Oxidative Stress and Human Ovarian Response—From Somatic Ovarian Cells to Oocytes Damage: A Clinical Comprehensive Narrative Review
Abstract
1. Background and Rationale
2. Materials and Methods
3. The Importance of Oxidative Balance
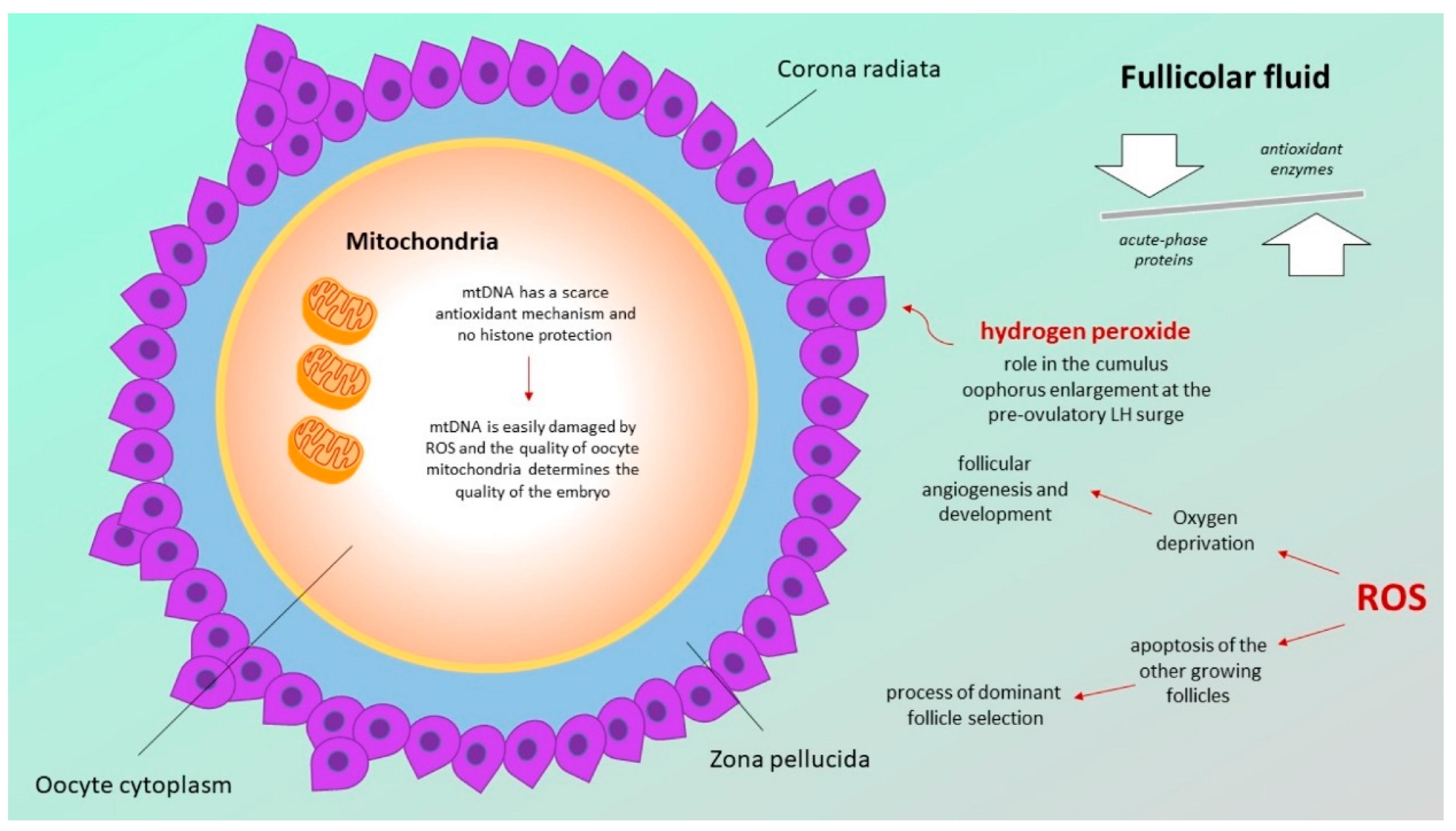
3.1. Follicular Fluid Redox Involvement in Ovarian Follicle Growth and Ovulation
3.2. Defects in the Mitochondrial Respiratory Chain
4. Oocytes Oxidative Stress and Endometriosis
5. Oocytes Oxidative Stress and Polycystic Ovary Syndrome
6. Oocytes Oxidative Stress and Aging
7. Oocytes Oxidative Stress and Cancer Treatment
8. Oocytes Oxidative Stress and Environmental Agents
9. Possible Therapeutic Strategies: Antioxidant Agents
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Agarwal, A.; Allamaneni, S.S. Role of free radicals in female reproductive diseases and assisted reproduction. Reprod. Biomed. Online 2004, 9, 338–347. [Google Scholar] [CrossRef]
- Ruder, E.H.; Hartman, T.J.; Goldman, M.B. Impact of oxidative stress on female fertility. Curr. Opin. Obstet. Gynecol. 2009, 21, 219–222. [Google Scholar] [CrossRef]
- Hamdan, M.; Jones, K.; Cheong, Y.; Lane, S.I.R. The sensitivity of the DNA damage checkpoint prevents oocyte maturation in endometriosis. Sci. Rep. 2016, 6, 36994. [Google Scholar] [CrossRef]
- Xu, J.; Osuga, Y.; Yano, T.; Morita, Y.; Tang, X.; Fujiwara, T.; Takaia, Y.; Matsumia, H.; Kogaa, K.; Taketani, Y.; et al. Bisphenol A induces apoptosis and G2-to-M arrest of ovarian granulosa cells. Biochem. Biophys. Res. Commun. 2002, 292, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Canipari, R. Oocyte-granulosa cell interactions. Hum. Reprod. Update 2000, 6, 279–289. [Google Scholar] [CrossRef]
- Roberts, M.J.; Gammill, H.S. Preeclampsia: Recent insights. Hypertension 2005, 46, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Sibai, B.; Dekker, G.; Kupferminc, M. Pre-eclampsia. Lancet 2005, 365, 785–799. [Google Scholar] [CrossRef]
- Gupta, S.; Agarwal, A.; Sharma, R.K. The Role of Placental Oxidative Stress and Lipid Peroxidation in Preeclampsia. Obstet. Gynecol. Surv. 2005, 60, 807–816. [Google Scholar] [CrossRef]
- Açikgoz, S.; Harma, M.; Harma, M.; Mungan, G.; Can, M.; Demirtaş, S. Comparison of Angiotensin-Converting Enzyme, Malonaldehyde, Zinc, and Copper Levels in Preeclampsia. Biol. Trace Element Res. 2006, 113, 1–8. [Google Scholar] [CrossRef]
- Duan, J.; Kasper, D.L. Oxidative depolymerization of polysaccharides by reactive oxygen/nitrogen species. Glycobiology 2010, 21, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Naskalski, J.W.; Bartosz, G. Oxidative modifications of protein structures. Adv. Clin. Chem. 2000, 35, 161–253. [Google Scholar] [CrossRef] [PubMed]
- Woodbine, L.; Brunton, H.; Goodarzi, A.A.; Shibata, A.; Jeggo, P.A. Endogenously induced DNA double strand breaks arise in heterochromatic DNA regions and require ataxia telangiectasia mutated and Artemis for their repair. Nucleic Acids Res. 2011, 39, 6986–6997. [Google Scholar] [CrossRef]
- Winship, A.L.; Stringer, J.M.; Liew, S.H.; Hutt, K.J. The importance of DNA repair for maintaining oocyte quality in response to anti-cancer treatments, environmental toxins and maternal ageing. Hum. Reprod. Update 2018, 24, 119–134. [Google Scholar] [CrossRef] [PubMed]
