release system. Appl. Clay Sci. 2016, 119, 236–242. [Google Scholar] [CrossRef]
- Gorni, G.; Pascual, M.J.; Caballero, A.; Velázquez, J.J.; Mosa, J.; Castro, Y.; Durán, A. Crystallization mechanism in sol-gel oxyfluoride glass-ceramics. J. Non Cryst. Solids 2018, 501, 145–152. [Google Scholar] [CrossRef]
- Chong, A.S.M.; Zhao, X.S. Functionalization of SBA-15 with APTES and characterization of functionalized materials. J. Phys. Chem. B 2003, 107, 12650–12657. [Google Scholar] [CrossRef]
- Qu, Q.; Zhou, G.; Ding, Y.; Feng, S.; Gu, Z. Adjustment of the morphology of MCM-41 silica in basic solution. J. Non Cryst. Solids 2014, 405, 104–115. [Google Scholar] [CrossRef]
- Qin, Y.; Wang, Y.; Wang, H.; Gao, J.; Qu, Z. Effect of morphology and pore structure of SBA-15 on toluene dynamic adsorption/desorption performance. Procedia Environ. Sci. 2013, 18, 366–371. [Google Scholar] [CrossRef][Green Version]
- Petrisor, G.; Ficai, D.; Motelica, L.; Trusca, R.D.; Bîrcă, A.C.; Vasile, B.S.; Voicu, G.; Oprea, O.C.; Semenescu, A.; Ficai, A.; et al. Mesoporous silica materials loaded with gallic acid with antimicrobial potential. Nanomaterials 2022, 12, 1648. [Google Scholar] [CrossRef]
- Luzi, F.; Puglia, D.; Dominici, F.; Fortunati, E.; Giovanale, G.; Balestra, G.; Torre, L. Effect of gallic acid and umbelliferone on thermal, mechanical, antioxidant and antimicrobial properties of poly (vinyl alcohol-co-ethylene) films. Polym. Degrad. Stab. 2018, 152, 162–176. [Google Scholar] [CrossRef]
- Li, I.; Kim, S.Y.; Chen, X.; Park, H.J. Calcium-alginate beads loaded with gallic acid: Preparation and characterization. LWT-Food Sci. Technol. 2016, 68, 667–673. [Google Scholar] [CrossRef]
- Zhang, B.; Li, S.; Wang, C.; Zou, T.; Pan, T.; Zhang, J.; Xu, Z.; Ren, G.; Zhao, H. Terahertz spectroscopic investigation of gallic acid and its monohydrate. Spectrochim. Acta A Mol. Biomol. 2018, 190, 40–46. [Google Scholar] [CrossRef]
- Mashhadi, S.M.A.; Yunus, U.; Bhatti, M.H.; Ahmed, I.; Tahir, M.N. Synthesis, characterization, solubility and stability studies of hydrate cocrystal of antitubercular isoniazid with antioxidant and anti-bacterial protocatechuic acid. J. Mol. Struct. 2016, 1117, 17–21. [Google Scholar] [CrossRef]
- Chao, J.; Wang, H.; Zhao, W.; Zhang, M.; Zhang, L. Investigation of the inclusion behavior of chlorogenic acid with hydroxypropyl-β-cyclodextrin. Int. J. Biol. Macromol. 2012, 50, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Vangala, V.R.; Chow, P.S.; Tan, R.B.H. Characterization, physicochemical and photo-stability of a co-crystal involving an antibiotic drug, nitrofurantoin, and 4-hydroxybenzoic acid. CrystEngComm 2011, 13, 759–762. [Google Scholar] [CrossRef]
