Citrus Essential Oils in Aromatherapy: Therapeutic Effects and Mechanisms
Abstract
1. Introduction
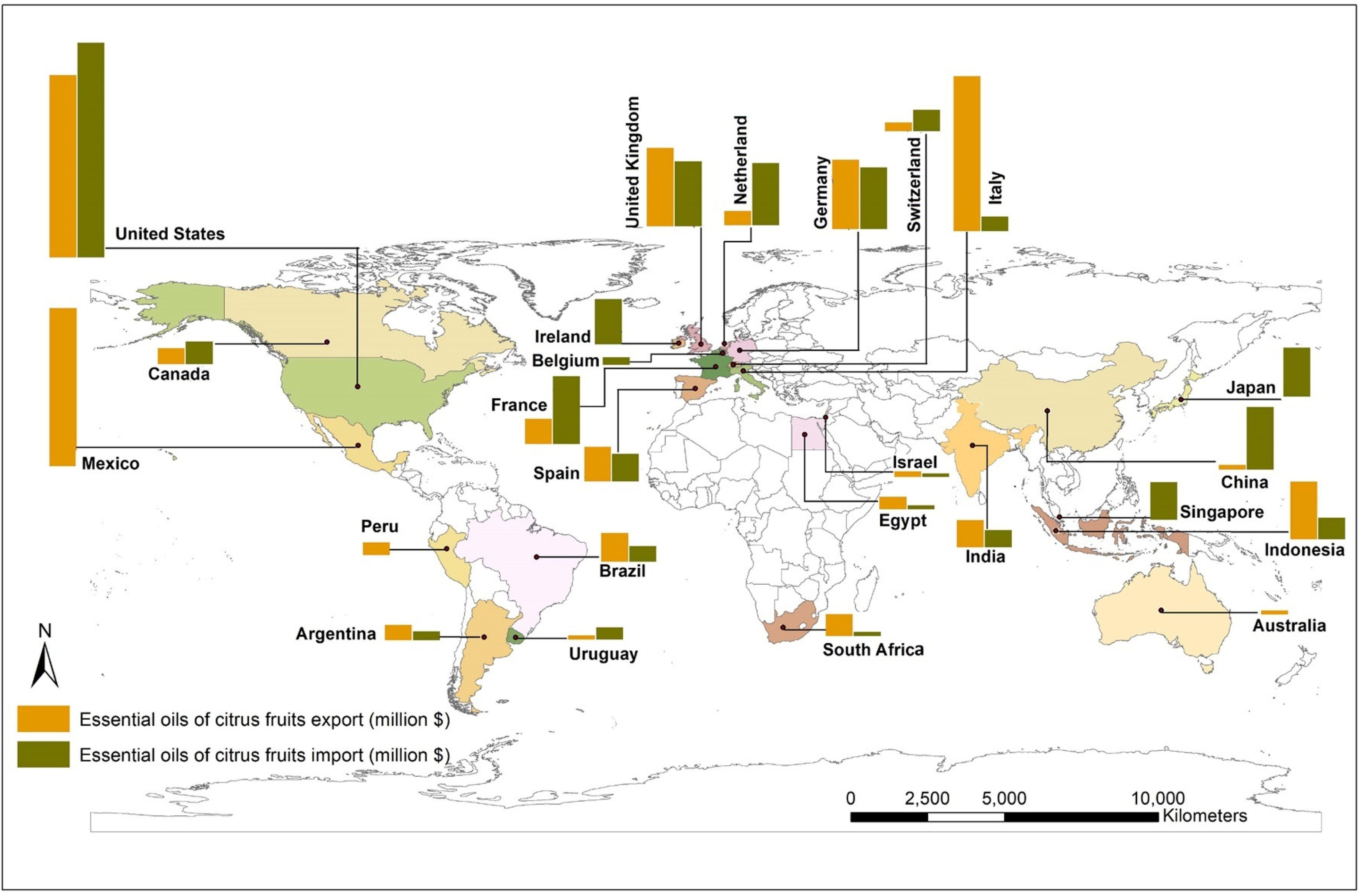
Global Production and Consumption of Citrus
2. Extraction, Characterization, and Authentication Methods for Citrus EOs
3. Components of Citrus EOs
4. Aromatherapy: Mechanisms
4.1. Evolution of Aromatherapy
4.2. Mechanism
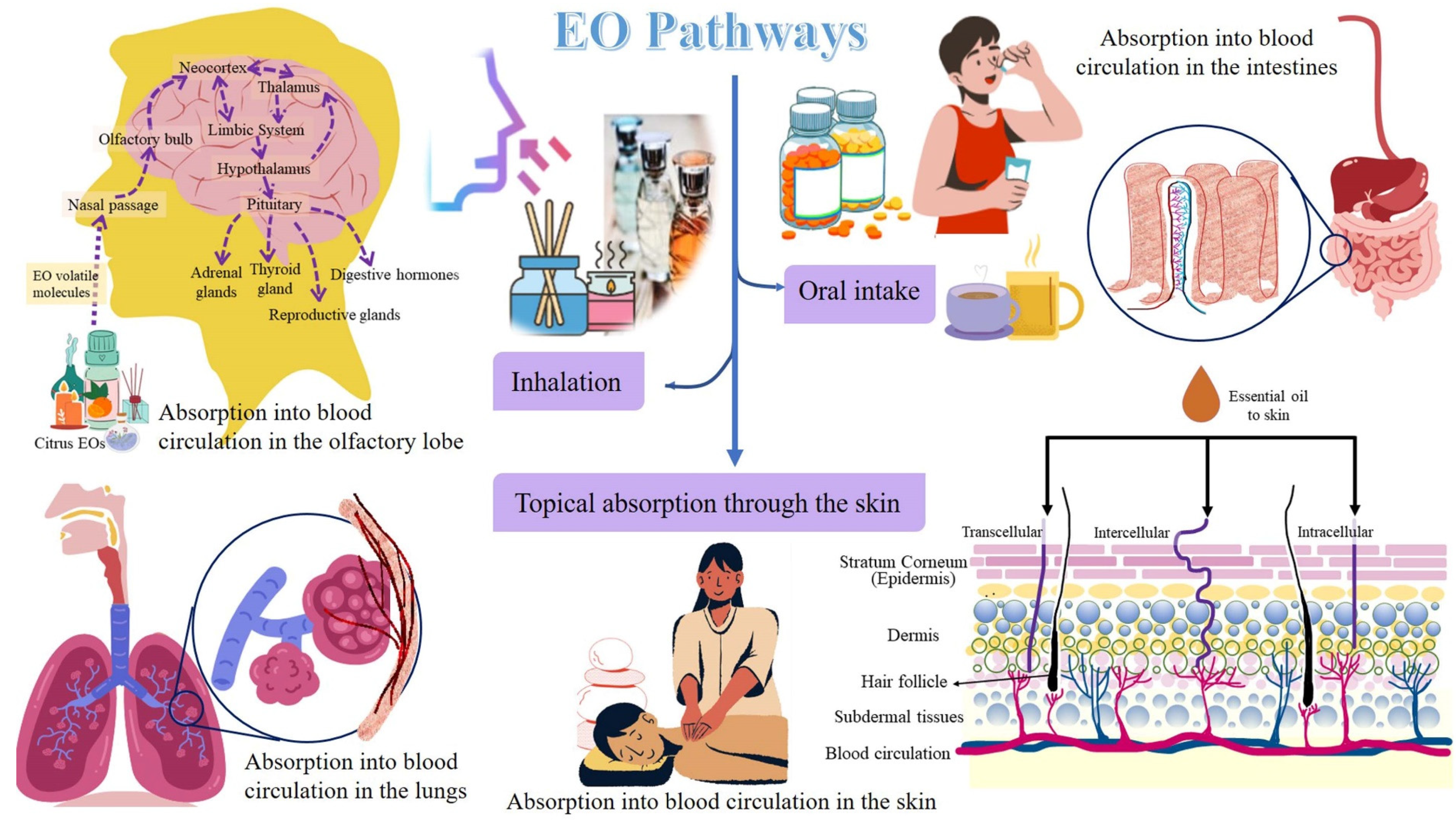
4.2.1. Inhalation
- (a)
- Activation of nasal olfactory chemoreceptors
- (b) Direct absorption of the EO active molecules into the neuronal pathway
- (c) Absorption of EO active molecules in the alveolar blood circulation
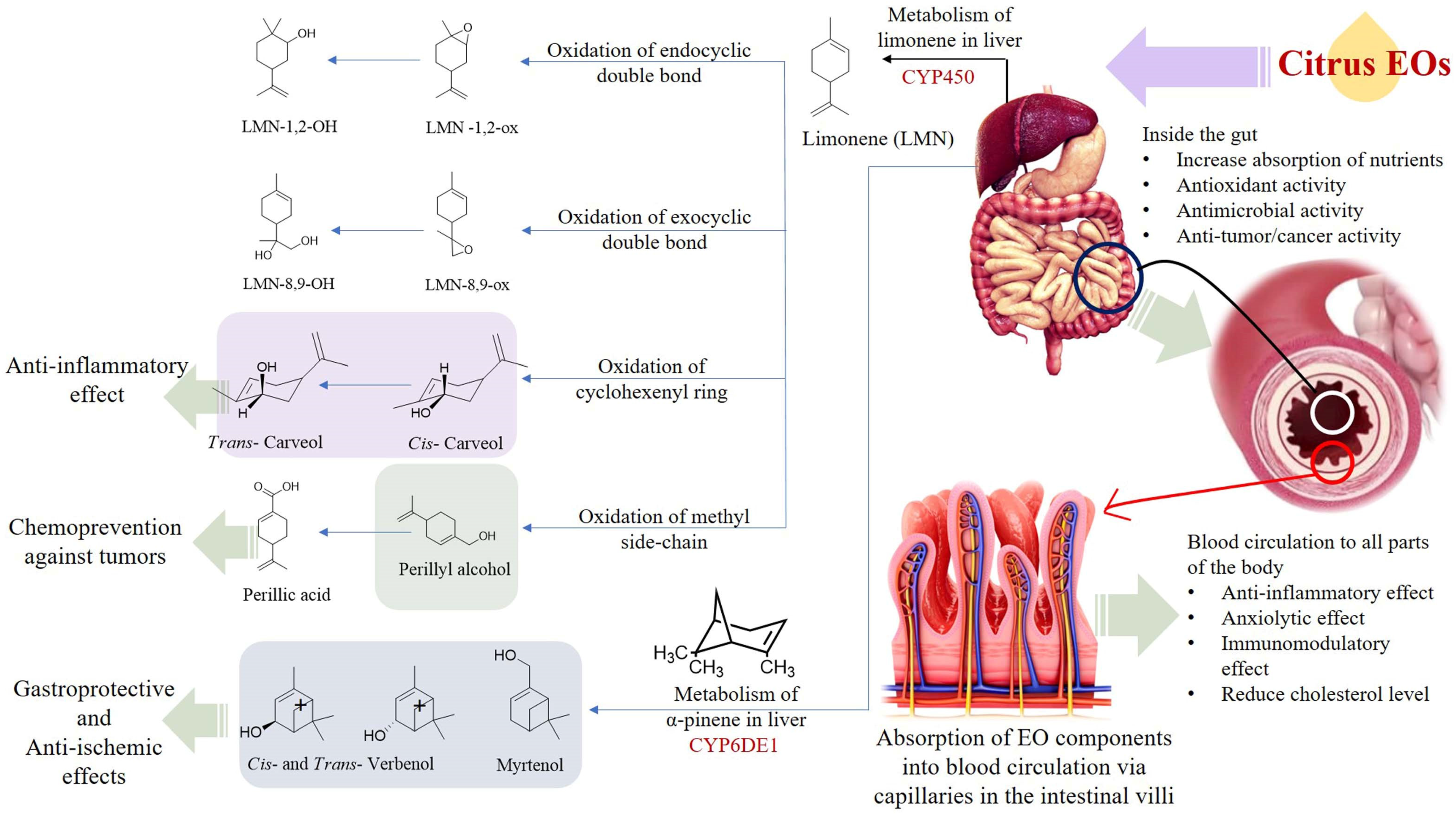
4.2.2. Oral Intake
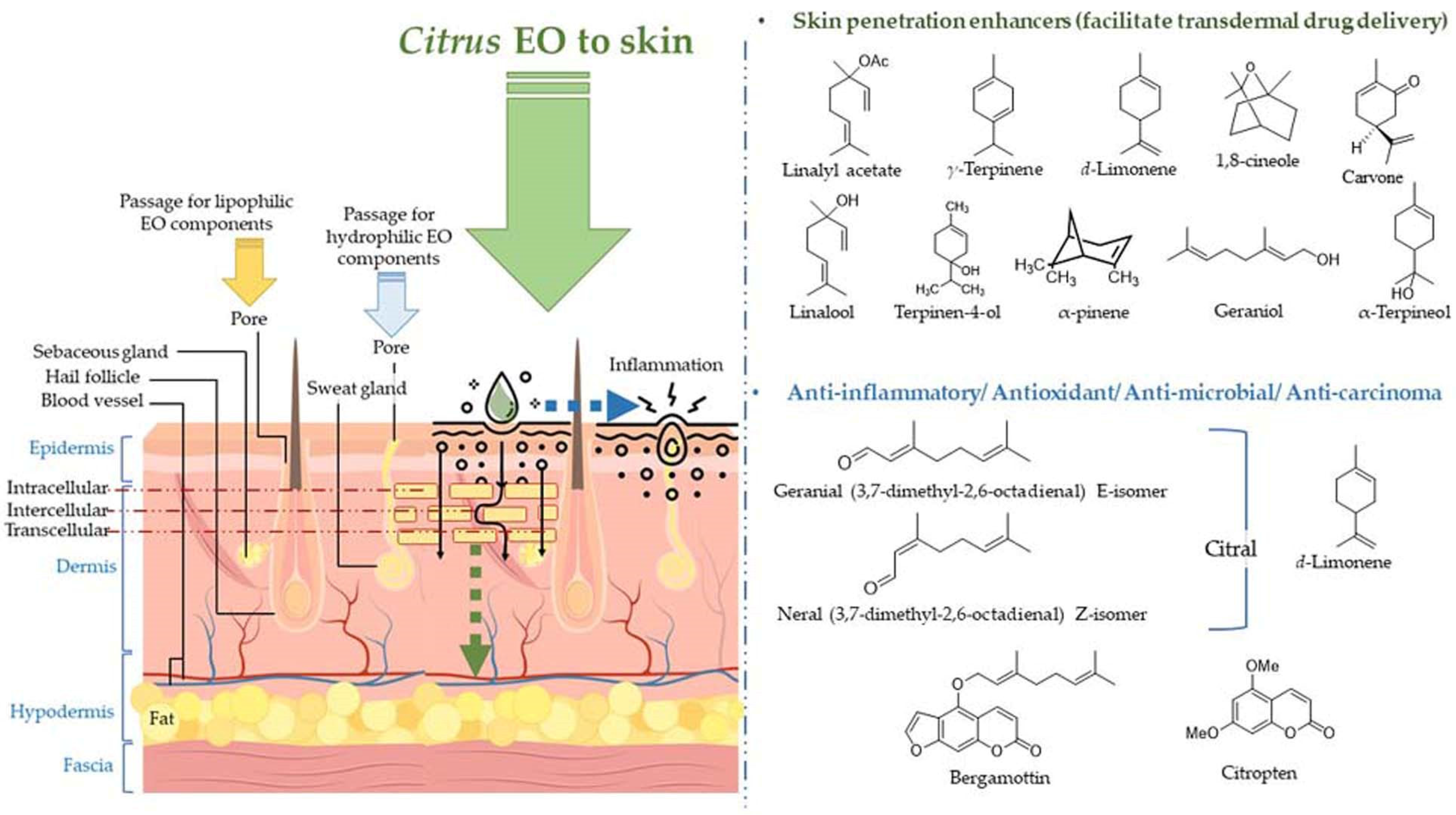
4.2.3. Applications on Skin
5. Aromatherapy Using Citrus EOs for Health and Treatment of Diseases
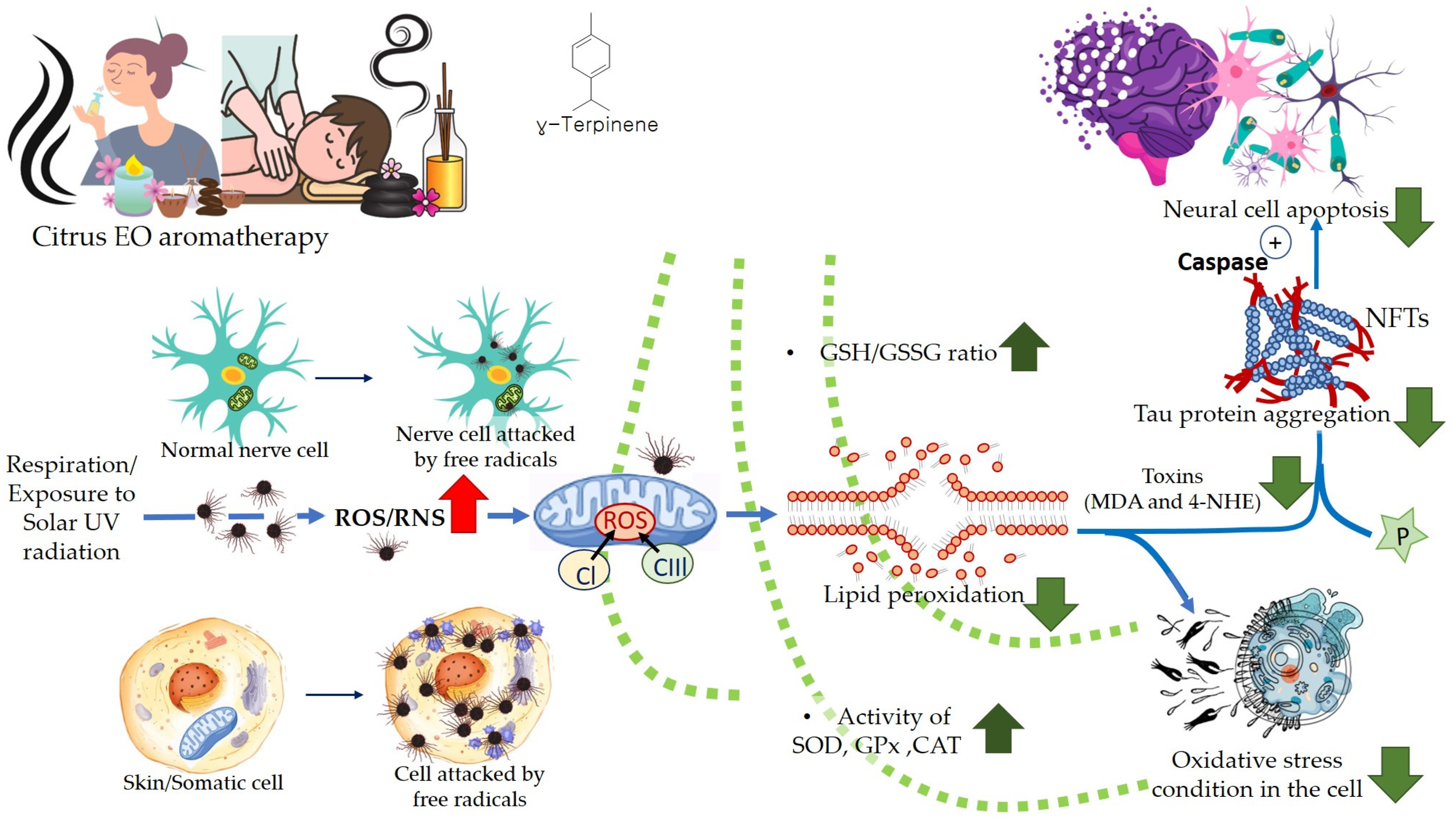
5.1. Oxidative Stress
5.2. Stress-Related Disorders/Mood Disorders
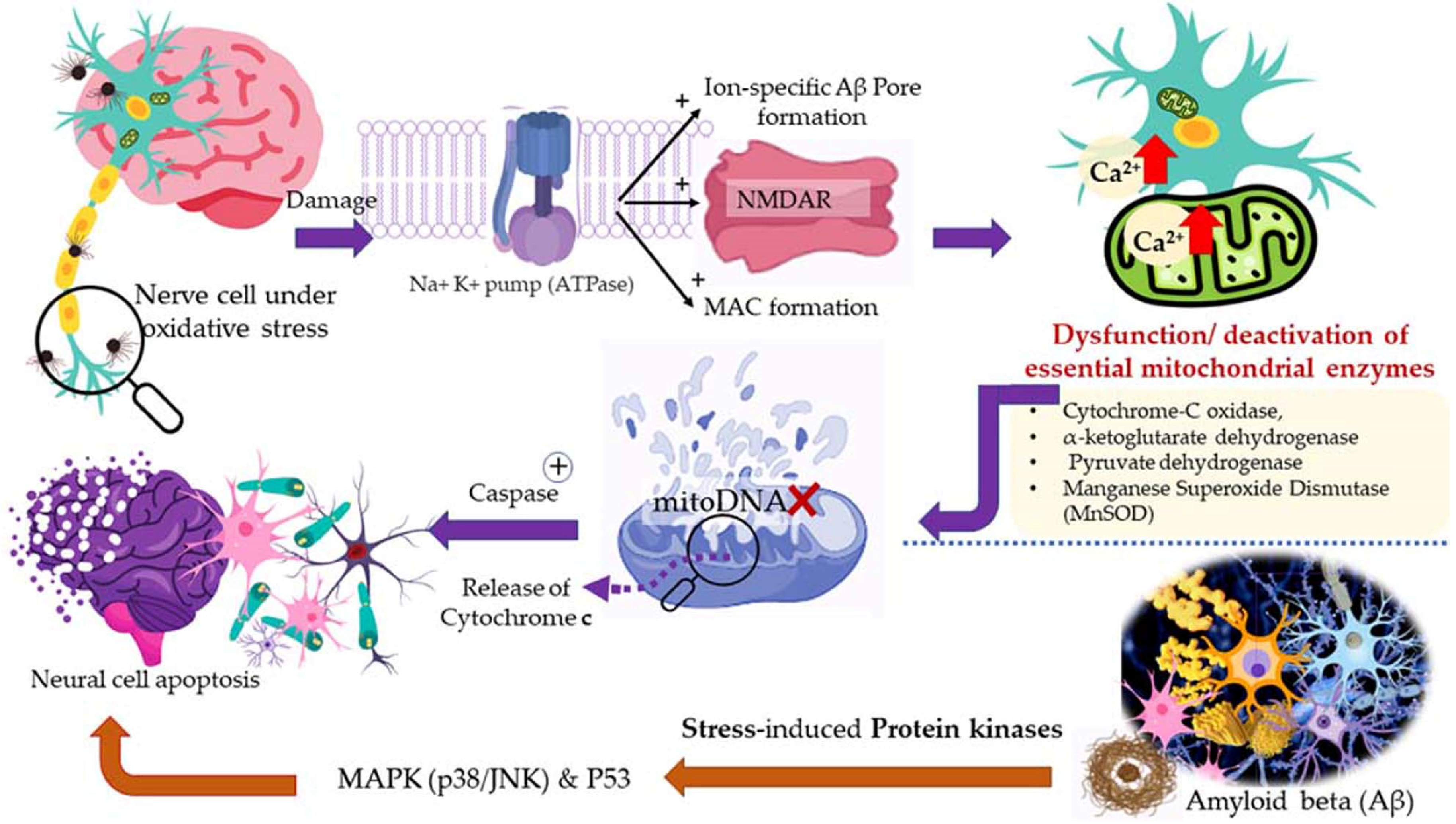
5.3. Diseased Conditions
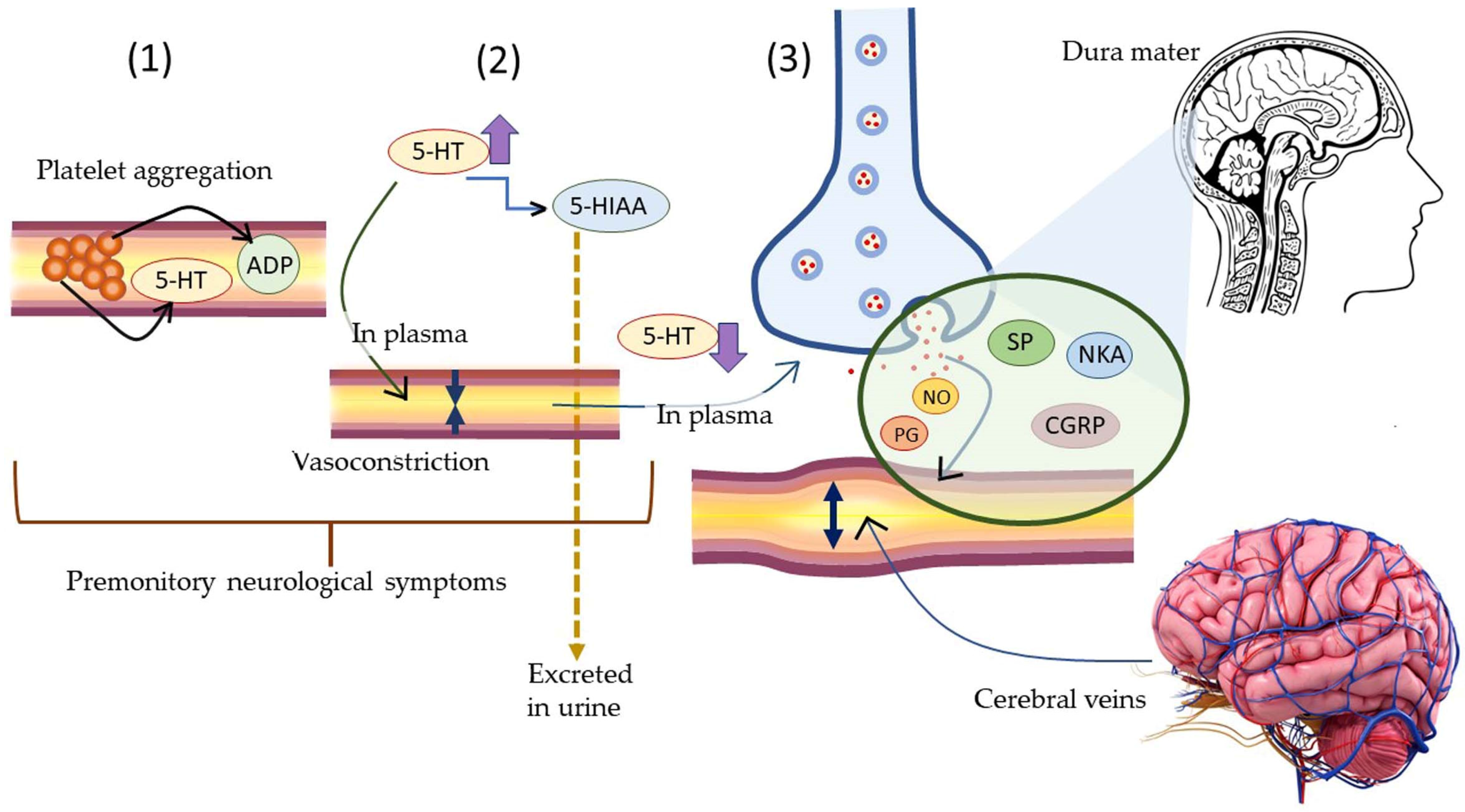
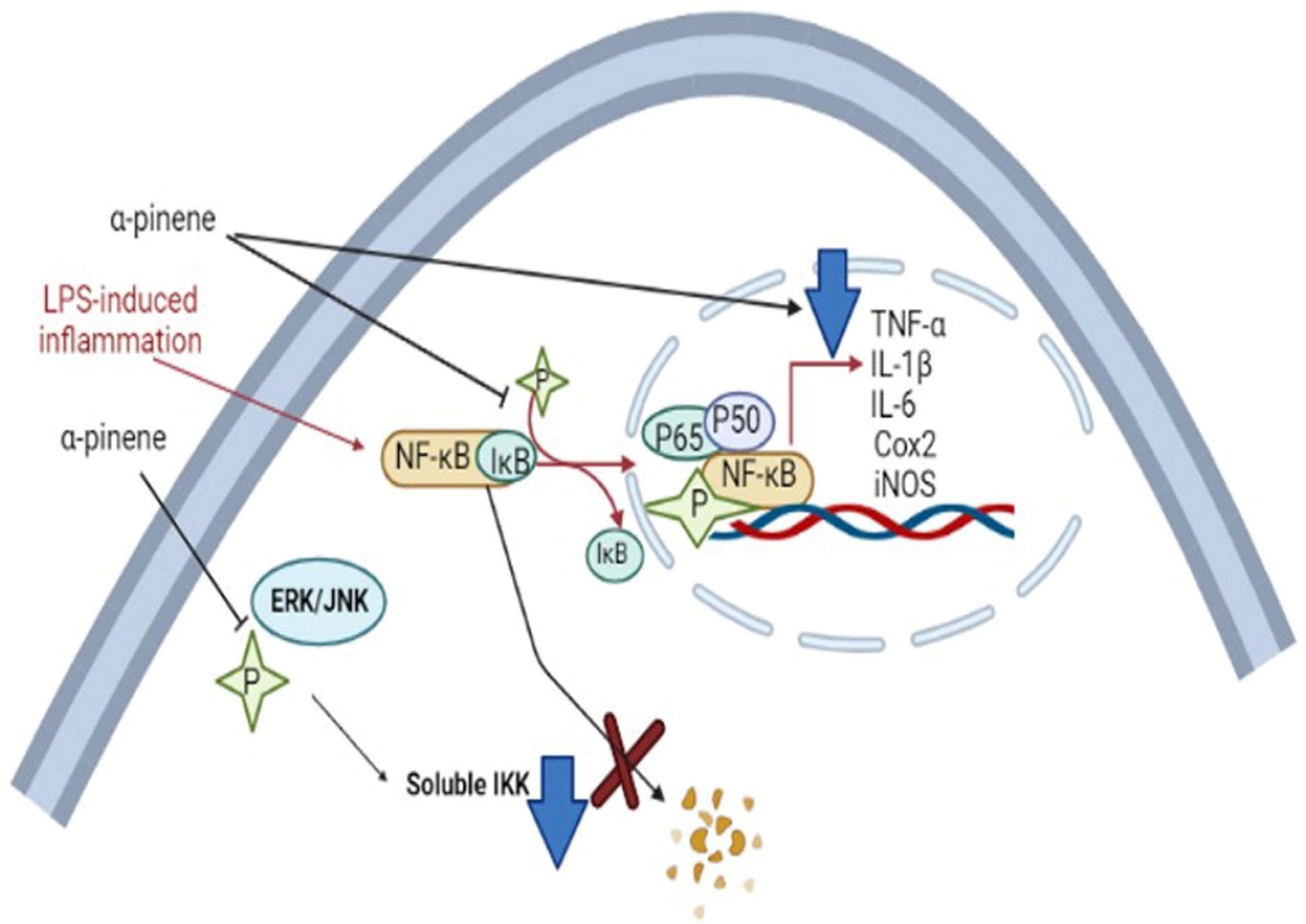
5.3.1. Neurogenic Inflammation
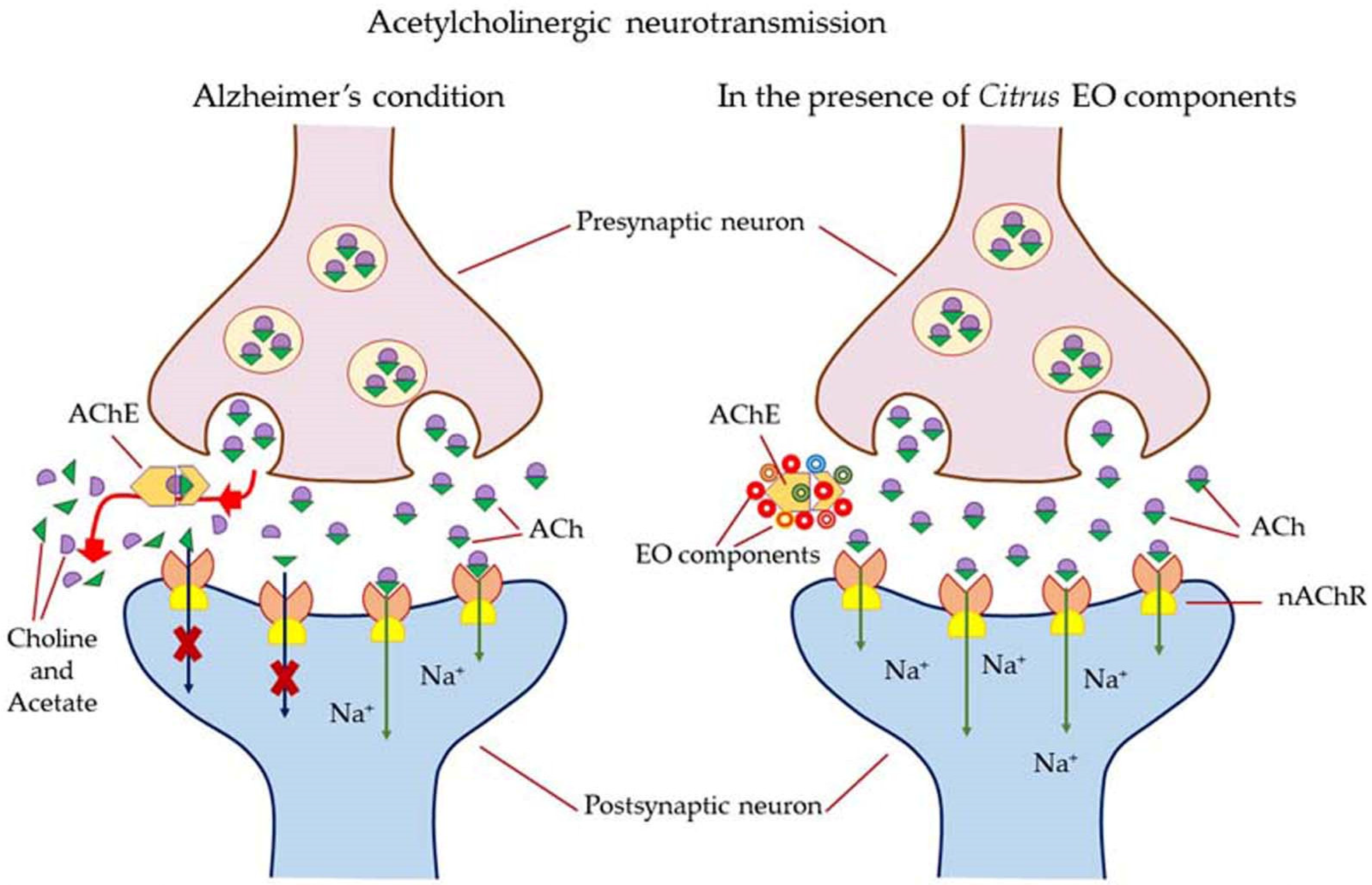
5.3.2. Dementia, Alzheimer’s Disease (AD), and Parkinson’s Disease (PD)
6. Summary
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zayed, A.; Badawy, M.T.; Farag, M.A. Valorization and extraction optimization of Citrus seeds for food and functional food applications. Food Chem. 2021, 355, 129609. [Google Scholar] [CrossRef] [PubMed]
- Fisher, K.; Phillips, C. Potential antimicrobial uses of essential oils in food: Is citrus the answer? Trends Food Sci. Technol. 2008, 19, 156–164. [Google Scholar] [CrossRef]
- Mahato, N.; Sharma, K.; Koteswararao, R.; Sinha, M.; Baral, E.R.; Cho, M.H. Citrus essential oils: Extraction, authentication and application in food preservation. Crit. Rev. Food Sci. Nutr. 2019, 59, 611–625. [Google Scholar] [CrossRef] [PubMed]
- Mahato, N.; Sinha, M.; Sharma, K.; Koteswararao, R.; Cho, M.H. Modern extraction and purification techniques for obtaining high purity food-grade bioactive compounds and value-added co-products from citrus wastes. Foods 2019, 8, 523. [Google Scholar] [CrossRef] [PubMed]