- Polo, S.E.; Jackson, S.P. Dynamics of DNA damage response proteins at DNA breaks: A focus on protein modifications. Genes Dev. 2011, 25, 409–433. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.B.; Hutt, K.J.; Michalak, E.M.; Cook, M.; Vandenberg, C.J.; Liew, S.H.; Bouillet, P.; Mills, A.; Scott, C.L.; Findlay, J.K.; et al. DNA damage-induced primordial follicle oocyte apoptosis and loss of fertility require TAp63-mediated induction of Puma and Noxa. Mol. Cell 2012, 48, 343–352. [Google Scholar] [CrossRef]
- Shaffer, L.G.; Lupski, J.R. Molecular Mechanisms for Constitutional Chromosomal Rearrangements in Humans. Annu. Rev. Genet. 2000, 34, 297–329. [Google Scholar] [CrossRef] [PubMed]
- McFadden, D.E.; Friedman, J.M. Chromosome abnormalities in human beings. Mutat. Res. 1997, 396, 129–140. [Google Scholar] [CrossRef]
- Zhang, Y.-X.; Zhang, Y.-P.; Gu, Y.; Guan, F.-J.; Li, S.-L.; Xie, J.-S.; Shen, Y.; Wu, B.-L.; Ju, W.; Jenkins, E.; et al. Genetic analysis of first-trimester miscarriages with a combination of cytogenetic karyotyping, microsatellite genotyping and arrayCGH. Clin. Genet. 2009, 75, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Ravel, C.; Letur, H.; Le Lannou, D.; Barthelemy, C.; Bresson, J.L.; Siffroi, J.P. High incidence of chromosomal abnormalities in oocyte donors. Fertil. Steril. 2007, 87, 439–441. [Google Scholar] [CrossRef] [PubMed]
- Lim, A.S.; Tsakok, M.F. Age-related decline in fertility: A link to degenerative oocytes? Fertil. Steril. 1997, 68, 265–271. [Google Scholar] [CrossRef]
- Agarwal, A.; Gupta, S.; Sekhon, L.; Shah, R. Redox Considerations in Female Reproductive Function and Assisted Reproduction: From Molecular Mechanisms to Health Implications. Antioxid. Redox Signal. 2008, 10, 1375–1403. [Google Scholar] [CrossRef] [PubMed]
- Gougeon, A. Human ovarian follicular development: From activation of resting follicles to preovulatory maturation. Ann. d’Endocrinol. 2010, 71, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Ambekar, A.S.; Nirujogi, R.S.; Srikanth, S.M.; Chavan, S.; Kelkar, D.S.; Hinduja, I.; Zaveri, K.; Prasad, T.K.; Harsha, H.; Pandey, A.; et al. Proteomic analysis of human follicular fluid: A new perspective towards understanding folliculogenesis. J. Proteom. 2013, 87, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Angelucci, S.; Ciavardelli, D.; Di Giuseppe, F.; Eleuterio, E.; Sulpizio, M.; Tiboni, G.M.; Giampietro, F.; Palumbo, P.; Di Ilio, C. Proteome analysis of human follicular fluid. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2006, 1764, 1775–1785. [Google Scholar] [CrossRef]
- Kishi, I.; Ohishi, M.; Akiba, Y.; Asada, H.; Konishi, Y.; Nakano, M.; Kamei, K.; Yoshimura, Y.; Maruyama, T. Thioredoxin, an antioxidant redox protein, in ovarian follicles of women undergoing in vitro fertilization. Endocr. J. 2016, 63, 9–20. [Google Scholar] [CrossRef]
- Shkolnik, K.; Tadmor, A.; Ben-Dor, S.; Nevo, N.; Galiani, D.; Dekel, N. Reactive oxygen species are indispensable in ovulation. Proc. Natl. Acad. Sci. USA 2011, 108, 1462–1467. [Google Scholar] [CrossRef]
- Juengel, J.L.; McNatty, K.P. The role of proteins of the transforming growth factor-beta superfamily in the intraovarian regulation of follicular development. Hum. Reprod. Update 2005, 11, 143–160. [Google Scholar] [CrossRef]
- Gordon, J.D.; Mesiano, S.; Zaloudek, C.J.; Jaffe, R.B. Vascular endothelial growth factor localization in human ovary and fallopian tubes: Possible role in reproductive function and ovarian cyst formation. J. Clin. Endocrinol. Metab. 1996, 81, 353–359. [Google Scholar]
