Potential Valorization of Edible Nuts By-Products: Exploring the Immune-Modulatory and Antioxidants Effects of Selected Nut Shells Extracts in Relation to Their Metabolic Profiles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Extraction
2.2. Total Phenolics Determination
2.3. Total Flavonoids Determination
2.4. Determination of Total Antioxidant Activity Using DPPH ASSAY
2.5. Determination of Total Antioxidant Activity Using FRAP Assay
2.6. Determination of Total Antioxidant Activity Using ABTS Assay
2.7. Ultra-Performance Liquid Chromatography Coupled with Tandem Mass Spectrometry (UPLC-MS/MS) Analysis
2.8. Biological Screening
2.8.1. In Vitro Cytotoxicity Using MTT Assay
2.8.2. LPS Induction in THP-1 Macrophage
2.8.3. RNA Extraction
2.8.4. Quantitative Polymerase Chain Reaction (q-PCR)
2.8.5. Cytokine Determination by ELISA
2.9. Docking Studies
2.9.1. Preparation of the Selected Compounds from the Studied Four Nut Shell Extracts
2.9.2. iNOS Receptor Pocket Preparation
2.9.3. Docking of the Mentioned Database into the iNOS Binding Pocket
2.10. Data Analysis and Visualization
3. Results
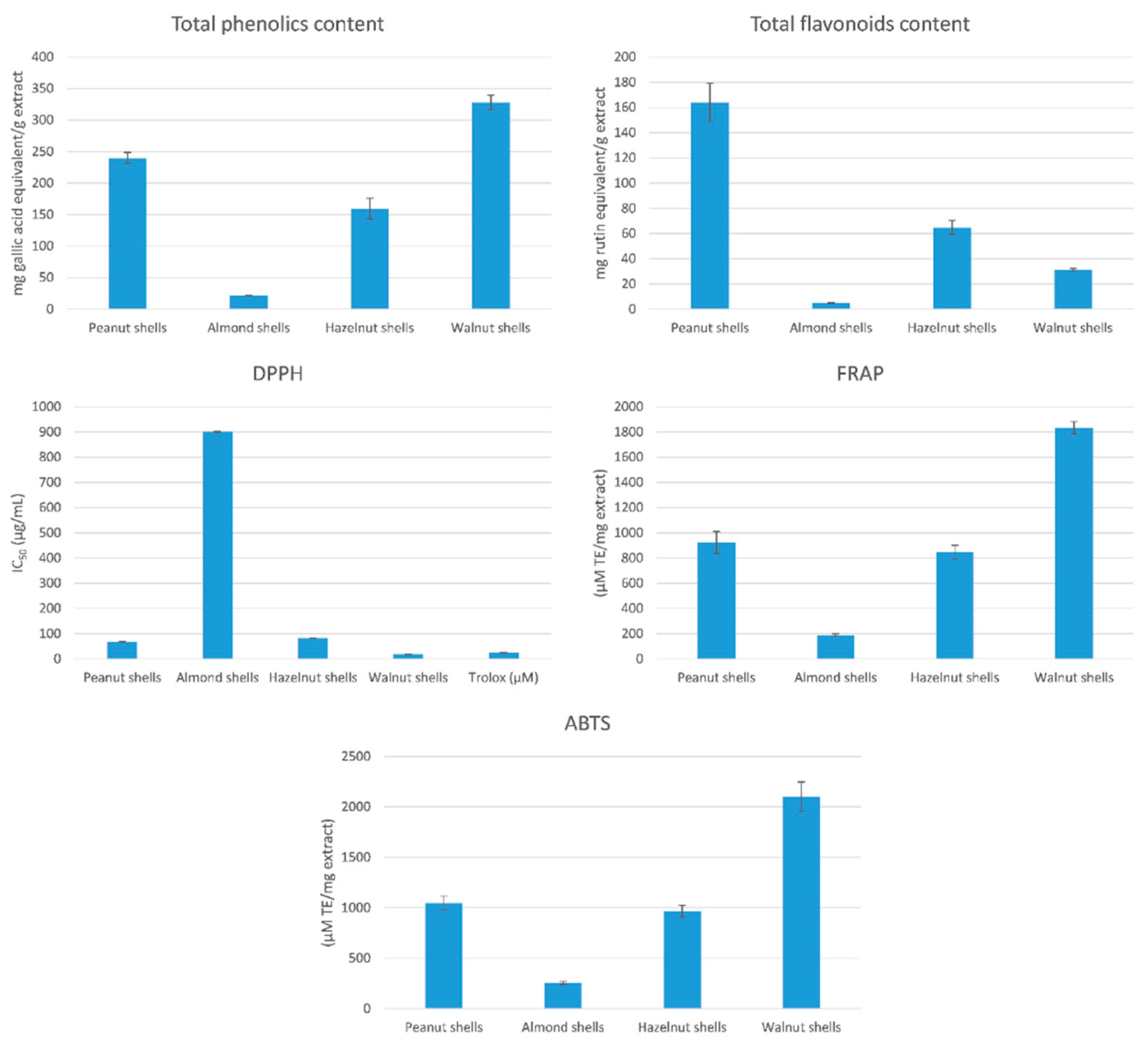
3.1. Total Phenolic and Flavonoid Contents
3.2. Determination of Total Antioxidant Activity
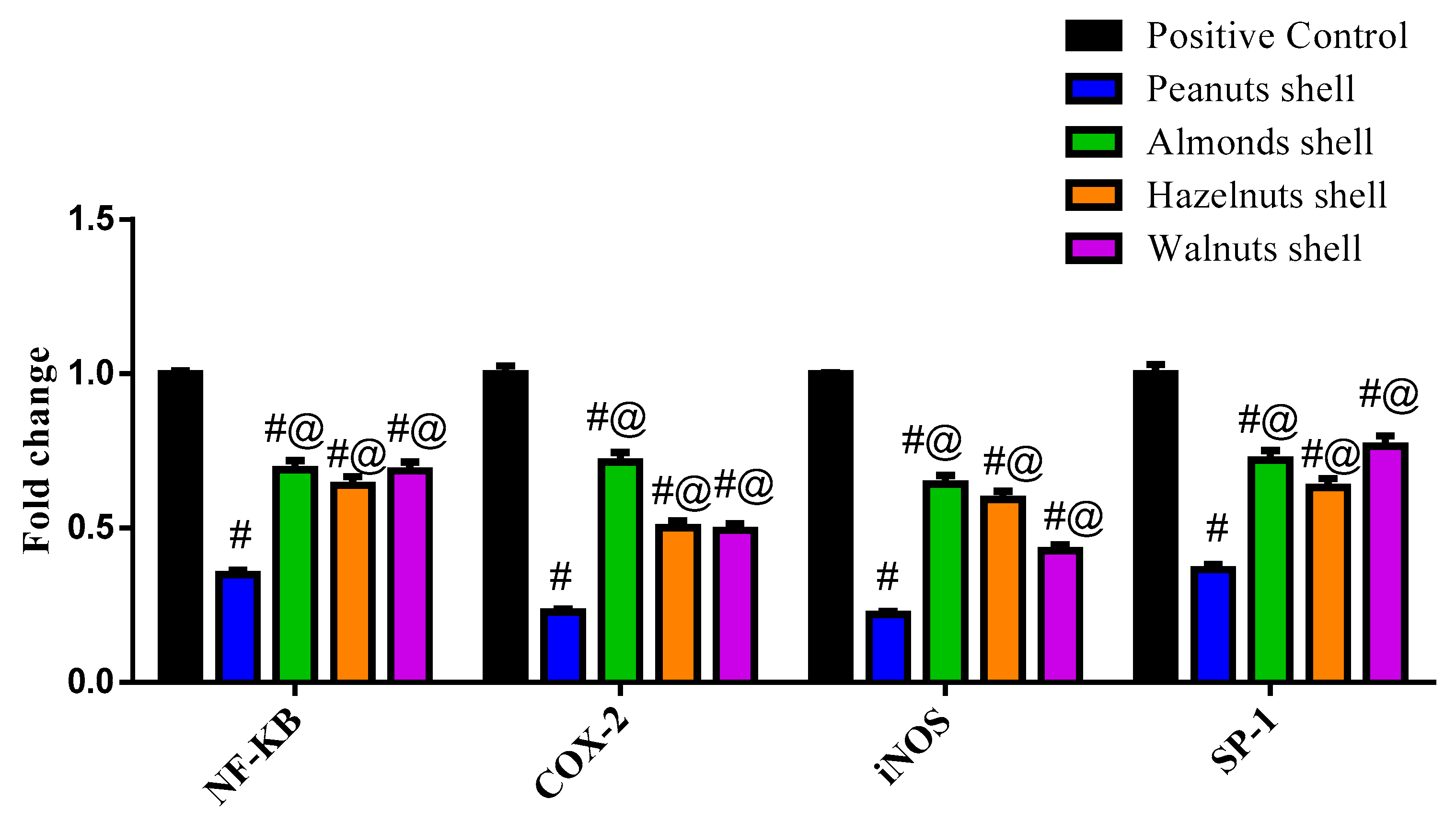
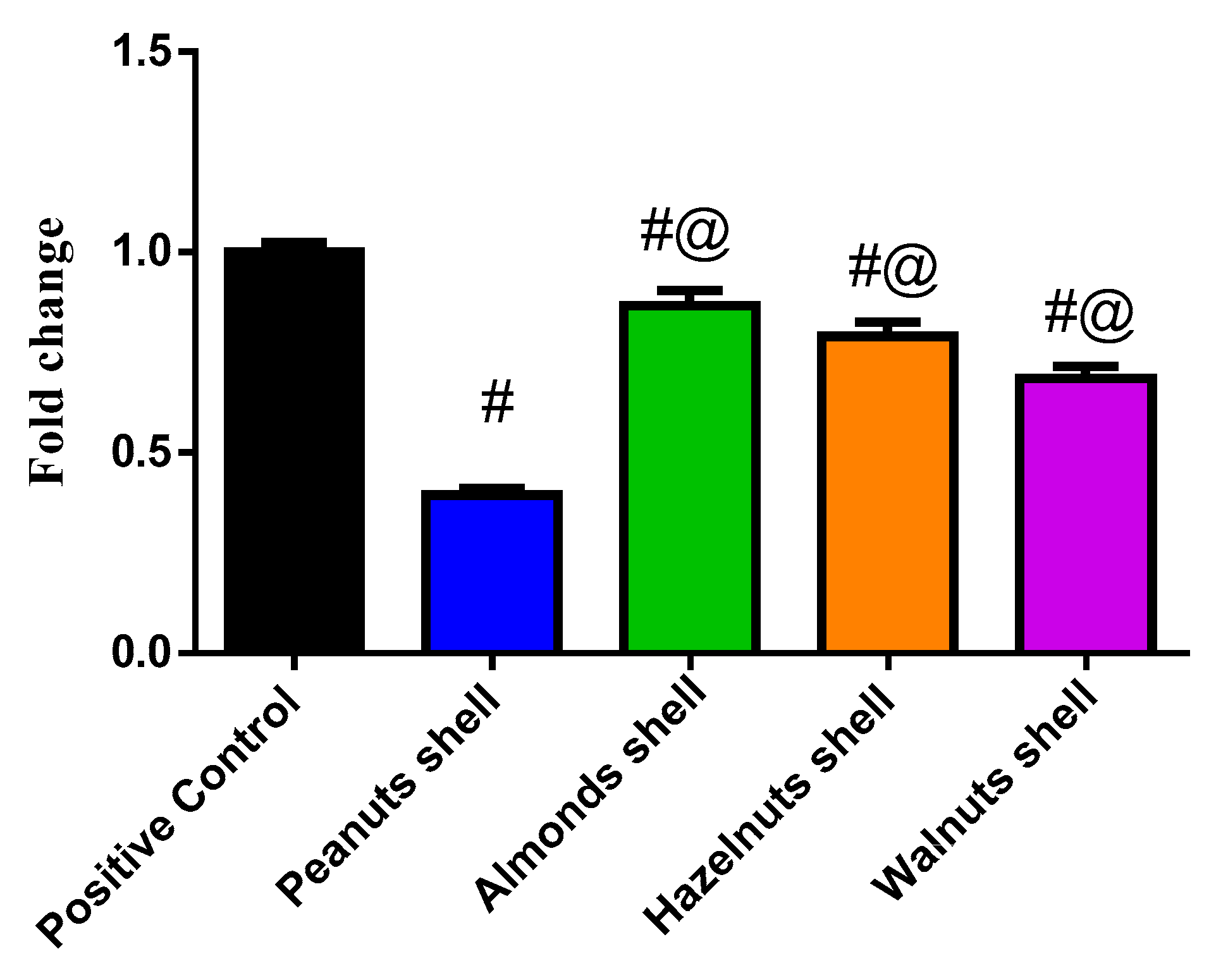
3.3. Evaluation of the Cytotoxicity Using MTT Assay and Gene Expression by q-PCR
3.4. Cytokines Determination by ELISA
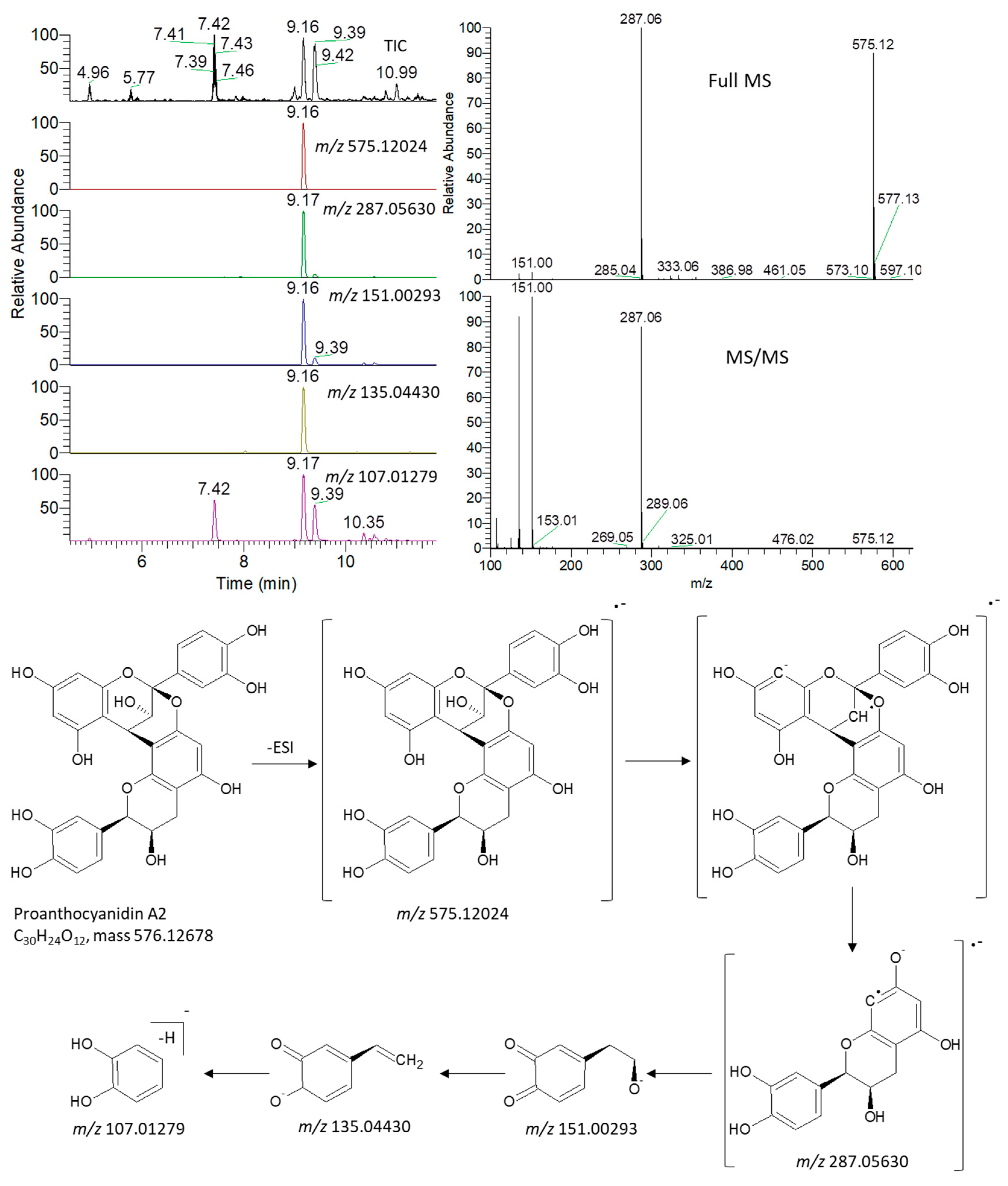
3.5. Metabolite Identification and Multivariate Data Analysis
3.6. Docking Studies
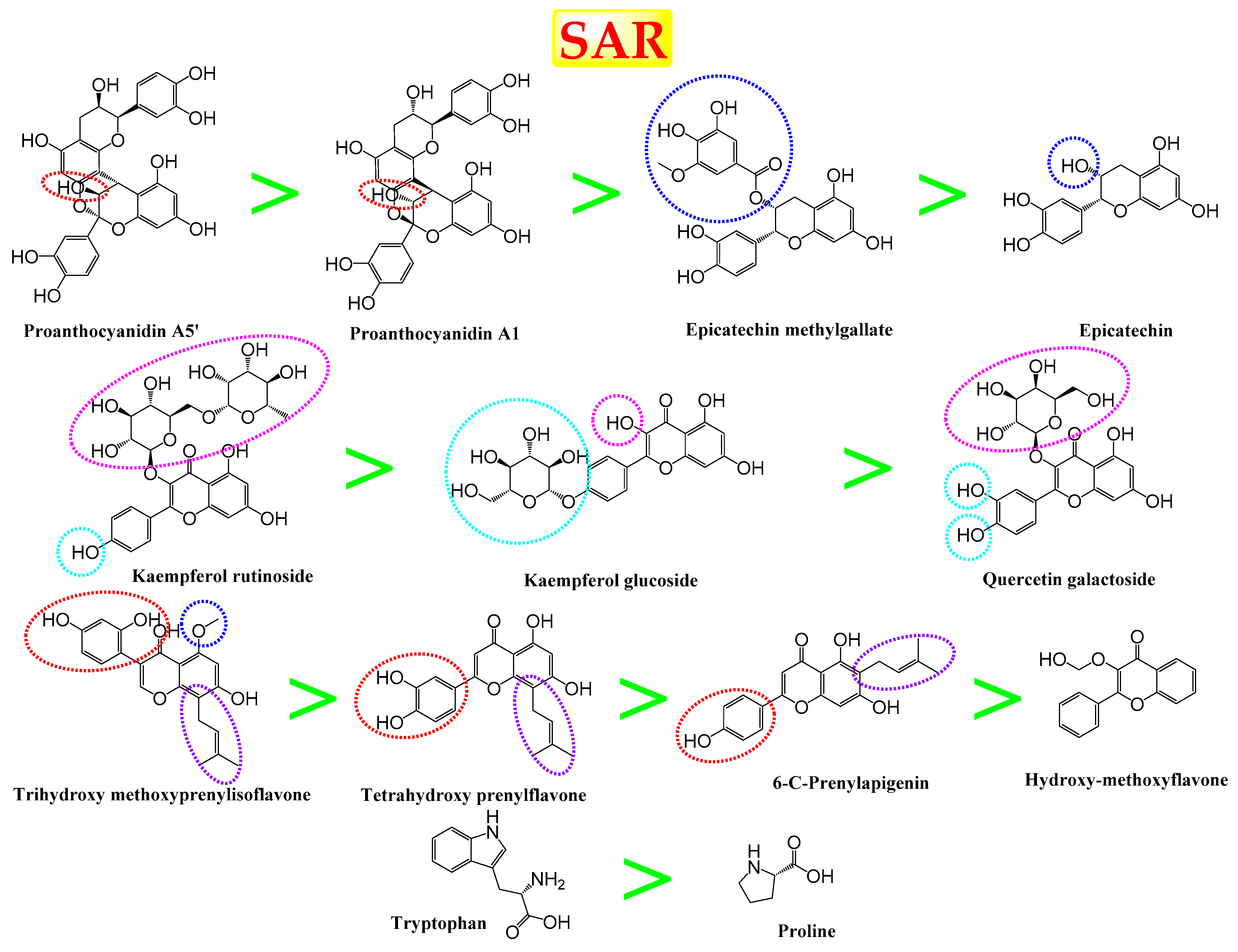
3.7. Structure–Activity Relationship (SAR)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nicholson, L.B. The immune system. Essays Biochem. 2016, 60, 275–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levy, O.; Netea, M.G. Innate immune memory: Implications for development of pediatric immunomodulatory agents and adjuvanted vaccines. Pediatr. Res. 2014, 75, 184–188. [Google Scholar] [CrossRef]
- Ongradi, J.; Kovesdi, V. Factors that may impact on immunosenescence: An appraisal. Immun. Ageing 2010, 7, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McLean-Tooke, A.; Spickett, G.P.; Gennery, A.R. Immunodeficiency and autoimmunity in 22q11.2 deletion syndrome. Scand. J. Immunol. 2007, 66, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Fontanet, A.; Cauchemez, S. COVID-19 herd immunity: Where are we? Nat. Rev. Immunol. 2020, 20, 583–584. [Google Scholar] [CrossRef] [PubMed]
- Patil, A.; Kakde, M. Medicinal plant as a natural immunity booster for COVID19-A review. Indian J. Integr. Med. 2020, 2, 24–27. [Google Scholar]
- Carmona, F.; Pereira, A.M.S. Herbal medicines: Old and new concepts, truths and misunderstandings. Rev. Bras. Farmacogn. 2013, 23, 379–385. [Google Scholar] [CrossRef] [Green Version]