- Ferhat, M.-A.; Boukhatem, M.N.; Hazzit, M.; Meklati, B.Y.; Chemat, F. Cold pressing, hydrodistillation and microwave dry distillation of Citrus essential oil from Algeria: A comparative study. Electron. J. Biol. S 2016, 1, 30–41. [Google Scholar]
- Boughendjioua, H.; Boughendjioua, Z. Chemical composition and biological activity of essential oil of mandarin (Citrus reticulata) cultivated in Algeria. Int. J. Pharm. Sci. Rev. Res. 2017, 44, 179–184. [Google Scholar]
- Farrar, A.J.; Farrar, F.C. Clinical Aromatherapy. Nurs. Clin. N. Am. 2020, 55, 489–504. [Google Scholar] [CrossRef]
- Goepfert, M.; Liebl, P.; Herth, N.; Ciarlo, G.; Buentzel, J.; Huebner, J. Aroma oil therapy in palliative care: A pilot study with physiological parameters in conscious as well as unconscious patients. J. Cancer Res. Clin. Oncol. 2017, 143, 2123–2129. [Google Scholar] [CrossRef]
- Fernández, L.F.; Palomino, O.M.; Frutos, G. Effectiveness of Rosmarinus officinalis essential oil as antihypotensive agent in primary hypotensive patients and its influence on health-related quality of life. J. Ethnopharmacol. 2014, 151, 509–516. [Google Scholar] [CrossRef]
- Choi, S.Y.; Kang, P.; Lee, H.S.; Seol, G.H. Effects of Inhalation of Essential Oil of Citrus aurantium L. var. amara on Menopausal Symptoms, Stress, and Estrogen in Postmenopausal Women: A Randomized Controlled Trial. Evid. Based. Complement. Alternat. Med. 2014, 2014, 796518. [Google Scholar] [CrossRef]
- Doweidar, H.; El-Damrawi, G.; El-Stohy, S. Structure and properties of CdO–B2O3 and CdO–MnO–B2O3 glasses; Criteria of getting the fraction of four coordinated boron atoms from infrared spectra. Phys. B Condens. Matter 2017, 525, 137–143. [Google Scholar] [CrossRef]
- Jimbo, D.; Kimura, Y.; Taniguchi, M.; Inoue, M.; Urakami, K. Effect of aromatherapy on patients with Alzheimer’s disease. Psychogeriatrics 2009, 9, 173–179. [Google Scholar] [CrossRef]
- Matsubara, E.; Tsunetsugu, Y.; Ohira, T.; Sugiyama, M. Essential oil of Japanese cedar (Cryptomeria japonica) wood increases salivary dehydroepiandrosterone sulfate levels after monotonous work. Int. J. Environ. Res. Public Health 2017, 14, 97. [Google Scholar] [CrossRef]
- Dosoky, N.S.; Setzer, W.N. Biological activities and safety of citrus spp. Essential oils. Int. J. Mol. Sci. 2018, 19, 1966. [Google Scholar] [CrossRef]