- Behrman, H.R.; Kodaman, P.H.; Preston, S.L.; Gao, S. Oxidative stress and the ovary. J. Soc. Gynecol. Investig. 2001, 8 (Suppl. 1), S40–S42. [Google Scholar]
- Chappel, S. The Role of Mitochondria from Mature Oocyte to Viable Blastocyst. Obstet. Gynecol. Int. 2013, 2013, 183024. [Google Scholar] [CrossRef]
- May-Panloup, P.; Chretien, M.; Malthiery, Y.; Reynier, P. Mitochondrial DNA in the Oocyte and the Developing Embryo. Curr. Top. Dev. Biol. 2007, 77, 51–83. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Keilty, D.; Zhang, Z.; Chian, R. Mitochondria in oocyte aging: Current understanding. Facts Views Vis. ObGyn 2017, 9, 29–38. [Google Scholar] [PubMed]
- Mahutte, N.G.; Arici, A. Endometriosis and assisted reproductive technologies: Are outcomes affected? Curr. Opin. Obstet. Gynecol. 2001, 13, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Van Langendonckt, A.; Casanas-Roux, F.; Donnez, J. Oxidative stress and peritoneal endometriosis. Fertil. Steril. 2002, 77, 861–870. [Google Scholar] [CrossRef]
- Bedaiwy, M.; Falcone, T. Peritoneal fluid environment in endometriosis. Clinicopathological implications. Minerva Ginecol. 2003, 55, 333–345. [Google Scholar] [PubMed]
- Gupta, S.; Agarwal, A.; Krajcir, N.; Alvarez, J.G. Role of oxidative stress in endometriosis. Reprod. Biomed. Online 2006, 13, 126–134. [Google Scholar] [CrossRef]
- Szczepańska, M.; Koźlik, J.; Skrzypczak, J.; Mikołajczyk, M. Oxidative stress may be a piece in the endometriosis puzzle. Fertil. Steril. 2003, 79, 1288–1293. [Google Scholar] [CrossRef]
- Portz, D.M.; Elkins, T.E.; White, R.; Warren, J.; Adadevoh, S.; Randolph, J. Oxygen free radicals and pelvic adhesion formation: I. Blocking oxygen free radical toxicity to prevent adhesion formation in an endometriosis model. Int. J. Fertil. 1991, 36, 39–42. [Google Scholar]
- Polak, G.; Kozioł-Montewka, M.; Gogacz, M.; Błaszkowska, I.; Kotarski, J. Total antioxidant status of peritoneal fluid in infertile women. Eur. J. Obstet. Gynecol. Reprod. Biol. 2001, 94, 261–263. [Google Scholar] [CrossRef]
- Murphy, A.A.; Palinski, W.; Rankin, S.; Morales, A.J.; Parthasarathy, S. Evidence for Oxidatively Modified Lipid-Protein Complexes in Endometrium and Endometriosis. Fertil. Steril. 1998, 69, 1092–1094. [Google Scholar] [CrossRef]
- Khorram, O.; Lessey, B.A. Alterations in expression of endometrial endothelial nitric oxide synthase and alpha(v)beta(3) integrin in women with endometriosis. Fertil. Steril. 2002, 78, 860–864. [Google Scholar] [CrossRef]
- Wu, M.-Y. Nitric oxide synthesis is increased in the endometrial tissue of women with endometriosis. Hum. Reprod. 2003, 18, 2668–2671. [Google Scholar] [CrossRef] [PubMed]
- Ota, H.; Igarashi, S.; Hatazawa, J.; Tanaka, T. Endothelial nitric oxide synthase in the endometrium during the menstrual cycle in patients with endometriosis and adenomyosis. Fertil. Steril. 1998, 69, 303–308. [Google Scholar] [CrossRef]
- Ota, H.; Igarashi, S.; Hatazawa, J.; Tanaka, T. Endometriosis and Free Radicals. Gynecol. Obstet. Investig. 1999, 48, 29–35. [Google Scholar] [CrossRef]
- Kamada, Y.; Nakatsuka, M.; Asagiri, K.; Noguchi, S.; Habara, T.; Takata, M.; Kudo, T. GnRH agonist-suppressed expression of nitric oxide synthases and generation of peroxynitrite in adenomyosis. Hum. Reprod. 2000, 15, 2512–2519. [Google Scholar] [CrossRef]
