Systemic Inflammation, Oxidative Stress and Cardiovascular Health in Children and Adolescents: A Systematic Review
Abstract
1. Systemic Inflammation and Oxidative Stress Influence Cardiovascular Health in Children and Adolescents
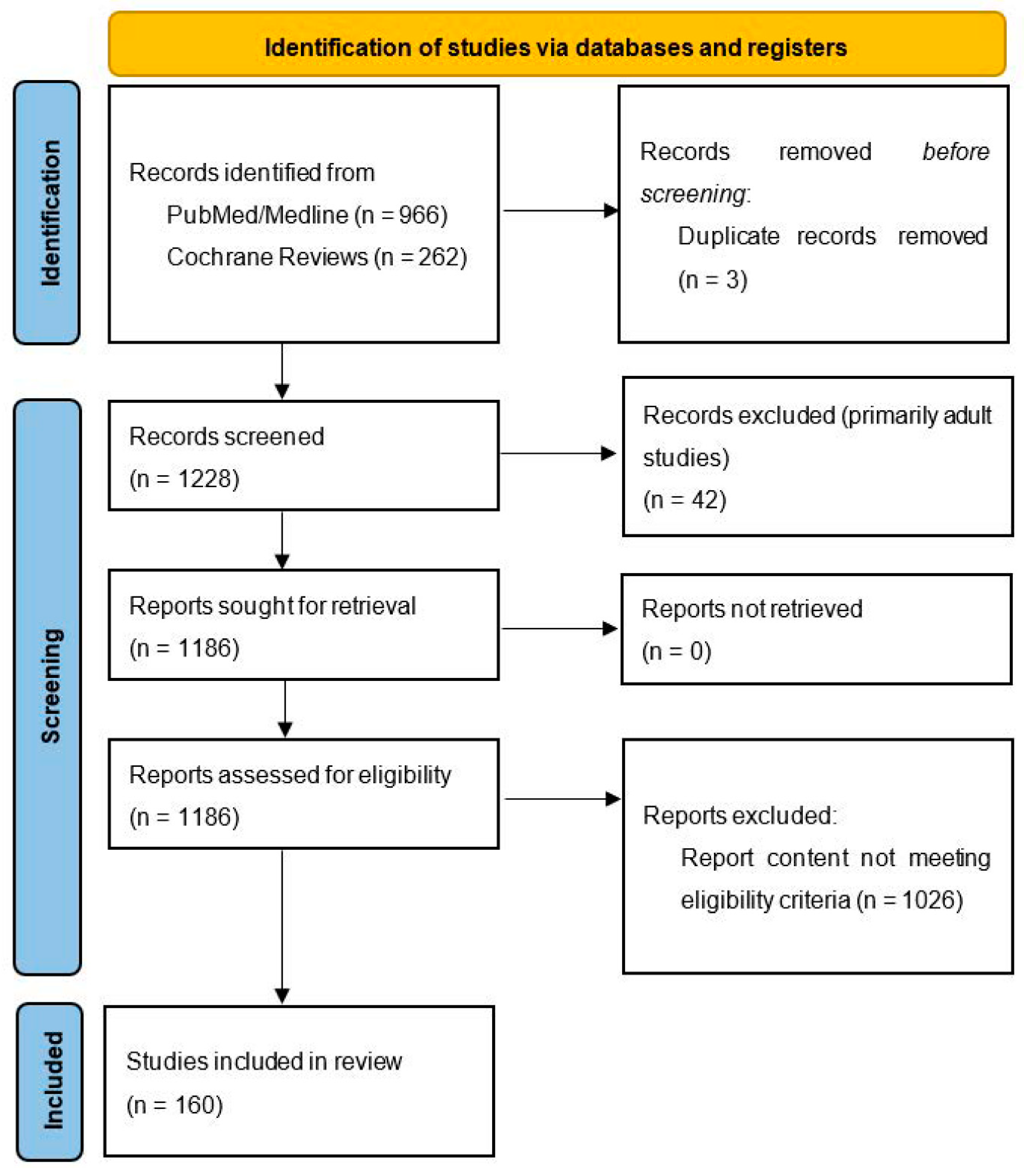
2. Materials and Methods
3. Genetic and Perinatal Factors Affect Oxidative Stress and Systemic Inflammation
4. Effect of Selected Chronic Diseases on Cardiovascular Health via Oxidative Stress and Systemic Inflammation
4.1. Hypertension and Vascular Dysfunction
4.2. Obesity
4.3. Metabolic Syndrome and Type 2 Diabetes Mellitus
4.4. Hyperlipidemia
4.5. Chronic Kidney Disease and Dialysis
4.6. Obstructive Sleep Apnea
4.7. Type 1 Diabetes Mellitus
5. Influence of Exercise and Diet on Oxidative Stress, Inflammation and Cardiovascular Health in Children and Adolescents
6. Use of Antioxidants and Their Effects on Cardiovascular Health
6.1. Vitamins, Minerals and Coenzymes
6.2. L-Arginine
6.3. Melatonin
6.4. Flavonoids and Natural Antioxidants
6.5. Alpha-Lipoic Acid
7. Other Influences on Systemic Inflammation and Oxidative Stress in Childhood and Adolescence
8. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Berrahmoune, H.; Lamont, J.; Fitzgerald, P.; Visvikis-Siest, S. Inter-individual variation of inflammatory markers of cardiovascular risks and diseases. Clin. Chem. Lab. Med. (CCLM) 2005, 43, 671–684. [Google Scholar] [CrossRef] [PubMed]
- Barton, M. Obesity and aging: Determinants of endothelial cell dysfunction and atherosclerosis. J. Physiol. 2010, 460, 825–837. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, M.; Amin, M.M.; Chavoshani, A.; Rafiei, N.; Ebrahimpour, K.; Kelishadi, R. Relationship of urinary phthalate metabolites with cardiometabolic risk factors and oxidative stress markers in children and adolescents. J. Environ. Public Health 2021, 2021, 5514073. [Google Scholar] [CrossRef] [PubMed]
- Eikendal, A.L.M.; Evelein, A.M.V.; Uiterwaal, C.S.P.M.; van der Ent, C.K.; Visseren, F.L.J.; Bots, M.L.; Hoefer, I.E.; den Ruijter, H.M.; Dalmeijer, G.W. Relation between circulating inflammatory chemokines and vascular characteristics in healthy, young children. J. Am. Heart Assoc. 2015, 4. [Google Scholar] [CrossRef]
- Kelishadi, R.; Sharifi, M.; Khosravi, A.; Adeli, K. Relationship Between C-reactive protein and atherosclerotic risk factors and oxidative stress markers among young persons 10–18 years old. Clin. Chem. 2007, 53, 456–464. [Google Scholar] [CrossRef]
- Mushenkova, N.V.; Bezsonov, E.E.; Orekhova, V.A.; Popkova, T.V.; Starodubova, A.V.; Orekhov, A.N. Recognition of oxidized lipids by macrophages and its role in atherosclerosis development. Biomedicines 2021, 9, 915. [Google Scholar] [CrossRef]
- De Souza, F.I.S.; D’Almeida, V.; Fonseca, F.L.A.; Hix, S.; Miranda, R.; Rossi, R.G.d.T.; Ribeiro, R.; Sarni, R.O.S. Lack of association of homocysteine concentrations with oxidative stress, alterations in carotid intima media thickness and endothelial reactivity in prepubertal children. Ann. Nutr. Metab. 2013, 63, 25–31. [Google Scholar] [CrossRef]
- Wilders-Truschnig, M.; Mangge, H.; Lieners, C.; Gruber, H.J.; Mayer, C.; März, W. IgG antibodies against food antigens are correlated with inflammation and intima media thickness in obese juveniles. Exp. Clin. Endocrinol. Diabetes 2008, 116, 241–245. [Google Scholar] [CrossRef]
- Tsukahara, H. Biomarkers for oxidative stress: Clinical application in pediatric medicine. Curr. Med. Chem. 2007, 14, 339–351. [Google Scholar] [CrossRef]
- Hara, T.; Yamamura, K.; Sakai, Y. The up-to-date pathophysiology of Kawasaki disease. Clin. Transl. Immunol. 2021, 10, e1284. [Google Scholar] [CrossRef]
- Graciano-Machuca, O.; Villegas-Rivera, G.; López-Pérez, I.; Macías-Barragán, J.; Sifuentes-Franco, S. Multisystem Inflammatory Syndrome in Children (MIS-C) following SARS-CoV-2 infection: Role of oxidative stress. Front. Immunol. 2021, 12, 723654. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Cruz, A.R.; Páez, A.; Jimenez-Flores, R.; Reyes-Reali, J.; Varela, E.; Cérbulo-Vázquez, A.; Rodriguez, E.; López-Marure, R.; Massó, F.A.; Flores-Romo, L.; et al. Increased expression of inflammation-related co-stimulatory molecules by HUVECs from newborns with a strong family history of myocardial infarction stimulated with TNF-α and oxLDL. Immunol. Lett. 2007, 111, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Schoeps, D.O.; Holzer, S.; Suano-Souza, F.I.; Hix, S.; Fonseca, F.L.A.; Sarni, R.O.S. Myeloperoxidase as cardiovascular risk marker in pre-pubertal preterm children? Nutr. Metab. Cardiovasc. Dis. 2019, 29, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Sakka, S.D.; Loutradis, D.; Kanaka-Gantenbein, C.; Margeli, A.; Papastamataki, M.; Papassotiriou, I.; Chrousos, G.P. Absence of insulin resistance and low-grade inflammation despite early metabolic syndrome manifestations in children born after in vitro fertilization. Fertil. Steril. 2010, 94, 1693–1699. [Google Scholar] [CrossRef]
- Kelishadi, R.; Sabri, M.; Motamedi, N.; Ramezani, M.A. Factor analysis of markers of inflammation and oxidation and echocardiographic findings in children with a positive family history of premature coronary heart disease. Pediatr. Cardiol. 2009, 30, 477–481. [Google Scholar] [CrossRef]
- de Giorgis, T.; Giannini, C.; Scarinci, A.; D’Adamo, E.; Agostinelli, S.; Chiarelli, F.; Mohn, A. Family history of premature cardiovascular disease as a sole and independent risk factor for increased carotid intima–media thickness. J. Hypertens. 2009, 27, 822–828. [Google Scholar] [CrossRef]
- Craig, A.; Mels, C.M.C.; Kruger, R. Thiobarbituric acid-reactive substances relate to arterial stiffness and blood pressure in 6 to 8-year-old boys stratified by maternal risk. Free Radic. Res. 2018, 52, 180–187. [Google Scholar] [CrossRef]
- Gonzalez-Enríquez, G.V.G.; Rubio-Benítez, M.I.; García-Gallegos, V.; Portila de Buen, E.P.; Troyo-Sanromán, R.; Leal-Cortés, C.A. Contribution of TNF-308A and CCL2-2518A to carotid intima-media thickness in obese Mexican children and adolescents. Arch. Med. Res. 2008, 39, 753–759. [Google Scholar] [CrossRef]
- Coelho, C.; Guerra, A.; Rego, C.; Breitenfeld, L.; Castro, E.; Rodrigues, P.; Laires, M.J.; Bicho, M. Genetic polymorphisms of angiotensin-I converting enzyme, haptoglobin and angiotensinogen and oxidative stress parameters in 12 to 15-year-old adolescents. Rev. Port. De Cardiol. 2006, 25, 677–690. [Google Scholar]
- Guerra, A.; Rego, C.; Castro, E.M.; Sinde, S.; Silva, D.; Rodrigues, P.; Crespo, M.E.; Albergaria, F.; Bicho, M. Acid phosphatase, genetic polymorphism and cardiovascular risk factors in a pediatric population. Rev. Port. Cardiol. 2000, 19, 679–691. [Google Scholar]
- Oztezcan, S.; Doğru-Abbasoğlu, S.; Mutlu-Türkoğlu, U.; Cetiner, S.; Eker-Omeroğlu, R.; Aykaç-Toker, G.; Uysal, M. The investigation of prooxidant-antioxidant balance in serum of children with family histories of essential hypertension. Res. Commun. Mol. Pathol. Pharmacol. 2002, 111, 167–174. [Google Scholar]
- Hapyn, E.; Czerwionka-Szaflarska, M.; Drewa, G. Enzymatic efficiency of erythrocyte antioxidant barrier and lipid peroxidation in children from families with high risk of early atherosclerosis. Med. Sci. Monit. 2000, 6, 112–116. [Google Scholar] [PubMed]
- Kolesnikova, L.I.; Prokhorova, Z.V.; Vlasov, B.Y.; Polyakov, V.M. Redox Status as a Metabolic Stage, Integrating Emotional pattern and blood pressure in adolescents. Bull. Exp. Biol. Med. 2014, 158, 9–12. [Google Scholar] [CrossRef]
- Ostalska-Nowicka, D.; Paszyńska, E.; Dmitrzak-Węglarz, M.; Neyman-Bartkowiak, A.; Rabiega, A.; Zachwieja, J.; Nowicki, M. Dental caries-related primary hypertension in children and adolescents: Cross-sectional study. Oral Dis. 2021, 27, 1822–1833. [Google Scholar] [CrossRef] [PubMed]
- Śladowska-Kozłowska, J.; Litwin, M.; Niemirska, A.; Wierzbicka, A.; Roszczynko, M.; Szperl, M. Associations of the eNOS G894T gene polymorphism with target organ damage in children with newly diagnosed primary hypertension. Pediatr. Nephrol. 2015, 30, 2189–2197. [Google Scholar] [CrossRef][Green Version]
- Baráth, Á.; Németh, I.; Karg, E.; Endreffy, E.; Bereczki, C.; Gellén, B.; Haszon, I.; Túri, S. Roles of Paraoxonase and oxidative stress in adolescents with uraemic, essential or obesity-induced hypertension. Kidney Blood Press. Res. 2006, 29, 144–151. [Google Scholar] [CrossRef]
- Litwin, M.; Michałkiewicz, J.; Niemirska, A.; Gackowska, L.; Kubiszewska, I.; Wierzbicka-Rucinska, A.; Wawer, Z.T.; Janas, R. Inflammatory activation in children with primary hypertension. Pediatr. Nephrol. 2010, 25, 1711–1718. [Google Scholar] [CrossRef]
- Túri, S.; Friedman, A.; Bereczki, C.; Papp, F.; Kovàcs, J.; Karg, E.; Németh, I. Oxidative stress in juvenile essential hypertension. J. Hypertens. 2003, 21, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Śladowska-Kozłowska, J.; Litwin, M.; Niemirska, A.; Płudowski, P.; Wierzbicka, A.; Skorupa, E.; Wawer, Z.T.; Janas, R. Oxidative stress in hypertensive children before and after 1 year of antihypertensive therapy. Pediatr. Nephrol. 2012, 27, 1943–1951. [Google Scholar] [CrossRef]
- Yamano, Y.; Miyakawa, S.; Nakadate, T. Association of arteriosclerosis index and oxidative stress markers in school children. Pediatr. Int. 2015, 57, 449–454. [Google Scholar] [CrossRef]
- Çakıcı, E.K.; Eroglu, F.K.; Yazılıtaş, F.; Bülbül, M.; Gur, G.; Aydoğ, O.; Güngör, T.; Erel, O.; Alisik, M.; Elhan, A.H. Evaluation of the level of dynamic thiol/disulphide homeostasis in adolescent patients with newly diagnosed primary hypertension. Pediatr. Nephrol. 2018, 33, 847–853. [Google Scholar] [CrossRef]
- Stoppa-Vaucher, S.; Dirlewanger, M.A.; Meier, C.A.; De Moerloose, P.; Reber, G.; Roux-Lombard, P.; Combescure, C.; Saudan, S.; Schwitzgebel, V.M. Inflammatory and prothrombotic states in obese children of European descent. Obesity 2012, 20, 1662–1668. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, M.V.; Estepa, R.M.; Camacho, R.M.M.; Estrada, R.C.; Luna, F.G.; Guitarte, F.B. Endothelial dysfunction is related to insulin resistance and inflammatory biomarker levels in obese prepubertal children. Eur. J. Endocrinol. 2007, 156, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Mauras, N.; DelGiorno, C.; Kollman, C.; Bird, K.; Morgan, M.; Sweeten, S.; Balagopal, P.; Damaso, L. Obesity without established comorbidities of the metabolic syndrome is associated with a proinflammatory and prothrombotic state, even before the onset of puberty in children. J. Clin. Endocrinol. Metab. 2010, 95, 1060–1068. [Google Scholar] [CrossRef]
- Morandi, A.; Corradi, M.; Piona, C.; Fornari, E.; Puleo, R.; Maffeis, C. Systemic anti-oxidant capacity is inversely correlated with systolic blood pressure and pulse pressure in children with obesity. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 508–513. [Google Scholar] [CrossRef]
- Hacıhamdioğlu, D.O.; Zeybek, C.; Gök, F.; Pekel, A.; Musabak, U. Elevated urinary T helper 1 chemokine levels in newly diagnosed hypertensive obese children. J. Clin. Res. Pediatr. Endocrinol. 2015, 7, 175–182. [Google Scholar] [CrossRef]
- Syrenicz, A.; Garanty-Bogacka, B.; Syrenicz, M.; Gebala, A.; Dawid, G.; Walczak, M. Relation of low-grade inflammation and endothelial activation to blood pressure in obese children and adolescents. Neuro Endocrinol. Lett. 2006, 27, 459–464. [Google Scholar]
- Aburawi, E.H.; Al Hamad, S.; Yasin, J.; Almekhaini, L.A.; Souid, A.-K. Dyslipidemia, subclinical inflammation, hepatic cholestasis and endothelial dysfunction in schoolchildren with excess fat: A study from the United Arab Emirates. PLoS ONE 2019, 14, e0210316. [Google Scholar] [CrossRef]
- Al-Shorman, A.; Al-Domi, H.; Faqih, A. Markers of subclinical atherosclerosis in schoolchildren with obesity and metabolic syndrome. Swiss Med. Wkly. 2017, 147, w14446. [Google Scholar] [CrossRef]
- Giannini, C.; de Giorgis, T.; Scarinci, A.; Ciampani, M.; Marcovecchio, M.L.; Chiarelli, F.; Mohn, A. Obese related effects of inflammatory markers and insulin resistance on increased carotid intima media thickness in pre-pubertal children. Atherosclerosis 2008, 197, 448–456. [Google Scholar] [CrossRef]
- Sinaiko, A.R.; Steinberger, J.; Moran, A.; Prineas, R.J.; Vessby, B.; Basu, S.; Tracy, R.; Jacobs, D.R., Jr. Relation of body mass index and insulin resistance to cardiovascular risk factors, inflammatory factors, and oxidative stress during adolescence. Circulation 2005, 111, 1985–1991. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.; Fischer, N.; Fritzenwanger, M.; Pernow, J.; Brehm, B.R.; Figulla, H.R. Association of waist circumference, traditional cardiovascular risk factors, and stromal-derived factor-1 in adolescents. Pediatr. Diabetes 2009, 10, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Codoñer-Franch, P.; Tavárez-Alonso, S.; Murria-Estal, R.; Tortajada-Girbés, M.; Simó-Jordá, R.; Alonso-Iglesias, E. Elevated advanced oxidation protein products (AOPPs) indicate metabolic risk in severely obese children. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 237–243. [Google Scholar] [CrossRef]
- Okur, I.; Tumer, L.; Ezgu, F.S.; Yesilkaya, E.; Aral, A.; Oktar, S.O.; Bideci, A.; Hasanoglu, A. Oxidized low-density lipoprotein levels and carotid intima-media thickness as markers of early atherosclerosis in prepubertal obese children. J. Pediatr. Endocrinol. Metab. 2013, 26, 657–662. [Google Scholar] [CrossRef]