- Lin, X.; Cao, S.; Sun, J.; Lu, D.; Zhong, B.; Chun, J. The chemical compositions, and antibacterial and antioxidant activities of four types of Citrus essential oils. Molecules 2021, 26, 3412. [Google Scholar] [CrossRef]
- Badalamenti, N.; Bruno, M.; Schicchi, R.; Geraci, A.; Leporini, M.; Gervasi, L.; Tundis, R.; Loizzo, M.R. Chemical compositions and antioxidant activities of essential oils, and their combinations, obtained from flavedo by-product of seven cultivars of Sicilian Citrus aurantium L. Molecules 2022, 27, 1580. [Google Scholar] [CrossRef]
- Zhang, L.L.; Yang, Z.Y.; Fan, G.; Ren, J.N.; Yin, K.J.; Pan, S.Y. Antidepressant-like Effect of Citrus sinensis (L.) Osbeck Essential Oil and Its Main Component Limonene on Mice. J. Agric. Food Chem. 2019, 67, 13817–13828. [Google Scholar] [CrossRef]
- Heydari, N.; Abootalebi, M.; Jamalimoghadam, N.; Kasraeian, M.; Emamghoreishi, M.; Akbarzadeh, M. Investigation of the effect of aromatherapy with Citrus aurantium blossom essential oil on premenstrual syndrome in university students: A clinical trial study. Complement. Ther. Clin. Pract. 2018, 32, 1–5. [Google Scholar] [CrossRef]
- Rombolà, L.; Scuteri, D.; Adornetto, A.; Straface, M.; Sakurada, T.; Sakurada, S.; Mizoguchi, H.; Corasaniti, M.T.; Bagetta, G.; Tonin, P.; et al. Anxiolytic-like effects of Bergamot Essential Oil are insensitive to Flumazenil in Rats. Evid.-Based Complement. Altern. Med. 2019, 2019, 2156873. [Google Scholar] [CrossRef]
- Bora, H.; Kamle, M.; Mahato, D.K.; Tiwari, P.; Kumar, P. Citrus essential oils (CEOs) and their applications in food: An overview. Plants 2020, 9, 357. [Google Scholar] [CrossRef]
- Dima, C.; Dima, S. Essential oils in foods: Extraction, stabilization, and toxicity. Curr. Opin. Food Sci. 2015, 5, 29–35. [Google Scholar] [CrossRef]
- Global Citrus Oil Market: By Origin: Organic, Conventional; By Oil Type: Orange, Lemon, Lime, and Bergamot Oil, Others; By Application: Food and Beverage, Cosmetic and Personal Care, Pharmaceuticals, Others; Regional Analysis; Historical Market and Forecast (2018–2028); Competitive Landscape; Industry Events and Developments; 2022. Available online: https://www.expertmarketresearch.com/reports/citrus-oil-market (accessed on 22 October 2022).