- Nuojua-Huttunen, S.; Tomas, C.; Bloigu, R.; Tuomivaara, L.; Martikainen, H. Intrauterine insemination treatment in subfertility: An analysis of factors affecting outcome. Hum. Reprod. 1999, 14, 698–703. [Google Scholar] [CrossRef]
- ESHRE, T.R.; ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil. Steril. 2004, 81, 19–25. [Google Scholar]
- Talbott, E.O.; Guzick, D.S.; Sutton-Tyrrell, K.; McHugh-Pemu, K.P.; Zborowski, J.V.; Remsberg, K.E.; Kuller, L.H. Evidence for Association between Polycystic Ovary Syndrome and Premature Carotid Atherosclerosis in Middle-Aged Women. Arter. Thromb. Vasc. Biol. 2000, 20, 2414–2421. [Google Scholar] [CrossRef]
- Fedorcsák, P.; Poleć, A.; Ráki, M.; Holm, R.; Jebsen, P.; Åbyholm, T. Differential Release of Matrix Metalloproteinases and Tissue Inhibitors of Metalloproteinases by Human Granulosa-Lutein Cells and Ovarian Leukocytes. Endocrinology 2010, 151, 1290–1298. [Google Scholar] [CrossRef][Green Version]
- Surazynski, A.; Liu, Y.; Miltyk, W.; Phang, J.M. Nitric oxide regulates prolidase activity by serine/threonine phosphorylation. J. Cell. Biochem. 2005, 96, 1086–1094. [Google Scholar] [CrossRef]
- Visse, R.; Nagase, H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: Structure, function, and biochemistry. Circ. Res. 2003, 92, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Lahav-Baratz, S.; Kraiem, Z.; Shiloh, H.; Koifman, M.; Ishai, D.; Dirnfeld, M. Decreased expression of tissue inhibitor of matrix metalloproteinases in follicular fluid from women with polycystic ovaries compared with normally ovulating patients undergoing in vitro fertilization. Fertil. Steril. 2003, 79, 567–571. [Google Scholar] [CrossRef]
- Baka, S.; Zourla, K.; Kouskouni, E.; Makrakis, E.; Demeridou, S.; Tzanakaki, D.; Hassiakos, D.; Creatsas, G. Matrix metalloproteinases 2 and 9 and their tissue inhibitors in the follicular fluid of patients with polycystic ovaries undergoing in vitro fertilisation. In Vivo 2010, 24, 293–296. [Google Scholar]
- Murri, M.; Luque-Ramírez, M.; Insenser, M.; Ojeda, M.; Escobar-Morreale, H.F. Circulating markers of oxidative stress and polycystic ovary syndrome (PCOS): A systematic review and meta-analysis. Hum. Reprod. Update 2013, 19, 268–288. [Google Scholar] [CrossRef] [PubMed]
- Zuo, T.; Zhu, M.; Xu, W. Roles of Oxidative Stress in Polycystic Ovary Syndrome and Cancers. Oxidative Med. Cell. Longev. 2016, 2016, 8589318. [Google Scholar] [CrossRef] [PubMed]
- Hilali, N.; Vural, M.; Camuzcuoglu, H.; Camuzcuoglu, A.; Aksoy, N. Increased prolidase activity and oxidative stress in PCOS. Clin. Endocrinol. 2013, 79, 105–110. [Google Scholar] [CrossRef]
- Liu, B.; Cai, L.-Y.; Lv, H.-M.; Xia, L.; Zhang, Y.-J.; Zhang, H.-X.; Guan, Y.-M. Raised serum levels of matrix metalloproteinase-9 in women with polycystic ovary syndrome and its association with insulin-like growth factor binding protein-1. Gynecol. Endocrinol. 2008, 24, 285–288. [Google Scholar] [CrossRef]
- He, C.; Lin, Z.; Robb, S.W.; Ezeamama, A.E. Serum Vitamin D Levels and Polycystic Ovary syndrome: A Systematic Review and Meta-Analysis. Nutrients 2015, 7, 4555–4577. [Google Scholar] [CrossRef]
- Krul-Poel, Y.H.M.; Snackey, C.; Louwers, Y.; Lips, P.; Lambalk, C.B.; Laven, J.S.E.; Simsek, S. The role of vitamin D in metabolic disturbances in polycystic ovary syndrome: A systematic review. Eur. J. Endocrinol. 2013, 169, 853–865. [Google Scholar] [CrossRef]