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The Traditional Medicine and Modern Medicine from Natural Products. Molecules 2016, 21, 559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Venkatachalam, M.; Sathe, S.K. Chemical composition of selected edible nut seeds. J. Agric. Food Chem. 2006, 54, 4705–4714. [Google Scholar] [CrossRef]
- Muradoglu, F.; Oguz, H.I.; Yildiz, K. Some chemical composition of walnut (Juglans regia L.) selections from Eastern Turkey. Afr. J. Agric. Res. 2010, 5, 2379–2385. [Google Scholar]
- Bottone, A.; Cerulli, A.; D’Urso, G.; Masullo, M.; Montoro, P.; Napolitano, A.; Piacente, S. Plant Specialized Metabolites in Hazelnut (Corylus avellana) Kernel and Byproducts: An Update on Chemistry, Biological Activity, and Analytical Aspects. Planta Med. 2019, 85, 840–855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Esposito, T.; Sansone, F.; Franceschelli, S.; Del Gaudio, P.; Picerno, P.; Aquino, R.P.; Mencherini, T. Hazelnut (Corylus avellana L.) Shells Extract: Phenolic Composition, Antioxidant Effect and Cytotoxic Activity on Human Cancer Cell Lines. Int. J. Mol. Sci. 2017, 18, 392. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.A.; Oliveira, I.; Sousa, A.; Valentao, P.; Andrade, P.B.; Ferreira, I.C.; Ferreres, F.; Bento, A.; Seabra, R.; Estevinho, L. Walnut (Juglans regia L.) leaves: Phenolic compounds, antibacterial activity and antioxidant potential of different cultivars. Food Chem. Toxicol. 2007, 45, 2287–2295. [Google Scholar] [CrossRef] [PubMed]
- De, L. Edible seeds and nuts in human diet for immunity development. Int. J. Recent Sci. Res. 2020, 6, 38877–38881. [Google Scholar]
- Karra, G.; Nadenla, R.; Kiran, R.S.; Srilatha, K.; Mamatha, P.; Rao, V.U. An overview on Arachis hypogaea plant. Int. J. Pharm. Sci. Res. (IJPSR) 2013, 4, 4508–4518. [Google Scholar]
- Al-Snafi, A. Chemical constituents and pharmacological activities of Arachis hypogaea—A review. Int. J. Pharm. Res. Sch. 2014, 3, 615–623. [Google Scholar]
- Attard, E. A rapid microtitre plate Folin-Ciocalteu method for the assessment of polyphenols. Open Life Sci. 2013, 8, 48–53. [Google Scholar] [CrossRef]
- Kiranmai, M.; Kumar, C.M.; Mohammed, I. Comparison of total flavanoid content of Azadirachta indica root bark extracts prepared by different methods of extraction. Res. J. Pharm. Biol. Chem. Sci. 2011, 2, 254–261. [Google Scholar]
- Boly, R.; Lamkami, T.; Lompo, M.; Dubois, J.; Guissou, I. DPPH free radical scavenging activity of two extracts from Agelanthus dodoneifolius (Loranthaceae) leaves. Int. J. Toxicol. Pharmacol. Res. 2016, 8, 29–34. [Google Scholar]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnao, M.B.; Cano, A.; Acosta, M. The hydrophilic and lipophilic contribution to total antioxidant activity. Food Chem. 2001, 73, 239–244. [Google Scholar] [CrossRef]
