Abstract
β-thalassemia and sickle cell disease (SCD) are inherited hemoglobinopathies that result in both quantitative and qualitative variations in the β-globin chain. These in turn lead to instability in the generated hemoglobin (Hb) or to a globin chain imbalance that affects the oxidative environment both intracellularly and extracellularly. While oxidative stress is not among the primary etiologies of β-thalassemia and SCD, it plays a significant role in the pathogenesis of these diseases. Different mechanisms exist behind the development of oxidative stress; the result of which is cytotoxicity, causing the oxidation of cellular components that can eventually lead to cell death and organ damage. In this review, we summarize the mechanisms of oxidative stress development in β-thalassemia and SCD and describe the current and potential antioxidant therapeutic strategies. Finally, we discuss the role of targeted therapy in achieving an optimal redox balance.
1. Introduction
The oxidative status of cells is dependent on the balance between oxidants and antioxidants. This balance is crucial to achieving normal physiology and maintaining cellular homeostasis. Thus, the thinning control of reactive oxygen species (ROS) is extremely important since low levels of ROS might participate in the signaling pathways involved in the differentiation and proliferation of the erythroid precursors [1,2,3,4].
Oxidative stress can manifest in several pathologies when the balance between oxidants and antioxidants is broken, as evident in β-thalassemia and sickle cell disease (SCD). Excessive levels of ROS can lead to cytotoxicity as these radicals often bind to cellular components such as proteins, membrane lipids, and DNA. Different mechanisms exist behind oxidative stress development. These include the accumulation of α-globin chains and free iron as seen in β-thalassemia or the cyclic polymerization/depolymerization of hemoglobin S (HbS) as seen in SCD. Thus, the activation and expression of potent anti-oxidant machinery are required to ensure the proper maturation of erythroid precursors and the survival of red blood cells (RBCs) in the peripheral circulation [5,6]. While more studies are necessary to better understand the role and mechanism of action of antioxidant agents, they have shown to be effective in improving the pathological manifestations of β-thalassemia or SCD by re-balancing the cellular redox state. Different molecules have been tested and these have acted as endogenous or as exogenous antioxidants that scavenge and inactivate ROS, leading to cellular protection against oxidation.
Progress in the knowledge of oxidation and cellular damages in β-thalassemia and SCD may lead to the identification of new antioxidant therapies that can prevent or delay the development of organ complications behind erythroid cells in patients. In this review, we summarize the mechanisms of oxidation in β-thalassemia and SCD and describe the current and potential antioxidant therapeutic strategies. Finally, we discuss the role of targeted therapy in achieving an optimal redox balance in these chronic invalidating disorders.
2. Evolutionary Perspective of a Redox Balance
Reactive oxygen species are chemically reactive molecules that are formed as a by-product of different cellular metabolic reactions. They include hydrogen peroxide (H2O2), superoxide free radicals (O2•−), as well as nitrogen-based free radical species such as peroxynitrite (ONOO−) or nitric oxide (NO) [7,8]. Reactive oxygen species are significant cellular entities that play a role in cellular proliferation, signal transduction, host defense mechanisms, homeostatic preservation, and gene expression [9].
Being heritable biological adaptations, ROS have evolved concurrently with natural and environmental modifications. Such modifications have been crucial for better understanding their role and mechanisms of action while providing insights into the evolutionarily preferred mechanistic physiologies of cells. One strategy of major significance in physiological functions is that cellular events revolve around fixed and coordinated set points. This phenomenon is physiologically referred to as homeostasis, and ROS are tightly counterbalanced by various cellular antioxidants to promote optimal redox homeostasis [10]. In fact, all cells possess effective antioxidant mechanisms that neutralize and remove ROS. Moreover, several enzyme systems present in our bodies neutralize ROS by metabolic conversion [11].
Reactive oxygen species are recognized as specific mediators and second messengers of cell signaling that play a role in vascular tone, immune responses, cell protection, and hormonal actions [12,13,14]. In sync with this homeostatic balance, these functions are primarily maintained and counter-balanced by anti-oxidative mechanisms that regulate the bioavailability of oxidative species. However, under pathological conditions, oxidant radicals lead to a state of oxidative stress. In cells, this results in the malfunctioning of many organelles, particularly the membrane, and may lead to cytotoxicity and eventually organ damage and failure [8].
Several reports have indicated increased ROS levels to be directly correlated with the irreversible oxidation of cellular components that eventually contribute to cellular dysfunction and necrosis [15]. Many studies have validated the implications of redox alternations in distinct pathologies such as diabetes and cancer. However, attempts to reduce bioactive ROS to very minimal levels have been shown to be detrimental because in lower concentrations these ROS can act as signaling molecules [16]. This highlights the significance of the redox system in cellular physiology and homeostatic balance between oxidant and antioxidant molecules [17]. Extensive research is currently aiming at identifying specific cellular sources of ROS production and those that are specifically altered in a cell- and disease-specific manner.
3. Sources of Reactive Oxygen Species in Red Blood Cells
Erythroid precursors and erythrocytes are unique cells since they are exposed to cyclic oxygenation/deoxygenation (auto-oxidation) events as long-term survival cells, and they require iron for hemoglobin (Hb) synthesis. Thus, anti-oxidant and cytoprotective systems are crucial for erythroid cell homeostasis. The process of auto-oxidation (from oxygenated Hb to methemoglobin) plays a major role in the generation of ROS inside RBCs [18,19]. This process is characterized by the rapid conversion of O2•− to H2O2 [19,20]. In β-thalassemia and SCD RBCs, Hb auto-oxidation is more pronounced as the Hb molecules in these diseases are highly unstable [19]. Moreover, in SCD, the Hb auto-oxidation process is further exacerbated under hypoxic conditions in the microcirculation and leads to the formation of unstable dimers at reduced Hb concentrations [19].
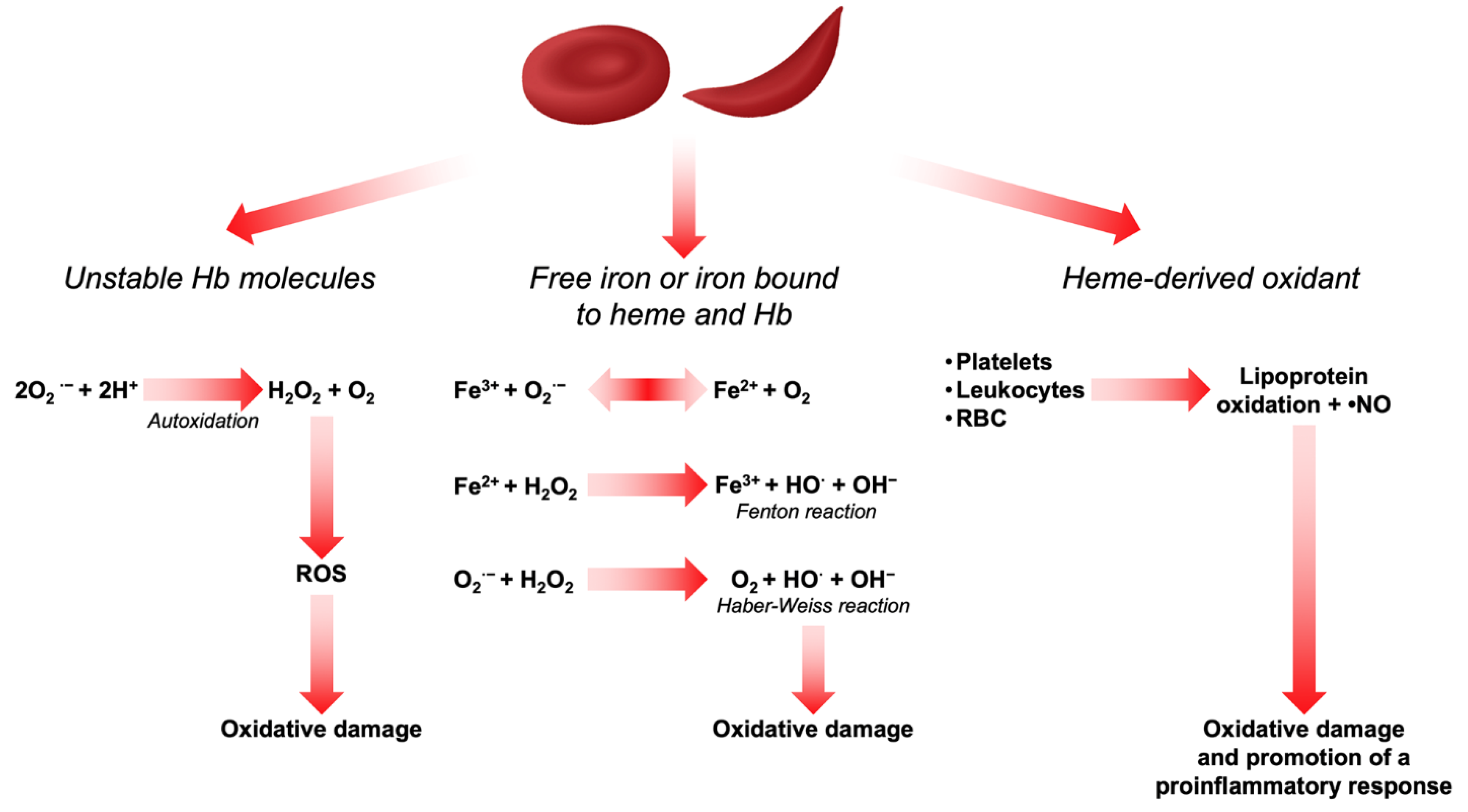
Heme and iron are also highly oxidizing agents and sources of ROS in RBCs. Whether iron is in its free form or bound to heme and Hb, it can act as a Fenton reagent in the Haber–Weiss cycle, thereby generating hydroxyl radicals (•OH) and promoting extensive oxidative damage [21,22]. Being a hydrophobic molecule, heme interacts with proteins and membrane lipids and thus promotes a series of oxidation reactions [23]. On the other hand, free iron bound to the RBC membrane has been described to participate in the oxidation of membrane proteins and lipid components, thereby affecting their mechanical properties. This increases erythrophagocytosis and contributes to the reduction in RBC survival in peripheral circulation [24,25,26,27]. In β-thalassemia and in SCD, chronic hemolysis overcomes physiologic buffer systems such as haptoglobin and hemopexin and results in increased plasma free Hb and heme [28,29]. Oxidants derived from heme can induce the recruitment of platelets, leukocytes, and RBCs to the vessel wall and produce lipoprotein oxidation and consume •NO to form strong oxidants [30]. In vascular endothelial cells, this activates the transcription factor nuclear factor-κB (NF-κB) which is redox-sensitive and promotes a proinflammatory response by binding to receptors, enzymes, and transcription factors that alter cell metabolism, cell function, and gene expression [31]. All the above-mentioned mechanisms of ROS production are depicted in Figure 1.
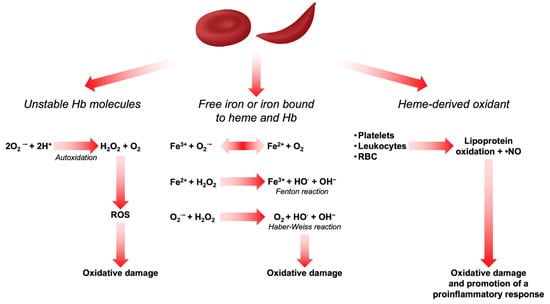
Figure 1.
Sources of ROS in RBCs.