- Shahbandeh, M. World Production of Citrus Fruits in 2020, by Region (in Thousand Metric Tons). 2022. Available online: https://www.statista.com/statistics/264002/production-of-citrus-fruits-worldwide-by-region/ (accessed on 22 October 2022).
- Citrus Oil Market by Product Types (Orange Oil, Lemon Lime Oil, Others), by Application (Food & Beverages, Personal Care, Cosmetics, Aromatherapy & Others), by Extraction Method, by Application, by Sales Channel & By Region. Available online: https://www.factmr.com/report/279/citrus-oil-market (accessed on 22 October 2022).
- Global And United States Citrus Oil Market Report & Forecast 2022–2028. 2022. Available online: https://www.industryresearch.biz/global-and-united-states-citrus-oil-market-20679913 (accessed on 22 October 2022).
- Simoes, A.J.G.; Hidalgo, C.A. The economic complexity observatory: An analytical tool for understanding the dynamics of economic development. In Proceedings of the Workshops at the Twenty-Fifth AAAI Conference on Artificial Intelligence, San Francisco, CA, USA, 7–8 August 2011; pp. 39–42. [Google Scholar]
- Bousbia, N.; Vian, M.A.; Ferhat, M.A.; Meklati, B.Y.; Chemat, F. A new process for extraction of essential oil from Citrus peels: Microwave hydrodiffusion and gravity. J. Food Eng. 2009, 90, 409–413. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Saito, M. Fractionation of lemon-peel oil by semi-preparative supercritical fluid chromatography. J. Chromatogr. A 1990, 505, 237–246. [Google Scholar] [CrossRef]
- Aresta, A.; Cotugno, P.; De Vietro, N.; Massari, F.; Zambonin, C. Determination of polyphenols and vitamins in wine-making by-products by supercritical fluid extraction (SFE). Anal. Lett. 2020, 53, 2585–2595. [Google Scholar] [CrossRef]
- Yousefi, M.; Rahimi-Nasrabadi, M.; Pourmortazavi, S.M.; Wysokowski, M.; Jesionowski, T.; Ehrlich, H.; Mirsadeghi, S. Supercritical fluid extraction of essential oils. TrAC—Trends Anal. Chem. 2019, 118, 182–193. [Google Scholar] [CrossRef]
- Rajput, S.; Kaur, S.; Panesar, P.S.; Thakur, A. Supercritical fluid extraction of essential oils from Citrus reticulata peels: Optimization and characterization studies. Biomass Convers. Biorefinery 2022, 1–10. [Google Scholar] [CrossRef]
- Rubiolo, P.; Sgorbini, B.; Liberto, E.; Cordero, C.; Bicchi, C. Essential oils and volatiles: Sample preparation and analysis. A review. Flavour Fragr. J. 2010, 25, 282–290. [Google Scholar] [CrossRef]
- Brinkman, U.A.T. Hyphenation: Hype and Fascination. J. Chromatogr. 1999, 856, 535. [Google Scholar]
- Hong, J.H.; Khan, N.; Jamila, N.; Hong, Y.S.; Nho, E.Y.; Choi, J.Y.; Lee, C.M.; Kim, K.S. Determination of volatile flavour profiles of Citrus spp. Fruits by SDE-GC–MS and enantiomeric composition of chiral compounds by MDGC–MS. Phytochem. Anal. 2017, 28, 392–403. [Google Scholar] [CrossRef]
- Bonaccorsi, I.; Sciarrone, D.; Schipilliti, L.; Dugo, P.; Mondello, L.; Dugo, G. Multidimensional enantio gas chromtography/mass spectrometry and gas chromatography-combustion-isotopic ratio mass spectrometry for the authenticity assessment of Lime essential oils (C. aurantifolia Swingle and C. latifolia Tanaka). J. Chromatogr. A 2012, 1226, 87–95. [Google Scholar] [CrossRef]
- Schipilliti, L.; Bonaccorsi, I.; Cotroneo, A.; Dugo, P.; Mondello, L. Evaluation of gas chromatography-combustion-isotope ratio mass spectrometry (GC-C-IRMS) for the quality assessment of Citrus liqueurs. J. Agric. Food Chem. 2013, 61, 1661–1670. [Google Scholar] [CrossRef]
- Marti, G.; Boccard, J.; Mehl, F.; Debrus, B.; Marcourt, L.; Merle, P.; Delort, E.; Baroux, L.; Sommer, H.; Rudaz, S.; et al. Comprehensive profiling and marker identification in non-volatile citrus oil residues by mass spectrometry and nuclear magnetic resonance. Food Chem. 2014, 150, 235–245. [Google Scholar] [CrossRef]
- González-Mas, M.C.; Rambla, J.L.; López-Gresa, M.P.; Amparo Blázquez, M.; Granell, A. Volatile compounds in citrus essential oils: A comprehensive review. Front. Plant Sci. 2019, 10, 12. [Google Scholar] [CrossRef]
- Tranchida, P.Q.; Bonaccorsi, I.; Dugo, P.; Mondello, L.; Dugo, G. Analysis of Citrus essential oils: State of the art and future perspectives. A review. Flavour Fragr. J. 2012, 27, 98–123. [Google Scholar] [CrossRef]
- Palazzolo, E.; Armando Laudicina, V.; Antonietta Germanà, M. Current and potential use of Citrus Essential Oils. Curr. Org. Chem. 2013, 17, 3042–3049. [Google Scholar] [CrossRef]
- Md Othman, S.; Hassan, M.; Nahar, L.; Basar, N.; Jamil, S.; Sarker, S. Essential Oils from the Malaysian Citrus (Rutaceae) Medicinal Plants. Medicines 2016, 3, 13. [Google Scholar] [CrossRef]
- Bonaccorsi, I.; Sciarrone, D.; Cotroneo, A.; Mondello, L.; Dugo, P.; Dugo, G. Enantiomeric distribution of key volatile components in Citrus essential oils. Rev. Bras. Farmacogn. 2011, 21, 841–849. [Google Scholar] [CrossRef]
- Sarrou, E.; Chatzopoulou, P.; Dimassi-Theriou, K.; Therios, I. Volatile constituents and antioxidant activity of peel, flowers and leaf oils of Citrus aurantium L. growing in Greece. Molecules 2013, 18, 10639–10647. [Google Scholar] [CrossRef]
- Svoboda, K.P.; Greenaway, R.I. Lemon scented plants. Int. J. Aromather. 2003, 13, 23–32. [Google Scholar] [CrossRef]
- Modzelewska, A.; Sur, S.; Kumar, S.K.; Khan, S.R. Sesquiterpenes: Natural products that decrease cancer growth. Curr. Med. Chem.—Anti-Cancer Agents 2005, 5, 477–499. [Google Scholar] [CrossRef]
- Perry, N.; Perry, E. Aromatherapy in the management of psychiatric disorders. CNS Drugs 2006, 20, 257–280. [Google Scholar] [CrossRef]
- Lillehei, A.S.; Halcon, L.L. A systematic review of the effect of inhaled essential oils on sleep. J. Altern. Complement. Med. 2014, 20, 441–451. [Google Scholar] [CrossRef]
- Shiina, Y.; Funabashi, N.; Lee, K.; Toyoda, T.; Sekine, T.; Honjo, S.; Hasegawa, R.; Kawata, T.; Wakatsuki, Y.; Hayashi, S.; et al. Relaxation effects of lavender aromatherapy improve coronary flow velocity reserve in healthy men evaluated by transthoracic Doppler echocardiography. Int. J. Cardiol. 2008, 129, 193–197. [Google Scholar] [CrossRef]
- Kotlik, B.A.; Petrova, L.M.; Red’ko, A.I.; EIu, K. Sources of equipment measurement error in stimulation electromyography. Fiziol. Cheloveka 1986, 12, 691–694. [Google Scholar]
- Zimmerman, W.B. Sleep mentation and auditory awakening thresholds. Psychophysiology 1970, 6, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Momjian, S.; Tyrand, R.; Landis, B.N.; Boëx, C. Intraoperative monitoring of olfactory function: A feasibility study. J. Neurosurg. 2019, 132, 1659–1664. [Google Scholar] [CrossRef] [PubMed]
- Marciniak-Firadza, R. The Sense of Smell in Logopaedic Theory and Practice. Logop. Lodz. 2021, 5, 123–145. [Google Scholar] [CrossRef]
- Naidich, T.P.; Som, P.M. The Olfactory System: Part III: Central processing of olfaction and central olfactory dysfunction. Neurographics 2018, 8, 298–316. [Google Scholar] [CrossRef]
- Loos, H.M.; Schreiner, L.; Karacan, B. A systematic review of physiological responses to odours with a focus on current methods used in event-related study designs. Int. J. Psychophysiol. 2020, 158, 143–157. [Google Scholar] [CrossRef]
- Liao, W.-C.; Yao, R.-A.; Chen, L.-Y.; Renn, T.-Y.; Klimenkov, I.V.; Sudakov, N.P.; Mai, F.-D.; Chen, Y.-T.; Chang, H.-M. Olfactory stimulation successfully modulates the neurochemical, biochemical and behavioral phenotypes of the visceral pain. Molecules 2022, 27, 7659. [Google Scholar] [CrossRef]
- Llorens, J. The physiology of taste and smell: How and why we sense flavors. Water Sci. Technol. 2004, 49, 1–10. [Google Scholar] [CrossRef]
- Lee, A.J.; Ling, R.S. Improved cementing techniques. Instr. Course Lect. 1981, 30, 407–413. [Google Scholar]
- Fung, T.K.H.; Lau, B.W.M.; Ngai, S.P.C.; Tsang, H.W.H. Therapeutic effect and mechanisms of essential oils in mood disorders: Interaction between the nervous and respiratory systems. Int. J. Mol. Sci. 2021, 22, 4844. [Google Scholar] [CrossRef]
- Fang, T.; Goto, M.; Sasaki, M.; Hirose, T. Combination of supercritical CO2 and vacuum distillation for the fractionation of bergamot oil. J. Agric. Food Chem. 2004, 52, 5162–5167. [Google Scholar] [CrossRef]
- Reverchon, E.; Iacuzio, G. Supercritical desorption of bergamot peel oil from silica gel—Experiments and mathematical modelling. Chem. Eng. Sci. 1997, 52, 3553–3559. [Google Scholar] [CrossRef]
- Adokoh, C.K.; Asante, D.B.; Acheampong, D.O.; Kotsuchibashi, Y.; Armah, F.A.; Sirikyi, I.H.; Kimura, K.; Gmakame, E.; Abdul-Rauf, S. Chemical profile and in vivo toxicity evaluation of unripe Citrus aurantifolia essential oil. Toxicol. Rep. 2019, 6, 692–702. [Google Scholar] [CrossRef]
- Smith, C.A.; Collins, C.T.; Crowther, C.A. Aromatherapy for pain management in labour. Cochrane Database Syst. Rev. 2011, 6, CD009215. [Google Scholar] [CrossRef]
- George, A.; Sanjay, M.R.; Srisuk, R.; Parameswaranpillai, J.; Siengchin, S. A comprehensive review on chemical properties and applications of biopolymers and their composites. Int. J. Biol. Macromol. 2020, 154, 329–338. [Google Scholar] [CrossRef]
- Lai, T.K.T.; Cheung, M.C.; Lo, C.K.; Ng, K.L.; Fung, Y.H.; Tong, M.; Yau, C.C. Effectiveness of aroma massage on advanced cancer patients with constipation: A pilot study. Complement. Ther. Clin. Pract. 2011, 17, 37–43. [Google Scholar] [CrossRef]
- Steps, S.B. An Introductory Guide to 1000′s of Uses for Essential Oils. Available online: http://www.sustainablebabysteps.com/uses-for-essential-oils.html (accessed on 14 October 2022).