- Dadej, A.; Woźniak-Braszak, A.; Bilski, P.; Piotrowska-Kempisty, H.; Józkowiak, M.; Stawny, M.; Dadej, D.; Mrotek, M.; Jelińska, A. APTES-Modified SBA-15 as a non-toxic carrier for phenylbutazone. Materials 2022, 15, 946. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zheng, S.; Liu, J.; Xu, Z. Tannic acid adsorption on amino-functionalized magnetic mesoporous silica. Chem. Eng. J. 2010, 165, 10–16. [Google Scholar] [CrossRef]
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeersch, T.; Zurek, E.; Hutchison, G.R. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 2012, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekar, G.; Vinu, A.; Murugesan, V.; Hartmann, M. Adsorption of vitamin E on mesoporous silica molecular sieves. In Studies in Surface Science and Catalysis; Čejka, J., Žilková, N., Nachtigall, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; Volume 158, pp. 1169–1176. [Google Scholar] [CrossRef]
- Richner, G.; Puxty, G. Assessing the chemical speciation during CO2 absorption by aqueous amines using in situ FTIR. Ind. Eng. Chem. Res. 2012, 51, 14317–14324. [Google Scholar] [CrossRef]
- Liu, H.; Yu, H.; Jin, P.; Jiang, M.; Zhu, G.; Duan, Y.; Yang, Z.; Qiu, H. Preparation of mesoporous silica materials functionalized with various amino-ligands and investigation of adsorption performances on aromatic acids. Chem. Eng. J. 2020, 379, 122405. [Google Scholar] [CrossRef]
- Ravinayagam, V.; Rabindran Jermy, B. Studying the loading effect of acidic type antioxidant on amorphous silica nanoparticle carriers. J. Nanopart. Res. 2017, 19, 190. [Google Scholar] [CrossRef]
- Berlier, G.; Gastaldi, L.; Ugazio, E.; Miletto, I.; Iliade, P.; Sapino, S. Stabilization of quercetin flavonoid in MCM-41 mesoporous silica: Positive effect of surface functionalization. J. Colloid Interface Sci. 2013, 393, 109–118. [Google Scholar] [CrossRef]
- Arriagada, F.; Günther, G.; Morales, J. Nanoantioxidant–based silica particles as flavonoid carrier for drug delivery applications. Pharmaceutics 2020, 12, 302. [Google Scholar] [CrossRef]
- Uejo, F.; Limwikrant, W.; Moribe, K.; Yamamoto, K. Dissolution improvement of fenofibrate by melting inclusion in mesoporous silica. Asian J. Pharm. Sci. 2013, 8, 329–335. [Google Scholar] [CrossRef]
- Van Speybroeck, M.; Barillaro, V.; Thi, T.D.; Mellaerts, R.; Martens, J.; Van Humbeeck, J.; Vermant, J.; Annaert, P.; Van Den Mooter, G.; Augustijns, P. Ordered mesoporous silica material SBA-15: A broad-spectrum formulation platform for poorly soluble drugs. J. Pharm. Sci. 2009, 98, 2648–2658. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Nie, L.; Feng, S.; Suo, J. Preparation, characterization and in vitro release study of gallic acid loaded silica nanoparticles for controlled release. Die Pharm.—Int. J. Pharm. Sci. 2013, 68, 401–405. [Google Scholar] [CrossRef]