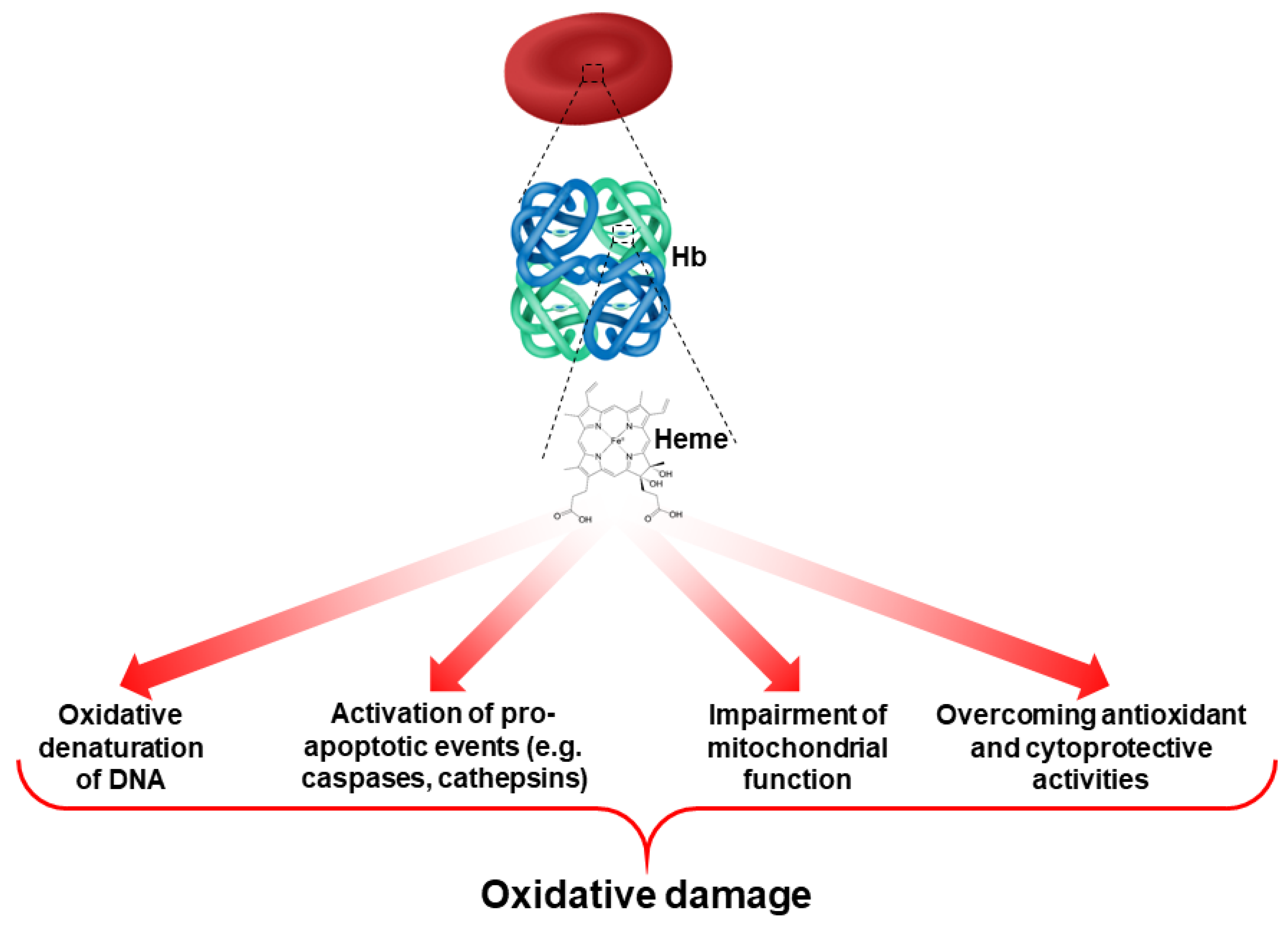
In normal erythropoiesis, heme biosynthesis is a key event in Hb production. In β-thalassemic erythropoiesis, different studies have shown an early phase of heme accumulation, followed by the activation of antioxidant and cytoprotective systems that generate a second phase of relative heme deficiency [3]. This is important since heme can act as a source of ROS in RBCs and cause oxidative damage [19,32,33] (Figure 2). Thus, prolonged and severe oxidation, due to the combination of abnormal heme biosynthesis and the accumulation of alpha-globin chains play a crucial role in the ineffective erythropoiesis of β-thalassemia.
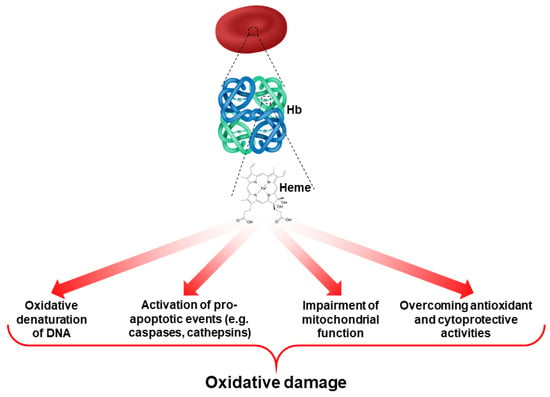
Figure 2.
Role of heme as a contributor to ROS production and oxidative damage in RBCs.
4. Oxidative Damage to Intracellular Components in Red Blood Cells
4.1. Oxidative Damage to Membrane-Cytoskeleton Proteins
Oxidative stress also has an effect on the overall cytoskeletal network and its associated proteins [34,35]. One study conducted on β-thalassemia RBCs showed that spectrins might be targeted by oxidation. This perturbates their interactions with other cytoskeletal proteins such as actin or with multiprotein complexes bridging the membrane to the cytoskeleton such as protein 4.1 or band 3 [35]. Consequently, impaired stability in the interaction between the cytoskeleton and the cellular membrane becomes evident. It is noteworthy to mention that the oxidation-induced and abnormal clusterization of band 3 contributes to increased RBC fragility and the generation of erythroid microvesicles which contributes to the pro-coagulant phenotype of both β-thalassemia and SCD [24,36,37].
Several cytoskeletal proteins in RBCs derived from SCD patients have been shown to undergo oxidation-mediated post-translational protein modifications (PTPM). Additionally, RBCs from transgenic SCD mice revealed that these irreversible PTPM detected in HbS molecules were found in the β-chain and included the ubiquitination of Lys96 and Lys145 and the irreversible oxidation of Cys93 and [38,39]. This ubiquitination process in HbS molecules occurs as a result of the accumulation of oxidatively damaged HbS molecules in RBCs as well as in microparticles and could likely be due to the redox imbalance-dependent proteasomal inhibition in SCD. A recent study conducted on RBCs and microparticle proteomes from SCD patients showed increased ubiquitination and phosphorylation of cytoskeletal proteins when compared to control cells [39]. Remarkably, these PTPM have been identified in spectrin, ankyrin, band 3, carbonic anhydrase, and band 4.1 [39]. Moreover, an increase in ROS production by auto-oxidized HbS led to an increase in the accumulation of oxidative lesions by membrane components. These, in turn, can degrade polyunsaturated lipids and thus lead to the formation of malondialdehyde as a by-product and damage proteins localized in the region near membrane-associated Hb [40]. Among PTPM, oxidation might also activate tyrosine kinases such as Syk or Lyn, a kinase of the Src family. Both Syk and Lyn target RBC membrane proteins such as band 3, contributing to both protein conformation state and protein–protein interaction [2,25,36,41,42]. This is important for the mechanical properties of the membrane and the volume/surface regulation of pathologic RBCs.
4.2. Oxidative Damage to Membrane Lipids
Lipid peroxidation and protein oxidation disturb the organization of the lipid mem-brane and lead to cellular deformability [43]. This disruption causes phosphatidylserine (PS) to be exposed to the outer membrane of the cell which in turn signals macrophages to engulf and degrade the PS-exposed RBCs [44], a phenomenon seen in β-thalassemia patients [44]. The exposure of PS and subsequent macrophage degradation is the mechanism of RBC removal during eryptosis. Eryptosis is the suicidal death process of RBCs that occurs prior to senescence and after injury. This phenomenon is characterized by cell shrinkage and the loss of membrane organization and can be further exacerbated by several factors, including oxidative stress.
As for SCD RBCs, the high rate of intracellular ROS production in addition to the presence of auto-oxidizing HbS in the cell membrane leads to the oxidative damage of membrane lipids and the loss of membrane lipid asymmetry. This alters membrane surface properties and permeability and exposes PS [45]. The externalization of PS is a critical event in the disease progression [46]. It is considered a key step in the premature senescence process, favoring the removal of RBCs from circulation and the release of erythroid microvesicles. This leads to a state of chronic anemia as well as endothelial dysfunction in SCD [47]. The pathophysiology of SCD is also characterized by increased plasma levels of secretory phospholipase A2 (sPLA2), a powerful inflammatory mediator that can selectively hydrolyze phospholipids in RBCs exposing PS, promoting their hemolysis [48,49]. The activation of sPLA2 also generates phospholipid breakdown products that can affect overall endothelial function. Moreover, PS-exposing RBCs can also activate phospholipase D, which catalyzes the hydrolysis of phosphatidylcholine into phosphatidic acid and choline [50].
5. A Focus on Oxidative Stress in β-Thalassemia
5.1. Oxidative Stress and Hemoglobin
β-thalassemia is characterized by the unbalanced production of globin chains, resulting in excess free α-globins [51]. These unstable α-globins tend to auto-oxidize, denature, and precipitate as hemichromes [52,53]. Hemichromes then bind to the cytoplasmic domain of band 3 and mediate the oxidative cross-linking through disulfide bonds. Subsequently, both heme and free iron are released and globin proteins precipitate. This initiates a self-amplifying redox reaction that oxidizes additional Hb molecules, depletes cellular reduction potential, and triggers phosphorylative responses, which lead to membrane destabilization and the acceleration of RBC removal by splenic macrophages [51,54,55]. In fact, deoxyhemoglobin (deoxyHb) binds avidly and reversibly to band-3 [56]. Because band 3 is associated with multiple metabolic, solute transport, signal transduction, and structural proteins, the oxygen-dependent regulation of erythrocyte properties is mediated by the reversible association of deoxyHb with band 3 [56].
5.2. Oxidative Stress and Ineffective Erythropoiesis
Ineffective erythropoiesis is one of the main pathophysiological culprits in β-thalassemia. The impaired ratio between α and β subunits results in the accumulation of unbounded α-chains during erythroblast maturation. These bind heme and eventually form hemichromes, which precipitate and bind to the plasma membrane [57,58]. Evidence of oxidative stress due to ineffective erythropoiesis comes from studies conducted on bone marrow cells and in developing thalassemia erythroid precursor cells. Many studies conducted with bone marrow cells of β-thalassemia patients have indicated an increased number of activated macrophages, which might cope with the several damaged erythroblasts [59,60]. Interestingly, one study showed that the presence of α-globin precipitates in cells at the polychromatophilic erythroblast stage of maturation, a stage characterized by increased hemoglobinization and apoptosis in β-thalassemia erythropoiesis [61,62]. Another study conducted in a mouse model of β-thalassemia intermedia (β-TI) showed that growth differentiation factor 11 (GDF11), a transforming growth factor β (TGF-β) superfamily ligand, blocks the terminal differentiation of erythroid precursors by promoting oxidative stress and α-globin precipitation [63]. The expression of GDF11 is also induced by oxidative stress, which indicates the presence of an autocrine amplification loop that can promote ineffective erythropoiesis [63]. Oxidative stress in developing thalassemia erythroid precursors has also been associated with increased apoptosis, as manifested by the externalization of PS. This notion suggests that oxidative stress can also be responsible for the ineffective erythropoiesis itself [64].