- Thomson, R.L.; Spedding, S.; Buckley, J. Vitamin D in the aetiology and management of polycystic ovary syndrome. Clin. Endocrinol. 2012, 77, 343–350. [Google Scholar] [CrossRef]
- Merhi, Z.; Doswell, A.; Krebs, K.; Cipolla, M. Vitamin D Alters Genes Involved in Follicular Development and Steroidogenesis in Human Cumulus Granulosa Cells. J. Clin. Endocrinol. Metab. 2014, 99, E1137–E1145. [Google Scholar] [CrossRef] [PubMed]
- Parikh, G.; Varadinova, M.; Suwandhi, P.; Araki, T.; Rosenwaks, Z.; Poretsky, L.; Seto-Young, D. Vitamin D Regulates Steroidogenesis and Insulin-like Growth Factor Binding Protein-1 (IGFBP-1) Production in Human Ovarian Cells. Horm. Metab. Res. 2010, 42, 754–757. [Google Scholar] [CrossRef]
- Yao, X.; Zhang, G.; Guo, Y.; Ei-Samahy, M.; Wang, S.; Wan, Y.; Han, L.; Liu, Z.; Wang, F.; Zhang, Y. Vitamin D receptor expression and potential role of vitamin D on cell proliferation and steroidogenesis in goat ovarian granulosa cells. Theriogenology 2017, 102, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Ni, K.; Cai, Y.; Shang, J.; Zhang, X.; Xiong, C. Effect of vitamin D supplementation on polycystic ovary syndrome: A systematic review and meta-analysis of randomized controlled trials. Complementary Ther. Clin. Pract. 2017, 26, 53–60. [Google Scholar] [CrossRef]
- Firouzabadi, R.D.; Aflatoonian, A.; Modarresi, S.; Sekhavat, L.; MohammadTaheri, S. Therapeutic effects of calcium & vitamin D supplementation in women with PCOS. Complementary Ther. Clin. Pract. 2012, 18, 85–88. [Google Scholar] [CrossRef]
- Pal, L.; Berry, A.; Coraluzzi, L.; Kustan, E.; Danton, C.; Shaw, J.; Taylor, H. Therapeutic implications of vitamin D and calcium in overweight women with polycystic ovary syndrome. Gynecol. Endocrinol. 2012, 28, 965–968. [Google Scholar] [CrossRef] [PubMed]
- Masjedi, F.; Keshtgar, S.; Zal, F.; Talaei-Khozani, T.; Sameti, S.; Fallahi, S.; Kazeroni, M. Effects of vitamin D on steroidogenesis, reactive oxygen species production, and enzymatic antioxidant defense in human granulosa cells of normal and polycystic ovaries. J. Steroid Biochem. Mol. Biol. 2020, 197, 105521. [Google Scholar] [CrossRef] [PubMed]
- Tatone, C. Oocyte senescence: A firm link to age-related female subfertility. Gynecol. Endocrinol. 2008, 24, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Balasch, J.; Gratacós, E. Delayed childbearing: Effects on fertility and the outcome of pregnancy. Curr. Opin. Obstet. Gynecol. 2012, 24, 187–193. [Google Scholar] [CrossRef]
- Hassold, T.; Hall, H.; Hunt, P. The origin of human aneuploidy: Where we have been, where we are going. Hum. Mol. Genet. 2007, 16, R203–R208. [Google Scholar] [CrossRef]
- Petrek, J.A.; Naughton, M.J.; Case, L.D.; Paskett, E.D.; Naftalis, E.Z.; Singletary, S.E.; Sukumvanich, P. Incidence, Time Course, and Determinants of Menstrual Bleeding After Breast Cancer Treatment: A Prospective Study. J. Clin. Oncol. 2006, 24, 1045–1051. [Google Scholar] [CrossRef]
- Letourneau, J.M.; Ebbel, E.E.; Katz, P.P.; Katz, A.; Ai, W.Z.; Chien, A.J.; Melisko, M.E.; Cedars, M.I.; Rosen, M.P. Pretreatment fertility counseling and fertility preservation improve quality of life in reproductive age women with cancer. Cancer 2012, 118, 1710–1717. [Google Scholar] [CrossRef]