- Salem, M.A.; Yoshida, T.; Perez de Souza, L.; Alseekh, S.; Bajdzienko, K.; Fernie, A.R.; Giavalisco, P. An improved extraction method enables the comprehensive analysis of lipids, proteins, metabolites and phytohormones from a single sample of leaf tissue under water-deficit stress. Plant J. 2020, 103, 1614–1632. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- El-Demerdash, A.; Al-Karmalawy, A.A.; Abdel-Aziz, T.M.; Elhady, S.S.; Darwish, K.M.; Hassan, A.H.E. Investigating the structure–activity relationship of marine natural polyketides as promising SARS-CoV-2 main protease inhibitors. RSC Adv. 2021, 11, 31339–31363. [Google Scholar] [CrossRef]
- Elebeedy, D.; Elkhatib, W.F.; Kandeil, A.; Ghanem, A.; Kutkat, O.; Alnajjar, R.; Saleh, M.A.; Abd El Maksoud, A.I.; Badawy, I.; Al-Karmalawy, A.A. Anti-SARS-CoV-2 activities of tanshinone IIA, carnosic acid, rosmarinic acid, salvianolic acid, baicalein, and glycyrrhetinic acid between computational and in vitro insights. RSC Adv. 2021, 11, 29267–29286. [Google Scholar] [CrossRef]
- Mahmoud, A.; Mostafa, A.; Al-Karmalawy, A.A.; Zidan, A.; Abulkhair, H.S.; Mahmoud, S.H.; Shehata, M.; Elhefnawi, M.M.; Ali, M.A. Telaprevir is a potential drug for repurposing against SARS-CoV-2: Computational and in vitro studies. Heliyon 2021, 7, e07962. [Google Scholar] [CrossRef]
- El-Shershaby, M.H.; Ghiaty, A.; Bayoumi, A.H.; Al-Karmalawy, A.A.; Husseiny, E.M.; El-Zoghbi, M.S.; Abulkhair, H.S. From triazolophthalazines to triazoloquinazolines: A bioisosterism-guided approach toward the identification of novel PCAF inhibitors with potential anticancer activity. Bioorg. Med. Chem. 2021, 42, 116266. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, R.J.; Garcin, E.D.; Panda, K.; Andersson, G.; Aberg, A.; Wallace, A.V.; Morris, G.M.; Olson, A.J.; Stuehr, D.J.; Tainer, J.A.; et al. Conformational changes in nitric oxide synthases induced by chlorzoxazone and nitroindazoles: Crystallographic and computational analyses of inhibitor potency. Biochemistry 2002, 41, 13915–13925. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, A.E.; Alesawy, M.S.; Eissa, S.I.; El-Fakharany, E.M.; Kalaba, M.H.; Sharaf, M.H.; Abo Shama, N.M.; Mahmoud, S.H.; Mostafa, A.; Al-Karmalawy, A.A.; et al. Design and synthesis of new 4-(2-nitrophenoxy)benzamide derivatives as potential antiviral agents: Molecular modeling and in vitro antiviral screening. New J. Chem. 2021, 45, 16557–16571. [Google Scholar] [CrossRef]
- Elshal, M.; Eid, N.; El-Sayed, I.; El-Sayed, W.; Al-Karmalawy, A.A. Concanavalin-A shows synergistic cytotoxicity with tamoxifen via inducing apoptosis in estrogen receptor-positive breast cancer: In vitro and molecular docking studies. Pharm. Sci. 2022, 28, 76–85. [Google Scholar] [CrossRef]
- Gaber, A.A.; El-Morsy, A.M.; Sherbiny, F.F.; Bayoumi, A.H.; El-Gamal, K.M.; El-Adl, K.; Al-Karmalawy, A.A.; Ezz Eldin, R.R.; Saleh, M.A.; Abulkhair, H.S. Pharmacophore-linked pyrazolo[3,4-d]pyrimidines as EGFR-TK inhibitors: Synthesis, anticancer evaluation, pharmacokinetics, and in silico mechanistic studies. Arch. Pharm. 2021, e2100258. [Google Scholar] [CrossRef]