- Mollace, V.; Sacco, I.; Janda, E.; Malara, C.; Ventrice, D.; Colica, C.; Visalli, V.; Muscoli, S.; Ragusa, S.; Muscoli, C. Hypolipemic and hypoglycaemic activity of bergamot polyphenols: From animal models to human studies. Fitoterapia 2011, 82, 309–316. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Bruschetta, G.; Di Paola, R.; Ahmad, A.; Campolo, M.; Cuzzocrea, S.; Esposito, E.; Navarra, M. The anti-inflammatory and antioxidant effects of bergamot juice extract (BJe) in an experimental model of inflammatory bowel disease. Clin. Nutr. 2015, 34, 1146–1154. [Google Scholar] [CrossRef] [PubMed]
- Delle Monache, S.; Sanità, P.; Trapasso, E.; Ursino, M.R.; Dugo, P.; Russo, M.; Ferlazzo, N.; Calapai, G.; Angelucci, A.; Navarra, M. Mechanisms underlying the anti-tumoral effects of Citrus bergamia juice. PLoS ONE 2013, 8, e61484. [Google Scholar] [CrossRef] [PubMed]
- Navarra, M.; Ursino, M.R.; Ferlazzo, N.; Russo, M.; Schumacher, U.; Valentiner, U. Effect of Citrus bergamia juice on human neuroblastoma cells in vitro and in metastatic xenograft models. Fitoterapia 2014, 95, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Visalli, G.; Ferlazzo, N.; Cirmi, S.; Campiglia, P.; Gangemi, S.; Di Pietro, A.; Calapai, G.; Navarra, M. Bergamot juice extract inhibits proliferation by inducing apoptosis in human colon cancer cells. Anticancer. Agents Med. Chem. 2014, 14, 1402–1413. [Google Scholar] [CrossRef] [PubMed]
- Pendino, G.M. Il bergamotto in terapia medica: Attualità e prospettive. Essenze Deriv. Agrum. 1998, 68, 57–62. [Google Scholar]
- Fisher, K.; Phillips, C.A. The effect of Lemon, Orange and Bergamot essential oils and their components on the survival of Campylobacter jejuni, Escherichia coli O157, Listeria monocytogenes, Bacillus cereus and Staphylococcus aureus in vitro and in food systems. J. Appl. Microbiol. 2006, 101, 1232–1240. [Google Scholar] [CrossRef]
- Karaca, M.; Özbek, H.; Him, A.; Tütüncü, M.; Akkan, H.A.; Kaplanoǧlu, V. Investigation of anti-inflammatory activity of bergamot oil. Eur. J. Gen. Med. 2007, 4, 176–179. [Google Scholar]
- Sanguinetti, M.; Posteraro, B.; Romano, L.; Battaglia, F.; Lopizzo, T.; De Carolis, E.; Fadda, G. In vitro activity of Citrus bergamia (bergamot) oil against clinical isolates of dermatophytes. J. Antimicrob. Chemother. 2007, 59, 305–308. [Google Scholar] [CrossRef]
- Romano, L.; Battaglia, F.; Masucci, L.; Sanguinetti, M.; Posteraro, B.; Plotti, G.; Zanetti, S.; Fadda, G. In vitro activity of bergamot natural essence and furocoumarin-free and distilled extracts, and their associations with boric acid, against clinical yeast isolates. J. Antimicrob. Chemother. 2005, 55, 110–114. [Google Scholar] [CrossRef]
- Sánchez-González, L.; Cháfer, M.; Chiralt, A.; González-Martínez, C. Physical properties of edible chitosan films containing bergamot essential oil and their inhibitory action on Penicillium italicum. Carbohydr. Polym. 2010, 82, 277–283. [Google Scholar] [CrossRef]
- Occhiuto, F.; Circosta, C. Antianginal and antiarrhythmic effects of bergamottine, a furocoumarin isolated from Bergamot oil. Phyther. Res. 1996, 10, 491–496. [Google Scholar] [CrossRef]
- Kang, P.; Suh, S.H.; Min, S.S.; Seol, G.H. The essential oil of Citrus bergamia Risso induces vasorelaxation of the mouse aorta by activating K+ channels and inhibiting Ca2+ influx. J. Pharm. Pharmacol. 2013, 65, 745–749. [Google Scholar] [CrossRef]
- You, T.; Arsenis, N.C.; Disanzo, B.L.; LaMonte, M.J. Effects of exercise training on chronic inflammation in obesity. Sports Med. 2013, 43, 243–256. [Google Scholar] [CrossRef]
- Mannucci, C.; Navarra, M.; Calapai, F.; Squeri, R.; Gangemi, S.; Calapai, G. Clinical Pharmacology of Citrus bergamia: A Systematic Review. Phyther. Res. 2017, 31, 27–39. [Google Scholar] [CrossRef]
- Karn, A.; Zhao, C.; Yang, F.; Cui, J.; Gao, Z.; Wang, F.; Xiao, H.; Zheng, J. In-vivo biotransformation of citrus functional components and their effects on health. Crit. Rev. Food Sci. Nutr. 2021, 61, 756–776. [Google Scholar] [CrossRef]
- Schmidt, L.; Göen, T. Human metabolism of α-pinene and metabolite kinetics after oral administration. Arch. Toxicol. 2017, 91, 677–687. [Google Scholar] [CrossRef]
- Igimi, H.; Nishimura, M.; Kodama, R.; Ide, H. Studies on the metabolism of d-limonene (p-mentha-1, 8-diene): I. the absorption, distribution and excretion of d-limonene in rats. Xenobiotica 1974, 4, 77–84. [Google Scholar] [CrossRef]
- Chen, T.C.; Da Fonseca, C.O.; Schönthal, A.H. Preclinical development and clinical use of perillyl alcohol for chemoprevention and cancer therapy. Am. J. Cancer Res. 2015, 5, 1580. [Google Scholar]
- Jayaprakasha, G.K.; Murthy, K.N.C.; Uckoo, R.M.; Patil, B.S. Chemical composition of volatile oil from Citrus limettioides and their inhibition of colon cancer cell proliferation. Ind. Crop. Prod. 2013, 45, 200–207. [Google Scholar] [CrossRef]
- Elegbede, J.A.; Maltzman, T.H.; Verma, A.K.; Tanner, M.A.; Elson, C.E.; Gould, M.N. Mouse skin tumor promoting activity of orange peel oil and d-limonene: A re-evaluation. Carcinogenesis 1986, 7, 2047–2049. [Google Scholar] [CrossRef]
- Guo, P.; Li, P.; Zhang, X.; Liu, N.; Wang, J.; Yang, S.; Yu, L.; Zhang, W. The effectiveness of aromatherapy on preoperative anxiety in adults: A systematic review and meta-analysis of randomized controlled trials. Int. J. Nurs. Stud. 2020, 111, 103747. [Google Scholar] [CrossRef] [PubMed]
- Patil, J.R.; Jayaprakasha, G.K.; Murthy, K.N.C.; Tichy, S.E.; Chetti, M.B.; Patil, B.S. Apoptosis-mediated proliferation inhibition of human colon cancer cells by volatile principles of Citrus aurantifolia. Food Chem. 2009, 114, 1351–1358. [Google Scholar] [CrossRef]
- Bardon, S.; Foussard, V.; Fournel, S.; Loubat, A. Monoterpenes inhibit proliferation of human colon cancer cells by modulating cell cycle-related protein expression. Cancer Lett. 2002, 181, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Karlson, J.; Borg-Karlson, A.-K.; Unelius, R.; Shoshan, M.C.; Wilking, N.; Ringborg, U.; Linder, S. Inhibition of tumor cell growth by monoterpenes in vitro: Evidence of a Ras-independent mechanism of action. Anticancer. Drugs 1996, 7, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Feng, K.; Zhu, X.; Liu, G.; Kan, Q.; Chen, T.; Chen, Y.; Cao, Y. Dietary citrus peel essential oil ameliorates hypercholesterolemia and hepatic steatosis by modulating lipid and cholesterol homeostasis. Food Funct. 2020, 11, 7217–7230. [Google Scholar] [CrossRef]
- Zhang, Y.; Gong, J.; Yu, H.; Guo, Q.; Defelice, C.; Hernandez, M.; Yin, Y.; Wang, Q. Alginate-whey protein dry powder optimized for target delivery of essential oils to the intestine of chickens. Poult. Sci. 2014, 93, 2514–2525. [Google Scholar] [CrossRef]
- Marques, F.M.; Figueira, M.M.; Schmitt, E.F.P.; Kondratyuk, T.P.; Endringer, D.C.; Scherer, R.; Fronza, M. In vitro anti-inflammatory activity of terpenes via suppression of superoxide and nitric oxide generation and the NF-κB signalling pathway. Inflammopharmacology 2019, 27, 281–289. [Google Scholar] [CrossRef]
- Price, L. How essential oils enter the body. In Aromatherapy for Health Professionals, 4th ed.; Price, S., Price, L., Eds.; Churchill Livingstone/Elsevier: Edinburgh, Scotland, 2012; pp. 125–139. ISBN 9780702035647. [Google Scholar]
- Buchbauer, G. Molecular interaction: Biological effects and modes of action of essential oils. Int. J. Aromather. 1993, 5, 11–14. [Google Scholar]
- Parker, R.A.; Gabriel, K.T.; Graham, K.D.; Butts, B.K.; Cornelison, C.T. Antifungal activity of select essential oils against Candida auris and Their interactions with antifungal drugs. Pathogens 2022, 11, 821. [Google Scholar] [CrossRef]
- Shutes, J.; Galper, A. The Aromatherapy Companion: A Portable Guide to Blending Essential Oils and Crafting Remedies for Body, Mind, and Spirit; Shutes, J., Galper, A., Eds.; Fair Winds Press: Beverly, MA, USA, 2022; ISBN 9780760377925. [Google Scholar]
- Scuteri, D.; Watanabe, C.; Sakurada, S.; Hamamura, K.; Sakurada, T.; Tonin, P.; Bagetta, G.; Corasaniti, M.T. Pharmacotechnological Advances for clinical translation of essential oils for the treatment of pain and agitation in severe dementia. Processes 2022, 10, 1340. [Google Scholar] [CrossRef]
- Mohammed, M.; Badeggi, U.M.; Mathew, J.T.; Umar, T.M. Therapeutic benefits of selected plant herbs with essential oils. Lapai J. Sci. Technol. 2021, 7, 158–168. [Google Scholar]
- Wilkinson, S.M.; Love, S.B.; Westcombe, A.M.; Gambles, M.A.; Burgess, C.C.; Cargill, A.; Young, T.; Maher, E.J.; Ramirez, A.J. Effectiveness of aromatherapy massage in the management of anxiety and depression in patients with cancer: A multicenter randomized controlled trial. J. Clin. Oncol. 2007, 25, 532–539. [Google Scholar] [CrossRef]
- Halcón, L.L. Aromatherapy: Therapeutic applications of plant essential oils. Minn. Med. 2002, 85, 42–46. [Google Scholar]
- Viana, M.D.M.; Cardoso, R.M.; Silva, N.K.G.T.; Falcão, M.A.P.; Vieira, A.C.S.; Alexandre-Moreira, M.S.; Campesatto, E.A. Anxiolytic-like effect of Citrus limon (L.) Burm f. essential oil inhalation on mice. Rev. Bras. Plantas Med. 2016, 18, 96–104. [Google Scholar] [CrossRef]
- Chen, M.; Chen, Y.; Lee, H. The effect of bergamot essential oil aromatherapy on improving depressive mood and sleep quality in postpartum women: A randomized controlled trial. J. Nurs. Res. 2022, 30, e201. [Google Scholar] [CrossRef]
- Igarashi, M.; Ikei, H.; Song, C.; Miyazaki, Y. Effects of olfactory stimulation with rose and orange oil on prefrontal cortex activity. Complement. Ther. Med. 2014, 22, 1027–1031. [Google Scholar] [CrossRef]
- Keyhanmehr, A.S.; Kolouri, S.; Heydarirad, G.; Mofid, B.; Mosavat, S.H. Aromatherapy for the management of cancer complications: A narrative review. Complement. Ther. Clin. Pract. 2018, 31, 175–180. [Google Scholar] [CrossRef]
- Dyer, J.; Cleary, L.; Ragsdale-Lowe, M.; McNeill, S.; Osland, C. The use of aromasticks at a cancer centre: A retrospective audit. Complement. Ther. Clin. Pract. 2014, 20, 203–206. [Google Scholar] [CrossRef]
- Kliszcz, A.; Danel, A.; Puła, J.; Barabasz-Krasny, B.; Możdżeń, K. Fleeting beauty—The world of plant fragrances and their application. Molecules 2021, 26, 2473. [Google Scholar] [CrossRef]
- Sebghatollahi, Z.; Ghanadian, M.; Agarwal, P.; Ghaheh, H.S.; Mahato, N.; Yogesh, R.; Hejazi, S.H. Citrus flavonoids: Biological activities, implementation in skin health, and topical applications: A Review. ACS Food Sci. Technol. 2022, 2, 1417–1432. [Google Scholar] [CrossRef]
- Herman, A.; Herman, A.P. Essential oils and their constituents as skin penetration enhancer for transdermal drug delivery: A review. J. Pharm. Pharmacol. 2015, 67, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Valgimigli, L.; Gabbanini, S.; Berlini, E.; Lucchi, E.; Beltramini, C.; Bertarelli, Y.L. Lemon (Citrus limon, Burm.f.) essential oil enhances the trans-epidermal release of lipid-(A, E) and water-(B6, C) soluble vitamins from topical emulsions in reconstructed human epidermis. Int. J. Cosmet. Sci. 2012, 34, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Q.; Wu, Y.; Zhang, H.; Liu, P.; Yao, J.; Yao, P.; Chen, J.; Duan, J. Development of essential oils as skin permeation enhancers: Penetration enhancement effect and mechanism of action. Pharm. Biol. 2017, 55, 1592–1600. [Google Scholar] [CrossRef] [PubMed]