- Meirow, D.; Biederman, H.; Anderson, R.A.; Wallace, W.H.B. Toxicity of Chemotherapy and Radiation on Female Reproduction. Clin. Obstet. Gynecol. 2010, 53, 727–739. [Google Scholar] [CrossRef]
- Barritt, J.A.; Cohen, J.; Brenner, C.A. Mitochondrial DNA point mutation in human oocytes is associated with maternal age. Reprod. Biomed. Online 2000, 1, 96–100. [Google Scholar] [CrossRef]
- Konstantinidis, M.; Alfarawati, S.; Hurd, D.; Paolucci, M.; Shovelton, J.; Fragouli, E.; Wells, D. Simultaneous assessment of aneuploidy, polymorphisms, and mitochondrial DNA content in human polar bodies and embryos with the use of a novel microarray platform. Fertil. Steril. 2014, 102, 1385–1392. [Google Scholar] [CrossRef]
- Suganuma, N.; Kitagawa, T.; Nawa, A.; Tomoda, Y. Human Ovarian Aging and Mitochondrial DNA Deletion. Horm. Res. 1993, 39 (Suppl. 1), 16–21. [Google Scholar] [CrossRef]
- Traver, S.; Scalici, E.; Mullet, T.; Molinari, N.; Vincens, C.; Anahory, T.; Hamamah, S. Cell-free DNA in Human Follicular Microenvironment: New Prognostic Biomarker to Predict in vitro Fertilization Outcomes. PLoS ONE 2015, 10, e0136172. [Google Scholar] [CrossRef] [PubMed]
- Bentov, Y.; Yavorska, T.; Esfandiari, N.; Jurisicova, A.; Casper, R.F. The contribution of mitochondrial function to reproductive aging. J. Assist. Reprod. Genet. 2011, 28, 773–783. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.W.; Keegan, T.; Hamilton, A.; Lynch, C.; Wu, X.-C.; Schwartz, S.M.; Kato, I.; Cress, R.; Harlan, L.; AYA HOPE Study Collaborative Group. Understanding care and outcomes in adolescents and young adults with cancer: A review of the AYA HOPE study. Pediatr. Blood Cancer 2019, 66, e27486. [Google Scholar] [CrossRef] [PubMed]
- Wallace, W.H.B.; Thomson, A.B.; Kelsey, T. The radiosensitivity of the human oocyte. Hum. Reprod. 2003, 18, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, S.W.; Han, S.J.; Lee, S.; Park, H.T.; Song, J.Y.; Kim, T. Molecular Mechanism and Prevention Strategy of Chemotherapy- and Radiotherapy-Induced Ovarian Damage. Int. J. Mol. Sci. 2021, 22, 7484. [Google Scholar] [CrossRef]
- Wallace, W.H.B.; Thomson, A.B.; Saran, F.; Kelsey, T.W. Predicting age of ovarian failure after radiation to a field that includes the ovaries. Int. J. Radiat. Oncol. 2005, 62, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Adriaens, I.; Smitz, J.; Jacquet, P. The current knowledge on radiosensitivity of ovarian follicle development stages. Hum. Reprod. Update 2009, 15, 359–377. [Google Scholar] [CrossRef] [PubMed]
- Stroud, J.S.; Mutch, D.; Rader, J.; Powell, M.; Thaker, P.H.; Grigsby, P.W. Effects of cancer treatment on ovarian function. Fertil. Steril. 2009, 92, 417–427. [Google Scholar] [CrossRef]
- Thomson, A.B.; Critchley, H.O.; Wallace, W.H. Fertility and progeny. Eur. J. Cancer 2002, 38, 1634–1644. [Google Scholar] [CrossRef]
- Chemaitilly, W.; Mertens, A.C.; Mitby, P.; Whitton, J.; Stovall, M.; Yasui, Y.; Robison, L.L.; Sklar, C.A. Acute Ovarian Failure in the Childhood Cancer Survivor Study. J. Clin. Endocrinol. Metab. 2006, 91, 1723–1728. [Google Scholar] [CrossRef] [PubMed]
- Kalich-Philosoph, L.; Roness, H.; Carmely, A.; Fishel-Bartal, M.; Ligumsky, H.; Paglin, S.; Wolf, I.; Kanety, H.; Sredni, B.; Meirow, D. Cyclophosphamide Triggers Follicle Activation and “Burnout”; AS101 Prevents Follicle Loss and Preserves Fertility. Sci. Transl. Med. 2013, 5, 185ra62. [Google Scholar] [CrossRef]