- Hamed, M.I.A.; Darwish, K.M.; Soltane, R.; Chrouda, A.; Mostafa, A.; Abo Shama, N.M.; Elhady, S.S.; Abulkhair, H.S.; Khodir, A.E.; Elmaaty, A.A.; et al. β-Blockers bearing hydroxyethylamine and hydroxyethylene as potential SARS-CoV-2 Mpro inhibitors: Rational based design, in silico, in vitro, and SAR studies for lead optimization. RSC Adv. 2021, 11, 35536–35558. [Google Scholar] [CrossRef]
- Khattab, M.; Al-Karmalawy, A.A. Computational repurposing of benzimidazole anthelmintic drugs as potential colchicine binding site inhibitors. Future Med. Chem. 2021, 13, 1623–1638. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, D.B.; Ismail, W.M.; Moatasim, Y.; Kutkat, O.; ElMeshad, A.N.; Ezzat, S.M.; El Deeb, K.S.; El-Fishawy, A.M.; Gomaa, M.R.; Kandeil, A.; et al. Delineating a potent antiviral activity of Cuphea ignea extract loaded nano-formulation against SARS-CoV-2: In silico and in vitro studies. J. Drug Deliv. Sci. Technol. 2021, 66, 102845. [Google Scholar] [CrossRef]
- Mahmoud, D.B.; Bakr, M.M.; Al-Karmalawy, A.A.; Moatasim, Y.; El Taweel, A.; Mostafa, A. Scrutinizing the Feasibility of Nonionic Surfactants to Form Isotropic Bicelles of Curcumin: A Potential Antiviral Candidate Against COVID-19. AAPS PharmSciTech 2021, 23, 44. [Google Scholar] [CrossRef] [PubMed]
- Shoala, T.; Al-Karmalawy, A.A.; Germoush, M.O.; ALshamrani, S.M.; Abdein, M.A.; Awad, N.S. Nanobiotechnological Approaches to Enhance Potato Resistance against Potato Leafroll Virus (PLRV) Using Glycyrrhizic Acid Ammonium Salt and Salicylic Acid Nanoparticles. Horticulturae 2021, 7, 402. [Google Scholar] [CrossRef]
- Pang, Z.; Chong, J.; Zhou, G.; de Lima Morais, D.A.; Chang, L.; Barrette, M.; Gauthier, C.; Jacques, P.E.; Li, S.; Xia, J. MetaboAnalyst 5.0: Narrowing the gap between raw spectra and functional insights. Nucleic Acids Res. 2021, 49, W388–W396. [Google Scholar] [CrossRef] [PubMed]
- Rue, E.A.; Rush, M.D.; van Breemen, R.B. Procyanidins: A comprehensive review encompassing structure elucidation via mass spectrometry. Phytochem. Rev. 2018, 17, 1–16. [Google Scholar] [CrossRef]
- Yuzuak, S.; Ballington, J.; Xie, D.Y. HPLC-qTOF-MS/MS-Based Profiling of Flavan-3-ols and Dimeric Proanthocyanidins in Berries of Two Muscadine Grape Hybrids FLH 13-11 and FLH 17-66. Metabolites 2018, 8, 57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jahanban-Esfahlan, A.; Jahanban-Esfahlan, R.; Tabibiazar, M.; Roufegarinejad, L.; Amarowicz, R. Recent advances in the use of walnut (Juglans regia L.) shell as a valuable plant-based bio-sorbent for the removal of hazardous materials. RSC Adv. 2020, 10, 7026–7047. [Google Scholar] [CrossRef] [Green Version]
- Ezzat, M.I.; Hassan, M.; Abdelhalim, M.A.; El-Desoky, A.M.; Mohamed, S.O.; Ezzat, S.M. Immunomodulatory effect of Noni fruit and its isolates: Insights into cell-mediated immune response and inhibition of LPS-induced THP-1 macrophage inflammation. Food Funct. 2021, 12, 3170–3179. [Google Scholar] [CrossRef] [PubMed]