- Elmas, B.; Karacan, M.; Dervişoğlu, P.; Kösecik, M.; İşgüven, Ş.P.; Bal, C. Dynamic thiol/disulphide homeostasis as a novel indicator of oxidative stress in obese children and its relationship with inflammatory-cardiovascular markers. Anatol. J. Cardiol. 2017, 18, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Correia-Costa, L.; Sousa, T.; Morato, M.; Cosme, D.; Afonso, J.; Areias, J.C.; Schaefer, F.; Guerra, A.; Afonso, A.C.; Azevedo, A.; et al. Oxidative stress and nitric oxide are increased in obese children and correlate with cardiometabolic risk and renal function. Br. J. Nutr. 2016, 116, 805–815. [Google Scholar] [CrossRef]
- Codoñer-Franch, P.; Tavárez-Alonso, S.; Murria-Estal, R.; Herrera-Martín, G.; Alonso-Iglesias, E. Polyamines are increased in obese children and are related to markers of oxidative/nitrosative stress and angiogenesis. J. Clin. Endocrinol. Metab. 2011, 96, 2821–2825. [Google Scholar] [CrossRef]
- Landgraf, K.; Friebe, D.; Ullrich, T.; Kratzsch, J.; Dittrich, K.; Herberth, G.; Adams, V.; Kiess, W.; Erbs, S.; Körner, A. Chemerin as a mediator between obesity and vascular inflammation in children. J. Clin. Endocrinol. Metab. 2012, 97, E556–E564. [Google Scholar] [CrossRef]
- Niklowitz, P.; Rothermel, J.; Lass, N.; Barth, A.; Reinehr, T. Link between chemerin, central obesity, and parameters of the metabolic syndrome: Findings from a longitudinal study in obese children participating in a lifestyle intervention. Int. J. Obes. 2018, 42, 1743–1752. [Google Scholar] [CrossRef]
- Wójcik, M.; Kozioł-Kozakowska, A.; Januś, D.; Furtak, A.; Małek, A.; Sztefko, K.; Starzyk, J.B. Circulating chemerin level may be associated with early vascular pathology in obese children without overt arterial hypertension–preliminary results. J. Pediatr. Endocrinol. Metab. 2020, 33, 729–734. [Google Scholar] [CrossRef]
- Simunovic, M.; Supe-Domic, D.; Karin, Z.; Degoricija, M.; Paradzik, M.; Bozic, J.; Unic, I.; Skrabic, V. Serum catestatin concentrations are decreased in obese children and adolescents. Pediatr. Diabetes 2019, 20, 549–555. [Google Scholar] [CrossRef]
- Makni, E.; Moalla, W.; Benezzeddine-Boussaidi, L.; Lac, G.; Tabka, Z.; Elloumi, M. Correlation of resistin with inflammatory and cardiometabolic markers in obese adolescents with and without metabolic syndrome. Obes. Facts 2013, 6, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Stringer, D.M.; Sellers, E.A.C.; Burr, L.L.; Taylor, C.G. Altered plasma adipokines and markers of oxidative stress suggest increased risk of cardiovascular disease in First Nation youth with obesity or type 2 diabetes mellitus. Pediatr. Diabetes 2009, 10, 269–277. [Google Scholar] [CrossRef]
- Stelzer, I.; Zelzer, S.; Raggam, R.B.; Prüller, F.; Truschnig-Wilders, M.; Meinitzer, A.; Schnedl, W.J.; Horejsi, R.; Möller, R.; Weghuber, D.; et al. Link between leptin and interleukin-6 levels in the initial phase of obesity related inflammation. Transl. Res. 2012, 159, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Mohanraj, L.; Kim, H.-S.; Li, W.; Cai, Q.; Kim, K.E.; Shin, H.-J.; Lee, Y.-J.; Lee, W.J.; Kim, J.H.; Oh, Y. IGFBP-3 inhibits cytokine-induced insulin resistance and early manifestations of atherosclerosis. PLoS ONE 2013, 8, e55084. [Google Scholar] [CrossRef]
- Ostrow, V.; Wu, S.; Aguilar, A.; Bonner, R., Jr.; Suarez, E.; De Luca, F. Association between oxidative stress and masked hypertension in a multi-ethnic population of obese children and adolescents. J. Pediatr. 2011, 158, 628–633.e1. [Google Scholar] [CrossRef] [PubMed]
- Selvaraju, V.; Ayine, P.; Fadamiro, M.; Babu, J.R.; Brown, M.; Geetha, T. Urinary biomarkers of inflammation and oxidative stress are elevated in obese children and correlate with a marker of endothelial dysfunction. Oxidative Med. Cell. Longev. 2019, 2019, 9604740. [Google Scholar] [CrossRef]
- Metzig, A.M.; Schwarzenberg, S.J.; Fox, C.K.; Deering, M.M.; Nathan, B.M.; Kelly, A.S. Postprandial endothelial function, inflammation, and oxidative stress in obese children and adolescents. Obesity 2011, 19, 1279–1283. [Google Scholar] [CrossRef]
- Codoner-Franch, P.; Tavarez-Alonso, S.; Simo-Jorda, R.; Laporta-Martin, P.; Carratala-Calvo, A.; Alonso-Iglesias, E. Vitamin D status is linked to biomarkers of oxidative stress, inflammation, and endothelial activation in obese children. J. Pediatr. 2012, 161, 848–854. [Google Scholar] [CrossRef]
- Rajakumar, K.; Moore, C.G.; Khalid, A.T.; Vallejo, A.N.; Virji, M.A.; Holick, M.F.; Greenspan, S.L.; Arslanian, S.; Reis, S.E. Effect of vitamin D3 supplementation on vascular and metabolic health of vitamin D–deficient overweight and obese children: A randomized clinical trial. Am. J. Clin. Nutr. 2020, 111, 757–768. [Google Scholar] [CrossRef]
- Al-Daghri, N.M.; Rahman, S.; Sabico, S.; Yakout, S.; Wani, K.; Al-Attas, O.S.; Saravanan, P.; Tripathi, G.; McTernan, P.G.; Alokail, M.S. Association of Vitamin B12 with pro-inflammatory cytokines and biochemical markers related to cardiometabolic risk in Saudi subjects. Nutrients 2016, 8, 460. [Google Scholar] [CrossRef] [PubMed]
- Economou, E.V.; Malamitsi-Puchner, A.V.; Pitsavos, C.P.; Kouskouni, E.E.; Magaziotou-Elefsinioti, I.; Damianaki-Uranou, D.; Stefanadis, C.I.; Creatsas, G. Negative association between circulating total homocysteine and proinflammatory chemokines MCP-1 and RANTES in prepubertal lean, but not in obese, children. J. Cardiovasc. Pharmacol. 2004, 44, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Giannini, C.; De Giorgis, T.; Scarinci, A.; Cataldo, I.; Marcovecchio, M.L.; Chiarelli, F.; Mohn, A. Increased carotid intima-media thickness in pre-pubertal children with constitutional leanness and severe obesity: The speculative role of insulin sensitivity, oxidant status, and chronic inflammation. Eur. J. Endocrinol. 2009, 161, 73–80. [Google Scholar] [CrossRef]
- Maggio, A.B.R.; Farpour-Lambert, N.J.; Aggoun, Y.; Galan, K.; Montecucco, F.; Mach, F.; Beghetti, M. Serum cardiovascular risk biomarkers in pre-pubertal obese children. Eur. J. Clin. Investig. 2018, 48, e12995. [Google Scholar] [CrossRef]
- Marti, A.; Martínez, I.; Ojeda-Rodríguez, A.; Azcona-Sanjulian, M.C. Higher lipopolysaccharide binding protein and chemerin concentrations were associated with metabolic syndrome features in pediatric subjects with abdominal obesity during a lifestyle intervention. Nutrients 2021, 13, 289. [Google Scholar] [CrossRef]
- Akinci, G.; Coskun, S.; Akinci, B.; Hekimsoy, Z.; Bayindir, P.; Onur, E.; Ozmen, B. Atherosclerosis risk factors in children of parents with the metabolic syndrome. Atherosclerosis 2007, 194, e165–e171. [Google Scholar] [CrossRef]
- Olza, J.; Aguilera, C.M.; Gil-Campos, M.; Leis, R.; Bueno, G.; Valle, M.; Cañete, R.; Tojo, R.; Moreno, L.A.; Gil, A. A Continuous metabolic syndrome score is associated with specific biomarkers of inflammation and CVD risk in prepubertal children. Ann. Nutr. Metab. 2015, 66, 72–79. [Google Scholar] [CrossRef]
- González-Jiménez, E.; Schmidt-Riovalle, J.; Sinausía, L.; Carmen Valenza, M.; Perona, J.S. Predictive value of ceruloplasmin for metabolic syndrome in adolescents. BioFactors 2016, 42. [Google Scholar] [CrossRef]
- Kelly, A.S.; Steinberger, J.; Kaiser, D.R.; Olson, T.P.; Bank, A.J.; Dengel, D.R. Oxidative stress and adverse adipokine profile characterize the metabolic syndrome in children. J. CardioMetabolic Syndr. 2006, 1, 248–252. [Google Scholar] [CrossRef]
- Scuteri, A.; Orru, M.; Morrell, C.; Piras, M.G.; Taub, D.; Schlessinger, D.; Uda, M.; Lakatta, E.G. Independent and additive effects of cytokine patterns and the metabolic syndrome on arterial aging in the SardiNIA Study. Atherosclerosis 2011, 215, 459–464. [Google Scholar] [CrossRef]
- Lin, L.-Y.; Kuo, H.-K.; Hwang, J.-J.; Lai, L.-P.; Chiang, F.-T.; Tseng, C.-D.; Lin, J.-L. Serum bilirubin is inversely associated with insulin resistance and metabolic syndrome among children and adolescents. Atherosclerosis 2009, 203, 563–568. [Google Scholar] [CrossRef]
- Rupérez, A.I.; Mesa, M.D.; Anguita-Ruiz, A.; González-Gil, E.M.; Vázquez-Cobela, R.; Moreno, L.A.; Gil, A.; Gil-Campos, M.; Leis, R.; Bueno, G.; et al. Antioxidants and oxidative stress in children: Influence of puberty and metabolically unhealthy status. Antioxidants 2020, 9, 618. [Google Scholar] [CrossRef] [PubMed]
- Huerta-Delgado, A.S.; Roffe-Vazquez, D.N.; Gonzalez-Gil, A.M.; Villareal-Calderon, J.R.; Tamez-Rivera, O.; Rodriguez-Gutierrez, N.A.; Castillo, E.C.; Silva-Platas, C.; Garcia-Rivas, G.; Elizondo-Montemayor, L. Serum irisin levels, endothelial dysfunction, and inflammation in pediatric patients with type 2 diabetes mellitus and metabolic syndrome. J. Diabetes Res. 2020, 2020, 1949415. [Google Scholar] [CrossRef]
- Reilly, M.P.; Praticò, D.; Delanty, N.; DiMinno, G.; Tremoli, E.; Rader, D.; Kapoor, S.; Rokach, J.; Lawson, J.; FitzGerald, G.A. Increased formation of distinct F2 isoprostanes in hypercholesterolemia. Circulation 1998, 98, 2822–2828. [Google Scholar] [CrossRef]
- Charakida, M.; Tousoulis, D.; Skoumas, I.; Pitsavos, C.; Vasiliadou, C.; Stefanadi, E.; Antoniades, C.; Latsios, G.; Siasos, G.; Stefanadis, C. Inflammatory and thrombotic processes are associated with vascular dysfunction in children with familial hypercholesterolemia. Atherosclerosis 2009, 204, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Holven, K.B.; Damås, J.K.; Yndestad, A.; Wæhre, T.; Ueland, T.; Halvorsen, B.; Heggelund, L.; Sandberg, W.J.; Semb, A.G.; Frøland, S.S.; et al. Chemokines in children with heterozygous familiar hypercholesterolemia: Selective upregulation of RANTES. Arter. Thromb. Vasc. Biol. 2006, 26, 200–205. [Google Scholar] [CrossRef]
- Loffredo, L.; Martino, F.; Carnevale, R.; Pignatelli, P.; Catasca, E.; Perri, L.; Calabrese, C.M.; Palumbo, M.M.; Baratta, F.; Del Ben, M.; et al. Obesity and hypercholesterolemia are associated with NOX2 generated oxidative stress and arterial dysfunction. J. Pediatr. 2012, 161, 1004–1009. [Google Scholar] [CrossRef] [PubMed]
- Martino, F.; Loffredo, L.; Carnevale, R.; Sanguigni, V.; Martino, E.; Catasca, E.; Zanoni, C.; Pignatelli, P.; Violi, F. Oxidative stress is associated with arterial dysfunction and enhanced intima-media thickness in children with hypercholesterolemia: The potential role of nicotinamide-adenine dinucleotide phosphate oxidase. Pediatrics 2008, 122, e648–e655. [Google Scholar] [CrossRef]
- Ece, A.; Gurkan, F.; Kervancioglu, M.; Kocamaz, H.; Güneş, A.; Atamer, Y.; Selek, S.; Yolbaş, I. Oxidative stress, inflammation and early cardiovascular damage in children with chronic renal failure. Pediatr. Nephrol. 2006, 21, 545–552. [Google Scholar] [CrossRef]
- Ece, A.; Atamer, Y.; Gürkan, F.; Davutoğlu, M.; Bilici, M.; Tutanç, M.; Güneş, A. Paraoxonase, anti-oxidant response and oxidative stress in children with chronic renal failure. Pediatr. Nephrol. 2006, 21, 239–245. [Google Scholar] [CrossRef]
- Hamed, E.A.; El-Abaseri, T.B.; Mohamed, A.O.; Ahmed, A.R.; El-Metwally, T.H. Hypoxia and oxidative stress markers in pediatric patients undergoing hemodialysis: Cross section study. BMC Nephrol. 2012, 13, 136. [Google Scholar] [CrossRef] [PubMed]
- Zachwieja, J.; Zaniew, M.; Runowski, D.; Lewandowska-Stachowiak, M.; Stefaniak, E.; Siwińska, A. Abnormal cytokine synthesis as a consequence of increased intracellular oxidative stress in children treated with dialysis. Nephron Clin. Pract. 2005, 101, c100–c108. [Google Scholar] [CrossRef] [PubMed]
- Maciejczyk, M.; Szulimowska, J.; Skutnik, A.; Taranta-Janusz, K.; Wasilewska, A.; Wiśniewska, N.; Zalewska, A. Salivary biomarkers of oxidative stress in children with chronic kidney disease. J. Clin. Med. 2018, 7, 209. [Google Scholar] [CrossRef] [PubMed]
- Al-Biltagi, M.; Tolba, O.A.; ElHafez, M.A.; Abo-Elezz, A.A.; EL Kady, E.K.; Hazza, S.M. Oxidative stress and cardiac dysfunction in children with chronic renal failure on regular hemodialysis. Pediatr. Nephrol. 2016, 31, 1329–1339. [Google Scholar] [CrossRef]
- Elshamaa, M.F.; Sabry, S.; Nabih, M.; ElGhoroury, E.A.; El-Saaid, G.S.; Ismail, A.A. Oxidative stress markers and c-reactive protein in pediatric patients on hemodialysis. Ann. Nutr. Metab. 2009, 55, 309–316. [Google Scholar] [CrossRef]
- El-Saeed, G.S.; Fadel, F.; Elshamaa, M.F.; Galal, R.E.; ElGhoroury, E.A.; Nasr, S.A.; Thabet, E.H.; Abdelrahman, S.M. Advanced glycation end products and soluble receptor as markers of oxidative stress in children on hemodialysis. Ren. Fail. 2015, 37, 1452–1456. [Google Scholar] [CrossRef]
- Zwolińska, D.; Grzeszczak, W.; Szczepańska, M.; Kiliś-Pstrusińska, K.; Szprynger, K. Vitamins A, E and C as Non-enzymatic antioxidants and their relation to lipid peroxidation in children with chronic renal failure. Nephron Clin. Pract. 2005, 103, c12–c18. [Google Scholar] [CrossRef]
- Zwołińska, D.; Grzeszczak, W.; Szczepańska, M.; Kiliś-Pstrusińska, K.; Szprynger, K. Lipid peroxidation and antioxidant enzymes in children on maintenance dialysis. Pediatr. Nephrol. 2006, 21, 705–710. [Google Scholar] [CrossRef]
- Badawy, A.; Nigm, D.A.; Ezzat, G.M.; Gamal, Y. Interleukin 18 as a new inflammatory mediator in left ventricular hypertrophy in children with end-stage renal disease. Saudi J. Kidney Dis. Transplant. 2020, 31, 1206–1216. [Google Scholar] [CrossRef]
- Drożdż, D.; Kwinta, P.; Sztefko, K.; Kordon, Z.; Drożdż, T.; Łątka, M.; Miklaszewska, M.; Zachwieja, K.; Rudziński, J.A.; Pietrzyk, J. Oxidative stress biomarkers and left ventricular hypertrophy in children with chronic kidney disease. Oxidative Med. Cell. Longev. 2016, 2016, 7520231. [Google Scholar] [CrossRef]