5.3. Oxidative Stress and Iron Overload
In β-thalassemia, iron overload is one of the most common disease-related complications and is a major cause of morbidity and mortality [65,66]. In the plasma, iron is normally bound to transferrin. In β-thalassemia patients, however, iron overload saturates the ability of the transferrin iron transport system, leading to the formation of non-transferrin bound iron (NTBI) and labile plasma iron (LPI). Both NTBI and LPI circulate in plasma and subsequently become deposited inside the susceptible cells [67,68]. The long-term uptake and accumulation of these molecules can lead to high levels of storage iron and labile cellular iron [69]. When the scale of the cellular production of the labile iron pool exceeds the capacity of the cell to synthesize new ferritin molecules, a critical concentration is reached. This intracellular labile iron pool is redox-active, catalyzing the Fenton and Haber-Weiss reactions, thus generating ROS [70]. The production of ROS due to iron overload by the metabolism of NTBI plays an essential role in inducing cellular dysfunction, apoptosis, and ferroptosis [71,72,73,74]. For example, one study showed that NTBI-triggered iron overload can aggravate atherosclerosis in ApoE-/- FPNwt/C326S mice, suggesting a pro-atherogenic role for iron [75]. This finding was characterized by endothelial permeabilization, activation, dysfunction, elevation in pro-inflammatory mediators, and the induction of highly vulnerable plaques [75].
6. A Focus on Oxidative Stress in Sickle Cell Disease
6.1. Connection between Oxidation and Oxidases in SCD
In the vascular compartment of patients with SCD, the overactivation of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, xanthine oxidase (XO), and uncoupled nitric oxide synthase (eNOS) can generate ROS [76,77,78,79]. NADPH oxidase is the major O2•−-producing enzyme in RBCs, vascular endothelial cells, and leucocytes. The production of ROS by NADPH oxidase can lead to a state of hemolysis that is often associated with infections or vessel-occlusive crises [78]. The O2•− derived from this process contributes to the pro-thrombogenic and pro-inflammatory responses that are often associated with SCD [24]. Intracellularly, NADPH oxidase activity is regulated by protein kinase C and Rac GTPases in RBCs. Extracellularly, however, it is regulated by signaling factors such as TGF-β1 and endothelin-1 [79]. NADPH oxidase-induced ROS may cause direct oxidative damage to several subcellular structures. This reduces the deformability of RBCs and results in increased fragility and hemolysis [79]. On the other hand, XO is considered to be another major source of O2•− and H2O2 in RBCs. In SCD patients, the activity of XO is often increased in the plasma. Episodes of hypoxia/reoxygenation in SCD patients can excite the release of this enzyme from the liver and into the circulation. These circulating XO molecules can then bind to vessel luminal cells, impair vascular function, and create an oxidative setting [25].
6.2. Oxidative Stress and Reduced •NO Bioavailability in SCD
SCD is characterized by a local reduction in NO, resulting in vascular dysfunction and abnormal vascular tone [80,81]. This occurs as a result of intravascular hemolysis and the consumption of physiologic buffer systems such as haptoglobin and hemopexin. Indeed, free heme and free Hb bind to NO, promoting the local reduction in NO availability [82,83,84,85]. The reaction of •NO with oxygenated Hb results in the formation of methemoglobin and nitrate [86]. Additionally, superoxides bind to NO to produce ONOO−. This contributes to a decrease in NO levels and leads to further ROS production [30,87]. Another NO-derived metabolite known as nitrite is consumed by the heme-containing myelop-eroxidase. Often found in neutrophils, this myeloperoxidase enzyme catalytically reacts with nitrite in the presence of H2O2 and generates powerful radicals such as nitrogen di-oxide (•NO2), which can create a state of oxidation [88].
In SCD, the release of arginase during hemolysis can also contribute to low NO levels. Arginase competes with nitric oxide synthase (NOS) for its substrate, L-arginine, which it uses to produce NO and L-citrulline [77]. The absence of tetrahydrobiopterin (BH4), due to peroxynitrite production, or L-arginine leads to the “uncoupling” of NOS and results in further ROS production in SCD [77,89]. This condition diminishes •NO bioavailability in both SCD and other vasculopathies [90].
Asymmetric dimethylarginine (ADMA) is the major endogenous inhibitor of NOS that competes with L-arginine for NOS [91,92]. ADMA can lead to the uncoupling of all NOS isoforms by converting them from •NO-producing enzymes to enzymes generating O2•− and other derived oxidant products, thus leading to an increase in oxidative stress and decreasing •NO bioavailability [93,94]. When plasma concentrations of ADMA are elevated, this can be a risk factor for the development of endothelial dysfunction and is used as a predictor for all-cause and cardiovascular mortality [95]. In SCD patients, increased plasma levels of ADMA have been reported. These were not only correlated with hemolytic markers but also associated with increased amounts of soluble vascular cell adhesion molecule-1 (sVCAM-1), SCD-related pulmonary hypertension, and early death [96,97,98].
7. Antioxidant Enzymes and Cytoprotective Defenses in β-Thalassemia and SCD
In the previous sections, we discussed how oxidation contributes to red cell damage and ineffective erythropoiesis in both β-thalassemia and SCD. Here, we focus on endogenous antioxidant and cytoprotective systems, which are crucial to limit prolonged oxidation and ensure cell survival.
7.1. Cytoprotective and Anti-Oxidant Systems in Erythroid Cells
7.1.1. Peroxiredoxin-2
Peroxiredoxins play a significant role in RBCs through their antioxidant properties and chaperone function. Proteasomes are multi-catalytic complexes with important roles in protein control. Their activity in stored RBCs is affected by both storage time and the donor’s characteristics. In fact, some recent studies on patients with β-thalassemia trait and SCD have provided significant information about the interaction of peroxiredoxins and the RBC proteasomal machinery [99,100,101,102]. Of interest is peroxiredoxin-2 (Prx2), one of the most abundant proteins in RBCs, which plays a major role against oxidation [24,27,103,104,105,106,107,108,109]. It is a member of the typical homodimeric 2-Cys Prxs, the wider group that uses two cysteine residues to detoxify many organic peroxides, including H2O2 and ONOO− [2,103,110]. The tyrosine phosphorylation of Prx2 increases its activity in response to severe oxidation [2]. In addition to its enzymatic function, and under oxidative stress conditions, peroxiredoxins have been shown to acquire a chaperone function, allowing them to migrate to the membrane and interact with numerous proteins [111,112]. The importance of Prx2 in RBCs and erythropoiesis has been supported by results in mouse models genetically lacking Prx2 (Prx2-/-), which display age-dependent chronic hemolysis and ineffective erythropoiesis [107,113]. We have recently shown that Prx2 expression is increased in β-thalassemic RBCs and plays an important role during both normal and pathologic erythropoiesis to support erythroid maturation against oxidative stress [25,114]. In addition, Prx2 acts as a backup mechanism in the presence of severe oxidation due to iron overload. This highlights the importance of Prx2 in chronic hemolytic anemia such as β-thalassemia [104,107]. In fact, one recent study showed that peroxiredoxin 1 cooperates with Prx2 in the antioxidant pathways of erythroid cells in patients with β-TI [115].
In SCD, Prx2 has been also shown to be involved in the dynamic cross-talk between the cytoplasm compartment and the RBC membrane of SCD mice exposed to hypoxia-reoxygenation stress, a condition used to mimic acute sickle cell-related vaso-occlusive crisis (VOC) [101,102,116]. Indeed, Prx2 membrane association might decrease local oxidation, preventing band 3 clusterization and the generation of erythroid microvesicles.
7.1.2. Superoxide Dismutase and Catalase
In RBCs, superoxide dismutase (SOD) acts as a first-line defense mechanism against free radicals. This cytosolic enzyme that contains both copper and zinc converts O2●− into the less reactive H2O2 [117]. Found at high concentrations in RBCs, catalase is an intracellular enzyme that protects cells and tissues from the toxic effects of H2O2 [118]. A major increase in SOD was found in beta-thalassemia major (β-TM) patients when compared to healthy controls [119]. Another study, however, showed no significant change in the levels of SOD and catalase in patients with β-thalassemia [120]. As for SCD, one study by Antwi-Boasiako et al. reported that levels of SOD and catalase in RBCs were significantly lower in SCD patients when compared to healthy controls [121]. This is in agreement with other studies that also showed low levels of antioxidant enzymes in SCD patients [122,123,124]. A previous study conducted by Manfredini et al., however, reported significantly higher levels of SOD in the RBCs of SCA patients compared to their healthy counterparts [125]. In the same study, levels of catalase were not found to be significantly different between the SCA and controls. Moreover, HbSS patients with VOCs had significantly lower levels of SOD in RBCs when compared to the other study subjects. The excessive amount of ROS produced during a VOC may have partly contributed to the lower levels of SOD and catalase in these patients. Findings from a 2019 study by Antwi-Boasiako et al. also proposed that the low levels of SOD and catalase in RBCs seen in SCD patients with the SS genotype may be due to the significant depletion of antioxidants, such as nitric oxide and vitamins [121]. As evident by many of these studies, there has been a discrepancy in reports on SOD and catalase activity. Increased levels of antioxidant enzymes such as SOD and catalase may be seen in various settings including an acute inflammatory phase, a state of trauma, and upon exposure to increased levels of pro-oxidants [126]. Decreased SOD levels could be due to increased oxidative stress which results in excessive antioxidant consumption and thus antioxidant deficiency [127]. On the other hand, a decrease in catalase activity might be due to the chronic level of oxidative stress itself, whereas an increase in its activity might be due to a protective measure by the body to scavenge ROS. Increased catalase levels could also be a consequence of higher reticulocyte content in SCD patients, for example [128]. Consequently, further studies with a much larger sample size and unified methodological approach are needed to better understand the reasons behind the varying levels of SOD and catalase that have been reported in β-thalassemia and SCD patients.
7.1.3. The Glutathione System
The glutathione system (GSH) is an important scavenger of free radicals and a potent endogenous antioxidant that can protect cells from oxidative injury [129,130]. In β-TM patients, higher levels of glutathione peroxidase (GPx) were observed as compared to healthy controls [119]. Another study, however, reported the levels of the antioxidant enzyme GPx to be significantly lower in β-thalassemia patients [131]. These findings are in agreement with the study of Garelnabi et al., which showed that the low levels of GPx in children with β-thalassemia seem to result from the enzyme inhibition or reduced activity due to the excessive production of H2O2 [132]. In a study on SCD patients, the oxidative status of RBCs was evaluated by exploring the glutathione system. The overall total content of GSH and reduced GSH in SCD RBCs was 32–36% lower in these patients [133]. This finding is in line with other previous studies and further highlights this increased oxidative stress status that is characteristic of sickle RBCs [134,135,136]. The same study also reported higher GPx activities in SCD patients compared to controls, and no significant differences in the activity of glutathione reductase activity among the studied cohorts [133]. The contradictory conclusions of these studies might be related to patient selection (age, presence/absence of the spleen, kidney disease), reticulocyte count, and/or the number of circulating erythroblasts. Moreover, the enzymatic levels/activity of the glutathione system is higher in young RBCs compared to mature/old RBCs.