- Aneklaphakij, C.; Saigo, T.; Watanabe, M.; Naake, T.; Fernie, A.R.; Bunsupa, S.; Satitpatipan, V.; Tohge, T. Diversity of Chemical Structures and Biosynthesis of Polyphenols in Nut-Bearing Species. Front. Plant Sci. 2021, 12, 642581. [Google Scholar] [CrossRef] [PubMed]
- Dourado, F.; Madureira, P.; Carvalho, V.; Coelho, R.; Coimbra, M.A.; Vilanova, M.; Mota, M.; Gama, F.M. Purification, structure and immunobiological activity of an arabinan-rich pectic polysaccharide from the cell walls of Prunus dulcis seeds. Carbohydr. Res. 2004, 339, 2555–2566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, D.; Wang, M.; Shen, M.; Liu, C.; Liu, W.; Min, W.; Liu, J. In vivo assessment of immunomodulatory activity of hydrolysed peptides from Corylus heterophylla Fisch. J. Sci. Food Agric. 2016, 96, 3508–3514. [Google Scholar] [CrossRef]
- Wang, S.; Giller, K.; Hillmann, E.; Marquardt, S.; Schwarm, A. Effect of supplementation of pelleted hazel (Corylus avellana) leaves on blood antioxidant activity, cellular immune response, and heart beat parameters in sheep. J. Anim. Sci. 2019, 97, 4496–4502. [Google Scholar] [CrossRef]
- Mythily, K.; Revathi, K. Effect of Peanut (Arachis hypogea L.) extracts as a feed additive on growth and hematological parameters of rohu fish (Labeo rohita) fingerlings. Int. J. Adv. Multidiscip. Res. 2017, 4, 1–8. [Google Scholar]
- Mao, R.; Wu, L.; Zhu, N.; Liu, X.; Hao, Y.; Liu, R.; Du, Q.; Li, Y. Immunomodulatory effects of walnut (Juglans regia L.) oligopeptides on innate and adaptive immune responses in mice. J. Funct. Foods 2020, 73, 104068. [Google Scholar] [CrossRef]
- Shahidi, F.; Alasalvar, C.; Liyana-Pathirana, C.M. Antioxidant phytochemicals in hazelnut kernel (Corylus avellana L.) and hazelnut byproducts. J. Agric. Food Chem. 2007, 55, 1212–1220. [Google Scholar] [CrossRef]
- Bottone, A.; Montoro, P.; Masullo, M.; Pizza, C.; Piacente, S. Metabolomics and antioxidant activity of the leaves of Prunus dulcis Mill. (Italian cvs. Toritto and Avola). J. Pharm. Biomed. Anal. 2018, 158, 54–65. [Google Scholar] [CrossRef]
- Lopes, R.M.; Agostini-Costa Tda, S.; Gimenes, M.A.; Silveira, D. Chemical composition and biological activities of Arachis species. J. Agric. Food Chem. 2011, 59, 4321–4330. [Google Scholar] [CrossRef]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Hawkins Byrne, D. Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J. Food Compos. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Dudonne, S.; Vitrac, X.; Coutiere, P.; Woillez, M.; Merillon, J.M. Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J. Agric. Food Chem. 2009, 57, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Katalinic, V.; Milos, M.; Kulisic, T.; Jukic, M. Screening of 70 medicinal plant extracts for antioxidant capacity and total phenols. Food Chem. 2006, 94, 550–557. [Google Scholar] [CrossRef]