- Garcia-Bello, J.A.; Gómez-Díaz, R.A.; Contreras-Rodríguez, A.; Talavera, J.O.; Mondragón-González, R.; Sanchez-Barbosa, L.; Diaz-Flores, M.; Valladares-Salgado, A.; Gallardo, J.M.; Aguilar-Kitsu, A.; et al. Carotid intima media thickness, oxidative stress, and inflammation in children with chronic kidney disease. Pediatr. Nephrol. 2014, 29, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Kotur-Stevuljević, J.; Peco-Antić, A.; Spasić, S.; Stefanović, A.; Paripović, D.; Kostić, M.; Vasić, D.; Vujović, A.; Jelić-Ivanović, Z.; Spasojević-Kalimanovska, V.; et al. Hyperlipidemia, oxidative stress, and intima media thickness in children with chronic kidney disease. Pediatr. Nephrol. 2013, 28, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Cengiz, N.; Baskin, E.; Sezgin, N.; Agras, P.; Haberal, M. Oxidative stress in children on hemodialysis: Value of autoantibodies against oxidized low-density lipoprotein. Pediatr. Nephrol. 2009, 24, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Srivaths, P.R.; Silverstein, D.M.; Leung, J.; Krishnamurthy, R.; Goldstein, S.L. Malnutrition-inflammation-coronary calcification in pediatric patients receiving chronic hemodialysis. Hemodial. Int. 2010, 14, 263–269. [Google Scholar] [CrossRef]
- Calò, L.A.; Dall’Amico, R.; Pagnin, E.; Bertipaglia, L.; Zacchello, G.; Davis, P.A. Oxidative stress and post-transplant hypertension in pediatric kidney-transplanted patients. J. Pediatr. 2006, 149, 53–57. [Google Scholar] [CrossRef]
- Al-Mashhadi, A.; Checa, A.; Wåhlin, N.; Neveus, T.; Fossum, M.; Wheelock, C.E.; Karanikas, B.; Stenberg, A.; Persson, A.E.G.; Carlstrom, M. Changes in arterial pressure and markers of nitric oxide homeostasis and oxidative stress following surgical correction of hydronephrosis in children. Pediatr. Nephrol. 2018, 33, 639–649. [Google Scholar] [CrossRef]
- Pavlova, E.L.; Lilova, M.I.; Savov, V.M. Oxidative stress in children with kidney disease. Pediatr. Nephrol. 2005, 20, 1599–1604. [Google Scholar] [CrossRef]
- Biltagi, M.A.; Maguid, M.A.; Ghafar, M.A.; Farid, E. Correlation of 8-isoprostane, interleukin-6 and cardiac functions with clinical score in childhood obstructive sleep apnoea. Acta Paediatr. 2008, 97, 1397–1405. [Google Scholar] [CrossRef]
- Gozal, D.; Serpero, L.D.; Capdevila, O.S.; Gozal, L.-K. Systemic inflammation in non-obese children with obstructive sleep apnea. Sleep Med. 2008, 9, 254–259. [Google Scholar] [CrossRef]
- Smith, D.F.; Schuler, C.L.; Hossain, M.M.; Huang, G.; McConnell, K.; Urbina, E.M.; Amin, R.S. Early Atherosclerotic inflammatory pathways in children with obstructive sleep apnea. J. Pediatr. 2021, 239, 168–174. [Google Scholar] [CrossRef]
- Loffredo, L.; Zicari, A.M.; Occasi, F.; Perri, L.; Carnevale, R.; Angelico, F.; Del Ben, M.; Martino, F.; Nocella, C.; Savastano, V.; et al. Endothelial dysfunction and oxidative stress in children with sleep disordered breathing: Role of NADPH oxidase. Atherosclerosis 2015, 240, 222–227. [Google Scholar] [CrossRef] [PubMed]
- van Hoorenbeeck, K.; Franckx, H.; Debode, P.; Aerts, P.; Wouters, K.; Ramet, J.; van Gaal, L.F.; Desager, K.N.; De Backer, W.A.; Verhulst, S.L. Weight loss and sleep-disordered breathing in childhood obesity: Effects on inflammation and uric acid. Obesity 2012, 20, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Babar, G.; Clements, M.; Dai, H.; Raghuveer, G. Assessment of biomarkers of inflammation and premature atherosclerosis in adolescents with type-1 diabetes mellitus. J. Pediatr. Endocrinol. Metab. 2019, 32, 109–113. [Google Scholar] [CrossRef]
- Suys, B.; de Beeck, L.O.; Rooman, R.; Kransfeld, S.; Heuten, H.; Goovaerts, I.; Vrints, C.; de Wolf, D.; Matthys, D.; Manuel-Y-Keenoy, B. Impact of oxidative stress on the endothelial dysfunction of children and adolescents with type 1 diabetes mellitus: Protection by superoxide dismutase? Pediatr. Res. 2007, 62, 456–461. [Google Scholar] [CrossRef]
- Sochett, E.; Noone, D.; Grattan, M.; Slorach, C.; Moineddin, R.; Elia, Y.; Mahmud, F.H.; Dunger, D.B.; Dalton, N.; Cherney, D.; et al. Relationship between serum inflammatory markers and vascular function in a cohort of adolescents with type 1 diabetes. Cytokine 2017, 99, 233–239. [Google Scholar] [CrossRef]
- Mylona-Karayanni, C.; Gourgiotis, D.; Bossios, A.; Kamper, E.F. Oxidative stress and adhesion molecules in children with type 1 diabetes mellitus: A possible link. Pediatr. Diabetes 2006, 7, 51–59. [Google Scholar] [CrossRef]
- Stanković, S.M.; Zivić, S.R.; Šaranac, L.; Cvetković, V.; Pešić, M.; Vasić, K.; Stanković, M.; Topalović, A.; Cvetkovic, T. Determinants of atherosclerosis in children and adolescents with diabetes type 1. Endokrynol. Polska 2012, 63, 414–419. [Google Scholar]
- Seckin, D.; Ilhan, N.; Ilhan, N.; Ertugrul, S. Glycaemic control, markers of endothelial cell activation and oxidative stress in children with type 1 diabetes mellitus. Diabetes Res. Clin. Pract. 2006, 73, 191–197. [Google Scholar] [CrossRef]
- Sharma, G.; Muller, D.P.; O’Riordan, S.M.; Bryan, S.; Dattani, M.T.; Hindmarsh, P.C.; Mills, K. Urinary conjugated α-tocopheronolactone—A biomarker of oxidative stress in children with type 1 diabetes. Free Radic. Biol. Med. 2013, 55, 54–62. [Google Scholar] [CrossRef][Green Version]
- Chiesa, S.T.; Charakida, M.; McLoughlin, E.; Nguyen, H.C.; Georgiopoulos, G.; Motran, L.; Elia, Y.; Marcovecchio, M.L.; Dunger, D.B.; Dalton, R.N.; et al. Elevated high-density lipoprotein in adolescents with Type 1 diabetes is associated with endothelial dysfunction in the presence of systemic inflammation. Eur. Heart J. 2019, 40, 3559–3566. [Google Scholar] [CrossRef]
- Yau, P.L.; Ross, N.; Tirsi, A.; Arif, A.; Ozinci, Z.; Convit, A. Insulin sensitivity and inflammation mediate the impact of fitness on cerebrovascular health in adolescents. Child. Obes. 2017, 13, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.K.; Chen, A.K.; Barnard, R.J. Effect of a short-term diet and exercise intervention in youth on atherosclerotic risk factors. Atherosclerosis 2007, 191, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Llorente-Cantarero, F.J.; Aguilar-Gómez, F.J.; Leis, R.; Bueno, G.; Rupérez, A.I.; Anguita-Ruiz, A.; Vázquez-Cobela, R.; Mesa, M.D.; Moreno, L.A.; Gil, A.; et al. Relationship between physical activity, oxidative stress, and total plasma antioxidant capacity in spanish children from the GENOBOX Study. Antioxidants 2021, 10, 320. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.S.; Wetzsteon, R.J.; Kaiser, D.R.; Steinberger, J.; Bank, A.J.; Dengel, D.R. Inflammation, insulin, and endothelial function in overweight children and adolescents: The role of exercise. J. Pediatr. 2004, 145, 731–736. [Google Scholar] [CrossRef] [PubMed]
- Leite-Almeida, L.; Morato, M.; Cosme, D.; Afonso, J.; Areias, J.C.; Guerra, A.; Afonso, A.C.; Albino-Teixeira, A.; Sousa, T.; Correia-Costa, L. Impact of physical activity on redox status and nitric oxide bioavailability in nonoverweight and overweight/obese prepubertal children. Free Radic. Biol. Med. 2021, 163, 116–124. [Google Scholar] [CrossRef]
- Platat, C.; Wagner, A.; Klumpp, T.; Schweitzer, B.; Simon, C. Relationships of physical activity with metabolic syndrome features and low-grade inflammation in adolescents. Diabetologia 2006, 49, 2078–2085. [Google Scholar] [CrossRef]
- Liu, M.; Lin, X.; Wang, X.; Liu, M.; Lin, X.; Wang, X. Decrease in serum chemerin through aerobic exercise plus dieting and its association with mitigation of cardio-metabolic risk in obese female adolescents. J. Pediatr. Endocrinol. Metab. 2018, 31, 127–135. [Google Scholar] [CrossRef]
- Dennis, B.A.; Ergul, A.; Gower, B.A.; Allison, J.D.; Davis, C.L. Oxidative stress and cardiovascular risk in overweight children in an exercise intervention program. Child. Obes. 2013, 9, 15–21. [Google Scholar] [CrossRef]
- Santiprabhob, J.; Limprayoon, K.; Aanpreung, P.; Charoensakdi, R.; Kalpravidh, R.W.; Phonrat, B.; Tungtrongchitr, R. Impact of a group-based treatment program on adipocytokines, oxidative status, inflammatory cytokines and arterial stiffness in obese children and adolescents. J. Pediatr. Endocrinol. Metab. 2018, 31, 733–742. [Google Scholar] [CrossRef]
- Cazeau, R.-M.; Huang, H.; Bauer, J.A.; Hoffman, R.P. Effect of vitamins C and E on endothelial function in type 1 diabetes mellitus. J. Diabetes Res. 2016, 2016, 3271293. [Google Scholar] [CrossRef]
- Goldstein, B.H.; Sandelin, A.M.; Golbus, J.R.; Warnke, N.; Gooding, L.; King, K.K.; Donohue, J.E.; Yu, S.; Gurney, J.G.; Goldberg, C.S.; et al. Impact of vitamin C on endothelial function and exercise capacity in patients with a Fontan circulation. Congenit. Heart Dis. 2012, 7, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, R.P.; Dye, A.S.; Bauer, J.A. Ascorbic acid blocks hyperglycemic impairment of endothelial function in adolescents with type 1 diabetes. Pediatr. Diabetes 2012, 13, 607–610. [Google Scholar] [CrossRef]
- Chiarelli, F.; Santilli, F.; Sabatino, G.; Blasetti, A.; Tumini, S.; Cipollone, F.; Mezzetti, A.; Verrotti, A. Effects of vitamin E supplementation on intracellular antioxidant enzyme production in adolescents with type 1 diabetes and early microangiopathy. Pediatr. Res. 2004, 56, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Németh, I.; Túri, S.; Haszon, I.; Bereczki, C. Vitamin E alleviates the oxidative stress of erythropoietin in uremic children on hemodialysis. Pediatr. Nephrol. 2000, 14, 13–17. [Google Scholar] [CrossRef]
- Kelishadi, R.; Hashemipour, M.; Adeli, K.; Tavakoli, N.; Movahedian-Attar, A.; Shapouri, J.; Poursafa, P.; Rouzbahani, A. Effect of zinc supplementation on markers of insulin resistance, oxidative stress, and inflammation among prepubescent children with metabolic syndrome. Metab. Syndr. Relat. Disord. 2010, 8, 505–510. [Google Scholar] [CrossRef]
- Soongswang, J.; Sangtawesin, C.; Durongpisitkul, K.; Laohaprasitiporn, D.; Nana, A.; Punlee, K.; Kangkagate, C. The effect of coenzyme Q10 on idiopathic chronic dilated cardiomyopathy in children. Pediatr. Cardiol. 2005, 26, 361–366. [Google Scholar] [CrossRef]
- Serag, H.; El Wakeel, L.; Adly, A. Coenzyme Q10 administration has no effect on sICAM-1 and metabolic parameters of pediatrics with type 1 diabetes mellitus. Int. J. Vitam. Nutr. Res. 2021, 91, 315–324. [Google Scholar] [CrossRef]
- Bennett-Richards, K.J.; Kattenhorn, M.; Donald, A.E.; Oakley, G.R.; Varghese, Z.; Bruckdorfer, K.R.; Deanfield, J.E.; Rees, L. Oral L-arginine does not improve endothelial dysfunction in children with chronic renal failure. Kidney Int. 2002, 62, 1372–1378. [Google Scholar] [CrossRef][Green Version]
- Lim, D.S.; Mooradian, S.J.; Goldberg, C.S.; Gomez, C.; Crowley, D.C.; Rocchini, A.P.; Charpie, J.R. Effect of oral l-arginine on oxidant stress, endothelial dysfunction, and systemic arterial pressure in young cardiac transplant recipients. Am. J. Cardiol. 2004, 94, 828–831. [Google Scholar] [CrossRef]
- Bo, Y.; Yuan, L.-P.; Zhang, J.-J.; Meng, D.-D.; Jing, H.; Dai, H.-J. Total flavonoids of Bidens bipinnata L. a traditional Chinese medicine inhibits the production of inflammatory cytokines of vessel endothelial cells stimulated by sera from Henoch–Schönlein purpura patients. J. Pharm. Pharmacol. 2012, 64, 882–887. [Google Scholar] [CrossRef]
- Hashemi, M.; Kelishadi, R.; Hashemipour, M.; Zakerameli, A.; Khavarian, N.; Ghatrehsamani, S.; Poursafa, P. Acute and long-term effects of grape and pomegranate juice consumption on vascular reactivity in paediatric metabolic syndrome. Cardiol. Young- 2010, 20, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, A.; Shahnaz, T.; Sorayya, K. Effect of 8 weeks’ supplementation grape seed extract on insulin resistance in iranian adolescents with metabolic syndrome: A randomized controlled trial. Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Sukardi, R.; Sastroasmoro, S.; Siregar, N.C.; Djer, M.M.; Suyatna, F.D.; Sadikin, M.; Ibrahim, N.; Rahayuningsih, S.E.; Witarto, A.B. The role of curcumin as an inhibitor of oxidative stress caused by ischaemia re-perfusion injury in tetralogy of Fallot patients undergoing corrective surgery. Cardiol. Young 2016, 26, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Elbarbary, N.S.; Ismail, E.A.R.; El-Naggar, A.R.; Hamouda, M.H.; El-Hamamsy, M. The effect of 12 weeks carnosine supplementation on renal functional integrity and oxidative stress in pediatric patients with diabetic nephropathy: A randomized placebo-controlled trial. Pediatr. Diabetes 2018, 19, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Gu, J.; Ansley, D.M.; Xia, F.; Yu, J. Antioxidant therapy with Salvia miltiorrhiza decreases plasma endothelin-1 and thromboxane B2 after cardiopulmonary bypass in patients with congenital heart disease. J. Thorac. Cardiovasc. Surg. 2003, 126, 1404–1410. [Google Scholar] [CrossRef]
- Cherukuri, L.; Birudaraju, D.; Kinninger, A.; Chaganti, B.T.; Shekar, C.; Hamal, S.; Shaikh, K.; Flores, F.; Roy, S.K.; Sotka, W.; et al. Effect of a plant-based bioequivalent inorganic nitrate (NO3−) complex with vitamins, antioxidants and phytophenol rich food extracts in hypertensive individuals-A randomized, double-blind, placebo-controlled study. Clin. Nutr. ESPEN 2020, 40, 327–335. [Google Scholar] [CrossRef]
- Hegazy, S.K.; Tolba, O.A.; Mostafa, T.M.; Eid, M.A.; El-Afify, D.R. Alpha-Lipoic Acid improves subclinical left ventricular dysfunction in asymptomatic patients with type 1 diabetes. Rev. Diabet. Stud. 2013, 10, 58–67. [Google Scholar] [CrossRef]
- Scaramuzza, A.; Giani, E.; Redaelli, F.; Ungheri, S.; MacEdoni, M.; Giudici, V.; Bosetti, A.; Ferrari, M.; Zuccotti, G.V. Alpha-lipoic acid and antioxidant diet help to improve endothelial dysfunction in adolescents with type 1 diabetes: A pilot trial. J. Diabetes Res. 2015, 2015, 474561. [Google Scholar] [CrossRef]