7.2. Cytoprotective Systems in Erythropoiesis
7.2.1. Heme-Regulated Inhibitor of Protein Translation
In β-thalassemic erythropoiesis, the thinning control of iron and heme is a priority for cell survival. Heme-regulated inhibitor of protein translation (HRI) has been shown to repress globin translation in heme-deficient erythroid precursors [137]. HRI is the heme-regulated eukaryotic initiation factor-2α (eIF2α) kinase that phosphorylates a subunit of eIF2. In vitro studies have shown that HRI activation also involves ROS and necessitates the presence of the heat shock proteins 70 and 90 [138]. In β-thalassemic mice that genetically lack HRI, a more severe hematological phenotype was shown when compared to normal β-thalassemia mice, supporting the key role of eIF2α in stress erythropoiesis [139]. Translational up-regulation of activating transcription factor 4 (ATF4) mRNA by the HRI-eIF2αP signaling pathway was necessary to mitigate oxidative stress and promote erythroid differentiation [140]. The repression of globin mRNA translation by HRI decreased proteotoxicity and allowed the ATF4 protein to be expressed. This is an important process in terminal erythropoiesis to maintain pivotal mitochondrial functions and oxidative homeostasis [141].
7.2.2. Heme Oxygenase-1 (HO-1)
The catabolism of heme is also important in normal and pathologic erythropoiesis. HO-1 (heme oxygenase-1) is an enzyme that catalyzes the degradation of heme [142]. It is generally considered to be a protective enzyme because of its ability to breakdown the pro-oxidant “free” heme and release biliverdin and bilirubin, which exhibit antioxidant properties. Thus, HO-1 might represent a potential therapeutic target in ineffective erythropoiesis and thus improve oxidative stress in β-thalassemia. In fact, the administration of tin-protoporphyrin IX, an HO-1 inhibitor, improved overall hematological parameters and decreased anemia, ineffective erythropoiesis, spleen size, liver iron, and erythropoietin levels, and increased the hepcidin serum levels in Th3/+ mice. Treatment with tin protoporphyrin IX also decreased apoptosis, increased RBC lifespan, and reduced ROS levels [143].
7.2.3. Alpha Hemoglobin-Stabilizing Protein
Among atypical chaperone systems, the α hemoglobin stabilizing protein (AHSP) has been reported to play an important role in β-thalassemic erythropoiesis. AHSP binds to α-Hb, prevents its precipitation, and limits free α-Hb toxicities. Alpha Hb bound to AHSP is more resistant to oxidant-induced precipitation and the phenotype of β-TI mice has been shown to be exacerbated by the concomitant loss of AHSP [144]. Moreover, AHSP knock-out mice showed pathological features and a degree of ineffective erythropoiesis that is similar to that seen in β-thalassemia [145]. One cross-sectional study on 37 patients with β-thalassemia and 12 sickle cell anemia (SCA) patients showed that AHSP levels were significantly higher in patients with SCA compared to those with β-thalassemia [146]. Moreover, no significant differences in the level of AHSP were seen between patients with β-TM and β-TI [146].
8. Antioxidant Therapeutic Agents
Because oxidative stress plays a significant role in the pathophysiology of diseases such as β-thalassemia and SCD, numerous molecules and pharmacological agents with antioxidant properties have been used as a potential therapeutic strategy. Evidence from the literature outlining these antioxidants and describing their therapeutic effect and contribution to a redox balance has been summarized in Table 1.

Table 1.
Antioxidant therapies in β-thalassemia and SCD patients.
9. Conclusions and Future Perspectives
In conclusion, while oxidative stress is not among the primary etiologies of β-thalassemia and SCD, it plays a significant role in the pathogenesis of these diseases. The mechanism of oxidative stress development in β-thalassemia and SCD is not only multifactorial in nature but also different. Oxidative stress can be ameliorated with antioxidative treatment modalities that act at various levels. The identification of cytoprotective and antioxidant enzymes and molecules has paved the way for a new era of new pharmacological targets for treating β-thalassemia and SCD. For optimal outcomes, future studies should aim at identifying specific sources of ROS, applying direct and targeted therapy, and improving overall outcomes for patients. A better understanding of the main oxidants and antioxidants involved, and the associated cascade of biological events will provide a better insight to achieve an optimal redox balance.
Author Contributions
Conceptualization, R.B.-F. and I.M.; investigation, R.B.-F. and L.D.F.; writing—original draft preparation, R.B.-F.; writing—review and editing, I.M., L.D.F., A.A.E., A.T.T. and M.D.C.; supervision, I.M. and M.D.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors would like to thank Luigi Flaminio Ghilardini for his support and assistance with the figures.
Conflicts of Interest
R.B.-F. and A.A.E. have nothing to disclose. L.D.F. serves on the advisory board of Novartis, Roche, and V.I.M. reports receiving honoraria from Sanofi-Genzyme and Amicus Therapeutics and serves on advisory boards for Bristol Myers Squibb/Celgene. M.D.C. serves on advisory boards for Bristol Myers Squibb/Celgene, Sanofi/Genzyme, Agios, Silence Therapeutics, Vertex, and Vifor. A.T.T. reports receiving consultancy from Novartis, Bristol Myers Squibb/Celgene, Vifor Pharma, Ionis Pharmaceuticals, Imara, and Agios Pharmaceuticals, and research funding from Novartis, Bristol Myers Squibb, Vifor Pharma, Imara, and Ionis Pharmaceuticals.
References
- Matte, A.; Federti, E.; Kung, C.; Kosinski, P.A.; Narayanaswamy, R.; Russo, R.; Federico, G.; Carlomagno, F.; Desbats, M.A.; Salviati, L.; et al. The pyruvate kinase activator mitapivat reduces hemolysis and improves anemia in a beta-thalassemia mouse model. J. Clin. Investig. 2021, 131, e144206. [Google Scholar] [CrossRef] [PubMed]
- Tibaldi, E.; Federti, E.; Matte, A.; Iatcenko, I.; Wilson, A.B.; Riccardi, V.; Pagano, M.A.; De Franceschi, L. Oxidation Impacts the Intracellular Signaling Machinery in Hematological Disorders. Antioxidants 2020, 9, 353. [Google Scholar] [CrossRef] [PubMed]
- Matte, A.; Federti, E.; Winter, M.; Koerner, A.; Harmeier, A.; Mazer, N.; Tomka, T.; Di Paolo, M.L.; De Falco, L.; Andolfo, I.; et al. Bitopertin, a selective oral GLYT1 inhibitor, improves anemia in a mouse model of beta-thalassemia. JCI Insight 2019, 4, e130111. [Google Scholar] [CrossRef] [PubMed]
- Matte, A.; De Franceschi, L. Oxidation and erythropoiesis. Curr. Opin. Hematol. 2019, 26, 145–151. [Google Scholar] [CrossRef]
- Brugnara, C.; de Franceschi, L. Effect of cell age and phenylhydrazine on the cation transport properties of rabbit erythrocytes. J. Cell Physiol. 1993, 154, 271–280. [Google Scholar] [CrossRef]
- de Franceschi, L.; Turrini, F.; Honczarenko, M.; Ayi, K.; Rivera, A.; Fleming, M.D.; Law, T.; Mannu, F.; Kuypers, F.A.; Bast, A.; et al. In vivo reduction of erythrocyte oxidant stress in a murine model of beta-thalassemia. Haematologica 2004, 89, 1287–1298. [Google Scholar]
- Voskou, S.; Aslan, M.; Fanis, P.; Phylactides, M.; Kleanthous, M. Oxidative stress in beta-thalassaemia and sickle cell disease. Redox Biol. 2015, 6, 226–239. [Google Scholar] [CrossRef]
- Fibach, E.; Dana, M. Oxidative stress in β-thalassemia. Mol. Diagn. Ther. 2019, 23, 245–261. [Google Scholar] [CrossRef]
- Turpaev, K.T. Reactive oxygen species and regulation of gene expression. Biochemistry 2002, 67, 281–292. [Google Scholar]
- Droge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. BioMed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef] [PubMed]
- Blackstone, N.W.; Kelly, M.M.; Haridas, V.; Gutterman, J.U. Mitochondria as integrators of information in an early-evolving animal: Insights from a triterpenoid metabolite. Proc. Biol. Sci. 2005, 272, 527–531. [Google Scholar] [CrossRef] [PubMed]
- van der Heyde, H.C.; Gu, Y.; Zhang, Q.; Sun, G.; Grisham, M.B. Nitric oxide is neither necessary nor sufficient for resolution of Plasmodium chabaudi malaria in mice. J. Immunol. 2000, 165, 3317–3323. [Google Scholar] [CrossRef] [PubMed]
- Holm, E.; Hildebrandt, W.; Kinscherf, R.; Droge, W. Low postabsorptive net protein degradation in male cancer patients: Lack of sensitivity to regulatory amino acids? Oncol. Rep. 2007, 17, 695–700. [Google Scholar] [CrossRef][Green Version]
- Scheiber, M.N.; Watson, P.M.; Rumboldt, T.; Stanley, C.; Wilson, R.C.; Findlay, V.J.; Anderson, P.E.; Watson, D.K. FLI1 expression is correlated with breast cancer cellular growth, migration, and invasion and altered gene expression. Neoplasia 2014, 16, 801–813. [Google Scholar] [CrossRef]
- Egea, J.; Fabregat, I.; Frapart, Y.M.; Ghezzi, P.; Gorlach, A.; Kietzmann, T.; Kubaichuk, K.; Knaus, U.G.; Lopez, M.G.; Olaso-Gonzalez, G.; et al. European contribution to the study of ROS: A summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS). Redox Biol. 2017, 13, 94–162. [Google Scholar] [CrossRef]
- Sies, H.; Cadenas, E. Oxidative stress: Damage to intact cells and organs. Philos. Trans. R Soc. Lond B Biol. Sci. 1985, 311, 617–631. [Google Scholar]
- Van Zwieten, R.; Verhoeven, A.J.; Roos, D. Inborn defects in the antioxidant systems of human red blood cells. Free Radic. Biol. Med. 2014, 67, 377–386. [Google Scholar] [CrossRef]