- Wong, C.-C.; Li, H.-B.; Cheng, K.-W.; Chen, F. A systematic survey of antioxidant activity of 30 Chinese medicinal plants using the ferric reducing antioxidant power assay. Food Chem. 2006, 97, 705–711. [Google Scholar] [CrossRef]
- Saad, B.; Abouatta, B.S.; Basha, W.; Hmade, A.; Kmail, A.; Khasib, S.; Said, O. Hypericum triquetrifolium-Derived Factors Downregulate the Production Levels of LPS-Induced Nitric Oxide and Tumor Necrosis Factor-alpha in THP-1 Cells. Evid.-Based Complementary Altern. Med. eCAM 2011, 2011, 586470. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S.; Lee, K.M.; Lee, J.A.; Choi, I. Peanut shell extract inhibits the development of dextran sulfate sodium (DSS)-induced colitis. Int. Immunopharmacol. 2019, 70, 235–240. [Google Scholar] [CrossRef]
- Moens, U.; Kostenko, S.; Sveinbjornsson, B. The Role of Mitogen-Activated Protein Kinase-Activated Protein Kinases (MAPKAPKs) in Inflammation. Genes 2013, 4, 101–133. [Google Scholar] [CrossRef]
- Hu, W.H.; Dai, D.K.; Zheng, B.Z.; Duan, R.; Chan, G.K.; Dong, T.T.; Qin, Q.W.; Tsim, K.W. The binding of kaempferol-3-O-rutinoside to vascular endothelial growth factor potentiates anti-inflammatory efficiencies in lipopolysaccharide-treated mouse macrophage RAW264.7 cells. Phytomed. Int. J. Phytother. Phytopharm. 2021, 80, 153400. [Google Scholar] [CrossRef]
- Iyer, S.S.; Cheng, G. Role of interleukin 10 transcriptional regulation in inflammation and autoimmune disease. Crit. Rev. Immunol. 2012, 32, 23–63. [Google Scholar] [CrossRef] [Green Version]
- Chanput, W.; Mes, J.; Vreeburg, R.A.; Savelkoul, H.F.; Wichers, H.J. Transcription profiles of LPS-stimulated THP-1 monocytes and macrophages: A tool to study inflammation modulating effects of food-derived compounds. Food Funct. 2010, 1, 254–261. [Google Scholar] [CrossRef]
- Soltane, R.; Chrouda, A.; Mostafa, A.; Al-Karmalawy, A.A.; Chouaib, K.; Dhahri, A.; Pashameah, R.A.; Alasiri, A.; Kutkat, O.; Shehata, M.; et al. Strong Inhibitory Activity and Action Modes of Synthetic Maslinic Acid Derivative on Highly Pathogenic Coronaviruses: COVID-19 Drug Candidate. Pathogens 2021, 10, 623. [Google Scholar] [CrossRef] [PubMed]
- Abo Elmaaty, A.; Hamed, M.I.A.; Ismail, M.I.; Elkaeed, E.B.; Abulkhair, H.S.; Khattab, M.; Al-Karmalawy, A.A. Computational Insights on the Potential of Some NSAIDs for Treating COVID-19: Priority Set and Lead Optimization. Molecules 2021, 26, 3772. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Taghour, M.S.; Belal, A.; Mehany, A.B.M.; Mostafa, N.; Nabeeh, A.; Eissa, I.H.; Al-Karmalawy, A.A. Design and Synthesis of New Quinoxaline Derivatives as Potential Histone Deacetylase Inhibitors Targeting Hepatocellular Carcinoma: In Silico, In Vitro, and SAR Studies. Front. Chem. 2021, 9, 725135. [Google Scholar] [CrossRef]