- Ben-Aharon, I.; Bar-Joseph, H.; Tzarfaty, G.; Kuchinsky, L.; Rizel, S.; Stemmer, S.M.; Shalgi, R. Doxorubicin-induced ovarian toxicity. Reprod. Biol. Endocrinol. 2010, 8, 20. [Google Scholar] [CrossRef]
- Spears, N.; Lopes, F.; Stefansdottir, A.; Rossi, V.; De Felici, M.; Anderson, R.A.; Klinger, F.G. Ovarian damage from chemotherapy and current approaches to its protection. Hum. Reprod. Update 2019, 25, 673–693. [Google Scholar] [CrossRef]
- Roness, H.; Kashi, O.; Meirow, D. Prevention of chemotherapy-induced ovarian damage. Fertil. Steril. 2016, 105, 20–29. [Google Scholar] [CrossRef]
- Utsunomiya, T.; Tanaka, T.; Utsunomiya, H.; Umesaki, N. A novel molecular mechanism for anticancer drug-induced ovarian failure: Irinotecan HCl, an anticancer topoisomerase I inhibitor, induces specific FasL expression in granulosa cells of large ovarian follicles to enhance follicular apoptosis. Int. J. Oncol. 2008, 32, 991–1000. [Google Scholar] [CrossRef][Green Version]
- Blumenfeld, Z.; von Wolff, M. GnRH-analogues and oral contraceptives for fertility preservation in women during chemotherapy. Hum. Reprod. Update 2008, 14, 543–552. [Google Scholar] [CrossRef]
- Molina, J.R.; Barton, D.L.; Loprinzi, C.L. Chemotherapy-induced ovarian failure: Manifestations and management. Drug Saf. 2005, 28, 401–416. [Google Scholar] [CrossRef]
- Basu, A.K. DNA Damage, Mutagenesis and Cancer. Int. J. Mol. Sci. 2018, 19, 970. [Google Scholar] [CrossRef]
- Meeker, J.D.; Ehrlich, S.; Toth, T.L.; Wright, D.L.; Calafat, A.M.; Trisini, A.T.; Ye, X.; Hauser, R. Semen quality and sperm DNA damage in relation to urinary bisphenol A among men from an infertility clinic. Reprod. Toxicol. 2010, 30, 532–539. [Google Scholar] [CrossRef]
- Tang, Z.-R.; Zhang, R.; Lian, Z.-X.; Deng, S.-L.; Yu, K. Estrogen-Receptor Expression and Function in Female Reproductive Disease. Cells 2019, 8, 1123. [Google Scholar] [CrossRef]
- Brieño-Enríquez, M.; Robles, P.; Camats-Tarruella, N.; García-Cruz, R.; Roig, I.; Cabero, L.; Martínez, F.; Caldés, M.G. Human meiotic progression and recombination are affected by Bisphenol A exposure during in vitro human oocyte development. Hum. Reprod. 2011, 26, 2807–2818. [Google Scholar] [CrossRef]
- Brieño-Enriquez, M.A.; Reig-Viader, R.; Cabero, L.; Toran, N.; Martinez, F.; Roig, I.; Caldes, M.G. Gene expression is altered after bisphenol A exposure in human fetal oocytes in vitro. Mol. Hum. Reprod. 2012, 18, 171–183. [Google Scholar] [CrossRef]
- Shamsuddin, A.; Gan, R. Immunocytochemical localization of benzo(a)pyrene-DNA adducts in human tissues. Hum. Pathol. 1988, 19, 309–315. [Google Scholar] [CrossRef]
- Lieber, M.R. The Mechanism of Double-Strand DNA Break Repair by the Nonhomologous DNA End-Joining Pathway. Annu. Rev. Biochem. 2010, 79, 181–211. [Google Scholar] [CrossRef]
- Longhese, M.P.; Bonetti, D.; Guerini, I.; Manfrini, N.; Clerici, M. DNA double-strand breaks in meiosis: Checking their formation, processing and repair. DNA Repair 2009, 8, 1127–1138. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.-X.; Manchester, C.; Burillo, S.L.; Juan, M.S.; Mayo, C. Melatonin: Detoxification of Oxygen And Nitrogen-Based Toxic Reactants. Dev. Tryptophan Serotonin Metab. 2003, 527, 539–548. [Google Scholar] [CrossRef]