- Rimoldi, S.F.; Sartori, C.; Rexhaj, E.; Bailey, D.M.; De Marchi, S.F.; McEneny, J.; Von Arx, R.; Cerny, D.; Duplain, H.; Germond, M.; et al. Antioxidants improve vascular function in children conceived by assisted reproductive technologies: A randomized double-blind placebo-controlled trial. Eur. J. Prev. Cardiol. 2015, 22, 1399–1407. [Google Scholar] [CrossRef]
- Roszkowska, R.; Taranta-Janusz, K.; Tenderenda-Banasiuk, E.; Wasilewska, A. Increased circulating inflammatory markers may indicate that formula-fed children are at risk of atherosclerosis. Acta Paediatr. 2014, 103, e354–e358. [Google Scholar] [CrossRef]
- Bernini, L.J.; Simão, A.N.C.; Alfieri, D.F.; Lozovoy, M.A.B.; Mari, N.L.; de Souza, C.H.B.; Dichi, I.; Costa, G.N. Beneficial effects of Bifidobacterium lactis on lipid profile and cytokines in patients with metabolic syndrome: A randomized trial. Effects of probiotics on metabolic syndrome. Nutrition 2016, 32, 716–719. [Google Scholar] [CrossRef] [PubMed]
- Gøbel, R.J.; Larsen, N.; Jakobsen, M.; Mølgaard, C.; Michaelsen, K.F. Probiotics to Adolescents with obesity: Effects on inflammation and metabolic syndrome. J. Pediatr. Gastroenterol. Nutr. 2012, 55, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Bresolin, A.C.; Pronsatti, M.M.; Pasqualotto, L.N.; Nassar, P.O.; Jorge, A.S.; da Silva, E.A.; Nassar, C.A. Lipid profiles and inflammatory markers after periodontal treatment in children with congenital heart disease and at risk for atherosclerosis. Vasc. Health Risk Manag. 2013, 9, 703–709. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zeigler, C.C.; Wondimu, B.; Marcus, C.; Modéer, T. Pathological periodontal pockets are associated with raised diastolic blood pressure in obese adolescents. BMC Oral Health 2015, 15, 41. [Google Scholar] [CrossRef]
- Parisi, F.; Milazzo, R.; Savasi, V.M.; Cetin, I. Maternal low-grade chronic inflammation and intrauterine programming of health and disease. Int. J. Mol. Sci. 2021, 22, 1732. [Google Scholar] [CrossRef]
- Skilton, M.R. Intrauterine risk factors for precocious atherosclerosis. Pediatrics 2008, 121, 570–574. [Google Scholar] [CrossRef]
- Leduc, L.; Levy, E.; Bouity-Voubou, M.; Delvin, E. Fetal programming of atherosclerosis: Possible role of the mitochondria. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 149, 127–130. [Google Scholar] [CrossRef]
- Silvestro, S.; Calcaterra, V.; Pelizzo, G.; Bramanti, P.; Mazzon, E. Prenatal hypoxia and placental oxidative stress: Insights from animal models to clinical evidences. Antioxidants 2020, 9, 414. [Google Scholar] [CrossRef]
- Sutherland, M.R.; Bertagnolli, M.; Lukaszewski, M.-A.; Huyard, F.; Yzydorczyk, C.; Luu, T.M.; Nuyt, A.M. Preterm birth and hypertension risk: The oxidative stress paradigm. Hypertension 2014, 63, 12–18. [Google Scholar] [CrossRef]
- Wang, H.-H. Oxidative stress and potential renal damage in neonates. Pediatr. Neonatol. 2015, 56, 209–210. [Google Scholar] [CrossRef]
- Yzydorczyk, C.; Armengaud, J.B.; Peyter, A.-C.; Chehade, H.; Cachat, F.; Juvet, C.; Siddeek, B.; Simoncini, S.; Sabatier, F.; Dignat-George, F.; et al. Endothelial dysfunction in individuals born after fetal growth restriction: Cardiovascular and renal consequences and preventive approaches. J. Dev. Orig. Health Dis. 2017, 8, 448–464. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-N.; Tain, Y.-L. Early Origins of hypertension: Should prevention start before birth using natural antioxidants? Antioxidants 2020, 9, 1034. [Google Scholar] [CrossRef] [PubMed]
- Radomski, M.W.; Salas, E. Nitric oxide-biological mediator, modulator and factor of injury: Its role in the pathogenesis of atherosclerosis. Atherosclerosis 1995, 118, S69–S80. [Google Scholar] [CrossRef]
- Sabri, M.; Gheissari, A.; Mansourian, M.; Mohammadifard, N.; Sarrafzadegan, N. Essential hypertension in children, a growing worldwide problem. J. Res. Med. Sci. 2019, 24, 109. [Google Scholar] [CrossRef] [PubMed]
- Wirix, A.J.; Kaspers, P.J.; Nauta, J.; Chinapaw, M.J.M.; Kirst-van Holthe, J.E. Pathophysiology of hypertension in obese children: A systematic review. Obes. Rev. 2015, 16, 831–842. [Google Scholar] [CrossRef] [PubMed]
- Filgueiras, M.S.; Rocha, N.P.; Novaes, J.F.; Bressan, J. Vitamin D status, oxidative stress, and inflammation in children and adolescents: A systematic review. Crit. Rev. Food Sci. Nutr. 2020, 60, 660–669. [Google Scholar] [CrossRef]
- Vincent, H.K.; Innes, K.E.; Vincent, K.R. Oxidative stress and potential interventions to reduce oxidative stress in overweight and obesity. Diabetes, Obes. Metab. 2007, 9, 813–839. [Google Scholar] [CrossRef]
- Espinola-Klein, C.; Gori, T.; Blankenberg, S.; Munzel, T. Inflammatory markers and cardiovascular risk in the metabolic syndrome. Front. Biosci. 2011, 16, 1663–1674. [Google Scholar] [CrossRef]
- Balat, A. Kidney is in trouble with mediators. Bosn. J. Basic Med. Sci. 2010, 10 (Suppl. 1), S29–S36. [Google Scholar] [CrossRef]
- Massy, Z.A.; Mazière, C.; Kamel, S.; Brazier, M.; Choukroun, G.; Tribouilloy, C.; Slama, M.; Andrejak, M. Impact of inflammation and oxidative stress on vascular calcifications in chronic kidney disease. Pediatr. Nephrol. 2005, 20, 380–382. [Google Scholar] [CrossRef]
- Avloniti, A.; Chatzinikolaou, A.; Deli, C.K.; Vlachopoulos, D.; Gracia-Marco, L.; Leontsini, D.; Draganidis, D.; Jamurtas, A.Z.; Mastorakos, G.; Fatouros, I.G. Exercise-induced oxidative stress responses in the pediatric population. Antioxidants 2017, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Montero, D.; Walther, G.; Perez-Martin, A.; Roche, E.; Vinet, A. Endothelial dysfunction, inflammation, and oxidative stress in obese children and adolescents: Markers and effect of lifestyle intervention. Obes. Rev. 2012, 13, 441–455. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Tain, Y.-L.; Sheen, J.-M.; Huang, L.-T. Melatonin utility in neonates and children. J. Formos. Med. Assoc. 2012, 111, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Gombert, M.; Codoñer-Franch, P. Melatonin in early nutrition: Long-term effects on cardiovascular system. Int. J. Mol. Sci. 2021, 22, 6809. [Google Scholar] [CrossRef]
- Page, M.J.; Mckenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaf, J.L.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Koide, S.-I.; Kugiyama, K.; Sugiyama, S.; Nakamura, S.-I.; Fukushima, H.; Honda, O.; Yoshimura, M.; Ogawa, H. Association of polymorphism in glutamate-cysteine ligase catalytic subunit gene with coronary vasomotor dysfunction and myocardial infarction. J. Am. Coll. Cardiol. 2003, 41, 539–545. [Google Scholar] [CrossRef]
- Kuller, L.H. Ethnic differences in atherosclerosis, cardiovascular disease and lipid metabolism. Curr. Opin. Lipidol. 2004, 15, 109–113. [Google Scholar] [CrossRef]
- Marseglia, L.; Manti, S.; D’Angelo, G.; Nicotera, A.; Parisi, E.; Di Rosa, G.; Gitto, E.; Arrigo, T. Oxidative stress in obesity: A critical component in human diseases. Int. J. Mol. Sci. 2014, 16, 378–400. [Google Scholar] [CrossRef]
- Orlando, A.; Nava, E.; Giussani, M.; Genovesi, S. Adiponectin and cardiovascular risk. From Pathophysiology to clinic: Focus on children and adolescents. Int. J. Mol. Sci. 2019, 20, 3228. [Google Scholar] [CrossRef]
- Choi, H.M.; Doss, H.M.; Kim, K.S. Multifaceted physiological roles of adiponectin in inflammation and diseases. Int. J. Mol. Sci. 2020, 21, 1219. [Google Scholar] [CrossRef]
- O’Gorman, C.S.; O’Neill, M.B.; Conwell, L.S. Considering statins for cholesterol-reduction in children if lifestyle and diet changes do not improve their health: A review of the risks and benefits. Vasc. Health Risk Manag. 2010, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chuang, J.; Zeller, M.H.; Inge, T.; Crimmins, N. Bariatric surgery for severe obesity in two adolescents with type 1 diabetes. Pediatrics 2013, 132, e1031–e1034. [Google Scholar] [CrossRef]
- Headid, R.J., III; Park, S.-Y. The impacts of exercise on pediatric obesity. Clin. Exp. Pediatr. 2021, 64, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Wärnberg, J.; Gómez_Martínez, S.; Romeo, J.; Díaz, L.-E.; Marcos, A. Nutrition, inflammation, and cognitive function. Ann. N. Y. Acad. Sci. 2009, 1153, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Greco, T.; Glenn, T.C.; Hovda, D.A.; Prins, M.L. Ketogenic diet decreases oxidative stress and improves mitochondrial respiratory complex activity. J. Cereb. Blood Flow Metab. 2016, 36, 1603–1613. [Google Scholar] [CrossRef] [PubMed]
- Orgeron, M.L.; Stone, K.P.; Wanders, D.; Cortez, C.C.; van, N.T.; Gettys, T.W. The impact of dietary methionine restriction on biomarkers of metabolic health. Prog. Mol. Biol. Transl. Sci. 2014, 121, 351–376. [Google Scholar] [CrossRef] [PubMed]
- Puchau, B.; Ochoa, M.C.; Zulet, M.A.; Marti, A.; Martínez, J.A.; Members, G. Dietary total antioxidant capacity and obesity in children and adolescents. Int. J. Food Sci. Nutr. 2010, 61, 713–721. [Google Scholar] [CrossRef]
- Aly, G.S.; Shaalan, A.H.; Mattar, M.K.; Ahmed, H.H.; Zaki, M.E.; Abdallah, H.R. Oxidative stress status in nutritionally stunted children. Egypt. Pediatr. Assoc. Gaz. 2014, 62, 28–33. [Google Scholar] [CrossRef]
- Berkner, K.L.; Runge, K.W. The physiology of vitamin K nutriture and vitamin K-dependent protein function in atherosclerosis. J. Thromb. Haemost. 2004, 2, 2118–2132. [Google Scholar] [CrossRef]
- Močnik, M.; Varda, N.M. Henoch–Schönlein purpura: The management and treatment of paediatric patients in north–east Slovenia. Acta Medico-Biotech. 2021, 14, 11–19. [Google Scholar]
- Kestilä, P.; Magnussen, C.G.; Viikari, J.S.; Kähönen, M.; Hutri-Kähönen, N.; Taittonen, L.; Jula, A.; Loo, B.-M.; Pietikäinen, M.; Jokinen, E.; et al. Socioeconomic status, cardiovascular risk factors, and subclinical atherosclerosis in young adults: The cardiovascular risk in Young Finns Study. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Packard, C.J.; Bezlyak, V.; McLean, J.S.; Batty, G.D.; Ford, I.; Burns, H.; Cavanagh, J.; Deans, K.A.; Henderson, M.; McGinty, A.; et al. Early life socioeconomic adversity is associated in adult life with chronic inflammation, carotid atherosclerosis, poorer lung function and decreased cognitive performance: A cross-sectional, population-based study. BMC Public Health 2011, 11, 42. [Google Scholar] [CrossRef] [PubMed]
- Senoner, T.; Dichtl, W. Oxidative stress in cardiovascular diseases: Still a therapeutic target? Nutrients 2019, 11, 2090. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Kang, P.M. Oxidative stress and antioxidant treatments in cardiovascular diseases. Antioxidants 2020, 9, 1292. [Google Scholar] [CrossRef] [PubMed]
- Valaei, K.; Taherkhani, S.; Arazi, H.; Suzuki, K. Cardiac Oxidative Stress and the Therapeutic approaches to the intake of antioxidant supplements and physical activity. Nutrients 2021, 13, 3483. [Google Scholar] [CrossRef] [PubMed]
- Tomasian, D.; Keaney, J.F.; Vita, J.A. Antioxidants and the bioactivity of endothelium-derived nitric oxide. Cardiovasc. Res. 2000, 47, 426–435. [Google Scholar] [CrossRef]
- Engler, M.M.; Engler, M.B.; Malloy, M.J.; Chiu, E.Y.; Schloetter, M.C.; Paul, S.M.; Stuehlinger, M.; Lin, K.Y.; Cooke, J.P.; Morrow, J.D.; et al. Antioxidant vitamins C and E improve endothelial function in children with hyperlipidemia: Endothelial Assessment of Risk from Lipids in Youth (EARLY) Trial. Circulation 2003, 108, 1059–1063. [Google Scholar] [CrossRef]
| Author, Year | Country | Population | Comparison | Outcomes |
|---|---|---|---|---|
| Original Research | ||||
| Hashemi, 2021 [3] | Iran | 108 children (51 ♂), aged 6 to 18 years | Urinary phthalate metabolites, cardiometabolic risk factors and oxidative stress markers | Exposure to phthalates correlates with cardiometabolic risk and oxidative stress markers (superoxide dismutase, malondialdehyde). |
| Eikendal, 2015 [4] | Netherlands | 139 healthy children (59 ♂), 8 years old | Circulating chemokines and vascular characteristics | The chemokine RANTES contributes to pre-atherosclerotic inflammatory vascular changes in youth |
| Kelishadi, 2007 [5] | Iran and Canada | 512 children (254 ♂), aged 10 to 18 years | C-reactive protein, oxidative stress markers and atherosclerotic risk factors | Oxidative stress and CRP may interact in the early inflammatory process of atherosclerosis |
| Suano de Souza, 2013 [7] | Brazil | 35 (15 ♂) children with elevated and 29 (14 ♂) with normal homocysteine levels, mean age 8.6 years | Homocysteine, oxidative stress, carotid IMT and endothelial reactivity | No differences observed in vascular parameters and homocysteine levels in prepubertal children |
| Wilders-Truschnig, 2008 [8] | Austria and Luxemburg | 30 obese (mean age 12.8 years) and 30 normal weight children (mean age 14.4 years) | IgG antibodies against food antigens, CRP and carotid IMT | Obese children have higher IgG antibody values against food antigens, associated with systemic inflammation and carotid IMT |
| Mendez-Cruz, 2007 [12] | Mexico | Human umbilical vein endothelial cells (HUVEC) of 11 mothers with strong family history of myocardial infarction | Expression of CD40 and CD40 ligand, CD80, CXCL8, tissue factor, and mono-/lymphocyte adhesion to stimulated HUVEC | HUVEC from newborns with a strong family history of MI show basal proinflammatory state and respond to proatherogenic stimuli |
| Schoeps, 2019 [13] | Brazil | 44 (19 ♂) preterm children and 30 (13 ♂) term children, aged 5 to 9 years | Evaluation of CVD risk markers in children born term and preterm | Prepuberal preterm children show high myeloperoxidase concentrations, associated with inflammation and oxidative stress |