- Rifkind, J.M.; Mohanty, J.G.; Nagababu, E. The pathophysiology of extracellular hemoglobin associated with enhanced oxidative reactions. Front. Physiol. 2014, 5, 500. [Google Scholar] [CrossRef]
- Kanias, T.; Acker, J.P. Biopreservation of red blood cells—The struggle with hemoglobin oxidation. FEBS J. 2010, 277, 343–356. [Google Scholar] [CrossRef]
- Reiter, R.J.; Melchiorri, D.; Sewerynek, E.; Poeggeler, B.; Barlow-Walden, L.; Chuang, J.; Ortiz, G.G.; Acuna-Castroviejo, D. A review of the evidence supporting melatonin’s role as an antioxidant. J. Pineal. Res. 1995, 18, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Sadrzadeh, S.M.; Graf, E.; Panter, S.S.; Hallaway, P.E.; Eaton, J.W. Hemoglobin. A biologic fenton reagent. J. Biol. Chem. 1984, 259, 14354–14356. [Google Scholar] [CrossRef]
- Zapora, E.; Jarocka, I. Hemoglobin—Source of reactive oxygen species. Postepy Hig. Med. Dosw. Online 2013, 67, 214–220. [Google Scholar] [CrossRef] [PubMed]
- De Franceschi, L.; Bertoldi, M.; Matte, A.; Santos Franco, S.; Pantaleo, A.; Ferru, E.; Turrini, F. Oxidative stress and beta-thalassemic erythroid cells behind the molecular defect. Oxid. Med. Cell Longev. 2013, 2013, 985210. [Google Scholar] [CrossRef] [PubMed]
- Matte, A.; Bertoldi, M.; Mohandas, N.; An, X.; Bugatti, A.; Brunati, A.M.; Rusnati, M.; Tibaldi, E.; Siciliano, A.; Turrini, F. Membrane association of peroxiredoxin-2 in red cells is mediated by the N-terminal cytoplasmic domain of band 3. Free Radic. Biol. Med. 2013, 55, 27–35. [Google Scholar] [CrossRef] [PubMed]
- de Franceschi, L.; Shalev, O.; Piga, A.; Collell, M.; Olivieri, O.; Corrocher, R.; Hebbel, R.P.; Brugnara, C. Deferiprone therapy in homozygous human beta-thalassemia removes erythrocyte membrane free iron and reduces KCl cotransport activity. J. Lab. Clin. Med. 1999, 133, 64–69. [Google Scholar] [CrossRef]
- Matte, A.; Low, P.S.; Turrini, F.; Bertoldi, M.; Campanella, M.E.; Spano, D.; Pantaleo, A.; Siciliano, A.; De Franceschi, L. Peroxiredoxin-2 expression is increased in β-thalassemic mouse red cells but is displaced from the membrane as a marker of oxidative stress. Free. Radic. Biol. Med. 2010, 49, 457–466. [Google Scholar] [CrossRef]
- Vinchi, F.; De Franceschi, L.; Ghigo, A.; Townes, T.; Cimino, J.; Silengo, L.; Hirsch, E.; Altruda, F.; Tolosano, E. Hemopexin therapy improves cardiovascular function by preventing heme-induced endothelial toxicity in mouse models of hemolytic diseases. Circulation 2013, 127, 1317–1329. [Google Scholar] [CrossRef]
- Ingoglia, G.; Sag, C.M.; Rex, N.; De Franceschi, L.; Vinchi, F.; Cimino, J.; Petrillo, S.; Wagner, S.; Kreitmeier, K.; Silengo, L.; et al. Hemopexin counteracts systolic dysfunction induced by heme-driven oxidative stress. Free Radic. Biol. Med. 2017, 108, 452–464. [Google Scholar] [CrossRef]
- Belcher, J.D.; Beckman, J.D.; Balla, G.; Balla, J.; Vercellotti, G. Heme degradation and vascular injury. Antioxid. Redox Signal. 2010, 12, 233–248. [Google Scholar] [CrossRef]
- Duvigneau, J.C.; Luis, A.; Gorman, A.M.; Samali, A.; Kaltenecker, D.; Moriggl, R.; Kozlov, A.V. Crosstalk between inflammatory mediators and endoplasmic reticulum stress in liver diseases. Cytokine 2019, 124, 154577. [Google Scholar] [CrossRef] [PubMed]
- Tracz, M.J.; Alam, J.; Nath, K.A. Physiology and pathophysiology of heme: Implications for kidney disease. J. Am. Soc. Nephrol. 2007, 18, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bandyopadhyay, U. Free heme toxicity and its detoxification systems in human. Toxicol. Lett. 2005, 157, 175–188. [Google Scholar] [CrossRef] [PubMed]
- De Franceschi, L.; Tomelleri, C.; Matte, A.; Brunati, A.M.; Bovee-Geurts, P.H.; Bertoldi, M.; Lasonder, E.; Tibaldi, E.; Danek, A.; Walker, R.H.; et al. Erythrocyte membrane changes of chorea-acanthocytosis are the result of altered Lyn kinase activity. Blood 2011, 118, 5652–5663. [Google Scholar] [CrossRef]
- Shinar, E.; Rachmilewitz, E.A.; Lux, S.E. Differing erythrocyte membrane skeletal protein defects in alpha and beta thalassemia. J. Clin. Investig. 1989, 83, 404–410. [Google Scholar] [CrossRef]
- Pantaleo, A.; Ferru, E.; Pau, M.C.; Khadjavi, A.; Mandili, G.; Mattè, A.; Spano, A.; De Franceschi, L.; Pippia, P.; Turrini, F. Band 3 erythrocyte membrane protein acts as redox stress sensor leading to its phosphorylation by p72 Syk. Oxidative Med. Cell. Longev. 2016, 2016, 6051093. [Google Scholar] [CrossRef]
- De Franceschi, L.; Brugnara, C.; Rouyer-Fessard, P.; Jouault, H.; Beuzard, Y. Formation of dense erythrocytes in SAD mice exposed to chronic hypoxia: Evaluation of different therapeutic regimens and of a combination of oral clotrimazole and magnesium therapies. Blood 1999, 94, 4307–4313. [Google Scholar] [CrossRef]
- Jana, S.; Strader, M.B.; Meng, F.; Hicks, W.; Kassa, T.; Tarandovskiy, I.; De Paoli, S.; Simak, J.; Heaven, M.R.; Belcher, J.D.; et al. Hemoglobin oxidation-dependent reactions promote interactions with band 3 and oxidative changes in sickle cell-derived microparticles. JCI Insight. 2018, 3, e120451. [Google Scholar] [CrossRef]
- Strader, M.B.; Jana, S.; Meng, F.; Heaven, M.R.; Shet, A.S.; Thein, S.L.; Alayash, A.I. Post-translational modification as a response to cellular stress induced by hemoglobin oxidation in sickle cell disease. Sci. Rep. 2020, 10, 14218. [Google Scholar] [CrossRef]
- Baliga, S.; Chaudhary, M.; Bhat, S.; Bhansali, P.; Agrawal, A.; Gundawar, S. Estimation of malondialdehyde levels in serum and saliva of children affected with sickle cell anemia. J. Indian Soc. Pedod. Prev. Dent. 2018, 36, 43–47. [Google Scholar]
- McNaughton-Smith, G.A.; Burns, J.F.; Stocker, J.W.; Rigdon, G.C.; Creech, C.; Arrington, S.; Shelton, T.; de Franceschi, L. Novel inhibitors of the Gardos channel for the treatment of sickle cell disease. J. Med. Chem. 2008, 51, 976–982. [Google Scholar] [CrossRef] [PubMed]
- Bordin, L.; Brunati, A.M.; Donella-Deana, A.; Baggio, B.; Toninello, A.; Clari, G. Band 3 is an anchor protein and a target for SHP-2 tyrosine phosphatase in human erythrocytes. Blood J. Am. Soc. Hematol. 2002, 100, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Kuypers, F.A.; de Jong, K. The role of phosphatidylserine in recognition and removal of erythrocytes. Cell Mol. Biol. 2004, 50, 147–158. [Google Scholar] [PubMed]
- Ibrahim, H.A.; Fouda, M.I.; Yahya, R.S.; Abousamra, N.K.; Abd Elazim, R.A. Erythrocyte phosphatidylserine exposure in beta-thalassemia. Lab. Hematol. 2014, 20, 9–14. [Google Scholar] [CrossRef]
- Kuypers, F.A. Hemoglobin s polymerization and red cell membrane changes. Hematol Oncol. Clin. North Am. 2014, 28, 155–179. [Google Scholar] [CrossRef]
- Hannemann, A.; Rees, D.C.; Brewin, J.N.; Noe, A.; Low, B.; Gibson, J.S. Oxidative stress and phosphatidylserine exposure in red cells from patients with sickle cell anaemia. Br. J. Haematol. 2018, 182, 567–578. [Google Scholar] [CrossRef]
- Setty, B.N.; Kulkarni, S.; Stuart, M.J. Role of erythrocyte phosphatidylserine in sickle red cell-endothelial adhesion. Blood 2002, 99, 1564–1571. [Google Scholar] [CrossRef]
- Kuypers, F.A.; Styles, L.A. The role of secretory phospholipase A2 in acute chest syndrome. Cell Mol. Biol. 2004, 50, 87–94. [Google Scholar]
- Neidlinger, N.A.; Larkin, S.K.; Bhagat, A.; Victorino, G.P.; Kuypers, F.A. Hydrolysis of phosphatidylserine-exposing red blood cells by secretory phospholipase A2 generates lysophosphatidic acid and results in vascular dysfunction. J. Biol. Chem. 2006, 281, 775–781. [Google Scholar] [CrossRef]
- Butikofer, P.; Yee, M.C.; Schott, M.A.; Lubin, B.H.; Kuypers, F.A. Generation of phosphatidic acid during calcium-loading of human erythrocytes. Evidence for a phosphatidylcholine-hydrolyzing phospholipase D. Eur. J. Biochem. 1993, 213, 367–375. [Google Scholar] [CrossRef]
- Rachmilewitz, E.A.; Weizer-Stern, O.; Adamsky, K.; Amariglio, N.; Rechavi, G.; Breda, L.; Rivella, S.; Cabantchik, Z.I. Role of iron in inducing oxidative stress in thalassemia: Can it be prevented by inhibition of absorption and by antioxidants? Ann. N. Y. Acad. Sci. 2005, 1054, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Pantaleo, A.; Giribaldi, G.; Mannu, F.; Arese, P.; Turrini, F. Naturally occurring anti-band 3 antibodies and red blood cell removal under physiological and pathological conditions. Autoimmun. Rev. 2008, 7, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Rachmilewitz, E.A.; Peisach, J.; Bradley, T.B.; Blumberg, W.E. Role of haemichromes in the formation of inclusion bodies in haemoglobin H disease. Nature 1969, 222, 248–250. [Google Scholar] [CrossRef]
- Pantaleo, A.; Ferru, E.; Giribaldi, G.; Mannu, F.; Carta, F.; Matte, A.; de Franceschi, L.; Turrini, F. Oxidized and poorly glycosylated band 3 is selectively phosphorylated by Syk kinase to form large membrane clusters in normal and G6PD-deficient red blood cells. Biochem. J. 2009, 418, 359–367. [Google Scholar] [CrossRef] [PubMed]