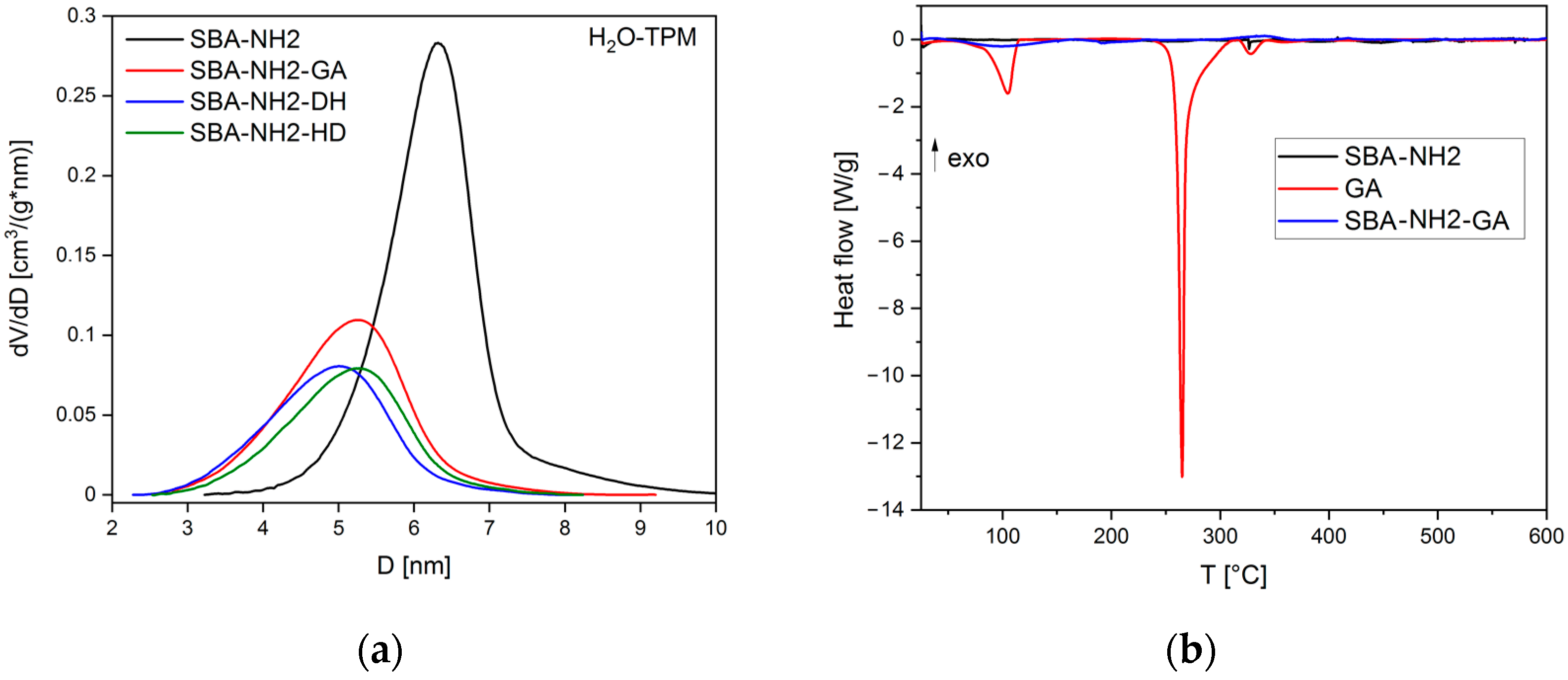
| Material Type | mAds ± SD (mg/g) | %Ads ± SD |
|---|---|---|
| MCM-41 | 97.6 ± 27.5 | 18.8 ± 5.3 |
| SBA-15 | 69.1 ± 7.8 | 13.3 ± 1.5 |
| SBA-NH2 | 133.4 ± 27.8 | 25.7 ± 5.4 |
| SBA-SH | 67.9 ± 13.3 | 13.1 ± 2.6 |
| Material Type | mAds ± SD (mg/g) | %Ads ± SD |
|---|---|---|
| SBA-NH2-GA | 133.4 ± 27.8 | 25.7 ± 5.4 |
| SBA-NH2-PCA | 52.7 ± 13.5 | 8.1 ± 2.1 |
| SBA-NH2-CGA | 290.8 ± 12.3 | 55.2 ± 2.3 |
| SBA-NH2-4HBA | 54.5 ± 4.0 | 2.8 ± 0.2 |
| Material Type | D (nm) | V (cm3/g) |
|---|---|---|
| SBA-NH2 | 6.3 | 0.41 |
| SBA-NH2-GA | 5.2 | 0.21 |
| SBA-NH2-PCA | 5.0 | 0.16 |
| SBA-NH2-CGA | below detection limit | below detection limit |
| SBA-NH2-4HBA | 5.3 | 0.15 |
| Material Type | HPLC | FC | AC | ||
|---|---|---|---|---|---|
| mDes ± SD (mg) | %Des ± SD | mDes ± SD (mg) | %Des ± SD | ||
| MCM-41-GA | 1.5 ± 0.02 | 31.2 ± 0.5 | 1.9 ± 0.09 | 39.2 ± 1.8 | 1.3 |
| SBA-15-GA | 1.7 ± 0.04 | 58.2 ± 7.7 | 2.21 ± 0.06 | 77.0 ± 10.0 | 1.3 |
| SBA-15-SH | 1.3 ± 0.06 | 39.1 ±1.8 | 1.58 ± 0.06 | 46.4 ± 1.6 | 1.2 |
| SBA-NH2-GA | 1.8 ± 0.02 | 27.4 ± 0.2 | 2.3 ± 0.1 | 35.1 ± 2.2 | 1.3 |
| SBA-NH2-PCA | 1.4 ± 0.01 | 53.1 ± 0.5 | 1.4 ± 0.06 | 52.4 ± 2.3 | 1.0 |
| SBA-NH2-CGA | 5.0 ± 0.14 | 33.8 ± 0.9 | 5.3 ± 0.05 | 35.4 ± 0.3 | 1.1 |
| SBA-NH2-4HBA | 0.71 ± 0.03 | 25.9 ± 1.1 | 1.04 ± 0.09 | 38.0 ± 3.3 | 1.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szewczyk, A.; Brzezińska-Rojek, J.; Ośko, J.; Majda, D.; Prokopowicz, M.; Grembecka, M. Antioxidant-Loaded Mesoporous Silica—An Evaluation of the Physicochemical Properties. Antioxidants 2022, 11, 1417. https://doi.org/10.3390/antiox11071417
Szewczyk A, Brzezińska-Rojek J, Ośko J, Majda D, Prokopowicz M, Grembecka M. Antioxidant-Loaded Mesoporous Silica—An Evaluation of the Physicochemical Properties. Antioxidants. 2022; 11(7):1417. https://doi.org/10.3390/antiox11071417
Chicago/Turabian StyleSzewczyk, Adrian, Joanna Brzezińska-Rojek, Justyna Ośko, Dorota Majda, Magdalena Prokopowicz, and Małgorzata Grembecka. 2022. "Antioxidant-Loaded Mesoporous Silica—An Evaluation of the Physicochemical Properties" Antioxidants 11, no. 7: 1417. https://doi.org/10.3390/antiox11071417
APA StyleSzewczyk, A., Brzezińska-Rojek, J., Ośko, J., Majda, D., Prokopowicz, M., & Grembecka, M. (2022). Antioxidant-Loaded Mesoporous Silica—An Evaluation of the Physicochemical Properties. Antioxidants, 11(7), 1417. https://doi.org/10.3390/antiox11071417