- Al-Karmalawy, A.A.; Farid, M.M.; Mostafa, A.; Ragheb, A.Y.; Mahmoud, S.H.; Shehata, M.; Shama, N.M.A.; GabAllah, M.; Mostafa-Hedeab, G.; Marzouk, M.M. Naturally Available Flavonoid Aglycones as Potential Antiviral Drug Candidates against SARS-CoV-2. Molecules 2021, 26, 6559. [Google Scholar] [CrossRef] [PubMed]
- El Gizawy, H.A.; Boshra, S.A.; Mostafa, A.; Mahmoud, S.H.; Ismail, M.I.; Alsfouk, A.A.; Taher, A.T.; Al-Karmalawy, A.A. Pimenta dioica (L.) Merr. Bioactive Constituents Exert Anti-SARS-CoV-2 and Anti-Inflammatory Activities: Molecular Docking and Dynamics, In Vitro, and In Vivo Studies. Molecules 2021, 26, 5844. [Google Scholar] [CrossRef]
- Ambiga, S.; Narayanan, R.; Gowri, D.; Sukumar, D.; Madhavan, S. Evaluation of wound healing activity of flavonoids from Ipomoea carnea Jacq. Anc. Sci. Life 2007, 26, 45–51. [Google Scholar] [PubMed]
- Clericuzio, M.; Tinello, S.; Burlando, B.; Ranzato, E.; Martinotti, S.; Cornara, L.; La Rocca, A. Flavonoid oligoglycosides from Ophioglossum vulgatum L. having wound healing properties. Planta Med. 2012, 78, 1639–1644. [Google Scholar] [CrossRef] [Green Version]
- Petpiroon, N.; Suktap, C.; Pongsamart, S.; Chanvorachote, P.; Sukrong, S. Kaempferol-3-O-rutinoside from Afgekia mahidoliae promotes keratinocyte migration through FAK and Rac1 activation. J. Nat. Med. 2015, 69, 340–348. [Google Scholar] [CrossRef] [PubMed]
- do Nascimento, J.E.T.; de Morais, S.M.; de Lisboa, D.S.; de Oliveira Sousa, M.; Santos, S.; Magalhaes, F.E.A.; Campos, A.R. The orofacial antinociceptive effect of Kaempferol-3-O-rutinoside, isolated from the plant Ouratea fieldingiana, on adult zebrafish (Danio rerio). Biomed. Pharm. 2018, 107, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Beecher, G.R. Proanthocyanidins: Biological Activities Associated with Human Health. Arch. Physiol. Biochem. 2004, 42, 2–20. [Google Scholar] [CrossRef]
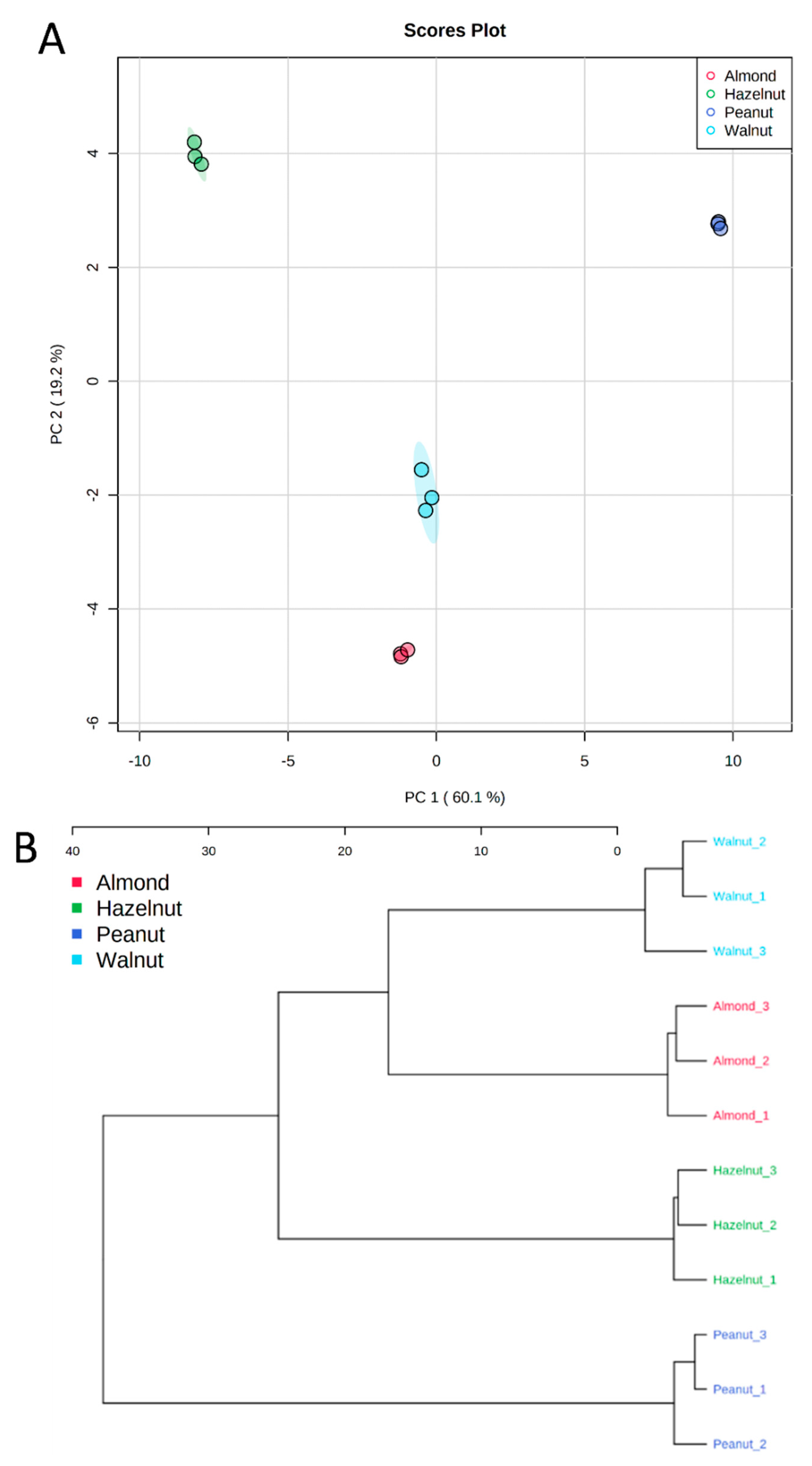
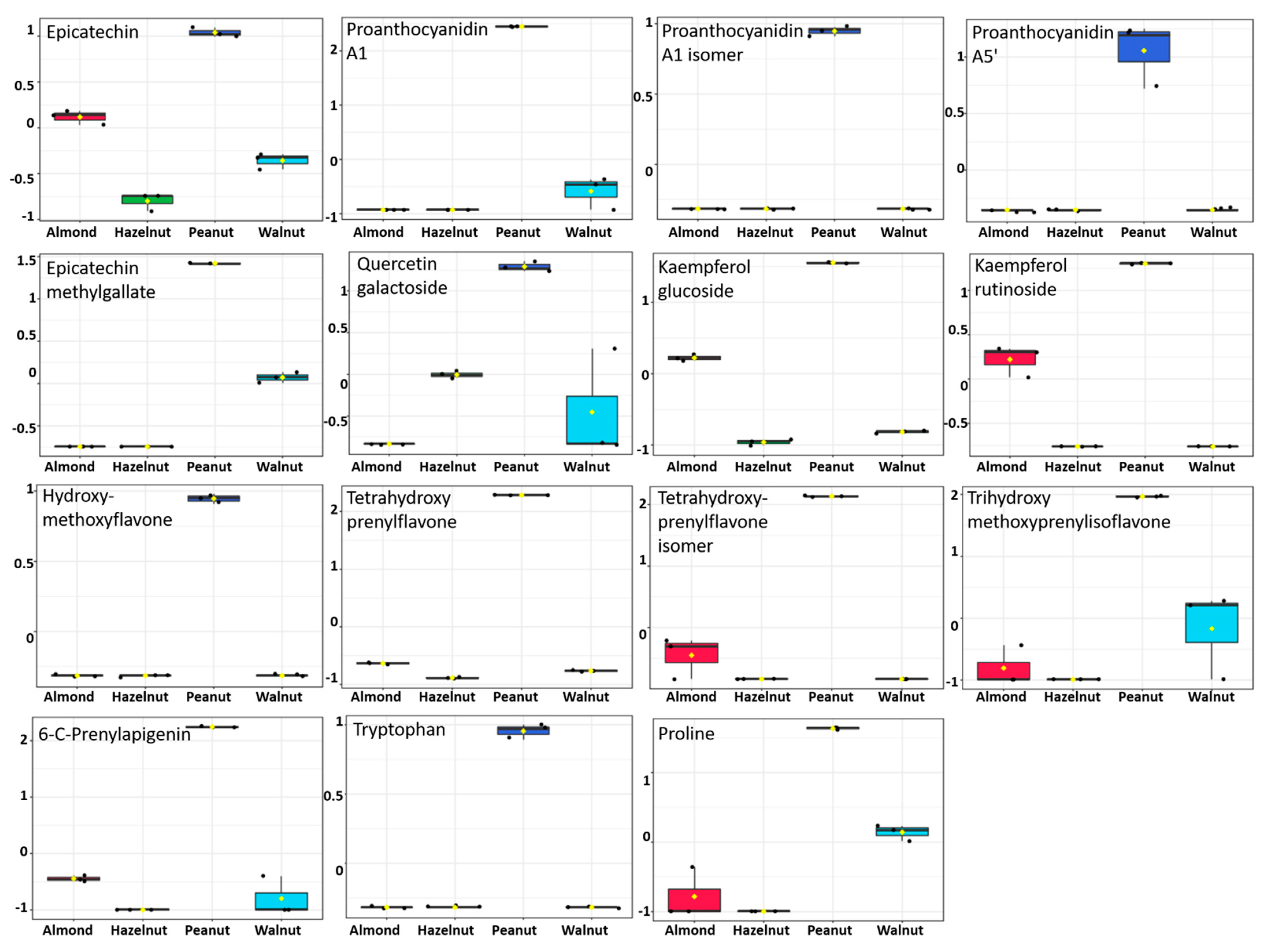

| Groups | IL-1β | IL-6 | IL-8 | IL-10 | TNF-α |
|---|---|---|---|---|---|
| Negative Control | 50.1 ± 5.9 | 41.6 ± 5.2 | 61.9 ± 2.25 | 165 ± 3.67 | 35.4 ± 2.1 |
| Positive Control | 289 ± 11 * | 347 ± 11.4 * | 610 ± 75.4 * | 29.3 ± 2.5 * | 273 ± 7.26 * |
| Peanut shells | 64.4 ± 4.6 # | 113.2 ± 5.46 *# | 135.6 ± 13.2 *# | 121 ± 11.2 # | 83.4 ± 4.35 *# |
| Almond shells | 171.1 ± 6.2 *#@ | 213.7 ± 21.4 *#@ | 458 ± 32 *#@ | 50.6 ± 1.97 *@ | 166 ± 13.9 *#@ |
| Hazelnut shells | 166.6 ± 5.6 *#@ | 212 ± 19.2 *#@ | 429 ± 41.5 *#@ | 52.9 ± 2.25 *@ | 178 ± 10.5 *#@ |
| Walnut shells | 204.2 ± 7.21 *#@ | 242 ± 28.5 *#@ | 505 ± 19.8 *#@ | 50.7 ± 1.91 *@ | 149 ± 1.93 *#@ |
| Compound | S a | RMSD b | Amino Acid Bond | L c |
|---|---|---|---|---|
| Epicatechin | −5.66 | 0.69 | Met368/H-acceptor | 3.16 |
| Epicatechin methylgallate | −7.54 | 1.92 | Trp366/H-piMet368/H-donor | 3.764.25 |
| Proanthocyanidin A1 | −7.73 | 1.90 | Met368/H-acceptor | 3.01 |
| Proanthocyanidin A5’ | −8.67 | 0.72 | Trp366/H-donorGlu371/H-donor | 2.853.02 |