- Parhi, R.; Suresh, P.; Mondal, S.; Kumar, P.M. Novel Penetration Enhancers for Skin Applications: A Review. Curr. Drug Deliv. 2012, 9, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Furuishi, T.; Kato, Y.; Fukami, T.; Suzuki, T.; Endo, T.; Nagase, H.; Ueda, H.; Tomono, K. Effect of terpenes on the skin permeation of lomerizine dihydrochloride. J. Pharm. Pharm. Sci. 2013, 16, 551–563. [Google Scholar] [CrossRef] [PubMed]
- Moghadam, S.H.; Saliaj, E.; Wettig, S.D.; Dong, C.; Ivanova, M.V.; Huzil, J.T.; Foldvari, M. Effect of chemical permeation enhancers on stratum corneum barrier lipid organizational structure and interferon alpha permeability. Mol. Pharm. 2013, 10, 2248–2260. [Google Scholar] [CrossRef]
- Bech-Thomsen, N.; Wulf, H.C. Carcinogenic and melanogenic effects of a filtered metal halide UVA source and a tubular fluorescent UVA tanning source with or without additional solar-simulated UV radiation in hairless mice. Photochem. Photobiol. 1995, 62, 773–779. [Google Scholar] [CrossRef]
- Kligman, A.M. Early destructive effect of sunlight on human skin. Jama 1969, 210, 2377–2380. [Google Scholar] [CrossRef]
- Oikarinen, A.; Karvonen, J.; Uitto, J.; Hannuksela, M. Connective tissue alterations in skin exposed to natural and therapeutic UV-radiation. Photodermatology 1985, 2, 15–26. [Google Scholar]
- Sakurai, H.; Yasui, H.; Yamada, Y.; Nishimura, H.; Shigemoto, M. Detection of reactive oxygen species in the skin of live mice and rats exposed to UVA light: A research review on chemiluminescence and trials for UVA protection. Photochem. Photobiol. Sci. 2005, 4, 715–720. [Google Scholar] [CrossRef]
- Watson, R.E.B.; Griffiths, C.E.M. Pathogenic aspects of cutaneous photoaging. J. Cosmet. Dermatol. 2005, 4, 230–236. [Google Scholar] [CrossRef]
- Okayama, Y. Oxidative stress in allergic and inflammatory skin diseases. Curr. Drug Targets Inflamm. Allergy 2005, 4, 517–519. [Google Scholar] [CrossRef]
- Ishfaq, M.; Akhtar, B.; Muhammad, F.; Sharif, A.; Akhtar, M.F.; Hamid, I.; Sohail, K.; Muhammad, H. Antioxidant and Wound healing potential of essential oil from Citrus reticulata peel and its chemical characterization. Curr. Pharm. Biotechnol. 2020, 22, 1114–1121. [Google Scholar] [CrossRef]
- Simitzis, P.E. Enrichment of animal diets with essential oils—A great perspective on improving animal performance and quality characteristics of the derived products. Medicines 2017, 4, 35. [Google Scholar] [CrossRef]
- Giannenas, I.; Bonos, E.; Christaki, E.; Florou-Paneri, P. Oregano: A feed additive with functional properties. In Therapeutic Foods; Holban, A.M., Grumezescu, A.M., Eds.; Academic Press: San Diego, CA, USA, 2018; pp. 179–208. ISBN 9780128115176. [Google Scholar]
- Del Prado-Audelo, M.L.; Cortés, H.; Caballero-Florán, I.H.; González-Torres, M.; Escutia-Guadarrama, L.; Bernal-Chávez, S.A.; Giraldo-Gomez, D.M.; Magaña, J.J.; Leyva-Gómez, G. Therapeutic applications of terpenes on inflammatory diseases. Front. Pharmacol. 2021, 12, 2114. [Google Scholar] [CrossRef]
- Maurya, A.K.; Mohanty, S.; Pal, A.; Chanotiya, C.S.; Bawankule, D.U. The essential oil from Citrus limetta Risso peels alleviates skin inflammation: In-vitro and in-vivo study. J. Ethnopharmacol. 2018, 212, 86–94. [Google Scholar] [CrossRef]
- Li, Z.H.; Cai, M.; Liu, Y.S.; Sun, P.L.; Luo, S.L. Antibacterial activity and mechanisms of essential oil from Citrus medica L. var. sarcodactylis. Molecules 2019, 24, 1577. [Google Scholar] [CrossRef]
- Hirota, R.; Roger, N.N.; Nakamura, H.; Song, H.S.; Sawamura, M.; Suganuma, N. Anti-inflammatory effects of limonene from yuzu (Citrus junos tanaka) essential oil on eosinophils. J. Food Sci. 2010, 75, H87–H92. [Google Scholar] [CrossRef]
- Bertuzzi, G.; Tirillini, B.; Angelini, P.; Venanzoni, R. Antioxidative Action of Citrus limonum Essential Oil on Skin. European J. Med. Plants 2013, 3, 1–9. [Google Scholar] [CrossRef]
- Ruberto, G.; Baratta, M.T. Antioxidant activity of selected essential oil components in two lipid model systems. Food Chem. 2000, 69, 167–174. [Google Scholar] [CrossRef]
- El Bouamri, M.; Gorrichon, J.P.; Braun, A.M.; Oliveros, E. The reactivity of citronellol and α-thujene with singlet oxygen. rate constants of chemical reaction and physical quenching. Photochem. Photobiol. 1991, 54, 619–623. [Google Scholar] [CrossRef]
- Ambrosio, C.M.S.; Diaz-Arenas, G.L.; Agudelo, L.P.A.; Stashenko, E.; Contreras-Castillo, C.J.; Da Gloria, E.M. Chemical composition and antibacterial and antioxidant activity of a citrus essential oil and its fractions. Molecules 2021, 26, 2888. [Google Scholar] [CrossRef]
- Song, H.-S.; Ukeda, H.; Sawamura, M. Antioxidative activities of citrus peel essential oils and their components against linoleic acid oxidation. Food Sci. Technol. Res. 2001, 7, 50–56. [Google Scholar] [CrossRef]
- Takahashi, Y.; Inaba, N.; Kuwahara, S.; Kuki, W. Antioxidative effect of citrus essential oil components on human low-density lipoprotein in vitro. Biosci. Biotechnol. Biochem. 2003, 67, 195–197. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.; Liu, G.; Parfitt, J.; Liu, X.; Van Herpen, E.; Stenmarck, Å.; O’Connor, C.; Östergren, K.; Cheng, S. Missing Food, Missing Data? A Critical Review of Global Food Losses and Food Waste Data. Environ. Sci. Technol. 2017, 51, 6618–6633. [Google Scholar] [CrossRef]
- Borkovec, T.D.; Ruscio, A.M. Psychotherapy for generalized anxiety disorder. J. Clin. Psychiatry 2001, 62, 37–45. [Google Scholar]
- WHO. Depression. Available online: https://www.who.int/news-room/fact-sheets/detail/depression (accessed on 14 October 2022).
- Association, A.S. What Is Alzheimer’s Disease? Available online: https://www.alz.org/alzheimers-dementia/what-is-alzheimers (accessed on 14 October 2022).
- Alzheimer’s Disease Fact Sheet. National Institute on Aging. Available online: https://www.nia.nih.gov/health/alzheimers-disease-fact-sheet (accessed on 14 October 2022).
- Santamaria, J.; Iranzo, A. Sleep disorders matter in neurology. Lancet Neurol. 2014, 13, 18–20. [Google Scholar] [CrossRef]
- Freeman, D.; Sheaves, B.; Waite, F.; Harvey, A.G.; Harrison, P.J. Sleep disturbance and psychiatric disorders. Lancet Psychiatry 2020, 7, 628–637. [Google Scholar] [CrossRef]
- Momen, N.C.; Plana-Ripoll, O.; Agerbo, E.; Benros, M.E.; Børglum, A.D.; Christensen, M.K.; Dalsgaard, S.; Degenhardt, L.; de Jonge, P.; Debost, J.-C.P.G. Association between mental disorders and subsequent medical conditions. N. Engl. J. Med. 2020, 382, 1721–1731. [Google Scholar] [CrossRef]
- Muz, G.; Taşcı, S. Effect of aromatherapy via inhalation on the sleep quality and fatigue level in people undergoing hemodialysis. Appl. Nurs. Res. 2017, 37, 28–35. [Google Scholar] [CrossRef]
- Takeda, A.; Watanuki, E.; Koyama, S. Effects of inhalation aromatherapy on symptoms of sleep disturbance in the elderly with dementia. Evid.-Based Complement. Altern. Med. 2017, 2017, 1902807. [Google Scholar] [CrossRef]
- Tang, Y.; Gong, M.; Qin, X.; Su, H.; Wang, Z.; Dong, H. The therapeutic effect of aromatherapy on insomnia: A meta-analysis. J. Affect. Disord. 2021, 288, 1–9. [Google Scholar] [CrossRef]
- Pause, B.M.; Miranda, A.; Göder, R.; Aldenhoff, J.B.; Ferstl, R. Reduced olfactory performance in patients with major depression. J. Psychiatr. Res. 2001, 35, 271–277. [Google Scholar] [CrossRef]
- Guillemain, J.; Rousseau, A.; Delaveau, P. Neurodepressive effects of the essential oil of Lavandula angustifolia Mill. Ann. Pharm. Fr. 1989, 47, 337–343. [Google Scholar]
- Tafet, G.E.; Idoyaga-Vargas, V.P.; Abulafia, D.P.; Calandria, J.M.; Roffman, S.S.; Chiovetta, A.; Shinitzky, M. Correlation between cortisol level and serotonin uptake in patients with chronic stress and depression. Cogn. Affect. Behav. Neurosci. 2001, 1, 388–393. [Google Scholar] [CrossRef]
- Raison, C.L.; Capuron, L.; Miller, A.H. Cytokines sing the blues: Inflammation and the pathogenesis of depression. Trends Immunol. 2006, 27, 24–31. [Google Scholar] [CrossRef]
- Chung, S.; Son, G.H.; Kim, K. Circadian rhythm of adrenal glucocorticoid: Its regulation and clinical implications. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2011, 1812, 581–591. [Google Scholar] [CrossRef]
- Slavich, G.M.; Irwin, M.R. From stress to inflammation and major depressive disorder: A social signal transduction theory of depression. Psychol. Bull. 2014, 140, 774. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, Q.D.; Chai, Y.P.; Zhang, H.; Peng, P.; Yang, X.X. Natural terpenes as penetration enhancers for transdermal drug delivery. Molecules 2016, 21, 1709. [Google Scholar] [CrossRef]
- Xiong, M.; Li, Y.; Tang, P.; Zhang, Y.; Cao, M.; Ni, J.; Xing, M. Effectiveness of aromatherapy massage and inhalation on symptoms of depression in Chinese community-dwelling older adults. J. Altern. Complement. Med. 2018, 24, 717–724. [Google Scholar] [CrossRef]
- Shimizu, E.; Hashimoto, K.; Watanabe, H.; Komatsu, N.; Okamura, N.; Koike, K.; Shinoda, N.; Nakazato, M.; Kumakiri, C.; Okada, S. Serum brain-derived neurotrophic factor (BDNF) levels in schizophrenia are indistinguishable from controls. Neurosci. Lett. 2003, 351, 111–114. [Google Scholar] [CrossRef]
- Sánchez-Vidaña, D.I.; Po, K.K.-T.; Fung, T.K.-H.; Chow, J.K.-W.; Lau, W.K.-W.; So, P.-K.; Lau, B.W.-M.; Tsang, H.W.-H. Lavender essential oil ameliorates depression-like behavior and increases neurogenesis and dendritic complexity in rats. Neurosci. Lett. 2019, 701, 180–192. [Google Scholar] [CrossRef]
- Ruan, L.; Lau, B.W.-M.; Wang, J.; Huang, L.; ZhuGe, Q.; Wang, B.; Jin, K.; So, K.-F. Neurogenesis in neurological and psychiatric diseases and brain injury: From bench to bedside. Prog. Neurobiol. 2014, 115, 116–137. [Google Scholar] [CrossRef] [PubMed]
- Malberg, J.E.; Eisch, A.J.; Nestler, E.J.; Duman, R.S. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J. Neurosci. 2000, 20, 9104–9110. [Google Scholar] [CrossRef] [PubMed]
- Ayuob, N.N. Evaluation of the antidepressant-like effect of musk in an animal model of depression: How it works. Anat. Sci. Int. 2017, 92, 539–553. [Google Scholar] [CrossRef] [PubMed]
- Atsumi, T.; Tonosaki, K. Smelling lavender and rosemary increases free radical scavenging activity and decreases cortisol level in saliva. Psychiatry Res. 2007, 150, 89–96. [Google Scholar] [CrossRef]
- Saiyudthong, S.; Marsden, C.A. Acute effects of bergamot oil on anxiety-related behaviour and corticosterone level in rats. Phyther. Res. 2011, 25, 858–862. [Google Scholar] [CrossRef]
- Zhong, Y.; Zheng, Q.; Hu, P.; Huang, X.; Yang, M.; Ren, G.; Du, Q.; Luo, J.; Zhang, K.; Li, J.; et al. Sedative and hypnotic effects of compound Anshen essential oil inhalation for insomnia. BMC Complement. Altern. Med. 2019, 19, 306. [Google Scholar] [CrossRef]
- Rombolà, L.; Tridico, L.; Scuteri, D.; Sakurada, T.; Sakurada, S.; Mizoguchi, H.; Avato, P.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Bergamot Essential Oil Attenuates Anxiety-Like Behaviour in Rats. Molecules 2017, 22, 614. [Google Scholar] [CrossRef]
- Moradi, K.; Ashtarian, H.; Danzima, N.Y.; Saeedi, H.; Bijan, B.; Akbari, F.; Mohammadi, M.M. Essential Oil from Citrus aurantium Alleviates Anxiety of Patients Undergoing Coronary Angiography: A Single-Blind, Randomized Controlled Trial. Chin. J. Integr. Med. 2021, 27, 177–182. [Google Scholar] [CrossRef]