| Sakka, 2010 [14] | Greece | 106 children born via IVF (48 ♂) and 68 matched controls (33 ♂), aged 4 to 14 years | IVF and insulin resistance, systemic inflammation, MetS | Children born via IVF display increased BP, but not insulin resistance or low-grade systemic inflammation |
| Kelishadi, 2009 [15] | Iran | 112 children of parents with premature coronary heart disease, aged 12 to 18 years and 127 matched controls | Family history of premature coronary heart disease and inflammation, oxidation, echocardiography | Clusters of inflammatory factors and markers of oxidation, carotid IMT and left ventricular mass associate with family history of coronary heart disease |
| de Giorgis, 2009 [16] | Italy | 24 prepubertal children with positive family history of premature CVD (10 ♂) and 25 healthy prepubertal controls (11 ♂) | Carotid IMT in children of family history of premature CVD and relationship to insulin resistance, oxidant status, lipid profile | Signs of precocious CVD risk are detectable in children with family history of premature CVD already during prepuberty |
| Craig, 2018 [17] | South Africa | 55 boys with maternal and/or lifestyle risk for CVD, aged 6 to 8 years and 26 boys without maternal risk | Associations between oxidative stress and vascular functions in boys, stratified by maternal risk | Oxidative stress is and early mediator of vascular changes in the studied population |
| Gonzalez-Enriquez, 2008 [18] | Mexico | 62 obese (34 ♂) and 21 lean controls (12 ♂), aged 6 to 19 years | Relationship between carotid IMT and functional polymorphic variants in genes for chemokines and proinflammatory cytokines associated with CVD risk | Genetic markers of an increased inflammatory response are already present in obese children and adolescents |
| Coelho, 2006 [19] | Portugal | 49 healthy adolescents | Genetic polymorphisms, somatic characteristics, blood pressure, biochemical markers of oxidative stress and CVD risk | Carriers of angiotensin-converting enzyme DD and haptoglobin 2-2 genotypes have a higher pro-oxidant status |
| Guerra, 2000 [20] | Portugal | 51 children, aged 9 to 12 years | Deleterious effect of reactive oxygen species on lipids, arterial endothelium and CVD | Polymorphic genetic variants of low molecular acid phosphatase and protein phosphotyrosine phosphatase are associated with oxidative stress indicators |
| Oztezcan, 2002 [21] | Turkey | NA | Lipid peroxidation and antioxidant system in children of hypertensive and normotensive parents | Serum malondialdehyde levels are increased in children of hypertensive parents |
| Hapyn, 2000 [22] | Poland | 56 children with family history of hypercholesterolemia or early atherosclerosis and 20 children without positive family history | Intensity of lipid peroxidation and the activity of antioxidant enzymes in children with risk of early atherosclerosis | Children with positive family history of hypercholesterolemia and early atherosclerosis may demonstrate intensive lipid peroxidation |
| Kolesnikova, 2014 [23] | Russia | 185 adolescents with essential hypertension (125 ♂) and 60 control subjects (36 ♂), aged 14 to 17 years | Emotional status, BP and lipid peroxidation-antioxidant protection system in adolescents | Opposite correlations between the emotional status parameters and components of lipid peroxidation–antioxidant protection system exist |
| Ostalska-Nowicka, 2021 [24] | Poland | 65 children with essential hypertension (32 ♂) and 44 normotensive controls (20 ♂), aged 6 to 18 years | Dental caries-related primary hypertension in children and adolescents | Tooth decay in children and adolescents may be a trigger factor of essential hypertension |
| Sladowska-Kozlowska, 2015 [25] | Poland | 126 children with arterial hypertension (95 ♂) and 83 healthy controls (40 ♂), aged 5 to 18 years | Endothelial nitric oxide (eNOS) gene polymorphism G894T and 24-h ambulatory blood pressure, carotid IMT, left ventricular mass, oxidative stress and urinary albumin secretion | The eNOS T allele is not more prevalent among hypertensive children than among healthy ones, but it is associated with early vascular damage in children with primary hypertension |
| Barath, 2006 [26] | Hungary | 49 adolescents with essential hypertension (35 ♂, mean age 14.0 years), 79 with obesity-induced hypertension (56 ♂, mean age 14.0 years), 20 uremic patients (11 ♂ mean age 14.9 years), 60 obese patients (33 ♂, mean age 13.2 years and 57 age-matched controls (33 ♂, mean age 13.7 years) | Roles of oxidative stress and paraoxonase 1 in uremic, essential or obesity-induced hypertension | No significant correlation between the biochemical parameters and neither genotypes nor paraoxonase activities |
| Litwin, 2010 [27] | Poland | 44 children with untreated primary hypertension (36 ♂, mean age 13.7 years) and 30 healthy controls (13 ♂, mean age 12.7 years) | Inflammatory activation in children with primary hypertension | Lack of correlation between CRP and chemokines suggests that vascular inflammation in primary hypertension precedes the systemic inflammatory changes |
| Turi, 2003 [28] | Hungary, USA | 52 children with essential hypertension (37 ♂, mean age 14.4 years) and 48 controls (27 ♂, mean age 14.3 years) | Plasma nitrites and nitrates, lipid peroxidation end-products and redox status of red blood cell glutathione in juvenile essential hypertension | Presence of systemic oxidative stress in hypertensive children and adolescents, irrespective of their body mass index |
| Sladowska-Kozlowska, 2012 [29] | Poland | 86 children with primary hypertension (66 ♂), aged 5 to 17 years | Primary hypertension, target organ damage and oxidative stress (reduced glutathione, glutathione peroxidase activity, thiobarbituric acid reactive substances, asymmetric dimethyloarginine and oxLDL) before and after 12 months of (non)pharmacological therapy | Oxidative stress in children with primary hypertension correlates with target organ damage, metabolic abnormalities, changes in fat amount and improvement of insulin sensitivity, but not with BP decrease |
| Yamano, 2015 [30] | Japan | 85 children (45 ♂), mean age 6.9 years | Arteriosclerosis index and oxidative stress markers in school children | Urinary 8-isoprostaglandin F2α may serve as early marker in predicting risk in children of developing lifestyle-related diseases |
| Cakici, 2018 [31] | Turkey | 30 children with primary hypertension (19 ♂, mean age 14.8 years) and 30 healthy controls (17 ♂, mean age 14.3 years) | Level of dynamic thiol/disulphide homeostasis in adolescents with newly diagnosed primary hypertension | Shift towards disulphide formation in adolescent patients with primary hypertension |
| Stoppa-Vaucher, 2012 [32] | Switzerland | 32 obese children (19 ♂, mean age 11.7 years) and 29 lean controls (19 ♂, mean age 11.3 years) | Inflammatory and prothrombotic states in obese children of European descent | Obese children present with inflammatory and prothrombotic states as early as 6 years of age |
| Valle Jimenez, 2007 [33] | Spain | 46 obese prepubertal children (19 ♂) and 46 lean controls (19 ♂), aged 6 to 9 years | Relationship between endothelial dysfunction and both insulin resistance and inflammation in prepubertal obese children | An association between endothelial dysfunction, insulin resistance, inflammation and inappropriate fibrinolysis was established in the children studied |
| Mauras, 2010 [34] | USA | 115 obese (59 ♂) and 88 lean children (47 ♂), aged 7 to 18 years | Markers of inflammation and prothrombosis in obese children without established metabolic syndrome comorbidities | Childhood obesity is associated with a proinflammatory and prothrombotic state before other comorbidities of the MetS are present and even before the onset of puberty |
| Morandi, 2020 [35] | Italy | 152 obese children and adolescents (79 ♂, mean age 11.9 years) | Association between total antioxidant capacity and BP | The systemic anti-oxidant capacity is inversely associated with systolic BP and pulse pressure in children and adolescents with obesity |
| Ovunc Hacihamdioglu, 2015 [36] | Turkey | 24 hypertensive obese children (16 ♂, mean age 13.1 years), 22 normotensive obese (14 ♂, mean age 11.5 years) and 27 healthy children (13 ♂, mean age 11.2 years) | Effect of obesity and anti-hypertensive treatment on urinary Th1 chemokines | Th1 cells could be activated in obese hypertensive children before the onset of clinical indicators of target organ damage |
| Syrenicz, 2006 [37] | Poland | 281 obese children (151 ♂), aged 6 to 18 years | Role of low-grade, systemic inflammation and endothelial activation in the modulation of BP independently of other traditional risk factors | Low-grade inflammation may play a role in the modulation of arterial BP relatively early in life |
| Aburawi, 2019 [38] | United Arab Emirates | 967 thin, normal, overweight and obese children, aged 7 to 16 years | Biomarkers for glycemic control, lipid metabolism, systemic inflammation, endothelial dysfunction and hepatic cholestasis | Children with excess fat had increased risks for developing systemic inflammation, dyslipidemia, endothelial dysfunction, cholestasis and diabetes |
| Al-Shorman, 2017 [39] | Jordan | 29 lean (17 ♂), 29 obese (17 ♂) and 29 severely obese children (17 ♂), aged 10 to 15 years | Levels of carotid IMT, endothelial dysfunction and inflammatory biomarkers | Biomarkers of inflammation and endothelial dysfunction were higher in obese schoolchildren, but are not increased by the degree of obesity nor the MetS cluster |
| Giannini, 2008 [40] | Italy | 53 obese pre-pubertal children (27 ♂, mean age 8 years) and 41 healthy pre-pubertal controls (21 ♂, mean age 7 years) | Relationship between carotid IMT, insulin resistance and oxidant status | Early changes in glucose metabolism and an alteration of oxidant–antioxidant status may be present in obese pre-pubertal children, which could lead to increased carotid IMT and early CVD |
| Sinaiko, 2005 [41] | USA, Sweden, Norway | Cohort of 295 adolescents (169 ♂, mean age 15 years) | Relation of fatness and insulin resistance and their interaction with CVD risk factors, inflammatory factors and oxidative stress | Insulin resistance may be acting interactively with fatness |
| Jung, 2009 [42] | Sweden | 79 ♂ adolescents, aged 13 to 17 years | Anthropometric measures, adiponectin, stromal-derived factor (SDF-1) and soluble E-selectin as parameters for beginning of insulin resistance and endothelial damage | SDF-1 might be a new marker for diagnosis of obesity-related diseases and help understand pathophysiologic mechanisms |
| Codoner-Franch, 2012 [43] | Spain | 54 severely obese (33 ♂) and 44 healthy children (25 ♂), aged 7 to 14 years | Plasma level of advanced oxidation protein products (mAOPPs) and relation to metabolic risk factors | Determination of plasma mAOPPs levels is an easy method that can evaluate protein oxidation and provide information related to metabolic risk and treatment effectiveness |
| Okur, 2013 [44] | Turkey | 27 prepubertal obese children (59% ♂, mean age 7 years,) and 30 healthy children (55% ♂, mean age 7 years) | Circulating oxidized low-density lipoprotein (LDL) concentrations and the carotid IMT and possible association with carotid atherosclerosis | Oxidation of LDL starts early in obese children, but the carotid IMT is not significantly affected |
| Elmas, 2017 [45] | Turkey | 65 children with exogenous obesity (34 ♂) and 64 healthy children (27 ♂), aged 5 to 17 years | Thiol/disulphide homeostasis as a novel and sensitive marker of oxidative stress and its relationship with some inflammatory and cardiovascular markers | The impairment in thiol/disulphide homeostasis, which is indicative of oxidative stress, is associated with inflammation in obesity |
| Correia-Costa, 2016 [46] | Portugal, Germany | 89 overweight (43 ♂), 61 obese (40 ♂) and 163 lean children (83 ♂), aged 8 to 9 years | Oxidative stress and NO production/ metabolism and correlations with cardiometabolic risk and renal function | Oxidant status and NO are increased in relation to fat accumulation and translate into higher values of cardiometabolic risk markers and affect renal function |
| Codoner-Franch, 2011 [47] | Spain | 60 obese and 42 lean children, aged 7 to 14 years | Childhood obesity and modification of blood polyamines, circulating markers of oxidative and nitrosative stress and endothelial dysfunction | Polyamine levels are increased in childhood obesity and correlate to markers of oxidative/nitrosative stress and angiogenesis |
| Landgraf, 2012 [48] | Germany | 69 lean and 105 obese children, aged 7 to 18 years | Association of chemerin with obesity and early-onset metabolic and vascular sequelae | There is an association of chemerin with obesity and inflammatory and endothelial activation markers |
| Niklowitz, 2018 [49] | Germany | 88 overweight children (39 ♂, mean age 11.9 years) and 23 lean children (10 ♂, mean age 11 years) | Relationships between chemerin, parameters of fat mass and MetS in obese children before and after weight reduction | Chemerin is related to parameters of central fat mass and MetS |
| Wojcik, 2020 [50] | Poland | 23 obese children (10 ♂, mean age 9 years) | Circulating chemerin level and 24 h blood pressure monitoring results and IMT | Elevated chemerin level may be associated with increased systolic BP in obese children |
| Simunovic, 2019 [51] | Croatia | 92 obese children (52 ♂) and 39 healthy, lean controls (18 ♂), aged 10 to 18 years | Catestatin in pediatric obesity, metabolic syndrome and correlations between catestatin and CVD risk | Serum catestatin concentrations are decreased in obese children and adolescents |