- De Franceschi, L.; Biondani, A.; Carta, F.; Turrini, F.; Laudanna, C.; Deana, R.; Brunati, A.M.; Turretta, L.; Iolascon, A.; Perrotta, S.; et al. PTPepsilon has a critical role in signaling transduction pathways and phosphoprotein network topology in red cells. Proteomics 2008, 8, 4695–4708. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; McKenna, M.M.; Krump, N.A.; Zheng, S.; Mendelsohn, L.; Thein, S.L.; Garrett, L.J.; Bodine, D.M.; Low, P.S. Reversible binding of hemoglobin to band 3 constitutes the molecular switch that mediates O2 regulation of erythrocyte properties. Blood J. Am. Soc. Hematol. 2016, 128, 2708–2716. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Kannan, R.; Shinar, E.; Rachmilewitz, E.A.; Low, P.S. Isolation, characterization, and immunoprecipitation studies of immune complexes from membranes of beta-thalassemic erythrocytes. Blood 1992, 79, 3007–3013. [Google Scholar] [CrossRef]
- Longo, F.; Piolatto, A.; Ferrero, G.B.; Piga, A. Ineffective Erythropoiesis in beta-Thalassaemia: Key Steps and Therapeutic Options by Drugs. Int. J. Mol. Sci. 2021, 22, 7229. [Google Scholar] [CrossRef]
- Angelucci, E.; Bai, H.; Centis, F.; Bafti, M.S.; Lucarelli, G.; Ma, L.; Schrier, S. Enhanced macrophagic attack on beta-thalassemia major erythroid precursors. Haematologica 2002, 87, 578–583. [Google Scholar]
- Centis, F.; Tabellini, L.; Lucarelli, G.; Buffi, O.; Tonucci, P.; Persini, B.; Annibali, M.; Emiliani, R.; Iliescu, A.; Rapa, S.; et al. The importance of erythroid expansion in determining the extent of apoptosis in erythroid precursors in patients with beta-thalassemia major. Blood 2000, 96, 3624–3629. [Google Scholar] [CrossRef]
- Mathias, L.A.; Fisher, T.C.; Zeng, L.; Meiselman, H.J.; Weinberg, K.I.; Hiti, A.L.; Malik, P. Ineffective erythropoiesis in beta-thalassemia major is due to apoptosis at the polychromatophilic normoblast stage. Exp. Hematol. 2000, 28, 1343–1353. [Google Scholar] [CrossRef]
- Wickramasinghe, S.N.; Bush, V. Observations on the ultrastructure of erythropoietic cells and reticulum cells in the bone marrow of patients with homozygous beta-thalassaemia. Br. J. Haematol. 1975, 30, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Dussiot, M.; Maciel, T.T.; Fricot, A.; Chartier, C.; Negre, O.; Veiga, J.; Grapton, D.; Paubelle, E.; Payen, E.; Beuzard, Y.; et al. An activin receptor IIA ligand trap corrects ineffective erythropoiesis in beta-thalassemia. Nat. Med. 2014, 20, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Schrier, S.L.; Centis, F.; Verneris, M.; Ma, L.; Angelucci, E. The role of oxidant injury in the pathophysiology of human thalassemias. Redox Rep. 2003, 8, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Taher, A.T.; Musallam, K.M.; Cappellini, M.D. beta-Thalassemias. N. Engl. J. Med. 2021, 384, 727–743. [Google Scholar] [CrossRef]
- Taher, A.T.; Weatherall, D.J.; Cappellini, M.D. Thalassaemia. Lancet 2018, 391, 155–167. [Google Scholar] [CrossRef]
- Leecharoenkiat, K.; Lithanatudom, P.; Sornjai, W.; Smith, D.R. Iron dysregulation in beta-thalassemia. Asian Pac. J. Trop. Med. 2016, 9, 1035–1043. [Google Scholar] [CrossRef]
- Hershko, C. Pathogenesis and management of iron toxicity in thalassemia. Ann. N. Y. Acad. Sci. 2010, 1202, 1–9. [Google Scholar] [CrossRef]
- Cabantchik, Z.I. Labile iron in cells and body fluids: Physiology, pathology, and pharmacology. Front. Pharmacol. 2014, 5, 45. [Google Scholar] [CrossRef]
- Valko, M.; Jomova, K.; Rhodes, C.J.; Kuca, K.; Musilek, K. Redox- and non-redox-metal-induced formation of free radicals and their role in human disease. Arch. Toxicol. 2016, 90, 1–37. [Google Scholar] [CrossRef]
- Bresgen, N.; Eckl, P.M. Oxidative stress and the homeodynamics of iron metabolism. Biomolecules 2015, 5, 808–847. [Google Scholar] [CrossRef] [PubMed]
- Mancardi, D.; Mezzanotte, M.; Arrigo, E.; Barinotti, A.; Roetto, A. Iron Overload, Oxidative Stress, and Ferroptosis in the Failing Heart and Liver. Antioxidants 2021, 10, 1864. [Google Scholar] [CrossRef] [PubMed]
- Menon, A.V.; Tsai, H.P.; Kim, J. Cardiac iron overload promotes ferroptosis and cardiac dysfunction in mice with sickle cell disease. FASEB J. 2020, 34, 1. [Google Scholar] [CrossRef]
- Menon, A.V.; Liu, J.; Tsai, H.P.; Zeng, L.; Yang, S.; Asnani, A.; Kim, J. Excess heme upregulates heme oxygenase 1 and promotes cardiac ferroptosis in mice with sickle cell disease. Blood 2022, 139, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Vinchi, F.; Porto, G.; Simmelbauer, A.; Altamura, S.; Passos, S.T.; Garbowski, M.; Silva, A.M.; Spaich, S.; Seide, S.E.; Sparla, R. Atherosclerosis is aggravated by iron overload and ameliorated by dietary and pharmacological iron restriction. Eur. Heart J. 2020, 41, 2681–2695. [Google Scholar] [CrossRef] [PubMed]
- Wood, K.C.; Hebbel, R.P.; Granger, D.N. Endothelial cell NADPH oxidase mediates the cerebral microvascular dysfunction in sickle cell transgenic mice. FASEB J. 2005, 19, 989–991. [Google Scholar] [CrossRef]
- Wood, K.C.; Hebbel, R.P.; Lefer, D.J.; Granger, D.N. Critical role of endothelial cell-derived nitric oxide synthase in sickle cell disease-induced microvascular dysfunction. Free Radic. Biol. Med. 2006, 40, 1443–1453. [Google Scholar] [CrossRef]
- Wood, K.C.; Granger, D.N. Sickle cell disease: Role of reactive oxygen and nitrogen metabolites. Clin. Exp. Pharmacol. Physiol. 2007, 34, 926–932. [Google Scholar] [CrossRef]
- George, A.; Pushkaran, S.; Konstantinidis, D.G.; Koochaki, S.; Malik, P.; Mohandas, N.; Zheng, Y.; Joiner, C.H.; Kalfa, T.A. Erythrocyte NADPH oxidase activity modulated by Rac GTPases, PKC, and plasma cytokines contributes to oxidative stress in sickle cell disease. Blood 2013, 121, 2099–2107. [Google Scholar] [CrossRef]
- De Franceschi, L.; Cappellini, M.D.; Olivieri, O. Thrombosis and sickle cell disease. Semin. Thromb. Hemost. 2011, 37, 226–236. [Google Scholar] [CrossRef]
- Matte, A.; Zorzi, F.; Mazzi, F.; Federti, E.; Olivieri, O.; De Franceschi, L. New Therapeutic Options for the Treatment of Sickle Cell Disease. Mediterr. J. Hematol. Infect. Dis. 2019, 11, e2019002. [Google Scholar] [CrossRef] [PubMed]
- De Franceschi, L.; Baron, A.; Scarpa, A.; Adrie, C.; Janin, A.; Barbi, S.; Kister, J.; Rouyer-Fessard, P.; Corrocher, R.; Leboulch, P. Inhaled nitric oxide protects transgenic SAD mice from sickle cell disease–specific lung injury induced by hypoxia/reoxygenation. Blood 2003, 102, 1087–1096. [Google Scholar] [CrossRef] [PubMed]
- De Franceschi, L.; Malpeli, G.; Scarpa, A.; Janin, A.; Muchitsch, E.M.; Roncada, P.; Leboeuf, C.; Corrocher, R.; Beuzard, Y.; Brugnara, C. Protective effects of S-nitrosoalbumin on lung injury induced by hypoxia-reoxygenation in mouse model of sickle cell disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2006, 291, L457–L465. [Google Scholar] [CrossRef] [PubMed]
- Gentinetta, T.; Belcher, J.D.; Brügger-Verdon, V.; Adam, J.; Ruthsatz, T.; Bain, J.; Schu, D.; Ventrici, L.; Edler, M.; Lioe, H. Plasma-Derived Hemopexin as a Candidate Therapeutic Agent for Acute Vaso-Occlusion in Sickle Cell Disease: Preclinical Evidence. J. Clin. Med. 2022, 11, 630. [Google Scholar] [CrossRef] [PubMed]
- Nath, K.A.; Grande, J.P.; Belcher, J.D.; Garovic, V.D.; Croatt, A.J.; Hillestad, M.L.; Barry, M.A.; Nath, M.C.; Regan, R.F.; Vercellotti, G.M. Antithrombotic effects of heme-degrading and heme-binding proteins. Am. J. Physiol. Heart Circ. Physiol. 2020, 318, H671–H681. [Google Scholar] [CrossRef] [PubMed]
- Herold, S.; Exner, M.; Nauser, T. Kinetic and mechanistic studies of the NO*-mediated oxidation of oxymyoglobin and oxyhemoglobin. Biochemistry 2001, 40, 3385–3395. [Google Scholar] [CrossRef] [PubMed]
- Beckman, J.S.; Koppenol, W.H. Nitric oxide, superoxide, and peroxynitrite: The good, the bad, and ugly. Am. J. Physiol. 1996, 271, C1424–C1437. [Google Scholar] [CrossRef]
- Davies, M.J.; Hawkins, C.L. The Role of Myeloperoxidase in Biomolecule Modification, Chronic Inflammation, and Disease. Antioxid. Redox Signal. 2020, 32, 957–981. [Google Scholar] [CrossRef]
- Alderton, W.K.; Cooper, C.E.; Knowles, R.G. Nitric oxide synthases: Structure, function and inhibition. Biochem. J. 2001, 357, 593–615. [Google Scholar] [CrossRef]
- Akinsheye, I.; Klings, E.S. Sickle cell anemia and vascular dysfunction: The nitric oxide connection. J. Cell Physiol. 2010, 224, 620–625. [Google Scholar] [CrossRef]
- Forstermann, U.; Munzel, T. Endothelial nitric oxide synthase in vascular disease: From marvel to menace. Circulation 2006, 113, 1708–1714. [Google Scholar] [CrossRef] [PubMed]
- Schnog, J.B.; Teerlink, T.; van der Dijs, F.P.; Duits, A.J.; Muskiet, F.A.; Group, C.S. Plasma levels of asymmetric dimethylarginine (ADMA), an endogenous nitric oxide synthase inhibitor, are elevated in sickle cell disease. Ann. Hematol. 2005, 84, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Smith, A.; Kumar, S.; Aggarwal, S.; Rehmani, I.; Snead, C.; Harmon, C.; Fineman, J.; Fulton, D.; Catravas, J.D.; et al. Mechanisms of nitric oxide synthase uncoupling in endotoxin-induced acute lung injury: Role of asymmetric dimethylarginine. Vasc. Pharm. 2010, 52, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, X.; Shang, R.; Chen, Y. Asymmetric dimethylarginine (ADMA) as an important risk factor for the increased cardiovascular diseases and heart failure in chronic kidney disease. Nitric Oxide 2018, 78, 113–120. [Google Scholar] [CrossRef]
- Vona, R.; Sposi, N.M.; Mattia, L.; Gambardella, L.; Straface, E.; Pietraforte, D. Sickle Cell Disease: Role of Oxidative Stress and Antioxidant Therapy. Antioxidants 2021, 10, 296. [Google Scholar] [CrossRef]
- Kato, G.J.; Wang, Z.; Machado, R.F.; Blackwelder, W.C.; Taylor, J.G.T.; Hazen, S.L. Endogenous nitric oxide synthase inhibitors in sickle cell disease: Abnormal levels and correlations with pulmonary hypertension, desaturation, haemolysis, organ dysfunction and death. Br. J. Haematol. 2009, 145, 506–513. [Google Scholar] [CrossRef]