| Kaempferol glucoside | −7.82 | 1.31 | Trp366/H-acceptorTrp366/H-donorTrp366/H-donor | 3.103.133.24 |
| Kaempferol rutinoside | −9.96 | 1.38 | Trp366/H-donorMet349/H-donorMet349/H-donor | 2.823.363.57 |
| Quercetin galactoside | −7.47 | 0.71 | Trp366/H-donor | 2.73 |
| Hydroxy-methoxyflavone | −5.87 | 1.34 | Met368/H-acceptorPro344/pi-H | 3.343.74 |
| Tetrahydroxy prenylflavone | −7.02 | 1.06 | Trp366/H-donorCys194/pi-H | 2.983.89 |
| Trihydroxy methoxyprenylisoflavone | −7.04 | 1.04 | Trp366/pi-H | 4.23 |
| 6-C-Prenylapigenin | −6.70 | 1.06 | Met368/H-acceptorTrp366/pi-H | 3.054.28 |
| Tryptophan | −5.51 | 1.44 | Trp366/H-donor | 2.81 |
| Proline | −4.60 | 0.72 | Trp366/H-acceptorMet428/H-donor | 3.544.49 |
| CLW | −4.78 | 1.46 | Trp366/H-donorMet368/H-acceptor | 2.973.04 |
| Compound | 3D Interactions | 3D Positioning |
|---|---|---|
| CLW | 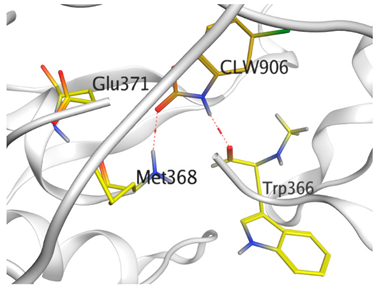 | 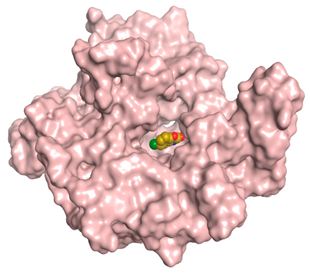 |
| Epicatechin-methyl gallate | 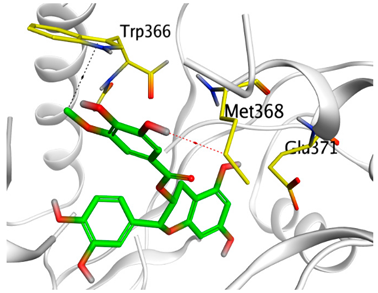 | 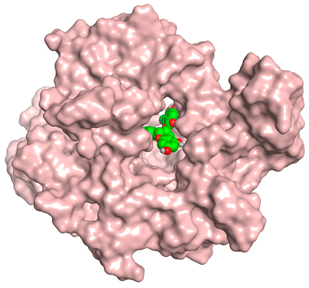 |
| Proanthocyanidin A1 | 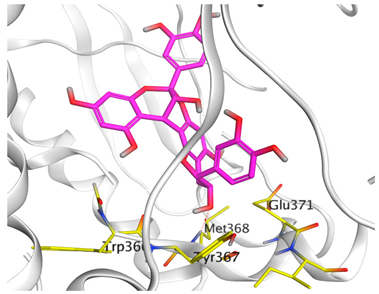 | 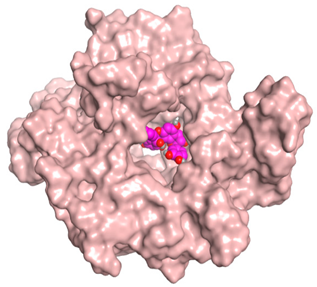 |
| Proanthocyanidin A5’ | 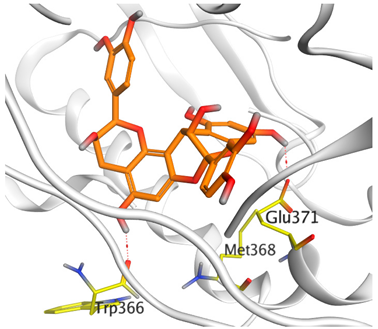 | 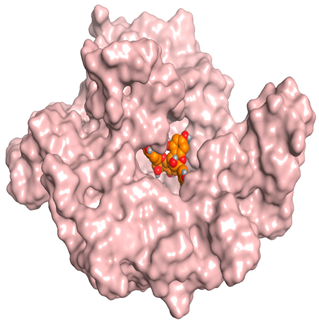 |
| Kaempferol glucoside |  | 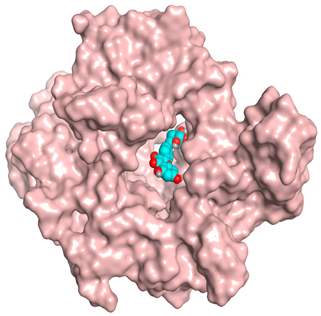 |
| Kaempferol rutinoside |  |  |
| Quercetin galactoside |  | 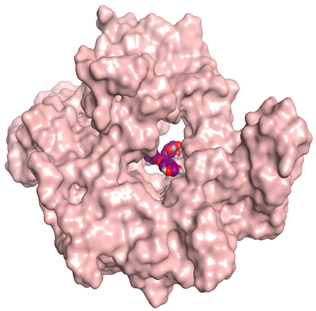 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salem, M.A.; Aborehab, N.M.; Al-Karmalawy, A.A.; Fernie, A.R.; Alseekh, S.; Ezzat, S.M. Potential Valorization of Edible Nuts By-Products: Exploring the Immune-Modulatory and Antioxidants Effects of Selected Nut Shells Extracts in Relation to Their Metabolic Profiles. Antioxidants 2022, 11, 462. https://doi.org/10.3390/antiox11030462
Salem MA, Aborehab NM, Al-Karmalawy AA, Fernie AR, Alseekh S, Ezzat SM. Potential Valorization of Edible Nuts By-Products: Exploring the Immune-Modulatory and Antioxidants Effects of Selected Nut Shells Extracts in Relation to Their Metabolic Profiles. Antioxidants. 2022; 11(3):462. https://doi.org/10.3390/antiox11030462
Chicago/Turabian StyleSalem, Mohamed A., Nora M. Aborehab, Ahmed A. Al-Karmalawy, Alisdair R. Fernie, Saleh Alseekh, and Shahira M. Ezzat. 2022. "Potential Valorization of Edible Nuts By-Products: Exploring the Immune-Modulatory and Antioxidants Effects of Selected Nut Shells Extracts in Relation to Their Metabolic Profiles" Antioxidants 11, no. 3: 462. https://doi.org/10.3390/antiox11030462
APA StyleSalem, M. A., Aborehab, N. M., Al-Karmalawy, A. A., Fernie, A. R., Alseekh, S., & Ezzat, S. M. (2022). Potential Valorization of Edible Nuts By-Products: Exploring the Immune-Modulatory and Antioxidants Effects of Selected Nut Shells Extracts in Relation to Their Metabolic Profiles. Antioxidants, 11(3), 462. https://doi.org/10.3390/antiox11030462