- Li, Z.; Wu, F.; Shao, H.; Zhang, Y.; Fan, A.; Li, F. Does the Fragrance of Essential Oils Alleviate the Fatigue Induced by Exercise? A Biochemical Indicator Test in Rats. Evid.-Based Complement. Altern. Med. 2017, 2017, 5027372. [Google Scholar] [CrossRef]
- Tanaka, M.; Szab, Á.; Spekker, E.; Tanaka, M.; Szabó, Á.; Vécsei, L. Neurogenic inflammation: The participant in migraine and recent advancements in translational research. Biomedicines 2021, 10, 76. [Google Scholar]
- Cutillas, A.-B.; Carrasco, A.; Martinez-Gutierrez, R.; Tomas, V.; Tudela, J. Rosmarinus officinalis L. essential oils from Spain: Composition, antioxidant capacity, lipoxygenase and acetylcholinesterase inhibitory capacities, and antimicrobial activities. Plant Biosyst. Int. J. Deal. All Asp. Plant Biol. 2018, 152, 1282–1292. [Google Scholar] [CrossRef]
- Ku, C.-M.; Lin, J.-Y. Anti-inflammatory effects of 27 selected terpenoid compounds tested through modulating Th1/Th2 cytokine secretion profiles using murine primary splenocytes. Food Chem. 2013, 141, 1104–1113. [Google Scholar] [CrossRef]
- De Almeida, A.A.C.; Silva, R.O.; Nicolau, L.A.D.; de Brito, T.V.; de Sousa, D.P.; Barbosa, A.L.d.R.; de Freitas, R.M.; Lopes, L.d.S.; Medeiros, J.-V.R.; Ferreira, P.M.P. Physio-pharmacological investigations about the anti-inflammatory and antinociceptive efficacy of (+)-limonene epoxide. Inflammation 2017, 40, 511–522. [Google Scholar] [CrossRef]
- Tang, M.Z.; Wang, Z.F.; Shi, Y.L. Involvement of cytochrome c release and caspase activation in toosendanin-induced PC12 cell apoptosis. Toxicology 2004, 201, 31–38. [Google Scholar] [CrossRef]
- Jackson, H.; Anzures-Cabrera, J.; Taylor, K.I.; Pagano, G. Hoehn and Yahr stage and striatal Dat-SPECT uptake are predictors of Parkinson’s disease motor progression. Front. Neurosci. 2021, 15, 765765. [Google Scholar]
- Zhou, X.; Hai-Yan, G.; Tun-Hai, X.; Tian, S. Physicochemical evaluation and essential oil composition analysis of Hyssopus cuspidatus Boriss from Xinjiang, China. Pharmacogn. Mag. 2010, 6, 278–281. [Google Scholar]
- Liu, J.L.; Fan, Y.G.; Yang, Z.S.; Wang, Z.Y.; Guo, C. Iron and Alzheimer’s disease: From pathogenesis to therapeutic implications. Front. Neurosci. 2018, 12, 632. [Google Scholar] [CrossRef]
- Swaminathan, S.; Fonseca, V.A.; Alam, M.G.; Shah, S.V. The role of iron in diabetes and its complications. Diabetes Care 2007, 30, 1926–1933. [Google Scholar] [CrossRef]
- Ademosun, A.O.; Oboh, G.; Olupona, A.J.; Oyeleye, S.I.; Adewuni, T.M.; Nwanna, E.E. Comparative study of chemical composition, in vitro inhibition of cholinergic and monoaminergic enzymes, and antioxidant potentials of essential oil from peels and seeds of sweet orange (Citrus sinensis L. Osbeck) Fruits. J. Food Biochem. 2016, 40, 53–60. [Google Scholar] [CrossRef]
- Oboh, G.; Puntel, R.L.; Rocha, J.B.T. Hot pepper (Capsicum annuum, Tepin and Capsicum chinese, Habanero) prevents Fe2+-induced lipid peroxidation in brain–in vitro. Food Chem. 2007, 102, 178–185. [Google Scholar] [CrossRef]
- Oboh, G.; Olasehinde, T.A.; Ademosun, A.O. Essential oil from lemon peels inhibit key enzymes linked to neurodegenerative conditions and pro-oxidant induced lipid peroxidation. J. Oleo Sci. 2014, 63, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Chaiyana, W.; Okonogi, S. Inhibition of cholinesterase by essential oil from food plant. Phytomedicine 2012, 19, 836–839. [Google Scholar] [CrossRef] [PubMed]
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Zhang, L.; Feng, L.; Yao, L. The anxiolytic effect of essential oil of Cananga odorata exposure on mice and determination of its major active constituents. Phytomedicine 2016, 23, 1727–1734. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Fukumoto, S.; Yokogoshi, H. Components of lemon essential oil attenuate dementia induced by scopolamine. Nutr. Neurosci. 2009, 12, 57–64. [Google Scholar] [CrossRef]
- Navarra, M.; Mannucci, C.; Delbò, M.; Calapai, G. Citrus bergamia essential oil: From basic research to clinical application. Front. Pharmacol. 2015, 6, 36. [Google Scholar] [CrossRef]
- Celia, C.; Trapasso, E.; Locatelli, M.; Navarra, M.; Ventura, C.A.; Wolfram, J.; Carafa, M.; Morittu, V.M.; Britti, D.; Di Marzio, L.; et al. Anticancer activity of liposomal Bergamot essential oil (BEO) on human neuroblastoma cells. Colloids Surf. B Biointerfaces 2013, 112, 548–553. [Google Scholar] [CrossRef]
- Da Pozzo, E.; De Leo, M.; Faraone, I.; Milella, L.; Cavallini, C.; Piragine, E.; Testai, L.; Calderone, V.; Pistelli, L.; Braca, A.; et al. Antioxidant and Antisenescence Effects of Bergamot Juice. Oxid. Med. Cell. Longev. 2018, 2018, 9395804. [Google Scholar] [CrossRef]
- Valussi, M.; Donelli, D.; Firenzuoli, F. Bergamot Oil: Botany, Production, Pharmacology. Encyclopedia 2021, 1, 16. [Google Scholar] [CrossRef]
- Scuteri, D.; Rombolà, L.; Tridico, L.; Mizoguchi, H.; Watanabe, C.; Sakurada, T.; Sakurada, S.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Neuropharmacological Properties of the Essential Oil of Bergamot for the Clinical Management of Pain-Related BPSDs. Curr. Med. Chem. 2019, 26, 3764–3774. [Google Scholar] [CrossRef]
- Toscano-Garibay, J.D.; Arriaga-Alba, M.; Sánchez-Navarrete, J.; Mendoza-García, M.; Flores-Estrada, J.J.; Moreno-Eutimio, M.A.; Espinosa-Aguirre, J.J.; González-Ávila, M.; Ruiz-Pérez, N.J. Antimutagenic and antioxidant activity of the essential oils of Citrus sinensis and Citrus latifolia. Sci. Rep. 2017, 7, 11479. [Google Scholar] [CrossRef]
- Goes, T.C.; Teixeira-silva, F. Effect of Sweet Orange Aroma on Experimental Anxiety in Humans. J. Altern. Complement. Med. 2012, 18, 798–804. [Google Scholar] [CrossRef]
- Hsouna, A.B.; Hamdi, N.; Halima, N.B.; Abdelkafi, S. Characterization of essential oil from Citrus aurantium L. flowers: Antimicrobial and antioxidant activities. J. Oleo Sci. 2013, 62, 763–772. [Google Scholar] [CrossRef]
- Pimenta, F.C.F.; Alves, M.F.; Pimenta, M.B.F.; Melo, S.A.L.; De Almeida, A.A.F.; Leite, J.R.; Pordeus, L.C.D.M.; Diniz, M.D.F.F.M.; Almeida, R.N. De Anxiolytic Effect of Citrus aurantium L. on Patients with Chronic Myeloid Leukemia. Phytother. Res. 2016, 30, 613–617. [Google Scholar] [CrossRef]
- Farshbaf-Khalili, A.; Kamalifard, M.; Namadian, M. Comparison of the effect of lavender and bitter orange on anxiety in postmenopausal women: A triple-blind, randomized, controlled clinical trial. Complement. Ther. Clin. Pract. 2018, 31, 132–138. [Google Scholar] [CrossRef]
- Namazi, M.; Amir Ali Akbari, S.; Mojab, F.; Talebi, A.; Alavi Majd, H.; Jannesari, S. Aromatherapy with Citrus aurantium oil and anxiety during the first stage of labor. Iran. Red Crescent Med. J. 2014, 16, e18371. [Google Scholar] [CrossRef]
- Moosavy, M.H.; Hassanzadeh, P.; Mohammadzadeh, E.; Mahmoudi, R.; Khatibi, S.A.; Mardani, K. Antioxidant and antimicrobial activities of essential oil of Lemon (Citrus limon) peel in vitro and in a food model. J. Food Qual. Hazards Control 2017, 4, 42–48. [Google Scholar]
- Johnson, C.E. Effect of aromatherapy on cognitive test anxiety among nursing students. Altern. Complement. Ther. 2014, 20, 84–87. [Google Scholar] [CrossRef]
- Viana, M.D.M.; Silva Neto, G.J.D.; Lima, A.A.D.; Leite, A.B.; Souza, I.T.; Santana, A.E.G.; Campesatto, E.A.; Moreira, M.S.A. Citrus limon (L.) Burm f. Essential oil has anxiolytic and sedative properties by modulating GABAA-receptors. Braz. Arch. Biol. Technol. 2020, 63, e20200206. [Google Scholar] [CrossRef]
- Khotimah, H.; Subagio, S.U. The Application of Acupressure Phytopharmaceutical Using Lemon Essential Oil Aromatherapy to Overcome Dysmenorrhea in Adolescents. Faletehan Health J. 2021, 8, 187–193. [Google Scholar] [CrossRef]
- Kustriyanti, D.; Putri, A.A. The effect of ginger and lemon aromatherapy on nausea and vomiting among pregnant women. J. Keperawatan Soedirman 2019, 14, 15–22. [Google Scholar] [CrossRef]
- Yi, F.; Jin, R.; Sun, J.; Ma, B.; Bao, X. Evaluation of mechanical-pressed essential oil from Nanfeng mandarin (Citrus reticulata Blanco cv. Kinokuni) as a food preservative based on antimicrobial and antioxidant activities. Lwt 2018, 95, 346–353. [Google Scholar] [CrossRef]
- Kwangjai, J.; Cheaha, D.; Manor, R.; Sa-ih, N.; Samerphob, N.; Issuriya, A.; Wattanapiromsakul, C.; Kumarnsit, E. Modification of brain waves and sleep parameters by Citrus reticulata Blanco. cv. Sai-Nam-Phueng essential oil. Biomed. J. 2021, 44, 727–738. [Google Scholar] [CrossRef]
- Zhou, X.M.; Zhao, Y.; He, C.C.; Li, J.X. Preventive effects of Citrus reticulata essential oil on bleomycin-induced pulmonary fibrosis in rats and the mechanism. J. Chin. Integr. Med. 2012, 10, 200–209. [Google Scholar] [CrossRef]
- Ali, M.; Akhter, R.; Narjish, S.N.; Shahriar, M.; Bhuiyan, M.A. Studies of preliminary phytochemical screening, membrane stabilizing activity, thrombolytic activity and in-vitro antioxidant activity of leaf extract of Citrus hystrix. Int. J. Pharm. Sci. Res. 2015, 6, 2367. [Google Scholar]
- Hongratanaworakit, T.; Buchbauer, G. Chemical composition and stimulating effect of Citrus hystrix oil on humans. Flavour Fragr. J. 2007, 22, 443–449. [Google Scholar] [CrossRef]
- Yoo, K.M.; Lee, K.W.; Park, J.B.; Lee, H.J.; Hwang, I.K. Variation in major antioxidants and total antioxidant activity of Yuzu (Citrus junos Sieb ex Tanaka) during maturation and between cultivars. J. Agric. Food Chem. 2004, 52, 5907–5913. [Google Scholar] [CrossRef]
- Satou, T.; Matsuura, M.; Takahashi, M.; Umezu, T.; Hayashi, S.; Sadamoto, K.; Koike, K. Anxiolytic-like effect of essential oil extracted from Abies sachalinensis. Flavour Fragr. J. 2011, 26, 416–420. [Google Scholar] [CrossRef]
- Ohata, M.; Zhou, L.; Ando, S.; Kaneko, S. Application of integrative physiological approach to evaluate human Application of integrative physiological approach to evaluate human physiological responses to the inhalation of essential oils of Japanese Citrus fruits. Biosci. Biotechnol. Biochem. 2021, 86, 109–116. [Google Scholar] [CrossRef]
- Matsumoto, T.; Kimura, T.; Hayashi, T. Aromatic effects of a Japanese citrus fruit-yuzu (Citrus junos Sieb. ex Tanaka)-on psychoemotional states and autonomic nervous system activity during the menstrual cycle: A single-blind randomized controlled crossover study. Biopsychosoc. Med. 2016, 10, 1–11. [Google Scholar] [CrossRef]
- Rahnama, S.; Rabiei, Z.; Alibabaei, Z.; Mokhtari, S.; Rafieian-Kopaei, M.; Deris, F. Anti-amnesic activity of Citrus aurantium flowers extract against scopolamine-induced memory impairments in rats. Neurol. Sci. 2015, 36, 553–560. [Google Scholar] [CrossRef]
- Azanchi, T.; Shafaroodi, H.; Asgarpanah, J. Anticonvulsant activity of Citrus aurantium blossom essential oil (neroli): Involvment of the GABAergic system. Nat. Prod. Commun. 2014, 9, 1615–1618. [Google Scholar]
- Scandurra, C.; Mezzalira, S.; Cutillo, S.; Zapparella, R.; Statti, G.; Maldonato, N.M.; Locci, M.; Bochicchio, V. The effectiveness of neroli essential oil in relieving anxiety and perceived pain in women during labor: A Randomized controlled trial. Healthcare 2022, 10, 366. [Google Scholar] [CrossRef]
- Satou, T.; Miyahara, N.; Murakami, S.; Hayashi, S.; Koike, K. Differences in the effects of essential oil from Citrus junos and (+)-limonene on emotional behavior in mice. J. Essent. Oil Res. 2012, 24, 493–500. [Google Scholar] [CrossRef]
- Eddin, L.B.; Jha, N.K.; Meeran, M.F.; Kesari, K.K.; Beiram, R.; Ojha, S. Neuroprotective potential of limonene and limonene containing natural products. Molecules 2021, 26, 4535. [Google Scholar] [CrossRef]