- Landburg, P.P.; Teerlink, T.; van Beers, E.J.; Muskiet, F.A.; Kappers-Klunne, M.C.; van Esser, J.W.; Mac Gillavry, M.R.; Biemond, B.J.; Brandjes, D.P.; Duits, A.J.; et al. Association of asymmetric dimethylarginine with sickle cell disease-related pulmonary hypertension. Haematologica 2008, 93, 1410–1412. [Google Scholar] [CrossRef]
- El-Shanshory, M.; Badraia, I.; Donia, A.; Abd El-Hameed, F.; Mabrouk, M. Asymmetric dimethylarginine levels in children with sickle cell disease and its correlation to tricuspid regurgitant jet velocity. Eur. J. Haematol. 2013, 91, 55–61. [Google Scholar] [CrossRef]
- Tzounakas, V.L.; Anastasiadi, A.T.; Dzieciatkowska, M.; Karadimas, D.G.; Stamoulis, K.; Papassideri, I.S.; Hansen, K.C.; D’Alessandro, A.; Kriebardis, A.G.; Antonelou, M.H. Proteome of stored RBC membrane and vesicles from heterozygous beta thalassemia donors. Int. J. Mol. Sci. 2021, 22, 3369. [Google Scholar] [CrossRef]
- Anastasiadi, A.T.; Tzounakas, V.L.; Arvaniti, V.-Z.; Dzieciatkowska, M.; Stamoulis, K.; Lekka, M.E.; Papassideri, I.S.; D’Alessandro, A.; Kriebardis, A.G.; Antonelou, M.H. Red Blood Cell Proteasome in Beta-Thalassemia Trait: Topology of Activity and Networking in Blood Bank Conditions. Membranes 2021, 11, 716. [Google Scholar] [CrossRef]
- Fujii, J.; Homma, T.; Kobayashi, S.; Warang, P.; Madkaikar, M.; Mukherjee, M.B. Erythrocytes as a preferential target of oxidative stress in blood. Free. Radic. Res. 2021, 55, 781–799. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Saha, S.; Karmakar, S.; Chakravarty, S.; Banerjee, D.; Dash, B.P.; Chakrabarti, A. 2D DIGE based proteomics study of erythrocyte cytosol in sickle cell disease: Altered proteostasis and oxidative stress. Proteomics 2013, 13, 3233–3242. [Google Scholar] [CrossRef] [PubMed]
- Mattè, A.; Federti, E.; Tibaldi, E.; Di Paolo, M.L.; Bisello, G.; Bertoldi, M.; Carpentieri, A.; Pucci, P.; Iatchencko, I.; Wilson, A.B.; et al. Tyrosine Phosphorylation Modulates Peroxiredoxin-2 Activity in Normal and Diseased Red Cells. Antioxidants 2021, 10, 206. [Google Scholar] [CrossRef] [PubMed]
- Matte, A.; De Falco, L.; Federti, E.; Cozzi, A.; Iolascon, A.; Levi, S.; Mohandas, N.; Zamo, A.; Bruno, M.; Lebouef, C.; et al. Peroxiredoxin-2: A Novel Regulator of Iron Homeostasis in Ineffective Erythropoiesis. Antioxid Redox Signal. 2018, 28, 1–14. [Google Scholar] [CrossRef]
- Federti, E.; Matté, A.; Ghigo, A.; Andolfo, I.; James, C.; Siciliano, A.; Leboeuf, C.; Janin, A.; Manna, F.; Choi, S.Y. Peroxiredoxin-2 plays a pivotal role as multimodal cytoprotector in the early phase of pulmonary hypertension. Free Radic. Biol. Med. 2017, 112, 376–386. [Google Scholar] [CrossRef]
- Matte, A.; Pantaleo, A.; Ferru, E.; Turrini, F.; Bertoldi, M.; Lupo, F.; Siciliano, A.; Ho Zoon, C.; De Franceschi, L. The novel role of peroxiredoxin-2 in red cell membrane protein homeostasis and senescence. Free Radic. Biol. Med. 2014, 76, 80–88. [Google Scholar] [CrossRef]
- Matte, A.; De Falco, L.; Iolascon, A.; Mohandas, N.; An, X.; Siciliano, A.; Leboeuf, C.; Janin, A.; Bruno, M.; Choi, S.Y. The interplay between peroxiredoxin-2 and nuclear factor-erythroid 2 is important in limiting oxidative mediated dysfunction in β-thalassemic erythropoiesis. Antioxid. Redox Signal. 2015, 23, 1284–1297. [Google Scholar] [CrossRef]
- Low, F.M.; Hampton, M.B.; Winterbourn, C.C. Peroxiredoxin 2 and peroxide metabolism in the erythrocyte. Antioxid Redox Signal. 2008, 10, 1621–1630. [Google Scholar] [CrossRef]
- Low, F.M.; Hampton, M.B.; Peskin, A.V.; Winterbourn, C.C. Peroxiredoxin 2 functions as a noncatalytic scavenger of low-level hydrogen peroxide in the erythrocyte. Blood 2007, 109, 2611–2617. [Google Scholar] [CrossRef]
- Nagababu, E.; Mohanty, J.G.; Friedman, J.S.; Rifkind, J.M. Role of peroxiredoxin-2 in protecting RBCs from hydrogen peroxide-induced oxidative stress. Free Radic. Res. 2013, 47, 164–171. [Google Scholar] [CrossRef]
- Jang, H.H.; Lee, K.O.; Chi, Y.H.; Jung, B.G.; Park, S.K.; Park, J.H.; Lee, J.R.; Lee, S.S.; Moon, J.C.; Yun, J.W.; et al. Two enzymes in one; two yeast peroxiredoxins display oxidative stress-dependent switching from a peroxidase to a molecular chaperone function. Cell 2004, 117, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Wood, Z.A.; Schroder, E.; Robin Harris, J.; Poole, L.B. Structure, mechanism and regulation of peroxiredoxins. Trends Biochem. Sci. 2003, 28, 32–40. [Google Scholar] [CrossRef]
- Lee, T.-H.; Kim, S.-U.; Yu, S.-L.; Kim, S.H.; Park, D.S.; Moon, H.-B.; Dho, S.H.; Kwon, K.-S.; Kwon, H.J.; Han, Y.-H. Peroxiredoxin II is essential for sustaining life span of erythrocytes in mice. Blood 2003, 101, 5033–5038. [Google Scholar] [CrossRef] [PubMed]
- De Franceschi, L.; Daraio, F.; Filippini, A.; Carturan, S.; Muchitsch, E.M.; Roetto, A.; Camaschella, C. Liver expression of hepcidin and other iron genes in two mouse models of beta-thalassemia. Haematologica 2006, 91, 1336–1342. [Google Scholar]
- Romanello, K.S.; Teixeira, K.K.L.; Silva, J.; Nagamatsu, S.T.; Bezerra, M.A.C.; Domingos, I.F.; Martins, D.A.P.; Araujo, A.S.; Lanaro, C.; Breyer, C.A.; et al. Global analysis of erythroid cells redox status reveals the involvement of Prdx1 and Prdx2 in the severity of beta thalassemia. PLoS ONE 2018, 13, e0208316. [Google Scholar] [CrossRef]
- Biondani, A.; Turrini, F.; Carta, F.; Matté, A.; Filippini, A.; Siciliano, A.; Beuzard, Y.; De Franceschi, L. Heat-shock protein-27,-70 and peroxiredoxin–II show molecular chaperone function in sickle red cells: Evidence from transgenic sickle cell mouse model. PROTEOMICS Clin. Appl. 2008, 2, 706–719. [Google Scholar] [CrossRef]
- Mondola, P.; Damiano, S.; Sasso, A.; Santillo, M. The Cu, Zn superoxide dismutase: Not only a dismutase enzyme. Front. Physiol. 2016, 7, 594. [Google Scholar] [CrossRef]
- Aslan, M.; Thornley-Brown, D.; Freeman, B.A. Reactive species in sickle cell disease. Ann. N. Y. Acad. Sci. 2000, 899, 375–391. [Google Scholar] [CrossRef]
- Şimşek Orhon, F.; Öztürk, G.; Kemahli, S.; Erbaş, D.; Hasanoğlu, A. Oxidant and antioxidant status in beta thalassemia major patients. Ank. Üniversitesi Tıp Fakültesi Mecmuası 2005, 58, 34–38. [Google Scholar]
- Gerli, G.; Beretta, L.; Bianchi, M.; Pellegatta, A.; Agostoni, A. Erythrocyte superoxide dismutase, catalase and glutathione peroxidase activities in β-thalassaemia (major and minor). Scand. J. Haematol. 1981, 25, 87–92. [Google Scholar] [CrossRef]
- Antwi-Boasiako, C.; Dankwah, G.B.; Aryee, R.; Hayfron-Benjamin, C.; Donkor, E.S.; Campbell, A.D. Oxidative Profile of Patients with Sickle Cell Disease. Med. Sci. 2019, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Al-Naama, L.M.; Hassan, M.K.; Mehdi, J.K. Association of erythrocytes antioxidant enzymes and their cofactors with markers of oxidative stress in patients with sickle cell anemia. Qatar. Med. J. 2015, 2015, 14. [Google Scholar] [CrossRef] [PubMed]
- Ama Moor, V.J.; Pieme, C.A.; Chetcha Chemegne, B.; Manonji, H.; Njinkio Nono, B.L.; Tchoula Mamiafo, C.; Moukette Moukette, B.; Tankeu Nzufo, F.; Tazoacha, A. Oxidative profile of sickle cell patients in a Cameroonian urban hospital. BMC Clin. Pathol. 2016, 16, 15. [Google Scholar] [CrossRef] [PubMed]
- Renoux, C.; Joly, P.; Faes, C.; Mury, P.; Eglenen, B.; Turkay, M.; Yavas, G.; Yalcin, O.; Bertrand, Y.; Garnier, N.; et al. Association between Oxidative Stress, Genetic Factors, and Clinical Severity in Children with Sickle Cell Anemia. J. Pediatr. 2018, 195, 228–235. [Google Scholar] [CrossRef]
- Manfredini, V.; Lazzaretti, L.L.; Griebeler, I.H.; Santin, A.P.; Brandao, V.D.; Wagner, S.; Castro, S.M.; Peralba Mdo, C.; Benfato, M.S. Blood antioxidant parameters in sickle cell anemia patients in steady state. J. Natl. Med. Assoc. 2008, 100, 897–902. [Google Scholar]
- Lü, J.M.; Lin, P.H.; Yao, Q.; Chen, C. Chemical and molecular mechanisms of antioxidants: Experimental approaches and model systems. J. Cell. Mol. Med. 2010, 14, 840–860. [Google Scholar] [CrossRef]
- Alsultan, A.; Seif, M.; Amin, T.; Naboli, M.; Alsuliman, A. Relationship between oxidative stress, ferritin and insulin resistance in sickle cell disease. Eur. Rev. Med. Pharmacol. Sci. 2010, 14, 527–538. [Google Scholar]
- Chirico, E.N.; Pialoux, V. Role of oxidative stress in the pathogenesis of sickle cell disease. IUBMB Life 2012, 64, 72–80. [Google Scholar] [CrossRef]
- Hanschmann, E.M.; Godoy, J.R.; Berndt, C.; Hudemann, C.; Lillig, C.H. Thioredoxins, glutaredoxins, and peroxiredoxins—Molecular mechanisms and health significance: From cofactors to antioxidants to redox signaling. Antioxid. Redox Signal. 2013, 19, 1539–1605. [Google Scholar] [CrossRef]
- Bindoli, A.; Rigobello, M.P. Principles in redox signaling: From chemistry to functional significance. Antioxid. Redox Signal. 2013, 18, 1557–1593. [Google Scholar] [CrossRef]
- Waseem, F.; Khemomal, K.A.; Sajid, R. Antioxidant status in beta thalassemia major: A single-center study. Indian J. Pathol. Microbiol. 2011, 54, 761–763. [Google Scholar] [PubMed]
- Garelnabi, M.; Paradhan, P. Splenectomy may not influence glutathione metabolism in children with beta-thalassaemia major. Turk. J. Haematol. 2005, 22, 25–30. [Google Scholar] [PubMed]