| Makni, 2013 [52] | France | 60 obese children (24 ♂), 54 obese children with MetS (33 ♂) and 37 lean controls (19 ♂), mean age 13 years | Correlation of resistin with inflammatory and cardiometabolic markers in obese adolescents with and without MetS | Plasma resistin showed higher correlations with anthropometric parameters, lipid profiles, systolic and diastolic BP and pro-inflammatory cytokines in obese children with MetS |
| Stringer, 2009 [53] | Canada | 24 children with T2DM, 19 obese children and 34 lean controls, aged 12 to 15 years | CVD risk in children with and without T2DM or obesity by comparing pro- and anti-inflammatory adipokines, markers of oxidative stress and the plasma phospholipid fatty acid profile | Altered plasma adipokines and markers of oxidative stress suggest increased risk of CVD in youth with obesity or T2DM |
| Stelzer, 2012 [54] | Austria, USA | 256 overweight and obese children (114 ♂), aged 8 to 18 years and 67 lean controls | Adipocytokines, interleukin-6 and leptin in overweight/obese and normal-weight subjects | Interleukin-6 is increased with the grade of overweight in every age group. Leptin is essentially involved in the early priming phase of obesity-related inflammation |
| Mohanraj, 2013 [55] | Korea, USA | 41 overweight (19 ♂), 56 obese children (25 ♂) and 100 lean controls (49 ♂), mean age 12 years | Comparison of obese adolescents to normal counterparts for total insulin-like growth factor-binding protein 3 (IGFBP-3) levels and proteolyzed IGFBP-3 in circulation | IGFBP-3 inhibits cytokine-induced insulin resistance and early manifestations of atherosclerosis |
| Ostrow, 2011 [56] | USA | 42 obese children (25 ♂, mean age 12.8 years) and 34 non-obese children (22 ♂, mean age 11.8 years) | Oxidative stress in obese vs. non-obese children with correlation to adiposity, obesity-related metabolic abnormalities and ambulatory BP | There is a correlation between oxidative stress, adiposity and BP in children |
| Selvaraju, 2019 [57] | USA | 24 overweight (14 ♂), 23 obese (9 ♂) and 41 normal weight children (23 ♂), mean age 8 years | Urinary biomarkers of inflammation (CRP, interleukin-6, and α-1-acid glycoprotein), and oxidative stress markers (8-isoprostane, 8-hydroxy-2′-deoxiguanosine and endothelin-1) | Urinary biomarkers of inflammation and oxidative stress are elevated in obese children and correlate with a marker of endothelial dysfunction |
| Metzig, 2011 [58] | USA | 34 obese children (17 ♂, mean age 12.4 years) | Endothelial function, glucose, insulin, CRP, interleukin-6, circulating oxidized low-density lipoprotein and myeloperoxidase in a fasting state and at 1- and 2-h following glucose ingestion | An acute oral glucose load does not reduce endothelial function or increase levels of inflammation or oxidative stress in obese youth |
| Codoner-Franch, 2012 [59] | Spain | 66 obese children (41 ♂) and 39 normal weight children (19 ♂), aged 8 to 13 years | Assessment of CVD risk in severely obese children | Insufficient 25(OH)D levels were detected in severely obese children with increased markers of oxidative/nitrosative stress, inflammation and endothelial activation. |
| Rajakumar, 2020 [60] | USA | 225 overweight or obese children, aged 10 to 18 years | Effect of vitamin D3 supplementation on vascular and metabolic health of vitamin D–deficient | Correction of vitamin D deficiency in overweight and obese children did not affect measures of arterial endothelial function or stiffness, systemic inflammation, or lipid profile, but lowered BP and fasting glucose concentration improved insulin sensitivity |
| Al-Daghri, 2016 [61] | Saudi Arabia | 224 children and 140 adults | Association of vitamin B12 with pro-inflammatory cytokines and biochemical markers related to cardiometabolic risk | Maintaining adequate vitamin B12 concentrations may lower inflammation-induced cardiometabolic risk |
| Economou, 2004 [62] | Greece | 72 prepubertal obese children (33 ♂), aged 7 to 10 years and 42 controls | Obese compared with lean children for a possible relation among plasma total homocysteine, monocyte chemoattractant protein-1 (MCP-1) and RANTES | Negative association between circulating total homocysteine and proinflammatory chemokines MCP-1 and RANTES in prepubertal lean, but not in obese children |
| Giannini, 2009 [63] | Italy | 40 obese children (19 ♂, mean age 8.5 years), 40 lean children (22 ♂, mean age 8.7 years) and 40 matched controls (24 ♂, mean age 8.1 years) | Oxidant–antioxidant status, inflammatory markers and carotid intima-media thickness | Prepubertal lean and obese children present increased oxidative stress and impaired inflammation and insulin sensitivity |
| Maggio, 2018 [64] | Switzerland, Italy | 48 children (28 ♂, mean age 9.7 years), 35 after behavioral obesity intervention and 13 controls | Cytokines, adiponectin, neutrophil product MMP-8, carotid IMT, flow-mediated dilation, nitroglycerin-mediated dilation, arterial stiffness, pulse wave velocity, resting and 24-hour BP after a 6-month behavioral intervention to treat obesity | Behavioral interventions resulted in a paradoxical increase in some biomarkers in children, with potentially beneficial effects detected with CCL2 changes |
| Marti, 2021 [65] | Spain | 29 children in a lifestyle intervention (14 ♂), aged 7 to 16 years | Lifestyle intervention with a 2-month intensive phase and a subsequent 10-month follow-up with a moderate calorie-restricted diet, physical activity and nutritional education | Higher lipopolysaccharide binding protein and chemerin concentrations were associated with MetS in children with abdominal obesity during a lifestyle intervention |
| Akinci, 2007 [66] | Turkey | 50 children (22 ♂, mean age 11.5 years) of parents with MetS and 38 matched controls | Comparison of anthropometric measurements, BP, echocardiography, flow-mediated vasodilatation and metabolic fasting blood measurements | Children of patients with MetS have higher values of the serum markers of inflammation |
| Olza, 2015 [67] | Spain | 146 overweight (65♂), 295 obese (161 ♂) and 236 normal weight children (127♂), aged 4 to 12 years | MetS score traits, markers of inflammation, endothelial damage and CVD risk | MetS score with specific risk biomarkers of inflammation, endothelial damage and CVD are useful in the early identification of children at increased risk of metabolic dysfunction |
| Gonzalez-Jimenez, 2016 [68] | Spain | 976 adolescents, mean age 13.2 years; 930 in non-MetS group (441 ♂) and 46 in MetS group (16 ♂) | Measurement of interleukin-6 (IL-6), tumor necrosis factor-a (TNFa), high- sensitivity CRP and ceruloplasmin | Subjects with MetS exhibited higher levels of TNF2a, IL-6, CRP and ceruloplasmin |
| Kelly, 2006 [69] | USA | 11 normal weight and healthy children (3 ♂), 13 overweight and healthy (7 ♂) and 10 overweight with the MetS (overweight + MetS) (5 ♂), aged 8 to 14 years | Body composition, BP, lipids, glucose tolerance, markers of insulin resistance, oxidative stress, and adipokines | Oxidative stress and adverse adipokine profile characterize the MetS in children |
| Scuteri, 2011 [70] | USA, Italy | 6148 children and adults, aged 14–102 years, enrolled in the SardiNIA Study | MetS components, common carotid artery diameter, IMT and aortic pulse wave velocity, adiponectin, leptin, high-sensitivity CRP, monocyte chemoattractant protein 1 and interleukin-6 levels | MetS and specific cytokine patterns are associated with arterial aging, the increases in arterial stiffness and thickness |
| Lin, 2009 [71] | Taiwan | 4723 children, aged 12 to 17 years | Measurements of various serum hepatic profiles and metabolic risks from Health and Nutrition Examination Survey 1999–2004 | Serum bilirubin is inversely associated with insulin resistance and MetS among children and adolescents |
| Ruperez, 2020 [72] | Spain | 1444 children (706 ♂), aged 3 to 17 years | Plasma antioxidants, oxidative stress biomarkers and associations with pro-inflammatory and endothelial damage biomarkers | Antioxidants and oxidative stress biomarkers showed novel associations with several pro-inflammatory and endothelial damage biomarkers |
| Huerta-Delgado, 2020 [73] | Mexico | 21 children with T2DM (10 ♂), 19 with MetS (11 ♂) and 17 healthy controls (8 ♂), aged 6 to 16 years | Associations among circulating irisin levels, soluble cell adhesion molecules (sCAMs) and inflammatory cytokines | MetS and T2DM patients have lower serum irisin levels. T2DM subjects have lower concentrations of most sCAMs compared to MetS patients |
| Reilly, 1998 [74] | USA, Italy, Australia | 38 children and adults (24 ♂), aged 3 to 24 years with homozygous familial hypercholesterolemia and 24 adults (16 ♂) with moderate hypercholesterolemia | Comparison of hyper- to normocholesterolemic control subjects for F2 isoprostanes iPF2a-III, iPF2a-VI and arachidonic acid (AA) | Asymptomatic patients with moderate and severe hypercholesterolemia have evidence of oxidant stress in vivo |
| Charakida, 2009 [75] | Greece | 38 children with familial hypercholesterolemia (19 ♂, mean age 14.8 years) and 41 healthy controls (22 ♂, mean age 15.4 years) | Endothelium dependent reactive hyperemia, endothelium-independent nitrate hyperemia dilatation, inflammatory and hemostatic parameters | Inflammatory and thrombotic processes are associated with vascular dysfunction in children with familial hypercholesterolemia |
| Holven, 2006 [76] | Norway | 33 children with familial hypercholesterolemia (13 ♂, mean age 14 years), 14 hypercholesterolemic adults (7 ♂, mean age 47 years) and 30 controls | Gene expression of chemokines in peripheral blood mononuclear cells from clinically healthy children with and without heterozygous familial hypercholesterolemia | A role of inflammation in the early stages of atherogenesis, possibly involving monocyte-derived RANTES |
| Loffredo, 2012 [77] | Italy | 20 children with hypercholesterolemia (10 ♂, mean age 10 years), 20 with obesity (10 ♂, mean age 10 years), 20 obese hypercholesterolemic children (10 ♂, mean age 10 years) and 40 healthy controls (19 ♂, mean age 10 years) | Interplay among oxidative stress, NOX2, the catalytic core of nicotinamide-adenine dinucleotide phosphate oxidase, and endothelial dysfunction in children with obesity and/or hypercholesterolemia | NOX2-generating oxidative stress may have a pathogenic role in the functional changes of the arterial wall occurring in obesity and/or hypercholesterolemia |
| Martino, 2008 [78] | Italy | 50 children with hypercholesterolemia (19 ♂, mean age 10 years) and 50 healthy controls (25 ♂, mean age 9.2 years) | Comparison of flow-mediated dilation, IMT, lipid profile, urinary isoprostanes, markers of oxidative stress and platelet expression of gp91phox | Gp91phox-mediated oxidative stress may have a pathogenic role in the anatomic and functional changes of the arterial wall occurring in children with premature atherosclerosis |
| Ece, 2006 [79] | Turkey | 29 children with CRF (14 ♂, mean age 10.2 years) and 25 healthy controls (14 ♂, mean age 8.3 years) | Markers of oxidative stress (superoxide dismutase, catalase activities, glutathione and malondialdehyde levels), inflammation and early cardiovascular abnormalities | Increased oxidant stress and inflammation together with early cardiovascular damage were found in children with CRF |
| Ece, 2006 [80] | Turkey | 29 children with CRF (14 ♂, mean age 10.2 years) and 25 healthy controls (14 ♂, mean age 8.3 years) | Investigation of PON activity, total antioxidant response, total peroxide, oxidative stress index and some pro-oxidant cytokines | Low levels of serum albumin and high levels of uremic metabolites might be responsible for increased oxidative stress in children with CRF |
| Hamed, 2012 [81] | Egypt | 40 children with CKD (25 ♂) and 20 healthy children (10 ♂), aged 6 to 15 years | Effect of CKD and hemodialysis (HD) on hypoxia and oxidative stress biomarkers | Patients with CKD succumb considerable tissue hypoxia with oxidative stress. HD ameliorates hypoxia but lowers antioxidants (decreased levels of hypoxia induced factor-1α and total antioxidant capacity) |
| Zachwieja, 2005 [82] | Poland | 21 children with ESRD; 10 on HD (4 ♂, mean age 15.2 years), 11 on peritoneal dialysis (8 ♂, mean age 10.2 years) and 9 healthy controls (4 ♂, mean age 8.9 years) | Cytokine synthesis and oxidative stress in peripheral blood lymphocytes of ESRD | Patients with ESRD, especially those treated with HD, present increased oxidative stress in T lymphocytes, which may lead to decreased cytokine synthesis and abnormal immune response |
| Maciejczyk, 2018 [83] | Poland | 25 children with CKD (15 ♂) and 25 healthy controls (15 ♂), aged 7 to 18 years | Evaluation of oxidative stress indicators in the non-stimulated and stimulated saliva | CKD is responsible for disturbances in salivary antioxidant systems and oxidative damage to proteins and lipids |
| Al-Biltagi, 2016 [84] | Egypt | 30 children with ESRD on regular hemodialysis (20 ♂, mean age 13.7 years) and 30 healthy controls | Cardiac function and plasma glutathione level, a marker of oxidative stress | Significant oxidative stress was present in children with ESRD and was correlated with the degree of cardiac dysfunction |
| Elshamaa, 2009 [85] | Egypt | 30 children on hemodialysis (17 ♂) and 20 controls (10 ♂), aged 4 to 18 years | Comparison of plasma total antioxidant capacity and lipid peroxidation products and correlation to high-sensitivity CRP | Children on HD have increased oxidative stress, which may explain the cardiovascular complications in HD patients |