- Ali, B.; Al-Wabel, N.A.; Shams, S.; Ahamad, A.; Khan, S.A.; Anwar, F. Essential oils used in aromatherapy: A systemic review. Asian Pac. J. Trop. Biomed. 2015, 5, 601–611. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Takemoto, H.; Fu, Z.; Shimizu, E.; Kinjo, Y. Enhancement of pentobarbital-induced sleep by the vaporized essential oil of citrus keraji var. Kabuchii and its characteristic component, γ-Terpinene. Nat. Prod. Commun. 2016, 11, 1175–1178. [Google Scholar] [CrossRef]
- Blanch, G.P.; Nicholson, G.J. Determination of the Enantiomeric Composition of Limonene and Limonene-1,2-epoxide in Lemon Peel by Multidimensional Gas Chromatography with Flame-Ionization Detection and Selected Ion Monitoring Mass Spectrometry. J. Chromatogr. Sci. 1998, 36, 37–43. [Google Scholar]
- Flores, G.; Blanch, G.P.; Ruiz del Castillo, M.L.; Herraiz, M. Enantiomeric composition studies in Lavandula species using supercritical fluids. J. Sep. Sci. 2005, 28, 2333–2338. [Google Scholar]
- Mosandl, A. Authenticity assessment: A permanent challenge in food flavor and essential oil analysis. J. Chromatogr. Sci. 2004, 42, 440–449. [Google Scholar]
- Nhu-Trang, T.T.; Casabianca, H.; Grenier-Loustalot, M.F. Authenticity control of essential oils containing citronellal and citral by chiral and stable-isotope gas-chromatographic analysis. Anal. Bioanal. Chem. 2006, 386, 2141–2152. [Google Scholar]
- Hör, K.; Ruff, C.; Weckerle, B.; König, T.; Schreier, P. Flavor authenticity studies by 2H/1H ratio determination using on-line gas chromatography pyrolysis isotope ratio mass spectrometry. J. Agric. Food Chem. 2001, 49, 21–25. [Google Scholar]
- Faulhaber, S.; Hener, U.; Mosandl, A. GC/IRMS Analysis of Mandarin Essential Oils. 2. δ13CPDB Values of Characteristic Flavor Components. J. Agric. Food Chem. 1997, 45, 4719–4725. [Google Scholar]
- König, W.A.; Hochmuth, D.H. Enantioselective gas chromatography in flavor and fragrance analysis: Strategies for the identification of known and unknown plant volatiles. J. Chromatogr. Sci. 2004, 42, 423–439. [Google Scholar]
- Schulz, H.; Schrader, B.; Quilitzsch, R.; Steuer, B. Quantitative analysis of various citrus oils by ATR/FT-IR and NIR-FT Raman spectroscopy. Appl. Spectrosc. 2002, 56, 117–124. [Google Scholar]







| Citrus Type | Particulars | In Vitro/In Vivo/Animal Model | Activity | References |
|---|---|---|---|---|
| Bergamot orange (C. bergamia) essential oil (CBEO) | Antioxidant behavior | In vivo model obtained from mouse hearts | Increase in transcription of genes involved in antioxidant responses Having lower IC50 O2•− value in scavenging activity test than ascorbic acid and higher FRAP activity | [182] |
| Mood disorder | BEO aromatherapy in alleviating depressive mood in postpartum women | Significantly improve the depressive mood Sleep quality was not significantly different | [103] | |
| Diseased condition | Acclimatization of the rats was performed | Relieve symptoms of stress-induced anxiety No overlapping between BEO and benzodiazepines behavioral effects Integrated effect on both 5-HT and GABA-A receptors | [183] | |
| Neuropharmacological studies | The elevated plus-maze and the hole-board tests were performed to study of BEO on rats | Usefulness in neuroprotection Chronic pain control Management of stress, anxiety, and anxiety-related conditions | [184] | |
| Antinociceptive effect | Effect of inhalation BEO on formalin-induced nociceptive response in mice. | Inhalation of BEO exerted antinociceptive activity. reduces formalin-induced licking/biting behavior. chronic pain relief in a stepwise therapeutic manner | [161] | |
| Sweet orange or navel orange (C. sinensis L.) essential oil (CSEO) | Antioxidant behavior | Evaluated against the ROS-generating compound | Activity in DPPH assay was in a range of 6–23% for C. sinensis Decreased apoptosis in HaCat cells stimulated with H2O2. The levels of intracellular superoxide ion found to be lower | [185] |
| Mood disorder | Aromatherapy during dental treatment | Lower degree of anxiety and a more cheerful attitude. To reduce salivary cortisol and pulse rate | [104] | |
| Physiological and psychological effect | Measurements were performed in a chamber with an artificial climate with 20 females | Significant decrease in oxyhemoglobin concentration in the right prefrontal cortex of the brain. Increases comfortable, relaxed, and natural feelings. | [104] | |
| Anxiolytic effect | Forty (40) male volunteers were allocated for the inhalation | Decreases the symptoms of anxiety Improves the mood | [186] | |
| Unpredictable mild stress | Randomized three-arm controlled trial | Significantly improved depression-like behaviors in CUMS mice by lowering sucrose preference, body weight, curiosity, and mobility Reducing immobility time and dyslipidemia | [17] | |
| Bitter orange (C. aurantium) essential oil (CAEO) | Antioxidant behavior | DPPH scavenging test | contribute to the prevention of oxidation as antioxidants and free radical scavengers | [187] |
| Mood disorder/anxiolytic effect | Collection of medullary material in patients with chronic myeloid leukemia (CML) | Anxiolytic effect and reduces the signs and symptoms associated with anxiety Decrease in the SBP and DBP | [188] | |
| Diseased condition/premenstrual syndrome (PMS) | Inhalation of 0.5 percent CAEO during the luteal phase of the menstrual cycle | Improved the symptoms of PMS Effective as a new and complementary therapeutic method for the emotions PMS symptoms in female. | [18] | |
| Sedative and hypnotic effects | Spielberger’s State-Trait Anxiety Inventory (STAI) was used after giving bitter orange flower powder capsule to post-menopausal women | Inhaling the CAEO greatly reduced anxiety | [189] | |
| Reduces pain | Study was a randomized clinical trial conducted with 126 eligible primiparous patients | Controls the enzymes in prostaglandins and reduces pain; controls the contractions caused by oxytocin and prostaglandins and exert anti-uterine pain effects | [190] | |
| Lemon (C. limon) essential oil (CLEO) | Antioxidant behavior | DPPH radical scavenging assay | Lemon peel EO showed 55.09% inhibition of DPPH considerable antioxidant properties both in vitro and barley soup as food model | [191] |
| Mood disorder/Anxiety | Thirty-nine sophomore nursing students (35 female and 4 males) | Positive effect on cognitive test anxiety | [192] | |
| Diseased condition/anxiolytic-like effect | Swiss mice model | Induce an anxiolytic behavior in mice no toxicity in vitro | [193] | |
| Treatment of dysmenorrhea | Population of this study amounted to 185 | Psychological and physical benefits | [194] | |
| Effect on nausea among pregnant women | Control trial on 90 pregnant women | Effective in reducing pregnancy nausea and vomiting | [195] | |
| Mandarin (C. reticulata) essential oil (CREO) | Antioxidant behavior | DPPH), 3-(N-morpholino) propane sulfonic acid (ABTS) | Exhibited moderate radical scavenging activity | [196] |
| Mood disorder/mood and as a relaxing hypnotic agent | Frontal and parietal skulls of male Wistar rats implanted with electrodes for electroencephalographic (EEG) | CREO reduces REM sleep latency and enhanced the overall time and number of REM sleep episodes | [197] | |
| Anti-proliferative | Protective effects on bleomycin (BLM)-induced lung fibrosis in rats | Preventive effects on BLM-induced pulmonary fibrosis in rats Anti-proliferative effect against human embryonic lung fibroblasts | [198] | |
| Kaffir lime (C. hystrix) essential oil (CHEO) | Antioxidant behavior | DPPH free radical scavenging assay | Potential antioxidant activity | [199] |
| Stimulating effect | Forty healthy volunteers participated in the experiments | Reducing depression and stress in humans more alert, attentive, cheerful attitude | [200] | |
| Yuzu (C. junos) essential oil (CJEO) | Antioxidant behavior | DPPH free radical scavenging test | Mature yuzu contains higher amounts of vitamin C and phenolics than other citrus fruits Significant dietary source of antioxidants | [201] |
| Mood disorder | Inhaled administration (i.h.) of EOCJ for 90 min on mouse | Increased locomotor activity The anxiolytic-like effect | [202] | |
| Autonomic nervous system (ANS) | Study on seventeen women with subjective premenstrual symptoms | Therapeutic effects of yuzu fragrance on premenstrual symptoms (PMS) Can reduce premenstrual emotional symptoms Increased parasympathetic activity | [203] | |
| Physiological effect | Effect of 10-min inhalation of the yuzu scent on 21 women | Reduced heart rate (HR) and enhanced high-frequency power of heart rate variability (HRV), exhibiting parasympathetic nervous system activation, alleviation of negative emotional stress | [204] | |
| Human psychology | 32 healthy participants enrolled in the study (16 men and 16 women, aged 20–24 years) | Oxyhemoglobin concentration in the prefrontal cortex increased Task performance improved after inhaling yuzu essential oil | [203] | |
| Neroli (C. aurantium) essential oil (CAEO) | Antioxidant behavior | DPPH test | Prevention of oxidation as antioxidants and free radical scavengers. Essential oils in the old leaves had the maximum antioxidant activity | [43] |
| Diseased condition/neurological disorder | Study on scopolamine-induced learning and memory deficit in rats | Repairing effects on memory and behavioral disorders Treatment of AD, insomnia, anxiety, and epilepsy | [205] | |
| Mood disorder/anxiolytic Effect | Study on patients with chronic myeloid leukemia (CML) | Diastolic pressure decreases Exhibits an anxiolytic effect and reduces the signs and symptoms associated with anxiety in patients with CML | [188] | |
| Antiseizure and anticonvulsant effect | Assessed in pentylenetetrazole (PTZ)-induced in mice | Anticonvulsant activity which supports the ethnomedicinal claims of the use of the plant in the management of seizure | [206] | |
| Effect on anxiety and perceived pain in women during labor | Study on 88 women during labor | Used as an alternative tool to relieve anxiety and perceived pain in women during all stages of labor | [207] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agarwal, P.; Sebghatollahi, Z.; Kamal, M.; Dhyani, A.; Shrivastava, A.; Singh, K.K.; Sinha, M.; Mahato, N.; Mishra, A.K.; Baek, K.-H. Citrus Essential Oils in Aromatherapy: Therapeutic Effects and Mechanisms. Antioxidants 2022, 11, 2374. https://doi.org/10.3390/antiox11122374
Agarwal P, Sebghatollahi Z, Kamal M, Dhyani A, Shrivastava A, Singh KK, Sinha M, Mahato N, Mishra AK, Baek K-H. Citrus Essential Oils in Aromatherapy: Therapeutic Effects and Mechanisms. Antioxidants. 2022; 11(12):2374. https://doi.org/10.3390/antiox11122374
Chicago/Turabian StyleAgarwal, Pooja, Zahra Sebghatollahi, Mehnaz Kamal, Archana Dhyani, Alpana Shrivastava, Kiran Kumari Singh, Mukty Sinha, Neelima Mahato, Awdhesh Kumar Mishra, and Kwang-Hyun Baek. 2022. "Citrus Essential Oils in Aromatherapy: Therapeutic Effects and Mechanisms" Antioxidants 11, no. 12: 2374. https://doi.org/10.3390/antiox11122374
APA StyleAgarwal, P., Sebghatollahi, Z., Kamal, M., Dhyani, A., Shrivastava, A., Singh, K. K., Sinha, M., Mahato, N., Mishra, A. K., & Baek, K.-H. (2022). Citrus Essential Oils in Aromatherapy: Therapeutic Effects and Mechanisms. Antioxidants, 11(12), 2374. https://doi.org/10.3390/antiox11122374