- Tamura, H.; Jozaki, M.; Tanabe, M.; Shirafuta, Y.; Mihara, Y.; Shinagawa, M.; Tamura, I.; Maekawa, R.; Sato, S.; Taketani, T.; et al. Importance of Melatonin in Assisted Reproductive Technology and Ovarian Aging. Int. J. Mol. Sci. 2020, 21, 1135. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Escames, G.; Venegas, C.; Díaz-Casado, M.E.; Lima-Cabello, E.; Lopez, L.C.; Rosales-Corral, S.; Tan, D.-X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation, and potential functions. Cell. Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef]
- Niles, L.; Wang, J.; Shen, L.; Lobb, D.; Younglai, E. Melatonin receptor mRNA expression in human granulosa cells. Mol. Cell. Endocrinol. 1999, 156, 107–110. [Google Scholar] [CrossRef]
- Yang, L.; Chen, Y.; Liu, Y.; Xing, Y.; Miao, C.; Zhao, Y.; Chang, X.; Zhang, Q. The Role of Oxidative Stress and Natural Antioxidants in Ovarian Aging. Front. Pharmacol. 2020, 11, 617843. [Google Scholar] [CrossRef] [PubMed]
- Al-Zubaidi, U.; Adhikari, D.; Cinar, O.; Zhang, Q.-H.; Yuen, W.S.; Murphy, M.P.; Rombauts, L.; Robker, R.L.; Carroll, J. Mitochondria-targeted therapeutics, MitoQ and BGP-15, reverse aging-associated meiotic spindle defects in mouse and human oocytes. Hum. Reprod. 2021, 36, 771–784. [Google Scholar] [CrossRef]
- Cajas, Y.N.; Cañón-Beltrán, K.; De Guevara, M.L.; De La Blanca, M.G.M.; Ramos-Ibeas, P.; Gutiérrez-Adán, A.; Rizos, D.; González, E.M. Antioxidant Nobiletin Enhances Oocyte Maturation and Subsequent Embryo Development and Quality. Int. J. Mol. Sci. 2020, 21, 5340. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Takasaki, A.; Miwa, I.; Taniguchi, K.; Maekawa, R.; Asada, H.; Taketani, T.; Matsuoka, A.; Yamagata, Y.; Shimamura, K.; et al. Oxidative stress impairs oocyte quality and melatonin protects oocytes from free radical damage and improves fertilization rate. J. Pineal Res. 2008, 44, 280–287. [Google Scholar] [CrossRef]
- Asma, A.; Marc-André, S. Melatonin Signaling Pathways Implicated in Metabolic Processes in Human Granulosa Cells (KGN). Int. J. Mol. Sci. 2022, 23, 2988. [Google Scholar] [CrossRef]
| Endometriosis | PCOS | Advanced Age | After Cancer Treatment |
|---|---|---|---|
|
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Immediata, V.; Ronchetti, C.; Spadaro, D.; Cirillo, F.; Levi-Setti, P.E. Oxidative Stress and Human Ovarian Response—From Somatic Ovarian Cells to Oocytes Damage: A Clinical Comprehensive Narrative Review. Antioxidants 2022, 11, 1335. https://doi.org/10.3390/antiox11071335
Immediata V, Ronchetti C, Spadaro D, Cirillo F, Levi-Setti PE. Oxidative Stress and Human Ovarian Response—From Somatic Ovarian Cells to Oocytes Damage: A Clinical Comprehensive Narrative Review. Antioxidants. 2022; 11(7):1335. https://doi.org/10.3390/antiox11071335
Chicago/Turabian StyleImmediata, Valentina, Camilla Ronchetti, Daria Spadaro, Federico Cirillo, and Paolo Emanuele Levi-Setti. 2022. "Oxidative Stress and Human Ovarian Response—From Somatic Ovarian Cells to Oocytes Damage: A Clinical Comprehensive Narrative Review" Antioxidants 11, no. 7: 1335. https://doi.org/10.3390/antiox11071335
APA StyleImmediata, V., Ronchetti, C., Spadaro, D., Cirillo, F., & Levi-Setti, P. E. (2022). Oxidative Stress and Human Ovarian Response—From Somatic Ovarian Cells to Oocytes Damage: A Clinical Comprehensive Narrative Review. Antioxidants, 11(7), 1335. https://doi.org/10.3390/antiox11071335