| El-Saeed, 2015 [86] | Egypt | 30 children on hemodialysis (23 ♂, mean age 8.7 years) and 30 controls | Advanced glycation end products AGEs, oxidized LDL, soluble receptor AGE as markers of oxidative stress | The low molecular weight form of AGEs is associated with oxidative stress in children receiving chronic HD |
| Zwolinska, 2006 [87] | Poland | 32 children with moderate CRF (18 ♂, mean age 12 years), 14 children with advanced CRF (7 ♂, mean age 12 years), 21 children on hemodialysis (11 ♂, mean age 14.8 years) and 27 controls | Evaluation of the plasma, erythrocyte and dialysate levels of vitamins A and E and the plasma and dialysate levels of vitamin C as exogenous non-enzymatic antioxidants | CRF in children is associated with decreased concentrations of plasma antioxidant vitamins, particularly plasma vitamin C and erythrocyte vitamin E . |
| Zwolinska, 2006 [88] | Poland | 10 children on continuous ambulatory peritoneal dialysis (2 ♂, mean age 13.2 years), 21 children on hemodialysis (11 ♂, mean age 14.8 years) and 27 controls | Lipid peroxidation in plasma and erythrocytes, erythrocyte antioxidant enzyme activity and concentrations of Cu and Zn and Se in erythrocytes, plasma and in dialysis fluid | Increased oxidative stress occurs in children on maintenance dialysis, independent of dialysis modality. Oxidative stress is aggravated during every single HD session in children |
| Badawy, 2020 [89] | Egypt | 50 children with ESRD on regular hemodialysis (24 ♂), aged 5 to 16 years | Some inflammatory markers and measurement of left ventricular hypertrophy (LVH) | Elevated hsCRP and IL-18 are independent determinants of LVH in children with HD |
| Drozdz, 2016 [90] | Poland | 65 children with stage 1 to 5 CKD (41 ♂, mean age 11.2 years) | Oxidative stress biomarkers, cardiovascular risk factors and LVH | Hypertension and dyslipidemia correlated with lipid oxidation and oxLDLs seem to be valuable markers of oxidative stress in CKD patients, correlating with LVH |
| Garcia-Bello, 2014 [91] | Mexico | 134 children with CKD (68 ♂), aged 6–17 years | Inflammation and oxidative stress with carotid IMT (cIMT) and elasticity increment module (Einc) | cIMT and Einc strongly associate with several biochemical parameters and glutathione |
| Kotur-Stevuljevic, 2013 [92] | Serbia | 52 children with CKD (28 ♂, mean age 14.2 years) and 36 healthy controls | Dyslipidemia and oxidative stress in the early phases of atherosclerosis | Early atherosclerosis in CKD children is caused also by dyslipidemia and oxidative stress |
| Cengiz, 2009 [93] | Turkey | 28 patients receiving HD (15 ♂, mean age 15.1 years) and 20 healthy children (7 ♂, mean age 14.3 years) | Oxidative stress and cardiovascular risk factors | Children treated by hemodialysis are exposed to oxidative stress and chronic inflammation |
| Srivaths, 2010 [94] | USA | 16 children receiving HD (10 ♂, mean age 17.2 years) | Inflammation, malnutrition, renal osteodystrophy and coronary calcification | Coronary calcification (CC) is prevalent in older children receiving maintenance HD with a longer dialysis vintage. Worse renal osteodystrophy control and malnutrition (low cholesterol) may contribute to CC development. |
| Calo, 2006 [95] | Italy, USA | 8 children with renal transplant and hypertension (1 ♂, mean age 15 years) and 8 renal transplant patients without hypertension (4 ♂, mean age 16 years) | Oxidative stress signaling and posttransplant endothelial dysfunction and hypertension | Oxidative stress is associated with posttransplant hypertension in hypertensive pediatric kidney-transplant patients |
| Al-Mashhadi, 2018 [96] | Sweden | 15 patients with unilateral congenital hydronephrosis (14 ♂) and 2 matched control groups, i.e., 8 healthy controls (6 ♂) and 8 operated controls (7 ♂), aged 1 to 12 years | Measurement of changes in arterial pressure, oxidative stress and NO homeostasis following correction of hydronephrosis | There is increased arterial pressure and oxidative stress in children with hydronephrosis |
| Pavlova, 2005 [97] | Bulgaria | 39 patients with kidney disease (14 ♂, aged 2,6 to 17 years) and 13 healthy controls (8 ♂, aged 2 to 8 years) | Dynamics of oxidative stress in the blood and urine | Products of lipid peroxidation, intensity of chemiluminescence, total and enzyme antioxidant capacity in combination with clinical parameters are a proper test for the dynamics of oxidative stress |
| Biltagi, 2008 [98] | Egypt | 40 children with adenoidal hypertrophy and 20 control children, aged 4 to 8 years | Obstructive sleep apnea (OSA), levels of 8-isoprostane, interleukin-6 (IL-6) and cardiac diastolic dysfunctions | OSA severity is positively correlated with the degree of elevation of 8-isoprostane and IL-6 in breath condensate of children with OSA and with the degree of cardiac dysfunction |
| Gozal, 2008 [99] | USA | 20 children with OSA before tonsillectomy and adenoidectomy and after (12 ♂, mean age 6.5, 7.2 years, respectively) and 20 controls (12 ♂, mean age 6.4) | Interleukins -6 and -10 before and 4–6 months after tonsillectomy and adenoidectomy | Systemic inflammation is a component of OSA, even in the absence of obesity and is reversible upon treatment in most patients |
| Smith, 2021 [100] | USA | 43 patients with OSA (16 ♂, mean age 9 years) and 53 healthy controls (22 ♂, mean age 10 years) | Structural, functional carotid changes and inflammatory profiles | IL-6 and IL-8 were associated with changes in carotid function in children with OSA |
| Loffredo, 2015 [101] | Italy | 22 children with OSA (14 ♂, mean age 7.6 years) and 45 children with primary snoring (31 ♂, mean age 8.9 years) and 67 healthy controls (45 ♂, mean age 8.7 years) | Oxidative stress, assessed by serum isoprostanes and soluble NOX2-dp and endothelial function, assessed by flow-mediated dilation | NOX2-derived oxidative stress is involved in artery dysfunction in sleep disordered breathing children |
| Van Hoorenbeeck, 2012 [102] | Belgium | 132 obese subjects (41 ♂, median age 15.4 years) | Sleep-disordered breathing (SDB), inflammation and oxidative stress before and after weight loss | Oxidative stress is reflected by uric acid at baseline and the concentration decreases after treatment according to improvements in SDB |
| Babar, 2019 [103] | USA | 52 children with T1DM, aged 12 to 18 years, from these 27 with hemoglobin A1c (HbA1c) ≤8.5% and 25 with HbA1c ≥9.5% | Glycemic control, vascular oxidative stress, inflammation and vascular health | Elevated E-selectin level is an early marker of oxidative stress in T1DM patients with an elevated HbA1c level |
| Suys, 2007 [104] | Belgium | 35 children with T1DM (16 ♂, mean age 14 years), matched with 35 non-diabetic controls (18 ♂, mean age 14 years) | Endothelial dysfunction and oxidative stress | Low Cu/Zn superoxide dismutase is a potential early marker of susceptibility to diabetic vascular disease |
| Sochett, 2017 [105] | Canada, UK | 51 children with T1DM (25 ♂, mean age 14.8 years) and 59 healthy controls (26 ♂, mean age 13.9 years) | Systemic inflammation and vascular function | The cytokine-chemokine signature in early type 1 diabetes mellitus, prior to the development of arterial disease, is significantly different from that seen in healthy controls |
| Mylona-Karayanni, 2006 [106] | Greece | 45 children with T1DM (27 ♂, mean age 13.8 years) and 20 healthy controls (11 ♂, mean age 13.2 years) | Oxidative stress parameters and adhesion molecules derived from endothelial/platelet activation | Decreased antioxidative protection from simultaneous nitrite and lipid hydroperoxide overproduction is evident in T1DM juveniles with a parallel endothelial/platelet activation even in the first years of the disease and contribute to the vascular complications |
| Stankovic, 2012 [107] | Serbia | 40 children with T1DM (30 ♂, mean age 12.8 years) and 20 healthy controls (13 ♂, mean age 11.8 years) | Traditional and novel risk factors, such as anti-oxidative capacity of circulating blood and level of lipid peroxidation and atherosclerosis | Despite the documented increased oxidative stress, there was no correlation between the oxidative stress parameters and coronary IMT values |
| Seckin, 2006 [108] | Turkey | 100 children with T1DM (54 ♂), aged 2 to 17 years | Effect of glycemic control on oxidative stress | Increased levels of MDA, ICAM-1, NO and VEGF present higher risk for atherosclerosis in poorly controlled T1DM in children |
| Sharma, 2013 [109] | UK | 32 young patients with T1DM and 32 controls | Novel method to measure metabolites of vitamin E and its measurement | Metabolites of vitamin E are increased in children with type 1 diabetes indicating an increased oxidative stress |
| Chiesa, 2019 [110] | UK | 70 adolescents with T1DM, aged 10 to 17 years | High-density lipoprotein and endothelial function | Impaired endothelial function in patients with high inflammatory risk score and high levels of HDL |
| Yau, 2017 [111] | USA | 157 adolescents (112 overweight), aged 16 to 22 years | Relationships between retinal vessel diameter, physical fitness, insulin sensitivity, systemic inflammation | Improved physical fitness and insulin function reduces inflammation in adolescents and improve cerebrovascular function later in life |
| Roberts, 2007 [112] | USA | 19 overweight children, aged 8 to 17 years | Effects of lifestyle modification on oxidative markers | Reduced several traditional and novel factors associated with atherosclerosis |
| Llorente-Cantarero, 2021 [113] | Spain | 216 children (111 ♂), aged 6 to 14 years | Physical fitness and oxidative stress | A high physical-activity-sedentarism score is associated to a better redox profile |
| Kelly, 2004 [114] | USA | 25 children with overweight (12 ♂, mean age 10.9 years) | Exercise in overweight children and subclinical inflammation and endothelial function | Aerobic exercise improves fitness, HDL cholesterol, endothelial function; CRP is associated with fasting insulin |
| Leite-Almeida, 2021 [115] | Portugal | 313 children (150 overweight/obese), aged 8 to 9 years | Physical activity and nitric oxide biomarkers in non-overweight and overweight/obese | Urinary nitrates were higher in children with higher physical activity and in non-overweight |
| Platat, 2006 [116] | France | 640 adolescents, aged 12 years | Effect of physical activity on symptoms of MetS and low-grade inflammation | In adolescents, physical activity is inversely related to HOMA and IL-6, independently of adiposity and fat localization |
| Liu, 2018 [117] | China | 50 obese female adolescents, aged 14 to 16 years | Exercise, diet and chemerin in study group | Aerobic exercise with diet showed decreased chemerin in comparison to diet only group |
| Dennis, 2013 [118] | USA | 112 overweight/obese children, aged 7 to 11 years | Exercise and plasma isoprostane levels | No effect of exercise on isoprostane levels; isoprostane levels were related to several cardiovascular risk at baseline |
| Santiprabhob, 2018 [119] | Thailand | 115 obese young individuals (mean age of 60 males 12,1 years and mean age of females 12,5 years) | Lifestyle modification program and CVD markers | Healthy lifestyle program for obese youths had beneficial effects on adipocytokines, inflammatory cytokines and arterial stiffness |
| Cazeau, 2016 [120] | USA | 2 males, 7 females with T1DM, mean age 12.9 years | Antioxidant supplementation and oxidative damage | Antioxidant therapy does not decrease oxidative damage, improve endothelial function or increase vascular repair capacity |
| Goldstein, 2012 [121] | USA | 23 vitamin C and 21 placebo assigned subject after Fontan completion, aged 8 to 25 years | Vitamin C and function health status in Fontan-palliated patients | Shorter therapy with vitamin C does not alter endothelial function or exercise capacity; it might provide benefit to those with abnormal vascular function |
| Hoffman, 2012 [122] | USA | 5 male, 3 female adolescents with T1DM, mean age 14.2 years | Ascorbic acid and hyperglycemia | Antioxidant treatment with vitamin C blocs acute hyperglycemic impairment of endothelial function |
| Chiarelli, 2004 [123] | Italy | 12 adolescents with T1DM, aged 11 to 21 years | Vitamin E and early signs of microangiopathy | Early diabetic angiopathy in adolescents cannot be modified by vitamin E |
| Németh, 2000 [124] | Hungary | 10 children, mean age 15.2 years | Chronic hemodialysis, recombinant human erythropoietin and vitamin E | Vitamin E supplementation improved the therapeutic effect of rhEPO in patients with chronic renal failure on hemodialysis |
| Kelishadi, 2010 [125] | Iran | 60 obese children, aged 6 to 10 years | Zinc and insulin resistance, oxidative stress and inflammation in obese | Significant decrease was documented for Apo B/ApoA-I ratio, ox-LDL, leptin and malondialdehyde, total and LDL-cholesterol after receiving zinc |
| Soongswang, 2005 [126] | Thailand | 15 patients with idiopathic dilated cardiomyopathy, aged 0.6 to 16.3 years | Coenzyme Q10, cardiac function and quality of life | CoQ10 may improve NYHA class and cardiothoracic ratio and shorten ventricular depolarization |
| Serag, 2021 [127] | Egypt | 49 T1DM pediatric patients and 7 healthy controls | Coenzyme Q10 and various metabolic parameters | CoQ10 had no favorable effect on endothelial dysfunction or metabolic parameters |
| Bennett-Richards, 2002 [128] | UK | 21 normotensive children with CRF, aged 7 to 17 years | L-arginine and endothelial function | Endothelial function was not improved with L-arginine |