- Gizi, A.; Papassotiriou, I.; Apostolakou, F.; Lazaropoulou, C.; Papastamataki, M.; Kanavaki, I.; Kalotychou, V.; Goussetis, E.; Kattamis, A.; Rombos, I.; et al. Assessment of oxidative stress in patients with sickle cell disease: The glutathione system and the oxidant-antioxidant status. Blood Cells Mol. Dis. 2011, 46, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Aslan, M.; Canatan, D. Modulation of redox pathways in neutrophils from sickle cell disease patients. Exp. Hematol. 2008, 36, 1535–1544. [Google Scholar] [CrossRef]
- Tsantes, A.E.; Bonovas, S.; Travlou, A.; Sitaras, N.M. Redox imbalance, macrocytosis, and RBC homeostasis. Antioxid. Redox Signal. 2006, 8, 1205–1216. [Google Scholar] [CrossRef]
- Walter, P.B.; Fung, E.B.; Killilea, D.W.; Jiang, Q.; Hudes, M.; Madden, J.; Porter, J.; Evans, P.; Vichinsky, E.; Harmatz, P. Oxidative stress and inflammation in iron-overloaded patients with beta-thalassaemia or sickle cell disease. Br. J. Haematol. 2006, 135, 254–263. [Google Scholar] [CrossRef]
- Chen, J.J. Regulation of protein synthesis by the heme-regulated eIF2alpha kinase: Relevance to anemias. Blood 2007, 109, 2693–2699. [Google Scholar] [CrossRef]
- Lu, L.; Han, A.P.; Chen, J.J. Translation initiation control by heme-regulated eukaryotic initiation factor 2alpha kinase in erythroid cells under cytoplasmic stresses. Mol. Cell Biol. 2001, 21, 7971–7980. [Google Scholar] [CrossRef]
- Han, A.P.; Yu, C.; Lu, L.; Fujiwara, Y.; Browne, C.; Chin, G.; Fleming, M.; Leboulch, P.; Orkin, S.H.; Chen, J.J. Heme-regulated eIF2α kinase (HRI) is required for translational regulation and survival of erythroid precursors in iron deficiency. EMBO J. 2001, 20, 6909–6918. [Google Scholar] [CrossRef]
- Chen, J.J. Translational control by heme-regulated eIF2alpha kinase during erythropoiesis. Curr. Opin. Hematol. 2014, 21, 172–178. [Google Scholar] [CrossRef]
- Chen, J.J.; Zhang, S. Heme-regulated eIF2alpha kinase in erythropoiesis and hemoglobinopathies. Blood 2019, 134, 1697–1707. [Google Scholar] [CrossRef] [PubMed]
- Pittala, V.; Salerno, L.; Romeo, G.; Modica, M.N.; Siracusa, M.A. A focus on heme oxygenase-1 (HO-1) inhibitors. Curr. Med. Chem. 2013, 20, 3711–3732. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Santos, D.; Hamdi, A.; Saxova, Z.; Fillebeen, C.; Pantopoulos, K.; Horvathova, M.; Ponka, P. Inhibition of heme oxygenase ameliorates anemia and reduces iron overload in a beta-thalassemia mouse model. Blood 2018, 131, 236–246. [Google Scholar] [CrossRef]
- Lai, M.I.; Jiang, J.; Silver, N.; Best, S.; Menzel, S.; Mijovic, A.; Colella, S.; Ragoussis, J.; Garner, C.; Weiss, M.J. α-Haemoglobin stabilising protein is a quantitative trait gene that modifies the phenotype of β-thalassaemia. Br. J. Haematol. 2006, 133, 675–682. [Google Scholar] [CrossRef]
- Kong, Y.; Zhou, S.; Kihm, A.J.; Katein, A.M.; Yu, X.; Gell, D.A.; Mackay, J.P.; Adachi, K.; Foster-Brown, L.; Louden, C.S.; et al. Loss of alpha-hemoglobin-stabilizing protein impairs erythropoiesis and exacerbates beta-thalassemia. J. Clin. Investig. 2004, 114, 1457–1466. [Google Scholar] [CrossRef]
- Mahmoud, H.M.; Shoeib, A.A.; Abd El Ghany, S.M.; Reda, M.M.; Ragab, I.A. Study of alpha hemoglobin stabilizing protein expression in patients with beta thalassemia and sickle cell anemia and its impact on clinical severity. Blood Cells Mol. Dis. 2015, 55, 358–362. [Google Scholar] [CrossRef]
- Dissayabutra, T.; Tosukhowong, P.; Seksan, P. The benefits of vitamin C and vitamin E in children with beta-thalassemia with high oxidative stress. J. Med. Assoc. Thai. 2005, 88 (Suppl. S4), S317–S321. [Google Scholar] [PubMed]
- Tesoriere, L.; D’Arpa, D.; Butera, D.; Allegra, M.; Renda, D.; Maggio, A.; Bongiorno, A.; Livrea, M.A. Oral supplements of vitamin E improve measures of oxidative stress in plasma and reduce oxidative damage to LDL and erythrocytes in beta-thalassemia intermedia patients. Free Radic. Res. 2001, 34, 529–540. [Google Scholar] [CrossRef]
- Ngarmpattarangkoon, D.; Pongtanakul, B.; Chanvorachote, P.; Meksawan, K. Effect of Vitamin E supplementation on oxidative stress in non-transfusion-dependent thalassemia. Prog. Nutr. 2016, 18, 258–265. [Google Scholar]
- Haghpanah, S.; Cohan, N.; Bordbar, M.; Bazrafshan, A.; Karimi, M.; Zareifar, S.; Safaei, S.; Aramesh, A.; Moghadam, M.; Fard, S.A.Z.; et al. Effects of three months of treatment with vitamin E and N-acetyl cysteine on the oxidative balance in patients with transfusion-dependent beta-thalassemia. Ann. Hematol. 2021, 100, 635–644. [Google Scholar] [CrossRef]
- Darvishi-Khezri, H.; Salehifar, E.; Kosaryan, M.; Karami, H.; Alipour, A.; Shaki, F.; Aliasgharian, A. The impact of silymarin on antioxidant and oxidative status in patients with beta-thalassemia major: A crossover, randomized controlled trial. Complement. Ther. Med. 2017, 35, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Hatairaktham, S.; Masaratana, P.; Hantaweepant, C.; Srisawat, C.; Sirivatanauksorn, V.; Siritanaratkul, N.; Panichkul, N.; Kalpravidh, R.W. Curcuminoids supplementation ameliorates iron overload, oxidative stress, hypercoagulability, and inflammation in non-transfusion-dependent beta-thalassemia/Hb E patients. Ann. Hematol. 2021, 100, 891–901. [Google Scholar] [CrossRef] [PubMed]
- Koonyosying, P.; Tantiworawit, A.; Hantrakool, S.; Utama-Ang, N.; Cresswell, M.; Fucharoen, S.; Porter, J.B.; Srichairatanakool, S. Consumption of a green tea extract-curcumin drink decreases blood urea nitrogen and redox iron in beta-thalassemia patients. Food Funct. 2020, 11, 932–943. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.; Prasad, A.S.; Beck, F.W.; Snell, D.; Suneja, A.; Sarkar, F.H.; Doshi, N.; Fitzgerald, J.T.; Swerdlow, P. Zinc supplementation decreases oxidative stress, incidence of infection, and generation of inflammatory cytokines in sickle cell disease patients. Transl. Res. 2008, 152, 67–80. [Google Scholar] [CrossRef]
- Ozdemir, Z.C.; Koc, A.; Aycicek, A.; Kocyigit, A. N-Acetylcysteine supplementation reduces oxidative stress and DNA damage in children with beta-thalassemia. Hemoglobin 2014, 38, 359–364. [Google Scholar] [CrossRef]
- Pattanakuhar, S.; Phrommintikul, A.; Tantiworawit, A.; Srichairattanakool, S.; Chattipakorn, S.C.; Chattipakorn, N. N-acetylcysteine Restored Heart Rate Variability and Prevented Serious Adverse Events in Transfusion-dependent Thalassemia Patients: A Double-blind Single Center Randomized Controlled Trial. Int. J. Med. Sci. 2020, 17, 1147–1155. [Google Scholar] [CrossRef]
- Nur, E.; Brandjes, D.P.; Teerlink, T.; Otten, H.M.; Oude Elferink, R.P.; Muskiet, F.; Evers, L.M.; Ten Cate, H.; Biemond, B.J.; Duits, A.J.; et al. N-acetylcysteine reduces oxidative stress in sickle cell patients. Ann. Hematol. 2012, 91, 1097–1105. [Google Scholar] [CrossRef]
- Sharifi-Zahabi, E.; Abdollahzad, H.; Mostafa Nachvak, S.; Moloudi, J.; Golpayegani, M.R.; Asiaei, S.; Rezavand, L.; Iraji, Z.; Jamshidi, K. Effects of alpha lipoic acid on iron overload, lipid profile and oxidative stress indices in beta-thalassemia major patients: A cross-over randomised controlled clinical trial. Int. J. Clin. Pract. 2021, 75, e14062. [Google Scholar] [CrossRef]
- Kapoor, S.; Little, J.A.; Pecker, L.H. Advances in the Treatment of Sickle Cell Disease. Mayo Clin. Proc. 2018, 93, 1810–1824. [Google Scholar] [CrossRef]
- Lal, A.; Suh, J.; Atamna, W.; Canty, B.; Hagar, W.; Vichinsky, E.; Kuypers, F.; Ames, B. Anti-Oxidant Treatment with α-Lipoic Acid and Acetyl L-Carnitine in Hemoglobinopathies. Blood 2007, 110, 3799. [Google Scholar] [CrossRef]
- Morris, C.R.; Brown, L.A.S.; Reynolds, M.; Dampier, C.D.; Lane, P.A.; Watt, A.; Kumari, P.; Harris, F.; Manoranjithan, S.; Mendis, R.D.; et al. Impact of arginine therapy on mitochondrial function in children with sickle cell disease during vaso-occlusive pain. Blood 2020, 136, 1402–1406. [Google Scholar] [CrossRef] [PubMed]
- Jaja, S.I.; Ogungbemi, S.O.; Kehinde, M.O.; Anigbogu, C.N. Supplementation with l-arginine stabilizes plasma arginine and nitric oxide metabolites, suppresses elevated liver enzymes and peroxidation in sickle cell anaemia. Pathophysiology 2016, 23, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Fibach, E.; Tan, E.S.; Jamuar, S.; Ng, I.; Amer, J.; Rachmilewitz, E.A. Amelioration of oxidative stress in red blood cells from patients with beta-thalassemia major and intermedia and E-beta-thalassemia following administration of a fermented papaya preparation. Phytother. Res. 2010, 24, 1334–1338. [Google Scholar] [CrossRef] [PubMed]
- Daak, A.A.; Ghebremeskel, K.; Mariniello, K.; Attallah, B.; Clough, P.; Elbashir, M.I. Docosahexaenoic and eicosapentaenoic acid supplementation does not exacerbate oxidative stress or intravascular haemolysis in homozygous sickle cell patients. Prostaglandins Leukot. Essent. Fat. Acids 2013, 89, 305–311. [Google Scholar] [CrossRef]
- Kaddam, L.; Fadl-Elmula, I.; Eisawi, O.A.; Abdelrazig, H.A.; Salih, M.A.; Lang, F.; Saeed, A.M. Gum Arabic as novel anti-oxidant agent in sickle cell anemia, phase II trial. BMC Hematol. 2017, 17, 4. [Google Scholar] [CrossRef]
- Kaddam, L.; Fadl-Elmula, I.; Eisawi, O.A.; Abdelrazig, H.A.; Saeed, A.M. Acacia senegal (Gum Arabic) supplementation modulate lipid profile and ameliorated dyslipidemia among sickle cell anemia patients. J. Lipids 2019, 2019, 3129461. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).