| Lim, 2004 [129] | USA | 8 children after heart transplantation (4 ♂), aged 9 to 29 years and 15 healthy controls | L-arginine, endothelial function and high BP | Oral L-arginine therapy reverses endothelial dysfunction and attenuates high blood pressure in hypertensive cardiac transplant recipients |
| Bo, 2012 [130] | China | 10 children with typical nonthrombocytopenic purpura and 10 healthy controls | Supernatant IL-8, TNF-alpha and NO levels of human umbilical vein endothelial cells and sera of participants and extracted flavonoids from B. bipinnata | Extracted flavonoids from B. bipinnata improve microvascular inflammation in Henoch–Schönlein patients |
| Hashemi, 2010 [131] | Iran | 30 adolescents with MetS, aged 12 to 15 years | Grape and pomegranate juice and endothelial function | Daily consumption of diets rich in antioxidants might improve endothelial function in adolescents with MetS |
| Mohammad, 2021 [132] | Iran | 42 adolescents with MetS, aged 13 to 19 years | Grape seed extract effect on insulin resistance | Grape seed extract improves insulin concentration and insulin resistance in adolescents with metabolic syndrome |
| Sukardi, 2016 [133] | Indonesia | 45 children after tetralogy of Fallot corrective surgery, aged 1 to 6 years | Curcumin and the concentrations of malondialdehyde and glutathione, activity of nuclear factor-kappa B, c-Jun N-terminal kinase, caspase-3 and post-operative clinical outcomes | Cardioprotective effects of curcumin may include inhibition of the c-Jun N-terminal kinase pathway and caspase-3 in cardiomyocytes |
| Elbarbary, 2018 [134] | Egypt | 90 patients with diabetic nephropathy; 45 in carnosine suppl. group (20 ♂, mean age 12.4) and 45 children in placebo group | Carnosine and urinary albumin excretion, alpha 1-microglobulin, oxidative stress | Oral supplementation with L-Carnosine for 12 weeks resulted in a significant improvement of oxidative stress, glycemic control and renal function |
| Xia, 2003 [135] | China | 20 children with congenital heart defects and pulmonary hypertension, aged 2 to 15 years | Placebo or Salvia miltiorrhiza before cardiac surgery | Antioxidant therapy reduces myocardial damage and attenuates postoperative vasoactive mediator imbalance |
| Cherukuri, 2020 [136] | USA | 67 individuals (26 ♂, mean age 59 years) | Plant based bioequivalent nitrate complex and NO in saliva | Endothelial function improved with reducing both systolic and diastolic BP in hypertensive individuals with daily supplementation of dietary NO3− |
| Hegazy, 2013 [137] | Egypt | 30 T1DM patients, aged 10 to 14 years | Insulin vs. insulin plus alpha-lipoic acid | Alpha-lipoic acid may have a role in preventing the development of diabetic cardiomyopathy in T1DM |
| Scaramuzza, 2015 [138] | Italy | 71 children and adolescents with T1DM, mean age 17 years | Antioxidant diet, alpha-lipoic acid, placebo | Alpha-lipoic acid might have an antioxidant effect in pediatric diabetes patients by reducing insulin |
| Rimoldi, 2015 [139] | Switzerland | 21 children conceived by assisted reproductive technology (8 ♂, mean age 12) and 21 controls | Vitamin C and placebo | Antioxidant administration to participants improved NO bioavailability and vascular responsiveness in the systemic and pulmonary circulation |
| Roszkowska, 2014 [140] | Poland | 87 babies and children, aged 5 to 32 months | Monocyte chemoattractant protein-1 and breast fed vs. bottle fed | Increased circulating inflammatory markers may indicate that formula-fed children are at risk of atherosclerosis |
| Bernini, 2016 [141] | Brazil | 51 participants with MetS, aged 18 to 60 years | Probiotics and placebo | Cardiovascular risk in patients with MetS might be reduced by probiotics (B. lactis HN019) |
| Gobel, 2012 [142] | Denmark | 50 adolescents with obesity, aged 12 to 15 years | Probiotic strain Lactobacillus salivarius and placebo | No beneficial effect of the probiotic intervention on inflammatory markers or parameters related to the MetS |
| Bresolin, 2013 [143] | Brazil | 33 children with congenital heart disease, aged 7 to 12 years | CV risk parameters and dental treatment (scaling and root planing or full-mouth scaling and root planing) | Both periodontal treatments were effective in children with congenital heart disease in reduction of CV markers |
| Zeigler, 2015 [144] | Sweden | 75 obese adolescents, aged 12 to 18 years | Obesity, periodontal disease and CV risk factors | Association between pathological periodontal pockets and diastolic BP |
| Reviews and editorials | ||||
| Berrahmoune, 2005 [1] | France | Human | Analysis of environmental, genetic and epigenetic determinants that influence inflammation in cardiovascular disease | Intermediate phenotype (protein and RNA level) variations in inflammation influence inter-individual risk of CVD |
| Barton, 2010 [2] | Switzerland | Human | Markers of oxidative stress and antioxidant capacity in obesity and aging and effect on vascular changes | Obesity and aging share similarities of pathomechanisms on vascular changes related to oxidative stress |
| Parisi, 2021 [145] | Italy | Animal and human | Intrauterine exposure to maternal low-grade chronic inflammation | Maternal obesity affects fetal programming of cardiovascular disease into adulthood |
| Skilton, 2008 [146] | France | Human | Intrauterine risk factors for precocious atherosclerosis | Impaired fetal growth, in utero exposure to maternal hypercholesterolemia and diabetic macrosomia as risk factors for fetal vascular changes |
| Leduc, 2010 [147] | Canada | Human | Fetal programming of atherosclerosis | Chronic in utero exposure to oxidative stress and inflammation, as also mitochondrial dysfunction may influence fetal programming of atherosclerosis |
| Silvestro, 2020 [148] | Italy | Animal and human | Role of placental oxidative stress on fetal development | Oxidative stress, mediated via placental hypoxia plays a key role in adverse effects on the developing offspring |
| Sutherland, 2014 [149] | Canada | Animal and human | Preterm birth, oxidative stress and hypertension risk | Oxidative stress associated with preterm birth affects neonatal development and future disease risk |
| Wang, 2015 [150] | Taiwan | Animal and human | Oxidative stress and renal damage in neonates | Recent studies elucidate the role of oxidative stress in neonatal renal damage |
| Yzydorczyk, 2017 [151] | Switzerland and France | Animal and human | Intrauterine growth restriction, endothelial dysfunction, CVD and renal consequences | Individuals born after IUGR exhibit vascular dysfunction, and preventive approaches (including antioxidant use) may mitigate long-term consequences |
| Hsu, 2020 [152] | Taiwan | Animal and human | Use of natural antioxidants as reprogramming strategies to precent developmental hypertension | Pregnant mothers and their children can benefit from natural antioxidant supplementation during pregnancy to reduce their risk for hypertension later in life |
| Radomski, 1995 [153] | Canada | Animal and human | The role of nitric oxide in the pathogenesis of atherosclerosis | It is possible that some of the detrimental effects of atherosclerosis on the nitric oxide pathway result from the generation of secondary oxidants such as peroxynitrite |
| Sabri, 2019 [154] | Iran | Human | Review of the epidemiology and risk factors in pediatric essential hypertension | Endothelial nitric oxide gene polymorphism is a possible factor to contribute to developing essential hypertension |
| Wirix, 2015 [155] | Netherlands | Human | Pathophysiology of hypertension in overweight and obese children | Development of hypertension in obese children is influenced by endocrine determinants, sympathetic nervous system activity, disturbed sodium homeostasis, as well as oxidative stress, inflammation and endothelial dysfunction |
| Filgueiras, 2020 [156] | Brazil | Human | Vitamin D status, oxidative stress and inflammation in children and adolescents | Vitamin D status is associated with oxidative stress and inflammation |
| Vincent, 2007 [157] | USA | Human | Oxidative stress and potential interventions to reduce oxidative stress in overweight and obesity | Oxidative stress may be corrected by improving antioxidant defenses through fat volume reduction via surgery, pharmacological agents, exercise and/or dietary modification |
| Espinola-Klein, 2011 [158] | Germany | Human | Inflammatory markers and cardiovascular risk in MetS | Elevations of inflammatory biomarkers impact the development of MetS and CVD |
| Balat, 2010 [159] | Turkey | Human | Nitric oxide (NO), adrenomedullin (AM), urotensin-II (U-II) in several pediatric renal diseases | NO, AM, and U-II may be important mediators in kidney diseases. They suggest a possible role in the pathophysiology of childhood glomerulonephritis |
| Massy, 2005 [160] | France | Human | Inflammation, oxidative stress, vascular calcifications in CKD | Inflammation and oxidative stress represent new features of the arterial and/or valvular calcification process |
| Avloniti, 2017 [161] | Greece | Animal and human | Oxidative stress responses to acute and chronic exercise in youth | Children and adolescents exhibit positive adaptations of their antioxidant system with exercise |
| Montero, 2012 [162] | France | Animal and human | Endothelial function, inflammation and oxidative stress in obese children | Obesity leads to endothelial dysfunction in children; early signs of CVD should be sought |
| Chen, 2012 [163] | Taiwan | Animal and human | Melatonin as antioxidant with anti-inflammatory effect | Melatonin modulates inflammation and increase mitochondrial biogenesis; potential therapeutic tool in a wide range of childhood disorders |
| Gombert, 2021 [164] | Spain | Animal and human | Melatonin and breast milk, antioxidant capacity, influence on weight gain | Melatonin has numerous benefits and is present in breast milk, making breastfeeding beneficial to long-term cardiovascular health, with an epigenetic effect on obesity |
| Risk Factors and Diseases | |
|---|---|
| Genetic predisposition | nitric oxide synthase gene polymorphism |
| gene polymorphisms of the renin–angiotensin–aldosterone system and aldosterone synthase genes | |
| single-nucleotide polymorphism of ATP2B1 (ATPase Plasma Membrane Ca2+ Transporting 1) familial hypercholesterolemia | |
| Perinatal factors | conception by assisted reproductive technology maternal hypercholesterolemia, obesity, metabolic syndrome placental insufficiency being small for gestational age preterm delivery formula-feeding |
| Environmental factors | lack of exercise |
| poor nutrition | |
| exposure to phthalates | |
| lower socioeconomic status | |
| Psycho-emotional factors | anxiety irritability predisposition to depressive reactions |
| Weight status | Underweight Overweight Obese |
| Immunosuppressive treatment | cyclosporine tacrolimus |
| Dialysis | |
| Hypovitaminosis | D and B12 |
| Hyperhomocysteinemia | |
| Kidney diseases | chronic kidney disease hydronephrosis glomerulonephritis pyelonephritis lower urinary tract infections |
| Arterial hypertension | essential and secondary |
| Obstructive sleep apnea | |
| Diabetes mellitus | type 1 and 2 |
| Metabolic syndrome |
| Oxidative Stress Markers | Antioxidant System Markers | Adipokines and Other Systemic Inflammation Markers |
|---|---|---|
| Lipid peroxidation F2 isoprostanes malondialdehyde (MDA) thiobarbituric acid reactive substances (TBARS) oxidized low density lipoprotein (oxLDL) Protein oxidation Advanced oxidation protein products (AOPPS) Carbohydrate oxidation Advanced glycosylation end-products (AGEs) Nucleic acid oxidation 8-hydroxy-2′-deoxyguanosine (8-OHdG) Reactive oxygen species (ROS) generation myeloperoxidase (MPO) NADPH 1 oxidase (NOX2) Nitric oxide system (NOx) polyamines derived from arginine asymmetric dimethyloarginine (ADMA) nitrite and nitrate NO | Thiol/disulphide homeostasis glutathione (GSH) superoxide dismutase (SOD) catalase (CAT) glutathione peroxidase (GPx) carotenes (vitamin A) ascorbic acid (vitamin C) tocopherols (vitamins E) bilirubin ceruloplasmin Total antioxidant capacity (TAC) | Chemerin adiponectin leptin resistin visfatin adipomyokine irisin RANTES 2 monocyte chemoattractant protein-1 (MCP-1) stromal-derived factor (SDF-1) interleukins IL-1, -1β, -6, -10, -18 tumor necrosis factor alpha (TNF-α) plasminogen activator-inhibitor-1 (PAI-1) α-1-acid glycoprotein (AGP) high sensitivity C-reactive protein (hsCRP) C-reactive protein (CRP) myeloperoxidase (MPO) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hertiš Petek, T.; Petek, T.; Močnik, M.; Marčun Varda, N. Systemic Inflammation, Oxidative Stress and Cardiovascular Health in Children and Adolescents: A Systematic Review. Antioxidants 2022, 11, 894. https://doi.org/10.3390/antiox11050894
Hertiš Petek T, Petek T, Močnik M, Marčun Varda N. Systemic Inflammation, Oxidative Stress and Cardiovascular Health in Children and Adolescents: A Systematic Review. Antioxidants. 2022; 11(5):894. https://doi.org/10.3390/antiox11050894
Chicago/Turabian StyleHertiš Petek, Tjaša, Tadej Petek, Mirjam Močnik, and Nataša Marčun Varda. 2022. "Systemic Inflammation, Oxidative Stress and Cardiovascular Health in Children and Adolescents: A Systematic Review" Antioxidants 11, no. 5: 894. https://doi.org/10.3390/antiox11050894
APA StyleHertiš Petek, T., Petek, T., Močnik, M., & Marčun Varda, N. (2022). Systemic Inflammation, Oxidative Stress and Cardiovascular Health in Children and Adolescents: A Systematic Review. Antioxidants, 11(5), 894. https://doi.org/10.3390/antiox11050894






